1. Introduction
Solar thermal energy is widely used in many countries [
1]. Solar water heaters (SWHs) are mainly composed of four parts: a solar collector, a water storage tank, piping, and a control system. The absorbing plate is generally made of copper, aluminum, stainless steel, or flexible polymer. Packing material between inside and outside barrels for a stainless-steel water storage tank is for thermal insulation. In Taiwan, the climatic conditions are predominantly sunny. The solar radiation ranges from 1200 to 1700 kWh/m
2/year. To disseminate SWHs, the Bureau of Energy, Ministry of Economic Affairs (BEMOEA, the first-stage national subsidy program: 1986–1991; the second-stage national subsidy program: 2000–present) and some regional governments have offered subsidy programs [
2]. The Energy Research Center at National Cheng Kung University (ERC/NCKU) has been authorized by the BEMOEA to organize an operation unit for the second national subsidy program starting in 2000. The accumulated area of solar collectors installed between 2000 and 2015 reached 1.62 million m
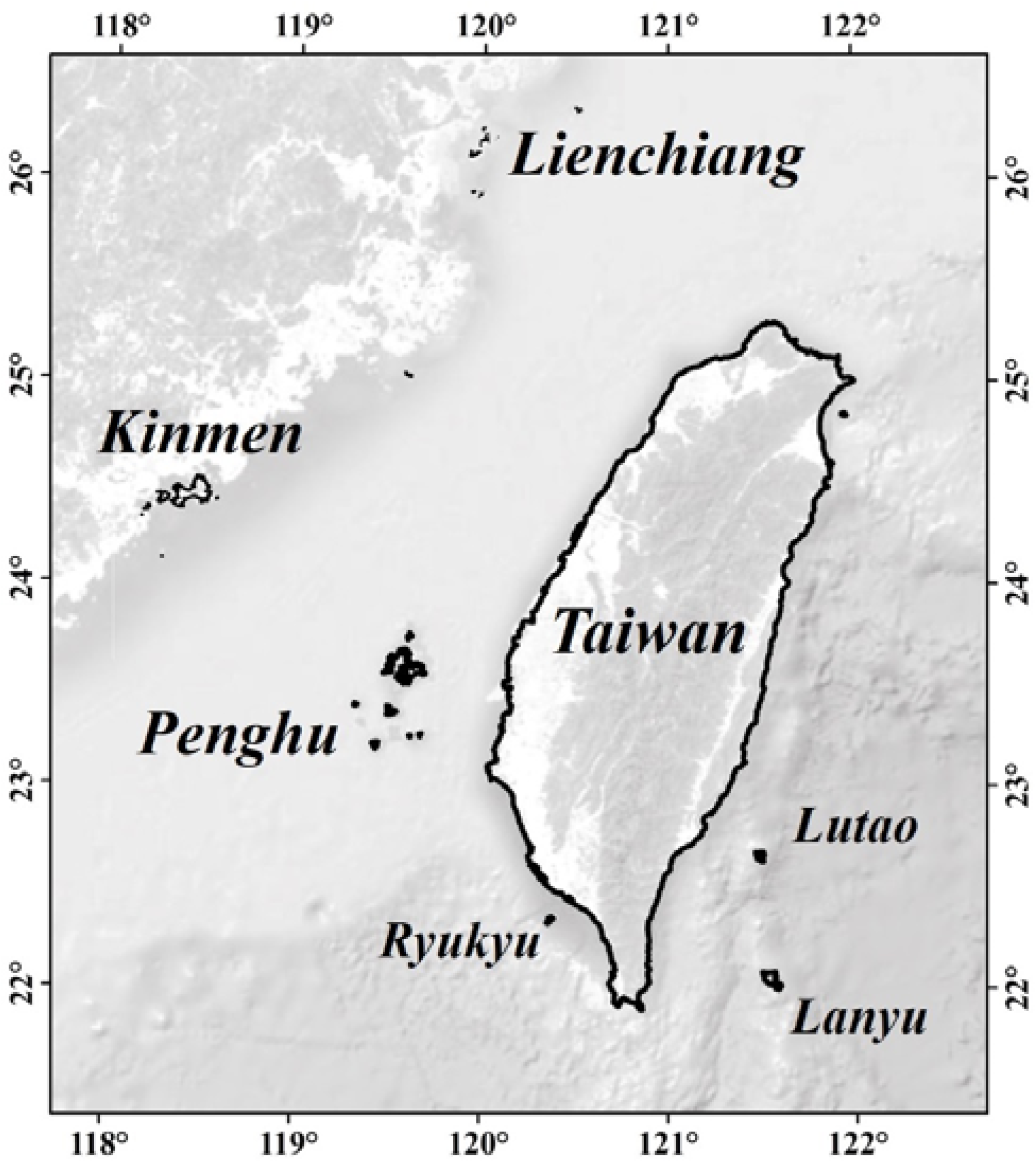
2. Taiwan, as a country (officially named Republic of China), is composed of a main island (named Taiwan island) and quite a few remote islands as shown in
Figure 1. There are three counties, namely Penghu, Kinmen, and Lienchiang, which are located out of the main island and constituted by remote islands. Note that the Low Carbon Island Project, including the Penghu and Kinmen counties, was also launched in 2012 and aimed at reducing the production of carbon dioxide in the remote islands of Taiwan. Promotion of SWHs was an initiative of the Project [
3].
A survey of promotion outcome of SWHs is of great concern to end users. In 2013, telephone interviews were conducted by the ERC/NCKU for 6482 SWHs installed in 2000, in which 3379 end users (52.1%) were reached. The survey showed that there were 95.3% systems in operation for 12 years at least. However, malfunctions of SWHs have been reported from time to time. In the period of 2000–2012, 942 cases were documented by the ERC/NCKU, in which 555 of them (58.9%) were damaged with leakage (corrosion or pitting) in absorbing plates, gate valves, storage tanks, or pipe joints.
In the main island of Taiwan, water resources are rich. Tap water is the principal domestic water supply (the population percentage is 91.56% in 2013) [
4], and its water quality is stable. In contrast, the principal domestic water sources in the remote islands are the tap water made mainly from desalination, ground water, or both [
3]. To assess the water quality in SWHs in the remote islands, this study sampled tap water and ground water from households with SWHs in the three counties constituted of the remote islands, namely Penghu, Kinmen, and Lienchiang. Analyses of water quality were conducted, including the parameters of pH, total alkalinity, total hardness, chloride, and sulfate. The water quality assessments by means of the methods of the Langelier Saturation Index (LSI), the Ryznar Stability Index (RSI), the Puckorius Scaling Index (PSI), and the Larson-Skold Corrosive Index (LSCI) were employed to estimate the tendency of scaling or corrosion [
5,
6,
7,
8,
9,
10,
11,
12,
13,
14]. The values of the LSI, the RSI, and the PSI are primarily used to estimate the scaling potential of calcium carbonate (CaCO
3) in water [
14]. Among these three methods, LSI is a forerunner and the most commonly used one [
11]. However, Seneviratne [
11] argued that LSI does not take into account the corrosivity of water because corrosion may still occur due to dissolved oxygen and solids such as chlorides or sulfates that increase the conductivity of water and corrosion rates. In contrast, the LSCI method proposed by Larson and Skold [
7] is suitable for assessing the corrosiveness of water on low-carbon steel. In this study, the water quality assessment is made with the LSI, RSI, PSI, and LSCI methods to estimate the levels of scaling and corrosion tendencies for either tap water or ground water sampled from SWHs in Penghu, Kinmen, and Lienchiang.
2. SWHs Installed in the Three Remote-Island Counties
As shown in
Figure 1, Penghu (119°19′–119°43′ E, 23°12′–23°47′ N), with 37,151 households, straddles the Tropic of Cancer (along 23°30′ N). There are five townships (Husi, Baisha, Siyu, Wang-An, and Chimei) and one city (Magong). Kinmen (118°24′–119°28′ E, 24°27′–24°59′ N), with 36,558 households, is made up of 12 jurisdictional islands and is located in the subtropical oceanic climate zone. There are six townships (Jincheng, Jingsha, Jinhu, Jinning, Lieyu, and Wuqiu). Lienchiang (119°54′–120°30′ E, 25°56′–26°44′ N), with 2423 households, which also lies within the subtropical oceanic climate zone, is the smallest county in Taiwan. There are four townships (Nangan, Beigan, Jyuguang, and Dongyin), of which Juguang is composed of two remote islands: Dongju Island and Xiju Island.
As previously mentioned, scaling and corrosion problems are of great concern for the promotion of SWHs in the remote islands of Taiwan. For instance, pipe blockage has stemmed from scaling (
Figure 2a, calcium carbonate analyzed by X-ray diffraction (XRD)).
Figure 2b shows pitting near the weld on a storage tank that is made from 304 stainless steel. The maximum diameter of pitting hole was larger than 1 mm. It has recently been reported [
15] that aragonite, which is a crystalline phase of the calcium carbonate scales, can be formed at a temperature in the range of 50–55 °C and causes severe pitting corrosion. The XRD results indeed show the existence of aragonite in the scales, shown in
Figure 2a. This may be one of the causes of the pitting corrosion problem.
Figure 2c shows the leaks (due to corrosion) and its subsequent scaling on the absorption plate (made from 304 stainless steel). The pitting problem shown in
Figure 2b was attributed to poor welding in the manufacturing processes in addition to the water environments of high chloride concentration and high temperature (in summer), which aggravates the corrosion rate of the welding materials.
3. Assessment Methods—Water Quality Index
Data on the quality of tap water and ground water were obtained from the households with SWHs installed in 2013 and 2014. LSI, RSI, PSI, and LSCI were employed to assess the tendency of scaling or corrosion in SWHs. All chemical analyses required in the present study of water quality were made by the Sustainable Environment Research Center at NCKU, which is a certified unit by Environmental Analysis Laboratory, Environmental Protection Agency (EPA) of Taiwan.
3.1. LSI Assessment Method
Langelier [
5] proposed a procedure for calculating the degree of saturation of water with respect to calcium carbonate, known as the Langelier Saturation Index (LSI). Prisyazhniuk [
12] pointed out that the LSI makes it possible to estimate the ability of water to corrode steel, or the tendency of water to form scaling. Interpretation of the LSI given by the Carrier Air Conditioning Company [
8] is compiled in
Table 1. In the “Handbook of Drinking Water Quality” [
16], the LSI is defined by
where pH is measured in a solution of hydrogen ion activity; pH
S is a saturated hydrogen ion activity in either a calcite or a calcium carbonate solution and is determined by
where
A = (log10 (TDS) − 1)/10; TDS: total dissolved solids, mg/L;
B = −13.12 log10 (T + 273) + 34.55; T: temperature, °C;
C = log10 (Hardness) − 0.4; Hardness, mg/L;
D = log10 (Alkalinity); Alkalinity concentration, mg/L.
3.2. RSI Assessment Method
The Ryznar Stability Index (RSI) was proposed by Ryznar [
6]. The RSI index, defined in Equation (3), is based on the calcium carbonate saturation index in water and can estimate the ability of scaling or corrosion. Interpretation of the RSI is summarized in
Table 2 [
8].
3.3. PSI Assessment Method
The Puckorius Scaling Index (PSI) was proposed by Puckorius [
9]. The PSI is a modification of the RSI but with a similar interpretation for the RSI, as indicated in
Table 2. When estimating the scaling or corrosion tendency of the solution, the PSI addresses the buffering capacity in water and the maximum quantity of precipitates that is formed at equilibrium state. Therefore, the equilibrium pH (pH
eq) is adopted, instead of pH, in the definition of the PSI as
where pH
eq is calculated by
The scaling tendency becomes more significant in the range of small PSI values (<6.0) than RSI, whereas only corrosion tendency is observed for PSI > 6.0. Therefore, the PSI value is considered to be more accurate, as compared to both the LSI and RSI values, in the assessment of water quality.
3.4. LSCI Assessment Method
It was reported that stainless steel, copper, and aluminum are subject to pitting corrosion under the environment of water that contains chloride ions [
17,
18,
19,
20,
21]. Nevertheless, the LSI, the RSI, and the PSI do not consider the effects of chloride ions in their assessment of water quality. The Larson–Skold Corrosive Index (LSCI) was originally proposed to assess the corrosivity of water on low-carbon steel [
7]. This index is defined as the ratio of equivalent parts per million (ppm) of sulfate (SO
42−) and chloride (Cl
−) to the ppm of alkalinity in the form of bicarbonate plus carbonate (HCO
3− + CO
32−):
where C is the concentration in mg/L (ppm).
Note that the LSCI does not consider temperature and pH buffering capacity as the factors affecting corrosion behavior. Interpretation of the LSCI on corrosion tendency of steel is summarized as follows [
12]:
LSCI < 0.8—chlorides and sulfates do not influence the natural formation of the protective film on steel surface;
0.8 < LSCI < 1.2—slight tendency of steel corrosion can be observed;
LSCI > 1.2—significant localized (pitting) corrosion of steel can be observed.
4. Results and Discussion
The water resources analyzed in the study were sampled from the SWH user sites in the three investigated counties constituted of remote islands in Taiwan. It was well agreed [
10] that scale formation/corrosion becomes more significant as the water temperature is elevated. In the case of a SWH, the temperature of 80 °C, which can be frequently reached in the summer of Taiwan, was used in the estimation of the B term in Equation (2).
As a contrast, the assessment of scaling/corrosion tendency of the tap water in the main (Taiwan) island (see
Figure 1) was firstly performed using the available data of water quality reported in between November 2013 and October 2014 [
22]. The calculated values of the LSI, the RSI, and the PSI are 0.72 ± 0.58, 6.15 ± 0.88, and 6.31 ± 0.93, respectively. Based on the judgments listed in
Table 1 and
Table 2, the tap water quality on the main island of Taiwan is subject to slight scaling or corrosive tendency. Since the penetration of tap water on the main island is as high as 91.6% [
4], the scaling and corrosion issues in the SWHs installed on the main island are not serious problems. The chloride ion concentrations of tap water on the main island fall within 8.25 ± 0.64 mg/L, which are far smaller than those measured in either tap water or ground water of the three investigated remote-island counties (cf.
Table 3,
Table 4,
Table 5,
Table 6,
Table 7 and
Table 8). The corresponding LSCI values are 0.58 ± 0.25. Following the interpretation of the LSCI on steel corrosion tendency, the steel corrosion issue due to the chloride ions in tap water can be overlooked in the SWHs installed on the main island of Taiwan.
Due to the prevention of salt damage, which is a serious problem encountered in all the remote islands, almost all of the SWHs installed in the three investigated remote-island counties were made from 300-series stainless steels. Among them, most are made from 304 stainless steel, and few from 316 stainless steel. Thus, the following discussion will be focused on the corrosivity of these two 300-series stainless steels that are due to the chloride ions in the water.
4.1. Penghu
The chemical analysis results and the calculated water quality indices for the tap water and ground water in Penghu are summarized in
Table 3 and
Table 4, respectively. As compared to the water quality indices of the tap water in the main (Taiwan) island, the values of the LSI, the RSI, and the PSI measured in Penghu (
Table 3) do not differ much from those measured on the main island. However, the LSCI values for Penghu (
Table 3) are remarkably higher than the values (0.58 ± 0.25) on the main island, which indicates a high corrosion tendency to stainless steel based on the interpretation of the LSCI. Note that the water source for tap water on the main island comes exclusively from rainfall. In contrast, the amounts of rainfall in all the remote islands of these three counties cannot generally afford to be self-sufficient supply for the demand of tap water. As a result, the tap water sources in these remote-island counties are made up from rainfall, ground water, desalinated water, or any combination of these. This leads to the situation of remarkably higher chloride ion concentrations, in comparison with 8.25 ± 6.40 mg/L of chloride ion concentrations on the main island, as shown in
Table 3. The chloride ion concentrations of the ground water are higher than those for the tap water, except for Chimei (cf.
Table 3 and
Table 4). This could be attributed to the fact that the tap-water source in Chimei relies more on the desalinated water than the ground water.
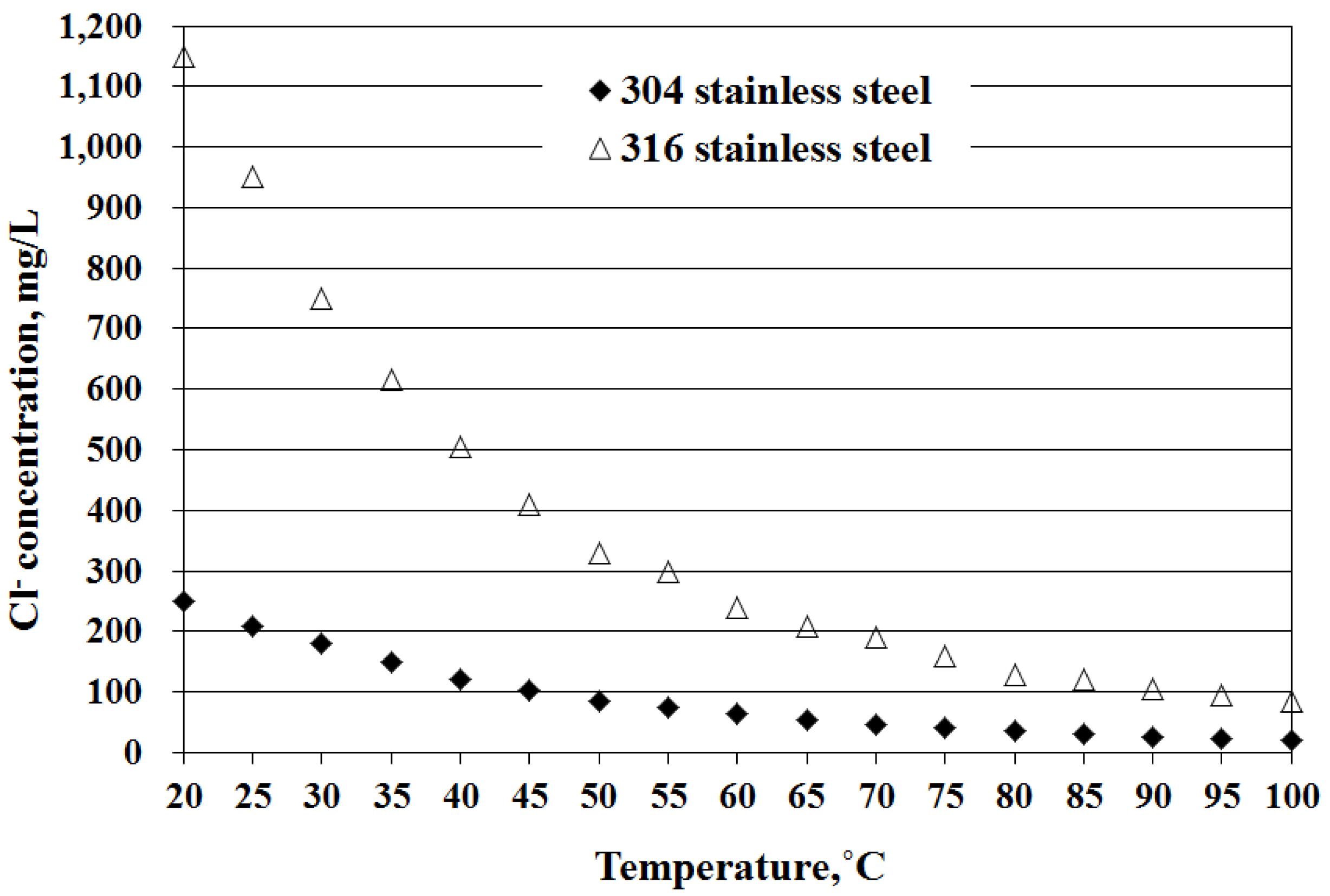
Both values of the LSCI for the tap water and ground water indicate a high corrosion tendency to stainless steel. Moreover, with such high concentrations of chloride ions in both tap and ground water, the pitting corrosion will easily occur, particularly in the SWHs made from 304 stainless steel according to the study of Huang and Huang [
23] (
Figure 3). In comparison with 304 stainless steel, 316 stainless steel of the passivation film on the surface has a higher corrosion resistance to chloride ions compared with 304 stainless steel in the measured range of chloride ion concentrations in Penghu.
Judging from the calculated values of the LSI, the RSI, and the PSI, the use of tap water in the SWHs leads to a slight scaling tendency, while the use of ground water in the SWHs leads to a relatively higher scaling tendency in Penghu, except for Siyu, in which its LSI, RSI, and PSI values for tap water indicate corrosion tendency.
4.2. Kinmen
Table 5 and
Table 6 summarize the chemical analysis results and the calculated water quality indices for the tap water and ground water, respectively, in Kinmen. Except for the tap water of Lieyu, in which the tap-water reservoir is exclusively supplied by rainfall, the chloride ion concentrations of the tap water are remarkable higher than those on the main island, while both chloride ion concentrations of the tap and ground water are relatively lower than those in Penghu. The calculated values of LSCI for the tap water (except for Lieyu) and ground water reveal significant corrosion tendency to stainless steel. Furthermore, in consideration of the working temperatures of SWHs (15–80 °C) and the chloride ion concentrations (
Table 5 and
Table 6) in Kinmen, 316 stainless steel is a better choice than 304 stainless steel in the manufacture of SWHs for the prevention of pitting corrosion at elevated water temperatures in SWHs.
Judging from the calculated values of the LSI, the RSI, and the PSI, there is a slight to serious corrosive tendency for SWHs using either tap water or ground water.
4.3. Lienchiang
Table 7 and
Table 8 summarize the chemical analysis results and the calculated water quality indices for the tap water and ground water, respectively, in Lienchiang. Similar to the cases of the other two remote-island counties, i.e., Penghu and Kinmen, the chloride ion concentrations of the tap water are remarkably higher than those on the main island, while both chloride ion concentrations of the tap and ground water are lower than those in Penghu. The calculated values of the LSCI for both tap and ground water reveal significant corrosion tendency to stainless steel. With such a high concentration of chloride ions, the same conclusion made for the SWHs installed in Penghu and Kinmen can also be drawn for the case in Lienchiang: 316 stainless steel is a better choice than 304 stainless steel in the manufacture of SWH for the prevention of pitting corrosion at elevated water temperatures in SWHs.
Judging from the calculated values of the LSI, the RSI, and the PSI, there is a slight to serious corrosive tendency, except for Dongju Island, in which the data of these three indices show a slight scaling tendency for SWHs using either tap water or ground water.
4.4. Comparison among Three Remote-Island Counties
Comparison of the LSCI and the chloride ion concentration for both ground and tap water among the three investigated counties is summarized in
Table 9. Excluding the tap water quality in Lieyu where the tap water is exclusively supplied by rainfall, and the ranges of chloride ion concentration and the LSCI fall within 45–104 mg/L and 1.7–2.8, respectively, in the other townships/cities of Kinmen. As revealed from
Table 9a, the ground water in Penghu has the highest concentrations of chloride ions, while the next highest level is Lienchiang. As mentioned previously, the tap-water sources in almost all townships/cities (except for Lieyu) of these three counties rely heavily on ground water, desalinated water, or both. As a result, the level of the chloride ion concentration for tap water in these three counties is close to that for ground water. However, all the LSCI data (except for the tap water of Lieyu) show a significant tendency of pitting corrosion no matter what the value of the chloride ion concentration is. This is because the LSCI is defined by the ratio of the sum of chloride ion concentration plus the sulfate concentration to the alkalinity concentration, i.e., Equation (6). However, the LSCI does not consider the temperature effect in its definition. Based on the study of Huang and Huang [
23], the concentration limits of 304 and 316 stainless steel corroded by chloride ions in the water temperature range of 20–100 °C drop as water temperature increases (
Figure 3). Due to the cost concern, most of the SWHs installed in these three remote-island counties were made from 304 stainless steel. In view of the measured chloride ion concentrations and the water temperatures in SWHs (80 °C in the sunny days of summer), pitting corrosion on 304 stainless steel definitely occurs. In contrast, 316 stainless steel can provide higher corrosion resistance to chloride ions but cannot completely prevent pitting corrosion. It is noted that both 304 and 316 steels are in the family of austenitic stainless steel, which easily suffers from “sensitization” from welding heat. Sensitization is one of the causes of the pitting corrosion near the weld shown in
Figure 2b. In fact, a small portion of SWHs installed in Taiwan is made from 444 steel, which is of the family of ferritic stainless steels that possess a higher resistance to pitting corrosion. It has also been reported [
24] that some ferritic stainless steels (444, 445, 446, and 447) have been used for the construction located in highly corrosive (caused mainly by chloride ions) coastal and offshore environments. Therefore, this series of ferritic stainless steels could be another good candidate in the manufacture of SWHs to be installed on the three remote-island counties studied herein.
5. Conclusions
An investigation of water quality in the three remote-island counties was conducted to understand the scaling or corrosion tendencies in SWHs. Three water quality indices including the LSI, the RSI, and the PSI were employed to assess the level of scaling or corrosion tendencies for both tap water and ground water. In addition, the method of the LSCI was also employed in the water quality assessment to understand the corrosion tendency on stainless steel, which is the primary material in the manufacture of SWHs installed in the remote islands of Taiwan. In Penghu, the values of the LSI, the RSI, and the PSI reveal a slight scaling tendency for both tap and ground water, except for the tap water of Siyu, which shows corrosion tendency. In Kinmen and Lienchiang, these three indices reveal a slight to serious corrosion tendency for both tap and ground water, except for Dongju Island, which shows a scaling tendency. As revealed from the high values of the LSCI in this study, the existence of a heavy corrosion tendency on stainless steel is indeed a critical issue to disseminate throughout the remote islands of Taiwan. In consideration of the high chloride ion concentrations and the high water temperatures (up to 80 °C) in SWHs, 304 stainless steel, commonly used in the manufacture of SWHs in Taiwan, cannot prevent the occurrence of pitting corrosion. Materials with a high corrosion resistance to chloride ions, such as 316 stainless steel, are preferable in the manufacture of SWHs installed in these three remote-island counties for the extension of the in-use lifetime of SWHs.