Modelling Beach Litter Accumulation on Mediterranean Coastal Landscapes: An Integrative Framework Using Species Distribution Models
Abstract
1. Introduction
2. Materials and Methods
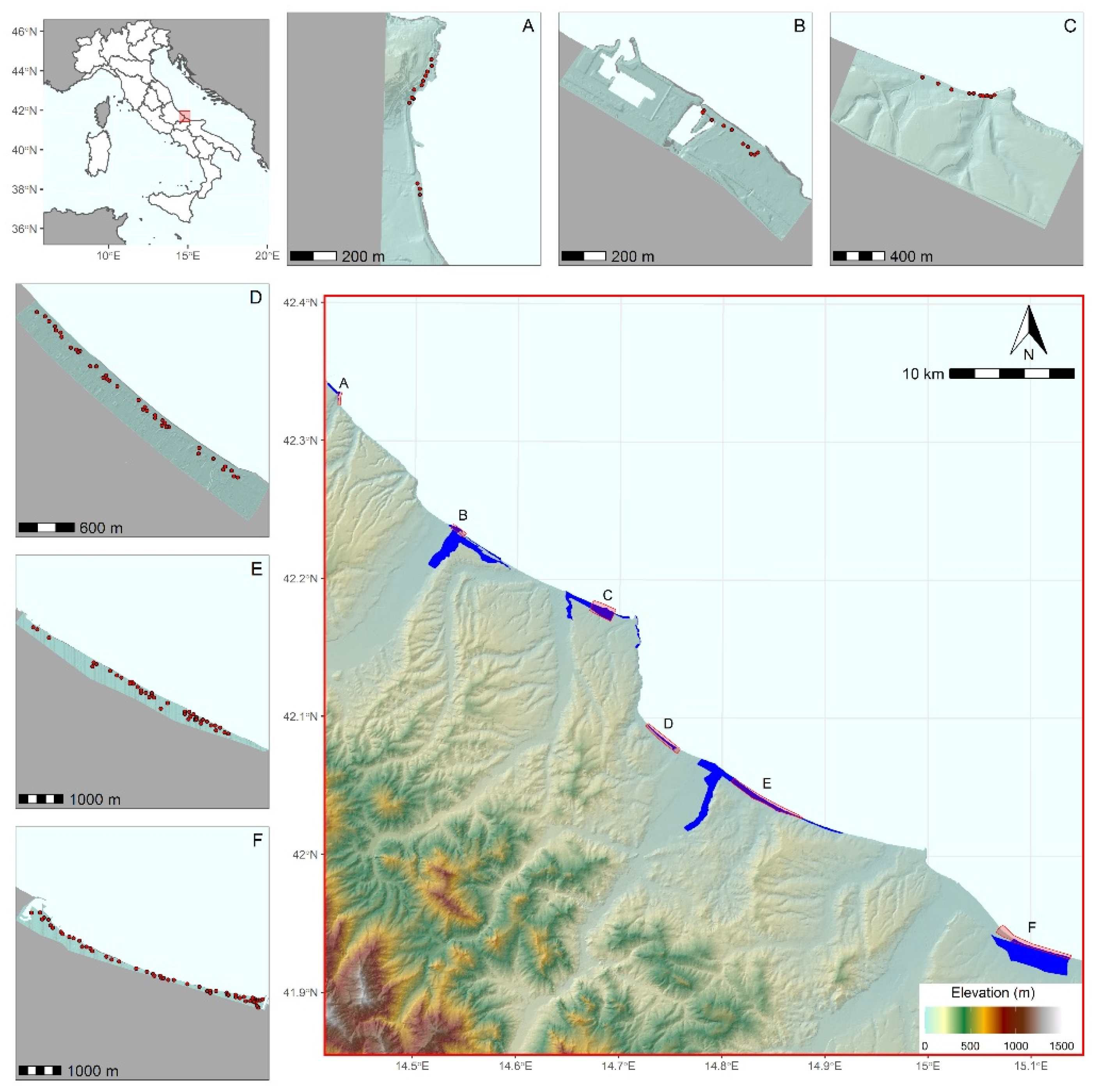
2.1. Study Area
2.2. Beach Litter Occurrence Data
2.3. Geographical Covariates
2.4. Litter Distribution Models
2.5. Hotspot Analysis
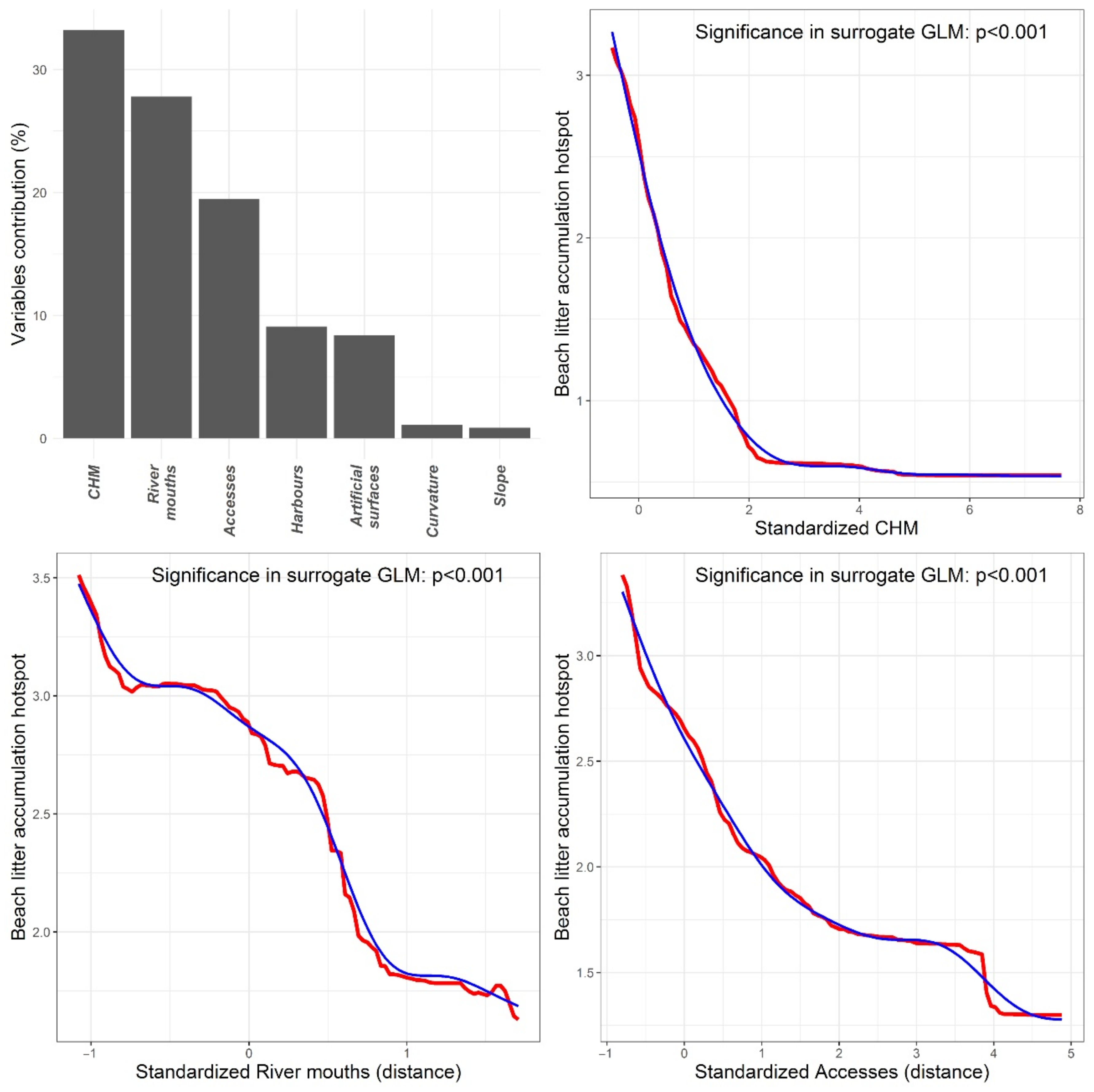
3. Results
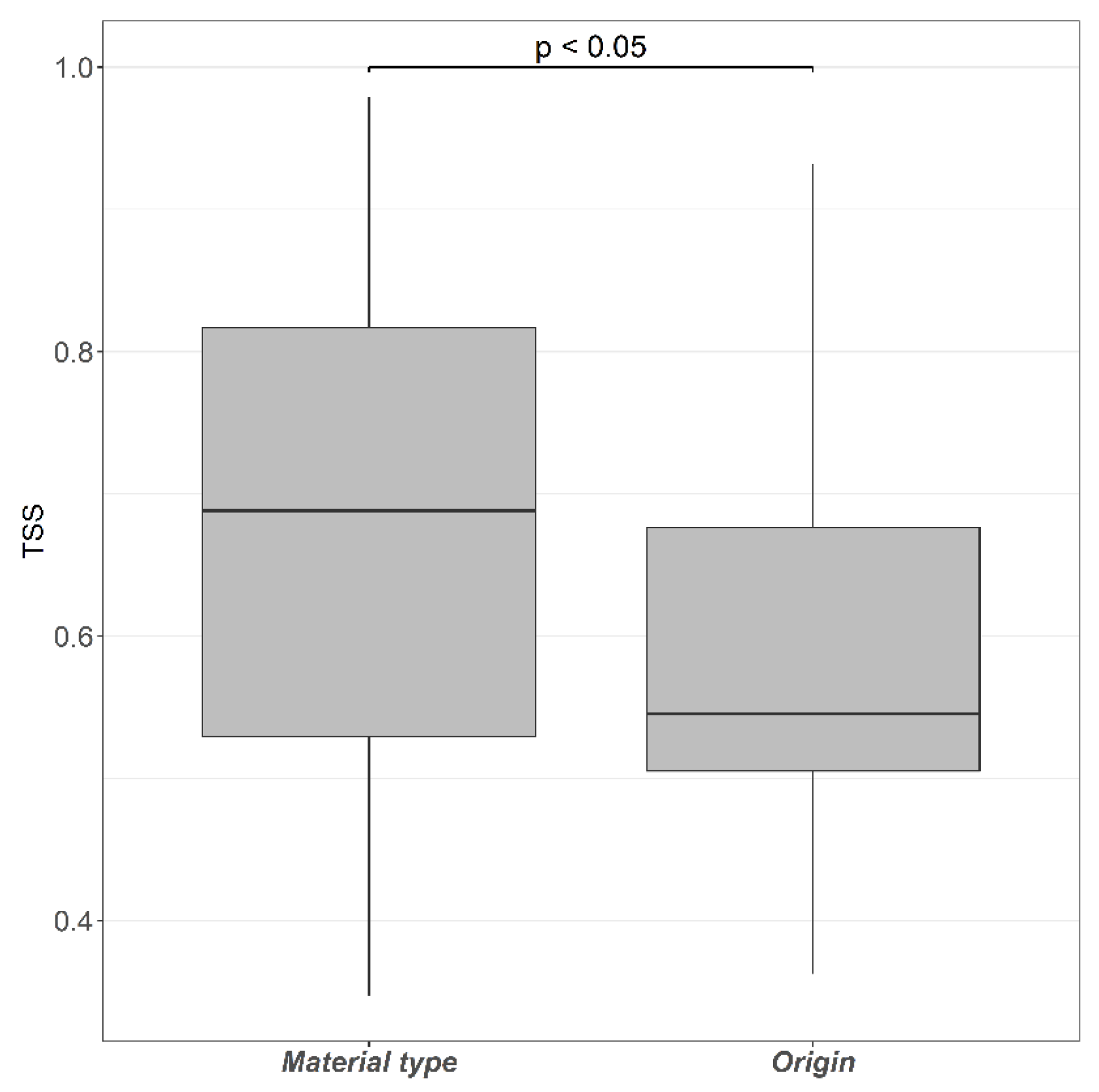
3.1. Litter Distribution Models
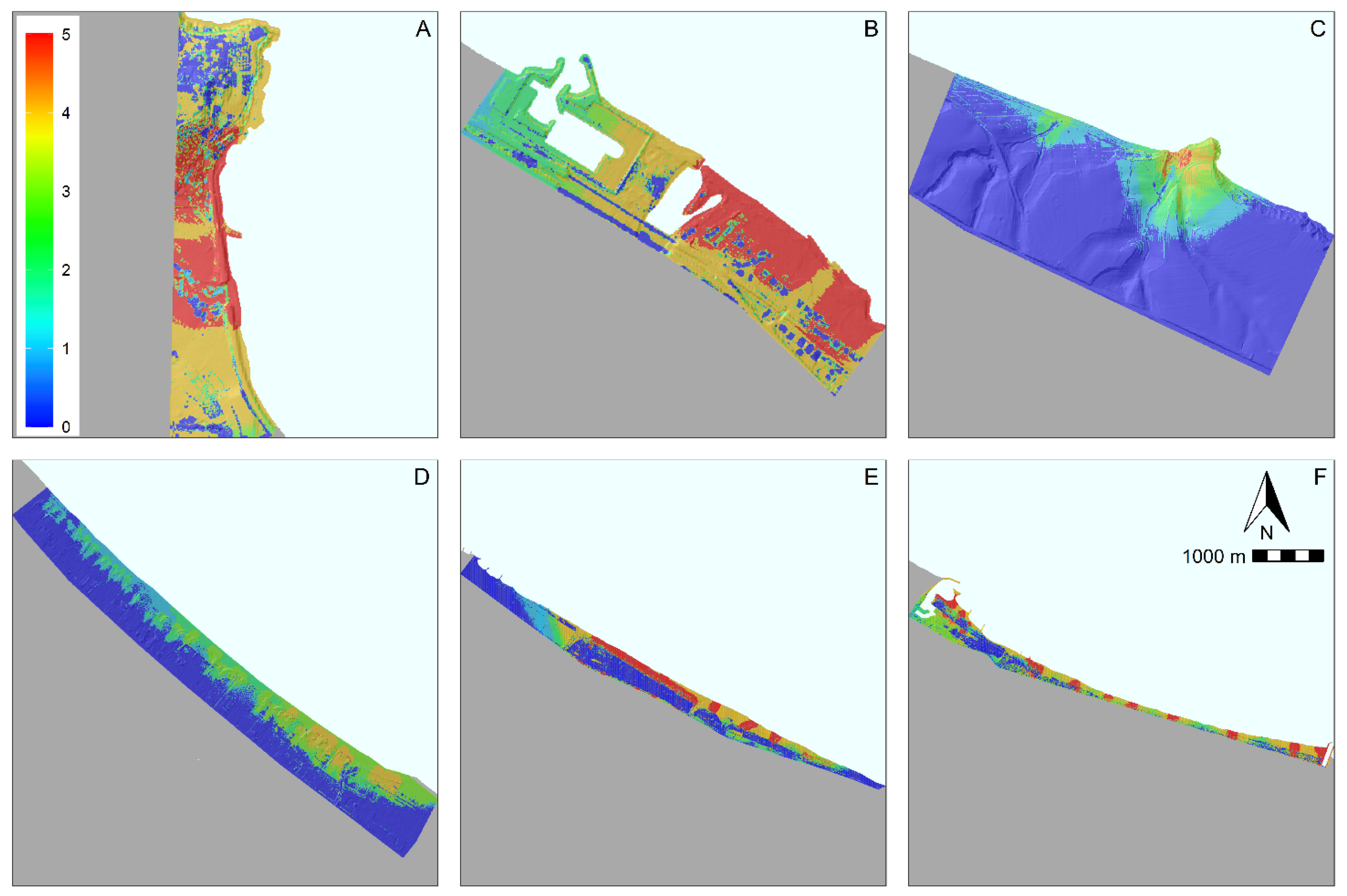
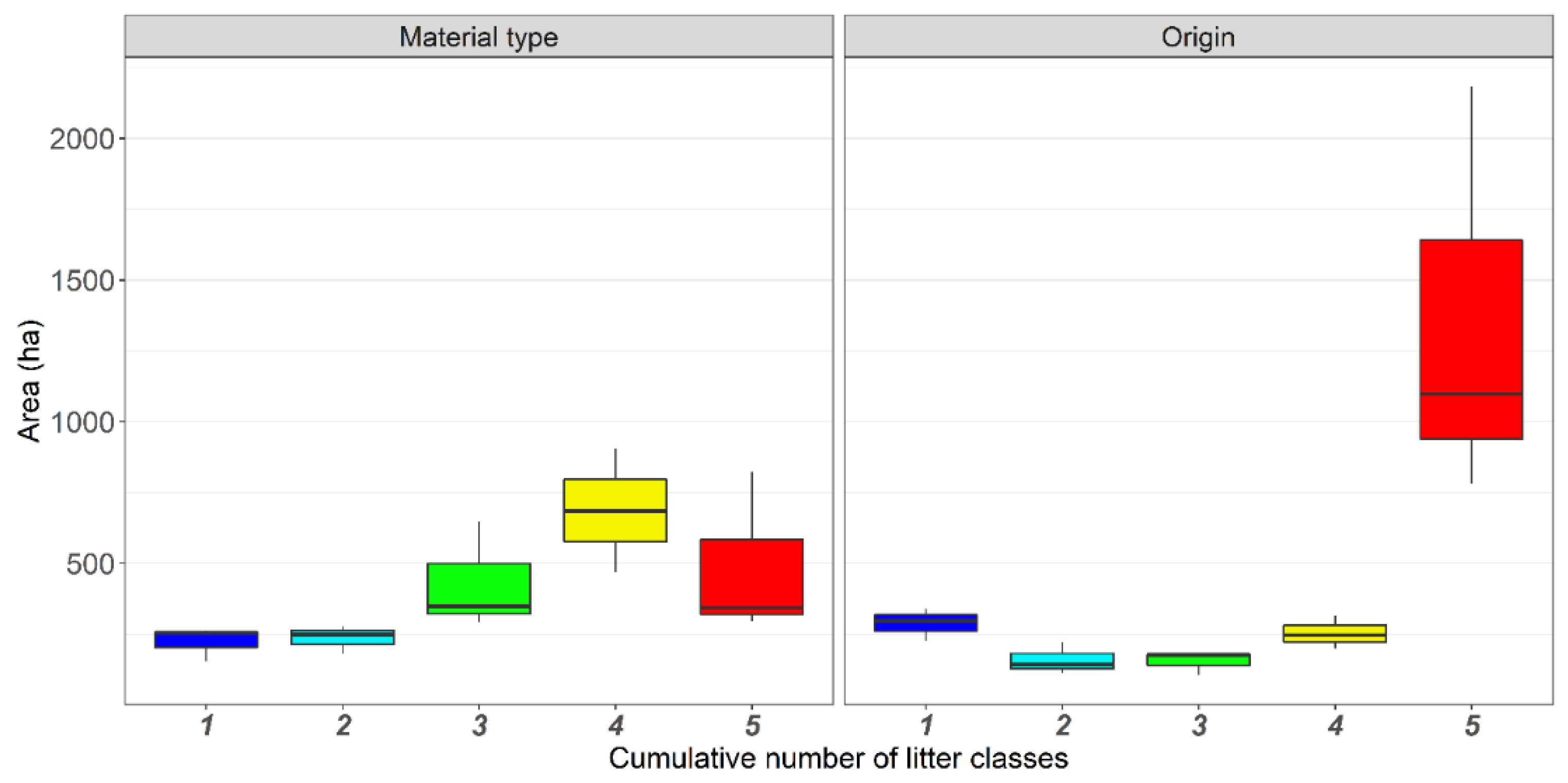
3.2. Hotspot Analysis
4. Discussion
4.1. Alternative Litter Classification Schemes Generate Different Predictive Accuracies
4.2. Geographical Factors Affecting Litter Accumulation Hotspots
4.3. Management Implications
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Appendix A. Checking for Significant Spatial Autocorrelation in LDM Residuals
Appendix A.1. Method
Appendix A.2. Results and Conclusions
References
- Vlachogianni, T.; Skocir, M.; Constantin, P.; Labbe, C.; Orthodoxou, D.; Pesmatzoglou, I.; Scannella, D.; Spika, M.; Zissimopoulos, V.; Scoullos, M. Plastic pollution on the Mediterranean coastline: Generating fit-for-purpose data to support decision-making via a participatory-science initiative. Sci. Total Environ. 2020, 711, 135058. [Google Scholar] [CrossRef] [PubMed]
- Barboza, L.G.A.; Dick Vethaak, A.; Lavorante, B.R.B.O.; Lundebye, A.K.; Guilhermino, L. Marine microplastic debris: An emerging issue for food security, food safety and human health. Mar. Pollut. Bull. 2018, 133, 336–348. [Google Scholar] [CrossRef] [PubMed]
- Fossi, M.C.; Vlachogianni, T.; Galgani, F.; Innocenti, F.D.; Zampetti, G.; Leone, G. Assessing and mitigating the harmful effects of plastic pollution: The collective multi-stakeholder driven Euro-Mediterranean response. Ocean Coast. Manag. 2020, 184, 105005. [Google Scholar] [CrossRef]
- Shen, M.; Mao, D.; Xie, H.; Li, C. The Social Costs of Marine Litter along the East China Sea: Evidence from Ten Coastal Scenic Spots of Zhejiang Province, China. Sustainability 2019, 11, 1807. [Google Scholar] [CrossRef]
- Gall, S.C.; Thompson, R.C. The impact of debris on marine life. Mar. Pollut. Bull. 2015, 92, 170–179. [Google Scholar] [CrossRef]
- Tabuenca, B.; Kalz, M.; Löhr, A. Massive open online education for environmental activism: The worldwide problem of marine litter. Sustainability 2019, 11, 2860. [Google Scholar] [CrossRef]
- Poeta, G.; Fanelli, G.; Pietrelli, L.; Acosta, A.T.R.; Battisti, C. Plastisphere in action: Evidence for an interaction between expanded polystyrene and dunal plants. Environ. Sci. Pollut. Res. 2017, 24, 11856–11859. [Google Scholar] [CrossRef]
- Good, T.P.; June, J.A.; Etnier, M.A.; Broadhurst, G. Derelict fishing nets in Puget Sound and the Northwest Straits: Patterns and threats to marine fauna. Mar. Pollut. Bull. 2010, 60, 39–50. [Google Scholar] [CrossRef]
- Galgani, F.; Hanke, G.; Maes, T. Global distribution, composition and abundance of marine litter. In Marine Anthropogenic Litter; Springer International Publishing: Cham, Switzerland, 2015; pp. 29–56. ISBN 9783319165103. [Google Scholar]
- UNEP/MAP. Integrated Monitoring and Assessment Guidance; UNEP/MAP: Athens, Greece, 2016. [Google Scholar]
- Battisti, C.; Poeta, G.; Pietrelli, L.; Acosta, A.T.R. An Unexpected Consequence of Plastic Litter Clean-Up on Beaches: Too Much Sand Might Be Removed. Environ. Pract. 2016, 18, 242–246. [Google Scholar] [CrossRef]
- Galgani, F.; Fleet, D.; Van Franeker, J.; Katsanevakis, S.; Maes, T.; Mouat, J.; Oosterbaan, L.; Poitou, I.; Hanke, G.; Thompson, R.; et al. Marine Strategy Framework Directive Task Group 10 Report: Marine Litte; European Commission: Ispra, VA, Italy; French Research Institute for Exploitation of the Sea: Issy-les-Moulineaux, France; International Council for the Exploration of the Sea Conseil International pour l’Exploration de la Mer: Copenhagen V, Denmark, 2010. [Google Scholar]
- OSPAR. Marine Litter in the North-East Atlantic Region: Assessment and Priorities for Response; OSPAR: London, UK, 2009. [Google Scholar]
- Vlachogianni, T. Marine Litter in Mediterranean Coastal and Marine Protected Areas—How Bad Is It. A Snapshot Assessment Report on the Amounts, Composition and Sources of Marine Litter Found on Beaches; UNEP/MAP: Athens, Greece, 2019. [Google Scholar]
- Ribic, C.A.; Sheavly, S.B.; Rugg, D.J.; Erdmann, E.S. Trends and drivers of marine debris on the Atlantic coast of the United States 1997–2007. Mar. Pollut. Bull. 2010, 60, 1231–1242. [Google Scholar] [CrossRef]
- de Francesco, M.C.; Carranza, M.L.; Varricchione, M.; Tozzi, F.P.; Stanisci, A. Natural protected areas as special sentinels of littering on coastal dune vegetation. Sustainability 2019, 11, 5446. [Google Scholar] [CrossRef]
- de Francesco, M.C.; Carranza, M.L.; Stanisci, A. Beach litter in Mediterranean coastal dunes: An insight on the Adriatic coast (central Italy). Rend. Lincei 2018, 29, 825–830. [Google Scholar] [CrossRef]
- Poeta, G.; Battisti, C.; Acosta, A.T.R. Marine litter in Mediterranean sandy littorals: Spatial distribution patterns along central Italy coastal dunes. Mar. Pollut. Bull. 2014, 89, 168–173. [Google Scholar] [CrossRef] [PubMed]
- Fortibuoni, T.; Amadesi, B.; Vlachogianni, T. Composition and abundance of macrolitter along the Italian coastline: The first baseline assessment within the European Marine Strategy Framework Directive. Environ. Pollut. 2021, 268, 115886. [Google Scholar] [CrossRef] [PubMed]
- Arcangeli, A.; David, L.; Aguilar, A.; Atzori, F.; Borrell, A.; Campana, I.; Carosso, L.; Crosti, R.; Darmon, G.; Gambaiani, D.; et al. Floating marine macro litter: Density reference values and monitoring protocol settings from coast to offshore. Results from the MEDSEALITTER project. Mar. Pollut. Bull. 2020, 160, 111647. [Google Scholar] [CrossRef]
- Browne, M.A. Sources and pathways of microplastics to habitats. In Marine Anthropogenic Litter; Springer: Cham, Switzerland, 2015; pp. 229–244. [Google Scholar]
- Turra, A.; Manzano, A.B.; Dias, R.J.S.; Mahiques, M.M.; Barbosa, L.; Balthazar-Silva, D.; Moreira, F.T. Three-dimensional distribution of plastic pellets in sandy beaches: Shifting paradigms. Sci. Rep. 2014, 4, 4435. [Google Scholar] [CrossRef] [PubMed]
- Topçu, E.N.; Tonay, A.M.; Dede, A.; Öztürk, A.A.; Öztürk, B. Origin and abundance of marine litter along sandy beaches of the Turkish Western Black Sea Coast. Mar. Environ. Res. 2013, 85, 21–28. [Google Scholar] [CrossRef] [PubMed]
- Keller, A.A.; Fruh, E.L.; Johnson, M.M.; Simon, V.; McGourty, C. Distribution and abundance of anthropogenic marine debris along the shelf and slope of the US West Coast. Mar. Pollut. Bull. 2010, 60, 692–700. [Google Scholar] [CrossRef] [PubMed]
- Munari, C.; Corbau, C.; Simeoni, U.; Mistri, M. Marine litter on Mediterranean shores: Analysis of composition, spatial distribution and sources in north-western Adriatic beaches. Waste Manag. 2016, 49, 483–490. [Google Scholar] [CrossRef] [PubMed]
- Araújo, M.C.; Costa, M. An analysis of the riverine contribution to the solid wastes contamination of an isolated beach at the Brazilian Northeast. Manag. Environ. Qual. An Int. J. 2007, 18, 6–12. [Google Scholar] [CrossRef]
- Poeta, G.; Conti, L.; Malavasi, M.; Battisti, C.; Acosta, A.T.R. Beach litter occurrence in sandy littorals: The potential role of urban areas, rivers and beach users in central Italy. Estuar. Coast. Shelf Sci. 2016, 181, 231–237. [Google Scholar] [CrossRef]
- Ariza, E.; Jiménez, J.A.; Sardá, R. Seasonal evolution of beach waste and litter during the bathing season on the Catalan coast. Waste Manag. 2008, 28, 2604–2613. [Google Scholar] [CrossRef] [PubMed]
- UNEP/MAP. Results of the Assessment of the Status of Marine Litter in the Mediterranean; UNEP/MAP: Kalamata, Greece, 2009. [Google Scholar]
- Vlachogianni, T.; Fortibuoni, T.; Ronchi, F.; Zeri, C.; Mazziotti, C.; Tutman, P.; Varezić, D.B.; Palatinus, A.; Trdan, Š.; Peterlin, M.; et al. Marine litter on the beaches of the Adriatic and Ionian Seas: An assessment of their abundance, composition and sources. Mar. Pollut. Bull. 2018, 131, 745–756. [Google Scholar] [CrossRef] [PubMed]
- Prevenios, M.; Zeri, C.; Tsangaris, C.; Liubartseva, S.; Fakiris, E.; Papatheodorou, G. Beach litter dynamics on Mediterranean coasts: Distinguishing sources and pathways. Mar. Pollut. Bull. 2018, 129, 448–457. [Google Scholar] [CrossRef] [PubMed]
- Núñez, P.; García, A.; Mazarrasa, I.; Juanes, J.A.; Abascal, A.J.; Méndez, F.; Castanedo, S.; Medina, R. A methodology to assess the probability of marine litter accumulation in estuaries. Mar. Pollut. Bull. 2019, 144, 309–324. [Google Scholar] [CrossRef]
- Mansui, J.; Molcard, A.; Ourmières, Y. Modelling the transport and accumulation of floating marine debris in the Mediterranean basin. Mar. Pollut. Bull. 2015, 91, 249–257. [Google Scholar] [CrossRef]
- Lebreton, L.C.M.; Greer, S.D.; Borrero, J.C. Numerical modelling of floating debris in the world’s oceans. Mar. Pollut. Bull. 2012, 64, 653–661. [Google Scholar] [CrossRef]
- Turrell, W.R. How litter moves along a macro tidal mid-latitude coast exposed to a coastal current. Mar. Pollut. Bull. 2020, 160, 111600. [Google Scholar] [CrossRef]
- Guisan, A.; Zimmermann, N.E. Predictive habitat distribution models in ecology. Ecol. Modell. 2000, 135, 147–186. [Google Scholar] [CrossRef]
- Gallardo, B.; Aldridge, D.C.; González-Moreno, P.; Pergl, J.; Pizarro, M.; Pyšek, P.; Thuiller, W.; Yesson, C.; Vilà, M. Protected areas offer refuge from invasive species spreading under climate change. Glob. Chang. Biol. 2017, 23, 5331–5343. [Google Scholar] [CrossRef]
- Innangi, M.; D’Alessandro, F.; Fioretto, A.; Di Febbraro, M.; D’Alessandro, F.; Fioretto, A.; Di Febbraro, M.; D’Alessandro, F.; Fioretto, A.; Di Febbraro, M. Modeling distribution of Mediterranean beech forests and soil carbon stock under climate change scenarios. Clim. Res. 2015, 66, 25–36. [Google Scholar] [CrossRef]
- Di Febbraro, M.; Sallustio, L.; Vizzarri, M.; De Rosa, D.; De Lisio, L.; Loy, A.; Eichelberger, B.A.; Marchetti, M. Expert-based and correlative models to map habitat quality: Which gives better support to conservation planning? Glob. Ecol. Conserv. 2018, 16, e00513. [Google Scholar] [CrossRef]
- Russo, L.F.; Barrientos, R.; Fabrizio, M.; Di Febbraro, M.; Loy, A. Prioritizing road-kill mitigation areas: A spatially explicit national-scale model for an elusive carnivore. Divers. Distrib. 2020, 26, 1093–1103. [Google Scholar] [CrossRef]
- Fabrizio, M.; Di Febbraro, M.; Loy, A. Where will it cross next? Optimal management of road collision risk for otters in Italy. J. Environ. Manag. 2019, 251, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Di Napoli, M.; Carotenuto, F.; Cevasco, A.; Confuorto, P.; Di Martire, D.; Firpo, M.; Pepe, G.; Raso, E.; Calcaterra, D. Machine learning ensemble modelling as a tool to improve landslide susceptibility mapping reliability. Landslides 2020, 17, 1897–1914. [Google Scholar] [CrossRef]
- Drius, M.; Jones, L.; Marzialetti, F.; de Francesco, M.C.; Stanisci, A.; Carranza, M.L. Not just a sandy beach. The multi-service value of Mediterranean coastal dunes. Sci. Total Environ. 2019, 668, 1139–1155. [Google Scholar] [CrossRef]
- Miccadei, E.; Mascioli, F.; Piacentini, T.; Ricci, F. Geomorphological features of coastal dunes along the Central Adriatic coast (Abruzzo, Italy). J. Coast. Res. 2011, 27, 1122–1136. [Google Scholar] [CrossRef]
- Aucelli, P.P.C.; Di Paola, G.; Rizzo, A.; Rosskopf, C.M. Present day and future scenarios of coastal erosion and flooding processes along the Italian Adriatic coast: The case of Molise region. Environ. Earth Sci. 2018, 77, 371. [Google Scholar] [CrossRef]
- Acosta, A.; Blasi, C.; Carranza, M.L.; Ricotta, C.; Stanisci, A. Quantifying ecological mosaic connectivity and hemeroby with a new topoecological index. Phytocoenologia 2003, 33, 623–631. [Google Scholar] [CrossRef]
- Bazzichetto, M.; Malavasi, M.; Acosta, A.T.R.; Carranza, M.L. How does dune morphology shape coastal EC habitats occurrence? A remote sensing approach using airborne LiDAR on the Mediterranean coast. Ecol. Indic. 2016, 71, 618–626. [Google Scholar] [CrossRef]
- Carranza, M.L.; Acosta, A.T.R.; Stanisci, A.; Pirone, G.; Ciaschetti, G. Ecosystem classification for EU habitat distribution assessment in sandy coastal environments: An application in central Italy. Environ. Monit. Assess. 2008, 140, 99–107. [Google Scholar] [CrossRef] [PubMed]
- Drius, M.; Malavasi, M.; Acosta, A.T.R.; Ricotta, C.; Carranza, M.L. Boundary-based analysis for the assessment of coastal dune landscape integrity over time. Appl. Geogr. 2013, 45, 41–48. [Google Scholar] [CrossRef]
- Malavasi, M.; Santoro, R.; Cutini, M.; Acosta, A.T.R.; Carranza, M.L. What has happened to coastal dunes in the last half century? A multitemporal coastal landscape analysis in Central Italy. Landsc. Urban Plan. 2013, 119, 54–63. [Google Scholar] [CrossRef]
- Carranza, M.L.; Drius, M.; Malavasi, M.; Frate, L.; Stanisci, A.; Acosta, A.T.R. Assessing land take and its effects on dune carbon pools. An insight into the Mediterranean coastline. Ecol. Indic. 2018, 85, 951–955. [Google Scholar] [CrossRef]
- Carboni, M.; Carranza, M.L.; Acosta, A. Assessing conservation status on coastal dunes: A multiscale approach. Landsc. Urban Plan. 2009, 91, 17–25. [Google Scholar] [CrossRef]
- Lippiatt, S.; Opfer, S.; Arthur, C. Marine Debris Monitoring and Assessment: Recommendations for Monitoring Debris Trends in the Marine Environment; NOAA Marine Debris Division: Silver Spring, MD, USA, 2013. [Google Scholar]
- OSPAR. Guideline for Monitoring Marine Litter on the Beaches in the OSPAR Maritime Area; OSPAR: London, UK, 2010. [Google Scholar]
- UNEP/MAP. Marine Litter Assessment in the Mediterranean; UNEP/MAP Athens: Nairobi, Kenya, 2015. [Google Scholar]
- Šilc, U.; Küzmič, F.; Caković, D.; Stešević, D. Beach litter along various sand dune habitats in the southern Adriatic (E Mediterranean). Mar. Pollut. Bull. 2018, 128, 353–360. [Google Scholar] [CrossRef]
- Zuur, A.F.; Ieno, E.N.; Elphick, C.S. A protocol for data exploration to avoid common statistical problems. Methods Ecol. Evol. 2010, 1, 3–14. [Google Scholar] [CrossRef]
- R Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2017. [Google Scholar]
- Phillips, S.S.J.; Anderson, R.P.R.; Schapire, R.E. Maximum entropy modeling of species geographic distributions. Ecol. Modell. 2006, 190, 231–259. [Google Scholar] [CrossRef]
- Merow, C.; Smith, M.J.; Silander, J.A. A practical guide to MaxEnt for modeling species’ distributions: What it does, and why inputs and settings matter. Ecography 2013, 36, 1058–1069. [Google Scholar] [CrossRef]
- Muscarella, R.; Galante, P.J.; Soley-Guardia, M.; Boria, R.A.; Kass, J.M.; Uriarte, M.; Anderson, R.P. ENMeval: An R package for conducting spatially independent evaluations and estimating optimal model complexity for Maxent ecological niche models. Methods Ecol. Evol. 2014, 5, 1198–1205. [Google Scholar] [CrossRef]
- Burnham, K.P.; Anderson, D. Model Selection and Multi-Model Inference: A Practical Information-Theoretic Approach; Springer: New York, NY, USA, 2002; ISBN 0387953647. [Google Scholar]
- Roberts, D.R.; Bahn, V.; Ciuti, S.; Boyce, M.S.; Elith, J.; Guillera-Arroita, G.; Hauenstein, S.; Lahoz-Monfort, J.J.; Schröder, B.; Thuiller, W.; et al. Cross-validation strategies for data with temporal, spatial, hierarchical, or phylogenetic structure. Ecography 2017, 40, 913–929. [Google Scholar] [CrossRef]
- Fourcade, Y.; Besnard, A.G.; Secondi, J. Paintings predict the distribution of species, or the challenge of selecting environmental predictors and evaluation statistics. Glob. Ecol. Biogeogr. 2018, 27, 245–256. [Google Scholar] [CrossRef]
- Hanley, J.A.; McNeil, B.J. The meaning and use of the area under a receiver operating characteristic (ROC) curve. Radiology 1982, 143, 29–36. [Google Scholar] [CrossRef] [PubMed]
- Warren, D.L.; Seifert, S.N. Ecological niche modeling in Maxent: The importance of model complexity and the performance of model selection criteria. Ecol. Appl. 2011, 21, 335–342. [Google Scholar] [CrossRef]
- Allouche, O.; Tsoar, A.; Kadmon, R. Assessing the accuracy of species distribution models: Prevalence, kappa and the true skill statistic (TSS). J. Appl. Ecol. 2006, 43, 1223–1232. [Google Scholar] [CrossRef]
- Swets, J.A. Measuring the accuracy of diagnostic systems. Science 1988, 240, 1285–1293. [Google Scholar] [CrossRef]
- Mesgaran, M.B.; Cousens, R.D.; Webber, B.L. Here be dragons: A tool for quantifying novelty due to covariate range and correlation change when projecting species distribution models. Divers. Distrib. 2014, 20, 1147–1159. [Google Scholar] [CrossRef]
- Liu, C.; Berry, P.; Dawson, T.; Pearson, R.G. Selecting thresholds of occurrence in the prediction of species distributions. Ecography 2005, 3, 385–393. [Google Scholar] [CrossRef]
- D’Amen, M.; Dubuis, A.; Fernandes, R.F.; Pottier, J.; Pellissier, L.; Guisan, A. Using species richness and functional traits predictions to constrain assemblage predictions from stacked species distribution models. J. Biogeogr. 2015, 42, 1255–1266. [Google Scholar] [CrossRef]
- Di Febbraro, M.; D’Amen, M.; Raia, P.; De Rosa, D.; Loy, A.; Guisan, A. Using macroecological constraints on spatial biodiversity predictions under climate change: The modelling method matters. Ecol. Modell. 2018, 390, 79–87. [Google Scholar] [CrossRef]
- Marzialetti, F.; Di Febbraro, M.; Malavasi, M.; Giulio, S.; Acosta, A.T.; Carranza, M.L. Mapping Coastal Dune Landscape through Spectral Rao’s Q Temporal Diversity. Remote Sens. 2020, 12, 2315. [Google Scholar] [CrossRef]
- Wright, M.N.; Ziegler, A. Ranger: A Fast Implementation of Random Forests for High Dimensional Data in C++ and R. J. Stat. Softw. 2017, 77, 1–17. [Google Scholar] [CrossRef]
- James, G.; Witten, D.; Hastie, T.; Tibshirani, R. An Introduction to Statistical Learning; Springer Texts in Statistics; Springer: New York, NY, USA, 2013; Volume 7, ISBN 978-1-4614-7137-0. [Google Scholar]
- Igras, K.; Biecek, P. Xspliner: Assisted Model Building, Using Surrogate Black-Box Models to Train Interpretable Spline Based Additive Models 2020. Available online: https://ModelOriented.github.io/xspliner/ (accessed on 1 September 2020).
- Landis, J.R.; Koch, G.G. The measurement of observer agreement for categorical data. Biometrics 1977, 33, 159–174. [Google Scholar] [CrossRef] [PubMed]
- Kusui, T.; Noda, M. International survey on the distribution of stranded and buried litter on beaches along the Sea of Japan. Mar. Pollut. Bull. 2003, 47, 175–179. [Google Scholar] [CrossRef]
- Williams, A.T.; Tudor, D.T. Litter burial and exhumation: Spatial and temporal distribution on a cobble pocket beach. Mar. Pollut. Bull. 2001, 42, 1031–1039. [Google Scholar] [CrossRef]
- Menicagli, V.; Balestri, E.; Vallerini, F.; Castelli, A.; Lardicci, C. Adverse effects of non-biodegradable and compostable plastic bags on the establishment of coastal dune vegetation: First experimental evidences. Environ. Pollut. 2019, 252, 188–195. [Google Scholar] [CrossRef] [PubMed]
- Bazzichetto, M.; Sperandii, M.G.; Malavasi, M.; Carranza, M.L.; Acosta, A.T.R. Disentangling the effect of coastal erosion and accretion on plant communities of Mediterranean dune ecosystems. Estuar. Coast. Shelf Sci. 2020, 241, 106758. [Google Scholar] [CrossRef]
- Maun, M.A. Burial of Plants as a Selective Force in Sand Dunes. In Coastal Dunes; Springer: Berlin/Heidelberg, Germany, 2008; pp. 119–135. [Google Scholar]
- Garcias, P.J.; Tavecchia, G. Apparent Survival and Long-Term Population Growth Rate of the Kentish Plover Charadrius alexandrinus in Mallorca, Archipelago, Spain. Ardeola 2018, 65, 59–68. [Google Scholar] [CrossRef]
- Berardo, F.; Carranza, M.L.; Frate, L.; Stanisci, A.; Loy, A. Seasonal habitat preference by the flagship species Testudo hermanni: Implications for the conservation of coastal dunes. Comptes Rendus Biol. 2015, 338, 343–350. [Google Scholar] [CrossRef]
- Crosti, R.; Arcangeli, A.; Campana, I.; Paraboschi, M.; González-Fernández, D. ‘Down to the river’: Amount, composition, and economic sector of litter entering the marine compartment, through the Tiber river in the Western Mediterranean Sea. Rend. Lincei 2018, 29, 859–866. [Google Scholar] [CrossRef]
- Gabrielides, G.P.; Golik, A.; Loizides, L.; Marino, M.G.; Bingel, F.; Torregrossa, M.V. Man-made garbage pollution on the Mediterranean coastline. Mar. Pollut. Bull. 1991, 23, 437–441. [Google Scholar] [CrossRef]
- Jayasiri, H.B.; Purushothaman, C.S.; Vennila, A. Quantitative analysis of plastic debris on recreational beaches in Mumbai, India. Mar. Pollut. Bull. 2013, 77, 107–112. [Google Scholar] [CrossRef] [PubMed]
- Rodríguez-Rodríguez, D. Littering in protected areas: A conservation and management challenge—A case study from the Autonomous Region of Madrid, Spain. J. Sustain. Tour. 2012, 20, 1011–1024. [Google Scholar] [CrossRef]
- Katsanevakis, S.; Katsarou, A. Influences on the distribution of marine debris on the seafloor of shallow coastal areas in Greece (eastern Mediterranean). Water. Air. Soil Pollut. 2004, 159, 325–337. [Google Scholar] [CrossRef]
- Martinez-Ribes, L.; Basterretxea, G.; Palmer, M.; Tintoré, J. Origin and abundance of beach debris in the Balearic Islands. Sci. Mar. 2007, 71, 305–314. [Google Scholar] [CrossRef]
- Laglbauer, B.J.L.; Franco-Santos, R.M.; Andreu-Cazenave, M.; Brunelli, L.; Papadatou, M.; Palatinus, A.; Grego, M.; Deprez, T. Macrodebris and microplastics from beaches in Slovenia. Mar. Pollut. Bull. 2014, 89, 356–366. [Google Scholar] [CrossRef] [PubMed]
- Gonçalves, G.; Andriolo, U.; Pinto, L.; Bessa, F. Mapping marine litter using UAS on a beach-dune system: A multidisciplinary approach. Sci. Total Environ. 2020, 706, 135742. [Google Scholar] [CrossRef]
| Material Type | Items | N. Plot | % Plot |
|---|---|---|---|
| PLASTIC | Bottles cups, pull tabs plastic, plastic bottles, plastic drums, fishing nets plastic, plastic plates, plastic forks, plastic bags, plastic sheets, soap containers, snack cards, straws, food trays, packaging of medicines, monofilament lines. | 113 | 38.0 |
| POLYSTYRENE | Polystyrene boxes, polystyrene cups. | 88 | 29.6 |
| GLASS | Glass bottles. | 19 | 6.4 |
| ALUMINUM | Drink cans. | 13 | 4.4 |
| MIXED MATERIALS | Cigarette butts, lighters, fluorescent light tubes, light globes, processed timber, rags, clothing, shoes, hats, tableware, toys, tires and inner tubes, rubber/chewing gum, wires, building materials, nappies, cotton buds, syringes, plasters, sanitary pads, foams, strapping bands, buoys, fishing nets not plastic, fishing related, ropes. | 59 | 19.9 |
| Origin | |||
| CONTAINERS | Bottle cups, pull tabs plastic, plastic bottles ≤ 2 L, plastic bottles > 2 L, plastic drums > 2 L, glass bottles. | 78 | 22.1 |
| FISHING AND BOATING | Buoys, fishing nets not plastic, fishing nets plastic, fishing related, monofilament lines, ropes, polystyrene boxes. | 100 | 28.3 |
| FOOD AND BEVERAGE | Drink and food packages, cups, food trays, drink cans, ice cream sticks, chip forks, plastic plates, straws, snack cards, chips bags. | 48 | 13.6 |
| PACKAGING | Foams, papers and cardboard, plastic bags, plastic sheets, strapping bends, soap containers. | 50 | 14.2 |
| OTHER | Fluorescent light tubes, light globes, processed timber, rags, clothing, shoes, hats, tableware, toys, tyres and inner tubes, rubber/chewing gum, wires, building materials. Cigarette packaging, cigarette butts, cigarettes lighters. Sanitary packaging, nappies, cotton buds, syringes, plasters, packaging of medicines, sanitary pads. | 77 | 21.8 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Di Febbraro, M.; Frate, L.; de Francesco, M.C.; Stanisci, A.; Tozzi, F.P.; Varricchione, M.; Carranza, M.L. Modelling Beach Litter Accumulation on Mediterranean Coastal Landscapes: An Integrative Framework Using Species Distribution Models. Land 2021, 10, 54. https://doi.org/10.3390/land10010054
Di Febbraro M, Frate L, de Francesco MC, Stanisci A, Tozzi FP, Varricchione M, Carranza ML. Modelling Beach Litter Accumulation on Mediterranean Coastal Landscapes: An Integrative Framework Using Species Distribution Models. Land. 2021; 10(1):54. https://doi.org/10.3390/land10010054
Chicago/Turabian StyleDi Febbraro, Mirko, Ludovico Frate, Maria Carla de Francesco, Angela Stanisci, Francesco Pio Tozzi, Marco Varricchione, and Maria Laura Carranza. 2021. "Modelling Beach Litter Accumulation on Mediterranean Coastal Landscapes: An Integrative Framework Using Species Distribution Models" Land 10, no. 1: 54. https://doi.org/10.3390/land10010054
APA StyleDi Febbraro, M., Frate, L., de Francesco, M. C., Stanisci, A., Tozzi, F. P., Varricchione, M., & Carranza, M. L. (2021). Modelling Beach Litter Accumulation on Mediterranean Coastal Landscapes: An Integrative Framework Using Species Distribution Models. Land, 10(1), 54. https://doi.org/10.3390/land10010054









