Detoxification of Pesticide-Containing Wastewater with FeIII, Activated Carbon and Fenton Reagent and Its Control Using Three Standardized Bacterial Inhibition Tests
Abstract
:1. Introduction
1.1. Motivation
1.2. Principle of Bacterial Inhibition Tests
1.3. Wastewater Treatment Methods for Detoxification
1.3.1. Precipitation/Flocculation via FeIII
1.3.2. Adsorption via Powdered Activated Carbon (PAC)
1.3.3. Fenton Method (FeII/H2O2)
2. Materials and Methods
2.1. Experimental Concept
2.2. Chemicals and Reagents
2.3. Bacterial Inhibition Tests
2.3.1. Preparation of Activated Sludge
2.3.2. Nitrification Inhibition Test (NIT)
2.3.3. Respiration Inhibition Test (RIT)
2.3.4. Luminescent Bacteria Test (LBT)
2.3.5. Calculation of the Effective Concentration (EC)
2.4. Analytical Methods
2.5. Experiments with Pesticide-Containing Wastewater
2.5.1. Wastewater Samples
2.5.2. Experiments Regarding Precipitation/Flocculation via FeIII
2.5.3. Experiments Regarding Adsorption via PAC
2.5.4. Experiments Regarding Fenton Method (FeII/H2O2)
3. Results and Discussion
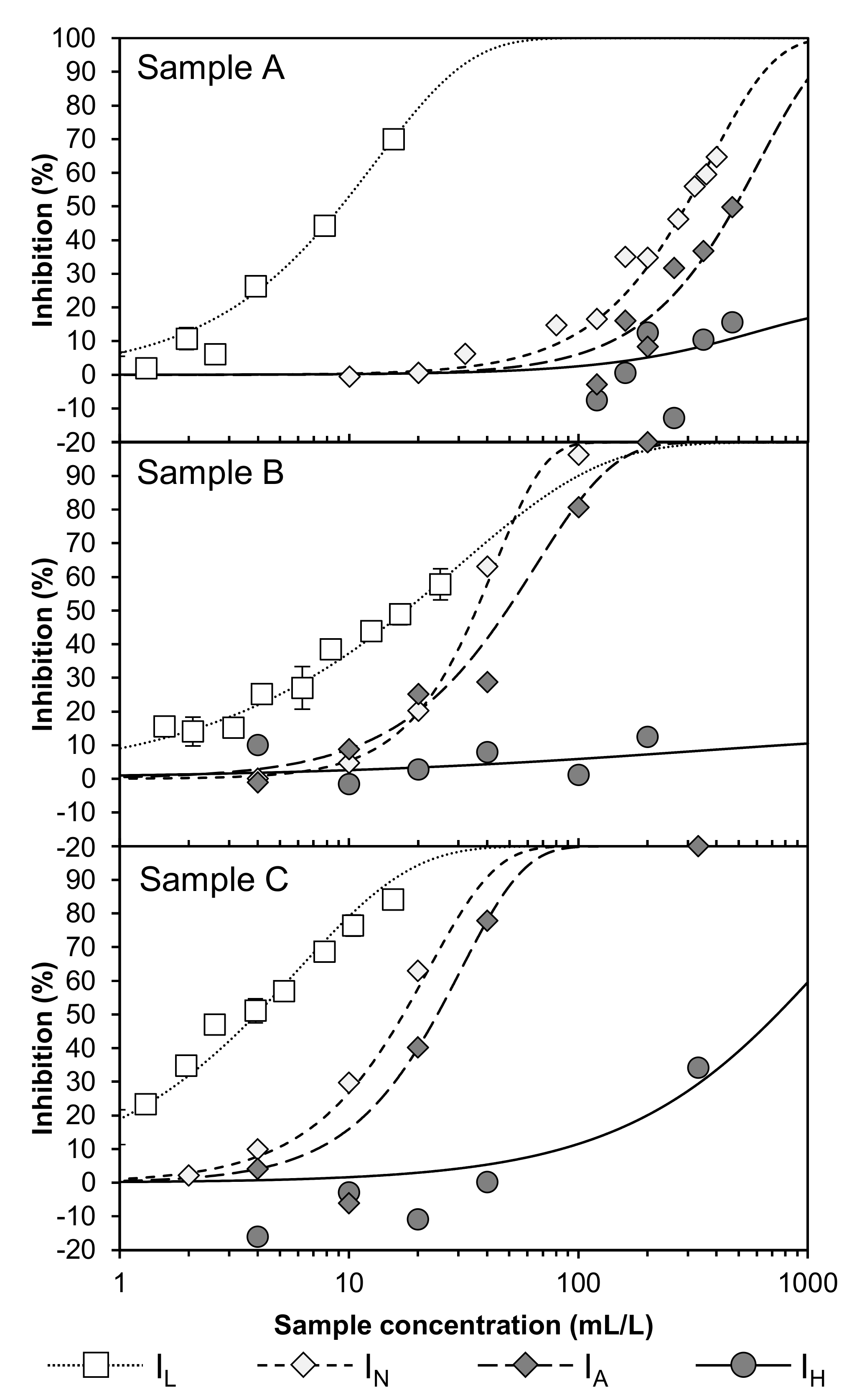
3.1. Raw Wastewater Samples
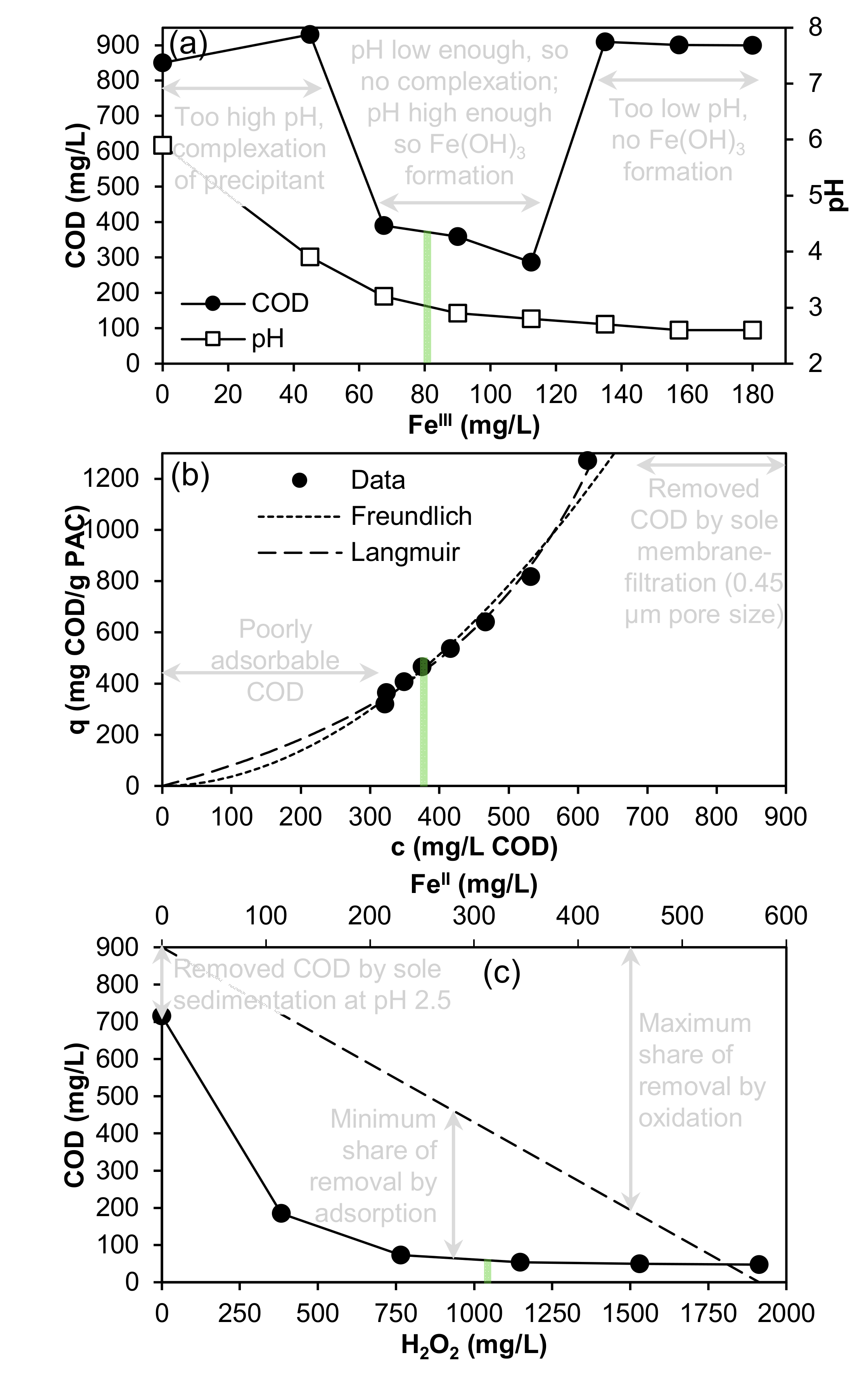
3.2. Experiment Regarding Precipitation/Flocculation via FeIII
3.3. Experiment Regarding Adsorption via PAC
3.4. Experiment Regarding Fenton Method (FeII/H2O2)
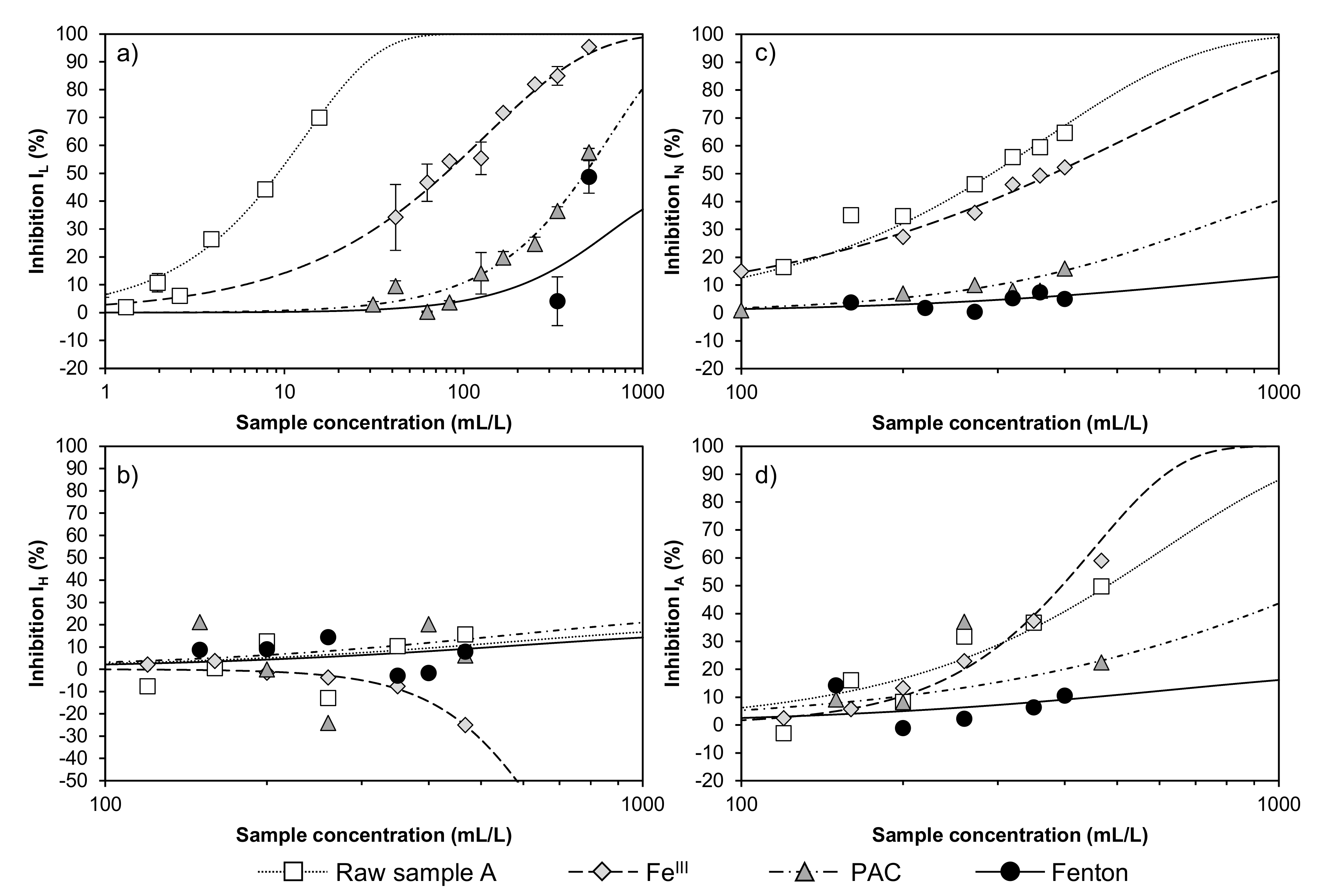
3.5. Experiment Regarding FeIII, PAC and FeII/H2O2 on a Larger Scale
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Geyer, H.J.; Scheunert, I.; Korte, F. Correlation between the bioconcentration potential of organic environmental chemicals in humans and their n-octanol/water partition coefficients. Chemosphere 1987, 16, 239–252. [Google Scholar] [CrossRef]
- Sankoh, A.I.; Whittle, R.; Semple, K.T.; Jones, K.C.; Sweetman, A.J. An assessment of the impacts of pesticide use on the environment and health of rice farmers in Sierra Leone. Environ. Int. 2016, 94, 458–466. [Google Scholar] [CrossRef] [PubMed]
- Karami-Mohajeri, S.; Abdollahi, M. Toxic influence of organophosphate, carbamate, and organochlorine pesticides on cellular metabolism of lipids, proteins, and carbohydrates: A systematic review. Hum. Exp. Toxicol. 2011, 30, 1119–1140. [Google Scholar] [CrossRef] [PubMed]
- Garry, V.F.; Harkins, M.E.; Erickson, L.L.; Long-Simpson, L.K.; Holland, S.E.; Burroughs, B.L. Birth Defects, Season of Conception, and Sex of Children Born to Pesticide Applicators Living in the Red River Valley of Minnesota, USA. Environ. Health Perspect. 2002, 110, 441–449. [Google Scholar] [CrossRef] [PubMed]
- Federico, G. Feeding the World: An Economic History of Agriculture, 1800–2000; Princeton University Press: Princeton, NJ, USA; Oxford, UK, 2009; ISBN 9780691120515. [Google Scholar]
- Akkan, Z.; Flaig, H.; Ballschmiter, K. Pflanzenbehandlungs- und Schädlingsbekämpfungsmittel in der Umwelt: Emissionen, Immissionen und Ihre Human- und Ökotoxikologische Bewertung; Erich Schmidt Verlag: Berlin, Germany, 2003; ISBN 3503070273. [Google Scholar]
- Jönsson, K. Occurrence of nitrification inhibition in Swedish municipal wastewaters. Water Res. 2000, 34, 2455–2462. [Google Scholar] [CrossRef]
- Grunditz, C.; Dalhammar, G. Development of nitrification inhibition assays using pure cultures of nitrosomonas and nitrobacter. Water Res. 2001, 35, 433–440. [Google Scholar] [CrossRef]
- Brack, A.; Strube, J.; Stolz, P.; Decker, H. Effects of ultrahigh dilutions of 3,5-dichlorophenol on the luminescence of the bacterium Vibrio fischeri. Biochim. Biophys. Acta Gen. Subj. 2003, 1621, 253–260. [Google Scholar] [CrossRef]
- Jennings, V.L.K.; Rayner-Brandes, M.H.; Bird, D.J. Assessing chemical toxicity with the bioluminescent photobacterium (Vibrio fischeri): A comparison of three commercial systems. Water Res. 2001, 35, 3448–3456. [Google Scholar] [CrossRef]
- Menz, J.; Schneider, M.; Kümmerer, K. Toxicity testing with luminescent bacteria—Characterization of an automated method for the combined assessment of acute and chronic effects. Chemosphere 2013, 93, 990–996. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.; Cho, K.; Lim, J.; Kim, W.; Hwang, S. Acclimation and activity of ammonia-oxidizing bacteria with respect to variations in zinc concentration, temperature, and microbial population. Bioresour. Technol. 2011, 102, 4196–4203. [Google Scholar] [CrossRef] [PubMed]
- Halling-Sørensen, B. Inhibition of aerobic growth and nitrification of bacteria in sewage sludge by antibacterial agents. Arch. Environ. Contam. Toxicol. 2001, 40, 451–460. [Google Scholar] [CrossRef] [PubMed]
- Gendig, C.; Domogala, G.; Agnoli, F.; Pagga, U.; Strotmann, U.J. Evaluation and further development of the activated sludge respiration inhibition test. Chemosphere 2003, 52, 143–149. [Google Scholar] [CrossRef]
- Cokgor, E.U.; Ozdemir, S.; Karahan, O.; Insel, G.; Orhon, D. Critical appraisal of respirometric methods for metal inhibition on activated sludge. J. Hazard. Mater. 2007, 139, 332–339. [Google Scholar] [CrossRef] [PubMed]
- Meriç, S.; Eremektar, G.; Çiner, F.; Tünay, O. An OUR-based approach to determine the toxic effects of 2,4-dichlorophenoxyacetic acid in activated sludge. J. Hazard. Mater. 2003, 101, 147–155. [Google Scholar] [CrossRef]
- Dalzell, D.J.B.; Christofi, N. An ATP luminescence method for direct toxicity assessment of pollutants impacting on the activated sewage sludge process. Water Res. 2002, 36, 1493–1502. [Google Scholar] [CrossRef]
- Foladori, P.; Bruni, L.; Tamburini, S. Toxicant inhibition in activated sludge: Fractionation of the physiological status of bacteria. J. Hazard. Mater. 2014, 280, 758–766. [Google Scholar] [CrossRef] [PubMed]
- Ricco, G.; Tomei, M.C.M.C.; Ramadori, R.; Laera, G. Toxicity assessment of common xenobiotic compounds on municipal activated sludge: Comparison between respirometry and Microtox®. Water Res. 2004, 38, 2103–2110. [Google Scholar] [CrossRef] [PubMed]
- Gutiérrez, M.; Etxebarria, J.; de las Fuentes, L. Evaluation of wastewater toxicity: Comparative study between Microtox® and activated sludge oxygen uptake inhibition. Water Res. 2002, 36, 919–924. [Google Scholar] [CrossRef]
- Ren, S. Assessing wastewater toxicity to activated sludge: Recent research and developments. Environ. Int. 2004, 30, 1151–1164. [Google Scholar] [CrossRef] [PubMed]
- Mamais, D.; Noutsopoulos, C.; Stasinakis, A.S.; Kouris, N.; Andreadakis, A.D. Comparison of bioluminescence and nitrification inhibition methods for assessing toxicity to municipal activated sludge. Water Environ. Res. 2008, 80, 484–489. [Google Scholar] [CrossRef] [PubMed]
- Pagga, U.; Bachner, J.; Strotmann, U. Inhibition of nitrification in laboratory tests and model wastewater treatment plants. Chemosphere 2006, 65, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Dalzell, D.J.B.; Alte, S.; Aspichueta, E.; de La Sota, A.; Etxebarria, J.; Gutierrez, M.; Hoffmann, C.C.; Sales, D.; Obst, U.; Christofi, N. A comparison of five rapid direct toxicity assessment methods to determine toxicity of pollutants to activated sludge. Chemosphere 2002, 47, 535–545. [Google Scholar] [CrossRef]
- Kilroy, A.C.; Gray, N.F. The toxicity of four organic solvents commonly used in the pharmaceutical industry to activated sludge. Water Res. 1992, 26, 887–892. [Google Scholar] [CrossRef]
- Arslan-Alaton, I.; Caglayan, A.E. Toxicity and biodegradability assessment of raw and ozonated procaine penicillin G formulation effluent. Ecotoxicol. Environ. Saf. 2006, 63, 131–140. [Google Scholar] [CrossRef] [PubMed]
- Sivrioğlu, Ö.; Yonar, T. Determination of the acute toxicities of physicochemical pretreatment and advanced oxidation processes applied to dairy effluents on activated sludge. J. Dairy Sci. 2015, 98, 2337–2344. [Google Scholar] [CrossRef] [PubMed]
- Bremner, J.M.; Blackmer, A.M. Nitrous oxide: Emission from Soils during Nitrification of Fertilizer Nitrogen. Science 1978, 199, 295–296. [Google Scholar] [CrossRef] [PubMed]
- Hastings, J.W. Chemistries and colors of bioluminescent reactions: A review. Gene 1996, 173, 5–11. [Google Scholar] [CrossRef]
- Langmuir, I. The Adsorption of Gases on Plane Surfaces of Glass, Mica and Platinum. J. Am. Chem. Soc. 1918, 40, 1361–1403. [Google Scholar] [CrossRef]
- Freundlich, H. Über die Adsorption in Lösungen. Z. Phys. Chem. 1907, 57U, 385–470. [Google Scholar] [CrossRef]
- Ho, Y.S.; Porter, J.F.; McKay, G. Equilibrium Isotherm Studies for the Sorption of Divalent Metal Ions onto Peat: Copper, Nickel and Lead Single Component Systems. Water Air Soil Pollut. 2002, 141, 1–33. [Google Scholar] [CrossRef]
- Sychev, A.Y.; Isak, V.G. Iron compounds and the mechanisms of the homogeneous catalysis of the activation of O2 and H2O2 and of the oxidation of organic substrates. Russ. Chem. Rev. 1995, 64, 1105–1129. [Google Scholar] [CrossRef]
- Bautista, P.; Mohedano, A.F.; Casas, J.A.; Zazo, J.A.; Rodriguez, J.J. An overview of the application of Fenton oxidation to industrial wastewaters treatment. J. Chem. Technol. Biotechnol. 2008, 83, 1323–1338. [Google Scholar] [CrossRef]
- International Organization for Standardization. Water Quality—Toxicity Test for Assessing the Inhibition of Nitrification of Activated Sludge Microorganisms; ISO 9509:2006; International Organization for Standardization: Geneva, Switzerland, 2006. [Google Scholar]
- International Organization for Standardization. Water Quality—Test for Inhibition of Oxygen Consumption by Activated Sludge for Carbonaceous and Ammonium Oxidation; ISO 8192:2007; International Organization for Standardization: Geneva, Switzerland, 2007. [Google Scholar]
- International Organization for Standardization. Water Quality—Determination of the Inhibitory Effect of Water Samples on the Light Emission of Vibrio Fischeri (Luminescent Bacteria Test)—Part 3: Method Using Freeze-Dried Bacteria; ISO 11348-3:2007; International Organization for Standardization: Geneva, Switzerland, 2007. [Google Scholar]
- Backhaus, T.; Froehner, K.; Altenburger, R.; Grimme, L.H. Toxicity testing with Vibrio fischeri: A comparison between the long term (24 h) and the short term (30 min) bioassay. Chemosphere 1997, 35, 2925–2938. [Google Scholar] [CrossRef]
- International Organization for Standardization. Water Quality—Determination of Nitrate—Part 1: 2,6-Dimethylphenol Spectrometric Method; ISO 7890-1:1986; International Organization for Standardization: Geneva, Switzerland, 1986. [Google Scholar]
- International Organization for Standardization. Water Quality—Determination of Nitrite—Molecular Absorption Spectrometric Method; ISO 6777:1984; International Organization for Standardization: Geneva, Switzerland, 1984. [Google Scholar]
- International Organization for Standardization. Water Quality—Determination of Phosphorus—Ammonium Molybdate Spectrometric Method; ISO 6878:2004; International Organization for Standardization: Geneva, Switzerland, 2004. [Google Scholar]
- Rott, E.; Minke, R.; Bali, U.; Steinmetz, H. Removal of phosphonates from industrial wastewater with UV/FeII, Fenton and UV/Fenton treatment. Water Res. 2017, 122, 345–354. [Google Scholar] [CrossRef] [PubMed]
- Aronson, D.; Citra, M.; Shuler, K.; Printup, H.; Howard, P.H. Aerobic Biodegradation of Organic Chemicals in Environmental Media: A Summary of Field and Laboratory Studies; Final Report; U.S. Environmental Protection Agency: Washington, DC, USA, 1999; Available online: http://energytomorrow.org/~/media/files/ehs/clean_water/gw_other/aerobicbiodegrateconstantrpt1999pdf.pdf (accessed on 11 December 2017).
- Babu, G.; Varghese, W. Biodegradation of Cyclohexanone and Cyclohexanol by the Activated Sludge Process. J. Sci. Ind. Res. 1999, 58, 864–868. [Google Scholar]
- Hach Lange. Unpublished List of EC50 Values Determined by the Luminescent Bacteria Test Issued by Hach Lange; Hach Lange: Berlin, Germany, 2012. [Google Scholar]
- Rott, E.; Minke, R.; Steinmetz, H. Removal of phosphorus from phosphonate-loaded industrial wastewaters via precipitation/flocculation. J. Water Process Eng. 2017, 17, 188–196. [Google Scholar] [CrossRef]
- Barja, B.C.; dos Santos Afonso, M. An ATR–FTIR Study of Glyphosate and Its Fe(III) Complex in Aqueous Solution. Environ. Sci. Technol. 1998, 32, 3331–3335. [Google Scholar] [CrossRef]
- Duan, J.; Gregory, J. Coagulation by hydrolysing metal salts. Adv. Colloid Interface Sci. 2003, 100–102, 475–502. [Google Scholar] [CrossRef]
- Barrera-Dı́az, C.; Ureña-Nuñez, F.; Campos, E.; Palomar-Pardavé, M.; Romero-Romo, M. A combined electrochemical-irradiation treatment of highly colored and polluted industrial wastewater. Radiat. Phys. Chem. 2003, 67, 657–663. [Google Scholar] [CrossRef]
- Bozkaya-Schrotter, B.; Daines, C.; Lescourret, A.-S.; Bignon, A.; Breant, P.; Schrotter, J.C. Treatment of trace organics in membrane concentrates I: Pesticide elimination. Water. Sci. Tech. 2008, 8, 223–230. [Google Scholar] [CrossRef]
- Huston, P.L.; Pignatello, J.J. Degradation of selected pesticide active ingredients and commercial formulations in water by the photo-assisted Fenton reaction. Water Res. 1999, 33, 1238–1246. [Google Scholar] [CrossRef]
- Sheals, J.; Sjöberg, S.; Persson, P. Adsorption of Glyphosate on Goethite: Molecular Characterization of Surface Complexes. Environ. Sci. Technol. 2002, 36, 3090–3095. [Google Scholar] [CrossRef] [PubMed]
- Goto, T.; Amano, Y.; Machida, M.; Imazeki, F. Effect of polarity of activated carbon surface, solvent and adsorbate on adsorption of aromatic compounds from liquid phase. Chem. Pharm. Bull. 2015, 63, 726–730. [Google Scholar] [CrossRef] [PubMed]
- Foo, K.Y.; Hameed, B.H. Detoxification of pesticide waste via activated carbon adsorption process. J. Hazard. Mater. 2010, 175, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Barbusiński, K.; Filipek, K. Use of fenton’s reagent for removal of pesticides from industrial wastewater. Pol. J. Environ. Stud. 2001, 10, 207–212. [Google Scholar]
- Ayranci, E.; Hoda, N. Adsorption of bentazon and propanil from aqueous solutions at the high area activated carbon-cloth. Chemosphere 2004, 57, 755–762. [Google Scholar] [CrossRef] [PubMed]
- Kim, T.-Y.; Park, S.-S.; Kim, S.-J.; Cho, S.-Y. Separation characteristics of some phenoxy herbicides from aqueous solution. Adsorption 2008, 14, 611–619. [Google Scholar] [CrossRef]

| Parameter | Raw Sample | FeΙΙΙ | PAC | Fenton |
|---|---|---|---|---|
| Turbidity (NTU) | 107 | 0.98 | 0.80 | 0.61 |
| Color | greenish | colorless | colorless | colorless |
| pH | 6.0 | 7.0 | 6.0 | 7.0 |
| COD (mg/L) | 903 | 318 | 354 | 50.3 |
| Total P (mg/L) | 71.2 | 19.3 | 70.4 | <0.50 |
| o-PO43−-P (mg/L) | 5.17 | 1.12 | 5.16 | <0.50 |
| EC20 (LBT) (mL/L) | 3.1 | 16.7 | 179 | 652 |
| EC50 (LBT) (mL/L) | 9.2 | 79.5 | 478 | 886 |
| EC80 (LBT) (mL/L) | 20.5 | 253 | 990 | >1000 |
| EC20 (NIT/RIT) (mL/L) | 139/228 | 137/260 | 701/399 | >1000/>1000 |
| EC50 (NIT/RIT) (mL/L) | 293/481 | 380/395 | 900/>1000 | >1000/>1000 |
| Parameter | NIT | RIT | LBT |
|---|---|---|---|
| ISO | 9509 | 8192 | 11348 |
| Sensitivity | High | Low–High * | Very High |
| Use of activated sludge | Yes | Yes | No |
| Nitrification inhibition | Yes | Yes | No |
| Heterotrophy inhibition | No | Yes | No |
| Reliability | Good | Little | Very good |
| Sample volume | Up to 1 L | Up to 1 L | mL-range |
| Personnel expenses | Extensive | Extensive | Low |
| Total working time a,b | 5.5 h | 4.25 h (2.75 h) c | 1.5 h |
| Sample preparation a,b | 0.75 h | 1.25 h (1 h) c | 0.75 h |
| Measurement a,b | 4.00 h | 3 h (1.75 h) c | 0.75 h |
| Analysis a | 0.75 h | - | - |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rott, E.; Pittmann, T.; Wasielewski, S.; Kugele, A.; Minke, R. Detoxification of Pesticide-Containing Wastewater with FeIII, Activated Carbon and Fenton Reagent and Its Control Using Three Standardized Bacterial Inhibition Tests. Water 2017, 9, 969. https://doi.org/10.3390/w9120969
Rott E, Pittmann T, Wasielewski S, Kugele A, Minke R. Detoxification of Pesticide-Containing Wastewater with FeIII, Activated Carbon and Fenton Reagent and Its Control Using Three Standardized Bacterial Inhibition Tests. Water. 2017; 9(12):969. https://doi.org/10.3390/w9120969
Chicago/Turabian StyleRott, Eduard, Timo Pittmann, Stephan Wasielewski, Amélie Kugele, and Ralf Minke. 2017. "Detoxification of Pesticide-Containing Wastewater with FeIII, Activated Carbon and Fenton Reagent and Its Control Using Three Standardized Bacterial Inhibition Tests" Water 9, no. 12: 969. https://doi.org/10.3390/w9120969
APA StyleRott, E., Pittmann, T., Wasielewski, S., Kugele, A., & Minke, R. (2017). Detoxification of Pesticide-Containing Wastewater with FeIII, Activated Carbon and Fenton Reagent and Its Control Using Three Standardized Bacterial Inhibition Tests. Water, 9(12), 969. https://doi.org/10.3390/w9120969





