Arsenic in Drinking Water and Diabetes
Abstract
1. Introduction—Water Quality and Importance
2. Health Impacts of Arsenic in Drinking Water
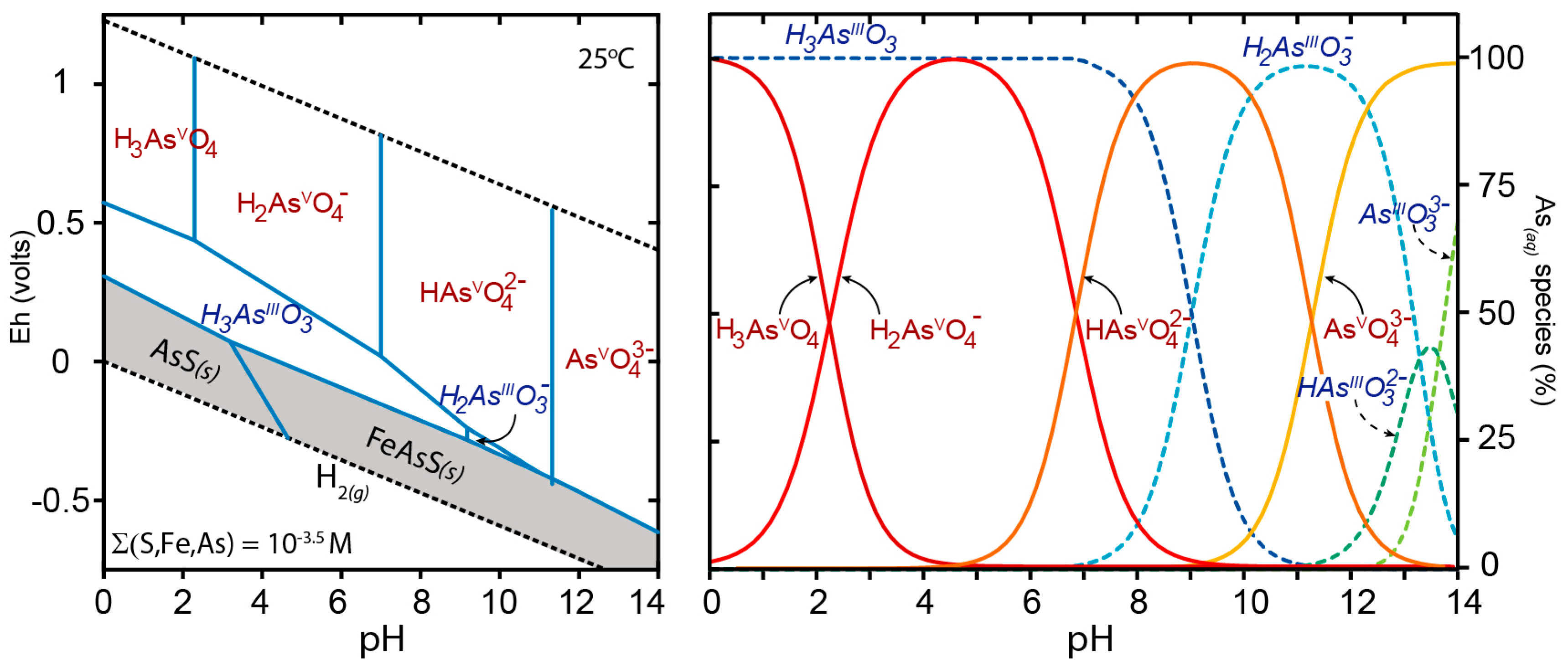
3. Arsenic Distribution in the Environment
4. Diabetes and Arsenic
4.1. Diabetes Types and Risk Factors
4.2. Epidemiological Link between iAs Exposure and Diabetes
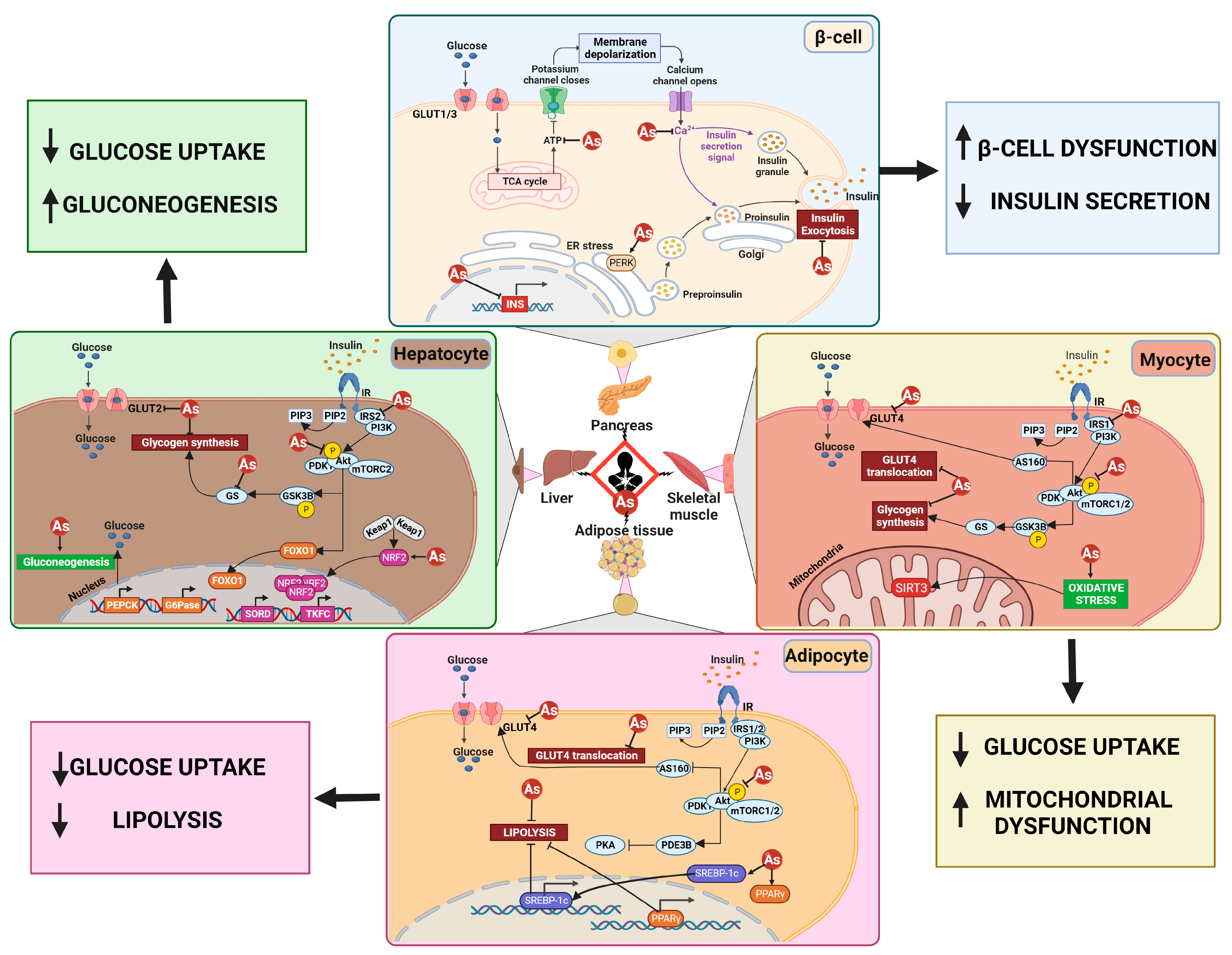
4.3. Mechanisms Associated with iAs-Induced Diabetogenesis
4.4. Future Research Needs
5. Regulation of Arsenic in Drinking Water
6. Approaches to Removal of Arsenic from Drinking Water
6.1. Technologies for Public Water Utilities
6.1.1. Blending
6.1.2. Coagulation/Filtration
6.1.3. Oxidation/Filtration
6.1.4. Metal Oxides
6.1.5. Anion Exchange Resins
6.1.6. Enhanced Lime Softening
6.1.7. Nanofiltration and Reverse Osmosis
6.2. Home Treatments
6.2.1. Distillation
6.2.2. Reverse Osmosis
6.2.3. Iron Filters
6.3. Summary—Challenges to Removal of Arsenic from Drinking Water
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- World Economic Forum. “The Global Risks Report 2021”. Available online: https://www.weforum.org/agenda/2021/01/global-risks-report-2021/ (accessed on 25 April 2023).
- World Health Organization; United Nations Children’s Fund. Progress on Household Drinking Water, Sanitation and Hygiene 2000–2017: Special Focus on Inequalities. World Health Organization. 2019. Available online: https://apps.who.int/iris/handle/10665/329370 (accessed on 25 April 2023).
- Li, P.; Karunanidhi, D.; Subramani, T.; Srinivasamoorthy, K. Sources and Consequences of Groundwater Contamination. Arch. Environ. Contam. Toxicol. 2021, 80, 1–10. [Google Scholar] [CrossRef]
- WHO; FAO; IAEA. Trace Elements in Human Nutrition and Health; World Health Organization: Geneva, Switzerland, 1996; Available online: https://apps.who.int/iris/handle/10665/37931 (accessed on 25 April 2023).
- Tchounwou, P.B.; Yedjou, C.G.; Patlolla, A.K.; Sutton, D.J. Heavy Metal Toxicity and the Environment. In Molecular, Clinical and Environmental Toxicology. Experientia Supplementum; Luch, A., Ed.; Springer: Basel, Switzerland, 2012; Volume 101. [Google Scholar] [CrossRef]
- Waseem, A.; Arshad, J.; Iqbal, F.; Sajjad, A.; Mehmood, Z.; Murtaza, G. Pollution status of Pakistan: A retrospective review on heavy metal contamination of water, soil, and vegetables. BioMed Res. Int. 2014, 2014, 813206. [Google Scholar] [CrossRef] [PubMed]
- Bacquart, T.; Frisbie, S.; Mitchell, E.; Grigg, L.; Cole, C.; Small, C.; Sarkar, B. Multiple inorganic toxic substances contaminating the groundwater of Myingyan Township, Myanmar: Arsenic, manganese, fluoride, iron, and uranium. Sci. Total Environ. 2015, 517, 232–245. [Google Scholar] [CrossRef]
- Rehman, K.; Fatima, F.; Waheed, I.; Akash, M.S.H. Prevalence of exposure of heavy metals and their impact on health consequences. J. Cell. Biochem. 2018, 119, 157–184. [Google Scholar] [CrossRef] [PubMed]
- Tomlinson, M.S.; Bommarito, P.; George, A.; Yelton, S.; Cable, P.; Coyte, R.; Karr, J.; Vengosh, A.; Gray, K.M.; Fry, R.C. Assessment of inorganic contamination of private wells and demonstration of effective filter-based reduction: A pilot-study in Stokes County, North Carolina. Environ. Res. 2019, 177, 108618. [Google Scholar] [CrossRef]
- Lotfi, S.; Chakit, M.; Belghyti, D. Groundwater Quality and Pollution Index for Heavy Metals in Saïs Plain, Morocco. J. Health Poll. 2020, 10, 200603. [Google Scholar] [CrossRef] [PubMed]
- ATSDR (Agency for Toxic Substances Disease Registry). Substance Priority List (1997). Available online: https://www.atsdr.cdc.gov/spl/resources/1997_atsdr_substance_priority_list.html (accessed on 25 April 2023).
- Bullard, R.D. Dumping in Dixie: Race, Class and Environmental Quality; Westview Press: Boulder, CO, USA, 1990. [Google Scholar]
- Bullard, R.D. Environmental Justice in the 21st Century: Race Still Matters. Phylon 2001, 49, 151–171. [Google Scholar] [CrossRef]
- U.S. Department of Health & Human Services, Biden-Harris Administration Establishes HHS Office of Environmental Justice. 2022. Available online: https://www.hhs.gov/about/news/2022/05/31/biden-harris-administration-establishes-hhs-office-of-environmental-justice.html (accessed on 25 April 2023).
- World Health Organization. Social Determinants of Health. Available online: https://www.who.int/health-topics/social-determinants-of-health#tab=tab_1 (accessed on 25 April 2023).
- Office of Disease Prevention and Health Promotion, Healthy People 2030. Available online: https://health.gov/healthypeople (accessed on 25 April 2023).
- CDC/ATSDR. Social Vulnerability Index. 2022. Available online: https://www.atsdr.cdc.gov/placeandhealth/svi/index.html (accessed on 25 April 2023).
- Adger, W.N. Vulnerability. Global Environ. Change 2006, 16, 268–281. [Google Scholar] [CrossRef]
- Cutter, S.L. Vulnerability to Environmental Hazards. Prog. Hum. Geogr. 1996, 20, 29–539. [Google Scholar] [CrossRef]
- Ramírez-Andreotta, M.D.; Walls, R.; Youens-Clark, K.; Blumberg, K.; Isaacs, K.E.; Kaufmann, D.; Maier, R.M. Alleviating Environmental Health Disparities through Community Science and Data Integration. Front. Sust. Food Sys. 2021, 5, 182. [Google Scholar] [CrossRef]
- Saeedi, P.; Petersohn, I.; Salpea, P.; Malanda, B.; Karuranga, S.; Unwin, N.; Colagiuri, S.; Guariguata, L.; Motala, A.A.; Ogurtsova, K.; et al. Global and regional diabetes prevalence estimates for 2019 and projections for 2030 and 2045: Results from the International Diabetes Federation Diabetes Atlas, 9th edition. Diabetes Res. Clin. Pract. 2019, 157, 107843. [Google Scholar] [CrossRef] [PubMed]
- Nordstrom, D.K. Public Health. Worldwide Occurrences of Arsenic in Ground Water. Science 2002, 296, 2143–2145. [Google Scholar] [CrossRef] [PubMed]
- Cullen, W.R.; Reimer, K.J. Arsenic Speciation in the Environment. Chem. Rev. 1989, 89, 713–764. [Google Scholar] [CrossRef]
- Shankar, S.; Shanker, U.; Shikha. Arsenic Contamination of Groundwater: A Review of Sources, Prevalence, Health Risks, and Strategies for Mitigation. Sci. World J. 2014, 2014, 304524. [Google Scholar] [CrossRef] [PubMed]
- Naujokas, M.F.; Anderson, B.; Ahsan, H.; Aposhian, H.V.; Graziano, J.H.; Thompson, C.; Suk, W.A. The Broad Scope of Health Effects from Chronic Arsenic Exposure: Update on a Worldwide Public Health Problem. Environ. Health Perspect. 2013, 121, 295–302. [Google Scholar] [CrossRef] [PubMed]
- Focazio, M.J.; Welch, A.H.; Watkins, S.A.; Helsel, D.R.; Horn, M.A. A Retrospective Analysis on the Occurrence of Arsenic in Ground-Water Resources of the United States and Limitations in Drinking-Water-Supply-Characterizations; Water-Resources Investigations Report 99-4279; US Geological Survey: Reston, VA, USA, 2000. [Google Scholar]
- Huang, L.; Wu, H.; van der Kuijp, T.J. The Health Effects of Exposure to Arsenic-Contaminated Drinking Water: A Review by Global Geographical Distribution. Int. J. Environ. Health Res. 2015, 25, 432–452. [Google Scholar] [CrossRef]
- Navas-Acien, A.; Silbergeld, E.K.; Streeter, R.A.; Clark, J.M.; Burke, T.A.; Guallar, E. Arsenic Exposure and Type 2 Diabetes: A Systematic Review of the Experimental and Epidemiological Evidence. Environ. Health Perspect. 2006, 114, 641–648. [Google Scholar] [CrossRef]
- Hill-Briggs, F.; Adler, N.E.; Berkowitz, S.A.; Chin, M.H.; Gary-Webb, T.L.; Navas-Acien, A.; Thornton, P.L.; Haire-Joshu, D. Social Determinants of Health and Diabetes: A Scientific Review. Diabetes Care 2021, 44, 258–279. [Google Scholar] [CrossRef]
- Ramírez-Andreotta, M.D.; Brusseau, M.L.; Beamer, P.; Maier, R.M. Home Gardening Near a Mining Site in an Arsenic-Endemic Region of Arizona: Assessing Arsenic Exposure Dose and Risk Via Ingestion of Home Garden Vegetables, Soils, and Water. Sci. Total Environ. 2013, 454–455, 373–382. [Google Scholar] [CrossRef]
- Manjón, I.; Ramírez-Andreotta, M.D.; Saez, A.E.; Root, R.A.; Hild, J.; Janes, K.; Alexander-Ozinskas, A. Ingestion and Inhalation of Metal(loid)s Through Preschool Gardening: An Exposure and Risk Assessment in Legacy Mining Communities. Sci. Total Environ. 2020, 718, 134639. [Google Scholar] [CrossRef]
- Rodriguez-Chavez, T.; Rine, K.; Almusawi, R.; O’Brien-Metzger, R.; Ramírez-Andreotta, M.D.; Betterton, E.; Saez, A.E. Outdoor/Indoor Contaminant Transport by Atmospheric Dust and Aerosol at an Active Smelter Site. Water Air Soil Pollut. 2021, 232, 226. [Google Scholar] [CrossRef]
- Heusinkveld, D.; Ramírez-Andreotta, M.D.; Rodriguez-Chavez, T.B.; Saez, E.A.; Betterton, E.; Rine, K. Assessing a Children’s Integrated Exposure Model in an Active Mining Town. Expo. Health 2021, 13, 517–533. [Google Scholar] [CrossRef] [PubMed]
- Centers for Disease Control and Prevention. National Diabetes Statistics Report. Available online: https://www.cdc.gov/diabetes/data/statistics-report/index.html (accessed on 25 April 2023).
- National Diabetes Association. The Cost of Diabetes. Available online: https://diabetes.org/about-us/statistics/cost-diabetes (accessed on 25 April 2023).
- Smith, N.M.; Lee, R.; Heitkemper, D.T.; Cafferky, K.D.; Haque, A.; Henderson, A.K. Inorganic Arsenic in Cooked Rice and Vegetables from Bangladeshi Households. Sci. Total Environ. 2006, 370, 294–301. [Google Scholar] [CrossRef]
- Prohaska, T.; Stingeder, G. Speciation of Arsenic. In Handbook of Elemental Speciation II—Species in the Environment, Food, Medicine and Occupational Health; Cornelis, R., Ed.; John Wiley & Sons Ltd.: West Sussex, UK, 2005; pp. 69–85. [Google Scholar]
- Muñoz, O.; Bastias, J.M.; Araya, M.; Morales, A.; Orellana, C.; Rebolledo, R. Estimation of the Dietary Intake of Cadmium, Lead, Mercury, and Arsenic by the Population of Santiago (Chile) Using a Total Diet Study. Food Chem. Toxicol. 2005, 43, 1647–1655. [Google Scholar] [CrossRef]
- Yost, L.J.; Schoof, R.A.; Aucoin, R. Intake of Inorganic Arsenic in the North American Diet. Hum. Ecol. Risk Assess. 1998, 4, 137–152. [Google Scholar] [CrossRef]
- Diaz, O.P.; Leyton, I.; Muñoz, O.; Nuñez, N.; Devesa, V.; Súñer, M.A.; Veles, D.; Mpntor, R. Contribution of Water, Bread and Vegetables (Raw and Cooked) to Dietary Intake of Inorganic Arsenic in a Rural Village of Northern Chile. J. Agric. Food Chem. 2004, 52, 1773–1779. [Google Scholar] [CrossRef]
- Diaz, O.P.; Arcos, R.; Tapia, Y.; Pastene, R.; Velez, D.; Devesa, V.; Montoro, R.; Aguilera, V.; Becerra, M. Estimation of Arsenic Intake from Drinking Water and Food (Raw and Cooked) in a Rural Village of Northern Chile. Urine as a Biomarker of Recent Exposure. Intl. J. Environ. Res. Public Health 2015, 12, 5614–5633. [Google Scholar] [CrossRef]
- Laparra, J.M.; Vélez, D.; Barberá, R.; Farré, R.; Montoro, R. Bioavailability of Inorganic Arsenic in Cooked Rice: Practical Aspects for Human Risk Assessments. J. Agric. Food Chem. 2005, 53, 8829–8833. [Google Scholar] [CrossRef]
- Bundschuh, J.; Nath, B.; Battacharya, P.; Liu, C.W.; Armienta, M.A.; López, M.V.; Lopez, D.L.; Jean, J.S.; Cornejo, L.; Macedo, L.F.; et al. Arsenic in the Human Food Chain: The Latin America Perspective. Sci. Total Environ. 2012, 429, 92–106. [Google Scholar] [CrossRef]
- Chung, J.Y.; Yu, S.D.; Hong, Y.S. Environmental Source of Arsenic Exposure. J. Prev. Med. Public Health 2014, 47, 253–257. [Google Scholar] [CrossRef]
- mindat.org. The Mineralogy of Arsenic. Available online: https://www.mindat.org/element/Arsenic (accessed on 25 April 2023).
- Savage, K.S.; Tingle, T.N.; O’Day, P.A.; Waychunas, G.A.; Bird, D.K. Arsenic Speciation in Pyrite and Secondary Weathering Phases, Mother Lode Gold District, Tuolumne County, California. Appl. Geochem. 2000, 15, 1219–1244. [Google Scholar] [CrossRef]
- Deditius, A.P.; Utsunomiya, S.; Renock, D.; Ewing, R.C.; Ramana, C.V.; Becker, U.; Kesler, S.E. A Proposed New Type of Arsenian Pyrite: Composition, Nanostructure and Geological Significance. Geochim. Cosmochim. Acta 2008, 72, 2919–2933. [Google Scholar] [CrossRef]
- Chen, C.J.; Chuang, Y.C.; Lin, T.M.; Wu, H.Y. Malignant Neoplasms Among Residents of a Blackfoot Disease-Endemic Area in Taiwan: High-Arsenic Atesian Well Water and Cancers. Cancer Res. 1985, 45 Pt 2, 5895–5899. [Google Scholar]
- Saha, K.C. Chronic Arsenical Dermatoses From Tube- Well Water In West Bengal During 1983–87. Indian J. Dermatol. 1995, 40, 1–12. [Google Scholar]
- Smith, A.H.; Lingas, E.O.; Rahman, M. Contamination of Drinking-Water by Arsenic in Bangladesh: A Public Health Emergency. Bull. World Health Org. 2000, 78, 1093–1103. [Google Scholar] [PubMed]
- Campbell, K.M.; Nordstrom, D.K. Arsenic Speciation and Sorption in Natural Environments. Rev. Mineral. Geochem. 2014, 79, 185–216. [Google Scholar] [CrossRef]
- Foster, A.L.; Kim, C.S. Arsenic Speciation in Solids Using X-ray Absorption Spectroscopy. Rev. Mineral. Geochem. 2014, 79, 257–369. [Google Scholar] [CrossRef]
- Polizzotto, M.L.; Kocar, B.D.; Benner, S.G.; Sampson, M.; Fendorf, S. Near-Surface Wetland Sediments as a Source of Arsenic Release to Ground Water in Asia. Nature 2008, 454, 505–508. [Google Scholar] [CrossRef]
- Masuda, H. Arsenic Cycling in the Earth’s Crust and Hydrosphere: Interaction Between Naturally Occurring Arsenic and Human Activities. Prog. Earth Planet. Sci. 2018, 5, 68. [Google Scholar] [CrossRef]
- Chowdhury, U.K.; Biswas, B.K.; Chowdhury, T.R.; Samanta, G.; Mandal, B.K.; Basu, G.C.; Chanda, C.R.; Lodh, D.; Saha, K.C.; Mukherjee, S.K. Groundwater Arsenic Contamination in Bangladesh and West Bengal, India. Environ. Health Perspec. 2000, 108, 393–397. [Google Scholar] [CrossRef]
- Charlet, L.; Polya, D.A. Arsenic in Shallow, Reducing Groundwaters in Southern Asia: An Environmental Health Disaster. Elements 2006, 2, 91–96. [Google Scholar] [CrossRef]
- Nriagu, J.O. (Ed.) Arsenic in the Environment (Part II, Human Health and Ecosystem Effects); Wiley Interscience: Hoboken, NJ, USA, 1994; ISBN 978-0-471-30436-4. [Google Scholar]
- Berg, M.; Tran, H.C.; Nguyen, T.C.; Pham, H.V.; Schertenleib, R.; Giger, W. Arsenic Contamination of Groundwater and Drinking Water in Vietnam: A Human Hhealth Threat. Environ. Sci. Technol. 2001, 35, 2621–2626. [Google Scholar] [CrossRef] [PubMed]
- Nickson, R.T.; McArthur, J.M.; Shrestha, B.; Kyaw-Myint, T.O.; Lowry, D. Arsenic and Other Drinking Water Quality Issues, Muzaffargarh District, Pakistan. Appl. Geochem. 2005, 20, 55–68. [Google Scholar] [CrossRef]
- Chen, S.L.; Dzeng, S.R.; Yang, M.H.; Chiu, K.H.; Shieh, G.M.; Wai, C.M. Arsenic Species in Groundwaters of the Blackfoot Disease Area, Taiwan. Environ. Sci. Technol. 1994, 28, 877–881. [Google Scholar] [CrossRef] [PubMed]
- Ayotte, J.D.; Medalie, L.; Qi, S.L.; Backer, L.C.; Nolan, B.T. Estimating the High-Arsenic Domestic-Well Population in the Conterminous United States. Environ. Sci. Technol. 2017, 51, 12443–12454. [Google Scholar] [CrossRef]
- Grantham, D.A.; Jones, J.F. Arsenic Contamination of Water wells in Nova Scotia. J.-Am. Water Work. Assoc. 1977, 69, 653–657. [Google Scholar] [CrossRef]
- Bundschuh, J.; Farias, B.; Martin, R.; Storniolo, A.; Bhattacharya, P.; Cortes, J.; Bonorino, G.; Albouy, R. Groundwater Arsenic in the Chaco-Pampean Plain, Argentina: Case Study from Robles County, Santiago del Estero Province. Appl. Geochem. 2004, 19, 231–243. [Google Scholar] [CrossRef]
- Bundschuh, J.; Litter, M.I.; Parvez, F.; Román-Ross, G.; Nicolli, H.B.; Jean, J.S.; Liu, C.W.; López, D.; Armienta, M.A.; Guilherme, L.R.G.; et al. One Century of Arsenic Exposure in Latin America: A Review of History and Occurrence from 14 Countries. Sci. Total Environ. 2012, 429, 2–35. [Google Scholar] [CrossRef]
- Smedley, P.; Kinniburgh, D.; Macdonald, D.; Nicolli, H.; Barros, A.; Tullio, J.; Pearce, J.; Alonso, M. Arsenic Associations in Sediments from the Loess Aquifer of La Pampa, Argentina. Appl. Geochem. 2005, 20, 989–1016. [Google Scholar] [CrossRef]
- Smedley, P.L.; Kinniburgh, D.G. A Review of the Source, Behaviour and Distribution of Arsenic in Natural Waters. Appl. Geochem. 2002, 17, 517–568. [Google Scholar] [CrossRef]
- Morin, G.; Calas, G. Arsenic in Soils, Mine Tailings, and Former Industrial Sites. Elements 2006, 2, 97–101. [Google Scholar] [CrossRef]
- Root, R.A.; Vlassopoulos, D.; Rivera, N.A.; Rafferty, M.T.; Andrews, C.; O’Day, P.A. Speciation and Natural Attenuation of Arsenic and Iron in a Tidally Influenced Shallow Aquifer. Geochim. Cosmochim. Acta 2009, 73, 5528–5553. [Google Scholar] [CrossRef]
- O’Day, P.A.; Vlassopoulos, D.; Root, R.; Rivera, N. The Influence of Sulfur and Iron on Dissolved Arsenic Concentrations in the Shallow Subsurface Under Changing Redox Conditions. Proc. Natl. Acad. Sci. USA 2004, 101, 13703–13708. [Google Scholar] [CrossRef] [PubMed]
- O’Day, P.A. Chemistry and Mineralogy of Arsenic. Elements 2006, 2, 77–83. [Google Scholar] [CrossRef]
- Welch, A.H.; Lico, M.S. Factors Controlling as and U in Shallow Ground Water, Southern Carson Desert, Nevada. Appl. Geochem. 1998, 13, 521–539. [Google Scholar] [CrossRef]
- Demergasso, C.S.; Ching, G.D.; Escudero, L.G.; Mur, J.J.P.; Alio, C.P. Microbial Precipitation of Arsenic Sulfides in Andean Salt Flats. Geomicrobiol. J. 2007, 24, 111–123. [Google Scholar] [CrossRef]
- Pierce, M.L.; Moore, C.B. Adsorption of Arsenite and Arsenate on Amorphous Iron Hydroxide. Water Res. 1982, 16, 1247–1253. [Google Scholar] [CrossRef]
- Fuller, C.C.; Davis, J.A.; Waychunas, G.A. Surface Chemistry of Ferrihydrite: Part 2. Kinetics of Arsenate Adsorption and Coprecipitation. Geochim. Cosmochim. Acta 1993, 57, 2271–2282. [Google Scholar] [CrossRef]
- Dixit, S.; Hering, J.G. Comparison of Arsenic(V) and Arsenic(III) Sorption onto Iron Oxide Minerals: Implications for Arsenic Mobility. Environ. Sci. Technol. 2003, 37, 4182–4189. [Google Scholar] [CrossRef]
- Gao, X.; Root, R.A.; Farrell, J.; Ela, W.; Chorover, J. Effect of Silicic Acid on Arsenate and Arsenite Retention Mechanisms on 6-L Ferrihydrite: A Spectroscopic and Batch Adsorption Approach. Appl. Geochem. 2013, 38, 110–120. [Google Scholar] [CrossRef]
- Waychunas, G.A.; Rea, B.A.; Fuller, C.C.; Davis, J.A. Surface Chemistry of Ferrihydrite: Part 1. EXAFS Studies of the Geometry of Coprecipitated and Adsorbed Arsenate. Geochim. Cosmochim. Acta 1993, 57, 2251–2269. [Google Scholar] [CrossRef]
- Root, R.A.; Dixit, S.; Campbell, K.M.; Jew, A.D.; Hering, J.G.; O’Day, P.A. Arsenic Sequestration by Sorption Processes in High-Iron Sediments. Geochim. Cosmochim. Acta 2007, 71, 5782–5803. [Google Scholar] [CrossRef]
- Root, R.A.; Fathordoobadi, S.; Alday, F.; Ela, W.; Chorover, J. Microscale Speciation of Arsenic and Iron in Ferric-Based Sorbents Subjected to Simulated Landfill Conditions. Environ. Sci. Technol. 2013, 47, 12992–13000. [Google Scholar] [CrossRef] [PubMed]
- Rodriguez-Freire, L.; Moore, S.E.; Sierra-Alvarez, R.; Root, R.A.; Chorover, J.; Field, J.A. Arsenic Remediation by Formation of Arsenic Sulfide Minerals in a Continuous Anaerobic Bioreactor. Biotechnol. Bioeng. 2016, 113, 522–530. [Google Scholar] [CrossRef] [PubMed]
- Ahmann, D.; Krumholz, L.R.; Hemond, H.F.; Lovley, D.R.; Morel, F.M.M. Microbial Mobilization of Arsenic from Sediments of the Aberjona Watershed. Environ. Sci. Technol. 1997, 31, 2923–2930. [Google Scholar] [CrossRef]
- Zobrist, J.; Dowdle, P.R.; Davis, J.A.; Oremland, R.S. Mobilization of Arsenite by Dissimilatory Reduction of Adsorbed Arsenate. Environ. Sci. Technol. 2000, 34, 4747–4753. [Google Scholar] [CrossRef]
- Podgorski, J.; Berg, M. Global threat of arsenic in groundwater. Science 2020, 368, 845–850. [Google Scholar] [CrossRef]
- Manning, B.A.; Fendorf, S.A.; Goldberg, S. Surface Structures and Stability of Arsenic (III) on Goethite: Spectroscopic Evidence for Inner-Sphere Complexes. Environ. Sci. Technol. 1998, 32, 2383–2388. [Google Scholar] [CrossRef]
- Bose, P.; Sharma, A. Role of Iron in Controlling Speciation and Mobilization of Arsenic in Subsurface Environment. Water Res. 2002, 36, 4916–4926. [Google Scholar] [CrossRef]
- Bradham, K.; Scheckel, K.; Nelson, C.; Seales, P.; Lee, G.; Hughes, M.; Miller, B.; Yeow, A.; Gilmore, T.; Serda, S.; et al. Relative Bioavailability and Bioaccessibility and Speciation of Arsenic in Contaminated Soils. Environ. Health Perspect. 2011, 119, 1629–1634. [Google Scholar] [CrossRef]
- Azcue, J.M.; Nriagu, J.O.; Schiff, S. Role of Sediment Porewater in the Cycling of Arsenic in a Mine-Polluted Lake. Environ. Int. 1994, 20, 517–527. [Google Scholar] [CrossRef]
- Inskeep, W.P.; Macur, R.E.; Harrison, G.; Bostick, B.C.; Fendorf, S. Biomineralization of As(V)-Hydrous Ferric Oxyhydroxide in Microbial Mats of an Acid-Sulfate-Chloride Geothermal Spring, Yellowstone National Park. Geochim. Cosmochim. Acta 2004, 68, 3141–3155. [Google Scholar] [CrossRef]
- Cherry, J.A.; Shaikh, A.U.; Tallman, D.E.; Nicholson, R.V. Arsenic Species as an Indicator of Redox Conditions in Groundwater. Devel. Water Sci. 1979, 12, 373–392. [Google Scholar] [CrossRef]
- Manning, B.A.; Goldberg, S. Modeling Competitive Adsorption of Arsenate with Phosphate and Molybdate on Oxide Minerals. Soil Sci. Soc. Amer. J. 1996, 60, 121–131. [Google Scholar] [CrossRef]
- Kneebone, P.E.; Hering, J.G. Behavior of Arsenic and Other Redox-Sensitive Elements in Crowley Lake, CA: A Reservoir in the Los Angeles Aqueduct System. Environ. Sci. Technol. 2000, 34, 4307–4312. [Google Scholar] [CrossRef]
- Beak, D.G.; Wilkin, R.T. Performance of a Zero valent Iron Reactive Barrier for the Treatment of Arsenic in Groundwater: Part 2. Geochemical Modeling and Solid Phase Studies. J. Contam. Hydrol. 2009, 106, 15–28. [Google Scholar] [CrossRef]
- Dzombak, D.A.; Morel, F.M.M. Surface Complexation Modeling: Hydrous Ferric Oxide; John Wiley & Sons: New York, NY, USA, 1990; p. 416. [Google Scholar]
- Newman, D.K.; Kennedy, E.K.; Coates, J.D.; Ahmann, D.; Ellis, D.J.; Lovley, D.R.; Morel, F.M.M. Dissimilatory Arsenate and Sulfate Reduction in Desulfotomaculum auripigmentum sp. nov. Arch. Microbiol. 1997, 168, 380–388. [Google Scholar] [CrossRef]
- Kocar, B.D.; Fendorf, S. Thermodynamic Constraints on Reductive Reactions Influencing the Biogeochemistry of Arsenic in Soils and Sediments. Environ. Sci. Technol. 2009, 43, 4871–4877. [Google Scholar] [CrossRef]
- Oremland, R.S.; Stolz, J.F. Arsenic, Microbes and Contaminated Aquifers. Trends Microbiol. 2005, 13, 45–49. [Google Scholar] [CrossRef]
- Stollenwerk, K.G.; Breit, G.N.; Welch, A.H.; Foster, A.L. Remediation of Arsenic-Contaminated Groundwater by Oxidized Sediments in Bangladesh. Geochim. Cosmochim. Acta 2006, 70 (Suppl. 18), A617. [Google Scholar] [CrossRef]
- Diagnosis and Classification of Diabetes Mellitus. Diabetes Care 2004, 27 (Suppl. 1), s5–s10. [CrossRef]
- DeFronzo, R.; Ferrannini, E.; Groop, L.; Henry, R.R.; Herman, W.H.; Holst, J.J.; Hu, F.B.; Kahn, C.R.; Raz, I.; Shulman, G.I.; et al. Type 2 Diabetes Mellitus. Nat. Rev. Dis. Prim. 2015, 1, 15019. [Google Scholar] [CrossRef]
- Katsarou, A.; Gudbjörnsdottir, S.; Rawshani, A.; Dabelea, D.; Bonifacio, E.; Anderson, B.J.; Jacobsen, L.M.; Schatz, D.A.; Lernmark, A. Type 1 Diabetes Mellitus. Nat. Rev. Dis. Prim. 2017, 3, 17016. [Google Scholar] [CrossRef]
- Chen, Y.W.; Yang, C.Y.; Huang, C.F.; Hung, D.Z.; Leung, Y.M.; Liu, S.H. Heavy Metals, Islet Function and Diabetes Development. Islets 2009, 1, 169–176. [Google Scholar] [CrossRef] [PubMed]
- Maull, E.A.; Ahsan, H.; Edwards, J.; Longnecker, M.P.; Navas-Acien, A.; Pi, J.; Silbergeld, E.K.; Styblo, M.; Tseng, C.H.; Thayer, K.A.; et al. Evaluation of the Association Between Arsenic and Diabetes: A National Toxicology Program Workshop Review. Environ. Health Perspect. 2012, 120, 1658–1670. [Google Scholar] [CrossRef] [PubMed]
- Del Razo, L.M.; García-Vargas, G.G.; Valenzuela, O.L.; Castellanos, E.H.; Sánchez-Peña, L.C.; Currier, J.M.; Drobná, Z.; Loomis, D.; Stýblo, M. Exposure to Arsenic in Drinking Water is Associated with Increased Prevalence of Diabetes: A Cross-Sectional Study in the Zimapán and Lagunera Regions in Mexico. Environ. Health 2011, 10, 73. [Google Scholar] [CrossRef] [PubMed]
- Lai, M.S.; Hsueh, Y.M.; Chen, C.J.; Shyu, M.P.; Chen, S.Y.; Kuo, T.L.; Wu, M.M.; Tai, T.Y. Ingested Inorganic Arsenic and Prevalence of Diabetes Mellitus. Am. J. Epidemiol. 1994, 139, 484–492. [Google Scholar] [CrossRef]
- Tsai, S.M.; Wang, T.N.; Ko, Y.-C. Mortality for Certain Diseases in Areas with High Levels of Arsenic in Drinking Water. Arch. Environ. Health 1999, 54, 186–193. [Google Scholar] [CrossRef]
- Jovanovic, D.; Rasic-Milutinovic, Z.; Paunovic, K.; Jakovljevic, B.; Plavsic, S.; Milosevic, J. Low Levels of Arsenic in Drinking Water and Type 2 Diabetes in Middle Banat Region, Serbia. Int. J. Hyg. Environ. Health 2013, 216, 50–55. [Google Scholar] [CrossRef]
- Islam, R.; Khan, I.; Hassan, S.N.; McEvoy, M.; D’Este, C.; Attia, J.; Peel, R.; Sultana, M.; Akter, S.; Milton, A.H. Association Between Type 2 Diabetes and Chronic Arsenic Exposure in Drinking Water: A Cross Sectional Study in Bangladesh. Environ. Health 2012, 11, 38. [Google Scholar] [CrossRef]
- Meliker, J.R.; Wahl, R.L.; Cameron, L.L.; Nriagu, J.O. Arsenic in Drinking Water and Cerebrovascular Disease, Diabetes Mellitus, and Kidney Disease in Michigan: A Standardized Mortality Ratio Analysis. Environ. Health 2007, 6, 4. [Google Scholar] [CrossRef]
- Balakrishnan, P.; Navas-Acien, A.; Haack, K.; Vaidya, D.; Umans, J.G.; Best, L.G.; Goessler, W.; Francesconi, K.A.; Franceschini, N.; North, K.E.; et al. Arsenic-Gene Interactions and Beta-Cell Function in the Strong Heart Family Study. Toxicol. Appl. Pharmacol. 2018, 348, 123–129. [Google Scholar] [CrossRef] [PubMed]
- Domingo-Relloso, A.; Makhani, K.; Riffo-Campos, A.L.; Tellez-Plaza, M.; Klein, K.O.; Subedi, P.; Zhao, J.; Moon, K.A.; Bozack, A.K.; Haack, K.; et al. Arsenic Exposure, Blood DNA Methylation, and Cardiovascular Disease. Circ. Res. 2022, 131, e51–e69. [Google Scholar] [CrossRef]
- Spratlen, M.J.; Grau-Perez, M.; Umans, J.G.; Yracheta, J.; Best, L.G.; Francesconi, K.; Goessler, W.; Balakrishnan, P.; Cole, S.A.; Gamble, M.V.; et al. Arsenic, one carbon metabolism and diabetes-related outcomes in the Strong Heart Family Study. Environ. Int. 2018, 121 Pt 1, 728–740. [Google Scholar] [CrossRef]
- Yang, K.; Xun, P.; Carnethon, M.; Carson, A.P.; Lu, L.; Zhu, J.; He, K. Low to Moderate Toenail Arsenic Levels in Young Adulthood and Incidence of Diabetes Later in Life: Findings from the CARDIA Trace Element Study. Environ. Res. 2019, 171, 321–327. [Google Scholar] [CrossRef] [PubMed]
- Nizam, S.; Kato, M.; Yatsuya, H.; Khalequzzaman, M.; Ohnuma, S.; Naito, H.; Nakajima, T. Differences in Urinary Arsenic Metabolites Between Diabetic and Non-Diabetic Subjects in Bangladesh. Int. J. Environ. Res. Public Health 2013, 10, 1006–1019. [Google Scholar] [CrossRef] [PubMed]
- Nabi, A.H.; Rahman, M.M.; Islam, L.N. Evaluation of Biochemical Changes in Chronic Arsenic Poisoning Among Bangladeshi Patients. Int. J. Environ. Res. Public Health 2005, 2, 385–393. [Google Scholar] [CrossRef]
- Rahman, M.; Tondel, M.; Ahmad, S.A.; Axelson, O. Diabetes Mellitus Associated with Arsenic Exposure in Bangladesh. Amer. J. Epidemiol. 1998, 148, 198–203. [Google Scholar] [CrossRef]
- Rahman, M.; Tondel, M.; Ahmad, S.A.; Chowdhury, I.A.; Faruquee, M.H.; Axelson, O. Hypertension and Arsenic Exposure in Bangladesh. Hypertension 1999, 33, 74–78. [Google Scholar] [CrossRef]
- Jensen, G.E.; Hansen, M.L. Occupational Arsenic Exposure and Glycosylated Haemoglobin. Analyst 1998, 123, 77–80. [Google Scholar] [CrossRef]
- Eick, S.M.; Ferreccio, C.; Acevedo, J.; Castriota, F.; Cordero, J.F.; Roh, T.; Smith, A.H.; Smith, M.T.; Steinmaus, C. Socioeconomic Status and the Association Between Arsenic Exposure and Type 2 Diabetes. Environ. Res. 2019, 172, 578–585. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Gao, D.; Zhang, G.; Zhang, X.; Li, Q.; Gao, Q.; Chen, R.; Xu, S.; Huang, L.; Zhang, Y.; et al. Exposure to multiple metals in early pregnancy and gestational diabetes mellitus: A prospective cohort study. Environ. Int. 2020, 135, 105370. [Google Scholar] [CrossRef] [PubMed]
- Jia, X.; Zhang, L.; Zhao, J.; Ren, M.; Li, Z.; Wang, J.; Wang, S.; Liu, Y.; An, H.; Li, Y.; et al. Associations between endocrine-disrupting heavy metals in maternal hair and gestational diabetes mellitus: A nested case-control study in China. Environ. Int. 2021, 157, 106770. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Chen, T.; Zhang, Y.; Hu, Q.; Wang, X.; Chang, H.; Mao, J.H.; Snijders, A.M.; Xia, Y. Contribution of trace element exposure to gestational diabetes mellitus through disturbing the gut microbiome. Environ. Int. 2021, 153, 106520. [Google Scholar] [CrossRef]
- Xia, X.; Liang, C.; Sheng, J.; Yan, S.; Huang, K.; Li, Z.; Pan, W.; Tao, R.; Hao, J.; Zhu, B.; et al. Association between serum arsenic levels and gestational diabetes mellitus: A population-based birth cohort study. Environ. Poll. 2018, 235, 850–856. [Google Scholar] [CrossRef]
- Lucio, M.; Barbir, R.; Vučić Lovrenčić, M.; Canecki Varžić, S.; Ljubić, S.; Smirčić Duvnjak, L.; Šerić, V.; Milić, M.; Tariba Lovaković, B.; Krivohlavek, A.; et al. Association Between Arsenic Exposure and Biomarkers of Type 2 Diabetes Mellitus in a Croatian Population: A Comparative Observational Pilot Study. Sci. Total Environ. 2020, 720, 137575. [Google Scholar] [CrossRef]
- Velmurugan, G.; Swaminathan, K.; Mohanraj, S.; Dhivakar, M.; Veerasekar, G.; Alexander, T.; Cherian, M.; Palaniswami, N.G.; Pradeep, T. Association of Co-Accumulation of Arsenic and Organophosphate Insecticides with Diabetes and Atherosclerosis in a Rural Agricultural Community: KMCH-NNCD-I study. Acta Diabetol. 2020, 57, 1159–1168. [Google Scholar] [CrossRef]
- Bartoli, D.; Battista, G.; De Santis, M.; Iaia, T.E.; Orsi, D.; Tarchi, M.; Pirastu, R.; Valiani, M. Cohort Study of Art Glass Workers in Tuscany, Italy: Mortality from Non-Malignant Diseases. Occup. Med. 1998, 48, 441–445. [Google Scholar] [CrossRef]
- Coronado-González, J.A.; Del Razo, L.M.; García-Vargas, G.; Sanmiguel-Salazar, F.; Escobedo-de la Peña, J. Inorganic Arsenic Exposure and Type 2 Diabetes Mellitus in Mexico. Environ. Res. 2007, 104, 383–389. [Google Scholar] [CrossRef]
- Mendez, M.A.; González-Horta, C.; Sánchez-Ramírez, B.; Ballinas-Casarrubias, L.; Cerón, R.H.; Morales, D.V.; Terrazas, F.A.; Ishida, M.C.; Gutiérrez-Torres, D.S.; Saunders, R.; et al. Chronic Exposure to Arsenic and Markers of Cardiometabolic Risk: A Cross-Sectional Study in Chihuahua, Mexico. Environ. Health Perspec. 2016, 124, 104–111. [Google Scholar] [CrossRef]
- Ruiz-Navarro, M.L.; Navarro-Alarcón, M.; Lopez González-de la Serrana, H.; Pérez-Valero, V.; López-Martinez, M.C. Urine Arsenic Concentrations in Healthy Adults as Indicators of Environmental Contamination: Relation with Some Pathologies. Sci. Total Environ. 1998, 216, 55–61. [Google Scholar] [CrossRef]
- Grau-Perez, M.; Navas-Acien, A.; Galan-Chilet, I.; Briongos-Figuero, L.S.; Morchon-Simon, D.; Bermudez, J.D.; Crainiceanu, C.M.; de Marco, G.; Rentero-Garrido, P.; Garcia-Barrera, T.; et al. Arsenic Exposure, Diabetes-Related Genes and Diabetes Prevalence in a General Population from Spain. Environ. Poll. 2018, 235, 948–955. [Google Scholar] [CrossRef] [PubMed]
- Lagerkvist, B.J.; Zetterlund, B. Assessment of Exposure to Arsenic Among Smelter Workers: A Five-Year Follow-Up. Am. J. Indus. Med. 1994, 25, 477–488. [Google Scholar] [CrossRef] [PubMed]
- Rahman, M.; Axelson, O. Diabetes Mellitus and Arsenic Exposure: A Second Look at Case-Control Data from a Swedish Copper Smelter. Occup. Environ. Med. 1995, 52, 773–774. [Google Scholar] [CrossRef]
- Rahman, M.; Wingren, G.; Axelson, O. Diabetes Mellitus Among Swedish Art Glass Workers- An Effect of Arsenic Exposure? Scand. J. Work Environ. Health 1996, 22, 146–149. [Google Scholar] [CrossRef] [PubMed]
- Tseng, C.H.; Tai, T.Y.; Chong, C.K.; Tseng, C.P.; Lai, M.S.; Lin, B.J.; Chiou, H.Y.; Hsueh, Y.M.; Hsu, K.H.; Chen, C.J. Long-Term Arsenic Exposure and Incidence of Non-Insulin-Dependent Diabetes Mellitus: A Cohort Study in Arseniasis-Hyperendemic Villages in Taiwan. Environ. Health Perspec. 2000, 108, 847–851. [Google Scholar] [CrossRef]
- Wang, S.L.; Chiou, J.M.; Chen, C.J.; Tseng, C.H.; Chou, W.L.; Wang, C.C.; Wu, T.N.; Chang, L.W. Prevalence of Non-Insulin-Dependent Diabetes Mellitus and Related Vascular Diseases in Southwestern Arseniasis-Endemic and Nonendemic Areas in Taiwan. Environ. Health Perspec. 2003, 111, 155–159. [Google Scholar] [CrossRef] [PubMed]
- Chiu, H.F.; Chang, C.C.; Tsai, S.S.; Yang, C.Y. Does Arsenic Exposure Increase the Risk for Diabetes Mellitus? J. Occup. Environ. Med. 2006, 48, 63–67. [Google Scholar] [CrossRef]
- Chen, J.W.; Wang, S.L.; Wang, Y.H.; Sun, C.W.; Huang, Y.L.; Chen, C.J.; Li, W.F. Arsenic Methylation, GSTO1 Polymorphisms, and Metabolic Syndrome in an Arseniasis Endemic Area of Southwestern Taiwan. Chemosphere 2012, 88, 432–438. [Google Scholar] [CrossRef]
- Ward, M.I.; Pim, B. Trace Element Concentrations in Blood Plasma from Diabetic Patients and Normal Individuals. Biol. Trace Elem. Res. 1984, 6, 469–487. [Google Scholar] [CrossRef]
- Kuo, C.C.; Howard, B.V.; Umans, J.G.; Gribble, M.O.; Best, L.G.; Francesconi, K.A.; Goessler, W.; Lee, E.; Guallar, E.; Navas-Acien, A. Arsenic Exposure, Arsenic Metabolism, and Incident Diabetes in the Strong Heart Study. Diabetes Care. 2015, 38, 620–627. [Google Scholar] [CrossRef]
- Mabuchi, K.; Lilienfeld, A.M.; Snell, L.M. Cancer and Occupational Exposure to Arsenic: A Study of Psticide Workers. Prev. Med. 1980, 9, 51–77. [Google Scholar] [CrossRef]
- Lubin, J.H.; Pottern, L.M.; Stone, B.J.; Fraumeni, J.F. Respiratory Cancer in a Cohort of Copper Smelter Workers: Results from more than 50 Years of Follow-Up. Am. J. Epidem. 2000, 151, 554–565. [Google Scholar] [CrossRef]
- Tollestrup, K.; Frost, F.J.; Harter, L.C.; McMillan, G.P. Mortality Among Children Residing near the American Smelting and Refining Company (ASARCO) Copper Smelter in Ruston, Washington. Arch. Environ. Health 2003, 58, 683–691. [Google Scholar] [CrossRef] [PubMed]
- Lewis, D.R.; Southwick, J.W.; Ouellet-Hellstrom, R.; Rench, J.; Calderon, R.L. Drinking water arsenic in Utah: A cohort Mortality Study. Environ. Health Perspec. 1999, 107, 359–365. [Google Scholar] [CrossRef] [PubMed]
- Zierold, K.M.; Knobeloch, L.; Anderson, H. Prevalence of Chronic Diseases in Adults Exposed to Arsenic-Contaminated Drinking Water. Am. J. Public Health 2004, 94, 1936–1937. [Google Scholar] [CrossRef] [PubMed]
- Navas-Acien, A.; Silbergeld, E.K.; Pastor-Barriuso, R.; Guallar, E. Arsenic Exposure and Prevalence of Type 2 Diabetes in US Adults. JAMA 2008, 300, 814–822. [Google Scholar] [CrossRef] [PubMed]
- Gribble, M.O.; Howard, B.V.; Umans, J.G.; Shara, N.M.; Francesconi, K.A.; Goessler, W.; Crainiceanu, C.M.; Silbergeld, E.K.; Guallar, E.; Navas-Acien, A. Arsenic Exposure, Diabetes Prevalence, and Diabetes Control in the Strong Heart Study. Am. J. Epidem. 2012, 176, 865–874. [Google Scholar] [CrossRef] [PubMed]
- Bulka, C.M.; Persky, V.W.; Daviglus, M.L.; Durazo-Arvizu, R.A.; Argos, M. Multiple metal exposures and metabolic syndrome: A cross-sectional analysis of the National Health and Nutrition Examination Survey 2011–2014. Environ. Res. 2019, 168, 397–405. [Google Scholar] [CrossRef]
- Chen, W.J.; Davis, E.M.; Stoner, J.A.; Robledo, C.; Goodman, J.R.; Garwe, T.; Janitz, A.E.; Xu, C.; Hwang, J.; Peck, J.D. Urinary total arsenic and arsenic methylation capacity in pregnancy and gestational diabetes mellitus: A case-control study. Chemosphere 2021, 271, 129828. [Google Scholar] [CrossRef] [PubMed]
- Grau-Pérez, M.; Kuo, C.C.; Spratlen, M.; Thayer, K.A.; Mendez, M.A.; Hamman, R.F.; Dabelea, D.; Adgate, J.L.; Knowler, W.C.; Bell, R.A.; et al. The Association of Arsenic Exposure and Metabolism with Type 1 and Type 2 Diabetes in Youth: The SEARCH Case-Control Study. Diabetes Care 2017, 40, 46–53. [Google Scholar] [CrossRef] [PubMed]
- Macfarlane, W.M.; Smith, S.B.; James, R.F.; Clifton, A.D.; Doza, Y.N.; Cohen, P.; Docherty, K. The p38/Reactivating Kinase Mitogen-Activated Protein Kinase Cascade Mediates the Activation of the Transcription Factor Insulin Upstream Factor 1 and Insulin Gene Transcription by High Glucose in Pancreatic Beta-Cells. J. Biol. Chem. 1997, 272, 20936–20944. [Google Scholar] [CrossRef]
- Elrick, L.J.; Docherty, K. Phosphorylation-Dependent Nucleocytoplasmic Shuttling of Pancreatic Duodenal Homeobox-1. Diabetes 2001, 50, 2244–2252. [Google Scholar] [CrossRef] [PubMed]
- Salazard, B.; Bellon, L.; Jean, S.; Maraninchi, M.; El-Yazidi, C.; Orsière, T.; Margotat, A.; Botta, A.; Bergé-Lefranc, J.L. Low-Level Arsenite Activates the Transcription of Genes Involved in Adipose Differentiation. Cell Biol. Toxicol. 2004, 20, 375–385. [Google Scholar] [CrossRef]
- Wauson, E.M.; Langan, A.S.; Vorce, R.L. Sodium Arsenite Inhibits and Reverses Expression of Adipogenic and Fat Cell-Specific Genes During in vitro Adipogenesis. Toxicol. Sci. 2002, 65, 211–219. [Google Scholar] [CrossRef] [PubMed]
- Walton, F.S.; Harmon, A.W.; Paul, D.S.; Drobna, Z.; Patel, Y.M.; Styblo, M. Inhibition of Insulin-Dependent Glucose Uptake by Trivalent Arsenicals: Possible Mechanism of Arsenic-Induced Diabetes. Toxicol. Appl. Pharmacol. 2004, 198, 424–433. [Google Scholar] [CrossRef]
- Paul, D.S.; Hernández-Zavala, A.; Walton, F.S.; Adair, B.M.; Dědina, J.; Matoušek, T.; Stýblo, M. Examination of the Effects of Arsenic on Glucose Homeostasis in Cell culture and Animal Studies: Development of a Mouse Model for Arsenic-Induced Diabetes. Toxicol. Appl. Pharm. 2007, 222, 305–314. [Google Scholar] [CrossRef] [PubMed]
- Yen, Y.P.; Tsai, K.S.; Chen, Y.W.; Huang, C.F.; Yang, R.S.; Liu, S.H. Arsenic Inhibits Myogenic Differentiation and Muscle Regeneration. Environ. Health Perspect. 2010, 118, 949–956. [Google Scholar] [CrossRef] [PubMed]
- Liu, S.; Guo, X.; Wu, B.; Yu, H.; Zhang, X.; Li, M. Arsenic Induces Diabetic Effects Through Beta-Cell Dysfunction and Increased Gluconeogenesis in Mice. Sci. Rep. 2014, 4, 6894. [Google Scholar] [CrossRef]
- Huang, C.F.; Yang, C.Y.; Chan, D.C.; Wang, C.C.; Huang, K.H.; Wu, C.C.; Tsai, K.S.; Yang, R.S.; Liu, S.H. Arsenic Exposure and Glucose Intolerance/Insulin Resistance in Estrogen-Deficient Female Mice. Environ. Health Perspect. 2015, 123, 1138–1144. [Google Scholar] [CrossRef]
- Díaz-Villaseñor, A.; Cruz, L.; Cebrián, A.; Hernández-Ramírez, R.U.; Hiriart, M.; García-Vargas, G.; Bassol, S.; Sordo, M.; Gandolfi, A.J.; Klimecki, W.T.; et al. Arsenic Exposure and Calpain-10 Polymorphisms Impair the Function of Pancreatic Beta-Cells in Humans: A Pilot Study of Risk Factors for T2DM. PLoS ONE 2013, 8, e51642. [Google Scholar] [CrossRef] [PubMed]
- Divya, S.P.; Pratheeshkumar, P.; Son, Y.O.; Roy, R.V.; Hitron, J.A.; Kim, D.; Dai, J.; Wang, L.; Asha, P.; Huang, B.; et al. Arsenic Induces Insulin Resistance in Mouse Adipocytes and Myotubes Via Oxidative Stress-Regulated Mitochondrial Sirt3-FOXO3a Signaling Pathway. Toxicological. Sci. 2015, 146, 290–300. [Google Scholar] [CrossRef] [PubMed]
- Sowell, M.O.; Boggs, K.P.; Robinson, K.A.; Dutton, S.L.; Buse, M.G. Effects of Insulin and Phospholipase C in Control and Denervated Rat Skeletal Muscle. Am. J. Physiol. 1991, 260 Pt 1, E247–E256. [Google Scholar] [CrossRef]
- Ambrosio, F.; Brown, E.; Stolz, D.; Ferrari, R.; Goodpaster, B.; Deasy, B.; Distefano, G.; Roperti, A.; Cheikhi, A.; Garciafigueroa, Y.; et al. Arsenic Induces Sustained Impairment of Skeletal Muscle and Muscle Progenitor Cell Ultrastructure and Bioenergetics. Free Radic. Biol. Med. 2014, 74, 64–73. [Google Scholar] [CrossRef] [PubMed]
- McDowell, H.E.; Walker, T.; Hajduch, E.; Christie, G.; Batty, I.H.; Downes, C.P.; Hundal, H.S. Inositol Phospholipid 3-Kinase is Activated by Cellular Stress But is Not Required for rhe Stress-Induced Activation of Glucose Transport in L6 Rat Skeletal Muscle Cells. Eur. J. Biochem. 1997, 247, 306–313. [Google Scholar] [CrossRef]
- Farkhondeh, T.; Samarghandian, S.; Azimi-Nezhad, M. The Role of Arsenic in Obesity and Diabetes. J. Cell. Physiol. 2019, 234, 12516–12529. [Google Scholar] [CrossRef]
- Gong, Y.; Liu, J.; Xue, Y.; Zhuang, Z.; Qian, S.; Zhou, W.; Li, X.; Qian, J.; Ding, G.; Sun, Z. Non-Monotonic Dose-Response Effects of Arsenic on Glucose Metabolism. Toxicol. Appl. Pharmacol. 2019, 377, 114605. [Google Scholar] [CrossRef] [PubMed]
- Adebayo, A.O.; Zandbergen, F.; Kozul-Horvath, C.D.; Gruppuso, P.A.; Hamilton, J.W. Chronic exposure to low-dose arsenic modulates lipogenic gene expression in mice. J. Biochem. Mol. Toxicol. 2015, 29, 1–9. [Google Scholar] [CrossRef]
- Singh, D.P.; Yadav, S.K.; Patel, K.; Patel, S.; Patil, G.P.; Bijalwan, V.; Singh, G.; Palkhade, R.; Kondepudi, K.K.; Boparai, R.K.; et al. Short-Term Trivalent Arsenic and Hexavalent Chromium Exposures Induce Gut Dysbiosis and Transcriptional Alteration in Adipose Tissue of Mice. Mol. Biol. Rep. 2023, 50, 1033–1044. [Google Scholar] [CrossRef]
- Liu, P.; Dodson, M.; Li, H.; Schmidlin, C.J.; Shakya, A.; Wei, Y.; Garcia, J.G.N.; Chapman, E.; Kiela, P.R.; Zhang, Q.Y.; et al. Non-Canonical NRF2 Activation Promotes a Pro-Diabetic Shift in Hepatic Glucose Metabolism. Mol. Metab. 2021, 51, 101243. [Google Scholar] [CrossRef]
- Hu, Y.; Jin, X.; Snow, E.T. Effect of Arsenic on Transcription Factor Ap-1 and Nf-Kappab DNA Binding Activity and Related Gene Expression. Toxicol. Lett. 2002, 133, 33–45. [Google Scholar] [CrossRef] [PubMed]
- Yang, C.; Frenkel, K. Arsenic-Mediated Cellular Signal Transduction, Transcription Factor Activation, and Aberrant Gene Expression: Implications in Carcinogenesis. J. Environ. Pathol. Toxicol. Oncol. 2002, 21, 331–342. [Google Scholar] [CrossRef] [PubMed]
- Lee, Y.S.; Lee, E.K.; Oh, H.H.; Choi, C.S.; Kim, S.; Jun, H.S. Sodium Meta-Arsenite Ameliorates Hyperglycemia in Obese Diabetic Db/Db Mice by Inhibition of Hepatic Gluconeogenesis. J. Diabetes Res. 2014, 2014, 961732. [Google Scholar] [CrossRef] [PubMed]
- Aposhian, H.V. Biochemical Toxicology of Arsenic. In Reviews in Biochemical Toxicology; Hodgson, E., Bend, J.R., Philpot, R.M., Eds.; Elsevier: Amsterdam, The Netherlands, 1989; Volume 10, pp. 265–289. [Google Scholar]
- NRC (National Research Council). Arsenic in Drinking Water; National Academy Press: Washington, DC, USA, 1999. [Google Scholar]
- Kennedy, E.P.; Lehninger, A.L. Oxidation of Fatty Acids and Tricarboxylic Acid Cycle Intermediates by Isolated Rat Liver Mitochondria. J. Biol. Chem. 1949, 179, 957–972. [Google Scholar] [CrossRef] [PubMed]
- Wu, M.M.; Chiou, H.Y.; Wang, T.W.; Hsueh, Y.M.; Wang, I.H.; Chen, C.J.; Lee, T.C. Association of Blood Arsenic Levels with Increased Reactive Oxidants and Decreased Antioxidant Capacity in a Human Population of Northeastern Taiwan. Environ. Health Perspec. 2001, 109, 1011–1017. [Google Scholar] [CrossRef] [PubMed]
- Thomas, D.J.; Styblo, M.; Lin, S. The Cellular Metabolism and Systemic Toxicity of Arsenic. Toxicol. Appl. Pharmacol. 2001, 176, 127–144. [Google Scholar] [CrossRef]
- Pan, W.C.; Kile, M.L.; Seow, W.J.; Lin, X.; Quamruzzaman, Q.; Rahman, M.; Mahiuddin, G.; Mostofa, G.; Lu, Q.; Christiani, D.C. Genetic susceptible locus in NOTCH2 interacts with arsenic in drinking water on risk of type 2 diabetes. PLoS ONE 2013, 8, e70792. [Google Scholar] [CrossRef]
- Argos, M. Arsenic Exposure and Epigenetic Alterations: Recent Findings Based on the Illumina 450K DNA Methylation Array. Curr. Environ. Health Rep. 2015, 2, 137–144. [Google Scholar] [CrossRef]
- Michailidi, C.; Hayashi, M.; Datta, S.; Sen, T.; Zenner, K.; Oladeru, O.; Brait, M.; Izumchenko, E.; Baras, A.; VandenBussche, C.; et al. Involvement of Epigenetics and EMT-Related miRNA in Arsenic-Induced Neoplastic Transformation and Their Potential Clinical Use. Cancer Prev. Res. 2015, 8, 208–221. [Google Scholar] [CrossRef]
- Bailey, K.A.; Wu, M.C.; Ward, W.O.; Smeester, L.; Rager, J.E.; García-Vargas, G.; Del Razo, L.M.; Drobná, Z.; Stýblo, M.; Fry, R.C. Arsenic and the Epigenome: Interindividual Differences in Arsenic Metabolism Related to Distinct Patterns of DNA Methylation. J. Biochem. Mol. Tox. 2013, 27, 106–115. [Google Scholar] [CrossRef]
- Rager, J.E.; Bailey, K.A.; Smeester, L.; Miller, S.K.; Parker, J.S.; Laine, J.E.; Drobná, Z.; Currier, J.; Douillet, C.; Olshan, A.F.; et al. Prenatal Arsenic Exposure and the Epigenome: Altered MicroRNAs Associated with Innate and Adaptive Immune Signaling in Newborn Cord Blood. Environ. Mol. Mutagen 2014, 55, 196–208. [Google Scholar] [CrossRef] [PubMed]
- Ren, X.; Gaile, D.P.; Gong, Z.; Qiu, W.; Ge, Y.; Zhang, C.; Huang, C.; Yan, H.; Olson, J.R.; Kavanagh, T.J.; et al. Arsenic Responsive Micrornas In Vivo and Their Potential Involvement in Arsenic-Induced Oxidative Stress. Toxicol. Appl. Pharmacol. 2015, 283, 198–209. [Google Scholar] [CrossRef] [PubMed]
- Abernathy, C.O.; Liu, Y.P.; Longfellow, D.; Aposhian, H.V.; Beck, B.; Fowler, B.; Goyer, R.; Menzer, R.; Rossman, T.; Thompson, C.; et al. Arsenic: Health Effects, Mechanisms of Actions, and Research Issues. Environ. Health Perspec. 1999, 107, 593–597. [Google Scholar] [CrossRef] [PubMed]
- All About Arsenic. 2022. Available online: https://www.allaboutarsenic.org/ (accessed on 25 April 2023).
- Bailey, C.; Farrell, A.; Purty, T.; Taylor, A.; Disney, J. Development of Privacy Features on Anecdata.org, a Free Citizen Science Platform for Collecting Datasets for Climate Change and Related Projects. Front. Clim. 2021, 3, 620100. [Google Scholar] [CrossRef] [PubMed]
- O’Fallon, L.; Finn, S. Citizen Science and Community-Engaged Research in Environmental Public Health. Lab Matters 2015, 4, 5. [Google Scholar]
- Pandya, R. A Framework for Engaging Diverse Communities in Citizen Science in the US. Front. Ecol. Environ. 2012, 10, 314–331. [Google Scholar] [CrossRef]
- Cooper, C.B.; Hawn, C.L.; Larson, L.R.; Parrish, J.K.; Bowser, G.; Cavalier, D.; Dunn, R.R.; Haklay, M.; Gupta, K.K.; Jelks, N.O.; et al. Inclusion in Citizen Science: The Conundrum of Rebranding. Science 2021, 372, 1386–1388. [Google Scholar] [CrossRef]
- Davis, L.F.; Ramírez-Andreotta, M.D. Participatory Research for Environmental Justice: A Critical Interpretive Synthesis. Environ. Health Perspec. 2021, 129, 026001-1-20. [Google Scholar] [CrossRef]
- Wallerstein, N.; Duran, B.; Oetzel, J.G.; Minkler, M. Community-Based Participatory Research for Health: Advancing Social and Health Equity; John Wiley & Sons: San Francisco, CA, USA, 2017. [Google Scholar]
- Ramirez-Andreotta, M.; Brusseau, M.; Artiola, J.; Maier, R.; Gandolfi, A. Building a Co-Created Citizen Science Program with Gardeners Neighboring a Superfund Site: The Gardenroots Case Study. Int. Public Health J. 2015, 7, 139. [Google Scholar]
- Ramirez-Andreotta, M.D.; Lothrop, N.; Wilkinson, S.T.; Root, R.A.; Artiola, J.F.; Klimecki, W.; Loh, M. Analyzing Patterns of Community Interest at a Legacy Mining Waste Site to Assess and Inform Environmental Health Literacy Efforts. J. Environ. Stud. Sci. 2016, 6, 543–555. [Google Scholar] [CrossRef]
- Sandhaus, S.; Kaufmann, D.; Ramirez-Andreotta, M.D. Public Participation, Trust and Data Sharing: Gardens as Hubs for Citizen Science and Environmental Health Literacy Efforts. Int. J. Sci. Edu. Part B 2019, 9, 54–71. [Google Scholar] [CrossRef]
- Zeider, K.; Van Overmeiren, N.; Rine, K.P.; Sandhaus, S.; Sáez, E.A.; Sorooshian, A.; Muñoz, H.C.S.; Ramírez-Andreotta, M.D. Foliar Surfaces as Dust and Aerosol Pollution Monitors: An Assessment by a Mining Site. Sci. Total Environ. 2021, 790, 148164. [Google Scholar] [CrossRef]
- Sorg, T.J.; Chen, A.S.C.; Wang, L. Arsenic Species in Drinking Water Wells in the USA with High Arsenic Concentrations. Water Res. 2014, 48, 156–169. [Google Scholar] [CrossRef] [PubMed]
- USEPA. Arsenic Mitigation Strategies. Available online: https://www.epa.gov/sites/default/files/2015-09/documents/train5-mitigation.pdf (accessed on 25 April 2023).
- Sorg, T.; Wang, L.; Chen, A. The Costs of Small Drinking Water Systems Removing Arsenic from Groundwater. J. Water Supply Res. Technol.-Aqua 2015, 64, 219–234. [Google Scholar] [CrossRef]
- Nicomel, N.R.; Leus, K.; Folens, K.; Van Der Voort, P.; Laing, G.D. Technologies for Arsenic Removal from Water: Curren Status and Future Perspectives. Int. J. Environ. Res. Public Health 2016, 13, 62. [Google Scholar] [CrossRef] [PubMed]
- Fields, K.A.; Chen, A.; Wang, L. Arsenic Removal from Drinking Water by Coagulation/Filtration and Lime Softening Plants. EPA/600/R-00/063. Available online: https://cfpub.epa.gov/si/si_public_record_report.cfm?Lab=NRMRL&dirEntryId=63171 (accessed on 25 April 2023).
- Singh, A.K. Approaches for Removal of Arsenic from Groundwater of Northeastern India. Curr. Sci. 2007, 92, 1506–1515. [Google Scholar]
- Artiola, J.F.; Wilkinson, S.T. How to Lower the Levels of Arsenic in Well Water: What Choices do Arizona Consumers Have? Available online: https://extension.arizona.edu/pubs/how-lower-levels-arsenic-well-water-what-choices-do-arizona-consumers-have (accessed on 25 April 2023).
| Country | Study Population | Age | Adjustments | Duration | As Concentration (In ppb or ppm) | Diabetic Assessment/Methods of Detection | Ref. |
|---|---|---|---|---|---|---|---|
| Bangladesh | 140 diabetic vs. 180 non-diabetic controls recruited with HbA1c level > 7% | ≥20 years | Age, sex, family history of diabetes, smoking habit, betel nut chewing, education | 2010 | 69.3–100.9 ppm in drinking water for 9.8–13.6 years | FBG ≥ 200mg/dL | [113] |
| 115 exposed subjects diagnosed as arsenicosis patients (>50 µg/L As water consumption and skin lesions) and 120 unexposed volunteers | 14–85 years | Age, height and body weight | 2001–2003 | drinking water (0.218 ppm) and spot urine (20.235 ppm) | FBG ≥ 140 mg/dL | [114] | |
| 163 subjects with keratosis exposed to arsenic and 854 unexposed individuals | >30 years | Age, sex and body mass index | NR | 0.01–2.1 ppm in drinking water | history of symptoms: previously diagnosed diabetes, glycosuria and blood sugar level after glucose intake (OGTT) | [115] | |
| 1595 subjects depending on drinking water from wells: 1841 drank arsenic-contaminated drinking water but 114 had not | ≥30 years | Age, sex and body mass index | NR | well water > 0.05 ppm | Glycosuria | [116] | |
| 40 workers occupationally exposed to arsenic, 26 without any known As exposure and 6 who directly handle As containing products | 20–60 years | Sex, occupation, age, smoking habit | NR | 22.3–294.5 nmol per mmol of creatinine in urine sample of the exposed group | glycosylated hemoglobin (HbA1c) 5.4% compared to reference group 4.4% | [117] | |
| Chile | population based cancer case-control study of 1301 participants in Northern Chile | ≥25 years | Age, sex, race, hypertension, cancer, socioeconomic status, smoking status | 2007–2010 | >0.8 ppm arsenic water concentration | physician diagnosed diabetes or oral hypoglycemic medication use | [118] |
| China | 2090 women with singleton pregnancy from the Tongji Maternal and Child Health Cohort (TMCHC) | ≥25 years | Pregnancy, education, income, ethnicity, fetal sex | 2013 | 0.3 ppb | Urine samples and oral glucose tolerance test, FBG ≥ 92 mg/dL | [119] |
| 335 gestational diabetes mellitus and 343 controls without GDM based on a prospective cohort established in Beijing, China | <35–≥35 years | Age, ethnicity, education, occupation, | 2017–2018 | 220 ppm | FBG ≥ 5.1 mmol/L, maternal hair samples | [120] | |
| 1527 pregnant women drawn from Mother and Child Microbiome Cohort (MCMC) study | <30–≥30 years | Education, BMI | 2017–2018 | 0.83 ppb | 75-g oral glucose tolerance test (OGTT), FBG ≥ 5.1 mmol/L, 1 h postprandial ≥ 10.0 mmol/L, or 2 h postprandial glucose ≥ 8.5 mmol/L | [121] | |
| 3474 women who were part of the Ma’anshan Birth Cohort (MABC) Study conducted from the City of Ma’anshan, Anhui Province of China | ≤24 years, 25–29 years, ≥30 years | Maternal age, BMI, gravidity, parity, income, education | 2013–2014 | 0.0047 ppb | FBG ≥ 5.1 mmol/L;1 h, ≥10.0 mmol/L; or 2 h, ≥8.5 mmol/L | [122] | |
| Croatia | 202 adult urban participants from the city of Osijek in eastern Croatia and city of Zagreb in western Croatia | ≥45 years | Age, gender, education, smoking, family history if diabetes, physical activity, dietary consumption, origin of water used for drinking | 2018 | 0.5–361 ppb total urine As | FBG ≥ 3.5 mmol/L, HbA1c ≥ 37 mmol/L, insulin ≥ 15 pmol/L | [123] |
| India | Natives to Nallampatti, an agricultural village in south India and part of the KMCH-NNCD cross-sectional study | ≥20 and ≤85 years | Age, sex, alcohol intake, smoking, tobacco use, BMI, education, occupation, familial diabetic history | 2015 | 4.10–63.30 ppm creatinine units of arsenic | blood investigation included a random glucose, HbA1c, cystatin-c, non-fasting lipid profile, uric acid and hemoglobin | [124] |
| Italy | 3390 art glass workers employed in 17 industrial facilities for at least 1 year | <40, 40–65 and >65 years | Age, sex, history of disease/mortality | 1950–1985 | 3.26 ppb in glassworks (>10 µg/m3 in glassworks) | All causes of death coded according to the 8th revision of the ICD | [125] |
| 258 subjectswith a minimum of two-year residency in the regions and without occupational exposure to As | ≥5 years | Age, sex, source of drinking water | 1993–2008 | 3–215 ppb iAs in drinking water, 2.3–233.7 ng/mL tAs in Urine | FBG ≥ 126 mg/dL, OGTT ≥ 200 mg/dL, HbA1c levels > 7%, self-reported diagnosis, or medication | [103] | |
| 200 diabetic cases and 200 controls | ≥30 years | Age, height, weight, body mass index, smoking habit, family history of diabetes, employment, location | 1960 | intermediate total As concentration in urine (63.5–104 µg/g creatinine) | FBG ≥ 126 mg/100 mL (> or =7.0 mmol/l) or a history of diabetes treated with insulin or oral hypoglycemic agents | [126] | |
| 1160 adults with a minimum 5 year residency in study area | ≥18 years | Age, gender, ethnicity, education/occupation, smoking status, alcohol consumption, recent seafood intake, drinking water sources (well, treatment plant or other) and use and medical history | 2008–2013 | <0.01–419.8 ppb As in drinking water, tAs 0.52–491.5 ppb in urinary As. | FBG ≥ 126 mg/dL, 2HPG ≥ 200 mg/dL, self-reported diagnosis, or medication | [127] | |
| 49 healthy individuals and 77 patients | NR | Age, sex, geographical location history of disease | NR | 0.32–9.82 ppb As in diabetic patients, mean As 3.44 ppb | Urine samples of diabetic patients to test As concentration | [128] | |
| 1451 randomly selected participants from Spain (representative sample of a general population) | ≥20 years | Age, sex, somking status, education, seafood consumption | 2001–2003 | 3.8 ppb of total plasma As, 106,000 ppb of total urine As, 14,900 ppb µg/g of iAs and 66,500 ppb of Asb in participants with diabetes | FBG ≥ 126 mg/dL and glycosylated hemoglobin (HbA1c) level > 6.5% or physician diagnosis or glucose lowering medication use | [129] | |
| Sweden | 43 smelter workers exposed to iAs dust for 13–45 years | 44–70 years | age, height, smoking habit, alcohol consumption | 1987 | 1.6–63 ppb As in work-room air at the smelter | self-reported type 2 diabetes | [130] |
| 12 cases with DM on death certificate and 31 controls employed in a Swedish copper smelter | 30–74 years | Age, history of diseas/death | 1960–1976 | <0.5–>0.5 ppb As | death certificate, medical record | [131] | |
| 5498 art glass workers in southeastern Sweden | ≥45 years | Age, occupation (glassworkers vs. glassblowers, other foundry workers and unspecified glass workers) | 1950–1982 | <1.9 ppb As in Swedish glassworks; <6 µg/m3 As in Swedish glassworks | All causes of death coded according to the 8th revision of the ICD | [132] | |
| Taiwan | 891 adults in southern Taiwan village where arseniasis if hyperendemic | ≥30 years | Age, sex, body mass index, activity level at work | 1960–1970 | 0.1–15 ppm-year or higher | oral glucose tolerance test (OGTT) or self-reported history of diabetes treated with sulfonylurea or insulin | [104] |
| Cancer and noncancer diseases | All age group | Sex, Age | 1971–1994 | 0.25–1.14 ppm As in artesian well water | All causes of death coded according to the 8th or 9th revision of the ICD | [105] | |
| 446 nondiabetic residents in a village in Taiwan | ≥30 years | Age, body mass index and cumulative arsenic exposure | 1988–1989 | median As of artesian well water from 0.7 to 0.93 ppm | FBG ≥ 7.8 mmol/L and/or a 2 h post-load glucose level > or = 11.1 mmol/L. | [133] | |
| 66,667 residents living in endemic areas and 639,667 in nonendemic areas | ≥25 years | Age, sex | 1999–2000 | artesian well water > 0.35 ppm | All causes of death coded according to the 9th revision of the ICD (ICD-9 code 250 and A181) | [134] | |
| 4 townships in southwestern Taiwan where blackfoot disease is endemic | NR | Age, Sex | 1971–2000 | arsenic concentration of artesian well water ranged from 0.35 to 1.14 ppm with a median of 0.78 ppm | All causes of death coded according to the 8th or 9th revision of the ICD (ICD-9 code 250). | [135] | |
| 1297 subjects from an arsenicosis endemic area in southwestern Taiwan | ≥40 years | Age, sex, smoking status, education, exercise, alcohol consumption, betel nut intake | 1990, 2002–2003 | 0.7–0.93 ppm As in well water | FBG, cholesterol, triglycerides, low and high density lipoproteins, urine acid and urine creatinine levels, arsenic methylation patterns and GSTO1 genotypes linked to metabolic syndrome as an early factor for diabetes | [136] | |
| UK | 32 insulin treated (ITDM), 55 non-insulin treated (NITDM) diabetic patients and 30 nondiabetic individuals (C-DNM) from Oxford, England | 18–78 years | Age, body mass index, glucose, insulin | NR | 0.018–0.2 ppm As | Glucose levels and insulin treatment | [137] |
| USA | 4549 American Indian participants | 45–75 years | Age, sociodemographic, smoking and alcohol status, height, weight, blood pressure | 1989–1991, 1998–1999 | 5.9–14 ppm iAs 14.3 ppb in Arizona, 11.9 ppb in Dakota, 7 ppb in Oklahoma | FBG ≥ 126 mg = dL, 2HPG ≥ 200 mg = dL, self-reported diagnosis, or medication | [138] |
| 1393 smelter workers | <20–40+ | Age, sex, race, occupation | 1946–1977 | 0.5–5 ppb As of air concentration in the insecticide building | All causes of death coded according to ICD | [139] | |
| 8014 copper smelter workers in Montana | <20–≥30 | Sex, Race | <1957, 1938–1989 | 0.29–11.3 ppb of airborne As | All causes of death coded according to the 8th or 9th revision of the ICD (ICD-8 codes 460–519) | [140] | |
| 1827 boys and 1305 girls | 2–14 years | Age, sex | 1907–1932 | 140–1600 ppm soil As concentration | All causes of death coded according to death records from the National Death Index, ≥47 and from Washington State (1900–1990), Oregon State (1971–1979), and California State (1960–1990), to locate deaths of cohort members | [141] | |
| Historical ward membership records of the Church of Jesus Christ of Latter-day Saints (LDS) (also known as the Mormons) | <50–80+ | Age, sex | 1977 | mean As 150 ppb, median As 14 to 166 ppb | Death certificate, mortality from hypertensive heart disease | [142] | |
| 1185 respondents from 19 townships in arsenic contaminated area | ≥35 years | Age | 1992–1993 | 2–>10 ppb As, with a median of 2 ppb As | Self reported | [143] | |
| 788 adults aged 20 years or older who participated in the 2003–2004 National Health and Nutrition Examination Survey (NHANES) and had urine arsenic determinations | ≥20 years | Age, sex, race, ethnicity; educational, smoking and alcohol consumption status; and dietary recall | 2003–2004 | 7.1 ppb total As, 3 ppb dmAs, 0.9 ppb arsenobetaine | FBG ≥ 126 mg/dL, self-reported physical diagnosis or use of insulin/oral hypoglycemic medication | [144] | |
| 3925 people on tribal tolls in 13 American Indian communities | <55–≥65 | Age, sex, education, body mass index, smoking status, alcohol consumption | 1989–1991 | 7.9–24.2 ppb urine As, median urine As 14.1 ppb | Glycated hemoglobin and insulin resistance, fasting glucose level of 126 mg/dL or higher, 2 h glucose levels of 200 mg/dL or higher, hemoglobin A1c (HbA1c) of 6.5% or higher, or diabetes treatment | [145] | |
| cohort of American Indians in Arizona, Oklahoma, North Dakota and South Dakota | ≥30 years | Age, ancestry, family relationships | 1998–1999, 2001–2003, 2005–2006, 2014–2015 | median exposure of 5.93 ppb | FBG ≥ 126 mg/dL, or use of insulin or oral hypoglycemic medications | [109] | |
| non-institutionalized civilian resident population from NHANES | ≥20 years | Body mass index, age, gender, race/ethnicity, education, income, cigarette use, alcohol intake and physical activity | 2011–2014 | 246–260.6 ng/h | Spot urine samples, FBG ≥ 100 mg/dL or use of medication to treat hyperglycemia | [146] | |
| 4549 members of 13 tribes based in Arizona, Oklahoma, North Dakota and South Dakota | 45–75 years | Age, sex, study region, medical history, smoking status | 1989–ongoing | 10.2–11.2 nmol per mmol of creatinine in urine sample of the exposed group | Urinary arsenic species measured using HPLC to identify differentially methylated position | [110] | |
| 2919 participants recruited by Strong Heart Family Study | ≥25 years | Age, sex, education, smoking history, alcohol use, medical history | 1998–1999, 2001–2003 | median 0.52 ppb | Urine arsenic, FBG ≥ 126 mg/dL, self-reported physician diagnosis or self-reported use of insulin or oral diabetes treatment | [111] | |
| Pregnant women with and without GDM who received prenatal care at the University of Oklahoma Health Sciences Center (OUHSC) Women’s Clinic and High Risk Pregnancy Clinic | ≥18 years | Maternal age, race/ethnicity, education, income, history of GDM diagnosis | 2009–2010 | 1.25 ppb total arsenic | BG ≥ 135 mg/dL | [147] | |
| 688 participants including type 1, type 2 and control participants from SEARCH, a study being conducted in South Carolina, Colorado and Columbia | 10–22 years | Age, sex, race, education, height, weight | 2003–2006 | 0.0429–0.0502 ppb iAs | Clinical diabetes assigned by the health provider | [148] | |
| 5114 African-American and white men and women who are part of the CRADIA study living in Birmingham, AL; Chicago, IL; Minneapolis, MN; and Oakland, CA | ≥25 years | Age, gender, race, education, smoking status, alcohol consumption, physical activity, BMI, dietary intake | 1987–88; 2015–2016 | <0.0593–≥0.1692 ppm toenail arsenic level | fasting glucose ≥ 126 mg/dL, non-fasting glucose ≥ 200 mg/dL, 2 h postchallenge glucose ≥ 200 mg/dL, hemoglobin A1c ≥ 6.5%, or use of glucose-lowering medications. | [112] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Shakya, A.; Dodson, M.; Artiola, J.F.; Ramirez-Andreotta, M.; Root, R.A.; Ding, X.; Chorover, J.; Maier, R.M. Arsenic in Drinking Water and Diabetes. Water 2023, 15, 1751. https://doi.org/10.3390/w15091751
Shakya A, Dodson M, Artiola JF, Ramirez-Andreotta M, Root RA, Ding X, Chorover J, Maier RM. Arsenic in Drinking Water and Diabetes. Water. 2023; 15(9):1751. https://doi.org/10.3390/w15091751
Chicago/Turabian StyleShakya, Aryatara, Matthew Dodson, Janick F. Artiola, Monica Ramirez-Andreotta, Robert A. Root, Xinxin Ding, Jon Chorover, and Raina M. Maier. 2023. "Arsenic in Drinking Water and Diabetes" Water 15, no. 9: 1751. https://doi.org/10.3390/w15091751
APA StyleShakya, A., Dodson, M., Artiola, J. F., Ramirez-Andreotta, M., Root, R. A., Ding, X., Chorover, J., & Maier, R. M. (2023). Arsenic in Drinking Water and Diabetes. Water, 15(9), 1751. https://doi.org/10.3390/w15091751







