1. Introduction
Ultramafic–mafic complexes are found everywhere in the world. They include various types of mineral deposits: Ni-Cu and PGE [
1], Cr [
2], Fe-Ti-V [
3], asbestos [
4], nephrite and jadeite [
5], talc, magnesite, vermiculate [
6] and others. Kimberlite and lamproite pipes contain diamonds [
7,
8].
In their natural state, the complexes do not have any impact on the environment. However, during development, they become a source of negative influence on nature. All these deposits contain a small proportion of useful components. This is especially true for diamonds and metal ore deposits. More than 90% of the extracted rock mass goes to dumps, considering dilution during mining. Both mining itself and its consequences are negative. Moreover, the load on ecosystems continues for a long time [
9,
10], it is global and represents a serious problem for all types of ecosystems [
11].
During the operation of the mining enterprise, the landscape of territories is changed, the land plots are withdrawn for waste rocks. The removal of soil cover, deforestation, the destruction of vegetation, the contamination of soil and water resources, and the loss of biodiversity in a mining zone change the environment on a local and regional scale [
12,
13,
14].
The destruction of mountain peaks by blasting operations changes the hydrological regime of river flows and water quality [
15,
16,
17]. The developed spaces are filled with water, turning into reservoirs that differ from natural lakes [
18]. Many of them are toxic, which poses a threat to adjacent ecosystems [
19], although some of them are used for entertainment purposes [
20].
Among the main problems of mining enterprises are dumps and tailings, which pose a great risk to the environment and people. When developing deposits, only a small part of the extracted rock mass is used. Overburden and host rocks, the amounts of which exceed 90% of the extracted ore, as well as enrichment waste, are stored in dumps. The dumps occupy large areas near the developed fields, and their impact on the environment is of a transboundary nature [
11,
21]. However, dump rocks should be considered not only as sources of pollution, but also as potential mineral resources [
22,
23]. This will allow us to achieve the rational use of natural resources, maintaining a balance between the extraction of mineral resources and environmental preservation [
24,
25,
26].
The use of waste rocks in the production of building materials allows us to dispose of waste, prevent contamination of the area and its occupation in dumps, save money in the delivery of construction materials, and reduce carbon dioxide emissions in cement production. However, magnesium-containing rocks that are part of the ultramafic–mafic complexes are not used in the production of construction materials, and remain in dumps due to the biased attitude towards them. Therefore, the issues of their disposal are relevant and require further study.
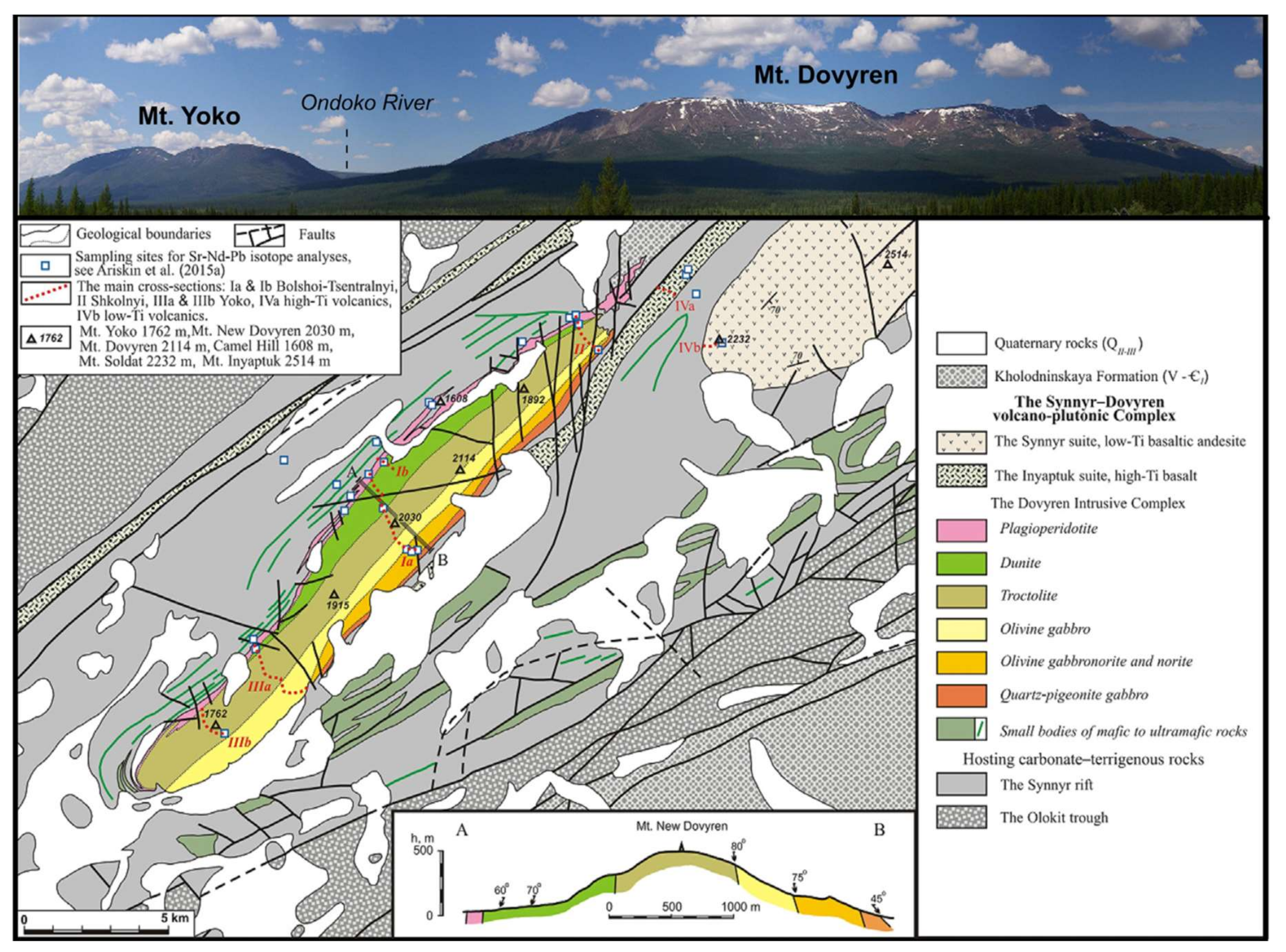
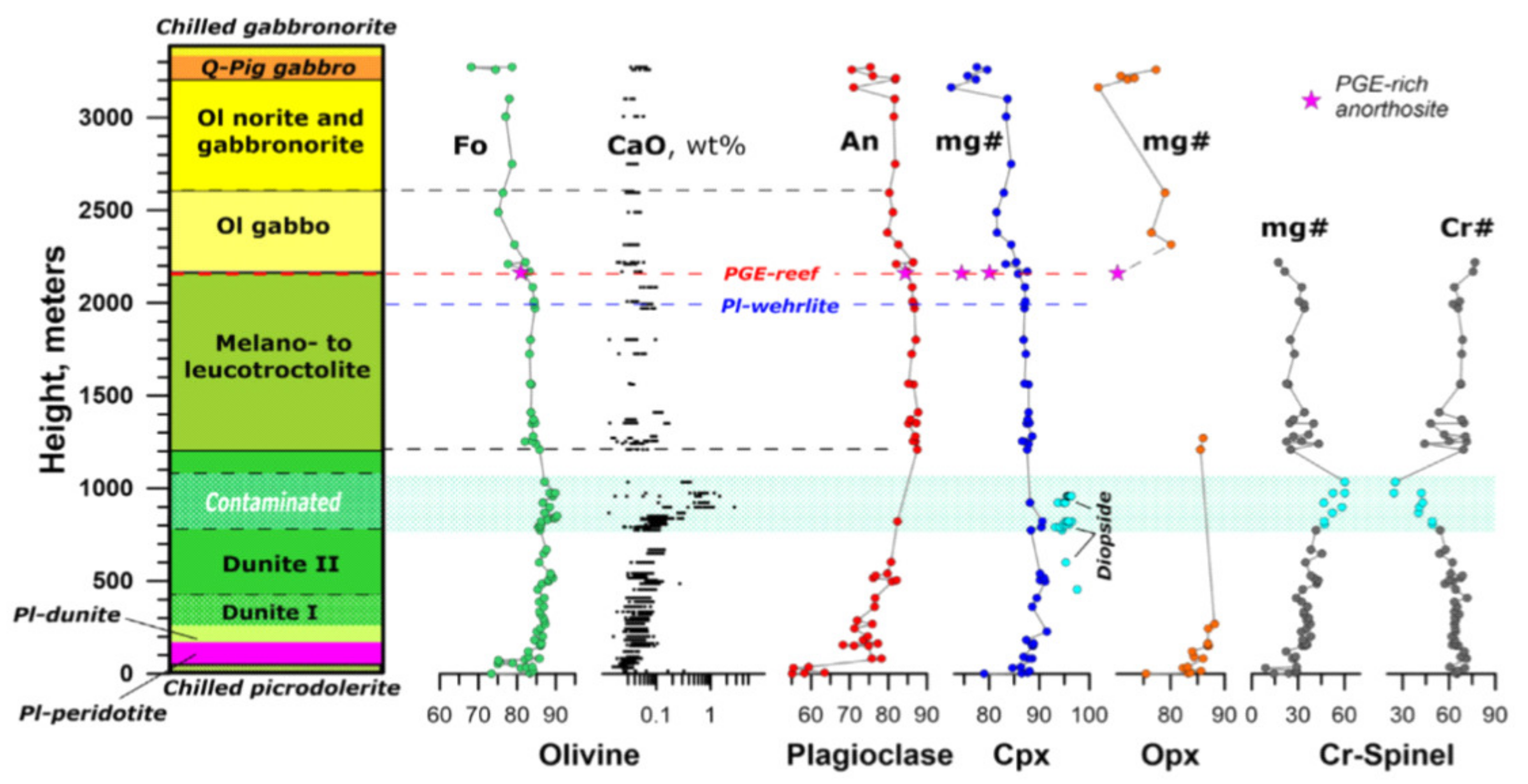
The solution to this problem is shown in the example of the Yoko–Dovyren layered dunite–troctolite–gabbro massif of the North Baikal region, Russia.
4. Results and Discussion
There are various ways to use the overburden and host rocks of mining enterprises [
55,
56]. These include the construction industry [
57,
58], metallurgy [
59,
60], agriculture [
61] and others. However, the main use of mining waste is in the construction industry. Using various technological approaches, a wide range of building materials can be derived from them [
62,
63,
64,
65,
66,
67], including concrete, where the use of mining waste in both large [
68,
69] and small [
70,
71,
72,
73] aggregate forms is promising.
Magnesium-containing rocks of the Yoko–Dovyren massif were studied to determine the possibility of using them in the production of building materials. Before involving any raw material in the technological cycle, it is necessary to assess its quality. First of all, its hygienic radiation assessment is performed. It is necessary to avoid exposure to ionizing radiation of natural origin [
74,
75,
76,
77]. The research is carried out in accordance with the requirements of the Interstate standard GOST 30108-94 “Building materials and elements. Determination of specific activity of natural radioactive nuclei” (the Russian Federation).
As a result of the conducted work, it has been concluded that magnesium silicate rocks belong to the materials of the first class (I). The values of the total specific effective activity of the natural radionuclide Aeff are as follows: dunite—85.69 Bq/kg, verlite—107.89 Bq/kg, troctolite—131.69 Bq/kg, dunite sand—94.45 Bq/kg. The radiation indicators of the rocks do not exceed 370 Bq/kg. According to the requirements of the Interstate standards of the Russian Federation GOST 8267-93 “Crushed stone and gravel of solid rocks for construction works. Specifications” and GOST 8736-2014 “Sand for construction works. Specifications”, they can be used for all types of construction work.
It is known that the quality of raw materials has a significant impact on the quality of finished products. Therefore, the physical and mechanical characteristics of magnesium silicate rocks were studied. Tests of the crushed stone and the sand were carried out using the methods of the Interstate standards of the Russian Federation GOST 8269.0–97 “Crashed stone and gravel from dense rocks and industrial waste for construction works. Physical and mechanical test methods” and GOST 8735–88 “Sand for construction work. Testing methods”.
The granulometric composition of crushed stone from magnesium silicate rocks of the Yoko–Dovyren massif was determined by sieving samples on a standard set of sieves and is presented in
Table 7.
Our analysis of the table data shows that more than 50% of the crushed stone is represented by particles from 20 to 40 mm in size. About 25% of the crushed stone has dimensions from 10 to 20 mm. Particles less than 5 mm in size make up 2.2% of the crushed stone for dunite and wehrlite and 1.6% for troctolite.
The following physical and mechanical parameters of crushed stone from magnesium silicate rocks were determined: crushability, abrasion, and frost resistance. The content of dusty and clay particles, clay in lumps and the presence of foreign clogging impurities were also determined. The stability of crushed stone to environmental influences and the chemical effects of concrete alkalis has been studied. The results obtained are presented in
Table 8.
The grade of crushed stone by crushability, the grade of crushed stone by abrasion and the content of grains in weak rocks are distinguished. The characteristics of the grade in terms of crushing are given in clause 4. The abrasion grade of crushed stone characterizes the wear resistance of the material and is determined by the loss of crushed stone mass after testing in a shelf drum with balls. The selection of grains of weak rocks is carried out as follows: the grains of weak rocks are easily broken by hands and destroyed by light blows of a hammer. When scratching with a needle, a trace of a steel needle remains on the surface of a weak grain. The crushed stone should not contain grains of weak rocks in an amount of more than 5%.
The compressive strength of crushed stone from dunite is 1365 kg/cm2, wehrlite—1312 kg/cm2, and troctolite—1294 kg/cm2. After crushing in a cylinder at a load of 200 kN, the weight loss of dunite is 9.5%, wehrlite is 9.7%, and troctolite is 10.3%. After abrasion in a shelf drum, the weight loss of dunite is 17.4%, wehrlite is 18.1%, and troctolite is 19.8%.
It has been determined that the magnesium silicate rocks are hard rocks that do not contain grains of weak rocks. According to the number of grains of lamellar and needle forms, they belong to the I group of the crushed stone (up to 10 wt.%). They have a high grade base on their crushing capacity (1200), abrasion (I) and a high specific weight. The crushed stone from these rocks is resistant to the environment and to the chemical effects of alkalis. Using the mineralogical–petrographic method, it was established that dunites, verlites, and troctolites do not contain minerals containing alkali-soluble silica and are inert with respect to alkalis. Crushed stone from these rocks is resistant to all types of decay. Harmful components and impurities in the studied rocks were not detected. They withstand 400 cycles of freezing and thawing and have a brand of frost resistance of F400.
The conducted tests show that the crushed stone from the magnesium silicate rocks in 5–40 mm fractions is of high quality, meets the requirements of GOST 8267-93 and can be used as a concrete aggregate, as well as for roads and other types of construction work.
The granulometric composition of the dunite sand of the Yoko–Dovyren massif, determined by sieving with a standard set of sieves, is shown in
Table 9.
The sieve analysis has shown that 48.2% of the sand is represented by particles larger than 0.63 mm. According to the fineness modulus (Mfn = 2.72) and the total residue on sieve number 0.63, it belongs to the group of large sands.
The properties of the dunite sand were defined and they are presented in
Table 10.
As shown by the research, the dunite sand belongs to the group of large sands. It consists of olivine, does not contain layered silicates, amorphous varieties of silicon dioxide, minerals containing sulfur, as well as other harmful components and impurities. A comparison of the color of the alkaline solution, settled over a sample of sand, with the color of the standard solution showed that it does not contain organic impurities. Dunite sand grains have an angular shape with a rough surface. Dunite sand, in its performance, meets the requirements for use as a filler and can be used for all types of construction work.
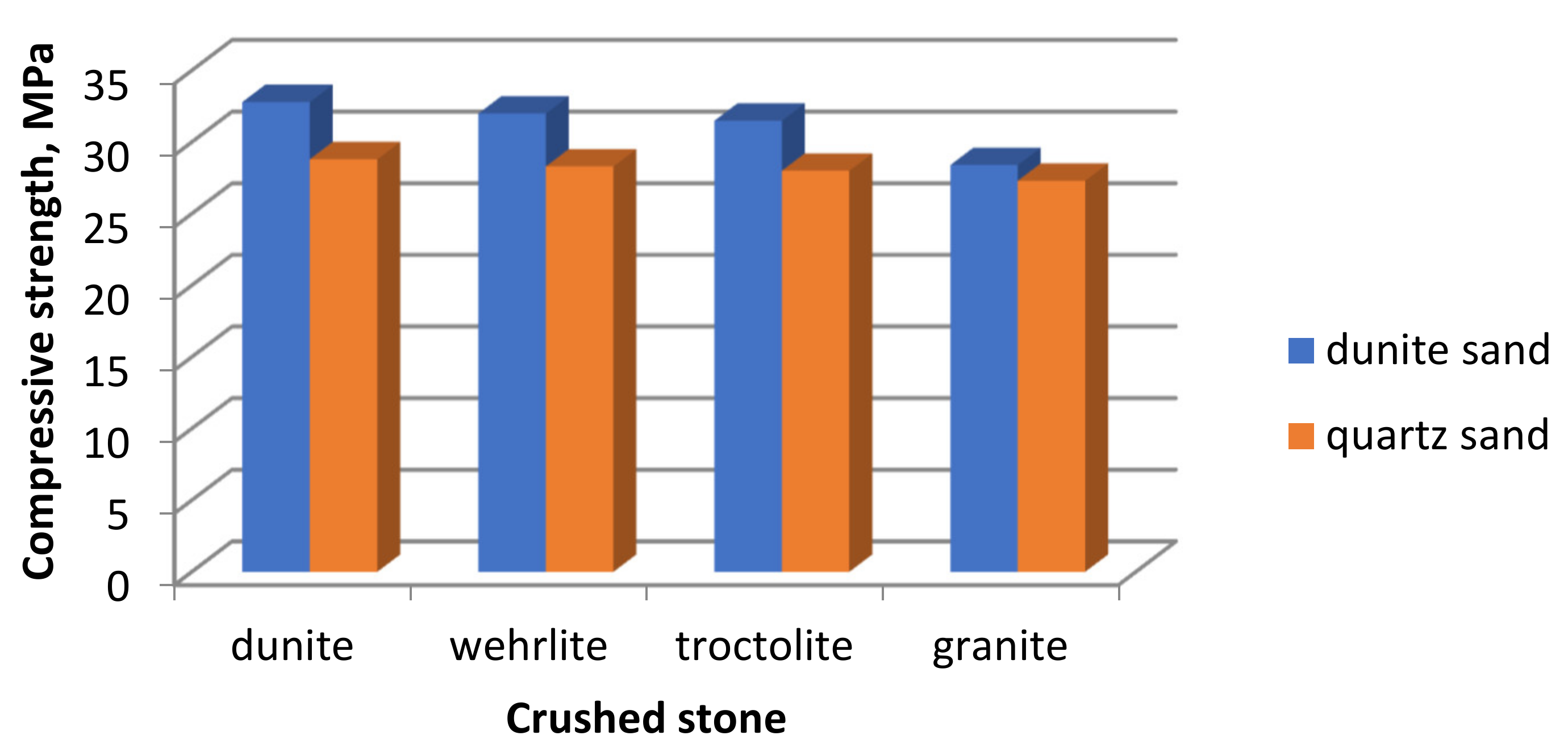
Since magnesium silicate rocks are of high quality, studies have been conducted on their use as large and small aggregates in the production of concretes. Concrete on granite rubble and quartz sand was taken as a control sample. The mechanical indicators of the concrete were determined, and their dependence on the types of aggregates used was established (
Figure 9).
The introduction of the crushed stone from magnesium silicate rocks into the concrete mix leads to an increase in their strength by 2.6–5.5% compared to the control sample. The greatest strength indicators are shown by concretes with the addition of dunite. Moreover, their values depend on the type of sand used. Replacing quartz sand with dunite increases their strength by 12.5–13.9%, depending on the type of crushed stone.
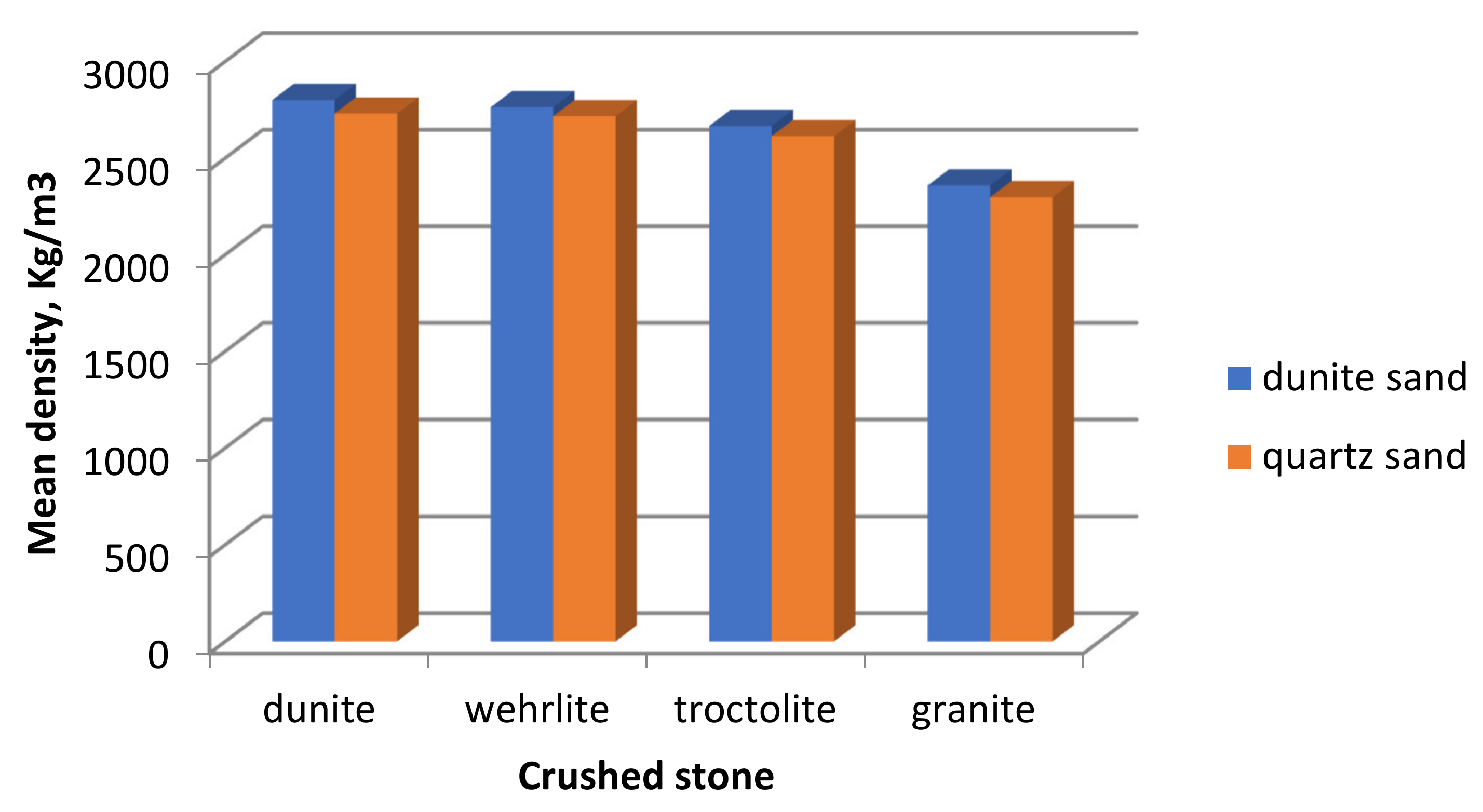
The average density of the resulting concretes (
Figure 10) also depends on the type of aggregates. The highest density is found in concretes where dunite crushed stone and dunite sand are used as aggregates.
The frost resistance of concrete with aggregates made of magnesium silicate rocks was determined. The samples withstood 50 freeze–thaw cycles. There was no destruction on their surface. The loss of mass in concrete samples with the addition of dunite aggregates was 1.18%, with the addition of wehrlite the loss was 1.34%, and with troctolite the loss was 1.67%. For the test sample, this figure was 1.83%. Based on the received values, as well as the strength indicators of compression after the completion of tests, the brand of concrete for frost resistance—F50—is defined.
The tests of concretes for abrasion showed that the loss of mass from the samples with magnesium-containing aggregates does not exceed the loss of mass of the control sample (0.63 g/cm2). Based on the classification of the abrasion, the resulting concretes belong to the G1 brand.
The use of magnesium silicate rocks in concrete production allows us to produce heavy concretes (average density 2000–2500 kg/m3), which are used in all load-bearing structures, and, particularly, heavy concretes (average density more than 2500 kg/m3), which are used for the manufacture of special constructions.
The data obtained show that the magnesium-containing rocks are of high quality and can be used in the production of building materials, in particular, concrete (
Figure 11).
When replacing traditional raw materials with them, it is possible to save about 2000 kg of mineral resources while obtaining 1 m3 of concrete.
5. Conclusions
Ultramafic–mafic complexes are widely developed in the Earth’s crust. They contain deposits of various minerals. The Yoko–Dovyren intrusive in the North Baikal Region, Russia, is considered an example of an intrusive containing diverse mineralization: Ni-Cu, PGE, Cr, Zr, B and blue diopside. Among the magnesium-containing rocks of the Yoko–Dovyren massif are dunites, wehrlites and troctolites, which are moved to dumps during the development of the deposit. The dunites and wehrlite are ultrabasic rocks and the troctolites are basic rocks.
Hygienic radiation assessments of the rocks have shown that they can be used in construction for all types of construction work. The values of the total specific effective activity of the natural radionuclide Aeff are as follows: dunite—85.69 Bq/kg, verlite—107.89 Bq/kg, troctolite—131.69 Bq/kg, dunite sand—94.45 Bq/kg. They do not exceed the normalized values.
The physical and mechanical parameters of the magnesium-containing rocks have been studied and their high quality has been determined. They do not contain lamellar and needle-shaped grains of weak rocks, have a high grade for crushing and a high specific weight, and do not contain harmful components and impurities. The rocks are a promising raw resource for obtaining new types of building materials.
Concretes in large and small aggregate forms were obtained from magnesium-containing rocks. It has been determined that the type of aggregates affects the mechanical characteristics of the concrete. The strength indicators of the concrete on crushed stone from the studied rocks are 2.6–5.5% higher than that of the control sample. The use of dunite sand increases the strength of the concretes by 12.5–13.9%, depending on the type of crushed stone.
The concrete density also depends on the types of aggregates. Using magnesium-containing rocks, one can obtain heavy concrete for load-bearing structures, and heavy concrete for special structures.
The use of magnesium-containing rocks in the development of deposits, without allowing them to fall into dumps, gives us the opportunity to create clean, environmentally friendly mining enterprises. In addition, replacing traditional raw materials with them will allow us to preserve mineral resources for future generations.