Creating Strategic Reserves to Protect Forest Carbon and Reduce Biodiversity Losses in the United States
Abstract
:1. Introduction
- ○
- Avoiding deforestation and forest degradation—keeping forests intact;
- ○
- Reducing carbon loss by increasing harvest intervals and decreasing harvest intensity;
- ○
- Carbon storage in long-lived forest products (e.g., in combination with shorter harvest intervals);
- ○
- Burning trees for bioenergy;
- ○
- Thinning to reduce fire risk or severity and thus carbon losses.
2. Strategies
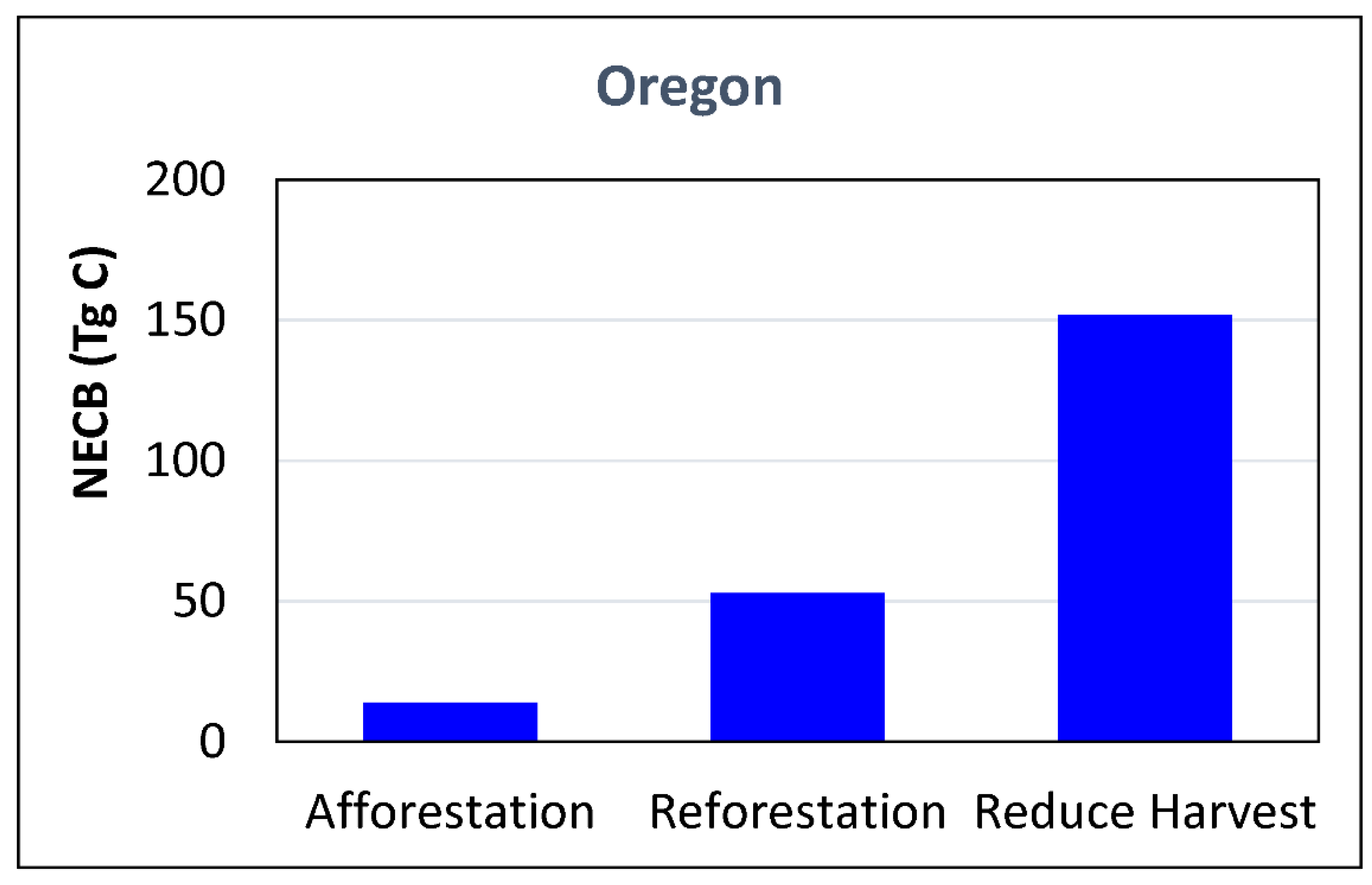
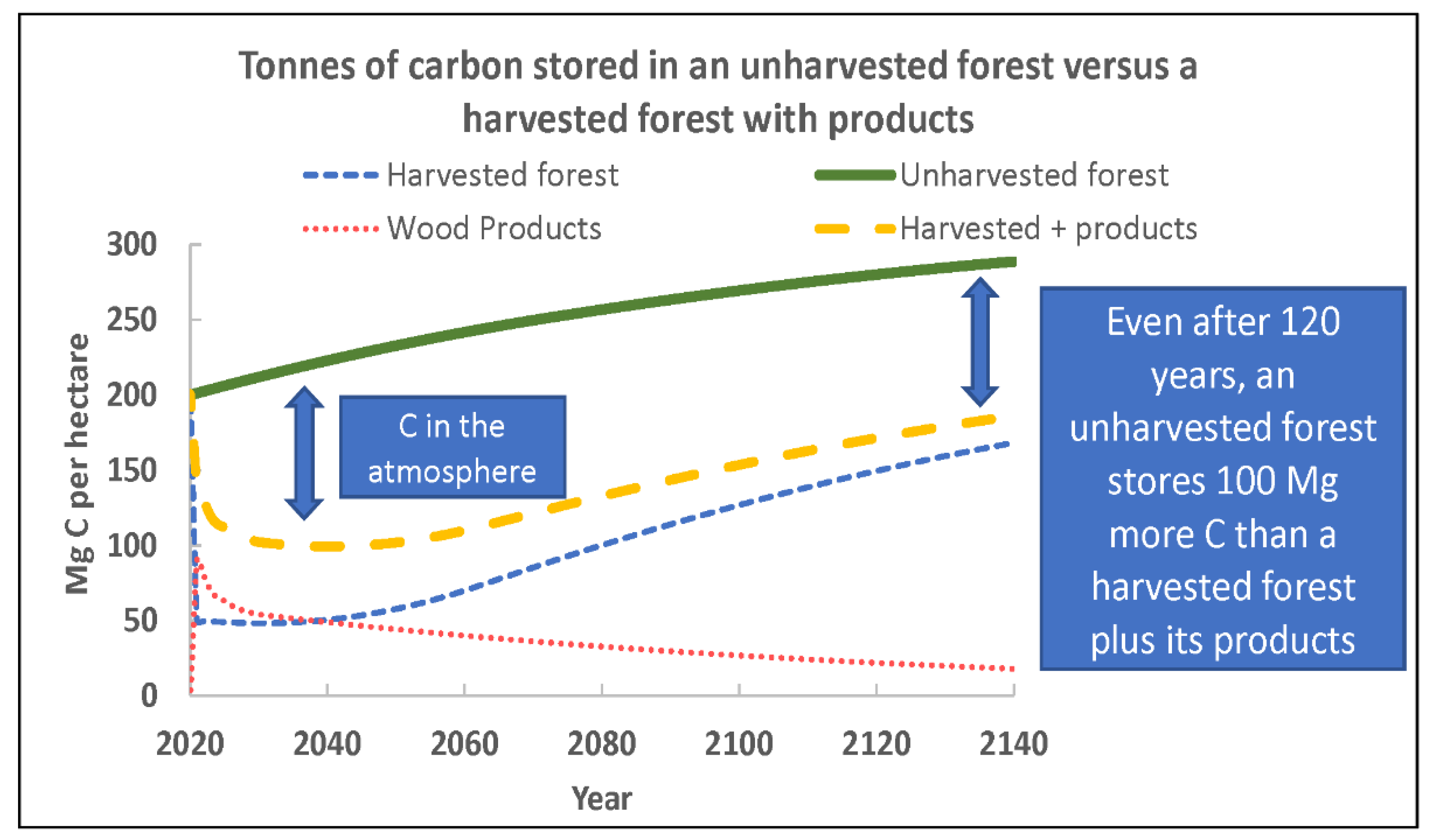
2.1. Avoid Deforestation and Forest Degradation, and Decrease Harvest-Related Carbon Losses
2.2. Harvesting Forests for Bioenergy Production
2.3. Thinning to Reduce Fire Risk or Severity and Carbon Loss
2.3.1. Broad-Scale Thinning to Reduce Fire Severity Conflicts with Climate Goals
2.3.2. Change Focus from Broadscale Thinning to the Home Ignition Zone
2.3.3. Post-Fire Harvest versus Natural Regeneration
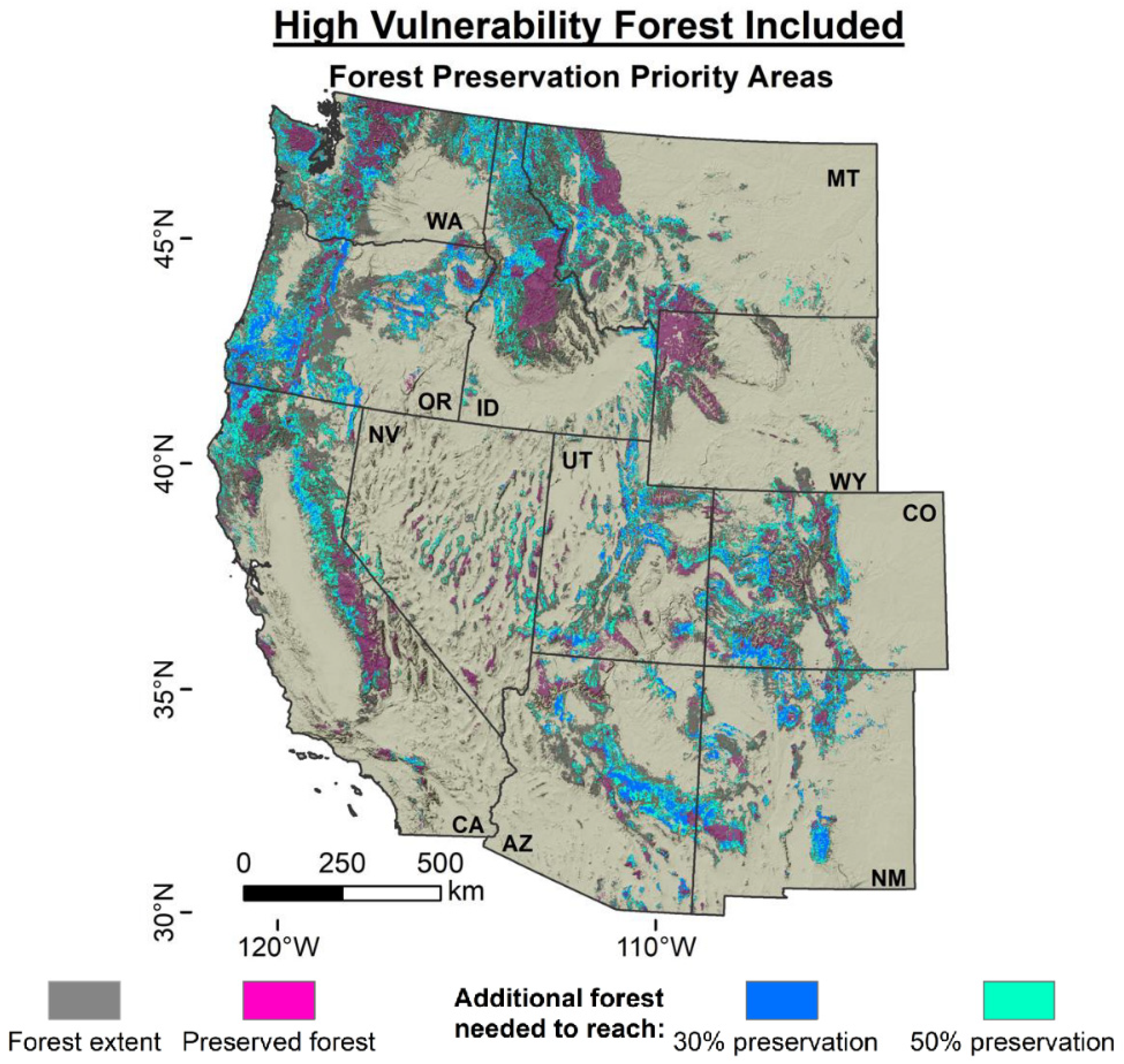
3. Solutions
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- IPCC. Summary for Policymakers. In Climate Change 2022: Impacts, Adaptation, and Vulnerability. Contribution of Working Group II to the Sixth Assessment Report of the Intergovernmental Panel on Climate Change; Pörtner, H.-O., Roberts, D.C., Poloczanska, E.S., Mintenbeck, K., Tignor, M., Alegría, A., Craig, M., Langsdorf, S., Löschke, S., Möller, V., et al., Eds.; Cambridge University Press: Cambridge, UK, 2022. [Google Scholar]
- IPCC. Summar for Policymakers. In Climate Change 2021: The Physical Science Basis. Contribution of Working Group I to the Sixth Assessment Report of the Intergovernmental Panel on Climate Change; Cambridge University Press: Cambridge, UK, 2021. [Google Scholar]
- Trisos, C.H.; Merow, C.; Pigot, A.L. The projected timing of abrupt ecological disruption from climate change. Nature 2020, 580, 496–501. [Google Scholar] [CrossRef] [PubMed]
- IPCC. Summary for Policymakers. In Global Warming of 1.5 °C; World Meteorological Organization: Geneva, Switzerland, 2018. [Google Scholar]
- Liu, P.R.; Raftery, A.E. Country-based rate of emissions reductions should increase by 80% beyond nationally determined contributions to meet the 2 C target. Commun. Earth Environ. 2021, 2, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Pan, Y.; Birdsey, R.A.; Phillips, O.L.; Jackson, R.B. The structure, distribution, and biomass of the world’s forests. Annu. Rev. Ecol. Evol. Syst. 2013, 44, 593–622. [Google Scholar] [CrossRef] [Green Version]
- Friedlingstein, P.; Jones, M.W.; O’Sullivan, M.; Andrew, R.M.; Bakker, D.C.E.; Hauck, J.; Le Quéré, C.; Peters, G.P.; Peters, W.; Pongratz, J.; et al. Global Carbon Budget 2021. Earth Syst. Sci. Data Discuss. 2021, 2021, 1917–2005. [Google Scholar] [CrossRef]
- Pandit, R.; Pörtner, H.-O.; Scholes, R.J.; Agard, J.; Archer, E.; Arneth, A.; Bai, X.; Barnes, D.; Burrows, M.; Chan, L. Scientific Outcome of the IPBES-IPCC Co-Sponsored Workshop on Biodiversity and Climate Change. 2021. Available online: https://zenodo.org/record/5101133#.YnqZFYfMLb0 (accessed on 20 April 2022).
- Law, B.E.; Berner, L.T.; Buotte, P.C.; Mildrexler, D.J.; Ripple, W.J. Strategic Forest Reserves can protect biodiversity in the western United States and mitigate climate change. Commun. Earth Environ. 2021, 2, 254. [Google Scholar] [CrossRef]
- Buotte, P.C.; Law, B.E.; Ripple, W.J.; Berner, L.T. Carbon sequestration and biodiversity co-benefits of preserving forests in the western United States. Ecol. Appl. 2020, 30, e02039. [Google Scholar] [CrossRef] [Green Version]
- Oswalt, S.N.; Smith, W.B.; Miles, P.D.; Pugh, S.A. Forest Resources of the United States, 2017: A Technical Document Supporting the Forest Service 2020 RPA Assessment; Gen. Tech. Rep. WO-97; US Department of Agriculture, Forest Service, Washington Office: Washington, DC, USA, 2019; Volume 97.
- FAO. Global Forest Resources Assessment 2020–Key Findings; FAO: Rome, Italy, 2020. [Google Scholar] [CrossRef]
- Novick, K.A.; Katul, G.G. The Duality of Reforestation Impacts on Surface and Air Temperature. J. Geophys. Res. Biogeosci. 2020, 125, e2019JG005543. [Google Scholar] [CrossRef]
- Lawrence, D.; Coe, M.; Walker, W.S.; Verchot, L.; Vandecar, K.L. The unseen effects of deforestation: Biophysical effects on climate. Front. For. Glob. Change 2022, 5, 756115. [Google Scholar] [CrossRef]
- Law, B.E.; Hudiburg, T.W.; Berner, L.T.; Kent, J.J.; Buotte, P.C.; Harmon, M.E. Land use strategies to mitigate climate change in carbon dense temperate forests. Proc. Natl. Acad. Sci. USA 2018, 115, 3663–3668. [Google Scholar] [CrossRef] [Green Version]
- Hudiburg, T.; Law, B.; Turner, D.P.; Campbell, J.; Donato, D.; Duane, M. Carbon dynamics of Oregon and Northern California forests and potential land-based carbon storage. Ecol. Appl. 2009, 19, 163–180. [Google Scholar] [CrossRef]
- Vynne, C.; Dovichin, E.; Fresco, N.; Dawson, N.; Joshi, A.; Law, B.E.; Lertzman, K.; Rupp, S.; Schmiegelow, F.; Trammell, E.J. The importance of Alaska for climate stabilization, resilience, and biodiversity conservation. Front. For. Glob. Change 2021, 121. [Google Scholar] [CrossRef]
- US Congress. Multiple Use-Sustained Yield Act. 1. PL 86-517; 74 Stat 1960, 215. Available online: https://www.fs.fed.us/emc/nfma/includes/musya60.pdf (accessed on 20 April 2022).
- 94th US Congress. Federal Land Management and Policy ACT OF 1976. PL 94–579. Available online: https://www.govinfo.gov/content/pkg/STATUTE-90/pdf/STATUTE-90-Pg2743.pdf#page=1 (accessed on 20 April 2022).
- Riddle, A.A. Timber Harvesting on Federal Lands; Congressional Research Service, R45688. Available online: https://crsreports.congress.gov/product/pdf/R/R45688 (accessed on 20 April 2022).
- Law, B.E.; Waring, R.H. Carbon implications of current and future effects of drought, fire and management on Pacific Northwest forests. For. Ecol. Manag. 2015, 355, 4–14. [Google Scholar] [CrossRef] [Green Version]
- Moomaw, W.R.; Masino, S.A.; Faison, E.K. Intact Forests in the United States: Proforestation Mitigates Climate Change and Serves the Greatest Good. Front. For. Glob. Change 2019, 2, 27. [Google Scholar] [CrossRef] [Green Version]
- National Academies of Sciences, Engineering, and Medicine. Negative Emissions Technologies and Reliable Sequestration: A Research Agenda. 2018. Available online: https://nap.nationalacademies.org/read/25259/chapter/1 (accessed on 15 April 2022).
- Lutz, J.A.; Furniss, T.J.; Johnson, D.J.; Davies, S.J.; Allen, D.; Alonso, A.; Anderson-Teixeira, K.J.; Andrade, A.; Baltzer, J.; Becker, K.M.L.; et al. Global importance of large-diameter trees. Glob. Ecol. Biogeogr. 2018, 27, 849–864. [Google Scholar] [CrossRef] [Green Version]
- Mildrexler, D.J.; Berner, L.T.; Law, B.E.; Birdsey, R.A.; Moomaw, W.R. Large Trees Dominate Carbon Storage in Forests East of the Cascade Crest in the United States Pacific Northwest. Front. For. Glob. Change 2020, 3, 17. [Google Scholar] [CrossRef]
- Luyssaert, S.; Schulze, E.D.; Borner, A.; Knohl, A.; Hessenmoller, D.; Law, B.E.; Ciais, P.; Grace, J. Old-growth forests as global carbon sinks. Nature 2008, 455, 213–215. [Google Scholar] [CrossRef]
- Erb, K.-H.; Kastner, T.; Plutzar, C.; Bais, A.L.S.; Carvalhais, N.; Fetzel, T.; Gingrich, S.; Haberl, H.; Lauk, C.; Niedertscheider, M. Unexpectedly large impact of forest management and grazing on global vegetation biomass. Nature 2018, 553, 73–76. [Google Scholar] [CrossRef]
- Lewis, S.L.; Wheeler, C.E.; Mitchard, E.T.; Koch, A. Regenerate natural forests to store carbon. Nature 2019, 568, 25–28. [Google Scholar] [CrossRef]
- Watson, J.E.; Evans, T.; Venter, O.; Williams, B.; Tulloch, A.; Stewart, C.; Thompson, I.; Ray, J.C.; Murray, K.; Salazar, A. The exceptional value of intact forest ecosystems. Nat. Ecol. Evol. 2018, 2, 599–610. [Google Scholar] [CrossRef]
- Houghton, R.A.; Nassikas, A.A. Negative emissions from stopping deforestation and forest degradation, globally. Glob. Change Biol. 2018, 24, 350–359. [Google Scholar] [CrossRef]
- Hudiburg, T.W.; Law, B.E.; Moomaw, W.R.; Harmon, M.E.; Stenzel, J.E. Meeting GHG reduction targets requires accounting for all forest sector emissions. Environ. Res. Lett. 2019, 14, 095005. [Google Scholar] [CrossRef]
- Harmon, M.E.; Marks, B. Effects of silvicultural practices on carbon stores in Douglas-fir western hemlock forests in the Pacific Northwest, USA: Results from a simulation model. Can. J. For. Res. 2002, 32, 863–877. [Google Scholar] [CrossRef] [Green Version]
- Harmon, M.E. Have product substitution carbon benefits been overestimated? A sensitivity analysis of key assumptions. Environ. Res. Lett. 2019, 14, 065008. [Google Scholar]
- Searchinger, T.D.; Beringer, T.; Holtsmark, B.; Kammen, D.M.; Lambin, E.F.; Lucht, W.; Raven, P.; van Ypersele, J.-P. Europe’s renewable energy directive poised to harm global forests. Nat. Commun. 2018, 9, 3741. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- EPA. Emissions Factors for Greenhouse Gas Inventories. Available online: https://www.epa.gov/sites/default/files/2018-03/documents/emission-factors_mar_2018_0.pdf (accessed on 23 February 2022).
- EPA. Compilation of Air Pollutant Emission Factors, AP-42; US Environemental Protection Agency: Washington, DC, USA, 1997.
- Röder, M.; Whittaker, C.; Thornley, P. How certain are greenhouse gas reductions from bioenergy? Life cycle assessment and uncertainty analysis of wood pellet-to-electricity supply chains from forest residues. Biomass Bioenerg. 2015, 79, 50–63. [Google Scholar] [CrossRef]
- Sterman, J.D.; Lori, S.; Juliette, N.R.-V. Does replacing coal with wood lower CO2 emissions? Dynamic lifecycle analysis of wood bioenergy. Environ. Res. Lett. 2018, 13, 015007. [Google Scholar]
- Mitchell, S.R.; Harmon, M.E.; O’Connell, K.E.B. Carbon debt and carbon sequestration parity in forest bioenergy production. GCB Bioenergy 2012, 4, 818–827. [Google Scholar] [CrossRef] [Green Version]
- Sterman, J.D.; Siegel, L.; Rooney-Varga, J.N. Reply to comment on ‘Does replacing coal with wood lower CO2 emissions? Dynamic lifecycle analysis of wood bioenergy’. Environ. Res. Lett. 2018, 13, 128003. [Google Scholar] [CrossRef]
- Obermeier, W.A.; Nabel, J.E.; Loughran, T.; Hartung, K.; Bastos, A.; Havermann, F.; Anthoni, P.; Arneth, A.; Goll, D.S.; Lienert, S. Modelled land use and land cover change emissions—A spatio-temporal comparison of different approaches. Earth Syst. Dyn. 2021, 12, 635–670. [Google Scholar] [CrossRef]
- Hudiburg, T.W.; Law, B.E.; Wirth, C.; Luyssaert, S. Regional carbon dioxide implications of forest bioenergy production. Nat. Clim. Change 2011, 1, 419–423. [Google Scholar] [CrossRef]
- Schlesinger, W.H. Are wood pellets a green fuel? Science 2018, 359, 1328–1329. [Google Scholar] [CrossRef] [PubMed]
- Körner, C. Slow in, Rapid out--Carbon Flux Studies and Kyoto Targets. Science 2003, 300, 1242–1243. [Google Scholar] [CrossRef] [PubMed]
- Solomon, S.; Plattner, G.-K.; Knutti, R.; Friedlingstein, P. Irreversible climate change due to carbon dioxide emissions. Proc. Natl. Acad. Sci. USA 2009, 106, 1704–1709. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Campbell, J.L.; Harmon, M.E.; Mitchell, S.R. Can fuel-reduction treatments really increase forest carbon storage in the western US by reducing future fire emissions? Front. Ecol. Environ. 2012, 10, 83–90. [Google Scholar] [CrossRef]
- Mitchell, S.R.; Harmon, M.E.; O’Connel, K.E.B. Forest fuel reduction alters fire severity and long-term carbon storage in three Pacific Northwest ecosystems. Ecol. Appl. 2009, 19, 643–655. [Google Scholar] [CrossRef]
- Rhodes, J.J.; Baker, W.L. Fire probability, fuel treatment effectiveness and ecological tradeoffs in western US public forests. Open For. Sci. J. 2008, 1, 1–7. [Google Scholar]
- Hudiburg, T.W.; Luyssaert, S.; Thornton, P.E.; Law, B.E. Interactive Effects of Environmental Change and Management Strategies on Regional Forest Carbon Emissions. Environ. Sci. Technol. 2013, 47, 13132–13140. [Google Scholar] [CrossRef]
- Zhou, D.; Zhao, S.; Liu, S.; Oeding, J. A meta-analysis on the impacts of partial cutting on forest structure and carbon storage. Biogeosciences 2013, 10, 3691–3703. [Google Scholar] [CrossRef] [Green Version]
- Stenzel, J.; Berardi, D.; Walsh, E.; Hudiburg, T. Restoration Thinning in a Drought-Prone Idaho Forest Creates a Persistent Carbon Deficit. J. Geophys. Res. Biogeosciences 2021, 126, e2020JG005815. [Google Scholar] [CrossRef]
- Zald, H.S.; Dunn, C.J. Severe fire weather and intensive forest management increase fire severity in a multi-ownership landscape. Ecol. Appl. 2018, 28, 1068–1080. [Google Scholar] [CrossRef]
- Schoennagel, T.; Balch, J.K.; Brenkert-Smith, H.; Dennison, P.E.; Harvey, B.J.; Krawchuk, M.A.; Mietkiewicz, N.; Morgan, P.; Moritz, M.A.; Rasker, R.; et al. Adapt to more wildfire in western North American forests as climate changes. Proc. Natl. Acad. Sci. USA 2017, 114, 4582–4590. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hurteau, M.D.; North, M.P.; Koch, G.W.; Hungate, B.A. Opinion: Managing for disturbance stabilizes forest carbon. Proc. Natl. Acad. Sci. USA 2019, 116, 10193–10195. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Stenzel, J.E.; Bartowitz, K.J.; Hartman, M.D.; Lutz, J.A.; Kolden, C.A.; Smith, A.M.S.; Law, B.E.; Swanson, M.E.; Larson, A.J.; Parton, W.J.; et al. Fixing a snag in carbon emissions estimates from wildfires. Glob. Change Biol. 2019, 25, 3985–3994. [Google Scholar] [CrossRef] [PubMed]
- Harmon, M.E.; Hanson, C.T.; DellaSala, D.A. Combustion of Aboveground Wood from Live Trees in Megafires, CA, USA. Forests 2022, 13, 391. [Google Scholar] [CrossRef]
- Meigs, G.; Donato, D.; Campbell, J.; Martin, J.; Law, B. Forest fire impacts on carbon uptake, storage, and emission: The role of burn severity in the Eastern Cascades, Oregon. Ecosystems 2009, 12, 1246–1267. [Google Scholar] [CrossRef]
- Campbell, J.; Donato, D.; Azuma, D.; Law, B. Pyrogenic carbon emission from a large wildfire in Oregon, United States. J. Geophys. Res. Biogeosciences 2007, 112, G04014. [Google Scholar] [CrossRef] [Green Version]
- Bartowitz, K.J.; Walsh, E.S.; Stenzel, J.E.; Kolden, C.A.; Hudiburg, T.W. Forest carbon emission sources arenot equal: Putting fire, harvest, and fossil fuel emissions in context. Front. For. Glob. Change 2022, 5, 867112. [Google Scholar] [CrossRef]
- Harris, N.L.; Hagen, S.C.; Saatchi, S.S.; Pearson, T.R.H.; Woodall, C.W.; Domke, G.M.; Braswell, B.H.; Walters, B.F.; Brown, S.; Salas, W.; et al. Attribution of net carbon change by disturbance type across forest lands of the conterminous United States. Carbon Balance Manag. 2016, 11, 24. [Google Scholar] [CrossRef] [Green Version]
- Downing, W.M.; Dunn, C.J.; Thompson, M.P.; Caggiano, M.D.; Short, K.C. Human ignitions on private lands drive USFS cross-boundary wildfire transmission and community impacts in the western US. Sci. Rep. 2022, 12, 1–14. [Google Scholar] [CrossRef]
- Ager, A.A.; Palaiologou, P.; Evers, C.R.; Day, M.A.; Ringo, C.; Short, K. Wildfire exposure to the wildland urban interface in the western US. Appl. Geogr. 2019, 111, 102059. [Google Scholar] [CrossRef]
- Smith, A.M.; Kolden, C.A.; Paveglio, T.B.; Cochrane, M.A.; Bowman, D.M.; Moritz, M.A.; Kliskey, A.D.; Alessa, L.; Hudak, A.T.; Hoffman, C.M. The science of firescapes: Achieving fire-resilient communities. Bioscience 2016, 66, 130–146. [Google Scholar] [CrossRef] [PubMed]
- Syphard, A.D.; Rustigian-Romsos, H.; Mann, M.; Conlisk, E.; Moritz, M.A.; Ackerly, D. The relative influence of climate and housing development on current and projected future fire patterns and structure loss across three California landscapes. Glob. Environ. Change 2019, 56, 41–55. [Google Scholar] [CrossRef]
- Keeley, J.E.; Syphard, A.D. Twenty-first century California, USA, wildfires: Fuel-dominated vs. wind-dominated fires. Fire Ecol. 2019, 15, 24. [Google Scholar] [CrossRef] [Green Version]
- Donato, D.C.; Fontaine, J.B.; Campbell, J.L.; Robinson, W.D.; Kauffman, J.B.; Law, B.E. Conifer regeneration in stand-replacement portions of a large mixed-severity wildfire in the Klamath–Siskiyou Mountains. Can. J. For. Res. 2009, 39, 823–838. [Google Scholar] [CrossRef] [Green Version]
- Cook-Patton, S.C.; Leavitt, S.M.; Gibbs, D.; Harris, N.L.; Lister, K.; Anderson-Teixeira, K.J.; Briggs, R.D.; Chazdon, R.L.; Crowther, T.W.; Ellis, P.W.; et al. Mapping carbon accumulation potential from global natural forest regrowth. Nature 2020, 585, 545–550. [Google Scholar] [CrossRef]
- Stevens-Rumann, C.S.; Kemp, K.B.; Higuera, P.E.; Harvey, B.J.; Rother, M.T.; Donato, D.C.; Morgan, P.; Veblen, T.T. Evidence for declining forest resilience to wildfires under climate change. Ecol. Lett. 2018, 21, 243–252. [Google Scholar] [CrossRef]
- Davis, K.T.; Dobrowski, S.Z.; Higuera, P.E.; Holden, Z.A.; Veblen, T.T.; Rother, M.T.; Parks, S.A.; Sala, A.; Maneta, M.P. Wildfires and climate change push low-elevation forests across a critical climate threshold for tree regeneration. Proc. Natl. Acad. Sci. USA 2019, 116, 6193–6198. [Google Scholar] [CrossRef] [Green Version]
- Fontaine, J.B.; Donato, D.C.; Robinson, W.D.; Law, B.E.; Kauffman, J.B. Bird communities following high-severity fire: Response to single and repeat fires in a mixed-evergreen forest, Oregon, USA. For. Ecol. Manag. 2009, 257, 1496–1504. [Google Scholar] [CrossRef] [Green Version]
- Beschta, R.L.; Frissell, C.A.; Gresswell, R.; Hauer, R.; Karr, J.R.; Minshall, G.W.; Perry, D.A.; Rhodes, J.J. Wildfire and Salvage Logging: Recommendations for Ecologically Sound Post-Fire Salvage Management And Other Post-Fire Treatments on Federal Lands in the West; Oregon State University: Corvallis, OR, USA, 1995. [Google Scholar]
- Rose, C.L.; Marcot, B.G.; Mellen, T.K.; Ohmann, J.L.; Waddell, K.L.; Lindley, D.L.; Schreiber, B. Decaying wood in Pacific Northwest forests: Concepts and tools for habitat management. In Wildlife-Habitat Relationships in Oregon and Washington; Oregon State University Press: Corvallis, OR, USA, 2001; pp. 580–623. [Google Scholar]
- Thorn, S.; Bässler, C.; Brandl, R.; Burton, P.J.; Cahall, R.; Campbell, J.L.; Castro, J.; Choi, C.-Y.; Cobb, T.; Donato, D.C.; et al. Impacts of salvage logging on biodiversity: A meta-analysis. J. Appl. Ecol. 2018, 55, 279–289. [Google Scholar] [CrossRef]
- Karr, J.R.; Rhodes, J.J.; Minshall, G.W.; Hauer, F.R.; Beschta, R.L.; Frissell, C.A.; Perry, D.A. The Effects of Postfire Salvage Logging on Aquatic Ecosystems in the American West. BioScience 2004, 54, 1029–1033. [Google Scholar] [CrossRef] [Green Version]
- Beschta, R.L.; Rhodes, J.J.; Kauffman, J.B.; Gresswell, R.E.; Minshall, G.W.; Karr, J.R.; Perry, D.A.; Hauer, F.R.; Frissell, C.A. Postfire Management on Forested Public Lands of the Western United States. Conserv. Biol. 2004, 18, 957–967. [Google Scholar] [CrossRef]
- Pehl, M.; Arvesen, A.; Humpenöder, F.; Popp, A.; Hertwich, E.G.; Luderer, G. Understanding future emissions from low-carbon power systems by integration of life-cycle assessment and integrated energy modelling. Nat. Energy 2017, 2, 939–945. [Google Scholar] [CrossRef]
- Elsen, P.R.; Monahan, W.B.; Dougherty, E.R.; Merenlender, A.M. Keeping pace with climate change in global terrestrial protected areas. Sci. Adv. 2020, 6, eaay0814. [Google Scholar] [CrossRef] [PubMed]
- Dinerstein, E.; Joshi, A.; Vynne, C.; Lee, A.; Pharand-Deschênes, F.; França, M.; Fernando, S.; Birch, T.; Burkart, K.; Asner, G. A “Global Safety Net” to reverse biodiversity loss and stabilize Earth’s climate. Sci. Adv. 2020, 6, eabb2824. [Google Scholar] [CrossRef]
- Timmers, R.; van Kuijk, M.; Verweij, P.A.; Ghazoul, J.; Hautier, Y.; Laurance, W.F.; Arriaga-Weiss, S.L.; Askins, R.A.; Battisti, C.; Berg, Å. Conservation of birds in fragmented landscapes requires protected areas. Front. Ecol. Environ. 2022. [Google Scholar] [CrossRef]
- IUCN. The IUCN Red List of Threatened Species. 2020. Available online: https://www.iucnredlist.org/ (accessed on 20 April 2022).
- Mackey, B.; Kormos, C.F.; Keith, H.; Moomaw, W.R.; Houghton, R.A.; Mittermeier, R.A.; Hole, D.; Hugh, S. Understanding the importance of primary tropical forest protection as a mitigation strategy. Mitig. Adapt. Strateg. Glob. Change 2020, 25, 763–787. [Google Scholar] [CrossRef] [Green Version]
- Dinerstein, E.; Olson, D.; Joshi, A.; Vynne, C.; Burgess, N.D.; Wikramanayake, E.; Hahn, N.; Palminteri, S.; Hedao, P.; Noss, R.; et al. An Ecoregion-Based Approach to Protecting Half the Terrestrial Realm. BioScience 2017, 67, 534–545. [Google Scholar] [CrossRef]
- Rosenberg, K.V.; Dokter, A.M.; Blancher, P.J.; Sauer, J.R.; Smith, A.C.; Smith, P.A.; Stanton, J.C.; Panjabi, A.; Helft, L.; Parr, M. Decline of the North American avifauna. Science 2019, 366, 120–124. [Google Scholar] [CrossRef]
- Wagner, D.L.; Grames, E.M.; Forister, M.L.; Berenbaum, M.R.; Stopak, D. Insect decline in the Anthropocene: Death by a thousand cuts. Proc. Natl. Acad. Sci. USA 2021, 118, e2023989118. [Google Scholar] [CrossRef]
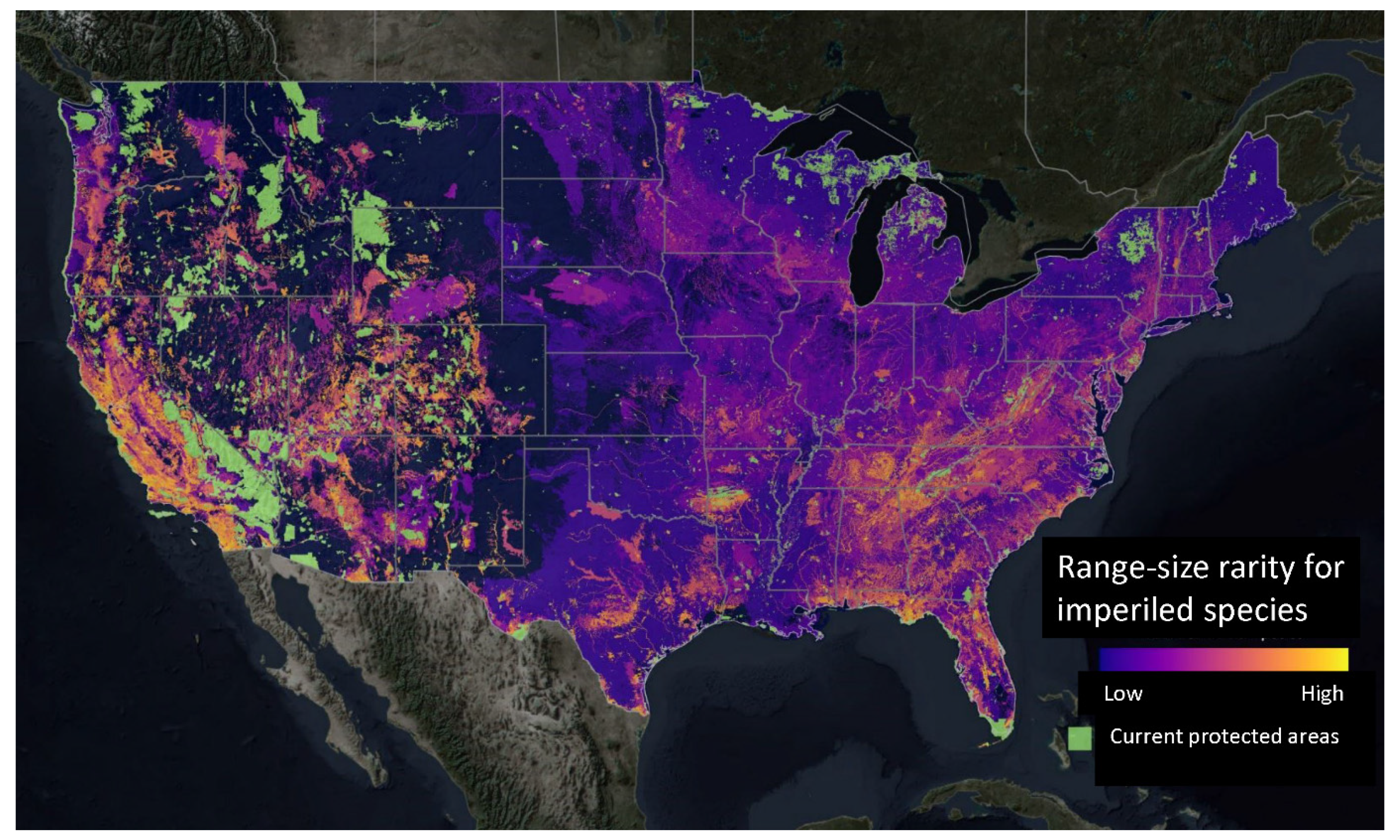
- Hamilton, H.; Smyth, R.L.; Young, B.E.; Howard, T.G.; Tracey, C.; Breyer, S.; Cameron, D.R.; Chazal, A.; Conley, A.K.; Frye, C.; et al. Increasing taxonomic diversity and spatial resolution clarifies opportunities for protecting US imperiled species. Ecol. Appl. 2022, 32, e2534. [Google Scholar] [CrossRef]
- Kauffman, J.B.; Beschta, R.L.; Lacy, P.M.; Liverman, M. Livestock Use on Public Lands in the Western USA Exacerbates Climate Change: Implications for Climate Change Mitigation and Adaptation. Environ. Manag. 2022, 69, 1137–1152. [Google Scholar] [CrossRef] [PubMed]
- Fa, J.E.; Watson, J.E.; Leiper, I.; Potapov, P.; Evans, T.D.; Burgess, N.D.; Molnár, Z.; Fernández-Llamazares, Á.; Duncan, T.; Wang, S. Importance of Indigenous Peoples’ lands for the conservation of Intact Forest Landscapes. Front. Ecol. Environ. 2020, 18, 135–140. [Google Scholar] [CrossRef]
- Liu, N.; Caldwell, P.V.; Dobbs, G.R.; Miniat, C.F.; Bolstad, P.V.; Nelson, S.A.; Sun, G. Forested lands dominate drinking water supply in the conterminous United States. Environ. Res. Lett. 2021, 16, 084008. [Google Scholar] [CrossRef]
- USDA. Forests to Faucets 2.0 [Spatial Data Set]. 2019. Available online: https://usfs-public.app.box.com/v/Forests2Faucets (accessed on 5 April 2022).
- USGS. Protected Areas Database of the United States (PAD-US) 2.1: U.S. Geological Survey Data Release. 2020. Available online: https://www.sciencebase.gov/catalog/item/5f186a2082cef313ed843257 (accessed on 30 March 2022).
- Cook, B.; Mankin, J.; Marvel, K.; Williams, A.; Smerdon, J.; Anchukaitis, K. Twenty-first century drought projections in the CMIP6 forcing scenarios. Earth’s Future 2020, 8, e2019EF001461. [Google Scholar] [CrossRef] [Green Version]
- Omernik, J.M.; Griffith, G.E. Ecoregions of the conterminous United States: Evolution of a hierarchical spatial framework. Environ. Manag. 2014, 54, 1249–1266. [Google Scholar] [CrossRef]
- UNFCCC. MLA, 7th ed.; United Nations Framework Convention on Climate Change; General Assembly: New York, NY, USA, 1992. [Google Scholar]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Law, B.E.; Moomaw, W.R.; Hudiburg, T.W.; Schlesinger, W.H.; Sterman, J.D.; Woodwell, G.M. Creating Strategic Reserves to Protect Forest Carbon and Reduce Biodiversity Losses in the United States. Land 2022, 11, 721. https://doi.org/10.3390/land11050721
Law BE, Moomaw WR, Hudiburg TW, Schlesinger WH, Sterman JD, Woodwell GM. Creating Strategic Reserves to Protect Forest Carbon and Reduce Biodiversity Losses in the United States. Land. 2022; 11(5):721. https://doi.org/10.3390/land11050721
Chicago/Turabian StyleLaw, Beverly E., William R. Moomaw, Tara W. Hudiburg, William H. Schlesinger, John D. Sterman, and George M. Woodwell. 2022. "Creating Strategic Reserves to Protect Forest Carbon and Reduce Biodiversity Losses in the United States" Land 11, no. 5: 721. https://doi.org/10.3390/land11050721
APA StyleLaw, B. E., Moomaw, W. R., Hudiburg, T. W., Schlesinger, W. H., Sterman, J. D., & Woodwell, G. M. (2022). Creating Strategic Reserves to Protect Forest Carbon and Reduce Biodiversity Losses in the United States. Land, 11(5), 721. https://doi.org/10.3390/land11050721




