Binding Analysis of Human Immunoglobulin G as a Zinc-Binding Protein
Abstract
:1. Introduction
2. Results and Discussion
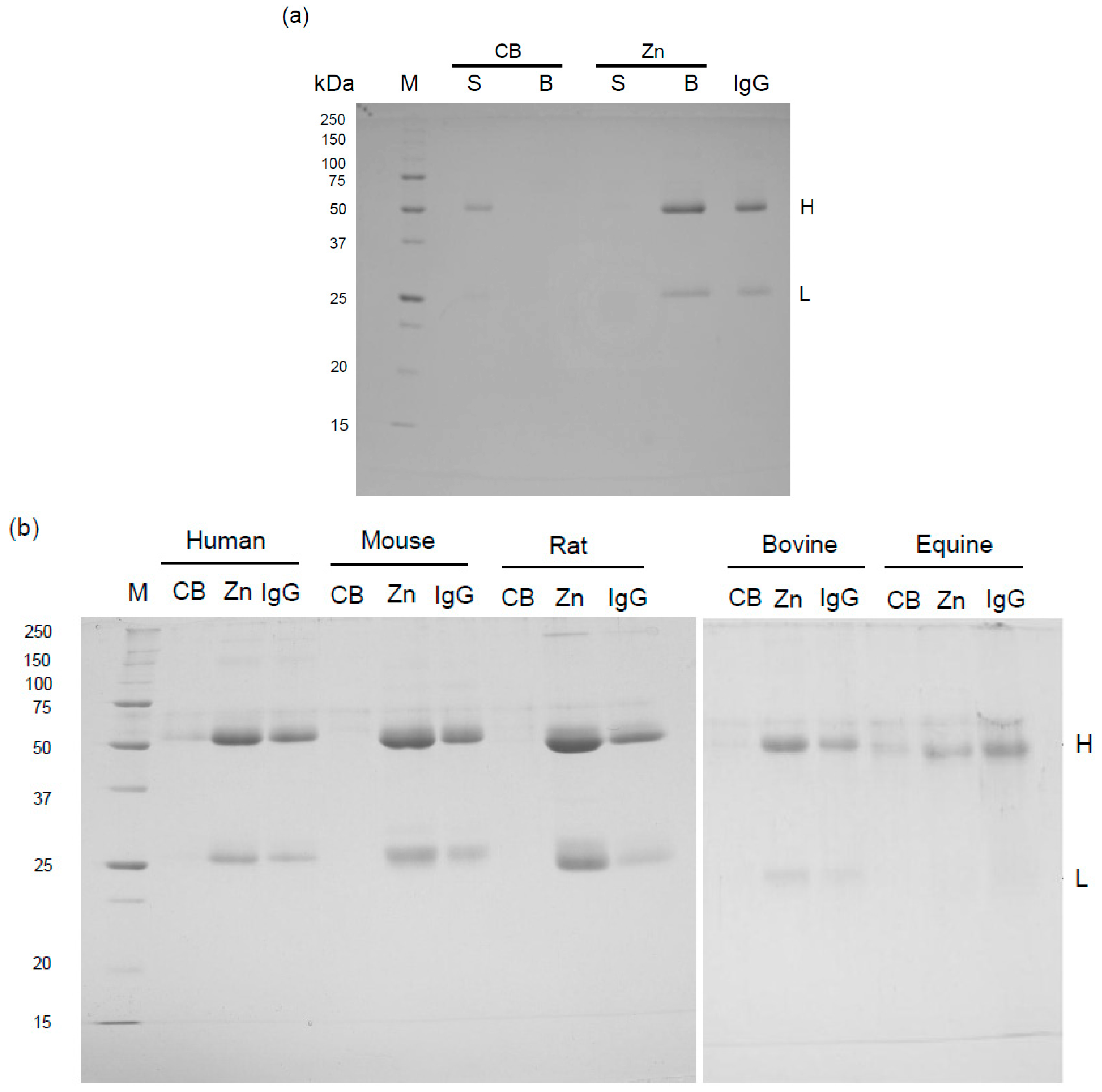
2.1. Binding of Mammalian IgGs to Zn-Beads
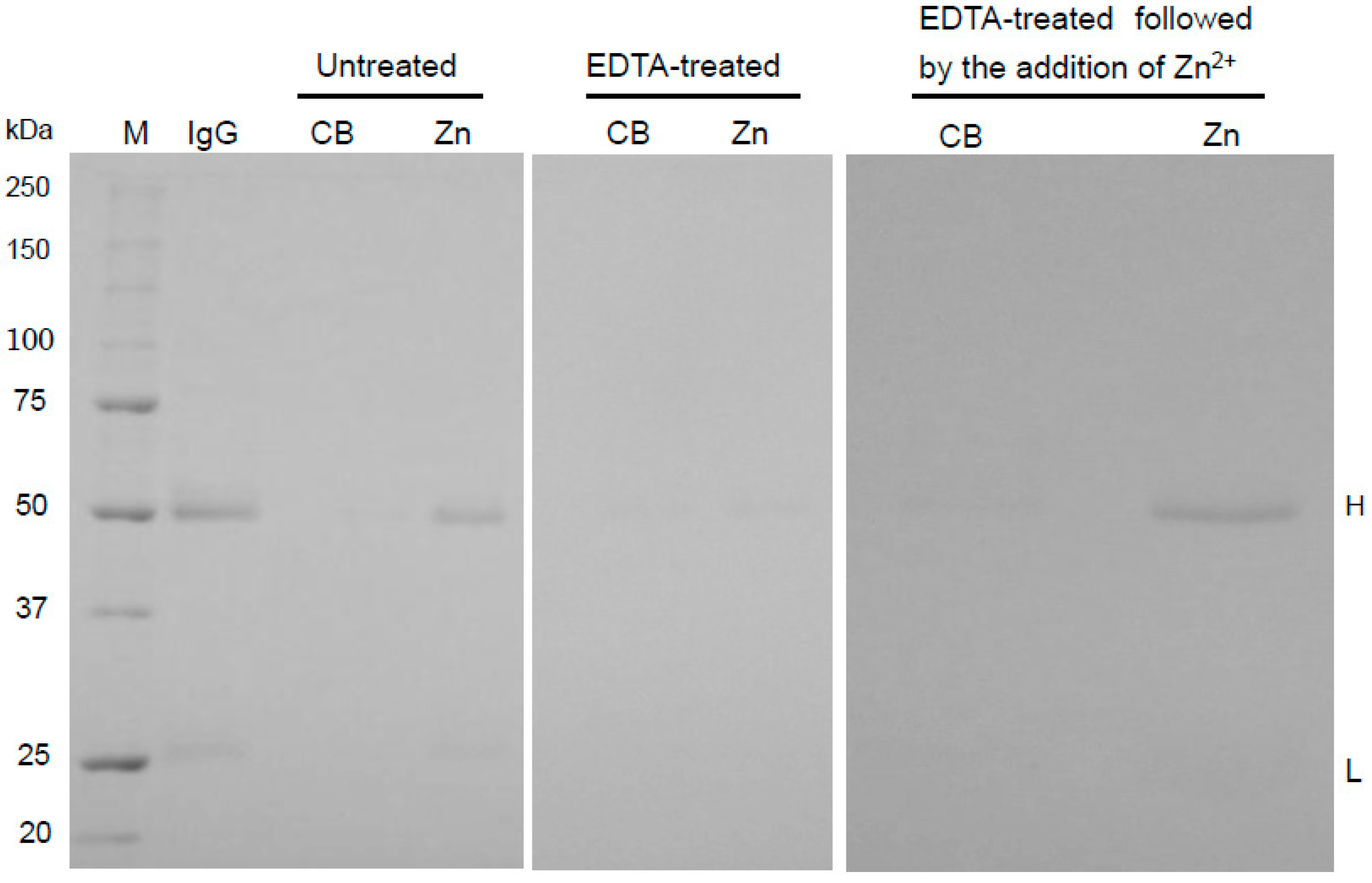
2.2. Binding Mechanism of Human IgG to Zn-Beads
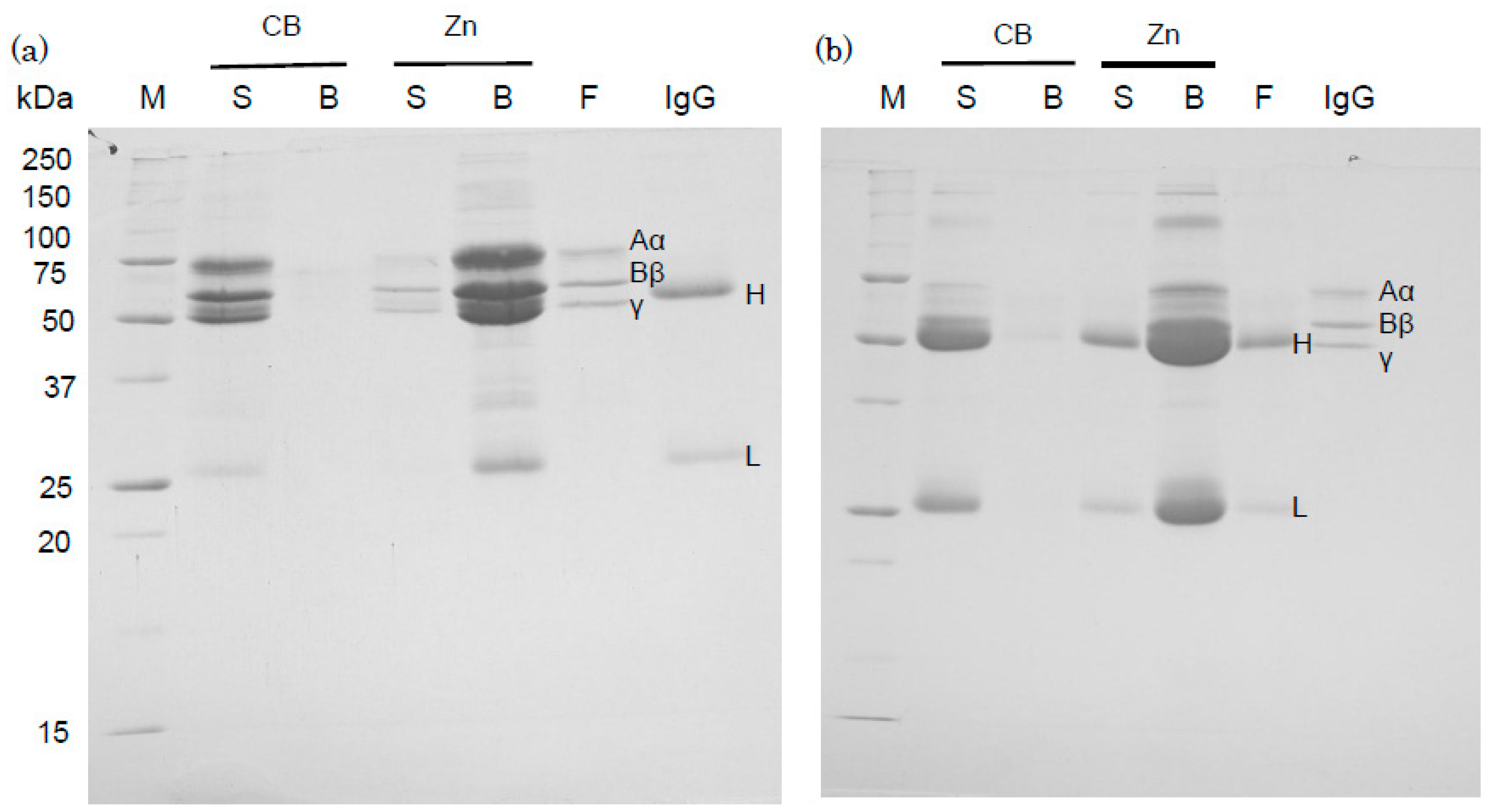
2.3. Binding of Human IgG and Fibrinogen to Zinc Ions
3. Experimental Section
3.1. Chemicals
3.2. SDS-PAGE
3.3. Binding of Mammalian IgGs to Zinc Ion–Immobilized Beads
3.4. Comparison of Binding Affinity of Human Fibrinogen and IgG to Zn-Beads
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of interest
References
- Prasad, A.S. Zinc: A miracle element. Its discovery and impact on human health. JSM Clin. Oncol. Res. 2014, 2, 1030–1036. [Google Scholar]
- Prasad, A.S. Zinc is an antioxidant and anti-inflammatory agent: Its role in human health. Front. Nutr. 2014, 1, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Prasad, A.S. Zinc: Role in immunity, oxidative stress and chronic inflammation. Curr. Opin. Clin. Nutr. Metab. Care 2009, 12, 646–652. [Google Scholar] [CrossRef] [PubMed]
- Hasse, H.; Rink, L. The immune system and the impact of zinc during aging. Immun. Ageing 2009, 6. [Google Scholar] [CrossRef] [PubMed]
- Bao, B.; Prasad, A.S.; Beck, F.W.J.; Bao, G.W.; Singh, T.; Ali, S.; Sarkar, F.H. Intracellular free zinc up-regulates IFN-γ and T-bet essential for Th1 differentiation in Con-A stimulated HUT-78 cells. Biochem. Biophys. Res. Acta 2011, 407, 703–707. [Google Scholar] [CrossRef] [PubMed]
- Hasse, H.; Rink, L. Signal transduction in monocytes: The role of zinc ions. Biometals 2007, 20, 579–585. [Google Scholar] [CrossRef] [PubMed]
- Margalit, O.; Simon, A.J.; Yakubov, E.; Puca, R.; Yosepovich, A.; Avivi, C.; Jacob-Hirsch, J.; Gelemter, I.; Harmelin, A.; Barshack, I.; et al. Zinc supplementation auguments in vivo antitumor effect of chemotherapy by restoring p53 function. Int. J. Cancer 2012, 131, E562–E568. [Google Scholar] [CrossRef] [PubMed]
- Cousins, R.J.; Liuzzi, J.P.; Lichten, L.A. Mammalian zinc transport, trafficking, and signals. J. Biol. Chem. 2006, 281, 24085–24089. [Google Scholar] [CrossRef] [PubMed]
- Scott, B.J.; Braswell, A.R. Identification of the serum binding proteins for iron, zinc, cadmium, nickel and calcium. Clin. Chem. 1983, 29, 629–633. [Google Scholar] [PubMed]
- Mocchegiani, E.; Costarelli, L.; Giacconi, R.; Cipriano, C.; Muti, E.; Malavolta, M. Zinc-binding proteins (metallothionein and α-2 macroglobulin) and immunosenescence. Exp. Gerontol. 2006, 41, 1094–1107. [Google Scholar] [CrossRef] [PubMed]
- Babaeva, E.E.; Vorobyova, U.A.; Denisova, E.A.; Medevedeva, D.A.; Cheknev, S.B. Binding of zinc cations to human serum γ-globulin. Bull. Exp. Biol. Med. 2006, 141, 602–605. [Google Scholar] [CrossRef] [PubMed]
- Higashi, S.; Nagasawa, K.; Yoshikawa, Y.; Watanabe, K.; Orino, K. Characterization analysis of human anti-ferritin autoantibodies. Antibodies 2014, 3, 169–181. [Google Scholar] [CrossRef]
- Orino, K. Functional binding analysis of human fibrinogen as an iron- and heme-binding protein. Biometals 2013, 26, 789–794. [Google Scholar] [CrossRef] [PubMed]
- Hammarström, L.; Persson, M.A.A.; Smith, C.I.E. Immunoglobulin subclass distribution of human anti-carbohydrate antibodies: Aberrant pattern in IgA-deficient donors. Immunology 1985, 54, 821–826. [Google Scholar] [PubMed]
- Boehm, T.K.; DeNardin, E. Fibrinogen binds IgG antibody and enhances IgG-mediated phagocytosis. Hum. Antibodies 2008, 17, 45–56. [Google Scholar] [PubMed]
- Porath, J.; Olin, B. Immobilized metal ion affinity adsorption and immobilized metal ion affinity chromatography of biomaterials. Serum protein affinities for gel-immobilized iron and nickel ions. Biochemistry 1983, 22, 1621–1630. [Google Scholar] [CrossRef] [PubMed]
- Tubek, S.; Grzanka, P.; Tubek, I. Role of zinc in hemostasis: A review. Biol. Trace Elem. Res. 2008, 121, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Olinescu, R.M.; Kummerow, F.A. Fibrinogen is an efficient antioxidant. J. Nutr. Biochem. 2001, 12, 162–169. [Google Scholar] [CrossRef]
- Kaplan, I.V.; Attaelmannan, M.; Levinson, S.S. Fibrinogen is an antioxidant that protects β-lipoprotein at physiological concentrations in a cell free system. Atherosclerosis 2001, 158, 455–463. [Google Scholar] [CrossRef]
- Nowak, P.; Zbikowska, H.M.; Ponczek, M.; Kolodziejczyk, J.; Wachowicz, B. Different vulnerability of fibrinogen subunits to oxidative/nitrative modifications induced by peroxynitrite: Functional consequences. Thromb. Res. 2007, 121, 163–174. [Google Scholar] [CrossRef] [PubMed]
- Prasad, A.S.; Meftah, S.; Abdallah, J.; Kaplan, J.; Brewer, G.J.; Bach, J.F.; Dardenne, M. Serum thymulin in human zinc deficiency. J. Clin. Investig. 1988, 8, 1202–1210. [Google Scholar] [CrossRef] [PubMed]
- Orino, K. Physiological implications of mammal ferritin-binding protein interacting with circulating ferritin and a new aspect of ferritin- and zinc-binding proteins. Biometals 2016, 29, 15–24. [Google Scholar] [CrossRef] [PubMed]
- Laemmli, U.K. Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature 1970, 227, 680–685. [Google Scholar] [CrossRef] [PubMed]

© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yamanaka, Y.; Matsugano, S.; Yoshikawa, Y.; Orino, K. Binding Analysis of Human Immunoglobulin G as a Zinc-Binding Protein. Antibodies 2016, 5, 13. https://doi.org/10.3390/antib5020013
Yamanaka Y, Matsugano S, Yoshikawa Y, Orino K. Binding Analysis of Human Immunoglobulin G as a Zinc-Binding Protein. Antibodies. 2016; 5(2):13. https://doi.org/10.3390/antib5020013
Chicago/Turabian StyleYamanaka, Yu, Sho Matsugano, Yasunaga Yoshikawa, and Koichi Orino. 2016. "Binding Analysis of Human Immunoglobulin G as a Zinc-Binding Protein" Antibodies 5, no. 2: 13. https://doi.org/10.3390/antib5020013
APA StyleYamanaka, Y., Matsugano, S., Yoshikawa, Y., & Orino, K. (2016). Binding Analysis of Human Immunoglobulin G as a Zinc-Binding Protein. Antibodies, 5(2), 13. https://doi.org/10.3390/antib5020013




