Multi-Step Recycling of BF Slag Heat via Biomass for CO2 Mitigation
Abstract
:1. Introduction
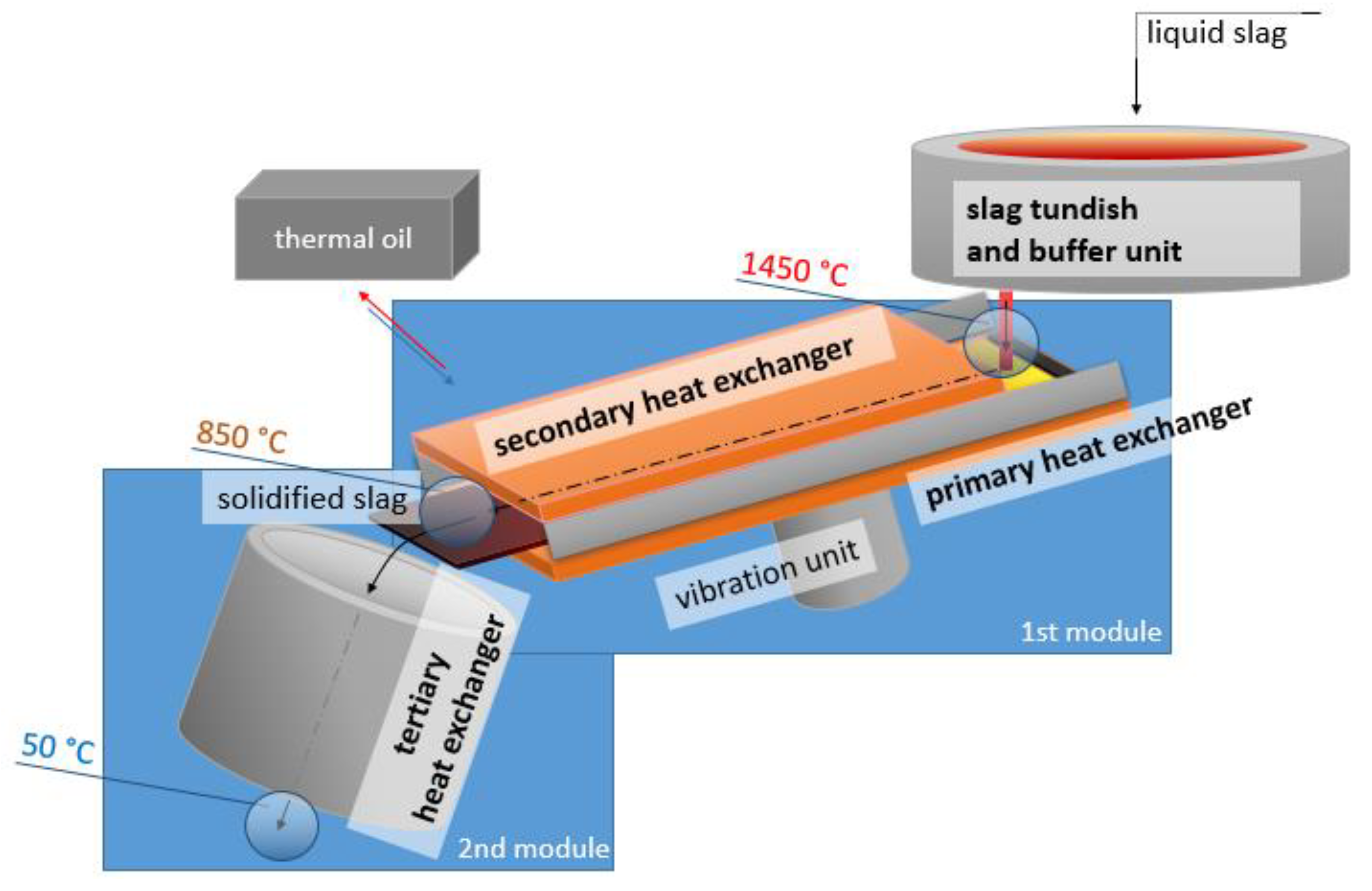
2. Description of the Slag Heat Recovery Concept
3. Production of Torrefied Biomass and Biogas
- Higher oxygen content (~40%);
- Lower carbon content (40–60%);
- Lower sulphur content (<0.1% S);
- Higher chlorine and alkali (particularly potassium) content in herbaceous biomass (grasses, straws);
- Low ash contents possible (<2%), depending on source very broad (0.1–25%);
- Lower heating values;
- Much lower bulk density;
- More fibrous and less friable structure;
- C/H and O/C ratios indicate the expected release.
4. Utilisation of Recuperated Thermal Energy
5. Conclusions
6. Patents
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Pikna, L.; Hezelova, M.; Morillon, A.; Algermissen, D.; Milkovic, O.; Findorak, R.; Cesnek, M.; Briancin, J. Recovery of Chromium from Slags Leachates by Electrocoagulation and Solid Product Characterization. Metals 2020, 10, 1593. [Google Scholar] [CrossRef]
- Kremer, D.; Wotruba, H. Separation of Products from Mineral Sequestration of CO2 with Primary and Secondary Raw Materials. Minerals 2020, 10, 1098. [Google Scholar] [CrossRef]
- Firsbach, F.; Senk, D.; Babich, A. Process Concept for the Dry Recovery of Thermal Energy of Liquid Ferrous Slags. J. Sustain. Metall. 2021, 7, 783–793. [Google Scholar] [CrossRef]
- Yang, J.; Firsbach, F.; Sohn, I. Pyrometallurgical processing of ferrous slag “co-product” zero waste full utilization: A critical review. Resour. Conserv. Recycl. 2022, 178, 106021. [Google Scholar] [CrossRef]
- Barati, M.; Jahanshahi, S. Granulation and Heat Recovery from Metallurgical Slags. J. Sustain. Metall. 2020, 6, 191–206. [Google Scholar] [CrossRef]
- Bisio, G. Energy recovery from molten slag and exploitation of the recovered energy. Energy 1997, 22, 501–509. [Google Scholar] [CrossRef]
- Shigaki, N.; Tobo, H.; Ozawa, S.; Ta, Y.; Hagiwara, K. Heat recovery process from packed bed of hot slag plates. ISIJ Int. 2015, 55, 2258–2265. [Google Scholar] [CrossRef] [Green Version]
- Fujii, H.; Tanaka, K.; Nakamura, M.; Okuno, R.; Hashizume, S.; Katayama, H.; Fujita, T. Apparatus for Heat Recovery from Molten Slag. US Patent US4350326A, 21 September 1982. [Google Scholar]
- Shimizu, H.; Watanabe, M.; Koide, M.; Ueno, T.; Yutaka, A. Development of molten blast furnace slag granulator. Tetsu-Hagane 1983, 69, 29. [Google Scholar]
- Ando, J.; Onoue, H.; Nakahara, T.; Ichimura, S.; Kondo, M. Development of slag blast granulating plant characterized by innovation of the slag treatment method, heat recovery and recovery of slag as resources. Mitsubishi Heavy Ind. Technol. Rev. 1985, 22, 136–142. [Google Scholar]
- Yoshinaga, M.; Fujii, K.; Shigematsu, T.; Nakata, T. Dry granulation and solidification of molten blast furnace slag. Trans. Iron Steel Inst. Jpn. 1982, 22, 823–829. [Google Scholar] [CrossRef]
- Xie, D.; Jahanshahi, S.; Norgate, T. Dry granulation to provide a sustainable option for slag treatment. In Proceedings of the Sustainable Mining Conference, Kalgoorlie, Australia, 17–19 August 2010; pp. 22–28. [Google Scholar]
- Fleischanderl, A.; Fenzl, T.; Neuhold, R. Dry Slag Granulation for Slag Sand Production and Heat Recovery–Development Status of the Pilotplant at Blast Furnace A at Voestalpine. Miner. Nebenprodukte Abfälle 2019, 6, 228–245. [Google Scholar]
- Kappes, H.; Michels, D. Dry Granulation with Energy Recovery: Pilot Campaign at ROGESA. In Proceedings of the AISTech 2014, Indianapolis, IN, USA, 5–8 May 2014. [Google Scholar]
- Hüttenmeister, D. Entwicklung einer Anlage zur Amorphen Erstarrung Flüssiger Hochofenschlacke mit Gleichzeitiger Wärmerückgewinnung. Ph.D. Thesis, RWTH Aachen University, Aachen, Germany, 2015. [Google Scholar]
- Firsbach, F. Konstruktion und Prozesstechnische Optimierung einer Anlage zur Amorphen und Rekuperativen Erstarrung von Schlackensystemen Integrierter Hüttenwerke. Ph.D. Thesis, RWTH Aachen University, Aachen, Germany, 2018. [Google Scholar]
- Babich, A.; Senk, D. Biomass use in the steel industry: Back to the future? Stahl Eisen 2013, 133, 57–67. [Google Scholar]
- Babich, A.; Senk, D.; Solar, J.; de Marco, I. Efficiency of Biomass Use for Blast Furnace Injection. ISIJ 2019, 59, 2212–2219. [Google Scholar] [CrossRef] [Green Version]
- Koppejan, J.; Sokhansanj, S.; Melin, S.; Madrali, S. Status Overview of Torrefaction Technologies; Task 32 Report; IEA Bioenergy: Paris, France, 2019; pp. 2–5. [Google Scholar]
- Solar, J.; de Marco, I.; Caballero, B.M.; Lopez-Urionabarrenechea, A.; Rodriguez, N.; Agirre, I.; Adrados, A. Influence of temperature and residence time in the pyrolysis of woody biomass waste in a continuous screw reactor. Biomass Bioenergy 2016, 95, 416–423. [Google Scholar] [CrossRef]
- Steinparzer, T. The Pathway to Green Steel. Steel Times International Webinar “Sustainability & Steelmaking”. Available online: https://www.steeltimesint.com/news/sustainability-steelmaking (accessed on 22 January 2021).
- Woo, H.; Turner, P. A Review of Recent Research on Carbon Neutrality in Forest Bioenergy Feedstocks. Int. J. Environ. Sci. Nat. Res. 2019, 19, 556014. [Google Scholar] [CrossRef]
- Schwarz, M. Beitrag zur Energetischen und Stofflichen Verwertung von Biomasse in der Eisenmetallurgie: Biokoks und Reduktionsgas, IEHK, 1st ed. Ph.D. Thesis, RWTH Aachen University, Shaker Verlag, Aachen, Germany, 2019. [Google Scholar]
- Zhang, W.; Zhang, J.; Xue, Z.; Zou, Z.; Qi, Y. Unsteady Analyses of the Top Gas Recycling Oxygen Blast Furnace. ISIJ Int. 2016, 56, 1358–1367. [Google Scholar] [CrossRef] [Green Version]
- Zhang, W.; Xue, Z.g; Zahng, J.; Wang, W.; Cheng, C.; Zou, Z. Medium oxygen enriched blast furnace with top gas recycling strategy. J. Iron Steel Res. Int. 2017, 8, 778–786. [Google Scholar] [CrossRef]
- Zhang, W.; Dai, J.; Li, C.; Yu, X.; Xue, Z.; Saxén, H. A review on Explorations of the Oxygen Blast Furnace Process. Steel Res. Int. 2021, 92, 202000326. [Google Scholar] [CrossRef]
- Mousa, E.; Ahmed, H. Utilization of biomass as an alternative fuel in iron and steel making. In Iron Ore, 2nd ed.; Lu, L., Ed.; Woodhead Publishing: Duxford, UK, 2022; pp. 665–690. [Google Scholar]
- Nielson, S.; Okosun, T.; Damstedt, B.; Jampani, M.; Zhou, C.Q. Tuyere-Level Syngas Injection in the Blast Furnace: A Computational Fluid Dynamics Investigation. Processes 2021, 9, 1447. [Google Scholar] [CrossRef]

| Technology | Developer | Source | Efficiency | Capacity | Pros | Cons |
|---|---|---|---|---|---|---|
| Rotating drum | Sumitomo, | [5] | 50–60% | 50 t/h |
|
|
| NKK, | [6] | 40% | - | |||
| JFE, | [7] | 34% | 60 t/h | |||
| Mechanical stirring | Kawasaki, | [8] | 59% | - |
|
|
| Sumitomo, | [9] | 50% | 30 t/h | |||
| Air blasting | NKK and Mitsubishi, | [5] | 40–50% | 60 t/h |
|
|
| Mitsubishi, | [10] | 81% | 80 t/h | |||
| Spinning disc/cup | Sumitomo, | [11] | 50–60% | 50 t/h |
|
|
| CSIRO, | [12] | 80% | 18–30 t/h | |||
| British Steel, | [6] | 60% | 40 t/h | |||
| Primetals, | [13] | 70% | 30–40 t/h | |||
| Steel sphere injection | Paul Wurth, | [14] | - | 150 t/h |
|
|
| Vibrating chute | RWTH Aachen, and Z&J Technologies, | [3] | 42% | 16 t/h |
|
|
| Parameter | Value | Unit |
|---|---|---|
| Blast furnace coke consumption rate | 350 | kg/tHM 1 |
| CO2 emission via coking | 270 | kg/tcoke 2 |
| CO2 emission via coke gasification in BF 3 | 3667 | kg/tcoke |
| CO2 emission via coking and coke gasification | 3937 | kg/tcoke |
| CO2 emission of coke per hot metal | 1378 | kg/tHM |
| Parameter | Value | Unit |
|---|---|---|
| Slag volume | 300 | kg/tHM |
| Slag heat 1 | 1.7 | GJ/tslag |
| Slag heat recovery yield 1 | 42 | % |
| Recovered slag heat 1 | 0.7 | GJ/tslag |
| Recovered slag heat 1 | 0.2 | GJ/tHM |
| Energy required for drying and torrefaction 2 | 1.3 | GJ/tWB |
| Torrefaction mass yield wet biomass to torrefied biomass 2 | 37 | % |
| Energy required for drying and torrefaction 2 | 3.5 | GJ/tTB |
| TB producible with recovered slag heat | 0.20 | tTB/tslag |
| TB producible with recovered slag heat | 0.06 | tTB/tHM |
| TB producible with recovered slag heat | 17 | % (tTB/tcoke) |
| Parameter | Value | Unit |
|---|---|---|
| Rate of coke substituted by TB as PC | 5 | % |
| Coke substituted by 5% TB as PC | −18 | kg/tHM |
| Coke substituted biogas injection into BF 1 | −10 | kg/tHM |
| Combined coke substitution by TB and biogas | −28 | kg/tHM |
| CO2 reduction by combined substitution with TB and biogas 2 | −108 | kg/tHM |
| Relative CO2 emission abatement via TB and biogas | −7.9 | % |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Firsbach, F.; Senk, D.; Babich, A. Multi-Step Recycling of BF Slag Heat via Biomass for CO2 Mitigation. Minerals 2022, 12, 136. https://doi.org/10.3390/min12020136
Firsbach F, Senk D, Babich A. Multi-Step Recycling of BF Slag Heat via Biomass for CO2 Mitigation. Minerals. 2022; 12(2):136. https://doi.org/10.3390/min12020136
Chicago/Turabian StyleFirsbach, Felix, Dieter Senk, and Alexander Babich. 2022. "Multi-Step Recycling of BF Slag Heat via Biomass for CO2 Mitigation" Minerals 12, no. 2: 136. https://doi.org/10.3390/min12020136
APA StyleFirsbach, F., Senk, D., & Babich, A. (2022). Multi-Step Recycling of BF Slag Heat via Biomass for CO2 Mitigation. Minerals, 12(2), 136. https://doi.org/10.3390/min12020136







