Removal of Arsenic Using Acid/Metal-Tolerant Sulfate Reducing Bacteria: A New Approach for Bioremediation of High-Arsenic Acid Mine Waters
Abstract
:1. Introduction
2. Materials and Methods
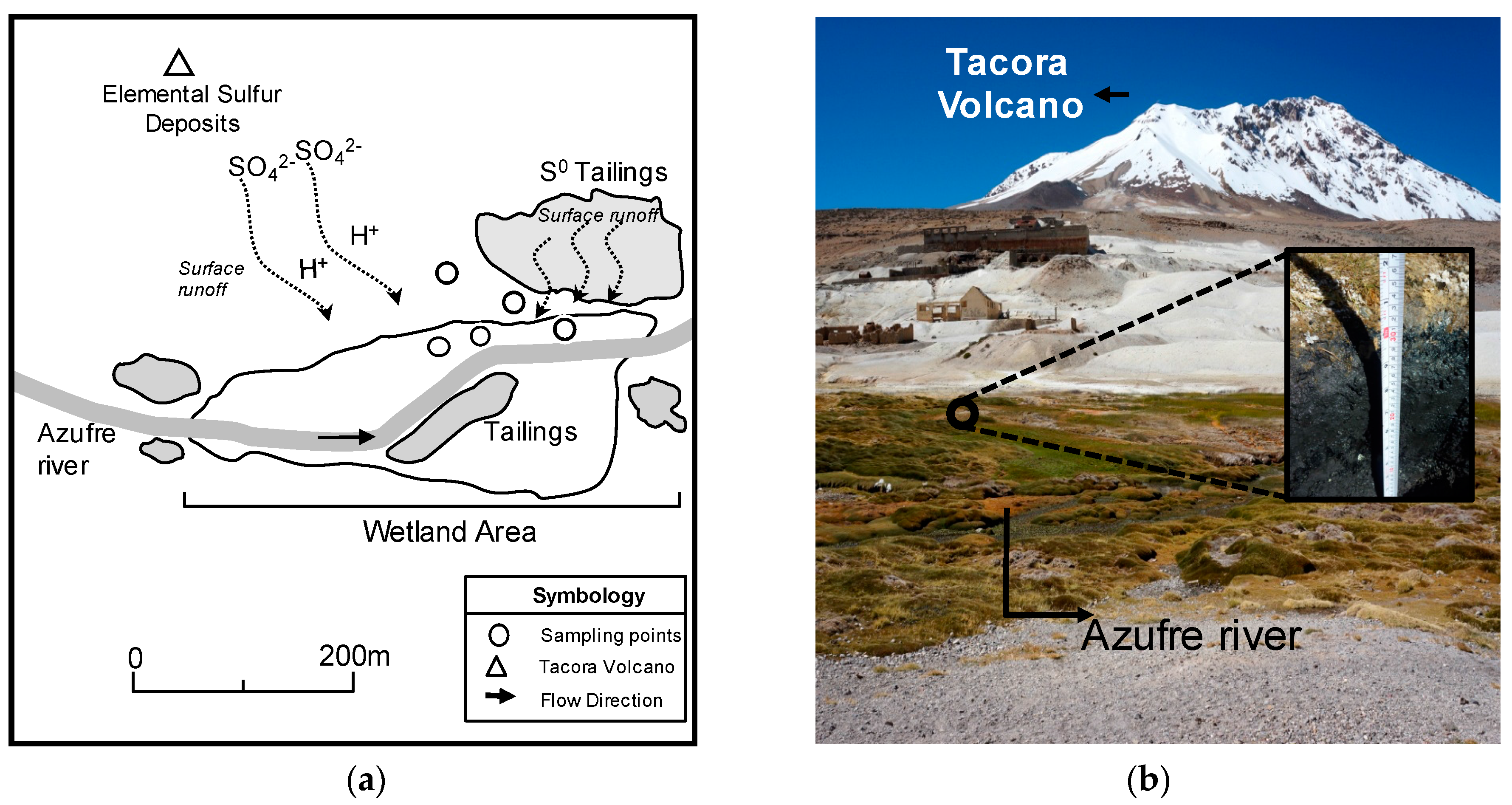
2.1. Study Site: Aguas Calientes Area in the Upper Section of the Azufre River Sub-Basin
2.2. Sampling
2.3. Batch Experiments
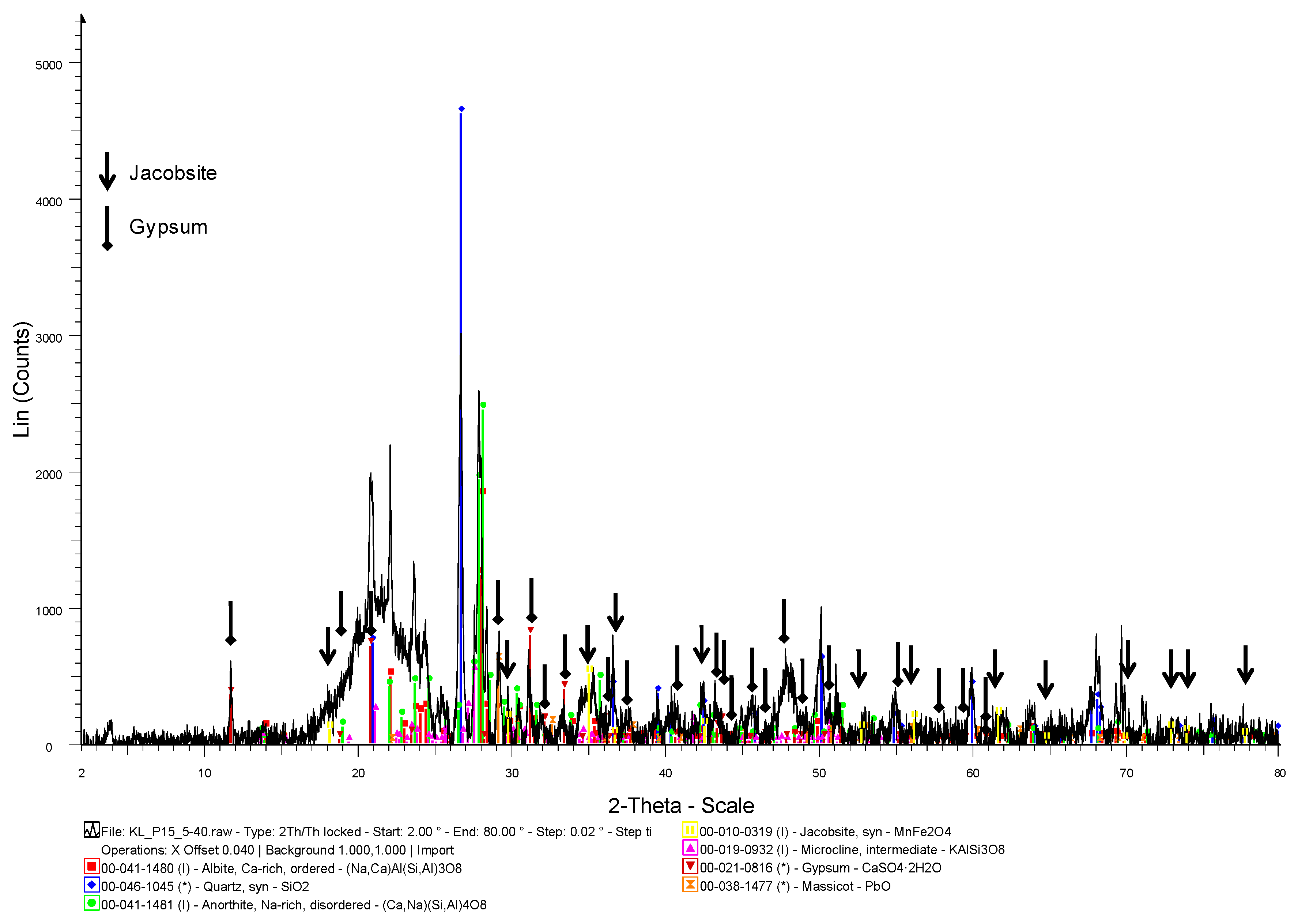
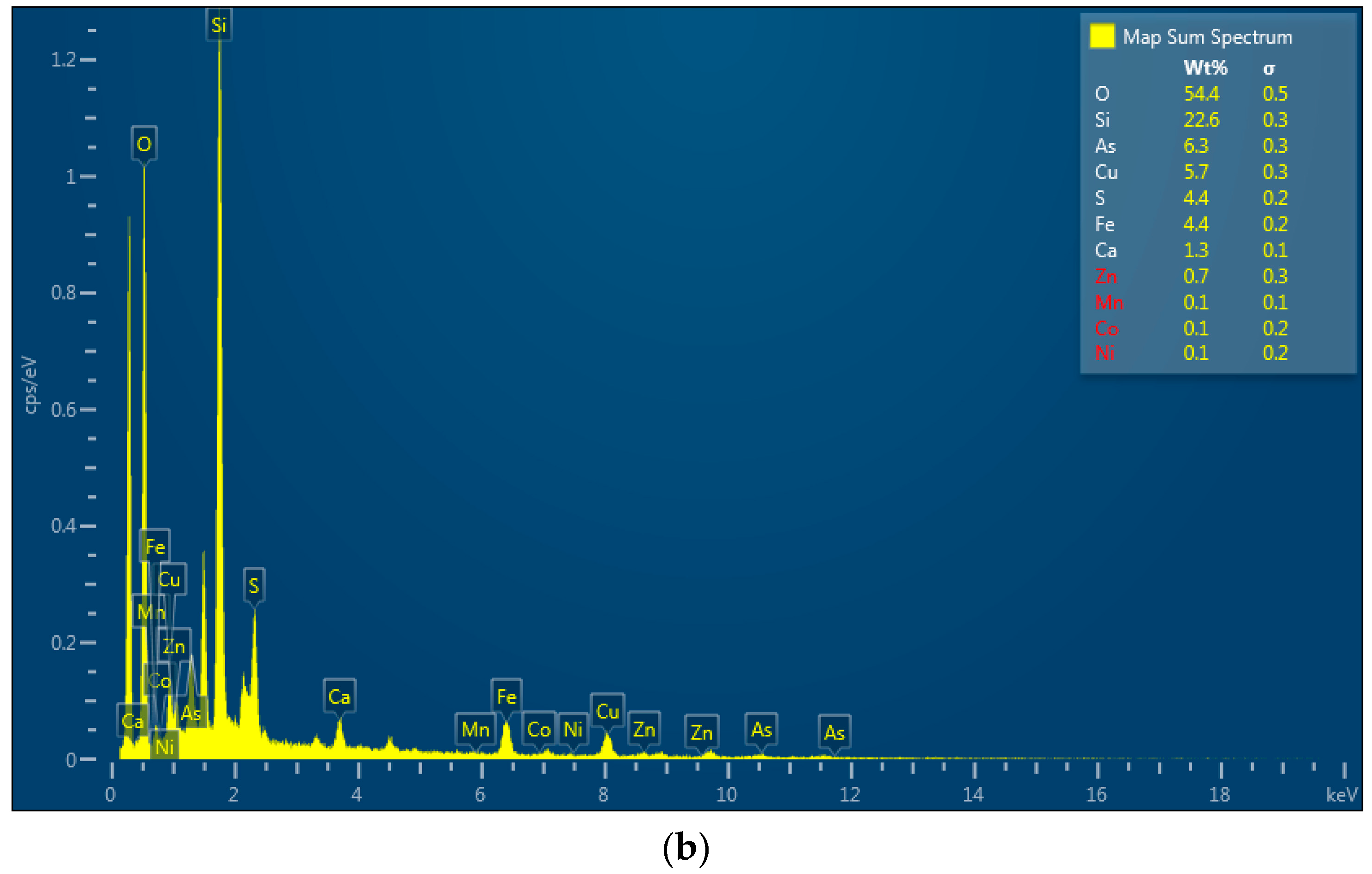
2.4. Geochemical Analyses
3. Results and Discussion
3.1. Metal-Tolerant Sulfate Reducing Bacteria
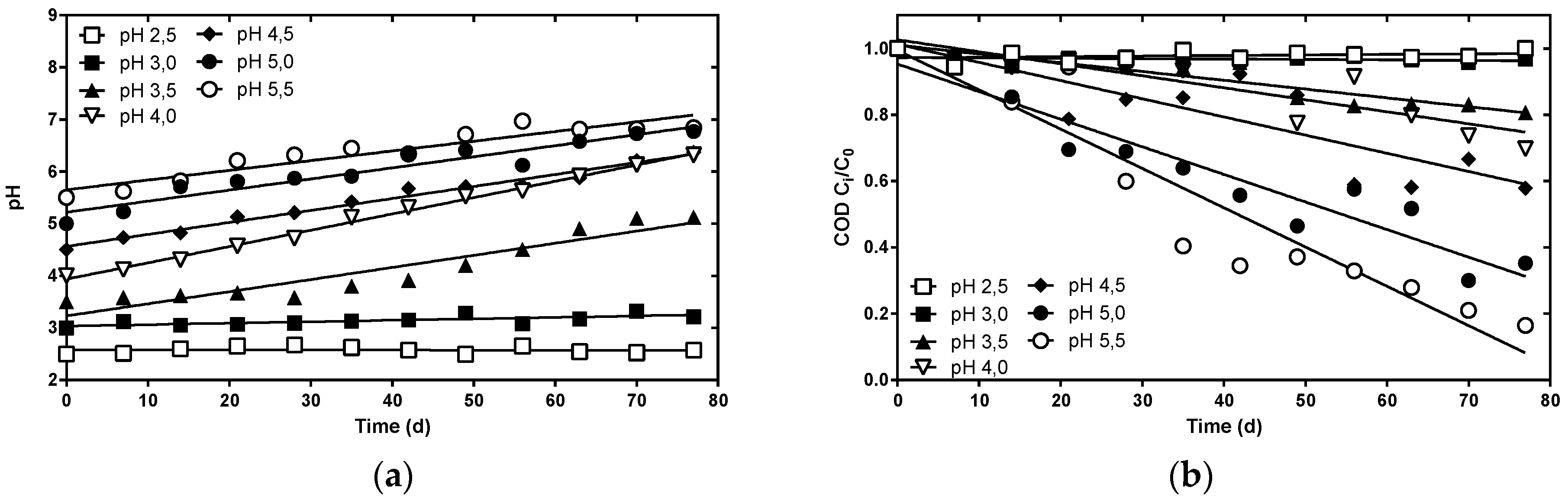
3.2. Metal-Tolerant Sulfate Reducing Bacteria Are Tolerant to Low pH
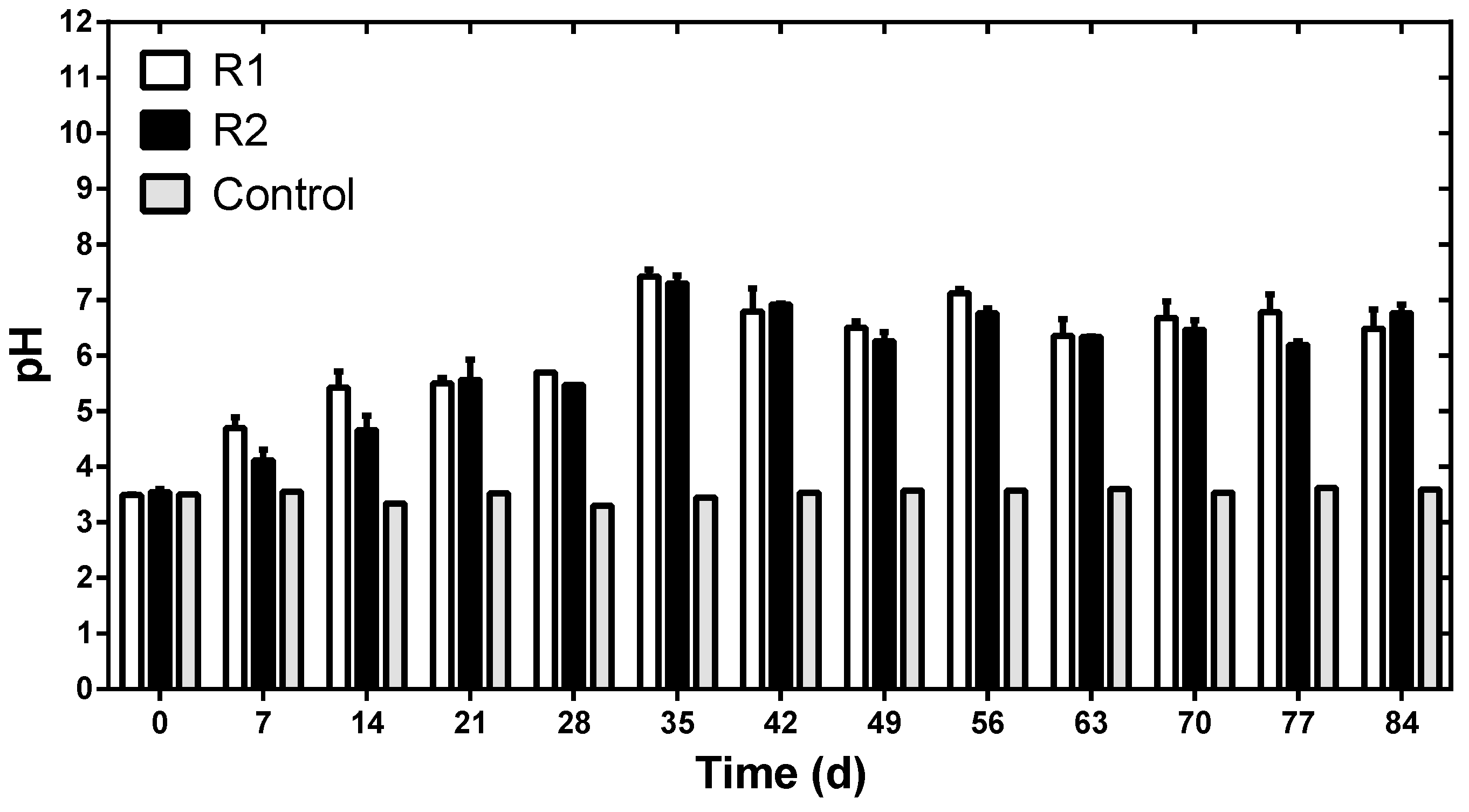
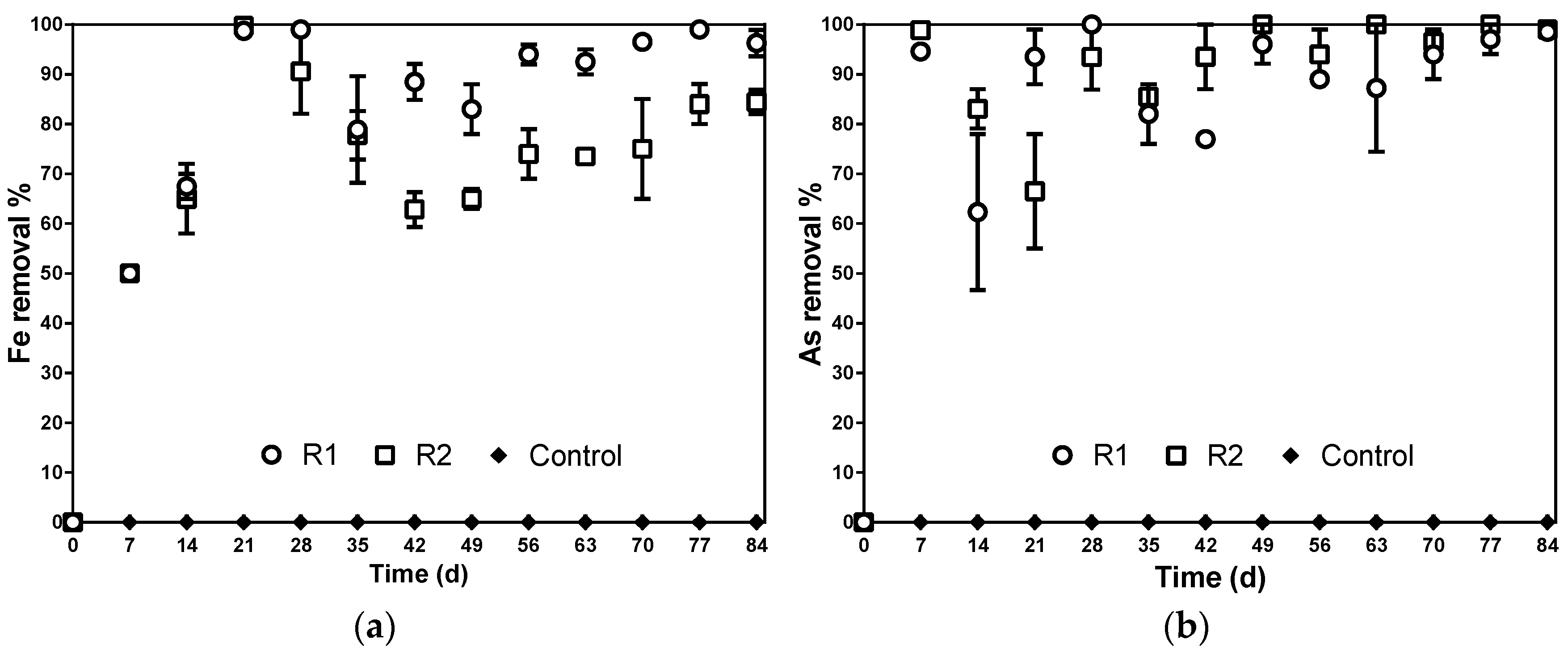
3.3. Arsenic and Iron Are Removed in Sulfate Reduction Reactors
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Smedley, P.L.; Kinniburgh, D.G. A review of the source, behaviour and distribution of arsenic in natural waters. Appl. Geochem. 2002, 17, 517–568. [Google Scholar] [CrossRef]
- Bugueño, M.P.; Acevedo, S.E.; Bonilla, C.A.; Pizarro, G.E.; Pasten, P.A. Differential arsenic binding in the sediments of two sites in Chile’s lower Loa River basin. Sci. Total Environ. 2014, 466, 387–396. [Google Scholar] [CrossRef] [PubMed]
- Landrum, J.T.; Bennett, P.C.; Engel, A.S.; Alsina, M.A.; Pastén, P.A.; Milliken, K. Partitioning geochemistry of arsenic and antimony, El Tatio Geyser Field, Chile. Appl. Geochem. 2009, 24, 664–676. [Google Scholar] [CrossRef]
- Leiva, E.D.; Rámila, C.D.P.; Vargas, I.T.; Escauriaza, C.R.; Bonilla, C.A.; Pizarro, G.E.; Regan, J.M.; Pasten, P.A. Natural attenuation process via microbial oxidation of arsenic in a high andean watershed. Sci. Total Environ. 2014, 466–467, 490–502. [Google Scholar] [CrossRef] [PubMed]
- Guerra, P.; Simonson, K.; González, C.; Gironás, J.; Escauriaza, C.; Pizarro, G.; Bonilla, C.A.; Pasten, P.A. Daily Freeze–Thaw Cycles Affect the Transport of Metals in Streams Affected by Acid Drainage. Water 2016, 8, 74. [Google Scholar] [CrossRef]
- Dixit, S.; Hering, J.G. Comparison of arsenic(V) and arsenic(III) sorption onto iron oxide minerals: Implications for arsenic mobility. Environ. Sci. Technol. 2003, 37, 4182–4189. [Google Scholar] [CrossRef] [PubMed]
- Dold, B.; Fontboté, L. Element cycling and secondary mineralogy in porphyry copper tailings as a function of climate, primary mineralogy, and mineral processing. J. Geochem. Explor. 2001, 74, 3–55. [Google Scholar] [CrossRef]
- Wang, S.L.; Mulligan, C.N. Natural attenuation processes for remediation of arsenic contaminated soils and groundwater. J. Hazard. Mater. 2006, 138, 459–470. [Google Scholar] [CrossRef] [PubMed]
- Cheng, H.; Hu, Y.; Luo, J.; Xu, B.; Zhao, J. Geochemical processes controlling fate and transport of arsenic in acid mine drainage (AMD) and natural systems. J. Hazard. Mater. 2009, 165, 13–26. [Google Scholar] [CrossRef] [PubMed]
- Courtin-Nomade, A.; Grosbois, C.; Bril, H.; Roussel, C. Spatial variability of arsenic in some iron-rich deposits generated by acid mine drainage. Appl. Geochem. 2005, 20, 383–396. [Google Scholar] [CrossRef]
- Majzlan, J.; Plášil, J.; Škoda, R.; Gescher, J.; Kögler, F.; Rusznyak, A.; Küsel, K.; Neu, T.R.; Mangold, S.; Rothe, J. Arsenic-rich acid mine water with extreme arsenic concentration: Mineralogy, geochemistry, microbiology, and environmental implications. Environ. Sci. Technol. 2014, 48, 13685–13693. [Google Scholar] [CrossRef] [PubMed]
- Blowes, D.W.; Ptacek, C.J.; Jambor, J.L.; Weisener, C.G. The geochemistry of acid mine drainage. Treatise Geochem. 2003, 9, 612. [Google Scholar]
- Akcil, A.; Koldas, S. Acid Mine Drainage (AMD): Causes, treatment and case studies. J. Clean. Prod. 2006, 14, 1139–1145. [Google Scholar] [CrossRef]
- Burgess, J.E.; Stuetz, R.M. Activated sludge for the treatment of sulphur-rich wastewaters. Miner. Eng. 2002, 15, 839–846. [Google Scholar] [CrossRef]
- Neculita, C.M.; Zagury, G.J.; Bussiere, B. Passive treatment of acid mine drainage in bioreactors using sulfate-reducing bacteria: Critical review and research needs. J. Environ. Qual. 2007, 36, 1–16. [Google Scholar] [CrossRef] [PubMed]
- Willow, M.A.; Cohen, R.R.H. pH, dissolved oxygen, and adsorption effects on metal removal in anaerobic bioreactors. J. Environ. Qual. 2003, 32, 1212–1221. [Google Scholar] [CrossRef] [PubMed]
- Dvorak, D.H.; Hedin, R.S.; Edenborn, H.M.; McIntire, P.E. Treatment of metal-contaminated water using bacterial sulfate reduction: Results from pilot-scale reactors. Biotechnol. Bioeng. 1992, 40, 609–616. [Google Scholar] [CrossRef] [PubMed]
- Utgikar, V.P.; Harmon, S.M.; Chaudhary, N.; Tabak, H.H.; Govind, R.; Haines, J.R. Inhibition of sulfate-reducing bacteria by metal sulfide formation in bioremediation of acid mine drainage. Environ. Toxicol. 2002, 17, 40–48. [Google Scholar] [CrossRef] [PubMed]
- Sánchez-Andrea, I.; Sanz, J.L.; Bijmans, M.F.; Stams, A.J. Sulfate reduction at low pH to remediate acid mine drainage. J. Hazard. Mater. 2014, 269, 98–109. [Google Scholar] [CrossRef] [PubMed]
- Meier, J.; Piva, A.; Fortin, D. Enrichment of sulfate-reducing bacteria and resulting mineral formation in media mimicking pore water metal ion concentrations and pH conditions of acidic pit lakes. FEMS Microbiol. Ecol. 2012, 79, 69–84. [Google Scholar] [CrossRef] [PubMed]
- Alazard, D.; Joseph, M.; Battaglia-Brunet, F.; Cayol, J.L.; Ollivier, B. Desulfosporosinus acidiphilus sp. nov.: A moderately acidophilic sulfate-reducing bacterium isolated from acid mining drainage sediments. Extremophiles 2010, 14, 305–312. [Google Scholar] [CrossRef] [PubMed]
- Le Pape, P.; Battaglia-Brunet, F.; Parmentier, M.; Joulian, C.; Gassaud, C.; Fernandez-Rojo, L.; Guigner, J.M.; Ikogou, M.; Stetten, L.; Olivi, L.; et al. Complete removal of arsenic and zinc from a heavily contaminated acid mine drainage via an indigenous SRB consortium. J. Hazard. Mater. 2017, 321, 764–772. [Google Scholar] [CrossRef] [PubMed]
- Srinath, T.; Verma, T.; Ramteke, P.W.; Garg, S.K. Chromium (VI) biosorption and bioaccumulation by chromate resistant bacteria. Chemosphere 2002, 48, 427–435. [Google Scholar] [CrossRef]
- Zouboulis, A.I.; Loukidou, M.X.; Matis, K.A. Biosorption of toxic metals from aqueous solutions by bacteria strains isolated from metal-polluted soils. Process Biochem. 2004, 39, 909–916. [Google Scholar] [CrossRef]
- Christensen, B.; Laake, M.; Lien, T. Treatment of acid mine water by sulfate-reducing bacteria; results from a bench scale experiment. Water Res. 1996, 30, 1617–1624. [Google Scholar] [CrossRef]
- Hao, O.J.; Chen, J.M.; Huang, L.; Buglass, R.L. Sulfate reducing bacteria. Crit. Rev. Environ. Sci. Technol. 1996, 26, 155. [Google Scholar] [CrossRef]
- Gyure, R.A.; Konopka, A.; Brooks, A.; Doemel, W. Microbial sulfate reduction in acidic (pH 3) strip-mine lakes. FEMS Microbiol. Ecol. 1990, 73, 193–201. [Google Scholar] [CrossRef]
- Tuttle, J.H.; Dugan, P.R.; Macmillan, C.B.; Randles, C.I. Microbial dissimilatory sulfur cycle in acid mine water. J. Bacteriol. 1969, 97, 594–602. [Google Scholar] [PubMed]
- Ghose, T.K.; Wikén, T. Inhibition of bacterial sulphate-reduction in presence of short chain fatty acids. Physiol. Plant. 1955, 8, 116–135. [Google Scholar] [CrossRef]
- Icgen, B.; Harrison, S. Exposure to sulfide causes populations shifts in sulfate reducing consortia. Res. Microbiol. 2006, 157, 784–791. [Google Scholar] [CrossRef] [PubMed]
- Moosa, S.; Harrison, S. Product inhibition by sulphide species on biological sulphate reduction for the treatment of acid mine drainage. Hydrometallurgy 2006, 83, 214–222. [Google Scholar] [CrossRef]
- Benner, S.G.; Blowes, D.W.; Gould, W.D.; Herbert, R.B.; Ptacek, C.J. Geochemistry of a permeable reactive barrier for metals and acid mine drainage. Environ. Sci. Technol. 1999, 33, 2793–2799. [Google Scholar] [CrossRef]
- Kalin, M.; Fyson, A.; Wheeler, W.N. The chemistry of conventional and alternative treatment systems for the neutralization of acid mine drainage. Sci. Total Environ. 2006, 366, 395–408. [Google Scholar] [CrossRef] [PubMed]
- Cowie, R.; Williams, M.; Wireman, M.; Runkel, R. Use of natural and applied tracers to guide targeted remediation efforts in an acid mine drainage system, colorado rockies, USA. Water 2014, 6, 745–777. [Google Scholar] [CrossRef]
- Dold, B. Basic Concepts in Environmental Geochemistry of Sulfidic Mine-Waste Management. In Waste Management; InTech, Inc.: Greenwood Village, CO, USA, 2010. [Google Scholar]
- Leiva, E.; Leiva-Aravena, E.; Vargas, I. Acid Water Neutralization Using Microbial Fuel Cells: An Alternative for Acid Mine Drainage Treatment. Water 2016, 8, 536. [Google Scholar] [CrossRef]
- Van Houten, R.T.; Pol, L.W.H.; Lettinga, G. Biological Sulfate Reduction Using Gas-Lift Reactors Fed with Hydrogen and Carbon-Dioxide as Energy and Carbon Source. Biotechnol. Bioeng. 1994, 44, 586–594. [Google Scholar] [CrossRef] [PubMed]
- Church, C.D.; Wilkin, R.T.; Alpers, C.N.; Rye, R.O.; McCleskey, R.B. Microbial sulfatereduction and metal attenuation in pH 4 acid mine water. Geochem. Trans. 2007, 8, 10. [Google Scholar] [CrossRef] [PubMed]
- Hard, B.C.; Friedrich, S.; Babel, W. Bioremediation of acid mine water using facultatively methylotrophic metal-tolerant sulfate-reducing bacteria. Microbiol. Res. 1997, 152, 65–73. [Google Scholar] [CrossRef]
- Kusel, K.; Roth, U.; Trinkwalter, T.; Peiffer, S. Effect of pH on the anaerobic micro-bial cycling of sulfur in mining-impacted freshwater lake sediments. Environ. Exp. Bot. 2001, 46, 213–223. [Google Scholar] [CrossRef]
- Kimura, S.; Hallberg, K.B.; Johnson, D.B. Sulfidogenesis in low pH (3.8–4.2) media by a mixed population of acidophilic bacteria. Biodegradation 2006, 17, 57–65. [Google Scholar] [CrossRef] [PubMed]
- Bijmans, M.F. Sulfate Reduction under Acidic Conditions for Selective Metals Recovery. Ph.D. Thesis, Wageningen Universiteit, Wageningen, The Netherlands, 2008; 156p. [Google Scholar]
- Battaglia-Brunet, F.; Crouzet, C.; Burnol, A.; Coulon, S.; Morin, D.; Joulian, C. Precipitation of arsenic sulphide from acidic water in a fixed-film bioreactor. Water Res. 2012, 46, 3923–3933. [Google Scholar] [CrossRef] [PubMed]
- Moore, J.N.; Ficklin, W.H.; Johns, C. Partitioning of Arsenic and Metals in Reducing Sulfidic Sediments. Environ. Sci. Technol. 1988, 22, 432–437. [Google Scholar] [CrossRef]
- O’Day, P.A.; Vlassopoulos, D.; Root, R.; Rivera, N. The influence of sulfur and iron on dissolved arsenic concentrations in the shallow subsurface under changing redox conditions. Proc. Natl. Acad. Sci. USA 2004, 101, 13703–13708. [Google Scholar] [CrossRef] [PubMed]
- Wolthers, M.; Charlet, L.; Van der Weijden, C.H.; Van der Linde, P.R.; Rickard, D. Arsenic mobility in the ambient sulfidic environment: Sorption of arsenic(V) and arsenic(III) onto disordered mackinawite. Geochim. Et Cosmochim. Acta 2005, 69, 3483–3492. [Google Scholar] [CrossRef]
- Cullen, W.R.; Reimer, K.J. Arsenic speciation in the environment. Chem. Rev. 1989, 89, 713–764. [Google Scholar] [CrossRef]
- Masscheleyn, P.H.; Delaune, R.D.; Patrick, W.H. Effect of Redox Potential and Ph on Arsenic Speciation and Solubility in a Contaminated Soil. Environ. Sci. Technol. 1991, 25, 1414–1419. [Google Scholar] [CrossRef]
- Demergasso, C.; Chong, G.; Escudero, L.; Pueyo, J.; Pedrós-Alió, C. Microbial precipitation of arsenic sulfides in Andean salt flats. Geomicrobiol. J. 2007, 24, 111–123. [Google Scholar] [CrossRef]
- Rodriguez-Freire, L.; Sierra-Alvarez, R.; Root, R.; Chorover, J.; Field, J. Biomineralization of arsenate to arsenic sulfides is greatly enhanced at midly acidic conditions. Water Res. 2014, 66, 242–253. [Google Scholar] [CrossRef] [PubMed]
- Rodriguez-Freire, L.; Sierra-Alvarez, R.; Root, R.; Chorover, J.; Field, J. Arsenic remediation by formation of arsenic sulfide minerals in a continuous anaerobic bioreactor. Biotechnol. Bioeng. 2015, 113, 522–530. [Google Scholar] [CrossRef] [PubMed]
- Jackson, C.; Koch, I.; Reimer, K. Mechanisms of dissolved arsenic removal by biochemical reactors: A bench-and field-scale study. Appl. Geochem. 2013, 29, 174–181. [Google Scholar] [CrossRef]
- Couture, R.M.; Rose, J.; Kumar, N.; Mitchell, K.; Wallschläger, D.; Van Cappellen, P. Sorption of arsenite, arsenate, and thioarsenates to iron oxides and iron sulfides: A kinetic and spectroscopic investigation. Environ. Sci. Technol. 2013, 47, 5652–5659. [Google Scholar] [CrossRef] [PubMed]
- Farquhar, M.L.; Charnock, J.M.; Livens, F.R.; Vaughan, D.J. Mechanisms of arsenic uptake from aqueous solution by interaction with goethite, lepidocrocite, mackinawite, and pyrite: An X-ray absorption spectroscopy study. Environ. Sci. Technol. 2002, 36, 1757–1762. [Google Scholar] [CrossRef] [PubMed]
- Gallegos, T.J.; Han, Y.S.; Hayes, K.F. Model predictions of realgar precipitation by reaction of as(III) with synthetic mackinawite under anoxic conditions. Environ. Sci. Technol. 2008, 42, 9338–9343. [Google Scholar] [CrossRef] [PubMed]

© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Serrano, J.; Leiva, E. Removal of Arsenic Using Acid/Metal-Tolerant Sulfate Reducing Bacteria: A New Approach for Bioremediation of High-Arsenic Acid Mine Waters. Water 2017, 9, 994. https://doi.org/10.3390/w9120994
Serrano J, Leiva E. Removal of Arsenic Using Acid/Metal-Tolerant Sulfate Reducing Bacteria: A New Approach for Bioremediation of High-Arsenic Acid Mine Waters. Water. 2017; 9(12):994. https://doi.org/10.3390/w9120994
Chicago/Turabian StyleSerrano, Jennyfer, and Eduardo Leiva. 2017. "Removal of Arsenic Using Acid/Metal-Tolerant Sulfate Reducing Bacteria: A New Approach for Bioremediation of High-Arsenic Acid Mine Waters" Water 9, no. 12: 994. https://doi.org/10.3390/w9120994
APA StyleSerrano, J., & Leiva, E. (2017). Removal of Arsenic Using Acid/Metal-Tolerant Sulfate Reducing Bacteria: A New Approach for Bioremediation of High-Arsenic Acid Mine Waters. Water, 9(12), 994. https://doi.org/10.3390/w9120994




