Photocatalytic Removal of Ciprofloxacin in Water by Novel Sandwich-like CuFe2O4 on rGO/Halloysite Material: Insights into Kinetics and Intermediate Reactive Radicals
Abstract
:1. Introduction
2. Experimental
2.1. Chemicals
2.2. Graphene Oxide (GO) Elaboration
2.3. Extraction of Halloysite Nanotubes
2.4. Elaboration of rGO/HNT
2.5. Elaboration of the CuFe2O4/Nanocomposite
2.6. Material Characterizations
2.7. Catalytic Activity Evaluation
2.8. Experimental Results Calculation
3. Results and Discussion
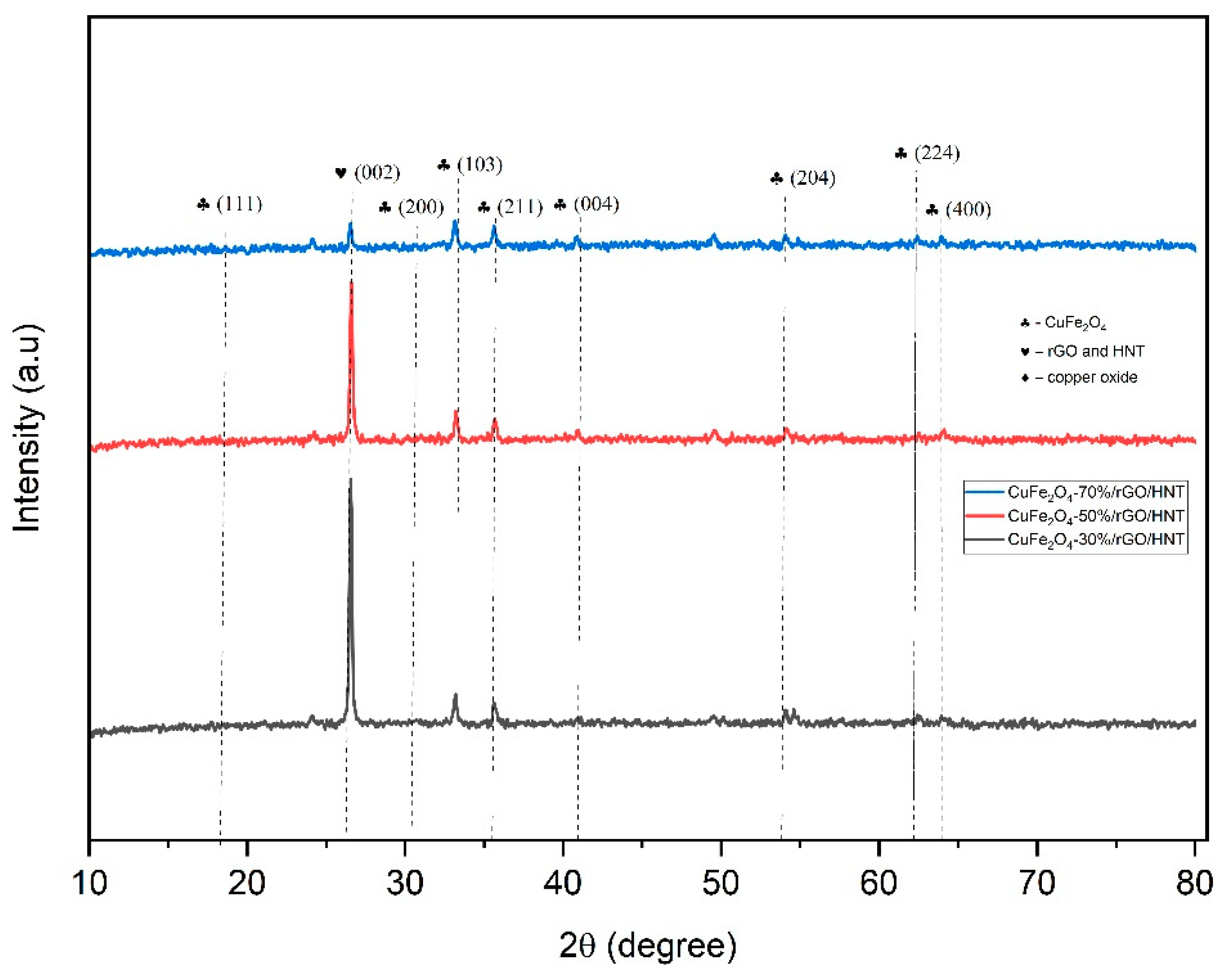
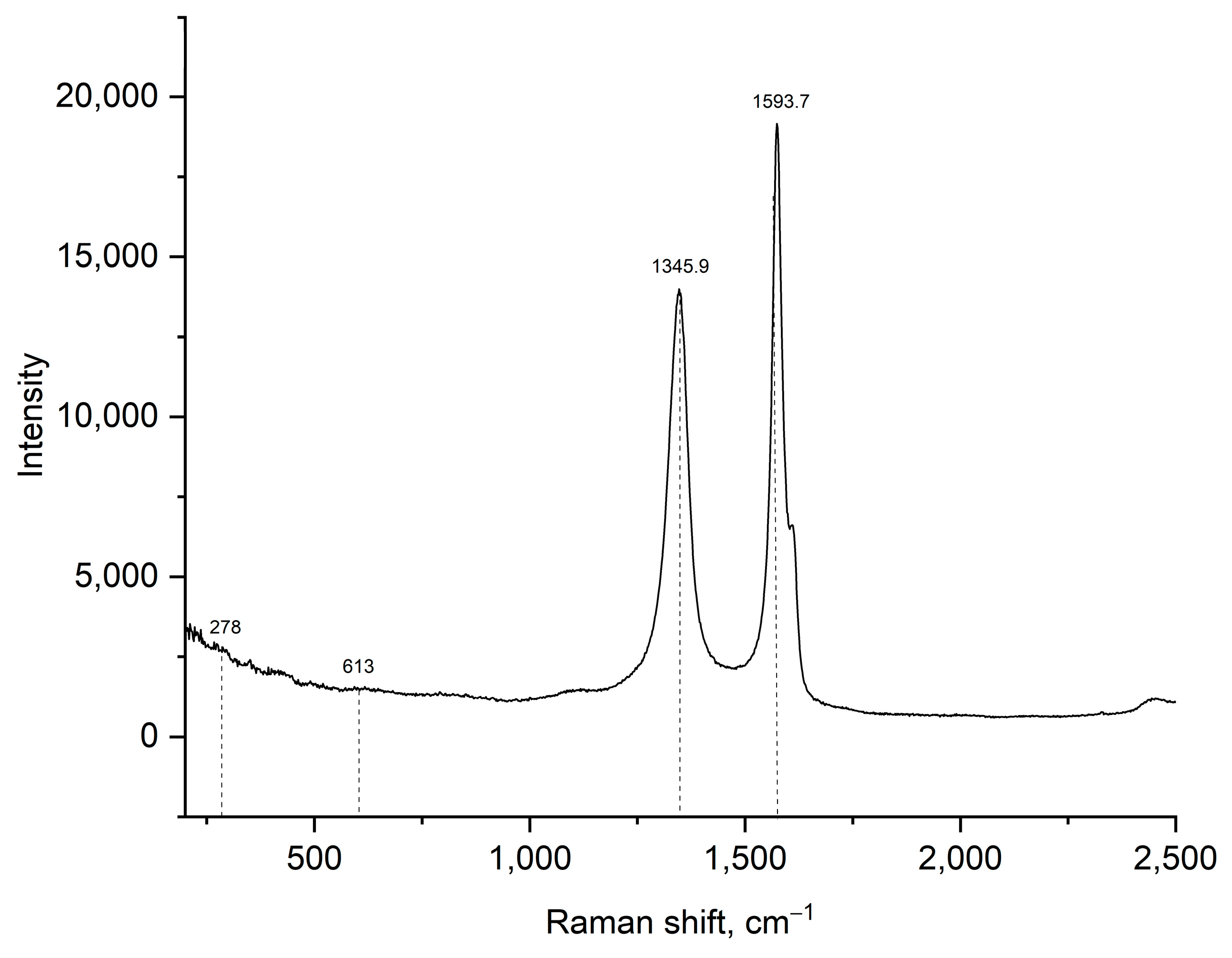
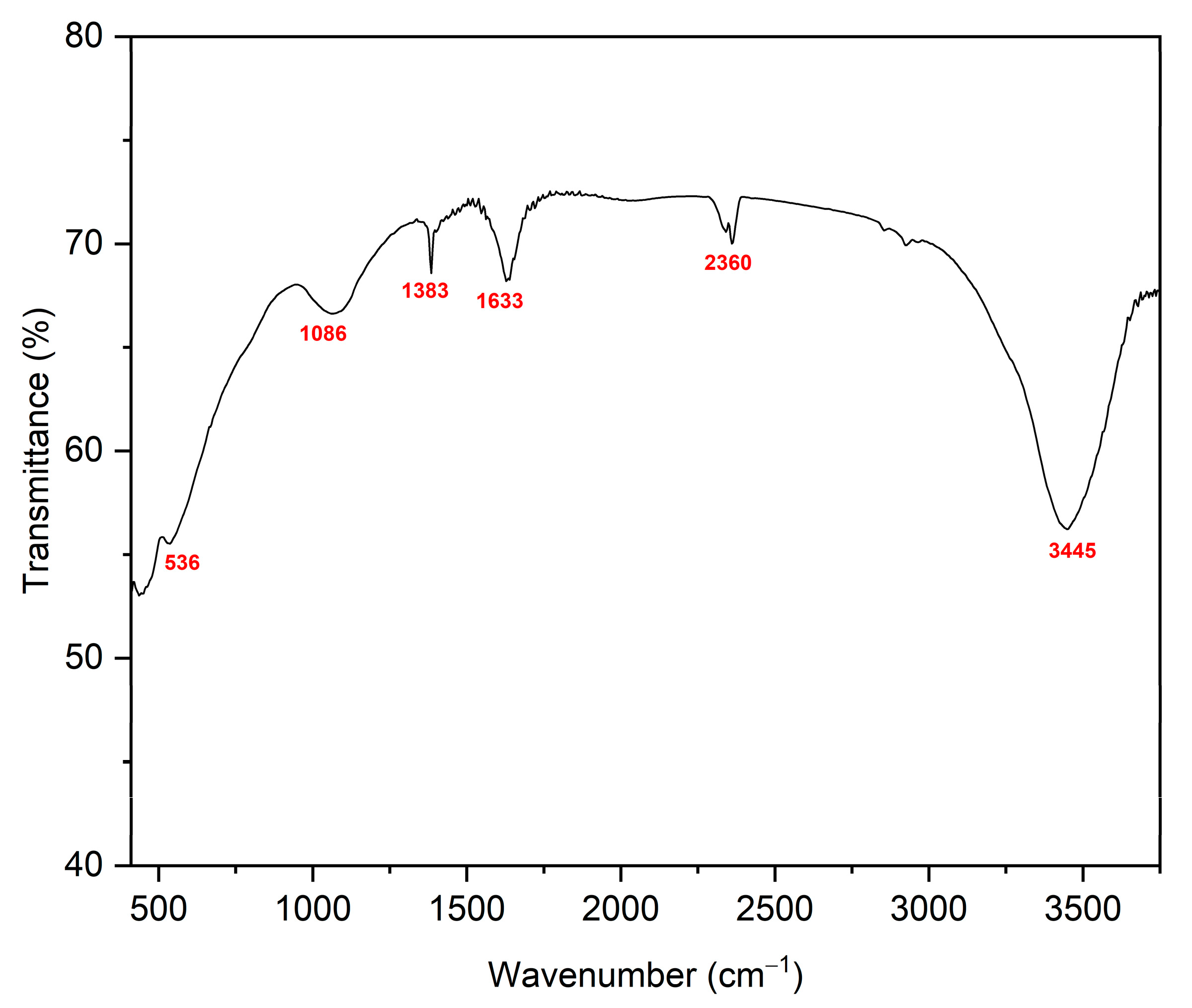
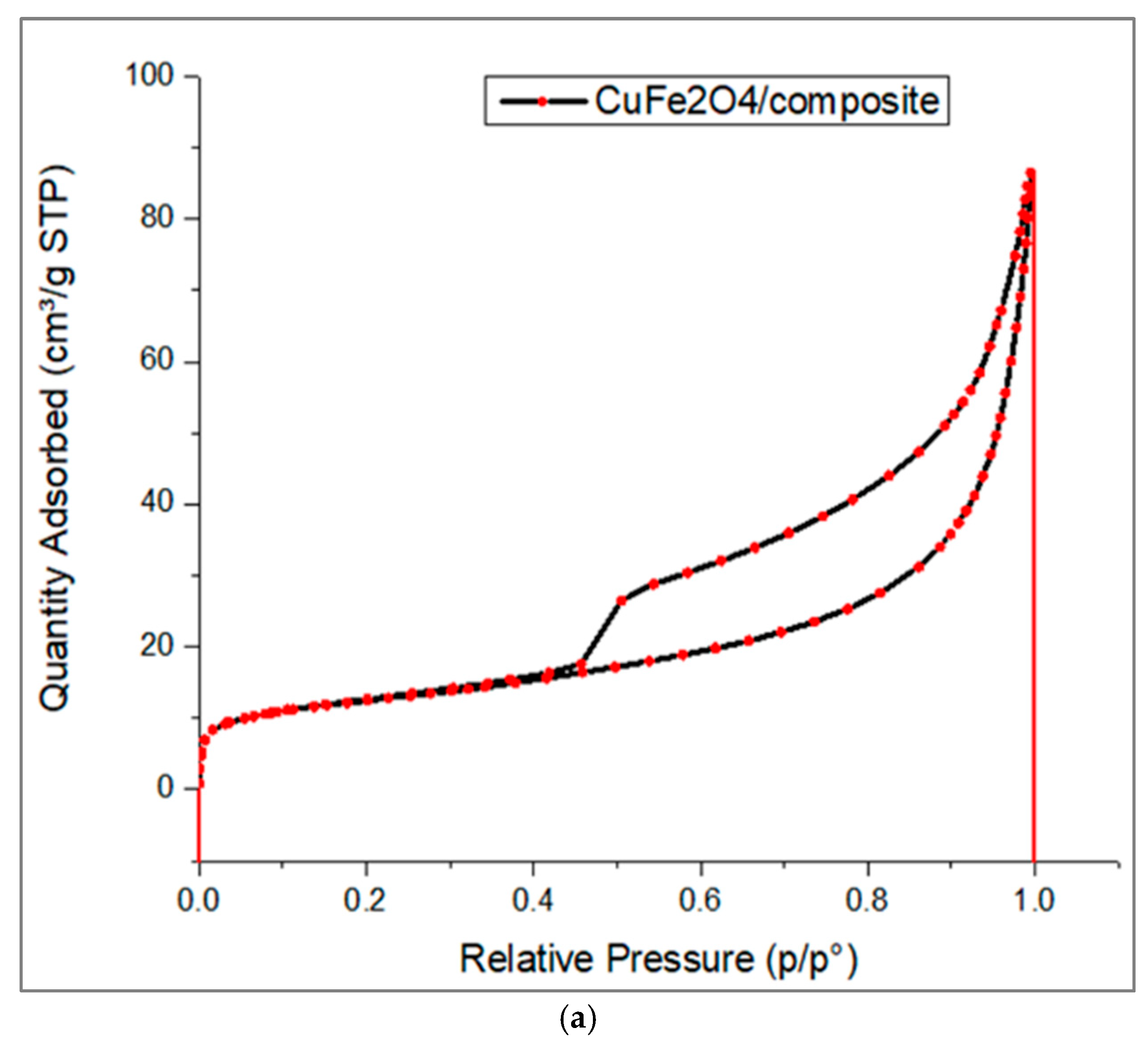
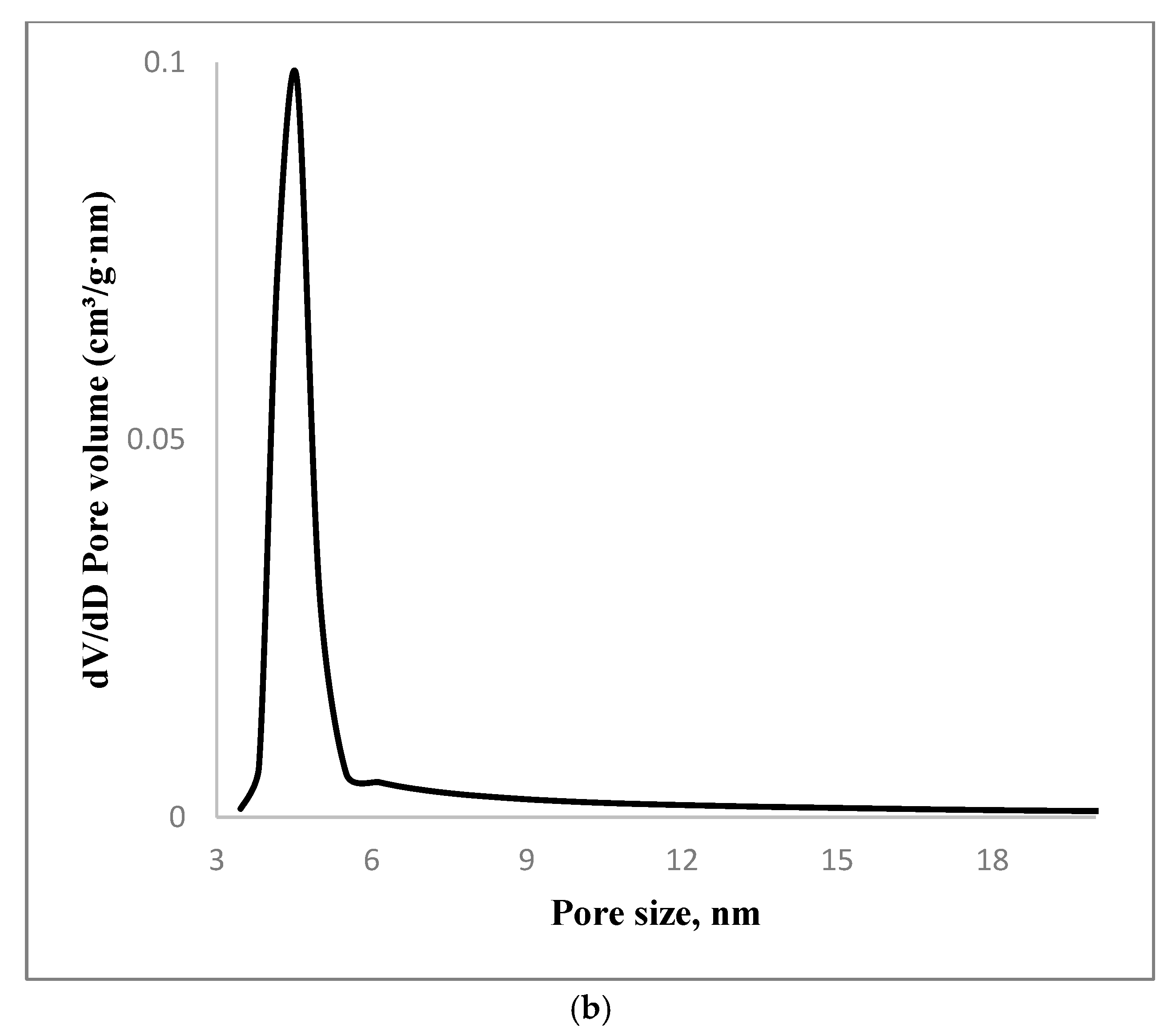
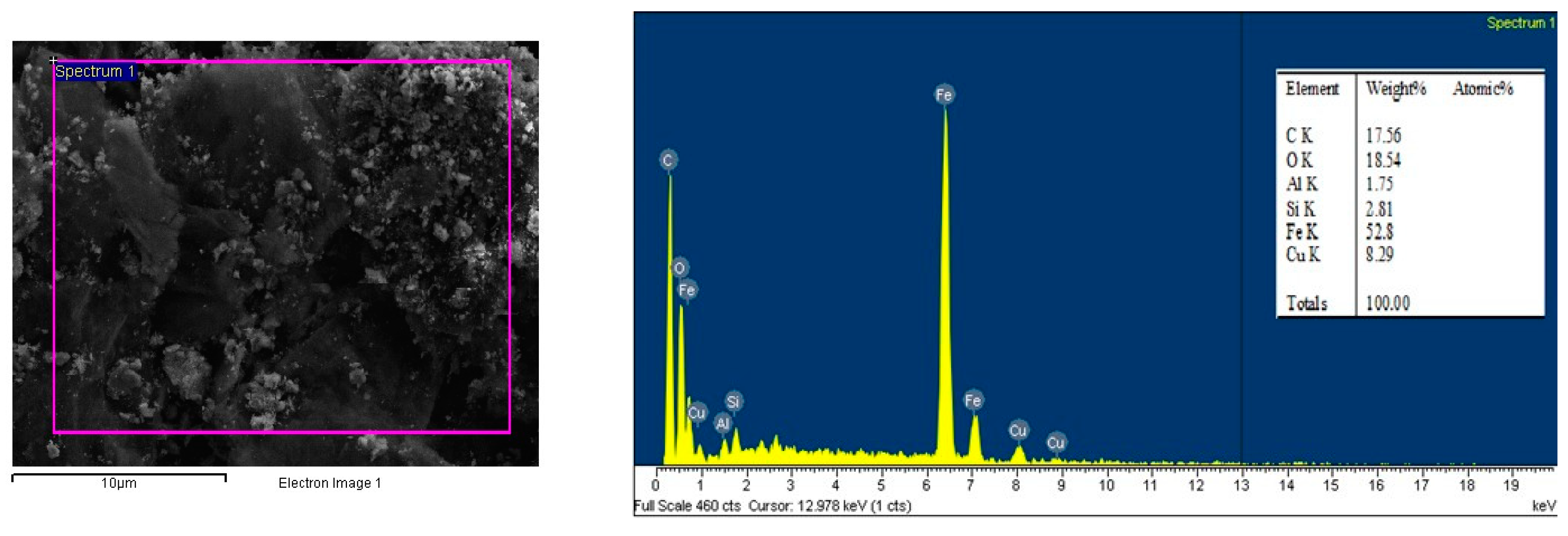
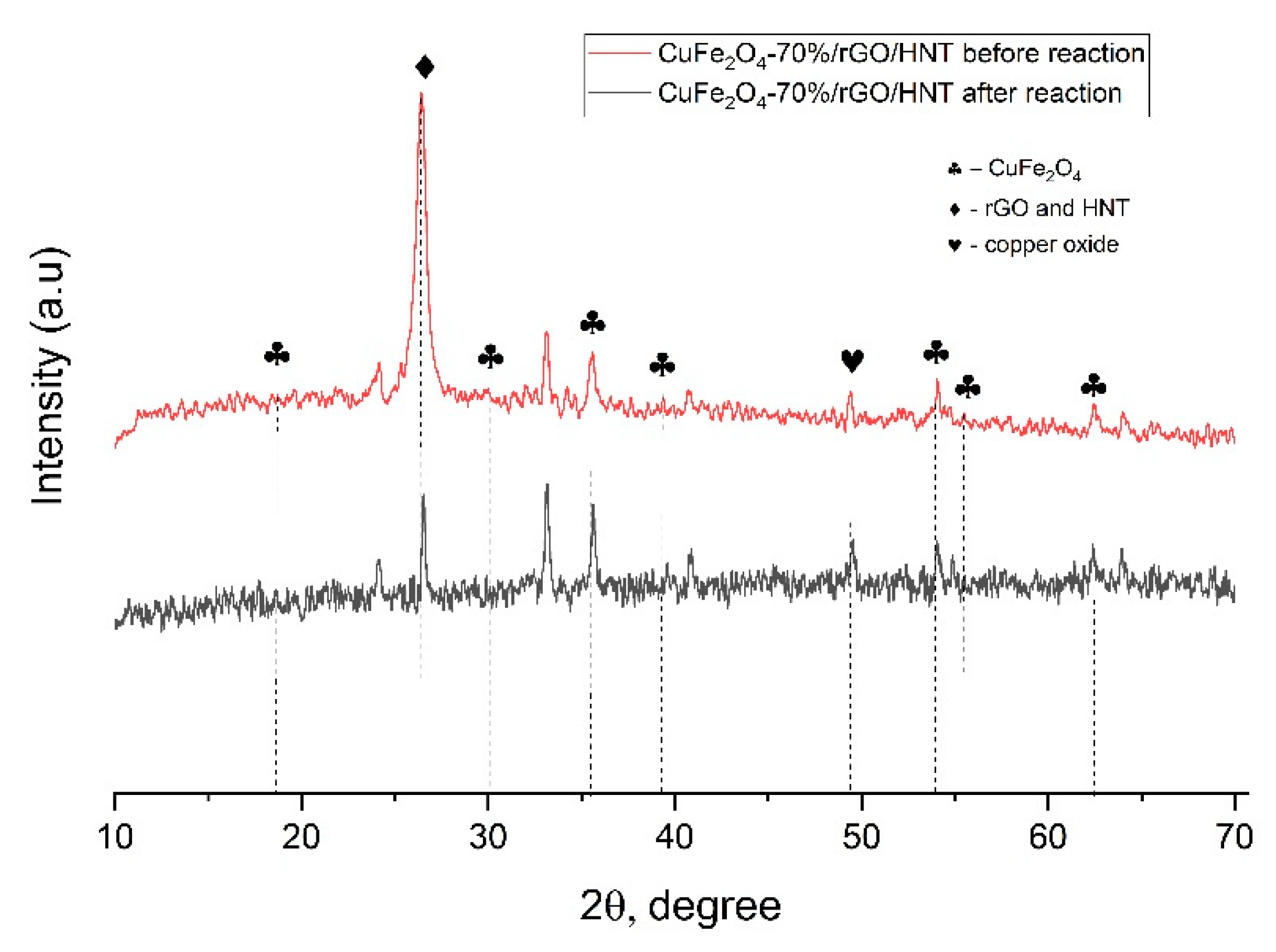
3.1. Material Characterizations
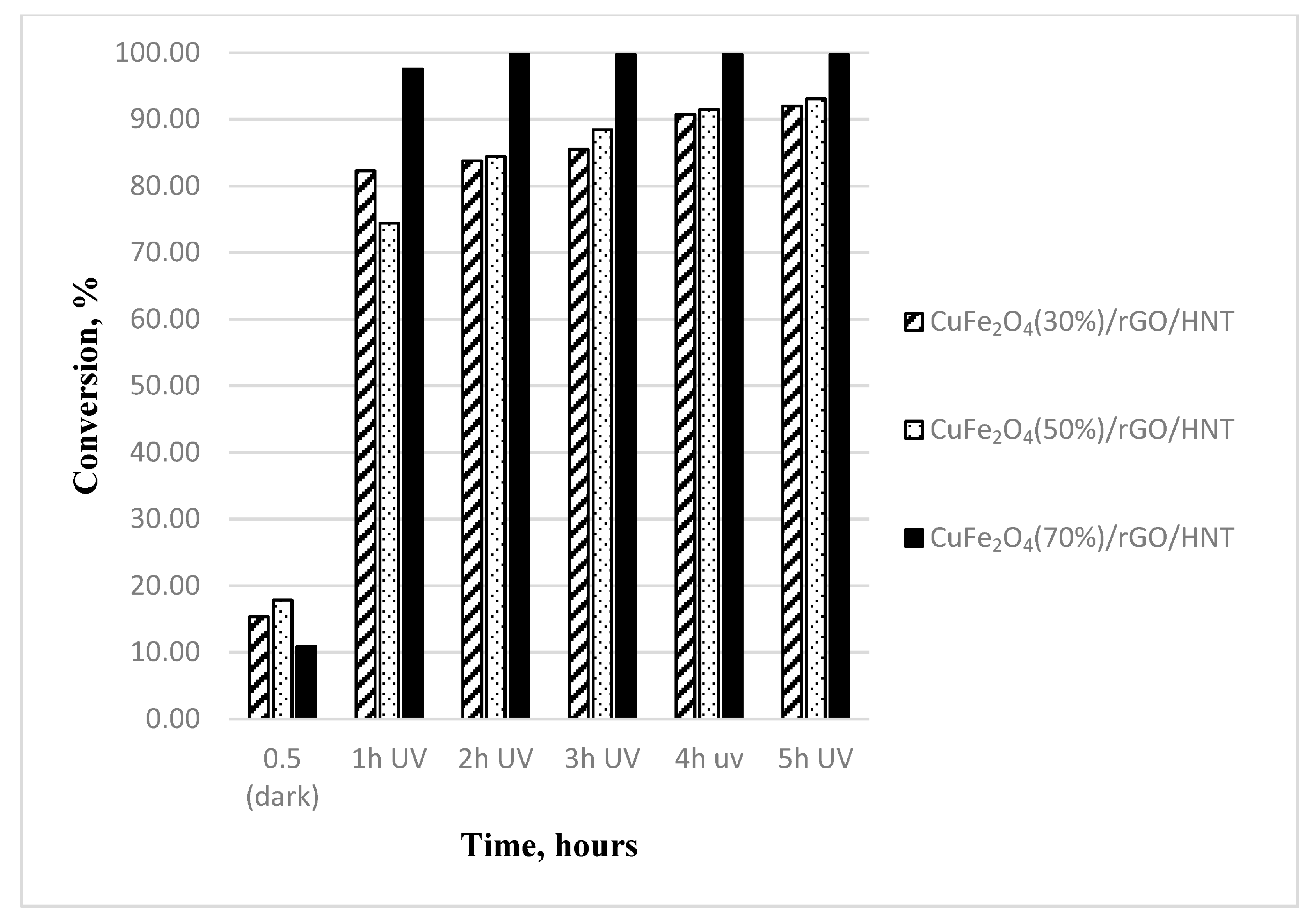
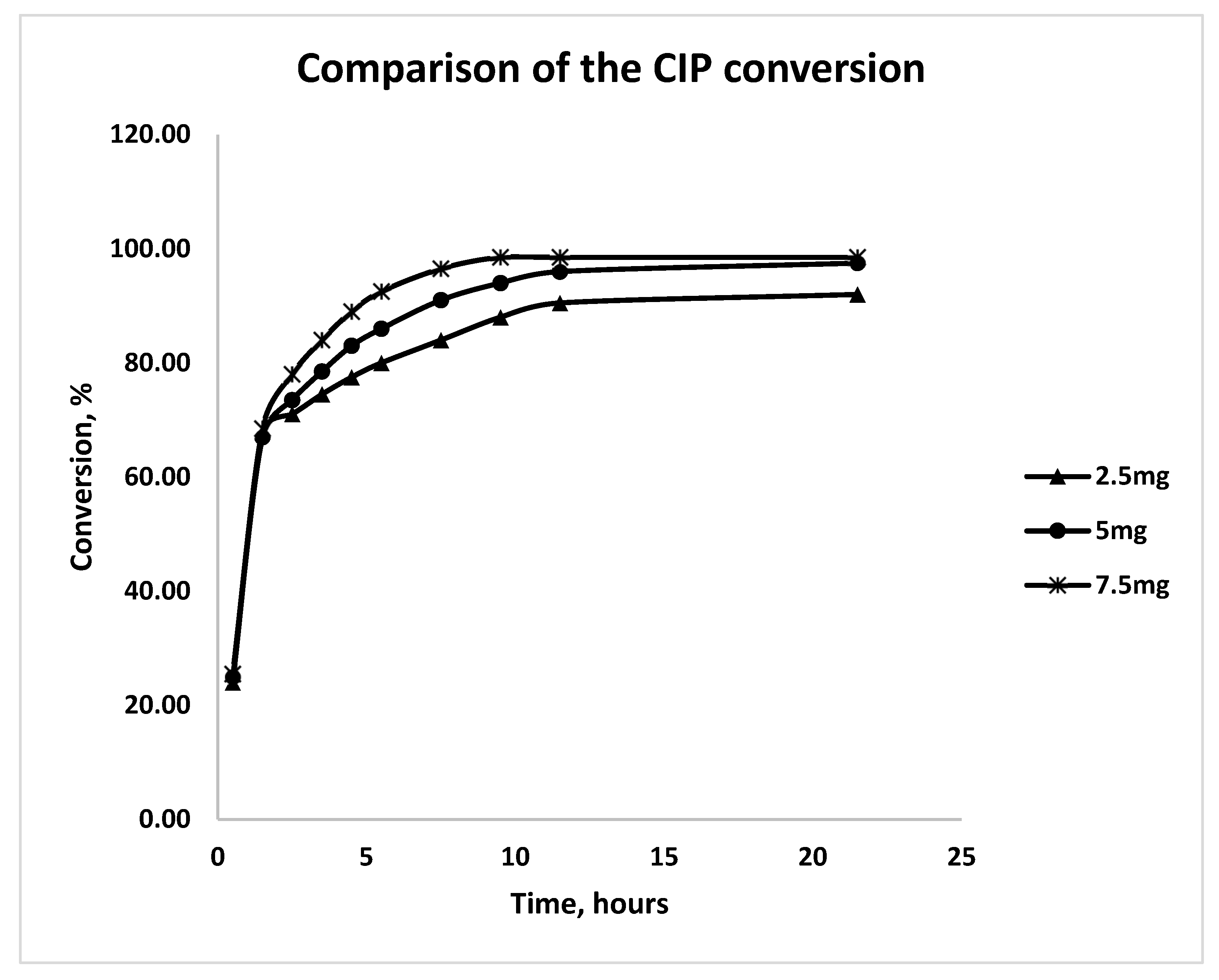
3.2. Impact of Active-Phase Loading
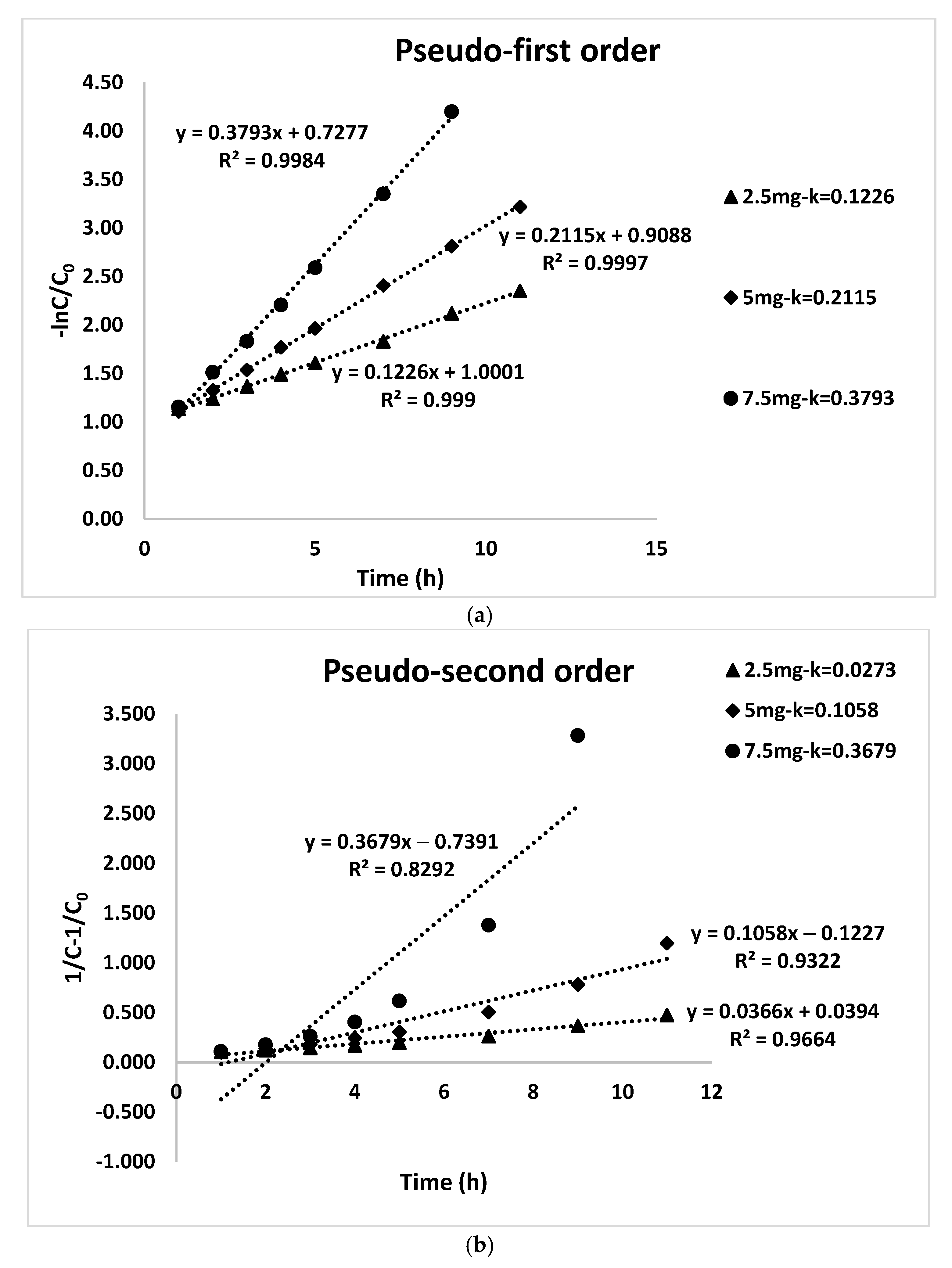
3.3. Photodegradation Kinetics of CIP
3.4. Impact of Solution pH
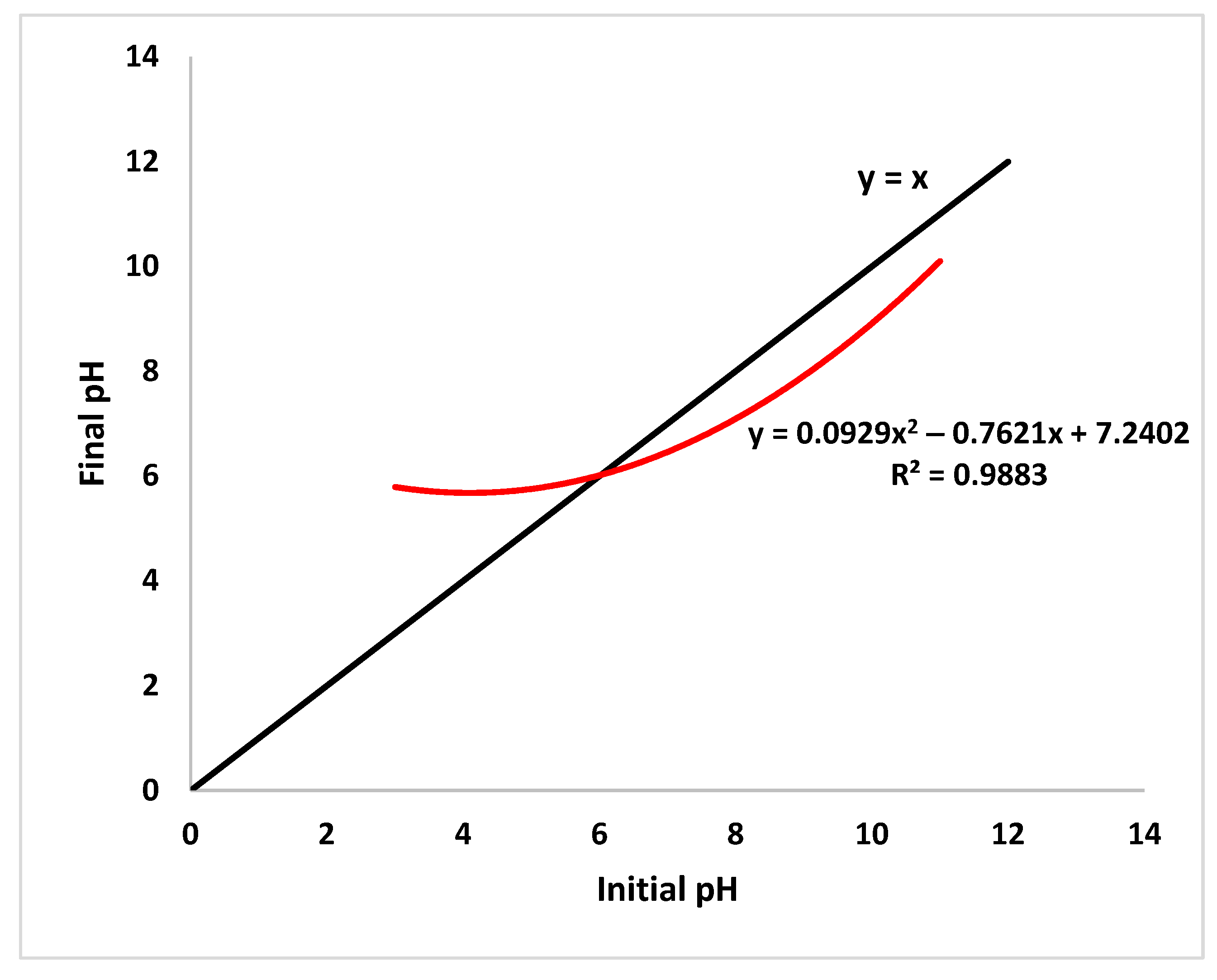
3.4.1. CuFe2O4/rGO/HNT Point of Zero Charge (pHpzc) Determination
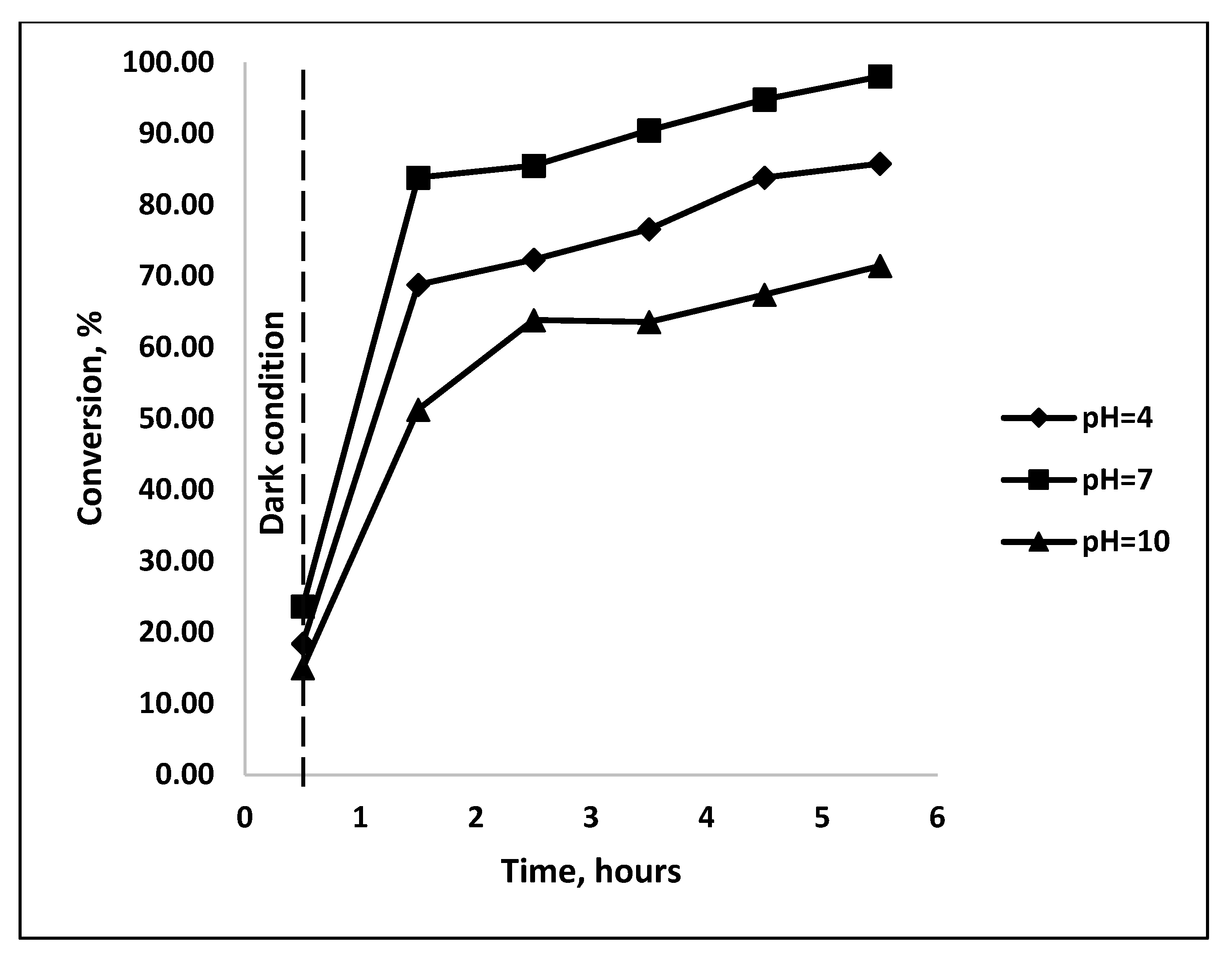
3.4.2. Influence of pH
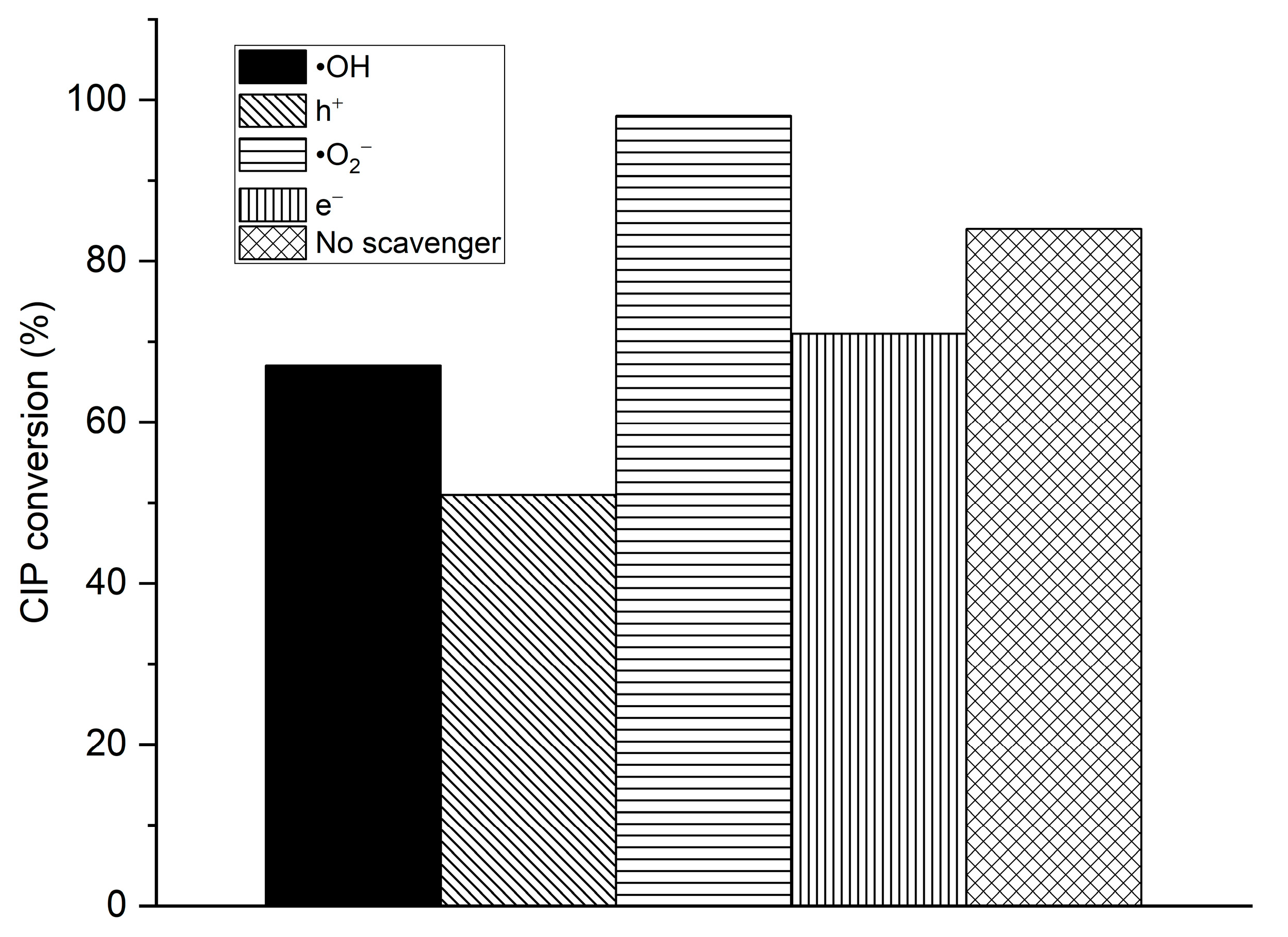
3.5. The Scavengers Test
3.6. The CIP Photodegradation Mechanism with CuFe2O4/rGO/HNT
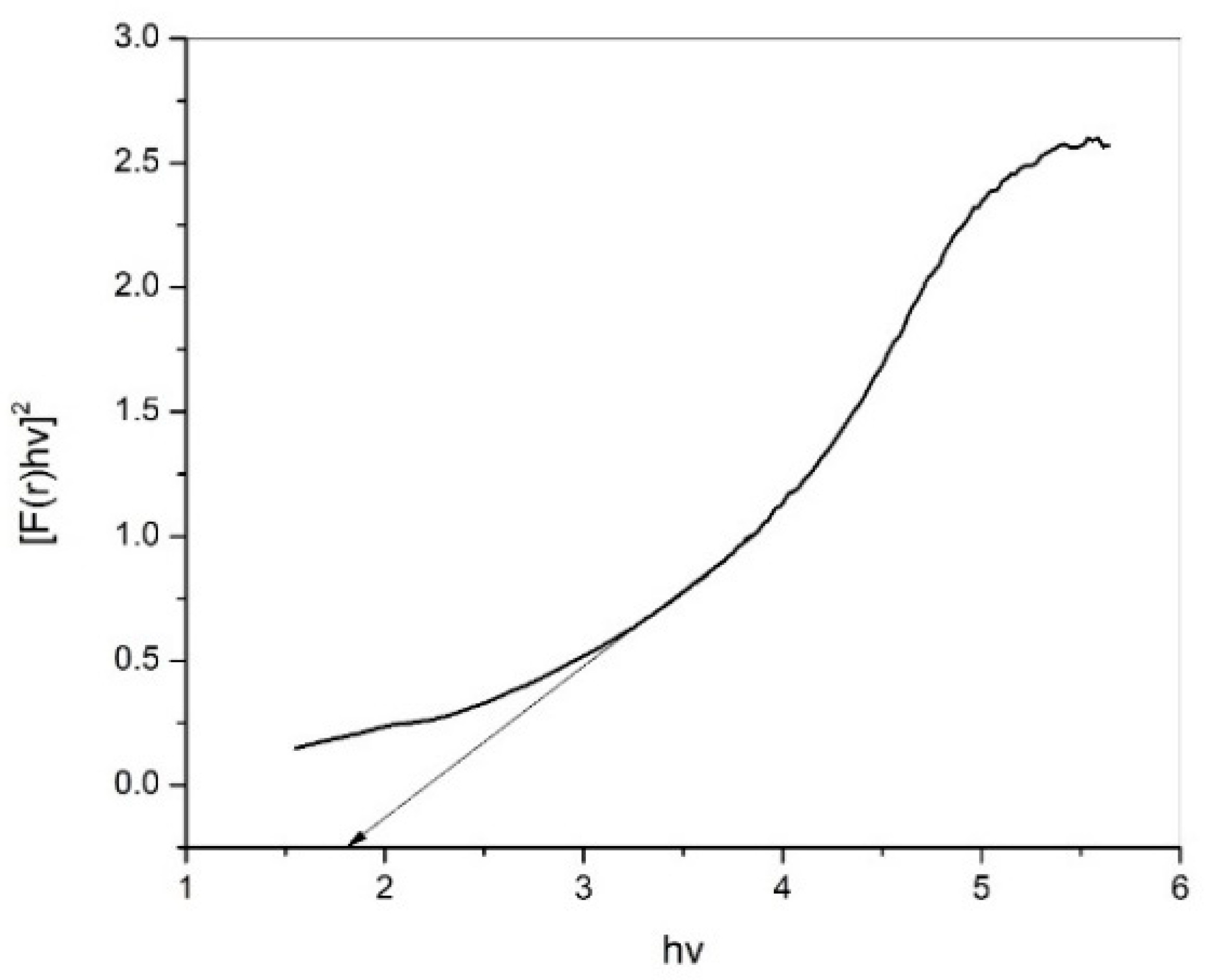
3.6.1. Calculation of the Conduction Band and Valence Band Energy (ECB, EVB)
3.6.2. The CIP Photochemical Degradation Mechanism with CuFe2O4/rGO/HNT
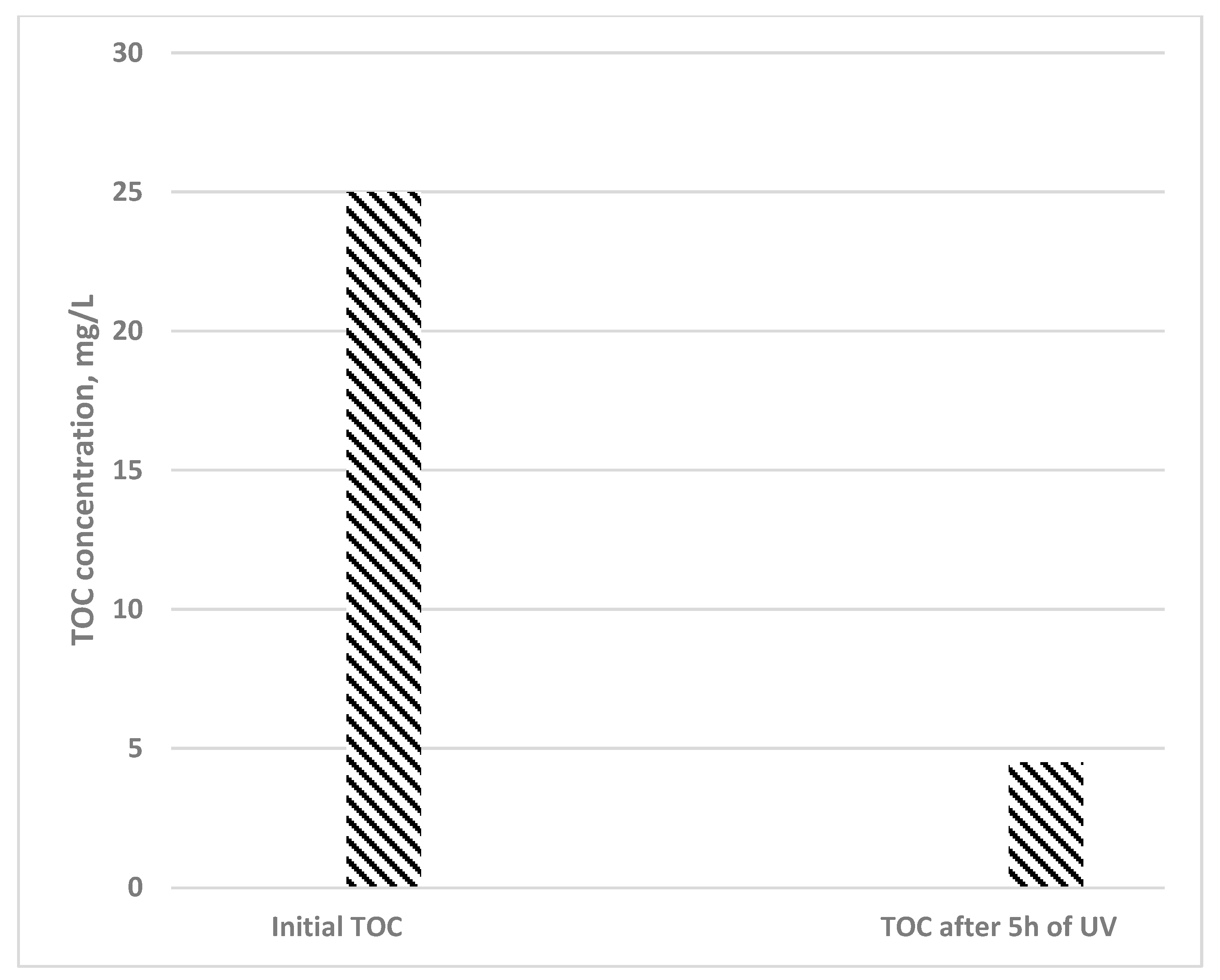
3.6.3. Total Organic Carbon Measurement
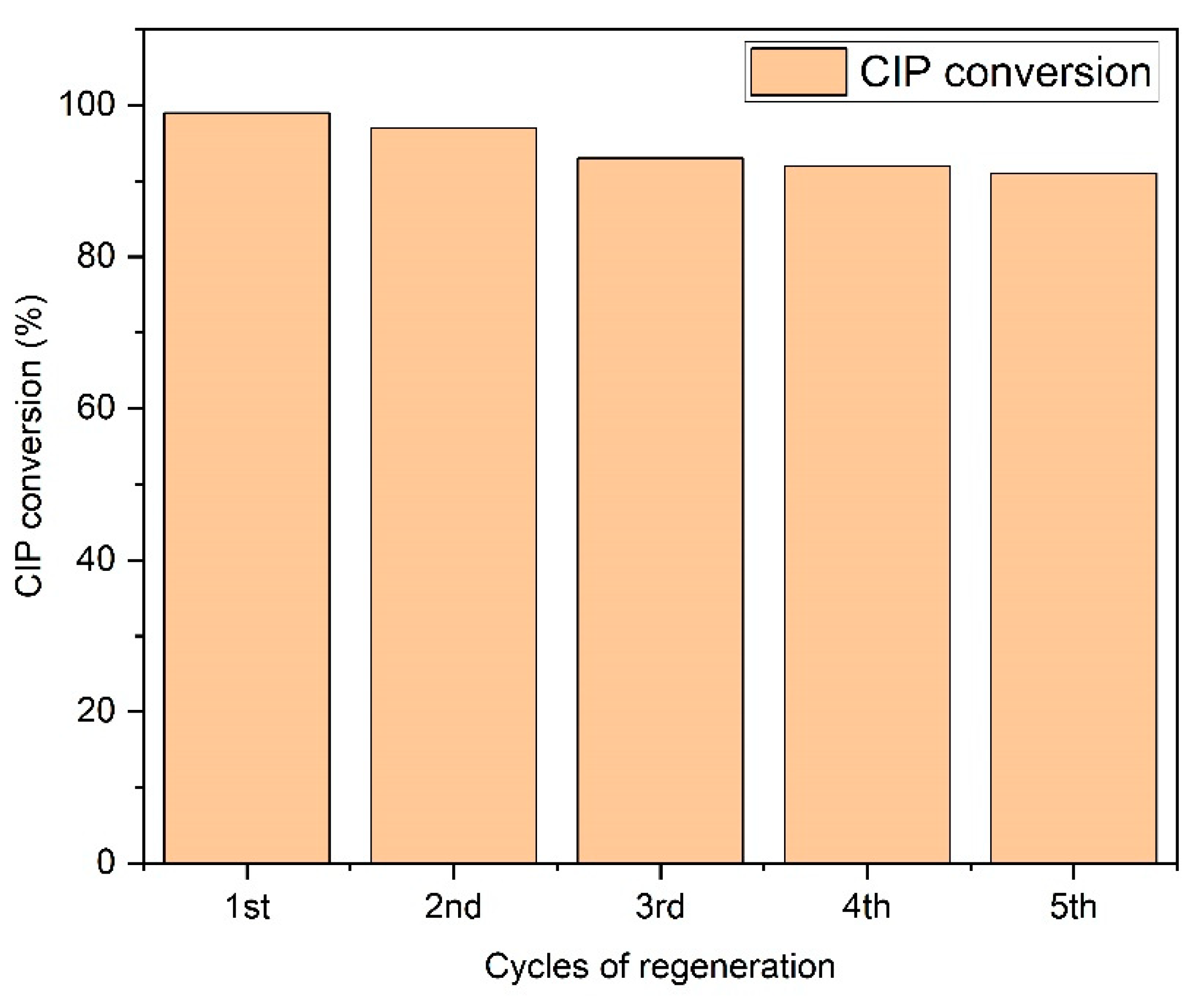
3.7. Reusability of the Photocatalyst
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Bilal, M.; Mehmood, S.; Rasheed, T.; Iqbal, H.M.N. Antibiotics Traces in the Aquatic Environment: Persistence and Adverse Environmental Impact. Curr. Opin. Environ. Sci. Health 2020, 13, 68–74. [Google Scholar] [CrossRef]
- Okeke, E.S.; Chukwudozie, K.I.; Nyaruaba, R.; Ita, R.E.; Oladipo, A.; Ejeromedoghene, O.; Atakpa, E.O.; Agu, C.V.; Okoye, C.O. Antibiotic Resistance in Aquaculture and Aquatic Organisms: A Review of Current Nanotechnology Applications for Sustainable Management. Environ. Sci. Pollut. Res. 2022, 29, 69241–69274. [Google Scholar] [CrossRef]
- Carvalho, I.T.; Santos, L. Antibiotics in the Aquatic Environments: A Review of the European Scenario. Environ. Int. 2016, 94, 736–757. [Google Scholar] [CrossRef] [PubMed]
- Zhang, S.; Huang, J.; Zhao, Z.; Cao, Y.; Li, B. Hospital Wastewater as a Reservoir for Antibiotic Resistance Genes: A Meta-Analysis. Front. Public Health 2020, 8, 574968. [Google Scholar] [CrossRef] [PubMed]
- Rodriguez-Mozaz, S.; Vaz-Moreira, I.; Varela Della Giustina, S.; Llorca, M.; Barceló, D.; Schubert, S.; Berendonk, T.U.; Michael-Kordatou, I.; Fatta-Kassinos, D.; Martinez, J.L.; et al. Antibiotic Residues in Final Effluents of European Wastewater Treatment Plants and Their Impact on the Aquatic Environment. Environ. Int. 2020, 140, 105733. [Google Scholar] [CrossRef]
- Akhil, D.; Lakshmi, D.; Senthil Kumar, P.; Vo, D.-V.N.; Kartik, A. Occurrence and Removal of Antibiotics from Industrial Wastewater. Environ. Chem. Lett. 2021, 19, 1477–1507. [Google Scholar] [CrossRef]
- Igwegbe, C.A.; Oba, S.N.; Aniagor, C.O.; Adeniyi, A.G.; Ighalo, J.O. Adsorption of Ciprofloxacin from Water: A Comprehensive Review. J. Ind. Eng. Chem. 2021, 93, 57–77. [Google Scholar] [CrossRef]
- Yuan, C.; Tebes-Stevens, C.; Weber, E.J. Reaction Library to Predict Direct Photochemical Transformation Products of Environmental Organic Contaminants in Sunlit Aquatic Systems. Environ. Sci. Technol. 2020, 54, 7271–7279. [Google Scholar] [CrossRef]
- Paul, T.; Dodd, M.C.; Strathmann, T.J. Photolytic and Photocatalytic Decomposition of Aqueous Ciprofloxacin: Transformation Products and Residual Antibacterial Activity. Water Res. 2010, 44, 3121–3132. [Google Scholar] [CrossRef]
- Guo, H.-G.; Gao, N.-Y.; Chu, W.-H.; Li, L.; Zhang, Y.-J.; Gu, J.-S.; Gu, Y.-L. Photochemical Degradation of Ciprofloxacin in UV and UV/H2O2 Process: Kinetics, Parameters, and Products. Environ. Sci. Pollut. Res. 2013, 20, 3202–3213. [Google Scholar] [CrossRef]
- Nakata, K.; Fujishima, A. TiO2 Photocatalysis: Design and Applications. J. Photochem. Photobiol. C Photochem. Rev. 2012, 13, 169–189. [Google Scholar] [CrossRef]
- Malato, S.; Fernández-Ibáñez, P.; Maldonado, M.I.; Blanco, J.; Gernjak, W. Decontamination and Disinfection of Water by Solar Photocatalysis: Recent Overview and Trends. Catal. Today 2009, 147, 1–59. [Google Scholar] [CrossRef]
- El-Kemary, M.; El-Shamy, H.; El-Mehasseb, I. Photocatalytic Degradation of Ciprofloxacin Drug in Water Using ZnO Nanoparticles. J. Lumin. 2010, 130, 2327–2331. [Google Scholar] [CrossRef]
- Machín, A.; Fontánez, K.; García, D.; Sampayo, P.; Colón-Cruz, C.; Claudio-Serrano, G.J.; Soto-Vázquez, L.; Resto, E.; Petrescu, F.I.; Morant, C.; et al. Hydrogen Production and Degradation of Ciprofloxacin by Ag@TiO2-MoS2 Photocatalysts. Catalysts 2022, 12, 267. [Google Scholar] [CrossRef]
- Beshkar, F.; Salavati-Niasari, M.; Amiri, O. Facile One-Pot In Situ Synthesis and Characterization of a Cu2O/Cu2(PO4)(OH) Binary Heterojunction Nanocomposite for the Efficient Photocatalytic Degradation of Ciprofloxacin from Aqueous Solution under Direct Sunlight Irradiation. Ind. Eng. Chem. Res. 2021, 60, 9578–9591. [Google Scholar] [CrossRef]
- Nhi, L.T.T.; Thuan, L.V.; Uyen, D.M.; Nguyen, M.H.; Thu, V.T.; Khieu, D.Q.; Sinh, L.H. Facile Fabrication of Highly Flexible and Floatable Cu2O/RGO on Vietnamese Traditional Paper toward High-Performance Solar-Light-Driven Photocatalytic Degradation of Ciprofloxacin Antibiotic. RSC Adv. 2020, 10, 16330–16338. [Google Scholar] [CrossRef]
- Noroozi, R.; Gholami, M.; Farzadkia, M.; Jonidi Jafari, A. Degradation of Ciprofloxacin by CuFe2O4/GO Activated PMS Process in Aqueous Solution: Performance, Mechanism and Degradation Pathway. Int. J. Environ. Anal. Chem. 2022, 102, 174–195. [Google Scholar] [CrossRef]
- Tamaddon, F.; Nasiri, A.; Yazdanpanah, G. Photocatalytic Degradation of Ciprofloxacin Using CuFe2O4@methyl Cellulose Based Magnetic Nanobiocomposite. MethodsX 2019, 7, 74–81. [Google Scholar] [CrossRef]
- A Critical Review on Surface-Modified Nano-Catalyst Application for the Photocatalytic Degradation of Volatile Organic Compounds—Environmental Science: Nano (RSC Publishing). Available online: https://pubs.rsc.org/en/content/articlelanding/2022/en/d1en00955a (accessed on 29 March 2023).
- Gupta, J.; Hassan, P.A.; Barick, K.C. 12—Defects in Nanomaterials for Visible Light Photocatalysis. In Nanostructured Materials for Visible Light Photocatalysis; Nayak, A.K., Sahu, N.K., Eds.; Micro and Nano Technologies; Elsevier: Amsterdam, The Netherlands, 2022; pp. 319–350. ISBN 978-0-12-823018-3. [Google Scholar]
- Zhang, Y.; He, X.; Ouyang, J.; Yang, H. Palladium Nanoparticles Deposited on Silanized Halloysite Nanotubes: Synthesis, Characterization and Enhanced Catalytic Property. Sci. Rep. 2013, 3, 2948. [Google Scholar] [CrossRef]
- Li, X.; Yao, C.; Lu, X.; Hu, Z.; Yin, Y.; Ni, C. Halloysite–CeO2–AgBr Nanocomposite for Solar Light Photodegradation of Methyl Orange. Appl. Clay Sci. 2015, 104, 74–80. [Google Scholar] [CrossRef]
- Li, X.; Zhu, W.; Yan, X.; Lu, X.; Yao, C.; Ni, C. Hierarchical La0.7Ce0.3FeO3/Halloysite Nanocomposite for Photocatalytic Degradation of Antibiotics. Appl. Phys. A 2016, 122, 723. [Google Scholar] [CrossRef]
- Dayana Priyadharshini, S.; Manikandan, S.; Kiruthiga, R.; Rednam, U.; Babu, P.S.; Subbaiya, R.; Karmegam, N.; Kim, W.; Govarthanan, M. Graphene Oxide-Based Nanomaterials for the Treatment of Pollutants in the Aquatic Environment: Recent Trends and Perspectives—A Review. Environ. Pollut. 2022, 306, 119377. [Google Scholar] [CrossRef] [PubMed]
- Liu, S.; Sun, H.; Suvorova, A.; Wang, S. One-Pot Hydrothermal Synthesis of ZnO-Reduced Graphene Oxide Composites Using Zn Powders for Enhanced Photocatalysis. Chem. Eng. J. 2013, 229, 533–539. [Google Scholar] [CrossRef]
- Zhang, J.; Xiong, Z.; Zhao, X.S. Graphene–Metal–Oxide Composites for the Degradation of Dyes under Visible Light Irradiation. J. Mater. Chem. 2011, 21, 3634–3640. [Google Scholar] [CrossRef]
- Liu, Y.; Jiang, X.; Li, B.; Zhang, X.; Liu, T.; Yan, X.; Ding, J.; Cai, Q.; Zhang, J. Halloysite Nanotubes@reduced Graphene Oxide Composite for Removal of Dyes from Water and as Supercapacitors. J. Mater. Chem. A 2014, 2, 4264–4269. [Google Scholar] [CrossRef]
- Zeng, G.; He, Y.; Ye, Z.; Yang, X.; Chen, X.; Ma, J.; Li, F. Novel Halloysite Nanotubes Intercalated Graphene Oxide Based Composite Membranes for Multifunctional Applications: Oil/Water Separation and Dyes Removal. Ind. Eng. Chem. Res. 2017, 56, 10472–10481. [Google Scholar] [CrossRef]
- Xiao, J.; Xie, S.; Jing, Y.; Yao, Y.; Wang, X.; Jia, Y. Preparation of Halloysite@graphene Oxide Composite and Its Application for High-Efficient Decontamination of U(VI) from Aqueous Solution. J. Mol. Liq. 2016, 220, 304–310. [Google Scholar] [CrossRef]
- Yu, L.; Zhang, Y.; Zhang, B.; Liu, J. Enhanced Antibacterial Activity of Silver Nanoparticles/Halloysite Nanotubes/Graphene Nanocomposites with Sandwich-Like Structure. Sci. Rep. 2014, 4, 4551. [Google Scholar] [CrossRef]
- Ngo, H.-S.; Nguyen, T.-L.; Tran, N.-T.; Le, H.-C. Experimental Study on Kinetics and Mechanism of Ciprofloxacin Degradation in Aqueous Phase Using Ag-TiO2/RGO/Halloysite Photocatalyst. Catalysts 2023, 13, 225. [Google Scholar] [CrossRef]
- Dadban Shahamat, Y.; Sadeghi, M.; Shahryari, A.; Okhovat, N.; Bahrami Asl, F.; Baneshi, M.M. Heterogeneous Catalytic Ozonation of 2, 4-Dinitrophenol in Aqueous Solution by Magnetic Carbonaceous Nanocomposite: Catalytic Activity and Mechanism. Desalination Water Treat. 2016, 57, 20447–20456. [Google Scholar] [CrossRef]
- Dhanda, R.; Kidwai, M. Magnetically Separable CuFe2O4/Reduced Graphene Oxide Nanocomposites: As a Highly Active Catalyst for Solvent Free Oxidative Coupling of Amines to Imines. RSC Adv. 2016, 6, 53430–53437. [Google Scholar] [CrossRef]
- Ali, K.; Bahadur, A.; Jabbar, A.; Iqbal, S.; Ahmad, I.; Bashir, M.I. Synthesis, Structural, Dielectric and Magnetic Properties of CuFe2O4/MnO2 Nanocomposites. J. Magn. Magn. Mater. 2017, 434, 30–36. [Google Scholar] [CrossRef]
- Mi, X.; Zhong, L.; Wei, F.; Zeng, L.; Zhang, J.; Zhang, D.; Xu, T. Fabrication of Halloysite Nanotubes/Reduced Graphene Oxide Hybrids for Epoxy Composites with Improved Thermal and Mechanical Properties. Polym. Test. 2019, 76, 473–480. [Google Scholar] [CrossRef]
- Ding, G.; Chen, C.; Tai, H.; Tang, Z.; Wang, Z.; Cheng, G.; Wan, X. Structural Characterization and Microwave Absorbing Performance of CuFe2O4/RGO Composites. J. Solid State Chem. 2021, 297, 122051. [Google Scholar] [CrossRef]
- Gao, C.; Li, B.; Chen, N.; Ding, J.; Cai, Q.; Zhang, J.; Liu, Y. Novel Fe3O4/HNT@rGO Composite via a Facile Co-Precipitation Method for the Removal of Contaminants from Aqueous System. RSC Adv. 2016, 6, 49228–49235. [Google Scholar] [CrossRef]
- Muthu, R.N.; Rajashabala, S.; Kannan, R. Facile Synthesis and Characterization of a Reduced Graphene Oxide/Halloysite Nanotubes/Hexagonal Boron Nitride (RGO/HNT/h-BN) Hybrid Nanocomposite and Its Potential Application in Hydrogen Storage. RSC Adv. 2016, 6, 79072–79084. [Google Scholar] [CrossRef]
- Dey, C.; De, D.; Nandi, M.; Goswami, M.M. A High Performance Recyclable Magnetic CuFe2O4 Nanocatalyst for Facile Reduction of 4-Nitrophenol. Mater. Chem. Phys. 2020, 242, 122237. [Google Scholar] [CrossRef]
- Zhang, X.; Feng, M.; Qu, R.; Liu, H.; Wang, L.; Wang, Z. Catalytic Degradation of Diethyl Phthalate in Aqueous Solution by Persulfate Activated with Nano-Scaled Magnetic CuFe2O4/MWCNTs. Chem. Eng. J. 2016, 301, 1–11. [Google Scholar] [CrossRef]
- Song, S.Y.; Chen, H.D.; Li, C.X.; Shi, D.S.; Ying, Y.; Han, Y.B.; Xu, J.C.; Hong, B.; Jin, H.X.; Jin, D.F.; et al. Magnetic Bi2WO6 Nanocomposites: Synthesis, Magnetic Response and Their Visible-Light-Driven Photocatalytic Performance for Ciprofloxacin. Chem. Phys. 2020, 530, 110614. [Google Scholar] [CrossRef]
- Li, C.; Chen, G.; Sun, J.; Rao, J.; Han, Z.; Hu, Y.; Xing, W.; Zhang, C. Doping Effect of Phosphate in Bi2WO6 and Universal Improved Photocatalytic Activity for Removing Various Pollutants in Water. Appl. Catal. B Environ. 2016, 188, 39–47. [Google Scholar] [CrossRef]
- Kočí, K.; Obalová, L.; Matějová, L.; Plachá, D.; Lacný, Z.; Jirkovský, J.; Šolcová, O. Effect of TiO2 Particle Size on the Photocatalytic Reduction of CO2. Appl. Catal. B Environ. 2009, 89, 494–502. [Google Scholar] [CrossRef]
- Chakrabarti, S.; Dutta, B.K. Photocatalytic Degradation of Model Textile Dyes in Wastewater Using ZnO as Semiconductor Catalyst. J. Hazard. Mater. 2004, 112, 269–278. [Google Scholar] [CrossRef] [PubMed]
- Zhang, D.; Lv, S.; Luo, Z. A Study on the Photocatalytic Degradation Performance of a [KNbO3]0.9-[BaNi0.5Nb0.5O3−δ]0.1 Perovskite. RSC Adv. 2020, 10, 1275–1280. [Google Scholar] [CrossRef] [PubMed]
- Das, S.; Ghosh, S.; Misra, A.J.; Tamhankar, A.J.; Mishra, A.; Lundborg, C.S.; Tripathy, S.K. Sunlight Assisted Photocatalytic Degradation of Ciprofloxacin in Water Using Fe Doped ZnO Nanoparticles for Potential Public Health Applications. Int. J. Environ. Res. Public Health 2018, 15, 2440. [Google Scholar] [CrossRef] [PubMed]
- Nasiri, A.; Tamaddon, F.; Mosslemin, M.H.; Amiri Gharaghani, M.; Asadipour, A. Magnetic Nano-Biocomposite CuFe2O4@methylcellulose (MC) Prepared as a New Nano-Photocatalyst for Degradation of Ciprofloxacin from Aqueous Solution. Environ. Health Eng. Manag. J. 2019, 6, 41–51. [Google Scholar] [CrossRef]
- Anjana Anand, A.S.; Adish Kumar, S.; Rajesh Banu, J.; Ginni, G. The Performance of Fluidized Bed Solar Photo Fenton Oxidation in the Removal of COD from Hospital Wastewaters. Desalination Water Treat. 2016, 57, 8236–8242. [Google Scholar] [CrossRef]
- Zhu, M.; Lu, J.; Hu, Y.; Liu, Y.; Hu, S.; Zhu, C. Photochemical Reactions between 1,4-Benzoquinone and O2•. Environ. Sci. Pollut. Res. Int. 2020, 27, 31289–31299. [Google Scholar] [CrossRef]
- Mejia-Bernal, J.R.; Mumanga, T.J.; Diaz-Torres, L.A.; Vallejo-Hernández, M.Á.; Gómez -Solís, C. Synthesis and Evaluation of MSiO3 (M = Ba, Sr, Mg) for Photocatalytic Hydrogen Generation under UV Irradiation. Mater. Lett. 2021, 295, 129851. [Google Scholar] [CrossRef]
- Koppenol, W.H.; Stanbury, D.M.; Bounds, P.L. Electrode Potentials of Partially Reduced Oxygen Species, from Dioxygen to Water. Free Radic. Biol. Med. 2010, 49, 317–322. [Google Scholar] [CrossRef]
- Monteoliva-García, A.; Martín-Pascual, J.; Muñío, M.M.; Poyatos, J.M. Removal of Carbamazepine, Ciprofloxacin and Ibuprofen in Real Urban Wastewater by Using Light-Driven Advanced Oxidation Processes. Int. J. Environ. Sci. Technol. 2019, 16, 6005–6018. [Google Scholar] [CrossRef]
- Liu, X.; Zhang, G.; Liu, Y.; Lu, S.; Qin, P.; Guo, X.; Bi, B.; Wang, L.; Xi, B.; Wu, F.; et al. Occurrence and Fate of Antibiotics and Antibiotic Resistance Genes in Typical Urban Water of Beijing, China. Environ. Pollut. 2019, 246, 163–173. [Google Scholar] [CrossRef] [PubMed]


| Pseudo-First-Order Rate Law | Pseudo-Second-Order Rate Law | |||||
|---|---|---|---|---|---|---|
| Catalyst Weight | Linear Equation | k | R2 | Linear Equation | k | R2 |
| 2.5 mg | y = 0.1226x + 1.0001 | 0.1226 | 0.999 | y = 0.0366x + 0.0394 | 0.0366 | 0.9664 |
| 5 mg | y = 0.2115x + 0.9088 | 0.2115 | 0.9997 | y = 0.1058x − 0.1227 | 0.1058 | 0.9322 |
| 7.5 mg | y = 0.3793x + 0.7277 | 0.3793 | 0.9984 | y = 0.3679x − 0.7391 | 0.3679 | 0.8292 |
| Initial pH | 11 | 8.94 | 7.02 | 5 | 3 |
| Final PH | 10.1 | 7.94 | 6.16 | 6.02 | 5.7 |
| Cu (eV) | Fe (eV) | O (eV) | |
|---|---|---|---|
| Ionization energy | 7.726 | 7.902 | 13.618 |
| Electron affinity | 1.2271 | 0.1627 | 1.4613 |
| χ | 4.47655 | 4.0324 | 7.53965 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ngo, H.-S.; Nguyen, T.-L.; Tran, N.-T.; Le, H.-C. Photocatalytic Removal of Ciprofloxacin in Water by Novel Sandwich-like CuFe2O4 on rGO/Halloysite Material: Insights into Kinetics and Intermediate Reactive Radicals. Water 2023, 15, 1569. https://doi.org/10.3390/w15081569
Ngo H-S, Nguyen T-L, Tran N-T, Le H-C. Photocatalytic Removal of Ciprofloxacin in Water by Novel Sandwich-like CuFe2O4 on rGO/Halloysite Material: Insights into Kinetics and Intermediate Reactive Radicals. Water. 2023; 15(8):1569. https://doi.org/10.3390/w15081569
Chicago/Turabian StyleNgo, Ha-Son, Thi-Linh Nguyen, Ngoc-Tuan Tran, and Hanh-Chi Le. 2023. "Photocatalytic Removal of Ciprofloxacin in Water by Novel Sandwich-like CuFe2O4 on rGO/Halloysite Material: Insights into Kinetics and Intermediate Reactive Radicals" Water 15, no. 8: 1569. https://doi.org/10.3390/w15081569
APA StyleNgo, H.-S., Nguyen, T.-L., Tran, N.-T., & Le, H.-C. (2023). Photocatalytic Removal of Ciprofloxacin in Water by Novel Sandwich-like CuFe2O4 on rGO/Halloysite Material: Insights into Kinetics and Intermediate Reactive Radicals. Water, 15(8), 1569. https://doi.org/10.3390/w15081569







