Growth and Muscle Quality of Grass Carp (Ctenopharyngodon idella) in In-Pond Raceway Aquaculture and Traditional Pond Culture
Abstract
:1. Introduction
2. Materials and Methods
2.1. Experimental Layout Set-Up
2.2. Water Quality Measurement
2.3. Sample Collection and Analysis
2.4. Microbial Diversity in Pond Water
2.5. Statistical Analysis
3. Results
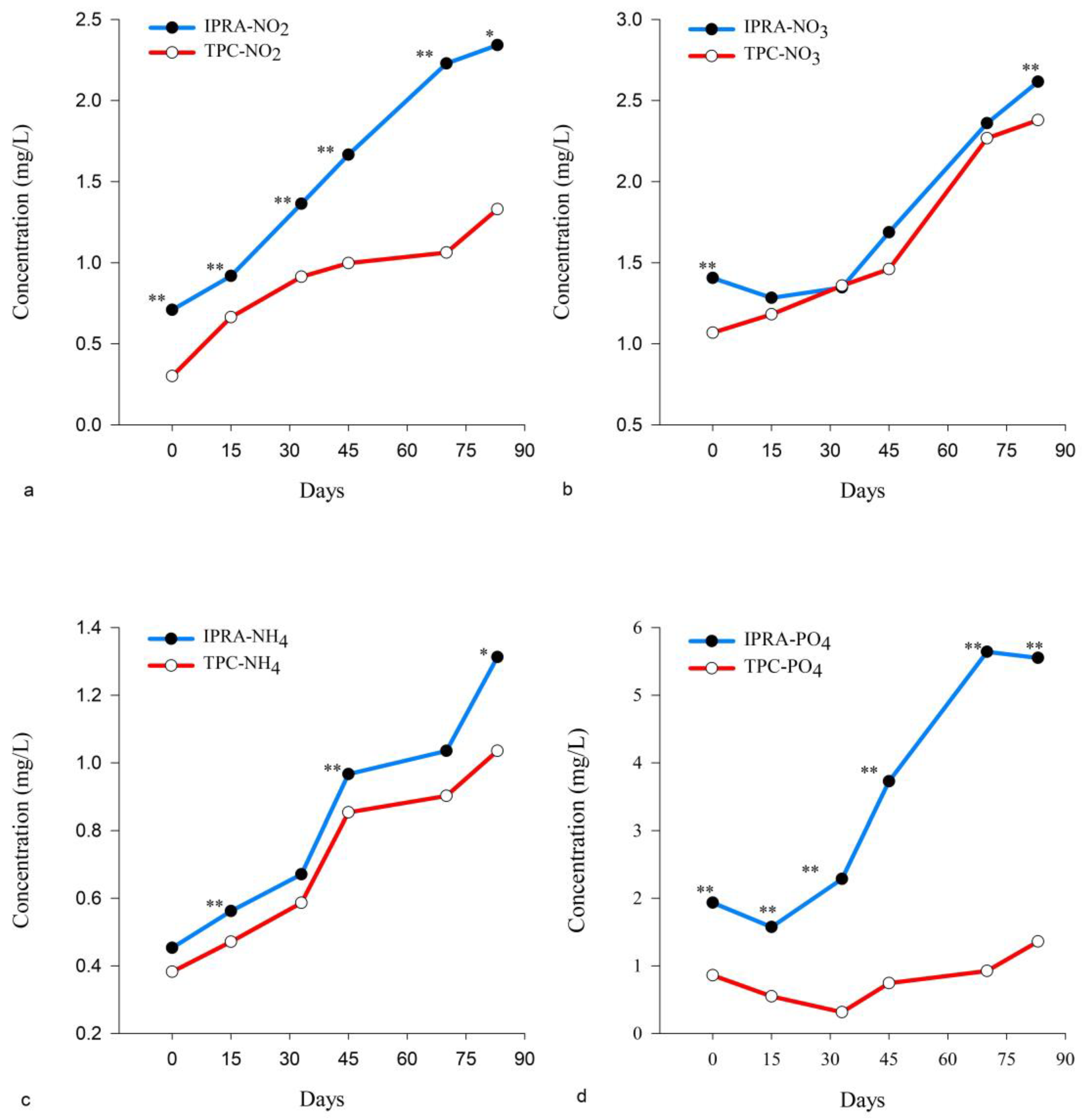
3.1. Water Quality Analysis
3.2. Growth Performance
3.3. Nutrient Composition of Muscle
3.4. Muscle Texture Profiles
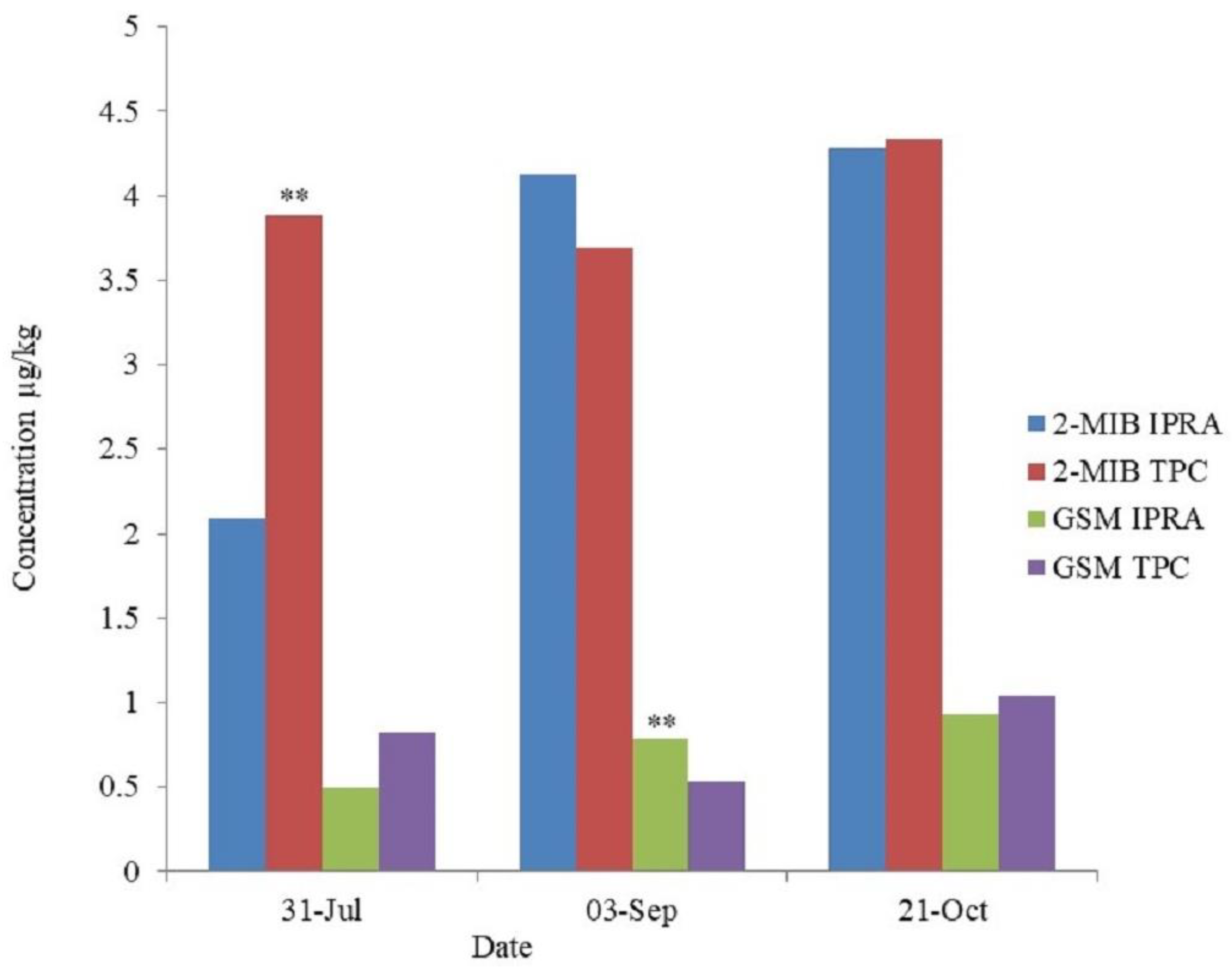
3.5. 2-MIB and Geosmin in Muscle
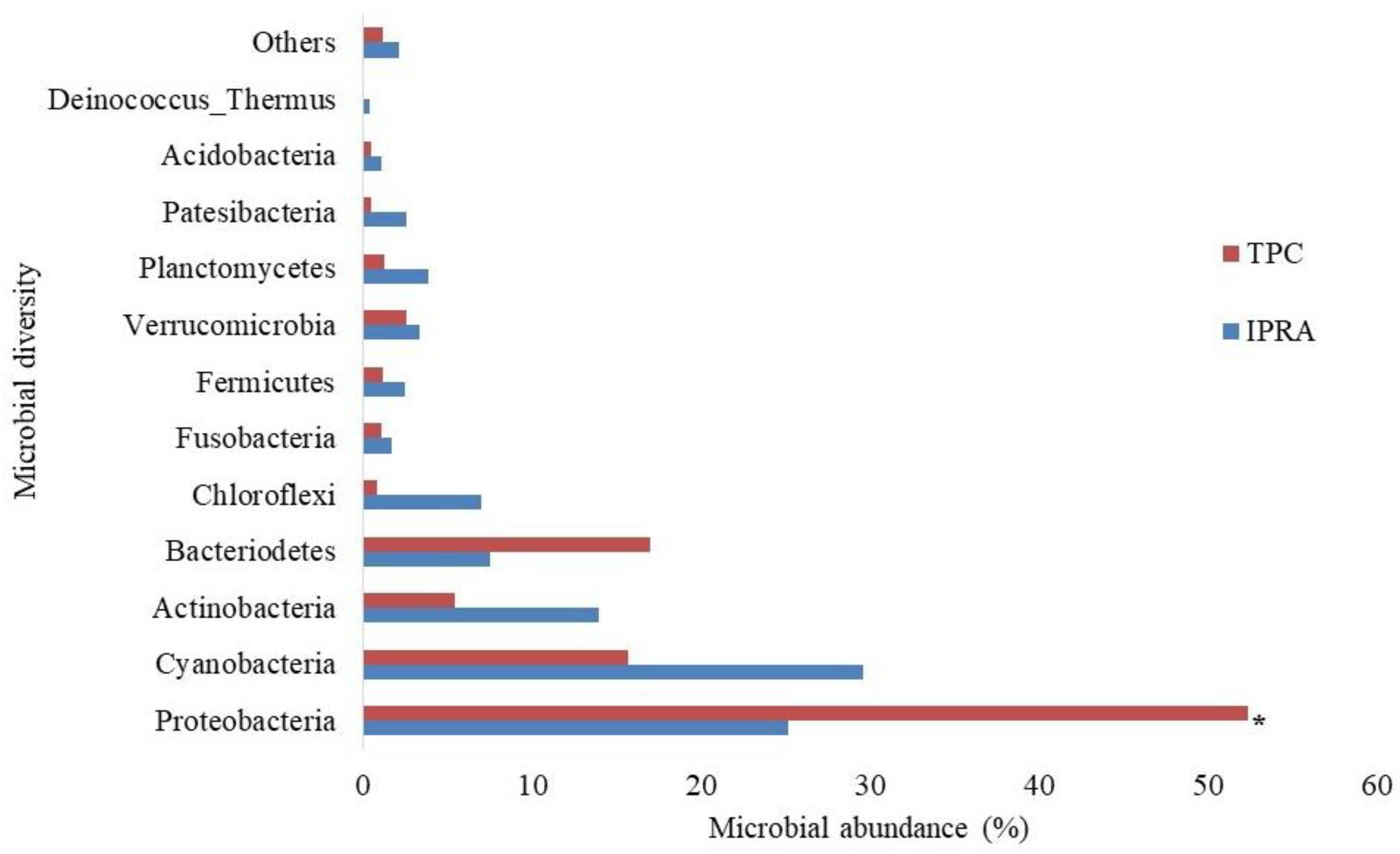
3.6. Microbial Diversity in Pond Water
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Subasinghe, R.; Soto, D.; Jia, J. Global aquaculture and its role in sustainable development. Rev. Aquac. 2009, 1, 2–9. [Google Scholar] [CrossRef]
- Crab, R.; Defoirdt, T.; Bossier, P.; Verstraete, W. Biofloc technology in aquaculture: Beneficial effects and future challenges. Aquaculture 2012, 356–357, 351–356. [Google Scholar] [CrossRef]
- Henriksson, P.J.G.; Troell, M.; Banks, L.K.; Belton, B.; Beveridge, M.C.M.; Klinger, D.H.; Pelletier, N.; Phillips, M.J.; Tran, N. Interventions for improving the productivity and environmental performance of global aquaculture for future food security. One Earth 2021, 4, 1220–1232. [Google Scholar] [CrossRef]
- Xie, C.; Li, J.; Li, D.; Shen, Y.; Gao, Y.; Zhang, Z. Grass carp: The fish that feeds half of china. In Aquaculture in China; Gui, J.-F., Tang, Q., Li, Z., Liu, J., De Silva, S.S., Eds.; John Wiley & Sons Ltd.: Chichester, UK, 2018; pp. 93–115. [Google Scholar]
- FAO. The State of World Fisheries and Aquaculture 2022. Towards Blue Transformation; FAO: Rome, Italy, 2022. [Google Scholar]
- Gisbert, E.; Mozanzadeh, M.T.; Kotzamanis, Y.; Estévez, A. Weaning wild flathead grey mullet (Mugil cephalus) fry with diets with different levels of fish meal substitution. Aquaculture 2016, 462, 92–100. [Google Scholar] [CrossRef]
- Zhao, H.; Xia, J.; Zhang, X.; He, X.; Li, L.; Tang, R.; Chi, W.; Li, D. Diet Affects Muscle Quality and Growth Traits of Grass Carp (Ctenopharyngodon idellus): A Comparison between Grass and Artificial Feed. Front. Physiol. 2018, 9, 283. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Xu, P.; Nie, Z.; Li, Q.; Shao, N.; Xu, G. Growth, digestive enzymes activities, serum biochemical parameters and antioxidant status of juvenile genetically improved farmed tilapia (Oreochromis niloticus) reared at different stocking densities in in-pond raceway recirculating culture system. Aquacult. Res. 2019, 50, 1338–1347. [Google Scholar] [CrossRef]
- Fatima, S.; Komal, W.; Manzoor, F.; Latif, A.A.; Liaqat, R.; Ameen, S.; Janjua, R.S. Analysis of the growth performance, stress, profile of fatty acids and amino acids and cortisol in Tilapia (Oreochromis niloticus), cultured at high stocking density using in-pond raceway system. Saudi J. Biol. Sci. 2021, 28, 7422–7431. [Google Scholar] [CrossRef]
- Martin, T.E. Are microhabitat preferences of coexisting species under selection and adaptive? Ecology 1998, 79, 656–670. [Google Scholar] [CrossRef]
- Nielsen, J.; Hyldig, G.; Larsen, E. ‘Eating Quality’ of Fish—A Review. J. Aquat. Food Prod. Technol. 2002, 11, 125–141. [Google Scholar] [CrossRef]
- Listrat, A.; Lebret, B.; Louveau, I.; Astruc, T.; Bonnet, M.; Lefaucheur, L.; Picard, B.; Bugeon, J. How Muscle Structure and Composition Influence Meat and Flesh Quality. Sci. World J. 2016, 2016, 3182746. [Google Scholar] [CrossRef] [PubMed]
- Tang, Y.; Zhao, L.; Cheng, Y.; Yang, Y.; Sun, Y.; Liu, Q. Control of cyanobacterial blooms in different polyculture patterns of filter feeders and effects of these patterns on water quality and microbial community in aquacultural ponds. Aquaculture 2021, 542, 736913. [Google Scholar] [CrossRef]
- Alamri, S.A.; Mohamed, Z.A. Selective inhibition of toxic cyanobacteria by β-carboline-containing bacterium Bacillus flexus isolated from Saudi freshwaters. Saudi J. Biol. Sci. 2013, 20, 357–363. [Google Scholar] [CrossRef] [PubMed]
- Cai, L.; Tong, F.; Tang, T.; Ao, Z.; Wei, Z.; Yang, F.; Shu, Y.; Liu, S.; Mai, K. Comparative evaluation of nutritional value and flavor quality of muscle in triploid and diploid common carp: Application of genetic improvement in fish quality. Aquaculture 2021, 541, 736780. [Google Scholar] [CrossRef]
- Wen, J.; Jiang, W.; Feng, L.; Kuang, S.; Jiang, J.; Tang, L.; Zhou, X.; Liu, Y. The influence of graded levels of available phosphorus on growth performance, muscle antioxidant and flesh quality of young grass carp (Ctenopharyngodon idella). Anim. Nutr. 2015, 1, 77–84. [Google Scholar] [CrossRef]
- Granada, L.; Lopes, S.; Novais, S.C.; Lemos, M.F.L. Modelling integrated multi-trophic aquaculture: Optimizing a three trophic level system. Aquaculture 2018, 495, 90–97. [Google Scholar] [CrossRef]
- Zhang, X.; Wang, J.; Tang, R.; He, X.; Li, L.; Takagi, Y.; Li, D. Improvement of Muscle Quality of Grass Carp (Ctenopharyngodon idellus) With a Bio-Floating Bed in Culture Ponds. Front. Physiol. 2019, 10, 683. [Google Scholar] [CrossRef]
- Zhang, X.; Shen, Z.; Qi, T.; Xi, R.; Liang, X.; Li, L.; Tang, R.; Li, D. Slight Increases in Salinity Improve Muscle Quality of Grass Carp (Ctenopharyngodon idellus). Fishes 2021, 6, 7. [Google Scholar] [CrossRef]
- Edwards, P. Aquaculture environment interactions: Past, present and likely future trends. Aquaculture 2015, 447, 2–14. [Google Scholar] [CrossRef]
- Dauda, A.B.; Ajadi, A.; Tola-Fabunmi, A.S.; Akinwole, A.O. Waste production in aquaculture: Sources, components and managements in different culture systems. Aquac. Fish. 2019, 4, 81–88. [Google Scholar] [CrossRef]
- Gutierrez, R.; Itayama, T.; Iwami, N.; Whangchai, N. Analysis of Geosmin and 2-Methylisoborneol Off-flavors In Tilapia Cage-Cultures in Thailand. In Proceedings of the 2nd MJU-Phrae National Research Conference, Phrae, Thailand, 1–2 September 2011. [Google Scholar]
- Hathurusingha, P.I.; Davey, K.R. A predictive model for taste taint accumulation in Recirculating Aquaculture Systems (RAS) farmed-fish–demonstrated with geosmin (GSM) and 2-methylisoborneol (MIB). Ecol. Model. 2014, 291, 242–249. [Google Scholar] [CrossRef]
- Qin, Y.; Hou, J.; Deng, M.; Liu, Q.; Wu, C.; Ji, Y.; He, X. Bacterial abundance and diversity in pond water supplied with different feeds. Sci. Rep. 2016, 6, 35232. [Google Scholar] [CrossRef] [PubMed]
- Lukassen, M.B.; de Jonge, N.; Bjerregaard, S.M.; Podduturi, R.; Jorgensen, N.O.G.; Petersen, M.A.; David, G.S.; da Silva, R.J.; Nielsen, J.L. Microbial Production of the Off-Flavor Geosmin in Tilapia Production in Brazilian Water Reservoirs: Importance of Bacteria in the Intestine and Other Fish-Associated Environments. Front. Microbiol. 2019, 10, 2447. [Google Scholar] [CrossRef] [PubMed]
- Brown, T.W.; Tucker, C.S. Pumping performance of a slow-rotating paddlewheel for split-pond aquaculture systems. N. Am. J. Aquacult. 2013, 75, 153–158. [Google Scholar] [CrossRef]
- Chopin, T.; Buschmann, A.H.; Halling, C.; Troell, M.; Kautsky, N.; Neori, A.; Kraemer, G.P.; Zertuche-González, J.A.; Yarish, C.; Neefus, C. Integrating seaweeds into marine aquaculture systems: A key toward sustainability. J. Phycol. 2001, 37, 975–986. [Google Scholar] [CrossRef]
- Schrader, K.K.; Tucker, C.S.; Brown, T.W.; Whitis, G.N. Earthy and Musty Off-Flavor Episodes in Catfish Split-Pond Aquaculture Systems. N. Am. J. Aquacult. 2018, 80, 26–41. [Google Scholar] [CrossRef]
- Wang, H.; Zhu, Y.; Zhang, J.; Wang, X.; Shi, W. Study on changes in the quality of grass carp in the process of postmortem. J. Food Biochem. 2018, 42, e12683. [Google Scholar] [CrossRef]
- Brown, T.W.; Chappell, J.A.; Boyd, C.E. A commercial-scale, in-pond raceway system for Ictalurid catfish production. Aquacult. Eng. 2011, 44, 72–79. [Google Scholar] [CrossRef]
- Zhang, X.; Zheng, W.; Zhang, H.; Chai, Y.; Ruan, G. Comparison of Muscle Quality of the Yellow Catfish Cultured in In-Pond Raceway Systems and Traditional Ponds. Water 2022, 14, 1223. [Google Scholar] [CrossRef]
- Akinwole, A.; Dauda, A.; Ololade, A. Haematological response of Clarias gariepinus juveniles reared in treated wastewater after waste solids removal using alum or Moringa oleifera seed powder. Int. J. Aquacult. 2016, 6, 1–8. [Google Scholar]
- Zhang, J.; Kaneko, G.; Sun, J.; Wang, G.; Xie, J.; Tian, J.; Li, Z.; Gong, W.; Zhang, K.; Xia, Y.; et al. Key Factors Affecting the Flesh Flavor Quality and the Nutritional Value of Grass Carp in Four Culture Modes. Foods 2021, 10, 2075. [Google Scholar] [CrossRef]
- Wang, Z.; Xu, Y.; Shao, J.; Wang, J.; Li, R. Genes associated with 2-methylisoborneol biosynthesis in cyanobacteria: Isolation, characterization, and expression in response to light. PLoS ONE 2011, 6, e18665. [Google Scholar] [CrossRef] [PubMed]
- Fadrosh, D.W.; Ma, B.; Gajer, P.; Sengamalay, N.; Ott, S.; Brotman, R.M.; Ravel, J. An improved dual-indexing approach for multiplexed 16S rRNA gene sequencing on the Illumina MiSeq platform. Microbiome 2014, 2, 6. [Google Scholar] [CrossRef] [PubMed]
- Johnston, I.A. Genetic and environmental determinants of muscle growth patterns. In Fish Physiology; Elsevier: Amsterdam, The Netherlands, 2001; Volume 18, pp. 141–186. [Google Scholar]
- Hua, K.; Koppe, W.; Fontanillas, R. Effects of dietary protein and lipid levels on growth, body composition and nutrient utilization of Channa striata. Aquaculture 2019, 501, 368–373. [Google Scholar] [CrossRef]
- Liu, L.; Zhou, Y.; Wu, J.; Zhang, W.; Abbas, K.; Xu-Fang, L.; Luo, Y. Supplemental graded levels of neutral phytase using pretreatment and spraying methods in the diet of grass carp, Ctenopharyngodon idellus. Aquacult. Res. 2014, 45, 1932–1941. [Google Scholar] [CrossRef]
- Masser, M.P. In-pond raceways. In Aquaculture Production Systems; John Wiley & Sons, Inc.: Hoboken, NJ, USA, 2012; pp. 387–394. [Google Scholar]
- Yuan, J.; Ni, M.; Liu, M.; Wang, H.; Zhang, C.; Mi, G.; Gu, Z. Analysis of the growth performances, muscle quality, blood biochemistry and antioxidant status of Micropterus salmoides farmed in in-pond raceway systems versus usual-pond systems. Aquaculture 2019, 511, 734241. [Google Scholar] [CrossRef]
- Brown, T.W.; Boyd, C.E.; Chappell, J.A. Approximate Water and Chemical Budgets for an Experimental, In-pond Raceway System. J. World Aquacult. Soc. 2012, 43, 526–537. [Google Scholar] [CrossRef]
- Wang, Y.; Xu, G.; Nie, Z.; Shao, N.; Li, Q.; Xu, P. Growth Performance of Bluntnose Black Bream, Channel Catfish, Yellow Catfish, and Largemouth Bass Reared in the In-Pond Raceway Recirculating Culture System. N. Am. J. Aquacult. 2019, 81, 153–159. [Google Scholar] [CrossRef]
- Liu, Q.; Hou, Z.; Wen, H.; Li, J.; He, F.; Wang, J.; Guan, B.; Wang, Q. Effect of stocking density on water quality and (Growth, Body Composition and Plasma Cortisol Content) performance of pen-reared rainbow trout (Oncorhynchus mykiss). J. Ocean. Univ. China 2016, 15, 667–675. [Google Scholar] [CrossRef]
- Li, W.; Cheng, X.; Xie, J.; Wang, Z.; Yu, D. Hydrodynamics of an in-pond raceway system with an aeration plug-flow device for application in aquaculture: An experimental study. R. Soc. Open Sci. 2019, 6, 182061. [Google Scholar] [CrossRef]
- Ni, M.; Liu, M.; Lou, J.; Mi, G.; Yuan, J.; Gu, Z. Stocking density alters growth performance, serum biochemistry, digestive enzymes, immune response, and muscle quality of largemouth bass (Micropterus salmoides) in in-pond raceway system. Fish Physiol. Biochem. 2021, 47, 1243–1255. [Google Scholar] [CrossRef]
- Yang, H.; He, J.; Lv, M.; Huang, G.; Wen, L.; Bi, X.; Hu, T.; Ma, H. Preliminary effects of dietary protein levels on muscle quality and digestive enzyme activities in GIFT-Oreochromis niloticus. Aquac. Stud. 2018, 18, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Pyz-Łukasik, R.; Chałabis-Mazurek, A.; Gondek, M. Basic and functional nutrients in the muscles of fish: A review. Int. J. Food Prop. 2020, 23, 1941–1950. [Google Scholar] [CrossRef]
- Younis, E.M.; Al-Quffail, A.S.; Al-Asgah, N.A.; Abdel-Warith, A.A.; Al-Hafedh, Y.S. Effect of dietary fish meal replacement by red algae, Gracilaria arcuata, on growth performance and body composition of Nile tilapia Oreochromis niloticus. Saudi J. Biol. Sci. 2018, 25, 198–203. [Google Scholar] [CrossRef] [PubMed]
- Pyz-Łukasik, R.; Paszkiewicz, W. Species Variations in the Proximate Composition, Amino Acid Profile, and Protein Quality of the Muscle Tissue of Grass Carp, Bighead Carp, Siberian Sturgeon, and Wels Catfish. J. Food Qual. 2018, 2018, 2625401. [Google Scholar] [CrossRef]
- He, Y.; Yu, H.; Zhao, H.; Zhu, H.; Zhang, Q.; Wang, A.; Shen, Y.; Xu, X.; Li, J. Transcriptomic analysis to elucidate the effects of high stocking density on grass carp (Ctenopharyngodon idella). BMC Genom. 2021, 22, 620. [Google Scholar] [CrossRef]
- Orban, E.; Lena, G.D.; Nevigato, T.; Casini, I.; Santaroni, G.; Marzetti, A.; Caproni, R. Quality characteristics of sea bass intensively reared and from lagoon as affected by growth conditions and the aquatic environment. J. Food Sci. 2002, 67, 542–546. [Google Scholar] [CrossRef]
- Grigorakis, K.; Taylor, K.; Alexis, M. Organoleptic and volatile aroma compounds comparison of wild and cultured gilthead sea bream (Sparus aurata): Sensory differences and possible chemical basis. Aquaculture 2003, 225, 109–119. [Google Scholar] [CrossRef]
- Valente, L.M.P.; Cornet, J.; Donnay-Moreno, C.; Gouygou, J.P.; Bergé, J.P.; Bacelar, M.; Escórcio, C.; Rocha, E.; Malhão, F.; Cardinal, M. Quality differences of gilthead sea bream from distinct production systems in Southern Europe: Intensive, integrated, semi-intensive or extensive systems. Food Control 2011, 22, 708–717. [Google Scholar] [CrossRef]
- Flos, R.; Reig, L.; Oca, J.; Ginovart, M. Influence of marketing and different land-based systems on gilthead sea bream (Sparus aurata) quality. Aquacult. Int. 2002, 10, 189–206. [Google Scholar] [CrossRef]
- Rasmussen, R.S. Quality of farmed salmonids with emphasis on proximate composition, yield and sensory characteristics. Aquacult. Res. 2001, 32, 767–786. [Google Scholar] [CrossRef]
- Wang, Y.; Lu, Y.; Zhang, Y.; Ning, Z.; Li, Y.; Zhao, Q.; Lu, H.; Huang, R.; Xia, X.; Feng, Q.; et al. The draft genome of the grass carp (Ctenopharyngodon idellus) provides insights into its evolution and vegetarian adaptation. Nat. Genet. 2015, 47, 625–631. [Google Scholar] [CrossRef] [PubMed]
- Xu, J.; Feng, L.; Jiang, W.-D.; Wu, P.; Liu, Y.; Jiang, J.; Kuang, S.-Y.; Tang, L.; Zhou, X.-Q. Different dietary protein levels affect flesh quality, fatty acids and alter gene expression of Nrf2-mediated antioxidant enzymes in the muscle of grass carp (Ctenopharyngodon idella). Aquaculture 2018, 493, 272–282. [Google Scholar] [CrossRef]
- Fernandez, X.; MÅGÅRd, M.; Tornberg, E.V.A. Glycolytic Potential in Porcine Longissimus Muscle before and after Transport: An in Vivo Study. J. Muscle Foods 1992, 3, 83–89. [Google Scholar] [CrossRef]
- Bee, G.; Anderson, A.L.; Lonergan, S.M.; Huff-Lonergan, E. Rate and extent of pH decline affect proteolysis of cytoskeletal proteins and water-holding capacity in pork. Meat Sci. 2007, 76, 359–365. [Google Scholar] [CrossRef]
- El Rammouz, R.; Abboud, J.; Abboud, M.; El Mur, A.; Yammine, S.; Jammal, B. pH, rigor mortis and physical properties of fillet in fresh water fish: The case of rainbow trout (Oncorynchus mykiss). J. Appl.Sci. Res. 2013, 9, 5746–5755. [Google Scholar]
- Fuentes, A.; Fernández-Segovia, I.; Serra, J.A.; Barat, J.M. Comparison of wild and cultured sea bass (Dicentrarchus labrax) quality. Food Chem. 2010, 119, 1514–1518. [Google Scholar] [CrossRef]
- Cheng, J.H.; Sun, D.W.; Han, Z.; Zeng, X.A. Texture and Structure Measurements and Analyses for Evaluation of Fish and Fillet Freshness Quality: A Review. Compr. Rev. Food Sci. Food Saf. 2014, 13, 52–61. [Google Scholar] [CrossRef]
- Andersen, U.B.; Thomassen, M.S.; Rørå, A.M.B. Texture properties of farmed rainbow trout (Oncorhynchus mykiss): Effects of diet, muscle fat content and time of storage on ice. J. Sci. Food Agric. 1997, 74, 347–353. [Google Scholar] [CrossRef]
- Pearce, K.L.; Rosenvold, K.; Andersen, H.J.; Hopkins, D.L. Water distribution and mobility in meat during the conversion of muscle to meat and ageing and the impacts on fresh meat quality attributes—A review. Meat Sci. 2011, 89, 111–124. [Google Scholar] [CrossRef]
- Refaey, M.M.; Li, D.; Tian, X.; Zhang, Z.; Zhang, X.; Li, L.; Tang, R. High stocking density alters growth performance, blood biochemistry, intestinal histology, and muscle quality of channel catfish Ictalurus punctatus. Aquaculture 2018, 492, 73–81. [Google Scholar] [CrossRef]
- Taylor, R.; Fjaera, S.; Skjervold, P. Salmon fillet texture is determined by myofiber-myofiber and myofiber-myocommata attachment. J. Food Sci. 2002, 67, 2067–2071. [Google Scholar] [CrossRef]
- Tucker, C.S. Off-flavor problems in aquaculture. Rev. Fish. Sci. 2000, 8, 45–88. [Google Scholar] [CrossRef]
- Guttman, L.; van Rijn, J. 2-Methylisoborneol and geosmin uptake by organic sludge derived from a recirculating aquaculture system. Water Res. 2009, 43, 474–480. [Google Scholar] [CrossRef] [PubMed]
- Robertson, R.F.; Hammond, A.; Jauncey, K.; Beveridge, M.C.M.; Lawton, L.A. An investigation into the occurrence of geosmin responsible for earthy–musty taints in UK farmed rainbow trout, Onchorhynchus mykiss. Aquaculture 2006, 259, 153–163. [Google Scholar] [CrossRef]
- Persson, P.-E. Sensory properties and analysis of two muddy odour compounds, geosmin and 2-methylisoborneol, in water and fish. Water Res. 1980, 14, 1113–1118. [Google Scholar] [CrossRef]
- Gutierrez, R.; Whangchai, N.; Sompong, U.; Prarom, W.; Iwami, N.; Itayama, T.; Nomura, N.; Sugiura, N. Off-flavour in Nile tilapia (Oreochromis niloticus) cultured in an integrated pond-cage culture system. Maejo Int. J. Sci. Technol. 2013, 7, 1. [Google Scholar]
- Vallod, D.; Cravedi, J.; Hillenweck, A.; Robin, J. Analysis of the off-flavor risk in carp production in ponds in Dombes and Forez (France). Aquacult. Int. 2007, 15, 287–298. [Google Scholar] [CrossRef]
- Lukassen, M.B.; Saunders, A.M.; Sindilariu, P.-D.; Nielsen, J.L. Quantification of novel geosmin-producing bacteria in aquaculture systems. Aquaculture 2017, 479, 304–310. [Google Scholar] [CrossRef]
- Varga, D.; Sándor, Z.; Hancz, C.; Csengeri, I.; Jeney, Z.; Papp, Z. Off-flavour compounds in common carp (Cyprinus carpio L.) flesh in context of type of fish pond. Acta Aliment. 2015, 44, 311–315. [Google Scholar] [CrossRef]
| Water Quality Indicators | IPRA | TPC | ||||
|---|---|---|---|---|---|---|
| Min | Max | Mean ± SD | Min | Max | Mean ± SD | |
| Temperature (°C) | 22.00 | 32.60 | 26.9 ± 4.29 | 22.80 | 34.00 | 27.4 ± 4.58 |
| Dissolve oxygen (mg/L) | 6.50 | 8.10 | 7.4 ± 0.63 | 6.90 | 8.00 | 7.7 ± 0.18 |
| pH | 7.20 | 7.60 | 7.4 ± 0.12 | 7.50 | 7.90 | 7.7 ± 0.18 |
| Total nitrogen (mg/L) | 3.05 | 9.51 | 5.69 ± 2.33 | 2.01 | 8.58 | 5.13 ± 2.65 |
| Total phosphorus (mg/L) | 2.11 | 7.71 | 4.32 ± 2.29 | 0.66 | 2.67 | 1.35 ± 1.00 |
| CODMn (mg/L) | 4.62 | 13.22 | 10.03 ± 4.79 | 6.39 | 10.96 | 7.74 ± 2.24 |
| Chlorophyll-a (mg/L) | 112.06 | 441.09 | 268 ± 155.41 | 80.96 | 351.49 | 173.91 ± 116.17 |
| Total soluble solid (mg/L) | 57.50 | 82.49 | 71.83 ± 50.0 | 64.67 | 108.90 | 85.85 ± 42.90 |
| Instruments | Used Conditions |
|---|---|
| Solid-phase microextraction (SPME) | Volume of vial 15 mL sample volume 5 mL |
| Gas Chromatography–Mass Spectrophotometry: 6890N Network (Agilent, Santa Clara, CA, USA) | Extraction time and temperature: 20 min and 50 °C |
| Detector: 5975 insert XL (Agilent, Santa Clara, CA, USA) | Desorption time: 5 min |
| Column: DB-WAX (30 m × 250 µm × 0.25 µm) | Injector temperature: 250 °C Oven temperature: 60 °C (2.5 min), 250 °C (8 °C/min) |
| Fiber SPME: Stable Flex 65µm DVB/ | Gas flow: He 1 mL/min Ionization voltage: 70 eV |
| PDMS/DVB (Supelco, Bellefonte, PA, USA) |
| IPRA | TPC | |
|---|---|---|
| (Means ± SD) | (Means ± SD) | |
| Stocking weight (Kg.) | 0.42 ± 0.02 | 2.72 ± 0.14 |
| Weight (Kg.) | 0.86 ± 0.03 | 3.28 ± 0.02 * |
| Weight gain rate (%) | 117.93 ± 16.79 ** | 22.34 ± 4.85 |
| Specific growth rate (%/d) | 1.35 ± 0.20 | 1.67 ± 0.87 |
| Length (cm) | 34.25 ± 0.50 | 53.05 ± 3.80 |
| Visceral mass ratio (VMR) | 11.82 ± 1.03 | 11.5 ± 1.06 |
| Hepato somatic index (HSI) | 2.89 ± 0.18 | 2.50 ± 0.91 |
| Condition factor (CF g/cm3) | 2.14 ± 0.075 | 2.21 ± 0.52 |
| IPRA | TPC | |
|---|---|---|
| (n = 24) | (n = 27) | |
| Moisture% | 79.64 ± 0.81 | 78.49 ± 2.33 |
| Ash% | 1.469 ± 0.24 | 1.326 ± 0.20 |
| Crude lipid (CL%) | 1.707 ± 0.47 * | 1.383 ± 0.21 |
| Crude protein (CP%) | 18.66 ± 1.93 | 18.71 ± 0.94 |
| pH, WHC, and Texture Profile Attributes | IPRA | TPC |
|---|---|---|
| pH | 6.26 ± 0.07 | 6.34 ± 0.22 |
| Water Holding Capacity (%) | 0.25 ± 0.07 | 0.24 ± 0.068 |
| Hardness | 411.75 ± 183.27 | 582.52 ± 398.45 |
| Springiness | 0.51 ± 0.04 | 0.50 ± 0.07 |
| Stickiness | 4.02 ± 1.78 | 4.17 ± 2.14 |
| Chewiness | 122.67 ± 49.82 | 180.28 ± 142.29 |
| Adhesiveness | 237.25 ± 91.71 | 344.14 ± 243.82 * |
| Cohesiveness | 0.59 ± 0.05 | 0.59 ± 0.06 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gharti, K.; Yan, L.; Li, K.; Boonpeng, N.; Liu, L. Growth and Muscle Quality of Grass Carp (Ctenopharyngodon idella) in In-Pond Raceway Aquaculture and Traditional Pond Culture. Water 2023, 15, 1771. https://doi.org/10.3390/w15091771
Gharti K, Yan L, Li K, Boonpeng N, Liu L. Growth and Muscle Quality of Grass Carp (Ctenopharyngodon idella) in In-Pond Raceway Aquaculture and Traditional Pond Culture. Water. 2023; 15(9):1771. https://doi.org/10.3390/w15091771
Chicago/Turabian StyleGharti, Kamala, Li Yan, Kang Li, Nattida Boonpeng, and Liping Liu. 2023. "Growth and Muscle Quality of Grass Carp (Ctenopharyngodon idella) in In-Pond Raceway Aquaculture and Traditional Pond Culture" Water 15, no. 9: 1771. https://doi.org/10.3390/w15091771





