Valorization of Rare Earth Elements from a Steenstrupine Concentrate Via a Combined Hydrometallurgical and Pyrometallurgical Method
Abstract
1. Introduction
2. Material and Methods
2.1. Material
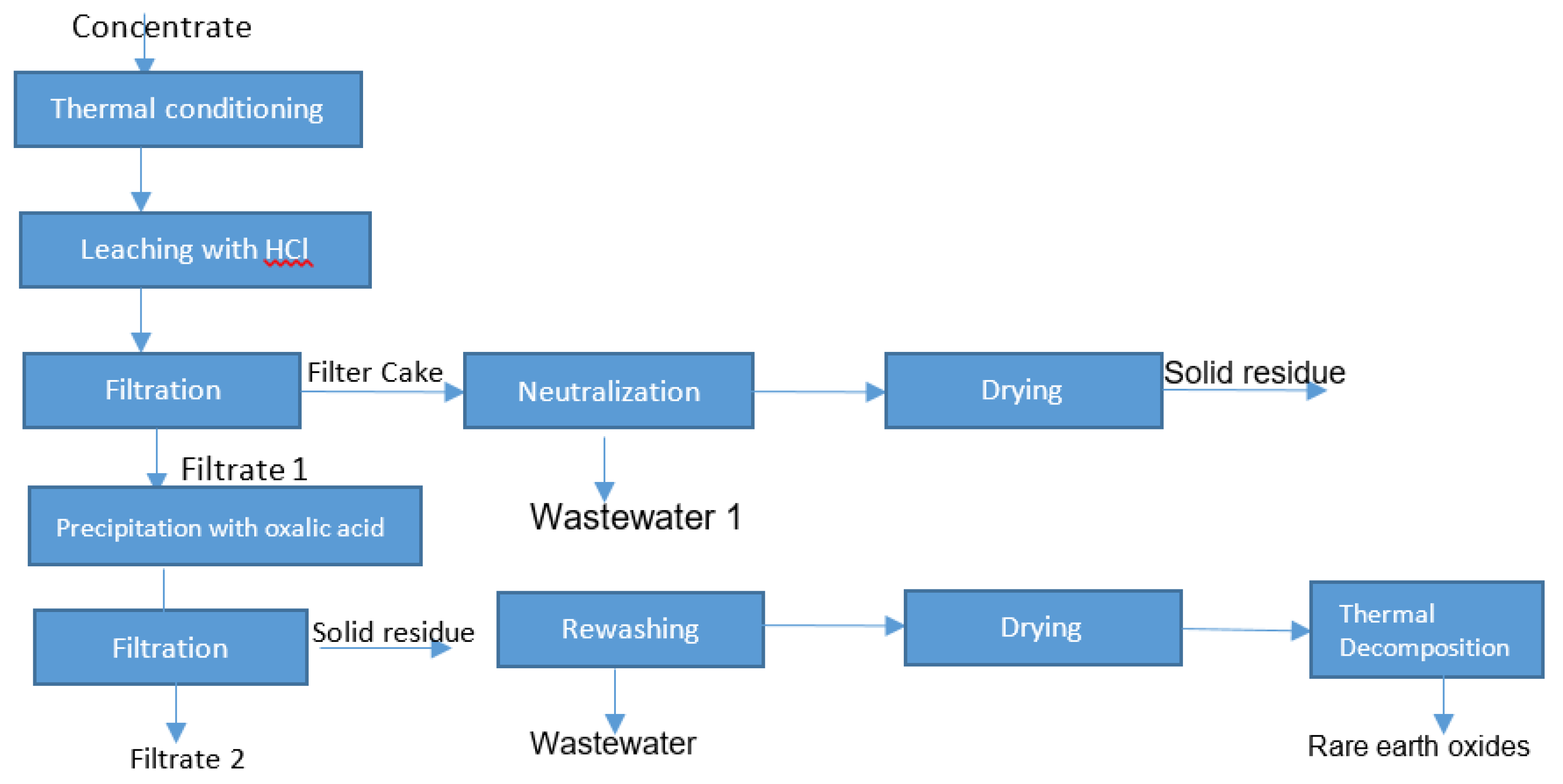
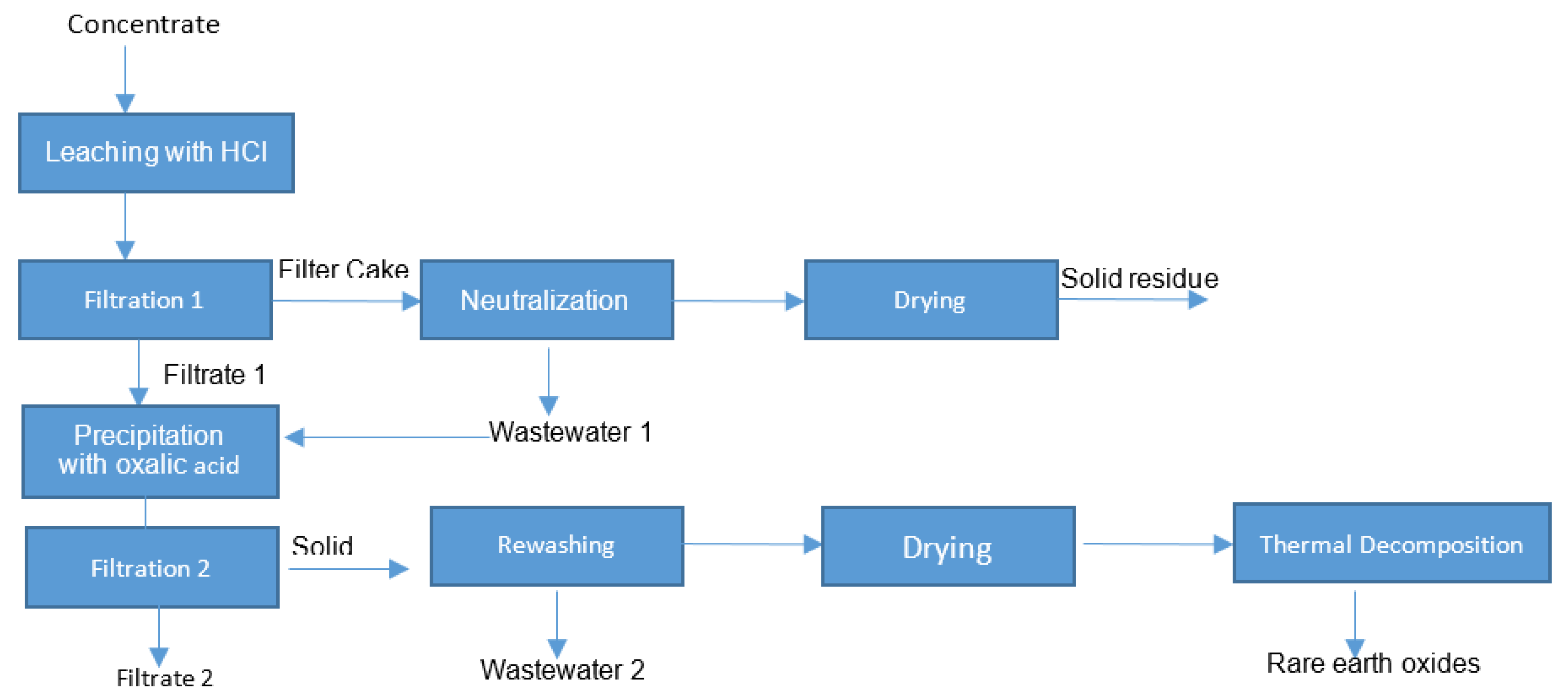
2.2. Methods
2.2.1. Thermal Conditioning
2.2.2. Leaching with Hydrochloric Acid and Filtration
3. Results and Discussion
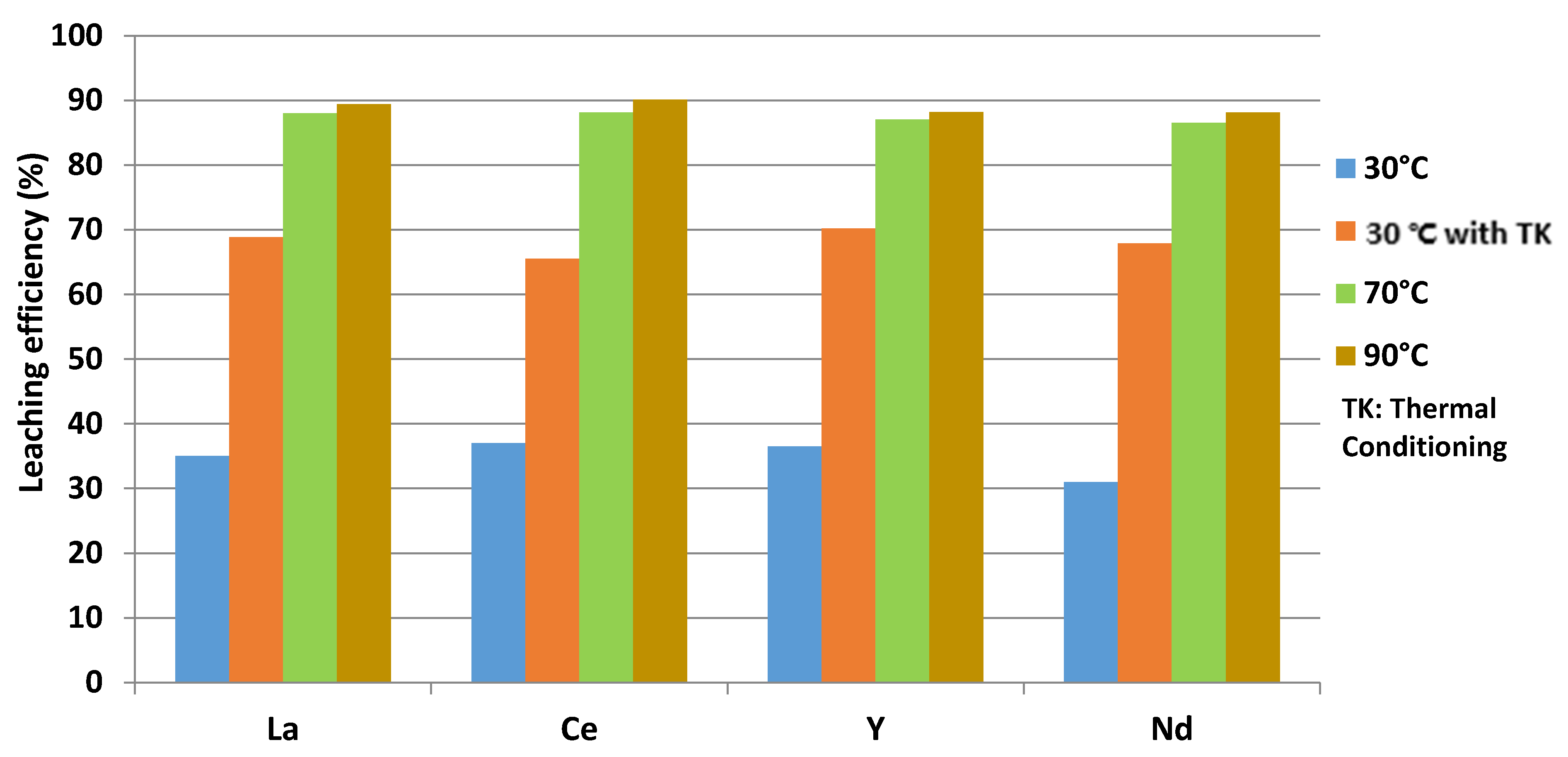
3.1. The Influence of Temperature on a Leaching Efficiency
3.2. The Influence of Liquid/Solid Ratio at a Leaching Efficiency
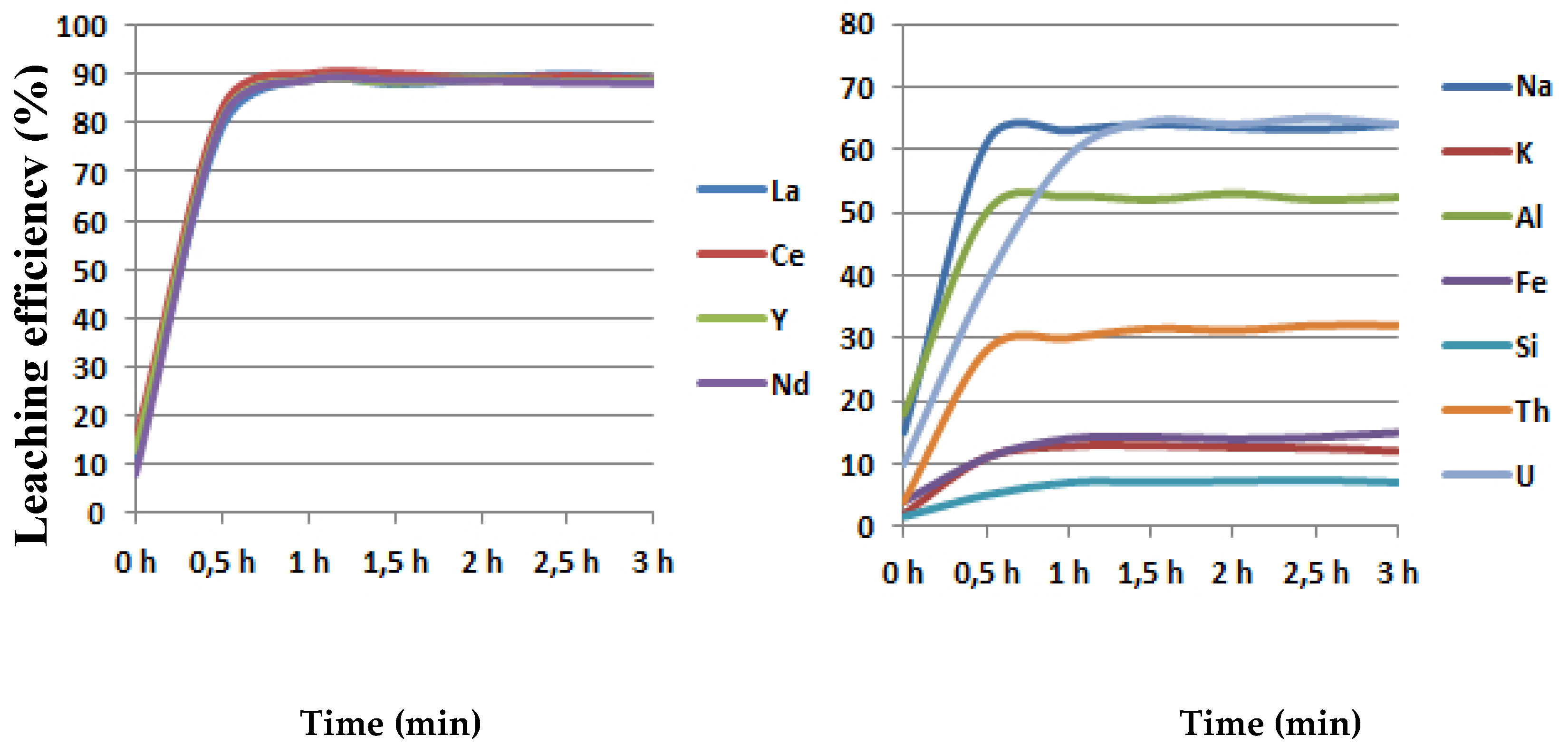
3.3. The Influence of Leaching Time on a Leaching Efficiency
3.4. Solid/Liquid Separation
3.5. Precipitation with Oxalic Acid
3.6. Thermal Decomposition of Rare Earth Oxalate
4. Conclusions
Author Contributions
Acknowledgments
Conflicts of Interest
References
- Zhang, G.; Huang, W. Bastnaesite smelting process review. Rare Met. 1997, 21, 193–199. [Google Scholar]
- Guocai, Z.; Jun, T. Rare earth extraction from bastnaesite with green chemistry method. Chemistry 2000, 12, 6–11. [Google Scholar]
- Spear, S.F.; Pyle, M.J. Apatite, monazite, and xenotime in metamorphic rocks. Rev. Mineral. Geochem. 2002, 48, 293–335. [Google Scholar] [CrossRef]
- Huang, X.; Li, H.; Wang, C.; Wang, G.; Xue, X.; Zhang, G. Development Status and Research Progress in Rare Earth Industry in China. Chin. J. Rare Met. 2007, 31, 279–288. [Google Scholar]
- Zhengfu, S. Study on the utilization of monazite resource. World Nonferrous Met. 2014, 31–33. [Google Scholar]
- Stopić, S.; Friedrich, B. Leaching of rare earth elements with sulfuric acid from bastnasite ores. Mil. Tech. Cour. 2018, 66, 757–770. [Google Scholar] [CrossRef]
- Furfaro, D.; Krebs, J. Removal of Uranium and Thorium from Kvanefjeld, Rare Earth İntermediate Product; Greenland Minerals and Energy Limited: Perth, Australia, 2013. [Google Scholar]
- Van Wyngaardt, M. GMEL looking to simplify refining at Kvanefjeld rare earths project. In Creamer Medias Mining Weekly; Creamer Media: Johannesburg, South Africa, 2018. [Google Scholar]
- Lebedev, V.N. Sulfuric Acid Technology for Processing of Eudialyte Concentrate. Russ. J. Appl. Chem. 2003, 76, 1559–1563. [Google Scholar] [CrossRef]
- Lebedev, V.N.; Shchur, T.E.; Maiorov, D.V.; Popova, L.A.; Serkova, R.P. Specific Features of Acid Decomposition of Eudialyte and Certain Rare-Metal Concentrates from Kola Peninsula. Russ. J. Appl. Chem. 2003, 76, 1191–1196. [Google Scholar] [CrossRef]
- Davris, P.; Stopic, S.; Balomenos, E.; Panias, D.; Paspaliaris, I.; Friedrich, B. Leaching of rare earth elements from Eudialyte concentrate by supressing silicon dissolution. Miner. Eng. 2017, 108, 115–122. [Google Scholar] [CrossRef]
- Voßenkaul, D.; Birich, A.; Müller, N.; Stoltz, N.; Friedrich, B. Hydrometallurgical processing of eudialyte bearing concentrates to recover rare earth elements via low-temperature dry digestion to prevent the silica gel formation. J. Sustain. Metall. 2017, 3, 79. [Google Scholar] [CrossRef]
- Stopic, S.; Ma, Y.; Friedrich, B. Leaching of Eudialyte concentrate and REE-Precipitation. In Proceedings of the 9th European Metallurgical Conference EMC 2017, Leipzig, Germany, 25–28 June 2017; Volume 2, pp. 595–602. [Google Scholar]
- Ma, Y.; Stopic, S.; Gronen, L.; Obradovic, S.; Milivojevic, M.; Friedrich, B. Neural Network Modeling for the Extraction of Rare Earth Elements from Eudialyte Concentrate by Dry Digestion and Leaching. Metals 2018, 8, 267. [Google Scholar] [CrossRef]
- Ma, Y.; Stopic, S.; Friedrich, B. Hydrometallurgical Treatment of a Eudialyte Concentrate for Preparation of Rare Earth Carbonate. Johns. Matthey Technol. Rev. 2019, 63, 2–13. [Google Scholar] [CrossRef]
- Ma, Y.; Stopic, S.; Friedrich, B. Selective Recovery and separation of Zr and Hf from sulfuric acid leach solution using anionic exchange resin. Hydrometallurgy 2019, 189, 105–143. [Google Scholar] [CrossRef]
- Balomenos, E.; Davris, P.; Deady, E.; Yang, J.; Panias, D.; Friedrich, B.; Binnemans, K.; Seisenbaeva, G.; Dittrich, C.; Kalvig, P.; et al. The EURARE Project: Development of a Sustainable Exploitation Scheme for Europe’s Rare Earth Ore Deposits. Johns. Matthey Technol. Rev. 2017, 61, 142–153. [Google Scholar] [CrossRef]
- Alkan, G.; Diaz, F.; Gronen, L.; Stopic, S.; Friedrich, B. A mineralogical assessment on bauxite residue (red mud) leaching for titanium recovery. Metals 2017, 7, 458. [Google Scholar] [CrossRef]
- Kaya, S.; Peters, E.; Forsberg, K.; Dittrich, C.; Stopic, S.; Friedrich, B. Scandium Recovery from an Ammonium Fluoride Strip Liquor by Anti-Solvent Crystallization. Metals 2018, 8, 767. [Google Scholar] [CrossRef]
- Alkan, G.; Yagmurlu, B.; Friedrich, B.; Ditrich, C.; Gronen, L.; Stopic, S.; Ma, Y. Selective Silica gel Scandium Extraction from, Iron–depleted Red Mud Slags by Dry Digestion. Hydrometallurgy 2019, 185, 266–272. [Google Scholar] [CrossRef]





| SiO2 | Al2O3 | Na2O | K2O | Fe2O3 | P2O5 | MnO | CaO | ZrO2 | MgO | TiO2 |
| 36.4 | 3.35 | 13.2 | 0.94 | 10.2 | 8.49 | 1.73 | 1.87 | 0.30 | 0.07 | 0.15 |
| F | La2O3 | CeO2 | Nd2O3 | Y2O3 | Sm2O3 | Gd2O3 | Dy2O3 | ThO2 | Total REO | |
| 0.54 | 4.06 | 7.76 | 2.78 | 1.99 | 0.40 | 0.45 | 0.23 | 1.48 | 17.66 | |
| La | Ce | Nd | Y | Pr | Sm | Eu | Gd | Tb | Dy | Er |
|---|---|---|---|---|---|---|---|---|---|---|
| 3.28 | 6.31 | 2.21 | 1.37 | 0.57 | 0.29 | 0.02 | 0.22 | 0.03 | 0.17 | 0.07 |
| Experiment | Thermal Conditioning | Solid-Liquid Ratio | Temperature (°C) | Conditions in Reactor | Silica Gel Formation |
|---|---|---|---|---|---|
| No.1 | No | 0.1 | 30 | open | Yes |
| No.2 | Yes | 0.1 | 30 | open | No |
| No.3 | No | 0.1 | 70 | open | Yes |
| No.4 | No | 0.1 | 70 | closed | No |
| No.5 | No | 0.1 | 90 | closed | No |
| Experiment | Solid/Liquid Ratio (g/mL) | Silica Gel Formation | Leaching Efficiency of Yttrium (%) |
|---|---|---|---|
| No.5 | 20/200 (0.73 g HCl/g concentrate) | No, but silica precipitation is observed during staying | 88.0 |
| No.6 | 20/250 (0.91 g HCl/g concentrate) | No | 88.3 |
| No.7 | 40/200 (0.37 g HCl/g concentrate) | Yes | − |
| No.8 | 20/300 (1.10 g HCl/g concentrate) | No | 88.0 |
| Element | Filtrate (%) | Wastewater from Filter Cake (%) | Solid Tesidue (%) |
|---|---|---|---|
| La | 89.50 | 8.10 | 4.03 |
| Ce | 90.20 | 7.85 | 4.82 |
| Nd | 8820 | 8.00 | 5.08 |
| Y | 88.30 | 8.80 | 5.18 |
| Element | Filtrate1 (%) | Wastewater 2 (%) | Solid Residue (%) |
|---|---|---|---|
| La | 95.28 | 2.02 | 2.02 |
| Ce | 95.69 | 1.93 | 2.10 |
| Nd | 96.36 | 1.90 | 2.53 |
| Y | 96.27 | 1.78 | 3.34 |
| La | Ce | Nd | Y | Pr | Sm | Gd | Dy | Er | Th | U |
| 98.98 | 99.57 | 98.19 | 98.53 | 98.30 | 98.60 | 97.83 | 97.99 | 97.86 | 95.59 | 10.24 |
| Si | Al | Na | K | Fe | P | Mn | Ca | Mg | ||
| 0.1 | 29.79 | 1.07 | 16.79 | 41.30 | 0.55 | 10.01 | 40.93 | 0.1 |
| La | Ce | Nd | Y | Pr | Sm | Eu | Gd | Dy | Er | Tb |
| 18.90 | 36.92 | 12.44 | 7.86 | 3.01 | 1.60 | 0.087 | 0.95 | 0.80 | 0.32 | 0.14 |
| Al | Fe | Mn | Na | Ca | K | P | Th | U | Total REO | |
| 1.37 | 0.08 | 0.04 | <0.01 | 0.54 | 0.08 | 0.10 | 1.72 | <0.01 | 99.80 | |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yun, Y.; Stopic, S.; Friedrich, B. Valorization of Rare Earth Elements from a Steenstrupine Concentrate Via a Combined Hydrometallurgical and Pyrometallurgical Method. Minerals 2020, 10, 248. https://doi.org/10.3390/min10030248
Yun Y, Stopic S, Friedrich B. Valorization of Rare Earth Elements from a Steenstrupine Concentrate Via a Combined Hydrometallurgical and Pyrometallurgical Method. Minerals. 2020; 10(3):248. https://doi.org/10.3390/min10030248
Chicago/Turabian StyleYun, Yunbo, Srecko Stopic, and Bernd Friedrich. 2020. "Valorization of Rare Earth Elements from a Steenstrupine Concentrate Via a Combined Hydrometallurgical and Pyrometallurgical Method" Minerals 10, no. 3: 248. https://doi.org/10.3390/min10030248
APA StyleYun, Y., Stopic, S., & Friedrich, B. (2020). Valorization of Rare Earth Elements from a Steenstrupine Concentrate Via a Combined Hydrometallurgical and Pyrometallurgical Method. Minerals, 10(3), 248. https://doi.org/10.3390/min10030248







