The Biomass and Physiological Responses of Vallisneria natans (Lour.) Hara to Epiphytic Algae and Different Nitrate-N Concentrations in the Water Column
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials and Study Site
2.2. Experimental Design
2.3. Epiphytic Algae Quantification and Classification
2.4. Chlorophyll, Malondialdehyde (MDA), Free Proline, Soluble Carbohydrates, and Soluble Proteins Analysis of V. natans
2.5. Enzyme Extraction and Assay of Superoxide Dismutase (SOD), Peroxidase (POD), and Catalase (CAT) Activity
2.6. Statistical Analysis
3. Result
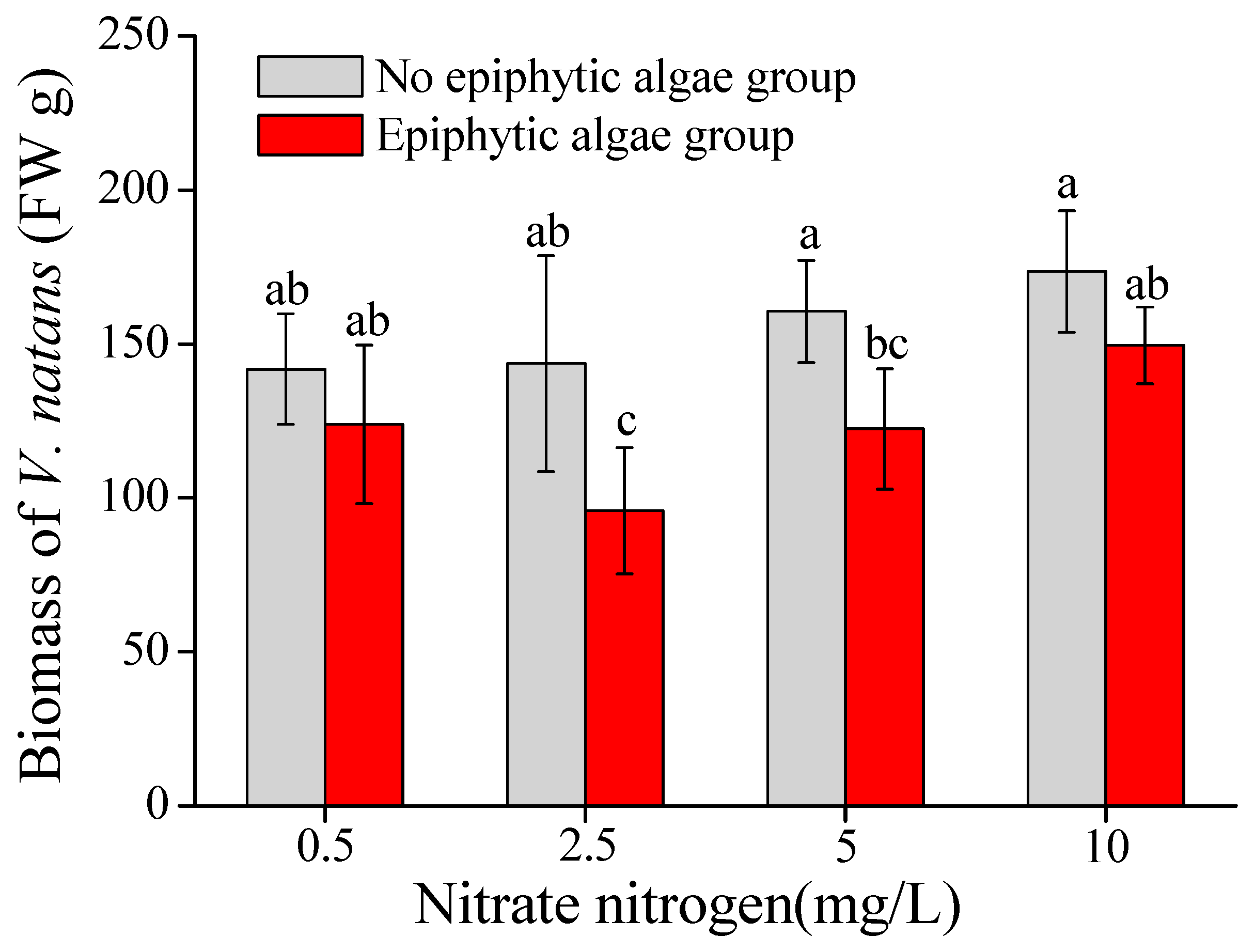
3.1. Biomass of V. natans
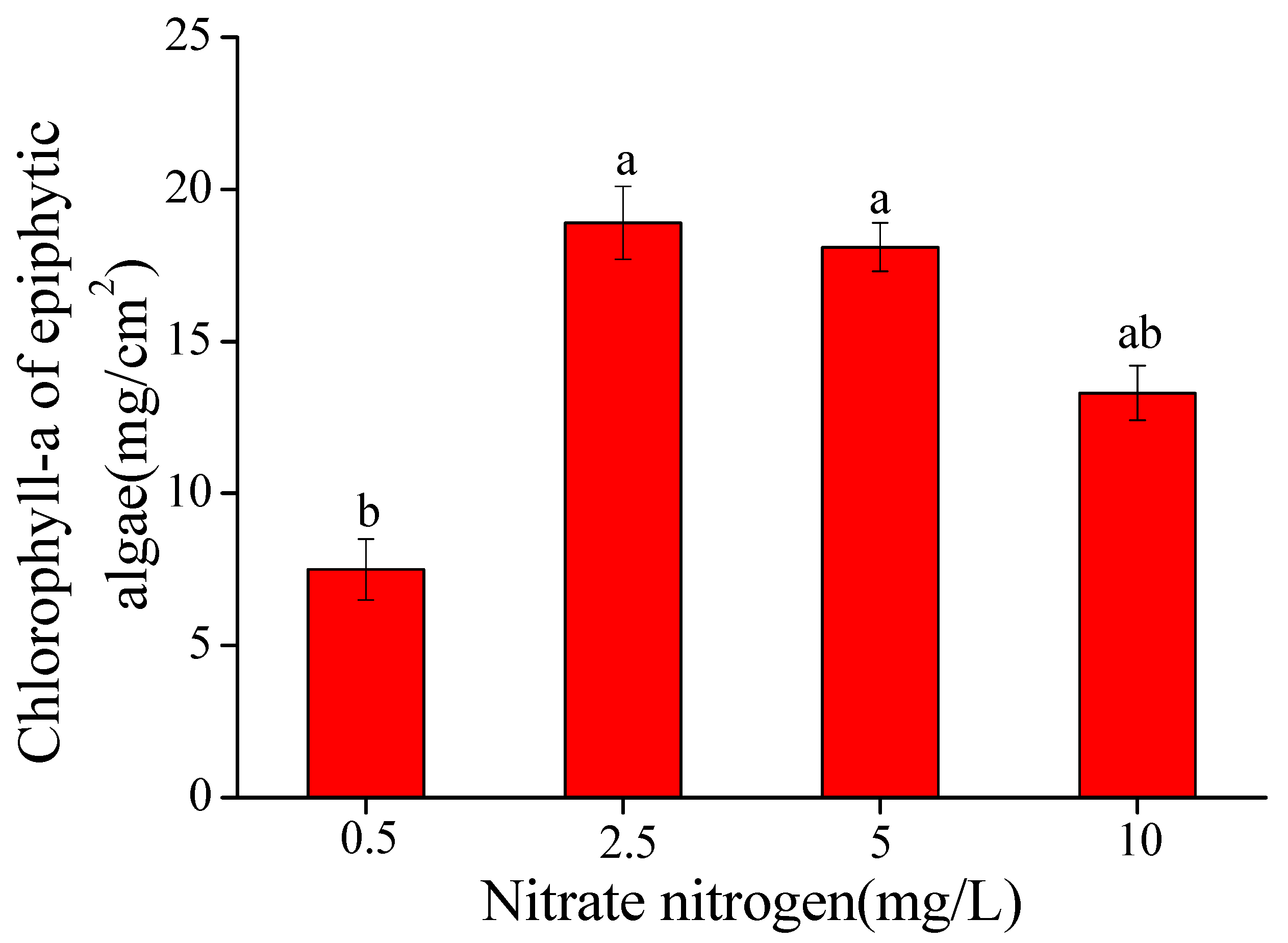
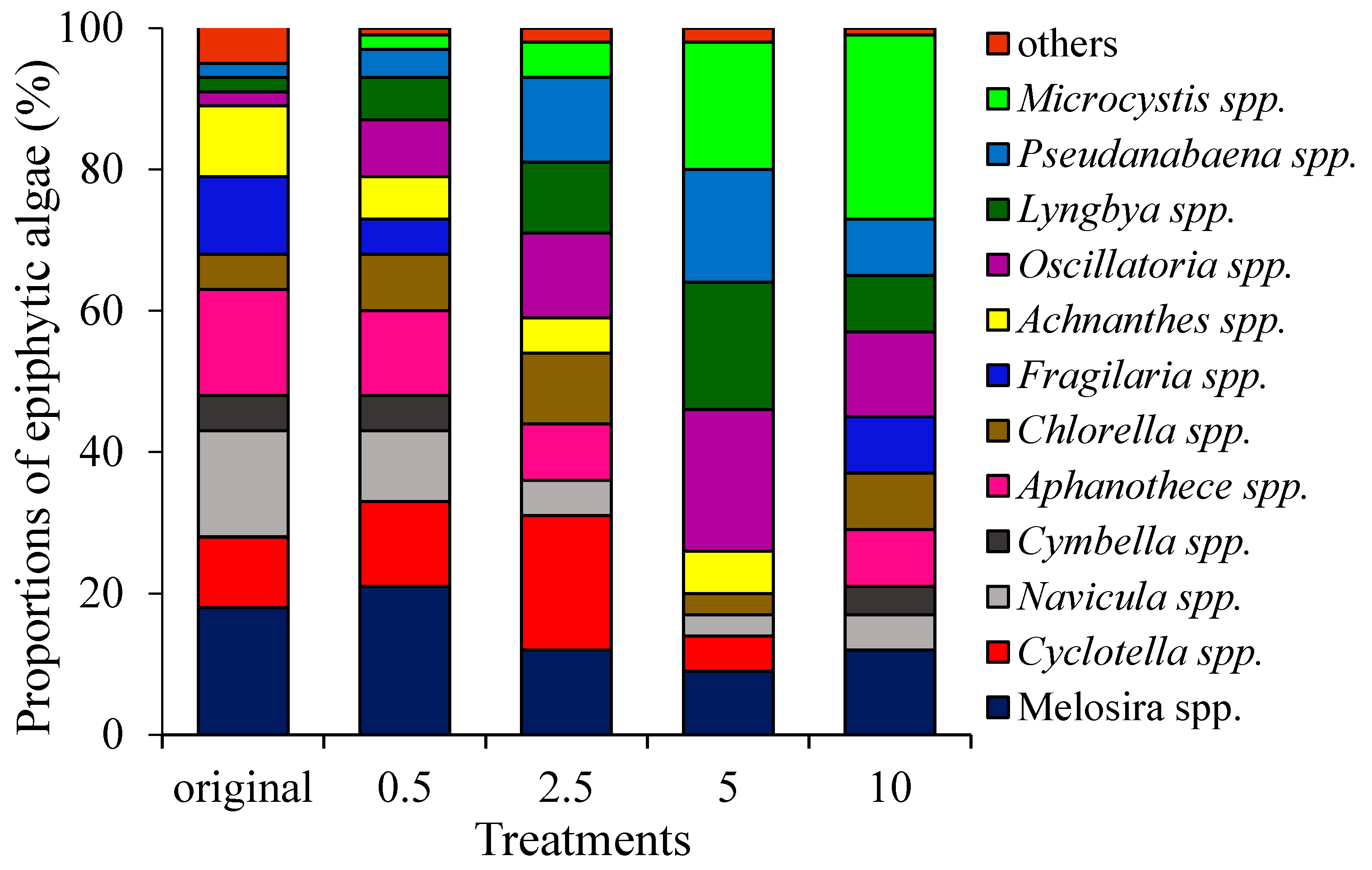
3.2. Biomass and Community Composition of Epiphytic Algae
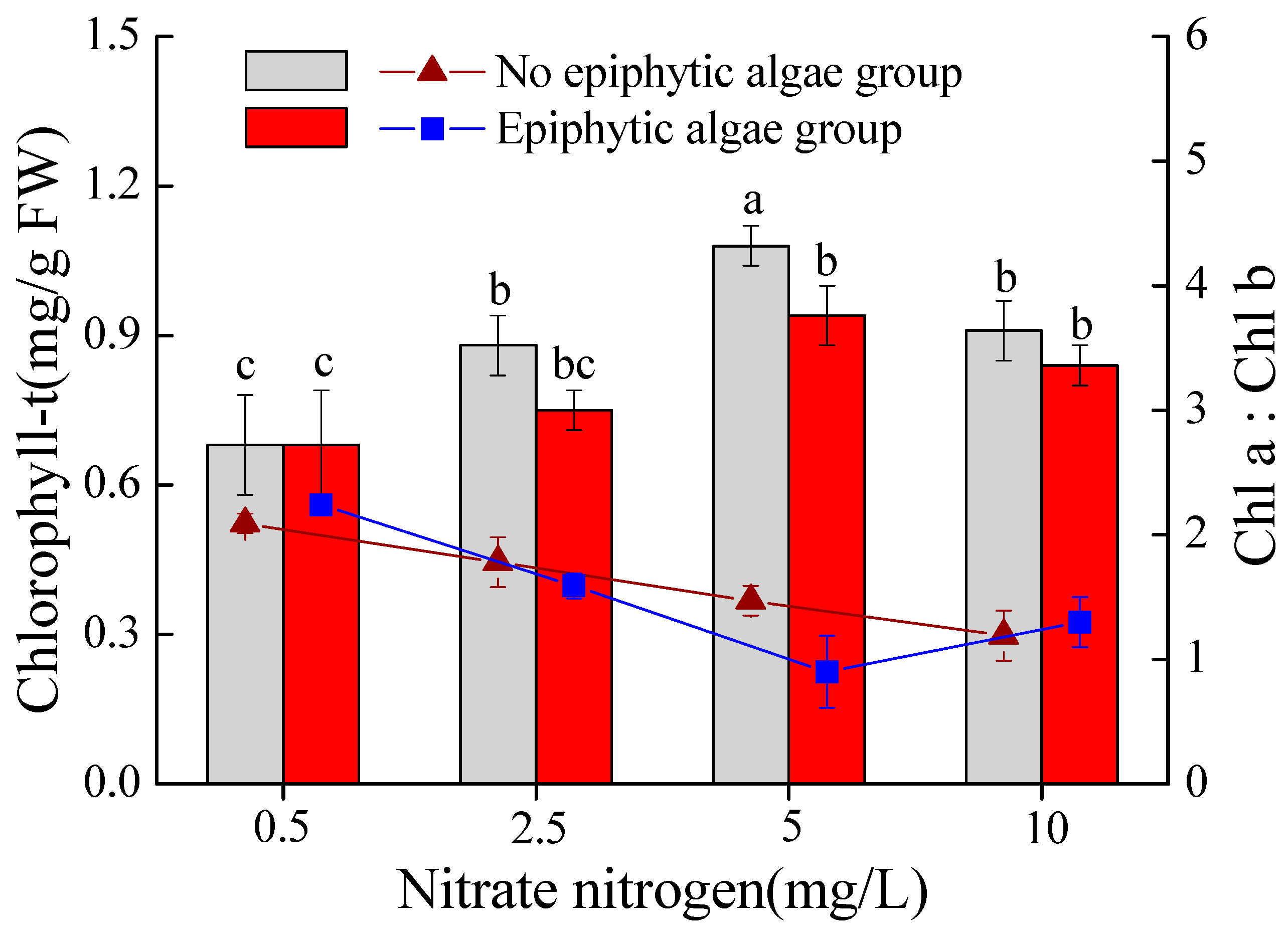
3.3. Total Chlorophyll Content and Chl-a/Chl-b Ratio in Leaves of V. natans
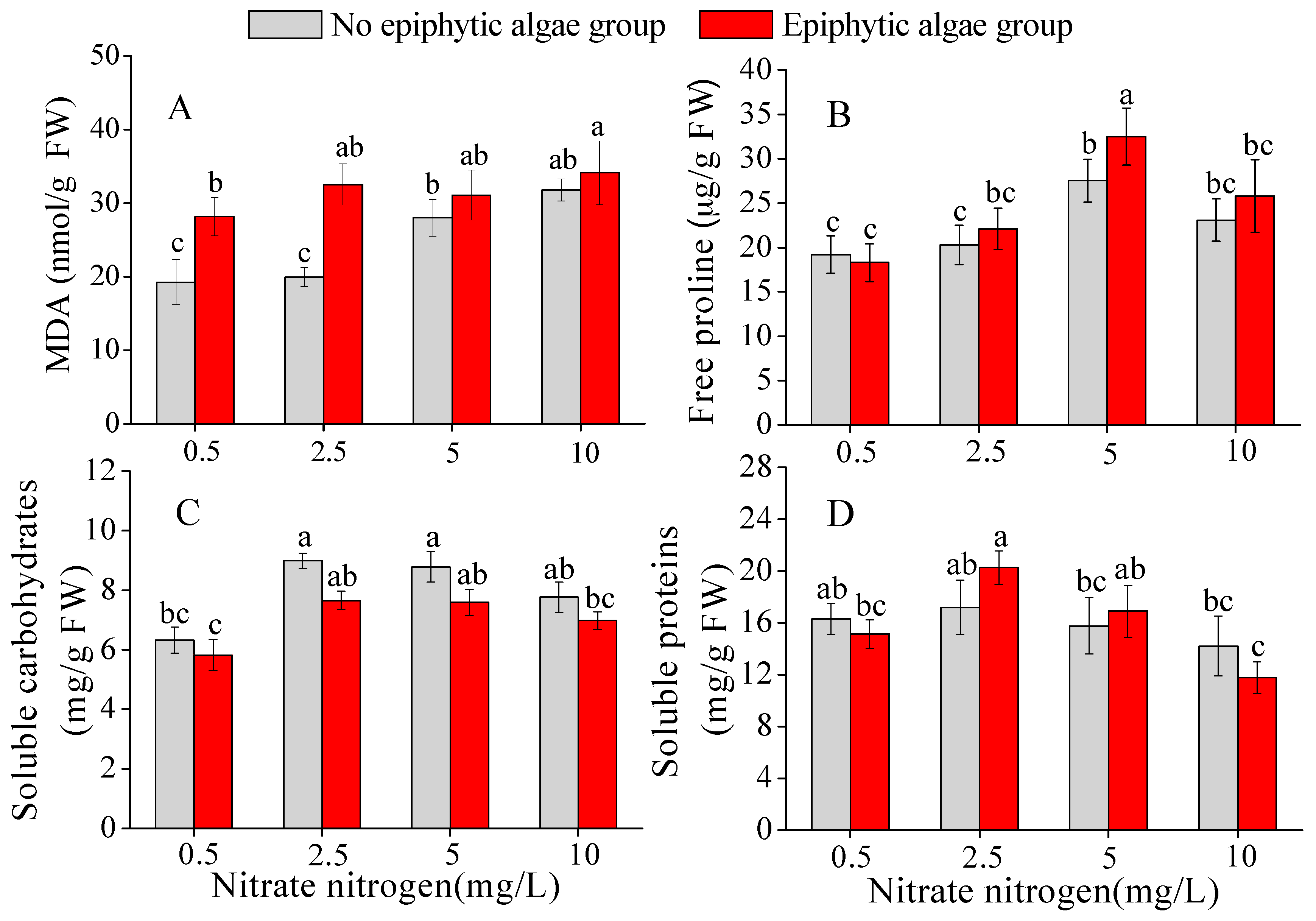
3.4. MDA Content in Leaves of V. natans
3.5. Free Proline Content in Leaves of V. natans
3.6. Soluble Carbohydrates in Leaves of V. natans
3.7. Soluble Proteins in Leaves of V. natans
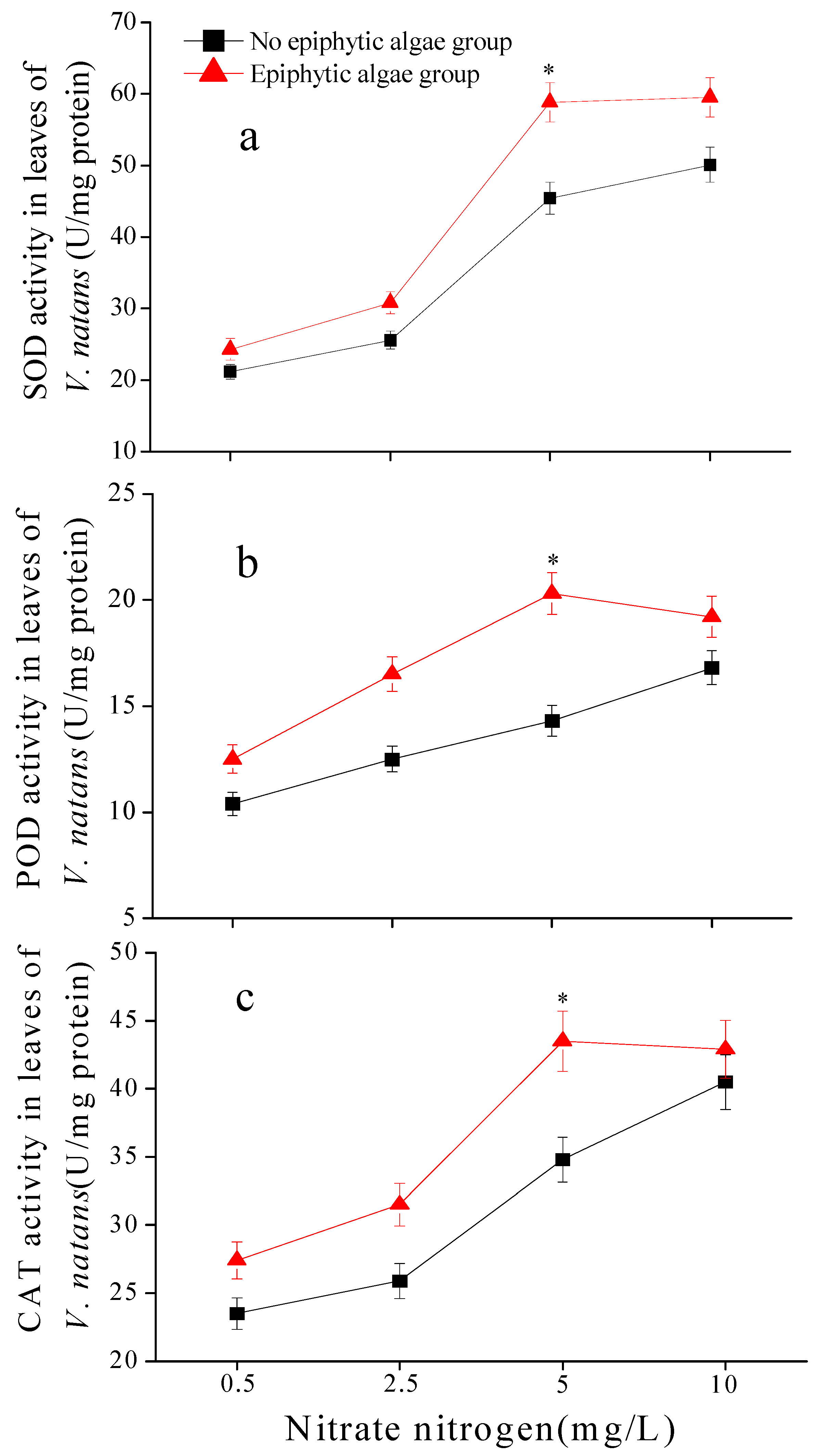
3.8. Antioxidant Enzyme Activity (SOD, POD, CAT) in Leaves of V. natans
4. Discussion
5. Conclusions
- Increasing nitrate-N concentration in the water column affected the growth of epiphytic algae on V. natans. Epiphytic algal biomass attached on leaves of V. natans reached the peak when the nitrate-N concentration was 2.5–5 mg/L, then decreased when nitrate-N was higher.
- High density of epiphytic algae could significantly inhibit the biomass accumulation of V. natans, while increasing the nitrate-N concentration (2.5 mg/L–10 mg/L) in the water column, which could counteract the adverse effect of epiphytic algae and promote the biomass accumulation of V. natans.
- Both epiphytic algae and a high nitrate-N concentration (above 5 mg/L) could severally or synergistically produce adverse effects on V. natans by influencing physiological indexes, which could hurt and inhibit the growth of V. natans.
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Beklioğlu, M.; Bucak, T.; Coppens, J.; Bezirci, G.; Tavşanoğlu, Ü.N.; Ҫakıroğlu, A.İ.; Levi, E.E.; Erdoğan, Ş.; Filiz, N.; Özkan, K.; et al. Restoration of eutrophic lakes with fluctuating water levels: A 20-year monitoring study of two inter-connected lakes. Water 2017, 9, 127. [Google Scholar] [CrossRef]
- Zhang, Y.; Liu, X.; Qin, B.; Shi, K.; Deng, J.; Zhou, Y. Aquatic vegetation in response to increased eutrophication and degraded light climate in Eastern Lake Taihu: Implications for lake ecological restoration. Sci. Rep. 2016, 6, 23867. [Google Scholar] [CrossRef] [PubMed]
- Orth, R.J.; Carruthers, T.J.B.; Dennison, W.C.; Duarte, C.M.; Fourqurean, J.W.; Heck, K.L., Jr.; Hughes, A.R.; Kendrick, G.A.; Kenworthy, W.J.; Olyarnik, S.; et al. A global crisis for seagrass ecosystems. AIBS Bull. 2006, 56, 987–996. [Google Scholar] [CrossRef]
- Van Donk, E.; van de Bund, W.J. Impact of submerged macrophytes including charophytes on phyto- and zooplankton communities: Allelopathy versus other mechanisms. Aquat. Bot. 2002, 72, 261–274. [Google Scholar] [CrossRef]
- Rodrigo, M.A.; Rojo, C.; Alonso-Guillén, J.L.; Vera, P. Restoration of two small Mediterranean lagoons: The dynamics of submerged macrophytes and factors that affect the success of revegetation. Ecol. Eng. 2013, 54, 1–15. [Google Scholar] [CrossRef]
- Gonzalez-Sagrario, M.A.; Jeppesen, E.; Goma, J.; Søndergaard, M.; Jensen, J.P.; Lauridsen, T.; Landkildehus, F. Does high nitrogen loading prevent clear-water conditions in shallow lakes at moderately high phosphorus concentrations? Freshw. Biol. 2005, 50, 27–41. [Google Scholar] [CrossRef]
- Wang, H.; Wang, H.; Liang, X.; Wu, S. Total phosphorus thresholds for regime shifts are nearly equal in subtropical and temperate shallow lakes with moderate depths and areas. Freshw. Biol. 2014, 59, 1659–1671. [Google Scholar] [CrossRef]
- Schindler, D.W. Eutrophication and recovery in experimental lakes: Implications for lake management. Science 1974, 184, 897–899. [Google Scholar] [CrossRef] [PubMed]
- Sand-Jensen, K.; Pedersen, N.L.; Thorsgaard, I.; Moeslund, B.; Borum, J.; Brodersen, K.P. 100 years of vegetation decline and recovery in Lake Fure, Denmark. J. Ecol. 2008, 96, 260–271. [Google Scholar] [CrossRef]
- Moss, B.; Jeppesen, E.; Søndergaard, M.; Lauridsen, T.L.; Liu, Z.W. Nitrogen, macrophytes, shallow lakes and nutrient limitation: Resolution of a current controversy? Hydrobiologia 2013, 710, 3–21. [Google Scholar] [CrossRef]
- Yu, Q.; Wang, H.; Li, Y.; Shao, J.; Liang, X.; Jeppesen, E.; Wang, H. Effects of high nitrogen concentrations on the growth of submersed macrophytes at moderate phosphorus concentrations. Water Res. 2015, 83, 385–395. [Google Scholar] [CrossRef] [PubMed]
- Barker, T.; Hatton, K.; O’Connor, M.; Connor, L.; Moss, B. Effects of nitrate load on submerged plant biomass and species richness: Results of a mesocosm experiment. Fundam. Appl. Limnol. 2008, 173, 89–100. [Google Scholar] [CrossRef]
- James, C.; Fisher, J.; Russell, V.; Collings, S.; Moss, B. Nitrate availability and hydrophyte species richness in shallow lakes. Freshw. Biol. 2005, 50, 1049–1063. [Google Scholar] [CrossRef]
- Food and Agriculture Organization Corporate Statistical Database (FAOSTAT). Fertilizers Consumption 2002–2012; Food and Agricultural Organization of the United Nations: Rome, Italy, 2014. [Google Scholar]
- Iversen, T.M.; Grant, K.; Nielsen, K. Nitrogen enrichment of European inland and marine waters with special attention to Danish policy measures. Environ. Pollut. 1998, 102, 771–780. [Google Scholar] [CrossRef]
- International Lake Environment Committee (ILEC). World Lakes Database, Survey of the State of World Lakes; ILEC: Kusatsu, Japan, 2004. [Google Scholar]
- Wetzel, R.G. Limnology, Lake and River Ecosystems; Academic Press: London, UK, 2001. [Google Scholar]
- Best, M.D.; Mantai, K.E. Growth of Myriophyllum: Sediment or lake water as the source of nitrogen and phosphorus. Ecology 1978, 59, 1075–1080. [Google Scholar] [CrossRef]
- Gu, J.; Xu, Z.; Jin, H.; Ning, X.; He, H.; Yu, J.; Jeppesen, E.; Li, K. Response of Vallisneria natans to increasing nitrogen loading depends on sediment nutrient characteristics. Water 2016, 8, 563. [Google Scholar] [CrossRef]
- Burkholder, J.M.; Mason, K.M.; Glasgow, H.B. Water column nitrate enrichment promotes decline of eelgrass Zostera marina: Evidence from seasonal mesocosm experiments. Mar. Ecol. Prog. Ser. 1992, 81, 163–178. [Google Scholar] [CrossRef]
- Wang, C.; Zhang, S.; Wang, P.; Hou, J.; Li, W.; Zhang, W.J. Metabolic adaptations to ammonia-induced oxidative stress in leaves of the submerged macrophyte Vallisneria natans (Lour.) Hara. Aquat. Toxicol. 2008, 87, 88–98. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Wang, S.; Jiao, L.; Zhao, H.; Zhang, Y.; Li, Y. Physiological response of a submerged plant (Myriophyllum spicatum) to different NH4Cl concentrations in sediments. Ecol. Eng. 2013, 58, 91–98. [Google Scholar] [CrossRef]
- Phillips, G.L.; Eminson, D.; Moss, B. A mechanism to account for macrophyte decline in progressively eutrophicated freshwaters. Aquat. Bot. 1978, 4, 103–126. [Google Scholar] [CrossRef]
- Romo, S.; Villena, M.M.A. Epiphyton, phytoplankton and macrophyte ecology in a shallow lake under in situ experimental conditions. Fundam. Appl. Limnol. 2007, 170, 197–209. [Google Scholar] [CrossRef]
- Xing, W.; Wu, H.; Hao, B.; Liu, G. Stoichiometric characteristics and responses of submerged macrophytes to eutrophication in lakes along the middle and lower reaches of the Yangtze River. Ecol. Eng. 2013, 54, 16–21. [Google Scholar] [CrossRef]
- He, H.; Liu, X.; Liu, X.; Yu, J.; Li, K.; Guan, B.; Jeppesen, E.; Liu, Z. Effects of cyanobacterial blooms on submerged macrophytes alleviated by the native Chinese bivalve Hyriopsis cumingii: A mesocosm experiment study. Ecol. Eng. 2014, 71, 363–367. [Google Scholar] [CrossRef]
- Van Geest, G.J.; Coops, H.; Roijackers, R.M.M.; Buijse, A.D.; Scheffer, M. Succession of aquatic vegetation driven by reduced water-level fluctuations in floodplain lakes. J. Appl. Ecol. 2005, 42, 251–260. [Google Scholar] [CrossRef]
- Sayer, C.D.; Burgess, A.; Kari, K.; Davidson, T.A.; Peglar, S.; Yang, H.; Rose, N. Long-term dynamics of submerged macrophytes and algae in a small and shallow, eutrophic lake: Implications for the stability of macrophyte-dominance. Freshw. Biol. 2010, 55, 565–583. [Google Scholar] [CrossRef]
- Jones, J.I.; Sayer, C. Does fish invertebrate periphyton cascade precipitate plant loss in shallow lakes? Ecology 2003, 84, 2155–2167. [Google Scholar] [CrossRef]
- Bécares, E.; Gomá, J.; Fernández-Aláez, M.; Fernández-Aláez, C.; Romo, S.; Miracle, M.R.; Ståhl-Delbanco, A.; Hansson, L.A.; Gyllström, M.; Van de Bund, W.J.; et al. Effects of nutrients and fish on periphyton and plant biomass across a European latitudinal gradient. Aquat. Ecol. 2008, 42, 561–574. [Google Scholar] [CrossRef]
- Fong, C.; Lee, S.; Wu, R. The effects of epiphytic algae and their grazers on the intertidal seagrass Zostera japonica. Aquat. Bot. 2000, 67, 251–261. [Google Scholar] [CrossRef]
- Sand-Jensen, K.; Søndergaard, M. Phytoplankton and epiphyte development and their shading effect on submerged macrophytes in lakes of different nutrient status. Int. Rev. Hydrobiol. 1981, 66, 529–552. [Google Scholar] [CrossRef]
- Asaeda, T.; Sultana, M.; Manatunge, J.; Fujino, T. The effect of epiphytic algae on the growth and production of Potamogeton perfoliatus L. in two light conditions. Environ. Exp. Bot. 2004, 52, 225–238. [Google Scholar] [CrossRef]
- Chen, C.; Yin, D.; Yu, B.; Zhu, H. Effect of epiphytic algae on photosynthetic function of Potamogeton crispus. J. Freshw. Ecol. 2007, 22, 411–420. [Google Scholar] [CrossRef]
- Hilt, S.; Gross, E.M. Can allelopathically active submerged macrophytes stabilize clear-water states in shallow lakes? Basic Appl. Ecol. 2008, 9, 422–432. [Google Scholar] [CrossRef]
- Dent, C.L.; Cumming, G.S.; Carpenter, S.R. Multiple states in river and lake ecosystems. Philos. Trans. R. Soc. Lond. B 2002, 357, 635–645. [Google Scholar] [CrossRef] [PubMed]
- Scheffer, M.; Carpenter, S.; Foley, J.A.; Folke, C.; Walker, B. Catastrophic shifts in ecosystems. Nature 2001, 413, 591–596. [Google Scholar] [CrossRef] [PubMed]
- Song, Y.; Kong, F.; Xue, Y.; Qin, B. Responses of chlorophyll and MDA of Vallisneria natans to nitrogen and phosphorus availability and epiphytic algae. J. Freshw. Ecol. 2015, 30, 85–97. [Google Scholar] [CrossRef]
- Yi, K.; Dai, Z.; Liu, B.; Lin, Q.; Zeng, L.; Xu, D.; He, F.; Wu, Z. Effects of vertical location and artificial substrate on colonization of algal periphyton. Acta Ecol. Sin. 2016, 36, 4864–4872. [Google Scholar]
- Arnon, D.I. Copper enzymes in isolated chloroplasts. Polyphenoloxidase in beta vulgaris. Plant Physiol. 1949, 24, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.; Huang, X. Freshwater Plankton Research Methods; Science Press: Beijing, China, 1991. [Google Scholar]
- Heath, R.L.; Packer, L. Photoperoxidation in isolated chloroplasts: I. Kinetics and stoichiometry of fatty acid peroxidation. Arch. Biochem. Biophys. 1968, 125, 189–198. [Google Scholar] [CrossRef]
- Bates, L.S.; Waldren, R.P.; Teare, I.D. Rapid determination of free proline for water-stress studies. Plant Soil 1973, 39, 205–207. [Google Scholar] [CrossRef]
- Yemm, E.W.; Willis, A.J. The estimation of carbohydrates in plant extracts by anthrone. Biochemistry 1954, 57, 508–514. [Google Scholar] [CrossRef]
- Bradford, M.M. Arapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal. Biochem. 1976, 72, 248–254. [Google Scholar] [CrossRef]
- Li, H. The Experiment Principle and Technology of Plant Physiology; Higher Education Press: Beijing, China, 2000; pp. 267–268. [Google Scholar]
- Reigosa, M.J.; Sánchez-Moreiras, A.; González, L. Ecophysiological approach in allelopathy. Crit. Rev. Plant Sci. 1999, 18, 577–608. [Google Scholar] [CrossRef]
- Song, Y.; Wang, J.; Gao, Y. Effects of epiphytic algae on biomass and physiology of Myriophyllum spicatum L. with the increase of nitrogen and phosphorus availability in the water body. Environ. Sci. Pollut. Res. 2017, 24, 9548–9555. [Google Scholar] [CrossRef] [PubMed]
- Shelford, V.E. Conditions of Existence; Wiley: New York, NY, USA, 1918; pp. 21–60. [Google Scholar]
- Wetzel, R.G. Attached algal-substrata interactions: Fact or myth, and when and how? In Periphyton of Freshwater Ecosystems; Junk: The Hague, The Netherlands, 1983; pp. 207–215. [Google Scholar]
- Allen, H.L. Primary productivity, chemo-organo-trophy, and nutritional interactions of epiphytic algae and bacteria on macrophytes in the littoral of a lake. Ecol. Monogr. 1971, 41, 97–127. [Google Scholar] [CrossRef]
- Thomas, J.D. Mutualistic interactions in freshwater modular systems with molluscan components. Adv. Ecol. Res. 1990, 20, 125–178. [Google Scholar]
- Jones, I.J.; Moss, B.; Eaton, J.W.; Young, J.O. Do submerged aquatic plants influence periphyton community composition for the benefit of invertebrate mutualists? Freshw. Biol. 2000, 43, 591–604. [Google Scholar] [CrossRef]
- Toporowska, M.; Pawlik-Skowrońska, B.; Wojtal, A.Z. Epiphytic algae on Stratiotes aloides L., Potamogeton lucens L., Ceratophyllum demersum L. and Chara spp. in a macrophyte-dominated lake. Oceanol. Hydrobiol. Stud. 2008, 2, 51–63. [Google Scholar] [CrossRef]
- Su, S.; Zhou, Y.; Qin, J.; Wang, W.; Yao, W.; Song, L. Physiological responses of Egeria densa to high ammonium concentration and nitrogen deficiency. Chemosphere 2012, 86, 538–545. [Google Scholar]
- Wu, W. Plant Physiology, 2nd ed.; Science Press: Beijing, China, 2012. [Google Scholar]
- Chen, Y.; He, Y.; Luo, Y.; Yu, Y.; Lin, Q.; Wong, M. Physiological mechanism of plant roots exposed to cadmium. Chemosphere 2003, 50, 789–793. [Google Scholar] [CrossRef]
- Huang, W.; Jia, Z.; Han, Q. Effects of herbivore stress by Aphis medicaginis Koch on the malondialdehyde contents and the activities of protective enzymes in different alfalfa varieties. Acta Ecol. Sin. 2007, 27, 2177–2183. [Google Scholar]
- Atanasova, E. Effect of nitrogen sources on the nitrogenous forms and accumulation of amino acid in Head Cabbage. Plant Soil Environ. 2008, 54, 66–71. [Google Scholar]
- Neuberg, M.; Pavlíková, D.; Pavlík, M.; Balík, J. The effect of different nitrogen nutrition on proline and asparagine content in plant. Plant Soil Environ. 2010, 56, 305–311. [Google Scholar]
- Harborne, J.B.; Turner, B.L. Plant Chemosysternatics; Academic Press: Cambridge, UK, 1984; pp. 216–232. [Google Scholar]
- Costa, G.; Spitz, E. Influence of cadmiumon soluble carbohydrates, free amino acids, protein content of In Vitro cultured Lupinus albus. Plant Sci. 1997, 128, 131–140. [Google Scholar] [CrossRef]
- Touchette, B.W.; Burkholder, J.M. Review of nitrogen and phosphorus metabolism in seagrasses. J. Exp. Mar. Biol. Ecol. 2000, 250, 133–167. [Google Scholar] [CrossRef]
- Amini, F.; Ehsanpour, A.A. Soluble proteins, proline, carbohydrates and Na+/K+ changes in two tomato (Lycopersicon esculentum Mill.) Cultivars under in vitro salt stress. Am. J. Biochem. Biotechnol. 2005, 1, 204–208. [Google Scholar] [CrossRef]
- Yan, C.; Zeng, A.; Jin, X.; Zhao, J.; Xu, Q.; Wang, X. Physiological effects of ammonia-nitrogen concentrations on Hydrilla verticillata. Acta Ecol. Sin. 2007, 27, 1050–1055. [Google Scholar]
- Palma, J.M.; Sandalio, L.M.; Corpas, F.J.; Romero-Puertas, M.C.; McCarthy, I.; Luis, A. Plant proteases protein degradation and oxidative stress: Role of peroxisomes. Plant Physiol. Biochem. 2002, 40, 521–530. [Google Scholar] [CrossRef]
- John, R.; Ahmad, P.; Sharma, S. Effect of cadmium and lead on growth, biochemical parameters and uptake in Lemna polyrrhiza L. Plant Soil Environ. 2008, 54, 262–270. [Google Scholar]
- Misra, N.; Gupta, A.K. Effect of salinity and different nitrogen sources on the activity of antioxidant enzymes and indole alkaloid content in Catharanthus roseus seedlings. J. Plant Physiol. 2006, 163, 11–18. [Google Scholar] [CrossRef] [PubMed]
- Yu, J.; Yang, Y. Physiological and biochemical response of seaweed Gracilaria lemaneiformis to concentration changes of N and P. J. Exp. Mar. Biol. Ecol. 2008, 367, 142–148. [Google Scholar] [CrossRef]
| Nitrate-N | Epiphytic Algae | Nitrate-N × Epiphytic Algae | ||||
|---|---|---|---|---|---|---|
| Parameters | F Value | p Value | F Value | p Value | F Value | p Value |
| Biomass | 4.987 | 0.012 * | 20.3 | <0.001 *** | 2.449 | 0.101 |
| Chl-t | 23.721 | <0.001 *** | 13.37 | 0.002 ** | 1.627 | 0.223 |
| Chl-a/Chl-b | 41.419 | <0.001 *** | 3.091 | 0.098 | 5.515 | 0.009 ** |
| MDA | 19.318 | <0.001 *** | 54.093 | <0.001 *** | 7.156 | 0.003 ** |
| Free proline | 22.356 | <0.001 *** | 1.628 | 0.22 | 1.77 | 0.193 |
| SC | 37.636 | <0.001 *** | 7.808 | 0.013 * | 9.697 | 0.001 ** |
| SP | 10.578 | <0.001 *** | 0.039 | 0.847 | 2.819 | 0.072 |
| SOD | 7.132 | 0.003 ** | 33.513 | <0.001 *** | 10.388 | <0.001 *** |
| POD | 75.899 | <0.001 *** | 16.473 | 0.001 ** | 20.205 | <0.001 *** |
| CAT | 32.843 | <0.001 *** | 16.065 | 0.001 ** | 13.838 | <0.001 *** |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Min, F.; Zuo, J.; Zhang, Y.; Lin, Q.; Liu, B.; Sun, J.; Zeng, L.; He, F.; Wu, Z. The Biomass and Physiological Responses of Vallisneria natans (Lour.) Hara to Epiphytic Algae and Different Nitrate-N Concentrations in the Water Column. Water 2017, 9, 863. https://doi.org/10.3390/w9110863
Min F, Zuo J, Zhang Y, Lin Q, Liu B, Sun J, Zeng L, He F, Wu Z. The Biomass and Physiological Responses of Vallisneria natans (Lour.) Hara to Epiphytic Algae and Different Nitrate-N Concentrations in the Water Column. Water. 2017; 9(11):863. https://doi.org/10.3390/w9110863
Chicago/Turabian StyleMin, Fenli, Jincheng Zuo, Yi Zhang, Qingwei Lin, Biyun Liu, Jian Sun, Lei Zeng, Feng He, and Zhenbin Wu. 2017. "The Biomass and Physiological Responses of Vallisneria natans (Lour.) Hara to Epiphytic Algae and Different Nitrate-N Concentrations in the Water Column" Water 9, no. 11: 863. https://doi.org/10.3390/w9110863
APA StyleMin, F., Zuo, J., Zhang, Y., Lin, Q., Liu, B., Sun, J., Zeng, L., He, F., & Wu, Z. (2017). The Biomass and Physiological Responses of Vallisneria natans (Lour.) Hara to Epiphytic Algae and Different Nitrate-N Concentrations in the Water Column. Water, 9(11), 863. https://doi.org/10.3390/w9110863





