Factorial Design Statistical Analysis and Optimization of the Adsorptive Removal of COD from Olive Mill Wastewater Using Sugarcane Bagasse as a Low-Cost Adsorbent
Abstract
:1. Introduction
2. Materials and Methods
2.1. OMW Sample
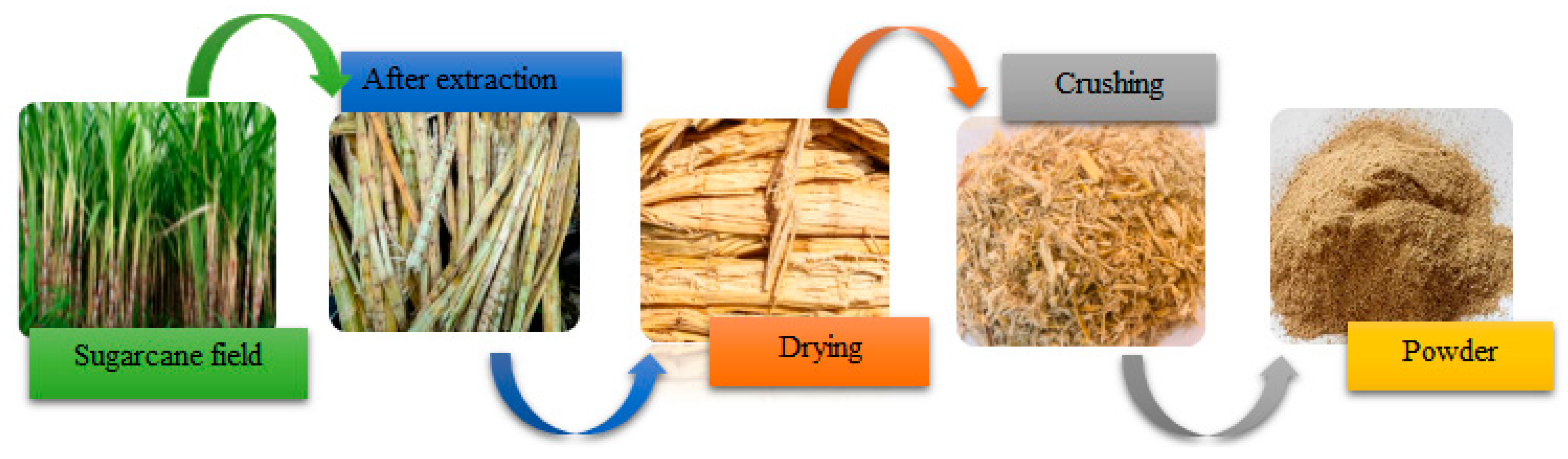
2.2. Sugarcane Bagasse Material
2.3. Batch Adsorption Experiments
2.4. Experimental Design
3. Results and Discussion
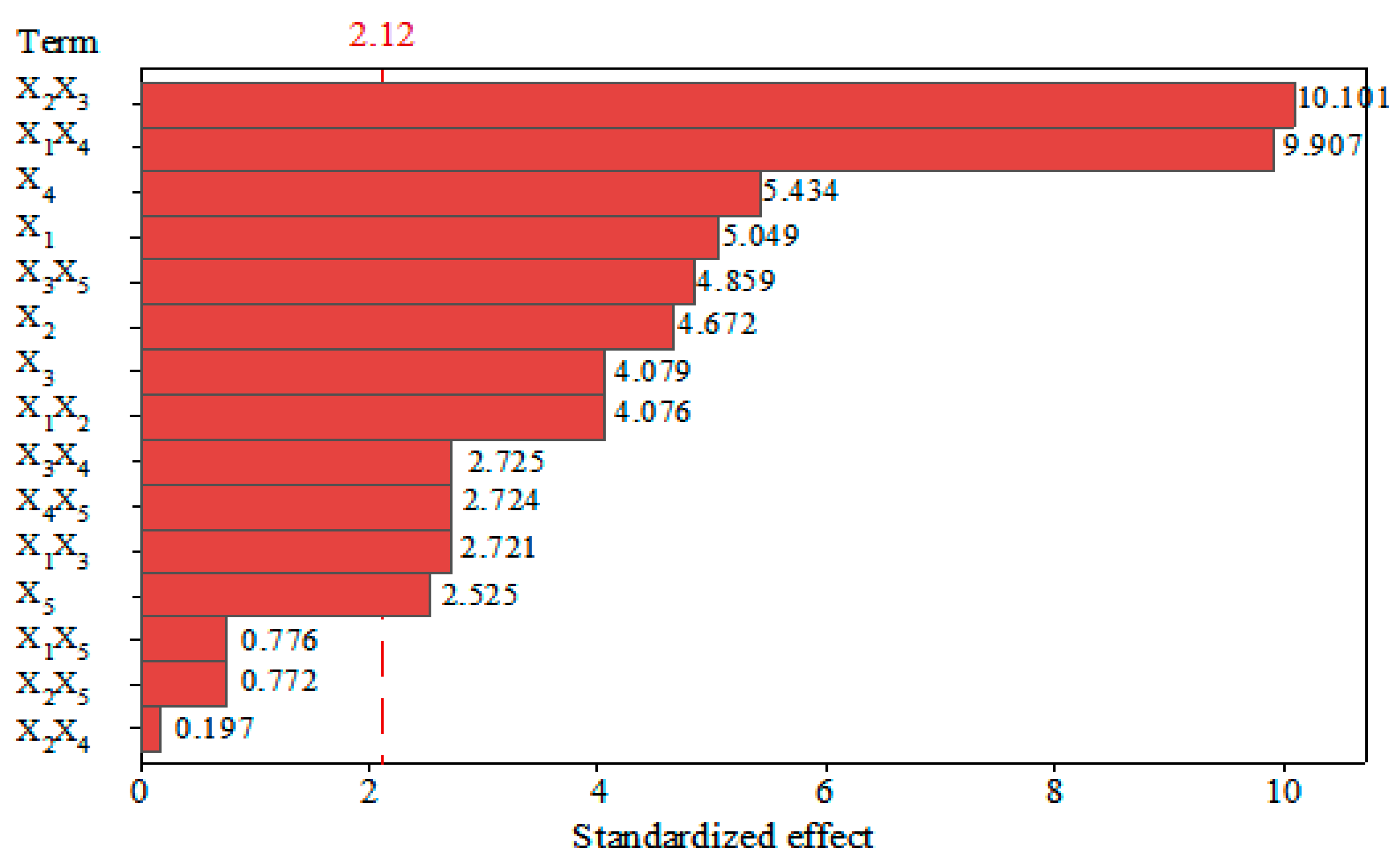
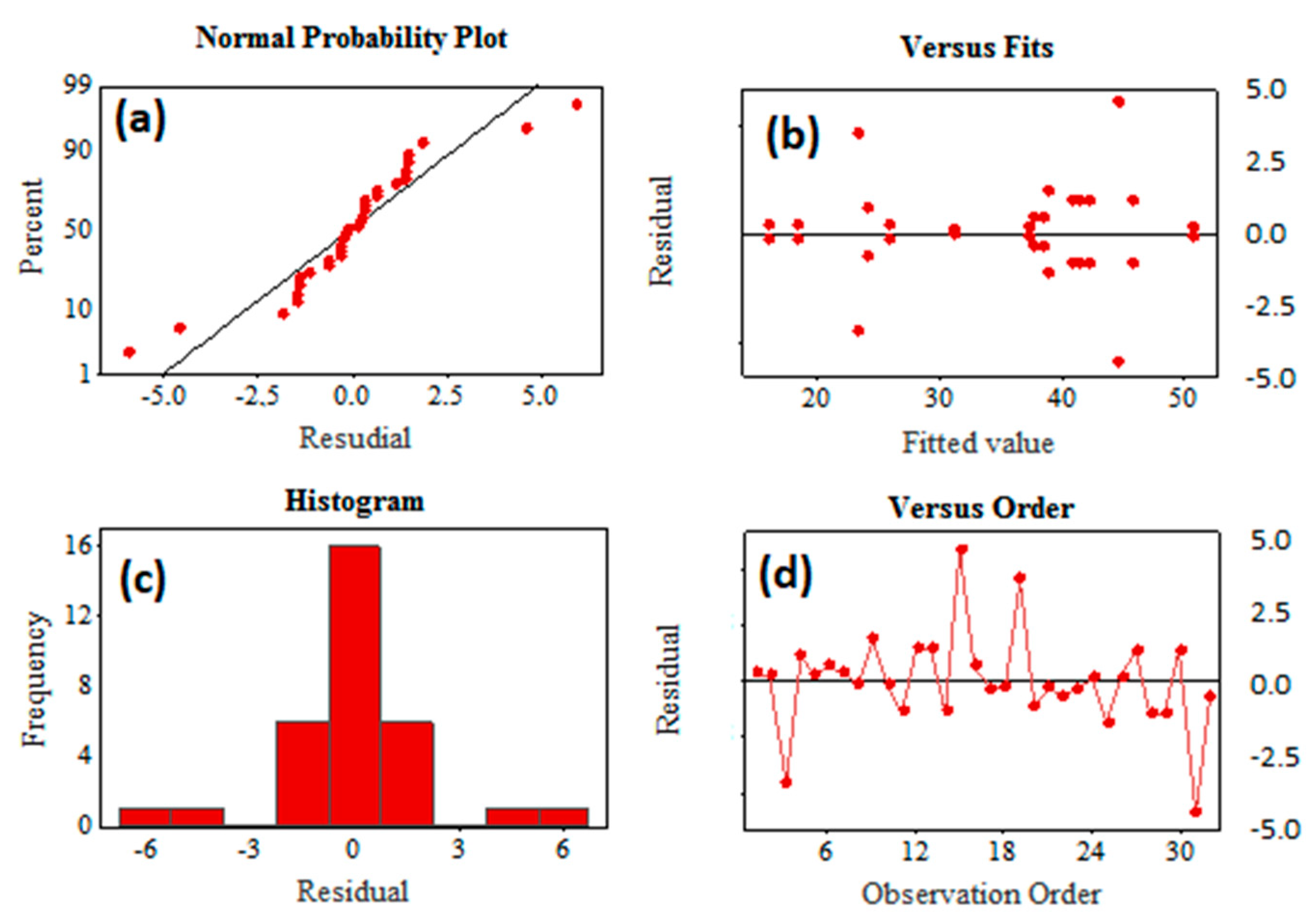
3.1. Analysis of Factorial Design
+b34 X3X4 + b25X2X5 + b35X3X5 + b45X4X5
− 0.00497 X1X3 + 0.001892 X1X4 − 0.09223 X2X3 − 0.001131 X3X4 − 0.01268 X3X5 − 0.000743 X4X5
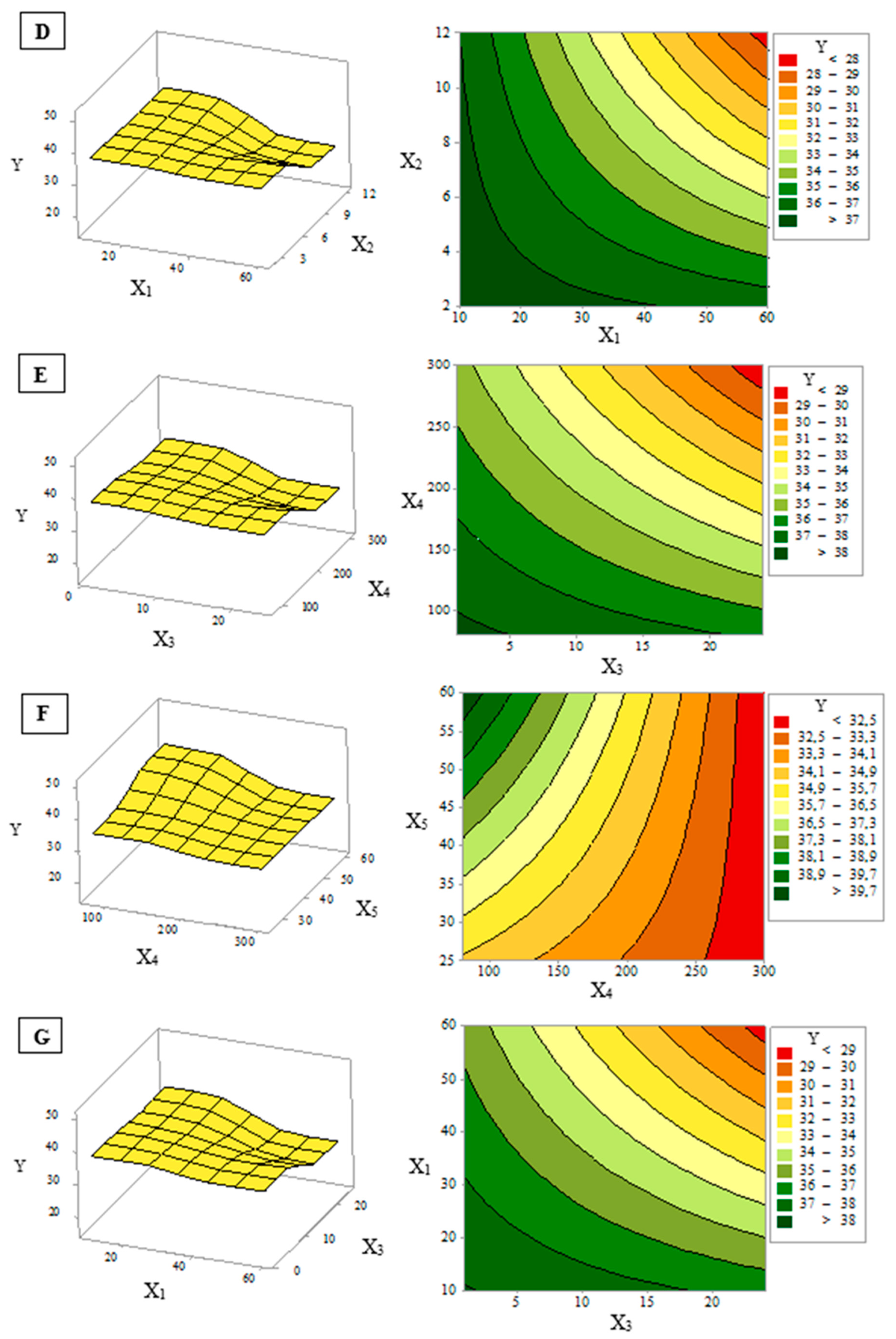
3.2. Response Surface Analysis
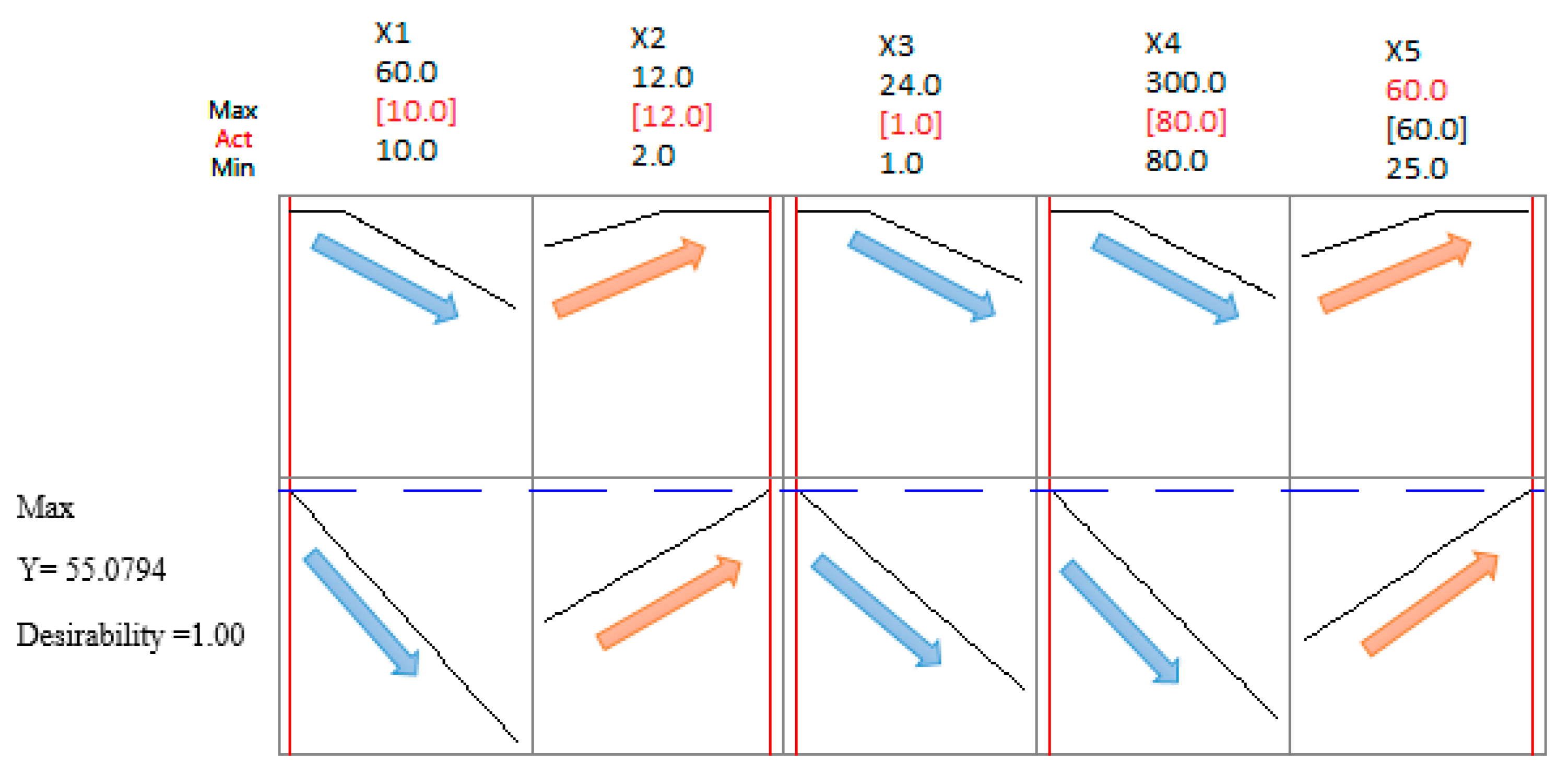
3.3. Optimization Process
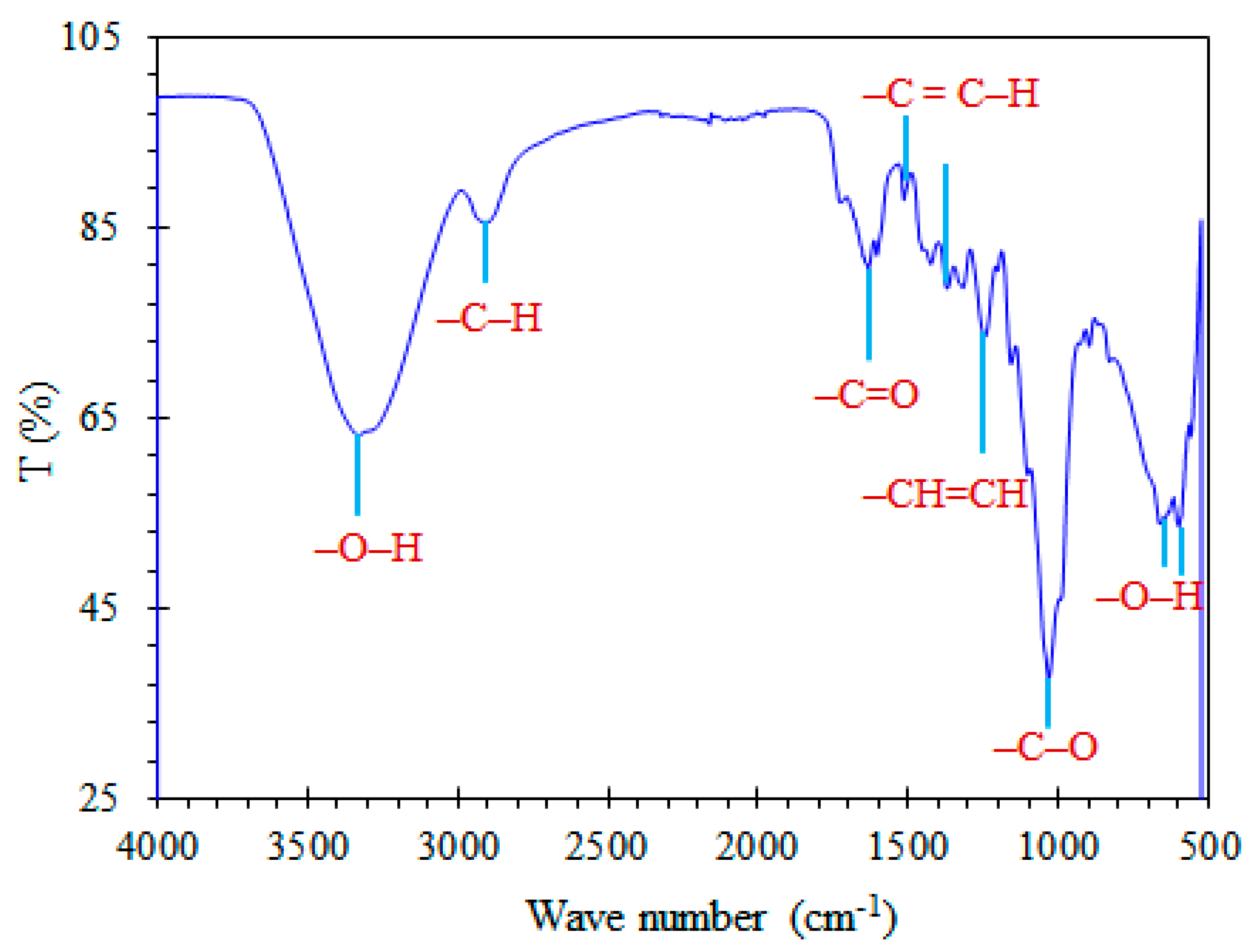
3.4. Characterization of Sugarcane Bagasse
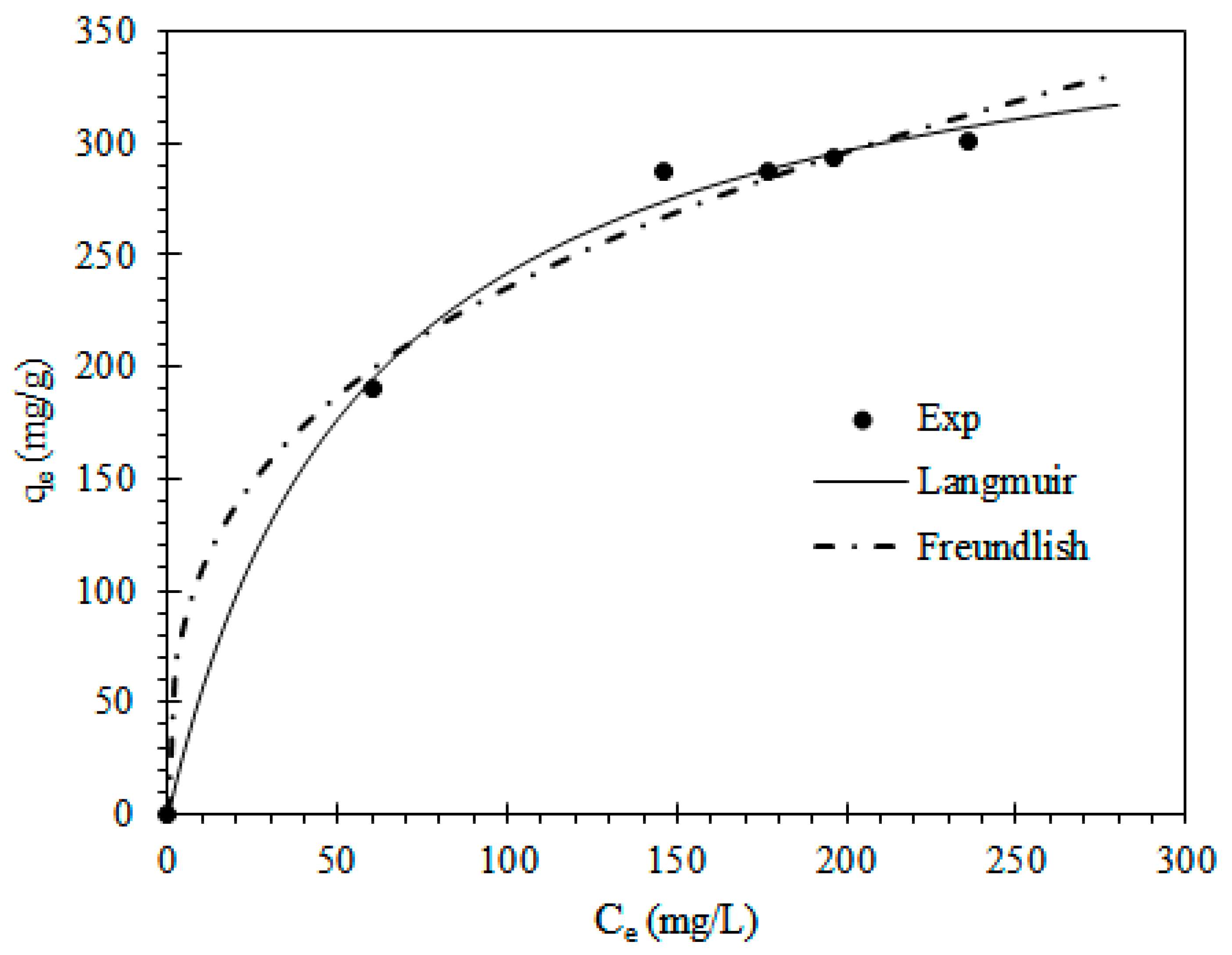
3.5. Adsorption Isotherms
3.6. Kinetic Studies
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Bayramoglu, M.; Kobya, M.; Can, O.T.; Sozbir, M. Operating cost analysis of electrocoagulation of textile dye wastewater. Sep. Purif. Technol. 2004, 37, 117–125. [Google Scholar] [CrossRef]
- Sayan, E. Optimization and modeling of decolorization and COD reduction of reactive dye solutions by ultrasound-assisted adsorption. Chem. Eng. J. 2006, 119, 175–181. [Google Scholar] [CrossRef]
- Rupani, P.F.; Singh, R.P.; Ibrahim, M.H.; Esa, N. Review of current palm oil mill effluent (POME) treatment methods: Vermicomposting as a sustainable practice. World Appl. Sci. J. 2010, 11, 70–81. [Google Scholar]
- Oladipo, A.A.; Adeleye, O.J.; Oladipo, A.S.; Aleshinloye, A.O. Bio-derived MgO nanopowders for BOD and COD reduction from tannery wastewater. J. Water Process Eng. 2017, 16, 142–148. [Google Scholar] [CrossRef]
- Elayadi, F.; El Adlouni, C.; Achak, M.; El Herradi, E.; El Krati, M.; Tahiri, S.; Naman, M.; Naman, F. Effects of raw and treated olive mill wastewater (OMW) by coagulation-flocculation, on the germination and the growth of three plant species (wheat, white beans, lettuce). Mor. J. Chem. 2019, 7, 111–122. [Google Scholar]
- Khatamian, M.; Divband, B.; Shahi, R. Ultrasound assisted co-precipitation synthesis of Fe3O4/bentonite nanocomposite: Performance for nitrate, BOD and COD water treatment. J. Water Process Eng. 2019, 3, 100870. [Google Scholar] [CrossRef]
- Aquilanti, L.; Taccari, M.; Bruglieri, D.; Osimani, A.; Clementi, F.; Comitini, F.; Ciani, M. Integrated biological approaches for olive mill wastewater treatment and agricultural exploitation. Int. Biodeterior. Biodegrad. 2014, 88, 162–168. [Google Scholar] [CrossRef]
- Mastoras, P.; Vakalis, S.; Fountoulakis, M.S.; Gatidou, G.; Katsianou, P.; Koulis, G.; Thomaidis, N.S.; Haralambopoulos, D.; Stasinakis, A.S. Evaluation of the performance of a pilot-scale solar still for olive mill wastewater treatment. J. Clean. Prod. 2022, 365, 132695. [Google Scholar] [CrossRef]
- Esteves, B.M.; Morales-Torres, S.; Madeira, L.M.; Maldonado-Hodar, F.J. Specific adsorbents for the treatment of OMW phenolic compounds by activation of bio-residues from the olive oil industry. J. Environ. Manag. 2022, 306, 114490. [Google Scholar] [CrossRef]
- Alaoui, S.B.; Lamy, E.; Achak, M. Assessment of the impact of diluted and pretreated olive mill wastewater on the treatment efficiency by infiltration-percolation using natural bio-adsorbents. Environ. Sci. Pollut. Res. 2022, 30, 16305–16320. [Google Scholar] [CrossRef]
- Jawad, A.H.; Abdulhameed, A.S.; Mastuli, M.S. Acid-factionalized biomass material for methylene blue dye removal: A comprehensive adsorption and mechanism study. J. Taibah Univ. Sci. 2020, 14, 305–313. [Google Scholar] [CrossRef]
- Mohan, D.; Pittman, C.U., Jr. Activated carbons and low cost adsorbents for remediation of tri- and hexavalent chromium from water. J. Hazard. Mater. 2006, 137, 762–811. [Google Scholar] [CrossRef] [PubMed]
- Hameed, B.H. Spent tea leaves: A new non-conventional and low-cost adsorbent for removal of basic dye from aqueous solutions. J. Hazard. Mater. 2009, 161, 753–759. [Google Scholar] [CrossRef]
- El-Naas, M.H.; Al-Zuhair, S.; Abu Alhaija, M. Reduction of COD in refinery wastewater through adsorption on date-pit activated carbon. J. Hazard. Mater. 2010, 173, 750–757. [Google Scholar] [CrossRef]
- Low, L.W.; Teng, T.T.; Alkarkhi, A.F.M.; Ahmad, A.; Morad, N. Optimization of the Adsorption Conditions for the Decolorization and COD Reduction of Methylene Blue Aqueous Solution using Low-Cost Adsorbent. Water Air Soil Pollut. 2011, 214, 185–195. [Google Scholar] [CrossRef]
- Ismail, M.N.; Aziz, H.A.; Ahmad, M.A.; Yusoff, N.A. Optimization of Areca cathecu Fronds as Adsorbent for Decolorization and COD Removal of Wastewater through the Adsorption Process. Sains Malaysiana. 2015, 11, 1609–1614. [Google Scholar]
- Nayl, A.E.A.; Elkhashab, R.A.; El Malah, T.; Sobhy, M.; Yakout Mohamed, A.; El-Khateeb Mahmoud, M.S.; Ali Hazim, M.A. Adsorption studies on the removal of COD and BOD from treated sewage using activated carbon prepared from date palm waste. Environ. Sci. Pollut. Res. 2017, 24, 22284–22293. [Google Scholar] [CrossRef]
- Mpatani, F.M.; Aryee, A.A.; Kani, A.N.; Wen, K.; Dovi, E.; Qu, L.; Li, Z.; Han, R. Removal of methylene blue from aqueous medium by citrate modified bagasse: Kinetic, Equilibrium and Thermodynamic study. Bioresour. Technol. Rep. 2020, 11, 100463. [Google Scholar] [CrossRef]
- Ge, M.; Du, M.; Zheng, L.; Wang, B.; Zhou, X.; Jia, Z.; Hu, G.; Jahangir Alam, S.M. A maleic anhydride grafted sugarcane bagasse adsorbent and its performance on the removal of methylene blue from related wastewater. Mater. Chem. Phys. 2017, 192, 147–155. [Google Scholar] [CrossRef]
- Surafel, M.B.; Venkatesa, P.S.; Tsegaye, S.T.; Abraham, G.A. Sugarcane bagasse based activated carbon preparation and its adsorption efficacy on removal of BOD and COD from textile effluents: RSM based modeling, optimization and kinetic aspects. Bioresour. Technol. Rep. 2021, 14, 100664. [Google Scholar] [CrossRef]
- Zhang, Z.; Moghaddam, L.; O’Hara, I.M.; Doherty, W.O.S. Congo Red adsorption by ball-milled sugarcane bagasse. Chem. Eng. J. 2011, 178, 122–128. [Google Scholar] [CrossRef]
- Akl, M.A.A.; Dawy, M.B.; Serage, A.A. Efficient removal of phenol from water samples using sugarcane bagasse based activated carbon. J. Anal Bioanal Tech. 2014, 5, 189. [Google Scholar] [CrossRef]
- Abdelhafez, A.; Jianhua, L. Removal of Pb (II) from aqueous solution by using biochars derived from sugar cane bagasse and orange peel. J. Taiwan Inst. Chem. Eng. 2016, 61, 367–375. [Google Scholar] [CrossRef]
- Elayadi, F.; Boumya, W.; Achak, M.; Chhiti, Y.; M’hamdi Alaoui, F.E.; Barka, N.; El Adlouni, C. Experimental and modelling studies of the removal of phenolic compounds from olive mill wastewater by adsorption on sugarcane bagasse. Environ. Chall. 2021, 4, 100184. [Google Scholar] [CrossRef]
- Tran, H.N. Applying Linear Forms of Pseudo-Second-Order Kinetic Model for Feasibly Identifying Errors in the Initial Periods of Time-Dependent Adsorption Datasets. Water 2023, 15, 1231. [Google Scholar] [CrossRef]
- Tawfik, A.; Saleh, S.; Omobayo, A.; Asif, M.; Dafalla, H. Response Surface Optimization and Statistical Analysis of Phenols Adsorption On Diethylenetriamine modified Activated Carbon. J. Clean. Prod. 2018, 182, 960–968. [Google Scholar]
- Moubarik, A.; Grimi, N. Valorization of olive stone and sugar cane bagasse by-products as biosorbents for the removal of cadmium from aqueous solution. Food Res. Int. 2015, 73, 169–175. [Google Scholar] [CrossRef]
- de Sales, P.F.; Magriotis, Z.M.; Rossi, M.A.L.S.; Resende, R.F.; Nunes, C.R. Optimization by Response Surface Methodology of the adsorption of Coomassie Blue dye on natural and acid-treated clays. J. Environ. Manag. 2013, 130, 417e428. [Google Scholar] [CrossRef]
- Leili, M.; Faradmal, J.; Kosravian, F.; Heydari, M. A Comparison study on the removal of phenol from aqueous solution using Organomodified Bentonite and commercial activated carbon. Avicenna J. Environ. Health Eng. 2015, 2, 2698. [Google Scholar] [CrossRef]
- Mondal, N.K.; Roy, S. Optimization study of adsorption parameters for removal of phenol on gastropod shell dust using response surface methodology. Clean Technol. Environ. Policy 2016, 18, 429–447. [Google Scholar] [CrossRef]
- Maleki, S.; Karimi-Jashni, A. Optimization of Ni(II) adsorption onto Cloisite Na+ clay using response surface2 methodology. Chemosphere 2019, 246, 125710. [Google Scholar] [CrossRef] [PubMed]
- Olusegun, S.J.; de Sousa Lima, L.F.; Santina Mohallem, N.D. Enhancement of adsorption capacity of clay through spray drying and surface modification process for wastewater treatment. Chem. Eng. J. 2018, 334, 1719–1728. [Google Scholar] [CrossRef]
- Hafshejani, L.D.; Hooshmand, A.; Naseri, A.A.; Mohammadi, A.S.; Abbasi, F.; Bhatnagar, A. Removal of nitrate from aqueous solution by modified sugarcanebagasse biochar. Ecol. Eng. 2016, 95, 101–111. [Google Scholar] [CrossRef]
- Yangui, A.; Abderrabba, M. Towards a high yield recovery of polyphenols from olive mill wastewater on activated carbon coated with milk proteins: Experimental design and antioxidant activity. J. Food Chem. 2018, 262, 102–109. [Google Scholar] [CrossRef] [PubMed]
- Mandal, A.; Bar, N.; Kumar Das, S. Phenol removal from wastewater using low-cost natural bioadsorbent neem (Azadirachta indica) leaves: Adsorption study and MLR modeling. Sustain. Chem. Pharm. 2020, 17, 100–308. [Google Scholar] [CrossRef]
- Geetha Devi, M.; Shinoon Al-Hashmi, Z.S.; Chandra Sekhar, G. Treatment of vegetable oil mill effluent using crab shell chitosan as adsorbent. Int. J. Environ. Sci. Technol. 2012, 9, 713–718. [Google Scholar] [CrossRef]
- Pourmortazavi, S.M.; Sahebi, H.; Zandavar, H.; Mirsadeghi, S. Fabrication of Fe3O4 nanoparticles coated by extracted shrimp peels chitosan as sustainable adsorbents for removal of chromium contaminates from wastewater: The design of experiment Compos B. J. Eng. 2019, 175, 107130. [Google Scholar] [CrossRef]
- Palin, D., Jr.; Rufato, K.B.; Linde, G.A.; Colauto, N.B.; Caetano, J.; Alberton, O.; Jesus, D.A.; Dragunski, D.C. Evaluation of Pb (II) biosorption utilizing sugarcane bagasse colonized by Basidiomycetes. Environ. Monit Assess. 2016, 188, 188–279. [Google Scholar] [CrossRef]
- Lei, S.; Dongmei, C.; Shungang, W.; Zebin, Y. Adsorption Studies of Dimetridazole and Metronidazole onto Biochar Derived from Sugarcane Bagasse: Kinetic, Equilibrium, and Mechanisms. J. Polym. Environ. 2017, 26, 765–777. [Google Scholar] [CrossRef]
- Srinivasan, A.; Viraraghavan, T. Decolorization of dye wastewaters by biosor-bents: A review. J. Environ. Manag. 2010, 91, 1915–1929. [Google Scholar] [CrossRef]
- Sharma, P.; Kaur, H. Sugarcane bagasse for the removal of erythrosin B and methylene blue from aqueous waste. Appl. Water Sci. 2011, 1, 135–145. [Google Scholar] [CrossRef]
- Kumar, R.; Mago, G.; Balan, V.; Wyman, C.E. Physical and chemical character-izations of corn stover and poplar solids resulting from leading pretreatment technologies. Bioresour. Technol. 2009, 100, 3948–3962. [Google Scholar] [CrossRef] [PubMed]
- Granados, D.A.; Ruiz, R.A.; Vega, L.Y.; Chejne, F. Study of reactivity reduction in sugarcane bagasse as consequence of a torrefaction process. Energy 2017, 139, 818–827. [Google Scholar] [CrossRef]
- Vimonses, V.; Lei, S.M.; Jin, B.; Chowd, C.W.K.; Saint, C. Kinetic study and equilibrium isotherm analysis of Congo Red adsorption by clay materials. J. Chem. Eng. 2009, 148, 354–364. [Google Scholar] [CrossRef]
- Ahmad, A.A.; Hameed, B.H. Reduction of COD and color of dyeing effluent from a cotton textile mill by adsorption onto bamboo-based activated carbon. J. Hazard. Mater. 2009, 172, 1538–1543. [Google Scholar] [CrossRef]
- Akter, S.; Sultana, F.; Kabir, M.R.; Brahma, P.P.; Tasneem, A.; Sarker, N.; Mst, M.; Akter, M.; Kabir, M.; Uddin, M.K. Comparative Study of COD Removal Efficacy from Pharmaceutical Wastewater by Areca Nut Husk Produced and Commercially Available Activated Carbons. Asian J. Environ. Ecol. 2021, 14, 47–56. [Google Scholar] [CrossRef]


| Factors | Variable | Unit | Levels | |
|---|---|---|---|---|
| Low (−) | High (+) | |||
| X1 | Adsorbent dose | g/L | 10 | 60 |
| X2 | pH | - | 2 | 12 |
| X3 | Contact time | h | 1 | 24 |
| X4 | Stirring speed | rpm | 80 | 300 |
| X5 | Temperature | °C | 25 | 60 |
| N° | X1 | X2 | X3 | X4 | X5 | Y (Experimental) | Y (Predicted) |
|---|---|---|---|---|---|---|---|
| 1 | −1 | −1 | −1 | −1 | +1 | 36.39 | 37.46 |
| 2 | 1 | −1 | −1 | −1 | −1 | 41.26 | 43.47 |
| 3 | −1 | 1 | −1 | −1 | −1 | 54.27 | 51.69 |
| 4 | 1 | 1 | −1 | −1 | +1 | 44.69 | 43.75 |
| 5 | −1 | −1 | 1 | −1 | −1 | 35.24 | 35.54 |
| 6 | 1 | −1 | 1 | −1 | +1 | 42.40 | 43.24 |
| 7 | −1 | 1 | 1 | −1 | +1 | 35.53 | 30.66 |
| 8 | 1 | 1 | 1 | −1 | −1 | 41.23 | 40.85 |
| 9 | −1 | −1 | −1 | 1 | −1 | 40.71 | 40.53 |
| 10 | 1 | −1 | −1 | 1 | +1 | 42.12 | 43.90 |
| 11 | −1 | 1 | −1 | 1 | +1 | 33.73 | 32.73 |
| 12 | 1 | 1 | −1 | 1 | −1 | 39.82 | 39.12 |
| 13 | −1 | −1 | 1 | 1 | +1 | 33.23 | 29.86 |
| 14 | 1 | −1 | 1 | 1 | −1 | 31.12 | 34.75 |
| 15 | −1 | 1 | 1 | 1 | −1 | 39.25 | 38.14 |
| 16 | 1 | 1 | 1 | 1 | +1 | 42.12 | 41.79 |
| Source | DF | Adj SS | Adj MS | F-Value | p-Value |
|---|---|---|---|---|---|
| Model | 16 | 3214.58 | 200.912 | 22.24 | 0.000 |
| Linear | 5 | 881.04 | 176.207 | 19.51 | 0.000 |
| X1 | 1 | 224.88 | 224.876 | 24.90 | 0.000 |
| X2 | 1 | 192.57 | 192.567 | 21.32 | 0.000 |
| X3 | 1 | 146.82 | 146.823 | 16.25 | 0.001 |
| X4 | 1 | 260.52 | 260.525 | 28.84 | 0.000 |
| X5 | 1 | 56.25 | 56.245 | 6.23 | 0.025 |
| Interactions | 10 | 2327.91 | 232.791 | 25.77 | 0.000 |
| X1 × X2 | 1 | 146.54 | 146.538 | 16.22 | 0.001 |
| X1 × X3 | 1 | 65.35 | 65.350 | 7.23 | 0.017 |
| X1 × X4 | 1 | 865.84 | 865.839 | 95.86 | 0.000 |
| X1 × X5 | 1 | 5.31 | 5.314 | 0.59 | 0.455 |
| X2 × X3 | 1 | 900.00 | 899.998 | 99.64 | 0.000 |
| X2 × X4 | 1 | 0.34 | 0.343 | 0.04 | 0.848 |
| X2 × X5 | 1 | 5.26 | 5.260 | 0.58 | 0.457 |
| X3 × X4 | 1 | 65.50 | 65.502 | 7.25 | 0.017 |
| X3 × X5 | 1 | 208.31 | 208.306 | 23.06 | 0.000 |
| X4 × X5 | 1 | 65.46 | 65.464 | 7.25 | 0.017 |
| Error | 15 | 135.49 | 9.033 | ||
| Total sum of squares | 31 | 3350.07 | |||
| S square | R2 | R2 (ajust) | R2 (prev) | ||
| 6.2538 | 99.98% | 87.54% | 62.23% | ||
| Composition | Concentration (%) |
|---|---|
| SiO2 | 62.23 |
| SO3 | 24.00 |
| CaO | 11.50 |
| MgO | 0.68 |
| Na2O | 0.60 |
| K2O | 0.51 |
| Al2O3 | 0.28 |
| P2O5 | 0.1 |
| Fe2O3 | 0.10 |
| Models | Equation a | Parameters | Values |
|---|---|---|---|
| Langmuir | qm (mg/g) | 331.92 | |
| KL | 0.019 | ||
| R2 | 0.996 | ||
| Adj-R2 | 0.994 | ||
| Red-χ2 | 78.33 | ||
| BIC | 25.59 | ||
| Freundlich | 1/n | 0.31 | |
| KF | 51.13 | ||
| R2 | 0.991 | ||
| Adj-R2 | 0.985 | ||
| χ2 | 208.11 | ||
| BIC | 31.45 |
| Adsorbent Types | qmax (mg/g) | Ref. |
|---|---|---|
| Sugarcane bagasse | 331.92 | This study |
| Date-pit activated carbon | 252.81 | [14] |
| Activated carbon | 41.3 | [17] |
| Raw bagasse | 77.95 | [15] |
| Bamboo-based activated carbon | 24.39 | [45] |
| Areca Nut Husk | 64.94 | [46] |
| Model | Equation b | Parameters | Value |
|---|---|---|---|
| Pseudo-first-order | qe.exp (mg/g) | 326.92 | |
| qecal (mg/g) | 337.45 | ||
| k1 | 0.20 | ||
| R12 | 0.978 | ||
| Adj-R2 | 0.967 | ||
| χ2 | 467.41 | ||
| BIC | 43.01 | ||
| Pseudo-second-order | qecal (mg/g) | 405.9 | |
| k2 | 0.0006 | ||
| R22 | 0.976 | ||
| Adj-R2 | 0.964 | ||
| χ2 | 503.52 | ||
| BIC | 43.52 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Elayadi, F.; Achak, M.; Boumya, W.; Elamraoui, S.; Barka, N.; Lamy, E.; Beniich, N.; El Adlouni, C. Factorial Design Statistical Analysis and Optimization of the Adsorptive Removal of COD from Olive Mill Wastewater Using Sugarcane Bagasse as a Low-Cost Adsorbent. Water 2023, 15, 1630. https://doi.org/10.3390/w15081630
Elayadi F, Achak M, Boumya W, Elamraoui S, Barka N, Lamy E, Beniich N, El Adlouni C. Factorial Design Statistical Analysis and Optimization of the Adsorptive Removal of COD from Olive Mill Wastewater Using Sugarcane Bagasse as a Low-Cost Adsorbent. Water. 2023; 15(8):1630. https://doi.org/10.3390/w15081630
Chicago/Turabian StyleElayadi, Fatima, Mounia Achak, Wafaa Boumya, Sabah Elamraoui, Noureddine Barka, Edvina Lamy, Nadia Beniich, and Chakib El Adlouni. 2023. "Factorial Design Statistical Analysis and Optimization of the Adsorptive Removal of COD from Olive Mill Wastewater Using Sugarcane Bagasse as a Low-Cost Adsorbent" Water 15, no. 8: 1630. https://doi.org/10.3390/w15081630







