Observations and Prediction of Recovered Quality of Desalinated Seawater in the Strategic ASR Project in Liwa, Abu Dhabi
Abstract
:1. Introduction
2. Materials and Methods
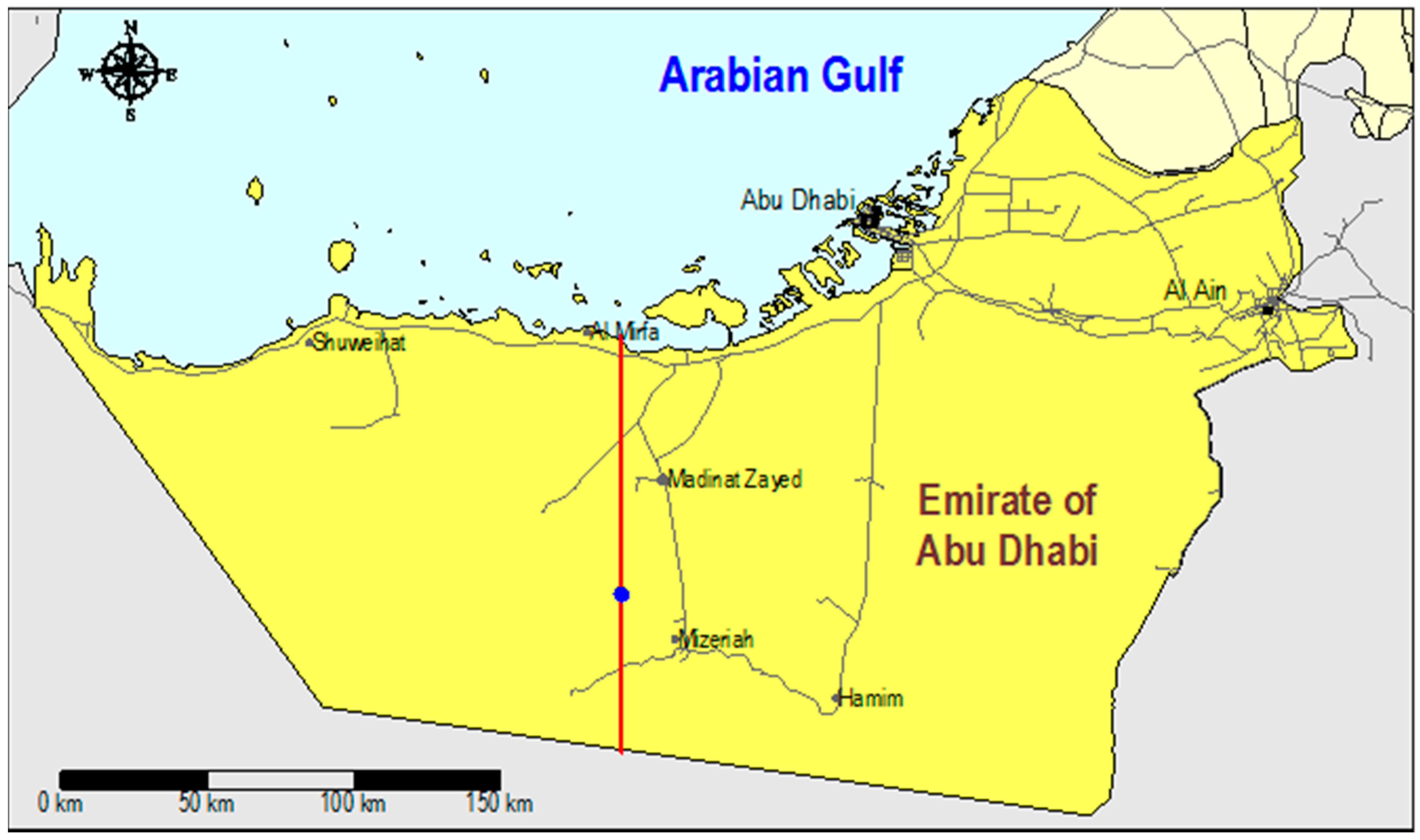
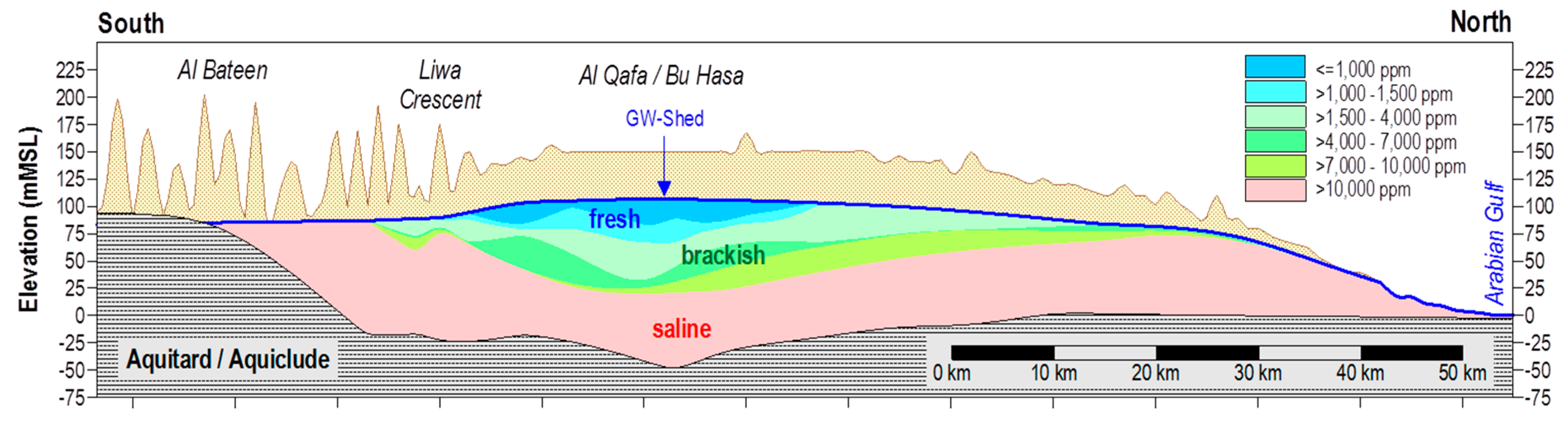
2.1. Field Site and Liwa ASR System
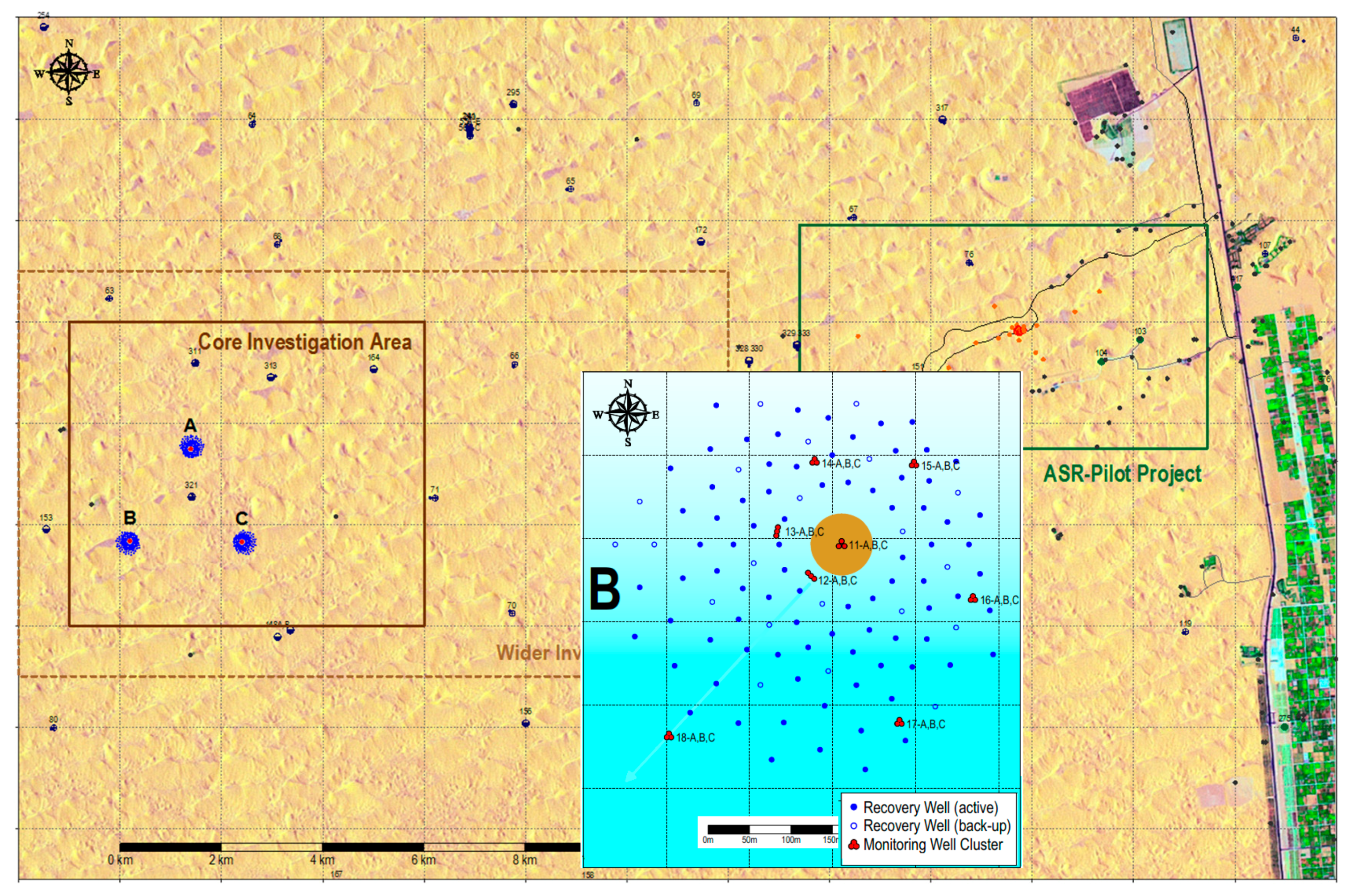
2.2. The Pilot
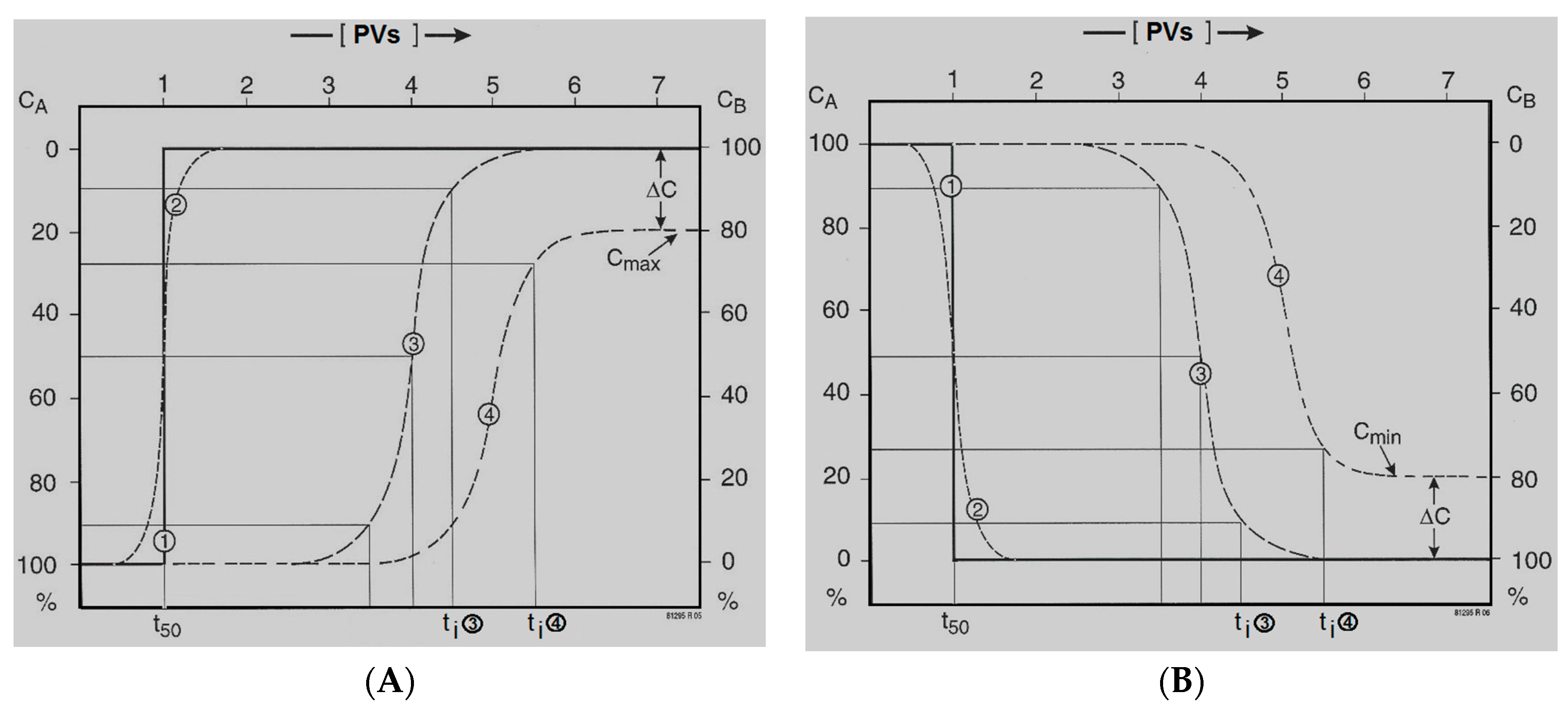
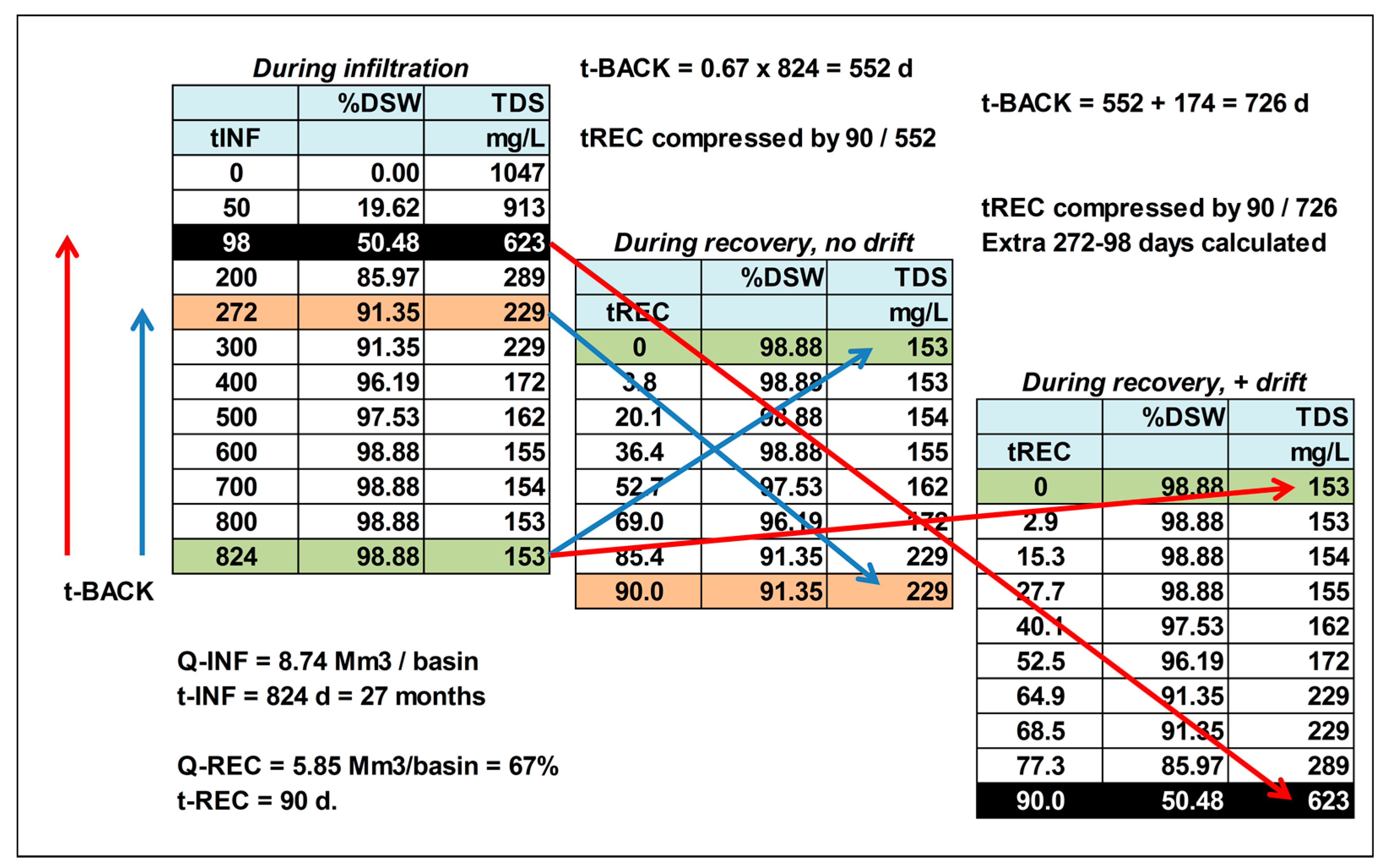
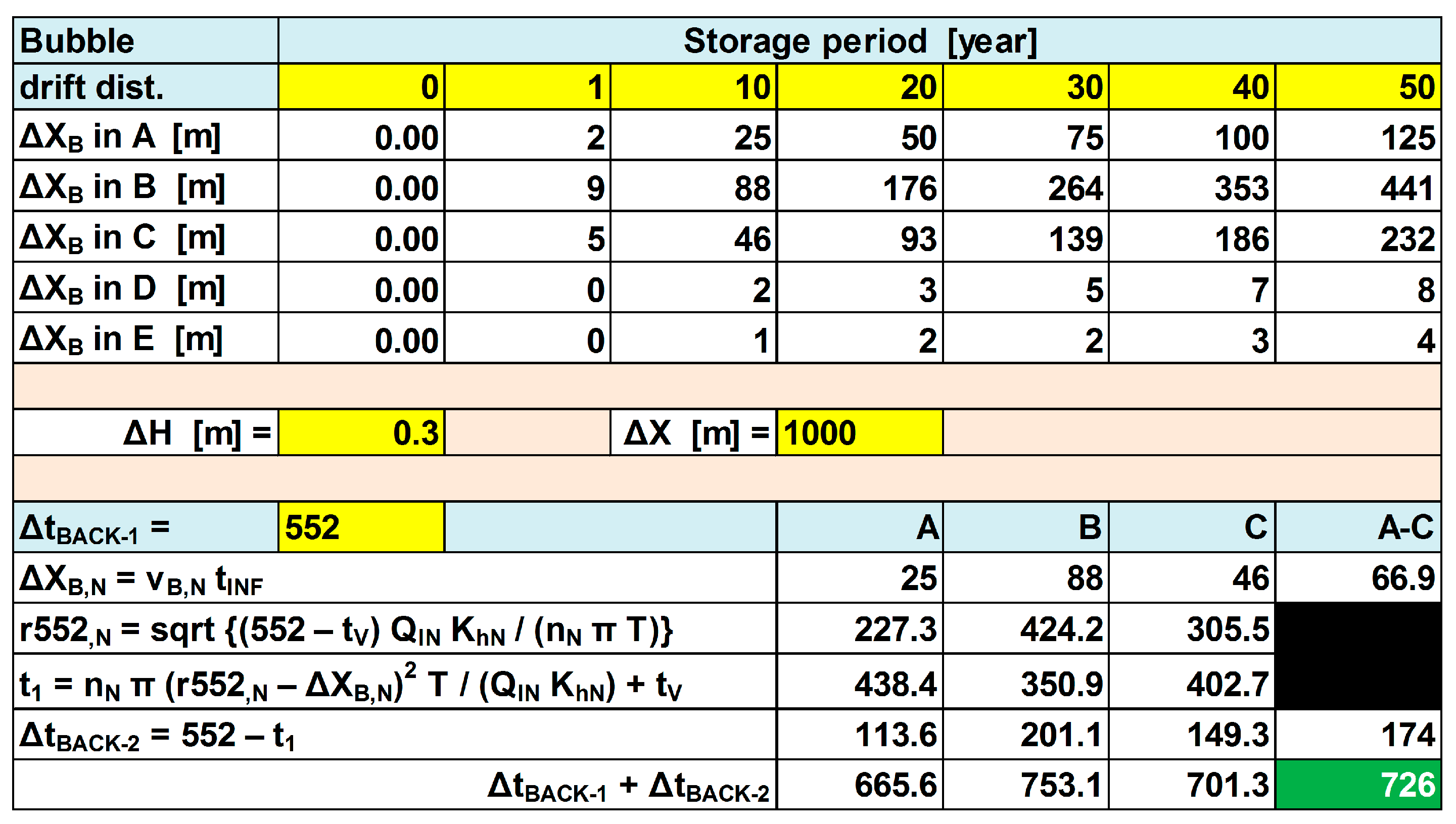
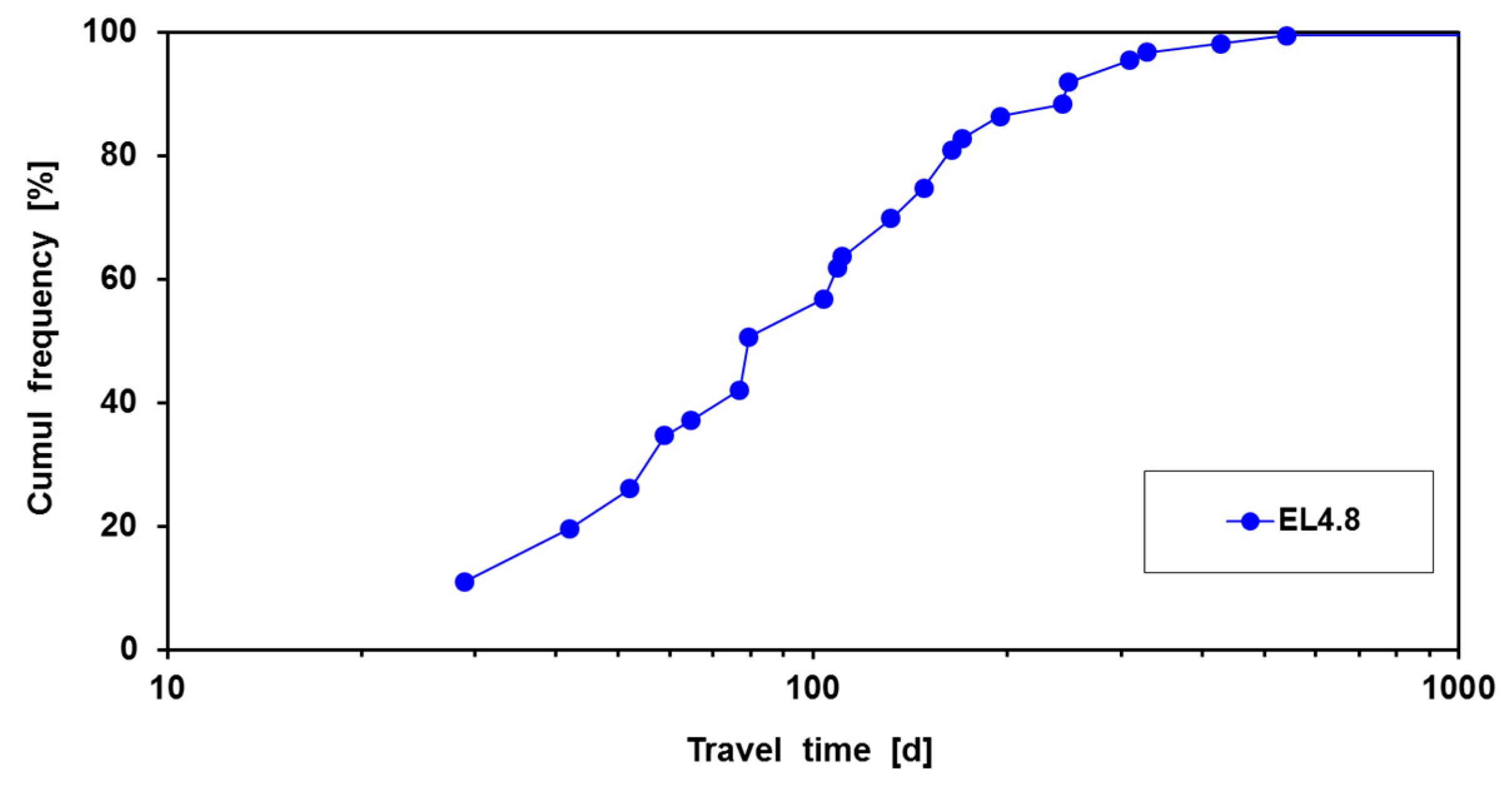
2.3. Quantitative Description of the Break-through Curve
2.4. Lithological and Geochemical Stratification
2.5. Hydrochemical Analyses in Period 2003–2013
2.6. Monitoring Campaign in August 2014
2.7. Predictions by EL Modeling
3. Results of Hydrogeological, Geochemical and Hydrochemical Stratification Analysis
3.1. Hydrogeology
3.2. Geochemistry
3.3. Chemistry of Native Groundwater
4. Results of Sampling Campaign in August 2014
5. Results of Pilot
- There are hardly any changes of O2, Cl, SO4, HCO3, TIC, NO3 and NH4, even after six years of storage. This indicates that redox reactions were practically nonexistent, and carbonates (calcite and dolomite) hardly dissolved or precipitated.
- A small TDS increase (20–30 mg/L) by dissolution of mainly SiO2, K and possibly Mg. Carbonate dissolution was noticed only where a very strong Ca/Na exchange was taking place (see next point).
- Ca/Na exchange in which Ca concentrations declined and Na increased was observed. This was especially important for samples close to the DSW intrusion front and typical for fresh water intrusion.
- Mobilization of the trace elements As, B, Ba, F, Cr, Sr and V from the aquifer, through desorption and/or mineral dissolution was observed. The strongest and most persistent increase is noticed for Sr, which does not exactly match Ca behavior, so that another mineral could be the source. Of the trace elements mentioned, Ba shows the smallest mobilization, whereas As, F and Cr show a significant leaching by DSW, which is also important from a drinking water quality perspective.
- Immobilization of PO4, Al, Cu, Fe and Ni during aquifer passage was mainly by sorption of PO4, Cu and Ni, and filtration of suspended colloidal particles of Al and Fe.
- Calculated mineral saturation indices show that the infiltrated water is more or less in equilibrium with calcite and dolomite as expected. The values for gypsum, barite and fluorite indicate a strong undersaturation.
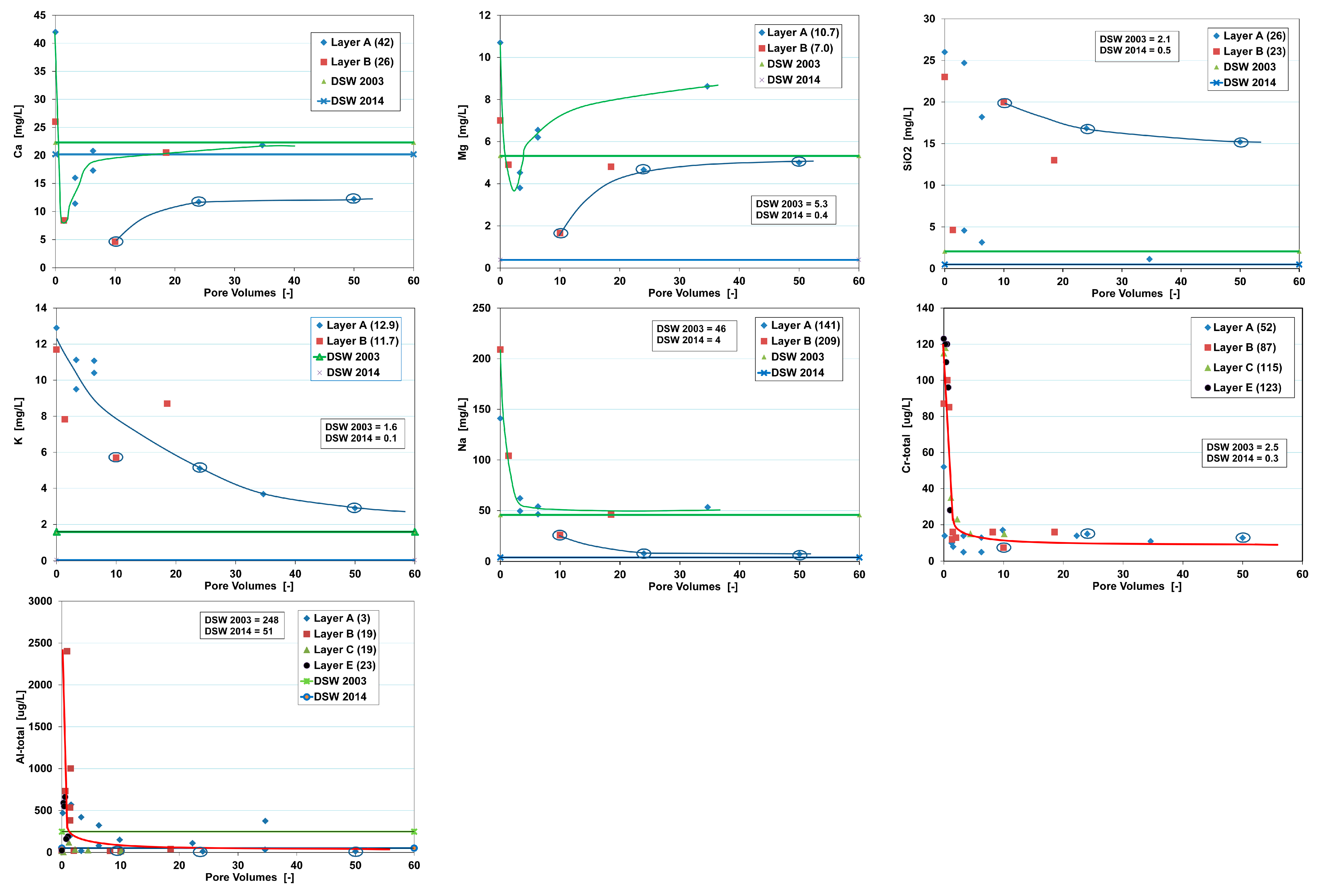
- Ca, Mg and Sr (Ca and Mg shown in Figure 9) concentrations start low and with increasing PVs rise to an asymptotic value. All 2014 data plot below those of 2003–2004, which is partly explained by the lower DSW input in 2008. Only Ca and Mg show an initial concentration below DSW input, which is probably linked to Na exchange, with Ca developing an asymptotic value at or below DSW input. Contrary, Mg and Sr approach an asymptotic value little (Mg) or far (Sr) above DSW input. Obviously, Mg and even more so Sr are dissolved from the aquifer, and Ca is not. Sources of Mg and Sr could be dolomite and/or silicate minerals.
- SiO2, K and V concentrations (SiO2 and K shown in Figure 9) decline in an exponential way, but relatively slowly compared to the next group, and the low concentration asymptote is also relatively high and in most cases far above DSW input. This suggests that these elements are initially desorbed and that later on they could be dissolving from the aquifer, possibly from quartz (SiO2), K-feldspar (K and SiO2) or for instance pyroxenes (SiO2 and V). A long storage time yields much higher concentrations for SiO2, pointing at slow dissolution kinetics. Vanadium is likely present as VO43−, which could desorb from iron (hydr)oxide coatings.
- Na, As, B, Cr, F and Mo (Na and Cr shown in Figure 9) concentrations also decline in an exponential way, but much more rapidly than the previous group. The low concentration asymptote is relatively high and in any case significantly above DSW input, especially for As, B and Cr. The asymptote approaches DSW input better for Na, F and Mo. This suggests that all these elements are very rapidly desorbed and that later on especially As, B and Cr could be steadily dissolving from the aquifer matrix. Except for Na and B (as H3BO3), this group is composed of anions, As as AsO43−, Cr as CrO42−, F as F− and Mo as MoO42−. The source mineral of chromate and vanadate could be pyroxenes [23].
- Al, Fe and Ni concentrations (Al shown in Figure 9) decline in an exponential way, approximately as rapidly as the previous group. Their concentration seems to be dictated by colloidal particles that become initially mobilized. On the one hand, lack of filtration may have contributed to this. On the other hand, it is well known [32], that clay minerals in aquifers tend to be mobilized by deflocculation when the sodium adsorption ratio (SAR) of the infiltration water and of the native groundwater is high, their salinity low, the clay mineral content high, and the dominant type of clay minerals unfavorable (smectite > illite > kaolinite). The SAR value of the native groundwater seems to be more important than the one of the infiltration water. With SAR values of ~20, the risk of mechanical clogging of the aquifer when the particles strand in the pore necks is relatively high. This risk is estimated higher in aquifer layer C than in A and B because SAR of native groundwater is higher and permeability is lower.
6. Results of Predicting the Quality of Recovered Water
6.1. Model Settings
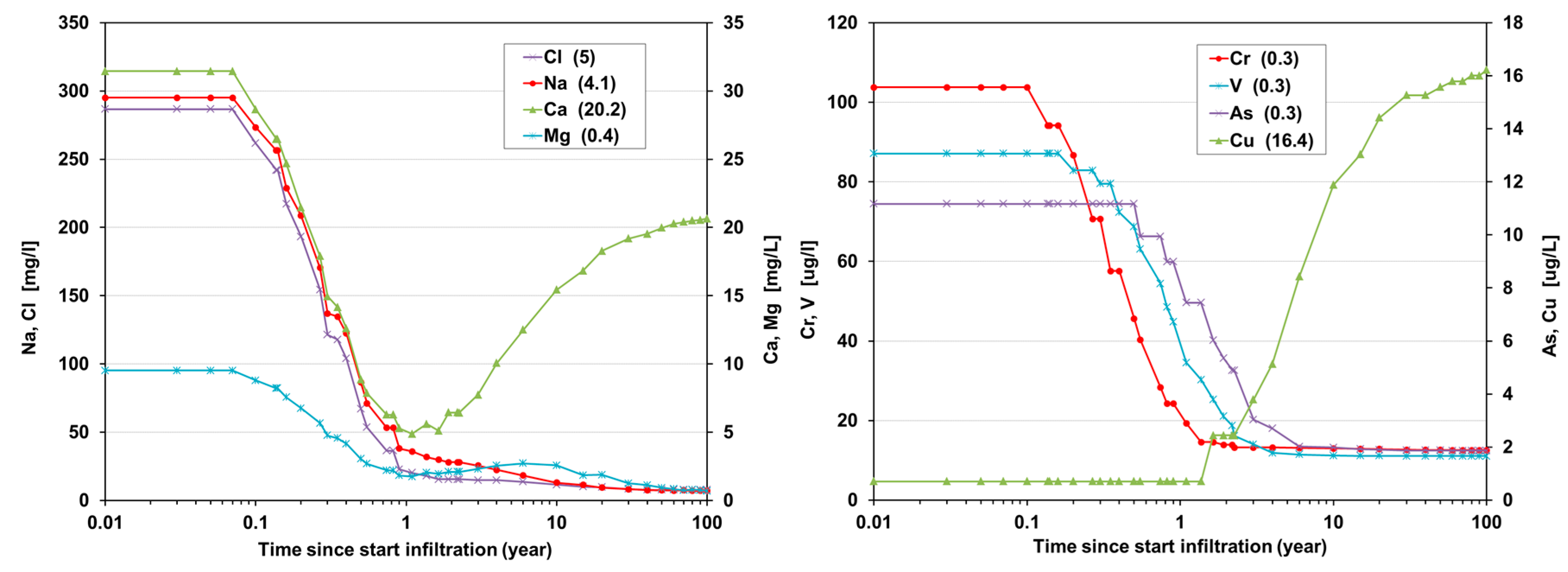
6.2. Quality Evolution during Nonstop Infiltration
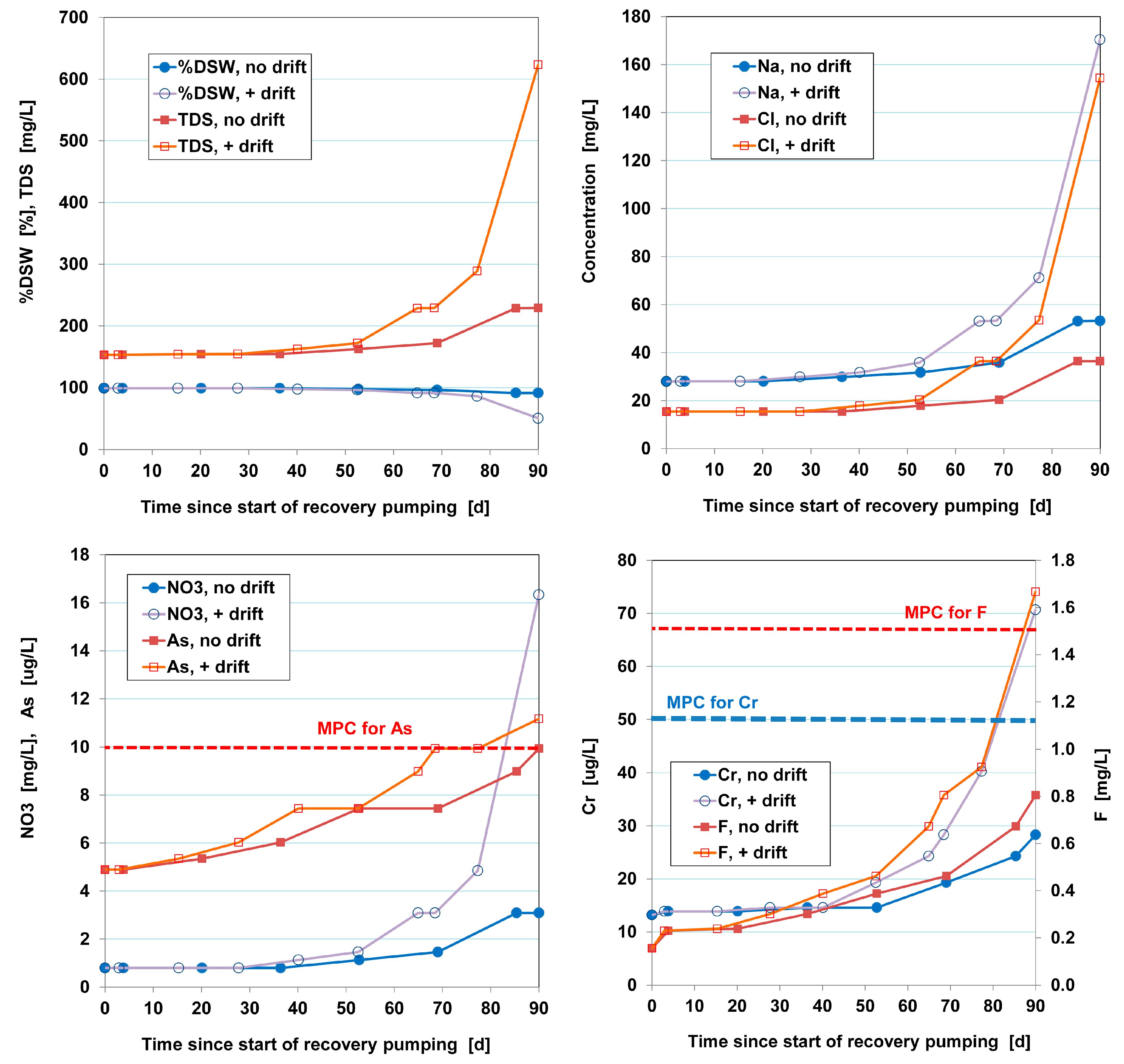
6.3. Quality Evolution during Recovery
7. Discussion
8. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
Abbreviations
| ΔC | (semi)permanent concentration change |
| ΔH/ΔX | mean regional hydraulic gradient in aquifer [-] |
| ΔtBACK-1 | setback time without horizontal bubble drift [d] |
| ρs | density of solids of porous medium [kg/L] |
| ASL | above sea level |
| ASR | Aquifer Storage and Recovery |
| BOM | bulk organic material in aquifer |
| BSL | below sea level |
| BTC | break through curve |
| CEC | cation exchange capacity |
| DSW | desalinated seawater |
| Kd | distribution coefficient [L/kg] |
| Kh,N | horizontal hydraulic conductivity of layer N [m/d] |
| Li | leach factor for compound i |
| MPC | maximum permissible concentration |
| MSL | mean sea level |
| n | porosity [L/L] |
| nN | porosity of layer N [-] |
| ORP | oxidation reduction potential |
| (prod) | concentration of reaction product in fluid during leaching [mmol/L] |
| PV | pore volume |
| QIN | mean infiltration rate [m3/d] |
| r | radial distance from the basin center [m] |
| r552,N | radial distance in aquifer layer N, to ASR well after 552 d recovery |
| (reac) | concentration of reactant in flushing fluid [mmol/L] |
| Ri | retardation factor for compound i |
| rP | reaction coefficient related to (prod) [-]. |
| rR | reaction coefficient related to (reac) [-] |
| (solid) | content of reactive phase in aquifer [mmol/kg dry weight] |
| T | transmissivity of the aquifer [m2/d] |
| t50 | observed 50% breakthrough time of conservative tracer or calculated travel time |
| TDS | total dissolved solids |
| ti | time for ≥90% break-through (Ri) or ≥90% leaching (Li) or till equilibrium [days] |
| tINF | total infiltration period [d] |
| tN | 50% break-through time in layer N [d] |
| TOC | total organic carbon |
| tV | vertical travel time [d] |
| vB,N | horizontal bubble drift in layer N [m/d] |
References
- Saif, O. The Future Outlook of Desalination in the Gulf: Challenges & Opportunities Faced by Qatar & the UAE. 2012. Available online: http://inweh.unu.edu/wp-content/uploads/2015/05/The-Future-Outlook-of-Desalination-in-the-Gulf1.pdf (accessed on 2 January 2017).
- Al Shehhi, M.R.; Gherboudj, I.; Ghedira, H. An overview of historical harmful algae blooms outbreaks in the Arabian Seas. Mar. Pollut. Bull. 2014, 86, 314–324. [Google Scholar] [CrossRef] [PubMed]
- Do Rosário Gomes, H.; Goes, J.L.; Matondkar, S.G.P.; Buskey, E.J.; Basu, S.; Parab, S.; Thoppil, P. Massive outbreaks of Noctiluca scintillans blooms in the Arabian Sea due to spread of hypoxia. Nat. Commun. 2014, 5. [Google Scholar] [CrossRef] [PubMed]
- Missimer, T.M.; Sinha, S.; Ghaffour, N. Strategic Aquifer Storage and Recovery of Desalinated Water to Achieve Water Security in the GCC/MENA Region. Int. J. Environ. Sustain. 2012, 1, 87–99. Available online: https://www.sciencetarget.com/Journal/index.php/IJES/article/viewFile/93/26 (accessed on 2 January 2017). [Google Scholar] [CrossRef]
- Hutchinson, C.B. Simulation of Aquifer Storage Recovery of Excess Desalinated Seawater; U.S. Geological Survey Open-File Report 98-410; USGS: Al Ain area, Abu Dhabi Emirate, 1998. Available online: https://pubs.usgs.gov/of/1998/0410/report.pdf (accessed on 2 January 2017).
- Pyne, R.D.G.; Howard, J.B. Desalination/aquifer storage recovery (DASR): A cost effective combination for Corpus Christi, Texas. Desalination 2004, 165, 363–367. [Google Scholar] [CrossRef]
- Dawoud, M.A.; Sallam, O.M. Sustainable Groundwater Resources Management in Arid Regions: Abu Dhabi Case Study; Environmental Agency Abu Dhabi Report; Environmental Agency Abu Dhabi: Abu Dhabi, United Arab Emirates, 2010.
- German Technical Cooperation Agency. Combined Artificial Recharge and Utilisation of the Groundwater Resource in the Greater Liwa Area (Western Region of the Emirate of Abu Dhabi); Pilot Project Final Technical Report; Deutsche Gesellschaft für Technische Zusammenarbeit (GTZ) GmbH in Co-operation with Dornier Consulting (DCo) GmbH: Bonn, Germany, 2005. [Google Scholar]
- German Technical Cooperation Agency. Detailed Design Groundwater Modelling Final Report; Consultancy Services for Artificial Recharge and Utilisation of the Groundwater Resource in the Liwa Area, Consultancy Contract No. G 4877; Deutsche Gesellschaft für Technische Zusammenarbeit (GTZ) GmbH in Co-operation with Dornier Consulting (DCo) GmbH: Bonn, Germany, 2009. [Google Scholar]
- German Technical Cooperation Agency. Detailed Design Drilling, Construction and Testing of Recovery and Monitoring Wells Draft Report; Consultancy Services for Artificial Recharge and Utilisation of the Groundwater Resource in the Liwa Area, Consultancy Contract No. G 4877; Deutsche Gesellschaft für Technische Zusammenarbeit (GTZ) GmbH in Co-operation with Dornier Consulting (DCo) GmbH: Bonn, Germany, 2009; p. 56. [Google Scholar]
- Earth Link & Advanced Resources Development. Hydrogeological Study and Numerical Groundwater Simulation; Construction Phase Strategic Water Storage and Recovery Project in Liwa Abu Dhabi—United Arab Emirates; Quarterly Report #6 Hydrogeological Structure and Groundwater Flow & Transport Model of the SWSR Region; Earth Link and Advanced Resources Development Sarl: Amaret Chalhoub, Lebanon, 2013. [Google Scholar]
- Al Futtaim Exova LLC. Test Report of Core Samples; 4 Reports on Respectively Well No: MW 01C, MW11C, MW21 and MW42; Al Futtaim Exova LLC, Lab: Dubai, United Arab Emirates, 2011–2012. [Google Scholar]
- Water Quality Regulations 2013-Final Draft Consultation; Regulation and Supervision Bureau Abu Dhabi: Abu Dhabi, United Arab Emirates, 2013.
- Stuyfzand, P.J. Quality changes upon injection into anoxic aquifers in the Netherlands: Evaluation of 11 experiments. In Artificial Recharge of Groundwater, Proceedings of the 3rd International Symposium on Artificial Recharge, Amsterdam, The Netherlands, 21–25 September 1998; Peters, J.H., Ed.; Balkema: Amsterdam, The Netherlands, 1998; pp. 283–291. [Google Scholar]
- Stuyfzand, P.J.; Timmer, H. Deep Well Injection at the Langerak and Nieuwegein sites in the Netherlands: Chemical Reactions and Their Modeling; Kiwa-SWE 99.006; KWR Watercycle Research Institute: Nieuwegein, The Netherlands, 1999; 44p. [Google Scholar]
- Stuyfzand, P.J. Pyrite oxidation and side-reactions upon deep well injection. WRI-10. In Proceedings of the 10th International Symposium on Water Rock Interaction, Villasimius, Italy, 10–15 June 2001; Volume 2, pp. 1151–1154.
- Prommer, H.; Stuyfzand, P.J. Identification of temperature-dependent water quality changes during a deep well injection experiment in a pyritic aquifer. Environ. Sci. Technol. 2005, 39, 2200–2209. [Google Scholar] [CrossRef] [PubMed]
- Antoniou, E.A.; van Breukelen, B.M.; Putters, B.; Stuyfzand, P.J. Hydrogeochemical patterns, processes and mass transfers during aquifer storage & recovery (ASR) in an anoxic sandy aquifer. Appl. Geochem. 2012, 27, 2435–2452. [Google Scholar]
- Zuurbier, K.G.; Hartog, N.; Stuyfzand, P.J. Reactive transport impacts on recovered freshwater quality during multiple partially penetrating wells (MPPW-)ASR in a brackish heterogeneous aquifer. Appl. Geochem. 2016, 71, 35–47. [Google Scholar] [CrossRef]
- Pyne, R.D.G. Aquifer Storage Recovery—A Guide to Groundwater Recharge through Wells; ASR Systems LLC: Gainesville, FL, USA, 2005. [Google Scholar]
- Price, R.E.; Pichler, T. Abundance and mineralogical association of arsenic in the Suwannee Limestone (Florida): Implications for arsenic release during water-rock interaction. Chem. Geol. 2006, 228, 44–56. [Google Scholar] [CrossRef]
- Wallis, I.; Prommer, H.; Simmons, C.T.; Post, V.; Stuyfzand, P.J. Evaluation of Conceptual and Numerical Models for Arsenic Mobilization and Attenuation during Managed Aquifer Recharge. Environ. Sci. Technol. 2010, 44, 5035–5041. [Google Scholar] [CrossRef] [PubMed]
- Stuyfzand, P.J.; Smidt, E.; Zuurbier, K.G. Baseline Hydrogeochemistry of the Strategic Water Storage and Recovery Project in Liwa, Abu Dhabi; KWR-Report KWR2014.073; KWR Watercycle Research Institute: Nieuwegein, The Netherlands, 2014. [Google Scholar]
- Stuyfzand, P.J.; Hartog, N.; Smidt, E. Prediction of Recovered Water Quality of the Strategic ASR Project in Liwa, Abu Dhabi; KWR-Report KWR2015.003; KWR Watercycle Research Institute: Nieuwegein, The Netherlands, 2014. [Google Scholar]
- Stuyfzand, P.J. Simple models for reactive transport of pollutants and main constituents during artificial recharge and bank filtration. In Artificial Recharge of Groundwater, Proceedings of the 3rd Internatioanl Symposium on Artificial Recharge, Amsterdam, The Netherlands, 21–25 September 1998; Peters, J.H., Ed.; Balkema: Amsterdam, The Netherlands, 1998; pp. 427–434. [Google Scholar]
- Stuyfzand, P.J. Hydrogeochemcal (HGC 2.1), for Storage, Management, Control, Correction and Interpretation of Water Quality Data in Excel (R) Spread Sheet; KWR-Report BTO2012.244(s); Updated in 2016 December; KWR Watercycle Research Institute: Nieuwegein, The Netherlands, 2016; 92p. [Google Scholar]
- Parkhurst, D.L.; Appelo, C.A.J. User’s Guide to PHREEQC (Version 2): A Computer Program for Speciation, Batch-Reaction, One-Dimensional Transport, and Inverse Geochemical Calculations; Water-Resources Investigations Report 99-4259.; Open-File Reports Section (Distributor); U.S. Geological Survey: Reston, VA, USA; Earth Science Information Center: Denver, CO, USA, 1999.
- Wood, W.W.; Clark, D.; Imes, J.L.; Councell, T.B. Eolian Transport of Geogenic Hexavalent Chromium to Ground Water. Ground Water 2010, 48, 19–29. [Google Scholar] [CrossRef] [PubMed]
- Wood, W.W.; Imes, J.L. Dating of Holocene groundwater recharge in the Rub al Khali of Abu Dhabi: Constraints on global climate-change models. In Water Resources Perspectives: Evaluation, Management and Policy; Developments in Water Science 50; Alsharhan, A.S., Wood, W.W., Eds.; Elsevier: Amsterdam, The Netherlands, 2003; pp. 379–385. [Google Scholar]
- Bottomley, N. Recent climate in Abu Dhabi. In Desert Ecology of Abu Dhabi; Osborne, P.E., Ed.; Pisces Publications: Newbury, UK, 1996; pp. 36–49. [Google Scholar]
- World Health Organization. Guidelines for Drinking-Water Quality, 4th ed.; World Health Organization: Geneva, Switzerland, 2011; Available online: http://apps.who.int/iris/bitstream/10665/44584/1/9789241548151_eng.pdf (accessed on 15 February 2017).
- Scheuerman, R.F.; Bergersen, B.M. Injection-water salinity, formation pretreatment, and well operations fluid-selection guidelines. J. Pet. Technol. 1990, 42, 836–845. [Google Scholar] [CrossRef]
- Al-Rahman, W.; Maraqa, M. Laboratory investigation of transport and treatment of chromium in groundwater at liwa district, Abu Dhabi. In Proceedings of the 1st International Conference on Natural Resources Engineering & Technology, Putrajaya, Malaysia, 24–25 July 2006; pp. 338–345.
- Yolcubal, İ.; Akyol, N.H. Retention and Transport of Hexavalent Chromium in Calcareous Karst Soils. Turk. J. Earth Sci. 2007, 16, 363–379. [Google Scholar]
- Richard, F.C.; Bourg, A.C.M. Aqueous geochemistry of chromium: A review. Water Res. 1991, 25, 807–816. [Google Scholar] [CrossRef]
- Siever, R. Silica solubility, 0–200 °C., and the diagenesis of siliceous sediments. J. Geol. 1992, 70, 127–150. [Google Scholar] [CrossRef]
- Ahmad, A.; Kools, S.; Schriks, M.; Stuyfzand, P.J.; Hofs, B. Arsenic and Chromium Concentrations and Their Speciation in Groundwater Resources and Drinking Water Supply in The Netherlands; KWR Report BTO 2015.017; KWR Watercycle Research Institute: Nieuwegein, The Netherlands, 2015. [Google Scholar]


| Layer No. | Top m ASL | Base m ASL | Quartz | Calcite | Dolomite | Feldspar | Clay Min. | Others | BOM | Gypsum | Apatite mg/kg | pH-H2O | CEC meq/kg |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| % d.w. | |||||||||||||
| A | 140 | 96 | 67.8 | 10.3 | 4.0 | 10.6 | 4.0 | 1.3 | 1.9 | <0.1 | 0 | 8.10 | 61.3 |
| B | 96 | 80 | 83.0 | 2.9 | 2.3 | 9.2 | 1.5 | 1.7 | 0.5 | <0.1 | 123 | 8.12 | 19.0 |
| C | 80 | 62 | 71.8 | 6.2 | 2.5 | 10.6 | 1.6 | 1.7 | 0.9 | <0.1 | 154 | 8.18 | 28.1 |
| D | 62 | 56 | 47.0 | 19.9 | 18.3 | 6.0 | 18.2 | 1.3 | 0.5 | <0.1 | 956 | 8.00 | 136.7 |
| E | 56 | −14 | 54.6 | 13.9 | 12.8 | 5.4 | 17.1 | 1.6 | 1.6 | 0.1 | 83 | 7.89 | 146.6 |
| F | −14 | <−40 | 0.9 | 16.6 | 80.1 | 0.0 | 0.2 | 6.9 | 1.8 | 2.7 | 0 | 7.90 | 31.5 |
| Sample | Layer # Figure 5 | a | b | c | d | e-Top | e-Base | Abu Dh. Drinking w. Standard | WHO 2011 Guidelines | |
|---|---|---|---|---|---|---|---|---|---|---|
| Depth | m ASL | 140 | 96 | 80 | 62 | 56 | 35 | |||
| 96 | 80 | 62 | 56 | 35 | −14 | |||||
| General | O2 | mg/L | 0.9 | 2.0 | 2.2 | 2.3 | 2.5 | 0.0 | >2 | |
| Temp. | °C | 34.0 | 34.1 | 34.5 | 34.5 | 34.8 | 31.0 | |||
| EC 20 °C | μS/cm | 969 | 1180 | 2132 | 4095 | 5650 | 17,647 | 1600 | ||
| TDS | mg/L | 682 | 803 | 1545 | 2957 | 4042 | 13,288 | 1000 | ||
| pH | lab | 7.83 | 8.25 | 8.09 | 7.92 | 7.94 | 7.40 | 7−9.2 | ||
| Main constituents | Cl | mg/L | 187 | 233 | 396 | 978 | 1506 | 5500 | 250 | |
| SO4 | 147 | 152 | 397 | 702 | 844 | 3137 | 250 | |||
| HCO3 | 81 | 101 | 154 | 148 | 144 | 39 | >60 | |||
| NO3 | 25.0 | 30.8 | 37.4 | 41.7 | 42.6 | 96.0 | 50 | 50 | ||
| PO4 | 0.022 | 0.034 | 0.029 | 0.028 | 0.030 | 0.015 | 2.2 | |||
| F | 1.29 | 1.50 | 3.61 | 3.29 | 2.39 | 0.75 | 1.5 | 1.5 | ||
| Na | 141 | 209 | 466 | 877 | 1204 | 3238 | 150 | |||
| K | 12.9 | 11.7 | 11.5 | 15.8 | 18.5 | 83.7 | 12 | |||
| Ca | 41.7 | 26.2 | 29.8 | 107.3 | 174.8 | 821.0 | 80 | |||
| Mg | 10.7 | 7.0 | 11.2 | 41.1 | 67.0 | 340.0 | 30 | |||
| Fe | 0.005 | 0.023 | 0.022 | 0.012 | 0.019 | <0.44 | 0.2 | |||
| Mn | 0.004 | 0.008 | 0.008 | 0.020 | 0.037 | 0.030 | 0.4 | |||
| NH4 | 0.015 | 0.025 | 0.019 | 0.015 | 0.015 | 0.035 | 0.5 | |||
| SiO2 | 26.2 | 23.1 | 28.4 | 27.8 | 23.6 | 14.3 | ||||
| DOC | 0.3 | 1.0 | 0.6 | 1.5 | 2.6 | 5.3 | 1 | |||
| Trace elements | Al | μg/L | 3 | 19 | 19 | 19 | 23 | <290 | 200 | |
| As | 2.1 | 11.4 | 9.6 | 6.0 | 4.7 | <10 | 10 | 10 | ||
| B | 605 | 774 | 1113 | 1330 | 1421 | 1340 | 2400 | 2400 | ||
| Ba | 40 | 38 | 31 | 33 | 36 | 23 | 700 | 700 | ||
| Br | 579 | 668 | 788 | 762 | 767 | 301 | ||||
| Cd | 0.08 | 0.05 | 0.05 | 0.06 | 0.08 | <10 | 3 | 3 | ||
| Co | 0.06 | 0.08 | 0.07 | 0.44 | 0.82 | |||||
| Cr-tot | 52 | 87 | 115 | 129 | 123 | <286 | 50 | 50 | ||
| Cr(VI) | 48 | 85 | 108 | 119 | 117 | |||||
| Cu | 0.3 | 0.7 | 0.9 | 0.3 | 0.3 | <1 | 1000 | 2000 | ||
| Hg | 0.02 | 0.23 | 0.13 | 0.02 | 0.04 | 6 | 6 | |||
| Mo | 6 | 11 | 39 | 47 | 39 | <10 | ||||
| Ni | 0.5 | 2.5 | 1.6 | 1.0 | 1.6 | 70 | 70 | |||
| Sb | <1 | 2.2 | 1.2 | <1 | <1 | 20 | 20 | |||
| Se | 3.2 | 3.2 | 3.3 | 4.1 | 5.2 | 17.0 | 40 | 40 | ||
| Sr | 3485 | 2846 | 7381 | 8486 | 6573 | 1700 | ||||
| Ti | <0.5 | <0.5 | <0.5 | 0.6 | 0.7 | |||||
| U | 0.6 | 0.7 | 3.0 | 3.1 | 3.1 | 30 | ||||
| V | 29 | 77 | 95 | 71 | 49 | <10 | ||||
| Zn | 3 | 16 | 7 | 2 | 2 | <50 | 5000 | |||
| Ratio’s | Cl/Br | mg/L-basis | 323 | 348 | 503 | 1284 | 1963 | 18297 | ||
| Ca/Sr | 12.0 | 9.2 | 4.0 | 12.6 | 26.6 | 482.9 | ||||
| Ca/Mg | 3.9 | 3.8 | 2.7 | 2.6 | 2.6 | 2.4 | ||||
| Mineral Sat. | Barite | Saturation Index | −0.12 | −0.13 | −0.14 | −0.16 | −0.20 | −0.50 | ||
| Calcite | −0.06 | 0.15 | 0.13 | 0.10 | 0.13 | −0.15 | ||||
| Dolomite | −0.26 | 0.18 | 0.30 | 0.29 | 0.39 | −0.15 | ||||
| Fluorite | −0.82 | −0.95 | −0.29 | −0.37 | −0.68 | −0.82 | ||||
| Gypsum | −1.72 | −1.96 | −1.70 | −1.42 | −1.43 | −0.02 | ||||
| TIC | mmol/L | 1.38 | 1.71 | 2.59 | 2.51 | 2.43 | 0.68 | |||
| Loc | Unit | First Pilot 2003-4 | Second Pilot 2008 (2014) | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| RB-01 | DSW | RB-01 | RB-02 | OB-01A | OB-07B | DSW | OB-01A | OB-07B | |||
| Ambient | Input | Recovery Wells | Obs. Wells | Input | Obs. Wells | ||||||
| Space & time | Top screen | m ASL | 93 | 121 | 93 | 91 | 104 | 100 | 121 | 104 | 100 |
| Base screen | m ASL | 75 | 121 | 75 | 73 | 95 | 91 | 121 | 95 | 91 | |
| Radial distance | m | 83 | 0.0 | 83 | 30 | 44 | 90 | 0 | 44 | 90 | |
| t50 | d | 0.0 | 11 | 50 | 0 | 11 | 50 | ||||
| PVs | - | 0.0 | 0.0 | >3 | >10 | 6.3 | 1.4 | 0.0 | 50.0 | 10.0 | |
| Digital date | year | 2003.70 | 2004.21 | 2004.35 | 2004.57 | 2003.94 | 2003.94 | 2014.59 | 2014.60 | 2014.60 | |
| Main composition | O2 | mg/L | 4.9 | 7.2 | 8.0 | 7.2 | 6.8 | 7.2 | |||
| EC 20 °C, lab | μS/cm | 1313 | 397 | 487 | 432 | 413 | 512 | 123 | 134 | 142 | |
| Temperature | °C | 31.0 | 35.0 | 31.0 | 31.0 | 31.0 | 31.0 | 35.0 | 36.4 | 35.8 | |
| pH-Lab | - | 8.94 | 7.90 | 9.11 | 7.69 | 8.57 | 8.50 | 8.11 | 8.43 | 8.90 | |
| Na | mg/L | 266.0 | 45.7 | 107.0 | 57.2 | 46.3 | 104.0 | 4.1 | 7.4 | 25.9 | |
| K | mg/L | 6.1 | 1.6 | 2.7 | 8.1 | 10.4 | 7.8 | <0.1 | 2.9 | 5.7 | |
| Ca | mg/L | 7.3 | 22.3 | 1.8 | 21.0 | 17.3 | 8.4 | 20.2 | 12.2 | 4.6 | |
| Mg | mg/L | 2.5 | 5.3 | 0.5 | 3.9 | 6.2 | 4.9 | 0.4 | 5.0 | 1.7 | |
| Fe | mg/L | 0.130 | 0.255 | 0.050 | 0.150 | 0.080 | 0.683 | 0.012 | 0.005 | 0.005 | |
| Mn | mg/L | 0.040 | 0.008 | 0.050 | <0.01 | 0.020 | 0.008 | 0.003 | 0.003 | 0.003 | |
| NH4 | mg NH4/L | <0.1 | <0.1 | <0.1 | <0.1 | <0.1 | <0.03 | <0.03 | <0.03 | ||
| SiO2 | mg SiO2/L | 27.3 | 2.1 | 24.0 | 24.0 | 18.2 | 4.6 | <1 | 15.2 | 20.0 | |
| Cl | mg/L | 216.0 | 80.8 | 80.3 | 78.0 | 83.1 | 89.5 | 5.0 | 10.0 | 11.0 | |
| SO4 | mg/L | 162.0 | 11.7 | 10.7 | 10.8 | 13.4 | 22.0 | 1.0 | <2 | 2.0 | |
| HCO3 | mg/L | 152 | 60 | 123 | 104 | 62 | 139 | 66 | 66 | 59 | |
| TIC | mmol/L | 2.64 | 1.01 | 2.16 | 1.78 | 1.04 | 2.33 | 1.11 | 1.11 | 1.01 | |
| NO3 | mg NO3/L | 23.5 | <0.5 | 0.0 | 0.0 | <0.5 | <0.5 | <1 | <1 | <1 | |
| NO2 | mg NO2/L | 0.01 | <0.01 | <0.01 | 0.004 | <0.01 | <0.01 | <0.01 | |||
| PO4-total | mg PO4/L | 0.21 | 0.06 | 0.02 | |||||||
| PO4-ortho | mg PO4/L | 0.021 | 0.062 | 0.021 | 0.358 | 0.060 | <0.03 | <0.03 | |||
| F | mg/L | 2.10 | 0.05 | 0.20 | 0.10 | 0.10 | 3.51 | <0.05 | 0.06 | 0.26 | |
| DOC | mg/L | 8.4 | <0.5 | <0.5 | <0.5 | ||||||
| Trace elements | Al | μg/L | 180 | 248 | 510 | 200 | 80 | 535 | 51 | 13 | 13 |
| As | μg/L | 11.0 | 0.0 | <3 | <0.5 | 2.2 | 8.4 | ||||
| B | μg/L | 850 | 29 | 58 | 52 | 52 | 17 | 56 | 55 | ||
| Ba | μg/L | 13 | 5 | 30 | 6 | 16 | 20 | 2 | 12 | 3 | |
| Br | μg/L | 570 | 20 | 301 | 301 | 140 | 161 | 186 | |||
| Cr | μg/L | 95.0 | 2.5 | 17.0 | 15.0 | 13.0 | <0.5 | 12.9 | 7.4 | ||
| Cu | μg/L | 5.0 | 20.3 | 2.0 | 4.0 | <2 | <2 | 16.4 | <0.5 | <0.5 | |
| Mo | μg/L | 10.0 | <3 | <3 | <3 | <3 | <3 | <1 | <1 | <1 | |
| Ni | μg/L | 3.0 | 9.5 | <3 | <3 | <3 | <3 | 4.3 | <1 | <1 | |
| Se | μg/L | <2 | <2 | 3.0 | 3.0 | <2 | <0.5 | <0.5 | <0.5 | ||
| Sr | μg/L | 1200 | 50 | 200 | 1600 | 1300 | 278 | 16 | 1000 | 274 | |
| V | μg/L | 66 | 1.5 | 140 | 36 | 17 | <0.5 | 12 | 64 | ||
| Zn | μg/L | 10 | 81 | 59 | 98 | 4 | 5 | 3 | <2 | <2 | |
| Mineral saturation | SI-B | Barite | −0.5 | −1.8 | −1.0 | −1.7 | −1.2 | −0.9 | −2.6 | −2.4 | −2.7 |
| SI-C | Calcite | 0.5 | −0.2 | 0.1 | −0.3 | 0.3 | 0.2 | 0.0 | 0.2 | 0.0 | |
| Si-D | Dolomite | 0.9 | −0.6 | 0.0 | −0.9 | 0.5 | 0.6 | −1.2 | 0.4 | 0.0 | |
| SI-G | Gypsum | −2.4 | −2.9 | −4.0 | −2.9 | −2.9 | −3.1 | −3.2 | −4.1 | −4.2 | |
| SI-B | Barite | −0.5 | −1.8 | −1.0 | −1.7 | −1.2 | −0.9 | −2.6 | −2.4 | −2.7 | |
| SI-F | Fluorite | −1.1 | −3.7 | −3.6 | −3.1 | −3.2 | −0.5 | −4.3 | −3.8 | −2.9 | |
| Parameter | L | R | ΔC [mg/L] | Parameter | L | R | ΔC [ug/L] | |||
|---|---|---|---|---|---|---|---|---|---|---|
| 2003-4 | 2014 | 2003-4 | 2014 | |||||||
| Na | 1.5–2 | -- | 3 | 3.5 | F | 1.5–2 | -- | 0.2 | 0.04 | |
| K | 20 | -- | 3 | 3 | Al | 1.5 | -- | −130 | −39 | |
| Ca | -- | 3.5 | 0 | −8 | As | 3 | -- | 1 | 2 | |
| Mg | -- | 3 | 3.3 | 4.5 | B | 2 | -- | 10 | 30 | |
| Fe | 2 | -- | 0 | 0 | Ba | -- | -- | 10 | 10 | |
| SiO2 | 2–4 | -- | 0.6 | 14.5 | Cr | 1.5–2 | -- | 10 | 13 | |
| Cl | 1 | -- | 0 | 0 | Mo | 1.5 | -- | 0 | 0 | |
| SO4 | 1 | -- | 0 | 0 | Ni | 1.5 | -- | -8 | −4 | |
| HCO3 | 1 | -- | 0 | 0 | Sr | -- | 3.5 | 1600 | 984 | |
| NO3 | 1 | -- | 0 | 0 | V | 5 | -- | 10 | 11 | |
| PO4-O | -- | -- | −0.04 | −0.05 | Zn | -- | -- | −60 | −1.5 | |
| Sample | Parameter | Unit | Native | RECOVERED WATER ‘SAMPLED’ AFTER | Input | Abu Dh. Drink. w. Standard | WHO Guide-Lines | |||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Measured | 0.1 | 0.27 | 0.74 | 2.26 | 10 | 100 | DSW | |||||
| at ABC | Year | Year | Year | Year | Year | Year | 2014 | |||||
| General | EC 20 °C | μS/cm | 1411 | 1304 | 829 | 295 | 199 | 184 | 170 | 123 | 1600 | |
| Temp. | °C | 34.2 | 34.2 | 34.4 | 34.8 | 35.0 | 35.0 | 35.0 | 35.0 | |||
| pH | 8.13 | 8.13 | 8.20 | 8.35 | 8.44 | 8.44 | 8.45 | 8.10 | 7–9.2 | |||
| O2 | mg/L | 1.9 | 2.5 | 4.5 | 6.7 | 7.1 | 7.1 | 7.1 | 7.2 | >2 | ||
| CH4 | mg/L | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 | |||
| DOC | mg/L | 0.8 | 0.8 | 0.6 | 0.4 | 0.3 | 0.3 | 0.3 | 0.3 | 1 | ||
| SI-calcite | 0.19 | 0.15 | 0.00 | −0.30 | −0.19 | 0.18 | 0.33 | 0.00 | ||||
| Main constituents | Cl | mg/L | 287.0 | 261.6 | 154.5 | 36.5 | 15.4 | 11.5 | 7.8 | 5.0 | 250 | |
| SO4 | mg/L | 235.0 | 217.8 | 130.7 | 28.0 | 8.3 | 5.9 | 3.6 | 1.0 | 250 | ||
| HCO3 | mg/L | 116 | 112 | 93 | 71 | 67 | 67 | 67 | 66 | >60 | ||
| NO3 | mg/L | 32.3 | 29.0 | 16.3 | 3.1 | 0.8 | 0.7 | 0.6 | 0.3 | 50 | 50 | |
| PO4 | mg/L | 0.03 | 0.03 | 0.03 | 0.03 | 0.05 | 0.16 | 0.21 | 0.21 | 2.2 | ||
| Na | mg/L | 295.0 | 273.2 | 170.4 | 53.3 | 28.0 | 13.1 | 7.2 | 4.1 | 150 | ||
| K | mg/L | 12.4 | 11.1 | 6.8 | 2.0 | 0.7 | 0.3 | 0.2 | 0.1 | 12 | ||
| Ca | mg/L | 31.5 | 28.7 | 17.9 | 6.3 | 6.4 | 15.4 | 20.6 | 20.2 | 80 | ||
| Mg | mg/L | 9.5 | 8.8 | 5.7 | 2.2 | 2.1 | 2.6 | 0.7 | 0.4 | 30 | ||
| NH4 | mg/L | 0.02 | 0.02 | 0.02 | 0.02 | 0.01 | 0.00 | 0.00 | 0.02 | 0.5 | ||
| Fe | mg/L | 0.02 | 0.02 | 0.02 | 0.02 | 0.02 | 0.01 | 0.00 | 0.012 | 0.2 | ||
| Mn | mg/L | 0.01 | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 | 0.003 | 0.4 | ||
| SiO2 | mg/L | 25.2 | 25.2 | 25.2 | 25.2 | 23.0 | 14.4 | 14.4 | 0.5 | |||
| Trace elements | As | μg/L | 11.2 | 11.2 | 11.2 | 9.9 | 4.9 | 2.0 | 1.9 | 0.3 | 10 | 10 |
| B | μg/L | 884 | 884 | 736 | 311 | 65 | 48 | 44 | 17 | 2400 | 2400 | |
| Ba | μg/L | 46 | 46 | 46 | 46 | 46 | 31 | 14 | 2 | 700 | 700 | |
| Cr | μg/L | 104.0 | 103.7 | 70.7 | 28.3 | 13.2 | 13.0 | 12.6 | 0.30 | 50 | 50 | |
| Cu | μg/L | 0.7 | 0.7 | 0.7 | 0.7 | 2.4 | 11.9 | 16.2 | 16.4 | 1000 | 2000 | |
| F | μg/L | 2190 | 2113 | 1667 | 895 | 173 | 101 | 92 | 30 | 1.5 | 1.5 | |
| Ni | μg/L | 2.0 | 2.0 | 2.0 | 2.0 | 2.2 | 3.4 | 4.3 | 4.3 | 70 | 70 | |
| Sr | μg/L | 4397 | 4397 | 4397 | 4397 | 4397 | 2889 | 228 | 16 | |||
| V | μg/L | 87 | 87 | 83 | 54 | 16 | 11 | 11 | 0.3 | |||
| Zn | μg/L | 12.0 | 11.6 | 11.6 | 11.6 | 11.6 | 5.4 | 3.0 | 3.0 | 5000 | ||
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Stuyfzand, P.J.; Smidt, E.; Zuurbier, K.G.; Hartog, N.; Dawoud, M.A. Observations and Prediction of Recovered Quality of Desalinated Seawater in the Strategic ASR Project in Liwa, Abu Dhabi. Water 2017, 9, 177. https://doi.org/10.3390/w9030177
Stuyfzand PJ, Smidt E, Zuurbier KG, Hartog N, Dawoud MA. Observations and Prediction of Recovered Quality of Desalinated Seawater in the Strategic ASR Project in Liwa, Abu Dhabi. Water. 2017; 9(3):177. https://doi.org/10.3390/w9030177
Chicago/Turabian StyleStuyfzand, Pieter J., Ebel Smidt, Koen G. Zuurbier, Niels Hartog, and Mohamed A. Dawoud. 2017. "Observations and Prediction of Recovered Quality of Desalinated Seawater in the Strategic ASR Project in Liwa, Abu Dhabi" Water 9, no. 3: 177. https://doi.org/10.3390/w9030177
APA StyleStuyfzand, P. J., Smidt, E., Zuurbier, K. G., Hartog, N., & Dawoud, M. A. (2017). Observations and Prediction of Recovered Quality of Desalinated Seawater in the Strategic ASR Project in Liwa, Abu Dhabi. Water, 9(3), 177. https://doi.org/10.3390/w9030177






