Formation of AOX and Cl/Br/I-THMs During NaClO Disinfection: A Comprehensive Study
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Experimental Procedure
2.3. Pretreatment Method
2.4. Analytical Method
2.5. Utilization Factors and Insertion Factor
3. Results and Discussions
3.1. The Formation of AOBr and AOI
3.2. Determination of THMs Formation from Lignin Chlorination Containing Only Br−
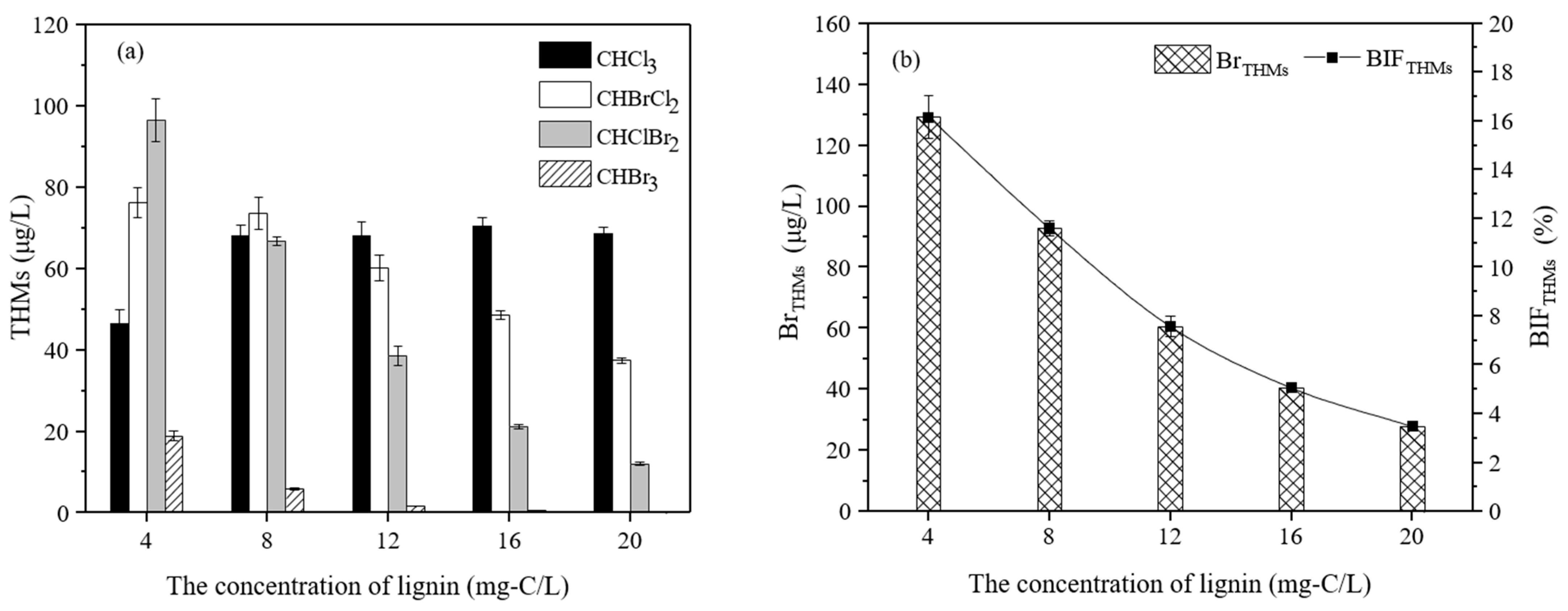
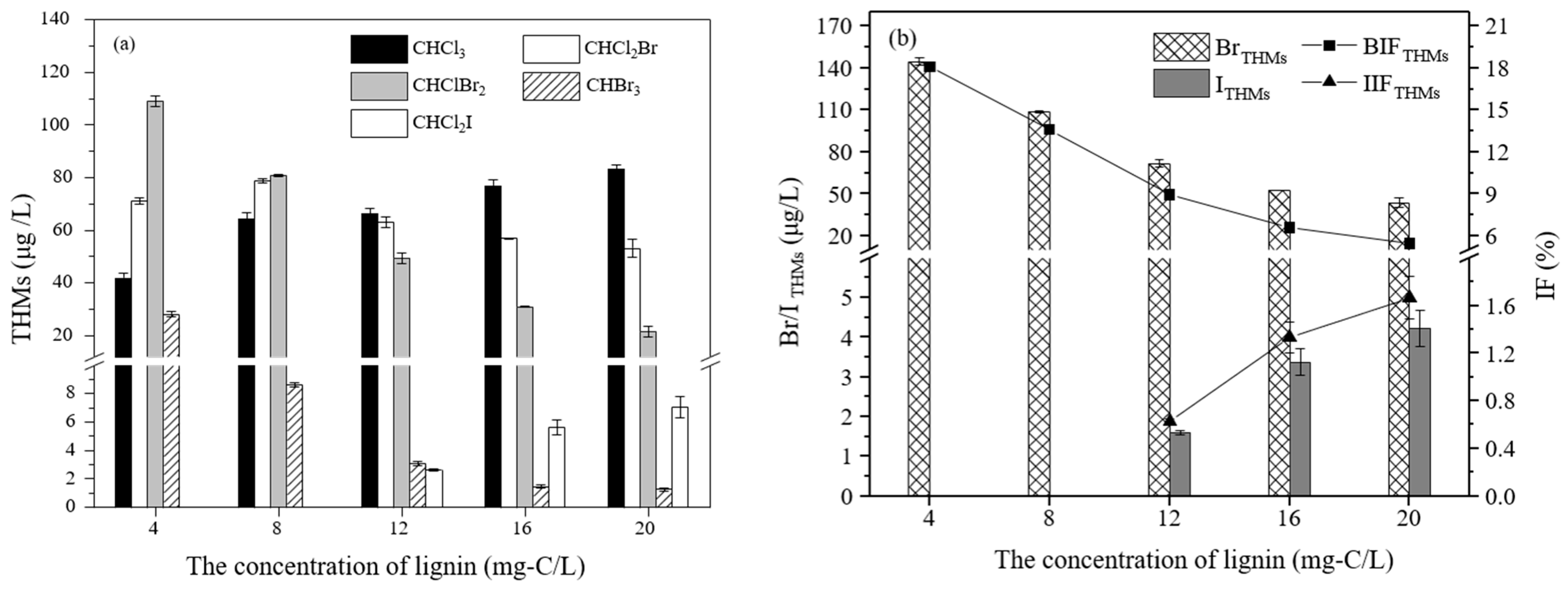
3.2.1. The Effect of the Lignin Concentration on the Formation of THMs
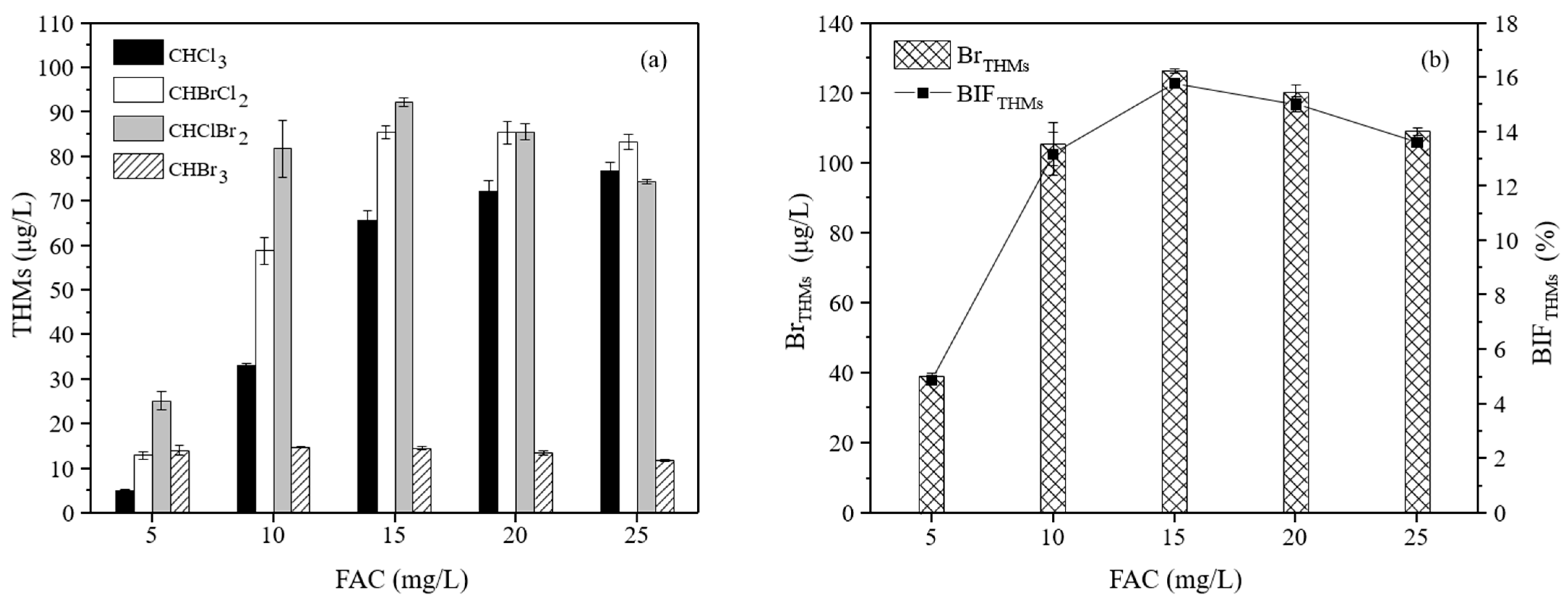
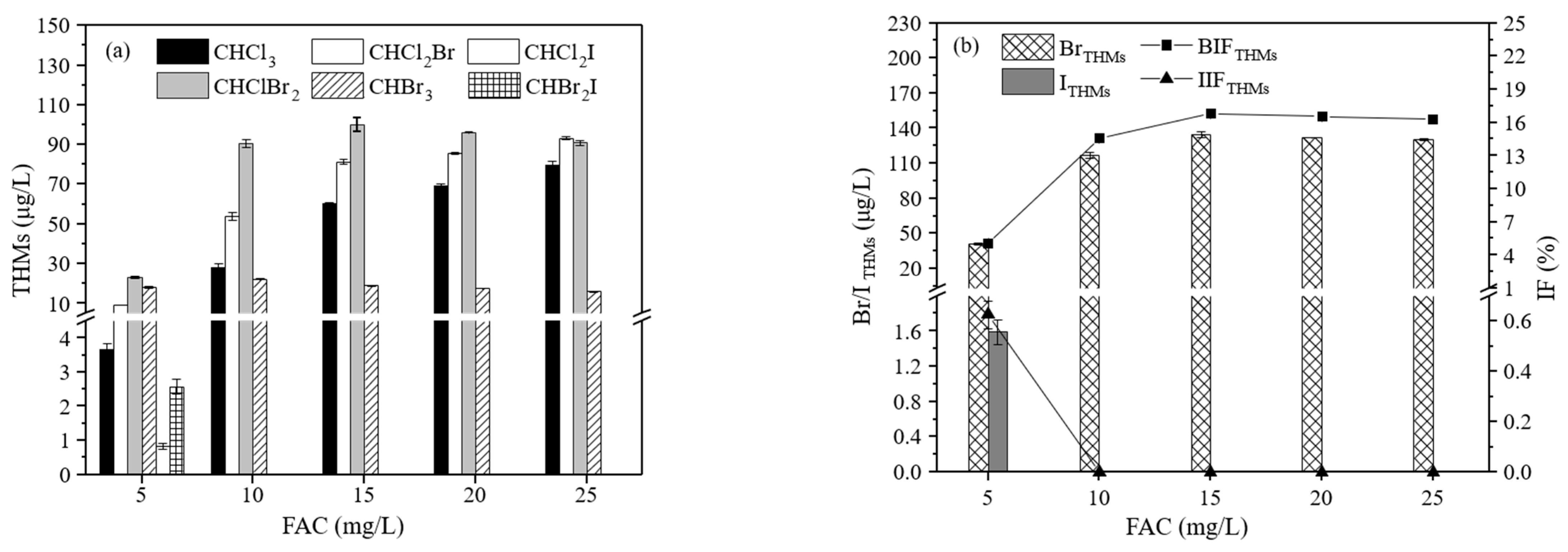
3.2.2. The Effect of the NaClO Dosage on the Formation of THMs
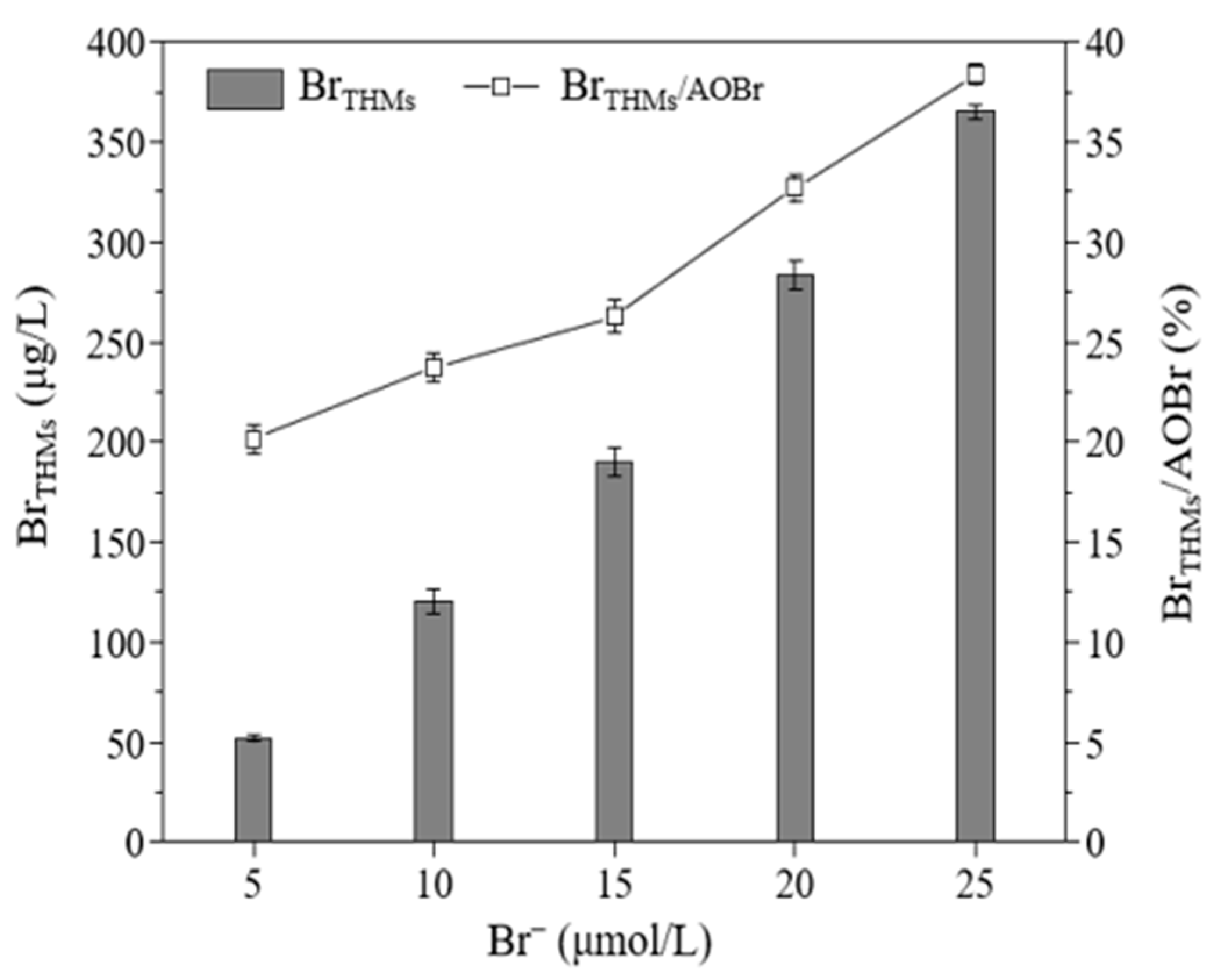
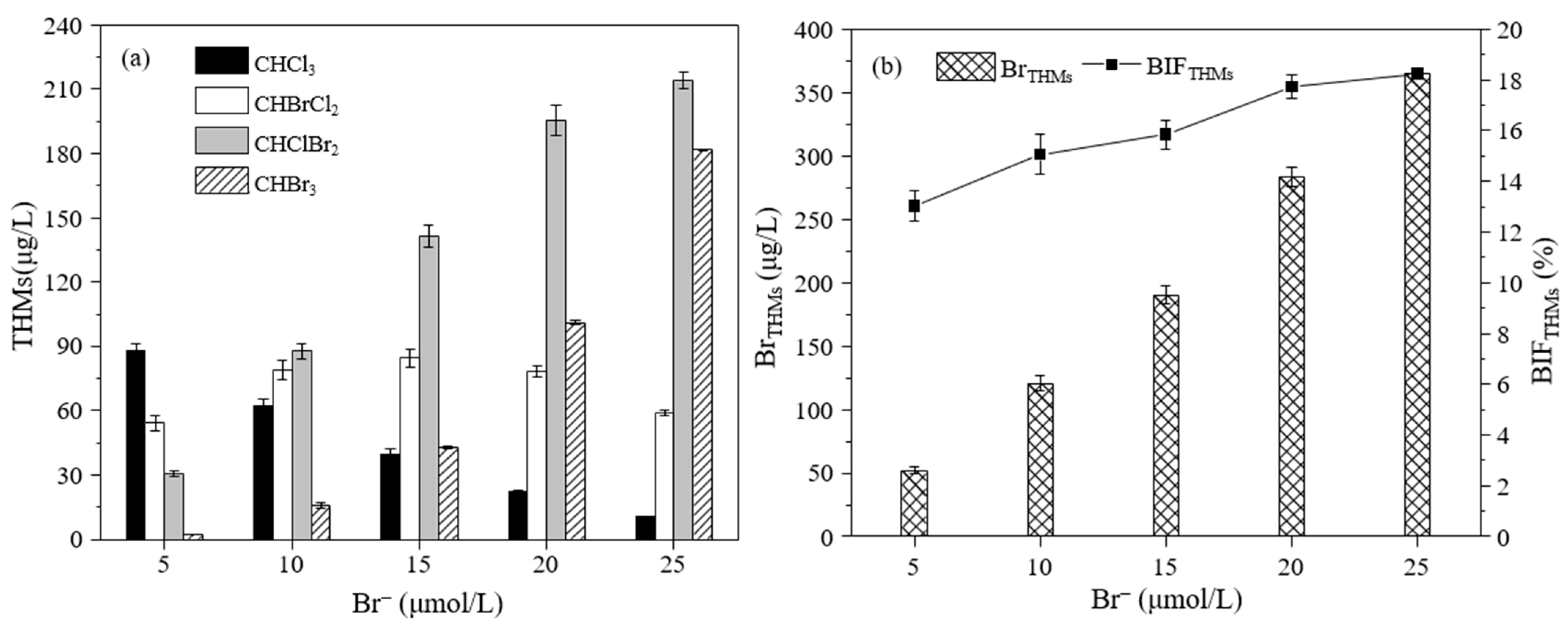
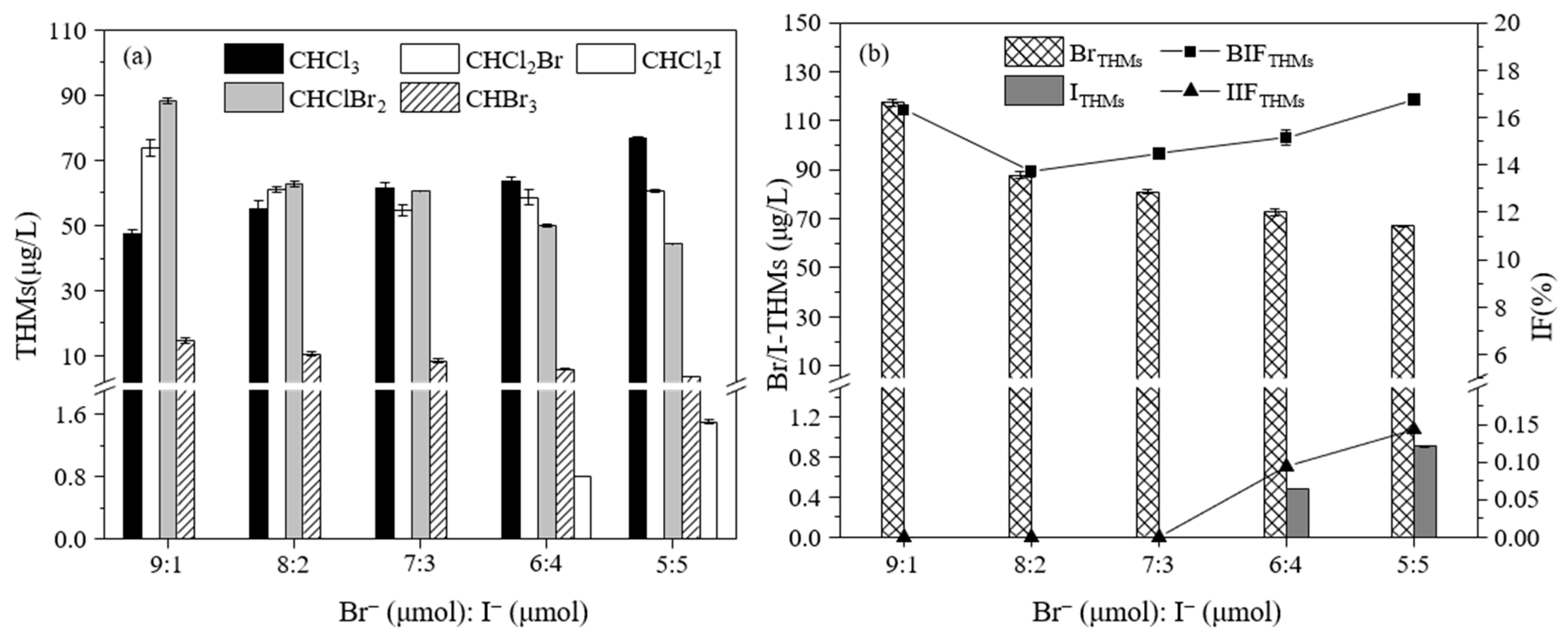
3.2.3. The Effect of the Br− Concentration on the Formation of THMs
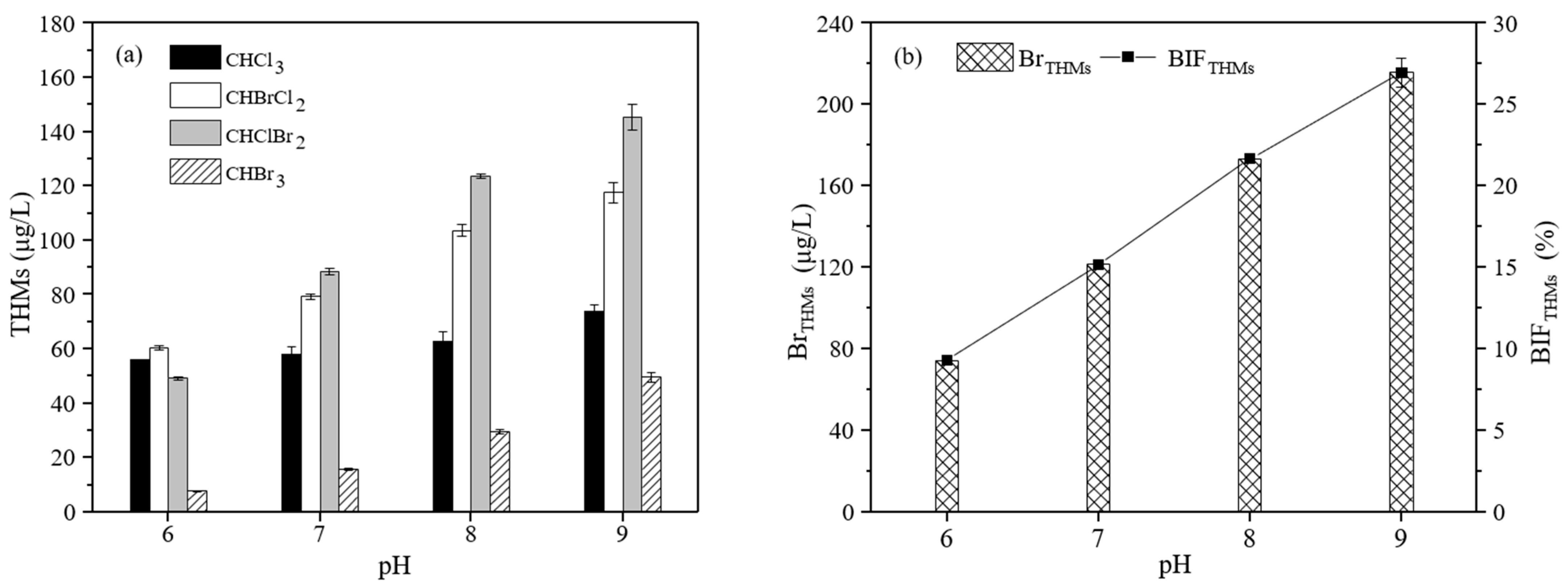
3.2.4. The Effect of pH on the Formation of THMs
3.3. Determination of THMs Formation from Lignin Chlorination Containing Br− and I−
3.3.1. The Effect of the Lignin Concentration on the Formation of THMs
3.3.2. The Effect of the NaClO Dosage on the Formation of THMs
3.3.3. The Effect of the Ratio of Br− and I− on the Formation of THMs
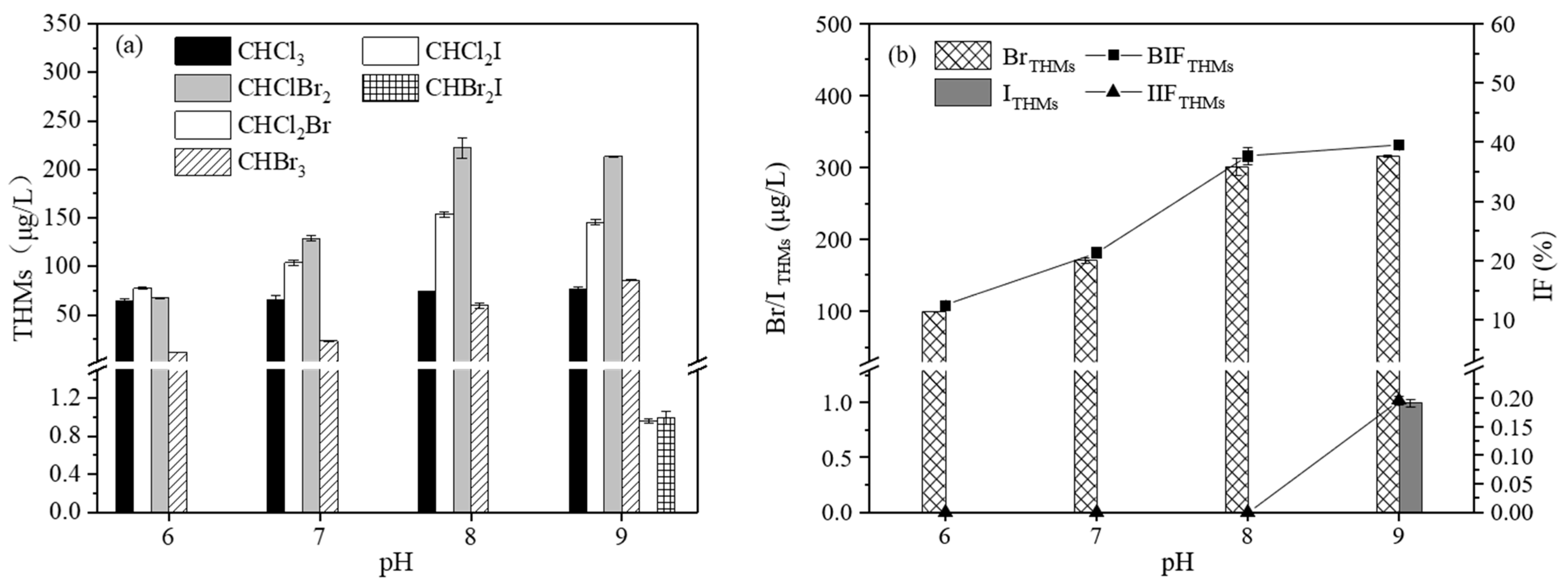
3.3.4. The Effect of pH on the Formation of THMs
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Wan, D.; Wang, H.; Sharma, V.K.; Selvinsimpson, S.; Dai, H.; Luo, F.; Wang, C.; Chen, Y. Mechanistic Investigation of Enhanced Photoreactivity of Dissolved Organic Matter after Chlorination. Environ. Sci. Technol. 2021, 55, 8937–8946. [Google Scholar] [CrossRef]
- Dong, H.; Cuthbertson, A.A.; Plewa, M.J.; Weisbrod, C.R.; McKenna, A.M.; Richardson, S.D. Unravelling High-Molecular-Weight DBP Toxicity Drivers in Chlorinated and Chloraminated Drinking Water: Effect-Directed Analysis of Molecular Weight Fractions. Environ. Sci. Technol. 2023, 57, 18788–18800. [Google Scholar] [CrossRef]
- Gao, Z.-C.; Lin, Y.-L.; Xu, B.; Xia, Y.; Hu, C.-Y.; Zhang, T.-Y.; Qian, H.; Cao, T.-C.; Gao, N.-Y. Effect of bromide and iodide on halogenated by-product formation from different organic precursors during UV/chlorine processes. Water Res. 2020, 182, 116035. [Google Scholar] [CrossRef] [PubMed]
- Komaki, Y.; Ibuki, Y. Inhibition of nucleotide excision repair and damage response signaling by dibromoacetonitrile: A novel genotoxicity mechanism of a water disinfection byproduct. J. Hazard. Mater. 2022, 423, 127194. [Google Scholar] [CrossRef]
- Lau, S.S.; Bokenkamp, K.; Tecza, A.; Wagner, E.D.; Plewa, M.J.; Mitch, W.A. Toxicological assessment of potable reuse and conventional drinking waters. Nat. Sustain. 2023, 6, 39–46. [Google Scholar] [CrossRef]
- Plewa, M.J.; Wagner, E.D.; Muellner, M.G.; Hsu, K.-M.; Richardson, S.D. Comparative Mammalian Cell Toxicity of N-DBPs and C-DBPs. In Disinfection By-Products in Drinking Water; ACS Symposium Series; American Chemical Society: Washington, DC, USA, 2008; Volume 995, pp. 36–50. [Google Scholar]
- Plewa, M.J.; Kargalioglu, Y.; Vankerk, D.; Minear, R.A.; Wagner, E.D. Mammalian cell cytotoxicity and genotoxicity analysis of drinking water disinfection by-products. Environ. Mol. Mutagen. 2002, 40, 134–142. [Google Scholar] [CrossRef]
- Wu, D.-X.; Wang, W.-L.; Du, Y.; He, L.; Wu, Q.-Y. Changes in toxicity and adsorbable organic bromine concentrations in ozonated reclaimed water irradiated with sunlight. Water Res. 2023, 230, 119512. [Google Scholar] [CrossRef] [PubMed]
- Ertel, J.R.; Hedges, J.I.; Perdue, E.M. Lignin signature of aquatic humic substances. Science 1984, 223, 485–487. [Google Scholar] [CrossRef]
- Kraus, T.E.C.; Anderson, C.A.; Morgenstern, K.; Downing, B.D.; Pellerin, B.A.; Bergamaschi, B.A. Determining Sources of Dissolved Organic Carbon and Disinfection Byproduct Precursors to the McKenzie River, Oregon. J. Environ. Qual. 2010, 39, 2100–2112. [Google Scholar] [CrossRef] [PubMed]
- Hua, G.H.; Kim, J.; Reckhow, D.A. Disinfection byproduct formation from lignin precursors. Water Res. 2014, 63, 285–295. [Google Scholar] [CrossRef]
- Federation, W.E.; Association, A. Standard Methods for the Examination of Water and Wastewater; American Public Health Association (APHA): Washington, DC, USA, 2005. [Google Scholar]
- Fang, C.; Krasner, S.W.; Chu, W.; Ding, S.; Zhao, T.; Gao, N. Formation and speciation of chlorinated, brominated, and iodinated haloacetamides in chloraminated iodide-containing waters. Water Res. 2018, 145, 103–112. [Google Scholar] [CrossRef] [PubMed]
- Deborde, M.; von Gunten, U. Reactions of chlorine with inorganic and organic compounds during water treatment—Kinetics and mechanisms: A critical review. Water Res. 2008, 42, 13–51. [Google Scholar] [CrossRef]
- Westerhoff, P.; Chao, P.; Mash, H. Reactivity of natural organic matter with aqueous chlorine and bromine. Water Res. 2004, 38, 1502–1513. [Google Scholar] [CrossRef] [PubMed]
- Liu, C.; Ersan, M.S.; Plewa, M.J.; Amy, G.; Karanfil, T. Formation of regulated and unregulated disinfection byproducts during chlorination of algal organic matter extracted from freshwater and marine algae. Water Res. 2018, 142, 313–324. [Google Scholar] [CrossRef]
- Pourmoghaddas, H.; Stevens, A.A. Relationship between trihalomethanes and haloacetic acids with total organic halogen during chlorination. Water Res. 1995, 29, 2059–2062. [Google Scholar] [CrossRef]
- Hua, G.; Reckhow, D.A. Evaluation of bromine substitution factors of DBPs during chlorination and chloramination. Water Res. 2012, 46, 4208–4216. [Google Scholar] [CrossRef] [PubMed]
- Lengyel, I.; Li, J.; Kustin, K.; Epstein, I.R. Rate Constants for Reactions between Iodine- and Chlorine-Containing Species: A Detailed Mechanism of the Chlorine Dioxide/Chlorite-Iodide Reaction. J. Am. Chem. Soc. 1996, 118, 3708–3719. [Google Scholar] [CrossRef]
- Zhu, X.; Zhang, X. Modeling the formation of TOCl, TOBr and TOI during chlor(am)ination of drinking water. Water Res. 2016, 96, 166–176. [Google Scholar] [CrossRef]
- Liu, R.; Tian, C.; Hu, C.; Qi, Z.; Liu, H.; Qu, J. Effects of bromide on the formation and transformation of disinfection by-products during chlorination and chloramination. Sci. Total Environ. 2018, 625, 252–261. [Google Scholar] [CrossRef]
- Bichsel, Y.; von Gunten, U. Oxidation of Iodide and Hypoiodous Acid in the Disinfection of Natural Waters. Environ. Sci. Technol. 1999, 33, 4040–4045. [Google Scholar] [CrossRef]
- Zhang, T.-Y.; Xu, B.; Hu, C.-Y.; Lin, Y.-L.; Lin, L.; Ye, T.; Tian, F.-X. A comparison of iodinated trihalomethane formation from chlorine, chlorine dioxide and potassium permanganate oxidation processes. Water Res. 2015, 68, 394–403. [Google Scholar] [CrossRef] [PubMed]
- Postigo, C.; Richardson, S.D.; Barceló, D. Formation of iodo-trihalomethanes, iodo-haloacetic acids, and haloacetaldehydes during chlorination and chloramination of iodine containing waters in laboratory controlled reactions. J. Environ. Sci. 2017, 58, 127–134. [Google Scholar] [CrossRef] [PubMed]
- Liu, S.; Li, Z.; Dong, H.; Goodman, B.A.; Qiang, Z. Formation of iodo-trihalomethanes, iodo-acetic acids, and iodo-acetamides during chloramination of iodide-containing waters: Factors influencing formation and reaction pathways. J. Hazard. Mater. 2017, 321, 28–36. [Google Scholar] [CrossRef]

Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Feng, C.; Liu, T.; Shi, Y.; Shen, J.; Yuan, L.; Guo, Y. Formation of AOX and Cl/Br/I-THMs During NaClO Disinfection: A Comprehensive Study. Water 2025, 17, 2563. https://doi.org/10.3390/w17172563
Feng C, Liu T, Shi Y, Shen J, Yuan L, Guo Y. Formation of AOX and Cl/Br/I-THMs During NaClO Disinfection: A Comprehensive Study. Water. 2025; 17(17):2563. https://doi.org/10.3390/w17172563
Chicago/Turabian StyleFeng, Chao, Tongyu Liu, Yongping Shi, Jimin Shen, Lei Yuan, and Yuanqing Guo. 2025. "Formation of AOX and Cl/Br/I-THMs During NaClO Disinfection: A Comprehensive Study" Water 17, no. 17: 2563. https://doi.org/10.3390/w17172563
APA StyleFeng, C., Liu, T., Shi, Y., Shen, J., Yuan, L., & Guo, Y. (2025). Formation of AOX and Cl/Br/I-THMs During NaClO Disinfection: A Comprehensive Study. Water, 17(17), 2563. https://doi.org/10.3390/w17172563







