Assessing Soil Quality in Areas Affected by Sulfide Mining. Application to Soils in the Iberian Pyrite Belt (SW Spain)
Abstract
: The characterization, evaluation and remediation of polluted soils is one of the present environmental challenges to be addressed in the coming years. The origin of trace elements in soils can be either geogenic or anthropogenic, but only the latter is interesting from a legal point of view. The hazard of the pollutants in the soils not only depends on their total concentration, but particularly on their availability. The mobility of the trace elements depends on their speciation, and it is also affected by several soil parameters. Mining activity is one of the most important anthropogenic causes of soil pollution. As a case study, this work is focused in the Riotinto mining area (Iberian Pyrite Belt, IPB, SW Spain). The IPB is one of the most important metallogenic provinces in the world and it has been exploited for thousands of years. The disposal of mining residues has produced important sources of contamination by trace elements and acidic waters affecting soils and rivers. In addition to these problems, the closure of mines in the Pyrite Belt at the end of the 20th Century has led to a great loss of employment, which has caused the development of an intensive agriculture of citrus fruits as a new source of income. The intensive growing of citrus fruits and the traditional subsistence agriculture have been developed surrounding the mining areas and on floodplains near to mining sites. The level of soil pollution has not been taken into account in these cases, nor has its impact on the health of the inhabitants of these areas. Therefore, it is of great interest to study the current state of the cultivated soils and the sources and types of contaminants derived from mining activity in order to program its decontamination, where appropriate, according to legislation. In order to know the present and future hazard posed by the soils chemical and mineralogical speciation has been carried out, given that the availability of a metal depends on the phase in which it is found. The results showed that mining activity has caused high levels of As, Cu, Pb and Zn in several cultivated soils. Moreover, Cu, Pb and Zn showed a high bioavailability. This suggests that at least other studies are necessary to preserve health in the inhabitants of this area. The methodology carried out in this work allowed to select potentially polluted areas where agricultural activities are not recommended.1. Introduction
1.1. Contamination and Contaminants
The presence of harmful concentrations of some chemical elements and compounds in soil is a special type of degradation known as contamination. Contaminants are always found in anomalous (higher than usual) concentrations and generally have a harmful impact on some organisms. The origin of contaminants is either geogenic if they come from soil parent rocks, volcanic activity or mineral leaching, or anthropogenic if they come from hazardous industrial waste, agricultural or mining activities, or urban solid waste from households. However, anthropogenic contaminants are the only ones legally considered as true contaminants.
The toxicity of a chemical element or compound is its ability to adversely affect biological functions. With the only exception of toxins (which are biogenic), toxic compounds and materials have a non-biological origin.
Contaminants are mobilized by physical, chemical and biological vectors. They can leave soil by volatilization, dissolution, leaching or erosion and enter organisms when they are in a bioavailable (relatively soluble) form. Thus, the potential of an element––whether a contaminant or otherwise––passing from soil into its solution is known as “availability”. The more specific term bioavailability is the degree to which a contaminant in a potential source is free for uptake (movement into or onto an organism) [1]. Usually, only a small fraction of each potentially contaminating substance in a medium is bioavailable. Although contaminants are generally noxious, some may be harmless to specific organisms.
Bioavailability is a function of (a) the specific physical and chemical form in which an element or substance occurs, and (b) the ability of organisms to absorb or ingest it. Bioavailable elements can be accumulated in an organism (i.e., bioaccumulation) at concentrations up to 3–5 orders of magnitude higher than those in their medium.
1.2. Heavy Metals and Trace Elements
The periodic table includes about 70 metals, 59 of which are heavy metals (i.e., metals with an atomic weight exceeding the weight of iron, which is 55.85 g/mol). This definition excludes metals with a lower atomic weight, which however can also include contaminants. Such is the case for V, Mn and Cr, which have an atomic weight of 50.95, 54.44 and 52.01 g/mol, respectively. It also happens with other non-metal elements such as As, F and P. Therefore it is more accurate to refer to them as trace elements, even though most inorganic contaminants are in fact heavy metals. Occasionally, soil is contaminated with high concentrations of major elements such as Na, Fe or Al.
Trace elements are present at low concentrations (mg·kg−1) in the earth crust, soils and plants. Many of them (Table 1 [2]) are essential for plant, animal and human growth, but can be toxic above certain concentration levels. As a rule, trace elements are toxic if inhaled or ingested in large quantities and over a long period of time. For example, the deficiency and toxicity thresholds for Se, F and Mo differ from one another only by a few parts-per-million (ppm) [3].
Most of trace elements in soil can be classified in five groups according to their chemical form in soil solution, namely: cations (Ag+, Cd2+, Co2+, Cr3+, Cu2+, Hg2+, Ni2+, Pb2+, Zn2+), native metals (Hg, V), oxianions (AsO43−, CrO42−, HSeO3−, SeO42−), halides (F−, Br−, I−) and organometals (Ag, As, Hg, Se, Te, Tl). These groups are not mutually exclusive and, in fact, some trace elements can fall into more than one. Typically, the concentrations of Cr, Ni, Pb and Zn in soil range from 1 to 1500 mg·kg−1; those of Co, Cu and As from 0.1 to 250 mg·kg−1; and those of Cd and Hg from 0.01 to 2 mg·kg−1 [4]. Table 2 lists other usual and anomalous geochemical concentrations of a variety of elements [5].
From all of the trace elements found in soil, the following 17 are especially toxic and available at concentrations exceeding toxic levels: Ag, As, Bi, Cd, Co, Cu, Hg, Ni, Pb, Pd, Pt, Sb, Se, Sn, Te, Tl and Zn. Ten of them (Ag, As, Cd, Cu, Hg, Ni, Pb, Sb, Sn and Tl) are readily mobilized in concentrations exceeding considerably their geological levels due to human activities [6]. The latest US Environmental Protection Agency (EPA) list of priority pollutants includes the following 13 trace elements: Sb, As, Cd, Cr, Cu, Hg, Ni, Ag, Pb, Se, Tl, Zn and Be, which was not included in previous lists.
1.3. Purifying Power of Soil
Soil usually acts as a protective barrier for more sensitive media such as water and organisms by filtering, decomposing, neutralizing or storing contaminants. Hence it substantially reduces their bioavailability. The purifying ability of soil depends on its contents in organic matter, carbonates, and Fe and Mn oxyhydroxides, as well as on its clay content and mineralogical composition, cation exchange capacity, pH, Eh, texture, permeability and microbial activity. Therefore, the purifying power of soil has a limit for each substance. If this limit is exceeded, the soil becomes contaminated and is a source for contamination.
The load capacity of soil shows its ability to control the adverse impacts of contaminants and render them inactive or harmless. Cheng et al. [7] proposed the Load Capacity of Agricultural Soils for Heavy Metals (LCASHM), which depends on soil properties, the type and contamination history, the presence of toxicity indicators and various environmental parameters.
1.4. The Origin of Trace Elements in Soil
Trace elements present in soil can be geogenic or anthropogenic. The presence of the former is largely determined by their geoavailability, which is that portion of the total content of a chemical element or a compound in an earth material that can be liberated to the surficial or near-surface environment (or biosphere) through mechanical, chemical, or biological processes [8].
Geoavailable trace elements typically reach the soil when they are released from their parent rock because of weathering. Together with metals from volcanic emissions and mineral leachates, they constitute the so-called “geogenic metals”. The ease with which trace elements can be released from their parent rock depends on the structure and the thermodynamic stability of the rock forming minerals. Thus, highly polymerized structures are especially stable and resistant to destruction, and their stability decreases according to the following sequence of atomic bonds: Si−O > Al−O > X−O.
The amount of geoavailable elements released from rocks into soil is negligible compared to those of anthropogenic origin. Weathering causes trace elements to concentrate in soil, but usually at levels below their toxic thresholds. Even if these levels are exceeded, there will be almost no risk of toxicity due to high stability and the low availability of these metals in soil.
Ultrabasic rocks can contain large amounts of some trace elements such as Cr, Ni, Cu or Mn. On the other hand, acid igneous rocks and sedimentary rocks (sandstone and limestone) contain very small amounts of them. The highest levels of trace metals in soils––inherited from the parent rock––are typically those of Cr, Mn and Ni, followed by Co, Cu, Zn and Pb, and, far beyond As, Cd and Hg, which are usually present at very low concentrations.
Anomalous high concentrations of trace elements in soil usually come from metal ores (sulfides, oxides). Metal mining and extraction produce tons of waste each year; most of it contains pyrite and other sulfides whose oxidation releases a large amount of trace elements into the environment, especially into soils. The upper layers of soil around mining and extraction areas can contain high concentrations of Cu, Ni, As, Se, Cd or Fe among other elements, depending on the particular ore.
Trace elements in soil can come from various other anthropogenic activities in addition to mining, namely:
Farming (irrigation, inorganic fertilizers, pesticides, manure, liming amendments and, especially, sewage sludge).
Electrical power production. Coal burning is one of the primary sources of metal deposition in soil. For example, thermal power plants typically release Pb, Ni and V contained in oil.
Industrial activities (particularly iron and steel making factories, which release metals contained in Fe and Ni ores). Heavy industrial areas are typical sources of As, Cd, Cr, Hg, Fe, Ni, Pb and Zn.
Household activities. Roughly 10% of all household waste consists of metals. Therefore, its burial can lead to the contamination of groundwater and its ashing to atmospheric contamination with volatile metals and their subsequent incorporation into soil.
2. Soil Contamination by Trace Elements
2.1. Physical State and Relative Availability of Soil Contaminants
Contaminants in soil and sediments can be present in six different forms, namely [9]: particles (particulate contaminants), liquid films, adsorbed, absorbed, and solids or dissolved matter in pore water. The behavior of a contaminant in soil depends on its particular form. As a result, total concentration of trace elements in soil may represent their potential toxicity in relation to agreed thresholds, but not an actual risk. Therefore, the accurate assessment of their toxicity requires determining not only their concentrations, but also their physical and chemical forms, as well as their availability, which provides a direct measure of their actual hazardousness. Lastly toxicity of trace element concentration in the soil should also be tested against some specific organisms. Nevertheless the ease with which a potentially toxic substance can enter the food chain from soil depends on whether the metal is in free form in the interstitial solution or in solid form, and also on the way the metal is bound to soil particles and on its specific chemical form (i.e., its speciation). According to Kabata-Pendias [10], how strongly a metal is retained by soil conditions dictates its relative availability to plants and hence the risk of its reaching organisms (Table 3).
2.2. Contaminant Mobility in Relation to Soil Parameters
The mobility of a metal depends not only on its chemical speciation, but also on various soil-related parameters such as pH, organic matter, carbonates and clay minerals. Thus, not all the exchange cations are equally available; rather, their availability depends on the particular minerals containing them in the exchange complex. For example, metals found in smectite or illite do not necessarily behave identically. Also, if the metal is found in its precipitated form, there is no reason why it has to behave identically, whether it is in a carbonate, sulfate or phosphate. Likewise, metals in sulfides—which are relatively easily oxidized and dissolved––will usually behave differently from metals in silicates, which are virtually stable in any medium.
Usually trace elements show a very low mobility and accumulate within the top few centimeters of soil as a result. Therefore they are leached in very small amounts to the lower horizons. Hence an anthropogenic metal present at high concentrations in the upper soil horizon will usually be at much lower levels in deeper layers. However, this behavior is additionally dependent on the properties of the particular soil. The following soil variables are crucial with a view to assessing the sensitivity of soil to contaminant aggressions:
pH. Most metals tend to be more readily available in acid media, where they are less strongly adsorbed. As an exception, As, Mo, Se and Cr are more mobile in alkaline media.
Texture. Clayey soils are highly effective in retaining metals by adsorption (or in their exchange complex of clay minerals). On the other hand, sandy soils are unable to fix metals, which can thus easily reach the water table.
Clay minerals. Clay minerals differ in specific surface area and electrical charge. Thus, the higher the specific surface area of a silicate is, the higher its ability to adsorb metals [11]. However, absorption by clay minerals is negligible in soils containing large amounts of organic matter and/or iron oxyhydroxides which are more effective in this respect [12].
Organic matter reacts with metals to form exchange complexes or chelates. Metals can be adsorbed to the extent of being stabilized as a result (Cu) or form highly stable chelates with organic matter (Pb, Zn).
Cation exchange capacity. The cation exchange capacity of soil depends on the clay minerals, organic matter, and valence and hydrated ionic radius of the metal. The greater the radius and lower the valence are, the less strongly the metal will be retained.
Redox potential. The redox potential dictates whether the metal is present in oxidized or reduced form. Eh–pH plots provide information about the stability of metal compounds and it is a convenient method for predicting the response of trace elements to a change in environmental conditions.
Carbonates. The presence of carbonates facilitates preservation of a highly alkaline medium and hence precipitation of trace elements. Also, Cd and other metals tend to be absorbed by carbonates.
Iron and manganese oxides and hydroxides. They are finely spread across soil and hence they are highly active. Their low crystallinity and small particle size make them highly sorptive for divalent metals especially Cu and Pb, and, to a lesser extent, Zn, Co, Cr, Mo, Ni and As.
Salinity. An increased salinity can result in the mobilization and retention of trace elements via two different mechanisms. First, Na and K can replace metals at cation exchange sites, and then chloride and sulfate ions can form more stable compounds with previously released metals such as Pb, Zn, Cu and Hg. The salts usually make soil alkaline.
2.3. Assessing the Hazardousness of Metals in Soil
The hazardousness (mobility and availability) of a contaminant can be assessed by using various chemical and mineralogical procedures. Whether a given element or compound is present in a water-soluble phase––and hence readily available to be transferred to a plant or living organism––can be checked by assessing its mobility via its water solubility. This process requires the extraction to happen with neutral or slightly acidic water.
Another way of assessing the transferability of metals (particularly divalent cations) to plants is extracting soil with DTPA or 0.05 M EDTA at pH 7 [13]. There is a wide consensus about the fact that this process provides quite an accurate measure of the amount of metal that plants can absorb from soil under regular conditions [14].
These simple methods can only be used to discriminate between residual metal and metal extracted by water or a mild organic solvent. However there are more elaborated methods involving sequential chemical extractions to carry out speciation of elements in soil (i.e., operational speciation). Also chemical speciation is used to discriminate between oxidation states of an element, and functional speciation, which distinguishes between molecular forms. The plant available fraction of an element in soil depends on its chemical and functional speciation (i.e., of its distribution among its different species and chemical forms).
Operational speciation, which is often referred to as simply “speciation”, is the most frequent choice for soils because it is the simplest analytically and can be virtually implemented in any laboratory with usual available means. However, proper speciation requires determining the total concentration of each metal and its different chemical species. The selectivity of each method used for this purpose depends largely on how fast the formation and dissociation reactions of the target species occur. Roughly, classical procedures used in this context that isolate and examine each constituent separately, are subject to two major shortcomings, namely: (a) complete isolation is very difficult or even impossible; and (b) the properties of a component in a complex mixture are not additive owing to the many interactions it can have with other components.
Sequential extractions do not allow us to complete full speciation processes but they can be useful to characterize the behavior of a contaminating element as they allow its content in the fraction associated to each soil component related to its mobility and transferability to living organisms. In summary, sequential extraction procedures can provide rough estimates of the distribution of trace elements in different soil phases and their relative mobility.
Sequential extraction procedures have been around for at least four decades. During that time they have been continuously modified and updated, or even replaced with others procedures providing us with improved results about specific elements and soil phases. Hall et al. [15] reviewed the most widely used speciation sequences for metals in soils and sediments. The procedure essentially involves obtaining various consecutive fractions by extracting procedures with different solvents to determine the distribution of trace elements in a solid sample between its constituent phases. Such a distribution is usually called partitioning. López Julián and Mandado [16] critically reviewed available speciation methods of this kind and their operational protocols.
One of the most popular protocols was initially proposed by Tessier et al. [17]. It has been the subject of a number of variations [18-21]. Briefly, the procedure is as follows: fractions 1 to 3 (F1–F3) are the results of extraction with ammonium acetate, a reductive solution made of hydroxylamine hydrochloride in acetic acid, and hydrogen peroxide in a nitric medium, respectively, whereas fraction 4 (F4) is the insoluble residue remaining after the three extractions (Table 4).
F1 contains metals in the exchange complex and those forming or being absorbed in carbonates. The treatment used to obtain this fraction involves two steps: extraction with 1 M MgCl2 for 1 h to separate exchangeable cations (i.e., those which can be replaced with other more competitive cations in clay minerals, amorphous materials, etc.) and extraction with sodium acetate-acetic acid at pH 5 for 5 h to attack carbonates. F2 contains metals in reducible compounds (Mn oxides, and amorphous or low-crystalline Fe- and, occasionally, also Al-hydroxides). F3 is the oxidizable phase and contains organically bound (complexed, adsorbed and chelated) cations in addition to some sulfides. Finally, the residual fraction (F4) contains metals in the mineral structure of each extraction-resistant mineral.
Overall, the performance of sequential extraction procedures is limited by factors such as (a) the selectivity of the reagents, (b) potential redistribution (or re-adsorption) of elements, (c) the experimental conditions used and (d) a scarcity of reference materials, which often prevents validation and precision assessment.
A number of extractants are not fully selective for some metals present in specific geochemical phases. Such is the case of soil containing large amounts of iron hydrous oxides, which require repeating their extraction with hydroxylamine hydrochloride several times. Altering the pH to ensure that all metals associated to the target phases will be completely released [20,21] is also needed.
In summary, sequential extraction protocols should be used with great caution, both when selecting the specific scheme to be applied and when interpreting the results. Some of the most important criteria for validating an analytical method (e.g., its theoretical background, cross-validation with other techniques and sample-independence of the results) cannot be applied to these procedures, which precludes validation of their operational results. However, they are bound to continue in use, constantly being improved and updated, until a universally accepted standard protocol is developed. It will happen this way because the information they provide about distributions bears a direct relationship to the geochemical behavior and the bioavailability of chemical elements in nature [22].
Currently, analyses often include a mineralogical study of the fractions obtained from each extraction. This not only shows us the particular elements released, but also the specific minerals dissolved by each extractant, to be known, i.e., chemical-mineralogical speciation. Dold and Fontboté and Dold [23,24], developed a speciation scheme adapted to sulfide mining residues which involves six steps and mineralogical analysis of each extract in relation to the mineralogical composition of the sample.
Indirect and semi-direct methods for characterizing solid phases by infrared, X-ray fluorescence spectrometry or energy dispersive spectrometry are being superseded by direct speciation methods based on more sophisticated techniques such as X-ray photoelectron spectroscopy (XPS), extended X-ray absorption fine structure spectroscopy (XANES), X-ray absorption near-edge structure spectroscopy (XAF) or X-ray absorption spectroscopy (XAS), all of which afford complete chemical speciation [25,26]. These techniques provide a more precise description of processes about solid surfaces, whether crystalline or not, and chemical species at atomic level. However, some analyses require using a synchrotron, which is usually inaccessible for many laboratories. Also, the routine work needed to process hundreds of samples makes these alternative procedures rather slow and expensive unless they are used to determine a single element.
Another classical way of assessing the mobility of trace elements is by leaching, whether on site or at the laboratory. By using test columns or batch tests, one can run isotherms or adsorption-desorption curves and model the results as a function of pH or the amount of each contaminant and its nature with a view to predicting the behavior of a soil. This methodology can be used in combination with one of the previous alternatives. For example, adsorption-desorption isotherms can be used jointly with speciation results obtained from sequential extractions and metal solubility modeling conditions to predict the geochemical mechanisms governing metal leaching and mobility in soil. The software PHREEQC-2 [27] is among the most widely used for modeling in this context.
Although all speciation methods (operational, functional, chemical, chemical-mineralogical, direct) and modeling of their results provide us with a broad picture of the potential “availability” of elements in soil and their ease of absorption by plants, each type of plant absorbs elements in a specific way. Thus, some elements are more readily absorbed, and some plants are more selective absorbers, than others. Chojnacka et al. [28] defined the so-called “transfer coefficient” for a metal as its concentration in plant tissue in relation to soil. This coefficient is maximal for Cd, Tl, Zn, Sn and Cu; intermediate for Ni; and minimal for Co, Cr, Pb, As and Sn. As a rule, the more persistent a given trace contaminant in soil is, the more stable it is and the lower its bioavailability and toxicity are.
3. Claim of Soils Contaminated by Trace Elements
Whereas to claim a soil is contaminated is purely an administrative action, to actually demonstrate it is not an easy task. Within the context of this current work, contamination could be defined as the presence of an anomalous high level of a given element in the soil. However since no soil in the world is pristine and completely uncontaminated, it is quite difficult to determine if it has been contaminated or not. In fact, every soil has been under some kind of human pressure leading to a greater or lesser alteration of its original nature. The contents in many elements have in fact risen in time over their original clark value (viz. the mean abundance in the 15–20 km deep layer of the earth crust). Another way of comparing the relative abundance of trace elements in soil is normalizing against a universal element assumed to be stable in most geochemical environments. Such is the case of Al; multiple comparisons have revealed increasing concentrations of Cd, Pb and As in soils.
The term contamination threshold defines the lowest level representing contamination or, conversely, the highest allowed level in uncontaminated soil. In any case, thresholds must always be checked against toxicological tests before they are deemed accurate. Also, any soil with element levels exceeding their respective thresholds does not have to be necessarily contaminated even if it contains trace elements at concentrations over their regional means.
The toxicity of an element depends on its biological activity. Moreover, it is unique for each species and a function of its way of access (breathing, ingestion, contact). Therefore, only a properly conducted toxicological study can determine the exact allowable bioavailable amount of a trace element. Because the highest toxicity is always what results from ingestion as an available element in solution, the maximum levels allowed are usually set foreseeing this way of access.
Overall, the available fractions of soil amount to that extracted by EDTA and account for about 10% of the whole mass––or as much as 40% depending on the above-described soil variables.
There are thus reference thresholds and contamination thresholds. Soils with intermediate trace element levels always require experimental confirmation showing they are in fact contaminated. Moreover thresholds vary with the use of soil, which obviously influences the particular elements it may contain and their hazardousness. Thus, some concentrations of elements can be critical in gardens and residential areas, but they have a minimal impact on industrial areas.
Despite these assumptions and the international consensus about the specific variables to be considered in setting reference levels, each country––or even each region, state or community––tends to establish its own maximum acceptable concentrations (MAC) (Table 5) [10]. That occurs because the maximum level of elements allowed for each soil differ with the local conditions, land use, regional history, soil type, climate and, obviously, the particular factors considered and the calculation methods used.
No regulations defining soil contamination by trace elements have been passed in Spain to date. The Royal Decree 9/2005, of 14 January (BOE 18 January 2005), which lists potentially soil contaminating activities and sets the criteria and standards for deeming a soil contaminated, sets limits for organic compounds. However it has been left to Spanish regional governments how to regulate the criteria and maximum levels for trace elements in the soil, owing to the strong influence that the parent rock has on the background. The regional governments of Madrid [29], the Basque Country [30], Aragon [31], Castilla-La Mancha [32], Catalonia and Galicia (Decree 60/2009, of 26 February) have issued their own regulations. Prior to issuance of Royal Decree 9/2005, the Andalusian government set the maximum regional levels for agricultural soils (Table 6) [33] and, subsequently, it provided guidelines to assess anomalous levels in eastern Andalusia [34].
The geochemical background of a chemical element represents its concentration in uncontaminated soil. Because it is virtually impossible to determine, it is usually replaced by the geochemical baseline, which is the average of superficial geochemical variations at the time of sampling. Geochemical baseline is obviously influenced by subsoil lithology and the presence of diffuse anthropic contamination [37-39]. Therefore, the measurements taken exclude areas directly exposed to contamination sources.
There is also the regional geochemical baseline, which is the geochemical background level for each region and element as determined with provision for the specific geological context of the region. This value allows us to discriminate between natural and anthropogenic contaminants. Also, they allow the potential allowable load of a contaminant in a specific soil to be controlled via contextual variables like geology, history and use, and also to provide the competent authorities with reference levels.
In any case, human health can only be assured by setting the so-called generic reference levels (GRL), which allow us to efficiently manage contaminated soils. GRL applied to an element provides us with the element concentration in soil, below which no hazards may be expected, and the risk threshold that determines if human health are affected. When GRL is exceeded in a specific site, the soil there should be carefully analyzed in order to assess potential contamination risks. Then generic reference levels are established in terms of two main criteria: guiding the assessment and controlling contaminated soils (viz. land use and human health hazards). This provides us with crucial means to investigate and manage them. These levels are determined using models shown in the US EPA Supplemental Guidance for Developing Soil Screening Levels for Superfund Sites, March 2001, which were constructed with data from the Agency for Toxic Substances and Disease Registry (ATSDR) and EPA. At least two reference values for different scenarios (viz. industrial use and miscellaneous uses) are established for each individual element. The exposure pathways considered include soil ingestion, dermal contact, ingestion plus dermal contact, and particle inhalation. As a rule, the GRL for an element is its lowest concentration having an impact through the most sensitive pathway(s).
4. Problems in Agricultural Soils Affected by Sulfide Mining
Soil contamination by trace elements and other trace elements is a matter of great concern for governments. Characterizing, assessing and reclaiming a soil, especially aiming to change its use (e.g., from mining to farming) is in fact one of the current greatest environmental challenges. All available methods for assessing soil contamination, and the criteria based on regional geochemical background levels and GRLs––even risk analyses––are mere approximations to the actual situation.
Assessing soil quality in mining areas is a difficult task because the closing-down of mines without implementing any environmental recovery measures has left a number of contamination sources that are currently affecting rivers and soils in their vicinity [40-43]. Mining activities have produced and still generate a variety of gaseous, liquid and solid contaminants that eventually reach soil. Regarding the impact of mining on soil through the occupation of previously fertile soil, the establishment of dumps, processing plants, etc., and the alterations caused by mining activities (e.g., deposition of sediment, rainborne or acid-leached particles), have grown steadily over the last decade. Mining activities can cause the complete loss of soil fertility. Moreover the contaminants present in soil mean a potential threat for plants, animals and humans since they can enter food chains and be accumulated in animal and/or plant tissues. Thus, trace elements absorbed by roots can easily be stored there or rapidly move to the aerial parts of the plant where they may remain accumulated [44]. Plants growing in contaminated soil can contain high concentrations of trace elements, not only in leaves, but also in roots [44-46], and occasionally become serious hazards for human health [47].
A number of studies on soils affected by mining activities have revealed a strong impact on soil worldwide. In Asian countries, where mining resources were intensively exploited during the first half of the 20th century, soils and waters in the vicinity of mines were highly contaminated [47-54]. Thus, some tested samples of soil found in SW Korea contained Pb and As concentrations up to 90 times higher than the internationally accepted maximum levels for agricultural use; based on bioavailability data, regular ingestion by farmers through inhalation and ingestion from dirty hands could pose a long-term health risk to the local population [55,56].
Countries such as USA, Australia and Canada [45,57] have large areas contaminated with mercury and sites affected by heavy acid drainage from abandoned mines. Other American countries with an important long mining tradition have assessed soil contamination in the vicinity of mines such as those of San Luis de Potosí in Mexico [58] or Santa Barbara [59]. Contamination by trace elements from Minas Gerais (Brazil) has been found to seriously affect the local flora and fauna within a range of 400 km [60].
In Europe, acid drainage from abandoned mines in the Swedish region of Dalarma has been found to result in serious contamination of the soil with Cu, Fe, Ni, Pb and Zn, as well as in strong acidification [61]. In the north of Somerset (England), soils around former metal mining zones are highly contaminated with Pb, Zn, Cu and Cd [62]. In the Polish region of High Silesia, soils are heavily contaminated with Cd, Zn and Pb [63,64]. Germany and Hungary have also reported soil contamination in various mining zones [65,66]. Finally, Portugal has conducted several studies on the impact of abandoned copper mines on water, soil and plants [67,68].
In Spain, a number of studies have assessed soil quality at previous mining sites in places such as Cartagena and Murcia, where soil near Pb and Zn mines was found to contain high levels of all metals studied [69-71]. In the historic mining district of Almadén, a number of studies on soil and plant contamination with mercury have revealed extremely high concentrations of this metal (up to 0.13 ppm) in relation to its reference levels for soil, which typically fall in the range of 0.01–0.03 ppm [72]. Heavy mining in the Sierra Almagrera-Las Herrerías district led to the production of large amounts of waste (dumps, flotation sludge, slag) that was indiscriminately disposed of and led to the presence of high concentrations of Pb (5,428 ppm), As (265 ppm), Zn (2,200 ppm), Ba (4.9%) and Ag (27.5 ppm), among other metals, in soil. Soils on alluvial materials in the vicinity of the waste dumping sites contained large amounts of Pb, As and Ba, and also of Zn and Cd at some sites, in all horizons [73]. Mining waste accumulated on soils in the Linares–La Carolina district contains high concentrations of Ag, Pb, As, Sb and Cd, with Pb levels up to 37,000 ppm near dumps. They can also be found in the exchangeable fraction, which poses a high environmental risk [74,75].
5. Soil Contamination by Mining Activities in the Iberian Pyrite Belt (SW Spain)
5.1. Study Area Description and Aim
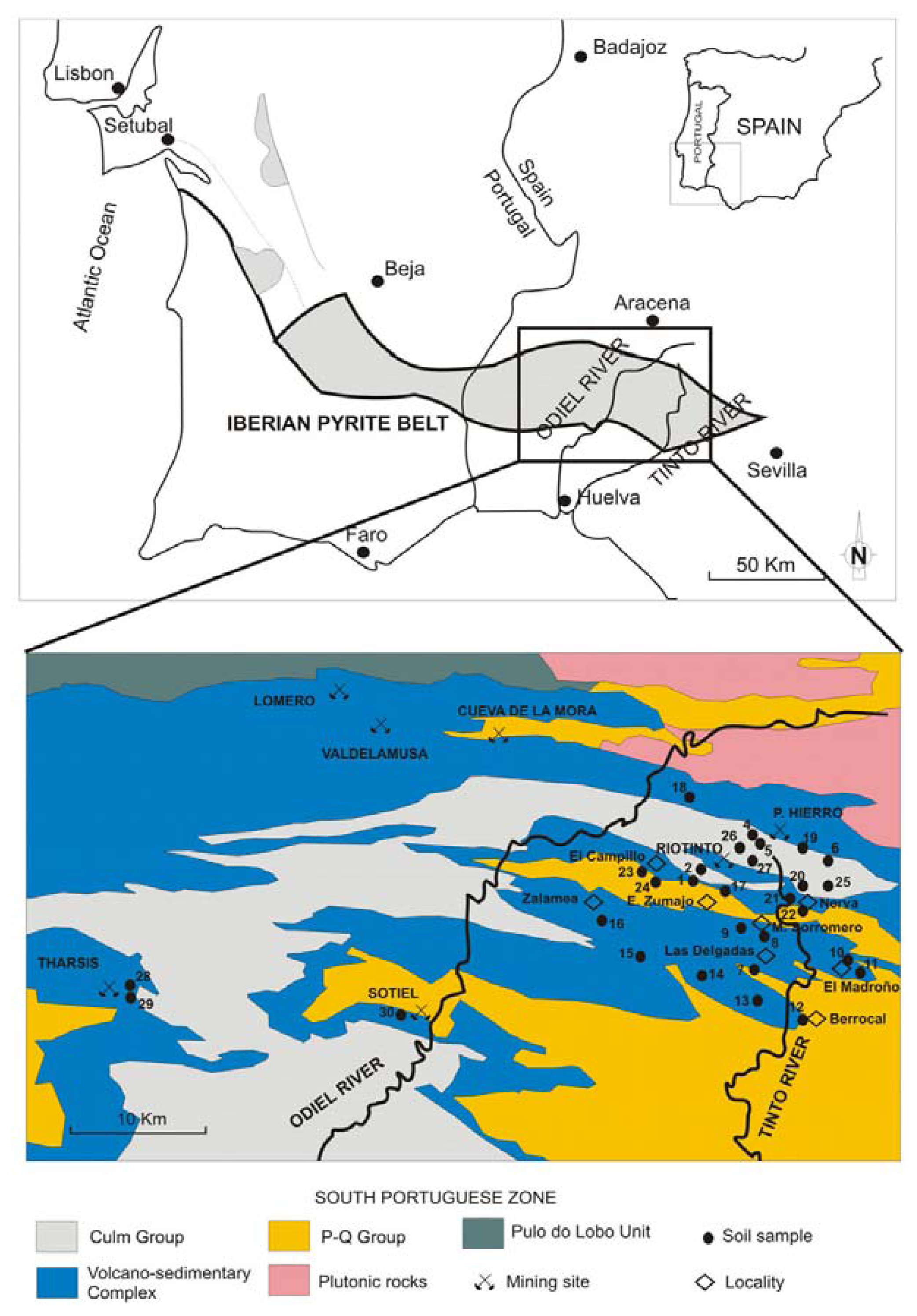
A retrospective view of the relationships between mining and the environment in the Southwest of the Iberian Peninsula (Figure 1) reveals an exemplary lesson on how to avoid potential geoenvironmental management errors in the future. Without a doubt, mining in the Iberian Pyrite Belt did contribute to the industrial development of some European countries producing a large amount of sulfur and the base metals required. Also, it has been an invaluable source for foreign currency, which did not help at all to power the socioeconomic progress in this mining region. Indeed, indiscriminate ore deposits mining, has deeply degraded sites and has produced deep scars in the landscape. It has also affected rivers and soils in such a way that now they are some of the most heavily contaminated in the world.
Social awakening regarding environmental awareness has promoted the development of research projects intended to accurately assess the current situation and facilitate the adoption of plans to recover the physical environment. This strategy seeks to reach an acceptable level for human health and ecosystems to take place while preserving a millennial mining and metallurgical tradition.
A number of studies about the current status of almost abandoned mine soils in the Pyrite Belt [76-84] have revealed the presence of high concentrations of trace metals. Thus, Chopin and Alloway [81] found high levels of trace elements within a range of 2 km around mines and foundries in Tharsis and Riotinto. Also, Barba et al. [82], López et al. [80] and González et al. [84] examined soils in Riotinto and realized they contained high concentrations of Pb, Cu, Zn and As largely exceeding the background levels for soils in the Sub-Portuguese zone––the geological setting for the Pyrite Belt––reported by Galán et al. [85].
The abandonment of mines in the Pyrite Belt during the second half of the 20th century has additionally had an adverse socioeconomic impact (e.g., a considerable loss of employment) which has been alleviated by shifting agriculture to be the new ground for social development. In fact, previous mine soils are now intensively cropped with citrus and other fruits. Also, nearby alluvial fertile plains have provided self-subsistence based on traditional farming on a small scale. At no time, however, the contamination of these soils and its potential impact on the quality of the products and population health has been considered. Therefore, it is crucial to assess the soil condition in these areas that were transformed from almost abandoned mines to cultivation fields, and to identify the contaminants they contain and their sources. If these areas are contaminated, a remediation strategy will be developed according to current regulations.
The aim of this research is to assess soil quality in the Riotinto mining area determining the total content of trace elements, their chemical and mineralogical speciation, and the mobility and bioavailability of potentially toxic elements in agricultural soils in the area on the basis of the methodology and recommendations described in Section 2. These data will show the actual degree of soil contamination to be evaluated with a view to assessing the hazards of their agricultural use.
5.2. Material and Methods
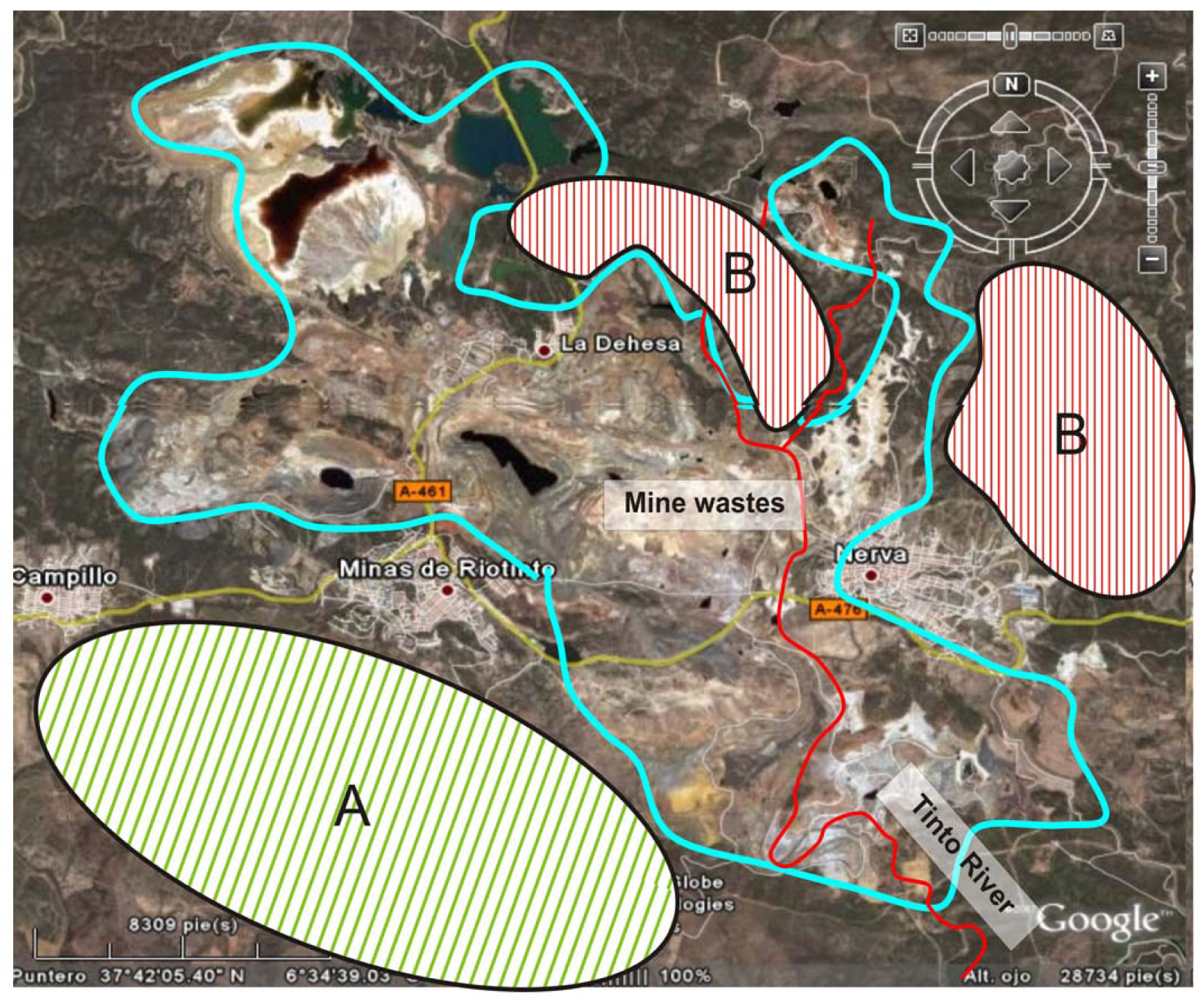
A total of 30 samples were collected from the topmost 20 cm of agricultural soils. These samples were obtained from sites nearby the mines, or from areas potentially affected by miming activities (Figure 1).
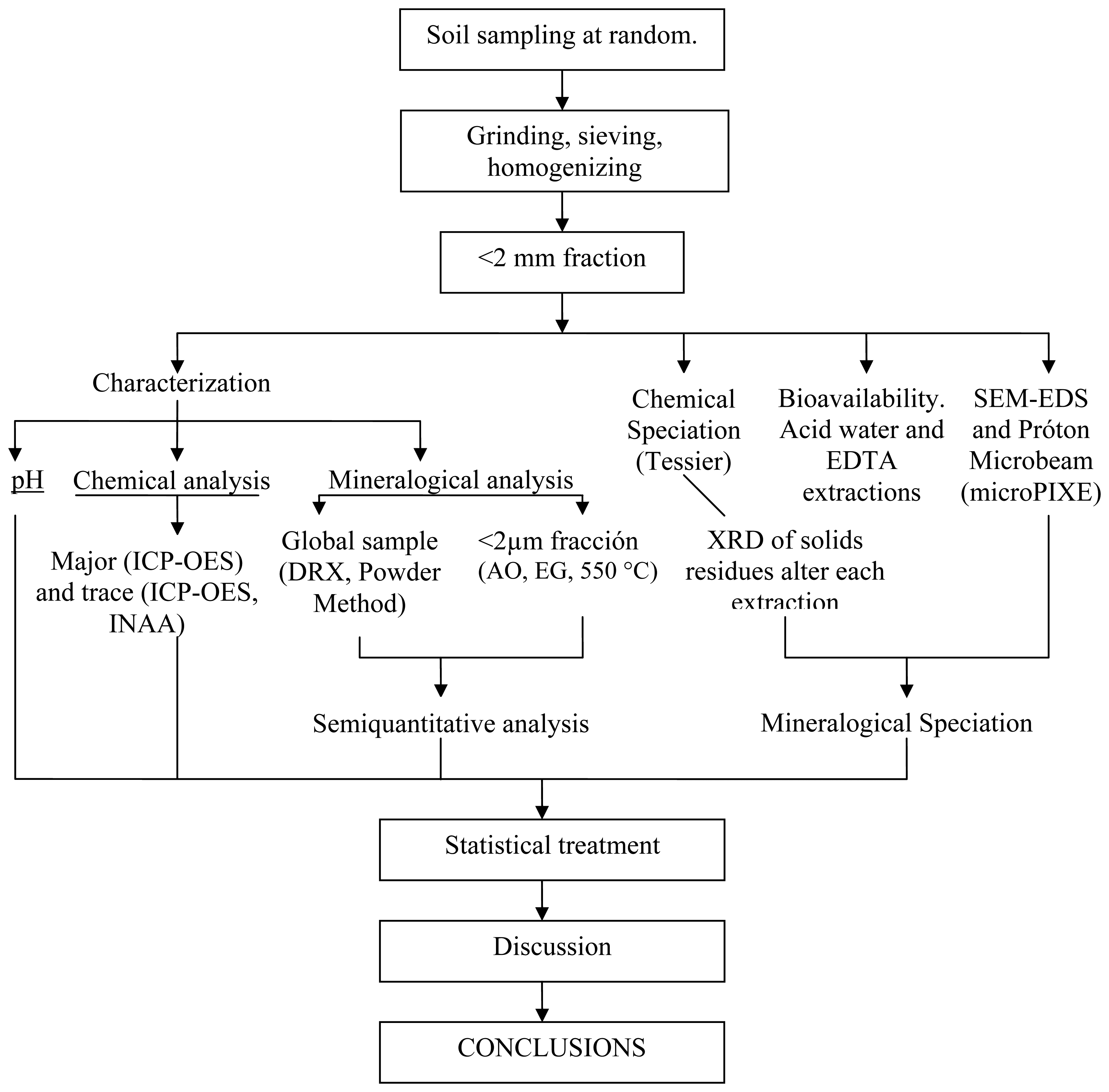
The following soil parameters were determined: pH, texture and clay mineralogy. Organic matter and carbonates were not determined since most of the soils were leptosols on shale and volcanic rocks in the Sub-Portuguese Zone (Iberian Massif, Spain). All tests were conducted on the <2 mm fraction as Figure 2 shows.
pH was measured with a Crisson microph 2002 pH-meter previously calibrated with two buffer solutions at pH 4 and 7. Measurements were made in suspensions obtained mixing 10 g of soil sample with 25 mL of distilled water under mechanical stirring for 10 min. The mineral composition of the soils was determined by X-ray diffraction (XRD) under standard conditions. Whole samples were analyzed by using the powder method on side-loaded sample holders. Clay minerals were determined in the <2 μm fraction, using the oriented aggregate (OA) method with ethylene-glycol and heating at 550 °C treatments.
Major elements were determined by inductively coupled plasma optical emission spectrometry (ICP-OES) and minor elements by both ICP-OES and instrumental neutron activation analysis (INAA). Chemical speciation was done in 11 samples of agricultural soils, following the scheme originally proposed by Tessier et al. [17] as subsequently modified by Galán et al. [21]. The trace elements extracted by each solvent were determined spectrophotometrically. Mineralogical speciation was performed on 7 samples selected in terms of their trace metals content, iron oxides, and phyllosilicates. That was done using XRD to identify the mineral phases present in the solid residues left by the different extractants in Tessier's protocol. For subsequent characterization, scanning electron microscopy (SEM-EDS) and microparticle induced X-ray emission (microPIXE) spectroscopy were also used.
Bioavailability was assessed extracting the samples with a 0.05 M solution of EDTA in sodium form at pH 7 under continuous stirring for 1 h. Acid-extractable metals were obtained treating the samples with water acidified with HNO3 to pH 1 under continuous stirring for 1 h. Both extracts were spectrophotometrically analyzed.
In order to confirm some relationships between elements with a similar geochemical behaviour in the soils, the results were subjected to correlation analysis and principal component analysis. Moreover, in order to facilitate spatial analysis of the data and mapping potentially dangerous zones for agricultural use, each sample was related to itself and to all others by using a Geographic Information System (GIS). Specifically, alphanumeric values were introduced as input into a database and georeferenced to obtain geographic location mapping outputs and develop interactive maps to display specific data for the sample concerned. That was done using the software ArcGIS v. 9.2. All information collected was recorded on a reference system used by the Spanish Geographic Institute (IGN) and Spain's Army Mapping Service (SCE) for zone 30 in the 1950 European Datum (ED-50), which was made compulsory in our country by Decree 2303/1970.
5.3. Results and Discussion
5.3.1. Characterization of Agricultural Soils
The soil samples studied were found to contain high proportions of gravel occasionally exceeding 50%. Therefore the soils were classified as sandy gravel. Because of their sandy nature, these soils have a low ability to absorb trace elements. In fact only those in Nerva zones, which were slightly more clayey, were able to retain greater amounts of the elements studied. Soil pH ranged from 4.6 to 7.6, the most acid samples being those collected very near the Peña del Hierro mine, Monte Sorromero and Tharsis (with pH < 5.5). These results are similar to those for contaminated soils in Mexico [58] and Mendips, England [86]. pH is a very important parameter since the concentration of hydrogen ions in soil influences the bioavailability of trace elements and nutrients. Also, it can be understood as a measure of soil deficiencies or alteration [87,88]. Most of the metals studied tend to be more readily available at an acid pH. As a rule, the contents in divalent metals such as Cu, Zn, Ni, Co, Cd and Mn decreased when increasing the pH [67,89].
The minerals found in the samples consisted mainly of quartz, phyllosilicates and feldspars. The latter are calcium plagioclases which explains the high Ca contents of some samples despite the absence of carbonates. There were also substantial amounts of hematite and goethite in some samples from Tharsis, Riotinto and Las Delgadas. The high content in iron oxides of some samples (5–13% Fe2O3)––particularly the ones form near the Tharsis and Sotiel mines, and from some sites in Riotinto––where no hematite or goehtite was detected, suggest the presence of iron gels. This is an interesting finding since the high specific surface area and chemical reactivity of these materials play a major role in the sorption of trace elements (particularly metals such as Cu, Pb, and As) [90].
The <2 μm fraction consisted of illite, kaolinite and chlorite as major minerals, in addition to smectite in some samples from zones with altered volcanic rocks (Berrocal, Marigenta, Zalamea). Some leptosols (soils with shales as parent rocks) were found to contain chlorite-vermiculite mixed layers (Peña del Hierro, Riotinto) and illite-smectite mixed layers (Las Delgadas). Unlike other contaminated soils [79], no jarosite was detected in any sample since their pH, above 3, precluded its formation.
The soils were poor in alkaline and alkaline-earth metals. Their contents in CaO, K2O, MgO and Na2O never exceeding 5%. These results are consistent with those reported by Chopin et al. [76] for Tharsis soils, and those reported by Fernández Caliani et al. [77], Barba et al. [83] and López et al. [80] for other similarly contaminated soils in the Iberian Pyrite Belt.
The spatial distribution of major elements in soils explains the existence of a zone with an increased abundance of iron and manganese associated to volcanic rocks (Figure 3), which is identified in the south of the study area [91].
The trace elements studied included As, Cd, Co, Ni, Cu, Pb and Zn, which are especially abundant in soils affected by sulphide mining [4]. All of them can be found in the US EPA priority pollutant list. The contents in As ranged from 19 to 994 ppm and peaked in the samples extracted from Tharsis (994 ppm) and Sotiel (882 ppm), which clearly exceeded the average value (227 ppm). This is logical if we consider these samples were under the direct influence of dumps and were thus mine soils. The contents in Pb ranged from 40 to 4,900 ppm. The highest values belong to the samples extracted from Tharsis (2,650 ppm) and Sotiel (4,890 ppm). Those of Zn ranged from 95 to 900 ppm and those of Cu from 27 to 1,160 ppm; the latter, however, were high in most samples and clearly exceeded the regional background level for this element [85]. The Co, Cr and Ni content, which were not too high, exceeded their respective regional background levels of Las Delgadas, Marigenta and Zalamea zones. The anomalous high levels they show were possibly geogenic and associated to the parent rock [84]. Except for Cd, all the elements exceeded the levels for the 95 percentile in Andalusian soils and the 90 percentile for the geological domain of the Sub-Portuguese Zone [85].
In the agriculture soils, which were not directly under the influence of acid mining drainage, the trace elements contents exceeded the critical ecotoxic concentration established by Kabata-Pendias [10], except for Cd and Zn (Table 7). Also, the mean values for all elements were similar to those previously found in Tharsis (Iberian Pyrite Belt) by Chopin and Alloway [79].
The factor analysis between trace elements suggests an association between Cu, Pb, Zn and As on the one hand, and Cr, Ni, Co and Fe on the other (Table 8). This is a logical result because of the presence of massive sulfides––and also, occasionally, veins of these elements––associated to the volcano sedimentary complex in the Sub-Portuguese Zone [91]. The principal component analysis confirmed these two associations, which were related to the potential origin of the elements. Thus, Cr, Ni and Co were essentially geogenic, whereas Pb, Zn, As and Cu were markedly anthropogenic. It should be noted that, unlike less contaminated soils [92-94], none of the trace elements were related to the phyllosilicate content found in the soil tested.
5.3.2. Chemical Speciation
Overall, the sum of trace element contents in each sequential extraction was comparable to those in the total sample according to the INAA and the ICP-OES (Table 9). Although some samples exhibited very different values, they would be accepted if the combined metal contents of their different fractions were within a range of ± 10% in relation to those in the total sample [95,96].
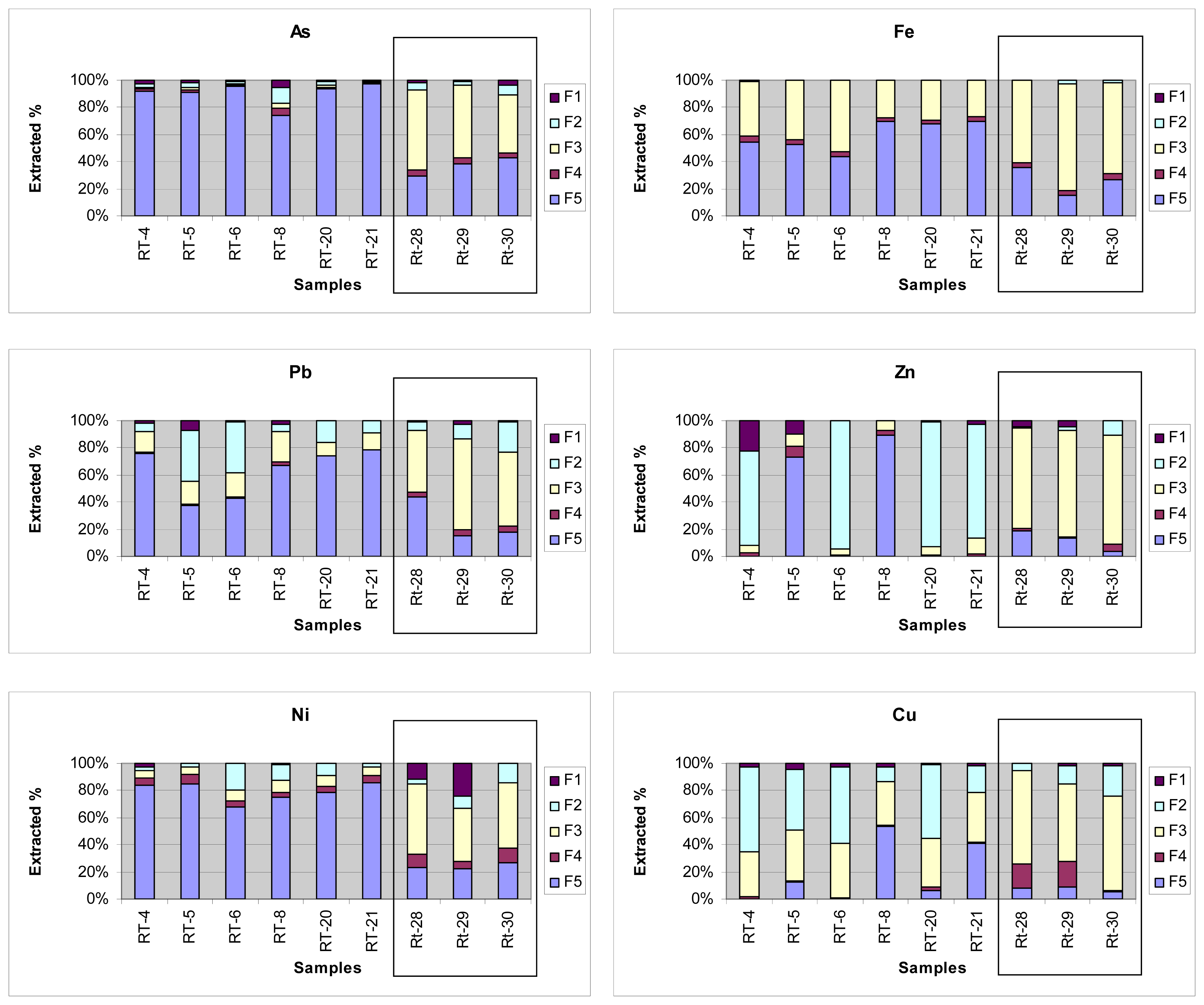
It should be noted that only Cd was present in high proportions in the exchangeable fraction. However its presence was not hazardous because of their low total content. All other trace elements were largely associated to iron oxyhydroxides and the residual fraction. These results confirm the presence of amorphous oxyhydroxides capable of incorporating large amounts of metal cations [24].
Arsenic and Ni were associated to the iron and manganese oxides fraction in the samples directly affected by mining wastes and to the residual fraction in all others. Hence it was scarcely present in the most mobile fractions (Figure 4). Copper was present in all fractions, but largely associated to labile fractions and iron and manganese oxides virtually in all samples. Lead and zinc were associated to iron and manganese oxides and in the insoluble residue, but also to labile fractions. The fact that Zn, Pb and Cu were partially associated to the exchangeable fraction that consisted of phyllosilicates and carbonates, suggests that their presence was a result of ion exchange in clay minerals or precipitation as oxides on their surface.
Similar results were previously reported for soils under the influence of mining waste in China and Korea [52,97], and the Pb-Zn mine in San Quintín (Spain) [94]. In conclusion, most of the elements studied were adsorbed in (or as structural component of) iron oxyhydroxides, or present in the residual fraction. Therefore, the present physical–chemical conditions of the soils facilitate the immobilization of the toxic elements studied, but their current levels pose a potential hazard that warrants assessment.
5.3.3. Availability and Bioavailability Tests
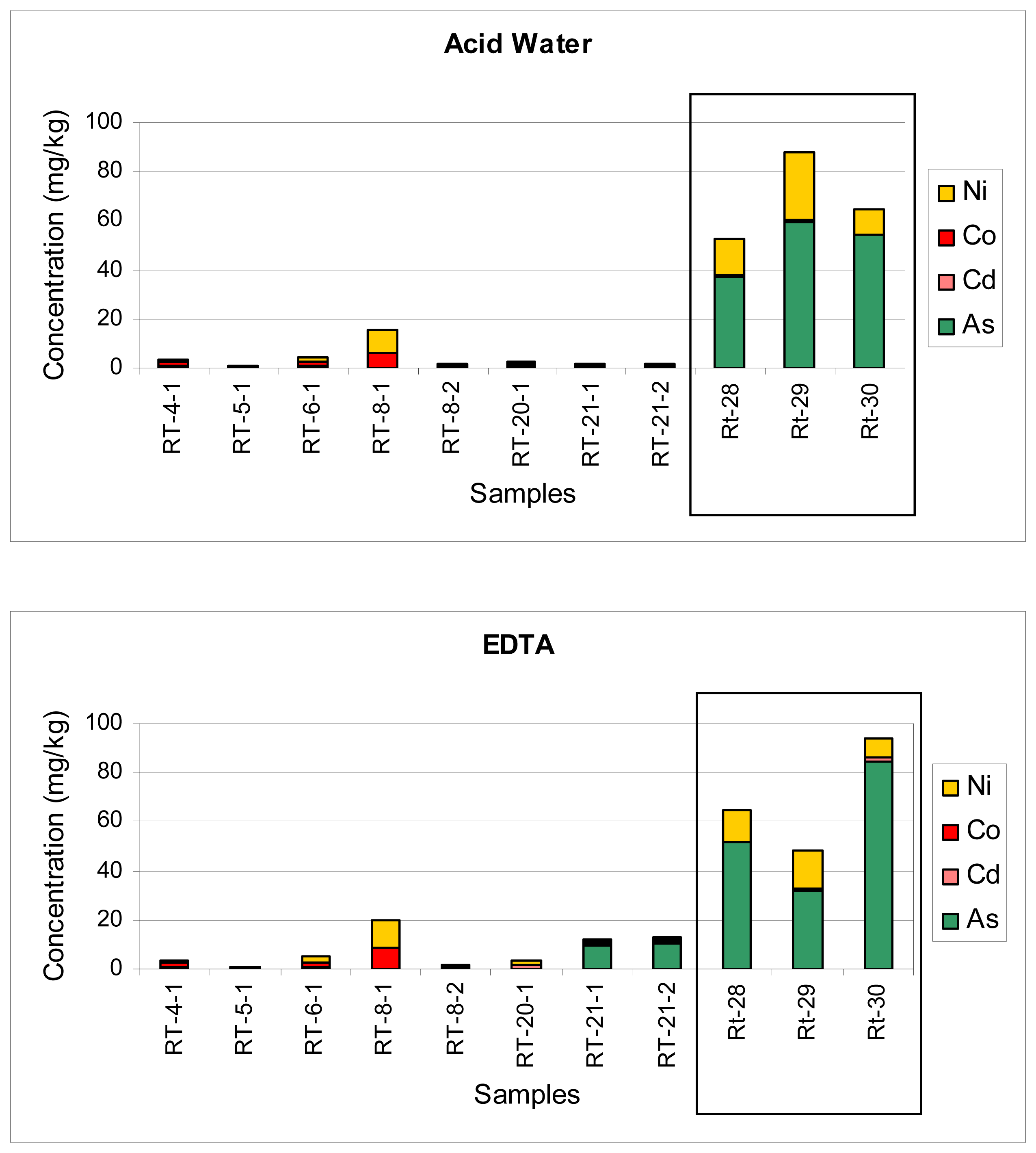
The fraction extracted with acid water contained all elements studied, some at virtually negligible concentrations and others, including Fe, Mn, Zn, Cu and Pb, at very high levels (close to the 40% in some samples). As it can be noticed in Figure 5, As and Ni exhibited a high availability in all samples under the direct influence of a mine dump, maybe because both elements were associated to iron oxides.
The fraction extracted with EDTA also contained all elements. As Figure 5 shows, As was highly available, especially in soils near the mines in Tharsis (Rt28-29) and Sotiel (Rt-30), which contained high levels of trace elements. Similar results were previously obtained in agricultural soils and rice fields in Korea, where As, Cu and Zn were highly bioavailable and posed serious health hazards [47].
5.3.4. Mineralogical Speciation
The XRD results obtained from the different fractions did not allow us to assign the different trace elements to specific mineral phases because the fractions were very similar in mineralogical terms, and the detection of minor mineral phases was not possible with this method.
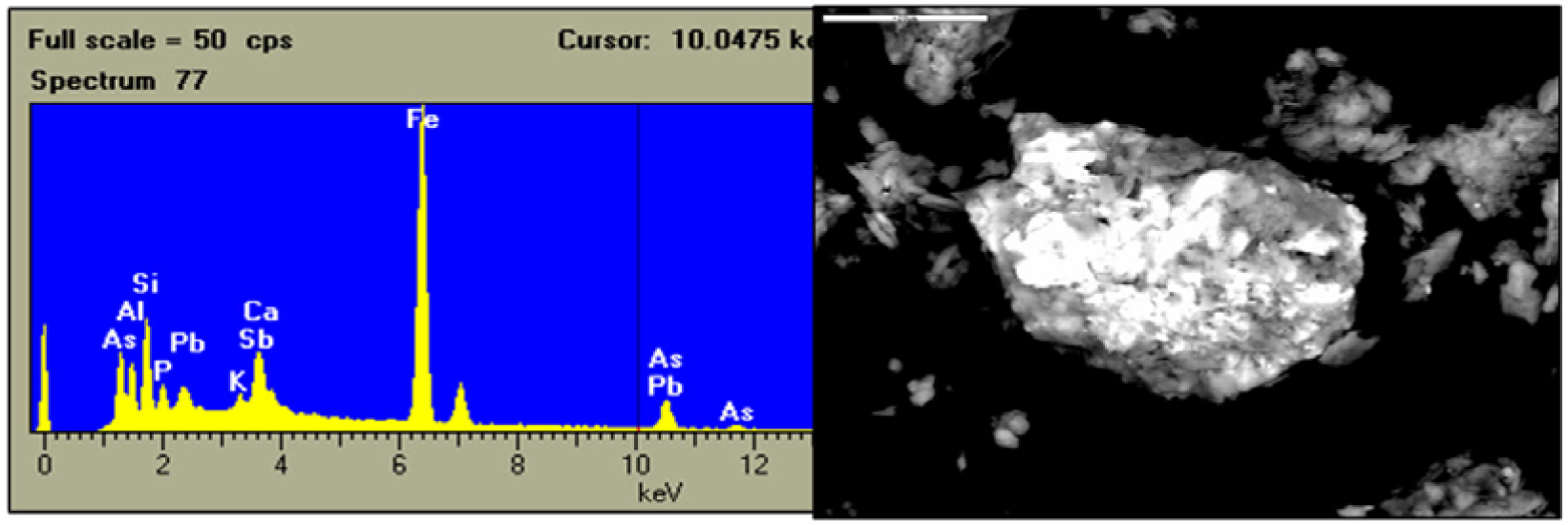
The SEM study confirmed the presence of the major minerals detected by XRD (quartz, phyllosilicates, feldspars and iron oxides) in addition to accessory minerals such as pyrite, barite, zircon, rutile, monazite and ilmenite (Table 10). No association of any trace element to clay minerals was detected though. Micrographs revealed the presence of iron oxide aggregates containing substantial concentrations of Pb and smaller amounts of As and Sb (Figure 6). There were also isolated pyrite crystals transformed into iron oxides (hematite) associated to no trace element. Some samples additionally contained galena.
Selected samples were subjected to a microproton probe (PIXE) study in their <63 μm fraction. The results were used to produce distribution maps for major and trace elements. Some of these elements such as Al, were uniformly distributed across the study area (Figure 7), whereas others (Si, Mn, As, Cu, Pb and Zn) appeared accumulated in small granules. The results were expressed in quantitative form using the data acquisition software OMDAQ to transform the study area into a matrix of 256 × 256 elements containing a PIXE spectrum each. That provided the overall PIXE spectrum for the area and the individual spectra for each zone of potential interest. This allowed us to identify numerical results for a zone of special accumulation of a given element to be obtained and whether the element concerned accumulated in any particular mineral phase to be confirmed (Tables 11 and 12).
According to the PIXE results As, Pb and Zn are associated to amorphous iron gels in some samples. That fact is significant since it had never been previously demonstrated using the techniques described above. In other samples Cu, Pb and Zn were associated to Fe and Mn oxides. Moreover, Cu-S and Pb-S associations were due to chalcopyrite and galena, respectively. These results are similar to those for Pb, Zn and As obtained by [98] through the synchrotron technique. In the present study, As was not associated to jarosite since their pH invariably exceeded 3 and was therefore unsuitable for its formation. Lead can be adsorbed by iron and manganese oxides, or on goethite particles, as well as lead oxides. Zn can be found as franklinite (ZnFe2O4) and willemite (Zn2SiO4) [99], or associated to manganese oxides (birnesite) and iron oxides (feroxyhyte).
5.3.5. Magnitude and Distribution of Trace Elements in the Study Area
Soil enrichment with trace elements, and hence anomalous element contents, are usually assessed thanks to the enrichment factor (EF) for the top soil level [80]. EF is defined as the ratio of the total concentration of an element to its regional geochemical level (background or baseline) in the geological domain concerned, which was the Sub-Portuguese Zone in our case [85]. An EF value = 1 denotes a natural origin, whereas a greater value shows enrichment of the soil with the element concerned via geogenic or anthropogenic ways.
The samples from both Tharsis and Sotiel mining zones, which were under the direct influence of acid drainage, were those exhibiting the greatest enrichment for As, Cu, Pb and Zn (Table 13). That particularly happened in Sotiel, where the Pb and As levels were nearly 200 and 50 times higher than their respective regional background levels. All other trace elements were found at lower concentrations. As an exception, some samples from Las Delgadas had moderate EF values for Cr, Cu, As and Pb.
The slightly increased levels of Cr, Ni and Co were due to natural sources: the soils developed on volcanic rocks with high content in these trace elements [80]. Overall, EF decreased with the increasing of the distance from mine dumps, as previously was also observed in Korean mining areas [51,97].
The EF values for Pb, As, Cr, Zn and Cd in the soils we analyzed were higher than those in Korea [97,100] and SE Spanish soils [34], but similar to those in soils from the Iberian Pyrite Belt [76-80].
The results permitted the spatial distribution of trace elements in the studied soils and, together with the mobility and bioavailability values, to assess the potential risks of using the soils in the Riotinto region for agricultural purposes (Figure 8). It also helped us to identify the potentially hazardous zones for the intensive growth of citrus since it showed the high contents in Cu, Pb, Zn and As in the NE. Thanks to this analysis we also identified other low or medium risk areas where we found a relatively high content in Cr, Ni and Co levels in the SW but low bioavailability. However, we could not develop more detailed information for small orchards, showing the risk and potential hazardous areas, since we used a fairly large scale.
Nevertheless, for those cases, we can provide with an interactive media (an html file exported from files in Flash format), that enables to show the content in major and minor elements as georeferenced data to be displayed with a view to assessing the cropability of the soil concerned.
6. Final Considerations
This work was intended to provide a general picture of soil contamination by trace elements and the best procedures for assessing soil quality in the vicinity of mining areas. Although analytical techniques based on ion beams for chemical and mineralogical speciation proved to be effective in addressing previously unsolved problems, the use of other classical characterization techniques facilitates speciation.
Based on the results of this study, the Iberian Pyrite Belt (SW Spain) has evolved in the opposite direction of modern societies (i.e., from an industrialized region to one where farming has become essential for the local economy). Specifically, there is an ongoing project that intends to recover the mining zones in the Belt establishing large-scale farming for citrus fruits over 3,000 ha of leptosols [101] in addition to small family orchards where the locals grow various types of products.
However the change in the use of the soil was preceded by no study of potential contamination in the area. That makes the present study specially interesting since plant species growing on contaminated soils are known precisely because they accumulate high concentrations of trace elements in leafs and branches [44-46,102,103]. Excessive accumulation of some elements in plant tissues might have toxic effects and produce an adverse impact on plant growth and production, or even affect the local population in the medium-to-long term.
The soils in the study area contain anomalous levels of some trace elements (As, Cu, Pb and Zn) and should not be used for farming. The results suggest widespread contamination, especially in small private orchards in the Tharsis and Sotiel zones.
The likelihood of trace elements sorption in soils is low by virtue of their sandy texture, low proportions of phyllosilicates and absence of carbonates. The fact that the majority of trace elements are found in the residual fraction, or are associated to iron and manganese oxides, results in a low mobility of the toxic elements under the current physical-chemical condition of the soils. However, the relatively high bioavailability of some elements such as As, Cu, Ni, Pb and Zn may pose potential risks for safe farming.


| Trace elements that are essential micronutrients (a few mg or μg/day) | As, Co, Cr, Cu, Fe, Mn, Mo, Se, V, Zn |
| Other essential micronutrients | F, I, Si |
| Macronutrients (100 mg or more per day) | Ca, Cl, Mg, P, K, Na, S |
| Non-essential trace elements * | Be, Cd, Hg, (Ni), Pb, Sb, (Sn), Ti |
*Metals in brackets can be essential.
| Element | Normal range (mg/kg) | Anomalous concentrations (mg/kg) |
|---|---|---|
| As | <5–40 | Up to 2,500 |
| Cd | <1–2 | Up to 30 |
| Cu | 2–60 | Up to 2,000 |
| Mo | <1–5 | 10–100 |
| Ni | 2–100 | Up to 8,000 |
| Pb | 10–150 | 10000 or more |
| Se | <1–2 | Up to 500 |
| Zn | 25–200 | 10,000 or more |
| Metal species and association | Availability-mobility |
|---|---|
| Simple or complex cations in solution phase | Easy |
| Exchangeable cations | Medium |
| Chelated cations | Slight |
| Metals adsorbed by soil particles | Slight |
| Metal compounds precipitated on soil particles | Available after dissolution |
| Metal bound or fixed inside organic substances | Available after decomposition |
| Metal bound or fixed inside mineral particles | Available after weathering and/or decomposition |
| Fraction | Procedure |
|---|---|
| Acid (F1) | NH4Ac 1 M (35 mL), pH 5, 1 h, 20 °C, continuous shaking (40 rpm) |
| Reducing (F2) | NH2OH.HCl 0.4 M in acetic acid 25% (20 mL), 6 h, 96 °C, manual shaking every 30 min. |
| Oxidant (F3) | HNO3 0.2 M (3 mL) + H2O2 30% (5 mL), pH 2 with HNO3, 2 h, 85 °C, manual shaking every 30 min.; add H2O2 30% (3mL), pH 2 with HNO3, 3 h, 85 °C, NH4OAc 3.2 M (5mL) v/v in HNO3 diluted in 20 ml with H2O2, 30 min., 20 °C, continuous shaking (40 rpm) |
| Residue (F4) | HF/HNO3/HCl 10/3/2.5 (20 mL), 2 h to dry |
| Metal | Austria | Canada | Poland | Japan | Great Britain a | Germany b |
|---|---|---|---|---|---|---|
| As | 50 | 25 | 30 | 15 | 20 | 40 (50) |
| Be | 10 | 10 | 10 (20) | |||
| Cd | 5 | 8 | 3 | 1 (3) | 2 (5) | |
| Co | 50 | 25 | 50 | 50 | ||
| Cr | 100 | 75 | 100 | 50 | 200 (500) | |
| Cu | 100 | 100 | 100 | 150 | 50 (100) | 50 (200) |
| Hg | 5 | 0.3 | 5 | 2 | 10 (50) | |
| Mo | 10 | 2 | 10 | |||
| Ni | 100 | 100 | 100 | 100 | 30 (50) | 100 (200) |
| Pb | 100 | 200 | 100 | 400 | 50 (100) | 500 (1,000) |
| Zn | 300 | 400 | 300 | 250 | 150 (300) | 300(600) |
aValues proposed for MAC in the EU for soils treated with residual lodos. Values in brackets are maximum concentrations given by Finnecy and Pearce [35];bAcceptable and toxic contents (in brackets), given by Kloke and Einkmann [36].
| Element | Agricultural soils | PN | AI | |||||||
|---|---|---|---|---|---|---|---|---|---|---|
| 1 | 21 | 22 | 3 | |||||||
| <7 | >7 | <7 | >7 | <7 | >7 | <7 | >7 | |||
| As | <20 | 20–30 | 30–50 | >50 | >100 | >300 | ||||
| Cd | <2 | <3 | 2–3 | 3–5 | 3–7 | 5–10 | >7 | >10 | >15 | >30 |
| Co | <20 | <50 | 20–50 | 50–150 | 50–100 | 150–300 | >100 | >300 | >300 | >400 |
| Cr | <100 | 100–250 | 250–450 | >450 | >500 | >1,000 | ||||
| Cu | <50 | <100 | 50–150 | 100–300 | 150–300 | 300–500 | >300 | >500 | >500 | >1,000 |
| Hg | <1 | 1–2 | 2–10 | >10 | >15 | >30 | ||||
| Mo | <10 | 10–40 | 40–200 | >200 | >200 | >500 | ||||
| Ni | <40 | <50 | 40–80 | 50–100 | 80–200 | 100–300 | >200 | >300 | >500 | >750 |
| Pb | <100 | <200 | 100–250 | 200–400 | 250–350 | 400–500 | >350 | >500 | >1,000 | >2,000 |
| Se | <2 | 2–5 | 5–10 | >10 | >20 | >30 | ||||
| Sn | <20 | 20–50 | 50–150 | >150 | >300 | >500 | ||||
| Tl | <1 | 1–3 | 3–5 | >5 | >10 | >30 | ||||
| Zn | <200 | <300 | 200–300 | 300–500 | 300–600 | 500–1,000 | >600 | >1,000 | >1,000 | >3,000 |
1: Maximum allowable values; 21: Investigation recommended; 22: Mandatory investigation; 3: Mandatory treatment; PN: Intervention value for natural parks and forest; AI = Intervention value for industrial areas; <7 = pH 5–7; >7 = pH > 7.
| Soils affected by mining exploitation | As | Cd | Cu | Pb | Zn | Cr |
|---|---|---|---|---|---|---|
| South Korea [56] | 23.3–75.9 (47.7) | 2.6–6.1 (4.3) | 40–99 (59) | 289–948 (477) | 275–568 (384) | ----- |
| Korea [47] | 64.8–131 (99.6) | ----- | 49.6–50.3 (50) | 2–11.3 (6.7) | 153–322 (237) | ----- |
| China [48] | ---- | 4–24 (8) | 11–100 (15) | 11–380 (15) | 33–1,600 (52) | 48 |
| Somerset, England [86] | ----- | 0.5–127 (2.1) | 2.8–145 (12) | 8–1,000 (52) | 14–8,344 (132) | |
| Mexico [58] | 7–1738 (569) | ----- | ----- | 30–3,450 (312) | ----- | ----- |
| “La Unión”, Spain [69] | ----- | 2–322 (41) | 36–2,255 (332) | 0–19,000 (8) | 1,200–53,100 (12.5) | |
| “San Quintín”, Spain [94] | ---- | (3.76) | (57.01) | (393) | (596) | ---- |
| “Tharsis” Spain [79] | 2–1519 (94) | ---- | 20–3,090 (216) 18–6,350 (172) | 45–870 (118) | ------ | |
| “Riotinto” Spain [80] | 12–204 (68) | 0.3–3.2 (0.53) | 47–586 (181) | 34–605 (177) | 72–795 (197) | 48–776 (149) |
| World normal values [4] | 6 | 0.35 | 70 | 30 | 35 | ------ |
| Allowable values [10] | 20 | 3 | 100 | 100 | 300 | 54 |
| Element | Factor 1 | Factor 2 |
|---|---|---|
| Cu | 0.840748 | −0.09327 |
| Cd | 0.647392 | −0.12501 |
| Pb | 0.916278 | 0.067878 |
| Ni | −0.16902 | 0.870814 |
| Zn | 0.882436 | 0.078646 |
| As | 0.910778 | 0.081364 |
| Co | 0.130555 | 0.892078 |
| Cr | −0.26457 | 0.883246 |
| Fe | 0.34058 | 0.843938 |
| SiO2 | −0.15414 | −0.72585 |
| Phillosilicates | -0.35041 | 0.568918 |
| Expl.Var | 3.951898 | 3.938739 |
| Prp.Totl | 0.359263 | 0.358067 |
| Fraction | Location | Sample | As | Fe | Pb | Zn | Cd | Cu | Co | Ni |
|---|---|---|---|---|---|---|---|---|---|---|
| Total sample | Peña Hierro | RT-4-1 | 77 | 37,400 | 145 | 186 | 1 | 212 | 24 | 38 |
| Peña Hierro | RT-5-1 | 54 | 47,500 | 66 | 91 | 0 | 97 | 11 | 39 | |
| Peña Hierro | RT-6-1 | 103 | 46,900 | 108 | 215 | 1 | 586 | 12 | 40 | |
| Las Delgadas | RT-8-1 | 18 | 10,900 | 39 | 127 | 0 | 80 | 88 | 424 | |
| Las Delgadas | RT-8-2 | 120 | 51,500 | 126 | 114 | 0 | 245 | 12 | 43 | |
| Nerva | RT-20-1 | 77 | 51,800 | 598 | 795 | 3 | 495 | 15 | 47 | |
| Nerva | RT-21-1 | 204 | 52,300 | 563 | 374 | 3 | 482 | 18 | 38 | |
| Nerva | RT-21-2 | 197 | 48,400 | 603 | 388 | 1 | 526 | 16 | 41 | |
| Tharsis | Rt-28 | 444 | 65,600 | 1,270 | 667 | 1 | 198 | 41 | 51 | |
| Tharsis | Rt-29 | 994 | 88,100 | 2,650 | 897 | 1 | 443 | 70 | 54 | |
| Sotiel | Rt-30 | 882 | 70,900 | 4,890 | 585 | 1 | 1160 | 19 | 48 | |
| Interchangeable | Peña Hierro | RT-4-1 | 2 | 0 | 2 | 109 | 0 | 6 | 0 | 1 |
| Peña Hierro | RT-5-1 | 1 | 1 | 5 | 9 | 0 | 4 | 0 | 0 | |
| Peña Hierro | RT-6-1 | 1 | 0 | 1 | 0 | 0 | 20 | 0 | 0 | |
| Las Delgadas | RT-8-1 | 1 | 0 | 1 | 0 | 0 | 2 | 0 | 5 | |
| Las Delgadas | RT-8-2 | 1 | 0 | 14 | 20 | 0 | 9 | 0 | 0 | |
| Nerva | RT-20-1 | 1 | 0 | 1 | 22 | 0 | 4 | 0 | 0 | |
| Nerva | RT-21-1 | 1 | 0 | 1 | 14 | 0 | 8 | 0 | 0 | |
| Nerva | RT-21-2 | 1 | 0 | 1 | 11 | 0 | 7 | 0 | 0 | |
| Tharsis | Rt-28 | 5 | 0 | 35 | 211 | 1 | 0 | N.D. | 7 | |
| Tharsis | Rt-29 | 8 | 0 | 114 | 337 | 1 | 10 | N.D. | 18 | |
| Sotiel | Rt-30 | 20 | 0 | 83 | 15 | 1 | 33 | N.D. | 0 | |
| Labil/carbonates | Peña Hierro | RT-4-1 | 2 | 180 | 10 | 336 | 0 | 132 | 4 | 1 |
| Peña Hierro | RT-5-1 | 2 | 126 | 25 | 0 | 0 | 43 | 0 | 1 | |
| Peña Hierro | RT-6-1 | 2 | 39 | 41 | 659 | 0 | 390 | 9 | 8 | |
| Las Delgadas | RT-8-1 | 2 | 248 | 2 | 0 | 0 | 9 | 6 | 47 | |
| Las Delgadas | RT-8-2 | 2 | 89 | 49 | 24 | 0 | 191 | 1 | 3 | |
| Nerva | RT-20-1 | 2 | 28 | 94 | 1713 | 2 | 268 | 2 | 4 | |
| Nerva | RT-21-1 | 2 | 27 | 48 | 428 | 0 | 96 | 2 | 1 | |
| Nerva | RT-21-2 | 2 | 25 | 54 | 404 | 0 | 98 | 0 | 1 | |
| Tharsis | Rt-28 | 20 | 180 | 162 | 26 | 1 | 17 | N.D. | 2 | |
| Tharsis | Rt-29 | 26 | 2,747 | 457 | 202 | 1 | 92 | N.D. | 7 | |
| Sotiel | Rt-30 | 42 | 1,371 | 1,574 | 342 | 1 | 455 | N.D. | 7 | |
| Fe-Mn Oxides | Peña Hierro | RT-4-1 | 1 | 15,174 | 22 | 24 | 2 | 72 | 5 | 2 |
| Peña Hierro | RT-5-1 | 1 | 20,484 | 11 | 8 | 3 | 36 | 1 | 2 | |
| Peña Hierro | RT-6-1 | 1 | 24,779 | 19 | 32 | 4 | 276 | 2 | 3 | |
| Las Delgadas | RT-8-1 | 1 | 30,348 | 9 | 9 | 5 | 26 | 17 | 39 | |
| Las Delgadas | RT-8-2 | 1 | 20,861 | 18 | 7 | 3 | 68 | 1 | 2 | |
| Nerva | RT-20-1 | 1 | 15,322 | 60 | 118 | 3 | 177 | 2 | 4 | |
| Nerva | RT-21-1 | 1 | 14,106 | 70 | 58 | 2 | 174 | 3 | 2 | |
| Nerva | RT-21-2 | 1 | 14,261 | 62 | 50 | 2 | 167 | 2 | 2 | |
| Tharsis | Rt-28 | 194 | 47,984 | 1,259 | 3,414 | 0 | 214 | N.D. | 31 | |
| Tharsis | Rt-29 | 513 | 70,834 | 2,781 | 5,713 | 1 | 390 | N.D. | 30 | |
| Sotiel | Rt-30 | 246 | 42,384 | 3,877 | 2,740 | 1 | 1,380 | N.D. | 23 | |
| Organic matter | Peña Hierro | RT-4-1 | 1 | 1,789 | 1 | 13 | 0 | 3 | 1 | 2 |
| Peña Hierro | RT-5-1 | 1 | 1,774 | 1 | 7 | 1 | 1 | 7 | 3 | |
| Peña Hierro | RT-6-1 | 1 | 1,384 | 1 | 6 | 0 | 5 | 0 | 2 | |
| Las Delgadas | RT-8-1 | 1 | 2,803 | 1 | 5 | 1 | 1 | 3 | 16 | |
| Las Delgadas | RT-8-2 | 1 | 1,137 | 1 | 4 | 0 | 2 | 0 | 2 | |
| Nerva | RT-20-1 | 1 | 1,306 | 2 | 14 | 0 | 13 | 0 | 2 | |
| Nerva | RT-21-1 | 1 | 1,730 | 3 | 9 | 0 | 6 | 0 | 2 | |
| Nerva | RT-21-2 | 1 | 1,487 | 3 | 7 | 0 | 4 | 0 | 2 | |
| Tharsis | Rt-28 | 15 | 2,892 | 99 | 79 | 0 | 57 | N.D. | 6 | |
| Tharsis | Rt-29 | 39 | 2,947 | 158 | 66 | 0 | 126 | N.D. | 4 | |
| Sotiel | Rt-30 | 24 | 2,861 | 292 | 180 | 0 | 17 | N.D. | 5 | |
| Residue | Peña Hierro | RT-4-1 | 71 | 20,257 | 110 | 0 | 0 | 0 | 14 | 32 |
| Peña Hierro | RT-5-1 | 50 | 25,114 | 25 | 67 | 0 | 12 | 2 | 33 | |
| Peña Hierro | RT-6-1 | 99 | 20,698 | 47 | 0 | 0 | 0 | 1 | 27 | |
| Las Delgadas | RT-8-1 | 13 | 75,601 | 26 | 113 | 0 | 43 | 62 | 317 | |
| Las Delgadas | RT-8-2 | 116 | 29,413 | 44 | 59 | 0 | 0 | 9 | 36 | |
| Nerva | RT-20-1 | 73 | 35,144 | 441 | 0 | 0 | 32 | 11 | 36 | |
| Nerva | RT-21-1 | 200 | 36,437 | 442 | 0 | 0 | 198 | 12 | 33 | |
| Nerva | RT-21-2 | 193 | 32,627 | 486 | 0 | 0 | 249 | 13 | 36 | |
| Tharsis | Rt-28 | 98 | 28,157 | 1,210 | 861 | 0 | 24 | N.D. | 14 | |
| Tharsis | Rt-29 | 366 | 14,135 | 644 | 961 | 0 | 61 | N.D. | 17 | |
| Sotiel | Rt-30 | 245 | 17,395 | 1,291 | 112 | 0 | 110 | N.D. | 13 | |
N.D.: not detected.
| Location | Sample | Q | Phy | Fd | Hem | V | K | Ch | I | Others |
|---|---|---|---|---|---|---|---|---|---|---|
| Peña Hierro | RT-4–1 | 55 | 34 | 6 | - | Tr | 15 | - | 83 | Ilmenite, zircon, barite, rutile, monazite and cerianite |
| Peña | RT-5–1 | 45 | 44 | 6 | - | 5 | 35 | - | 60 | Ilmenite, zircon, rutile and monazite |
| Peña Hierro | RT-6–1 | 57 | 38 | Tr | - | 5 | 5 | - | 90 | Rutile, monazite and ilmenite |
| Delgadas | RT-8–1 | 7 | 61 | 27 | Tr | Non quantifiable | Pyrite | |||
| Delgadas | RT-8–2 | 52 | 43 | Tr | X | Monazite, barite and rutile | ||||
| Nerva | RT-20–1 | 55 | 40 | Tr | X | 5 | - | 5 | 90 | Barite, monazite, rutile, ilmenite |
| Nerva | RT-21–1 | 53 | 35 | 7 | - | 5 | - | 5 | 90 | Zircon, barite, rutile, monazite |
| Nerva | RT-21–2 | 59 | 31 | 5 | - | 5 | - | 5 | 90 | Pb antimonide, cassiterite, pyrite, Pb associated to sulphides. |
| Tharsis | Rt-28 | 56 | 34 | 5 | Tr | - | 21 | - | 79 | Fe oxy-hydroxides with Pb |
| Tharsis | Rt-29 | 76 | 24 | 5 | - | - | 30 | - | 70 | Fe oxy-hydroxides with Pb |
| Sotiel | Rt-30 | 80 | 16 | 4 | - | - | 15 | - | 85 | Fe oxy-hydroxides with Pb |
Q: quartz; Phy: Phyllosilicates; Fd: Feldspar; Hem: Hematite; V: Vermiculite: K: Kaolinite; Chl: Chlorite; I: Illita; Tr: <5%.
| Elements | SiO2 | Al2O3 | Fe2O3 | MnO | MgO | CaO | Na2O | K2O | TiO2 | P2O5 |
|---|---|---|---|---|---|---|---|---|---|---|
| RT-21-1 Total | 48.6 | 14.8 | 7.5 | 0.2 | 1.3 | 1.8 | 0.4 | 3.4 | 0.7 | 0.8 |
| RT-21-1 Al | 49.6 | 17.1 | 5.8 | 0.2 | 1.2 | 1.2 | 0.9 | 3.7 | 0.8 | 0.6 |
| RT-21-1 As | 21.2 | 7.5 | 31.7 | 0.1 | 0.5 | 1.1 | N.D. | 1.9 | 0.4 | 0.6 |
| RT-21-1 Ca | 37.2 | 14.6 | 4.7 | 0.2 | 1.5 | 6.0 | N.C. | 3.3 | 0.5 | 1.0 |
| RT-21-1 Cu | 37.0 | 10.5 | 5.5 | 0.1 | N.C. | 1.1 | N.D. | 2.3 | 0.4 | 0.5 |
| RT-21-1 P | 31.7 | 10.1 | 7.9 | 0.1 | N.C. | 3.5 | N.D. | 2.8 | 0.5 | 3.9 |
| RT-21-1 Fe | 30.0 | 9.9 | 27.9 | 0.2 | 1.1 | 1.3 | 0.1 | 2.1 | 0.5 | 0.6 |
| RT-21-1 Mg | 38.5 | 6.5 | 7.2 | 1.2 | 5.0 | 1.3 | N.C. | 1.6 | 0.4 | N.C. |
| RT-21-1 Mn | 37.7 | 10.0 | 9.2 | 2.6 | 1.4 | 2.7 | N.C. | 2.3 | 0.6 | 0.9 |
| RT-21-1Pb | 19.0 | 6.5 | 31.4 | 0.1 | 0.5 | 1.0 | N.C. | 1.7 | 0.4 | 0.6 |
| RT-21-1 K | 44.2 | 14.3 | 3.5 | 0.1 | 1.1 | 1.1 | 0.2 | 7.9 | 0.4 | 0.6 |
| RT-21-1 Si | 69.0 | 13.3 | 4.5 | 0.1 | 0.9 | 0.6 | N.C. | 3.0 | 0.6 | 0.5 |
| RT-21-1Ti | 54.1 | 14.4 | 7.0 | 0.2 | 1.4 | 1.5 | N.C. | 3.4 | 2.8 | 0.7 |
| RT-21-1 Zn | 30.3 | 9.6 | 29.8 | 0.2 | 1.1 | 1.4 | N.C. | 2.0 | 0.5 | 0.8 |
ND: Not detected; N.C.: Not quantifiable.
| S | Cu | Zn | As | Zr | Ba | Pb | |
|---|---|---|---|---|---|---|---|
| RT-21-1 Total | 1,751 | 496 | 501 | 115 | 157 | 791 | 567 |
| RT-21-1 Al | 1,667 | 290 | 452 | 113 | N.C. | N.D. | 383 |
| RT-21-1 As | 1,767 | 658 | 1,837 | 1,849 | N.D. | N.D. | 4,276 |
| RT-21-1 Ba | 1,800 | N.C. | N.C. | N.C. | N.D. | 49,832 | 2,901 |
| RT-21-1 Ca | 1,603 | N.C. | N.C. | N.C. | N.C. | N.D. | 395 |
| RT-21-1 Cu | 14,111 | 5911 | N.C. | N.C. | 0 | N.D. | N.C. |
| RT-21-1 P | 1,377 | N.C. | 579 | N.C. | N.C. | N.D. | 495 |
| RT-21-1 Fe | 1,857 | 472 | 1,362 | 319 | 110 | 820 | 1,061 |
| RT-21-1 Mg | N.D. | N.C | N.C. | N.C. | N.C. | N.D. | N.C. |
| RT-21-1 Mn | 1,716 | 463 | 530 | 187 | N.C | N.D. | 577 |
| RT-21-1 Pb | N.C. | 772 | 1,480 | 2,551 | 0 | N.D. | 5,806 |
| RT-21-1 Zn | 1,854 | 497 | 1,470 | 355 | 115 | N.C. | 1,060 |
N.D.: Not detected; N.C.: Not quantifiable.
| Location | As | Co | Cr | Cu | Ni | Pb | Zn |
|---|---|---|---|---|---|---|---|
| Cueva de la Mora | 6.76 | 1.35 | 1.59 | 7.23 | 1.33 | 7.04 | 3.98 |
| Valdelamusa | 1.78 | 0.59 | 0.44 | 4.09 | 0.42 | 7.13 | 5.00 |
| Lomero | 1.32 | 1.06 | 0.85 | 3.05 | 0.44 | 3.88 | 3.00 |
| Tharsis 1 | 26.12 | 2.41 | 1.33 | 9.00 | 1.13 | 52.92 | 11.70 |
| Tharsis 2 | 58.47 | 4.12 | 1.33 | 20.14 | 1.20 | 11.42 | 15.74 |
| Sotiel 1 | 21.24 | 1.53 | 1.77 | 29.45 | 1.56 | 58.75 | 6.25 |
| Sotiel 2 | 51.88 | 1.12 | 1.24 | 52.73 | 1.07 | 203.75 | 10.26 |
| P. Hierro 1 | 2.27 | 1.06 | 1.49 | 4.55 | 1.18 | 2.08 | 2.09 |
| P. Hierro 2 | 3.54 | 1.00 | 1.37 | 5.05 | 1.22 | 2.75 | 1.86 |
| Riotinto 1 | 4.63 | 1.65 | 1.41 | 9.64 | 0.58 | 7.25 | 2.58 |
| Riotinto 2 | 1.11 | 2.00 | 2.41 | 1.23 | 0.31 | 1.71 | 1.67 |
| Riotinto 3 | 1.28 | 0.94 | 1.18 | 2.59 | 0.62 | 8.21 | 3.53 |
| Delgadas 1 | 11.12 | 1.88 | 2.99 | 11.50 | 4.67 | 20.13 | 3.21 |
| Delgadas 2 | 5.69 | 1.88 | 2.96 | 9.86 | 1.62 | 9.08 | 3.61 |
| Delgadas 3 | 2.91 | 1.65 | 2.19 | 6.95 | 0.96 | 11.17 | 5.30 |
| Background | 20 | 16 | 85 | 31 | 27 | 31 | 72 |
Acknowledgments
This work was conducted by the Research Group RNM 135 of Junta de Andalucía within the framework of the project “Assessing contamination in soils affected by mining activities: Environmental problems in the Riotinto area (SW Spain)”, funded by Spain's Ministry of Science and Innovation (CTM2005–05832).
References
- Newman, M.C.; Jagoe, C.H. Inorganic Toxicants-Ligands and the Bioavailability of Metals in Aquatic Environments. In Bioavailability-Physical, Chemical, and Biological Interactions, Spec. Pub. Series; Hameling, J.L., Landrum, P.F., Bergman, H.L., Benson, W.H., Eds.; CRC Press Inc.: Boca Raton, FL, USA, 1994; pp. 39–61. [Google Scholar]
- Siegel, F.R. Environmental Geochemistry of Potentially Toxic Metals; Springer: Berlin, Germany, 2002. [Google Scholar]
- Plant, J.; Smith, D.; Smith, B.; Williams, L. Environmental geochemistry at the global scale. Appl. Geochem. 2001, 16, 1291–1308. [Google Scholar]
- Bowen, H.J.M. Environmental Chemistry of the Elements; Academic Press: London, UK, 1979. [Google Scholar]
- Bowie, S.H.U. Thornton Environmental Geochemistry and Health; Kluwer Academic Publisher: Hingham, MA, USA, 1985. [Google Scholar]
- Novotny, V. Diffuse sources of pollution by toxic metals and impact on receiving waters. In Heavy Metals; Salomon, W., Förstner, U., Madre, P., Eds.; Springer-Verlag: Berlin, German, 1995; pp. 33–35. [Google Scholar]
- Cheng, H.M.; Zheng, C.R.; Tu, C.; Zhou, D.M. Studies on loading capacity of agricultural soils for heavy metals and its applications in China. Appl. Geochem. 2001, 16, 1397–1403. [Google Scholar]
- Plumlee, G.S. The environmental geochemistry of mineral deposits. SEG Newsletter 1994, 371. [Google Scholar]
- Rulkens, W.H.; Grotenhuis, J.T.C.; Tichy, R. Methods for cleaning contaminated soils and sediments. In Heavy Metals; Salomons, W., Förstner, U., Mader, P., Eds.; Springer-Verlag: Berlin, German, 1995; pp. 165–191. [Google Scholar]
- Kabata-Pendias, A. Agricultural problems related to excessive trace metal contents of soils. In Heavy Metals; Salomons, W., Förstner, U., Mader, P., Eds.; Springer-Verlag: Berlin, German, 1995; pp. 3–18. [Google Scholar]
- Sposito, G. The Chemistry of Soils; Oxford University Press: Oxford, UK, 1989. [Google Scholar]
- Galán, E. The role of clay minerals in removing and immobilising heavy metals from contaminated soils. 351–361.
- Quevauvillier, P.; Lachica, M.; Barahona, M.; Gómez, A.; Rauret, G.; Muntuau, H. Certified reference material for the quality control of EDTA and DTPA extractable trace metal contents in calcareous soils (CRM 6000). Presenius J. Anal. Chem. 1998, 360, 505–511. [Google Scholar]
- Ure, A.M.; Davidson, C.M.; Thomas, R.P. Single and sequential extraction schemes for trace metal speciation in soil and sediment. Tech. Instrum. Anal. Chem. 1995, 17, 505–523. [Google Scholar]
- Hall, G.E.M.; Vaive, J.E.; Beer, R.; Hoashi, M. Selective leaches revisited, with emphasis on the amorphous Fe oxy-hydroxide phase extraction. J. Geochem. Explor. 1996, 56, 59–78. [Google Scholar]
- López Julián, P.L.; Mandado Collado, J.M. Extracciones químicas secuenciales de metales pesados. Aplicación en Ciencias Geológicas. Estud. Geol. 2002, 58, 133–144. [Google Scholar]
- Tessier, A.; Campbell, P.G.C.; Bisson, M. Sequential extraction procedure for the speciation of particulate trace metals. Anal. Chem. 1979, 57, 844–851. [Google Scholar]
- Ure, A.M.; Quevauviller, P.H.; Muntau, H.; Griepink, B. Speciation of heavy metals in soils and sediment. An account of the improvement and harmonisation of extraction techniques undertaken under the auspecies of the BCR of the Commission of the European Communities. Int. J. Environ. Anal. Chem. 1993, 51, 135–151. [Google Scholar]
- Quevauvillier, P.; Rauret, G.; Muntuau, H.; Ure, A.M.; Rubio, R.; López-Sánchez, J.F.; Fielder, H.D.; Griepink, B. Evaluation of a sequential extraction procedure for the determination of extractable trace metal contents in sediments. Fresenius' J. Anal. Chem. 1994, 349, 808–814. [Google Scholar]
- Gómez Ariza, J.L.; Giraldez, I.; Sánchez-Rodas, D.; Morales, E. Metal sequential extraction procedure optimized for heavily polluted and iron oxide rich sediments. Anal. Chem. Acta 2000, 414, 151–164. [Google Scholar]
- Galán, E.; Gómez Ariza, J.L.; González, I.; Fernández Caliani, J.C.; Morales, E.; Giráldez, I. Utilidad de las Técnicas de Extracción Secuencial en la Mejora y Caracterización Mineralógica por DRX de Suelos y Sedimentos con altos Contenidos de Óxidos de Hierro. In Integración de Ciencia y Tecnología de Arcillas en el Contexto Tecnológico-Social del Nuevo Milenio; Pascual, J., Zapatero, J., Ramírez del Valle, A.J., Moya García, M.V., Eds.; Sociedad Espanola de Arcillas y Diputación Provincial de Málaga: Sevilla, Spain, 2000; pp. 337–347. [Google Scholar]
- Nirel, P.M.V.; Morel, F.M.M. Pitfalls of sequential extractions. Water Resour. 1990, 24, 1055–1056. [Google Scholar]
- Dold, B.; Fontboté, L. A mineralogical and geochemical study of element mobility in sulfide mine tailings of the Fe-oxide Cu-Au deposits from the Punta del Cobre district, northern Chile. Chem. Geol. 2002, 189, 135–163. [Google Scholar]
- Dold, B. Speciation of the most soluble phases in a sequential extraction procedure adapted for geochemical studies of copper sulphide mine waste. J. Geochem. Explor. 2003, 80, 55–68. [Google Scholar]
- Manceau, A.; Kersten, M.; Marcus, M.A.; Geoffroy, N.; Granina, L. Ba and Ni speciation in a nodule of binary Mn oxide phase composition from lake Baikal. Geochim. Cosmochim. Acta 2007, 71, 1967–1981. [Google Scholar]
- Esbrí, J.M.; Bernaus, A.; Ávila, M.; Kocman, D.; García-Noguero, E.M.; Guerrero, B.; Gaona, X.; Álvarez, R.; Perez-Gonzalez, G.; Valiente, M.; Higueras, P.; Horvat, M.; Loredo, J. XANES speciation of mercury in three mining districts––Almadén, Asturias (Spain), Idria (Slovenia). J. Synchrotron Radiat. 2010, 17, 179–186. [Google Scholar]
- Parkhust, D.L.; Appelo, C.A.J. Users' guide to PHREEQC (version 2)––A computer program for speciation, reaction-path, advenctive-transport and inverse geochemical calculation. US. Geological Survey. Water Resour. Investig. Rep. 1999, 99, 4259–4312. [Google Scholar]
- Chojnacka, K.; Chojnacki, A.; Górecka, H.; Górecki, H. Bioavailability of heavy metals from polluted soils to plants. Sci. Total Environ. 2005, 337, 175–182. [Google Scholar]
- De Miguel, E.; Callaba, A.; Arranz, J.C.; Cala, V.; Chacón, E.; Gallego, E.; Alberruche, E.; Alonso, C.; Fernández-Canteli, P.; Iribarren, I.; et al. Determinación de Niveles de Fondo y Niveles de Referencia de Metales Pesados y Otros Elementos Traza en Suelos de la Comunidad de Madrid; Instituto Geológico y Minero de Espana: Madrid, Spain, 2002; p. 167. [Google Scholar]
- IHOBE. Investigación de la Contaminación del Suelo. Vol. 8. Calidad del Suelo. Valores Indicativos de Evaluación; Departamento de Urbanismo, Vivienda y Medio Ambiente: Gobierno Vasco: San Sebastián, Spain, 1993. [Google Scholar]
- Navas, A.; Machín, J. Spatial distribution of heavy metals and arsenic in soils of Aragón (northeast Spain): Controlling factors and environmental implications. Appl. Geochem. 2002, 17, 961–973. [Google Scholar]
- Jiménez Ballesta, R.; Conde Bueno, P.; Martín Rubí, J.A.; García Giménez, R. Niveles de fondo geoquímico e influencia del marco geológico en las concentraciones edafogeoquímicas de base de suelos seleccionados de Castilla-La Mancha. Estud. Geol. 2010, 66, 123–130. [Google Scholar]
- Aguilar, J.; Dorronsoro, C.; Galán, E.; Gómez Ariza, J.L. Los criterios y Estándares Para Declarar un Suelo Como Contaminado en Andalucía y la Metodología y Técnica de Toma de Muestras y Análisis Para su Investigación. In Investigación y Desarrollo Medioambiental en Andalucía; OTRI. Universidad de Sevilla: Sevilla, Spain, 1999; pp. 61–64. [Google Scholar]
- Sierra, M.; Díez, M.; Martinez, J.; Fernández, J.; Aguilar, J. A new index to evaluate the anomalies of trace elements in soils. The case of SE Spain. Span. J. Agric. Res. 2009, 7, 933–943. [Google Scholar]
- Finnecy, K.; Pearce, I. Land Contamination and Reclamation. In Understanding of Our Environment; Hester, R.E., Ed.; Royal Soc. of Chemistry: London, UK, 1986; pp. 329–337. [Google Scholar]
- Kloke, A.; Einkmann, T. Nutzung- und schutzbezogene Orientierungsdaten für (Schad) Stoffe in Böden, Sonderdruck aus Heft 1.8.; Erich Schmidt Verlag: Berlin, Germany, 1991. [Google Scholar]
- Salminen, R.; Tarvainen, T. The problem of defining geochemical baselines. A case study of selected elements and geological materials in Finland. J. Geochem. Explor. 1997, 60, 91–98. [Google Scholar]
- Baize, D.; Sterckeman, T. Of the necessity of knowledge of the natural pedo-geochemical background content in the evaluation of the contamination of soils by trace elements. Sci. Total Environ. 2001, 264, 127–139. [Google Scholar]
- Tarvainen, T.; Kallio, E. Baselines of certain bioavailable and total heavy metal concentrations in Finland. Appl. Geochem. 2002, 17, 975–980. [Google Scholar]
- Salomons, W. Environmental impact of metals derived from mining activities. J. Geochem. Explor. 1995, 53, 53–56. [Google Scholar]
- Merefield, J.R. Sediment Mineralogy and the Enviromental Impact of Mining. In Sediment and Water Quality in River Catchments; Foster, I.D.L., Gurnell, A.M., Webb, B.W., Eds.; Wiley: Hoboken, NJ, USA, 1995; pp. 145–160. [Google Scholar]
- Macías, F. Contaminación de Suelos: Algunos Hechos y Perspectivas. In Problemática Geoambiental y Desarrollo; Ortiz Silla, R., Ed.; V Reunión Nacional de Geología. Ambiental y Ordenación del: Territorio, Spain, 1993; volume I; pp. 53–74. [Google Scholar]
- Macías, F. Los Suelos de Mina: Su Recuperación. In Evaluación y Manejo de Suelos; Aguilar, J., Martínez, A., Roca, A., Eds.; Universidad de Granada: Granada, Spain, 1996; pp. 227–243. [Google Scholar]
- Reimann, C.; Koller, F.; Kashulina, G.; Niskavaara, H.; Englmaier, P. Influencie of extreme pollution on the inorganic chemical composition of some plants. Environ. Pollut. 2001, 115, 239–252. [Google Scholar]
- Gratton, W.S.; Nkongolo, K.K.; Spiers, G.A. Heavy metal accumulation in soil and Jack pine (Pinus banksiana) needles in Sudbury, Ontario, Canada. Bull. Environ. Contam. Toxicol. 2000, 64, 550–557. [Google Scholar]
- Robinson, B.; Mills, T.M.; Petit, D.; Fung, L.E.; Green, F.S.; Clothier, B.E. Natural and induced cadmium accumulation in poplar and willows; Implications for phytoremediation. Plant Soils 2001, 227, 301–306. [Google Scholar]
- Lee, S.; Lee, B.; Kim, J.; Kim, J.; Lee, J. Human risk assessment for heavy metals and as contamination in the abandoned metal mine Areas, Korea. Environ. Monit. Assess. 2006, 119, 233–244. [Google Scholar]
- Zabowski, D.; Henry, C.L.; Zheng, Z.; Zhang, X. Mining impacts on trace metal content of water, soil and stream sediments in the Hei River Basin, China. Water Air Soil Pollut. 2001, 131, 261–273. [Google Scholar]
- Lin, C.; Tong, X.; Lu, W.; Yan, L.; Wu, Y.; Nie, C.; Chu, C.; Long, J. Environmental impacts of surface mining on mined lands, affected streams and agricultural lands in the Dabaoshan mine region, southern China. Land Degrad. Dev. 2005, 16, 463–474. [Google Scholar]
- Lee, C. Assessment of contamination load on water, soil and sediment affected by the Kongjujeil mine drainage, Republic of Korea. Environ. Geol. 2003, 44, 501–515. [Google Scholar]
- Sun, Y.; Xie, Z.; Li, J.; Xu, J.; Chen, Z.; Naidu, R. Assessment of toxicity of heavy metal contaminated soils by the toxicity characteristic leaching procedure. Environ. Geochem. Health 2006, 28, 73–78. [Google Scholar]
- Zhou, J.; Dang, Z.; Cai, M.F.; Liu, C. Soil heavy metal pollution around the dabaoshan mine, Guangdong Province, China. Pedosphere 2007, 17, 588–594. [Google Scholar]
- Chae, Jung, M. Heavy metals concentrations and factor affecting metal uptake by plants in the vicinity of a Korea Cu-W mine. Sensors 2008, 8, 2413–2423. [Google Scholar]
- Chae Jung, M. Contamination by Cd, Cu, Pb, and Zn in mine wastes from abandoned metal mines classified as mineralization types in Korea. Environ. Geochem. Health 2008, 30, 205–217. [Google Scholar]
- Chae Jung, M. Heavy metal contamination of soils and waters in and around the Imcheon Au-Ag mine, Korea. Appl. Geochem. 2001, 16, 1369–137. [Google Scholar]
- Kim, J.; Kim, K.; Lee, J.; Lee, J.; Jenny Cook, J. Assessment of as and heavy metal contamination in the vicinity of Duckum Au-Ag mine, Korea. Environ. Geochem. Health 2002, 24, 213–225. [Google Scholar]
- Noble, R.; Hough, R.M.; Watkins, R.T. Enrichment and exposure assessment of As, Cr and Pb of the soils in the vicinity of Stawell, Victoria, Australia. Environ. Geochem. Health 2010, 32, 193–205. [Google Scholar]
- Razo, I.; Carrizales, L.; Castro, J.; Díaz-Barriga, F.; Monroy, M. Arsenic and heavy metal pollution of soil, water and sediments in a semi-arid climate mining area in Mexico. Water Air Soil Pollut. 2004, 152, 129–152. [Google Scholar]
- Gutierrez Ruiz, M. Suelos y sedimentos afectados por la dispersión de jales inactivos de sulfuros metálicos en la zona minera de Santa Bárbara, Chihuahua, México. Rev. Mex. Cienc. Geol. 2007, 24, 170–184. [Google Scholar]
- Veado, M.A.; Arantes, I.A.; Oliveira, A.H.; Almeida, M.R.; Miguel, M.I.; Severo, M.I.; Cabaleiro, H.L. Metal pollution in the environment of minas gerais state––Brazil. Environ. Monit. Assess. 2007, 117, 157–172. [Google Scholar]
- Herbet, R.B. Partitioning of heavy metals in podzol soils contaminated by mine drainage waters, Dalarna, Sweden. Water Air Soil Pollut. 1997, 96, 39–59. [Google Scholar]
- Davies, E.; Bailinger, R. Heavy metals in soils in north Somerset, England, with special reference to contamination from base metal mining in the Mendips. Environ. Geochem. Health 1990, 12, 291–300. [Google Scholar]
- Dudka, S.; Piotrowska, M.; Chlopecka, A.; Witek, T. Trace metal contamination of soils and crop plants by the mining and smelting industry in Upper Silesia, South Poland. J. Geochem. Explor. 1995, 52, 237–250. [Google Scholar]
- Aleksander-Kwaterczak, U.; Helios-Rybicka, E. Contaminated sediments as a potential source of Zn, Pb, and Cd for a river system in the historical metalliferous ore mining and smelting industry area of South Poland. J. Soils Sediments 2009, 9, 13–22. [Google Scholar]
- Hofmann, T.; Schuwirth, N. Zn and Pb release of sphalerite (ZnS)-bearing mine waste tailings. J. Soils Sediments 2008, 8, 433–441. [Google Scholar]
- Gazdag, E.R.; Sipter, E. Geochemical background in heavy metals and human health risk assessment at an ore mine site, Gyongyosoroszi (North Hungary). Carpathian J. Earth Environ. Sci. 2008, 3, 83–92. [Google Scholar]
- Abreu, M.M.; Matias, M.J.; Clara, M.; Magalhães, F.; Basto, M.J. Impacts on water, soil and plants from the abandoned Miguel Vacas copper mine, Portugal. J. Geochem. Explor. 2008, 96, 161–170. [Google Scholar]
- Tavares, M.J.; Sousa, J.; Abreu, M.M. Ordinary kriging and indicator kriging in the cartography of trace elements contamination in São Domingos mining site (Alentejo, Portugal). J. Geochem. Explor. 2008, 98, 43–56. [Google Scholar]
- Navarro, M.C.; Pérez-Sirvent, C.; Martínez-Sánchez, M.J.; Vidal, J.; Tovar, P.J.; Bech, J. Abandoned mine sites as a source of contamination by heavy metals: A case study in a semi-arid zone. J. Geochem. Explor. 2008, 96, 183–193. [Google Scholar]
- García Lorenzo, M.L. Evaluación de la contaminación por vía hídríca de elementos traza en áreas con influencia minero-metalúrgicas. Aplicación a la Sierra Minera de Cartagena-La Unión (Murcia); Memoria Tesis Doctoral, Universidad de Murcia: Murcia, Spain, 2009. [Google Scholar]
- Martínez-Sánchez, M.J.; García-Lorenzo, M.L.; Molina-Ruiz, J.; Pérez-Sirvent, M.C. Mapping of trace elements contamination in an abandoned mining site in a semiarid zone. J. Geochem. Explor. 2011, 109, 1–7. [Google Scholar]
- Molina, J.; Oyarzun, R.; Esbrí, J.M.; Higueras, P. Mercury accumulation in soils and plants in the Almadén mining district, Spain: One of the most contaminated sites on Earth. Environ. Geochem. Health 2006, 28, 487–498. [Google Scholar]
- Collado Fernández, D.; Sánchez Garrido, J.; Navarro Flores, A.; del Moral Torres, F. Suelos desarrollados sobre materiales aluviales afectados por actividades mineras en la zona de El Hártela (Cuevas de Almanzora, Almeria). Edafología 2000, 7, 217–225. [Google Scholar]
- Martínez, J. Caracterización Geoquímica y Ambiental de Los Suelos del Sector Minero de Linaes; Tesis doctoral de la Universidad Politecnica de Madrid: Madrid, Spain, 2002. [Google Scholar]
- Adelino, M.A.; Romero, A.; González, I. Geochemical Anomalies in Soils of the Jaen Province (Andalusia, Spain). In Science and Technology for Environmental Protection; SETAC: Pensacola, FL, USA, 2010; p. 214. [Google Scholar]
- Chopin, E.; Black, S.; Hodson, M.E.; Coleman, M.L.; Alloway, B.J. A preliminary investigation into mining and smelting impacts on trace element concentrations in the soils and vegetation around Tharsis, SW Spain. Mineral. Mag. 2003, 67, 279–288. [Google Scholar]
- Fernández-Caliani, J.C.; González, I.; Aparicio, P.; Barba, C.; Galán, E. Niveles de concentración de arsénico y metales pesados en los suelos del entorno de las minas abandonadas de la Faja Pirítica Ibérica. Macla 2005, 3, 73–74. [Google Scholar]
- López, M. Estudio de la Contaminación por Metales Pesados en Suelos Agrícolas de la Comarca Minera de Riotinto; Trabajo de investigación, para la obtención del Diploma de Estudios Avanzados, Universidad de Sevilla: Sevilla, Spain, 2006. [Google Scholar]
- Chopin, E.I.B.; Alloway, B.J. Trace element partitioning and soil particle characterisation around mining and smelting areas at Tharsis, Ríotinto and Huelva, SW Spain. Sci. Total Environ. 2007, 373, 488–500. [Google Scholar]
- López, M.; González, I.; Romero, A. Trace Elements contamination of agricultural soils affected by sulphide exploitation (Iberian Pyrite Belt, SW Spain). Environ. Geol. 2008, 54, 805–818. [Google Scholar]
- Barba, C. Especiación Química y Mineralógica de Elementos Traza de Origen Minero en Suelos Agrícolas; Trabajo realizado para la obtención del Diploma de Estudios avanzados, Universidad de Huelva: Huelva, Spain, 2007. [Google Scholar]
- Barba Brioso, C.; Sánchez Blanco, E.; Fernández Caliani, J.C. Efectos del Drenaje Ácido de Minas Sobre la Composición Química y Mineralógica de Suelos Agrícolas. Una Aproximación experimental. In II Congreso Ibérico de la Ciencia del Suelo; Bellinfante, N., Parrales, I.A., Jordán, A., Martínez-Zavala, L., Eds.; IICICS2006: Huelva, Spain, 2007; p. 61. [Google Scholar]
- González, I.; López, M.; Romero, A.; Galán, E. Consideraciones químico-mineralógicas sobre la movilidad de elementos tóxicos en suelos afectados por explotaciones mineras de sulfuros (Faja Pirítica Ibérica). In Libro de Resúmenes Reunión SEA; SEA: Sevilla, Spain, 2007; pp. 22–24. [Google Scholar]
- González, I.; López, M.; Romero, A.; Galán, E.; Fernández Caliani, J.C.; Aparicio, P. Consideraciones mineralógicas sobre la movilidad de elementos tóxicos en suelos agrícolas afectados por explotaciones mineras de sulfuros (Faja Pirítica Ibérica). Geotemas 2008, 25, 72–78. [Google Scholar]
- Galán, E.; Fernández Caliani, J.C.; González, I.; Aparicio, P.; Romero, A. Influence of geological setting on geochemical baselines of trace elements in soils. Application to soils of South-West Spain. J. Geochem. Explor. 2008, 98, 89–106. [Google Scholar]
- Davies, B.E.; Ballinger, R.C. Heavy metals in soils in north Somerset, England, with special reference to contamination from base metal mining in the Mendips. Environ. Geochem. Health 1990, 12, 291–300. [Google Scholar]
- Wen, X.; Allen, H.E. Mobilization of heavy metals from Le An River Sediment. Sci. Total Environ. 1999, 227, 101–108. [Google Scholar]
- Galán, E. Mineralogía Aplicada; Síntesis, S.A: Madrid, Spain, 2003. [Google Scholar]
- Cama, J.; Ayora, C.; Querol, X.; Moreno, N. Metal adsorption on cáliz from pyrite contaminated soil. J. Environ. Eng. 2005, 131, 1052–1056. [Google Scholar]
- Galán, E.; Gómez Ariza, J.L.; González, I.; Fernández Caliani, J.C.; Morales, E.; Giraldez, I. Heavy metals partitioning in river sediments severely polluted by acid mine drainage in the Iberian Pyrite Belt. Appl. Geochem. 2003, 18, 409–421. [Google Scholar]
- Tornos, F. La geología y metalogenía de la faja pirítica ibérica. Macla 2008, 10, 13–24. [Google Scholar]
- Papadopaulos, A.; Prochaska, F.; Papadopoulos, F.; Gantidis, N.; Metaxa, E. Determination and evaluation of cadmium, copper, nickel and zinc in agricultural soils of western Macedonia, Greece. Environ. Manag. 2007, 40, 719–726. [Google Scholar]
- Bech, J.; Tume, P.; Longan, L.; Reverter, F.; Bech, J.; Tume, L.; Tempio, M. Concentration of Cd, Cu, Pb, Zn, Al, and Fe in soils Manresa, NE Spain. Environ. Monit. Assess. 2008, 145, 256–266. [Google Scholar]
- Rodríguez, L.; Ruiz, L.; Alonso-Azcárate, J.; Rincón, J. Heavy metal distribution and chemical speciation in tailings and soils around a Pb-Zn mine in Spain. J. Environ. Manag. 2008, 90, 1106–1116. [Google Scholar]
- Pickering, W.F. Metal ion speciation—Soils and sediment. Ore Geol. Rev. 1986, 1, 83–146. [Google Scholar]
- Yan, X.P.; Kerrich, R.; Hendry, M.J. Sequencial leachates of multiple grain size fractions from a clay-rich till, Sakatchewan, Canada: Implications for controls on the rare earth element gepchemistry of porewaters in an aquitard. Chem. Geol. 1999, 158, 53–79. [Google Scholar]
- Lee, S. Geochemistry and partitioning of trace metals in paddy soils affected by metal mine tailings in Korea. Geoderma 2006, 135, 26–37. [Google Scholar]
- Vodyanitskii, N. Arsenic, lead, and zinc compounds in contaminated soils according to EXAFS spectroscopic data: A review. Eurasian Soil Sci. 2006, 39, 611–621. [Google Scholar]
- Maskall, J.E.; Thornton, I. Chemical partitioning of heavy metals in soils, clays and rocks as historical lead smelting sites. Water Air Soil Pollut. 1998, 108, 391–409. [Google Scholar]
- Song, Y.; Wilson, M.J.; Moon, H.S.; Bacon, J.R.; Bain, D.C. Chemical and mineralogical forms of lead, zinc and cadmium in particle size fractions some wastes, sediments and soils in Korea. Appl. Geochem. 1999, 14, 621–633. [Google Scholar]
- Ortiz, M. Aproximación a la Minería y Metalurgia de Minas de Riotinto Desde la Antigüedad Hasta el Siglo XX; Tesis doctoral. Universidad Politécnica de Madrid: Madrid, Spain, 2003. [Google Scholar]
- Legaz, F.; Serna, M.D.; Ferrer, P.; Cebolla, V.; Primo-Milla, E. Análisis de Hojas, Suelos y Aguas Para Diagnóstico Nutricional de Plantaciones de Cítricos. Procedimiento de Toma de Muestras. Generalitat Valenciana; Conselleria d'Agricultura, Pesca i Alimentació: Generalitat Valenciana, Spain, 1995. [Google Scholar]
- Puga, S.; Sosa, M.; de la Mora, A.; Pinedo, C.; Jiménez, J. Concentraciones de As y Zn en vegetación nativa cercana a una presa de Jales. Rev. Int. Contm. Ambient 2006, 22, 75–82. [Google Scholar]
© 2011 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
González, I.; Galán, E.; Romero, A. Assessing Soil Quality in Areas Affected by Sulfide Mining. Application to Soils in the Iberian Pyrite Belt (SW Spain). Minerals 2011, 1, 73-108. https://doi.org/10.3390/min1010073
González I, Galán E, Romero A. Assessing Soil Quality in Areas Affected by Sulfide Mining. Application to Soils in the Iberian Pyrite Belt (SW Spain). Minerals. 2011; 1(1):73-108. https://doi.org/10.3390/min1010073
Chicago/Turabian StyleGonzález, Isabel, Emilio Galán, and Antonio Romero. 2011. "Assessing Soil Quality in Areas Affected by Sulfide Mining. Application to Soils in the Iberian Pyrite Belt (SW Spain)" Minerals 1, no. 1: 73-108. https://doi.org/10.3390/min1010073
APA StyleGonzález, I., Galán, E., & Romero, A. (2011). Assessing Soil Quality in Areas Affected by Sulfide Mining. Application to Soils in the Iberian Pyrite Belt (SW Spain). Minerals, 1(1), 73-108. https://doi.org/10.3390/min1010073




