Gemological Characteristics of Lvwen Stone and Its Color Genesis
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Specimen Preparation and Testing Methods
2.2.1. Specimen Preparation
2.2.2. X-ray Diffraction Analysis
2.2.3. Scanning Electron Microscope
2.2.4. Fourier-Transform Infrared Spectroscopy
2.2.5. Laser Ablation Inductively Coupled Plasma Mass Spectrometry
2.2.6. Ultraviolet–Visible Spectroscopy
3. Results
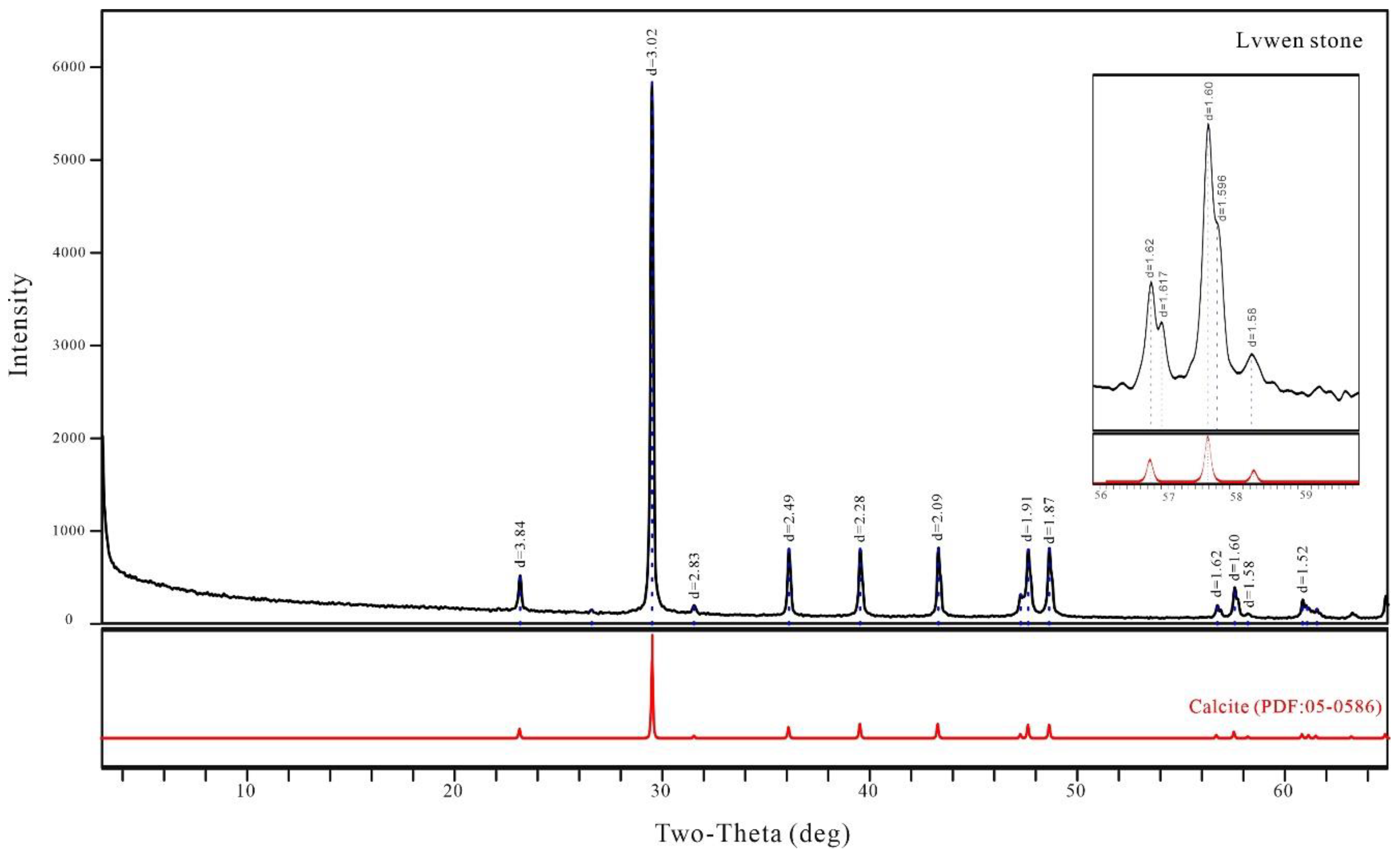
3.1. X-ray Diffraction Analysis
3.2. Spectroscopic Characterization
3.2.1. Fourier-Transform Infrared Spectroscopy Analysis
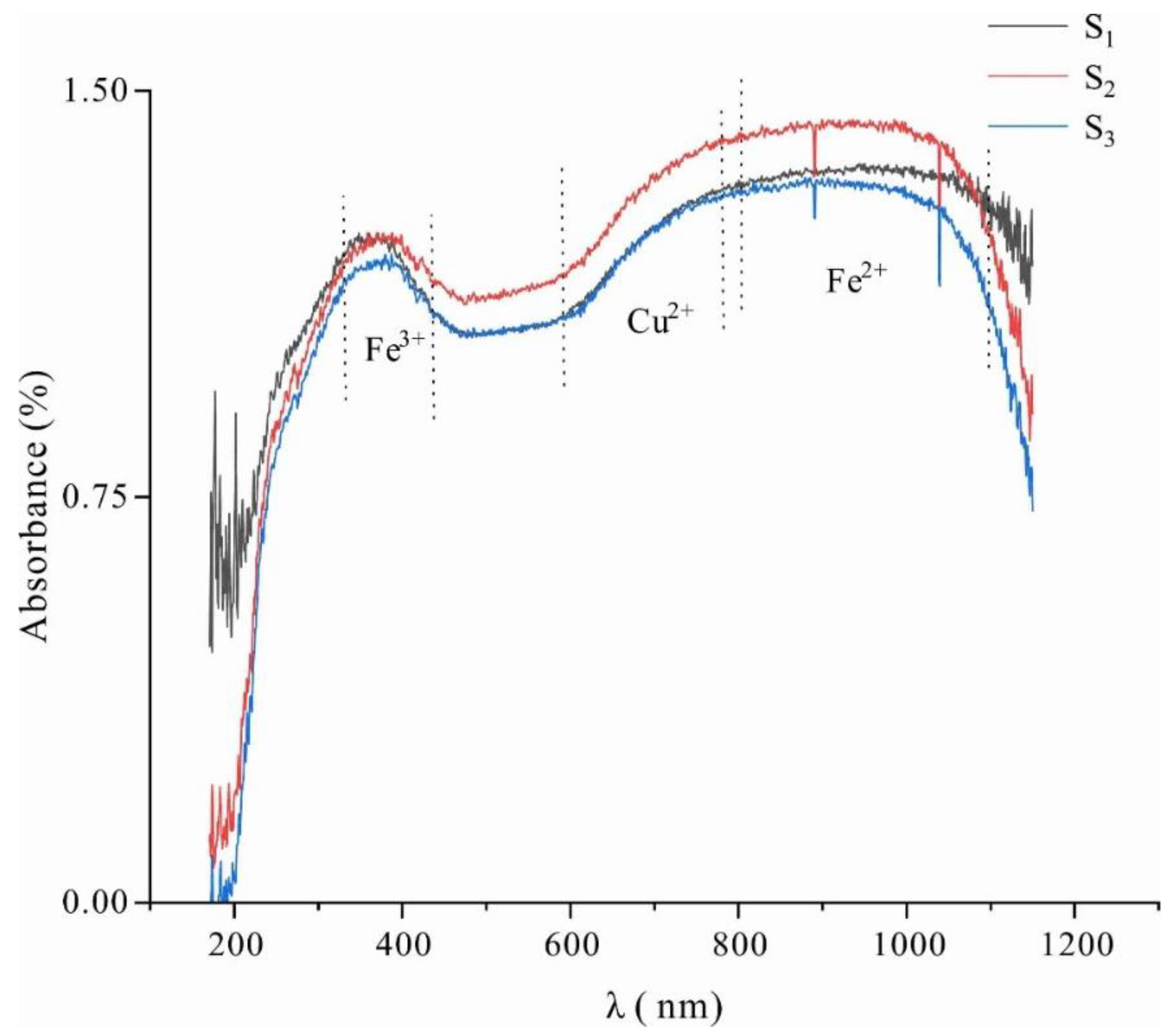
3.2.2. Ultraviolet–Visible Spectroscopy Analysis
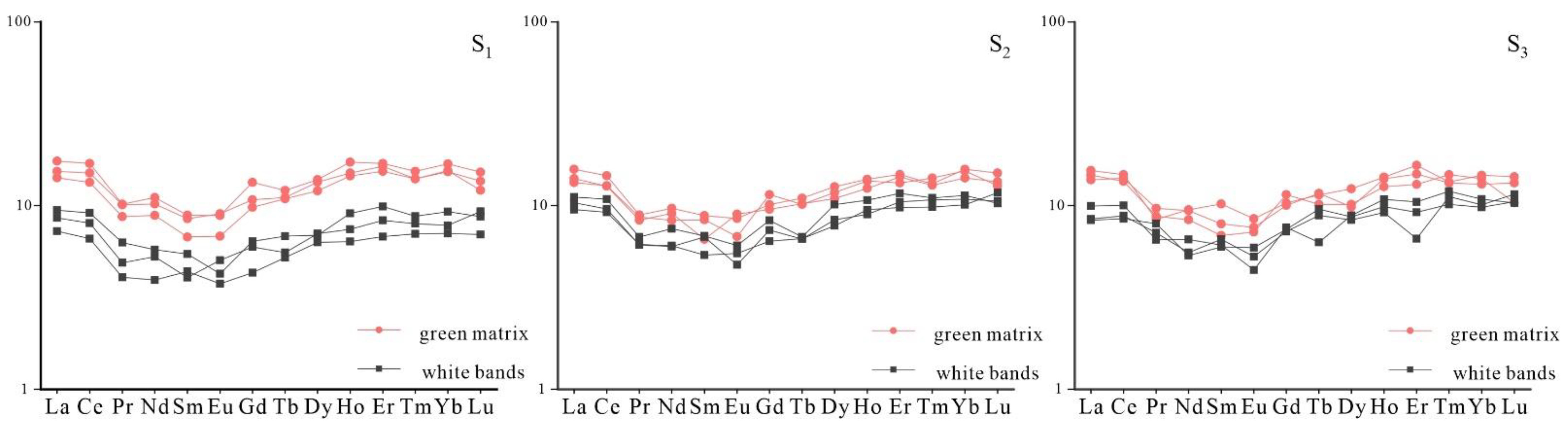
3.3. Chemical Compositions
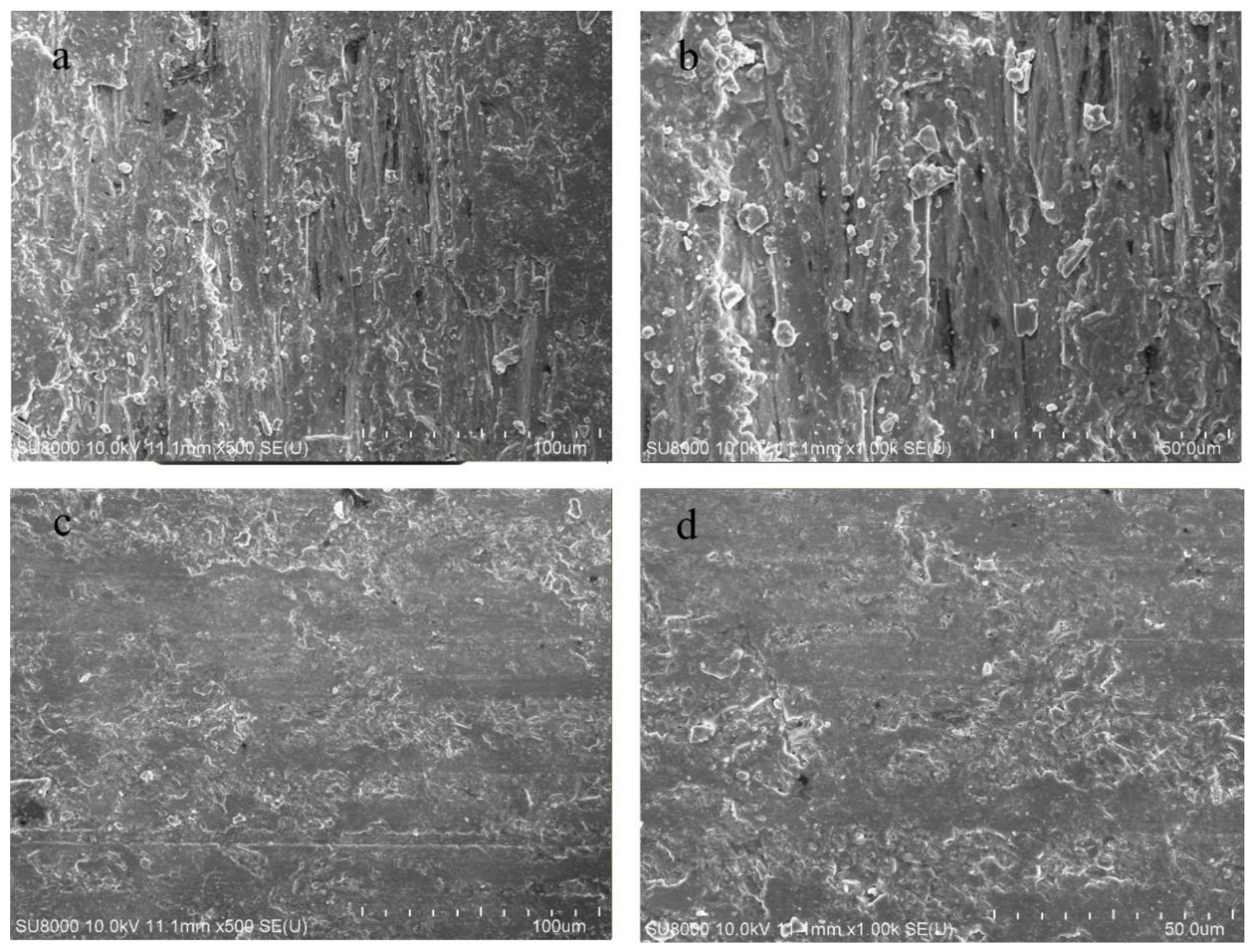
3.4. Microstructural Characteristics
4. Discussion
5. Conclusions
- (1)
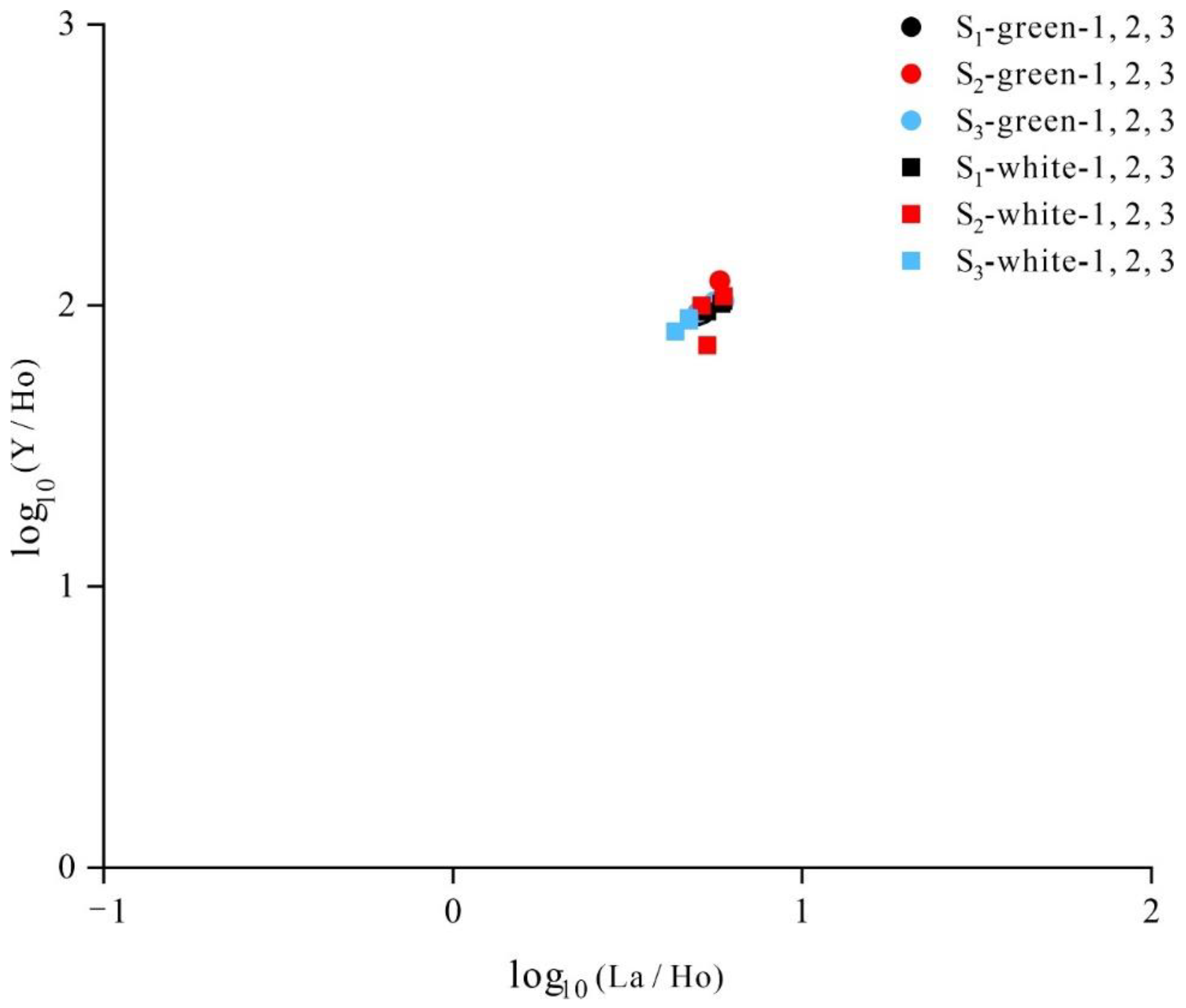
- The mineral composition of Lvwen stone is calcite, with crystal cell parameters of a = 4.97 and c = 17.00 Å and calcite crystals with an average grain size of 1737 Å. The cabochons used in this study have obvious white bands even after being cut into blocks, indicating that the banding is not due to chatoyancy (“cat’s-eye effect”). The white bands are composed of parallel-arranged, ~100 μm–sized calcite crystals. In contrast, the green matrix is dominated by smaller (~10 μm) cryptocrystalline calcite particles. These differences in crystallinity contribute to the contrasting transparencies of the white bands and green matrix.
- (2)
- The chemical formula of Lvwen stone is Ca0.97Mn0.02Mg0.01 [CO3], and the content of ΣREE in the green matrix is higher than that in the white bands. There is a significantly larger content of Fe and Cu in the green matrix as compared with the white bands, showing that the green color of the matrix is formed by the absorption of light by Fe3+ and Cu2+ in the calcite lattice, resulting from the 6A1→4E(4D)d-d intra-ion electron transition of Fe3+ and 2E→2T2(2D)d-d intra-ion electron transition of Cu2+, respectively. In addition, different crystal particle sizes may also affect the color of the green matrix and the white bands.
- (3)
- The occurrence of the ~1125 cm−1 absorption band and the broadening of the ~1490 cm−1 band on the FTIR spectrum suggest that the reduced symmetry of the [CO3]2− distribution of calcite crystals in Lvwen stone, compared with that of ideal calcite, may be related to isomorphic substitution of Mg2+ and Mn2+ for Ca2+. The double absorptions at 2923 and 2854 cm−1 reveal the presence of aliphatic hydrocarbon inclusions in the study specimens.
- (4)
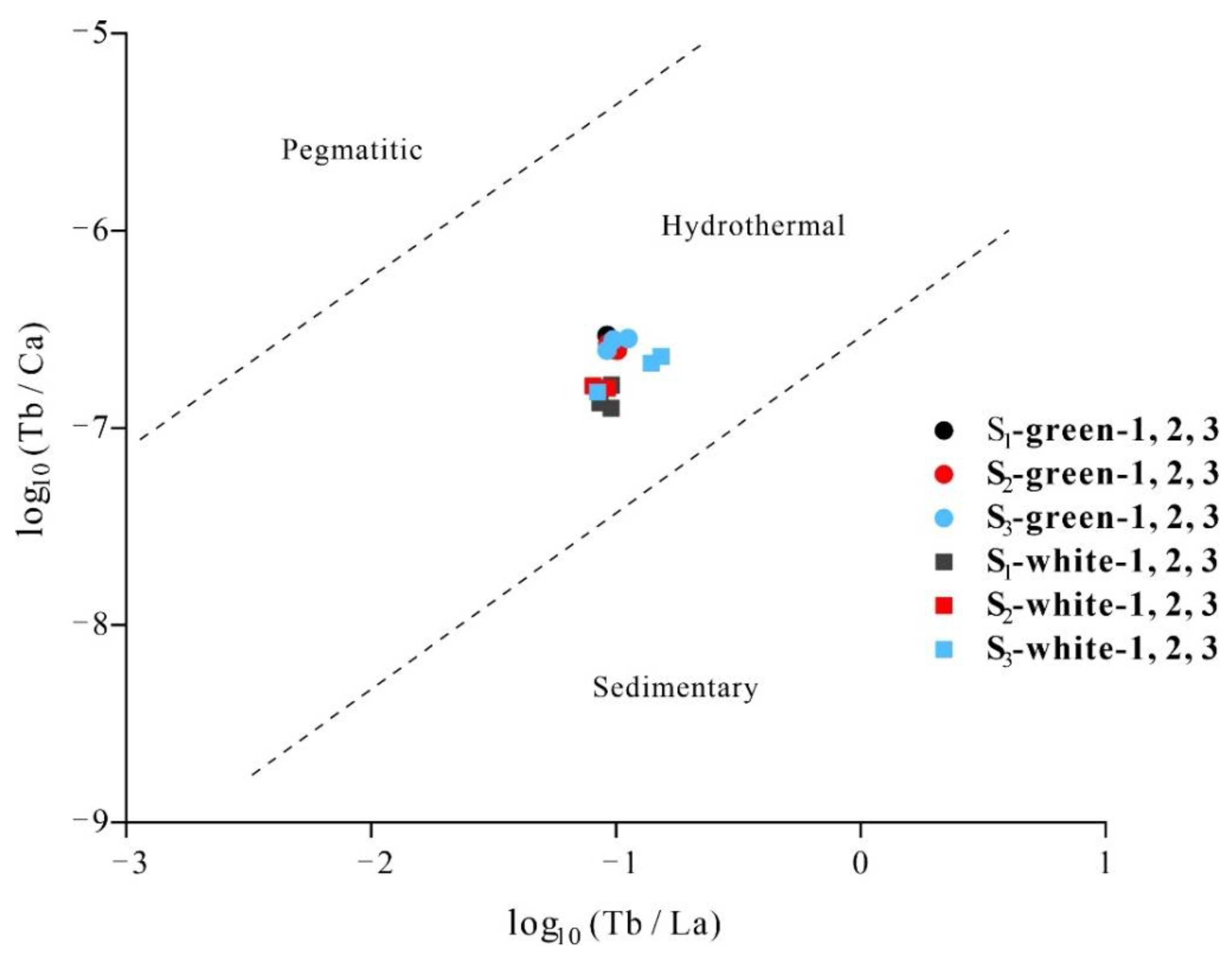
- Discriminant diagrams show that the green matrix and white bands of Lvwen stone had the same origin, and that both are the products of hydrothermal fluids. The presence of positive Ce anomalies in both the green matrix and white bands is an indication that the mineralizing fluid reductively remobilized earlier-formed Ce-oxides that had precipitated under oxidizing conditions. The orientation of crystals in the white bands perpendicular to the band plane shows that the study specimens formed in a tectonically controlled stress regime that permitted the formation of stretching veins.
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Hyrsl, J. Cat’s-eye calcite from Pakistan. Gems. Gemol. 2015, 51, 199–200. [Google Scholar]
- Zong, K.; Klemd, R.; Yuan, Y.; He, Z.; Guo, J.; Shi, X.; Liu, Y.; Hu, Z.; Zhang, Z. The assembly of Rodinia: The correlation of early Neoproterozoic (ca. 900Ma) high-grade metamorphism and continental arc formation in the southern Beishan Orogen, southern Central Asian Orogenic Belt (CAOB). Precambrian Res. 2017, 290, 32–48. [Google Scholar] [CrossRef]
- Hu, Z.; Zhang, W.; Liu, Y.; Gao, S.; Li, M.; Zong, K.; Chen, H.; Hu, S. “Wave” Signal-Smoothing and Mercury-Removing Device for Laser Ablation Quadrupole and Multiple Collector ICPMS Analysis: Application to Lead Isotope Analysis. Anal. Chem. 2015, 87, 1152–1157. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Hu, Z.; Gao, S.; Günther, D.; Xu, J. In situ analysis of major and trace elements of anhydrous minerals by LA-ICP-MS without applying an internal standard. Chem. Geol. 2008, 257, 34–43. [Google Scholar] [CrossRef]
- Wen, L.; Liang, W.; Zhang, Z.; Huang, J. The Infrared Spectroscopy of Minerals; Chongqing University Press: Chongqing, China, 1989. [Google Scholar]
- Plav, B.; Kobe, S.; Orel, B. Identification of crystallization forms of CaCO3 with FTIR spectroscopy. Kovine Zlitine Tehnol. 1999, 33, 517–521. [Google Scholar]
- Li, R.; Dong, S.; Lehrmann, D.; Duan, L. Tectonically driven organic fluid migration in the Dabashan Foreland Belt: Evidenced by geochemistry and geothermometry of vein-filling fibrous calcite with organic inclusions. J. Asian Earth Sci. 2013, 75, 202–212. [Google Scholar] [CrossRef]
- Marfunin, A.d.S.; Egorova, N. Physics of Minerals and Inorganic Materials: An introduction; Springer: Berlin/Heidelberg, Germany, 1979. [Google Scholar]
- Farges, F.; Benzerara, K.; Brown, G.E., Jr. Chrysocolla redefined as spertiniite. Am. Inst. Phys. 2007, 882, 223–225. [Google Scholar]
- Gaffey, S.J. Spectral reflectance of carbonate minerals in the visible and near infrared (0.35–2.55 um): Anhydrous carbonate minerals. J. Geophys. Res. Solid Earth 1987, 92, 1429–1440. [Google Scholar] [CrossRef]
- McDonough, W.F.; Sun, S.S. The composition of the Earth. Chem Geol. Chem. Geol. 1995, 120, 223–253. [Google Scholar] [CrossRef]
- Zhao, S. Crystallography and Mineralogy (Third Edition); Higher Education Press: Beijing, China, 2017. [Google Scholar]
- Bau, M.; Möller, P. Rare earth element fractionation in metamorphogenic hydrothermal calcite, magnesite and siderite. Mineral. Petrol. 1992, 45, 231–246. [Google Scholar] [CrossRef]
- Möller, P.; Parekh, P.P.; Schneider, H.J. The application of Tb/Ca-Tb/La abundance ratios to problems of fluorspar genesis. Miner. Depos. 1976, 11, 111–116. [Google Scholar] [CrossRef]
- Zhong, S.; Mucci, A. Partitioning of rare earth elements (REEs) between calcite and seawater solutions at 25 C and 1 atm, and high dissolved REE concentrations. Geochim. Cosmochim. Acta 1995, 59, 443–453. [Google Scholar] [CrossRef]
- Lottermoser, B. Rare earth elements and hydrothermal ore formation processes. Ore Geol. Rev. 1992, 7, 25–41. [Google Scholar] [CrossRef]
- Haas, J.R.; Shock, E.L.; Sassani, D.C. Rare earth elements in hydrothermal systems: Estimates of standard partial molal thermodynamic properties of aqueous complexes of the rare earth elements at high pressures and temperatures. Geochim. Cosmochim. Acta 1995, 59, 4329–4350. [Google Scholar] [CrossRef]
- Migdisov, A.; Williams-Jones, A.; Brugger, J.; Caporuscio, F.A. Hydrothermal transport, deposition, and fractionation of the REE: Experimental data and thermodynamic calculations. Chem. Geol. 2016, 439, 13–42. [Google Scholar] [CrossRef]
- Machel, H.G. Fibrous gypsum and fibrous anhydrite in veins. Sedimentology 1985, 32, 443–454. [Google Scholar] [CrossRef]
- Al-Aasm, I.S.; Muir, I.; Morad, S. Diagenetic conditions of fibrous calcite vein formation in black shales: Petrographic, chemical and isotopic evidence. Bull. Can. Pet. Geol. 1993, 41, 46–56. [Google Scholar]
- Badertscher, N.P.; Beaudoin, G.; Therrien, R.; Burkhard, M. Glarus overthrust: A major pathway for the escape of fluids out of the Alpine orogen. Geology 2002, 30, 875–878. [Google Scholar] [CrossRef]
- Parnell, J.; Honghan, C.; Middleton, D.; Haggan, T.; Carey, P. Significance of fibrous mineral veins in hydrocarbon migration: Fluid inclusion studies. J. Geochem. Explor. 2000, 69, 623–627. [Google Scholar] [CrossRef]
- Bons, P.D.; Montenari, M. The formation of antitaxial calcite veins with well-developed fibres, Oppaminda Creek, South Australia. J. Struct. Geol. 2005, 27, 231–248. [Google Scholar] [CrossRef]
- Bons, P.D.; Elburg, M.A.; Gomez-Rivas, E. A review of the formation of tectonic veins and their microstructures. J. Struct. Geol. 2012, 43, 33–62. [Google Scholar] [CrossRef]
- Wang, M.; Chen, Y.; Xu, X. Progress on formation mechanism of the fibrous veins in mudstone and its implications to hydrocarbon migration. Adv. Earth Sci. 2015, 30, 1107–1118. [Google Scholar]
- Durney, D.W.; Ramsay, J.G. Incremental strains measured by syntectonic crystal growths. In Gravity and tectonics; De Jong, K.A., Scholten, R., Eds.; Wiley: New York, NY, USA, 1973; pp. 67–96. [Google Scholar]
- Ramsay, J.G. The crack-seal mechanism of rock deformation. Nature 1980, 284, 135–139. [Google Scholar] [CrossRef]
- Hilgers, C.; Urai, J.L. Microstructural observations on natural syntectonic fibrous veins: Implications for the growth process. Tectonophysics 2002, 352, 257–274. [Google Scholar] [CrossRef]




| NaO | MgO | SiO2 | CaO | MnO | CuO | SrO | |
|---|---|---|---|---|---|---|---|
| S1-green-1 | 0.38 | 0.49 | 0.21 | 52.56 | 1.62 | 0.32 | 0.36 |
| S1-green-2 | 0.40 | 0.47 | 0.21 | 52.45 | 1.65 | 0.34 | 0.36 |
| S1-green-3 | 0.38 | 0.47 | 0.26 | 52.43 | 1.68 | 0.35 | 0.36 |
| Average | 0.39 | 0.48 | 0.24 | 52.48 | 1.65 | 0.33 | 0.36 |
| S2-green-1 | 0.36 | 0.51 | 0.30 | 52.44 | 1.63 | 0.30 | 0.36 |
| S2-green-2 | 0.35 | 0.47 | 0.31 | 52.29 | 1.77 | 0.36 | 0.35 |
| S2-green-3 | 0.35 | 0.47 | 0.30 | 52.38 | 1.72 | 0.34 | 0.36 |
| Average | 0.35 | 0.48 | 0.30 | 52.37 | 1.71 | 0.33 | 0.36 |
| S3-green-1 | 0.35 | 0.49 | 0.31 | 52.57 | 1.54 | 0.29 | 0.36 |
| S3-green-2 | 0.36 | 0.50 | 0.31 | 52.44 | 1.62 | 0.28 | 0.37 |
| S3-green-3 | 0.35 | 0.48 | 0.31 | 52.33 | 1.73 | 0.35 | 0.35 |
| Average | 0.35 | 0.49 | 0.31 | 52.45 | 1.63 | 0.31 | 0.36 |
| S1-white-1 | 0.38 | 0.56 | 0.21 | 53.00 | 1.16 | 0.19 | 0.40 |
| S1-white-2 | 0.40 | 0.57 | 0.21 | 52.84 | 1.27 | 0.21 | 0.39 |
| S1-white-3 | 0.42 | 0.58 | 0.26 | 52.85 | 1.19 | 0.18 | 0.39 |
| Average | 0.40 | 0.57 | 0.24 | 52.90 | 1.20 | 0.19 | 0.39 |
| S2-white-1 | 0.36 | 0.56 | 0.30 | 53.02 | 1.14 | 0.17 | 0.38 |
| S2-white-2 | 0.37 | 0.59 | 0.30 | 52.98 | 1.17 | 0.17 | 0.37 |
| S2-white-3 | 0.37 | 0.59 | 0.32 | 52.92 | 1.17 | 0.17 | 0.38 |
| Average | 0.37 | 0.58 | 0.31 | 52.98 | 1.16 | 0.17 | 0.38 |
| S3-white-1 | 0.37 | 0.57 | 0.32 | 52.96 | 1.18 | 0.17 | 0.37 |
| S3-white-2 | 0.36 | 0.57 | 0.31 | 53.04 | 1.14 | 0.16 | 0.37 |
| S3-white-3 | 0.36 | 0.57 | 0.33 | 53.11 | 1.08 | 0.15 | 0.36 |
| Average | 0.36 | 0.57 | 0.32 | 53.04 | 1.14 | 0.15 | 0.37 |
| V | Cr | Fe | Co | Ni | Zn | |
|---|---|---|---|---|---|---|
| S1-green-1 | 0.34 | 0.51 | 185.88 | 38.88 | 3.20 | 399.46 |
| S1-green-2 | 0.26 | 0.76 | 204.03 | 38.36 | 3.96 | 416.37 |
| S1-green-3 | 0.28 | 0.78 | 237.38 | 39.29 | 3.22 | 453.73 |
| Average | 0.29 | 0.68 | 209.10 | 38.84 | 3.46 | 423.19 |
| S2-green-1 | 0.18 | 0.46 | 218.61 | 36.80 | 2.98 | 407.60 |
| S2-green-2 | 0.24 | 0.12 | 268.16 | 36.89 | 3.70 | 471.20 |
| S2-green-3 | 0.29 | 0.00 | 233.86 | 36.55 | 1.71 | 400.73 |
| Average | 0.24 | 0.20 | 240.21 | 36.75 | 2.80 | 426.51 |
| S3-green-1 | 0.31 | 0.00 | 223.69 | 34.95 | 2.30 | 382.36 |
| S3-green-2 | 0.29 | 0.00 | 231.72 | 37.30 | 3.44 | 375.34 |
| S3-green-3 | 0.28 | 0.00 | 276.33 | 36.38 | 1.80 | 443.82 |
| Average | 0.29 | 0.00 | 243.91 | 37.30 | 2.51 | 400.51 |
| S1-white-1 | 0.21 | 0.74 | 135.69 | 34.99 | 3.47 | 226.68 |
| S1-white-2 | 0.30 | 1.40 | 151.05 | 35.70 | 2.63 | 251.09 |
| S1-white-3 | 0.25 | 0.46 | 109.16 | 35.07 | 3.62 | 220.96 |
| Average | 0.25 | 0.87 | 131.97 | 35.25 | 3.24 | 232.91 |
| S2-white-1 | 0.26 | 0.99 | 165.77 | 36.05 | 3.23 | 211.17 |
| S2-white-2 | 0.17 | 0.74 | 112.69 | 37.96 | 2.95 | 232.60 |
| S2-white-3 | 0.18 | 0.00 | 137.22 | 38.77 | 3.07 | 242.52 |
| Average | 0.20 | 0.58 | 138.56 | 37.59 | 3.08 | 228.76 |
| S3-white-1 | 0.29 | 1.28 | 167.06 | 36.46 | 2.29 | 243.10 |
| S3-white-2 | 0.27 | 0.00 | 124.00 | 36.97 | 2.78 | 241.07 |
| S3-white-3 | 0.25 | 0.00 | 117.14 | 36.46 | 1.54 | 255.99 |
| Average | 0.27 | 0.43 | 136.07 | 36.63 | 2.20 | 246.72 |
| The Number of Cations Based on Of.u. = 3 | Stoichiometric Number | |||||||
|---|---|---|---|---|---|---|---|---|
| S1-green | S2-green | S3-green | S1-white | S2-white | S3-white | Total Average | ||
| Mg2+ | 0.01 | 0.01 | 0.01 | 0.01 | 0.01 | 0.01 | 0.01 | 1.00 |
| Ca2+ | 0.96 | 0.96 | 0.96 | 0.97 | 0.97 | 0.97 | 0.97 | |
| Mn2+ | 0.02 | 0.02 | 0.02 | 0.02 | 0.02 | 0.02 | 0.02 | |
| C4+ | 1.00 | 1.00 | 1.00 | 1.00 | 1.00 | 1.00 | 1.00 | 1.00 |
| S1-green-1 | S1-green-2 | S1-green-3 | S2-green-1 | S2-green-2 | S2-green-3 | S3-green-1 | S3-green-2 | S3-green-3 | Average | ||
| La | 3.36 | 3.64 | 4.14 | 3.33 | 3.74 | 3.16 | 3.27 | 3.48 | 3.68 | 3.53 | |
| Ce | 8.20 | 9.23 | 10.40 | 7.84 | 8.92 | 7.87 | 8.64 | 8.32 | 9.04 | 8.72 | |
| Pr | 0.81 | 0.94 | 0.94 | 0.77 | 0.83 | 0.80 | 0.90 | 0.81 | 0.78 | 0.84 | |
| Nd | 4.05 | 4.66 | 5.06 | 4.15 | 4.42 | 3.80 | 4.29 | 3.83 | 4.34 | 4.29 | |
| Sm | 1.00 | 1.25 | 1.31 | 0.97 | 1.31 | 1.24 | 1.18 | 1.02 | 1.51 | 1.20 | |
| Eu | 0.38 | 0.51 | 0.50 | 0.51 | 0.48 | 0.38 | 0.43 | 0.40 | 0.48 | 0.45 | |
| Gd | 1.95 | 2.14 | 2.67 | 1.90 | 2.02 | 2.29 | 1.99 | 2.29 | 2.06 | 2.14 | |
| Tb | 0.39 | 0.40 | 0.44 | 0.37 | 0.40 | 0.37 | 0.42 | 0.37 | 0.41 | 0.40 | |
| Dy | 2.96 | 3.30 | 3.41 | 2.68 | 3.11 | 2.90 | 2.42 | 2.50 | 3.04 | 2.92 | |
| Ho | 0.79 | 0.82 | 0.94 | 0.68 | 0.76 | 0.74 | 0.77 | 0.69 | 0.78 | 0.77 | |
| Er | 2.46 | 2.62 | 2.71 | 2.29 | 2.36 | 2.12 | 2.38 | 2.08 | 2.65 | 2.41 | |
| Tm | 0.35 | 0.35 | 0.38 | 0.32 | 0.32 | 0.35 | 0.33 | 0.36 | 0.33 | 0.34 | |
| Yb | 2.46 | 2.50 | 2.72 | 2.27 | 2.54 | 2.49 | 2.11 | 2.26 | 2.35 | 2.41 | |
| Lu | 0.33 | 0.30 | 0.37 | 0.33 | 0.37 | 0.32 | 0.33 | 0.26 | 0.35 | 0.33 | |
| ΣREE | 29.49 | 32.66 | 35.98 | 28.42 | 31.57 | 28.83 | 29.46 | 28.67 | 31.80 | 30.76 | |
| δCe | 1.20 | 1.21 | 1.27 | 1.18 | 1.23 | 1.19 | 1.22 | 1.20 | 1.29 | ||
| δEu | 0.84 | 0.95 | 0.81 | 1.14 | 0.90 | 0.69 | 0.85 | 0.81 | 0.83 | ||
| S1-white-1 | S1-white-2 | S1-white-3 | S2-white-1 | S2-white-2 | S2-white-3 | S3-white-1 | S3-white-2 | S3-white-3 | Average | Chondrite * | |
| La | 2.03 | 2.24 | 1.72 | 2.46 | 2.26 | 2.64 | 2.36 | 1.97 | 2.00 | 2.19 | 0.24 |
| Ce | 4.93 | 5.59 | 4.05 | 5.87 | 5.65 | 6.64 | 6.14 | 5.21 | 5.39 | 5.50 | 0.61 |
| Pr | 0.45 | 0.58 | 0.38 | 0.57 | 0.57 | 0.63 | 0.61 | 0.74 | 0.66 | 0.58 | 0.09 |
| Nd | 2.41 | 2.63 | 1.80 | 2.74 | 2.76 | 3.42 | 2.99 | 2.45 | 2.53 | 2.64 | 0.46 |
| Sm | 0.60 | 0.81 | 0.65 | 1.00 | 0.80 | 1.01 | 0.90 | 0.88 | 0.97 | 0.85 | 0.15 |
| Eu | 0.28 | 0.24 | 0.21 | 0.27 | 0.31 | 0.34 | 0.25 | 0.33 | 0.30 | 0.28 | 0.06 |
| Gd | 1.19 | 1.27 | 0.86 | 1.46 | 1.28 | 1.65 | 1.51 | 1.48 | 1.44 | 1.35 | 0.20 |
| Tb | 0.20 | 0.25 | 0.19 | 0.24 | 0.24 | 0.24 | 0.23 | 0.35 | 0.32 | 0.25 | 0.04 |
| Dy | 1.73 | 1.70 | 1.55 | 2.06 | 1.91 | 2.49 | 2.18 | 2.14 | 2.06 | 1.98 | 0.25 |
| Ho | 0.41 | 0.50 | 0.35 | 0.49 | 0.52 | 0.58 | 0.59 | 0.54 | 0.50 | 0.50 | 0.05 |
| Er | 1.33 | 1.58 | 1.09 | 1.68 | 1.56 | 1.86 | 1.68 | 1.47 | 1.06 | 1.48 | 0.16 |
| Tm | 0.20 | 0.22 | 0.17 | 0.27 | 0.24 | 0.27 | 0.30 | 0.25 | 0.28 | 0.24 | 0.02 |
| Yb | 1.26 | 1.49 | 1.13 | 1.75 | 1.63 | 1.83 | 1.75 | 1.58 | 1.63 | 1.56 | 0.16 |
| Lu | 0.23 | 0.21 | 0.17 | 0.26 | 0.29 | 0.25 | 0.25 | 0.26 | 0.28 | 0.25 | 0.02 |
| ΣREE | 17.24 | 19.31 | 14.32 | 21.11 | 20.01 | 23.87 | 21.73 | 19.64 | 19.42 | 19.63 | |
| δCe | 1.24 | 1.18 | 1.22 | 1.20 | 1.20 | 1.25 | 1.24 | 1.04 | 1.13 | ||
| δEu | 1.02 | 0.72 | 0.86 | 0.68 | 0.94 | 0.81 | 0.65 | 0.89 | 0.77 | ||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Liu, Z.; Wang, W.; Yin, K.; Hong, H.; Algeo, T.J.; Yin, Z.; Pan, Y.; Lu, Z.; Han, W.; Wang, Y.; et al. Gemological Characteristics of Lvwen Stone and Its Color Genesis. Minerals 2022, 12, 1584. https://doi.org/10.3390/min12121584
Liu Z, Wang W, Yin K, Hong H, Algeo TJ, Yin Z, Pan Y, Lu Z, Han W, Wang Y, et al. Gemological Characteristics of Lvwen Stone and Its Color Genesis. Minerals. 2022; 12(12):1584. https://doi.org/10.3390/min12121584
Chicago/Turabian StyleLiu, Zhendong, Wenjie Wang, Ke Yin, Hanlie Hong, Thomas J. Algeo, Zuowei Yin, Yong Pan, Zhuo Lu, Wen Han, Yiming Wang, and et al. 2022. "Gemological Characteristics of Lvwen Stone and Its Color Genesis" Minerals 12, no. 12: 1584. https://doi.org/10.3390/min12121584
APA StyleLiu, Z., Wang, W., Yin, K., Hong, H., Algeo, T. J., Yin, Z., Pan, Y., Lu, Z., Han, W., Wang, Y., & Yang, Y. (2022). Gemological Characteristics of Lvwen Stone and Its Color Genesis. Minerals, 12(12), 1584. https://doi.org/10.3390/min12121584







