Near Real-Time Flow Cytometry Monitoring of Bacterial and Viral Removal Efficiencies during Water Reclamation Processes
Abstract
:1. Introduction
2. Materials and Methods
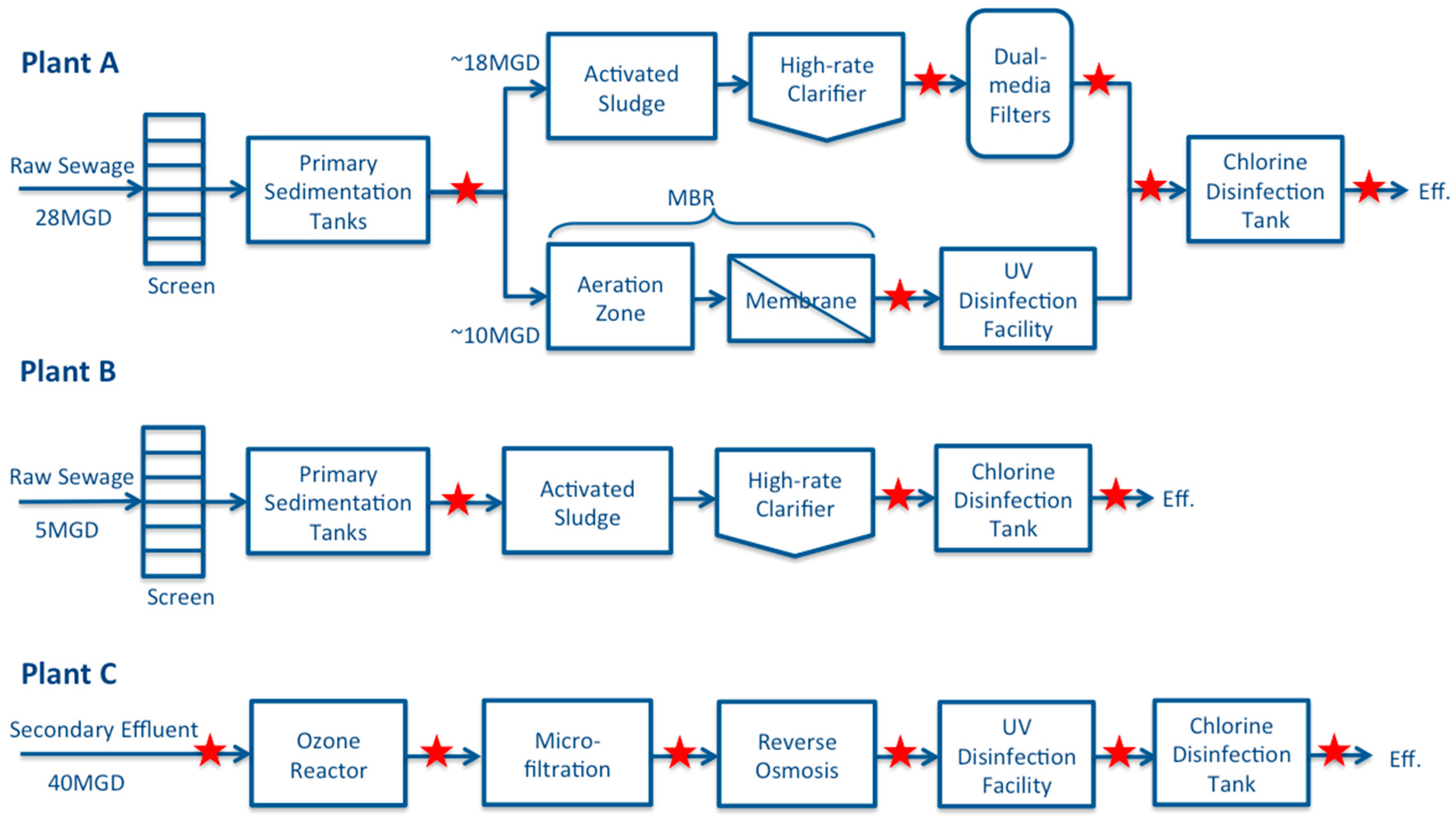
2.1. Study Sites and Sampling Scheme
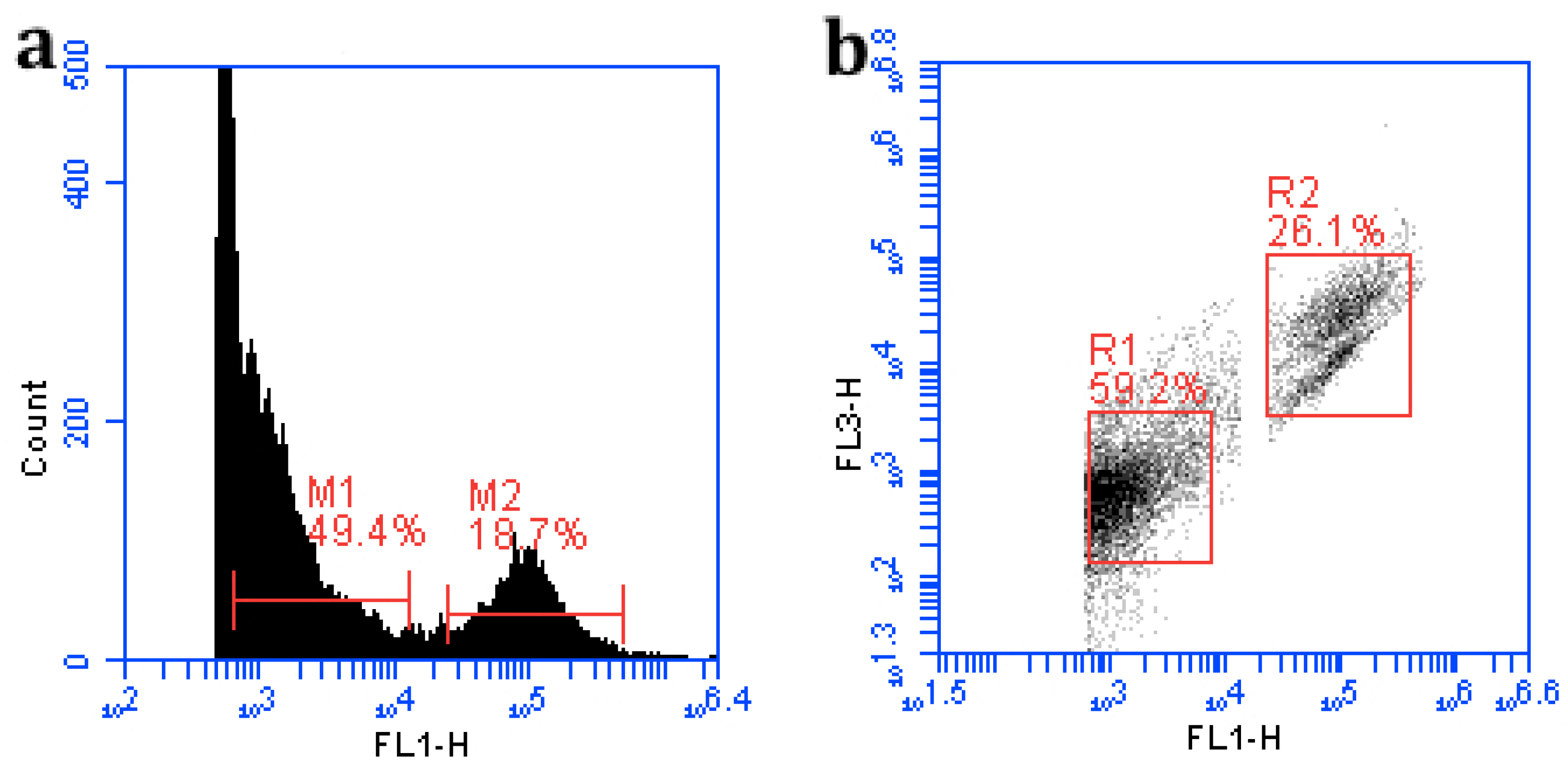
2.2. Quantification of Total Bacteria and Viruses
2.3. Bacterial and Viral Removal Efficiency
3. Results and Discussion
3.1. Quantification of Both Bacteria and Viruses by FCM
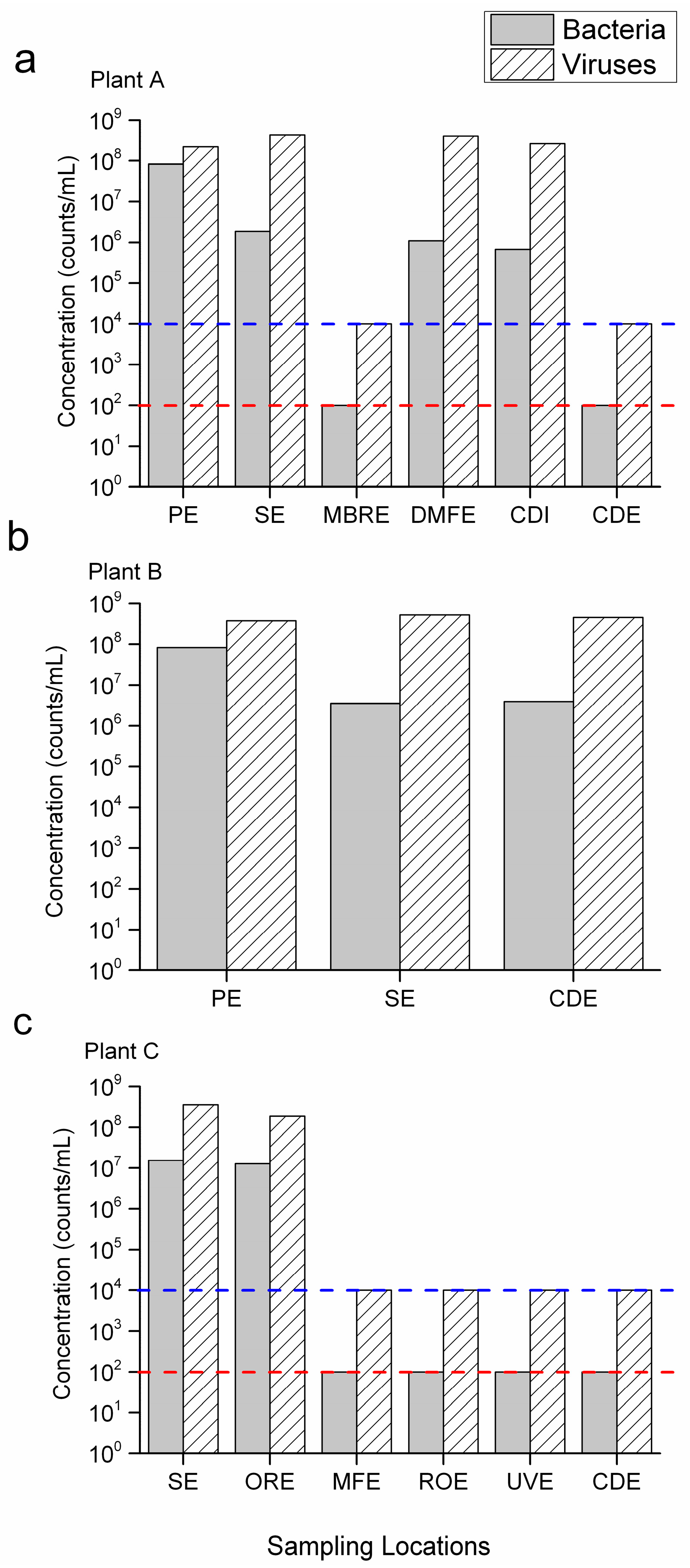
3.2. Removal through AS, High Rate Clarification and Dual Medium Filtration
3.3. Removal through MBR, MF and RO
3.4. Removal through Ozonation and Chlorination
3.5. Inadequacy of Current Microbial Water Reuse Criteria
4. Conclusions
- The traditional secondary AS process with a high rate of clarification removed 1–2 log10 of bacteria but was not effective at viral removal.
- The MBR achieved similar bacterial (5 log10) and viral (4 log10) removal rates in comparison with the standalone MF process.
- For disinfection, both chloramine and free chlorine are equally effective in reducing total coliform bacteria to meet the water reuse criteria. High concentrations of bacterial and viral particles were still present in the final effluent after chloramine disinfection, while both organisms were below the LLOD of FCM in the final effluent of the plant using free chlorine as its main disinfectant.
- Current water reuse criteria, using total coliform bacteria as the sole indicator of microbial quality, may underestimate the potential health risk under certain conditions. FCM targeting indigenous total bacteria and viruses shows potential as a rapid monitoring tool for the evaluation of microbial removal.
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Natural Resources Defense Council (NRDC). Water Reuse Potential in California. 2014. Available online: http://pacinst.org/app/uploads/2014/06/ca-water-reuse.pdf (accessed on 13 October 2016).
- Drewes, J.E.; Horstmeyer, N. Recent Developments in potable water reuse. In Advanced Treatment Technologies for Urban Wastewater Reuse; Springer: Berlin, Germany, 2015; pp. 269–290. [Google Scholar]
- Harris-Lovett, S.R.; Binz, C.; Sedlak, D.L.; Kiparsky, M.; Truffer, B. Beyond user acceptance: A legitimacy framework for potable water reuse in California. Environ. Sci. Technol. 2015, 49, 7552–7561. [Google Scholar] [CrossRef] [PubMed]
- Olivieri, A.W.; Seto, E.; Cooper, R.C.; Cahn, M.D.; Colford, J.; Crook, J.; Debroux, J.-F.; Mandrell, R.; Suslow, T.; Tchobanoglous, G. Risk-Based Review of California’s Water-Recycling Criteria for Agricultural Irrigation. J. Environ. Eng. 2014, 140. [Google Scholar] [CrossRef]
- Jiang, S.C.; Lim, K.Y.; Huang, X.; McCarthy, D.; Hamilton, A.J. Human and environmental health risks and benefits associated with use of urban stormwater. Wiley Interdiscip. Rev. Water 2015, 2, 683–699. [Google Scholar] [CrossRef]
- Hall, A.J.; Lopman, B.A.; Payne, D.C.; Patel, M.M.; Gastañaduy, P.A.; Vinjé, J.; Parashar, U.D. Norovirus disease in the United States. Emerg. Infect. Dis. 2013, 19, 1198–1205. [Google Scholar] [CrossRef] [PubMed]
- Scott, T.; McLaughlin, M.; Harwood, V.; Chivukula, V.; Levine, A.; Gennaccaro, A.; Lukasik, J.; Farrah, S.; Rose, J. Reduction of pathogens, indicator bacteria, and alternative indicators by wastewater treatment and reclamation processes. Water Sci. Technol. Water Supply 2003, 3, 247–252. [Google Scholar]
- Zhang, C.-M.; Xu, L.-M.; Xu, P.-C.; Wang, X.C. Elimination of viruses from domestic wastewater: Requirements and technologies. World J. Microbiol. Biotechnol. 2016, 32, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Tree, J.A.; Adams, M.R.; Lees, D.N. Chlorination of indicator bacteria and viruses in primary sewage effluent. Appl. Environ. Microbiol. 2003, 69, 2038–2043. [Google Scholar] [CrossRef] [PubMed]
- Stewart, E.J. Growing unculturable bacteria. J. Bacteriol. 2012, 194, 4151–4160. [Google Scholar] [CrossRef] [PubMed]
- Research Method 366.1 Determining the Total Cell Count and Ratios of High and Low Nucleic Acid Content Cells in Freshwater Using Flow Cytometry. 2012. Available online: http://www.isme-microbes.org/sites/isme-microbes.org/files/2012_SLMB_366%201%20ME%20EN%20Final.pdf (accessed on 13 October 2016).
- Drinking Water Unexpectedly Rich in Microbial Life. 2013. Available online: https://www.admin.ch/gov/en/start/documentation/media-releases.msg-id-47549.html (accessed on 13 October 2016).
- Ramseier, M.K.; von Gunten, U.; Freihofer, P.; Hammes, F. Kinetics of membrane damage to high (HNA) and low (LNA) nucleic acid bacterial clusters in drinking water by ozone, chlorine, chlorine dioxide, monochloramine, ferrate(VI), and permanganate. Water Res. 2011, 45, 1490–1500. [Google Scholar] [CrossRef] [PubMed]
- Huang, X.; Min, J.H.; Lu, W.; Jaktar, K.; Yu, C.; Jiang, S.C. Evaluation of methods for reverse osmosis membrane integrity monitoring for wastewater reuse. J. Water Proc. Eng. 2015, 7, 161–168. [Google Scholar] [CrossRef]
- Huang, X.; Qu, Y.; Cid, C.A.; Finke, C.; Hoffmann, M.R.; Lim, K.; Jiang, S.C. Electrochemical disinfection of toilet wastewater using wastewater electrolysis cell. Water Res. 2016, 92, 164–172. [Google Scholar] [CrossRef] [PubMed]
- Tamaki, H.; Zhang, R.; Angly, F.E.; Nakamura, S.; Hong, P.Y.; Yasunaga, T.; Kamagata, Y.; Liu, W.T. Metagenomic analysis of DNA viruses in a wastewater treatment plant in tropical climate. Environ. Microbiol. 2012, 14, 441–452. [Google Scholar] [CrossRef] [PubMed]
- Wu, Q.; Liu, W.-T. Determination of virus abundance, diversity and distribution in a municipal wastewater treatment plant. Water Res. 2009, 43, 1101–1109. [Google Scholar] [CrossRef] [PubMed]
- Shin, G.-A.; Sobsey, M.D. Removal of norovirus from water by coagulation, flocculation and sedimentation processes. Water Sci. Technol. Water Supply 2015, 15, 158–163. [Google Scholar] [CrossRef]
- Shirasaki, N.; Matsushita, T.; Matsui, Y.; Marubayashi, T.; Murai, K. Investigation of enteric adenovirus and poliovirus removal by coagulation processes and suitability of bacteriophages MS2 and φX174 as surrogates for those viruses. Sci. Total Environ. 2016, 563, 29–39. [Google Scholar] [CrossRef] [PubMed]
- Rosario, K.; Nilsson, C.; Lim, Y.W.; Ruan, Y.; Breitbart, M. Metagenomic analysis of viruses in reclaimed water. Environ. Microbiol. 2009, 11, 2806–2820. [Google Scholar] [CrossRef] [PubMed]
- Nieuwstad, T.J.; Mulder, E.; Havelaar, A.; Van Olphen, M. Elimination of micro-organisms from wastewater by tertiary precipitation and simultaneous precipitation followed by filtration. Water Res. 1988, 22, 1389–1397. [Google Scholar] [CrossRef]
- Marti, E.; Monclús, H.; Jofre, J.; Rodriguez-Roda, I.; Comas, J.; Balcázar, J.L. Removal of microbial indicators from municipal wastewater by a membrane bioreactor (MBR). Bioresour. Technol. 2011, 102, 5004–5009. [Google Scholar] [CrossRef] [PubMed]
- Hirani, Z.M.; DeCarolis, J.F.; Adham, S.S.; Jacangelo, J.G. Peak flux performance and microbial removal by selected membrane bioreactor systems. Water Res. 2010, 44, 2431–2440. [Google Scholar] [CrossRef] [PubMed]
- Branch, A.; Trinh, T.; Carvajal, G.; Leslie, G.; Coleman, H.M.; Stuetz, R.M.; Drewes, J.E.; Khan, S.J.; Le-Clech, P. Hazardous events in membrane bioreactors—Part 3: Impacts on microorganism log removal efficiencies. J. Membr. Sci. 2016, 497, 514–523. [Google Scholar] [CrossRef]
- Miura, T.; Okabe, S.; Nakahara, Y.; Sano, D. Removal properties of human enteric viruses in a pilot-scale membrane bioreactor (MBR) process. Water Res. 2015, 75, 282–291. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Z. Personal communication, 15 January 2015.
- Tam, L.; Tang, T.; Lau, G.N.; Sharma, K.; Chen, G. A pilot study for wastewater reclamation and reuse with MBR/RO and MF/RO systems. Desalination 2007, 202, 106–113. [Google Scholar] [CrossRef]
- Pype, M.-L.; Lawrence, M.G.; Keller, J.; Gernjak, W. Reverse osmosis integrity monitoring in water reuse: The challenge to verify virus removal—A review. Water Res. 2016, 98, 384–395. [Google Scholar] [CrossRef] [PubMed]
- Xu, P.; Janex, M.-L.; Savoye, P.; Cockx, A.; Lazarova, V. Wastewater disinfection by ozone: Main parameters for process design. Water Res. 2002, 36, 1043–1055. [Google Scholar] [CrossRef]
- Hunt, N.K.; Mariñas, B.J. Kinetics of Escherichia coli inactivation with ozone. Water Res. 1997, 31, 1355–1362. [Google Scholar] [CrossRef]
- Gehr, R.; Wagner, M.; Veerasubramanian, P.; Payment, P. Disinfection efficiency of peracetic acid, UV and ozone after enhanced primary treatment of municipal wastewater. Water Res. 2003, 37, 4573–4586. [Google Scholar] [CrossRef]
- Rose, L.J.; Rice, E.W.; Hodges, L.; Peterson, A.; Arduino, M.J. Monochloramine inactivation of bacterial select agents. Appl. Environ. Microbiol. 2007, 73, 3437–3439. [Google Scholar] [CrossRef] [PubMed]
- Jacangelo, J.G.; Olivieri, V.P.; Kawata, K. Investigating the Mechanism of Inactivation of Escherichia-Coli-B by Monochloramine. J. Am. Water Works Assoc. 1991, 83, 80–87. [Google Scholar]
- Cho, M.; Kim, J.; Kim, J.Y.; Yoon, J.; Kim, J.-H. Mechanisms of Escherichia coli inactivation by several disinfectants. Water Res. 2010, 44, 3410–3418. [Google Scholar] [CrossRef] [PubMed]
- Cromeans, T.L.; Kahler, A.M.; Hill, V.R. Inactivation of Adenoviruses, Enteroviruses, and Murine Norovirus in Water by Free Chlorine and Monochloramine. Appl. Environ. Microbiol. 2010, 76, 1028–1033. [Google Scholar] [CrossRef] [PubMed]
| Plant A | AS | DMF | MBR | CD |
| Bacteria | 1.66 ± 0.4 | 0.25 ± 0.1 | 5.9 ± 0.3 | 3.7 ± 0.4 |
| Virus | −0.30 ± 0.16 | 0.09 ± 0.3 | 4.3 ± 0.2 | 4.3 ± 0.4 |
| Plant B | AS | CD | ||
| Bacteria | 1.33 ± 0.2 | −0.02 ± 0.2 | ||
| Virus | 0.12 ± 0.1 | 0.12 ± 0.2 | ||
| Plant C | Ozone | MF | RO | UV |
| Bacteria | 0.07 ± 0.1 | 5.10 ± 0.1 | N.A. * | N.A. * |
| Virus | 0.3 ± 0.1 | 4.25 ± 0.2 | N.A. * | N.A. * |
| NH3 (mg/L) | NO2− (mg/L) | NO3− (mg/L) | |
|---|---|---|---|
| Plant A | 0.39 ± 1.10 | 0.15 ± 0.10 | 10.90 ± 1.55 |
| Plant B | 38.88 ± 5.48 | 1.71 ± 0.99 | 1.85 ± 1.04 |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Huang, X.; Zhao, Z.; Hernandez, D.; Jiang, S.C. Near Real-Time Flow Cytometry Monitoring of Bacterial and Viral Removal Efficiencies during Water Reclamation Processes. Water 2016, 8, 464. https://doi.org/10.3390/w8100464
Huang X, Zhao Z, Hernandez D, Jiang SC. Near Real-Time Flow Cytometry Monitoring of Bacterial and Viral Removal Efficiencies during Water Reclamation Processes. Water. 2016; 8(10):464. https://doi.org/10.3390/w8100464
Chicago/Turabian StyleHuang, Xiao, Zheng Zhao, Dana Hernandez, and Sunny C. Jiang. 2016. "Near Real-Time Flow Cytometry Monitoring of Bacterial and Viral Removal Efficiencies during Water Reclamation Processes" Water 8, no. 10: 464. https://doi.org/10.3390/w8100464
APA StyleHuang, X., Zhao, Z., Hernandez, D., & Jiang, S. C. (2016). Near Real-Time Flow Cytometry Monitoring of Bacterial and Viral Removal Efficiencies during Water Reclamation Processes. Water, 8(10), 464. https://doi.org/10.3390/w8100464







