Effects of the “Run-of-River” Hydro Scheme on Macroinvertebrate Communities and Habitat Conditions in a Mountain River of Northeastern China
Abstract
:1. Introduction
2. Materials and Methods
2.1. Study Area
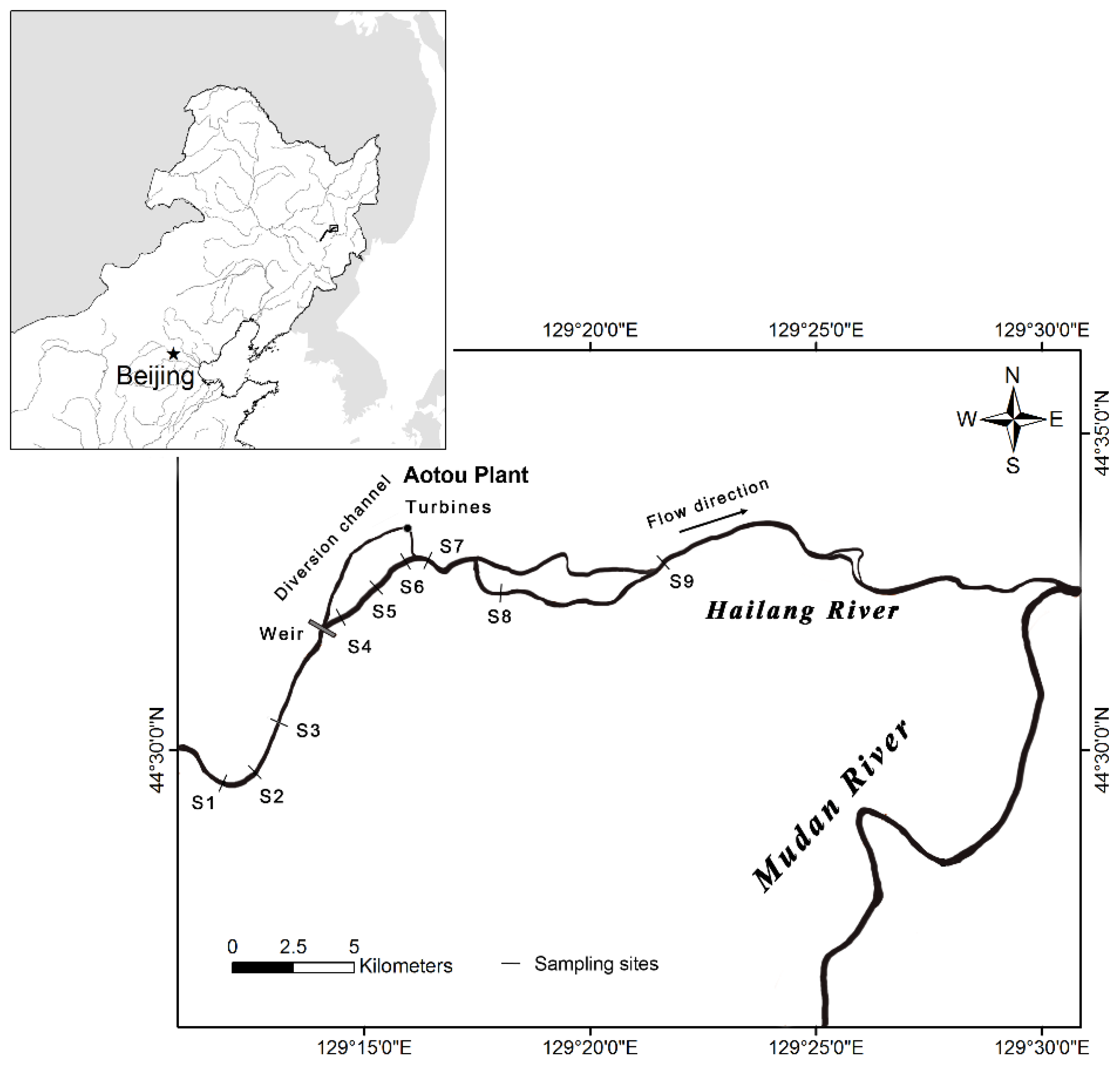
2.2. Sampling and Identification
2.3. Physical Habitat Assessment
2.4. Methods of Analysis
3. Results
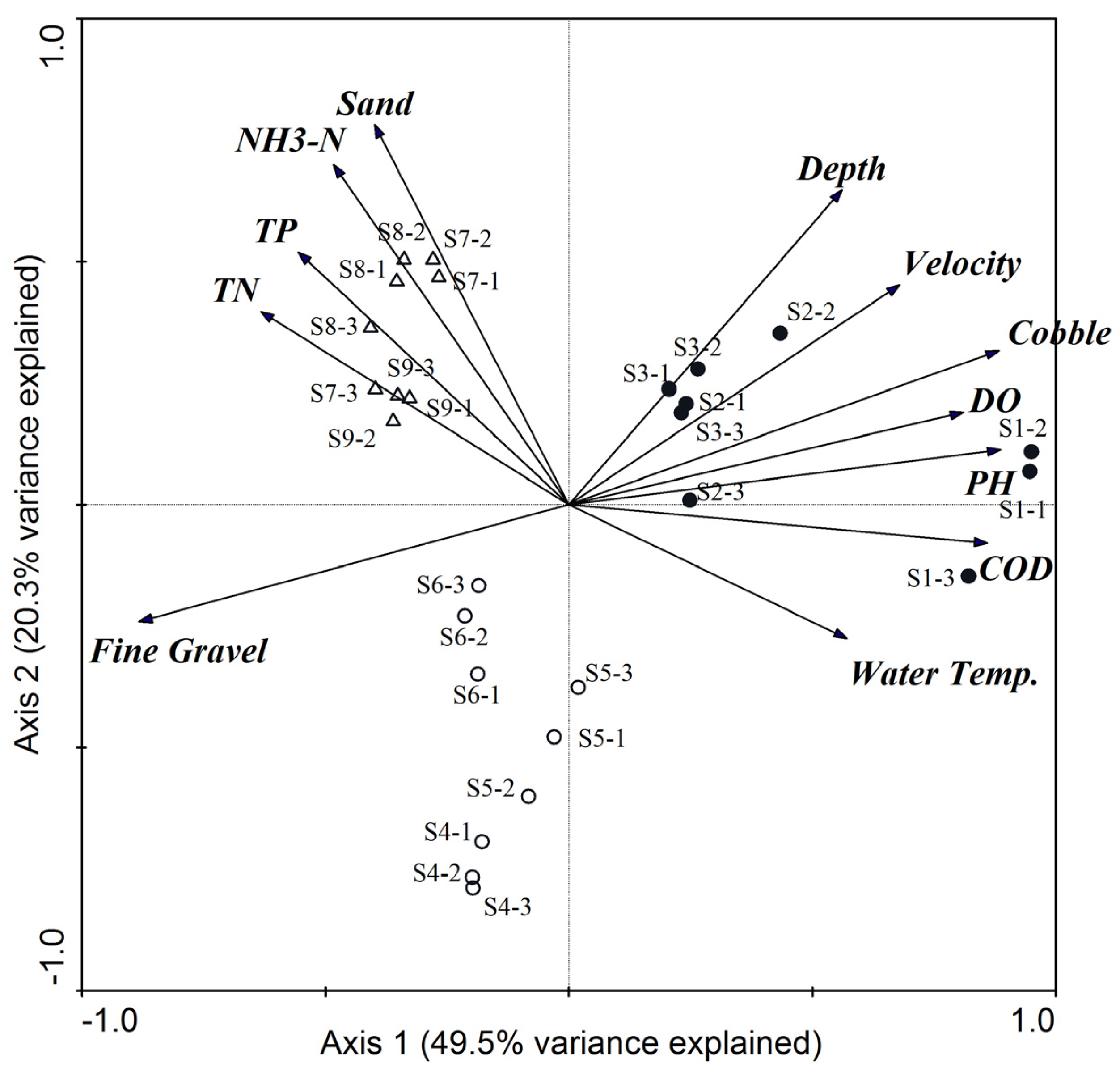
3.1. Physical and Chemical Variables
| Variables | Units | Upper Sites | Depleted Sites | Lower Sites | ||||||
|---|---|---|---|---|---|---|---|---|---|---|
| S1 | S2 | S3 | S4 | S5 | S6 | S7 | S8 | S9 | ||
| Depth | m | 0.34 (0.03) | 0.31 (0.06) | 0.24 (0.03) | 0.09 (0.02) | 0.17 (0.04) | 0.18 (0.03) | 0.28 (0.04) | 0.19 (0.04) | 0.23 (0.02) |
| Velocity | m/s | 0.56 (0.12) | 0.53 (0.11) | 0.56 (0.01) | 0.21 (0.01) | 0.24 (0.06) | 0.27 (0.02) | 0.34 (0.09) | 0.21 (0.01) | 0.37 (0.05) |
| DO | mg/L | 10.13 (0.01) | 10.38 (0.09) | 10.20 (0.08) | 9.22 (0.08) | 9.29 (0.01) | 9.57 (0.04) | 9.27 (0.01) | 9.19 (0.03) | 9.11 (0.02) |
| Temp | °C | 20.23 (0.03) | 20.80 (0.31) | 19.50 (0.20) | 19.97 (0.12) | 19.73 (0.03) | 20.07 (0.17) | 19.43 (0.03) | 19.37 (0.03) | 19.33 (0.03) |
| PH | − | 7.69 | 7.36 | 7.30 | 7.27 | 7.23 | 7.19 | 7.25 | 7.27 | 7.25 |
| CODMn | mg/L | 5.70 | 4.60 | 4.70 | 4.50 | 4.80 | 4.80 | 4.60 | 4.50 | 4.40 |
| COD | mg/L | 19.50 | 16.10 | 16.10 | 15.40 | 16.30 | 16.50 | 15.90 | 15.40 | 14.60 |
| BOD5 | mg/L | 2.00 | 2.00 | 2.00 | 2.00 | 2.00 | 2.00 | 2.00 | 2.00 | 2.00 |
| NH3-N | mg/L | 0.12 | 0.17 | 0.16 | 0.13 | 0.14 | 0.15 | 0.19 | 0.17 | 0.15 |
| TP | mg/L | 0.08 | 0.08 | 0.07 | 0.07 | 0.07 | 0.11 | 0.10 | 0.11 | 0.12 |
| TN | mg/L | 0.24 | 0.25 | 0.25 | 0.25 | 0.25 | 0.28 | 0.30 | 0.29 | 0.26 |
| CB Cobble | % | 60.00 | 37.00 | 42.40 | 22.89 | 32.92 | 28.32 | 30.73 | 34.61 | 27.92 |
| GC Gravel | % | 27.01 | 38.86 | 35.35 | 50.66 | 45.73 | 48.16 | 38.90 | 34.10 | 39.70 |
| GF Gravel | % | 7.80 | 15.69 | 13.23 | 21.94 | 17.20 | 19.90 | 20.90 | 16.20 | 20.70 |
| SA Sand | % | 4.00 | 8.10 | 9.02 | 4.20 | 3.70 | 3.10 | 9.10 | 14.50 | 11.10 |

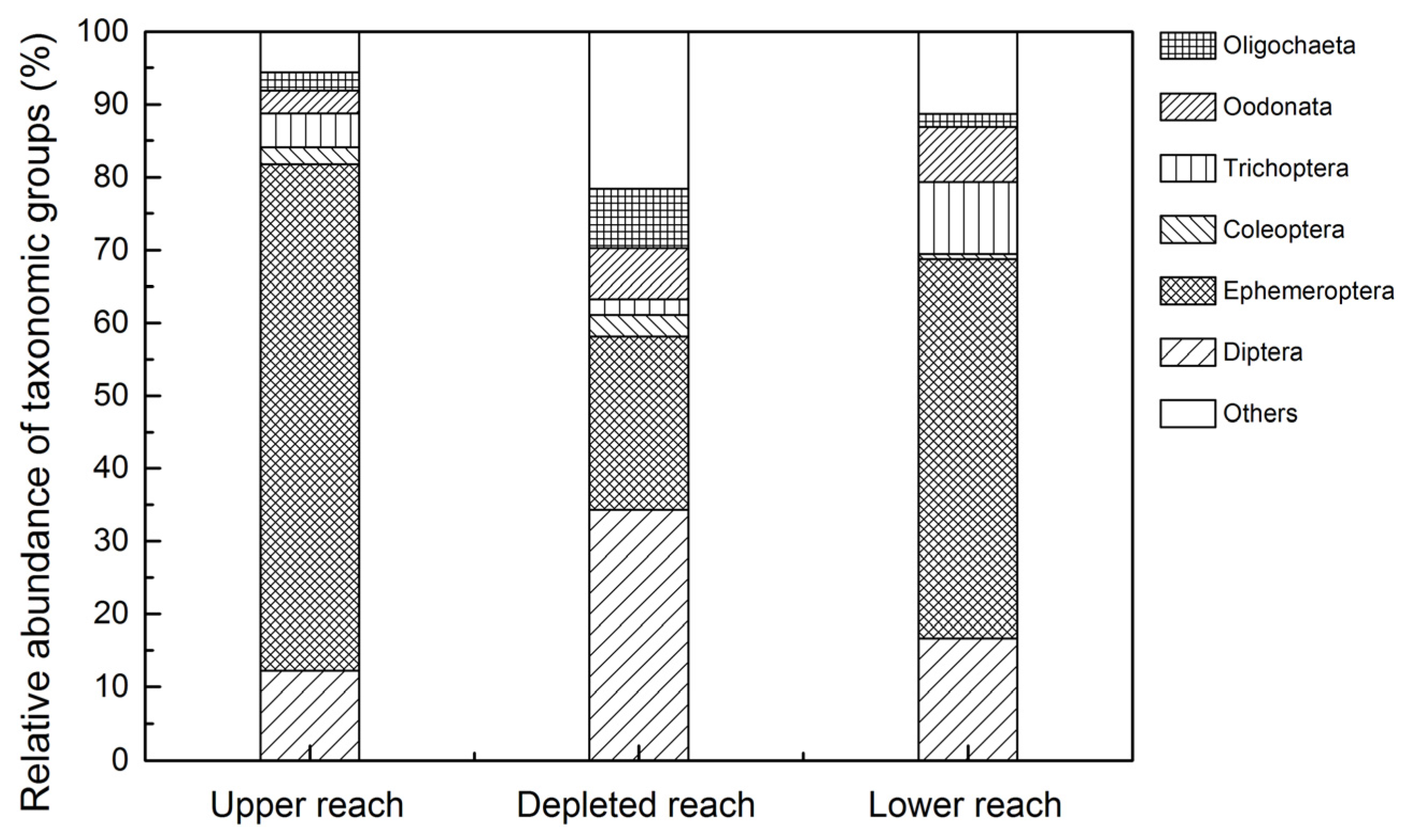
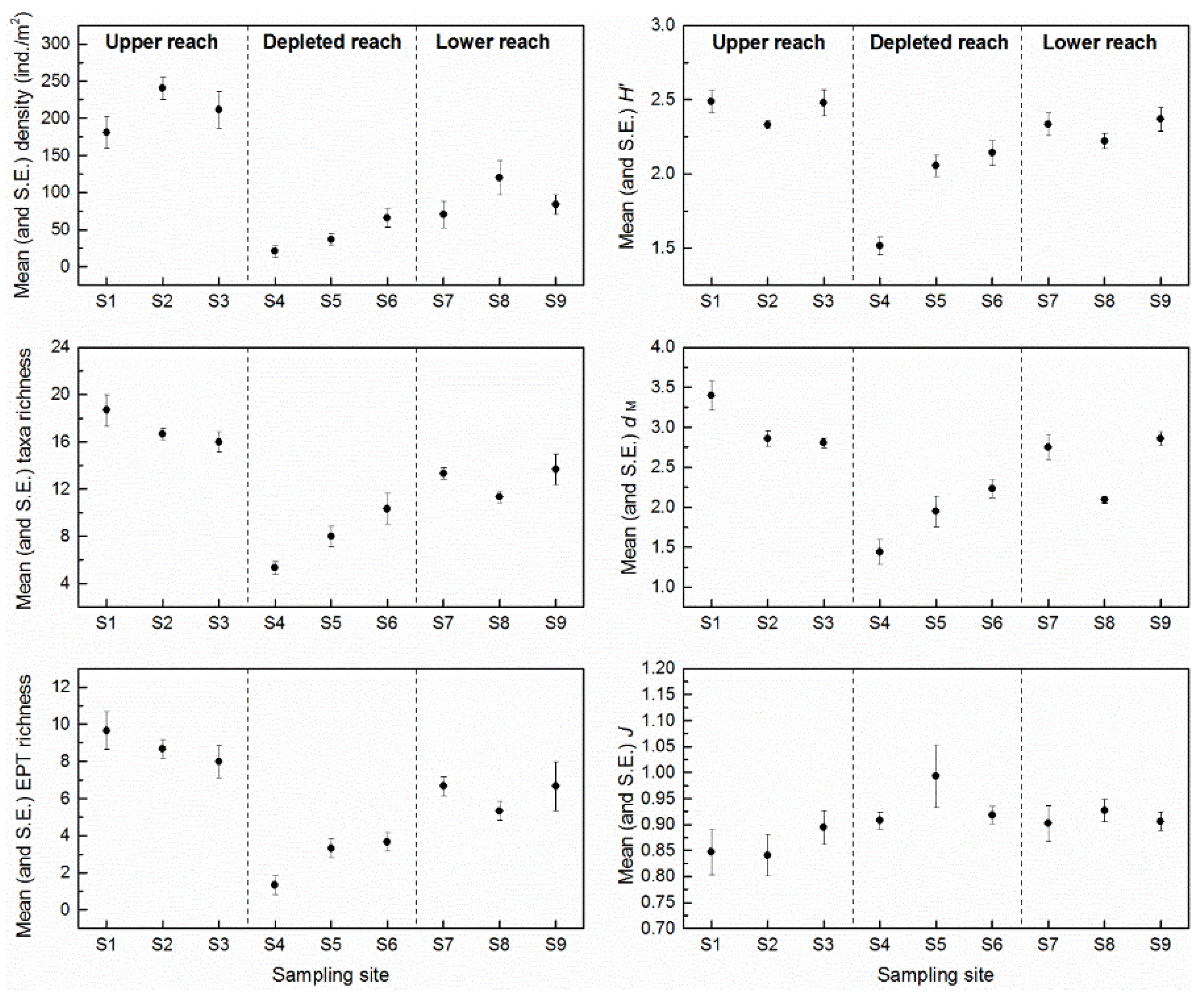
3.2. Assemblage Composition
| Class Genus/Species | Relative Abundances (%) | ||
|---|---|---|---|
| Upper Reach | Depleted Reach | Lower Reach | |
| Insecta | |||
| Polypedilum sordens | 0.79 | 6.49 | 2.19 |
| Cryptochironomus defectus | 3.95 | 14.32 | 4.50 |
| Chironomus plumosus | 5.89 | 9.19 | 7.30 |
| Pocladius choreus | 1.58 | 2.43 | 2.68 |
| Cinygma sp1 | 4.58 | − | − |
| Cinygma sp2 | 4.42 | − | − |
| Epeorus uenoi | 11.26 | 7.03 | 3.53 |
| Drunella sp1 | 7.53 | 0.00 | 5.60 |
| Drunella sp2 | 14.32 | 0.00 | 17.76 |
| Ephemerella sp | 14.47 | 3.24 | 8.52 |
| Ephemera sp | 10.53 | 7.84 | 10.22 |
| Baetis sp | 0.63 | 0.00 | 4.38 |
| Potamanthus huoshanensis | 1.84 | 5.68 | 2.07 |
| Elmidae | 2.16 | − | 0.73 |
| Dytiscidae | 0.11 | 2.97 | − |
| Ieptoceridae | 0.37 | − | − |
| Hydropsychidae | 4.32 | 2.16 | 9.85 |
| Gomphidae | 3.16 | 7.03 | 7.54 |
| Muscidae | − | 1.89 | − |
| Oligochaeta | |||
| Tubificidae | 2.47 | 8.11 | 1.82 |
| Clitellata | |||
| Glossiphonia sp | 1.42 | − | 2.43 |
| Whitmania sp | 1.95 | − | 0.61 |
| Gastropoda | |||
| Radix ovata | 1.00 | 5.41 | 2.92 |
| Oncomelania | 1.26 | 15.41 | 5.35 |
| Bivalvia | |||
| Corbicula | − | 0.81 | − |
3.3. FFG Variations
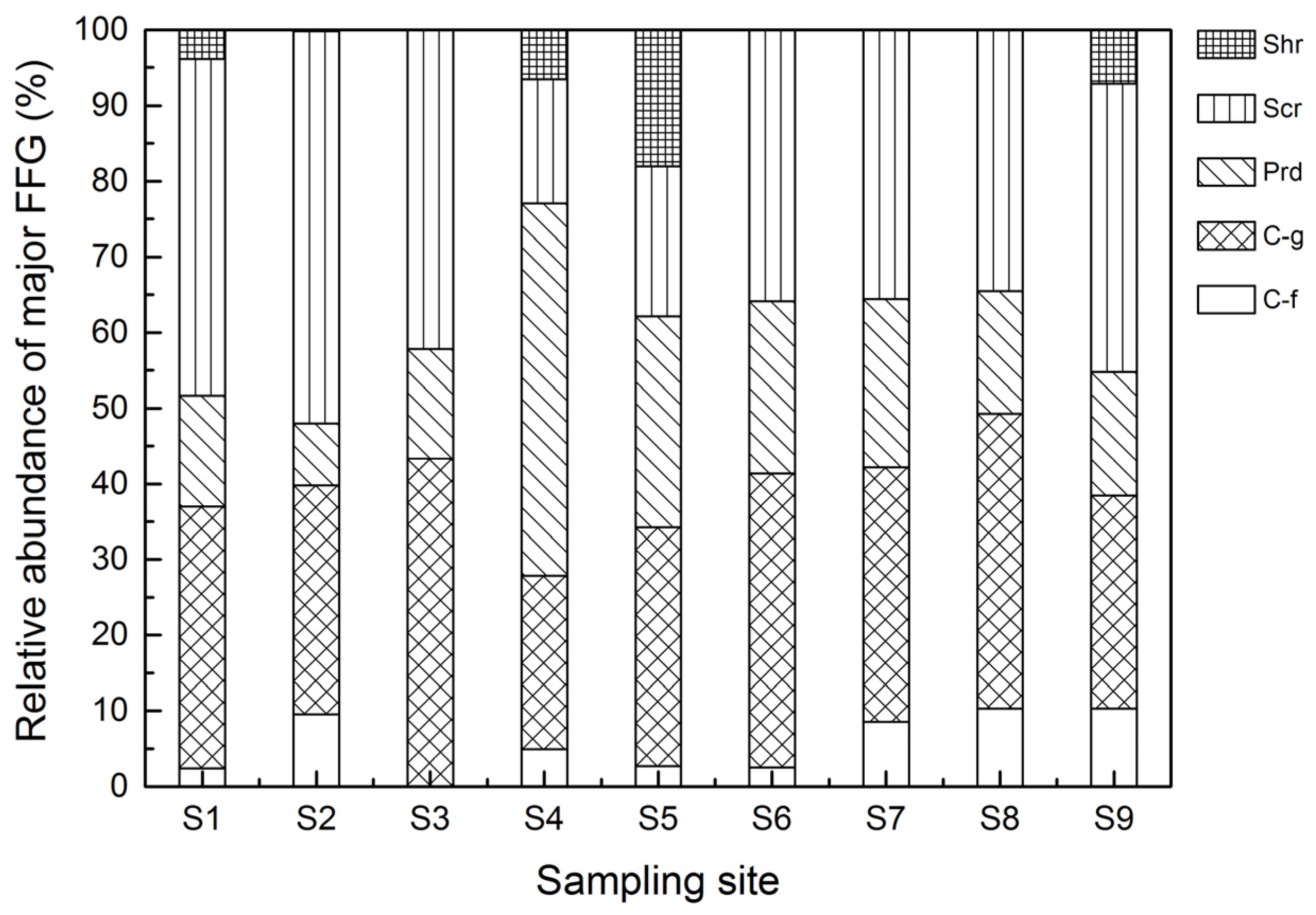
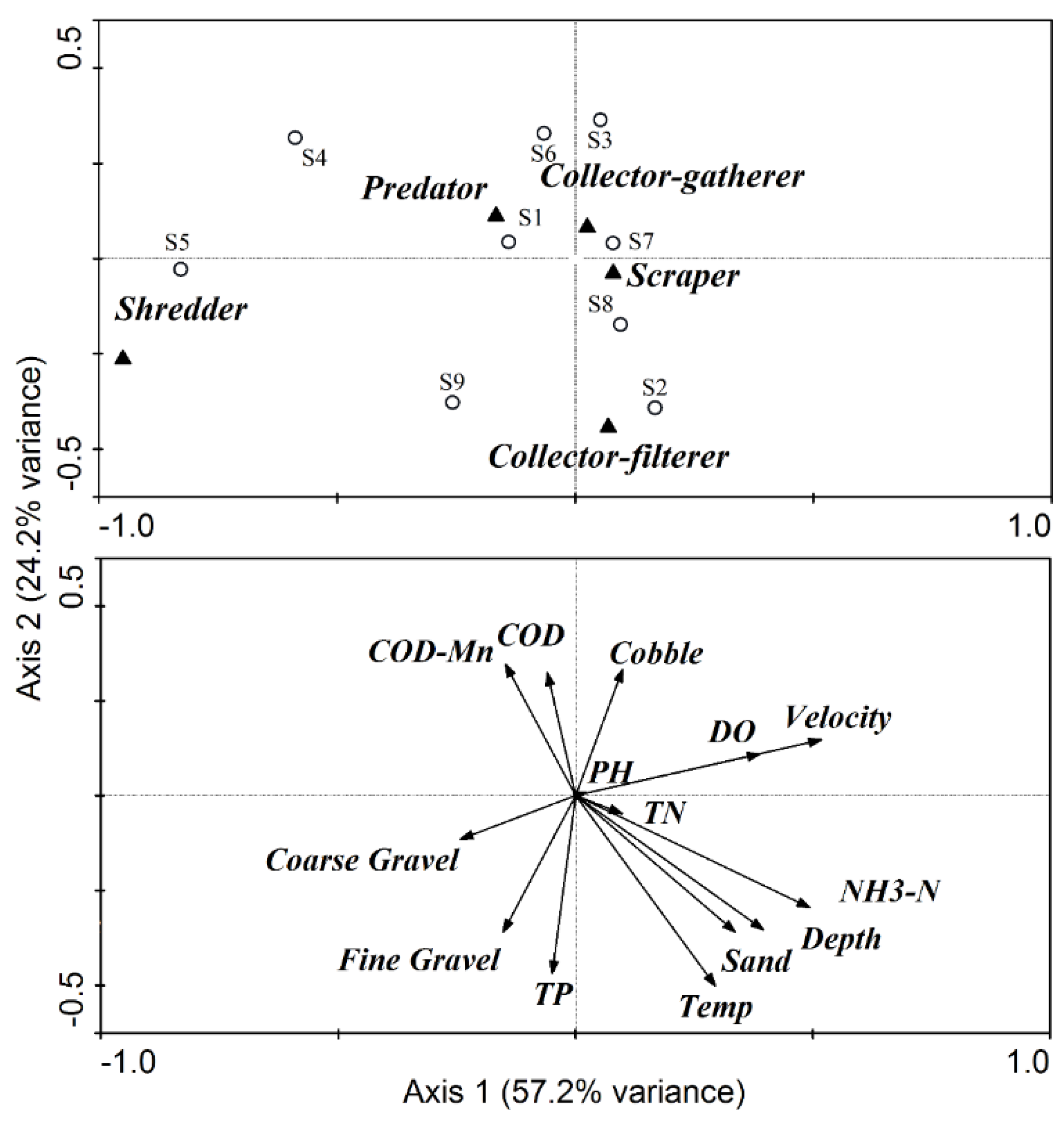
| Group | Positive Correlation Coefficients | Negative Correlation Coefficients |
|---|---|---|
| Scr | Velocity * (0.453) DO ** (0.783) Cobble * (0.408) | TN † (−0.321) Fine Gravel † (−0.346) |
| Shr | − | NH3-N * (−0.373) |
| Prd | Coarse Gravel * (0.377) | − |
| C-g | Velocity * (0.452) DO ** (0.646) Cobble * (0.434) | Fine Gravel * (−0.370) |
| C-f | Water Temp.** (0.770) NH3-N * (0.138) Sand ** (0.694) | − |
4. Discussion
4.1. The Effects of ROR Scheme on Spatial Variations in Habitat Conditions
4.2. Response of Macroinvertebrate Structure and Biodiversity to ROR Scheme
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Anderson, D.; Moggridge, H.; Warren, P.; Shucksmith, J. The impacts of “run-of-river” hydropower on the physical and ecological condition of rivers. Water Environ. J. 2015, 29, 268–276. [Google Scholar] [CrossRef]
- Humborg, C.; Ittekkot, V.; Cociasu, A.; Bodungen, B. Effect of danube river dam on black sea biogeochemistry and ecosystem structure. Nature 1997, 386, 385–388. [Google Scholar] [CrossRef]
- Nilsson, C.; Reidy, C.A.; Dynesius, M.; Revenga, C. Fragmentation and flow regulation of the world’s large river systems. Science 2005, 308, 405–408. [Google Scholar] [CrossRef] [PubMed]
- Stone, R. The legacy of the three gorges dam. Science 2011, 333, 817. [Google Scholar] [CrossRef] [PubMed]
- Parsley, M.J.; Beckman, L.G.; McCabe, G.T., Jr. Spawning and rearing habitat use by white sturgeons in the columbia river downstream from mcnary dam. Trans. Am. Fish. Soc. 1993, 122, 217–227. [Google Scholar] [CrossRef]
- Kanehl, P.D.; Lyons, J.; Nelson, J.E. Changes in the habitat and fish community of the milwaukee river, wisconsin, following removal of the woolen mills dam. N. Am. J. Fish. Manag. 1997, 17, 387–400. [Google Scholar] [CrossRef]
- Beasley, C.A.; Hightower, J.E. Effects of a low-head dam on the distribution and characteristics of spawning habitat used by striped bass and american shad. Trans. Am. Fish. Soc. 2000, 129, 1316–1330. [Google Scholar] [CrossRef]
- Zhang, G.; Wu, L.; Li, H.; Liu, M.; Cheng, F.; Murphy, B.R.; Xie, S. Preliminary evidence of delayed spawning and suppressed larval growth and condition of the major carps in the yangtze river below the three gorges dam. Environ. Biol. Fish. 2012, 93, 439–447. [Google Scholar] [CrossRef]
- Paish, O. Micro-hydropower: Status and prospects. Proc. Inst. Mech. Eng. Part A J. Power Energy 2002, 216, 31–40. [Google Scholar] [CrossRef]
- Dewson, Z.S.; James, A.B.; Death, R.G. Invertebrate community responses to experimentally reduced discharge in small streams of different water quality. J. N. Am. Benthol. Soc. 2007, 26, 754–766. [Google Scholar] [CrossRef]
- Finn, M.A.; Boulton, A.J.; Chessman, B.C. Ecological responses to artificial drought in two australian rivers with differing water extraction. Fundam. Appl. Limnol. 2009, 175, 231–248. [Google Scholar] [CrossRef]
- Resh, V.H.; Norris, R.H.; Barbour, M.T. Design and implementation of rapid assessment approaches for water resource monitoring using benthic macroinvertebrates. Aust. J. Ecol. 1995, 20, 108–121. [Google Scholar] [CrossRef]
- Duan, X.; Wang, Z.; Xu, M.; Zhang, K. Effect of streambed sediment on benthic ecology. Int. J. Sedim. Res. 2009, 24, 325–338. [Google Scholar] [CrossRef]
- Cummins, K.W.; Klug, M.J. Feeding ecology of stream invertebrates. Ann. Rev. Ecol. Syst. 1979, 147–172. [Google Scholar] [CrossRef]
- Cummins, K.W.; Merritt, R.W.; Andrade, P.C. The use of invertebrate functional groups to characterize ecosystem attributes in selected streams and rivers in south brazil. Stud. Neotro. Fauna Environ. 2005, 40, 69–89. [Google Scholar] [CrossRef]
- Ter Braak, C.J.; Verdonschot, P.F. Canonical correspondence analysis and related multivariate methods in aquatic ecology. Aquat. Sci. 1995, 57, 255–289. [Google Scholar] [CrossRef]
- Barbour, M.T.; Gerritsen, J.; Snyder, B.; Stribling, J. Rapid Bioassessment Protocols for Use in Streams and Wadeable Rivers; USEPA: Washington, DC, USA, 1999.
- Lake, P. Disturbance, patchiness, and diversity in streams. J. N. Am. Benthol. Soc. 2000, 19, 573–592. [Google Scholar] [CrossRef]
- Lytle, D.A.; Poff, N.L. Adaptation to natural flow regimes. Trends Ecol. Evol. 2004, 19, 94–100. [Google Scholar] [CrossRef] [PubMed]
- Poff, N.L.; Allan, J.D.; Bain, M.B.; Karr, J.R.; Prestegaard, K.L.; Richter, B.D.; Sparks, R.E.; Stromberg, J.C. The natural flow regime. BioScience 1997, 769–784. [Google Scholar] [CrossRef]
- Bunn, S.E.; Arthington, A.H. Basic principles and ecological consequences of altered flow regimes for aquatic biodiversity. Environ. Manag. 2002, 30, 492–507. [Google Scholar] [CrossRef]
- Arthington, A.H.; Bunn, S.E.; Poff, N.L.; Naiman, R.J. The challenge of providing environmental flow rules to sustain river ecosystems. Ecol. Appl. 2006, 16, 1311–1318. [Google Scholar] [CrossRef]
- Anderson, D.; Moggridge, H.; Shucksmith, J.; Warren, P. Quantifying the impact of water abstraction for low head “run of the river” hydropower on localized river channel hydraulics and benthic macroinvertebrates. River Res. Appl. 2015, 2015. [Google Scholar] [CrossRef]
- Irvine, R.L.; Oussoren, T.; Baxter, J.S.; Schmidt, D.C. The effects of flow reduction rates on fish stranding in british columbia, canada. River Res. Appl. 2009, 25, 405–415. [Google Scholar] [CrossRef]
- Nakamura, Y.; Stefan, H.G. Effect of flow velocity on sediment oxygen demand: Theory. J. Environ. Eng. 1994, 120, 996–1016. [Google Scholar] [CrossRef]
- Matthews, K.; Berg, N.H. Rainbow trout responses to water temperature and dissolved oxygen stress in two southern california stream pools. J. Fish Biol. 1997, 50, 50–67. [Google Scholar] [CrossRef]
- Williams, G.P.; Wolman, M.G. Downstream Effects of Dams on Alluvial Rivers. Avaliable online: http://relicensing.pcwa.net/documents/Library/PCWA-L-307.pdf (accessed on 11 November 2011).
- Friedman, J.M.; Lee, V.J. Extreme floods, channel change, and riparian forests along ephemeral streams. Ecol. Monogr. 2002, 72, 409–425. [Google Scholar] [CrossRef]
- Stromberg, J.; Beauchamp, V.; Dixon, M.; Lite, S.; Paradzick, C. Importance of low-flow and high-flow characteristics to restoration of riparian vegetation along rivers in arid south-western united states. Freshw. Biol. 2007, 52, 651–679. [Google Scholar] [CrossRef]
- Rood, S.B.; Mahoney, J.M.; Reid, D.E.; Zilm, L. Instream flows and the decline of riparian cottonwoods along the st. Mary river, Alberta. Can. J. Bot. 1995, 73, 1250–1260. [Google Scholar] [CrossRef]
- Jansson, R.; Nilsson, C.; Dynesius, M.; Andersson, E. Effects of river regulation on river-margin vegetation: A comparison of eight boreal rivers. Ecol. Appl. 2000, 10, 203–224. [Google Scholar] [CrossRef]
- Simon, A.; Collison, A.J. Quantifying the mechanical and hydrologic effects of riparian vegetation on streambank stability. Earth Surf. Proces. Landf. 2002, 27, 527–546. [Google Scholar] [CrossRef]
- Huo, T. Community structures of macrobenthos and biodiversity in natural aquatic germplasm resource reserve zone in hailang river. Chin. J. Fish. 2012, 3, 006. [Google Scholar]
- Fei, T. Macrobenthos Community Structure and Biological Evaluational Research of Water Quality in Hailang River. Master’s Thesis, Northeast Forestry University, Harbin, China, 2012. [Google Scholar]
- Wright, J.; Berrie, A. Ecological effects of groundwater pumping and a natural drought on the upper reaches of a chalk stream. Regul. Rivers Res. Manag. 1987, 1, 145–160. [Google Scholar] [CrossRef]
- McKay, S.; King, A. Potential ecological effects of water extraction in small, unregulated streams. River Res. Appl. 2006, 22, 1023–1037. [Google Scholar] [CrossRef]
- McIntosh, M.D.; Benbow, M.E.; Burky, A.J. Effects of stream diversion on riffle macroinvertebrate communities in a maui, hawaii, stream. River Res. Appl. 2002, 18, 569–581. [Google Scholar] [CrossRef]
- Cazaubon, A.; Giudicelli, J. Impact of the residual flow on the physical characteristics and benthic community (algae, invertebrates) of a regulated mediterranean river: The durance, france. Regul. Rivers Res. Manag. 1999, 15, 441–461. [Google Scholar] [CrossRef]
- Dewson, Z.S.; James, A.B.; Death, R.G. A review of the consequences of decreased flow for instream habitat and macroinvertebrates. J. N. Am. Benthol. Soc. 2007, 26, 401–415. [Google Scholar] [CrossRef]
- Pilière, A.; Schipper, A.M.; Breure, A.M.; Posthuma, L.; de Zwart, D.; Dyer, S.D.; Huijbregts, M.A. Comparing responses of freshwater fish and invertebrate community integrity along multiple environmental gradients. Ecol. Indic. 2014, 43, 215–226. [Google Scholar] [CrossRef]
- Miller, S.W.; Wooster, D.; Li, J.L. Does species trait composition influence macroinvertebrate responses to irrigation water withdrawals: Evidence from the intermountain west, USA. River Res. Appl. 2010, 26, 1261–1280. [Google Scholar] [CrossRef]
- Quinn, J.M.; Hickey, C.W. Hydraulic parameters and benthic invertebrate distributions in two gravel-bed new zealand rivers. Freshw. Biol. 1994, 32, 489–500. [Google Scholar] [CrossRef]
- Heino, J. Lentic macroinvertebrate assemblage structure along gradients in spatial heterogeneity, habitat size and water chemistry. Hydrobiologia 2000, 418, 229–242. [Google Scholar] [CrossRef]
- Englund, G.; Malmqvist, B. Effects of flow regulation, habitat area and isolation on the macroinvertebrate fauna of rapids in north swedish rivers. Regul. Rivers Res. Manag. 1996, 12, 433–446. [Google Scholar] [CrossRef]
- Likens, G.E. River Ecosystem Ecology: A global Perspective; Academic Press: Cambridge, MA, USA, 2010. [Google Scholar]
- Morin, A. Intensity and importance of abiotic control and inferred competition on biomass distribution patterns of simuliidae and hydropsychidae in southern quebec streams. J. N. Am. Benthol. Soc. 1991, 10, 388–403. [Google Scholar] [CrossRef]
- Parr, L.; Mason, C. Long-term trends in water quality and their impact on macroinvertebrate assemblages in eutrophic lowland rivers. Water Res. 2003, 37, 2969–2979. [Google Scholar] [CrossRef]
- González, R.A.; Díaz, F.; Licea, A.; Re, A.D.; Sánchez, L.N.; García-Esquivel, Z. Thermal preference, tolerance and oxygen consumption of adult white shrimp litopenaeus vannamei (boone) exposed to different acclimation temperatures. J. Therm. Biol. 2010, 35, 218–224. [Google Scholar] [CrossRef]
- Korte, T. Current and substrate preferences of benthic invertebrates in the rivers of the hindu kush-himalayan region as indicators of hydromorphological degradation. Hydrobiologia 2010, 651, 77–91. [Google Scholar] [CrossRef]
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons by Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wang, H.; Chen, Y.; Liu, Z.; Zhu, D. Effects of the “Run-of-River” Hydro Scheme on Macroinvertebrate Communities and Habitat Conditions in a Mountain River of Northeastern China. Water 2016, 8, 31. https://doi.org/10.3390/w8010031
Wang H, Chen Y, Liu Z, Zhu D. Effects of the “Run-of-River” Hydro Scheme on Macroinvertebrate Communities and Habitat Conditions in a Mountain River of Northeastern China. Water. 2016; 8(1):31. https://doi.org/10.3390/w8010031
Chicago/Turabian StyleWang, Haoran, Yongcan Chen, Zhaowei Liu, and Dejun Zhu. 2016. "Effects of the “Run-of-River” Hydro Scheme on Macroinvertebrate Communities and Habitat Conditions in a Mountain River of Northeastern China" Water 8, no. 1: 31. https://doi.org/10.3390/w8010031
APA StyleWang, H., Chen, Y., Liu, Z., & Zhu, D. (2016). Effects of the “Run-of-River” Hydro Scheme on Macroinvertebrate Communities and Habitat Conditions in a Mountain River of Northeastern China. Water, 8(1), 31. https://doi.org/10.3390/w8010031






