Abstract
Starch-based flocculants have been emerged as a promising alternative to conventional synthetic flocculants in wastewater treatment, especially for the treatment of oil sand tailings, as they are low cost, safe, biodegradable, fairly shear-stable, readily available from reproducible agricultural resources, and do not result in secondary pollution. In this paper, three types of polymer-grafted starches (St-g-Polymer) with different charge properties were synthesized and their molecular structures were controlled by atom transfer radical polymerization (ATRP). The correlations between the charge properties of starch-based flocculants, external environmental parameters, and flocculation performance were systematically investigated by conducting jar tests under various environmental conditions. It was found that the charge properties of the branch chain had a significant impact on flocculation performance. The cationic St-g-Polymer demonstrated the best performance due to the grafting of the cationic monomer to the starch backbone which improved the solubility of the copolymer and aided in the removal of small/water-soluble particles. The results obtained could assist in guiding the selection and design of suitable biodegradable flocculants when treating targeted wastewater.
1. Introduction
Flocculation is a traditional chemical water treatment technique typically applied to separate suspended solids from water. Specifically, in the flocculation process, synthetic polymer flocculants are used to form molecular bridges between the solid particles facilitating impurities to be separated from the water. Starch-based flocculants, as alternatives to synthetic polyacrylamide in wastewater treatment, especially for the treatment of oil sand tailings, are high-performance and low-cost natural polymeric flocculants which have attracted much attention because they are inexpensive, biodegradable, and shear stable [1]. In order to increase the flocculation efficiency, the natural starches were commonly modified to alter their functional groups. There are different approaches for starch modification, such as esterification, oxidation, graft reaction, etc. [2,3,4]. Among them, polymer grafted starches (St-g-Polymer) have gained extensive interest because they have properties of both natural and synthetic polymers, and they can also significantly improve the flocculation performance for water treatment. Atom transfer radical polymerization (ATRP) is one of the most effective and widely used methods among controlled radical polymerization (CRP) [5]. In recent decades, many kinds of St-g-Polymer have been produced by ATRP. For example, Liu and coworkers prepared poly(butyl methacrylate) grafted starch (Starch-g-PBA), polystyrene grafted starch (Starch-g-PS), and poly(methyl methacrylate) grafted starch (Starch-g-PMMA) by ATRP [6,7]. Nurmi and coworkers grafted methyl methacrylate (MMA) from starch acetate [8]. Moghaddam and coworkers prepared polyacrylamide grafted carboxymethyl starch (CMS-g-PAA) or polyhydroxyethylacrylate grafted carboxymethyl starch (CMS-g-PHEA) by ATRP [9]. However, little work has been reported regarding St-g-Polymer prepared by ATRP used as flocculants in the application of water treatment. This is either because the modified starch was synthesized by surface-initiated ATRP that limits the graft density and ratio of the grafted polymer for better flocculation performance, or because the synthesized materials have poor water solubility due to the chemical structure of the grafted polymer.
In the applications of wastewater treatment, the performance of flocculants is highly determined by their molecular structures [10,11,12] as well as the external operation conditions such as pH, temperature, and dosage [13,14]. Therefore, it is very important to understand the structure-activity relationships when preparing starch-based polymer flocculants. Various structural factors can affect the flocculation performance, such as charge properties, grafting ratio, degree of functional group substitution, and distributions. Knowledge of the effects of the charge properties of starch-based flocculants on their flocculation performance is limited and unsystematic in spite of some previous investigations [15].
In the present study, three types of polymer grafted starches (St-g-Polymer), were synthesized and their molecular structures were controlled by ATRP. Three types of polymers consist of either nonionic dimethylacrylamide groups without charge, cationic quaternary ammonium salt groups with positive charges, or anionic sulfonate groups with negative charges [16]. As a first step, in order to evaluate the effectiveness of the modified starches, flocculation tests were conducted on the model tailings, i.e., the suspensions of kaolin clays. The correlations between charge properties of the starch-based flocculants, external environmental parameters, and the flocculation performances were systematically investigated by the jar tests under various environmental conditions including pH, dosage, and type of kaolin clay suspensions. This was carried out in order to create a predictive model that could forecast the final performance of selected and designed flocculants in the application of water treatment. In addition, the flocculation behavior of one of the most effective synthesized St-g-Polymer as synthetic flocculants for water treatment was evaluated and compared with the commercial flocculants, polyacrylamide (PAM). Investigating the flocculation behaviors of the modified starches in actual wastewater streams such as oil sand tailings is planned for the next-step research work.
2. Materials and Methods
2.1. Materials
Starch, N,N-Dimethylacrylamide (DMA), 2-Acrylamido-2-methyl-1-propanesulfonic acid sodium salt solution (AMPS), [2-(Methacryloyloxy) ethyl] trimethylammonium chloride solution (DMC), 2-Bromo-2-methylpropionyl bromide (BIBB), dimethylacetamide (DMAc), and polyacrylamide (PAM) were purchased from Sigma-Aldrich (Oakville, ON, Canada). All the reagents were used directly without further purification.
Kaolin clay samples (two brands, Pioneer and Calcined) were obtained from Greenbarn Potters Supply Ltd. (Surrey, BC, Canada) and used without further treatment. The particle size distribution (PSD) of the two clay samples was measured by the laser light scattering method (Malvern Instrument) and the PSD curves are reported in the supplemental material section (Figure S1). The Pioneer clay and Calcined clay have an average size of ~1.1 µm and 1.5 µm, respectively.
2.2. Synthesis of St-g-Polymer by Atom Transfer Radical Polymerization (ATRP)
2.2.1. Synthesis of Macroinitiator (St-Br)
20 mL of LiCl/DMAc (5%) solution and 0.5 g of starch (9.26 mmol OH) were added to a 50 mL round-bottom flask equipped with a magnetic stirring bar and condensation column. The content of the flask was mixed and under reflux at 80 °C for about 2 h and then cooled down to room temperature. Pyridine (1.6 g, 20.22 mmol) and 2-bromo-2-methylpropionyl bromide (BIBB, 4.34 g, 18.52 mmol) were then added to the flask and the content was stirred overnight at room temperature (RT). The reaction product was dispersed in 200 mL 2 M HCl, filtered, washed three times with distilled water and methanol, and then dried under vacuum for 12 h at 50 °C.
2.2.2. Synthesis of Polymer-Grafted Starch by ATRP
A pre-determined amount of macroinitiator (St-Br), monomer, ligand, CuCl, CuCl2, and solvent (1:1 v/v DMF/H2O solution) was added to a 25 mL Schlenk flask (Sigma-Aldrich, Oakville, ON, Canada) and stirred until completely dissolved. The mixture was degassed with three freeze–evacuate–thaw cycles, flushed with argon, and transferred into the glove box. The polymerization was stopped through exposure to air. Then the mixture was added into 150 mL anhydrous methanol, filtered, washed with anhydrous methanol three times, and dried at 50 °C under a vacuum for 12 h.
2.3. Characterizations of St-g-Polymer
The St-g-Polymer and the original starch were characterized by measuring the 1H nuclear magnetic resonance (1H NMR) spectra, the Fourier Transform Infrared Spectroscopy (FTIR) spectra, and weight loss at elevated temperatures from thermogravimetric analysis (TGA). The 1H NMR spectra were measured using a Bruker Avance 300 NMR Spectrometer (Bruker Ltd., Milton, ON, Canada). The FTIR spectra were recorded on NICOLET 8700 Spectrometer (Thermo Fisher Scientific, Mississauga, ON, Canada) in the range of 4000-400 cm−1 using a potassium bromide disc technique. The weight loss was determined using a thermal analyzer (TGA-50, Shimadzu Co., Tokyo, Japan) under nitrogen atmosphere with a heating rate of 20 °C/min from RT to 800 °C.
2.4. Jar Test
As the most extensively used flocculants for wastewater treatment, non-ionic PAM (MW 3 × 106) was selected as a benchmark to evaluate the flocculation performance of the modified starches in this study. Flocculation performances of original starch, St-g-Polymer, and PAM were evaluated using 0.1 wt% of kaolin clay suspension. The flocculation tests were conducted using Phipps and Bird jar test unit. Each flocculant was first dissolved in water to form flocculant solution with a concentration of 1000 mg/L. The flocculant solution was added to 500 mL of the kaolin suspension in a jar. The suspension was then mixed at speed of 200 rpm for 2 min, followed by a slow stirring at 40 rpm for 10 min. After mixing was stopped, the suspension was let settle for 5 min before a supernatant sample was collected from the top of the jar for measurement of the turbidity. Turbidity was measured using a turbidimeter (model HACH 2100AN, Hach Company, Loveland, CO, USA) and reported in nephelometric turbidity units (NTU). The flocculation performance was evaluated by the turbidity change as calculated using Equation (1).
where Turbidity0 is the turbidity of the supernatant of pure clay after settling for 5 min and Turbidity1 is the turbidity of the supernatant after flocculant addition and settling for 5 min.
3. Results and Discussion
3.1. Synthesis and Characterization of St-g-Polymer
3.1.1. Synthesis and Characterization of Starch-Based Macroinitiator
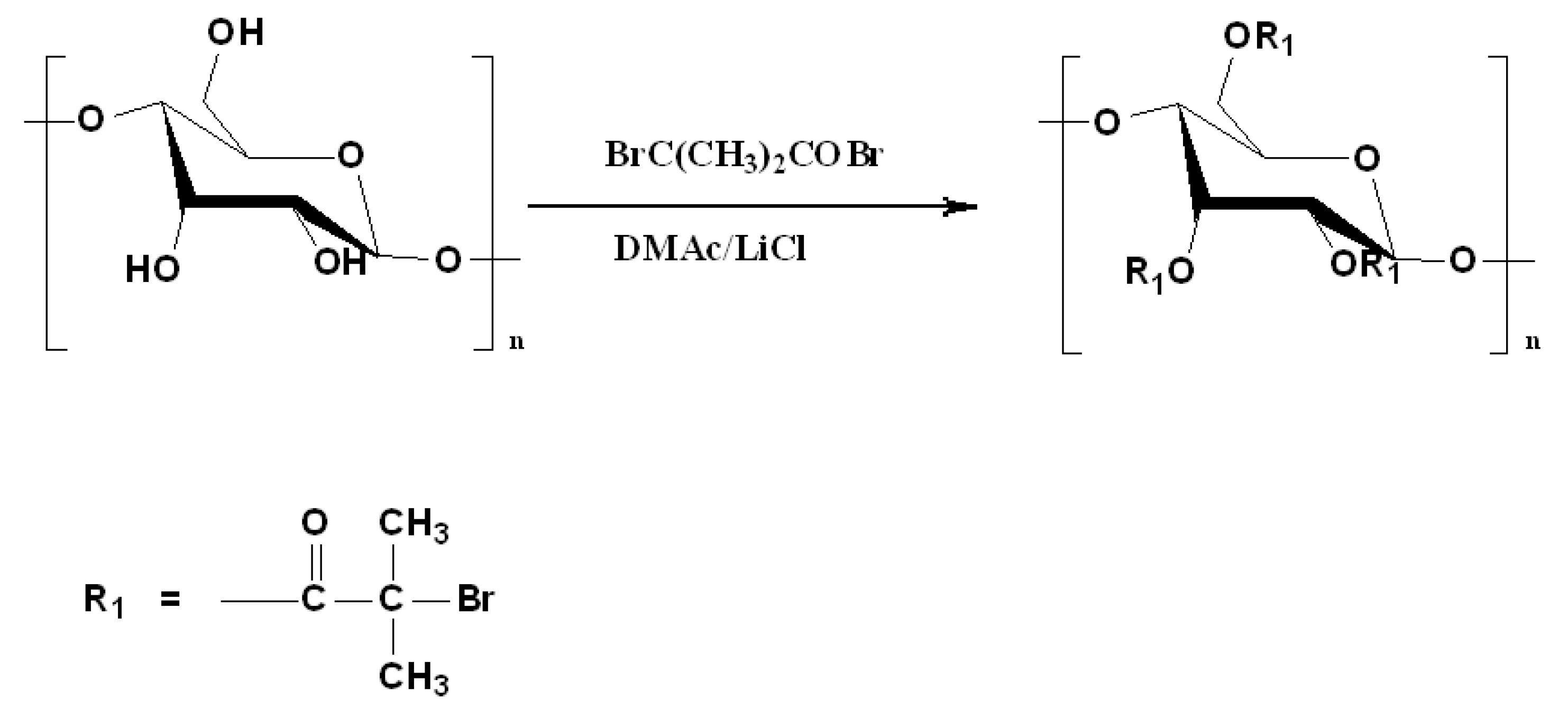
Figure 1 shows the synthetic route for starch-based macroinitiator (Starch-Br) via partial esterification of the hydroxyl groups in the glucose units of starch with BiBB in the presence of pyridine. It is a one-step homogeneous process in LiCl/DMAc solution at room temperature. Parts of the hydroxyl groups of starch were replaced with 2-bromo-2-methylpropionyl groups, which are able to initiate polymerization by the ATRP mechanism.
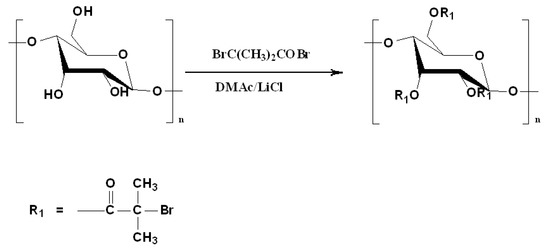
Figure 1.
Synthesis of starch-based ATRP macroinitiator.
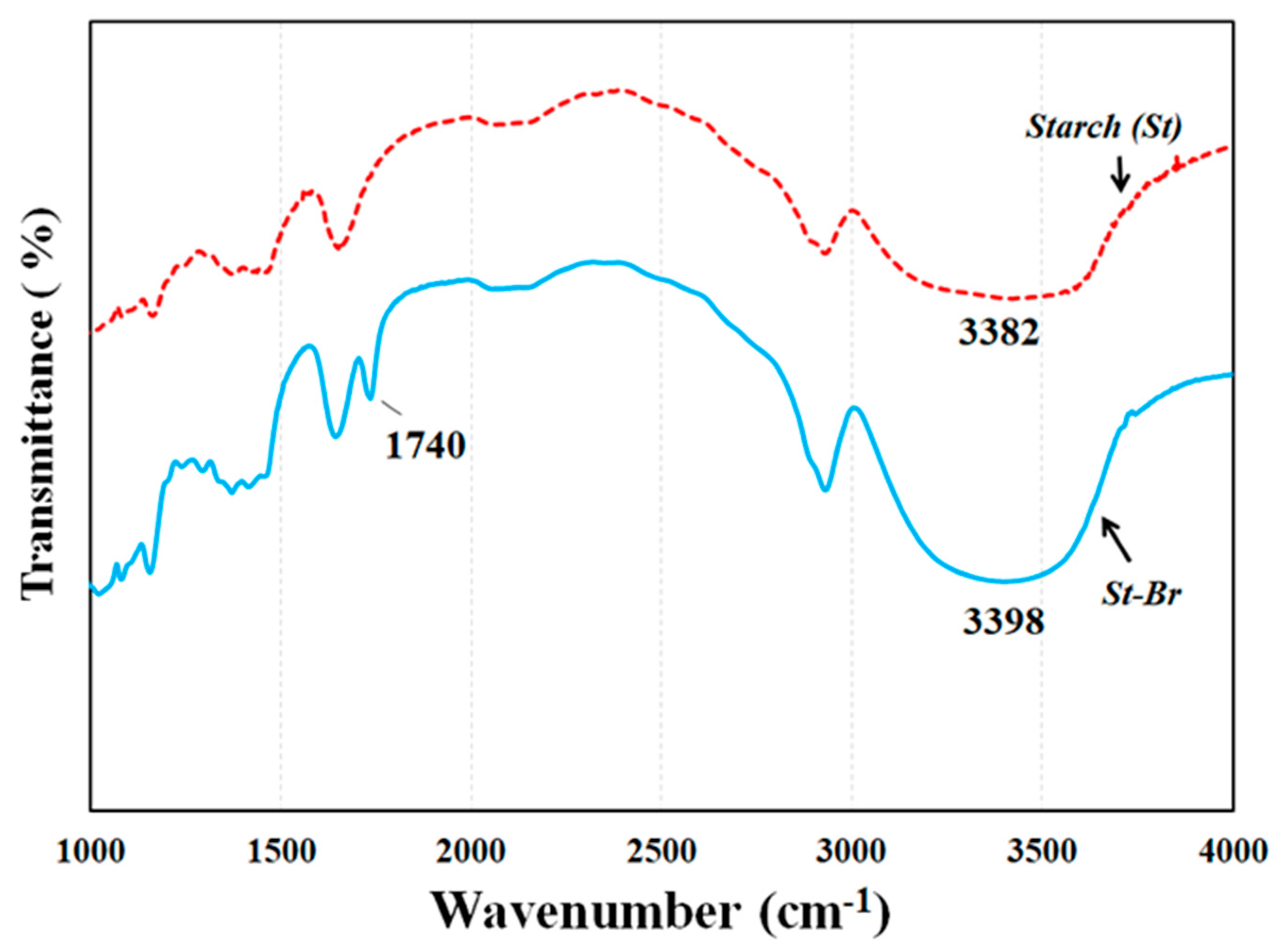
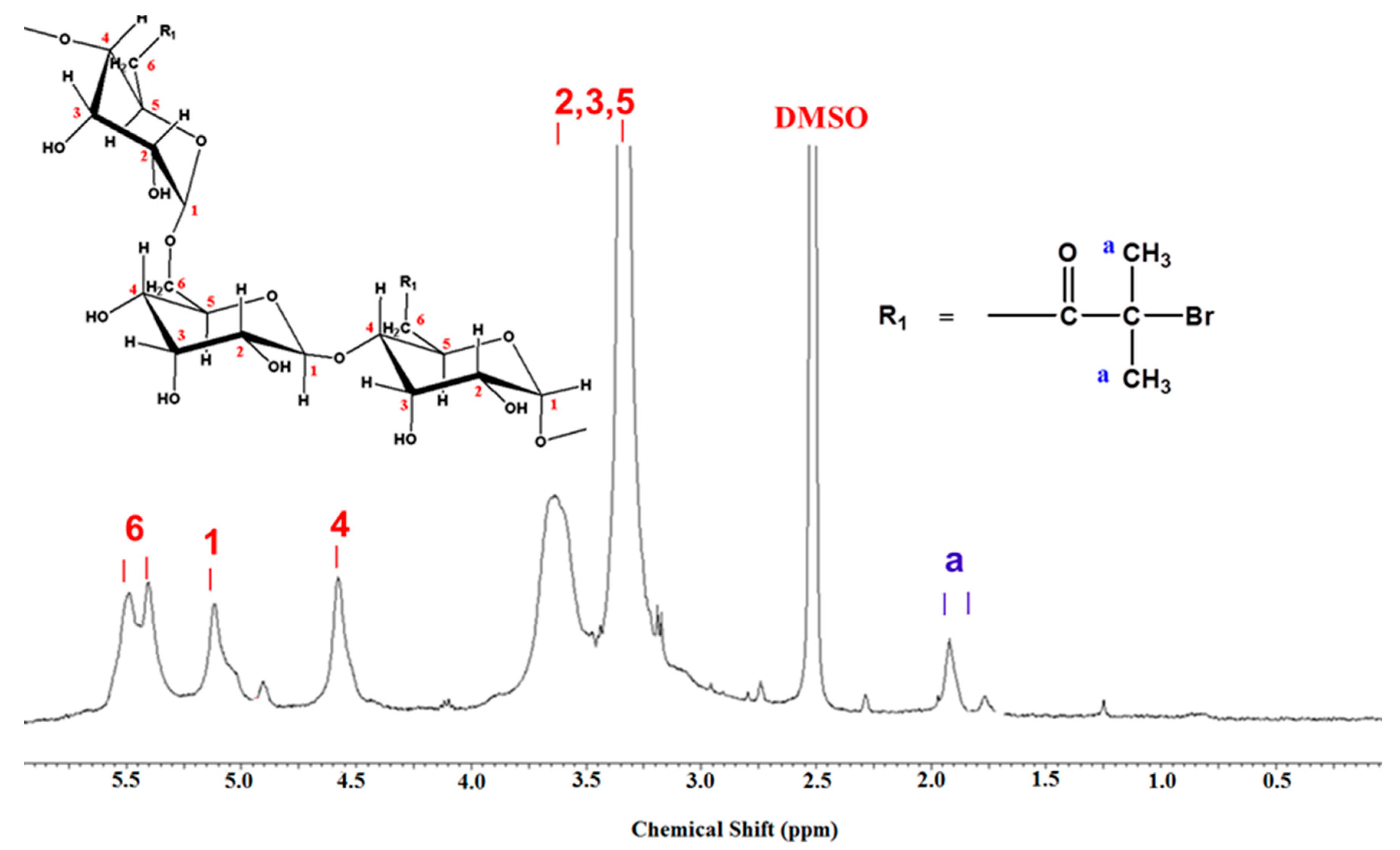
FTIR spectra (Figure 2) and 1H NMR (Figure 3) were employed to demonstrate the successful synthesis of the starch-based macroinitiator. Figure 2 shows the FTIR spectra of starch before and after modification. Because the product was washed three times with distilled water and methanol, unreacted BIBB was removed from the macroinitiator product. Compared to the original starch, the appearance of a peak at 1740 cm−1 confirmed the successful substitution of the BIBB group on the starch molecular chains. This peak is due to C=O stretching of the carbonyl group from the BIBB. The slight shift of the broad and asymmetric peak from 3382 cm−1 to 3398 cm−1 is due to diminished O–H vibrations after esterification further proved the formation of the macroinitiator. The substitution of the hydroxyl group on the starch backbone with BIBB was also verified by 1H-NMR (Figure 3). The chemical shifts in the range of 4.5–5.7 ppm and 1.7–2.2 ppm are attributed to the hydrogen linked to the C6, C1, and C4 carbons and the methyl protons in the ester group of bromoisobutyryl (BiBB) respectively. According to the ratio of the integral of the methyl groups to the integral of protons linked to C1, the total degree of substitution (DS) of BiBB is ~0.1.
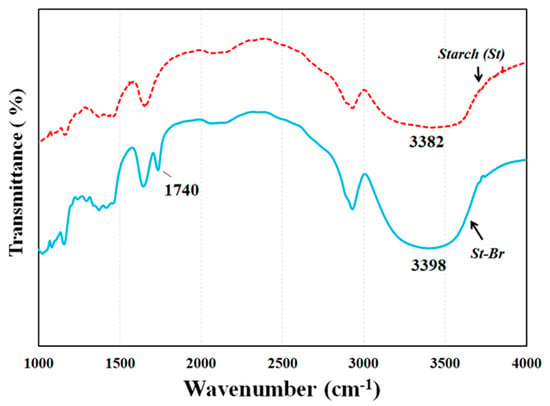
Figure 2.
FTIR spectra of starch (St) and starch based macroinitiator (St-Br).
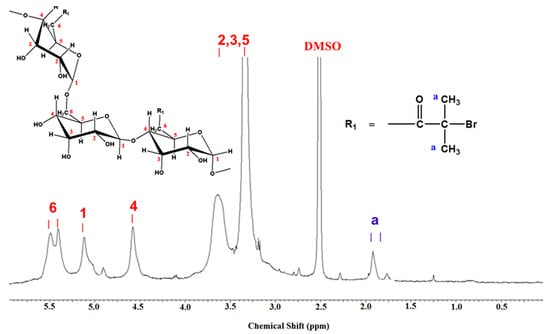
Figure 3.
1H NMR spectra of starch based macroinitiator (St-Br).
3.1.2. Synthesis of St-g-Polymer
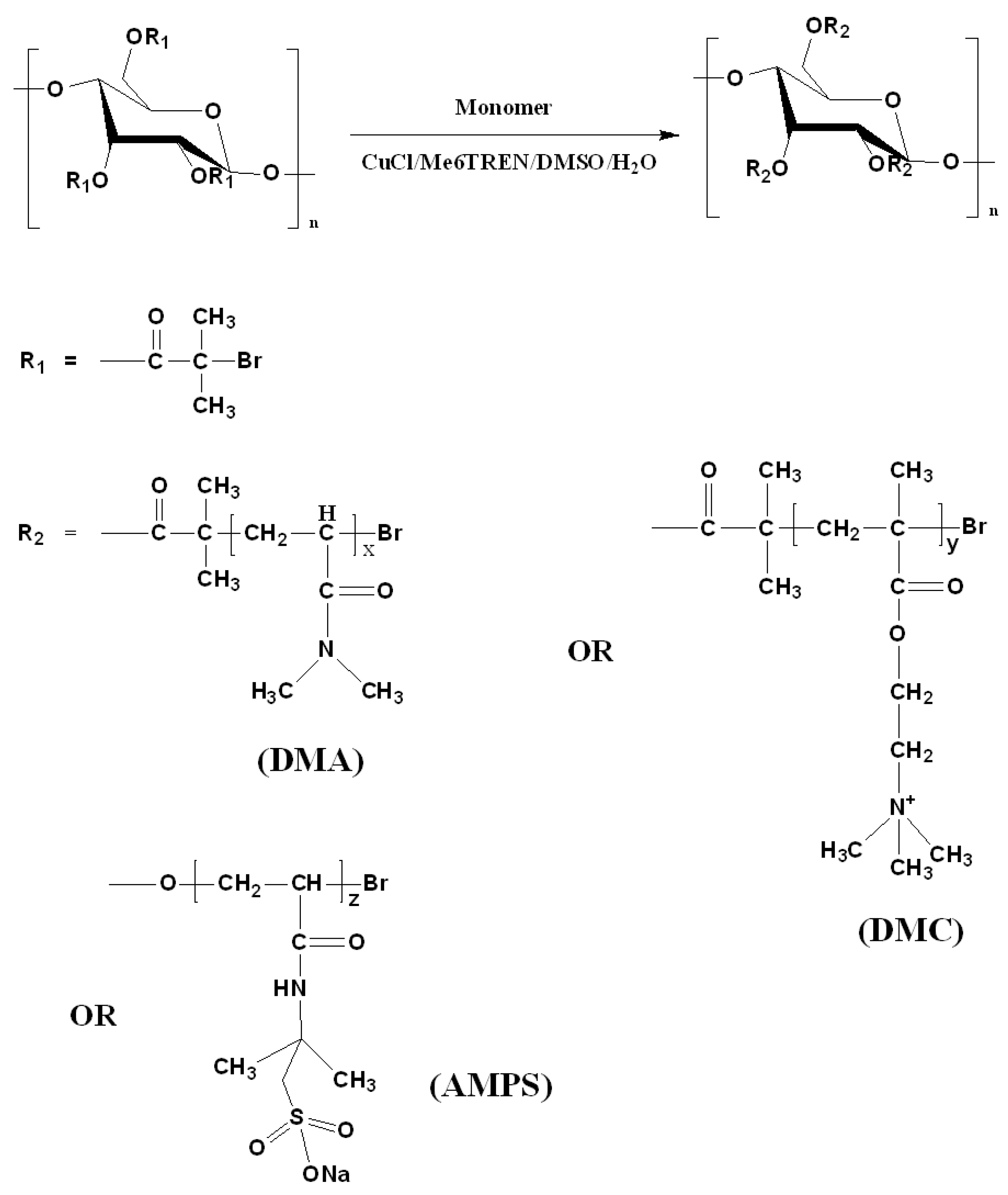
Three types of polymer-grafted starches (St-g-Polymer) with different ionic groups were polymerized using the pre-synthesized macroinitiator with DS of 0.1 (based on 1H NMR). The polymerizations of DMA, AMPS, and DMC are described in Figure 4. ATRP was conducted with constant [Monomer]/[CuCl]/[CuCl2]/[Me6TREN]/[Initiator] ratio of 270:2.2:0.1:5.9:1, using starch-Br as macroinitiator and CuCl/CuCl2/Me6TREN as catalysts. All macroinitiators were grafted in 1:1 v/v DMF/H2O solution. It has been reported that the starch-based macroinitiator can dissolve better in DMF [7]. The DMF addition in this work was to improve solubility of the macroinitiator due to the hydrophobicity increment of starch after esterification of the hydroxyl groups with BiBB. The reason for selecting H2O as a co-solvent was to accelerate the reaction in order to improve the polymer grafting percentage. Thus, a mixed solvent of DMF/H2O was selected for higher solubility of the macroinitiator and to improve the polymer graft density. Although DMF is miscible with H2O, all of the polymerization mixtures were partly heterogeneous with respect to the catalyst. In most of the polymerizations, the reaction mixtures were observed to form a highly viscous mass. The polymerizations were ended when the reaction mixture became too viscous to be mixed. The reason for the observed high viscosity is probably due to the polyfunctionality of the growing graft copolymers which makes them prone to intermolecular termination and therefore also to gelation [8,17,18,19].
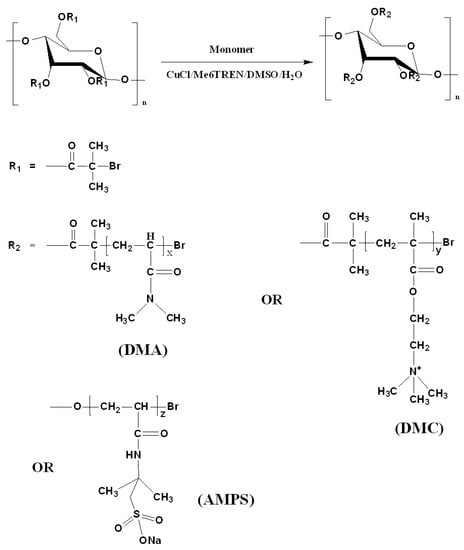
Figure 4.
Synthetic route for St-g-Polymer by ATRP.
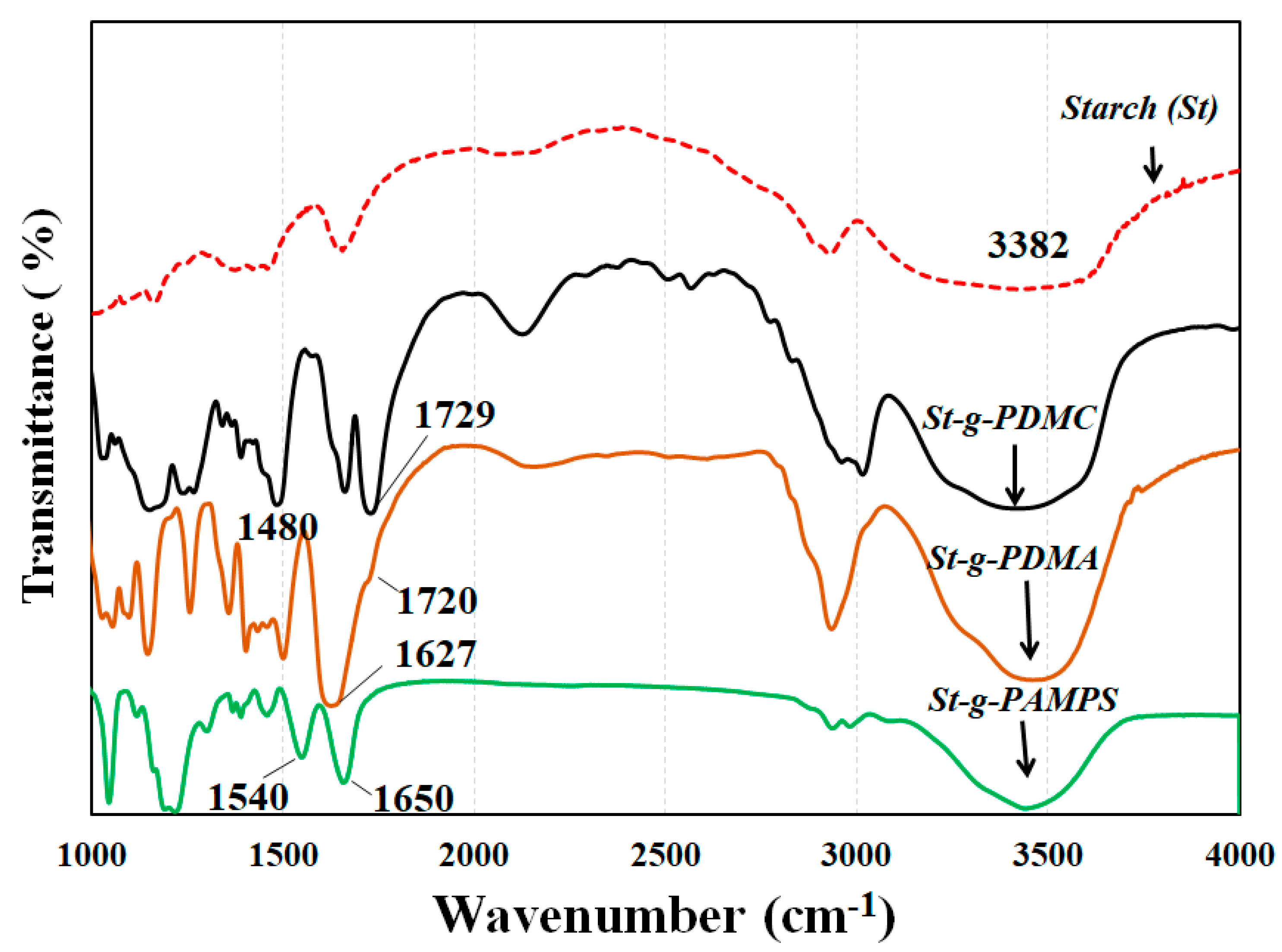
The chemical structures of the St-g-Polymer were characterized by FTIR spectroscopy. Figure 5 shows the representative FTIR spectra of the original starch, poly([(2-methacryloyloxy)ethyl]trimethylammonium chloride) grafted starch (St-g-PDMC), poly(N,N-dimethylacrylamide) grafted starch (St-g-PDMA) and poly(2-acrylamido-2-methyl-1-propanesulfonic acid) grafted starch (St-g-PAMPS). Compared to the original starch, three new peaks appeared. The peak at 1729 cm−1 is due to the vibrating adsorption of carbonyl groups in the grafting PDMC, and the peaks at 1480 cm−1 and 951 cm−1 are assigned to the methyl groups of ammonium and quaternary ammonium in PDMC. This demonstrated that PDMC was successfully grafted onto the starch. When the FT-IR spectrum of St-g-PDMA was compared with that of original starch and St-Br, the characteristic absorptions observed were 1720 cm−1 for carboxyl C=O, 1627 cm−1 for amide C=O, and 1027 cm−1 for the “C–O” group, indicating PDAM was grafted onto the starch backbone. Compared to original starch, the spectra of St-PAMPS shown in Figure 5 had the characteristic peaks at 1540 cm−1 and 1650 cm−1 which are due to the vibration of the -NH group and the carbonyl absorption, respectively. The appearance of the new absorption bands at about 1040, 1180, and 1384 cm−1, which are due to the symmetric, asymmetric, and antisymmetric stretching of S=O groups, confirmed the successful grafting PAMPS onto the backbone of starch.
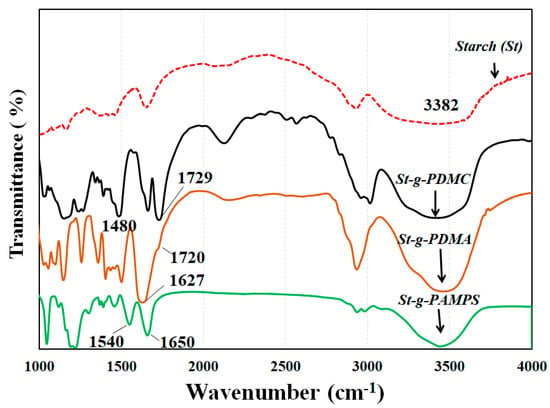
Figure 5.
FTIR spectra of starch (St) and St-g-PDMC, St-g-PDMA, and St-g-PAMPS.
The thermal stabilities of the original starch, St-Br, and St-g-Polymer were studied by thermogravimetric analysis (TGA) in a nitrogen stream at a heating rate of 20 °C/min and depicted in Figure S2. Based on TGA data, the polymer grafting percentages for St-g-PDMC, St-PAMPS, and St-PMDA are estimated as of ~40%, ~50%, and ~50%, respectively.
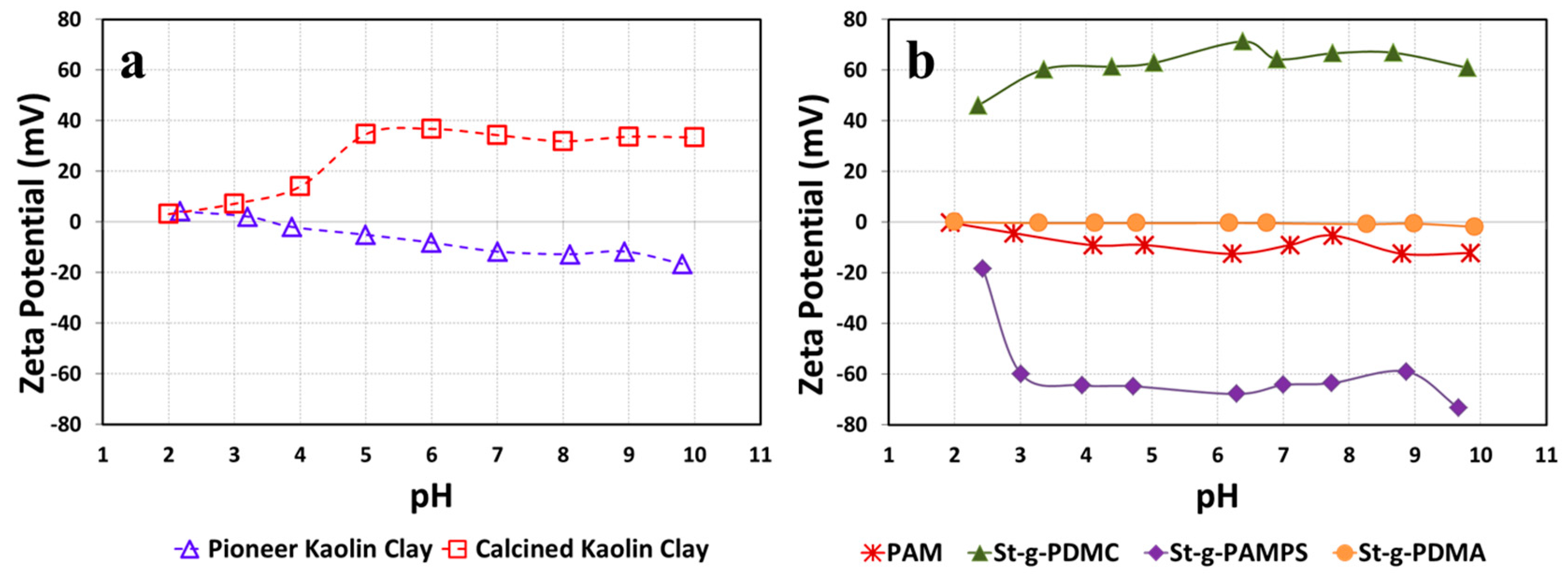
3.2. Charge Properties by Zeta Potentials (ZPs)
Zeta potentials (ZPs) measured in a typical working pH range (2–10) for two types of kaolin clay suspensions are presented in Figure 6a. It was found that the Pioneer clay exhibited an almost neutral charge at a relatively low pH range (2–5) whereas it bore negative charges in the pH range of 5–10. The decreased ZPs with increasing pH for Pioneer clay are probably due to the increment of the counter ion concentrations with respect to the change in pH. In contrast, the Calcined clay carried positive charges under most of the measured pH range (from 3 to 10). Most kaolin clay reported in the literature is negatively charged. However, we noticed that the Calcined clay is positively charged. It is believed that the positive charge of the Calcined clay surface may be because the clay has been modified and the surface chemistry and physical properties were altered [20]. The ZPs of the Pioneer and Calcined kaolin clays averaged −7 mV and 25 mV respectively, indicating a difference in the surface properties of these kaolin clays. The zeta potential indicates the stability of colloidal dispersions. The magnitude of the zeta potential measures the degree of electrostatic repulsion between adjacent, similarly charged particles in the clay suspension. The clay particles with a high zeta potential value will confer stability, i.e., resist aggregation. Therefore, the Calcined kaolin clay is more difficult to flocculate than the Pioneer clay since the Calcined kaolin clay is more stable than the Pioneer clay as shown by its higher ZPs.
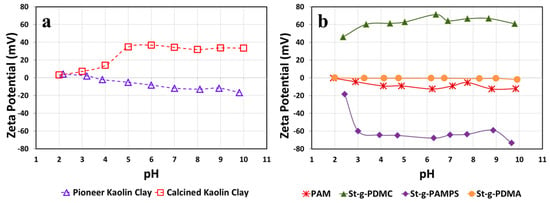
Figure 6.
Zeta potentials (ZPs) for (a) two types of kaolin clay suspensions and (b) various St-g-Polymer and PAM solutions measured on pH range of 2–10.
Meanwhile, the ZPs of various St-g-Polymer solutions also have been measured and reported in Figure 6b, as the charge properties of ionic flocculants would highly affect their flocculation efficiency. St-g-PDMC bore positive charges and presented the characteristics of a strong cationic polyelectrolyte under most of the measured pH range (from 2 to 10). This is due to the introduction of the tertiary amine groups onto the starch backbone which increases the ZPs. This cationic flocculant shows little to no change in ZP with increasing pH, which can be explained by the fact that all the bonding sites in quaternary ammonium are completely occupied leaving no room for protonation or deprotonation of nitrogen. However, as for St-g-PAMPS, after sulfonic acid groups were introduced onto the starch backbones, the negative charges were grafted. This shows the typical characteristics of an anionic polyelectrolyte. It is worth mentioning that a sharp change in the zeta potential of St-g-PAMPS was observed at a pH of 2, which can account for pKa1 (SO32−) = 1.9. Non-ionic PAM was selected as a control flocculant and has ZPs close to zero. Similar to PAM, St-g-PDMA does not bear any surface charge since the introduction of non-charged amide groups onto the starch backbone does not impact the ZPs. The zeta potential of St-g-PDMC, St-g-PAMPS, and St-g-PDMA averaged 62 mV, −60 mV, and −0.6 mV, respectively.
3.3. Effects of Charge Properties on the Flocculation Performance
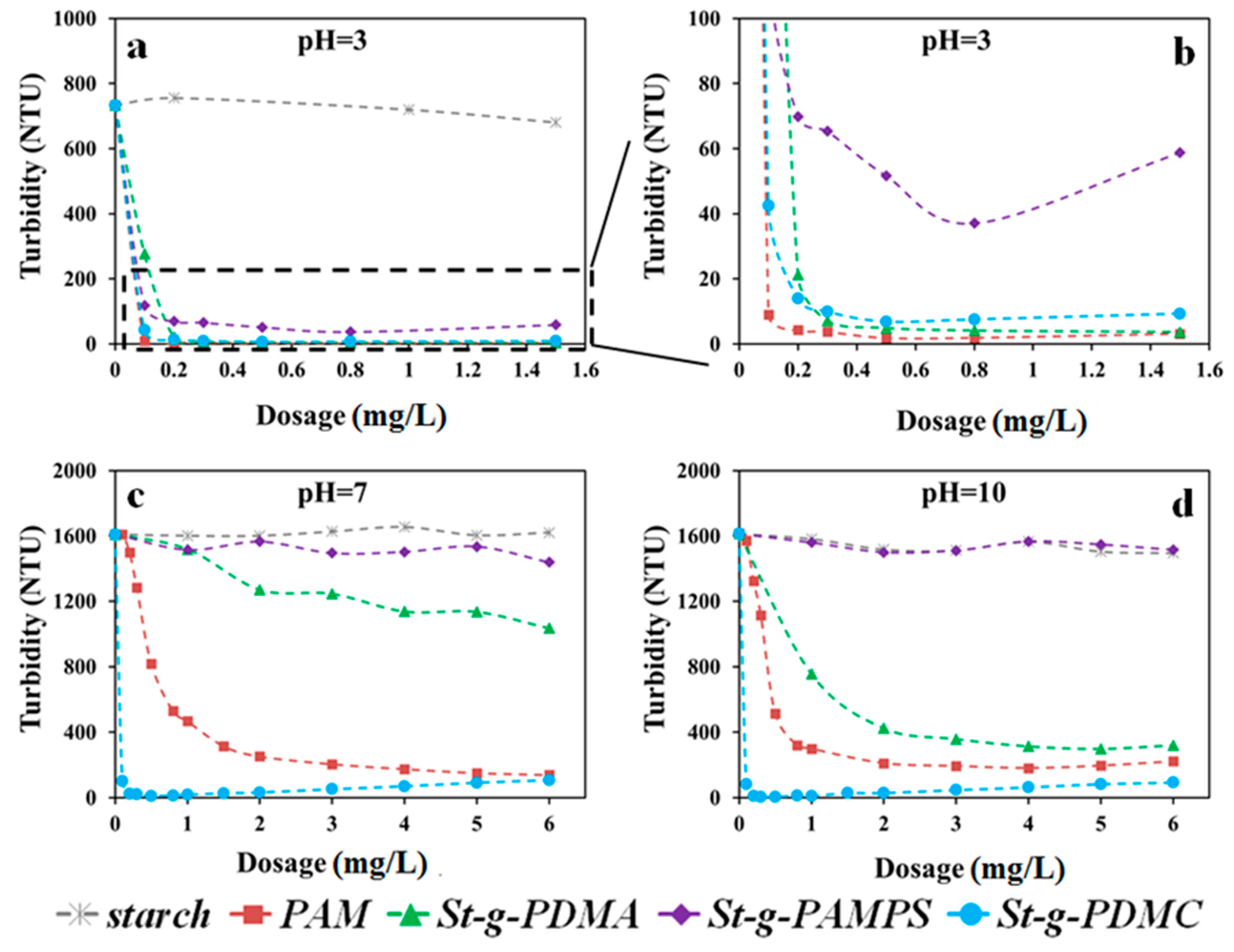
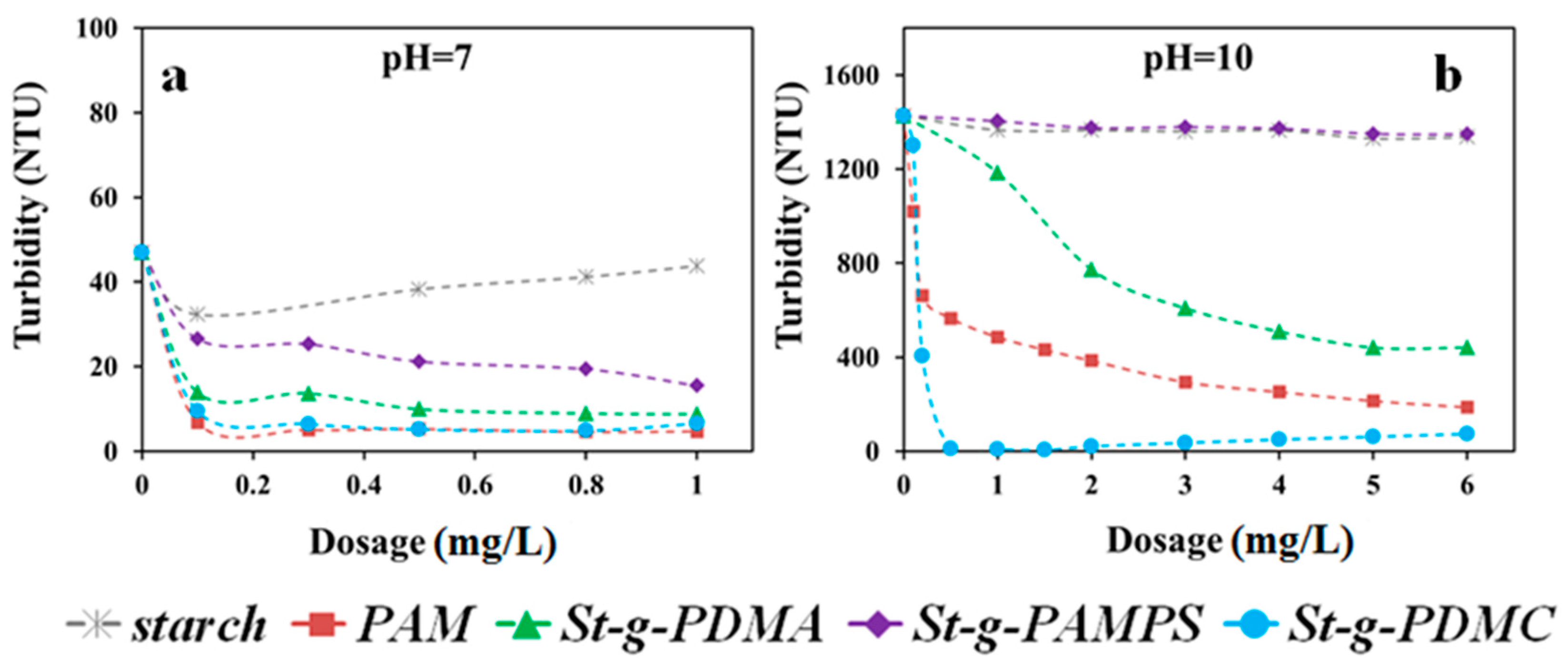
Two types of clay suspension with different characteristics of surface charge, i.e., Pioneer kaolin and Calcined kaolin clays, were employed for comprehensive investigation of the flocculation behaviors using the synthesized St-g-Polymer. Since the dosage and pH are the two major factors in the flocculation process, the flocculation performances for original starch, PAM, and three types of St-g-Polymer flocculants were evaluated under various pH conditions and dosages (Figure 7 in Calcined clay and Figure 8 in Pioneer clay).
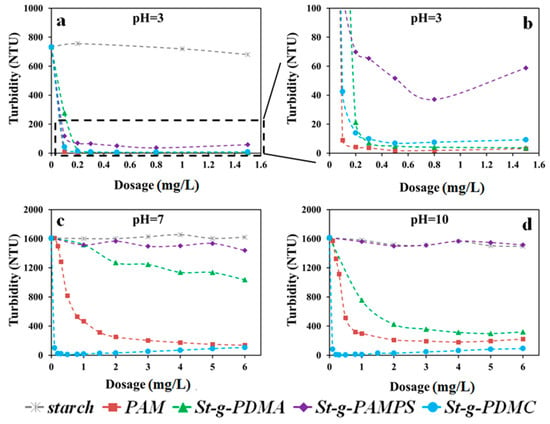
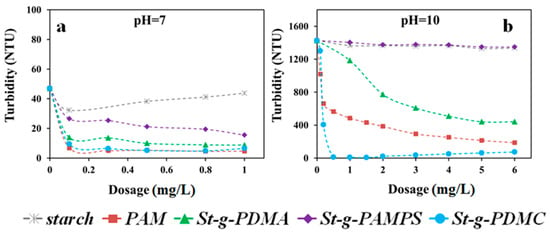
Figure 8.
Turbidity versus dosage for the investigated polymer flocculants in Pioneer kaolin suspensions under (a) pH 7.0 and (b) pH 10.0.
Figure 7 shows the turbidity of the supernatant against the dosage of the investigated polymer flocculants in Calcined kaolin suspensions under pH 3.0, 7.0, and 10.0. As a comparison, the turbidity of pure clay suspensions without any flocculants was measured after settling for 5 min and presented in Figure 7 as the dosage of zero (0 mg/L). The original starch did not show any flocculation ability under the tested conditions, therefore it is not an effective flocculant. For St-g-PDMA, the flocculation performance increased with increasing dosage. For example, at pH of 10.0, the turbidity of St-g-PDMA decreases from 750 NTU to 320 NTU with the increment of the dosage from 1 mg/L to 6 mg/L (corresponding to 1000 g/ton to 6000 g/ton in commonly-used industrial flocculation process). In contrast, St-g-PAMPS exhibited relatively low flocculation performance under acidic conditions compared to the other synthetic flocculants and did not show any flocculation ability under neutral and alkaline conditions. Therefore, it is not suitable as a flocculant in neutral and alkaline conditions. The difference in flocculation performance between these three conditions (pH of 3.0, 7.0, and 10.0) mainly resulted from the difference in zeta potential for Calcined kaolin suspension under each condition (7 mV at pH of 3.0, 33 mV at pH of 7.0, and 33 mV at pH of 10.0). Generally, there are four primary flocculation/aggregation mechanisms in water treatment: (1) double layer compression; (2) charge neutralization; (3) sweep flocculation; (4) adsorption and inter-particle bridging. Although St-g-PAMPS carried charges opposite to that of Calcined kaolin clay particles and charge neutralization could take effect, the flocculation performance of St-g-PAMPS was the lowest among these three types of copolymers. This suggests that adsorption and inter-particle bridging could be the major destabilization mechanisms in this specific clay suspension system.
The St-g-PDMC exhibited the best flocculation performance among the three types of St-g-Polymers under acidic, neutral, and alkaline conditions. For example, at a pH of 7.0, the turbidity of the Calcined kaolin suspension decreased from 1610 NTU to 103 NTU, 1519 NTU, and 1515 NTU by St-g-PDMC, St-g-PDMA, and St-g-PAMPS respectively, at the flocculant dosage of 0.1 mg/L. The St-g-PDMC presented high flocculation performance at the flocculant dosage range from 0.1 to 6 mg/L, implying that it has a broad flocculation window. This is one of the advantages of St-g-Polymer. The grafting chain of PDMC on the semi-rigid starch backbone increased the adsorption bridging ability and the flexibility of the flocculants. Thus, the dispersion re-stabilization for the colloidal particles caused by charge reversion is weakened. With increasing dosage, the flocculation performance of copolymer St-g-PDMC increased to reach a maximum value and then slightly decreased. For example, the lowest turbidity of the Calcined kaolin suspension at pH of 10.0 was 11 NTU with a dosage of 0.8 mg/L. It was indicated that there was a limit to the extent to which St-g-PDMC improved the flocculation efficiency. Under alkaline conditions (pH 10.0), the zeta potential of the Calcined kaolin suspension was high (33 mV) making the clay colloids quite stable in the suspension. Meanwhile, St-g-PDMC carried positive charges that are the same as the Calcined kaolin clay colloids. Therefore, when St-g-PDMC is used as a flocculant for the Calcined kaolin clay suspension, charge neutralization hardly takes place, while adsorption and inter-particle bridging play a critical role. Furthermore, both turbidity and turbidity change rate are useful parameters for determining the flocculation performance. The turbidity change rate for St-g-PDMC is ~94% with a dosage of 0.1 mg/L under acidic conditions, neutral conditions, or alkaline conditions, indicating it was a high efficient flocculant under various conditions at a low dosage. It should also point out that the St-g-PDMC showed a better flocculation performance under neutral and alkaline conditions than PAM. For example, at pH 10 and a dosage of 0.1 mg/L, the turbidity change rate of St-g-PDMC is ~94%, whereas PAM as the baseline is only 0.03%. This is 3000 times higher than the baseline.
Figure 8 shows the effects of dosage on the flocculation performance of the investigated flocculants in Pioneer kaolin suspensions under pH 7.0 and 10.0. Since the St-g-Polymer already exhibited high flocculation performance under pH 7.0 (turbidity less than 20 NTU), the jar test under pH 3.0 was skipped. In theory, the flocculation performance under acidic conditions should be better than that under neutral and alkaline conditions, owing to the difference in the zeta potential of Pioneer clay colloids under these conditions. Similar to the previous tests in Calcined kaolin clay, St-g-PDMC was the best flocculant among the three types of St-g-Polymer under neutral and alkaline conditions. St-g-PDMA gave better performance than St-g-PAMPS. For example, at pH of 10.0, the turbidity of the Pioneer kaolin suspension decreased from 1427 NTU to 8 NTU, 1404 NTU, and 1428 NTU by St-g-PDMC, St-g-PDMA, and St-g-PAMPS, respectively, at a flocculant dosage of 1 mg/L. In addition to the adsorption and inter-particle bridging mechanism, St-g-PDMC may exhibit charge attraction for Pioneer kaolin particles under alkaline conditions, and thus charge neutralization could happen.
The difference in flocculation behavior between the three types of flocculants in kaolin suspension is mainly due to the differences in charge properties, especially for Calcined kaolin suspension under basic conditions. Cationic St-g-PDMC which has a positive charge shows the best flocculation performance amongst these three types of flocculants. In further comparison, the flocculation efficiency follows the order of St-g-PDMC (positive charge) > PAM ≈ St-g-PDMA (no charge) > St-g-PAMPS (negative charge). All of these experimental results could be well explained in terms of the distinct charge properties of the three starch-based flocculants. The grafting of polymer onto starch backbone enhanced the charge properties and increased molecular weight of the copolymer. For the St-g-PDMC, the branch structures resulted in enhancement of bridging effects, which brings significant benefits to the subsequent floc growth and effective Calcined kaolin clay removal. On the other hand, those grafted chains would also cause the floc structures to be slightly loose due to the steric hindrance effects. Overall, adsorption and inter-particle bridging mechanism play a more important role than charge neutralization in the Calcined kaolin clay flocculation. Moreover, the flocculation method is popularly employed for removal of both insoluble suspended colloids with particle sizes from nanometer to micrometers as well as for dissolved matters.
4. Conclusions
Three types of polymer-grafted starches (St-g-polymer) with different charge properties were synthesized and their molecular structures were controlled by ATRP. The success of the grafting was confirmed by FTIR, 1H NMR, and TGA characterization. The flocculation behaviors in kaolin suspension using these three starch-based flocculants were systematically investigated and compared with commercially widely used polyacrylamide (PAM). Besides the effects of dosage, the flocculation performances of the various modified starch flocculants in Pioneer kaolin and Calcined kaolin suspensions were evaluated in acidic, basic & neutral pH environments. The experimental results indicated that the charge properties of the grafted polymers distinctly contributed to flocculation performance at different flocculation conditions (pH, dosage et al.). All of the starch-based flocculants exhibited a highly efficient flocculation ability for 0.1 wt% kaolin suspensions (both Pioneer kaolin and Calcined kaolin) under acidic conditions due to the relatively low ZPs of the clay suspension. Adsorption and inter-particle bridging is the major destabilization mechanism. Under basic conditions, especially for Calcined kaolin suspension, charge properties played an important role in the flocculation. The cationic St-g-PDMC having positive charges showed the best flocculation performance amongst the three types of flocculants, likely due to the benefits from the positive charges for the removal of small-size/water-soluble particles. The results obtained are of importance as guidance in the selection and design of suitable starch-based polymeric flocculants for targeted wastewater treatments. However, this conclusion needs further experimental validation and systematic study to confirm if the flocculants with a positive charge and branch structure can perform efficient flocculation of other dissolved contaminants such as water-soluble dyes, proteins, and polysaccharides. Further investigating the flocculation of these modified starches in oil sand tailings will be conducted in the future.
Supplementary Materials
The following are available online at https://www.mdpi.com/2075-163X/10/12/1054/s1, Figure S1: Particle size distribution (PSD) of Pioneer clay and Calcined clay by intensity. Figure S2: TGA curves of starch, starch-Br, St-g-PDMC, St-g-PAMPS, and St-g-PDMA.
Author Contributions
Conceptualization, N.Z., Z.S. and Y.X.; methodology, N.Z., H.A.B., and Y.Z.; formal analysis, N.Z., H.A.B., Z.S. and Y.X.; writing—original draft preparation, N.Z.; writing—review and editing, Z.S., and Y.X.; supervision, Z.S. and Y.X.; project administration, Z.S. and Y.X.; funding acquisition, Z.S. and Y.X. All authors have read and agreed to the published version of the manuscript.
Funding
This research work was financially supported by The Program of Energy Research and Development (PERD), Natural Resources Canada (NRCan). The APC was funded by Energy, Mining and Environment (EME) research centre, National Research Council Canada.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Huang, M.; Wang, Y.; Cai, J.; Bai, J.; Yang, H.; Li, A. Preparation of dual-function starch-based flocculants for the simultaneous removal of turbidity and inhibition of Escherichia coli in water. Water Res. 2016, 98, 128–137. [Google Scholar] [CrossRef] [PubMed]
- Malafaya, P.B.; Elvira, C.; Gallardo, A.; San Román, J.; Reis, R.L. Porous starch-based drug delivery systems processed by a microwave route. J. Biomater. Sci. Polym. Ed. 2001, 12, 1227–1241. [Google Scholar] [CrossRef] [PubMed]
- Tan, Y.; Xu, K.; Wang, P.; Li, W.; Sun, S.; Dong, L. High mechanical strength and rapid response rate of poly(N-isopropyl acrylamide) hydrogel crosslinked by starch-based nanospheres. Soft Matter 2010, 6, 1467–1471. [Google Scholar] [CrossRef]
- Tan, Y.; Xu, K.; Li, Y.; Sun, S.; Wang, P. A robust route to fabricate starch esters vesicles. Chem. Commun. 2010, 46, 4523–4525. [Google Scholar] [CrossRef] [PubMed]
- Matyjaszewski, K. Atom Transfer Radical Polymerization (ATRP): Current Status and Future Perspectives. Macromolecules 2012, 45, 4015–4039. [Google Scholar] [CrossRef]
- Liu, P.; Su, Z. Surface-initiated atom transfer radical polymerization (SI-ATRP) of n-butyl acrylate from starch granules. Carbohydr. Polym. 2005, 62, 159–163. [Google Scholar] [CrossRef]
- Wang, L.; Shen, J.; Men, Y.; Wu, Y.; Peng, Q.; Wang, X.; Yang, R.; Mahmood, K.; Liu, Z. Corn starch-based graft copolymers prepared via ATRP at the molecular level. Polym. Chem. 2015, 6, 3480–3488. [Google Scholar] [CrossRef]
- Nurmi, L.; Holappa, S.; Mikkonen, H.; Seppälä, J. Controlled grafting of acetylated starch by atom transfer radical polymerization of MMA. Eur. Polym. J. 2007, 43, 1372–1382. [Google Scholar] [CrossRef]
- Avval, M.E.; Moghaddam, P.N.; Fareghi, A.R. Modification of starch by graft copolymerization: A drug delivery system tested for cephalexin antibiotic. Starch-Stärke 2013, 65, 572–583. [Google Scholar] [CrossRef]
- Ashby, M.F.; Shercliff, H.; Cebon, D. Materials: Engineering, Science, Processing and Design; Butterworth-Heinemann: Oxford, UK, 2018. [Google Scholar]
- Ravve, A. Principles of Polymer Chemistry, 3th ed.; Springer: New York, NY, USA, 2012. [Google Scholar] [CrossRef]
- Wessel, J.K. (Ed.) Handbook of Advanced Materials: Enabling New Designs; John Wiley & Sons, Inc.: Hoboken, NJ, USA, 2004. [Google Scholar] [CrossRef]
- Khayet, M.; Zahrim, A.Y.; Hilal, N. Modelling and optimization of coagulation of highly concentrated industrial grade leather dye by response surface methodology. Chem. Eng. J. 2011, 167, 77–83. [Google Scholar] [CrossRef]
- Li, H.; Du, Y.; Wu, X.; Zhan, H. Effect of molecular weight and degree of substitution of quaternary chitosan on its adsorption and flocculation properties for potential retention-aids in alkaline papermaking. Colloids Surf. A 2004, 242, 1–8. [Google Scholar] [CrossRef]
- Hu, P.; Xi, Z.; Li, Y.; Li, A.; Yang, H. Evaluation of the structural factors for the flocculation performance of a co-graft cationic starch-based flocculant. Chemosphere 2020, 240, 124866. [Google Scholar] [CrossRef] [PubMed]
- Guo, Y.; Kong, F.; Fatehi, P. Generation and use of lignin-g-AMPS in extended DLVO theory for evaluating the flocculation of colloidal particles. ACS Omega 2020, 5, 21032. [Google Scholar] [CrossRef] [PubMed]
- Pyun, J.; Kowalewski, T.; Matyjaszewski, K. Synthesis of Polymer Brushes Using Atom Transfer Radical Polymerization. Macromol. Rapid Commun. 2003, 24, 1043–1059. [Google Scholar] [CrossRef]
- Vlček, P.; Janata, M.; Látalová, P.; Kríž, J.; Čadová, E.; Toman, L. Controlled grafting of cellulose diacetate. Polymer 2006, 47, 2587–2595. [Google Scholar] [CrossRef]
- Masař, B.; Janata, M.; Látalová, P.; Netopilík, M.; Vlček, P.; Toman, L. Graft copolymers and high-molecular-weight star-like polymers by atom transfer radical polymerization. J. Appl. Polym. Sci. 2006, 100, 3662–3672. [Google Scholar] [CrossRef]
- Jordan, J.W. Industrial Applications of Kaolin. Clays Clay Miner. 1961, 10, 291–298. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).