Emerging Contaminants and Their Removal from Aqueous Media Using Conventional/Non-Conventional Adsorbents: A Glance at the Relationship between Materials, Processes, and Technologies
Abstract
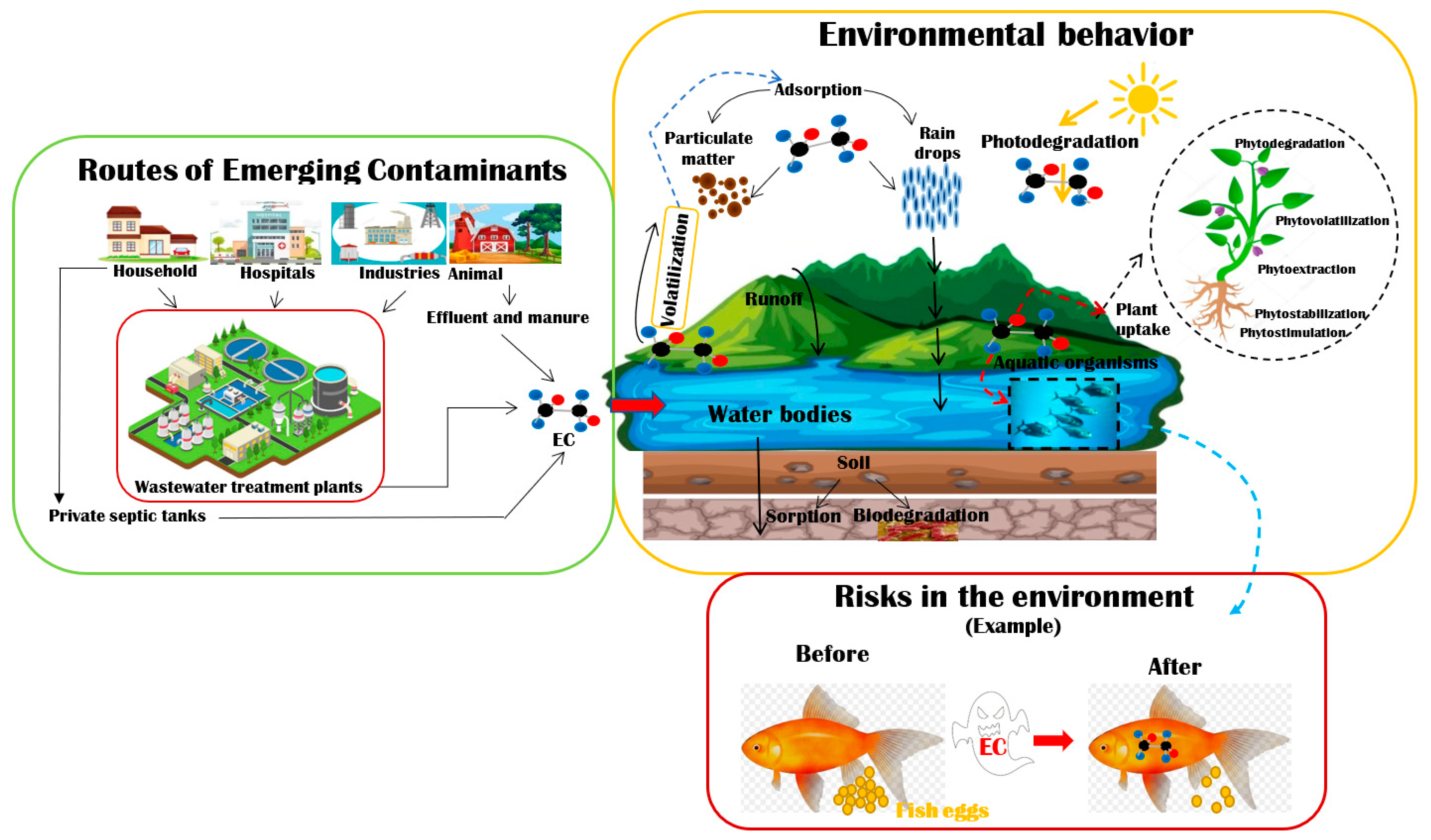
:1. Introduction
2. Characteristics, Concentration, and Toxicity of Some ECs in Water Bodies
2.1. Pharmaceuticals and Personal Care Products (PPCPs)
2.2. Pesticides
2.3. Steroid Hormones
2.4. Micellaneous ECs
2.5. Microplastics
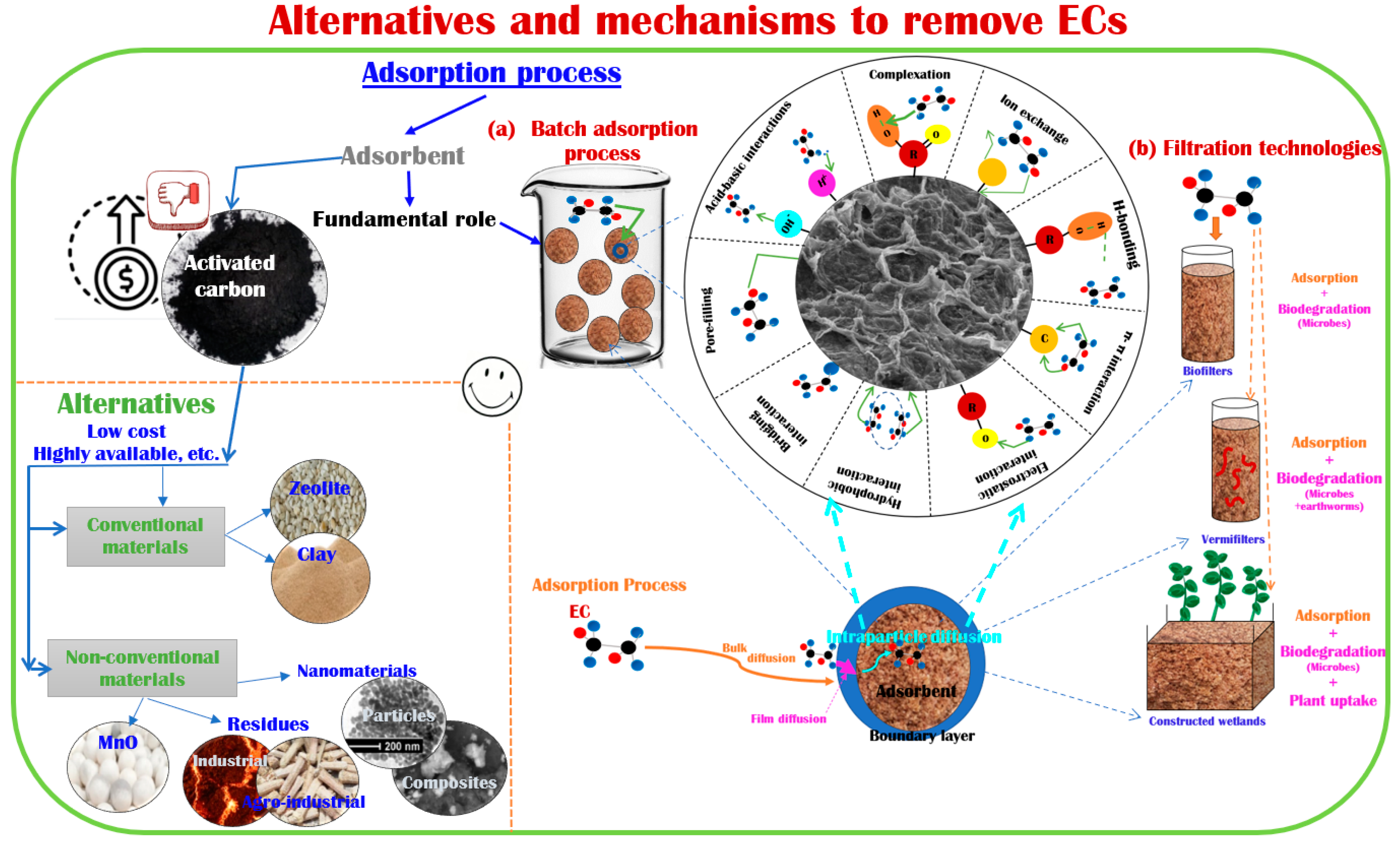
3. Adsorbents Used to Remove ECs from Water
3.1. Conventional Materials
3.1.1. Activated Carbon
3.1.2. Zeolites
3.1.3. Clays
3.2. Inorganic Non-Conventional Materials
3.3. Organic Non-Conventional Materials
3.3.1. Chitosan
3.3.2. Peat
3.3.3. Agricultural/Agro-Industrial Residues
3.3.4. Biochar
3.3.5. Industrial Waste
3.3.6. Polymeric Adsorbents
| Adsorbent Characteristics | Adsorption Behavior | ||||||||
|---|---|---|---|---|---|---|---|---|---|
| Adsorbent | Composition/ Functional Groups/Ions | SA[m2/g] P [cm3/g] | Adsorbent | EC Removed | Adsorption Conditions | Adsorption Mechanism | Removal [%] | Adsorption Capacity [mg/g] | Reference |
| Biochar: S.O. [139] | C, H, O, N [139]/ COOH, C=O, OH−, RCOOR, C6H5OH, (RCO)2O etc. [158] | AS: 2.46–1500 [139] P: 0.21–0.95 [144,158] | Methanol-modified biochar from rice husk | Tetracycline | CT = 0.33, T = 30, pH = 2.0, AD = 1.0 [EC] = 100 | Ion exchange, electrostatic, hydrophobic, pore-filling, and bridging interactions [144] | - | ~80 | [143] |
| Modified biochar + Spiruline sp. | Tetracycline | CT = 120, T = 20–40, pH = 3–9, AD = 0.1 [EC] = 100 | - | 132 | [142] | ||||
| Chitosan: S.O. [131] | α-D-glucosamine [159,160]/ OH−, -NH2 [131] | AS: 1.1–23.4 [122,127] P: 0.002–0.7 [122,127] | Chitosan and Modified chitosan with 2-hydroxy-1-naphthaldehyde | Phenol 2-chlorophenol 4-chlorophenol 2,4-dichlorophenol 2,4,6-trichlorophenol | CT = 3, T = 20, pH = 7.0, AD = 1.0 [EC] = 150 | Electrostatic and hydrophobic interactions and hydrogen bonding [131] | - | 59.7 70.5 96.4 315.5 375.9 | [122] |
| Chitosan grafted with sulfonic acid and cross-linked with glutaraldehyde | Pramipexole | CT = 24, T = 25, pH = 10, [EC] = 0–500 | - | 181 | [125] | ||||
| Sulfonate–grafted chitosan | Pramipexole | CT = 0–24, T = 25, 45, 65, pH = 2–12, AD = 1.0 [EC] = 0–500 | 11–82 | 181–367 | [124] | ||||
| Magnetic modified chitosan | Phenol BPA | CT = 0.67, T = 45, pH = 4.5, AD = 0.6 [EC] = 376–913 | 96 85.5 | - | [123] | ||||
| Peat: N.O. [71] | Cellulose, lignin, humic acid, fulvic acid [127,131]/ alcohols, aldehydes, ketones, carboxylic acids, and phenolic hydroxides [71,131] | AS: 0.9–>200 [71] P: 70–95% [71] | Fibric peat modified. | BPA | CT = 4, T = 25, pH = 6.9, AD = 0.05, [EC] = 45.0 | Hydrophobic interactions, hydrogen bonding [128,129] | - | 31.4 | [128] |
| Raw/modified peat | BPA | CT = 4, T = 25, pH = 7.0, AD = 1.0 [EC] = 2.0 | 80 | 1.7 | [129] | ||||
| Commercial peat soil | Sulfamethoxazole, sulfapyridine | CT = 168, T = 25 ± 3, AD = 20.0, pH = 4.4–9.5 [EC] = 0.15–13.46 | - | Up to 4.05 Up to 0.40 | [130] | ||||
| Agro-industrial residues: N.O. [131] | Cellulose (35–50%), lignin (20–30%), hemicellulose (15–30%), and pectin, among others [131]/ OH−, COH, COOH, ROR, RCO-H, RCOR, C6H5OH, etc. [16] | SA: 0.034–120 [131,134] P: 0.03–4.8 [17,134] | Rice husk | Norfloxacin | CT = 1, T = 25, pH = 6.2, AD= 3.0 [EC] = 5 | Π–π interactions, the formation of H-, -COOH, and C = O bonds, and electrostatic interactions [29,160,161] | 96.95 | 20.1 | [16] |
| Coffee husk | 99.66 | 33.6 | |||||||
| Rice husk | E1 + E2 + E3 | CT = 4, T = 25, AD = 12, [EC] = 3.5–7 | 45–90 | 1.0–2.7 | [69] | ||||
| Pinecone + Pig manure (BCP) | Sulfamethazine, ciprofloxacin, oxytetracycline, florfenicol | CT = 48, T = 20, pH = 7.6–8.5, AD = 1.0 w/w %, [EC] = 1.2 | - | - | [133] | ||||
| Mansonia wood sawdust | 4-nitrophenol | CT = 2.5, T = 26 ± 4, pH = 4.0, AD = 1.5 [EC] = 120 | 22.5–55.5 | 7.4–18.0 | [136] | ||||
| Granulated cork | Phenols Phenol, 2-chlorophenol, 2-nitrophenol, 2,4-dichlorophenol, Pentachlorophenol PPCPs Carbamazepine, naproxen, ketoprofen, diclofenac, triclosan, methyl paraben | CT = 0.5, T = 20 ± 2, pH = 6.0, AD = 5–20 [EC](phenols) = 30.0, [EC](PPCPs) = 1.0 | Phenols 20–100 PPCPs 50–100 | Phenols 0.6–1.6 PPCPs 1.8–3.6 | [135] | ||||
| Charred sawdust of sheesham | Phenol | CT = 1.5, T = 25 ± 2, pH = 2–6, AD = 0.1–10 [EC] = 10–1000 | >95.0 | 300.6–337.5 | [95] | ||||
| Sawdust from Finland wood | Phenol | CT = 3, T = 22, pH = 5.79, AD = 10 [EC] = 20 | Up to 70.4 | Up to 5.5 | [137] | ||||
| Industrial residues: N.O. [145,148] | SiO2, Fe2O3, Al2O3, CaO, MgO, organic compounds [148]/ OH−, C=O, COOH, lactone, quinone, etc. [148,162] | Raw: 2–34 Treated or activated: up to 1800. [145,149,161] Treated or activated: 0.098–0.145 [149,161] | Fertilizer and steel industry wastes | 2,4-dichlorophenoxyacetic acid, carbofuran | CT = 1.67, T = 25, pH = 6.5–7.5, AD = 1.0 [EC] = 88.01–132.76 | Polar interaction, hydrogen bonding, π–π electron–donor–acceptor, acid–base interactions [133,136,137] | - | 212 208 | [150] |
| Sewage Sludge and fish waste | Carbamazepine+ sulfamethoxazole+ trimethoprim | CT = 5, T = 30, pH = 9.39–11.82, AD = 5.0 [EC] = 100 (Of each one) | - | Up to 41.3 Up to 3.8 Up to 13.6 | [149] | ||||
| Sewage Sludge and fish waste | Trimethoprim, sulfamethoxazole | CT = 180, T = 30, pH = 4.53–7.64, AD = 5.0, [EC] = 100 | - | 90.0 5.3 | [148] | ||||
| Paper pulp-based adsorbents + H3PO4 | Carbamazepine, sulfamethoxazole | T = 25, pH = 7.5, [EC] = 5 | - | 92 ± 19 13 ± 0.6 | [163] | ||||
| Phosphoric acid activated corn porous straw carbon | Tetracycline | CT = 12, T = 30, pH = 4, AD = 0.2–1.0 [EC] = 50 | 8.03–97.0 | 227.3 | [147] | ||||
| Activated carbon. from gas mask | Triclosan | CT = 10, T = 24 ± 1, pH = -, AD = 1 × 10−3 [EC] = 10–400 | 2–100 | 85 | [152] | ||||
| Carbon slurry waste | 2-bromophenol, 4-bromophenol, 2,4-dibromophenol | CT = 11, T = 25 ± 2, pH = 5.8–6.8, AD = 1.0 [EC] = 69.2–100.76 | - | 40.7 170.4 190.2 | [162] | ||||
| Activated carbon from sewage sludge | Nimesulide | CT = 2, pH = 8, AD = 1.0 | 98.6 | 275.0 | [151] | ||||
| Polymeric adsorbents: S.O. [63,152] | Depends on the polymer [63,154]/ Tertiary amino, Carboxyl, sulfonic acid, dicyandiamide, amidocyanogen, polyethylene glycol, and 2-Carboxy-3/4-nitrobenzoyl, among others [63] | SA: 15–1000 [63,152] P: 0.16–1.22 [63,152] | Post-cross-linked polystyrene/poly (methyl acryloyl diethylenetriamine) | Phenol, benzoic acid, P-hydroxybenzoic acid | CT = 2.0, T = 25, 35, 45, AD = 2.0 [EC] = 507.3 | Hydrophobic interaction, π–π interaction, ionic attraction, hydrogen bonding; complex formation [63,154] | >90 | 50 190 242.1 | [155] |
| Hyper-crosslinked β-cyclodextrin | BPA | CT = 0.5–12, T = 25, AD = 0.25, [EC] = 40 | 94.45 | 278 | [156] | ||||
| Hyper-crosslinked β-cyclodextrin porous polymer | BPA | CT = 0.17, T = room, AD = 1 [EC] = 22.8 | 80–95 | 22.2 | |||||
| Molecularly Imprinted Polymer | Diclofenac | CT = 1, T = room temperature, pH = 7, AD = 5 [EC] = 1–25 | 100 | 160 | [164] | ||||
3.4. Nanomaterials
3.4.1. Carbon-Based Nanomaterials
- Carbon Nanotubes (CNTs)
- b.
- Graphene
3.4.2. Metal-Based Nano Adsorbents
3.4.3. Nanocomposites
4. Technologies Using Adsorbent Materials to Remove ECs from Wastewater
4.1. Filtration Technologies
4.1.1. Rapid Filtration
4.1.2. Slow Filtration
4.2. Biofiltration
4.2.1. Biofilters
4.2.2. Constructed Wetlands
4.2.3. Vermifilters
4.3. Outlook and Future Perspectives
5. Concluding Remarks
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Rout, P.R.; Zhang, T.C.; Bhunia, P.; Surampalli, R.Y. Treatment technologies for emerging contaminants in wastewater treatment plants: A review. Sci. Total Environ. 2021, 753, 141990. [Google Scholar] [CrossRef]
- Delgado, N.; Bermeo, L.; Hoyos, D.A.; Peñuela, G.A.; Capparelli, A.; Marino, D.; Navarro, A.; Casas-Zapata, J.C. Occurrence and removal of pharmaceutical and personal care products using subsurface horizontal flow constructed wetlands. Water Res. 2020, 187, 116448. [Google Scholar] [CrossRef]
- Stuart, M.; Lapworth, D. Emerging Organic Contaminants in Groundwater. In Smart Sensors, Measurement and Instrumentation; Mukhopadhyay, S., Mason, A., Eds.; Springer: Berlin/Heidelberg, Germany, 2013; pp. 259–284. ISBN 9783642370069. [Google Scholar]
- Tran, N.H.; Reinhard, M.; Gin, K.Y.H. Occurrence and fate of emerging contaminants in municipal wastewater treatment plants from different geographical regions-a review. Water Res. 2018, 133, 182–207. [Google Scholar] [CrossRef]
- Wilkinson, J.; Hooda, P.S.; Barker, J.; Barton, S.; Swinden, J. Occurrence, fate and transformation of emerging contaminants in water: An overarching review of the field. Environ. Pollut. 2017, 231, 954–970. [Google Scholar] [CrossRef]
- Luo, Y.; Guo, W.; Ngo, H.H.; Nghiem, L.D.; Hai, F.I.; Zhang, J.; Liang, S.; Wang, X.C. A review on the occurrence of micropollutants in the aquatic environment and their fate and removal during wastewater treatment. Sci. Total Environ. 2014, 473–474, 619–641. [Google Scholar] [CrossRef]
- Wilkinson, J.L.; Hooda, P.S.; Swinden, J.; Barker, J.; Barton, S. Spatial (bio) accumulation of pharmaceuticals, illicit drugs, plasticisers, per fl uorinated compounds and metabolites in river sediment, aquatic plants and benthic organisms *. Environ. Pollut. 2018, 234, 864–875. [Google Scholar] [CrossRef]
- Zheng, C.; Feng, S.; Liu, P.; Fries, E.; Wang, Q.; Shen, Z.; Liu, H.; Zhang, T. Sorption of Organophosphate Flame Retardants on Pahokee Peat Soil. Clean-Soil Air Water 2016, 44, 1163–1173. [Google Scholar] [CrossRef]
- Catherine, H.N.; Tan, K.-H.; Shih, Y.; Doong, R.; Manu, B.; Ding, J. Surface interaction of tetrabromobisphenol A, bisphenol A and phenol with graphene-based materials in water: Adsorption mechanism and thermodynamic effects. J. Hazard. Mater. Adv. 2023, 9, 100227. [Google Scholar] [CrossRef]
- Baloo, L.; Isa, M.H.; Sapari, N.B.; Jagaba, A.H.; Wei, L.J.; Yavari, S.; Razali, R.; Vasu, R. Adsorptive removal of methylene blue and acid orange 10 dyes from aqueous solutions using oil palm wastes-derived activated carbons. Alex. Eng. J. 2021, 60, 5611–5629. [Google Scholar] [CrossRef]
- Birniwa, A.H.; Abubakar, A.S.; Huq, A.K.O.; Mahmud, H.N.M.E. Polypyrrole-polyethyleneimine (Ppy-PEI) nanocomposite: An effective adsorbent for nickel ion adsorption from aqueous solution. J. Macromol. Sci. Part A Pure Appl. Chem. 2021, 58, 206–217. [Google Scholar] [CrossRef]
- Birniwa, A.H.; Kehili, S.; Ali, M.; Musa, H.; Ali, U.; Rahman, S.; Kutty, M.; Jagaba, A.H.; Sa, S.; Tag-eldin, E.M.; et al. Polymer-Based Nano-Adsorbent for the Removal of Lead Ions: Kinetics Studies and Optimization by Response Surface Methodology. Separations 2022, 9, 356. [Google Scholar] [CrossRef]
- Castro, D.; Rosas-Laverde, N.M.; Aldás, M.B.; Almeida-Naranjo, C.; Guerrero, V.H.; Pruna, A.I. Chemical modification of agro-industrial waste-based bioadsorbents for enhanced removal of Zn(II) ions from aqueous solutions. Materials 2021, 14, 2134. [Google Scholar] [CrossRef]
- Almeida-Naranjo, C.E.; Morillo, B.; Aldás, M.B.; Garcés, N.; Debut, A.; Guerrero, V.H. Zinc removal from synthetic waters using magnetite/graphene oxide composites. Remediation 2023, 33, 135–150. [Google Scholar] [CrossRef]
- Sellaoui, L.; Bouzidi, M.; Franco, D.S.P.; Alshammari, A.S.; Gandouzi, M.; Georgin, J.; Mohamed, N.B.H.; Erto, A.; Badawi, M. Exploitation of Bauhinia angmuir residual fruit powder for the adsorption of cationic dyes. Chem. Eng. J. 2023, 456, 141033. [Google Scholar] [CrossRef]
- Paredes-Laverde, M.; Silva-agredo, J.; Torres-palma, R.A. Removal of norfloxacin in deionized, municipal water and urine using rice (Oryza sativa) and coffee (Coffee angmu) husk wastes as natural adsorbents. J. Environ. Manag. 2018, 213, 98–108. [Google Scholar] [CrossRef]
- Quesada, H.B.; Alves Baptista, A.T.; Cusioli, L.F.; Seibert, D.; de Oliveira Bezerra, C.; Bergamasco, R. Surface water pollution by pharmaceuticals and an alternative of Removal by low-cost adsorbents: A review. Chemosphere 2019, 222, 766–780. [Google Scholar] [CrossRef] [PubMed]
- Ebele, A.J.; Abou-Elwafa Abdallah, M.; Harrad, S. Pharmaceuticals and personal care products (PPCPs) in the freshwater aquatic environment. Emerg. Contam. 2017, 3, 1–16. [Google Scholar] [CrossRef]
- Wang, J.; Wang, S. Removal of pharmaceuticals and personal care products (PPCPs) from wastewater: A review. J. Environ. Manag. 2016, 182, 620–640. [Google Scholar] [CrossRef] [PubMed]
- Brausch, J.M.; Rand, G.M. A review of personal care products in the aquatic environment: Environmental concentrations and toxicity. Chemosphere 2011, 82, 1518–1532. [Google Scholar] [CrossRef]
- Dhillon, G.S.; Kaur, S.; Pulicharla, R.; Brar, S.K.; Cledón, M.; Verma, M.; Surampalli, R.Y. Triclosan: Current status, occurrence, environmental risks and bioaccumulation potential. Int. J. Environ. Res. Public Health 2015, 12, 5657–5684. [Google Scholar] [CrossRef]
- Kaur, H.; Bansiwal, A.; Hippargi, G.; Pophali, G.R. Effect of hydrophobicity of pharmaceuticals and personal care products for adsorption on activated carbon: Adsorption isotherms, kinetics and mechanism. Environ. Sci. Pollut. Res. 2017, 25, 20473–20485. [Google Scholar] [CrossRef]
- Adeel, M.; Song, X.; Wang, Y.; Francis, D.; Yang, Y. Environmental impact of estrogens on human, animal and plant life: A critical review. Environ. Int. 2017, 99, 107–119. [Google Scholar] [CrossRef]
- Kaur, H.; Hippargi, G.; Pophali, G.R.; Bansiwal, A. Biomimetic lipophilic activated carbon for enhanced removal of triclosan from water. J. Colloid Interface Sci. 2019, 535, 111–121. [Google Scholar] [CrossRef]
- Zepon Tarpani, R.R.; Azapagic, A. Life cycle environmental impacts of advanced wastewater treatment techniques for removal of pharmaceuticals and personal care products (PPCPs). J. Environ. Manag. 2018, 215, 258–272. [Google Scholar] [CrossRef] [PubMed]
- Valle-Sistac, J.; Molins-delgado, D.; Díaz, M.; Ibáñez, L.; Barceló, D.; Díaz-cruz, M.S. Determination of parabens and benzophenone-type UV fi lters in human placenta. First description of the existence of benzyl paraben and benzophenone-4. Environ. Int. 2016, 88, 243–249. [Google Scholar] [CrossRef] [PubMed]
- Pietrzak, D.; Kmiecik, E.; Malina, G.; Wa, K. Fate of selected neonicotinoid insecticides in soil e water systems: Current state of the art and knowledge gaps. Chemosphere 2020, 255, 126981. [Google Scholar] [CrossRef]
- Glinski, D.A.; Purucker, S.T.; Van Meter, R.J.; Black, M.C.; Henderson, W.M. Analysis of pesticides in surface water, stemflow, and throughfall in an agricultural area in South Georgia, USA. Chemosphere 2018, 209, 496–507. [Google Scholar] [CrossRef]
- de Souza, R.M.; Seibert, D.; Quesada, H.B.; de Jesus Bassetti, F.; Fagundes-Klen, M.R.; Bergamasco, R. Occurrence, impacts and general aspects of pesticides in surface water: A review. Process Saf. Environ. Prot. 2020, 135, 22–37. [Google Scholar] [CrossRef]
- Villamar-Ayala, C.A.; Carrera-Cevallos, J.V.; Espinoza-Montero, P.J.; Alejandra, C.; Carrera-Cevallos, J.V. Technology Fate, eco-toxicological characteristics, and treatment processes applied to water polluted with glyphosate: A critical review. Crit. Rev. Environ. Sci. Technol. 2019, 49, 1476–1514. [Google Scholar] [CrossRef]
- Kaur, Y.; Bhatia, Y.; Chaudhary, S.; Chaudhary, G.R. Comparative performance of bare and functionalize ZnO nanoadsorbents for pesticide removal from aqueous solution. J. Mol. Liq. 2017, 234, 94–103. [Google Scholar] [CrossRef]
- Franco, D.S.P.; Georgin, J.; Lima, E.C.; Silva, L.F.O. Advances made in removing paraquat herbicide by adsorption technology: A review. J. Water Process Eng. 2022, 49, 102988. [Google Scholar] [CrossRef]
- Zavareh, S.; Farrokhzad, Z.; Darvishi, F. Modification of zeolite 4A for use as an adsorbent for glyphosate and as an antibacterial agent for water. Ecotoxicol. Environ. Saf. 2018, 155, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Ilyas, H.; van Hullebusch, E.D. Performance comparison of different constructed wetlands designs for the removal of personal care products. Int. J. Environ. Res. Public Health 2020, 17, 3091. [Google Scholar] [CrossRef] [PubMed]
- Ahmad, J.; Naeem, S.; Ahmad, M.; Usman, A.R.A.; Al-wabel, M.I. A critical review on organic micropollutants contamination in wastewater and removal through carbon nanotubes. J. Environ. Manag. 2019, 246, 214–228. [Google Scholar] [CrossRef] [PubMed]
- Adeel, M.; Yang, Y.S.; Wang, Y.Y.; Song, X.M.; Ahmad, M.A.; Rogers, H. Uptake and transformation of steroid estrogens as emerging contaminants influence plant development. Environ. Pollut. 2018, 243, 1487–1497. [Google Scholar] [CrossRef]
- Diniz, V.; Gasparini Fernandes Cunha, D.; Rath, S. Adsorption of recalcitrant contaminants of emerging concern onto activated carbon: A laboratory and pilot-scale study. J. Environ. Manag. 2023, 325, 116489. [Google Scholar] [CrossRef]
- Palmer, M.; Hatley, H. The role of surfactants in wastewater treatment: Impact, removal and future techniques: A critical review. Water Res. 2018, 147, 60–72. [Google Scholar] [CrossRef]
- Praveena, S.M.; Cheema, M.S.; Guo, H.-R. Non-nutritive artificial sweeteners as an emerging contaminant in environment: A global review and risks perspectives. Ecotoxicol. Environ. Saf. 2019, 170, 699–707. [Google Scholar] [CrossRef]
- Luo, J.; Zhang, Q.; Cao, M.; Wu, L.; Cao, J.; Fang, F.; Li, C. Ecotoxicity and environmental fates of newly recognized contaminants-artificial sweeteners: A review. Sci. Total Environ. 2019, 653, 1149–1160. [Google Scholar] [CrossRef]
- Lapworth, D.J.; Baran, N.; Stuart, M.E.; Ward, R.S. Emerging organic contaminants in groundwater: A review of sources, fate and occurrence. Environ. Pollut. 2012, 163, 287–303. [Google Scholar] [CrossRef]
- Kumar, K.S. Fluorinated Organic Chemicals: A Review. Res. J. Chem. Environ. 2005, 9, 50–79. [Google Scholar]
- Wang, Y.; Chang, W.; Wang, L.; Zhang, Y.; Zhang, Y.; Wang, M.; Wang, Y.; Li, P. A review of sources, multimedia distribution and health risks of novel fluorinated alternatives. Ecotoxicol. Environ. Saf. 2019, 182, 109402. [Google Scholar] [CrossRef]
- Ivanković, T.; Hrenović, J. Surfactants in the environment. J. Surfactants Environ. 2010, 61, 95–110. [Google Scholar] [CrossRef] [PubMed]
- Horton, A.A.; Walton, A.; Spurgeon, D.J.; Lahive, E.; Svendsen, C. Microplastics in freshwater and terrestrial environments: Evaluating the current understanding to identify the knowledge gaps and future research priorities. Sci. Total Environ. 2017, 586, 127–141. [Google Scholar] [CrossRef] [PubMed]
- Silva, A.B.; Bastos, A.S.; Justino, C.I.L.; Duarte, A.C.; Rocha-santos, T.A.P. Microplastics in the environment: Challenges in analytical chemistry—A review. Anal. Chim. Acta 2018, 1017, 1–19. [Google Scholar] [CrossRef]
- Avio, C.G.; Gorbi, S.; Regoli, F. Plastics and microplastics in the oceans: From emerging pollutants to emerged threat. Mar. Environ. Res. 2017, 128, 2–11. [Google Scholar] [CrossRef]
- Padervand, M.; Lichtfouse, E.; Robert, D.; Wang, C. Removal of microplastics from the environment. A review. Environ. Chem. Lett. 2020, 18, 807–828. [Google Scholar] [CrossRef]
- Li, J.; Liu, H.; Paul Chen, J. Microplastics in freshwater systems: A review on occurrence, environmental effects, and methods for microplastics detection. Water Res. 2018, 137, 362–374. [Google Scholar] [CrossRef]
- Zhou, Y.; Wang, T.; Zhi, D.; Guo, B.; Zhou, Y. Applications of nanoscale zero-valent iron and its composites to the removal of antibiotics: A review. J. Mater. Sci. 2019, 54, 12171–12188. [Google Scholar] [CrossRef]
- Abuwatfa, W.H.; Al-Muqbel, D.; Al-Othman, A.; Halalsheh, N.; Tawalbeh, M. Insights into the removal of microplastics from water using biochar in the era of COVID-19: A mini review. Case Stud. Chem. Environ. Eng. 2021, 4, 100151. [Google Scholar] [CrossRef]
- Shi, X.; Zhang, X.; Gao, W.; Zhang, Y.; He, D. Removal of microplastics from water by magnetic nano-Fe3O4. Sci. Total Environ. 2022, 802, 149838. [Google Scholar] [CrossRef] [PubMed]
- León, G.R.; Aldás, M.B.; Guerrero, V.H.; Landázuri, A.C.; Almeida-Naranjo, C.E. Caffeine and irgasan removal from water using bamboo, laurel and moringa residues impregnated with commercial TiO2 nanoparticles. MRS Adv. 2019, 4, 3553–3567. [Google Scholar] [CrossRef]
- Gavrilescu, M.; Demnerová, K.; Aamand, J.; Agathos, S.; Fava, F. Emerging pollutants in the environment: Present and future challenges in biomonitoring, ecological risks and bioremediation. New Biotechnol. 2015, 32, 147–156. [Google Scholar] [CrossRef] [PubMed]
- Almeida-Naranjo, C.E.; Frutos, M.; Tejedor, J.; Cuestas, J.; Valenzuela, F.; Rivadeneira, M.I.; Villamar, C.A.; Guerrero, V.H. Caffeine adsorptive performance and compatibility characteristics (Eisenia foetida Savigny) of agro-industrial residues potentially suitable for vermifilter beds. Sci. Total Environ. 2021, 801, 149666. [Google Scholar] [CrossRef]
- Ghauch, A.; Tuqan, A.M.; Kibbi, N. Ibuprofen removal by heated persulfate in aqueous solution: A kinetics study. Chem. Eng. J. 2012, 197, 483–492. [Google Scholar] [CrossRef]
- Deng, Y.; Ok, Y.S.; Mohan, D.; Pittman, C.U.; Dou, X. Carbamazepine removal from water by carbon dot-modified magnetic carbon nanotubes. Environ. Res. 2018, 169, 434–444. [Google Scholar] [CrossRef]
- Jiang, N.; Erdős, M.; Moultos, O.A.; Shang, R.; Vlugt, T.J.H.; Heijman, S.G.J.; Rietveld, L.C. The adsorption mechanisms of organic micropollutants on high-silica zeolites causing S-shaped adsorption isotherms: An experimental and Monte Carlo simulation study. Chem. Eng. J. 2020, 389, 123968. [Google Scholar] [CrossRef]
- Boudrahem, N.; Delpeux-ouldriane, S.; Khenniche, L. Single and mixture adsorption of clofibric acid, tetracycline and paracetamol onto Activated carbon developed from cotton cloth residue. Process Saf. Environ. Prot. 2017, 111, 544–559. [Google Scholar] [CrossRef]
- Duan, W.; Wang, N.; Xiao, W.; Zhao, Y.; Zheng, Y. Ciprofloxacin adsorption onto different micro-structured tourmaline, halloysite and biotite. J. Mol. Liq. 2018, 269, 874–881. [Google Scholar] [CrossRef]
- Peng, J.; Wang, X.; Yin, F.; Xu, G. Characterizing the removal routes of seven pharmaceuticals in the activated sludge process. Sci. Total Environ. 2019, 650, 2437–2445. [Google Scholar] [CrossRef]
- Kim, H.; Hwang, Y.S.; Sharma, V.K. Adsorption of antibiotics and iopromide onto single-walled and multi-walled carbon nanotubes. Chem. Eng. J. 2014, 255, 23–27. [Google Scholar] [CrossRef]
- Akhtar, J.; Aishah, N.; Amin, S.; Shahzad, K. A review on removal of pharmaceuticals from water by adsorption. Desalin. Water Treat. 2015, 57, 12842–12860. [Google Scholar] [CrossRef]
- Lozano-Morales, V.; Gardi, I.; Nir, S.; Undabeytia, T. Removal of pharmaceuticals from water by clay-cationic starch sorbents. J. Clean. Prod. 2018, 190, 703–711. [Google Scholar] [CrossRef]
- Käfferlein, H.U.; Göen, T.; Angerer, J. Musk xylene: Analysis, occurrence, kinetics, and toxicology. Crit. Rev. Toxicol. 1998, 28, 431–476. [Google Scholar] [CrossRef] [PubMed]
- Meffe, R.; Bustamante, I. De Science of the Total Environment Emerging organic contaminants in surface water and groundwater: A fi rst overview of the situation in Italy. Sci. Total Environ. 2014, 481, 280–295. [Google Scholar] [CrossRef]
- Shoiful, A.; Ueda, Y.; Nugroho, R.; Honda, K. Degradation of organochlorine pesticides (OCPs) in water by iron (Fe)-based materials. J. Water Process Eng. 2016, 11, 110–117. [Google Scholar] [CrossRef]
- Man, Y.B.; Chow, K.L.; Cheng, Z.; Kang, Y.; Wong, M.H. Profiles and removal efficiency of organochlorine pesticides with emphasis on DDTs and HCHs by two different sewage treatment works. Environ. Technol. Innov. 2018, 9, 220–231. [Google Scholar] [CrossRef]
- Honorio, J.F.; Veit, M.T.; Regina, C.; Tavares, G. Alternative adsorbents applied to the removal of natural hormones from pig farming effluents and characterization of the biofertilizer. Environ. Sci. Pollut. Res. 2018, 26, 28429–28435. [Google Scholar] [CrossRef]
- Akhbarizadeh, R.; Dobaradaran, S.; Schmidt, T.C.; Nabipour, I.; Spitz, J. Worldwide bottled water angmuire of emerging contaminants: A review of the recent scientific literature. J. Hazard. Mater. 2020, 392, 122271. [Google Scholar] [CrossRef]
- Grassi, M.; Kaykioglu, G.; Belgiorno, V.; Lofrano, G. Removal of Emerging Contaminants from Water and Wastewater by Adsorption Process. In Emerging Compounds Removal from Wastewater: Natural and Solar Based Treatments; Springer: Berlin/Heidelberg, Germany, 2012; pp. 15–37. [Google Scholar]
- Sophia A., C.; Lima, E.C. Removal of emerging contaminants from the environment by adsorption. Ecotoxicol. Environ. Saf. 2018, 150, 1–17. [Google Scholar] [CrossRef] [PubMed]
- Pallarés, J.; González-Cencerrado, A.; Arauzo, I. Production and characterization of activated carbon from barley straw by physical activation with carbon dioxide and steam. Biomass Bioenergy 2018, 115, 64–73. [Google Scholar] [CrossRef]
- Forouzesh, M.; Ebadi, A.; Aghaeinejad-meybodi, A. Separation and Puri fi cation Technology Degradation of metronidazole antibiotic in aqueous medium using activated carbon as a persulfate activator. Sep. Purif. Technol. 2019, 210, 145–151. [Google Scholar] [CrossRef]
- García-Mateos, F.J.; Ruiz-Rosas, R.; Marqués, M.D.; Cotoruelo, L.M.; Rodríguez-Mirasol, J.; Cordero, T. Removal of paracetamol on biomass-derived activated carbon: Modeling the fixed bed breakthrough curves using batch adsorption experiments. Chem. Eng. J. 2015, 279, 18–30. [Google Scholar] [CrossRef]
- Raupp, Í.N.; Filho, A.V.; Arim, A.L.; Muniz, A.R.C.; da Rosa, G.S. Development and characterization of activated carbon from olive pomace: Experimental design, kinetic and equilibrium studies in nimesulide adsorption. Materials 2021, 14, 6820. [Google Scholar] [CrossRef]
- Lin, S.H.; Juang, R.S. Adsorption of phenol and its derivatives from water using synthetic resins and low-cost natural adsorbents: A review. J. Environ. Manag. 2009, 90, 1336–1349. [Google Scholar] [CrossRef]
- Jiang, N.; Shang, R.; Heijman, S.G.J.; Rietveld, L.C. High-silica zeolites for adsorption of organic micro-pollutants in water treatment: A review. Water Res. 2018, 144, 145–161. [Google Scholar] [CrossRef]
- Lye, J.W.P.; Saman, N.; Sharuddin, S.S.N.; Othman, N.S.; Mohtar, S.S.; Noor, A.M.M.; Buhari, J.; Cheu, S.C.; Kong, H.; Mat, H. Removal performance of tetracycline and oxytetracycline from aqueous solution via natural zeolites: An equilibrium and kinetic study. Clean-Soil Air Water 2017, 45, 1600260. [Google Scholar] [CrossRef]
- Rasamimanana, S.; Mignard, S.; Batonneau-Gener, I. Hierarchical zeolites as adsorbents for mesosulfuron-methyl removal in aqueous phase. Microporous Mesoporous Mater. 2016, 226, 153–161. [Google Scholar] [CrossRef]
- Sun, K.; Shi, Y.; Wang, X.; Li, Z. Sorption and retention of diclofenac on zeolite in the presence of cationic surfactant. J. Hazard. Mater. 2017, 323, 584–592. [Google Scholar] [CrossRef]
- Pukcothanung, Y.; Siritanon, T.; Rangsriwatananon, K. The efficiency of zeolite Y and surfactant-modified zeolite Y for removal of 2,4-dichlorophenoxyacetic acid and 1,1″-dimethyl-4,4″-bipyridinium ion. Microporous Mesoporous Mater. 2018, 258, 131–140. [Google Scholar] [CrossRef]
- de Sousa, D.N.R.; Insa, S.; Mozeto, A.A.; Petrovic, M.; Chaves, T.F.; Fadini, P.S. Equilibrium and kinetic studies of the adsorption of antibiotics from aqueous solutions onto powdered zeolites. Chemosphere 2018, 205, 137–146. [Google Scholar] [CrossRef]
- Bouras, O.; Bollinger, J.C.; Baudu, M.; Khalaf, H. Adsorption of diuron and its degradation products from aqueous solution by surfactant-modified pillared clays. Appl. Clay Sci. 2007, 37, 240–250. [Google Scholar] [CrossRef]
- Nassar, M.M.; Farrag, T.E.; Ahmed, M.H. Removal of an insecticide (methomyl) from aqueous solutions using natural clay. Alexandria Eng. J. 2012, 51, 11–18. [Google Scholar] [CrossRef]
- Srinivasan, R. Advances in application of natural clay and its composites in removal of biological, organic, and inorganic contaminants from drinking water. Adv. Mater. Sci. Eng. 2011, 2011, 872531. [Google Scholar] [CrossRef]
- Thiebault, T. Raw and modified clays and clay minerals for the removal of pharmaceutical products from aqueous solutions: State of the art and future perspectives. Crit. Rev. Environ. Sci. Technol. 2019, 50, 1451–1514. [Google Scholar] [CrossRef]
- Thiebault, T.; Guégan, R.; Boussafir, M. Adsorption mechanisms of emerging micro-pollutants with a clay mineral: Case of tramadol and doxepine pharmaceutical products. J. Colloid Interface Sci. 2015, 453, 1–8. [Google Scholar] [CrossRef] [PubMed]
- León, H.; Almeida-Naranjo, C.; Aldás, M.B.; Guerrero, V.H. Methomyl removal from synthetic water using natural and modified bentonite clays. IOP Conf. Ser. Earth Environ. Sci. 2021, 776, 012002. [Google Scholar] [CrossRef]
- Zaghouane-Boudiaf, H.; Boutahala, M. Preparation and characterization of organo-montmorillonites. Application in adsorption of the 2,4,5-trichlorophenol from aqueous solution. Adv. Powder Technol. 2011, 22, 735–740. [Google Scholar] [CrossRef]
- Torrellas, S.Á.; Lovera, R.G.; Escalona, N.; Sepúlveda, C.; Sotelo, J.L.; García, J. Chemical-activated carbons from peach stones for the adsorption of emerging contaminants in aqueous solutions. Chem. Eng. J. 2015, 279, 788–798. [Google Scholar] [CrossRef]
- Reeve, P.J.; Fallow, H.J. Natural and surfactant modified zeolites: A review of their applications for water remediation with a focus on surfactant desorption and toxicity towards microorganisms. J. Environ. Manag. 2018, 205, 253–261. [Google Scholar] [CrossRef]
- Yu, F.; Li, Y.; Han, S.; Ma, J. Adsorptive removal of antibiotics from aqueous solution using carbon materials. Chemosphere 2016, 153, 365–385. [Google Scholar] [CrossRef]
- Álvarez-Torrellas, S.; Rodríguez, A.; Ovejero, G.; Gómez, J.M.; García, J. Removal of caffeine from pharmaceutical wastewater by adsorption: Influence of NOM, textural and chemical properties of the adsorbent. Env. Tech. J. 2016, 37(13), 1618–1630. [Google Scholar] [CrossRef]
- Mubarik, S.; Saeed, A.; Mehmood, Z.; Iqbal, M. Phenol adsorption by charred sawdust of sheesham (Indian rosewood; Dalbergia sissoo) from single, binary and ternary contaminated solutions. J. Taiwan Inst. Chem. Eng. 2012, 43, 926–933. [Google Scholar] [CrossRef]
- Benjedim, S.; Romero-cano, L.A.; Pérez-cadenas, A.F.; Bautista-toledo, M.I.; Lotfi, E.M. Removal of emerging pollutants present in water using an E-coli biofilm supported onto activated carbons prepared from argan wastes: Adsorption studies in batch and fixed bed. Sci. Total Environ. 2020, 720, 137491. [Google Scholar] [CrossRef]
- Yanan, C.; Srour, Z.; Ali, J.; Guo, S.; Taamalli, S.; Fèvre-Nollet, V.; da Boit Martinello, K.; Georgin, J.; Franco, D.S.P.; Silva, L.F.O.; et al. Adsorption of paracetamol and ketoprofenon activated charcoal prepared from the residue of the fruit of Butiacapitate: Experiments and theoretical interpretations. Chem. Eng. J. 2023, 454, 139943. [Google Scholar] [CrossRef]
- Thiebault, T.; Boussafir, M. Adsorption Mechanisms of Psychoactive Drugs onto Montmorillonite. Colloid Interface Sci. Commun. 2019, 30, 100183. [Google Scholar] [CrossRef]
- Kuila, U.; Prasad, M. Specific surface area and pore-size distribution in clays and shales. Geophys. Prospect. 2013, 61, 341–362. [Google Scholar] [CrossRef]
- Islam, M.A.; Morton, D.W.; Johnson, B.B.; Mainali, B.; Angove, M.J. Manganese oxides and their application to metal ion and contaminant removal from wastewater. J. Water Process Eng. 2018, 26, 264–280. [Google Scholar] [CrossRef]
- Kasprzyk-Hordern, B. Chemistry of alumina, reactions in aqueous solution and its application in water treatment. Adv. Colloid Interface Sci. 2004, 110, 19–48. [Google Scholar] [CrossRef]
- Turku, I.; Sainio, T.; Paatero, E. Thermodynamics of tetracycline adsorption on silica. Environ. Chem. Lett. 2007, 5, 225–228. [Google Scholar] [CrossRef]
- Nguyen, N.T.; Dao, T.H.; Truong, T.T.; Minh, T.; Nguyen, T.; Pham, T.D. Adsorption characteristic of ciprofloxacin antibiotic onto synthesized alpha alumina nanoparticles with surface modification by polyanion. J. Mol. Liq. 2020, 309, 113150. [Google Scholar] [CrossRef]
- Aazza, M.; Ahla, H.; Moussout, H.; Maghat, H. Adsorption of metha-nitrophenol onto alumina and HDTMA modified alumina: Kinetic, isotherm and mechanism investigations. J. Mol. Liq. 2018, 268, 587–597. [Google Scholar] [CrossRef]
- Aazza, M.; Ahlafi, H.; Moussout, H.; Maghat, H. Ortho-Nitro-Phenol adsorption onto alumina and surfactant modified alumina: Kinetic, isotherm and mechanism. J. Environ. Chem. Eng. 2017, 5, 3418–3428. [Google Scholar] [CrossRef]
- Khan, H.; Gul, K.; Ara, B.; Khan, A.; Ali, N.; Ali, N.; Bilal, M. Adsorptive removal of acrylic acid from the aqueous environment using raw and chemically modified alumina: Batch adsorption, kinetic, equilibrium and thermodynamic studies. J. Environ. Chem. Eng. 2020, 8, 103927. [Google Scholar] [CrossRef]
- Doan, T.H.Y.; Le, T.T.; Nguyen, T.M.T.; Chu, T.H.; Pham, T.N.M.; Nguyen, T.A.H.; Pham, T.D. Simultaneous adsorption of anionic alkyl sulfate surfactants onto alpha alumina particles: Experimental consideration and modeling. Env. Tech. & Innovation. 2021, 24, 101920. [Google Scholar] [CrossRef]
- Bui, T.X.; Choi, H. Adsorptive removal of selected pharmaceuticals by mesoporous silica SBA-15. J. Hazard. Mater. 2009, 168, 602–608. [Google Scholar] [CrossRef]
- Chen, W.; Li, X.; Pan, Z.; Bao, Y.; Ma, S.; Li, L. Efficient adsorption of Norfloxacin by Fe-MCM-41 molecular sieves: Kinetic, isotherm and thermodynamic studies. Chem. Eng. J. 2015, 281, 397–403. [Google Scholar] [CrossRef]
- Tran, T.H. Adsorption and transformation of the anthelmintic drug niclosamide by manganese oxide. Chemosphere 2018, 201, 425–431. [Google Scholar] [CrossRef]
- Remucal, C.K.; Ginder-Vogel, M. A critical review of the reactivity of manganese oxides with organic contaminants. Environ. Sci. Process. Impacts 2014, 16, 1247–1266. [Google Scholar] [CrossRef]
- Feitosa-Felizzola, J.; Hanna, K.; Chiron, S.; Poincare, H. Adsorption and transformation of selected human-used macrolide antibacterial agents with iron (III) and manganese (IV) oxides. Environ. Pollut. 2009, 157, 1317–1322. [Google Scholar] [CrossRef]
- Liu, W.; Langenhoff, A.A.M.; Sutton, N.B.; Rijnaarts, H.H.M. Application of manganese oxides under anoxic conditions to remove diclofenac from water. J. Environ. Chem. Eng. 2018, 6, 5061–5068. [Google Scholar] [CrossRef]
- Zhao, F.; Li, X.; Graham, N. Treatment of a model HA compound (resorcinol) by potassium manganate. Sep. Purif. Technol. 2012, 91, 52–58. [Google Scholar] [CrossRef]
- Zhang, L.; Ma, J.; Yu, M. The microtopography of manganese dioxide formed in situ and its adsorptive properties for organic micropollutants. Solid State Sci. 2008, 10, 148–153. [Google Scholar] [CrossRef]
- Diagboya, P.N.E.; Dikio, E.D. Silica-based mesoporous materials; emerging designer adsorbents for aqueous pollutants removal and water treatment. Microporous Mesoporous Mater. 2018, 266, 252–267. [Google Scholar] [CrossRef]
- Gupta, V.K. Suhas Application of low-cost adsorbents for dye removal—A review. J. Environ. Manag. 2009, 90, 2313–2342. [Google Scholar] [CrossRef] [PubMed]
- Dao, T.H.; Vu, T.Q.M.; Nguyen, N.T.; Pham, T.T.; Nguyen, T.L.; Yusa, S.I.; Pham, T.D. Adsorption characteristics of synthesized polyelectrolytes onto alumina nanoparticles and their application in antibiotic removal. Langmuir 2020, 36, 13001–13011. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Zhuang, S.; Wang, J. Removal of various pollutants from water and wastewater by modified chitosan adsorbents. Crit. Rev. Environ. Sci. Technol. 2018, 47, 2331–2386. [Google Scholar] [CrossRef]
- Ali, I.; Asim, M.; Khan, T.A. Low cost adsorbents for the removal of organic pollutants from wastewater. J. Environ. Manag. 2012, 113, 170–183. [Google Scholar] [CrossRef]
- Lessa, E.F.; Nunes, M.L.; Fajardo, A.R. Chitosan/waste coffee-grounds composite: An efficient and eco-friendly adsorbent for removal of pharmaceutical contaminants from water. Carbohydr. Polym. 2018, 189, 257–266. [Google Scholar] [CrossRef]
- Zhou, L.-C.; Meng, X.; Fu, J.; Yang, Y. Highly efficient adsorption of chlorophenols onto chemically modified chitosan. Appl. Surf. Sci. 2014, 292, 735–741. [Google Scholar] [CrossRef]
- Tarasi, R.; Alipour, M.; Gorgannezhad, L.; Imanparast, S.; Yousefi-Ahmadipour, A.; Ramezani, A.; Ganjali, M.R.; Shafiee, A.; Faramarzi, M.A.; Khoobi, M. Laccase Immobilization onto Magnetic β-Cyclodextrin-Modified Chitosan: Improved Enzyme Stability and Efficient Performance for Phenolic Compounds Elimination. Macromol. Res. 2018, 26, 755–762. [Google Scholar] [CrossRef]
- Kyzas, G.Z.; Kostoglou, M.; Lazaridis, N.K.; Lambropoulou, D.A.; Bikiaris, D.N. Environmental friendly technology for the removal of pharmaceutical contaminants from wastewaters using modified chitosan adsorbents. Chem. Eng. J. 2013, 222, 248–258. [Google Scholar] [CrossRef]
- Kyzas, G.Z.; Bikiaris, D.N.; Lambropoulou, D.A. Effect of humic acid on pharmaceuticals adsorption using sulfonic acid grafted chitosan. J. Mol. Liq. 2017, 230, 1–5. [Google Scholar] [CrossRef]
- Zhou, A.; Chen, W.; Liao, L.; Xie, P.; Zhang, T.C.; Wu, X.; Feng, X. Comparative adsorption of emerging contaminants in water by functional designed magnetic poly (N-isopropylacrylamide)/chitosan hydrogels. Sci. Total Environ. 2019, 671, 377–387. [Google Scholar] [CrossRef]
- Nie, Y.; Deng, S.; Wang, B.; Huang, J.; Yu, G. Removal of clofibric acid from aqueous solution by polyethylenimine-modified chitosan beads. Front. Environ. Sci. Eng. 2014, 8, 675–682. [Google Scholar] [CrossRef]
- Zhou, Y.; Chen, L.; Lu, P.; Tang, X.; Lu, J. Removal of bisphenol A from aqueous solution using modified fibric peat as a novel biosorbent. Sep. Purif. Technol. 2011, 81, 184–190. [Google Scholar] [CrossRef]
- Zhou, Y.; Lu, P.; Lu, J. Application of natural biosorbent and modified peat for bisphenol a removal from aqueous solutions. Carbohydr. Polym. 2012, 88, 502–508. [Google Scholar] [CrossRef]
- Chen, K.; Liu, L.; Chen, W. Adsorption of sulfamethoxazole and sulfapyridine antibiotics in high organic content soils. Environ. Pollut. 2017, 231, 1163–1171. [Google Scholar] [CrossRef]
- Mo, J.; Yang, Q.; Zhang, N.; Zhang, W.; Zheng, Y.; Zhang, Z. A review on agro-industrial waste (AIW) derived adsorbents for water and wastewater treatment. J. Environ. Manag. 2018, 227, 395–405. [Google Scholar] [CrossRef]
- FAO. Setting up Fuel Supply Strategies for Large-Scale Bio-Energy Projects Using Agricultural and Forest Residues; Junginger, M., Ed.; FAO: Bangkok, Thailand, 2000; ISBN 907395858X. [Google Scholar]
- Ngigi, A.N.; Ok, Y.S.; Thiele-Bruhn, S. Biochar-mediated sorption of antibiotics in pig manure. J. Hazard. Mater. 2018, 364, 663–670. [Google Scholar] [CrossRef] [PubMed]
- Bhatnagar, A.; Sillanpää, M.; Witek-Krowiak, A. Agricultural waste peels as versatile biomass for water purification—A review. Chem. Eng. J. 2015, 270, 244–271. [Google Scholar] [CrossRef]
- Mallek, M.; Chtourou, M.; Portillo, M.; Monclús, H.; Walha, K.; Salvadó, V. Granulated cork as biosorbent for the removal of phenol derivatives and emerging contaminants. J. Environ. Manag. 2018, 223, 576–585. [Google Scholar] [CrossRef] [PubMed]
- Ofomaja, A.E. Kinetics and pseudo-isotherm studies of 4-nitrophenol adsorption onto mansonia wood sawdust. Ind. Crops Prod. 2011, 33, 418–428. [Google Scholar] [CrossRef]
- Larous, S.; Meniai, A.-H. The use of sawdust as by product adsorbent of organic pollutant from wastewater: Adsorption of phenol. Energy Procedia 2012, 18, 905–914. [Google Scholar] [CrossRef]
- Thompson, K.A.; Shimabuku, K.K.; Kearns, J.P.; Knappe, D.R.U.; Summers, R.S.; Cook, S.M.; Cook, S.M. Environmental Comparison of Biochar and Activated Carbon for Tertiary Wastewater Treatment. Environ. Sci. Technol. 2016, 50, 11253–11262. [Google Scholar] [CrossRef]
- Rangabhashiyam, S.; Balasubramanian, P. Industrial Crops & Products The potential of lignocellulosic biomass precursors for biochar production: Performance, mechanism and wastewater application—A review. Ind. Crops Prod. 2019, 128, 405–423. [Google Scholar] [CrossRef]
- Li, L.; Zou, D.; Xiao, Z.; Zeng, X.; Zhang, L.; Jiang, L.; Wang, A.; Ge, D.; Zhang, G.; Liu, F. Biochar as a sorbent for emerging contaminants enables improvements in waste management and sustainable resource use. J. Clean. Prod. 2018, 210, 1324–1342. [Google Scholar] [CrossRef]
- Bedia, J.; Peñas-garz, M.; Almudena, G.; Rodriguez, J.J. A Review on the Synthesis and Characterization of Biomass-Derived Carbons for Adsorption of Emerging Contaminants from Water. J. Carbon Res. 2018, 4, 63. [Google Scholar] [CrossRef]
- Choi, Y.K.; Choi, T.R.; Gurav, R.; Bhatia, S.K.; Park, Y.L.; Kim, H.J.; Kan, E.; Yang, Y.H. Adsorption behavior of tetracycline onto Spirulina sp. (microalgae)-derived biochars produced at different temperatures. Sci. Total Environ. 2020, 710, 136282. [Google Scholar] [CrossRef]
- Jing, X.R.; Wang, Y.Y.; Liu, W.J.; Wang, Y.K.; Jiang, H. Enhanced adsorption performance of tetracycline in aqueous solutions by methanol-modified biochar. Chem. Eng. J. 2014, 248, 168–174. [Google Scholar] [CrossRef]
- Kim, E.; Jung, C.; Han, J.; Her, N.; Park, C.; Jang, M.; Son, A.; Yoon, Y. Sorptive removal of selected emerging contaminants using biochar in aqueous solution. J. Ind. Eng. Chem. 2016, 36, 364–371. [Google Scholar] [CrossRef]
- Devi, P.; Saroha, A.K. Utilization of sludge based adsorbents for the removal of various pollutants: A review. Sci. Total Environ. 2016, 578, 16–33. [Google Scholar] [CrossRef]
- Tran, H.N.; Nguyen, H.C.; Woo, S.H.; Nguyen, T.V.; Vigneswaran, S.; Hosseini-Bandegharaei, A.; Rinklebe, J.; Kumar Sarmah, A.; Ivanets, A.; Dotto, G.L.; et al. Removal of various contaminants from water by renewable lignocellulose-derived biosorbents: A comprehensive and critical review. Crit. Rev. Environ. Sci. Technol. 2019, 49, 2155–2219. [Google Scholar] [CrossRef]
- Yang, Q.; Wu, P.; Liu, J.; Rehman, S.; Ahmed, Z.; Ruan, B. Batch interaction of emerging tetracycline contaminant with novel phosphoric acid activated corn straw porous carbon: Adsorption rate and nature of mechanism. Environ. Res. 2019, 181, 108899. [Google Scholar] [CrossRef]
- Nielsen, L.; Bandosz, T.J. Analysis of sulfamethoxazole and trimethoprim adsorption on sewage sludge and fi sh waste derived adsorbents. Microporous Mesoporous Mater. 2016, 220, 58–72. [Google Scholar] [CrossRef]
- Nielsen, L.; Bandosz, T.J. Analysis of the competitive adsorption of pharmaceuticals on waste derived materials. Chem. Eng. J. 2015, 287, 139–147. [Google Scholar] [CrossRef]
- Gupta, V.K.; Ali, I.; Saini, V.K. Adsorption of 2,4-D and carbofuran pesticides using fertilizer and steel industry wastes. J. Colloid Interface Sci. 2006, 299, 556–563. [Google Scholar] [CrossRef]
- Filho, A.V.; Tholozan, L.V.; Arim, A.L.; de Almeida, A.R.F.; da Rosa, G.S. High-performance removal of anti-inflammatory using activated carbon from water treatment plant sludge: Fixed-bed and batch studies. Int. J. Environ. Sci. Technol. 2023, 20, 36333644. [Google Scholar] [CrossRef]
- Sharipova, A.A.; Aidarova, S.B.; Bekturganova, N.E.; Tleuova, A.; Schenderlein, M.; Lygina, O.; Lyubchik, S.; Miller, R. Triclosan as model system for the adsorption on recycled adsorbent materials. Colloids Surfaces A Physicochem. Eng. Asp. 2016, 505, 193–196. [Google Scholar] [CrossRef]
- Alsbaiee, A.; Smith, B.J.; Xiao, L.; Ling, Y.; Helbling, D.E.; Dichtel, W.R. Rapid removal of organic micropollutants from water by a porous β-cyclodextrin polymer. Nature 2016, 529, 190–194. [Google Scholar] [CrossRef]
- Pan, B.; Pan, B.; Zhang, W.; Lv, L.; Zhang, Q.; Zheng, S. Development of polymeric and polymer-based hybrid adsorbents for pollutants removal from waters. Chem. Eng. J. 2009, 151, 19–29. [Google Scholar] [CrossRef]
- Li, X.; Zhou, M.; Jia, J.; Ma, J.; Jia, Q. Design of a hyper-crosslinked β-cyclodextrin porous polymer for highly efficient removal toward bisphenol a from water. Sep. Purif. Technol. 2018, 195, 130–137. [Google Scholar] [CrossRef]
- Li, H.; Fu, Z.; Yan, C.; Huang, J.; Liu, Y.N.; Kirin, S.I. Hydrophobic–hydrophilic post-cross-linked polystyrene/poly (methyl acryloyl diethylenetriamine) interpenetrating polymer networks and its adsorption properties. J. Colloid Interface Sci. 2016, 463, 61–68. [Google Scholar] [CrossRef] [PubMed]
- Tejedor, J.; Cóndor, V.; Almeida-Naranjo, C.E.; Guerrero, V.H.; Villamar, C.A. Performance of wood chips/peanut shells biofilters used to remove organic matter from domestic wastewater. Sci. Total Environ. 2020, 738, 139589. [Google Scholar] [CrossRef]
- Ahmed, M.B.; Zhou, J.L.; Ngo, H.H.; Guo, W.; Chen, M. Progress in the preparation and application of modified biochar for improved contaminant removal from water and wastewater. Bioresour. Technol. 2016, 214, 836–851. [Google Scholar] [CrossRef] [PubMed]
- Zhai, X.; Ren, Y.; Wang, N.; Guan, F.; Agievich, M.; Duan, J.; Hou, B. Microbial Corrosion Resistance and Antibacterial Property of Electrodeposited Zn–Ni–Chitosan Coatings. Molecules 2019, 24, 1974. [Google Scholar] [CrossRef]
- Anastopoulos, I.; Pashalidis, I.; Orfanos, A.G.; Manariotis, I.D.; Tatarchuk, T.; Sellaoui, L.; Bonilla-Petriciolet, A.; Mittal, A.; Núñez-Delgado, A. Removal of caffeine, nicotine and amoxicillin from (waste) waters by various adsorbents. A review. J. Environ. Manag. 2020, 261, 110236. [Google Scholar] [CrossRef]
- Silva, C.P.; Jaria, G.; Otero, M.; Esteves, V.I.; Calisto, V. Waste-based alternative adsorbents for the remediation of pharmaceutical contaminated waters: Has a step forward already been taken? Bioresour. Technol. 2017, 250, 888–901. [Google Scholar] [CrossRef]
- Bhatnagar, A. Removal of bromophenols from water using industrial wastes as low cost adsorbents. J. Hazard. Mater. 2007, 139, 93–102. [Google Scholar] [CrossRef]
- Oliveira, G.; Calisto, V.; Santos, S.M.; Otero, M.; Esteves, V.I. Paper pulp-based adsorbents for the removal of pharmaceuticals from wastewater: A novel approach towards diversification. Sci. Total Environ. 2018, 631–632, 1018–1028. [Google Scholar] [CrossRef]
- Samah, N.A.; Sánchez-Martín, M.J.; Sebastián, R.M.; Valiente, M.; López-Mesas, M. Molecularly imprinted polymer for the removal of diclofenac from water: Synthesis and characterization. Sci. Total Environ. 2018, 631–632, 1534–1543. [Google Scholar] [CrossRef] [PubMed]
- Zhao, L.; Deng, J.; Sun, P.; Liu, J.; Ji, Y.; Nakada, N.; Qiao, Z.; Tanaka, H.; Yang, Y. Nanomaterials for treating emerging contaminants in water by adsorption and photocatalysis: Systematic review and bibliometric analysis. Sci. Total Environ. 2018, 627, 1253–1263. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Wu, B.; Xu, H.; Liu, H.; Wang, M.; He, Y.; Pan, B. Nanomaterials-enabled water and wastewater treatment. NanoImpact 2016, 3–4, 22–39. [Google Scholar] [CrossRef]
- Almeida-Naranjo, C.E.; Aldás, M.B.; Cabrera, G.; Guerrero, V.H. Caffeine removal from synthetic wastewater using peel composites: Material characterization, isotherm and kinetic studies. Environ. Challenges 2021, 5, 100343. [Google Scholar] [CrossRef]
- Pham, T.D.; Vu, T.N.; Nguyen, H.L.; Le, P.H.P.; Hoang, T.S. Adsorptive removal of antibiotic ciprofloxacin from aqueous solution using protein-modified nanosilica. Polymers 2020, 12, 57. [Google Scholar] [CrossRef]
- Yu, F.; Ma, J.; Han, S. Adsorption of tetracycline from aqueous solutions onto multi-walled carbon nanotubes with different oxygen contents. Sci. Rep. 2014, 4, 5326. [Google Scholar] [CrossRef]
- Gupta, V.K.; Kumar, R.; Nayak, A.; Saleh, T.A.; Barakart, M.A. Adsorptive removal of dyes from aqueous solution onto carbon nanotubes: A review. Adv. Colloid Interface Sci. 2013, 193–194, 24–34. [Google Scholar] [CrossRef] [PubMed]
- Joseph, L.; Heo, J.; Park, Y.; Flora, J.R.V.; Yoon, Y. Adsorption of bisphenol A and 17 α-ethinyl estradiol on single walled carbon nanotubes from seawater and brackish water. Desalination 2011, 281, 68–74. [Google Scholar] [CrossRef]
- Hu, J.; Tong, Z.; Hu, Z.; Chen, G.; Chen, T. Adsorption of roxarsone from aqueous solution by multi-walled carbon nanotubes. J. Colloid Interface Sci. 2012, 377, 355–361. [Google Scholar] [CrossRef]
- Patiño, Y.; Díaz, E.; Ordóñez, S.; Gallegos-Suarez, E.; Guerrero-Ruiz, A.; Rodríguez-Ramos, I. Adsorption of emerging pollutants on functionalized multiwall carbon nanotubes. Chemosphere 2015, 136, 174–180. [Google Scholar] [CrossRef]
- Hu, X.; Cheng, Z. Removal of diclofenac from aqueous solution with multi-walled carbon nanotubes modified by nitric acid. Chinese J. Chem. Eng. 2015, 23, 1551–1556. [Google Scholar] [CrossRef]
- Hiew, B.Y.Z.; Lee, L.Y.; Lee, X.J.; Gan, S.; Thangalazhy-Gopakumar, S.; Lim, S.S.; Pan, G.T.; Yang, T.C.K. Adsorptive removal of diclofenac by graphene oxide: Optimization, equilibrium, kinetic and thermodynamic studies. J. Taiwan Inst. Chem. Eng. 2018, 98, 150–162. [Google Scholar] [CrossRef]
- Li, M.; Liu, Y.; Zeng, G.; Liu, N.; Liu, S. Graphene and graphene-based nanocomposites used for antibiotics removal in water treatment: A review. Chemosphere 2019, 226, 360–380. [Google Scholar] [CrossRef]
- Naseem, T.; Waseem, M. A comprehensive review on the role of some important nanocomposites for antimicrobial and wastewater applications. Int. J. Environ. Sci. Technol. 2021, 19, 2221–2246. [Google Scholar] [CrossRef]
- Baig, N.; Sajid, M.; Saleh, A. Graphene-based adsorbents for the removal of toxic organic pollutants: A review. J. Environ. Manag. 2019, 244, 370–382. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Yin, R.; Zeng, L.; Zhu, M. A review of graphene-based nanomaterials for removal of antibiotics from aqueous environments. Environ. Pollut. 2019, 253, 100–110. [Google Scholar] [CrossRef]
- Borthakur, P.; Boruah, P.K.; Das, M.R.; Kulik, N.; Minofar, B. Adsorption of 17α-ethynyl estradiol and β-estradiol on graphene oxide surface: An experimental and computational study. J. Mol. Liq. 2018, 269, 160–168. [Google Scholar] [CrossRef]
- Moussavi, G.; Hossaini, Z.; Pourakbar, M. High-rate adsorption of acetaminophen from the contaminated water onto double-oxidized graphene oxide. Chem. Eng. J. 2016, 287, 665–673. [Google Scholar] [CrossRef]
- Balasubramani, K.; Sivarajasekar, N.; Naushad, M. Effective adsorption of antidiabetic pharmaceutical (metformin) from aqueous medium using graphene oxide nanoparticles: Equilibrium and statistical modelling. J. Mol. Liq. 2020, 301, 112426. [Google Scholar] [CrossRef]
- Liu, S.; Tang, W.; Yang, Y. Adsorption of nicotine in aqueous solution by a defective graphene oxide. Sci. Total Environ. 2018, 643, 507–515. [Google Scholar] [CrossRef]
- Kunduru, K.R.; Nazarkovsky, M.; Farah, S.; Pawar, R.P.; Basu, A.; Domb, A.J. Nanotechnology for water purification: Applications of nanotechnology methods in wastewater treatment. In Water Purification; Elsevier Inc.: Amsterdam, The Netherlands, 2017; pp. 33–74. ISBN 9780128043004. [Google Scholar]
- Hamdy, A.; Mostafa, M.K.; Nasr, M. Zero-valent iron nanoparticles for methylene blue removal from aqueous solutions and textile wastewater treatment, with cost estimation. Water Sci. Technol. 2018, 78, 367–378. [Google Scholar] [CrossRef] [PubMed]
- Kumar, B.; Smita, K.; Cumbal, L.; Debut, A.; Galeas, S.; Guerrero, V.H. Phytosynthesis and photocatalytic activity of magnetite (Fe3O4) nanoparticles using the Andean blackberry leaf. Mater. Chem. Phys. 2016, 179, 310–315. [Google Scholar] [CrossRef]
- Crane, R.A.; Scott, T.B. Nanoscale zero-valent iron: Future prospects for an emerging water treatment technology. J. Hazard. Mater. 2012, 211–212, 112–125. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Blowes, D.W.; Ptacek, C.J.; Groza, L.G. Removal of pharmaceutical compounds, arti fi cial sweeteners, and perfluoroalkyl substances from water using a passive treatment system containing zero-valent iron and biochar. Sci. Total Environ. 2019, 691, 165–177. [Google Scholar] [CrossRef] [PubMed]
- Al-Jabari, M.H.; Sulaiman, S.; Ali, S.; Barakat, R.; Mubarak, A.; Khan, S.A. Adsorption study of levofloxacin on reusable magnetic nanoparticles: Kinetics and antibacterial activity. J. Mol. Liq. 2019, 291, 111249. [Google Scholar] [CrossRef]
- Minh, T.D.; Lee, B.K.; Nguyen-Le, M.T. Methanol-dispersed of ternary Fe3O4@γ-APS/graphene oxide-based nanohybrid for novel removal of benzotriazole from aqueous solution. J. Environ. Manag. 2018, 209, 452–461. [Google Scholar] [CrossRef]
- Rakshit, S.; Sarkar, D.; Punamiya, P.; Datta, R. Kinetics of oxytetracycline sorption on magnetite nanoparticles. Int. J. Environ. Sci. Technol. 2013, 11, 1207–1214. [Google Scholar] [CrossRef]
- Leone, V.O.; Pereira, M.C.; Aquino, S.; Oliveira, L.; Correa, S.; Ramalho, T.C.; Gurgel, L.; da Silva, A.C. Adsorption of diclofenac on a magnetic adsorbent based on maghemite: Experimental and theoretical studies. New J. Chem. 2018, 42, 437–449. [Google Scholar] [CrossRef]
- Zhao, Y.; Liu, F.; Qin, X. Adsorption of diclofenac onto goethite: Adsorption kinetics and effects of pH. Chemosphere 2017, 180, 373–378. [Google Scholar] [CrossRef]
- Qin, X.; Liu, F.; Wang, G.; Weng, L.; Li, L. Adsorption of levofloxacin onto goethite: Effects of pH, calcium and phosphate. Colloids Surfaces B Biointerfaces 2014, 116, 591–596. [Google Scholar] [CrossRef]
- Yin, R.; Sun, J.; Xiang, Y.; Shang, C. Recycling and Reuse of Rusted Iron Particles Containing Core-shell Fe-FeOOH for Ibuprofen Removal: Adsorption and Persulfate-Based Advanced Oxidation. J. Clean. Prod. 2018, 178, 441–448. [Google Scholar] [CrossRef]
- Nassar, M.Y.; Ahmed, I.S.; Hendy, H. A facile one-pot hydrothermal synthesis of hematite (α-Fe2O3) nanostructures and cephalexin antibiotic sorptive removal from polluted aqueous media. J. Mol. Liq. 2018, 271, 844–856. [Google Scholar] [CrossRef]
- Rajendran, K.; Sen, S. Adsorptive removal of carbamazepine using biosynthesized hematite nanoparticle. Environ. Nanotechnol. Monit. Manag. 2018, 9, 122–127. [Google Scholar] [CrossRef]
- Fakhri, A.; Behrouz, S. Comparison studies of adsorption properties of MgO nanoparticles and ZnO-MgO nanocomposites for linezolid antibiotic removal from aqueous solution using response surface methodology. Process Saf. Environ. Prot. 2015, 94, 37–43. [Google Scholar] [CrossRef]
- Sivaselvam, S.; Premasudha, P.; Viswanathan, C.; Ponpandian, N. Enhanced Removal of Emerging Pharmaceutical Contaminant Ciprofloxacin and Pathogen Inactivation Using Morphologically Tuned MgO Nanostructures; Elsevier B.V.: Amsterdam, The Netherlands, 2020; Volume 8, ISBN 9142224284. [Google Scholar]
- Danalıoğlua, S.T.; Bayazit, Ş.S.; Kuyumcu, Ö.K.; Salam, M.A. Efficient removal of antibiotics by a novel magnetic adsorbent: Magnetic activated carbon/chitosan (MACC) nanocomposite. J. Mol. Liq. J. 2017, 240, 589–596. [Google Scholar] [CrossRef]
- Lompe, K.M.; Vo Duy, S.; Peldszus, S.; Sauvé, S.; Barbeau, B. Removal of micropollutants by fresh and colonized magnetic powdered activated carbon. J. Hazard. Mater. 2018, 360, 349–355. [Google Scholar] [CrossRef] [PubMed]
- Wang, B.; Wan, Y.; Zheng, Y.; Lee, X.; Liu, T.; Yu, Z.; Huang, J.; Ok, Y.S.; Chen, J.; Gao, B.; et al. Alginate-based composites for environmental applications: A critical review. Crit. Rev. Environ. Sci. Technol. 2018, 49, 318–356. [Google Scholar] [CrossRef]
- Boruah, P.K.; Sharma, B.; Hussain, N.; Das, M.R. Magnetically recoverable Fe3O4/graphene nanocomposite towards efficient removal of triazine pesticides from aqueous solution: Investigation of the adsorption phenomenon and specific ion effect. Chemosphere 2016, 168, 1058–1067. [Google Scholar] [CrossRef] [PubMed]
- Sauvé, S.; Desrosiers, M. A review of what is an emerging contaminant. Chem. Cent. J. 2014, 8, 15. [Google Scholar] [CrossRef]
- Azhar, M.R.; Abid, H.R.; Periasamy, V.; Sun, H.; Tade, M.O.; Wang, S. Adsorptive removal of antibiotic sulfonamide by UiO-66 and ZIF-67 for wastewater treatment. J. Colloid Interface Sci. 2017, 500, 88–95. [Google Scholar] [CrossRef] [PubMed]
- Alizadeh Fard, M.; Barkdoll, B. Magnetic activated carbon as a sustainable solution for removal of micropollutants from water. Int. J. Environ. Sci. Technol. 2018, 16, 1625–1636. [Google Scholar] [CrossRef]
- Wong, K.T.; Yoon, Y.; Snyder, S.A.; Jang, M. Phenyl-functionalized magnetic palm-based powered activated carbon for the effective removal of selected pharmaceutical and endocrine-disruptive compounds. Chemosphere 2016, 152, 71–80. [Google Scholar] [CrossRef] [PubMed]
- Wang, F.; Yang, B.; Wang, H.; Song, Q.; Tan, F.; Cao, Y. Removal of ciprofloxacin from aqueous solution by a magnetic chitosan grafted graphene oxide composite. J. Mol. Liq. 2016, 222, 188–194. [Google Scholar] [CrossRef]
- Hossein, M.; Mohammadirad, M.; Shemirani, F.; Akbar, A. Magnetic cellulose ionomer/layered double hydroxide: An efficient anion exchange platform with enhanced diclofenac adsorption property. Carbohydr. Polym. 2017, 157, 438–446. [Google Scholar] [CrossRef]
- Ma, Y.; Yang, L.; Wu, L.; Li, P.; Qi, X.; He, L.; Cui, S.; Ding, Y.; Zhang, Z. Carbon nanotube supported sludge biochar as an efficient adsorbent for low concentrations of sulfamethoxazole removal. Sci. Total Environ. 2020, 718, 137299. [Google Scholar] [CrossRef]
- Yuan, F.; Yue, L.; Zhao, H.; Wu, H. Study on the adsorption of polystyrene microplastics by three-dimensional reduced graphene oxide. Water Sci. Technol. 2020, 81, 2163–2175. [Google Scholar] [CrossRef]
- Sun, C.; Wang, Z.; Zheng, H.; Chen, L.; Li, F. Biodegradable and re-usable sponge materials made from chitin for efficient removal of microplastics. J. Hazard. Mater. 2021, 420, 126599. [Google Scholar] [CrossRef]
- Alidadi, H.; Dolatabadi, M.; Davoudi, M.; Askari, F.B.; Farideh, J.-B.; Hosseinzadeh, A. Enhanced Removal of Tetracycline Using Modified Sawdust: Optimization, Isotherm, Kinetics, and Regeneration Studies. Process Saf. Environ. Prot. 2018, 117, 51–60. [Google Scholar] [CrossRef]
- Baghdadi, M.; Ghaffari, E.; Aminzadeh, B. Removal of carbamazepine from municipal wastewater effluent using optimally synthesized magnetic activated carbon: Adsorption and sedimentation kinetic studies. J. Environ. Chem. Eng. 2016, 4, 3309–3321. [Google Scholar] [CrossRef]
- Peng, X.; Hu, F.; Zhang, T.; Qiu, F.; Dai, H. Amine-functionalized magnetic bamboo-based activated carbon adsorptive removal of ciprofloxacin and norfloxacin: A batch and fixed-bed column study. Bioresour. Technol. 2018, 249, 924–934. [Google Scholar] [CrossRef]
- Reguyal, F.; Sarmah, A.K. Site energy distribution analysis and influence of Fe3O4 nanoparticles on sulfamethoxazole sorption in aqueous solution by magnetic pine sawdust biochar. Environ. Pollut. 2018, 233, 510–519. [Google Scholar] [CrossRef]
- Zaheer, M.; Sun, X.; Liu, J.; Song, C.; Wang, S.; Javed, A. Enhancement of ciprofloxacin sorption on chitosan/biochar hydrogel beads. Sci. Total Environ. 2018, 639, 560–569. [Google Scholar] [CrossRef]
- Liu, Y.; Tourbin, M.; Lachaize, S.; Guiraud, P. Nanoparticles in wastewaters: Hazards, fate and remediation. Powder Technol. 2014, 255, 149–156. [Google Scholar] [CrossRef]
- Wanjeri, V.W.O.; Sheppard, C.J.; Prinsloo, A.R.E.; Ngila, J.C.; Ndungu, P.G. Isotherm and kinetic investigations on the adsorption of organophosphorus pesticides on graphene oxide based silica coated magnetic nanoparticles functionalized with 2-phenylethylamine. J. Environ. Chem. Eng. 2018, 6, 1333–1346. [Google Scholar] [CrossRef]
- Catherine, H.N.; Ou, M.-H.; Manu, B.; Shih, Y. Adsorption mechanism of emerging and conventional phenolic compounds on graphene oxide nano flakes in water. Sci. Total Environ. 2018, 635, 629–638. [Google Scholar] [CrossRef]
- Cao, Z.; Liu, X.; Xu, J.; Zhang, J.; Yang, Y.; Zhou, J.; Xu, X.; Lowry, G.V. Removal of Antibiotic Florfenicol by Sulfide-Modified Nanoscale Zero-Valent Iron. Environ. Sci. Technol. 2017, 51, 11269–11277. [Google Scholar] [CrossRef]
- Al-Ahmari, S.D.; Watson, K.; Fong, B.N.; Ruyonga, R.M.; Ali, H. Adsorption Kinetics of 4-n-Nonylphenol on Hematite and Goethite. Environ. Chem. Eng. 2018, 6, 4030–4036. [Google Scholar] [CrossRef]
- Marcelo, L.R.; de Gois, J.S.; Araujo, A.; Vargas, D. Synthesis of iron-based magnetic nanocomposites and applications in adsorption processes for water treatment: A review. Environ. Chem. Lett. 2020, 19, 1229–1274. [Google Scholar] [CrossRef]
- Taha, M.R.; Mobasser, S. Adsorption of DDT and PCB by nanomaterials from residual soil. PLoS ONE 2015, 10, e0144071. [Google Scholar] [CrossRef]
- Kerkes-Kuyumcu, Ö.; Bayazit, S.S.; Alam, M.A. Antibiotic amoxicillin removal from aqueous solution using magnetically modified graphene nanoplatelets. J. Ind. Eng. Chem. 2016, 36, 198–205. [Google Scholar] [CrossRef]
- Yin, Y.; Guo, X.; Peng, D. Iron and manganese oxides modified maize straw to remove tylosin from aqueous solutions. Chemosphere 2018, 205, 156–165. [Google Scholar] [CrossRef]
- Choong, C.E.; Ibrahim, S.; Yoon, Y.; Jang, M. Removal of lead and bisphenol A using magnesium silicate impregnated palm-shell waste powdered activated carbon: Comparative studies on single and binary pollutant adsorption. Ecotoxicol. Environ. Saf. 2018, 148, 142–151. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; de Ridder, D.; van der Wal, A.; Sutton, N.B. Harnessing biodegradation potential of rapid sand filtration for organic micropollutant removal from drinking water: A review. Crit. Rev. Environ. Sci. Technol. 2021, 51, 2086–2118. [Google Scholar] [CrossRef]
- Zhang, L.; Carvalho, P.N.; Bollmann, U.E.; Ei-taliawy, H.; Bester, K. Enhanced removal of pharmaceuticals in a biofilter: Effects of manipulating co-degradation by carbon feeding. Chemosphere 2019, 236, 124303. [Google Scholar] [CrossRef]
- Di Marcantonio, C.; Bertelkamp, C.; van Bel, N.; Pronk, T.E.; Timmers, P.H.A.; van der Wielen, P.; Brunner, A.M. Organic micropollutant removal in full-scale rapid sand filters used for drinking water treatment in The Netherlands and Belgium. Chemosphere 2020, 260, 127630. [Google Scholar] [CrossRef]
- Hedegaard, M.J.; Prasse, C.; Albrechtsen, H.-J. Microbial degradation pathways of the herbicide bentazone in filter sand used for drinking water treatment. Environ. Sci. Water Res. Technol. 2019, 5, 521–532. [Google Scholar] [CrossRef]
- Hedegaard, M.J.; Albrechtsen, H. Microbial pesticide removal in rapid sand filters for drinking water treatment e Potential and kinetics. Water Res. 2014, 48, 71–81, Erratum in Water Res. 2017, 122, 708–713. [Google Scholar] [CrossRef]
- Brunner, A.M.; Vughs, D.; Siegers, W.; Bertelkamp, C.; Kolkman, A.; Laak, T. Monitoring transformation product formation in the drinking water treatments rapid sand filtration and ozonation. Chemosphere 2018, 214, 801–811. [Google Scholar] [CrossRef]
- Papadopoulou, A.; Hedegaard, M.J.; Musovic, S.; Smets, B.F. Methanotrophic contribution to biodegradation of phenoxy acids in cultures enriched from a groundwater-fed rapid sand filter. Appl. Microbiol. Biotechnol. 2018, 103, 1007–1019. [Google Scholar] [CrossRef]
- Pompei, C.M.E.; Ciric, L.; Canales, M.; Karu, K.; Vieira, E.M.; Campos, L.C. Influence of PPCPs on the performance of intermittently operated slow sand filters for household water purification. Sci. Total Environ. 2017, 581–582, 174–185. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Zhou, Q.; Campos, L.C. The application of GAC sandwich slow sand filtration to remove pharmaceutical and personal care products. Sci. Total Environ. 2018, 635, 1182–1190. [Google Scholar] [CrossRef]
- Verma, S.; Daverey, A.; Sharma, A. Slow sand filtration for water and wastewater treatment—A review. Environ. Technol. Rev. 2017, 6, 47–58. [Google Scholar] [CrossRef]
- Georgin, J.; Franco, D.S.P.; Da Boit Martinello, K.; Lima, E.C.; Silva, L.F.O. A review of the toxicology presence and removal of ketoprofen through adsorption technology. J. Environ. Chem. Eng. 2022, 10, 107798. [Google Scholar] [CrossRef]
- Georgin, J.; Franco, D.S.P.; Netto, M.S.; Gama, B.M.V.; Fernandes, D.P.; Sepúlveda, P.; Silva, L.F.O.; Meili, L. Effective adsorption of harmful herbicide diuron onto novel activated carbon from Hovenia dulcis. Colloids Surfaces A Physicochem. Eng. Asp. 2022, 654, 129900. [Google Scholar] [CrossRef]
- Zhou, Y.; Zhang, L.; Cheng, Z. Removal of organic pollutants from aqueous solution using agricultural wastes: A review. J. Mol. Liq. 2015, 212, 739–762. [Google Scholar] [CrossRef]
- Thuptimdang, P.; Siripattanakul-Ratpukdi, S.T.R.; Youngwilai, A.; Khan, E. Biofiltration for Treatment of Recent Emerging Contaminants in Water: Current and Future Perspectives. Water Environ. Res. 2020, 93, 972–992. [Google Scholar] [CrossRef]
- Greenstein, K.E.; Lew, J.; Dickenson, E.R.V.; Wert, E.C. Investigation of biotransformation, sorption, and desorption of multiple chemical contaminants in pilot-scale drinking water biofilters. Chemosphere 2018, 200, 248–256. [Google Scholar] [CrossRef]
- Zhang, S.; Courtois, S.; Gitungo, S.; Raczko, R.F.; Dyksen, J.E.; Li, M.; Axe, L. Microbial community analysis in biologically active filters exhibiting efficient removal of emerging contaminants and impact of operational conditions. Sci. Total Environ. 2018, 640–641, 1455–1464. [Google Scholar] [CrossRef]
- Zhang, Y.; Zhu, H.; Szewzyk, U.; Lübbecke, S.; Geissen, S.U. Removal of emerging organic contaminants with a pilot-scale biofilter packed with natural manganese oxides. Chem. Eng. J. 2017, 317, 454–460. [Google Scholar] [CrossRef]
- Ahmed, M.B.; Zhou, J.L.; Ngo, H.H.; Guo, W.; Thomaidis, N.S.; Xu, J. Progress in the biological and chemical treatment technologies for emerging contaminant removal from wastewater: A critical review. J. Hazard. Mater. 2017, 323, 274–298. [Google Scholar] [CrossRef] [PubMed]
- Ávila, C.; García-Galán, M.J.; Borrego, C.M.; Rodríguez-Mozaz, S.; García, J.; Barceló, D. New insights on the combined removal of antibiotics and ARGs in urban wastewater through the use oftwo configurations of vertical subsurface flow constructed wetlands. Sci. Total Environ. 2021, 755, 142554. [Google Scholar] [CrossRef]
- Gorito, A.M.; Ribeiro, A.R.; Almeida, C.M.R. A review on the application of constructed wetlands for the removal of priority substances and contaminants of emerging concern listed in recently launched EU legislation *. Environ. Pollut. 2017, 227, 428–443. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.; Liu, Y.; Deng, W.; Ying, G. Removal of steroid hormones and biocides from rural wastewater by an integrated constructed wetland. Sci. Total Environ. 2019, 660, 358–365. [Google Scholar] [CrossRef]
- Arora, S.; Saraswat, S.; Rajpal, A.; Shringi, H.; Mishra, R.; Sethi, J.; Rajvanshi, J.; Nag, A.; Saxena, S.; Kazmi, A.A. Effect of earthworms in reduction and fate of antibiotic resistant bacteria (ARB) and antibiotic resistant genes (ARGs) during clinical laboratory wastewater treatment by vermifiltration. Sci. Total Environ. 2021, 773, 145152. [Google Scholar] [CrossRef]
- Bhat, S.A.; Vig, A.P.; Li, F.; Ravindran, B. (Eds.) Earthworm Assisted Remediation of Effluents and Wastes; Springer: Singapore, 2020; ISBN 9789811545214. [Google Scholar]
- Shokouhi, R.; Ghobadi, N.; Godini, K.; Hadi, M.; Atashzaban, Z. Antibiotic detection in a hospital wastewater and comparison of their removal rate by activated sludge and earthworm-based vermifilteration: Environmental risk assessment. Process Saf. Environ. Prot. 2019, 134, 169–177. [Google Scholar] [CrossRef]
- Alessio, M.D.; Yoneyama, B.; Kirs, M.; Kisand, V.; Ray, C. Pharmaceutically active compounds: Their removal during slow sand filtration and their impact on slow sand fi ltration bacterial removal. Sci. Total Environ. 2015, 524–525, 124–135. [Google Scholar] [CrossRef] [PubMed]
- Sabogal-Paz, L.P.; Cintra, L.; Bogush, A.; Canales, M. Household slow sand filters in intermittent and continuous flows to treat water containing low mineral ion concentrations and Bisphenol A. Sci. Total Environ. 2020, 702, 135078. [Google Scholar] [CrossRef] [PubMed]
- Ma, B.; Arnold, W.A.; Hozalski, R.M. The relative roles of sorption and biodegradation in the removal of contaminants of emerging concern (CECs) in GAC-sand biofilters. Water Res. 2018, 146, 67–76. [Google Scholar] [CrossRef]
- Chen, J.; Deng, W.; Liu, Y.; Hu, L.; He, L.; Zhao, J.; Wang, T.; Ying, G. Fate and removal of antibiotics and antibiotic resistance genes in hybrid constructed wetlands. Environ. Pollut. 2019, 249, 894–903. [Google Scholar] [CrossRef] [PubMed]
- Sgroi, M.; Pelissari, C.; Roccaro, P.; Sezerino, P.H.; García, J.; Vagliasindi, F.G.A.; Ávila, C. Removal of organic carbon, nitrogen, emerging contaminants and fluorescing organic matter in different constructed wetland configurations. Chem. Eng. J. 2018, 332, 619–627. [Google Scholar] [CrossRef]
- Oliveira, M.D.; Atalla, A.A.; Emanuel, B.; Frihling, F.; Cavalheri, P.S.; Migliolo, L.; Filho, F.J.C.M. Ibuprofen and caffeine removal in vertical flow and free-floating macrophyte constructed wetlands with Heliconia rostrata and Eichornia crassipes. Chem. Eng. J. 2019, 373, 458–467. [Google Scholar] [CrossRef]

| EC Type | EC Subtype | Contaminant | Water Solubility [mg/L] | Log Kow | Concentration Found [ng/L] | Type of Water | Reference |
|---|---|---|---|---|---|---|---|
| PPCPs | Stimulant | Caffeine | 21,600 | −0.07 | 753,500–1.0 × 106 | Surface water | [22,54,55] |
| 20–23,970 | Ground water | ||||||
| 500–5000 | Drinking water | ||||||
| Anti-inflammatory | Ibuprofen | 21 | 0.35 | 13.5–89,500 | Effluent WWTP | [19,22,54,56] | |
| Anticonvulsant | Carbamazepine | 112–236 | 13.90 | 589–3.5 × 108 | Influent WWTP | [19,57,58] | |
| 1200–6.6 × 107 | Effluent WWTP | ||||||
| Lipid regulator | Clofibric acid | 214,650 | 2.88 | nd–420 | Influent WWTP | [19,59] | |
| Antibiotic | Ciprofloxacin | 650 | 0.28 | 2200–14,000 | Influent WWTP | [19,60,61] | |
| 1100–44,000 | Hospital wastewater | ||||||
| Diagnostic Contrast Media | Iopromide | 23.8 | −2.1 | 780–11,4000 | River water | [19,62] | |
| 1170–4030 | Urban effluents | ||||||
| Antidepressant | Diazepan | 50 | 3.08 | nd–100 | Surface water | [19,63] | |
| nd–100 | Influent WWTP | ||||||
| Beta blocker | Atenolol | 300 | 0.16 | 90–255 | Influent WWTP | [19,64] | |
| Fragrance | Musk xylene | 0.49–1.0 | 4.40 | 200,000–400,000 | Drinking water | [19,65] | |
| Disinfectant | Triclosan | 10.00 | 4.76 | 200–1854 | Influent WWTP | [24,25,54] | |
| Pesticides | Herbicide | Glyphosate | 15.70 | −3.40 | up to 6.01 × 109 | Surface water | [30,66] |
| Insecticide | Acetamiprid | 2950 | 0.80 | 0.08–249 | Surface water | [27,29] | |
| Clothianidin | 304 | 0.70 | 1.0–740 | Surface water | [27,29,67,68] | ||
| Thiamethoxam | 4100 | −0.13 | 1.0–914 | Surface water | |||
| Steroid hormones | Natural | E2 | 13 | 2.45 | 3.8–188.0 | Influent WWTP | [19,58,60,69] |
| E1 | 13 | 3.43 | 12–196 | Effluent WWTP | [23] | ||
| 17α-estradiol | 13.3 | 4.01 | 6.4–12.6 | ||||
| Synthetic | EE2 | 4.8 | 3.67 | 0.59–5.6 | |||
| Industrial and chemical substances | Anticorrosive | Methylbenzotriazole | 366 | 2.72 | nd–2900 | Effluent WWTP | [29] |
| Plasticizer | BPA | 120 | 2.2–3.4 | 0.01–8800 | Bottled water | [5,70] | |
| 140–12,000 | Surface waster | ||||||
| Microplastics | - | - | - | - | 8.3–3.1 × 105 * | Effluent WWTP | [45,49] |
| 0.27–30 * | Lake water | ||||||
| 0.05–320 * | River water |
| Adsorbent Characteristics | Adsorption Behavior | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Adsorbent | Composition/ Functional Groups/Ions | SA[m2/g] P [cm3/g] | Adsorbent | EC Removed | Adsorption Conditions | Adsorption Mechanism | Removal [%] | Adsorption Capacity [mg/g] | Reference | |
| Activated carbon (AC): Carbonized carbonaceous materials (S.O.) [71] | C, H, N, S and O [78] COOH, C=O, OH−, -NH2, CHO, etc. [91] | SA = 300–2500 [78] P = 0.1–16.9 [91,93,94,95] | AC from argan waste | Paracetamol Amoxicillin | CT = 0.8–12, T = 25, Ph = 3–11, AD = 0.1, [EC] = 100 | van der Waals forces, H-bonding, dipole–dipole interactions, ion exchange, covalent bonding, cation bridging and water bridging [16,63,79,93] | ~95 | 512 319 | [96] | |
| Commercial AC | Caffeine | CT = 4, T = 30 ± 2, pH = 3–8, AD = 0.1, [EC] = 1.125–1.252 | 44.1 | 71.7 | [22] | |||||
| Ibuprofen | 52.7 | 72.3 | ||||||||
| Triclosan | 60.8 | 70.0 | ||||||||
| Commercial GAC | Tetracycline | CT = 180, T = 30 AD = 2.4, [EC] = 100 | - | 845.9 | [91] | |||||
| Ibuprofen | - | 239.8 | ||||||||
| AC from olive pomace | Niesulide | AD = 0.1–0.5, CT = 6, [EC] = 10–30, pH = 8–11 | - | 353.3 | [76] | |||||
| Commercial GAC | Metronidazole | CT = 2, T = 20 ± 1 pH = 3.9–10.2, AD = 5 [EC] = 99.27 | 69.0–80.0 | - | [74] | |||||
| Modified commercial AC | Triclosan | CT = 4, T = 30 ± 2 pH = 6, AD = 0.07, [EC] = 1.0 | 98.3 | 395.2 | [24] | |||||
| AC from biomass | Paracetamol | CT = 240, T = 15–35, AD = 0.1, [EC] = 1–20 | - | 100 | [75] | |||||
| AC from fruit of Butiacapitate | Paracetamol Ketoprofenon | T = 25–55, AD = 0.9, [EC] = 50–300 pH = 7 | 73.0–98.2 | 101.2–134.5 | [97] | |||||
| GAC from coconut shells | Mix: Caffeine, hydro-chlorothiazide, saccharin, sucralose, sulfamethoxazole | T = 25, AD = 0.07, [EC] = 0.5, pH = 2–12, CT = up to 5.5 | - | 1.21–4.33 | [37] | |||||
| Zeolites: Crystalline microporous aluminosilicate (S.O. and N.O.) [72] | Si, Al, O and cations [72] K+, Na+, Ca2+, and Mg2+ [92] | SA = 300–2300 [72] P = 0.10–0.35 [92] | Zeolite + Hexadecyltrimethylammonium bromide or chloride | Diclofenac | CT = 24, Ta = 32, Tb = 42, Tc = 52, pH = 2–11, AD = 25, [EC] = 636.28 | van der Waals forces and acid-base forces, donor–acceptor interaction [60,66] | - | Up to 47.4 | [81] | |
| Natural zeolite | Tetracycline Oxytetracycli-ne | CT = 2, pH = 6, AD = 6.0, [EC] = 44.4–46.0 | 90 75 | - | [79] | |||||
| Modified zeolite | Mesosulfuron-methyl | CT = 72, T = 25, pH = 6, AD = 2.0, [EC] = 8 | - | 2.4–3.4 | [80] | |||||
| Cu2+ zeolite 4A | Glyphosate | CT = 2, T = room, pH = 6–8, AD = 2.0, [EC] = 100 | - | 112.7 | [33] | |||||
| Zeolite Y (ZY) | 2,4-dichlorphe- noxyacetic acid, paraquat | CT = 24, T = 28 ± 2 pH = 3, AD = 1.0 [EC] = 20–250 | - | 82.6–175.4 - 71.4–92.6 | [82] | |||||
| Powdered zeolites: FAU 1/FAU 2 | Azithromycin, ofloxacin, sulfamethoxazole | CT = 6, pH = 6.5, [EC] = 0.1 | ≥80 | 7.0–8.5 25.3–31.2 - | [83] | |||||
| Clays: Hydrous aluminosilicates (N.O.) [86,87,98] | SiO2, CO32−, MOx [86,98]/ Ca2+, Mg2+, H+, K+, NH4+, Na+, SO42−, Cl−, PO43−, NO3−, and OH− [73,84] | SA: 10–426 [73,84] P: 0.1–0.5 [99] | Natural clay | Methomyl | CT = 3, T = 25 ± 1 pH = 6.6, AD = 10 [EC] = 19.99–43.71 | Ion exchange, coordination, dipole–dipole interactions, van der Waals forces, H-bonding, acid-base forces, donor–acceptor interaction, acid-base forces [87] | 27.6–32.9 | 0.3–0.5 | [85] | |
| Bentonite | Atenolol, sulfamethoxazole, diclofenac | CT = 24, T = 20 pH = 6.5–8.0, AD = 1.6 [EC] = 1–50 | - | 5.3–24.5 1.1–3.5 1.1–4.0 | [64] | |||||
| Sodium smectite | Tramadol, doxepin | CT = 16, T = 20 pH = 6–7, AD = - [EC] = 10 | - | 210.7 279.4 | [88] | |||||
| Bentonite + Surfactant | Diuron and their byproducts | CT = 24, T = 18–20 pH = 3, AD = 0.5 [EC] = 0.25 | - | 0.1–0.6 | [84] | |||||
| Organo-montmorillonites, organo-pillared-montmorillon, organo-acid-activated montmorillonite | 2,4,5-trichlorophenol | CT = 2, T = 20 ± 1 pH = 4, AD = - [EC] = 10–200 | - | 374.9 | [90] | |||||
| Natural/modified bentonite | Methomyl | CT = 2, T = 20, 30, 40 pH = 4, AD = 10 [EC] = 0.1 | 66–76 | 5 | [89] | |||||
| Adsorbent Characteristics | Adsorption Behavior | ||||||||
|---|---|---|---|---|---|---|---|---|---|
| Adsorbent | Composition/ Functional Groups/Ions | SA[m2/g] P [cm3/g] | Adsorbent | EC Removed | Adsorption Conditions | Adsorption Mechanism | Removal [%] | Adsorption Capacity [mg/g] | Reference |
| Manganese oxide: N.O. and S.O. [111] | Mn(II), Mn(III), Mn(IV) or Mn(VII) [100,111]/ Mn-O, OH− [115] | SA: 12–236 [111] | Manganese oxide | Diclofenac | CT = 33, T = 30, pH = 7.0, AD = 0.61 [EC] = 49,946.41 | Electrostatic, cation–exchange interactions, and van der Waals forces [113] | Up to 90 ± 0.7 | - | [113] |
| Manganese oxide | Phenol | CT = 1, T = 19, pH = 6.7 ± 0.5, AD = 10.0 [EC] = 0.5 | ~40 | - | [114] | ||||
| Manganese oxide | Clarithromycin Roxithromycin | CT = 24, T = 20, pH = 5.0, AD = 1.0 [EC] = 4.94–83.70 | 85–90 | - | [112] | ||||
| Manganese oxide + Fe | Resorcinol | CT = 0.33–2.0, pH = 5.0, AD = 0.0157 [EC] = 35.0 | ~100 | - | [114] | ||||
| Manganese oxide birnessite | Niclosamide | CT = 24, T = 22 ± 2, pH = 5.0, AD = 0.015 [EC] = 0.042 | - | - | [110] | ||||
| Alumina: N.O. and S.O. [105,117] | MOx, FeO, SO42−, Al2O3 (α, β, γ) [101]/ O-Al-O, OH− [115] | AS: 50–300 [101,118] | Alumina and surfactant modified alumina | Ortho-Nitro-Phenol | CT = 1, pH = 6.0, AD = 5.0 [EC] = 55.64 | Electrostatic interactions and hydrogen bonding [103,119] | - | 4.4–7.3 | [104] |
| α-Alumina nanoparticles and Modified α-Alumina nanoparticles | Ciprofloxacin | CT = 1.5, T= 25 ± 2, pH = 6, AD = 5, [EC] = 10 | 33.6–97.8 | 34.5 | [103] | ||||
| MCM-41 | Norfloxacin | CT = 1, T = 15, pH = 3.0–7.0, AD = 0.5 [EC] = 60.0 | >80 | 52 | [106] | ||||
| Raw alumina and Raw alumina modified with HCl | Acrylic acid | CT = 0.5–120, T = 20–50, pH = 4.5, AD = 66.7, [EC] = 0.08 | 32.1–36.2 | 0.29–0.31 | [109] | ||||
| Alumina and HDTMA modified alumina | Metha-nitrophenol | CT = 1, T = 25–45, pH = 6.0, AD = 5.0 [EC] = 0.4 | - | 3.0–8.1 | [104] | ||||
| Silica: N.O. and S.O. [63,108] | SiO2− [63]/ Si-O-H, OH− [108] | SA: 7.5–up to 1000 [108,116] P: 0.53–1.03 [109] | Silica | Tetracycline | T = 23, pH = 6.0, AD = - [EC] = - | H bonds or cationic exchange [108,109] | - | - | [102] |
| Mesoporous silica SBA-15 | Carbamazepine Diclofenac Ibuprofen Ketoprofen Clofibric acid | CT = 2, T = 25, pHa-d = 5, pHe = 3, AD = 2.0 [EC] = 0.1 | 35.6–94.3 | 0.16 0.34 0.41 0.28 0.07 | [108] | ||||
| Adsorbent Characteristics | Adsorption Behavior | ||||||||
|---|---|---|---|---|---|---|---|---|---|
| Adsorbent | Composition/ Functional Groups/ions | SA[m2/g] P [cm3/g] | Adsorbent | EC Removed | Adsorption Conditions | Adsorption Mechanism | Removal [%] | Adsorption Capacity [mg/g] | Reference |
| CNT: S.O. [170,171] | Graphene or graphite sheet with π conjugative structure and highly hydrophobic surface [170]/ –OH, –C=O, –COOH [72] | SWCNT SA: 400–1020 [62,93] MWCNTs SA: 38.7–>500 [93] P = 0.59 [93] | *SWCNT | 17a-ethinyl estradiol BPA | CT = 4, pH = 3.5–11, T = room, AD = 0.05 [EC] = 2.28–2.96 | Hydrophobic effect, π–π interactions, hydrogen bonding, covalent bonding, and electrostatic interactions [165,166] | 95–98 75–80 | 35.5–35.7 13.4–16.1 | [171] |
| SWCNT MWCNT | Lincomycine, Sulfamethoxazole, iopromide | CT = 72, T = 20 ± 1, pH = 6.0 ± 0.2, AD = - [EC] = 12,000 | - | - | [62] | ||||
| MWCNT | Roxarsone | CT = -, T = 10, pH = 2–11.7, AD = 2 [EC] = 10 | - | Up to 13.5 | [172] | ||||
| MWCNT MWCNT-COOH MWCNT-NH2 N-CNT | 1,8dichlorooctane, nalidixic acid, 2-(4-ethylphenoxy) ethanol | CT = 72, T = 25, pH = 7.0 ± 0.5, AD = 0.02–0.2 [EC] = 20–80 | - | 248–380 79–111- | [173] | ||||
| MWCNT modified with HNO3 | Diclofenac | CT = 1, T = 25, pH = 7.0, AD = 5.4, [EC] = 50 | Up to 95 | Up to 8.6 | [174] | ||||
| Graphene: S.O. [180] | 2D single layer of sp2 hybridized carbon atom [180]/ Epoxide, carbonyl, carboxyl, and hydroxyl groups [180] | SA: 46.4–2630 (theoretical) [179,180] P: 0.065 [219,220] | Graphene oxide | β-estradiol 17α-ethynyl estradiol | CT = 0.83, T = 25, pH = 3.0, AD = 0.40 [EC] = 8.0 | Hydrophobic effect, π–π interactions, hydrogen bonding, covalent bonding, and electrostatic interactions [153,180] | 97.2 98.5 | - - | [180] |
| Graphene oxide | Diclofenac | CT = 0.25, T = 60, pH = 6.0, AD = 0.16 [EC] = 400 | 96.2 | 653.9 | [175] | ||||
| Graphene oxide | TetrabromoBPA BPA | CT = 24, T = 15–35, pH = 6.0, AD = 1.25 [EC] = 20 | 19.1 17.5 | [9] | |||||
| Graphene oxide | Metformin | CT = 1–3, T = 1545, pH = 4.5–8.5, AD = 0.05–0.15 [EC] = 300–700 | 59–97.6 | 122.6 | [182] | ||||
| Graphene oxide | Nicotine | CT = 0.5, T = 25–55 pH = 3–10.5 AD = 0.1 [EC] = 5–150 | - | 96.5 | [183] | ||||
| Double-oxidized graphene oxide | Acetaminophen | CT = 0.17, T = 25, pH = 8.0, AD = 0.02 [EC] = 10 | 83.7 | 704 | [181] | ||||
| Graphene oxide nanoflakes | BPA, 4-nonylphenol, tetrabromineBPA | CT = 0.08–24, T = 25, pH = 4–9, AD = 1.25, [EC] = 20 | - | 19–30 | [220] | ||||
| 3D Graphene oxide | Polystyrene microplastics | CT = 2, T = 26, pH = ~7, AD = 1.25, [EC] = 600 | - | 617.3 | [212] | ||||
| Iron oxides: N.O. and S.O. [192,193] | FeOx [193,221]/ FeO, OH-, COOH-, C=O [193] | SA: ZVI > 10 Hematite ~30 Goethite 18–83.5 Magnetite 40–300 Maghemite 31–178 [40,167,192,193] P: Magnetite ~0.22 [216] | Magnetite | Levofloxacin | CT = 0–4, T = 15–45, pH = 6.5, AD = 1.0, [EC] = 2.5–20 | Ion-dipole, van der Waals forces, or ion exchange, hydroxyethyl cleavage and chelation [40,167,192,193,196] | 36.7–80.1 | 6.1–6.8 | [189] |
| Maghemite | Diclofenac | CT = 6.25, T = 25 ± 1, pH = 7, AD = 1.0, [EC] = 100–500 | ~90 | 261 | [192] | ||||
| Goethite | Ibuprofen | CT = 2, T = 25, pH = 7, AD = 0.25, [EC] = 1.65–2.06 | 89–91 | 0.72–3.47 | [195] | ||||
| Goethite | Diclofenac | CT = 0.08–48, T = 25, pH = 5.3–10, AD = 10, [EC] = 1 | 75 | - | [193] | ||||
| Hematite | Cephalexin | CT = 0.08–6.67, T = 25, pH = 2–10, AD = 2, [EC] = 25–250 | ~99.7 | 70 | [196] | ||||
| Hematite and goethite | 4-n-nonylphenol | CT = 24, T = Room T, pH = 7.0–9.0, AD = 100, [EC] = 1.5 | - | - | [222] | ||||
| Magnetite | Oxytetracycline | T = 5–35, pH = 5.6, AD = 2.5–20, [EC] = 868.86 | 60–100 | - | [191] | ||||
| Nano-alumina: S.O. [101] | MOx, FeO, SO42−, Al2O3 (α, β, γ) [101]/ O-Al-O, OH-, C=O [101] | SA: 143.7 [223] P: 3 [223] | Nano-alumina | Dichlorodiphenyltri-chloroethane, Polychlorinated biphenyls | CT = 0.33, T = 28–34, [EC] = 10–60 | Hydrophobic, hydrogen bonding, and van der Waals, Electrostatic interactions [63,101,102,223] Π–π interaction, electron donor acceptors [200] | 54–68 | Up to 0.18 Up to 0.16 | [224] |
| Nano-silica: S.O. [63,102] | SiO2 [102]/ Si-O-H, OH− [63] | Information not available | Nano-silica/modified nano-silica | Ciprofloxacin | CT = 3.33, T = 25 ± 2, pH = 3–7, AD = 1–50, [EC] = 20 | 56.8–89.9 | 85 | [168] | |
| ZnO: S.O. | Zn, O/ Zn-O, O-H, C-H | SA: ~14.8 | ZnO | Naphthalene | CT = 1, T = 25 ± 2, pH = 2–12, AD = 600 [EC] = 25 | 100 | 148.3 | [26] | |
| MgO: S.O. [200] | MgO [202] Mg-O, OH- [200] | SA: 48–108 [200] P: 0.3–24.8 [200] | MgO | Linezolid | CT = 1, T = 35, pH = 3–8, AD = 0.2–0.8 [EC] = 10–100 | ~62 | 123.4 | [198] | |
| Nano-composites: S.O. [177,179,202,203] | Depends on the components of the composite, e.g., carbon atom, FeOx, graphene, and graphite sheet, among others/ Depends on the components of the composite, e.g., OH-, COOH-, C=O, epoxy, and amino, among others [178,179,223,225] | SA: 3.18- around 1260 [201] P: 0.15–0.72 [178,223,225] | MOFs UIO-66 | Sulfachloropyra-dazine | CT = 2, T = 25, pH = 5.5, AD = 0.1, [EC] = 10–100 | Depends on the components of the composite, are an example hydrogen bonding, π–π interaction, cation–π bonding, and amidation reaction, electrostatic interaction, hydrophobic interaction, ligand exchange, cation–π bonding, chemisorption, etc. [178,225,226] | 80 | 417 | [205] |
| Magnetic activated carbon | Triclosan Bisphenol-A Tonalide Metolachlor Ketoprofen E2 | CT = 1, T = 25–45, pH = 7, AD = -, [EC] = 0.025–0.25 | 96–98 | 21.32 31.05 29.41 22.37 28.49 20.20 | [206] | ||||
| Magnetic activated carbon | Carbamazepine | CT = 0.5, T = 25, pH = 6.65, AD = 0.05, [EC] = 20.0 | 93 | 189.5 | [214] | ||||
| Triethoxyphenylsilane (TECs)-functionalized magnetic palm-based powdered AC | BPA, carbamazepine, ibuprofen, clofibric acid | CT = 6, T = Room T, pH = 7, AD = 0.1, [EC] = 10 | - | 58.1–166.7 | [207] | ||||
| Magnetic activated carbon/chitosan | Ciprofloxacin, erythromycin, amoxicillin | CT = 2, T = 25, pH = -, AD = 1.5, [EC] = - | 54–82 | 90.1 178.6 526.3 | [200] | ||||
| Magnetic cellulose ionomer/layered double hydroxide | Diclofenac | CT = 0.5, T = -, pH = 9, AD = 1.0, [EC] = 0.5 | ~100 | 268 | [209] | ||||
| Magnetic chitosan grafted graphene oxide | Ciprofloxa-cin | CT= 8, T= -, pH = 5, AD = 0.33, [EC] = 20 | - | 282.9 | [208] | ||||
| Fe3O4/graphene oxide reduced | Ametryn, atrazine prometryn, simazine, simeton | CT = 1.17, T= 25, pH = 5.0, AD = 0.5, [EC] = 10 | 93.6 | 54.8–63.7 | [203] | ||||
| Magnetically modified graphene nanoplatelets | Amoxicillin | CT = 1.5, T = 20, pH = 5, AD = 2.0, [EC] = 10.0 | 84 | 14.1 | [225] | ||||
| Fe3O4@SiO2-Chitosan/GO | Tetracycline | CT = 8, T = 25, pH = 6, AD = 0.4, [EC] = 44.44 | >90 | - | [176] | ||||
| Fe/Cu-GO | Tetracycline | CT = 0.25, T = 22, pH = 6.5, AD = 0.25, [EC] = 20–100 | ~100% | - | |||||
| GO-BC | Sulfamethazine | CT = 12, T = 25, pH = 6, AD = 1, [EC] = 2031.8–20,332 | - | - | |||||
| Sawdust+ FeCl3 | Tetracycline | CT = 2.0, T = 22 ± 2, pH = 7.8, AD = 4.0, [EC] = 20 | - | ~5.4 | [213] | ||||
| Biochar + Chitosan | Ciprofloxacin | CT = 48, T = 20, pH = 3.0, AD = 5.0, [EC] = 160 | >76 | [217] | |||||
| Magnetic bamboo-based activated carbon | Ciprofloxacin, norfloxacin | CT = 24, T = 25, pH = 6, AD = 2.0, [EC] = 300 | - | 245.6 293.2 | [215] | ||||
| Magnetic pine sawdust biochar | Sulfamethoxazole | CT = 24, pH = 7 ± 0.10, [EC] = 0.17–78 | - | 51.8–87.9 | [216] | ||||
| Maize straw + manganese/iron oxides | Tylosin | CT = 0.25–24, T = 25, pH = 3–11, AD = 2, [EC] = 20 | - | 3070 | [226] | ||||
| Palm-shell waste AC + magnesium silicate | BPA | CT = 24, T = 24 ± 1, pH = 4.5, AD = 0.1, [EC] = 10–100 | - | 168.4 | [227] | ||||
| Chitin + GO+ O-C3N4 | Carboxylate-modified PE, amine-modified PE, PE (~1 µm) | CT = 48, T = 25, pH = 7, [EC] = 1.0 | 71.6–92.1 | 5.4–9.7 | [213] | ||||
| Type of Filtration (Scale) | Bed Filter Material (Size, mm) | Influent Type | EC Removed | Operational Conditions | Removal Mechanism | Removal [%] | Reference |
|---|---|---|---|---|---|---|---|
| Aerated rapid filtration. (microcosms scale) | Sand (3–5) | Anaerobic groundwater | Mecoprop, bentazone, glyphosate, p-nitrophenol | [EC] = 3 × 10−5–2.4 × 10−3, RT = 0.17 | Biodegradation | 1–85 | [232] |
| Dichlorprop | [EC] = 2 × 10−4, RT = 0.93 | >50 | [232] | ||||
| Rapid filtration (microcosms scale) | Sand (3–5) | Anaerobic groundwater | Bentazone | [EC] = 5, RT = 312 | Biodegradation | 92 | [231] |
| Rapid filtration (microcosms scale) | Filtralite clay (0.8–1.6) | Groundwater enriched with Ecs | 2,6-dichlorobenzamide, bromoxynil, chlorotoluron, diuron, ioxynil, isoproturon, linuron, 4-chloro-2-methylphenoxy acetic acid | [EC] = 2.1 × 10−3–6.6 × 10−3, RT = 0.023, FR = 21 | Biodegradation | 13–98 | [234] |
| Rapid filtration (lab scale) | Sand | Influent water from the RSF filters (WRK, Nieuwegein, The Netherlands) enriched with Ecs | Atrazine, bentazon, metolachlor and clofibric acid, carbamazepine | [EC] = 0.01, RT = 8 and 96, FR = 80 | Biodegradation/Sorption | - | [233] |
| Rapid filtration: downflow, upflow, dual media down flow (field scale) | Sand (0.7–2.5) Sand + hydroanthracite (1.4–2.5) Sand + anthracite (1.6–2.5) | Surface water from The Netherlands and Belgium | Caffeine, acesulfame-K, sucralose, metformin, phenazone, chloridazon, valsartan, sulfadiazine, sotalol, etc | [EC] ≤ 1 × 10−5–5.7 × 10−4 RT = 15–240 | Biodegradation/Sorption (probably) | 0–93 | [230] |
| Slow filtration with rapid pulses of a carbon source (lab scale) | Quartz sand (0.210–0.297) | WWTP effluent | Atenolol, metoprolol, iopromide, iomeprol, carbamazepine, diclofenac, sulfadiazine, sulfamethoxazole, etc. | RT = 150, FR = 0.15 | Biodegradation | - | [229] |
| GAC sandwich slow filtration (lab scale) | Coarse sand (0.6) GAC (0.4–1.7) Coarse sand+ GAC | Synthetic wastewater | Mix of DEET, paracetamol, caffeine and triclosan | [EC] = 0.025, HLR = 5, 10, 20 | Adsorption (GAC) + Biodegradation | 18.8–100 | [236] |
| Slow filtration (pilot scale) | Silica sand (0.15–0.30) Support: pea gravel | Stream water/Stream water+ 1% of primary effluent added, both enriched with Ecs | Caffeine, carbamazepine, 17-β estradiol, E1, gemfibrozil, phenazone | [EC] = 0.05, HLR = 5 | Sorption and/or biodegradation | <10–100 | [252] |
| Household slow filtration with intermittent and continuous flows (pilot scale) | Sand (0.09–0.5) Support: coarse sand (1–3)+ fine gravel (3–6) + coarse gravel (10–12) Top: non-woven polyester | Synthetic wastewater | BPA | [EC] = 2.35 Continuous flow HLR = 1.58 Intermittent flow HLR = 0–875 | Biodegradation | Continuous flow 14 ± 6 Intermittent flow 3 ± 8 | [253] |
| Biofilters (bench scale) | GAC (1.0–1.2) and anthracite (0.8–2.0)/sand (0.55–0.65) dual media | Municipal waste streams | Acetaminophen, ibuprofen, erythromycin, sulfamethoxazole, trimethoprim, carbamazepine, atenolol, gemfibrozil, tri(2-chloroethyl) phosphate, DEET, cotinine, aminotrizaole, atrazine, caffeine, E2, iopromide | [EC] = 2.27 × 10−4–6.44 × 10−3 RT = 0.17 and 0.30 | Biodegradation | >75 | [242] |
| Biofilters (pilot scale) | Anthracite/sand (1.07/0.52) | Superficial water of Grand River enriched with Ecs | DEET, atrazine, naproxen, ibuprofen, nonylphenol, carbamazepine | [EC] = 5 × 10−4–5 × 10−3, RT = 0.08 and 0.23, HLR = 500 | Adsorption (non-biodegradable Ecs) and biodegradation (biodegradable Ecs) | <20–100 | [243] |
| Biofilters (pilot scale) | GAC/sand and anthracite/sand | Water from the full-scale recarbonation chambers enriched with Ecs | Atenolol, atrazine, carbamazepine, fluoxetine, gemfibrozil, metolachlor, sulfamethoxazole, tris(2-chloroethyl) phosphate | [EC] = 1 × 10−4–2 × 10−4, 1 × 10−3–3 × 10−3, RT = 8.4 and 4.2, HLR = 488 and 976 | Adsorption and biodegradation | GAC/sand: 49.1–94.4 anthracite/sand: 0–66.1 | [254] |
| Biofilters (pilot scale) | Anthracite-sand and previously used biological activated carbon (BAC)-sand dual media, BAC = (0.9) | Raw surface water (Colorado River) enriched with Ecs | Sulfamethoxazole, caffeine, gemfibrozil, naproxen, DEET, trimethoprim, acetaminophen, ibuprofen, sucralose, meprobamate | [EC]= 1 × 10−4–×10−3, RT = 0.17, HLR = 904.56 | Biodegradation and BAC sorption | <50–>99 | [242] |
| Biofilters (pilot scale) | Natural manganese oxides (3–5) | Secondary effluent of WWTP | 1-hydroxybenzotriazol, 4’-hydroxydiclofenac, 10.11-dihydro-10.11-dihydroxycarbamazepine, acyclovir, benzotriazole, diclofenac, carbamazepine, carboxy-acyclovir, diatrizoic acid, erythromycin, gabapentin, iomeprol, tolyltriazole, sulfamethoxazole, tramadol, | RT = 5 and 10, FR = 8000, HLR = 400, | Adsorption, biodegradation, oxidation | 70–98 | [244] |
| Horizontal/vertical subsurface flow and hybrid CWs, aerated/unaerated (mesocosm scale) | Zeolite (20–30) | Domestic sewage enriched with Ecs | Sulfamonomethoxine, sulfamethazine, sulfameter, trimethoprim, norfloxacin, ofloxacin, enrofloxacin, erythromycin-H2O, roxithromycin, oxytetracycline, lincomycin | [EC] = 5 × 10−3, HLR = 1.67, PT = Iris tectorum | Sorption and biological processes | 87.4–99.1 | [255] |
| Four CWs of subsurface horizontal flow (pilot scale) | Gravel (12.7–19.05) | Synthetic wastewater | Carbamazepine, sildenafil, methylparaben | [EC] = 0.2, FR = 15, RT = 72, PT = Heliconea Zingiberales and Cyperus Haspan | Biodegradation, adsorption, plant absorption | <10–97 | [2] |
| Combination of partially saturated and unsaturated vertical subsurface flow CWs (experimental scale) | Top: sand layer (1–2) Underneath: gravel (3–8) | Urban wastewater (surrounding residential area) from primary treatment | Ciprofloxacin, ofloxacin, pipemidic acid, azithromycin | [EC] = 5 × 10−4, HLR = 0.55, PT = Phragmites australis | Sorption and biodegradation | <−200–>90 | [246] |
| Line 1: Partially vertical flow Line 2: unsaturated vertical flow+ horizontal subsurface flow + free water surface CWs (experimental scale) | - | Urban wastewater from primary treatment | Caffeine, trimethoprim; sulfamethoxazole, DEET, sucralose | HLR = 0.55, FR = 138.89 | Biodegradation | <10–~100 | [256] |
| Vertical flow CW | Top: gravel (4.8–9.5) Filter media: sand (0.27) Bottom: medium gravel (4.8–9.5) + coarse gravel (25–32) | Wastewater | Ibuprofen and caffeine | [EC] ≤ 0.1, HLR = 16, RT = 168, PT = Heliconia rostrata | Biodegradation, adsorption, plant absorption | 90–97 | [257] |
| Vermifiltration (pilot scale) | Soil Sand (0.1–0.8) Detritus (3–10) Support: cobblestone (10–50) | Hospital effluent from sedimentation basin | Ciprofloxacin, ofloxacin, sulfamethoxazole, trimethoprim, tetracycline, metronidazole | HLR = 4.17, ET = Eisenia foetida, ED = 10,000 | Adsorption, earthworm absorption (mineralization/transformation), biodegradation | 40–98 | [251] |
| Integrated CW | Chaff and soil | Domestic + livestock wastewater | Androsta-1,4-diene-3,17-dione, 17α-trenbolone, 17α-boldenone, 17β-boldenone, testosterone, stanozolol, progesterone, ethynyl testosterone, 19-norethindrone, norgestrel, medroxyprogesterone, cortisol, cortisone, prednisone, miconazole, fluconazole, itraconazole, etc. | [EC] = 6.3 × 10−7–1.05 × 10−4, RT = 36, PT = Myriophyllum verticillatum L. and Pontederia cordata | Biodegradation, adsorption, plant absorption | <10–97.6 | [249] |
| Vermifiltration (pilot scale) | Top: vermigratings (0.118) and cow-dung (0.05–5), Small gravel (2–4), Medium gravel (6–8) Support: coarse gravel (12–14) | Clinical laboratory wastewater | Amoxicillin, ampicillin, ticarcillin, ceftazidime, cefotaxime, ceftriaxone, streptomycin, gentamicin, erythromycin, tetracycline, chloramphenicol, ciprofloxacin | HLR = 4.17, RT = 7–8, ET = Eisenia foetida, ED = 10,000 | Earthworms/microorganisms degradation, biofilm adsorption, filter media sorption | - | [250] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Almeida-Naranjo, C.E.; Guerrero, V.H.; Villamar-Ayala, C.A. Emerging Contaminants and Their Removal from Aqueous Media Using Conventional/Non-Conventional Adsorbents: A Glance at the Relationship between Materials, Processes, and Technologies. Water 2023, 15, 1626. https://doi.org/10.3390/w15081626
Almeida-Naranjo CE, Guerrero VH, Villamar-Ayala CA. Emerging Contaminants and Their Removal from Aqueous Media Using Conventional/Non-Conventional Adsorbents: A Glance at the Relationship between Materials, Processes, and Technologies. Water. 2023; 15(8):1626. https://doi.org/10.3390/w15081626
Chicago/Turabian StyleAlmeida-Naranjo, Cristina E., Víctor H. Guerrero, and Cristina Alejandra Villamar-Ayala. 2023. "Emerging Contaminants and Their Removal from Aqueous Media Using Conventional/Non-Conventional Adsorbents: A Glance at the Relationship between Materials, Processes, and Technologies" Water 15, no. 8: 1626. https://doi.org/10.3390/w15081626







