Nutrient Content at the Sediment-Water Interface of Tile-Fed Agricultural Drainage Ditches
Abstract
:1. Introduction
2. Experiments
2.1. Study Site
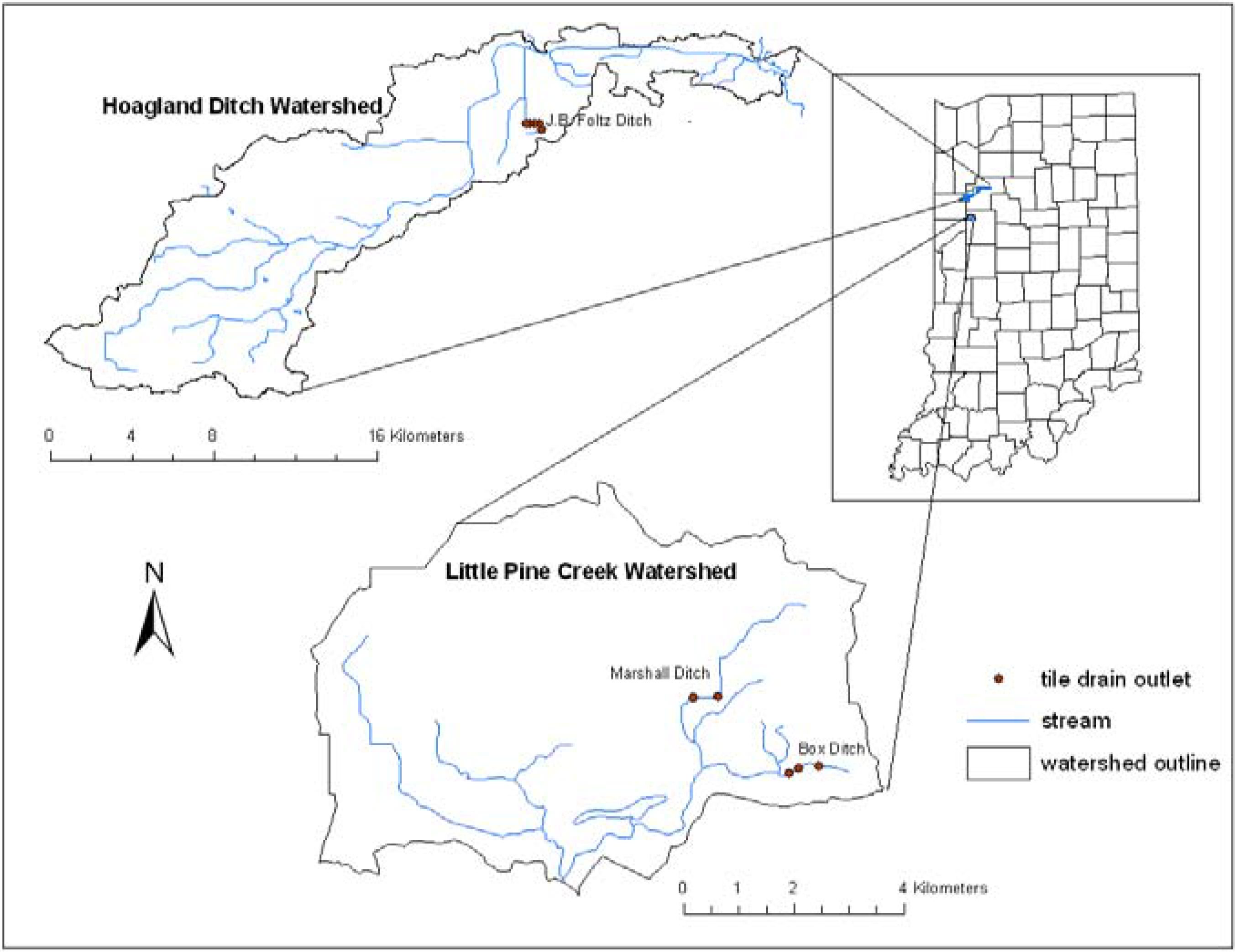
2.2. Field Techniques
| Ditch | Tile outlet | Diameter (cm) | Longitude | Latitude | Distance (m) |
| Box Ditch, Reynolds, IN | 1a | 183 | W 86º 59.899 | N 40º 29.659 | 0 |
| 2 | 23 | W 86º 59.897 | N 40º 29.658 | 7 | |
| 3 | 23 | W 87º 00.114 | N 40º 29.668 | 336 | |
| 4 | 23 | W 87º 00.269 | N 40º 29.593 | 456 | |
| J.B. Foltz Ditch, West Lafayette, IN | 1 | 17 | W 86º 55.215 | N 40º 46.868 | 0 |
| 2 | 15 | W 86º 55.295 | N 40º 47.020 | 385 | |
| 3 | 20 | W 86º 55.464 | N 40º 47.017 | 621 | |
| 4 | 20 | W 86º 55.608 | N 40º 47.010 | 826 | |
| 5 | 20 | W 86º 55.756 | N 40º 47.006 | 1,036 | |
| Marshall Ditch, West Lafayette, IN | 1 | 36 | W 87º 01.186 | N 40º 30.351 | 0 |
| 2b | 31 | W 87º 01.506 | N 40º 30.336 | 431 | |
| 1a : small ditch entering the main ditch through a pipe | |||||
| 2b: conventional weir flow control structure with circular spillway | |||||
2.3. Laboratory Techniques
2.4. Statistics
3. Results and Discussion
3.1. Chemical Characteristics of the Ditch Water Column
| Season | Ditch | pH | Salinity | DO | Cond | Temp |
| (g kg-1) | (mg L-1) | (µS) | (°C) | |||
| Jan-Mar | ||||||
| (Winter) | Box | 7.9 | 0.3 | 6.7 | 329 | 1.10 |
| J.B. Foltz | 7.8 | 0.3 | 5.5 | 343 | 3.03 | |
| Marshall | 7.3 | 0.3 | 6.5 | 324 | 1.57 | |
| Apr-Jun | ||||||
| (Spring) | Box | 8.2 | 0.3 | 3.8 | 423 | 11.1 |
| J.B. Foltz | 7.7 | 0.2 | 2.1 | 462 | 20.2 | |
| Marshall | 8.0 | 0.3 | 4.4 | 426 | 8.37 | |
| Jul-Sep | ||||||
| (Summer) | Box | 7.7 | 0.3 | 3.6 | 509 | 19.0 |
| J.B. Foltz | 8.1 | 0.3 | 5.9 | 537 | 23.8 | |
| Marshall | 8.0 | 0.4 | 4.6 | 664 | 14.8 | |
| Oct-Dec | ||||||
| (Fall) | Box | 8.0 | 0.3 | 8.5 | 325 | 3.82 |
| J.B. Foltz | 7.9 | 0.3 | 8.9 | 346 | 6.69 | |
| Marshall | 7.9 | 0.2 | 5.6 | 385 | 3.71 |
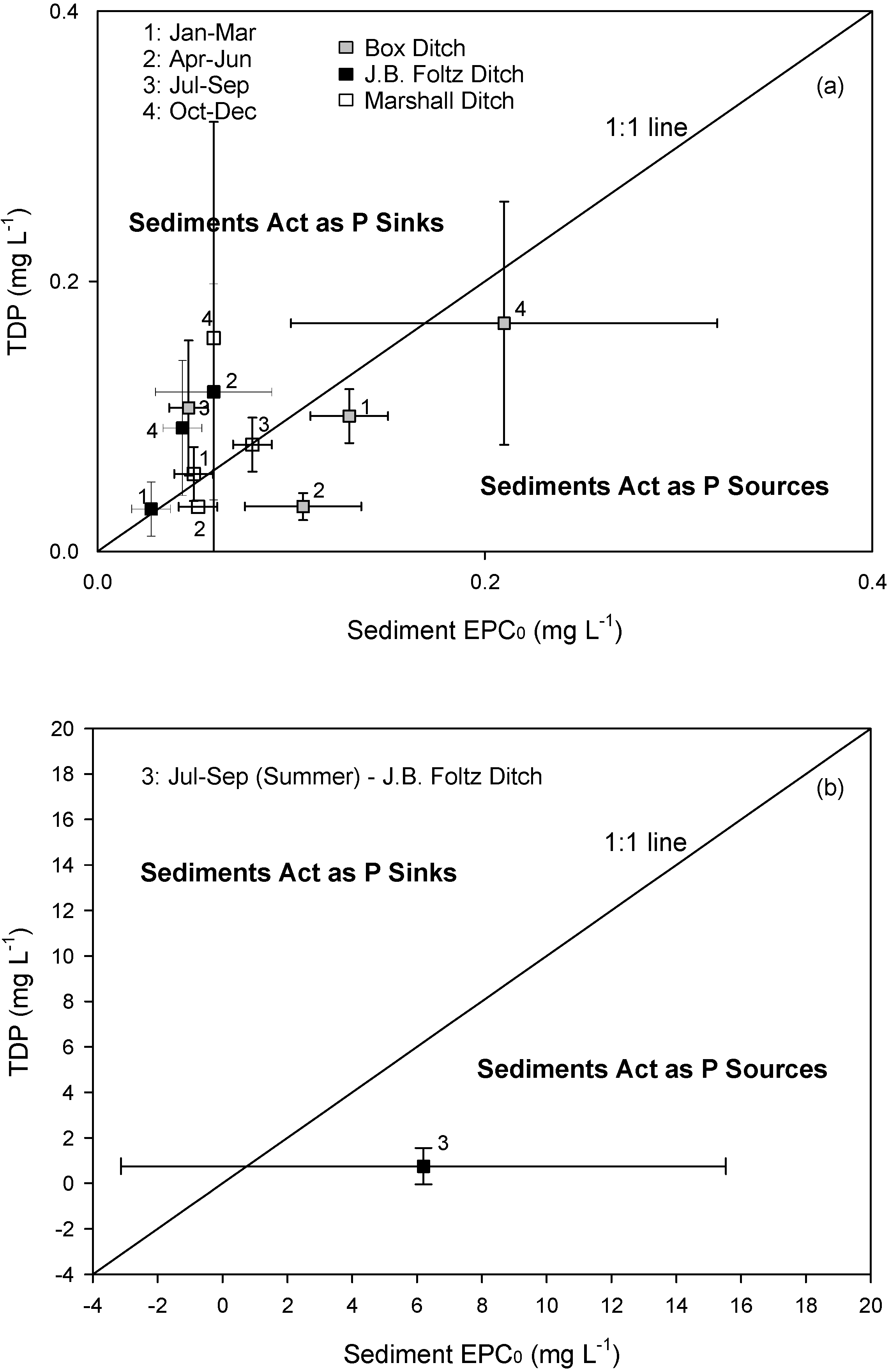
3.2. Sediment Equilibrium Characteristics
| EPC0 | TDP | |||||
| (mg L–1) | (mg L–1 ) | |||||
| Mean | SD | r2 range | p–value | Mean | SD | |
| Jan–Mar | ||||||
| Box | 0.13 | 0.02 | 0.24–0.94 | 0.09 | 0.10 | 0.02 |
| J.B. Foltz | 0.03 | 0.01 | 0.50–0.99 | 0.005 | 0.03 | 0.02 |
| Marshall | 0.05 | 0.01 | 0.41–0.99 | <0.08 | 0.06 | 0.02 |
| Apr–Jun | ||||||
| Box | 0.11* | 0.03 | 0.50–0.99 | <0.06 | 0.03* | 0.01 |
| J.B. Foltz | 0.06* | 0.03 | 0.60–0.98 | <0.05 | 0.12* | 0.08 |
| Marshall | 0.05* | 0.01 | 0.89–0.99 | <0.02 | 0.03* | 0.00 |
| Jul–Sep | ||||||
| Box | 0.05* | 0.01 | 0.90–0.99 | 0.01 | 0.11* | 0.05 |
| J.B. Foltz | 6.20* | 9.33 | 0.08–0.99 | – | 0.75* | 0.80 |
| Marshall | 0.08 | 0.01 | 0.96–0.99 | <0.003 | 0.08 | 0.02 |
| Oct–Dec | ||||||
| Box | 0.21 | 0.11 | 0.41–0.99 | 0.09 | 0.17 | 0.09 |
| J.B. Foltz | 0.04* | 0.01 | 0.87–0.99 | 0.02 | 0.09* | 0.05 |
| Marshall | 0.06* | 0.00 | 0.45–0.99 | <0.04 | 0.16* | 0.03 |
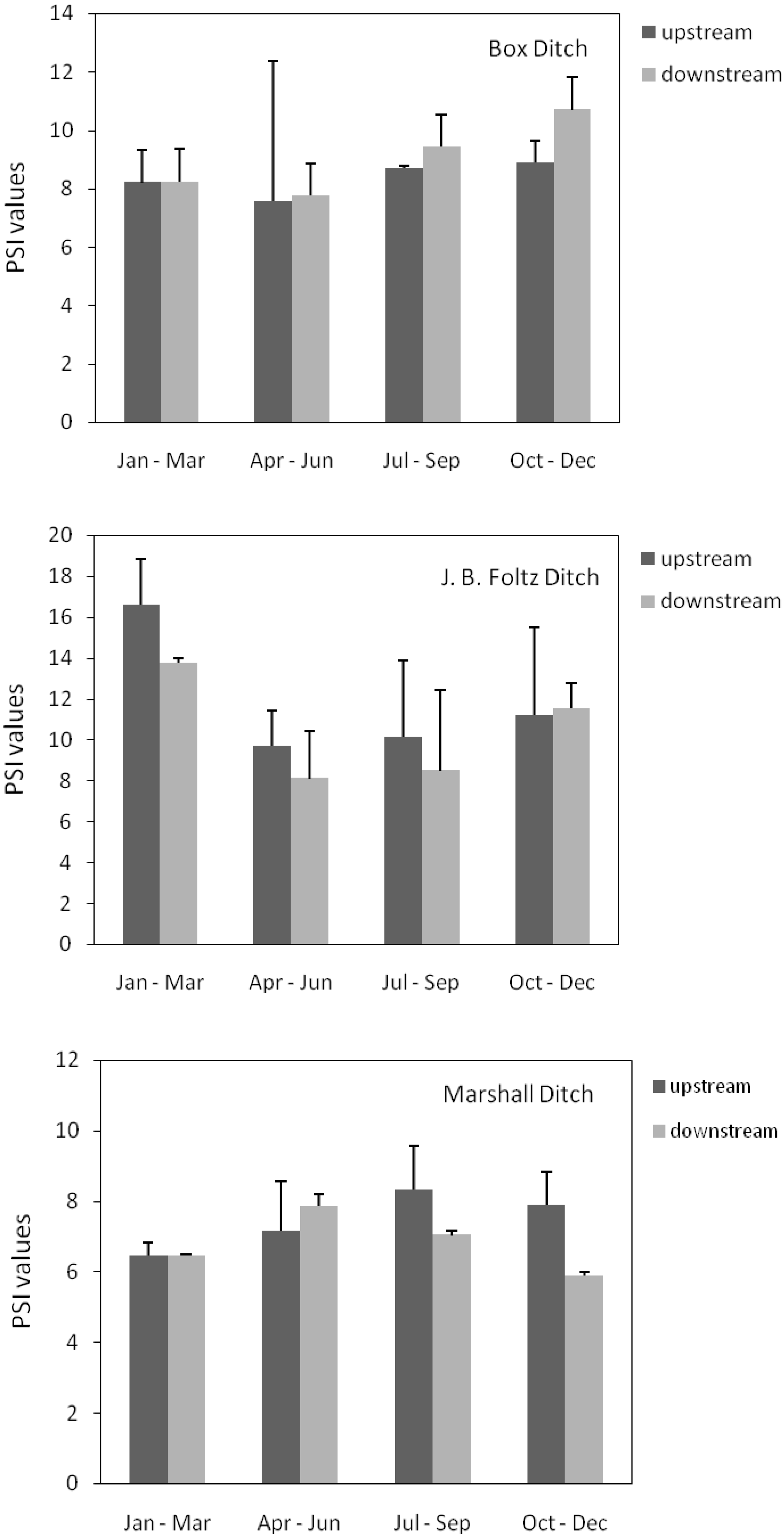
| EPC0 | TDP | |||||||||
| Upstream | Downstream | Upstream | Downstream | |||||||
| Mean | SD | Mean | SD | Mean | SD | Mean | SD | |||
| Jan-Mar | ||||||||||
| Box | 0.143 | 0.010 | 0.104 | 0.000 | 0.100 | 0.020 | 0.101 | 0.002 | ||
| J.B. Foltz | 0.025 | 0.014 | 0.027 | 0.012 | 0.027 | 0.019 | 0.036 | 0.018 | ||
| Marshall | 0.049 | 0.018 | 0.051 | 0.000 | 0.049 | 0.006 | 0.075 | 0.000 | ||
| Apr-Jun | ||||||||||
| Box | 0.107 | 0.024 | 0.104 | 0.026 | 0.033 | 0.008 | 0.033 | 0.011 | ||
| J.B. Foltz | 0.046 | 0.020 | 0.067 | 0.029 | 0.105 | 0.088 | 0.131 | 0.073 | ||
| Marshall | 0.052 | 0.007 | 0.051 | 0.010 | 0.032 | 0.005 | 0.036 | 0.001 | ||
| Jul-Sep | ||||||||||
| Box | 0.046 | 0.010 | 0.051 | 0.000 | 0.114 | 0.040 | 0.095 | 0.055 | ||
| J.B. Foltz | 4.776 | 0.924 | 7.714 | 10.959 | 0.580 | 0.782 | 0.911 | 0.908 | ||
| Marshall | 0.076 | 0.004 | 0.085 | 0.002 | 0.088 | 0.021 | 0.062 | 0.020 | ||
| Oct-Dec | ||||||||||
| Box | 0.252 | 0.090 | 0.144 | 0.006 | 0.159 | 0.081 | 0.184 | 0.079 | ||
| J.B. Foltz | 0.046 | 0.019 | 0.042 | 0.010 | 0.084 | 0.058 | 0.099 | 0.056 | ||
| Marshall | 0.063 | 0.001 | 0.109 | 0.000 | 0.140 | 0.020 | 0.194 | 0.010 | ||
3.3. Phosphorus Uptake of Upstream and Downstream Sediments
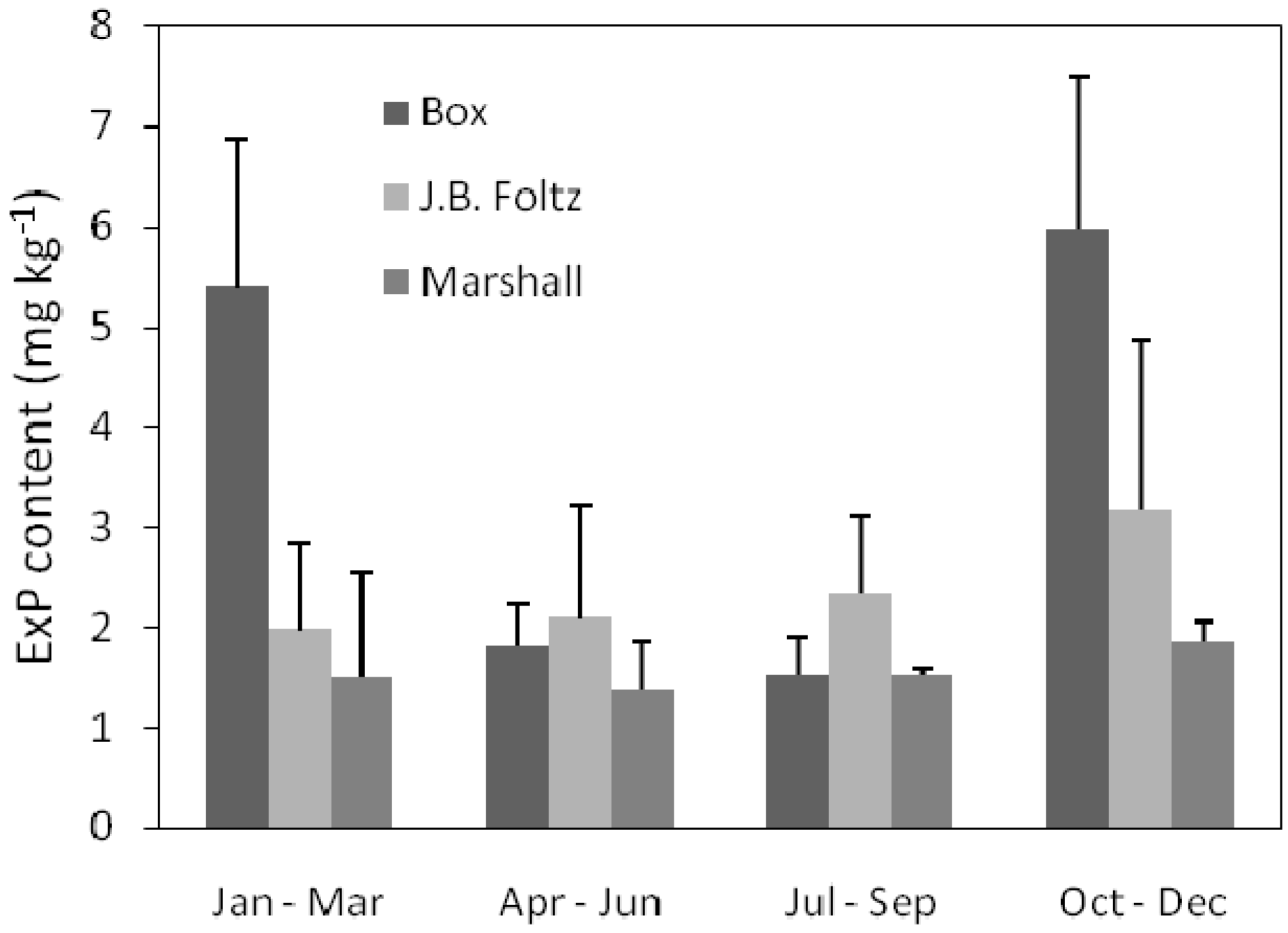
3.4. Sediment Exchangeable Nutrient
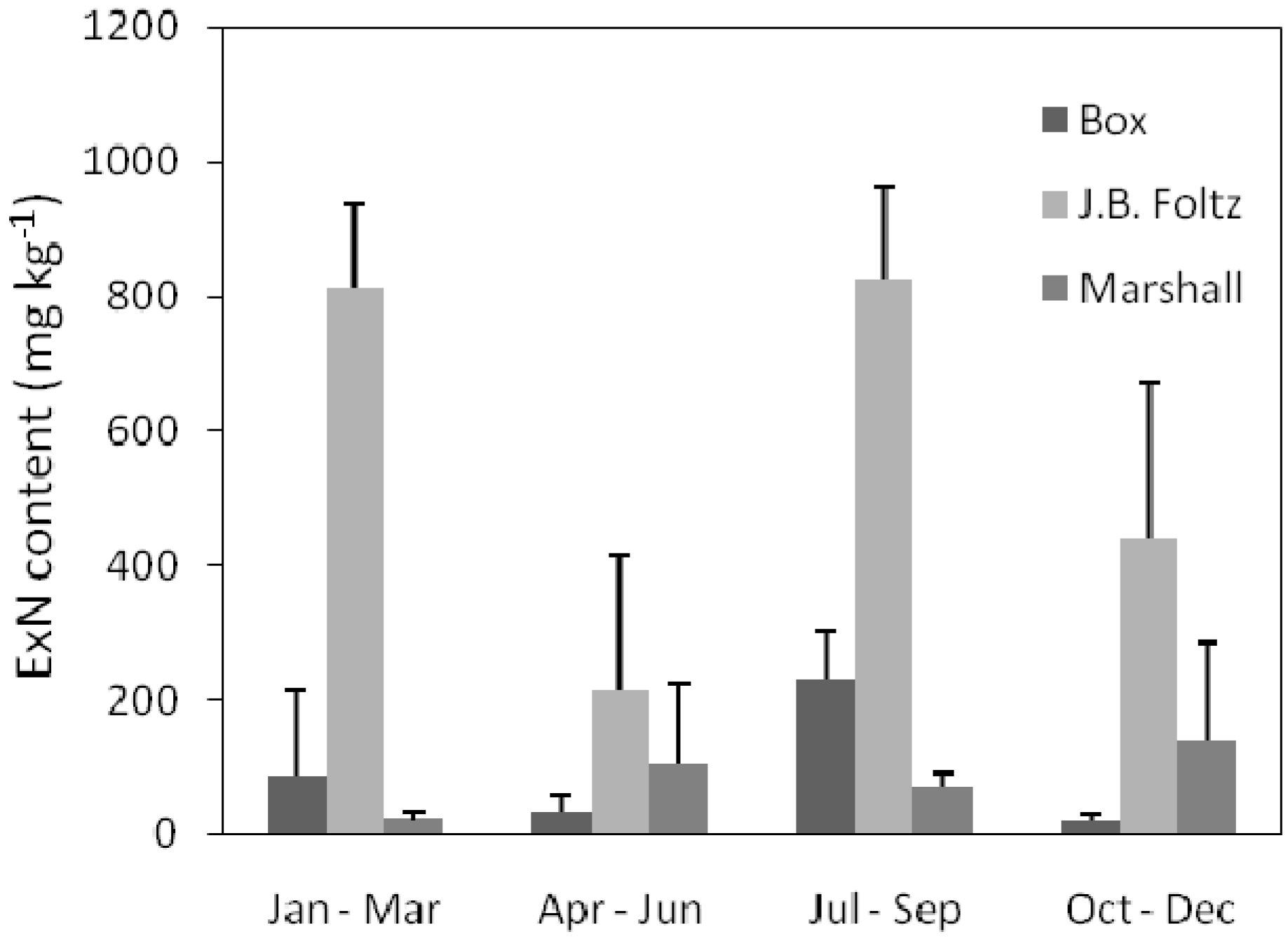
4. Conclusions
Acknowledgements
References
- Smith, D.R.; Haggard, B.E.; Warnemuende, E.A.; Huang, C. Sediment phosphorus dynamics for three tile fed drainage ditches in northeast Indiana. Agric. Water Manag. 2005, 71, 19–32. [Google Scholar] [CrossRef]
- Kleinman, P.J.A.; Allen, A.L.; Needleman, B.A.; Sharpley, A.N.; Vadas, P.A.; Saporito, L.S.; Folmar, G.J.; Bryant, R.B. Dynamics of phosphorus transfers from heavily manured coastal plain soils to drainage ditches. J. Soil Water Conserv. 2007, 62, 225–234. [Google Scholar]
- Sharpley, A.N.; Krogstad, T.; Kleinman, P.J.A.; Haggard, B.; Shigaki, F.; Saporito, L.S. Managing natural processes in drainage ditches for nonpoint source phosphorus control. J. Soil Water Conserv. 2007, 62, 197–205. [Google Scholar]
- Gentry, L.E.; David, M.B.; Royer, T.V. Phosphorus transport pathways to streams in tile-drained agricultural watersheds. J. Environ. Qual. 2007, 36, 408–415. [Google Scholar] [CrossRef] [PubMed]
- Alexander, R.B.; Smith, R.A.; Schwarz, G.E.; Boyer, E.W.; Nolan, J.V.; Brakebill, J.W. Differences in phosphorus and nitrogen delivery to the Gulf of Mexico from the Mississippi River Basin. Environ. Sci. Technol. 2008, 42, 822–830. [Google Scholar] [CrossRef] [PubMed]
- Sims, J.T.; Simard, R.R.; Joern, B.C. Phosphorus loss in agricultural drainage: Historical perspective and current research. J. Environ. Qual. 1998, 27, 277–293. [Google Scholar] [CrossRef]
- Dunne, E.; McKee, K.A.; Clark, M.W.; Grunwald, S.; Reddy, K.R. Phosphorus in agricultural ditch soil and potential implications for water quality. J. Soil Water Conserv. 2007, 62, 244–252. [Google Scholar]
- DeLaune, P.B.; Moore, P.A., Jr.; Carman, D.K.; Sharpley, A.N.; Haggard, B.E.; Daniel, T.C. Development of a phosphorus index for pastures fertilized with poultry litter-Factors affecting phosphorus runoff. J. Environ. Qual. 2004, 33, 2183–2191. [Google Scholar] [CrossRef] [PubMed]
- Smith, D.R.; Linvingston, S.J.; Zuercher, B.W.; Larose, M.; Heathman, G.C.; Huang, C. Nutrient losses from row crop agriculture in Indiana. J. Soil Water Conserv. 2008, 63, 396–409. [Google Scholar] [CrossRef]
- House, W.A.; Donaldson, L. Adsorption and coprecipitation of phosphate with calcite. J. Colloid Interface Sci. 1986, 112, 309–324. [Google Scholar] [CrossRef]
- Lottig, N.R.; Stanley, E.H. Benthic sediment influence on phosphorus concentrations in a headwater stream. Biogeochemistry 2007, 84, 297–309. [Google Scholar] [CrossRef]
- Haggard, B.E.; Ekka, S.A.; Matlock, M.D.; Chaubey, I. Phosphate equilibrium between stream sediments and water: Potential effect of chemical amendments. Trans. ASAE 2004, 47, 1113–1118. [Google Scholar] [CrossRef]
- McDaniel, M.D.; David, M.B.; Royer, T.V. Relationships between benthic sediments and water column phosphorus in illinois streams. J. Environ. Qual. 2009, 38, 607–617. [Google Scholar]
- Smith, D.R.; Warnemuende, E.A.; Haggard, B.E.; Huang, C. Changes in sediment-water column phosphorus interactions following sediment disturbance. Ecol. Engng. 2006, 27, 71–78. [Google Scholar] [CrossRef]
- Meyer, J.L. The role of sediments and bryophytes in phosphorus dynamics in a headwater ecosystem. Limnol. Oceanogr. 1979, 24, 365–375. [Google Scholar]
- Smith, D.R. Assessment of in-stream phosphorus dynamics in agricultural drainage ditches. Sci. Total Environ. 2009, 407, 3883–3889. [Google Scholar] [CrossRef] [PubMed]
- Kröger, R; Holland, M.M. Hydrolodical variability and agricultural drainage ditch inorganic nitrogen reduction capacity. J. Environ. Qual. 2007, 36, 1646–1652. [Google Scholar] [CrossRef] [PubMed]
- Dorioz, J.M.; Cassell, E.A.; Orand, A.; Eisenman, K.G. Phosphorus storage, transport and export dynamics in the Foron River watershed. Hydrol.l Process. 1998, 12, 285–309. [Google Scholar] [CrossRef]
- McDowell, R.W.; Sharpley, A.; Folmar, G. Phosphorus export from an agricultural watershed: Linking source and transport mechanisms. J. Environ. Qual. 2001, 30, 1587–1595. [Google Scholar] [CrossRef] [PubMed]
- Haggard, B.E.; Storm, D.E.; Stanley, E.H. Effect of a point source input on stream nutrient retention. J. Am. Wat. Resour. Ass. 2001, 35, 1291–1299. [Google Scholar] [CrossRef]
- Smith, D.R.; Moore, P.A., Jr.; Maxwell, C.V.; Haggard, B.E.; Daniel, T.C. Reducing phosphorus runoff from swine manure with dietary phytase and aluminum chloride. J. Environ. Qual. 2004, 33, 1048–1054. [Google Scholar] [CrossRef] [PubMed]
- Smith, D.R.; Moore, P.A., Jr.; Miles, D.M.; Haggard, B.E.; Daniel, T.C. Reducing phosphorus runoff from land applied poultry litter with dietary modifications and alum addition. J. Environ. Qual. 2004, 33, 2210–2216. [Google Scholar] [CrossRef] [PubMed]
- Alexander, R.B.; Smith, R.A.; Schwarz, G.E. Effect of stream channel size on the delivery of nitrogen to the Gulf of Mexico. Nature 2000, 403, 758–761. [Google Scholar] [CrossRef] [PubMed]
- Bernot, M.J.; Tank, J.L.; Royer, T.V.; David, M.B. Nutrient uptake in streams draining agricultural catchments of the midwestern United States. Freshw. Biol. 2006, 51, 499–509. [Google Scholar]
- Gentry, L.E.; David, M.B.; Smith, K.M.; Kovacic, D.A. Nitrogen cycling and tile drainage nitrate loss in a corn/soybean watershed. Agricult. Ecosys. Environ. 1998, 68, 85–97. [Google Scholar] [CrossRef]
- Kladivko, E.J.; Frankenberger, J.R.; Jaynes, D.B.; Meek, D.W.; Jenkinson, B.J.; Faussey, N.R. Nitrate leaching to subsurface drains as affected by drain spacing and changes in crop production system. J. Environ. Qual. 2004, 33, 1803–1813. [Google Scholar] [CrossRef] [PubMed]
- Royer, T.V.; Tank, J.L.; David, M.B. Transport and fate of nitrate in headwater agricultural streams in Illinois. J. Environ. Qual. 2004, 33, 1296–1304. [Google Scholar] [CrossRef] [PubMed]
- Strock, J.S.; Dell, C.J.; Schmidt, J.P. Managing natural processes in drainage ditches or nonpoint source nitrogen control. J. Soil Water Conserv. 2007, 62, 188–196. [Google Scholar]
- Clark, G.M.; Mueller, D.K.; Mast, M.A. Nutrient concentrations and yields in undeveloped stream basins of the United States. J. Am. Wat. Resour. Ass. 2000, 36, 849–860. [Google Scholar] [CrossRef]
- Kladivko, E.J.; Van Scoyoc, G.E.; Monke, E.J.; Oates, K.M.; Pask, W. Pesticide and nutrient movement into subsurface tile drains on a silt loam soil in Indiana. J. Environ. Qual. 1991, 20, 264–270. [Google Scholar] [CrossRef]
- Randall, G.W.; Vetsch, J.A.; Huffman, J.R. Nitrate losses in subsurface drainage from a corn-soybean rotation as affected by time of nitrogen application and use of nitrapyrin. J. Environ. Qual. 2003, 32, 1764–1772. [Google Scholar] [CrossRef] [PubMed]
- Randall, G.W.; Vetsch, J.A. Nitrate losses in subsurface drainage from a corn-soybean rotation as affected by fall and spring application of nitrogen and nitrapyrin. J. Environ. Qual. 2005, 34, 590–597. [Google Scholar] [CrossRef] [PubMed]
- Petrolia, D.R.; Gowda, P.H. Missing the Boat: Midwest Farm Drainage and Gulf of Mexico Hypoxia. Rev. Agric. Econ. 2006, 28, 240–253. [Google Scholar] [CrossRef]
- Marti, E.; Autmatell, J.; Godé, L.; Poch, M.; Sabater, F. Nutrient retention efficiency in streams receiving inputs from wastewater treatment plants. J. Environ. Qual. 2004, 33, 285–293. [Google Scholar] [CrossRef] [PubMed]
- Naz, B.S.; Ale, S.; Bowling, L.C. Detecting subsurface drainage systems and estimating drain spacing in intensively managed agricultural landscapes. Agric. Water Manag. 2008, 96, 627–637. [Google Scholar] [CrossRef]
- Popova, Y.A. Sediment-Phosphorus Chemistry in Ozark Plateau Streams in Northeast Oklahoma. M.S. Thesis, Oklahoma State University, Oklahoma City, OK, USA, 2000. [Google Scholar]
- Chaubey, I.; Sahoo, D.; Haggard, B.E.; Matlock, M.D.; Costello, T.A. Nutrient retention, nutrient limitation, and sediment-nutrient interactions in a pasture-dominated stream. Trans. ASABE 2007, 50, 35–44. [Google Scholar] [CrossRef]
- Haggard, B.E.; Stanley, E.H.; Hyler, R. Sediment-Phosphorus relationships in three Northcentral Oklahoma streams. Trans. ASAE 1999, 42, 1709–1714. [Google Scholar] [CrossRef]
- Taylor, A.W.; Kunishi, H.M. Phosphate equilibria on stream sediment and soil in a watershed draining an agricultural region. J. Agric. Food Chem. 1971, 19, 827–831. [Google Scholar] [CrossRef]
- Klotz, R.L. Sediment control of soluble reactive phosphorus in Hoxie Gorge Creek, New York. Can. J. Fish Aquat. Sci. 1988, 45, 2026–2034. [Google Scholar] [CrossRef]
- Bache, B.W.; Williams, E.G. Phosphate sorption index for soils. J. Soil Sci. 1971, 22, 289–301. [Google Scholar] [CrossRef]
- Richey, J.S.; McDowell, W.H.; Likens, G.E. Nitrogen transformation in a small mountain stream. Hydrobiogia 1985, 124, 129–139. [Google Scholar] [CrossRef]
- Ruttenburg, K.C. Development of a sequential extraction method of different forms of phosphorus in marine sediments. Limnol. Oceanogr. 1992, 37, 1460–1482. [Google Scholar] [CrossRef]
- Popova, Y.A.; Keyworth, V.G.; Haggard, B.E.; Storm, D.E.; Lynch, R.A.; Payton, M.E. Stream nutrient limitation and sediment interactions in the Eucha-Spavinaw Basin. J. Soil Water Conserv. 2006, 61, 105–115. [Google Scholar]
- Bremner, J.M. Inorganic forms of nitrogen. In Methods of soil analysis; Black, C.A., Ed.; American Society of Agronomy: Madison, WI, USA, 1965; Part 2; pp. 1152–1191. [Google Scholar]
- Rowland, A.P.; Haygarth, P.M. Determination of Total Dissolved Phosphorus in Soil Solutions. J. Environ. Qual. 1997, 26, 410–415. [Google Scholar] [CrossRef]
- Jarvie, H.P.; Withers, P.J.A.; Neal, C. Review of robust measurement of phosphorus in river water: Sampling, storage, fractionation and sensitivity. Hydrol. Earth Syst. Sci. 2002, 6, 113–132. [Google Scholar] [CrossRef]
- House, W.A.; Denison, F.H.; Armitage, P.D. Comparison of the uptake of inorganic phosphorus to a suspended and stream bed-sediment. Wat. Res. 1995, 29, 767–779. [Google Scholar] [CrossRef]
- Klotz, R.L. Factors controlling phosphorus limitation in stream sediments. Limnol. Oceanogr. 1985, 30, 543–553. [Google Scholar] [CrossRef]
- Triska, F.J.; Jackman, A.P.; Duff, J.H.; Avanzino, R.J. Ammonium sorption to channel and riparian sediments: A transient storage pool for dissolved inorganic nitrogen. Biogeochemistry 1994, 2, 67–83. [Google Scholar] [CrossRef]
- SAS, version 9.1; SAS Institute Inc.: Cary, NC, USA, 2003.
- Indiana Administrative Code (IAC). Water Quality Standards. Article 2. Available online: www.in.gov/legislative/iac/pdf-iac/iac2008 (accessed on 30 November 2008).
- Vaughan, R.E.; Needelman, B.A.; Kleinman, P.J.A.; Allen, A.L. Vertical distribution of phosphorus in agricultural drainage ditch soils. J. Environ. Qual. 2007, 36, 1895–1903. [Google Scholar] [CrossRef] [PubMed]
- Ekka, S.A.; Haggard, B.E.; Matlock, M.D.; Chaubey, I. Dissolved phosphorus concentrations and sediment interactions in effluent dominated Ozark streams. Ecol. Engng. 2006, 26, 375–391. [Google Scholar] [CrossRef]
- Jarvie, H.P.; Juergens, M.D.; Williams, R.J.; Neal, C.; Davies, J.J.L.; Barrett, C.; White, J. Role of river bed sediments as sources and sinks of phosphorus across two major eutrophic UK river basins: The Hampshire Avon and Herefordshire Wye. J. Hydrol. 2005, 305, 51–74. [Google Scholar] [CrossRef]
- Smith, D.R.; Warnemuende, E.A.; Haggard, B.E.; Huang, C. Dredging of drainage ditches increases short-term transport of soluble phosphorus. J. Environ. Qual. 2006, 35, 611–616. [Google Scholar] [CrossRef] [PubMed]
- David, M.B.; Gentry, L.E.; Kovacic, D.A. Nitrogen balance in and export from an agricultural watershed. J. Environ. Qual. 1997, 26, 1038–1048. [Google Scholar] [CrossRef]
- Zheng, A.; Shen, H.; Chen, D. Study of exchangeable nitrogen and their bioavailability in the sediments. EIA 2006, 4, 26–34. [Google Scholar]
- Haggard, B.E.; Smith, D.R.; Brye, K.R. Variations in stream water and sediment phosphorus among select Ozark Catchments. J. Environ. Qual. 2007, 36, 1725–1734. [Google Scholar] [CrossRef] [PubMed]
© 2010 by the authors; licensee MDPI, Basel, Switzerland. This article is an Open Access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Ahiablame, L.; Chaubey, I.; Smith, D. Nutrient Content at the Sediment-Water Interface of Tile-Fed Agricultural Drainage Ditches. Water 2010, 2, 411-428. https://doi.org/10.3390/w2030411
Ahiablame L, Chaubey I, Smith D. Nutrient Content at the Sediment-Water Interface of Tile-Fed Agricultural Drainage Ditches. Water. 2010; 2(3):411-428. https://doi.org/10.3390/w2030411
Chicago/Turabian StyleAhiablame, Laurent, Indrajeet Chaubey, and Douglas Smith. 2010. "Nutrient Content at the Sediment-Water Interface of Tile-Fed Agricultural Drainage Ditches" Water 2, no. 3: 411-428. https://doi.org/10.3390/w2030411
APA StyleAhiablame, L., Chaubey, I., & Smith, D. (2010). Nutrient Content at the Sediment-Water Interface of Tile-Fed Agricultural Drainage Ditches. Water, 2(3), 411-428. https://doi.org/10.3390/w2030411




