Gypsum Amendment Induced Rapid Pyritization in Fe-Rich Mine Tailings from Doce River Estuary after the Fundão Dam Collapse
Abstract
1. Introduction
2. Materials and Methods
2.1. Site Characterization
2.2. Characterization of Mine Tailings and Amendments
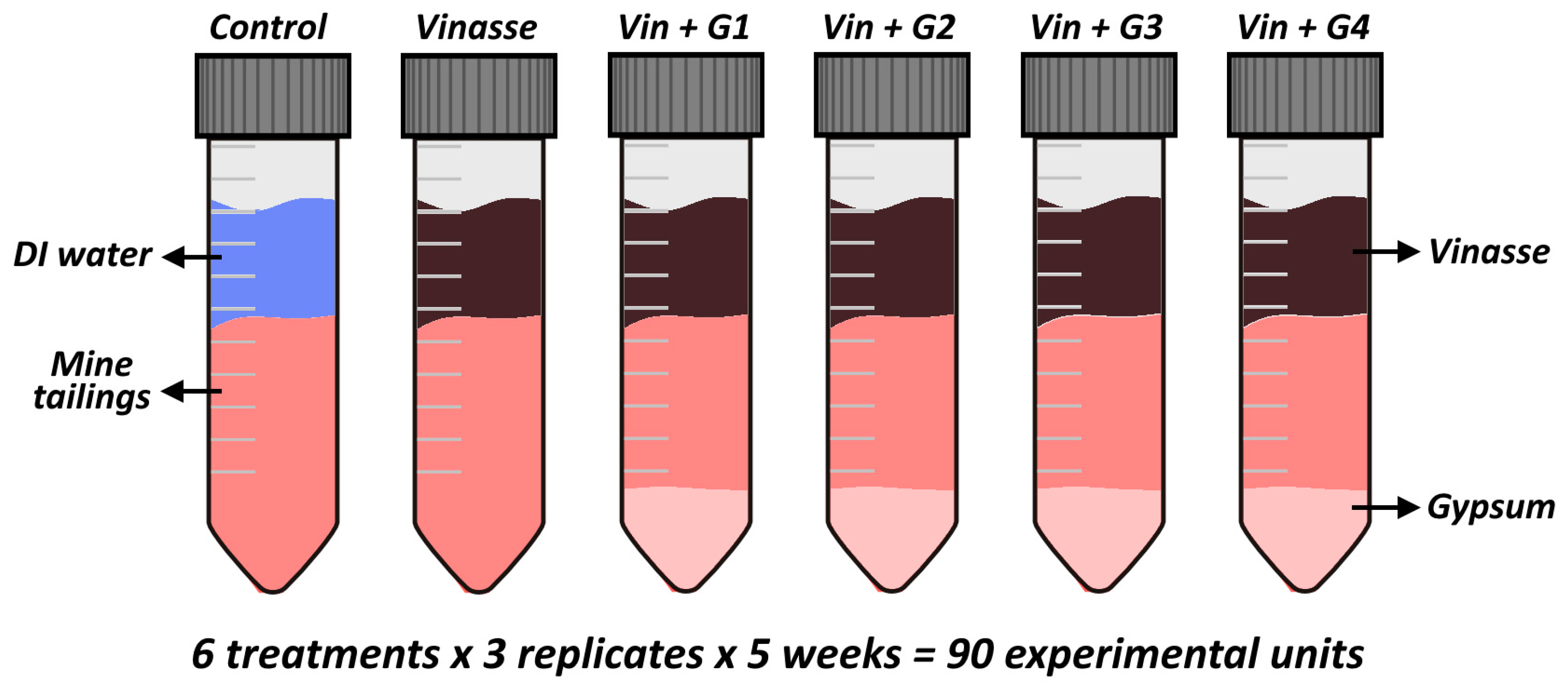
2.3. Experimental Design
- -
- Control: only deionized water (0 mg of S);
- -
- Vinasse: only 6.2 mL of vinasse addition (as a carbon source: 51.7 mg of C, but with 2.4 mg of S);
- -
- Vin + G1: 6.2 mL of vinasse + 0.5 g of gypsum (89.7 mg of S);
- -
- Vin + G2: 6.2 mL of vinasse + 2.5 g of gypsum (439 mg of S);
- -
- Vin + G3: 6.2 mL of vinasse + 5 g of gypsum (875 mg of S);
- -
- Vin + G4: 6.2 mL of vinasse + 10 g of gypsum (1748 mg of S).
2.4. Aqueous and Solid-Phase Analysis
2.5. Statistical Analysis
3. Results and Discussion
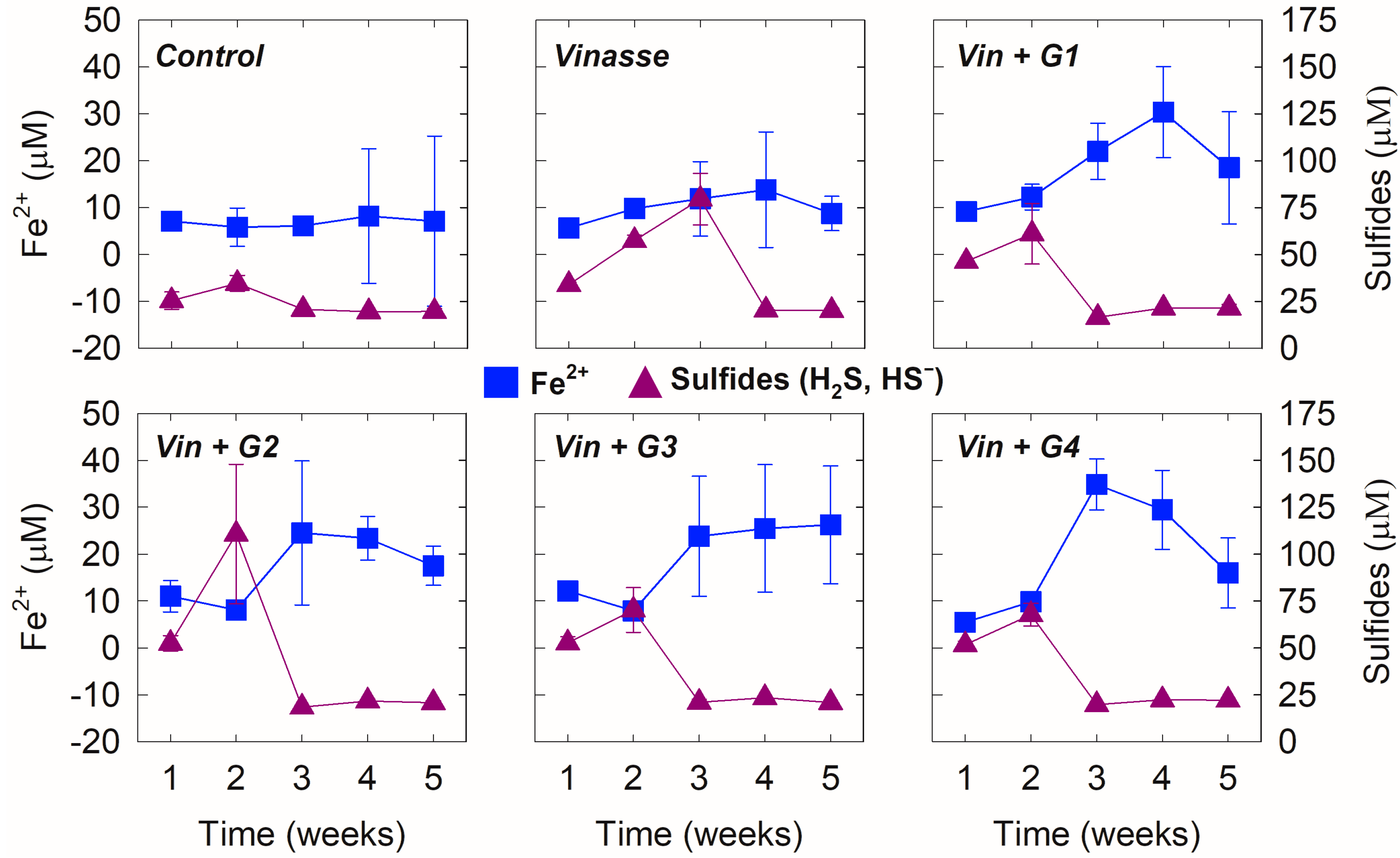
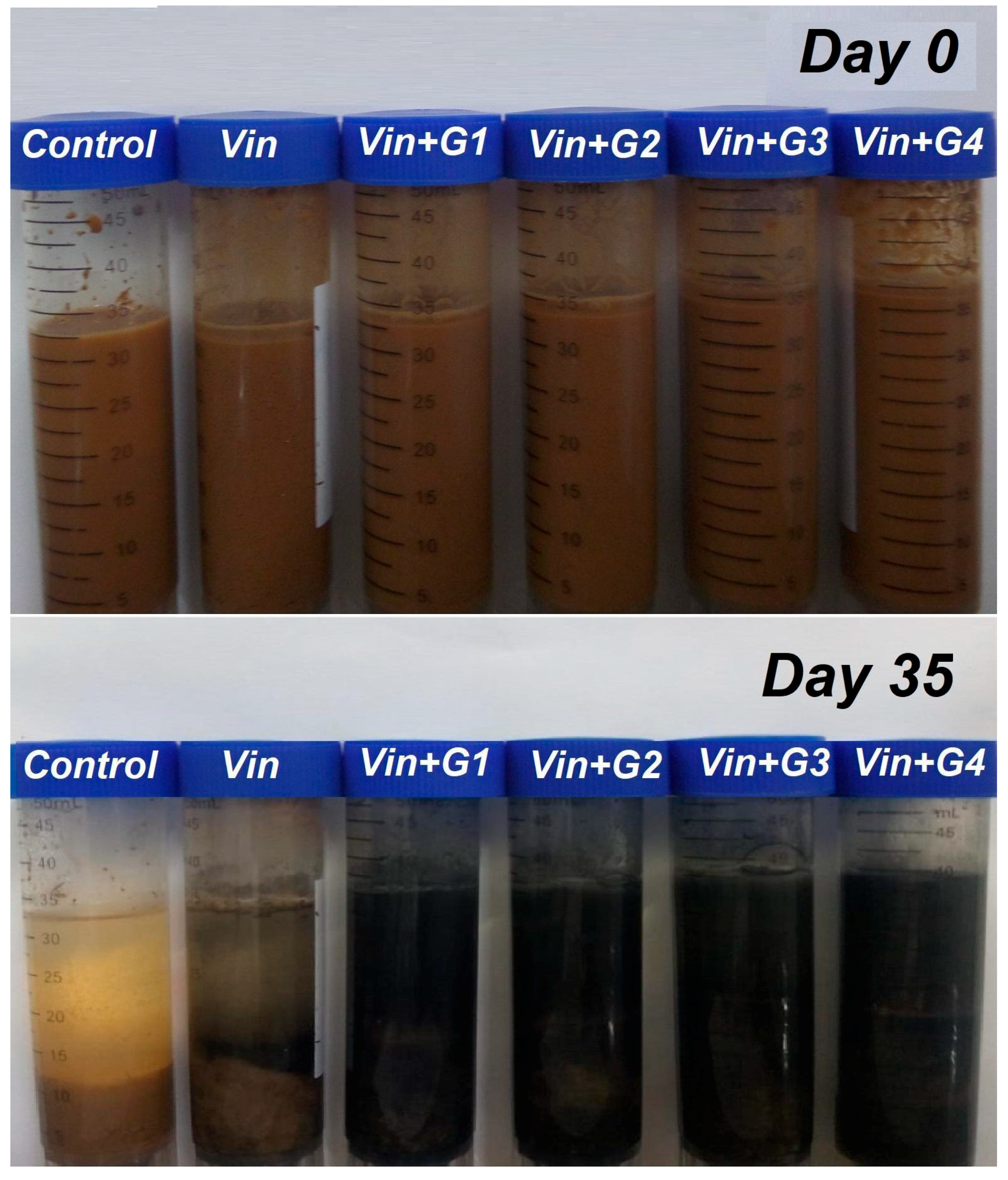
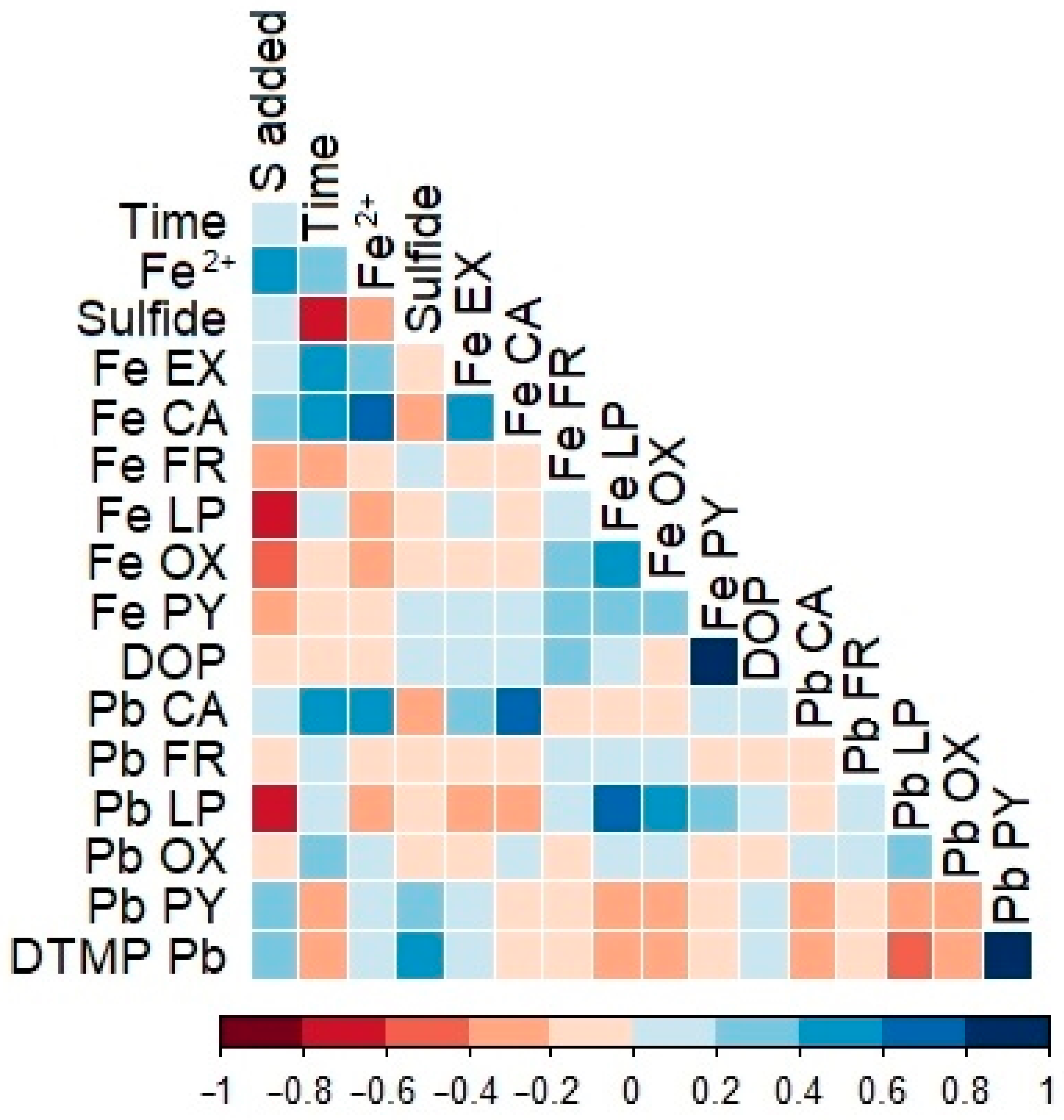
3.1. Effects of Time and Amendment Dose
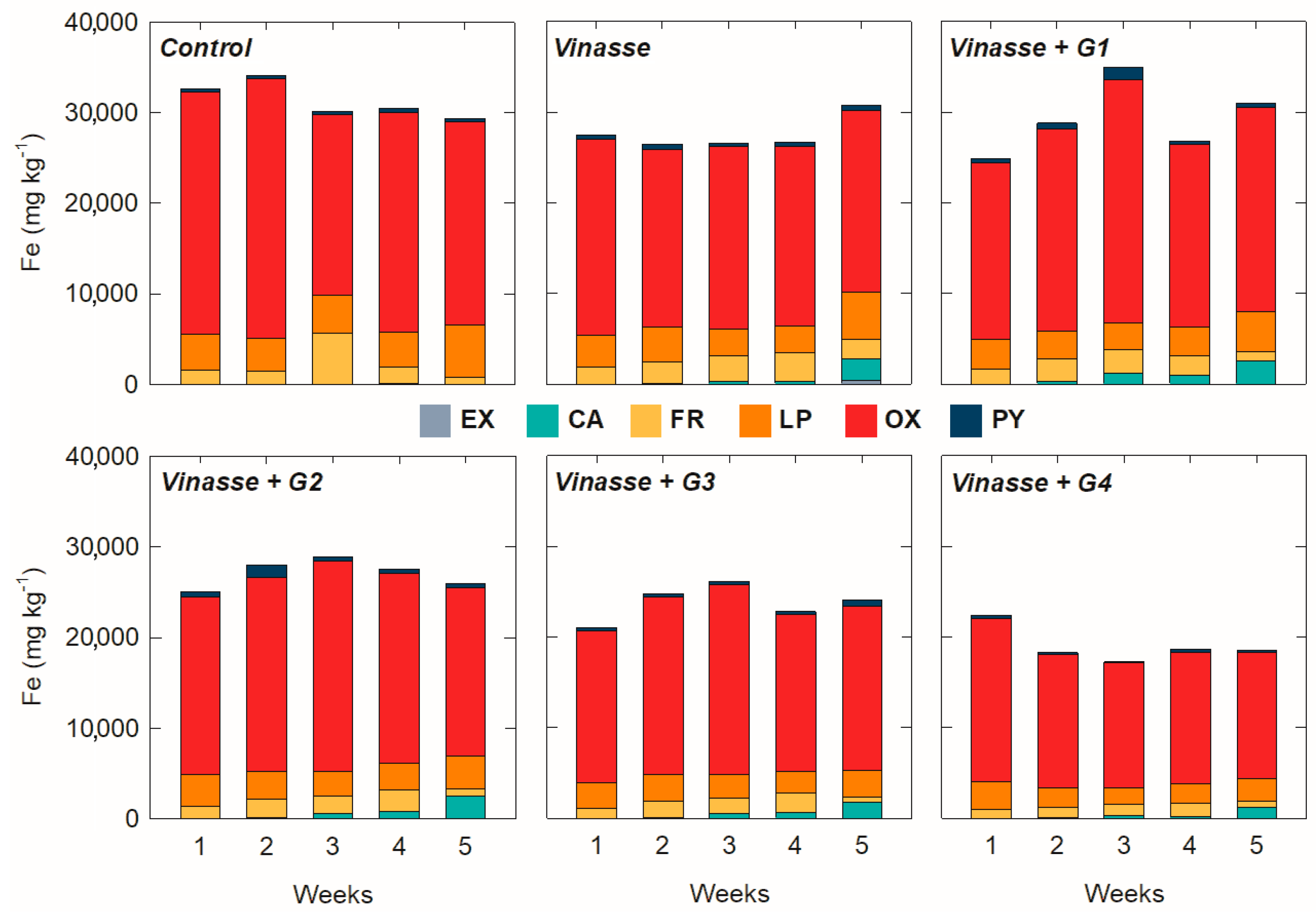
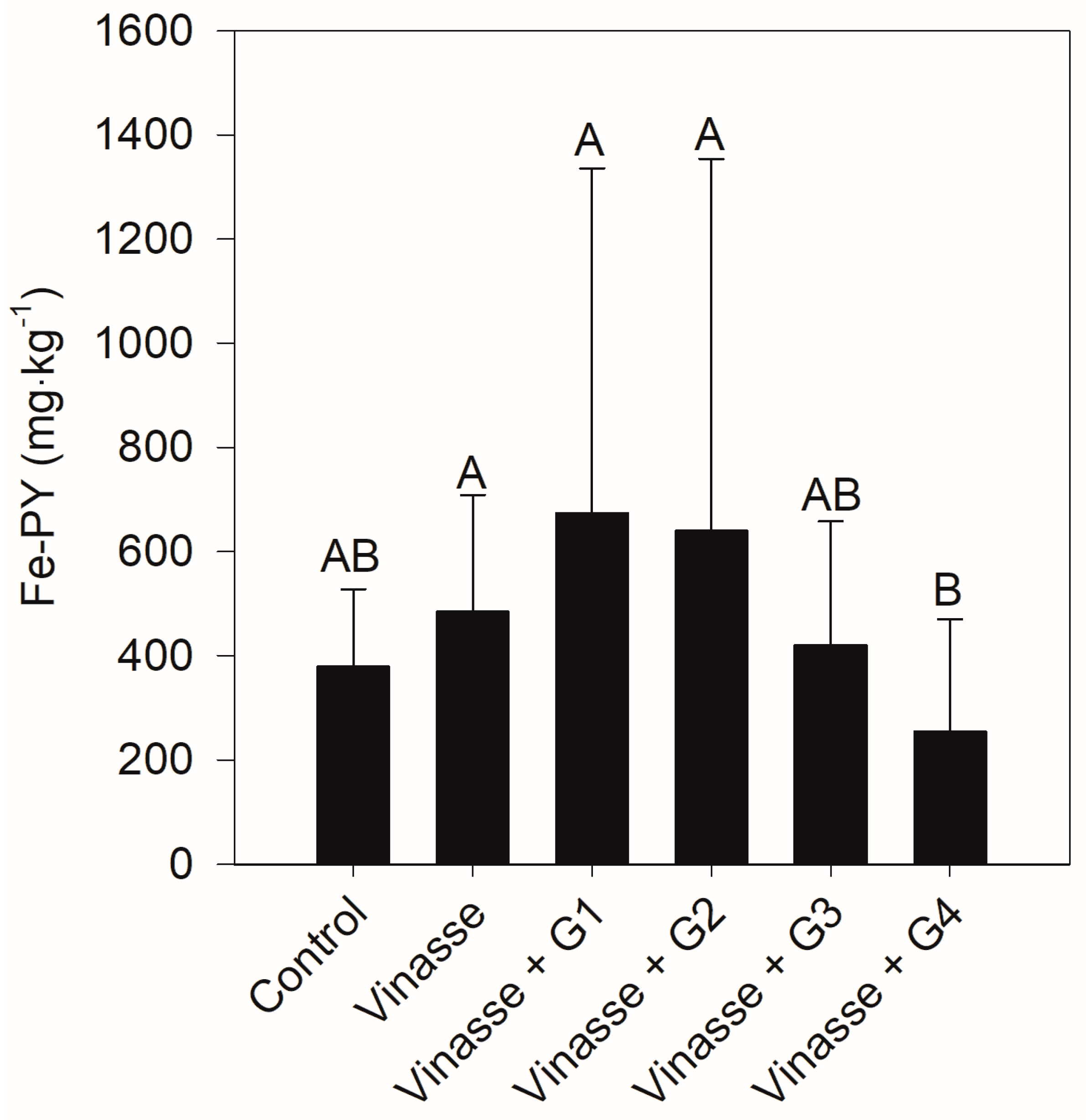
3.2. Variation of Solid-Phase Content over Time and as a Function of Amendment Dose
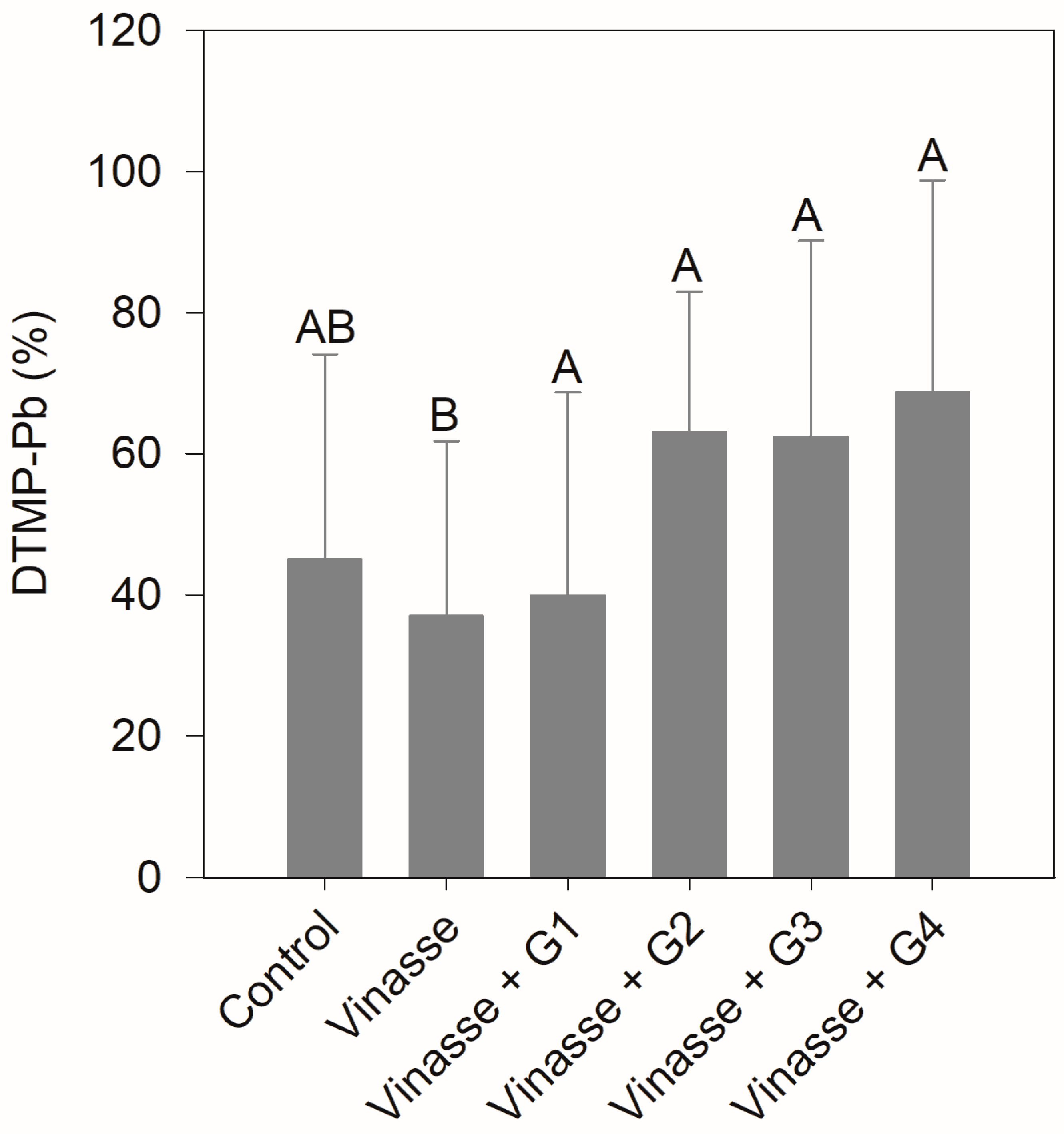
3.3. Pb Pyritization and Modeling
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Nóbrega, G.N.; Ferreira, T.O.; Neto, M.S.; Queiroz, H.M.; Artur, A.G.; Mendonça, E.D.S.; Silva, E.D.O.; Otero, X.L. Edaphic factors controlling summer (rainy season) greenhouse gas emissions (CO2 and CH4) from semiarid mangrove soils (NE-Brazil). Sci. Total. Environ. 2016, 542, 685–693. [Google Scholar] [CrossRef]
- Machado, W.; Borrelli, N.; Ferreira, T.O.; Marques, A.; Osterrieth, M.; Guizan, C. Trace metal pyritization variability in response to mangrove soil aerobic and anaerobic oxidation processes. Mar. Pollut. Bull. 2014, 79, 365–370. [Google Scholar] [CrossRef]
- Otero, X.L.; Ferreira, T.O.; Huerta-Diaz, M.A.; Partiti, C.S.D.M.; Souza, V.; Torrado, P.V.; Macías, F. Geochemistry of iron and manganese in soils and sediments of a mangrove system, Island of Pai Matos (Cananeia—SP, Brazil). Geoderma 2009, 148, 318–335. [Google Scholar] [CrossRef]
- Andrade, R.A.; Sanders, C.J.; Boaventura, G.; Patchineelam, S.R. Pyritization of trace metals in mangrove sediments. Environ. Earth Sci. 2012, 67, 1757–1762. [Google Scholar] [CrossRef]
- Berner, R.A. Sedimentary pyrite formation. Am. J. Sci. 1970, 268, 1–23. [Google Scholar] [CrossRef]
- Huerta-Diaz, M.A.; Morse, J.W. Pyritization of trace metals in anoxic marine sediments. Geochim. Cosmochim. Acta 1992, 56, 2681–2702. [Google Scholar] [CrossRef]
- Rickard, D.; Morse, J.W. Acid volatile sulfide (AVS). Mar. Chem. 2005, 97, 141–197. [Google Scholar] [CrossRef]
- Ferreira, T.O.; Nóbrega, G.N.; Albuquerque, A.G.B.M.; Sartor, L.R.; Gomes, I.S.; Artur, A.G.; Otero, X. Pyrite as a proxy for the identification of former coastal lagoons in semiarid NE Brazil. Geo-Mar. Lett. 2015, 35, 355–366. [Google Scholar] [CrossRef]
- Alongi, D.; Wattayakorn, G.; Boyle, S.; Tirendi, F.; Payn, C.; Dixon, P. Influence of roots and climate on mineral and trace element storage and flux in tropical mangrove soils. Biogeochemistry 2004, 69, 105–123. [Google Scholar] [CrossRef]
- Hellige, K.; Pollok, K.; Larese-Casanova, P.; Behrends, T.; Peiffer, S. Pathways of ferrous iron mineral formation upon sulfidation of lepidocrocite surfaces. Geochim. Cosmochim. Acta 2012, 81, 69–81. [Google Scholar] [CrossRef]
- Devolder, P.S.; Brown, S.L.; Hesterberg, D.; Pandya, K. Metal Bioavailability and Speciation in a Wetland Tailings Repository Amended with Biosolids Compost, Wood Ash, and Sulfate. J. Environ. Qual. 2003, 32, 851–864. [Google Scholar] [CrossRef] [PubMed]
- Berkowitz, J.F.; Vanzomeren, C.M.; Fresard, N.D. Rapid formation of iron sulfides alters soil morphology and chemistry following simulated marsh restoration. Geoderma 2019, 351, 76–84. [Google Scholar] [CrossRef]
- Carmo, F.F.D.; Kamino, L.H.Y.; Junior, R.T.; De Campos, I.C.; Carmo, F.F.D.; Silvino, G.; Castro, K.J.D.S.X.D.; Mauro, M.L.; Rodrigues, N.U.A.; Miranda, M.P.D.S.; et al. Fundão tailings dam failures: The environment tragedy of the largest technological disaster of Brazilian mining in global context. Perspect. Ecol. Conserv. 2017, 15, 145–151. [Google Scholar] [CrossRef]
- Queiroz, H.M.; Nóbrega, G.N.; Ferreira, T.O.; Almeida, L.S.; Romero, T.B.; Santaella, S.T.; Bernardino, A.F.; Otero, X.L. The Samarco mine tailing disaster: A possible time-bomb for heavy metals contamination? Sci. Total. Environ. 2018, 498–506. [Google Scholar] [CrossRef] [PubMed]
- Gomes, L.E.D.O.; Correa, L.B.; Sá, F.; Neto, R.R.; Bernardino, A.F. The impacts of the Samarco mine tailing spill on the Rio Doce estuary, Eastern Brazil. Mar. Pollut. Bull. 2017, 120, 28–36. [Google Scholar] [CrossRef]
- Gabriel, F.A.; Silva, A.G.; Queiroz, H.M.; Ferreira, T.O.; Hauser-Davis, R.A.; Bernardino, A.F. Ecological Risks of Metal and Metalloid Contamination in the Rio Doce Estuary. Integr. Environ. Assess. Manag. 2020, 16, 655–660. [Google Scholar] [CrossRef]
- Bernardino, A.F.; Pais, F.S.; Oliveira, L.S.; Gabriel, F.A.; Ferreira, T.O.; Queiroz, H.M.; Mazzuco, A.C.A. Chronic trace metals effects of mine tailings on estuarine assemblages revealed by environmental DNA. PeerJ 2019, 7, e8042. [Google Scholar] [CrossRef]
- Queiroz, H.M.; Ferreira, T.O.; Barcellos, D.; Nóbrega, G.N.; Antelo, J.; Otero, X.L.; Bernardino, A.F. From sinks to sources: The role of Fe oxyhydroxide transformations on phosphorus dynamics in estuarine soils. J. Environ. Manag. 2021, 278, 111575. [Google Scholar] [CrossRef]
- Burton, J.G.A. Sediment quality criteria in use around the world. Limnology 2002, 3, 65–76. [Google Scholar] [CrossRef]
- Lwin, C.S.; Seo, B.-H.; Kim, H.-U.; Owens, G.; Kim, K.-R. Application of soil amendments to contaminated soils for heavy metal immobilization and improved soil quality—A critical review. Soil Sci. Plant Nutr. 2018, 64, 156–167. [Google Scholar] [CrossRef]
- Andrunik, M.; Wołowiec, M.; Wojnarski, D.; Zelek-Pogudz, S.; Bajda, T. Transformation of Pb, Cd, and Zn Minerals Using Phosphates. Minerals 2020, 10, 342. [Google Scholar] [CrossRef]
- Guo, G.; Zhou, Q.; Ma, L.Q. Availability and Assessment of Fixing Additives for The in Situ Remediation of Heavy Metal Contaminated Soils: A Review. Environ. Monit. Assess. 2006, 116, 513–528. [Google Scholar] [CrossRef] [PubMed]
- Evans, K. The History, Challenges, and New Developments in the Management and Use of Bauxite Residue. J. Sustain. Met. 2016, 2, 316–331. [Google Scholar] [CrossRef]
- Mahmoodabadi, M.; Yazdanpanah, N.; Sinobas, L.R.; Pazira, E.; Neshat, A. Reclamation of calcareous saline sodic soil with different amendments (I): Redistribution of soluble cations within the soil profile. Agric. Water Manag. 2013, 120, 30–38. [Google Scholar] [CrossRef]
- Seixas, F.L.; Gimenes, M.L.; Fernandes, N.R. Treatment of vinasse by adsorption on carbon from sugar cane bagasse. Química Nova 2016, 39, 172–179. [Google Scholar] [CrossRef]
- Gee, G.W.; Bauder, J.W. Particle-Size Analysis. In Methods of Soil Analysis: Part 1—Physical and Mineralogical Methods; Soil Science Society of America, American Society of Agronomy: Madison, WI, USA, 1986; pp. 383–411. [Google Scholar]
- Howard, J.; Hoyt, S.; Isensee, K.; Pidgeon, E. Maciej Telszewski Coastal Blue Carbon: Methods for Assessing Carbon Stocks and Emissions Factors in Mangroves, Tidal Salt Marshes, and Seagrass Meadows, 1st ed.; International Union for Conservation of Nature (IUCN): Arlington, VA, USA, 2014. [Google Scholar]
- USEPA. Method 3052: Microwave Assisted Acid Digestion of Siliceous and Organically Based Matrices; United States Environmental Protection Agency: Washington, DC, USA, 1996.
- USEPA. Method 6010C Inductively Coupled Plasma-Atomic Emission Spectometry; United States Environmental Protection Agency: Washington, DC, USA, 1997; Volume 2, pp. 1–19.
- MAPA, Instrução Normativa n. 027 de 05 de Junho de 2006. Dispõe Sobre Fertilizantes, Corretivos, Inoculantes e Biofertilizantes, Para Serem Produzidos, Importados Ou Comercializados, Deverão Atender Aos Limites Estabelecidos Nesta Instrução; Esplanada dos Ministérios Bloco D-Brasília: Brasília Brazil, 2006.
- Viollier, E.; Inglett, P.; Hunter, K.; Roychoudhury, A.; Van Cappellen, P. The ferrozine method revisited: Fe(II)/Fe(III) determination in natural waters. Appl. Geochem. 2000, 15, 785–790. [Google Scholar] [CrossRef]
- Cline, J.D. Spectrophotometric determination of hydrogen sulfide in natural waters. Limnol. Oceanogr. 1969, 14, 454–458. [Google Scholar] [CrossRef]
- Huerta-Diaz, M.A.; Morse, J.W. A quantitative method for determination of trace metal concentrations in sedimentary pyrite. Mar. Chem. 1990, 29, 119–144. [Google Scholar] [CrossRef]
- Fortin, D.; Leppard, G.G.; Tessier, A. Characteristics of lacustrine diagenetic iron oxyhydroxides. Geochim. Cosmochim. Acta 1993, 57, 4391–4404. [Google Scholar] [CrossRef]
- Tessier, A.; Campbell, P.G.C.; Bisson, M. Sequential extraction procedure for the speciation of particulate trace metals. Anal. Chem. 1979, 51, 844–851. [Google Scholar] [CrossRef]
- Ferreira, T.O.; Otero, X.L.; Vidal-Torrado, P.; Macías, F. Redox Processes in Mangrove Soils under Rhizophora mangle in Relation to Different Environmental Conditions. Soil Sci. Soc. Am. J. 2007, 71, 484–491. [Google Scholar] [CrossRef]
- Reimann, C.; Filzmoser, P.; Garrett, R.G.; Dutter, R. Statistical Data Analysis Explained; John Wiley & Sons, Ltd.: Chichester, UK, 2008; ISBN 9780470987605. [Google Scholar]
- RStudio Team; RStudio: Boston, MA, USA, 2020.
- Peiffer, S.; Behrends, T.; Hellige, K.; Larese-Casanova, P.; Wan, M.; Pollok, K. Pyrite formation and mineral transformation pathways upon sulfidation of ferric hydroxides depend on mineral type and sulfide concentration. Chem. Geol. 2015, 400, 44–55. [Google Scholar] [CrossRef]
- Wan, M.; Schröder, C.; Peiffer, S. Fe(III):S(-II) concentration ratio controls the pathway and the kinetics of pyrite formation during sulfidation of ferric hydroxides. Geochim. Cosmochim. Acta 2017, 217, 334–348. [Google Scholar] [CrossRef]
- Kumar, N.; Lezama-Pacheco, J.; Noël, V.; Dublet, G.; Brown, G.E. Sulfidation mechanisms of Fe(iii)-(oxyhydr)oxide nanoparticles: A spectroscopic study. Environ. Sci. Nano 2018, 5, 1012–1026. [Google Scholar] [CrossRef]
- Burton, E.D.; Bush, R.T.; Sullivan, L.A. Fractionation and extractability of sulfur, iron and trace elements in sulfidic sediments. Chemosphere 2006, 64, 1421–1428. [Google Scholar] [CrossRef] [PubMed]
- Nóbrega, G.N.; Ferreira, T.O.; Romero, R.E.; Marques, A.G.B.; Otero, X. Iron and sulfur geochemistry in semi-arid mangrove soils (Ceará, Brazil) in relation to seasonal changes and shrimp farming effluents. Environ. Monit. Assess. 2013, 185, 7393–7407. [Google Scholar] [CrossRef] [PubMed]
- Noël, V.; Marchand, C.; Juillot, F.; Ona-Nguema, G.; Viollier, E.; Marakovic, G.; Olivi, L.; Delbes, L.; Gelebart, F.; Morin, G. EXAFS analysis of iron cycling in mangrove sediments downstream a lateritized ultramafic watershed (Vavouto Bay, New Caledonia). Geochim. Cosmochim. Acta 2014, 136, 211–228. [Google Scholar] [CrossRef]
- Lian, B.; Hu, Q.; Chen, J.; Ji, J.; Teng, H.H. Carbonate biomineralization induced by soil bacterium Bacillus megaterium. Geochim. Cosmochim. Acta 2006, 70, 5522–5535. [Google Scholar] [CrossRef]
- Dhami, N.K. Biomineralization of Calcium Carbonate Polymorphs by the Bacterial Strains Isolated from Calcareous Sites. J. Microbiol. Biotechnol. 2013, 23, 707–714. [Google Scholar] [CrossRef]
- Castanier, S.; Le Métayer-Levrel, G.; Perthuisot, J.-P. Bacterial Roles in the Precipitation of Carbonate Minerals. In Microbial Sediments; Springer: Berlin/Heidelberg, Germany, 2000; pp. 32–39. [Google Scholar]
- Sundaray, S.K.; Nayak, B.B.; Lin, S.; Bhatta, D. Geochemical speciation and risk assessment of heavy metals in the river estuarine sediments—A case study: Mahanadi basin, India. J. Hazard. Mater. 2011, 186, 1837–1846. [Google Scholar] [CrossRef]

| pH | Eh * | TOC | TN | Sand | Silt | Clay | Fe | Al |
| mV | % | |||||||
| 6.0 | 360 | 0.37 | 0.03 | 16.2 | 42.8 | 41.0 | 8.02 | 0.31 |
| Mn | Ba | Cd | Cr | Co | Cu | Ni | Pb | Zn |
| mg·kg−1 | ||||||||
| 854.92 | 95.44 | 6.03 | 20.79 | 20.08 | 24.75 | 7.88 | 68.41 | 86.27 |
| pH | C | N | P | K | Mg | Ca | S (SO2−4) | |
|---|---|---|---|---|---|---|---|---|
| g·kg−1 | ||||||||
| Gypsum | - | 223.2 | 174.6 | |||||
| Vinasse | 4.10 | 8.34 | 0.66 | 0.18 | 1.90 | 0.16 | 0.48 | 0.38 |
| Variable | AIC * | R²c ** | Model (Significant Predictors in Bold, p < 0.05) |
|---|---|---|---|
| Pb-PY | 341.04 | 0.73 | Pb-PY= 49.32 × Sulfides + 13.32 × DOP + 0.0005 × Fe-OX − 2.29 × Pb-CA − 3.93 |
| DTMP-Pb | 467.55 | 0.53 | DTMP-Pb= 248.48 × Sulfides +73.1 × DOP – 19 × Pb-CA + 9.87 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ferreira, A.D.; Queiroz, H.M.; Kaneagae, M.P.; Nóbrega, G.N.; Otero, X.L.; Bernardino, Â.F.; Ferreira, T.O. Gypsum Amendment Induced Rapid Pyritization in Fe-Rich Mine Tailings from Doce River Estuary after the Fundão Dam Collapse. Minerals 2021, 11, 201. https://doi.org/10.3390/min11020201
Ferreira AD, Queiroz HM, Kaneagae MP, Nóbrega GN, Otero XL, Bernardino ÂF, Ferreira TO. Gypsum Amendment Induced Rapid Pyritization in Fe-Rich Mine Tailings from Doce River Estuary after the Fundão Dam Collapse. Minerals. 2021; 11(2):201. https://doi.org/10.3390/min11020201
Chicago/Turabian StyleFerreira, Amanda D., Hermano M. Queiroz, Maira P. Kaneagae, Gabriel N. Nóbrega, Xosé L. Otero, Ângelo F. Bernardino, and Tiago O. Ferreira. 2021. "Gypsum Amendment Induced Rapid Pyritization in Fe-Rich Mine Tailings from Doce River Estuary after the Fundão Dam Collapse" Minerals 11, no. 2: 201. https://doi.org/10.3390/min11020201
APA StyleFerreira, A. D., Queiroz, H. M., Kaneagae, M. P., Nóbrega, G. N., Otero, X. L., Bernardino, Â. F., & Ferreira, T. O. (2021). Gypsum Amendment Induced Rapid Pyritization in Fe-Rich Mine Tailings from Doce River Estuary after the Fundão Dam Collapse. Minerals, 11(2), 201. https://doi.org/10.3390/min11020201








