Diagnostic Performance of PD-L1 versus PD-1 Expression in Circulating CD20 Cells in Diffuse Large B-Cell Lymphoma
Abstract
1. Introduction
2. Materials and Methods
2.1. Patients
2.2. Clinical Criteria and Laboratory Samples Collection
2.3. Antibodies
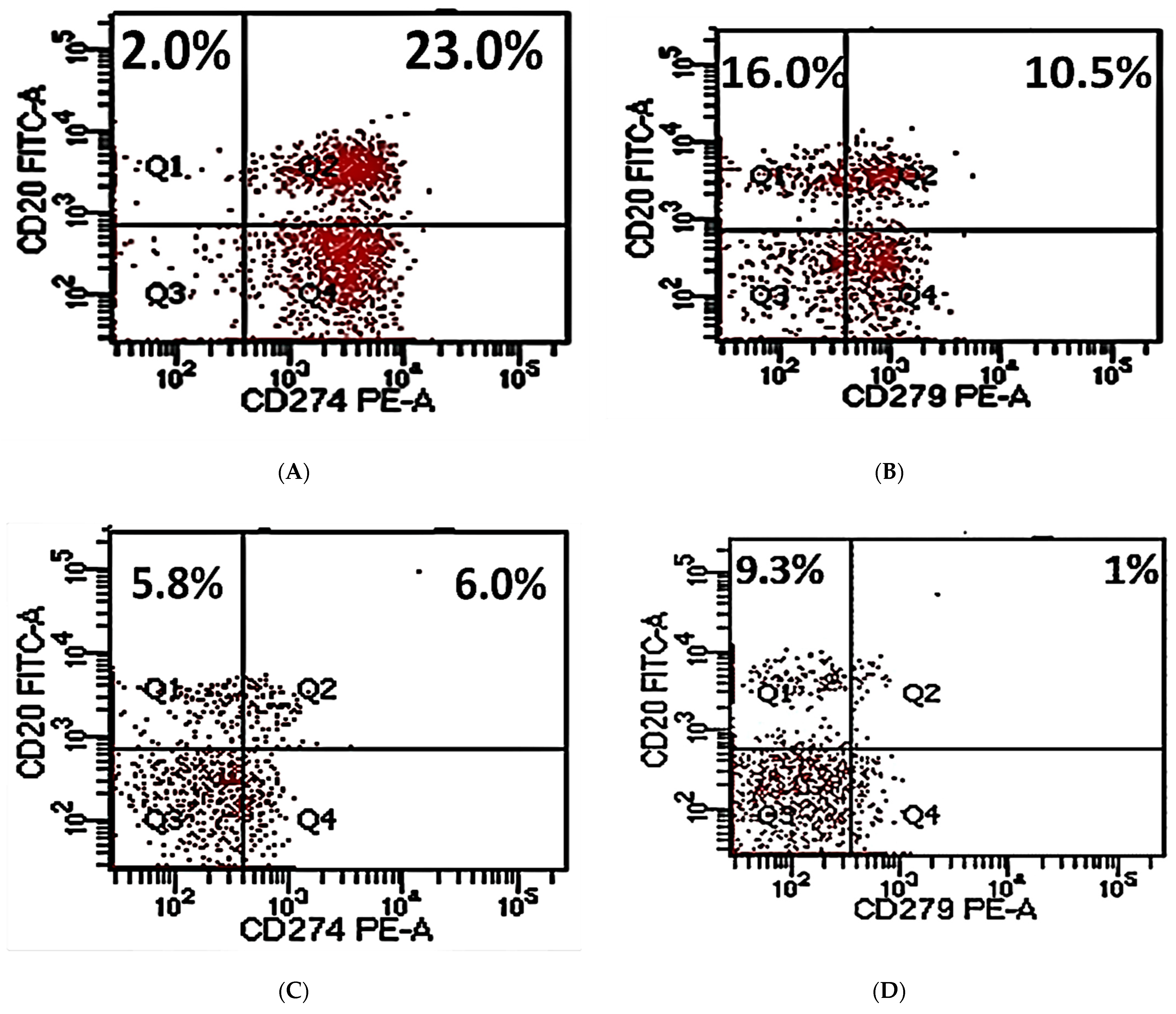
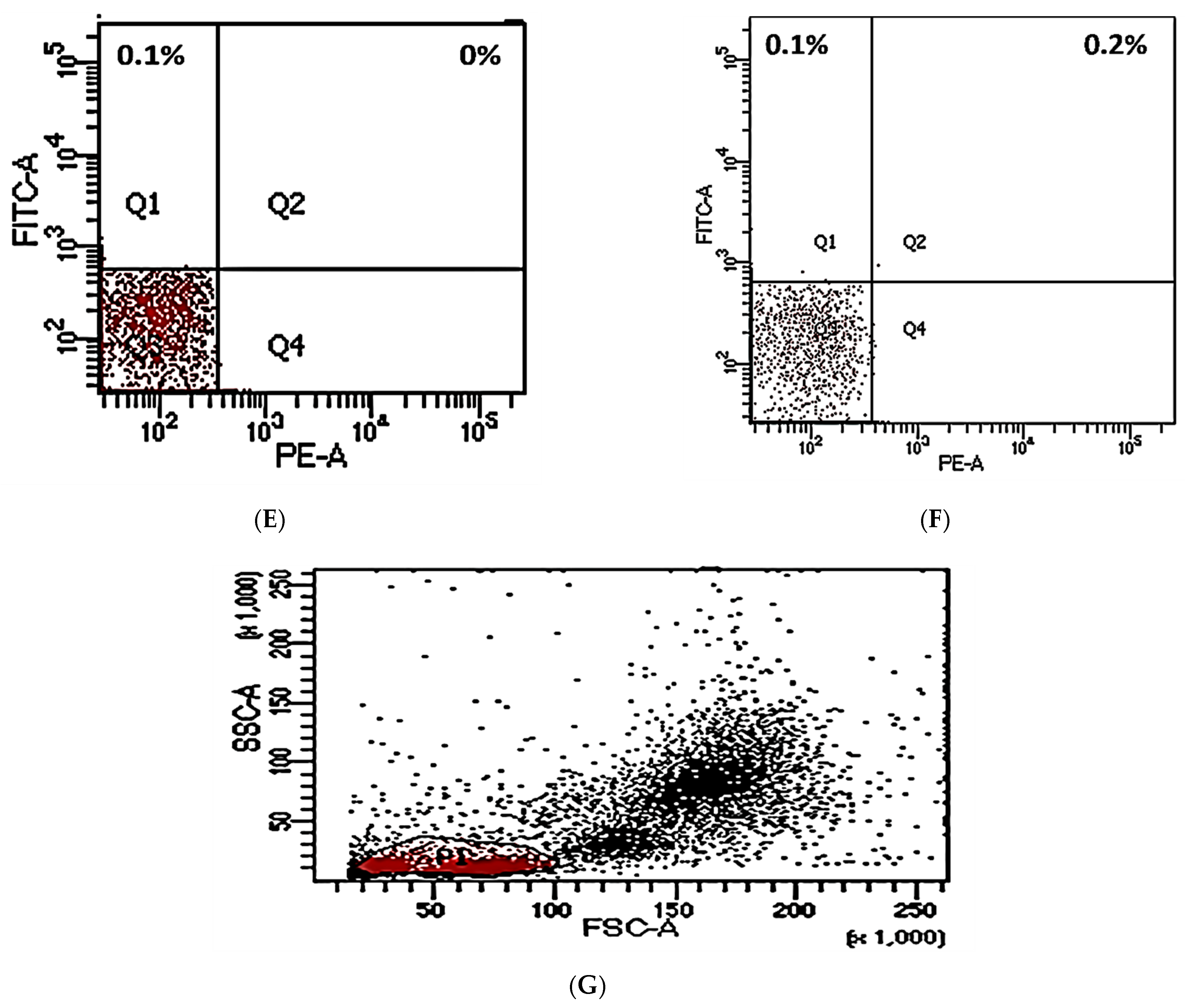
2.4. Flow Cytometry Analysis
2.5. Laboratory Methods
2.6. Statistics
3. Results
3.1. Patients’ Characteristics
3.2. PD-L1/PD-1 Expression in Peripheral CD20+ in DLBCL
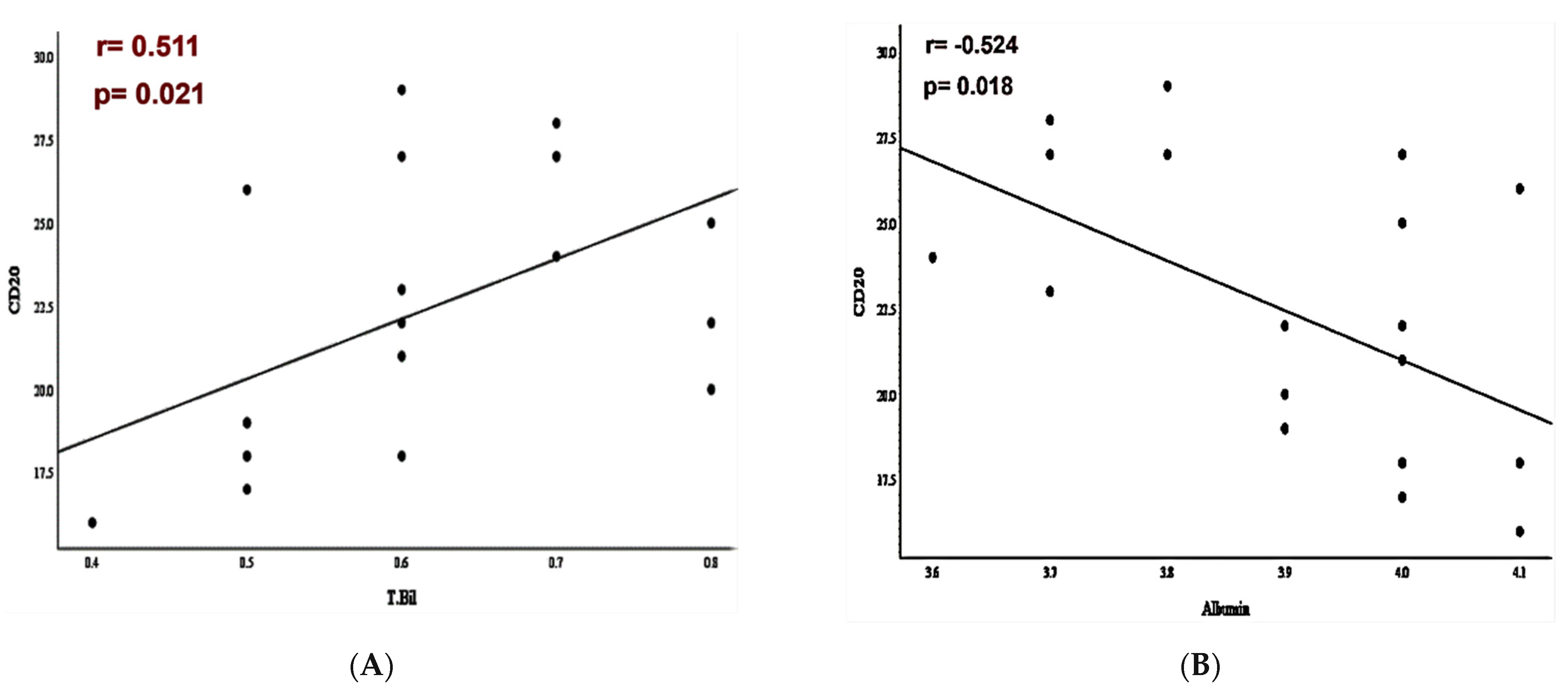
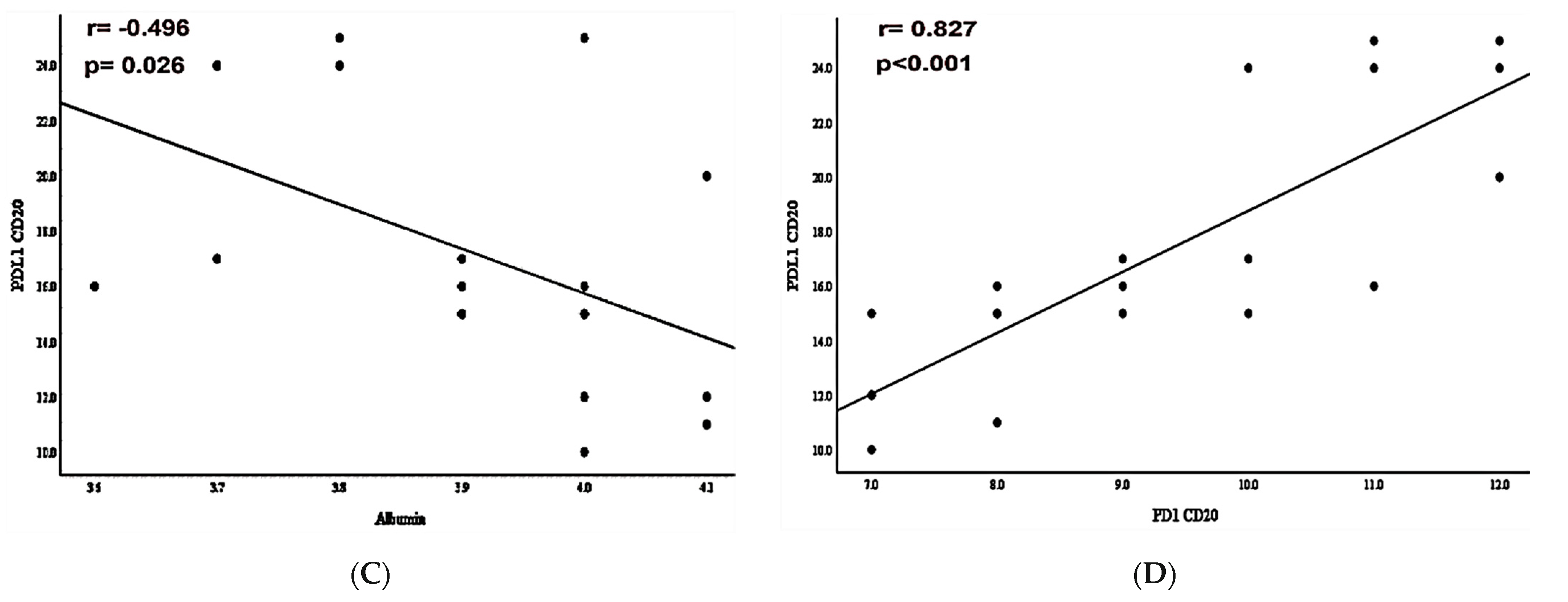
3.3. PD-L1/PD-1 Expression and Laboratory-Systemic Data
3.4. Clinicopathological Analysis and PD-L1/PD-1 Expressions in DLBCL
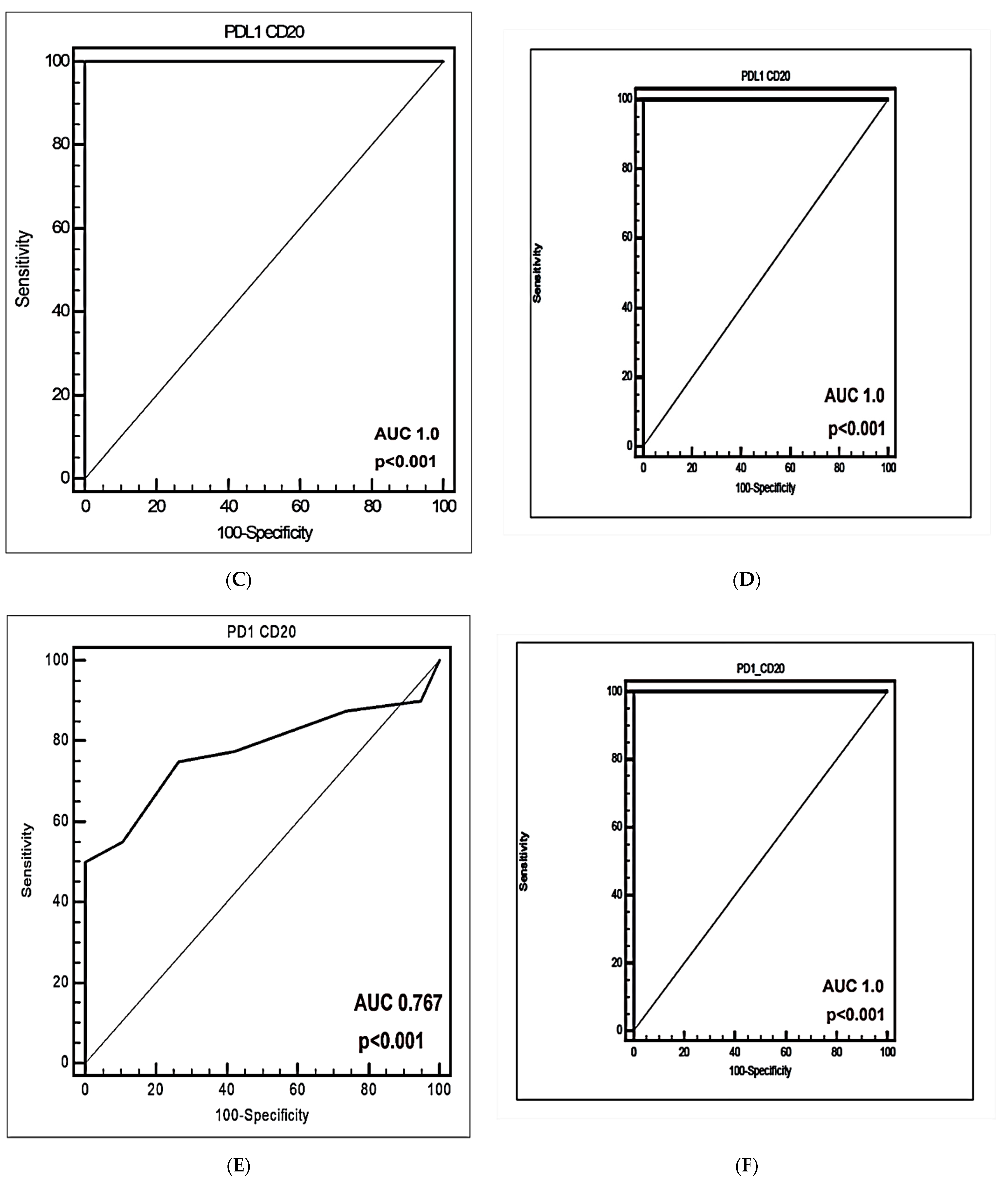
3.5. The Predictive and Diagnostic Efficacies of Circulating PD-L1+CD20+ and PD-1+CD20+ Cells
4. Discussion
5. Study Limitations
6. Conclusions
Supplementary Materials
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Swerdlow, S.H.; Campo, E.; Harris, N.L.; Jaffe, E.S.; Pileri, S.A.; Stein, H.; Thiele, J. WHO Classification of Tumours of Haematopoietic and Lymphoid Tissues; Revised, 4th ed.; International Agency for Research on Cancer: Lyon, France, 2017; Volume 2, pp. 190–198. [Google Scholar]
- Siegel, R.L.; Miller, K.D.; Jemal, A. Cancer statistics. CA Cancer J. Clin. 2017, 67, 7–30. [Google Scholar] [CrossRef] [PubMed]
- Armitage, J.O.; Gascoyne, R.D.; Lunning, M.A.; Cavalli, R. Non-Hodgkin lymphoma. Lancet 2017, 390, 298–310. [Google Scholar]
- Lodhi, N.; Tun, M.; Nagpal, P.; Inamdar, A.; Ayoub, N.M.; Siyam, N.; Oton-Gonzalez, L.; Gerona, A.; Morris, D.; Sandhu, R.; et al. Biomarkers and novel therapeutic approaches for diffuse large B-cell lymphoma in the era of precision medicine. Oncotarget 2020, 11, 4045–4073. [Google Scholar] [CrossRef] [PubMed]
- Alizadeh, A.A.; Eisen, M.B.; Davis, R.E.; Ma, V.; Lossos, I.S.; Rosenwald, A.; Boldrick, J.C.; Sabet, H.; Tran, T.; Yu, X.; et al. Distinct types of diffuse large B-cell lymphoma identified by gene expression profiling. Nature 2000, 403, 503–511. [Google Scholar]
- Read, J.A.; Koff, J.L.; Nastoupil, L.J.; Williams, J.N.; Cohen, J.B.; Flowers, C.R. Evaluating cell-of-origin subtype methods for predicting diffuse large B-cell lymphoma survival: A meta-analysis of gene expression profiling and immunohistochemistry algorithms. Clin. Lymphoma Myeloma Leuk. 2014, 14, 460–467. [Google Scholar] [CrossRef]
- McKelvey, E.M.; Gottlieb, J.A.; Wilson, H.E.; Haut, A.; Talley, R.W.; Stephens, R.; Lane, M.; Gamble, J.F.; Jones, S.E.; Grozea, P.N.; et al. Hydroxy daunomycin (Adriamycin) combination chemotherapy in malignant lymphoma. Cancer 1976, 38, 1484–1493. [Google Scholar] [CrossRef]
- Pfreundschuh, M.; Kuhnt, E.; Trümper, L.; Osterborg, A.; Trneny, M.; Shepherd, L.; Gill, D.S.; Walewski, J.; Pettengell, R.; Jaeger, U.; et al. CHOP-like chemotherapy with or without rituximab in young patients with good-prognosis diffuse large-B-cell lymphoma: 6-year results of an open-label randomized study of the MabThera International Trial (MInT) Group. Lancet Oncol. 2011, 12, 1013–1022. [Google Scholar] [CrossRef]
- Friedberg, J.W. Relapsed/refractory diffuse large B-cell lymphoma. Hematol. Am. Soc. Hematol. Educ. Program 2011, 2011, 498–505. [Google Scholar] [CrossRef] [PubMed]
- Vital, E.M.; Dass, S.; Emery, P. B-cell depletion. In Rheumatology, 6th ed.; Hochberg, M.C., Silman, A.J., Smolen, J.S., Eds.; ScienceDirect: Philadelphia, PA, USA, 2015; pp. 472–478. [Google Scholar]
- Middleton, O.; Wheadon, H.; Michie, A.M. Classical complement pathway. In Encyclopedia of Immunobiology; Ratcliffe, M.J.H., Ed.; Oxford Academic Press: New York, NY, USA, 2016; pp. 318–324. [Google Scholar]
- Clark, E.A.; Shu, G.; Ledbetter, J.A. Role of the Bp35 cell surface polypeptide in human B-cell activation. Proc. Natl. Acad. Sci. USA 1985, 82, 1766–1770. [Google Scholar] [CrossRef]
- Golay, J.T.; Clark, E.A.; Beverley, P.C. The CD20 (Bp35) antigen is involved in the activation of B cells from the G0 to the G1 phase of the cell cycle. J. Immunol. 1985, 135, 3795–3801. [Google Scholar] [PubMed]
- Huh, Y.O.; Keating, M.J.; Saffer, H.L.; Jilani, I.; Lerner, S.; Albitar, M. Higher Levels of Surface CD20 Expression on Circulating Lymphocytes Compared with Bone Marrow and Lymph Nodes in B-Cell Chronic Lymphocytic Leukemia. Am. J. Clin. Pathol. 2001, 116, 437–443. [Google Scholar] [CrossRef]
- Küppers, R.; Engert, A.; Hansmann, M.L. Hodgkin lymphoma. J. Clin. Investig. 2012, 122, 3439–3447. [Google Scholar] [CrossRef] [PubMed]
- Olejniczak, S.H.; Stewart, C.C.; Donohue, K.; Czuczman, M.S. A quantitative exploration of surface antigen expression in common B-cell malignancies using flow cytometry. Immunol. Investig. 2006, 35, 93–114. [Google Scholar] [CrossRef]
- Stein, H.; Chan, J.K.C.; Warnke, R.A.; Gatter, K.C.; Chan, W.C.; Campo, E.; Jaffe, E.S. Diffuse large B-cell lymphoma, not otherwise specified. In WHO Classification of Tumors of Hematopoietic and Lymphoid Tissues; Swerdlow, S.H., Campo, E., Harris, N.L., Jaffe, E.S., Pileri, S.A., Stein, H., Thiele, J., Eds.; WHO: Geneva, Switzerland, 2008; pp. 233–237. [Google Scholar]
- Castillo, J.J.; Chavez, J.C.; Hernandez-Ilizaliturri, F.J.; Montes-Moreno, S. CD20-negative diffuse large B-cell lymphomas: Biology and emerging therapeutic options. Expert Rev. Hematol. 2015, 8, 343–354. [Google Scholar] [CrossRef] [PubMed]
- Montes-Moreno, S.; Montalban, C.; Piris, M.A. Large B-cell lymphomas with plasmablastic differentiation: A biological and therapeutic challenge. Leuk. Lymphoma 2012, 53, 185–194. [Google Scholar] [CrossRef]
- His, E.D.; Lorsbach, R.B.; Fend, F.; Dogan, A. Plasmablastic lymphoma and related disorders. Am. J. Clin. Pathol. 2011, 136, 183–194. [Google Scholar]
- Laurent, C.; Do, C.; Gascoyne, R.D.; Lamant, L.; Ysebaert, L.; Laurent, G.; Delsol, G.; Brousset, P. Anaplastic lymphoma kinase-positive diffuse large B-cell lymphoma: A rare clinicopathologic entity with poor prognosis. J. Clin. Oncol. 2009, 27, 4211–4216. [Google Scholar] [CrossRef]
- Feugier, P.; Van Hoof, A.; Sebban, C.; Solal-Celigny, P.; Bouabdallah, R.; Fermé, C.; Christian, B.; Lepage, E.; Tilly, H.; Morschhauser, F.; et al. Long-term results of the R-CHOP study in the treatment of elderly patients with diffuse large B-cell lymphoma: A study by the Groupe d’ Etude des Lymphomes de l’ Adulte. J. Clin. Oncol. 2005, 23, 4117–4126. [Google Scholar] [CrossRef]
- Pfreundschuh, M.; Trümper, L.; Osterborg, A.; Pettengell, R.; Trneny, M.; Imrie, K.; Ma, D.; Gill, D.; Walewski, J.; Zinzani, P.L.; et al. International Trial Group: CHOP-like chemotherapy plus rituximab versus CHOP-like chemotherapy alone in young patients with good-prognosis diffuse large-B-cell lymphoma: A randomised controlled trial by the MabThera International Trial (MinT) Group. Lancet Oncol. 2006, 7, 379–391. [Google Scholar] [CrossRef]
- Habermann, T.M.; Weller, E.A.; Morrison, V.A.; Gascoyne, R.D.; Cassileth, P.A.; Cohn, J.B.; Dakhil, S.R.; Woda, B.; Fisher, R.I.; Peterson, B.A.; et al. Rituximab-CHOP versus CHOP alone or with maintenance rituximab in older patients with diffuse large B-cell lymphoma. J. Clin. Oncol. 2006, 24, 3121–3127. [Google Scholar] [CrossRef] [PubMed]
- Pfreundschuh, M.; Schubert, J.; Ziepert, M.; Schmits, R.; Mohren, M.; Lengfelder, E.; Reiser, M.; Nickenig, C.; Clemens, M.; Peter, N.; et al. German High-Grade Non-Hodgkin Lymphoma Study Group (DSHNHL): Six versus eight cycles of bi-weekly CHOP-14 with or without rituximab in elderly patients with aggressive CD20+ B-cell lymphomas: A randomized controlled trial (RICOVER-60). Lancet Oncol. 2008, 9, 105–116. [Google Scholar] [CrossRef]
- Rassidakis, G.Z.; Medeiros, J.; Viviani, S.; Bonfante, V.; Nadali, G.P.; Vassilakopoulos, T.P.; Mesina, O.; Herling, M.; Angelopoulou, M.K.; Giardini, R.; et al. CD20 expression in Hodgkin and Reed-Sternberg cells of classical Hodgkin’s disease: Associations with presenting features and clinical outcome. J. Clin. Oncol. 2002, 20, 1278–1287. [Google Scholar] [PubMed]
- Cohen, Y.; Solal-Celigny, P.; Polliack, A. Rituximab therapy for follicular lymphoma: A comprehensive review of its efficacy as primary treatment, treatment for relapsed disease, retreatment, and maintenance. Haematologica 2003, 88, 811–823. [Google Scholar] [PubMed]
- Coiffier, B. Rituximab and CHOP-like chemotherapy in good-prognosis diffuse large-B-cell lymphoma. Nat. Clin. Pract. Oncol. 2006, 3, 594–595. [Google Scholar] [CrossRef] [PubMed]
- Coiffier, B.; Thieblemont, C.; Van Den Neste, E.; Lepeu, G.; Plantier, I.; Castaigne, S.; Lefort, S.; Marit, G.; Macro, M.; Sebban, C.; et al. Long-term outcome of patients in the LNH-98.5 trial, the first randomized study comparing rituximab-CHOP to standard CHOP chemotherapy in DLBCL patients: A study by the Groupe d’Etudes des Lymphomes de l’Adulte. Blood 2010, 116, 2040–2045. [Google Scholar] [CrossRef] [PubMed]
- Atchi, T.; Liu, D. Diagnosis and treatment of CD20 negative B cell lymphomas. Biomark Res. 2017, 16, 1–5. [Google Scholar]
- Sanmamed, M.F.; Chen, L. Inducible expression of B7-H1 (PD-L1) and its selective role in tumor site immune modulation. Cancer J. 2004, 20, 256–261. [Google Scholar] [CrossRef] [PubMed]
- Freeman, G.J.; Long, A.J.; Iwai, Y.; Bourque, K.; Chernova, T.; Nishimura, H.; Fitz, L.J.; Malenkovich, N.; Okazaki, T.; Byrne, M.C.; et al. Engagement of the PD-1 immunoinhibitory receptor by a novel B7 family member leads to negative regulation of lymphocyte activation. J. Exp. Med. 2000, 192, 1027–1034. [Google Scholar] [CrossRef] [PubMed]
- Blank, C.; Gajewski, T.F.; Mackensen, A. Interaction of PD-L1 on tumor cells with PD-1 on tumor-specific T cells as a mechanism of immune evasion: Implications for tumor immunotherapy. Cancer Immunol. Immunother. 2005, 54, 307–314. [Google Scholar]
- Sharpe, A.H.; Wherry, E.J.; Ahmed, R.; Freeman, G.J. The function of programmed cell death 1 and its ligands in regulating autoimmunity and infection. Nat. Immunol. 2007, 8, 239–245. [Google Scholar] [PubMed]
- He, J.; Hu, Y.; Hu, M.; Li, B. Development of PD-1/PD-L1 pathway in tumor immune microenvironment and treatment for non-small cell lung cancer. Sci. Rep. 2015, 5, 13110. [Google Scholar]
- Ishida, M.; Iwai, Y.; Tanaka, Y.; Okazaki, T.; Freeman, G.J.; Minato, N.; Honjo, T. Differential expression of PD-L1 and PD-L2, ligands for an inhibitory receptor PD-1, in the cells of lymphohematopoietic tissues. Immunol. Lett. 2002, 84, 57–62. [Google Scholar] [CrossRef]
- Andorsky, D.J.; Yamada, R.E.; Said, J.; Pinkus, G.S.; Betting, D.J.; Timmerman, J.M. Programmed death-ligand 1 is expressed by non-Hodgkin lymphomas and inhibits the activity of tumor-associated T cells. Clin. Cancer. Res. 2011, 17, 4232–4244. [Google Scholar] [CrossRef] [PubMed]
- Kiyasu, J.; Miyoshi, H.; Hirata, A.; Arakawa, F.; Ichikawa, A.; Niino, D.; Sugita, Y.; Yufu, Y.; Choi, I.; Abe, Y.; et al. Expression of programmed cell death ligand 1 is associated with poor overall survival in patients with diffuse large B-cell lymphoma. Blood 2015, 126, 2193–2201. [Google Scholar] [CrossRef] [PubMed]
- Hu, L.Y.; Xu, X.L.; Rao, H.L.; Chen, J.; Lai, R.C.; Huang, H.Q.; Jiang, W.-Q.; Lin, T.-Y.; Xia, Z.-J.; Cai, Q.-Q. Expression and clinical value of programmed cell death-ligand 1 (PD-L1) in diffuse large B cell lymphoma: A retrospective study. Chin. J. Cancer 2017, 36, 94. [Google Scholar] [PubMed]
- Fang, X.; Xiu, B.; Yang, Z.; Qiu, W.; Zhang, L.; Zhang, S.; Wu, Y.; Zhu, X.; Chen, X.; Xie, S.; et al. The expression and clinical relevance of PD-1, PD-L1, and TP63 in patients with diffuse large B-cell lymphoma. Medicine 2017, 96, e6398. [Google Scholar] [CrossRef] [PubMed]
- Gatalica, Z.; Bilalovic, N.; Vranic, S.; Arguello, D.; Reddy, S.; Ghosh, N. PD-L1 and PD1 expression in lymphomas. Blood 2015, 126, 3899. [Google Scholar] [CrossRef]
- Armand, P. Immune checkpoint blockade in hematologic malignancies. Blood 2015, 125, 3393–3400. [Google Scholar] [CrossRef] [PubMed]
- Goodman, A.; Patel, S.P.; Kurzrock, R. PD-1–PD-L1 immune-checkpoint blockade in B-cell lymphomas. Nat. Rev. Clin. Oncol. 2016, 14, 203. [Google Scholar]
- Non-Hodgkin Lymphoma Treatment; American Cancer Society: Kennesaw, GA, USA, 2019.
- Swerdlow, S.H.; Campo, E.; Pileri, S.A.; Harris, N.L.; Stein, H.; Siebert, R.; Advani, R.; Ghielmini, M.; Salles, G.A.; Zelenetz, A.D.; et al. The 2016 Revision of the World Health Organization Classification of Lymphoid Neoplasms. Blood 2016, 127, 2375–2390. [Google Scholar] [PubMed]
- Hans, C.P.; Weisenburger, D.D.; Greiner, T.C.; Gascoyne, R.D.; Delabie, J.; Ott, G.; Müller-Hermelink, H.K.; Campo, E.; Braziel, R.M.; Jaffe, E.S.; et al. Confirmation of the molecular classification of diffuse large B-cell lymphoma by immunohistochemistry using a tissue microarray. Blood 2004, 103, 275–282. [Google Scholar] [CrossRef] [PubMed]
- Jang, R.W.; Caraiscos, V.B.; Swami, N.; Banerjee, S.; Mak, E.; Kaya, E.; Rodin, G.; Bryson, J.; Ridley, J.Z.; Le, L.W.; et al. Simple prognostic model for patients with advanced cancer based on performance status. J. Politics 2014, 10, e335–e341. [Google Scholar] [CrossRef] [PubMed]
- Johnson, S.A.; Kumar, A.; Matasar, M.J.; Schöder, H.; Rademaker, J. Imaging for staging and response assessment in lymphoma. Radiology 2015, 276, 323–338. [Google Scholar] [CrossRef] [PubMed]
- Brown Ja Dorfman, D.M.; Ma, F.-R.; Sullivan, E.L.; Munoz, O.; Wood, C.R.; Greenfield, E.A.; Freeman, G.J. Blockade of Programmed Death-1 Ligands on Dendritic Cells Enhances T Cell Activation and Cytokine Production. J. Immunol. 2003, 170, 1257. [Google Scholar] [CrossRef] [PubMed]
- Nakamoto, N.; Cho, A.; Shaked, A.; Olthoff, K.; Valiga, M.E.; Kaminski, M.; Gostick, E.; Price, D.A.; Freeman, G.J.; Wherry, E.J.; et al. Synergistic Reversal of Intrahepatic HCV-Specific CD8 T Cell Exhaustion by Combined PD-1/CTLA-4 Blockade. PLoS Pathog. 2009, 5, e100031. [Google Scholar] [CrossRef] [PubMed]
- Velu, V.; Kannanganat, S.; Ibegbu, C.; Chennareddi, L.; Villinger, F.; Freeman, G.J.; Ahmed, R.; Amara, R.R. Elevated expression levels of inhibitory receptor programmed death 1 on simian immunodeficiency virus-specific CD8 T cells during chronic infection but not after vaccination. J. Virol. 2007, 81, 5819. [Google Scholar] [CrossRef]
- Li, H.; Pauza, C.D. CD25(+) Bcl6(low) T follicular helper cells provide help to maturing B cells in germinal centers of human tonsil. Eur. J. Immunol. 2015, 45, 298. [Google Scholar] [CrossRef]
- Polyak, M.J.; Deans, J.P. Alanine-170 and proline-172 are critical determinants for extracellular CD20 epitopes; heterogeneity in the fine specificity of CD20 monoclonal antibodies is defined by additional requirements imposed by both amino acid sequence and quaternary structure. Blood 2002, 99, 3256. [Google Scholar] [CrossRef] [PubMed]
- Mack, C.L.; Tucker, R.M.; Sokol, R.J.; Karrer, F.M.; Kotzin, B.L.; Whitington, P.F.; Miller, S.D. Biliary atresia is associated with CD4+ Th1 cell-mediated portal tract inflammation. Pediatr. Res. 2004, 56, 79. [Google Scholar] [CrossRef] [PubMed]
- Hawkes, E.A.; Grigg, A.; Chong, G. Programmed cell death-1 inhibition in lymphoma. Lancet Oncol. 2015, 16, e234–e245. [Google Scholar] [PubMed]
- Madore, J.; Vilain, R.E.; Menzies, A.M.; Kakavand, H.; Wilmott, J.S.; Hyman, J.; Yearley, J.H.; Kefford, R.F.; Thompson, J.F.; Long, G.V.; et al. PD-L1 expression in melanoma shows marked heterogeneity within and between patients: Implications for anti—PD. 1/PD-L1 clinical trials. Pigment. Cell Melanoma Res. 2014, 28, 245–253. [Google Scholar] [CrossRef]
- Liu, D.; Wang, S.; Bindeman, W. Clinical applications of PD-L1 bioassays for cancer immunotherapy. J. Hematol. Oncol. 2017, 10, 110. [Google Scholar] [CrossRef] [PubMed]
- Diggs, L.P.; Hsueh, E.C. Utility of PD-L1 immunohistochemistry assays for predicting PD-1/PD-L1 inhibitor response. Biomark Res. 2017, 5, 12. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Wang, G.; Wang, Z.; Liu, B.; Han, N.; Li, J. PD-1-expressing B cells suppress CD4+ and CD8+ T cells via PD-1/PD-L1- dependent pathway. Mol. Immunol. 2019, 109, 20–26. [Google Scholar] [CrossRef] [PubMed]
- Xiao, X.; Lao, X.-M.; Chen, M.-M.; Liu, R.-X.; Wei, Y.; Ouyang, F.-Z. PD-1hi identifies a novel regulatory B-cell population in human hepatoma that promotes disease progression. Cancer Discov. 2016, 6, 546–559. [Google Scholar] [CrossRef] [PubMed]
- Khan, A.R.; Hams, E.; Floudas, A.; Sparwasser, T.; Weaver, C.T.; Fallon, P.G. PDL1hi B cells are critical regulators of humoral immunity. Nat. Commun. 2015, 6, 5997. [Google Scholar] [CrossRef] [PubMed]
- Howitt, B.E.; Shukla, S.A.; Sholl, L.M.; Ritterhouse, L.L.; Watkins, J.C.; Rodig, S.; Stover, E.; Strickland, K.C.; D’Andrea, A.D.; Wu, C.J. Association of polymerase e-mutated and microsatellite-instable endometrial cancers with neoantigen load, number of tumor—Infiltrating lymphocytes, and expression of PD-1 and PD-L1. JAMA Oncol. 2015, 1, 1319–1323. [Google Scholar] [CrossRef] [PubMed]
- Brahmer, J.R.; Tykodi, S.S.; Chow, L.Q.; Hwu, W.J.; Topalian, S.L.; Hwu, P.; Drake, C.G.; Camacho, L.H.; Kauh, J.; Odunsi, K.; et al. Safety and activity of anti-PD-L1 antibody in patients with advanced cancer. N. Engl. J. Med. 2012, 366, 2455–2465. [Google Scholar] [CrossRef]
- Ma, X.J.; Zhang, F.H.; Sun, L. Expression and significance of PDL-1, HSP90, and HSP90 α in serum of patients with acute leukemia. Zhongguo Shi Yan Xue Ye Xue Za Zhi 2017, 25, 1384–1389. [Google Scholar]
- Herbst, R.S.; Baas, P.; Kim, D.W.; Felip, E.; Pérez-Gracia, J.L.; Han, J.Y.; Molina, J.; Kim, J.H.; Arvis, C.D.; Ahn, M.-J.; et al. Pembrolizumab versus docetaxel for previously treated, PD-L1-positive, advanced non-small-cell lung cancer (KEYNOTE-010): A randomized controlled trial. Lancet 2016, 387, 1540–1550. [Google Scholar] [CrossRef]
- Mataki, N.; Kikuchi, K.; Kawai, T.; Higashiyama, M.; Okada, Y.; Kurihara, C.; Hokari, R.; Kawaguchi, A.; Nagao, S.; Kondo, T.; et al. Expression of PD-1, PD-L1, and PD-L2 in the liver in autoimmune liver diseases. Am. J. Gastroenterol. 2007, 102, 302–312. [Google Scholar] [CrossRef]
- Fei, Y.; Yu, J.; Li, Y.; Li, L.; Zhou, S.; Zhang, T.; Li, L.; Qiu, L.; Meng, B.; Pan, Y.; et al. Plasma soluble PD-L1 and STAT3 predict the prognosis in diffuse large B cell lymphoma patients. J. Cancer 2020, 11, 7001–7008. [Google Scholar] [CrossRef]
- Zhang, W.; Bai, J.F.; Zuo, M.-X.; Cao, X.-X.; Chen, M.; Zhang, Y.; Han, X.; Zhong, D.-R.; Zhou, D.-B. PD-1 expression on the surface of peripheral blood CD4(+) T cell and its association with the prognosis of patients with diffuse large B-cell lymphoma. Cancer Med. 2016, 5, 3077–3084. [Google Scholar] [CrossRef] [PubMed]
- Gravelle, P.; Burroni, B.; Péricart, S.; Rossi, C.; Bezombes, C.; Tosolini, M.; Damotte, D.; Brousset, P.; Fournié, J.-J.; Laurent, C. Mechanisms of PD-1/PD-L1 expression and prognostic relevance in non-Hodgkin lymphoma: A summary of immunohistochemical studies. Oncotarget 2017, 8, 44960–44975. [Google Scholar] [CrossRef] [PubMed]
- Xu-Monette, Z.Y.; Zhou, J.; Young, K.H. PD-1 expression and clinical PD-1 blockade in B-cell lymphomas. Blood 2018, 131, 68–83. [Google Scholar]
- Montgomery, N.D.; Fedoriw, Y. Pathology consultation on intermediate-to-large B-cell lymphomas. Am. J. Clin. Pathol. 2014, 141, 305–317. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Laurent, C.; Charmpi, K.; Gravelle, P.; Tosolini, M.; Franchet, C.; Ysebaert, L.; Brousset, P.; Bidaut, A.; Ycart, B.; Fournie, J.J. Several immune escape patterns in non-Hodgkin’s lymphomas. Oncoimmunology 2015, 4, e1026530. [Google Scholar] [CrossRef] [PubMed]
- Georgiou, K.; Chen, L.; Berglund, M.; Ren, W.; de Miranda, N.F.; Lisboa, S.; Fangazio, M.; Zhu, S.; Hou, Y.; Wu, K. Genetic basis of PD-L1 overexpression in diffuse large B-cell lymphomas. Blood 2016, 127, 3026–3034. [Google Scholar] [PubMed]
- Siddiqi, I.N.; Thodima, V.; Friedman, J.; Violeta, A.; Tulpule, A.; Shaknovich, R.; Houldsworth, J. PD-L1 expression identifies high risk diffuse large B-cell lymphoma and is associated with several genomic markers. Blood 2016, 128, 153. [Google Scholar] [CrossRef]
- Kleinovink, J.W.; Marijt, K.A.; Schoonderwoerd, M.J.A.; van Hall, T.; Ossendorp, F.; Fransen, M.F. PD-L1 expression on malignant cells is no prerequisite for checkpoint therapy. Oncoimmunology 2017, 6, e1294299. [Google Scholar] [CrossRef]





| KERRYPNX | Control | DLBCL | p-Value | |
|---|---|---|---|---|
| N = 19 | N = 40 | |||
| Age | Median | 45 | 46.5 | 0.955 |
| IQR | (30–57) | (34–55.8) | ||
| Sex | Male | 9 (47.4%) | 20 (50%) | 0.850 |
| Female | 10 (52.6%) | 20 (50%) | ||
| Hepatomegaly | No | 19 (100%) | 20 (50%) | <0.001 ** |
| Yes | 0 (0%) | 20 (50%) | ||
| Splenomegaly | No | 19 (100%) | 24 (60%) | <0.001 ** |
| Yes | 0 (0%) | 16 (40%) | ||
| Treatment | None | 20 (50%) | ||
| CHOP | 20 (50%) |
| Control | DLBCL | p-Value | ||
|---|---|---|---|---|
| N = 19 | N = 40 | |||
| CD20+ % | Median | 24 | 14.5 | 0.001 * |
| IQR | (20–25) | (10–22) | ||
| PDL1+CD20+ % | Median | 1 | 9.5 | <0.001 ** |
| IQR | (0.8–1) | (6.7–16) | ||
| PD1+CD20+ % | Median | 0.7 | 4.1 | 0.001 ** |
| IQR | (0.6–1) | (0.9–9) |
| Controls (I) | Pre-Therapy DLBCL (II) | Post-Therapy DLBCL (III) | p | ||||
|---|---|---|---|---|---|---|---|
| N = 19 | N = 20 | N = 20 | I vs. II | I vs. III | II vs. III | ||
| CD20+ % | Median | 24 | 22 | 10 | 0.525 | <0.001 * | <0.001 ** |
| IQR | (20–25) | (18.3–26.8) | (9.6–11.8) | ||||
| PDL-1+CD20+ % | Median | 1 | 16 | 6.8 | <0.001 ** | <0.001 * | <0.001 ** |
| IQR | (0.8–1) | (15–23) | (6–7.9) | ||||
| PD-1+CD20+ % | Median | 0.7 | 9 | 0.9 | <0.001 ** | 0.575 | <0.001 ** |
| IQR | (0.6–1) | (8–11) | (0.6–1) |
| Control (I) | Pre-Therapy DLBCL (II) | Post-Therapy DLBCL (III) | p | ||||
|---|---|---|---|---|---|---|---|
| N = 19 | N = 20 | N = 20 | I vs. II | I vs. III | II vs. III | ||
| CD20+ % | Low (<19) | 4 (21.1%) | 7 (35%) | 20 (100%) | 0.333 | <0.001 ** | <0.001 ** |
| High (>19) | 15 (78.9%) | 13 (65%) | 0 (0%) | ||||
| PDL-1+CD20+ % | Low (<6.8) | 19 (100%) | 0 (0%) | 10 (50%) | <0.001 ** | <0.001 ** | <0.001 ** |
| High (>6.8) | 0 (0%) | 20 (100%) | 10 (50%) | ||||
| PD-1+CD20+ % | Low (<1) | 17 (89.5%) | 0 (0%) | 18 (90%) | <0.001 ** | 1.0 | <0.001 ** |
| High (>1) | 2 (10.5%) | 20 (100%) | 2 (10%) |
| High CD20+ % | High PD-L1+CD20+ % | High PD-1+CD20+ % | ||||
|---|---|---|---|---|---|---|
| r | p-Value | r | p-Value | r | p-Value | |
| High PD-L1+CD20+ % (P) | 0.401 | 0.010 * | ||||
| High PD-1+CD20+ % (P) | 0.628 | <0.001 ** | 0.638 | <0.001 ** | ||
| L D H (P) U/L | 0.479 | 0.002 * | 0.365 | 0.021 * | 0.549 | <0.001 ** |
| Random glucose (P) (mg/dL) | 0.019 | 0.910 | 0.368 | 0.020 * | 0.152 | 0.348 |
| Splenomegaly (S) | 0.235 | 0.151 | 0.251 | 0.123 | 0.354 | 0.027 * |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Saber, M.M. Diagnostic Performance of PD-L1 versus PD-1 Expression in Circulating CD20 Cells in Diffuse Large B-Cell Lymphoma. Antibodies 2022, 11, 15. https://doi.org/10.3390/antib11010015
Saber MM. Diagnostic Performance of PD-L1 versus PD-1 Expression in Circulating CD20 Cells in Diffuse Large B-Cell Lymphoma. Antibodies. 2022; 11(1):15. https://doi.org/10.3390/antib11010015
Chicago/Turabian StyleSaber, Manal Mohamed. 2022. "Diagnostic Performance of PD-L1 versus PD-1 Expression in Circulating CD20 Cells in Diffuse Large B-Cell Lymphoma" Antibodies 11, no. 1: 15. https://doi.org/10.3390/antib11010015
APA StyleSaber, M. M. (2022). Diagnostic Performance of PD-L1 versus PD-1 Expression in Circulating CD20 Cells in Diffuse Large B-Cell Lymphoma. Antibodies, 11(1), 15. https://doi.org/10.3390/antib11010015





