Reactivity of Rheumatoid Arthritis-Associated Citrulline-Dependent Antibodies to Epstein-Barr Virus Nuclear Antigen1-3
Abstract
:1. Introduction
2. Materials and Methods
2.1. Reagents
2.2. Peptides
2.3. Generation of Biotinylated Synthetic EBNA3 Peptides
2.4. Patient Material
2.5. Detection of Antibodies by Enzyme-Linked Immunosorbent Assay and Streptavidin-Capture Enzyme-Linked Immunosorbent Assay
2.6. Data Interpretation and Statistics
3. Results
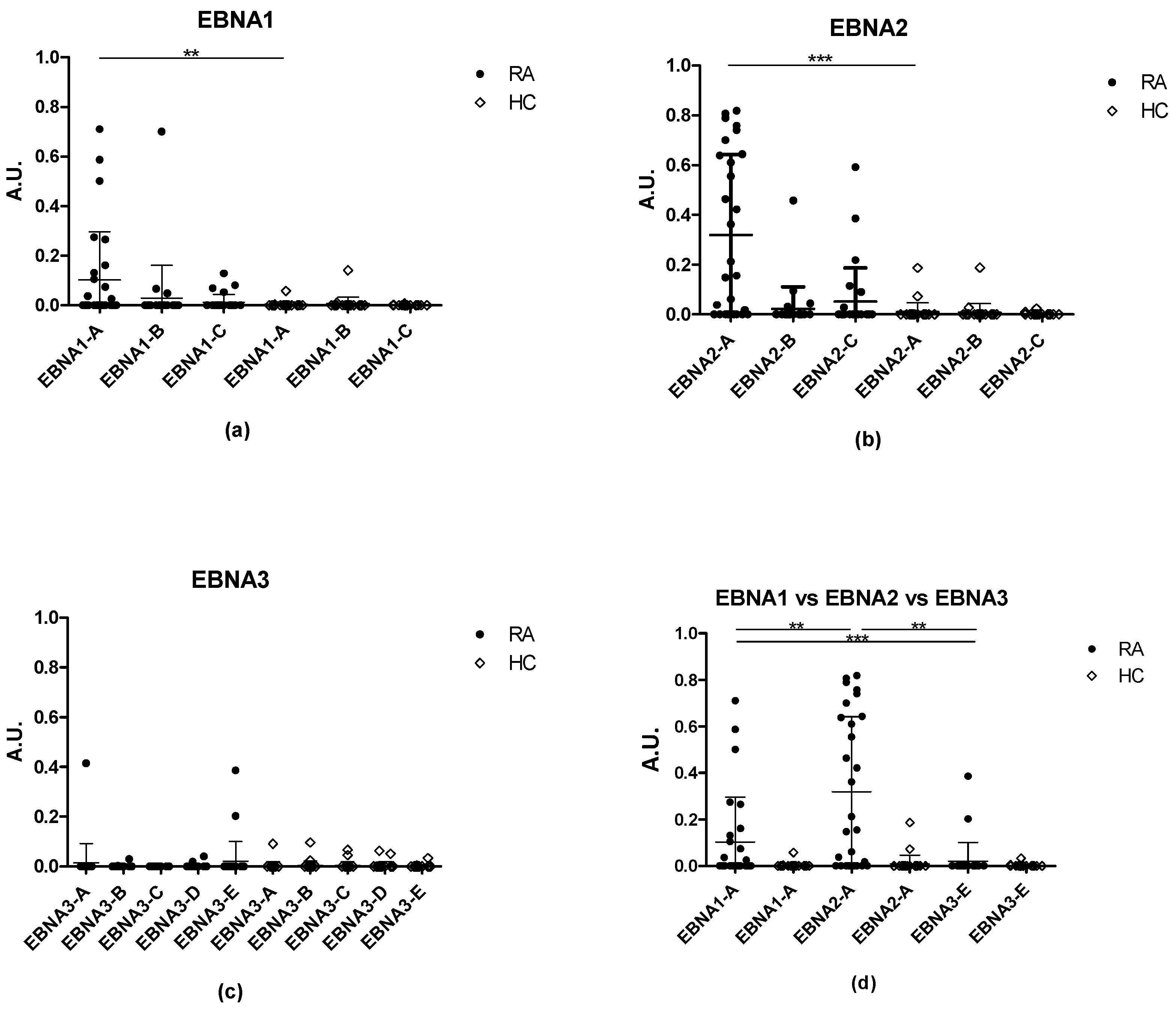
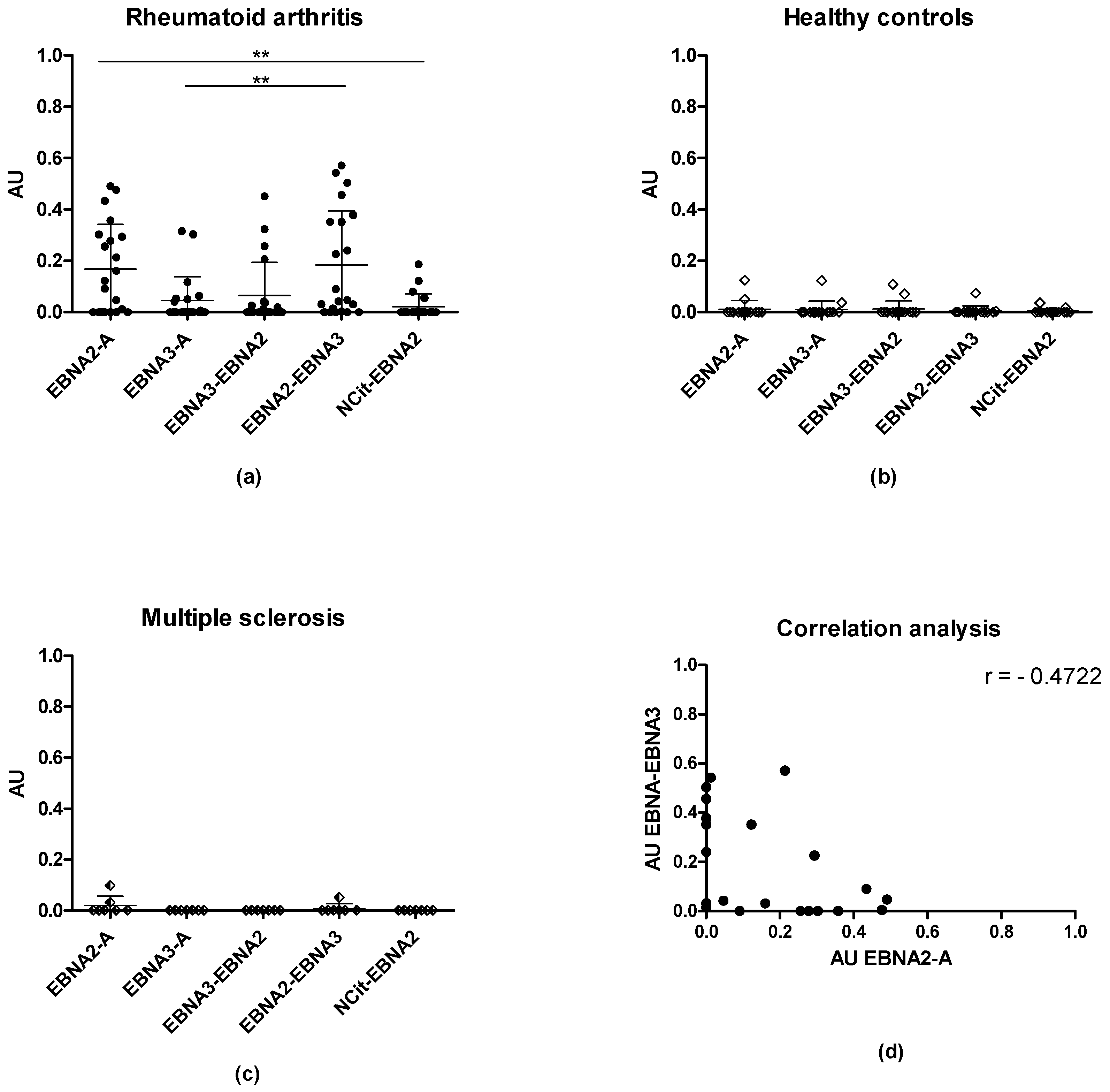
3.1. Reactivity of Rheumatoid Arthritis Sera and Healthy Control Sera to Citrullinated Epstein-Barr Nuclear Antigen Peptides
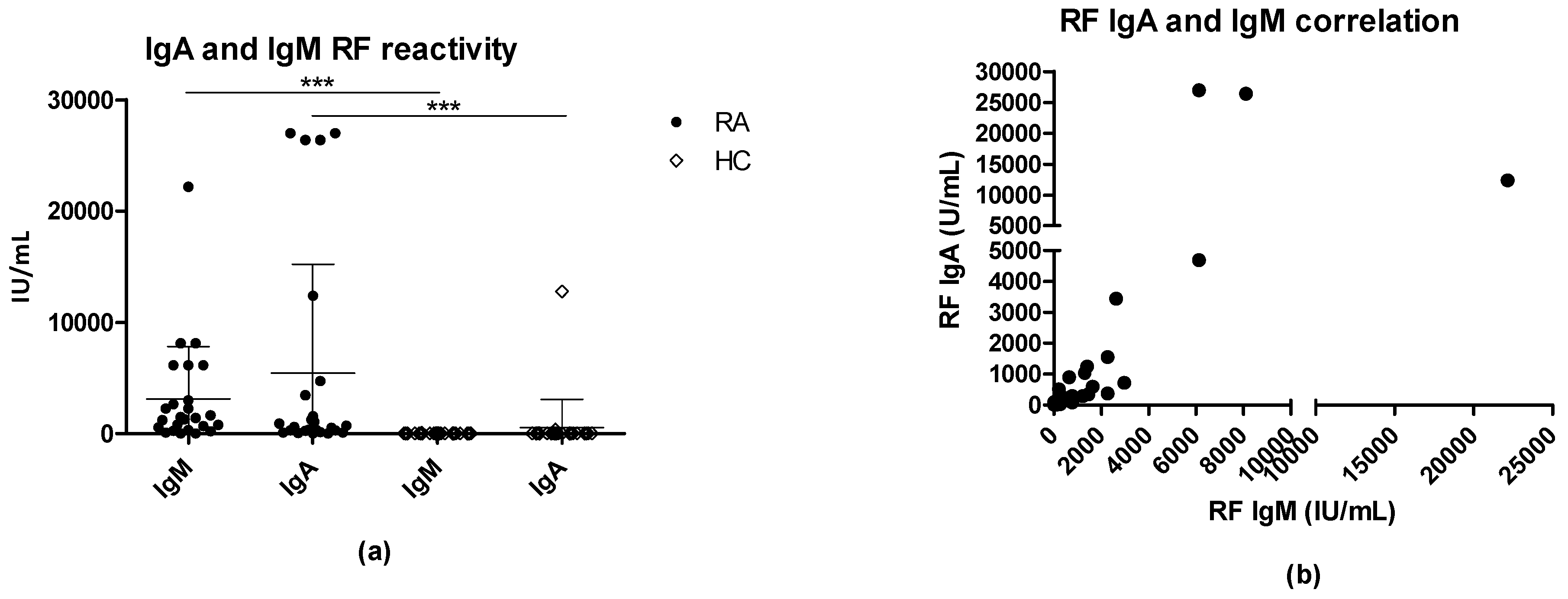
3.2. Serologic Correlations between Rheumatoid Factors and EBNA-Specific Proteins and Peptides
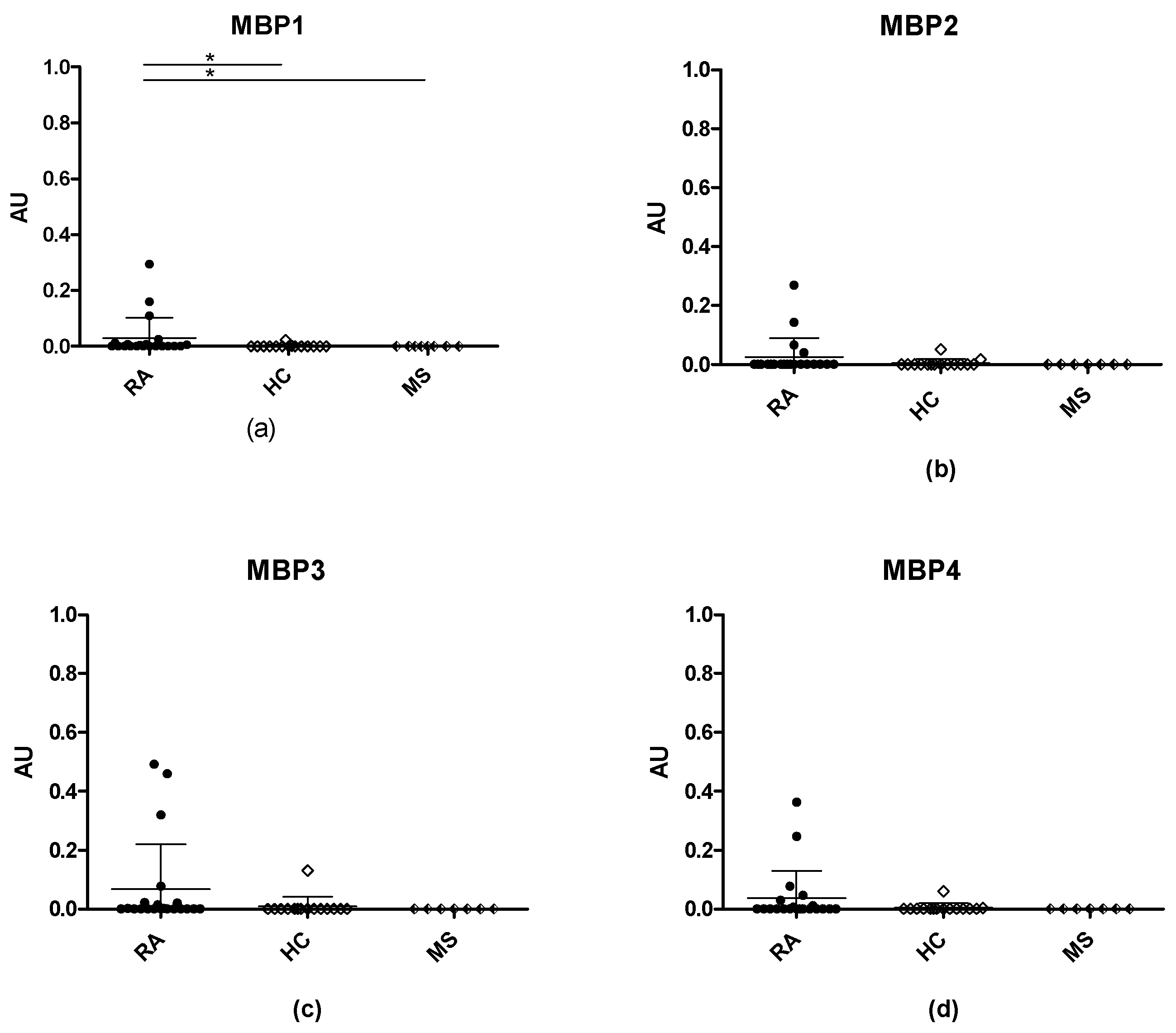
3.3. Anti-Citrullinated Protein Antibody Reactivities to Citrullinated MBP Peptides
4. Discussion
Author Contributions
Funding
Informed Consent Statement
Conflicts of Interest
References
- Littlejohn, E.A.; Monrad, S.U. Early Diagnosis and Treatment of Rheumatoid Arthritis. Prim. Care Clin. Off. Pract. 2018, 45, 237–255. [Google Scholar] [CrossRef] [PubMed]
- Scott, D.L.; Wolfe, F.; Huizinga, T.W.J. Rheumatoid arthritis. Lancet 2010, 376, 1094–1108. [Google Scholar] [CrossRef]
- Aletaha, D.; Smolen, J.S. Diagnosis and Management of Rheumatoid Arthritis. JAMA 2018, 320, 1360–1372. [Google Scholar] [CrossRef] [PubMed]
- Smolen, J.S.; Aletaha, D.; McInnes, I.B. Rheumatoid arthritis. Lancet 2016, 388, 2023–2038. [Google Scholar] [CrossRef]
- Klareskog, L.; Catrina, A.I.; Paget, S. Rheumatoid arthritis. Lancet 2009, 373, 659–672. [Google Scholar] [CrossRef]
- Aletaha, D.; Neogi, T.; Silman, A.; Funovits, J.; Felson, D.; Bingham, C.; Birnbaum, N.; Burmester, G.; Bykerk, V.; Costenbader, K.; et al. 2010 Rheumatoid arthritis classification criteria: An American College of Rheumatology/European League Against Rheumatism collaborative initiative. Arthritis Rheum. 2010, 62, 2569–2581. [Google Scholar] [CrossRef] [PubMed]
- Maibom-Thomsen, S.L.; Trier, N.H.; Holm, B.; Hansen, K.B.; Rasmussen, M.I.; Chailyan, A.; Marcatili, P.; Hojrup, P.; Houen, G. Immunoglobulin G structure and rheumatoid factor epitopes. PLoS ONE 2019, 14, e0217624. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Schellekens, G.A.; de Jong, B.A.; van de Putte, L.B.; van Venrooij, W.; van den Hoogen, F. Citrulline is an essential constituent of antigenic determinants recognized by rheumatoid arthritis-specific autoantibodies. J. Clin. Investig. 1998, 101, 273–281. [Google Scholar] [CrossRef] [Green Version]
- Schmerling, R.H.; Delbanco, T.L. How useful is the rheumatoid factor? An analysis of sensitivity, specificity, and predictive value. Arch. Intern. Med. 1992, 152, 2417–2420. [Google Scholar] [CrossRef]
- Ingegnoli, F.; Castelli, R.; Gualtierotti, R. Rheumatoid factors: Clinical applications. Dis. Markers 2013, 35, 727–734. [Google Scholar] [CrossRef] [Green Version]
- Lisse, J.R. Does rheumatoid factor always mean arthritis? Postgrad. Med. 1993, 94, 133–134. [Google Scholar] [CrossRef] [PubMed]
- Trier, N.H.; Holm, B.E.; Heiden, J.; Slot, O.; Locht, H.; Jensen, B.; Lindegaard, H.; Svendsen, A.; Tandrup Nielsen, C.; Jacobsen, S.; et al. The use of synthetic peptides for detection of anti-citrullinated protein antibodies in rheumatoid arthritis. J. Immunol. Methods 2018, 454, 6–14. [Google Scholar] [CrossRef] [PubMed]
- Trier, N.H.; Holm, B.E.; Slot, O.; Locht, H.; Lindegaard, H.; Svendsen, A.; Houen, G. Physical Characteristics of a Citrullinated Pro-Filaggrin Epitope Recognized by Anti-Citrullinated Protein Antibodies in Rheumatoid Arthritis Sera. PLoS ONE 2016, 11, e0168542. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Heidari, B.; Firouzjahi, A.; Heidari, P.; Hajian, K. The prevalence and diagnostic performance of anti-cyclic citrullinated peptide antibody in rheumatoid arthritis: The predictive and discriminative ability of serum antibody level in recognizing rheumatoid arthritis. Ann. Saudi Med. 2009, 29, 467–470. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sun, J.; Liu, G.; Zhang, Y.; Liu, L. Diagnostic accuracy of combined tests of anti cyclic citrullinated peptide antibody and rheumatoid factor for rheumatoid arthritis: A meta-analysis. Clin. Exp. Rheumatol. 2014, 32, 11–21. [Google Scholar] [PubMed]
- Saigal, R.; Bhakal, S.S.; Goyal, L.; Goal, A.D. Seroprevalence of anti-citrullinated protein antibodies (ACPA) in patients with rheumatic diseases other than rheumatoid arthritis. J. Assoc. Physicians India 2018, 66, 26–28. [Google Scholar] [PubMed]
- Trier, N.; Holm, B.; Slot, O.; Locht, H.; Lindegaard, H.; Svendsen, A.; Nielsen, C.; Jacobsen, S.; Theander, E.; Houen, G. Application of synthetic peptides for detection of anti-citrullinated peptide antibodies. Peptides 2016, 76, 87–95. [Google Scholar] [CrossRef]
- Van de Stadt, L.; de Koning, M.; van de Stadt, R.; Wolbink, G.; Dijkmans, B.; Hamann, D.; van Schaardenburg, D. Development of the anti-citrullinated protein antibody repertoire prior to the onset of rheumatoid arthritis. Arthritis Rheum. 2011, 63, 3226–3233. [Google Scholar] [CrossRef]
- Vossenaar, E.; Zendman, A.; van Venrooij, W.; Pruijn, G. PAD, a growing family of citrullinating enzymes: Genes, features and involvement in disease. Bioassays 2003, 25, 1106–1118. [Google Scholar] [CrossRef]
- Trier, N.; Houen, G. Epitope Specificity of Anti-Citrullinated Protein Antibodies. Antibodies 2017, 6, 5. [Google Scholar] [CrossRef]
- Tarcsa, E.; Marekov, L.; Mei, G.; Melino, G.; Lee, S.; Steinert, P. Protein unfolding by peptidylarginine deiminase. Substrate specificity and structural relationships of the natural substrates trichohyalin and filaggrin. J. Biol. Chem. 1996, 271, 30709–30716. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Schellekens, G.; Visser, H.; de Jong, B.; van den Hoogen, F.; Hazes, J.; Breedveld, F.; van Venrooij, W. The diagnostic properties of rheumatoid arthritis antibodies recognizing a cyclic citrullinated peptide. Arthritis Rheum. 2000, 43, 155–163. [Google Scholar] [CrossRef]
- Aggarwal, R.; Liao, K.; Nair, R.; Ringold, S.; Costenbader, K. Anti-citrullinated peptide antibody assays and their role in the diagnosis of rheumatoid arthritis. Arthritis Rheum. 2009, 61, 1472–1483. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Saraux, A.; Berthelot, J.; Devauchelle, V.; Bendaoud, B.; Chales, G.; Le Henaff, C.; Thorel, J.; Hoang, S.; Jousse, S.; Baron, D.; et al. Value of antibodies to citrulline-containing peptides for diagnosing early rheumatoid arthritis. J. Rheumatol. 2003, 30, 2535–2539. [Google Scholar] [PubMed]
- Vander Cruyssen, B.; Noqueira, L.; Van Praet, J.; Deforce, D.; Elewaut, E.; Serre, G.; De Keyser, F. Do all anti-citrullinated protein/peptide antibody tests measure the same? Evaluation of discrepancy between anti-citrullinated protein/peptide antibody tests in patients with and without rheumatoid arthritis. Ann. Rheum. Dis. 2008, 67, 542–546. [Google Scholar] [CrossRef] [PubMed]
- Vittecoq, O.; Incaurgarat, B.; Jouen-Beades, F.; Legoedec, J.; Letourneur, O.; Rolland, D.; Gervasi, G.; Menard, J.; Gayet, A.; Fardellone, P.; et al. Autoantibodies recognizing citrullinated rat filaggrin in an ELISA using citrullinated and non-citrullinated recombinant proteins as antigens are highly diagnostic for rheumatoid arthritis. Clin. Exp. Immunol. 2004, 135, 173–180. [Google Scholar] [CrossRef] [PubMed]
- Trier, N.H.; Holm, B.E.; Heiden, J.; Slot, O.; Lindegaard, H.; Svendsen, A.; Tandrup Nielsen, C.; Jacobsen, S.; Theander, E.; Houen, G. Antibodies to a strain-specific citrullinated Epstein-Barr virus peptide diagnoses rheumatoid arthritis. Sci. Rep. 2018, 8, 3684. [Google Scholar] [CrossRef] [PubMed]
- Dam, C.; Houen, G.; Trier, N. The dependency on neighboring amino acids for reactivity of anti-citrullinated protein antibodies to citrullinated proteins. Scand. J. Clin. Lab. Investig. 2016, 76, 417–425. [Google Scholar] [CrossRef]
- Kinloch, A.; Tatzer, V.; Wait, R.; Peston, D.; Lundberg, K.; Donatien, P.; Moyes, D.; Taylor, P.; Venables, P. Identification of citrullinated alpha-enolase as a candidate autoantigen in rheumatoid arthritis. Arthritis Res. Ther. 2005, 7, R1421–R1429. [Google Scholar] [CrossRef] [Green Version]
- Trier, N.; Dam, C.; Olsen, D.; Hansen, P.; Houen, G. Contribution of peptide backbone to anti-citrullinated peptide antibody reactivity. PLoS ONE 2015, 10, e0144707. [Google Scholar] [CrossRef]
- Trier, N.; Leth, M.; Hansen, P.; Houen, G. Cross-reactivity of a human IgG1 anticitrullinated fibrinogen monoclonal antibody to a citrullinated profilaggrin peptide. Protein Sci. 2012, 21, 1929–1941. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fanelli, I.; Rovero, P.; Hansen, P.R.; Frederiksen, J.L.; Houen, G.; Trier, N.H. Specificity of Anti-Citrullinated Protein Antibodies to Citrullinated α-Enolase Peptides as a Function of Epitope Structure and Composition. Antibodies 2021, 10, 27. [Google Scholar] [CrossRef] [PubMed]
- Snir, O.; Widhe, M.; von Spee, C.; Lindberg, J.; Padyukov, L.; Lundberg, K.; Engstrom, A.; Venables, P.; Lundeberg, J.; Holmdahl, R.; et al. Multiple antibody reactivities to citrullinated antigens in sera from patients with rheumatoid arthritis: Association with hla-drb1 alleles. Ann. Rheum. Dis. 2009, 68, 736–743. [Google Scholar] [CrossRef] [PubMed]
- Scherer, H.U.; Häupl, T.; Burmester, G.R. The etiology of rheumatoid arthritis. J. Autoimmun. 2020, 110, 102400. [Google Scholar] [CrossRef] [PubMed]
- Goeb, V.; Dieude, P.; Daveau, R.; Thomas-L’otellie, M.; Jouen, F.; Hau, F.; Boumier, P.; Tron, F.; Gilbert, D.; Fardellone, P.; et al. Contribution of PTPN22 1858T, TNFRII 196R and HLA-shared epitope alleles with rheumatoid factor and anti-citrullinated protein antibodies to very early rheumatoid arthritis diagnosis. Rheumatology 2008, 47, 1208–1212. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Raychaudhuri, S.; Remmers, E.F.; Lee, A.T.; Hacket, R.; Guiducci, C.; Burtt, N.P.; Gianniny, L.; Korman, B.D.; Padyukov, L.; Kurreeman, F.A.S.; et al. Common variants at CD40 and other loci confer risk of rheumatoid arthritis. Nat. Genet. 2008, 40, 1216–1223. [Google Scholar] [CrossRef] [PubMed]
- Szodoray, P.; Szabo, Z.; Kapitany, A.; Gyetvai, A.; Lakos, G.; Szanto, S.; Szucs, G.; Szekanecz, Z. Anti-citrullinated protein/peptide autoantibodies in association with genetic and environmental factors as indicators of disease outcome in rheumatoid arthritis. Autoimmun. Rev. 2010, 9, 140–143. [Google Scholar] [CrossRef] [Green Version]
- de Vries, R. Genetics of rheumatoid arthritis: Time for a change! Curr. Opin. Rheumatol. 2011, 23, 227–232. [Google Scholar] [CrossRef]
- Gregersen, P.; Silver, J.; Winchester, R. The shared epitope hypothesis. An approach to understanding the molecular genetics of susceptibility to rheumatoid arthritis. Arthritis Rheum. 1987, 30, 1205–1213. [Google Scholar] [CrossRef] [PubMed]
- Deane, K.; Demoruelle, M.; Kelmenson, L.; Kelmenson, L.; Kuhn, K.; Norris, J.; Holers, V. Genetic and environmental risk factors for rheumatoid arthritis. Best Pract. Res. Clin. Rheumatol. 2017, 31, 3–18. [Google Scholar] [CrossRef]
- Klareskog, L.; Gregersen, P.; Huizinga, T. Prevention of autoimmune rheumatic disease: State of the art and future perspectives. Ann. Rheum. Dis. 2010, 69, 2062–2066. [Google Scholar] [CrossRef] [PubMed]
- Houen, G.; Trier, N.H. Epstein-Barr Virus and systemic autoimmune diseases. Front. Immunol. 2021, 11, 587380. [Google Scholar] [CrossRef] [PubMed]
- Costenbader, K.; Karlson, E. Epstein–Barr virus and rheumatoid arthritis: Is there a link? Arthritis Res. Ther. 2006, 8, 204. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Westergaard, M.W.; Draborg, A.H.; Troelsen, L.; Jacobsen, S.; Houen, G. Isotypes of Epstein-Barr Virus Antibodies in Rheumatoid Arthritis: Association with Rheumatoid Factors and Citrulline-Dependent Antibodies. BioMed Res. Int. 2015, 2015, 472174. [Google Scholar] [CrossRef] [PubMed]
- Houen, G.; Trier, N.H.; Frederiksen, J.L. Epstein-Barr Virus and multiple sclerosis. Front. Immunol. 2020, 11, 587078. [Google Scholar] [CrossRef] [PubMed]
- Alspaugh, M.; Jensen, F.; Rabin, H.; Tan, E. Lymphocytes transformed by Epstein-Barr virus. Induction of nuclear antigen reactive with antibody in rheumatoid arthritis. J. Exp. Med. 1978, 147, 1018–1027. [Google Scholar] [CrossRef] [Green Version]
- Balandraud, N.; Roudier, J. Epstein-Barr virus and rheumatoid arthritis. Joint Bone Spine 2018, 85, 165–170. [Google Scholar] [CrossRef]
- Alspaugh, M.; Henle, G.; Lennette, E.T.; Henle, W. Elevated levels of antibodies to Epstein-Barr virus antigens in sera and synovial fluids of patients with rheumatoid arthritis. J. Clin. Investig. 1981, 67, 1134–1140. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Valleala, H.; Kauppi, M.; Kouri, V.; Korpela, M. Epstein-Barr virus in peripheral blood is associated with response to rituximab therapy in rheumatoid arthritis patients. Clin. Rheumatol. 2015, 34, 1485–1488. [Google Scholar] [CrossRef]
- Balandraud, N.; Meynard, J.; Auger, I.; Sovran, H.; Mugnier, B.; Reviron, D.; Roudier, J.; Roudier, C. Epstein-Barr virus load in the peripheral blood of patients with rheumatoid arthritis accurate quantification using real-time polymerase chain reaction. Arthritis Rheum. 2003, 48, 1223–1228. [Google Scholar] [CrossRef]
- Trier, N.; Izarzugaza, J.; Chailyan, A.; Marcatili, P.; Houen, G. Human MHC-II with Shared Epitope Motifs Are Optimal Epstein Barr Virus Glycoprotein 42 Ligands—Relation to Rheumatoid Arthritis. Int. J. Mol. Sci. 2018, 19, 317. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Harley, J.B.; Chen, X.; Pujato, M.; Miller, D.; Maddox, A.; Forney, C.; Magnusen, A.; Lynch, A.; Chetal, K.; Yukawa, M.; et al. Transcription factors operate across disease loci, with EBNA2 implicated in autoimmunity. Nat. Genet. 2018, 50, 699–707. [Google Scholar] [CrossRef] [PubMed]
- Trier, N.H.; Houen, G. Changes in future rheumatoid arthritis treatment in the light of Epstein-Barr virus infection. In Translational Autoimmunity; Elsevier: Amsterdam, The Netherlands, 2022; Volume 6, in press. [Google Scholar]
- Trier, N.H.; Hansen, P.R.; Vedeler, C.A.; Somnier, F.E.; Houen, G. Identification of continuous epitopes of HuD antibodies related to paraneoplastic diseases/small cell lung cancer. J. Neuroimmunol. 2012, 243, 25–33. [Google Scholar] [CrossRef] [PubMed]
- Mäde, V.; Els-Heindl, S.; Beck-Sickinger, A.G. Automated solid-phase peptide synthesis to obtain therapeutic peptides. Beilstein J. Org. Chem. 2014, 10, 1197–1212. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fini, A.; Breccia, A. Chemistry by microwaves. Pure Appl. Chem. 1999, 71, 573–579. [Google Scholar] [CrossRef]
- Yang, L.; Tan, D.; Piao, H. Myelin basic protein citrullination in multiple sclerosis: A potential therapeutic target for the pathology. Neurochem. Res. 2016, 41, 1845–1856. [Google Scholar] [CrossRef] [PubMed]
- Deraos, G.; Chatzantoni, K.; Matsoukas, M.T.; Tselios, T.; Deraos, S.; Katsara, M.; Papathanasopoulos, P.; Vynios, D.; Apostolopoulos, V.; Mouzaki, A.; et al. Citrullination of linear and cyclic altered peptide ligands from myelin basic protein (MBP (87-99)) epitope elicits a Th1 polarized response by T cells isolated from multiple sclerosis patients: Implications in triggering disease. J. Med. Chem. 2008, 51, 7834–7842. [Google Scholar] [CrossRef]
- Derksen, V.F.A.M.; Huizinga, T.W.J.; van der Woude, D. The role of autoantibodies in the pathophysiology of rheumatoid arthritis. In Seminars in Immunopathology; Springer: Berlin/Heidelberg, Germany, 2017; Volume 39, pp. 437–446. [Google Scholar]
- Suwannalai, P.; Trouw, L.A.; Toes, R.E.M.; Huizinga, T.W.J. Anti-citrullinated protein antibodies (ACPA) in early rheumatoid arthritis. Mod. Rheumatol. 2012, 22, 15–20. [Google Scholar] [CrossRef] [PubMed]
- Ioan-Facsinay, A.; el-Bannoudi, H.; Scherer, H.U.; van der Woude, D.; Ménard, H.A.; Lora, M.; Trouw, L.A.; Huizinga, T.W.; Toes, R.E. Anti-cyclic citrullinated peptide antibodies are a collection of anti-citrullinated protein antibodies and contain overlapping and non-overlapping reactivities. Ann. Rheum. Dis. 2011, 70, 188–193. [Google Scholar] [CrossRef] [Green Version]
- Svendsen, A.J.; Westergaard, M.C.W.; Draborg, A.H.; Holst, R.; Kyvik, K.O.; Jakobsen, M.A.; Junker, P.; Houen, G. Altered antibody response to epstein-barr virus in patients with rheumatoid arthritis and healthy subjects predisposed to the disease. A twin study. Front. Immunol. 2021, 12, 650713. [Google Scholar] [CrossRef]
- Cho, J.; Pyo, J.Y.; Fadriquela, A.; Uh, Y.; Lee, J.-H. Comparison of the analytical and clinical performances of four anti-cyclic citrullinated peptide antibody assays for diagnosing rheumatoid arthritis. Clin. Rheumatol. 2021, 40, 565–573. [Google Scholar] [CrossRef] [PubMed]
- Correia, M.L.; Carvalho, S.; Fortuna, J.; Pereira, M.H. Comparison of three anti-CCP antibody tests and rheumatoid factor in RA and control patients. Clin. Rev. Allergy Immunol. 2008, 34, 21–25. [Google Scholar] [CrossRef] [PubMed]
- van der Helm-van Mil, A.H.M.; Verpoort, K.N.; Breedveld, F.C.; Toes, R.E.M.; Huizinga, T.W.J. Antibodies to citrullinated proteins and differences in clinical progression of rheumatoid arthritis. Arthritis Res. Ther. 2005, 7, R949–R958. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Klareskog, L.; Padyukov, L.; Lorentzen, J.; Alfredsson, L. Mechanisms of disease: Genetic susceptibility and environmental triggers in the development of rheumatoid arthritis. Nat. Clin. Pract. Rheumatol. 2006, 2, 425–433. [Google Scholar] [CrossRef] [PubMed]
- Kuhn, K.; Kulik, L.; Tomooka, B.; Braschler, K.; Arend, W.; Robinson, W.; Holers, V. Antibodies against citrullinated proteins enhance tissue injury in experimental autoimmune arthritis. J. Clin. Investig. 2006, 116, 961–973. [Google Scholar] [CrossRef] [PubMed]
- Trouw, L.; Haisma, E.; Levarht, E.; van der Woude, D.; Ioan-Fascinay, A.; Daha, M.; Huizinga, T.; Toes, R. Anti-cyclic citrullinated peptide antibodies from rheumatoid arthritis patients activate complement via both the classical and alternative pathways. Arthritis Rheum. 2009, 60, 1923–1931. [Google Scholar] [CrossRef]
- Bax, M.; van Heemst, J.; Huizinga, T.W.J.; Toes, R.E.M. Genetics of rheumatoid arthritis: What have we learned? Immunogenetics 2011, 63, 459–466. [Google Scholar] [CrossRef] [Green Version]
- Vassalo, R.; Luckey, D.; Behrens, M.; Madden, B.; Luthra, H.; David, C.; Taneja, V. Cellular and humoral immunity in arhritis are profoundly influenced by the interaction between cigarette smoke effect effects and host HLA-DR and DQ genes. Clin. Immunol. 2014, 152, 25–35. [Google Scholar] [CrossRef] [Green Version]
- Kallberg, H.; Padyukov, L.; Plenge, R.; Ronnelid, J.; Gregersen, P.; van der Helm-van Mil, A.; Toes, R.; Huizinga, T.; Klareskog, L.; Alfredsson, L. Epidemiological Investigation of Rheumatoid Arthritis study group. Gene-gene and gene-environment interactions involving HLA-DRB1, PTPN22, and smoking in two subsets of Rheumatoid Arthritis. Am. J. Hum. Genet. 2007, 80, 867–875. [Google Scholar] [CrossRef] [Green Version]

| Origin | Name | Sequence |
|---|---|---|
| EBNA1 | EBNA1-A | A-Cit-GGSRERARGRGRGRGEKR |
| EBNA1-B | ARGGSRERARGRGRG-Cit-GEKR | |
| EBNA1-C | GGSKTSLYNLR-Cit-GTALAIPQ | |
| EBNA2 | EBNA2-A | GQGRGRWRG-Cit-GRSKGRGRMH |
| EBNA2-B | GQSRGRGRG-Cit-GRGRGKGKSR | |
| EBNA2-C | KQGPDQGQG-Cit-GRWRGRGRSK | |
| NCit-EBNA2 | G-Cit-GSKGRGRMHKLPEPRRPGPD | |
| EBNA3 | EBNA3-A | EPDSRDQQC-Cit-GQRRGDENRG |
| EBNA3-B | PNENPYHAR-Cit-GIKEHVIQNA | |
| EBNA3-C | DQLPGVPKG-Cit-GACAPVPALA | |
| EBNA3-D | EDAHLEPSQ-Cit-GKKRKRVDDD | |
| EBNA3-E | AQAWNAGLL-Cit-GRAYGQDLL | |
| EBNA2-EBNA3 | GQGRGRWRG-Cit-GQRRGDENRG | |
| EBNA2-EBNA2 | EPDSRDQQS-Cit-GRSKGRGRMH | |
| MBP | MBP1 | KASTNSETN-Cit-GESEKKRNLG |
| MBP2 | SIGRFFGGD-Cit-GAPKRGSGKD | |
| MBP3 | FGGDRGAPK-Cit-GSGKDSHHPA | |
| MBP4 | TPPPSQGKG-Cit-GLSLSRFSWG |
| Single Positive | Double Positive | Triple Positive | ||||||
|---|---|---|---|---|---|---|---|---|
| Reactivities | None | EBNA2 | RF IgM | RF IgA | RF IgM/A | EBNA2 + RF IgA | EBNA2 + RF IgM | RF IgA/M + EBNA2 |
| Number (n) | 0 | 19 | 24 | 23 | 23 | 18 | 19 | 17 |
| Percentage (%) | 0 | 76 | 96 | 88 | 88 | 72 | 76 | 68 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Fanelli, I.; Rovero, P.; Hansen, P.R.; Frederiksen, J.L.; Houen, G.; Trier, N.H. Reactivity of Rheumatoid Arthritis-Associated Citrulline-Dependent Antibodies to Epstein-Barr Virus Nuclear Antigen1-3. Antibodies 2022, 11, 20. https://doi.org/10.3390/antib11010020
Fanelli I, Rovero P, Hansen PR, Frederiksen JL, Houen G, Trier NH. Reactivity of Rheumatoid Arthritis-Associated Citrulline-Dependent Antibodies to Epstein-Barr Virus Nuclear Antigen1-3. Antibodies. 2022; 11(1):20. https://doi.org/10.3390/antib11010020
Chicago/Turabian StyleFanelli, Ilaria, Paolo Rovero, Paul Robert Hansen, Jette Lautrup Frederiksen, Gunnar Houen, and Nicole Hartwig Trier. 2022. "Reactivity of Rheumatoid Arthritis-Associated Citrulline-Dependent Antibodies to Epstein-Barr Virus Nuclear Antigen1-3" Antibodies 11, no. 1: 20. https://doi.org/10.3390/antib11010020
APA StyleFanelli, I., Rovero, P., Hansen, P. R., Frederiksen, J. L., Houen, G., & Trier, N. H. (2022). Reactivity of Rheumatoid Arthritis-Associated Citrulline-Dependent Antibodies to Epstein-Barr Virus Nuclear Antigen1-3. Antibodies, 11(1), 20. https://doi.org/10.3390/antib11010020






