Influence of Seawater Characteristics on Antibiotic Pollutant Removal via Fe(II)-Peroxymonosulfate-Modified Clay
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Experimental
2.2.1. Preparation of the Modified Clay
2.2.2. Preparation of the SMX Solution and Artificial Seawater
2.2.3. Cultivation of the Microalgae
2.2.4. SMX Removal Experiments
2.3. Analysis Methods
2.3.1. Analysis of SMX
2.3.2. Analysis of the PMS Concentration
2.3.3. Analysis of the Fe(II) Concentration
2.3.4. Statistical Analysis
3. Results and Discussion
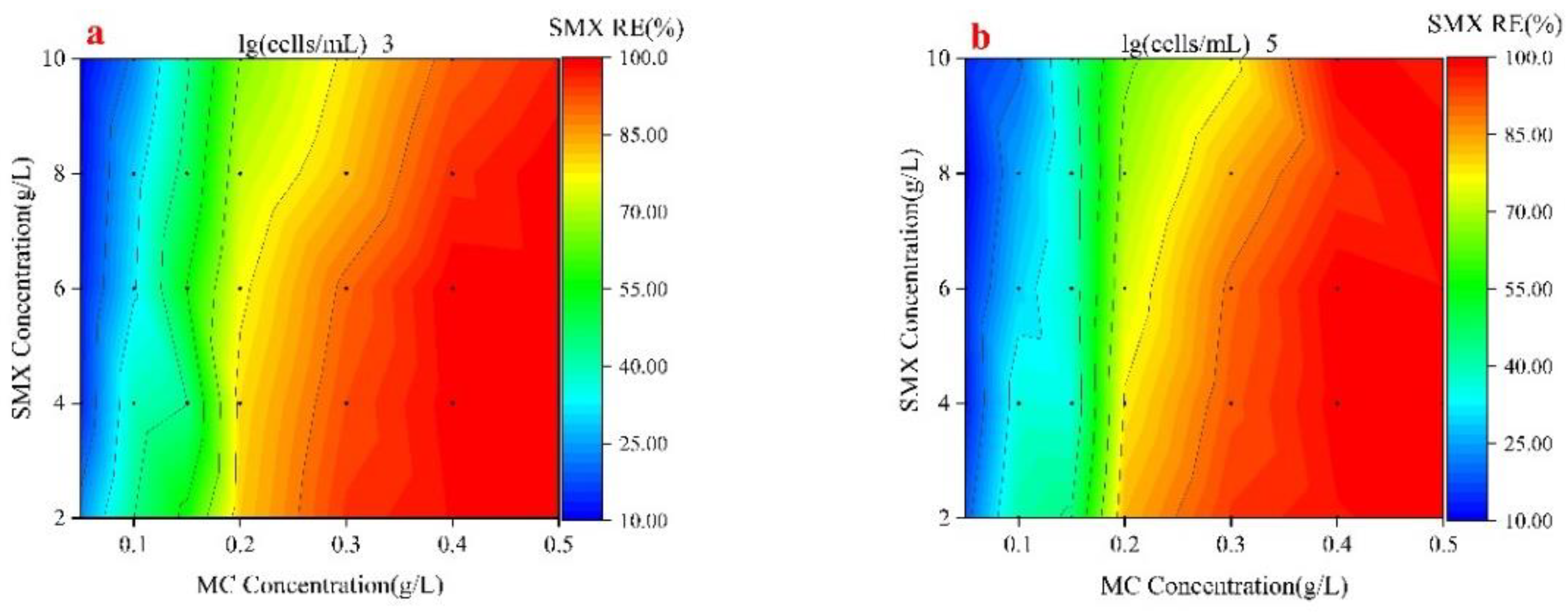
3.1. SMX Removal Efficiency of Fe-PMS-MC
3.2. Influences of Various Seawater Features
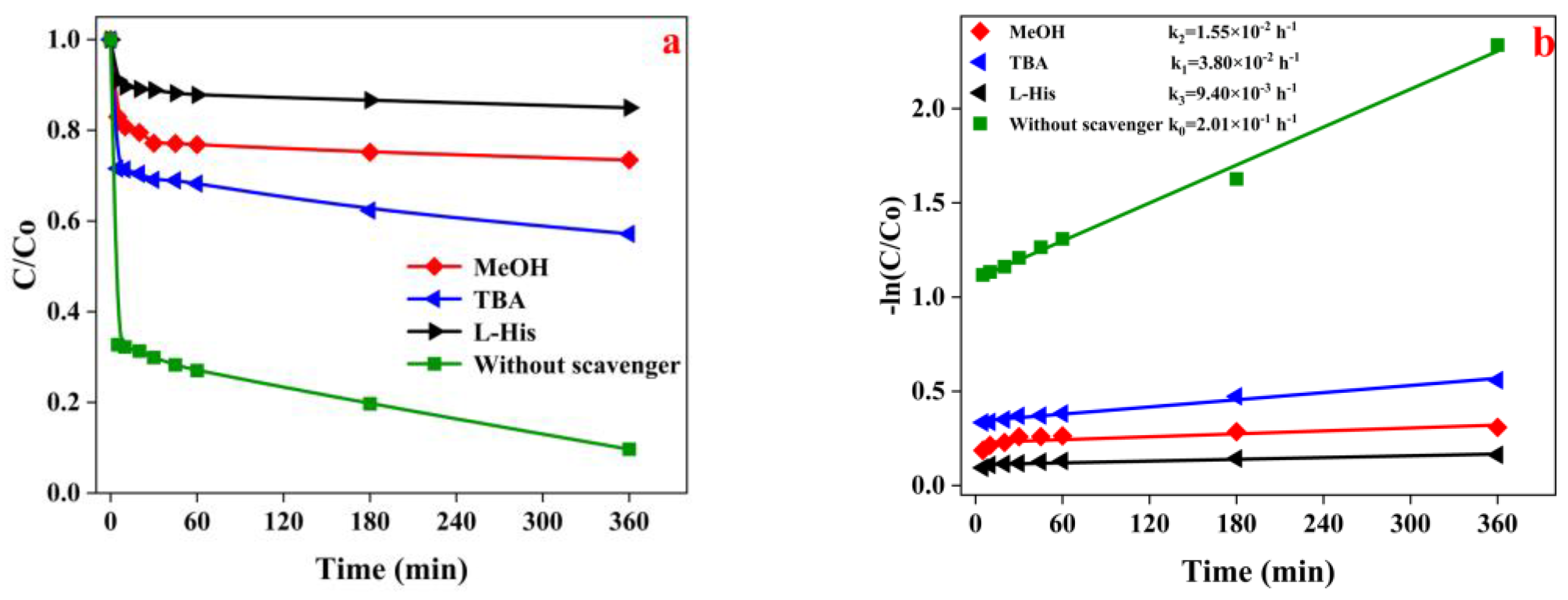
3.3. Mechanism of SMX Removal by the Fe-PMS-MC
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Yu, J.K.; Han, Q.C. Food security of mariculture in China: Evolution, future potential and policy. Mar. Policy 2020, 115, 3892. [Google Scholar] [CrossRef]
- Han, Q.F.; Zhao, S.; Zhang, X.R.; Wang, X.L.; Song, C.; Wang, S.G. Distribution, combined pollution and risk assessment of antibiotics in typical marine aquaculture farms surrounding the Yellow Sea, North China. Environ. Int. 2020, 138, 105551. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.T.; Lin, Y.F.; Zheng, Y.; Meng, F.P. Antibiotics in mariculture systems: A review of occurrence, environmental behavior, and ecological effects. Environ. Pollut. 2022, 293, 118541. [Google Scholar] [CrossRef] [PubMed]
- Li, S.; Shi, W.Z.; Liu, W.; Li, H.M.; Zhang, W.; Hu, J.R.; Ke, Y.C.; Sun, W.L.; Ni, J.R. A duodecennial national synthesis of antibiotics in China’s major rivers and seas (2005–2016). Sci. Total Environ. 2018, 615, 906–917. [Google Scholar] [CrossRef]
- Shi, B.S.; Cheng, X.J.; Chen, H.Z.; Xie, J.; Zhou, Z.H.; Jiang, S.Q.; Peng, X.M.; Zhang, Y.D.; Zhu, D.T.; Lu, Z.Y. Occurrence, source tracking and removal of antibiotics in recirculating aquaculture systems (RAS) in southern China. J. Environ. Manag. 2022, 324, 116311. [Google Scholar] [CrossRef]
- Walsh, T.R.; Weeks, J.; Livermore, D.M.; Toleman, M.A. Dissemination of NDM-1 positive bacteria in the New Delhi environment and its implications for human health: An environmental point prevalence study. Lancet Infect. Dis. 2011, 11, 355–362. [Google Scholar] [CrossRef]
- Berendonk, T.U.; Manaia, C.M.; Merlin, C.; Fatta-Kassinos, D.; Cytryn, E.; Walsh, F.; Burgmann, H.; Sorum, H.; Norstrom, M.; Pons, M.N.; et al. Tackling antibiotic resistance: The environmental framework. Nat. Rev. Microbiol. 2015, 13, 310–317. [Google Scholar] [CrossRef]
- Cui, G.J.; Liu, Z.B.; Xu, W.; Gao, Y.H.; Yang, S.; Grossart, H.P.; Li, M.; Luo, Z.H. Metagenomic exploration of antibiotic resistance genes and their hosts in aquaculture waters of the semi-closed Dongshan Bay (China). Sci. Total Environ. 2022, 838, 155784. [Google Scholar] [CrossRef]
- Xu, S.J.; Liu, Y.; Zhang, J. Transcriptomic mechanisms for the promotion of cyanobacterial growth against eukaryotic microalgae by a ternary antibiotic mixture. Environ. Sci. Pollut. Res. 2022, 29, 58881–58891. [Google Scholar] [CrossRef]
- Xin, R.X.; Yu, X.; Fan, J. Physiological, biochemical and transcriptional responses of cyanobacteria to environmentally relevant concentrations of a typical antibiotic-roxithromycin. Sci. Total Environ. 2022, 814, 152703. [Google Scholar] [CrossRef]
- Ahmed, M.B.; Zhou, J.L.; Ngo, H.H.; Guo, W.S. Adsorptive removal of antibiotics from water and wastewater: Progress and challenges. Sci. Total Environ. 2015, 532, 112–126. [Google Scholar] [CrossRef] [PubMed]
- Blasioli, S.; Martucci, A.; Paul, G.; Gigli, L.; Cossi, M.; Johnston, C.T.; Marchese, L.; Braschi, I. Removal of sulfamethoxazole sulfonamide antibiotic from water by high silica zeolites: A study of the involved host-guest interactions by a combined structural, spectroscopic, and computational approach. J. Colloid Interface Sci. 2014, 419, 148–159. [Google Scholar] [CrossRef]
- Prasannamedha, G.; Kumar, P.S. A review on contamination and removal of sulfamethoxazole from aqueous solution using cleaner techniques: Present and future perspective. J. Clean. Prod. 2020, 250, 119553. [Google Scholar] [CrossRef]
- de Ilurdoz, M.S.; Sadhwani, J.J.; Reboso, J.V. Antibiotic removal processes from water & wastewater for the protection of the aquatic environment-a review. J. Water Process Eng. 2022, 45, 102474. [Google Scholar] [CrossRef]
- Wang, J.L.; Wang, S.Z. Activation of persulfate (PS) and peroxymonosulfate (PMS) and application for the degradation of emerging contaminants. Chem. Eng. J. 2018, 334, 1502–1517. [Google Scholar] [CrossRef]
- Zhou, Y.; Jiang, J.; Gao, Y.; Ma, J.; Pang, S.Y.; Li, J.; Lu, X.T.; Yuan, L.P. Activation of Peroxymonosulfate by Benzoquinone: A Novel Nonradical Oxidation Process. Environ. Sci. Technol. 2015, 49, 12941–12950. [Google Scholar] [CrossRef] [PubMed]
- Ji, Y.F.; Lu, J.H.; Wang, L.; Jiang, M.D.; Yang, Y.; Yang, P.Z.; Zhou, L.; Ferronato, C.; Chovelon, J.M. Non-activated peroxymonosulfate oxidation of sulfonamide antibiotics in water: Kinetics, mechanisms, and implications for water treatment. Water Res. 2018, 147, 82–90. [Google Scholar] [CrossRef]
- Zhang, Y.; Li, L.; Pan, Z.; Zhu, Y.; Shao, Y.; Wang, Y.; Yu, K. Degradation of sulfamethoxazole by UV/persulfate in different water samples: Influential factors, transformation products and toxicity. Chem. Eng. J. 2020, 379, 122354. [Google Scholar] [CrossRef]
- Yang, S.Y.; Wang, P.; Yang, X.; Shan, L.; Zhang, W.Y.; Shao, X.T.; Niu, R. Degradation efficiencies of azo dye Acid Orange 7 by the interaction of heat, UV and anions with common oxidants: Persulfate, peroxymonosulfate and hydrogen peroxide. J. Hazard. Mater. 2010, 179, 552–558. [Google Scholar] [CrossRef]
- Ji, Y.F.; Fan, Y.; Liu, K.; Kong, D.Y.; Lu, J.H. Thermo activated persulfate oxidation of antibiotic sulfamethoxazole and structurally related compounds. Water Res. 2015, 87, 1–9. [Google Scholar] [CrossRef]
- Yu, Z.M.; Song, X.X.; Cao, X.H.; Liu, Y. Mitigation of harmful algal blooms using modified clays: Theory, mechanisms, and applications. Harmful Algae 2017, 69, 48–64. [Google Scholar] [CrossRef] [PubMed]
- Yu, Z.; Chen, N. Emerging trends in red tide and major research progresses. Oceanol. Limnol. Sin. 2019, 50, 474–486. [Google Scholar]
- Lin, Y.X.; Cao, X.H.; Song, X.X.; Wang, N.; Yu, Z.M. Mechanisms and factors affecting the adsorption of sodium alginate onto modified clays. Chin. J. Oceanol. Limnol. 2013, 31, 867–875. [Google Scholar] [CrossRef]
- Qiu, L.X.; Yu, Z.M.; Cao, X.H.; Ji, H.N.; Song, X.X. The mechanism of a new type of modified clay controlling Phaeocystis globosagrowth. J. Oceanol. Limnol. 2020, 38, 1270–1282. [Google Scholar] [CrossRef]
- Lu, G.Y.; Song, X.X.; Yu, Z.M.; Cao, X.H. Application of PAC-modified kaolin to mitigate Prorocentrum donghaiense: Effects on cell removal and phosphorus cycling in a laboratory setting. J. Appl. Phycol. 2017, 29, 917–928. [Google Scholar] [CrossRef]
- Song, W.; Song, X.; Li, J.; Zhang, Y.; Shen, H.; Zhang, P.; Yu, Z. Toxin remained in residual alexandrium pacificum after flocculation with modified clay. Oceanol. Limnol. Sin. 2021, 52, 917–924. [Google Scholar]
- Lu, G.Y.; Song, X.X.; Yu, Z.M.; Cao, X.H.; Yuan, Y.Q. Environmental effects of modified clay flocculation on Alexandrium tamarense and paralytic shellfish poisoning toxins (PSTs). Chemosphere 2015, 127, 188–194. [Google Scholar] [CrossRef]
- Lu, G.Y.; Song, X.X.; Yu, Z.M.; Cao, X.H.; Yuan, Y.Q. Effects of modified clay flocculation on major nutrients and diatom aggregation during Skeletonema costatum blooms in the laboratory. Chin. J. Oceanol. Limnol. 2015, 33, 1007–1019. [Google Scholar] [CrossRef]
- Zhang, Y.; Yu, Z.; Song, X.; Cao, X.; Liu, Y. Study on removal of brown tideAureococcus anophagefferens by modified clay. Acta Oceanol. Sin. 2013, 35, 197–203. [Google Scholar]
- Yu, Z.M.; Zou, J.Z.; Ma, X.N. Application of clays to removal of red tide organisms I. Coagulation of red tide organisms with clays. Chin. J. Oceanol. Limnol. 1994, 12, 193–200. [Google Scholar]
- GB/T 12763.4-2007; The Spesifications for Oceanographic Survey. Part 4: Survey of Chemical Parameters in Sea Water. Standardization Administration of China: Beijing, China, 2007.
- Lai, J.X.; Yu, Z.M.; Song, X.X.; Cao, X.H.; Han, X.T. Responses of the growth and biochemical composition of Prorocentrum donghaiense to different nitrogen and phosphorus concentrations. J. Exp. Mar. Biol. Ecol. 2011, 405, 6–17. [Google Scholar] [CrossRef]
- Liang, C.J.; Huang, C.F.; Mohanty, N.; Kurakalva, R.M. A rapid spectrophotometric determination of persulfate anion in ISCO. Chemosphere 2008, 73, 1540–1543. [Google Scholar] [CrossRef] [PubMed]
- Yang, L.; Chen, Y.; Ouyang, D.; Yan, J.C.; Qian, L.B.; Han, L.; Chen, M.F.; Li, J.; Gu, M.Y. Mechanistic insights into adsorptive and oxidative removal of monochlorobenzene in biochar-supported nanoscale zero-valent iron/persulfate system. Chem. Eng. J. 2020, 400, 125811. [Google Scholar] [CrossRef]
- Wang, Y.; Wang, Y.; Liu, Y.X. Fe2+/heat-coactivated PMS oxidation-absorption system for H2S removal from gas phase. Sep. Purif. Technol. 2022, 286, 120458. [Google Scholar] [CrossRef]
- Wu, G.; Wang, D.; Wang, Y.; Wang, S.; Dong, P.; Zhao, C. Research progresses on activation of peroxymonosulfate and its degradation mechanism to organic pollutants in aqueous solutions. Environ. Prot. Chem. Ind. 2018, 38, 505–513. [Google Scholar]
- Lente, G.; Kalmar, J.; Baranyai, Z.; Kun, A.; Kek, I.; Bajusz, D.; Takacs, M.; Veres, L.; Fabian, I. One- Versus Two-Electron Oxidation with Peroxomonosulfate Ion: Reactions with Iron(II), Vanadium(IV), Halide Ions, and Photoreaction with Cerium(III). Inorg. Chem. 2009, 48, 1763–1773. [Google Scholar] [CrossRef]
- Jiang, M.D.; Lu, J.H.; Ji, Y.F.; Kong, D.Y. Bicarbonate-activated persulfate oxidation of acetaminophen. Water Res. 2017, 116, 324–331. [Google Scholar] [CrossRef]
- Oyekunle, D.T.; Gendy, E.A.; Ifthikar, J.; Chen, Z.Q. Heterogeneous activation of persulfate by metal and non-metal catalyst for the degradation of sulfamethoxazole: A review. Chem. Eng. J. 2022, 437, 135277. [Google Scholar] [CrossRef]
- Ao, X.W.; Liu, W.J. Degradation of sulfamethoxazole by medium pressure UV and oxidants: Peroxymonosulfate, persulfate, and hydrogen peroxide. Chem. Eng. J. 2017, 313, 629–637. [Google Scholar] [CrossRef]
- Leng, L.J.; Wei, L.; Xiong, Q.; Xu, S.Y.; Li, W.T.; Lv, S.; Lu, Q.; Wan, L.P.; Wen, Z.Y.; Zhou, W.G. Use of microalgae based technology for the removal of antibiotics from wastewater: A review. Chemosphere 2020, 238, 124680. [Google Scholar] [CrossRef]
- Xiong, Q.; Hu, L.X.; Liu, Y.S.; Zhao, J.L.; He, L.Y.; Ying, G.G. Microalgae-based technology for antibiotics removal: From mechanisms to application of innovational hybrid systems. Environ. Int. 2021, 155, 106594. [Google Scholar] [CrossRef] [PubMed]
- Wei, L.X.; Li, H.X.; Lu, J.F. Algae-induced photodegradation of antibiotics: A review. Environ. Pollut. 2021, 272, 115589. [Google Scholar] [CrossRef]
- Milh, H.; Cabooter, D.; Dewil, R. Degradation of sulfamethoxazole by ferrous iron activated peroxymonosulfate: Elucidation of the degradation mechanism and influence of process parameters. Chem. Eng. J. 2022, 430, 132875. [Google Scholar] [CrossRef]
- Ji, Y.; Wang, L.; Jiang, M.; Yang, Y.; Yang, P.; Lu, J.; Ferronato, C.; Chovelon, J.-M.J.E.S.; Research, P. Ferrous-activated peroxymonosulfate oxidation of antimicrobial agent sulfaquinoxaline and structurally related compounds in aqueous solution: Kinetics, products, and transformation pathways. Environ. Sci. Pollut. Res. 2017, 24, 19535–19545. [Google Scholar] [CrossRef] [PubMed]
- Tan, C.Q.; Dong, Y.J.; Shi, L.; Chen, Q.; Yang, S.W.; Liu, X.Y.; Ling, J.F.; He, X.F.; Fu, D.F. Degradation of Orange II in ferrous activated peroxymonosulfate system: Efficiency, situ EPR spin trapping and degradation pathway study. J. Taiwan Inst. Chem. Eng. 2018, 83, 74–81. [Google Scholar] [CrossRef]
- Ghanbari, F.; Moradi, M. Application of peroxymonosulfate and its activation methods for degradation of environmental organic pollutants: Review. Chem. Eng. J. 2017, 310, 41–62. [Google Scholar] [CrossRef]
- Lee, H.; Kim, H.I.; Weon, S.; Choi, W.; Hwang, Y.S.; Seo, J.; Lee, C.; Kim, J.H. Activation of Persulfates by Graphitized Nanodiamonds for Removal of Organic Compounds. Environ. Sci. Technol. 2016, 50, 10134–10142. [Google Scholar] [CrossRef]
- Li, Z.; Sun, Y.Q.; Yang, Y.; Han, Y.T.; Wang, T.S.; Chen, J.W.; Tsang, D.C.W. Biochar-supported nanoscale zero-valent iron as an efficient catalyst for organic degradation in groundwater. J. Hazard. Mater. 2020, 383, 121240. [Google Scholar] [CrossRef]
- Wang, B.W.; Wang, Y. A comprehensive review on persulfate activation treatment of wastewater. Sci. Total Environ. 2022, 831, 154906. [Google Scholar] [CrossRef]
- Wang, J.; Shen, M.; Wang, H.L.; Du, Y.S.; Zhou, X.Q.; Liao, Z.W.; Wang, H.B.; Chen, Z.Q. Red mud modified sludge biochar for the activation of peroxymonosulfate: Singlet oxygen dominated mechanism and toxicity prediction. Sci. Total Environ. 2020, 740, 140388. [Google Scholar] [CrossRef]



Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jiang, Y.; Cao, X.; Li, F.; Song, X. Influence of Seawater Characteristics on Antibiotic Pollutant Removal via Fe(II)-Peroxymonosulfate-Modified Clay. Water 2023, 15, 1680. https://doi.org/10.3390/w15091680
Jiang Y, Cao X, Li F, Song X. Influence of Seawater Characteristics on Antibiotic Pollutant Removal via Fe(II)-Peroxymonosulfate-Modified Clay. Water. 2023; 15(9):1680. https://doi.org/10.3390/w15091680
Chicago/Turabian StyleJiang, Yuxin, Xihua Cao, Fang Li, and Xiuxian Song. 2023. "Influence of Seawater Characteristics on Antibiotic Pollutant Removal via Fe(II)-Peroxymonosulfate-Modified Clay" Water 15, no. 9: 1680. https://doi.org/10.3390/w15091680
APA StyleJiang, Y., Cao, X., Li, F., & Song, X. (2023). Influence of Seawater Characteristics on Antibiotic Pollutant Removal via Fe(II)-Peroxymonosulfate-Modified Clay. Water, 15(9), 1680. https://doi.org/10.3390/w15091680





