Abstract
The simultaneous occurrence of various pollutants in the aquatic environment raises questions about their mutual interactions. There is a gap in research on the sorption of polar substances on microplastics. This study aimed to assess the adsorption of the antidepressants sertraline, fluoxetine and duloxetine on microplastic polystyrene, polyethylene terephthalate and polyvinyl chloride, each in two versions: virgin and aged. To assess the affinity of the tested drugs for plastic and planktonic organisms, the experiment was conducted in microplastic suspensions and in a mixture of microplastics with the protozoan Spirostomum ambiguum. The Fourier transform infrared technique assessed the identity of microplastics and changes during ageing. No significant differences were found between the sorption of the tested drugs on virgin and aged microplastics. The sorption of sertraline onto microplastics was 1.5–3 times lower in the presence of the protozoa than in samples with microplastics alone. Moreover, its concentration in the protozoan cells was 10–30 times higher than in the microplastics. Considering that the amount of plankton in freshwaters is much greater than that of microplastics, it should be concluded that microplastics have a negligible share in the transport of antidepressants in surface waters.
1. Introduction
The development of synthetic drugs is one of the key achievements of civilisation. Drugs are widely used all over the world, and the annual consumption of the most popular ones reaches tens of thousands of tons [1]. After consumption, the unmetabolised and metabolised drugs are discharged to the aquatic environment via sewage water. Studies conducted since the beginning of the 21st century indicate the presence of numerous pharmaceuticals in rivers around the world [2,3]. Their presence in the environment can have a negative impact on various organisms, from bacteria to fish [4].
Depression affects a large part of societies around the world. This complication has become more severe during the COVID-19 pandemic, resulting in a significant increase in the use of antidepressant pharmaceuticals. Monoamine reuptake inhibitors have become a breakthrough in the treatment of depression and are currently one of the most frequently used groups of drugs in the U.S. and EU [1]. Sertraline (SER, (1S,4S)-4-(3,4-Dichlorophenyl)-N-methyl-1,2,3,4-tetrahydronaphthalen-1-amine) and fluoxetine (FLX, N-methyl-3-phenyl-3-[4-(trifluoromethyl)phenoxy]propan-1-amine) are serotonin reuptake inhibitors (SSRIs), while duloxetine (DLX, (+)-(S)-N-Methyl-3-(naphthalen-1-yloxy)-3-(thiophen-2-yl)propan-1-amine) is a serotonin-norepinephrine reuptake inhibitor (SNRI) (Figure 1). In 2021, SER, FLX and DLX were the 11th, 25th and 27th best-selling prescription drugs, respectively, accounting for one-third of all antidepressants in the U.S. [5]. The concentration of antidepressants in wastewater ranges from several ng to even one µg per litre [6,7,8,9]. At the time of the greatest popularity of Prozac, “The Happy Pill”, FLX was detected in most U.S. rivers [7]. Several studies have shown that FLX and SER affected aquatic organisms at concentrations as low as 14 and 0.47 µg L−1, respectively [7,10]. Our recent studies indicate that SER and DLX significantly influence the food intake of protozoan Spirostomum ambiguum [11]. Antidepressant pharmaceuticals bioconcentrate in aquatic organisms [9,12,13] and can also be transferred in the aquatic food chain [6]. FLX adsorbs to a significant extent on suspended particles in wastewater treatment plants [14]. SER had one of the highest adsorption rates on sewage sludge among the 21 drugs tested [15].
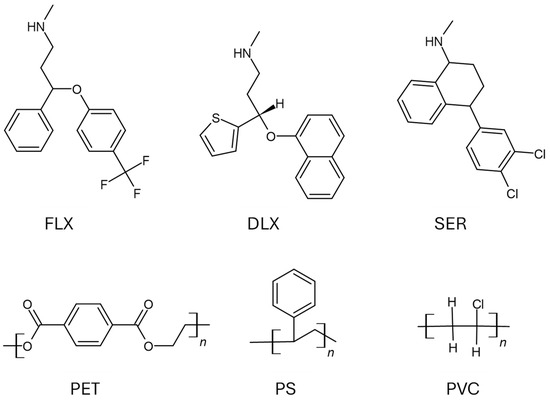
Figure 1.
Antidepressant pharmaceuticals and polymers used in the study.
The ability to absorb toxic substances is one of the most important challenges related to pollution of the aquatic environment by microplastics [16,17,18]. The absorption may increase the bioavailability of adsorbed xenobiotics and/or facilitate their transport in the aquatic environment [19]. The sorption of nonpolar compounds onto microplastics has been documented by many researchers [16,17]. The most frequently studied nonpolar compounds have been benzene derivatives, polycyclic aromatic hydrocarbons and polychlorinated biphenyls [16]. However, research on the sorption of polar compounds, including pharmaceuticals, has been conducted less commonly [16,20,21,22].
The adsorption process of organic substances onto microparticles is related to the physicochemical properties of the sorbate and sorbent as well as the properties of the medium. Concerning the sorbate properties, sorption is mainly associated with the polarity and charge of the molecule [23]. Considering the sorbent, the size of the surface depending on the particle diameter and the number of micropores is also vital. In a natural aquatic environment, in addition to pH and salinity, the presence of suspensions, including microplankton, should also be considered [18,20,24].
In natural waters, microplastics undergo ageing processes. UV irradiation, free radicals and dissolved oxygen may degrade polymer chains, which leads to the formation of microcracks and oxidation of the polymer surface functional groups [23,25,26,27]. As a result, functional groups, e.g., peroxide, carbonyl and carboxyl, are formed, which decreases the hydrophobicity of the polymer and, therefore, the ability to adsorb nonpolar substances. Hüffer et al. [23] observed a more than ten times lower naphthalene sorption on aged PS than on virgin PS. On the other hand, the adsorption of polar and/or ionised compounds increases [28].
In this study, sorption experiments of three antidepressant pharmaceuticals onto three types of microplastics, polystyrene (PS), polyethylene terephthalate (PET) and polyvinyl chloride (PVC), each in two versions, virgin and aged, were performed. The aging of microplastics by UV irradiation and hydrogen peroxide oxidation led to a change in the surface chemistry of particles, measured by Fourier transform infrared spectroscopy. As the antidepressants analysed in this study SER, FLX and DLX have pKa > 9.5; they exist mainly in the cation form in freshwaters (pH between 6 and 8.5). Therefore, we hypothesised that their adsorption onto more polar surfaces of aged plastics would be greater than on nonpolar virgin ones.
To assess the affinity of the tested drugs for plastic and planktonic organisms, the experiment was conducted in microplastic suspensions and a mixture of microplastics with the protozoan S. ambiguum. The concentration of SER, FLX and DLX was 2–3 orders of magnitude higher than the measured environmental concentration to ensure an appropriate balance between the equipment capabilities and real, environmental conditions.
2. Materials and Methods
2.1. Reagents
High purity grade (>90%) standard reagents of SER, DLX, FLX, deuterated sertraline hydrochloride, duloxetine hydrochloride and fluoxetine hydrochloride were obtained from TRC (Toronto, ON, Canada). The standard stock solutions were prepared in 50% methanol at concentrations of 1 mg mL−1 and stored at −20 °C. Working solutions were prepared just before the tests by diluting the stock solution with the culture medium. An internal standard working solution (500 ng mL−1) was prepared by diluting of the deuterated compounds stock solution with acetonitrile. The solvents, namely HPLC–MS grade acetonitrile (LiChrosolv) and formic acid 98%, were provided by Merck (Darmstadt, Germany). Ultrapure water was obtained from a Millipore water purification system (Milli-Q water). Three types of virgin microplastics (vMPs), polystyrene (PS), polyethylene terephthalate (PET) and polyvinyl chloride (PVC), were prepared by grinding in liquid nitrogen using a cryomill, type 6770, SPEX (Metuchen, NJ, USA), according to Chojnacka et al. [11].
2.2. Aging of Microplastics
An amount of 100 mg of MPs was placed in a 40 mL glass Petri dish, and 10 mL of 30% hydrogen peroxide was added. The dish was covered with a quartz lid and placed in a SunTest CPS+ sunlight simulator (Klimatest, Warsaw, Poland) for two hours. Aged MPs (aMPs) were rinsed with deionised water and dried at 40 °C.
2.3. Accumulation Tests
The experiments were carried out in 150 mL glass beakers filled with 100 mL of sample. An inorganic medium (Tyrode’s solution) was used as the diluent, with the pH adjusted to 7.4 [29]. The accumulation experiments were performed for antidepressant pharmaceuticals, each at two concentrations, 10 µg L−1 (low level, LL) and 100 µg L−1 (high level, HL). We added 100 protozoan cells and/or 10 mg of microplastics to each beaker, and the beakers were incubated for 6 days (144 h) at 25 °C in darkness. The arrangement of the tested samples is presented in Table 1.

Table 1.
Abbreviations used in the accumulation test with microplastics and the protozoan S. ambiguum.
The accumulation test with SER was performed on all kinds of MPs: PET, PS and PVC. Meanwhile, the accumulation tests with DLX and FLX were only performed with PS. These tests were performed in triplicate.
2.4. Determination of Antidepressant Concentrations in MPs and S. ambiguum with LC-MS
To determine antidepressant concentrations in S. ambiguum, all protozoan cells were sampled from each test beaker at the end of incubation and placed into a 1.5 mL Eppendorf tube. The volume of protozoans and the remaining medium was 50 µL. To determine the antidepressants’ concentration sorbed on the MPs, the MPs were separated from the medium by filtering the sample with 2 µm nylon membrane filters and transferred into 1.5 mL Eppendorf tubes. Then, 100 µL of acetonitrile with an internal standard (IS, 50 ng mL−1 deuterated standard in acetonitrile) was added to the tubes. The samples were vortexed for 10 min, placed in a freezer (at −20 °C) for 10 min and then centrifuged (5 min at 10,000× g). The supernatant (80–100 µL) was transferred to the autosampler vial. No cleanup procedure was used. The concentration of pharmaceuticals in organisms was calculated using the measured concentration of the pharmaceutical in the extract and the volume of S. ambiguum. The average volume of 100 cells of S. ambiguum was 0.50 µL [13].
Quantitative analyses were performed using Agilent 1260 Infinity (Agilent Technologies, Santa Clara, CA, USA), equipped with a degasser, a thermostated autosampler and a binary pump and connected in series to a QTRAP®4000 (AB SCIEX, Framingham, MA, USA) equipped with a turbo ion spray source operated in the positive mode according to method described and validated previously [13].
2.5. Assessment of Physicochemical Properties of Virgin and Aged MPs
To analyse the physicochemical properties of virgin and aged MPs, mid-infrared spectroscopy (FT-IR) was performed using two techniques: (1) Attenuated total reflectance (ATR) technique using an IRAffinity-1 (Shimadzu, Kyoto, Japan) spectrometer working at the mid-infrared range with a Quest diamond crystal attachment (Specac Ltd., London, UK). The conditions were as follows: 50 repetitions and a resolution of 2 cm−1. The spectra were recorded from 400 to 3600 cm−1 in triplicate repetitions. (2) Potassium bromide pellet (KBr) technique using a Spectrum 1000 (Perkin Elmer, Cleveland, OH, USA) spectrometer working at the mid-infrared range. The conditions were as follows: 50 repetitions and resolution of 2 cm−1. The obtained samples were mixed with KBr in a weight ratio of 1:100 and pressed into pellets under a pressure of 9 tons. The spectra were recorded from 400 to 3600 cm−1 in triplicate repetitions.
FT-IR spectra obtained by both methods were processed using the GRAMS/AI 8.0 software (Thermo Scientific, Burlington, ON, USA, 2006). The processing included baseline correction, measurement of surface area under the band in the ranges from 1650 to 1850 cm−1 and 1420 to 1500 cm−1 and smoothing the lines only to present graphs. Combining these two research techniques in mid-infrared spectroscopy allows for the analysis of the surface and interior of the analysed samples. The ATR technique allows for the sample surface analysis adjacent to the diamond crystal, while the KBr pellets technique shows the entire volume of the MPs sample. Research shows that the penetration depth of ATR allows for examination no deeper than 2 µm [30].
The carbonyl index (CI) was used to examine the ageing of plastics, showing the increase in the band from carbonyl stretching (C=O), which is responsible for the oxidation process. CI was measured by dividing the area of the carbonyl stretching band (C=0) in the range from 1650 to 1850 cm−1 by the reference band (CH2) from 1420 to 1500 cm−1 as in the Equation (1) [30,31]:
2.6. Statistical Analysis
Statistical analysis was conducted using Analysis ToolPak in Microsoft Excel 365. To compare the relationship between the results, a paired Student’s t-test was used. For all tests, the significance threshold was set at p < 0.05.
3. Results
3.1. Physicochemical Properties of Virgin and Aged Microplastics
The physicochemical studies carried out using mid-infrared spectroscopy using the ATR and KBr pellet techniques are shown in Figure 2. These spectra confirm that the environmentally derived plastics that were ground are indeed PET, PS and PVC.
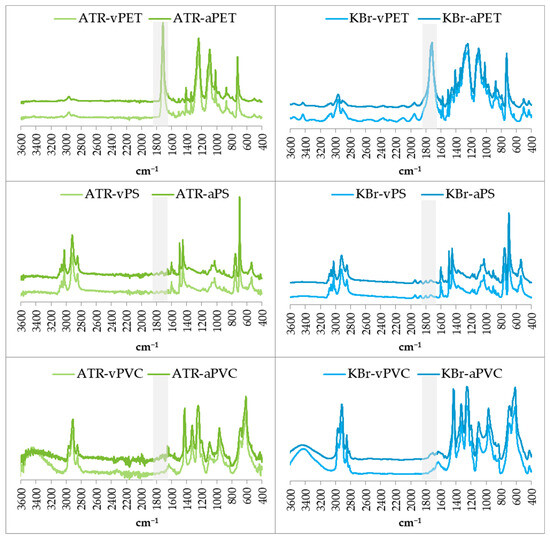
Figure 2.
Attenuated total reflectance Fourier transform infrared (ATR FT-IR) and potassium bromide pellet (KBr) spectra of virgin and aged MPs. The presented spectra on the left for the ATR technique and on the right for the KBr technique confirm the acquisition from the PET, PS and PVC environment, confirmed by the characteristic bands visible in the spectra. The carbonyl functional group band range used to calculate the carbonyl index (1650–1850 cm−1) has been highlighted.
For the PET sample analysed by both techniques, the spectra have the strongest C=O stretching band at about 1712 cm−1 and two strong C–O stretching bands at 1241 cm−1 and 1094 cm−1. At 720 cm−1, an aromatic C–H out-of-plane beading was observed [30,32].
In the spectra obtained for PS, we see the bends 3024 cm−1, 2847 cm−1, 1601 cm−1, 1492 cm−1, 1451 cm−1, 1027 cm−1, 694 cm−1 and 537 cm−1, respectively, originating from aromatic C–H stretching, C–H stretching, aromatic ring stretching, aromatic ring stretching, C–H2 bending, aromatic C–H bending, aromatic C–H out-of-plane bending and aromatic ring out-of-plane bending [30].
For the PVC sample, characteristic bends described in the literature were also observed at 1427 cm−1, 1331 cm−1, 1255 cm−1, 1099 cm−1, 966 cm−1 and 616 cm−1, respectively, originating from C–H2 bending, C–H bending, C–H bending, C–C stretching, C–H2 rocking and C–Cl stretching [30,33], respectively.
Mid-infrared spectroscopy studies using two techniques also allow for the observation of the formation of the carbonyl group during ageing [25,26,32]. The region of carbonyl group occurrence is highlighted in the spectra presented in Figure 2.
Following earlier reports, the presented spectra show a slight increase in the C=O stretching band at about 1712 cm−1 in the marked region. This change is most visible for the ATR-vPET and ATR-aPET pairs and for the vPVC and aPVC samples tested with both techniques. Previously, the increase in this band was observed under the influence of different ageing techniques for plastics such as PET, PS and PVC [30,31,32,33].
For PS samples, we can also observe a very small band originating from the C=O group at 1725 cm−1, previously reported for photo-oxidised PS [34].
Figure 3 shows the carbonyl index, which presents the actual change in the carbonyl band relative to the reference band. An increase in CI calculated from the spectra obtained using the ATR and KBr techniques is visible for all MPs. In comparing ATR and KBr techniques, the increase in CI was higher for ATR, which may be because this method only analyses surfaces up to 2 µm. A significant difference between the aged and virgin samples (p < 0.05) was observed for the PET samples tested by both techniques and for the PVC sample tested by the KBr technique. These results confirm previous observations made for the presented FT-IR spectrum in the highlighted range and are consistent with the changes described in the literature following ageing [30,31,32,33]. The greater significant difference for the KBr technique is due to its much greater repeatability.
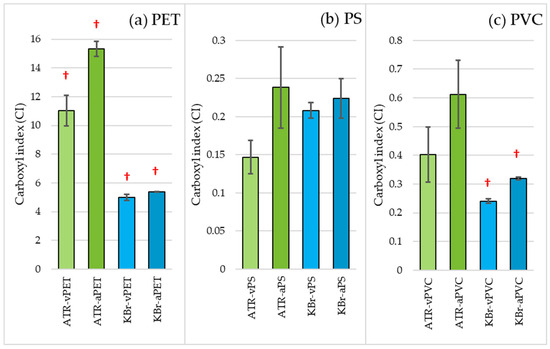
Figure 3.
Carbonyl index (CI) of virgin and aged MPs obtained by ATR and KBr pellet FT-IR techniques for (a) PET, (b) PS and (c) PVC. The CI was calculated using Equation (1). †—Significant difference between the aged and virgin samples (p < 0.05).
3.2. Sorption and Bioaccumulation of Sertraline
SER was incubated with three microplastics: virgin vPET, vPS, vPVC and their aged counterparts aPET, aPS and aPVC in the presence and absence of the protozoan S. ambiguum. Tyrode’s medium was used in the experiment to simulate river-like conditions, with a pH range of 6.5–8.0, low hardness and salinity. It contains the minimal amount of inorganic ions necessary for the proper functioning of protozoa [29] and was not buffered. The pH remained stable at 7.4 throughout the experiment. After six days of incubation, the concentration of SER adsorbed on MPs and in protozoan cells was determined. The results are shown in Figure 4.
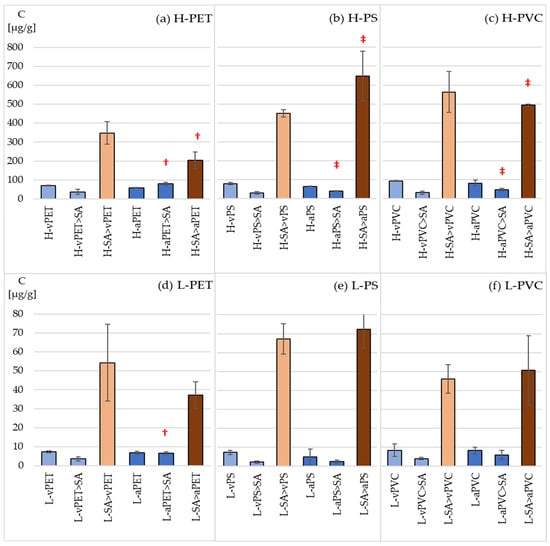
Figure 4.
The concentration of SER sorbed on virgin and aged MPs and in the protozoan S. ambiguum. (a,d) PET, high (100 µg L−1) and low (10 µg L−1) levels of SER; (b,e) PS, high (100 µg L−1) and low (10 µg L−1) levels of SER; (c,f) PVC, high (100 µg L−1) and low (10 µg L−1) levels of SER. Descriptions according to Table 1. †—Significant difference between the aged and virgin samples (p < 0.05); ‡—significant difference between this sample and its corresponding PET sample (p < 0.05).
Because the tests were carried out on low concentrations of drugs, their concentrations in water were not determined. Therefore, sorption coefficients on microplastics and bioconcentration factors in protozoa were not calculated. The SER adsorbed on microplastic particles was 200–900 times greater than its initial solution concentration. No significant differences were observed between sorption on virgin and aged microplastics. The amount of SER sorbed onto microplastics was 1.5–3 times lower in samples containing both microplastics and protozoa than in samples with only microplastics. The only exception was aPET.
The protozoa accumulated SER to a much greater extent than microplastics did. The nominal bioconcentration factor (the ratio of the concentration of a compound in organisms to the initial/nominal concentration in water) exceeded 2000 in all cases. Thus, according to a European Union regulation, Registration, Evaluation, Authorisation and Restriction of Chemicals (REACH), SER is considered bioaccumulative [35]. In each sample tested, the concentration of SER was 10 to even 30 times higher in the protozoa than in the MP suspension.
3.3. Sorption and Bioaccumulation of Fluoxetine and Duloxetine
In the case of DLX and FLX, the sorption analyses were performed only with PS due to the similar sorption of SER onto all three tested microplastics. Both vPS and aPS absorb FLX and DLX much less than SER. Their concentrations in PS samples are 3–5 and 4–9 times lower than in the case of SER, respectively (Figure 5, Tables S2 and S3). Similarly to SER in the case of FLX, no significant differences in sorption on virgin and aged microplastics were found for both drug concentration levels. However, in the higher level of DLX, the drug sorption on aged microplastics was twice as high, both in the case of aPS suspension and aPS with protozoa. In the latter case, the bioaccumulation of DLX in protozoa was two times lower than for vPS, which may indicate competition between sorption on microplastics and bioaccumulation in protozoa.
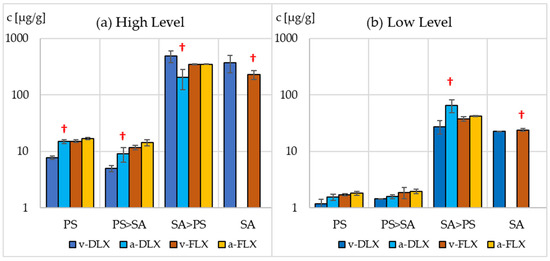
Figure 5.
The concentration of DLX and FLX sorbed on virgin (v-DLX, v-FLX)) and aged (a-DLX, a-FLX) microplastic (PS), microplastic incubated with protozoa (PS > SA), in protozoa S. ambiguum (SA) and in protozoa incubated with microplastic (SA > PS). (a) High level of pharmaceuticals, 100 µg L−1; (b) low level of pharmaceuticals, 10 µg L−1. †—Significantly different compared with the virgin PS samples (p < 0.05).
4. Discussion
4.1. Physicochemical Properties of Virgin and Aged Microplastics
Plastics are considered to be very durable materials in the environment. Their decomposition may take up to several hundred years. Environmental factors, including UV radiation, free radicals and oxidising compounds, may cause the ageing of materials. Ageing processes include physical degradation of the material, e.g., the formation of microcracks, and chemical changes in the polymers, e.g., molecular weight reduction and modification in surface functional groups [36]. In the accelerated ageing process caused by H2O2 and UV radiation, oxygen radicals are formed, which oxidise functional groups to peroxides [23]. The photooxidation processes are faster than other degradation processes and may be prevented by some additives of the plastics [36]. Oxidation of polymer functional groups may lead to the formation of peroxides and aldehyde and carboxyl groups [25,26,28]. In the research, the oxidation of the tested microplastics was confirmed using the FTIR technique. A higher increase in carbonyl index was received in the ATR method, indicating the formation of the C=O bonds on the surface of the materials. Less pronounced changes in the carbonyl index were obtained for the KBr method, which implies smaller changes in the chemical structure inside the material. Combining these two research methods allows for a more comprehensive analysis of the photooxidation process. ATR spectra similar to ours were obtained after PS ageing using H2O2 and UV irradiation [23].
The oxidation of polymer functional groups may increase the surface’s hydrophilicity, influencing the sorption behaviour of polar and/or ionised xenobiotics.
4.2. Sorption and Bioaccumulation of Pharmaceuticals
The sorption of only a few pharmaceuticals on microplastics has been studied so far [21,22,37,38]. The sorption of pharmaceuticals was studied on microplastics, which are environmental pollutants [37,38], and polymers fabricated to remove micropollutants from the environment [1]. The adsorption of an analyte on a sorbent depends on many factors, including their hydrophobic properties and charges, as well as surface quality, microcracks, etc. Analyte/Sorbent interactions largely depend on the degree of ionisation of the compound and the charge on the sorbent surface. Also, the medium’s pH and pH of zero point (pHpzc), corresponding to the pH at which the negative and positive charges on the sorbent’s surface are equal, are essential factors in determining adsorption [39]. The pHpzc is a crucial parameter and has been extensively used to study metal adsorption on nanoparticles. At pH > pHpzc, the sorbent surface is negatively charged, enhancing electrostatic attraction between cations and the sorbent [39,40]. However, the pHpzc parameter has not yet been considered in studies of pharmaceutical sorption on microplastics. This is likely due to the low charge density on the surface of unmodified polymers, which complicates the analysis of pHpzc values. In contrast, this parameter should be taken into account for aged polymers with a significant number of carboxyl groups. The analysis of polar substance sorption on polyethylene indicates a strong dependence of sorption efficiency on the medium’s pH and the compound’s acidity [41]. Triclosan was adsorbed at 70% at pH = 4; at pH = 10, where it was mostly present in its ionic form, no adsorption occurred [41]. Analysing the dependence of sorption on water pH is crucial when searching for sorbents to remove the maximum number of micropollutants from water [39]. However, this is of lesser importance in analysing environmental samples as the pH of most European rivers ranges from 7.0 to 8.5 [42]. Therefore, in this study, we conducted the research at only one pH value: 7.4. A number of studies also indicate the important contribution of other interactions, including hydrogen binding, van der Waals forces, and π–π and ion–dipole interactions [1,43]. In the present work, the mechanism of the adsorption of antidepressants to polymers was not investigated. However, all types of interactions can occur due to the presence of ionic, hydrophobic and aromatic rings in drug molecules (Figure 1) [1].
In this study, we found that the antidepressants SER, FLX and DLX sorbed onto the microplastics, and the sorption of SER was stronger than in the case of the other two drugs. The adsorption of SER on polyethylene (PE) was also the highest among the drugs tested by Razanajatovo et al. [38], and at an initial concentration of SER in the solution of 60 µg L−1, the maximum sorbed concentration was 88.8 µg g−1. Our studies obtained a similar sorbed concentration at an initial SER concentration of 100 µg L−1. This result indicates a similar sorption capacity of PET, PS and PVC compared with PE. As these polymers differ significantly in their chemical structure, the results suggest the lack of specific molecular interactions between SER and polymers. Razanajatovo et al. [38] explained these results by strong electrostatic interactions between positively charged drug molecules and the negatively charged PE surface. They showed a relationship between the pKa values of drugs (the charge of molecules at neutral pH) and their affinity to microplastics. Moura et al. [31] demonstrated the high potential of virgin and aged microplastics for drug adsorption in aqueous environments. Of the five drugs tested, FLX showed the greatest ability to adsorb onto microplastics which, according to the authors, is due to its higher hydrophobicity and positive molecule charge at pH = 7. Zwitterions have the strongest nonpolar interaction at their isoelectric points [20]. The tested antidepressants have a pKa > 9.5. Therefore, they always occur as positive ions in natural waters with a pH ranging between 6 and 8.5. Most microplastic surfaces have a net negative charge at the neutral pH of water [24]. Therefore, the positively charged molecules interact with microplastic surfaces through electrostatic attraction [20]. Microplastics with ionisable groups, e.g., PVC, should interact with cations more strongly than nonpolar polymers, e.g., PS, because they can induce dipole–dipole interactions. On the other hand, polymers made of aromatic monomers, e.g., PS and PET, can attract aromatic compounds due to π–π interactions [20]. In the current study, we found no differences between the adsorption of SER onto different microplastics: PET, PS and PVC. Similar sorption capacity onto both PS and PVC was received for antibiotic ciprofloxacin [37]. On the other hand, different sorption onto four microplastics was determined for the macrolide antibiotic tylosin [44]. In the latter case, in addition to electrostatic interactions, surface complexation and hydrophobic interactions could be responsible for the interaction of tylosin with microplastics.
Several authors have observed an increase in adsorption capacity due to the ageing of microplastics [20,31,37]. In this work, the artificial ageing of materials using H2O2 and UV irradiation was used to investigate the effect of ageing on the sorption properties of microplastics. The ageing process resulted in the formation of carbonyl groups on the surface of the microparticles, which was confirmed by the APR-FTIR technique. Microplastic ageing did not increase drug sorption. The only exception was the HL–DLX–PS sample, where twice as much drug was adsorbed onto aged PS. FLX sorption on virgin and aged microplastics depends on the type of polymer [31]. They observed higher FLX sorption on aged PS and PE compared with virgin polymers, while no differences were noted for PVC, polyamide and polypropylene. In studies of ciprofloxacin sorption on PS, a significant difference was observed in the sorption behaviour of this zwitterionic antibiotic, depending on the pH [37]. At pH = 5, sorption was substantially higher on virgin PS compared with aged PS, while at pH = 9, the trend was reversed [37]. However, at a neutral pH of 7, the differences in sorption between aged and virgin plastics were much smaller. In the case of PVC, such variations were not observed. According to Liu et al. [37], the primary mechanisms of ciprofloxacin adsorption on PS and PVC are electrostatic interactions and, for aged microplastics, hydrogen bonding. On the other hand, UV-induced surface functionalisation decreased sorption coefficients for aged PS and 21 substances, including nonpolar and polar aliphatics, as well as nonpolar and polar aromatics [23].
Most natural surface waters, in addition to dissolved substances, contain suspensions of organic material and phyto- and zooplankton. According to Baker and Kasprzyk-Hordern [14], more than 50% of FLX in wastewater is associated with suspended particles. FLX adsorbs to a significant extent on suspended particles in wastewater treatment plants [10]. SER had one of the highest adsorption rates on sewage sludge among the 21 drugs tested, and the adsorption process occurred very quickly. After 5 h, the SER concentration in the aqueous phase dropped to only several percent of the initial level [15]. SER, its metabolite norsertraline and norfluoxetine showed the highest bioaccumulation rate in fish tissues out of 22 pharmaceuticals observed in the Niagara River [6]. Similar results were presented by Grabicova et al. [45], who detected high concentrations of SER in brown trout tissue in effluent polluted rivers, and the bioaccumulation factor was as high as 4400 and 2400 in the kidney and liver, respectively. An equally high bioconcentration factor was obtained in a field for benthic invertebrates [46]. High bioconcentration factors of SER and FLX were also obtained in laboratory experiments on the invertebrate Asellus aquaticus and fish Pungitius pungitius [16].
4.3. Ecological Consequences
The presence of a mixture of xenobiotics in the aquatic environment raises questions about their mutual interactions. One of the most important problems related to environmental pollution with microplastics is their sorption behaviour towards substances dissolved in water. However, most researchers analysing this problem use pure suspensions of microplastics without paying attention to the surface waters’ natural biotic and abiotic components.
In our current research, we obtained several to several dozen times higher concentrations of the tested antidepressants in the cells of the protozoan S. ambiguum compared with plastic microparticles. Additionally, sorption onto microplastics was lower in the presence of protozoa than in samples with microplastics alone. This means that the affinity of these drugs for microplastics is lower than for natural, organic matter. Therefore, to obtain real results for the xenobiotic sorption on microplastic suspensions, comparative studies should be performed concerning sorption on natural suspensions and/or organisms.
The concentration of microplastics, even in the most polluted surface waters, ranges from a few to several hundred particles per litre [47]. However, plankton density in European waters is several orders of magnitude higher [48,49]. It should, therefore, be concluded that both virgin and aged microplastics have a negligible share in the transport of antidepressants in surface waters.
5. Conclusions
Antidepressants were adsorbed equally onto virgin and aged microplastics, with sertraline being the most strongly adsorbed.
To assess the affinity of the tested pharmaceuticals for plastic and planktonic organisms, the experiment was conducted in microplastic suspensions and in a mixture of microplastics with the protozoan S. ambiguum. The sorption of sertraline onto microplastics was 1.5–3 lower in the presence of the protozoa than in samples with the microplastics alone. Moreover, its concentration in the protozoan cells was 10–30 times higher than the concentration sorbed onto the microplastics.
Considering that the amount of plankton in freshwaters is much greater than that of microplastics, it should be concluded that microplastics have a negligible share in the transport of antidepressants in surface waters.
Supplementary Materials
The following supporting information can be downloaded at: https://www.mdpi.com/article/10.3390/w16172514/s1, Table S1: The concentration of SER sorbed on virgin and aged MPs and in protozoan S. ambiguum; Table S2: The concentration of FLX sorbed on virgin and aged PSs and in protozoan S. ambiguum.; Table S3: The concentration of DLX sorbed on virgin and aged PSs and in protozoan S. ambiguum.
Author Contributions
Conceptualisation, G.N.-J., J.G., Ł.P. and A.D.; methodology, G.N.-J., J.G., J.C., Ł.P. and A.D.; software, G.N.-J., J.G. and Ł.P.; validation, G.N.-J., J.G., Ł.P. and A.D.; formal analysis, G.N.-J., J.G. and Ł.P.; investigation, G.N.-J., J.G., J.C., Ł.P. and A.D.; resources, G.N.-J., J.G., Ł.P. and A.D.; data curation, G.N.-J., Ł.P. and A.D.; writing—original draft preparation, G.N.-J., J.G., Ł.P. and A.D.; writing—G.N.-J., J.G., Ł.P. and A.D.; visualisation, G.N.-J., Ł.P. and A.D.; supervision, G.N.-J. and A.D.; project administration, G.N.-J.; funding acquisition, G.N.-J. All authors have read and agreed to the published version of the manuscript.
Funding
This research was funded by the National Science Centre in Poland (grant number: 2019/35/B/NZ8/01388).
Data Availability Statement
Data are contained within the article.
Conflicts of Interest
The authors declare no conflicts of interest. The funders had no role in the design of this study; in the collection, analyses, or interpretation of data; in the writing of the manuscript; or in the decision to publish the results.
References
- Khan, S.A.; Abbasi, N.; Hussain, D.; Khan, T.A. Sustainable mitigation of paracetamol with a novel dual-functionalised pullulan/kaolin hydrogel nanocomposite from simulated wastewater. Langmuir 2022, 38, 8280–8295. [Google Scholar] [CrossRef] [PubMed]
- Boyd, G.R.; Reemtsma, H.; Grimm, D.A.; Mitra, S. Pharmaceuticals and personal care products (PPCPs) in surface and treated waters of Louisiana, USA and Ontario, Canada. Sci. Total Environ. 2003, 311, 135–149. [Google Scholar] [CrossRef]
- Mole, R.A.; Brooks, B.W. Global scanning of selective serotonin reuptake inhibitors: Occurrence, wastewater treatment and hazards in aquatic systems. Environ. Pollut. 2019, 250, 1019–1031. [Google Scholar] [CrossRef] [PubMed]
- Santos, L.H.M.L.M.; Araújo, A.N.; Fachini, A.; Pena, A.; Delerue-Matos, C.; Montenegro, M.C.B.S.M. Ecotoxicological aspects related to the presence of pharmaceuticals in the aquatic environment. J. Hazard. Mat. 2010, 175, 45–95. [Google Scholar] [CrossRef] [PubMed]
- ClinCalc DrugStats Database. Available online: https://clincalc.com (accessed on 1 June 2024).
- Arnnok, P.; Singh, R.R.; Burakham, R.; Perez-Fuentetaja, A.; Aga, D.S. Selective uptake and bioaccumulation of antidepressants in fish from effluent-impacted Niagara River. Environ. Sci. Technol. 2017, 51, 10652–10662. [Google Scholar] [CrossRef] [PubMed]
- Brooks, B.W.; Turner, P.K.; Stanley, J.K.; Weston, J.J.; Glidewell, E.A.; Foran, C.M.; Slattery, M.; La Point, T.W.; Huggett, D.B. Waterborne and sediment toxicity of fluoxetine to select organisms. Chemosphere 2003, 52, 135–142. [Google Scholar] [CrossRef]
- Giebułtowicz, J.; Nałęcz-Jawecki, G. Occurrence of antidepressant residues in the sewage-impacted Vistula and Utrata rivers and in tap water in Warsaw (Poland). Ecotoxicol. Environ. Saf. 2014, 104, 103–109. [Google Scholar] [CrossRef]
- Bojanowska-Czajka, A.; Pyszyńska, M.; Majkowska-Pilip, A.; Wawrowicz, K. Degradation of selected antidepressants sertraline and citalopram in ultrapure water and surface water using gamma radiation. Processes 2022, 10, 63. [Google Scholar] [CrossRef]
- Figuiere, R.; Waara, S.; Ahrens, L.; Golovko, O. Risk-based screening for prioritisation of organic micropollutants in Swedish freshwater. J. Hazard. Mat. 2022, 429, 128302. [Google Scholar] [CrossRef]
- Chojnacka, J.; Drobniewska, A.; Lenga, W.; Misztal, J.; Wawryniuk, M.; Nałęcz-Jawecki, G. The mutual effect of microparticles and antidepressants on the protozoan Spirostomum ambiguum (Müller, 1786) Ehrenberg, 1835. Water 2023, 15, 552. [Google Scholar] [CrossRef]
- Boström, M.L.; Ugge, G.; Jonsson, J.A.; Berglund, O. Bioaccumulation and trophodynamics of the antidepressants sertraline and fluoxetine in laboratory-constructed, 3-level aquatic food chains. Environ. Toxicol. Chem. 2017, 36, 1029–1037. [Google Scholar] [CrossRef] [PubMed]
- Nałęcz-Jawecki, G.; Wawryniuk, M.; Giebułtowicz, J.; Olkowski, A.; Drobniewska, A. Influence of selected antidepressants on the ciliated protozoan Spirostomum ambiguum: Toxicity, bioaccumulation, and biotransformation products. Molecules 2020, 25, 1476. [Google Scholar] [CrossRef] [PubMed]
- Baker, D.R.; Kasprzyk-Hordern, B. Multi-residue determination of the sorption of illicit drugs and pharmaceuticals to wastewater suspended particulate matter using pressurised liquid extraction, solid phase extraction and liquid chromatography coupled with tandem mass spectrometry. J. Chromatogr. A 2011, 1218, 7901–7913. [Google Scholar] [CrossRef] [PubMed]
- Burzio, C.; Mohammadi, A.S.; Smith, S.; Abadikhah, M.; Svahn, O.; Modin, O.; Persson, F.; Wilen, B.-M. Sorption of pharmaceuticals to foam and aerobic granular sludge with different morphologies. Resour. Environ. Sustain. 2024, 15, 100149. [Google Scholar] [CrossRef]
- Costigan, E.; Collins, A.; Hatinoglu, M.D.; Bhagat, K.; MacRae, J.; Perreault, F.; Apul, O. Adsorption of organic pollutants by microplastics: Overview of a dissonant literature. J. Hazard. Mat. Adv. 2022, 6, 100091. [Google Scholar] [CrossRef]
- Guo, X.; Wang, X.; Zhou, X.; Kong, X.; Tao, S.; Xing, B. Sorption of four hydrophobic organic compounds by three chemically distinct polymers: Role of chemical and physical composition. Environ. Sci. Technol. 2012, 46, 7252–7259. [Google Scholar] [CrossRef]
- Wagstaff, A.; Petrie, B. Enhanced desorption of fluoxetine from polyethylene terephthalate microplastics in gastric fluid and sea water. Environ. Chem. Lett. 2022, 20, 975–982. [Google Scholar] [CrossRef]
- Athey, S.N.; Albotra, A.D.; Gordon, C.A.; Monteleone, B.; Seaton, P.; Andrady, A.L.; Taylor, A.R.; Brander, S.M. Trophic transfer of microplastics in an estuarine food chain and the effects of a sorbed legacy pollutant. Limnol. Oceanol. Lett. 2020, 5, 154–162. [Google Scholar] [CrossRef]
- Atugoda, T.; Vithanage, M.; Wijesekara, H.; Bolan, N.; Sarmah, A.K.; Bank, M.S.; You, S.; Ok, Y.S. Interactions between microplastics, pharmaceuticals and personal care products: Implications for vector transport. Environ. Int. 2021, 149, 106367. [Google Scholar] [CrossRef]
- Li, J.; Zhang, K.; Zhang, H. Adsorption of antibiotics on microplastics. Environ. Pollut. 2018, 237, 460–467. [Google Scholar] [CrossRef]
- Elizalde- Velázquez, A.; Subbiah, S.; Anderson, T.A.; Green, M.J.; Zhao, X.; Cañas-Carrell, J.E. Sorption of three common nonsteroidal anti-inflammatory drugs (NSAIDs) to microplastics. Sci. Total Environ. 2020, 715, 136974. [Google Scholar] [CrossRef] [PubMed]
- Hüffer, T.; Weniger, A.-K.; Hofmann, T. Sorption of organic compounds by aged polystyrene microplastic particles. Environ. Pollut. 2018, 236, 218–225. [Google Scholar] [CrossRef] [PubMed]
- Tourinho, P.S.; Koci, V.; Loureiro, S.; van Gestel, C.A.M. Partitioning of chemical contaminants to microplastics: Sorption mechanisms, environmental distribution and effects on toxicity and bioaccumulation. Environ. Pollut. 2019, 252, 1246–1256. [Google Scholar] [CrossRef] [PubMed]
- Almond, J.; Sugumaar, P.; Wenzel, M.N.; Hill, G.; Wallis, C. Determination of the carbonyl index of polyethylene and polypropylene using specified area under band methodology with ATR-FTIR spectroscopy. e-Polymers 2020, 20, 369–381. [Google Scholar] [CrossRef]
- Zvekic, M.; Richards, L.C.; Tong, C.C.; Krogh, E.T. Characterizing photochemical ageing processes of microplastic materials using multivariate analysis of infrared spectra. Environ. Sci. Process. Impacts 2022, 24, 52. [Google Scholar] [CrossRef]
- Zhu, Z.; Cao, X.; Wang, K.; Guan, Y.; Ma, Y.; Li, Z.; Guan, J. The environmental effects of microplastics and microplastic derived dissolved organic matter in aquatic environments: A review. Sci. Total Environ. 2024, 933, 173163. [Google Scholar] [CrossRef]
- Bhagat, K.; Barrios, A.C.; Rajwade, K.; Kumar, A.; Oswald, J.; Apul, O.; Perreault, F. Aging of microplastics increases their adsorption affinity towards organic contaminants. Chemosphere 2022, 298, 134238. [Google Scholar] [CrossRef]
- Nałęcz-Jawecki, G. Spirotox test—Spirostomum ambiguum acute toxicity test. In Small-Scale Freshwater Toxicity Investigations; Blaise, C., Férard, J., Eds.; Springer: Berlin/Heidelberg, Germany, 2005; Volume 1, pp. 299–322. [Google Scholar]
- Veerasingam, S.; Ranjani, M.; Venkatachalapathy, R.; Bagaev, A.; Mukhanov, V.; Litvinyuk, D.; Mugilarasan, M.; Gurumoorthi, K.; Guganathan, L.; Aboobacker, V.M.; et al. Contributions of Fourier transform infrared spectroscopy in microplastic pollution research: A review. Crit. Rev. Environ. Sci. Technol. 2021, 51, 2681–2743. [Google Scholar] [CrossRef]
- Moura, D.S.; Pestana, C.J.; Moffat, C.F.; Gkoulemani, N.; Hui, J.; Irvine, J.T.S.; Lawton, L.A. Aging microplastics enhances the adsorption of pharmaceuticals in freshwater. Sci. Tot. Environ. 2024, 912, 169467. [Google Scholar] [CrossRef]
- Korolkov, I.; Mashentseva, A.; Güven, O.; Niyazova, D.; Barsbay, M.; Zdorovets, M. The effect of oxidising agents/systems on the properties of track-etched PET membranes. Polym. Degrad. Stab. 2014, 107, 150–157. [Google Scholar] [CrossRef]
- Shi, W.; Zhang, J.; Shi, X.; Jiang, G. Different photodegradation processes of PVC with different average degrees of polymerisation. J. Appl. Polym. Sci. 2006, 107, 528–540. [Google Scholar] [CrossRef]
- Lucas, P.C.; Porter, R.S. Carbonyl functional groups in photo-oxidised polystyrene. Polym. Degrad. Stab. 1985, 13, 287–295. [Google Scholar] [CrossRef]
- Regulation (EC) No 1907/2006 of the European Parliament and of the Council of 18 December 2006 Concerning the Registration, Evaluation, Authorisation and Restriction of Chemicals, Establishing a European Chemicals Agency. Available online: http://data.europa.eu/eli/reg/2006/1907/oj (accessed on 9 July 2024).
- Vieira, Y.; Lima, E.C.; Foletto, E.L.; Dotto, G.L. Microplastics physicochemical properties, specific adsorption modeling and their interaction with pharmaceuticals and other emerging contaminants. Sci. Total Environ. 2021, 753, 141981. [Google Scholar] [CrossRef]
- Liu, G.; Zhu, Z.; Yang, Y.; Sun, Y.; Yu, F.; Ma, J. Sorption behavior and mechanism of hydrophilic organic chemicals to virgin and aged microplastics in freshwater and seawater. Environ. Pollut. 2019, 246, 26–33. [Google Scholar] [CrossRef]
- Razanajatovo, R.M.; Ding, J.N.; Zhang, S.S.; Jiang, H.; Zou, H. Sorption and desorption of selected pharmaceuticals by polyethylene microplastics. Mar. Pollut. Bull. 2018, 136, 516–523. [Google Scholar] [CrossRef]
- Parmanbek, N.; Sütekin, D.S.; Barsbay, M.; Mashentseva, A.A.; Zheltov, D.A.; Aimanova, N.A.; Jakupova, Z.J.; Zdorovets, M.V. Hybrid PET track-etched membranes grafted by well-defined poly(2-(dimethylamino)ethyl methacrylate) brushes and loaded with silver nanoparticles for the removal of As(III). Polymers 2022, 13, 4026. [Google Scholar] [CrossRef] [PubMed]
- Leiva, E.; Tapia, C.; Rodriguez, C. Highly efficient removal of Cu(II) ions from acidic aqueous solution using ZnO nanoparticles as nano-adsorbents. Water 2021, 13, 2960. [Google Scholar] [CrossRef]
- Seidensticker, S.; Grathwohl, P.; Lamprecht, J.; Zarfl, C. A combined experimental and modeling study to evaluate pH-dependent sorption of polar and non-polar compounds to polyethylene and polystyrene microplastics. Environ. Sci. Eur. 2018, 30, 30. [Google Scholar] [CrossRef] [PubMed]
- Bundchuh, M.; Weyers, A.; Ebeling, M.; Elsaesser, D.; Schulz, R. Narrow pH range of surface water bodies receiving pesticide input in Europe. Bull. Environ. Contam. Toxicol. 2016, 96, 3–8. [Google Scholar] [CrossRef]
- Rybacka, A.; Andersson, P.L. Considering ionic state in modeling sorption of pharmaceuticals to sewage sludge. Chemosphere 2016, 165, 284–293. [Google Scholar] [CrossRef]
- Guo, X.; Pang, J.; Chen, S.; Jia, H. Sorption properties of tylosin on four different microplastics. Chemosphere 2018, 209, 240–245. [Google Scholar] [CrossRef] [PubMed]
- Grabicova, K.; Grabic, R.; Fedorova, G.; Fick, J.; Cerveny, D.; Kolarova, J.; Turek, J.; Zlabek, V.; Randak, T. Bioaccumulation of psychoactive pharmaceuticals in fish in an effluent dominated streams. Water Res. 2017, 124, 654–662. [Google Scholar] [CrossRef] [PubMed]
- Grabicova, K.; Grabic, R.; Blaha, L.; Kumar, V.; Cerveny, D.; Fedorova, G.; Randak, T. Presence of pharmaceuticals in benthic fauna living in a small stream affected by effluent from a municipal sewage treatment plant. Water Res. 2015, 72, 145–153. [Google Scholar] [CrossRef] [PubMed]
- Anbumani, S.; Kakkar, P. Ecotoxicological effects of microplastics on biota: A review. Environ. Sci. Pollut. Res. 2018, 25, 14373–14396. [Google Scholar] [CrossRef]
- Grabowska, M.; Mazur-Marzec, H. The influence of hydrological conditions on phytoplankton community structure and cyanopeptide concentration in dammed lowland river. Environ. Monit. Assess. 2016, 188, 488. [Google Scholar] [CrossRef]
- Izydorczyk, K.; Jurczak, T.; Wojtal-Frankiewicz, A.; Skowron, A.; Mankiewicz-Boczek, J.; Tarczyńska, M. Influence of abiotic and biotic factors on microcystin content in Microcystis aeruginosa cells in a eutrophic temperate reservoir. J. Plankton Res. 2008, 30, 393–400. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).