Colloids in Thermokarst Lakes along a Permafrost and Climate Gradient of Permafrost Peatlands in Western Siberia Using In Situ Dialysis Procedure
Abstract
:1. Introduction
2. Materials and Methods
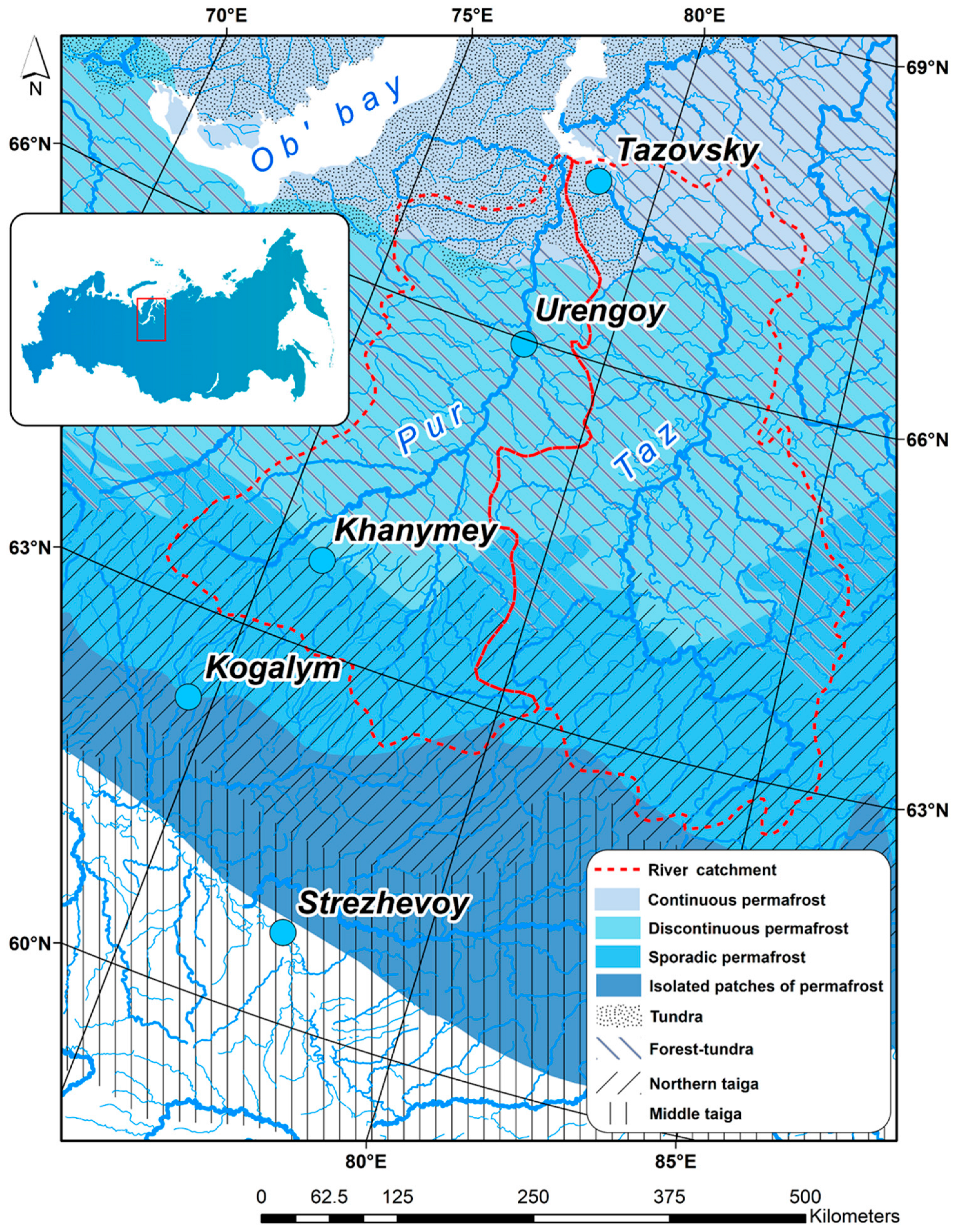
2.1. Study Sites in Western Siberia
2.2. Water Sampling and Size Fractionation
2.3. Analyses
2.4. Statistical Treatment
2.5. Element Speciation Modeling
3. Results and Discussion
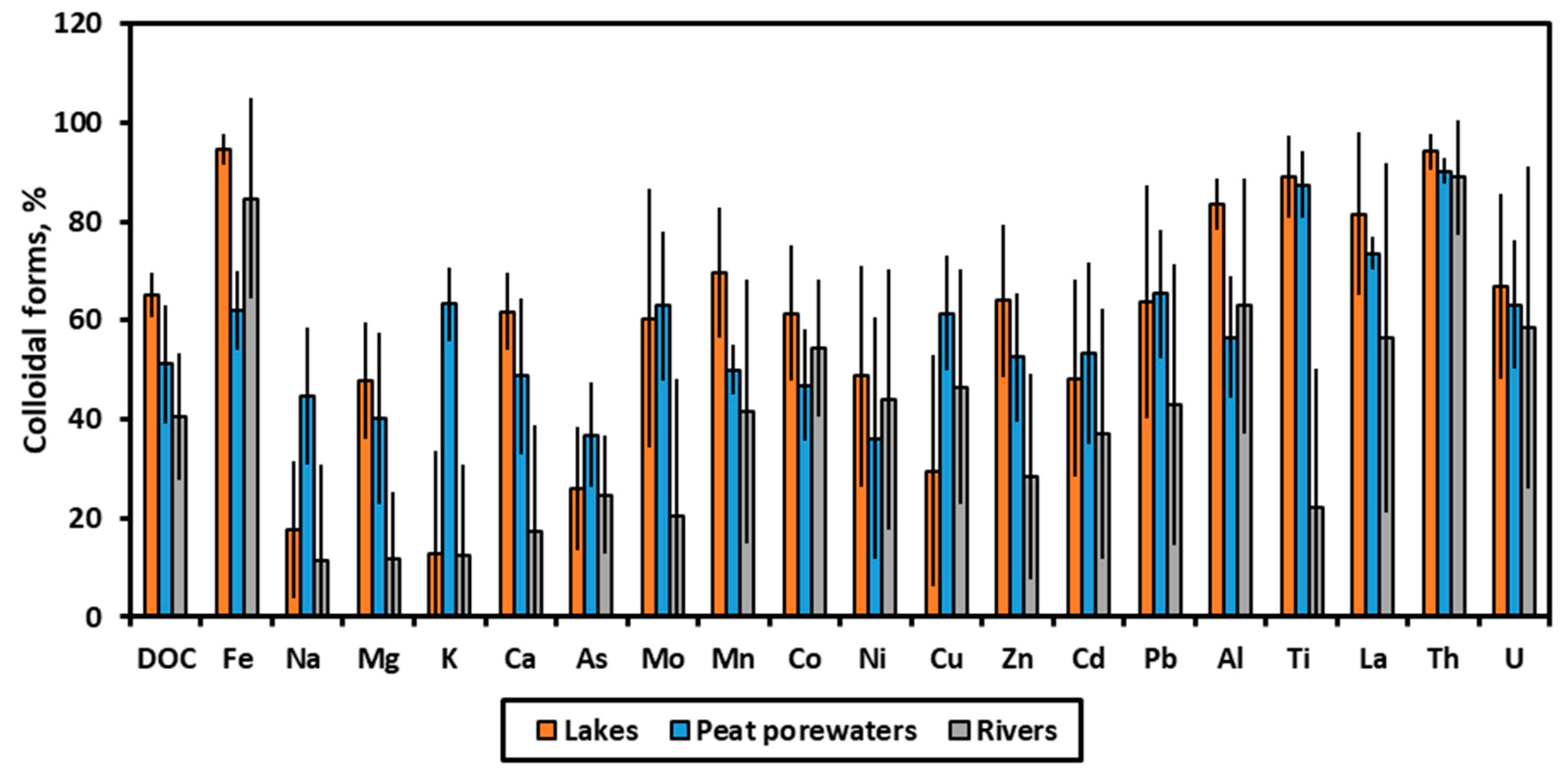
3.1. Effect of Lake Surface Area on the Colloidal Status of Solutes in Thermokarst Lake Waters
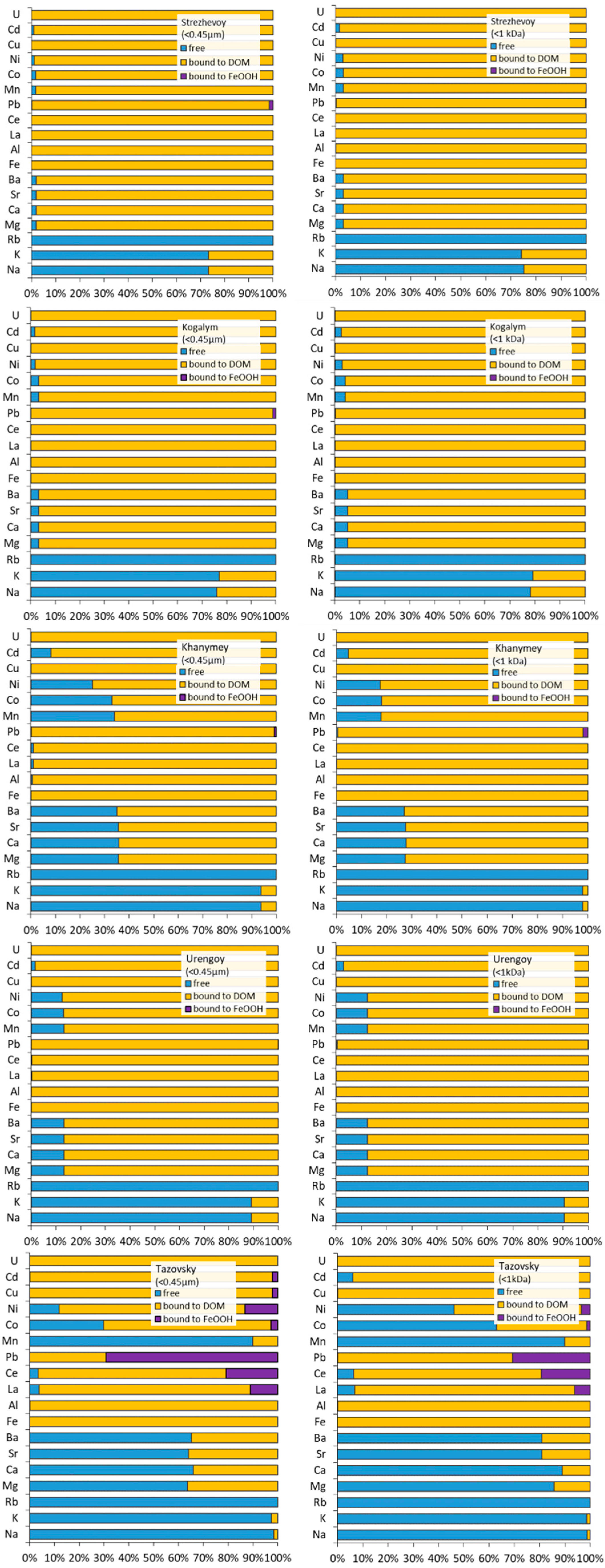
3.2. Groups of Solutes According to Their Colloidal Status and Stoichiometry of Colloids
3.3. Element Speciation in Colloids via Thermodynamic Modeling
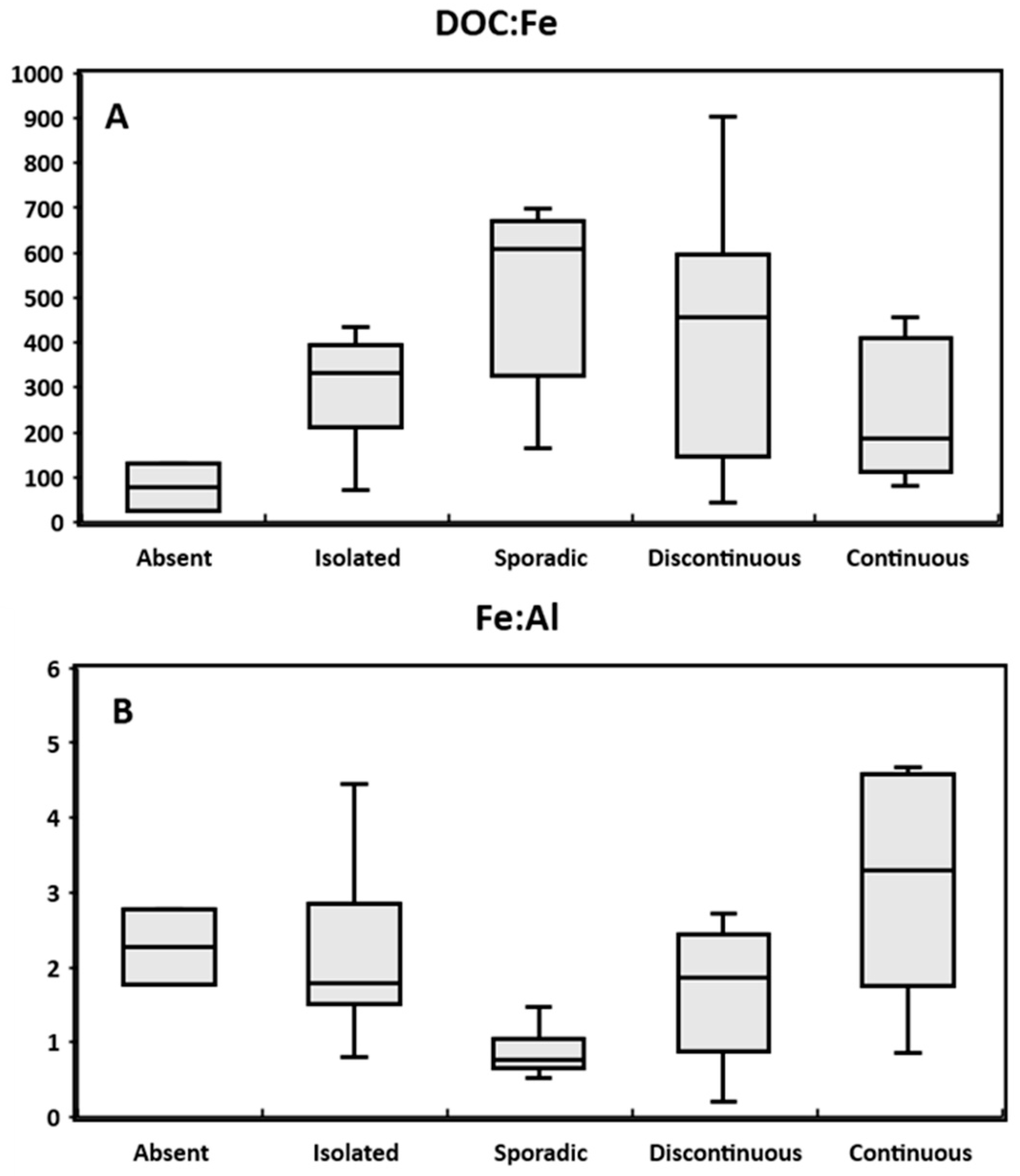
3.4. Effect of Permafrost Distribution on Colloidal Status of Trace Elements: Possible Evolution in Case of Permafrost Thaw
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Appendix A
| Element | Absent (60°48′ N/77°41′ E) | Isolated (62°15′ N/74°10′ E) | Sporadic (63°47′ N/75°33′ E) | Discontinuous (66°00′ N/78°31′ E) | Continuous (67°21′ N/78°37′ E) | |||||
|---|---|---|---|---|---|---|---|---|---|---|
| <0.45 μm | <1 kDa | <0.45 μm | <1 kDa | <0.45 μm | <1 kDa | <0.45 μm | <1 kDa | <0.45 μm | <1 kDa | |
| SC | 16.3 ± 3.2 | – | 15.7 ± 6.6 | – | 21.6 ± 8.7 | – | 22.1 ± 13 | – | 20 ± 9.2 | – |
| pH | 4.64 ± 0.3 | – | 5.03 ± 0.7 | – | 4 ± 0.2 | – | 4.11 ± 0.4 | – | 6.24 ± 0.4 | – |
| SUVA254 | 3.6 ± 0.8 | 1.5 ± 0.6 | 2.9 ± 0.5 | 0.6 ± 0.3 | 4.1 ± 0.5 | 1.5 ± 0.6 | 3.9 ± 0.4 | 2.1 ± 0.3 | 3.2 ± 0.6 | 2.0 ± 0.1 |
| Cl | 0.14 ± 0.1 | 0.25 ± 0.2 | 0.24 | – | – | – | 0.19 ± 0.2 | 0.29 ± 0.3 | – | – |
| SO4 | 0.49 ± 0.5 | 0.36 ± 0.3 | 0.21 | – | – | – | 0.1 ± 0.06 | 0.05 ± 0.01 | – | – |
| DOC | 17.4 ± 3.9 | 6.7 ± 0.9 | 12.1 ± 3.4 | 3.97 ± 1.2 | 23.6 ± 10.8 | 8.6 ± 4.7 | 21.3 ± 5.5 | 7.56 ± 2.4 | 10.1 ± 1.7 | 2.94 ± 0.7 |
| DIC | 0.81 ± 0.2 | 0.9 ± 0.03 | 0.92 ± 0.6 | 0.69 ± 0.2 | 0.65 ± 0.1 | 0.6 ± 0.1 | 0.77 ± 0.2 | 0.74 ± 0.2 | 1.98 ± 1.1 | 2.23 ± 0.98 |
| Li | 0.53 ± 0.3 | 0.32 ± 0.2 | 0.37 ± 0.4 | 0.24 ± 0.26 | 0.67 ± 0.4 | 0.4 ± 0.1 | 0.33 ± 0.2 | 0.202 ± 0.1 | 0.88 ± 0.5 | 0.75 ± 0.4 |
| Be | 0.011 ± 0.01 | 0.009 | 0.004 ± 0.002 | 0.001 ± 0.0006 | 0.02 ± 0.01 | 0.04 ± 0.001 | 0.006 ± 0.003 | 0.002 ± 0.001 | 0.008 ± 0.004 | 0.001 ± 0.0007 |
| B | 3.94 ± 1.5 | 3.54 ± 1.6 | 3.17 ± 1.4 | 2.27 ± 1.5 | 6.27 ± 2.3 | 10.4 ± 3.7 | 2.22 ± 1.5 | 2.36 ± 1.5 | 1.68 ± 1.67 | 1.97 ± 2 |
| Na | 0.27 ± 0.2 | 0.24 ± 0.1 | 0.2 ± 0.1 | 0.15 ± 0.09 | 0.45 ± 0.4 | 0.33 ± 0.1 | 0.25 ± 0.3 | 0.18 ± 0.16 | 0.45 ± 0.2 | 0.43 ± 0.2 |
| Mg | 0.25 ± 0.2 | 0.1 ± 0.09 | 0.14 ± 0.1 | 0.05 ± 0.04 | 0.26 ± 0.1 | 0.2 ± 0.03 | 0.12 ± 0.1 | 0.04 ± 0.04 | 1.07 ± 0.6 | 0.82 ± 0.6 |
| Al | 0.15 ± 0.1 | 0.036 ± 0.036 | 0.038 ± 0.01 | 0.0065 ± 0.003 | 0.092 ± 0.05 | 0.0094 ± 0.004 | 0.11 ± 0.08 | 0.014 ± 0.007 | 0.035 ± 0.03 | 0.0037 ± 0.002 |
| Si | 0.23 ± 0.1 | 0.2 ± 0.1 | 0.22 ± 0.2 | 0.19 ± 0.20 | 0.25 ± 0.2 | 0.19 ± 0.06 | 0.37 ± 0.3 | 0.35 ± 0.3 | 0.22 ± 0.1 | 0.23 ± 0.1 |
| K | 0.37 ± 0.5 | 0.29 ± 0.3 | 0.047 ± 0.04 | 0.037 ± 0.027 | 0.078 ± 0.071 | 0.082 ± 0.02 | 0.04 ± 0.02 | 0.04 ± 0.02 | 0.11 ± 0.1 | 0.096 ± 0.1 |
| Ca | 0.66 ± 0.5 | 0.27 ± 0.2 | 0.39 ± 0.3 | 0.15 ± 0.1 | 0.29 ± 0.2 | 0.065 ± 0.01 | 0.42 ± 0.3 | 0.14 ± 0.11 | 2.29 ± 1.3 | 1.69 ± 1.1 |
| Ti | 0.51 ± 0.4 | 0.056 ± 0.004 | 0.3 ± 0.2 | 0.025 ± 0.02 | 0.8 ± 0.5 | 0.07 ± 0.01 | 0.8 ± 0.6 | 0.044 ± 0.02 | 0.49 ± 0.44 | 0.02 ± 0.009 |
| V | 0.32 ± 0.3 | 0.31 ± 0.3 | 0.13 ± 0.1 | 0.094 ± 0.03 | 1.02 ± 0.5 | 0.99 ± 0.2 | 0.33 ± 0.2 | 0.22 ± 0.2 | 0.26 ± 0.2 | 0.11 ± 0.09 |
| Cr | 0.38 ± 0.3 | 0.19 ± 0.1 | 0.15 ± 0.1 | 0.23 ± 0.6 | 0.52 ± 0.2 | 0.29 ± 0.07 | 0.31 ± 0.2 | 0.12 ± 0.04 | 0.19 ± 0.09 | 0.039 ± 0.02 |
| Mn | 0.043 ± 0.04 | 0.017 ± 0.02 | 0.0092 ± 0.004 | 0.0032 ± 0.001 | 0.013 ± 0.007 | 0.0035 ± 0.002 | 0.022 ± 0.02 | 0.007 ± 0.006 | 0.0054 ± 0.004 | 0.0008 ± 0.001 |
| Fe | 0.31 ± 0.3 | 0.029 ± 0.03 | 0.18 ± 0.2 | 0.0075 ± 0.007 | 0.15 ± 0.1 | 0.0039 ± 0.002 | 0.35 ± 0.4 | 0.0088 ± 0.005 | 0.21 ± 0.1 | 0.0023 ± 0.001 |
| Co | 0.34 ± 0.4 | 0.14 ± 0.2 | 0.05 ± 0.01 | 0.021 ± 0.007 | 0.11 ± 0.07 | 0.036 ± 0.01 | 0.17 ± 0.1 | 0.052 ± 0.03 | 0.15 ± 0.07 | 0.035 ± 0.01 |
| Ni | 0.66 ± 0.9 | 0.29 ± 0.3 | 0.096 ± 0.03 | 0.065 ± 0.05 | 0.35 ± 0.2 | 0.19 ± 0.06 | 0.45 ± 0.3 | 0.15 ± 0.04 | 1.21 ± 0.4 | 0.32 ± 0.1 |
| Cu | 0.2 ± 0.2 | 0.25 ± 0.1 | 0.15 ± 0.07 | 0.19 ± 0.09 | 0.28 ± 0.1 | 0.14 ± 0.04 | 0.27 ± 0.2 | 0.26 ± 0.04 | 0.48 ± 0.3 | 0.35 ± 0.07 |
| Zn | 6.2 ± 2.8 | 2.14 ± 1.7 | 2.35 ± 0.5 | 1.13 ± 0.5 | 4.42 ± 2.4 | 1.32 ± 0.9 | 5.33 ± 1.8 | 1.25 ± 0.3 | 1.64 ± 0.9 | 0.62 ± 0.3 |
| Ga | 0.017 ± 0.008 | 0.0049 ± 0.004 | 0.0091 ± 0.004 | 0.0019 ± 0.001 | 0.021 ± 0.01 | 0.0053 ± 0.0009 | 0.019 ± 0.008 | 0.0031 ± 0.0009 | 0.0058 ± 0.005 | 0.0014 ± 0.001 |
| Ge | 0.0019 ± 0.001 | 0.0014 | 0.001 ± 0.0007 | 0.0009 ± 0.0004 | 0.0035 ± 0.001 | 0.0033 ± 0.0007 | 0.001 ± 0.0006 | 0.0022 ± 0.0009 | 0.001 ± 0.0008 | 0.0004 ± 0.0007 |
| As | 0.54 ± 0.2 | 0.56 ± 0.09 | 0.38 ± 0.1 | 0.29 ± 0.07 | 0.47 ± 0.2 | 0.3 ± 0.09 | 0.31 ± 0.1 | 0.18 ± 0.05 | 0.44 ± 0.1 | 0.28 ± 0.05 |
| Rb | 0.86 ± 1.1 | 0.59 ± 0.7 | 0.1 ± 0.096 | 0.067 ± 0.065 | 0.15 ± 0.17 | 0.064 ± 0.05 | 0.11 ± 0.09 | 0.077 ± 0.05 | 0.16 ± 0.1 | 0.14 ± 0.1 |
| Sr | 3.35 ± 3.2 | 1.32 ± 1.4 | 2.32 ± 2.4 | 0.8 ± 0.8 | 2.72 ± 1.6 | 0.63 ± 0.4 | 2.59 ± 2.3 | 0.78 ± 0.7 | 9.72 ± 4.9 | 7.22 ± 4.4 |
| Y | 0.053 ± 0.05 | 0.0083 ± 0.007 | 0.026 ± 0.03 | 0.0015 ± 0.001 | 0.037 ± 0.02 | 0.0025 ± 0.001 | 0.06 ± 0.06 | 0.0026 ± 0.002 | 0.083 ± 0.03 | 0.0035 ± 0.002 |
| Zr | 0.067 ± 0.07 | 0.0048 ± 0.004 | 0.031 ± 0.04 | 0.0005 ± 0.0003 | 0.094 ± 0.06 | 0.0081 ± 0.018 | 0.088 ± 0.07 | 0.0041 ± 0.003 | 0.075 ± 0.04 | 0.0011 ± 0.0006 |
| Nb | 0.0044 ± 0.004 | 0.0002 ± 0.00004 | 0.0013 ± 0.0006 | 0.002 ± 0.004 | 0.0006 ± 0.0003 | 0.00004 ± 0.00002 | 0.0048 ± 0.002 | 0.0004 ± 0.0003 | 0.0018 ± 0.0016 | 0.0001 ± 0.0001 |
| Mo | 0.072 ± 0.03 | 0.015 ± 0.01 | 0.031 ± 0.04 | 0.0039 ± 0.002 | 0.011 ± 0.01 | 0.0095 ± 0.004 | 0.045 ± 0.047 | 0.0084 ± 0.01 | 0.061 ± 0.052 | 0.02 ± 0.02 |
| Cd | 0.03 ± 0.02 | 0.013 ± 0.01 | 0.014 ± 0.006 | 0.0071 ± 0.004 | 0.019 ± 0.01 | 0.015 ± 0.008 | 0.012 ± 0.003 | 0.0067 ± 0.004 | 0.0022 ± 0.002 | 0.01 ± 0.008 |
| Sb | 0.039 ± 0.01 | 0.055 ± 0.02 | 0.035 ± 0.008 | 0.037 ± 0.008 | 0.043 ± 0.01 | 0.054 ± 0.01 | 0.034 ± 0.01 | 0.042 ± 0.01 | 0.017 ± 0.007 | 0.018 ± 0.006 |
| Cs | 0.0093 ± 0.005 | 0.0056 ± 0.003 | 0.0044 ± 0.004 | 0.0026 ± 0.002 | 0.0099 ± 0.007 | 0.0044 ± 0.003 | 0.0039 ± 0.004 | 0.0021 ± 0.002 | 0.0008 ± 0.0008 | 0.0005 ± 0.0002 |
| Ba | 3.75 ± 4.6 | 1.48 ± 1.9 | 1.88 ± 2.1 | 0.66 ± 0.61 | 2.84 ± 1.9 | 0.65 ± 0.4 | 4.03 ± 3.3 | 1.16 ± 0.9 | 1.61 ± 0.64 | 0.96 ± 0.4 |
| La | 0.046 ± 0.06 | 0.0081 ± 0.006 | 0.044 ± 0.1 | 0.0027 ± 0.004 | 0.02 ± 0.01 | 0.0015 ± 0.0008 | 0.047 ± 0.06 | 0.0024 ± 0.001 | 0.061 ± 0.03 | 0.017 ± 0.04 |
| Ce | 0.13 ± 0.1 | 0.02 ± 0.018 | 0.071 ± 0.15 | 0.0065 ± 0.01 | 0.046 ± 0.04 | 0.0027 ± 0.002 | 0.12 ± 0.1 | 0.0053 ± 0.003 | 0.14 ± 0.07 | 0.12 ± 0.3 |
| Pr | 0.014 ± 0.02 | 0.0022 ± 0.002 | 0.0043 ± 0.005 | 0.0006 ± 0.001 | 0.0055 ± 0.004 | 0.0003 ± 0.0002 | 0.013 ± 0.02 | 0.0006 ± 0.0004 | 0.018 ± 0.008 | 0.0015 ± 0.002 |
| Nd | 0.061 ± 0.07 | 0.0096 ± 0.01 | 0.018 ± 0.02 | 0.0027 ± 0.004 | 0.022 ± 0.02 | 0.0014 ± 0.0007 | 0.055 ± 0.06 | 0.0024 ± 0.002 | 0.076 ± 0.03 | 0.0053 ± 0.007 |
| Sm | 0.014 ± 0.01 | 0.0033 ± 0.003 | 0.0048 ± 0.005 | 0.0007 ± 0.0008 | 0.0075 ± 0.005 | 0.0009 ± 0.0006 | 0.014 ± 0.01 | 0.0008 ± 0.0004 | 0.018 ± 0.009 | 0.0008 ± 0.0004 |
| Eu | 0.0031 ± 0.003 | 0.0008 ± 0.0006 | 0.0015 ± 0.0015 | 0.0002 ± 0.0002 | 0.0014 ± 0.0009 | 0.0004 ± 0.0001 | 0.0035 ± 0.003 | 0.0005 ± 0.0003 | 0.0039 ± 0.002 | 0.0003 ± 0.0003 |
| Gd | 0.014 ± 0.01 | 0.0027 ± 0.002 | 0.0054 ± 0.006 | 0.0008 ± 0.0006 | 0.0068 ± 0.004 | 0.0008 ± 0.0004 | 0.013 ± 0.01 | 0.0011 ± 0.0007 | 0.018 ± 0.008 | 0.0036 ± 0.005 |
| Tb | 0.002 ± 0.002 | 0.0003 ± 0.0001 | 0.0008 ± 0.0008 | 0.0001 ± 0.0001 | 0.0011 ± 0.0006 | 0.0001 ± 0.0001 | 0.002 ± 0.002 | 0.0001 ± 0.0001 | 0.0026 ± 0.001 | 0.0001 ± 0.0001 |
| Dy | 0.011 ± 0.01 | 0.0017 ± 0.002 | 0.0049 ± 0.005 | 0.0006 ± 0.0004 | 0.0071 ± 0.004 | 0.0011 ± 0.0006 | 0.012 ± 0.01 | 0.0005 ± 0.0003 | 0.016 ± 0.006 | 0.0009 ± 0.0002 |
| Ho | 0.0019 ± 0.002 | 0.0003 ± 0.0001 | 0.0011 ± 0.001 | 0.0001 ± 0.0001 | 0.0017 ± 0.001 | 0.0002 ± 0.0002 | 0.0024 ± 0.002 | 0.0001 ± 0.0001 | 0.003 ± 0.001 | 0.0001 ± 0.0001 |
| Er | 0.005 ± 0.006 | 0.001 ± 0.0008 | 0.0032 ± 0.003 | 0.0003 ± 0.0001 | 0.0048 ± 0.003 | 0.0007 ± 0.0002 | 0.0064 ± 0.006 | 0.0007 ± 0.0004 | 0.0083 ± 0.004 | 0.0004 ± 0.0002 |
| Tm | 0.0007 ± 0.0008 | 0.0002 ± 0.0002 | 0.0005 ± 0.0005 | 0.0001 ± 0.0001 | 0.0008 ± 0.0004 | 0.0001 ± 0.00004 | 0.001 ± 0.001 | 0.0001 ± 0.0001 | 0.0013 ± 0.0005 | 0.0001 ± 0.00005 |
| Yb | 0.0045 ± 0.005 | 0.001 ± 0.0007 | 0.0026 ± 0.003 | 0.0006 ± 0.0007 | 0.004 ± 0.002 | 0.0007 ± 0.0005 | 0.0055 ± 0.0055 | 0.0004 ± 0.0002 | 0.0094 ± 0.004 | 0.0008 ± 0.0003 |
| Lu | 0.0006 ± 0.0005 | 0.0002 ± 0.0001 | 0.0005 ± 0.0006 | 0.0001 ± 0.0001 | 0.0007 ± 0.0004 | 0.0002 ± 0.0001 | 0.0007 ± 0.0007 | 0.0001 ± 0.00004 | 0.0013 ± 0.0005 | 0.0001 ± 0.0001 |
| Hf | 0.0025 ± 0.003 | 0.0005 ± 0.0004 | 0.0016 ± 0.001 | 0.0005 ± 0.0003 | 0.0039 ± 0.003 | 0.0011 ± 0.001 | 0.0032 ± 0.003 | 0.0002 ± 0.0001 | 0.0023 ± 0.002 | 0.0004 ± 0.0002 |
| Tl | 0.0038 ± 0.003 | 0.002 ± 0.002 | 0.0019 ± 0.0018 | 0.0014 ± 0.0009 | 0.0049 ± 0.003 | 0.0013 ± 0.0008 | 0.0016 ± 0.0009 | 0.0005 ± 0.0004 | 0.0003 ± 0.0002 | 0.0002 ± 0.0002 |
| Pb | 0.31 ± 0.2 | 0.22 ± 0.2 | 0.21 ± 0.1 | 0.062 ± 0.05 | 0.19 ± 0.1 | 0.034 ± 0.02 | 0.21 ± 0.1 | 0.063 ± 0.05 | 0.081 ± 0.09 | 0.049 ± 0.07 |
| Th | 0.011 ± 0.01 | 0.0005 ± 0.0004 | 0.0038 ± 0.002 | 0.0003 ± 0.0002 | 0.011 ± 0.007 | 0.0002 ± 0.0002 | 0.011 ± 0.01 | 0.0007 ± 0.0005 | 0.011 ± 0.006 | 0.0005 ± 0.0003 |
| U | 0.0012 ± 0.0007 | 0.001 ± 0.0008 | 0.0017 ± 0.001 | 0.0003 ± 0.0002 | 0.015 ± 0.03 | 0.0008 ± 0.0004 | 0.0019 ± 0.0019 | 0.0004 ± 0.0002 | 0.0085 ± 0.006 | 0.0008 ± 0.0004 |
References
- Frey, K.E.; McClelland, J.W.; Holmes, R.M.; Smith, L.C. Impacts of climate warming and permafrost thaw on the riverine transport of nitrogen and phosphorus to the Kara Sea. J. Geophys. Res. Biogeosci. 2007, 112, G04S58. [Google Scholar] [CrossRef]
- Vonk, J.E.; Tank, S.E.; Bowden, W.B.; Laurion, I.; Vincent, W.F.; Alekseychik, P.; Amyot, M.; Billet, M.F.; Canário, J.; Cory, R.M.; et al. Reviews and Syntheses: Effects of Permafrost Thaw on Arctic Aquatic Ecosystems. Biogeosciences 2015, 12, 7129–7167. [Google Scholar] [CrossRef]
- Walvoord, M.A.; Kurylyk, B.L. Hydrologic Impacts of Thawing Permafrost—A Review. Vadose Zone J. 2016, 15, vzj2016.01.0010. [Google Scholar] [CrossRef]
- Wik, M.; Varner, R.K.; Anthony, K.W.; MacIntyre, S.; Bastviken, D. Climate-Sensitive Northern Lakes and Ponds Are Critical Components of Methane Release. Nat. Geosci. 2016, 9, 99–105. [Google Scholar] [CrossRef]
- Turetsky, M.R.; Abbott, B.W.; Jones, M.C.; Anthony, K.W.; Olefeldt, D.; Schuur, E.A.G.; Koven, C.; McGuire, A.D.; Grosse, G.; Kuhry, P.; et al. Permafrost Collapse Is Accelerating Carbon Release. Nature 2019, 569, 32–34. [Google Scholar] [CrossRef]
- Polishchuk, Y.M.; Bogdanov, A.N.; Polishchuk, V.Y.; Manasypov, R.M.; Shirokova, L.S.; Kirpotin, S.N.; Pokrovsky, O.S. Size Distribution, Surface Coverage, Water, Carbon, and Metal Storage of Thermokarst Lakes in the Permafrost Zone of the Western Siberia Lowland. Water 2017, 9, 228. [Google Scholar] [CrossRef]
- Polishchuk, Y.M.; Bogdanov, A.N.; Muratov, I.N.; Polishchuk, V.Y.; Lim, A.; Manasypov, R.M.; Shirokova, L.S.; Pokrovsky, O.S. Minor contribution of small thaw ponds to the pools of carbon and methane in the inland waters of the permafrost-affected part of the Western Siberian Lowland. Environ. Res. Lett. 2018, 13, 045002. [Google Scholar] [CrossRef]
- Serikova, S.; Pokrovsky, O.S.; Laudon, H.; Krickov, I.V.; Lim, A.G.; Manasypov, R.M.; Karlsson, J. High carbon emissions from thermokarst lakes of Western Siberia. Nat. Commun. 2019, 10, 1552. [Google Scholar] [CrossRef] [PubMed]
- Karlsson, J.; Serikova, S.; Vorobyev, S.N.; Rocher-Ros, G.; Denfeld, B.; Pokrovsky, O.S. Carbon emission from Western Siberian inland waters. Nat. Commun. 2021, 12, 825. [Google Scholar] [CrossRef] [PubMed]
- Shirokova, L.S.; Chupakov, A.V.; Ivanova, I.S.; Moreva, O.Y.; Zabelina, S.A.; Shutskiy, N.A.; Loiko, S.V.; Pokrovsky, O.S. Lichen, moss and peat control of C, nutrient and trace metal regime in lakes of permafrost peatlands. Sci. Total Environ. 2021, 782, 146737. [Google Scholar] [CrossRef] [PubMed]
- Pokrovsky, O.S.; Shirokova, L.S.; Kirpotin, S.N.; Audry, S.; Viers, J.; Dupré, B. Effect of permafrost thawing on organic carbon and trace element colloidal speciation in the thermokarst lakes of western Siberia. Biogeosciences 2011, 8, 565–583. [Google Scholar] [CrossRef]
- Pokrovsky, O.S.; Shirokova, L.S.; Kirpotin, S.N.; Kulizhsky, S.P.; Vorobiev, S.N. Impact of western Siberia heat wave 2012 on greenhouse gases and trace metal concentration in thaw lakes of discontinuous permafrost zone. Biogeosciences 2013, 10, 5349–5365. [Google Scholar] [CrossRef]
- Shirokova, L.S.; Pokrovsky, O.S.; Kirpotin, S.N.; Desmukh, C.; Pokrovsky, B.G.; Audry, S.; Viers, J. Biogeochemistry of organic carbon, CO2, CH4, and trace elements in thermokarst water bodies in discontinuous permafrost zones of Western Siberia. Biogeochemistry 2013, 113, 573–593. [Google Scholar] [CrossRef]
- Pokrovsky, O.S.; Manasypov, R.M.; Loiko, S.V.; Shirokova, L.S. Organic and organo-mineral colloids in discontinuous permafrost zone. Geochim. Cosmochim. Acta 2016, 188, 1–20. [Google Scholar] [CrossRef]
- Manasypov, R.M.; Vorobyev, S.N.; Loiko, S.V.; Kritzkov, I.V.; Shirokova, L.S.; Shevchenko, V.P.; Kirpotin, S.N.; Kulizhsky, S.P.; Kolesnichenko, L.G.; Zemtzov, V.A.; et al. Seasonal dynamics of organic carbon and metals in thermokarst lakes from the discontinuous permafrost zone of western Siberia. Biogeosciences 2015, 12, 3009–3028. [Google Scholar] [CrossRef]
- Manasypov, R.M.; Lim, A.G.; Krickov, I.V.; Shirokova, L.S.; Vorobyev, S.N.; Kirpotin, S.N.; Pokrovsky, O.S. Spatial and seasonal variations of C, nutrient, and metal concentration in thermokarst lakes of Western Siberia across a permafrost gradient. Water 2020, 12, 1830. [Google Scholar] [CrossRef]
- Pavlova, O.A.; Pokrovsky, O.S.; Manasypov, R.M.; Shirokova, L.S.; Vorobyev, S.N. Seasonal dynamics of phytoplankton in acidic and humic environment in thaw ponds of discontinuous permafrost zone. Ann. Limnol.—Int. J. Lim. 2016, 52, 47–60. [Google Scholar] [CrossRef]
- Pokrovsky, O.S.; Manasypov, R.M.; Pavlova, O.A.; Shirokova, L.S.; Vorobyev, S.N. Carbon, nutrient and metal controls on phytoplankton concentration and biodiversity in thermokarst lakes of latitudinal gradient from isolated to continuous permafrost. Sci. Total Environ. 2022, 806, 151250. [Google Scholar] [CrossRef]
- Audry, S.; Pokrovsky, O.S.; Shirokova, L.S.; Kirpotin, S.N.; Dupré, B. Organic matter mineralization and trace element post-depositional redistribution in Western Siberia thermokarst lake sediments. Biogeosciences 2011, 8, 3341–3358. [Google Scholar] [CrossRef]
- Manasypov, R.M.; Lim, A.G.; Krickov, I.V.; Shirokova, L.S.; Shevchenko, V.P.; Aliev, R.A.; Karlsson, J.; Pokrovsky, O.S. Carbon storage and burial in thermokarst lakes of permafrost peatlands. Biogeochemistry 2022, 159, 69–86. [Google Scholar] [CrossRef]
- Manasypov, R.M.; Shirokova, L.S.; Pokrovsky, O.S. Experimental modeling of thaw lake water evolution in discontinuous permafrost zone: Role of peat, lichen leaching and ground fire. Sci. Total Environ. 2017, 580, 245–257. [Google Scholar] [CrossRef] [PubMed]
- Kuzmina, D.; Lim, A.G.; Loiko, S.V.; Pokrovsky, O.S. Experimental assessment of tundra fire impact on element export and storage in permafrost peatlands. Sci. Total Environ. 2022, 853, 158701. [Google Scholar] [CrossRef] [PubMed]
- Loiko, S.V.; Pokrovsky, O.S.; Raudina, T.V.; Lim, A.; Kolesnichenko, L.G.; Shirokova, L.S.; Vorobyev, S.N.; Kirpotin, S.N. Abrupt permafrost collapse enhances organic carbon, CO2, nutrient and metal release into surface waters. Chem. Geol. 2017, 471, 153–165. [Google Scholar] [CrossRef]
- Krickov, I.V.; Pokrovsky, O.S.; Manasypov, R.M.; Lim, A.G.; Shirokova, L.S.; Viers, J. Colloidal Transport of Carbon and Metals by Western Siberian Rivers during Different Seasons across a Permafrost Gradient. Geochim. Cosmochim. Acta 2019, 265, 221–241. [Google Scholar] [CrossRef]
- Raudina, T.V.; Loiko, S.V.; Kuzmina, D.M.; Shirokova, L.S.; Kulizhskiy, S.P.; Golovatskaya, E.A.; Pokrovsky, O.S. Colloidal Organic Carbon and Trace Elements in Peat Porewaters across a Permafrost Gradient in Western Siberia. Geoderma 2021, 390, 114971. [Google Scholar] [CrossRef]
- Lim, A.G.; Loiko, S.V.; Kuzmina, D.M.; Krickov, I.V.; Shirokova, L.S.; Kulizhsky, S.P.; Pokrovsky, O.S. Organic Carbon, and Major and Trace Elements Reside in Labile Low-Molecular Form in the Ground Ice of Permafrost Peatlands: A Case Study of Colloids in Peat Ice of Western Siberia. Environ. Sci. Process. Impacts 2022, 24, 1443–1459. [Google Scholar] [CrossRef]
- Krickov, I.V.; Lim, A.G.; Vorobyev, S.N.; Shevchenko, V.P.; Pokrovsky, O.S. Colloidal associations of major and trace elements in the snow pack across a 2800-km south-north gradient of western Siberia. Chem. Geol. 2022, 610, 121090. [Google Scholar] [CrossRef]
- Carpita, N.; Sabularse, D.; Montezinos, D.; Delmer, D. Determination of the pore size of cell walls of living plant cells. Science 1979, 205, 1144–1147. [Google Scholar] [CrossRef]
- Trias, J.; Jarlier, V.; Benz, R. Porins in the cell wall of mycobacteria. Science 1992, 258, 1479–1481. [Google Scholar] [CrossRef]
- Vasyukova, E.; Pokrovsky, O.S.; Viers, J.; Oliva, P.; Dupré, B.; Martin, F.; Candaudap, F. Trace elements in organic- and iron-rich surficial fluids of the boreal zone: Assessing colloidal forms via dialysis and ultrafiltration. Geochim. Cosmochim. Acta 2010, 74, 449–468. [Google Scholar] [CrossRef]
- Vasyukova, E.; Pokrovsky, O.S.; Viers, J.; Dupré, B. New operational method of testing colloid complexation with metals in natural waters. Appl. Geochem. 2012, 6, 1226–1237. [Google Scholar] [CrossRef]
- Magaritz, M.; Wells, M.; Amiel, A.J.; Ronen, D. Application of a multi-layer sampler based on the dialysis cell technique for the study of trace metals in groundwater. Appl. Geochem. 1989, 4, 617–624. [Google Scholar] [CrossRef]
- Alfaro-De la Torre, M.C.; Beaulieu, P.Y.; Tessier, A.T. In situ measurement of trace metals in lake water using the dialysis and DGT techniques. Anal. Chim. Acta 2000, 418, 53–68. [Google Scholar] [CrossRef]
- Gimpel, J.; Zhang, H.; Davison, W.; Edwards, A. In situ trace metal speciation in lake surface waters using DGT, dialysis, and filtration. Environ. Sci. Technol. 2003, 37, 138–146. [Google Scholar] [CrossRef]
- Beneš, P.; Steinnes, E. In situ dialysis for the determination of the state of trace elements in natural waters. Water Res. 1974, 8, 947–953. [Google Scholar] [CrossRef]
- Jansen, B.; Kotte, M.C.; van Wijk, A.J.; Verstraten, J.M. Comparison of diffusive gradients in thin films nd equilibrium dialysis for the determination of Al, Fe(III) and Zn complexed with dissolved organic matter. Sci. Total Environ. 2001, 277, 45–55. [Google Scholar] [CrossRef]
- Carignan, R.; Rapin, F.; Tessier, A. Sediment porewater sampling for metal analysis: A comparison of techniques. Geochim. Cosmochim. Acta 1985, 49, 2493–2497. [Google Scholar] [CrossRef]
- Vesely’, J.; Majer, V.; Kučera, J.; Havránek, V. Solid–water partitioning of elements in Czech freshwaters. Appl. Geochem. 2001, 16, 437–450. [Google Scholar] [CrossRef]
- Huerta-Diaz, M.A.; Rivera-Duarte, I.; Sañudo-Wilhelmy, S.A.; Flegal, R. Comparative distributions of size fractionated metals in pore waters sampled by in situ dialysis and whole-core sediment squeezing: Implications for diffusive flux calculations. Appl. Geochem. 2007, 22, 2509–2525. [Google Scholar] [CrossRef]
- Masing, V.; Botch, M.; Läänelaid, A. Mires of the former Soviet Union. Wetl. Ecol Manag. 2010, 18, 397–433. [Google Scholar] [CrossRef]
- Trofimova, I.E.; Balybina, A.S. Classification of climates and climatic regionalization of the West-Siberian plain. Geogr. Nat. Resour. 2014, 35, 114–122. [Google Scholar] [CrossRef]
- Raudina, T.V.; Loiko, S.V.; Lim, A.; Manasypov, R.M.; Shirokova, L.S.; Istigechev, G.I.; Kuzmina, D.M.; Kulizhsky, S.P.; Vorobyev, S.N.; Pokrovsky, O.S. Permafrost thaw and climate warming may decrease the CO2, carbon, and metal concentration in peat soil waters of the Western Siberia Lowland. Sci. Total Environ. 2018, 634, 1004–1023. [Google Scholar] [CrossRef] [PubMed]
- Manasypov, R.M.; Pokrovsky, O.S.; Kirpotin, S.N.; Shirokova, L.S. Thermokarst lake waters across the permafrost zones of western Siberia. Cryosphere 2014, 8, 1177–1193. [Google Scholar] [CrossRef]
- Pokrovsky, O.S.; Shirokova, L.S.; Zabelina, S.A.; Vorobieva, T.Y.; Moreva, O.Y.; Klimov, S.I.; Chupakov, A.V.; Shorina, N.V.; Kokryatskaya, N.M.; Audry, S.; et al. Size Fractionation of Trace Elements in a Seasonally Stratified Boreal Lake: Control of Organic Matter and Iron Colloids. Aquat. Geochem. 2012, 18, 115–139. [Google Scholar] [CrossRef]
- Shirokova, L.S.; Pokrovsky, O.S.; Moreva, O.Y.; Chupakov, A.V.; Zabelina, S.A.; Klimov, S.I.; Shorina, N.V.; Vorobieva, T.Y. Decrease of concentration and colloidal fraction of organic carbon and trace elements in response to the anomalously hot summer 2010 in a humic boreal lake. Sci. Total Environ. 2013, 463–464, 78–90. [Google Scholar] [CrossRef]
- Yeghicheyan, D.; Bossy, C.; Coz, M.B.L.; Douchet, C.; Granier, G.; Heimburger, A.; Lacan, F.; Lanzanova, A.; Rousseau, T.C.C.; Seidel, J.-L.; et al. A compilation of silicon, rare earth element and twenty-one other trace element concentrations in the natural river water reference material SLRS-5 (NRC-CNRC). Geostand. Geoanal. Res. 2013, 37, 449–467. [Google Scholar] [CrossRef]
- Gustafsson, J. Visual MINTEQ Ver. 3.1. 2014. Available online: http://vminteq.lwr.kth.se (accessed on 8 February 2020).
- Benedetti, M.F.; Milne, C.; Kinniburgh, D.; van Riemsdijk, W.; Koopal, L. Metal ion binding to humic substances: Application of the non ideal competitive adsorption model. Environ. Sci. Technol. 1995, 29, 446–457. [Google Scholar] [CrossRef]
- Kinniburgh, D.G.; Van Riemsdijk, W.H.; Koopal, L.K.; Borkovec, M.; Benedetti, M.F.; Avena, M.J. Ion binding to natural organic matter: Competition, heterogeneity, stoichiometry and thermodynamic consistency. Colloids Surf. A 1999, 151, 147–166. [Google Scholar] [CrossRef]
- Milne, C.J.; Kinniburgh, D.G.; van Riemsdijk, W.H.; Tipping, E. Generic NICA−Donnan Model Parameters for Metal-Ion Binding by Humic Substances. Environ. Sci. Technol. 2003, 37, 958–971. [Google Scholar] [CrossRef]
- Dzombak, D.A.; Morel, F.M.M. Surface Complexation Modeling: Hydrous Ferric Oxide; John Wiley & Sons: New York, NY, USA, 1990. [Google Scholar]
- Sjöstedt, C.; Gustafsson, J.-P.; Köhler, S.J. Chemical equilibrium modeling of organic acids, pH, aluminium, and iron in Swedish surface waters. Environ. Sci. Technol. 2010, 44, 8587–8593. [Google Scholar] [CrossRef]
- Downing, J.A. Emerging global role of small lakes and ponds: Little things mean a lot. Limnetica 2010, 29, 9–24. [Google Scholar] [CrossRef]
- Holgerson, M.A.; Raymond, P.A. Large Contribution to Inland Water CO2 and CH4 Emissions from Very Small Ponds. Nat. Geosci. 2016, 9, 222–226. [Google Scholar] [CrossRef]
- Arsenault, J.; Talbot, J.; Brown, L.E.; Holden, J.; Martinez-Cruz, K.; Sepulveda-Jauregui, A.; Swindles, G.T.; Wauthy, M.; Lapierre, J.-F. Biogeochemical Distinctiveness of Peatland Ponds, Thermokarst Waterbodies, and Lakes. Geophys. Res. Lett. 2022, 49, e2021GL097492. [Google Scholar] [CrossRef]
- Pokrovsky, O.S.; Manasypov, R.M.; Loiko, S.; Shirokova, L.S.; Krickov, I.A.; Pokrovsky, B.G.; Kolesnichenko, L.G.; Kopysov, S.G.; Zemtzov, V.A.; Kulizhsky, S.P.; et al. Permafrost coverage, watershed area and season control of dissolved carbon and major elements in western Siberian rivers. Biogeosciences 2015, 12, 6301–6320. [Google Scholar] [CrossRef]
- Savenko, A.V. Coprecipitation of uranium with iron(III) hydroxide formed in sea water by oxidation of iron (II). Geochem. Int. 1996, 33, 1–9. [Google Scholar]
- Savenko, A.V. Coprecipitation of manganese, copper, zinc, lead and cadmium with iron hydroxide in hydrothermal plumes (by the data of laboratory modeling). Oceanology 2001, 41, 502–507. [Google Scholar]
- Pokrovsky, O.S.; Viers, J.; Shirokova, L.S.; Shevchenko, V.P.; Filipov, A.S.; Dupré, B. Dissolved, suspended, and colloidal fluxes of organic carbon, major and trace elements in the Severnaya Dvina River and its tributary. Chem. Geol. 2010, 273, 136–149. [Google Scholar] [CrossRef]
- Pourret, O.; Dia, A.; Davranche, M.; Gruau, G.; Hénin, O.; Angée, M. Organo-Colloidal Control on Major- and Trace-Element Partitioning in Shallow Groundwaters: Confronting Ultrafiltration and Modelling. Appl. Geochem. 2007, 22, 1568–1582. [Google Scholar] [CrossRef]
- Pédrot, M.; Dia, A.; Davranche, M.; Bouhnik-Le Coz, M.; Henin, O.; Gruau, G. Insights into Colloid-Mediated Trace Element Release at the Soil/Water Interface. J. Colloid Interface Sci. 2008, 325, 187–197. [Google Scholar] [CrossRef]
- Guénet, H.; Davranche, M.; Vantelon, D.; Gigault, J.; Prévost, S.; Taché, O.; Jaksch, S.; Pédrot, M.; Dorcet, V.; Boutier, A.; et al. Characterization of Iron–Organic Matter Nano-Aggregate Networks through a Combination of SAXS/SANS and XAS Analyses: Impact on As Binding. Environ. Sci. Nano 2017, 4, 938–954. [Google Scholar] [CrossRef]
- Ratié, G.; Vantelon, D.; Lotfi Kalahroodi, E.; Bihannic, I.; Pierson-Wickmann, A.C.; Davranche, M. Iron Speciation at the Riverbank Surface in Wetland and Potential Impact on the Mobility of Trace Metals. Sci. Total Environ. 2019, 651, 443–455. [Google Scholar] [CrossRef] [PubMed]
- Vantelon, D.; Davranche, M.; Marsac, R.; Fontaine, C.L.; Guénet, H.; Jestin, J.; Campaore, G.; Beauvois, A.; Briois, V. Iron Speciation in Iron–Organic Matter Nanoaggregates: A Kinetic Approach Coupling Quick-EXAFS and MCR-ALS Chemometrics. Environ. Sci. Nano 2019, 6, 2641–2651. [Google Scholar] [CrossRef]


Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Manasypov, R.M.; Lim, A.G.; Kriсkov, I.V.; Raudina, T.V.; Kurashev, D.G.; Shirokova, L.S.; Pokrovsky, O.S. Colloids in Thermokarst Lakes along a Permafrost and Climate Gradient of Permafrost Peatlands in Western Siberia Using In Situ Dialysis Procedure. Water 2023, 15, 1783. https://doi.org/10.3390/w15091783
Manasypov RM, Lim AG, Kriсkov IV, Raudina TV, Kurashev DG, Shirokova LS, Pokrovsky OS. Colloids in Thermokarst Lakes along a Permafrost and Climate Gradient of Permafrost Peatlands in Western Siberia Using In Situ Dialysis Procedure. Water. 2023; 15(9):1783. https://doi.org/10.3390/w15091783
Chicago/Turabian StyleManasypov, Rinat M., Artem G. Lim, Ivan V. Kriсkov, Tatiana V. Raudina, Danil G. Kurashev, Liudmila S. Shirokova, and Oleg S. Pokrovsky. 2023. "Colloids in Thermokarst Lakes along a Permafrost and Climate Gradient of Permafrost Peatlands in Western Siberia Using In Situ Dialysis Procedure" Water 15, no. 9: 1783. https://doi.org/10.3390/w15091783





