Role of Hydrology in Cyanobacterial Blooms in the Floodplain Lakes
Abstract
1. Introduction
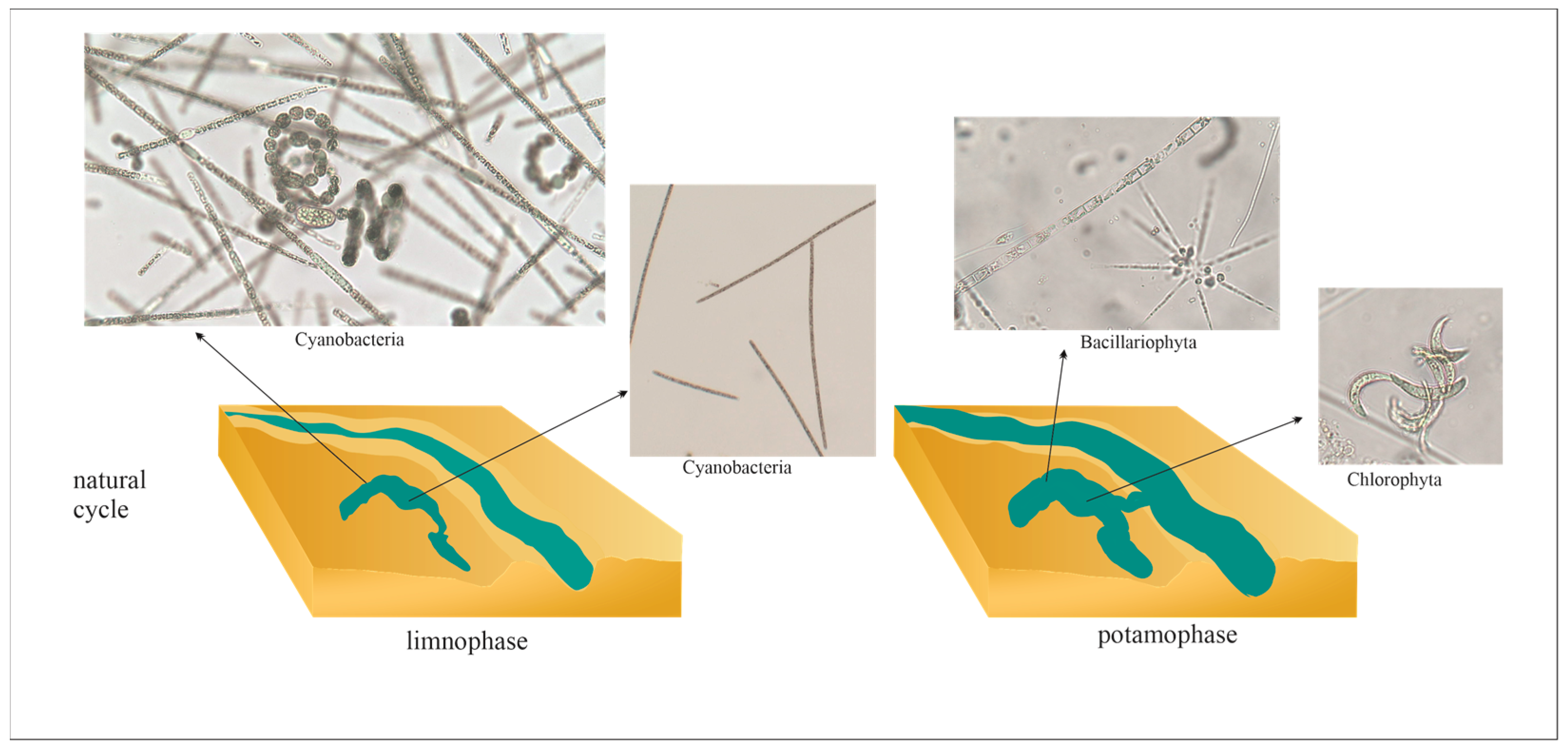
2. Hydrological Connectivity
3. Cyanobacterial Blooms in Floodplain Lakes
| Lakes Trophy, Names and Location, the Study Period | Cyanobacterial Taxa Creating Blooms | Maximal Cyanobacterial Abundance and/or Biomass, Share or Total Phytoplankton Biomass Expressed as the Concentration of Chl-a | Cyanotoxins and Their Concentrations | Factors That Favored Blooms | References |
|---|---|---|---|---|---|
| Eutrophic/hypertrophic Laguna Grande Lake, floodplain Ramsar Paraná de las Palmas and Luján Rivers (Argentina) 1998–2013 | Very high variability of cyanobacterial species from year to year: Planktothrix agardhii and Raphidiopsis mediterranea; Dolichospermum spp. and Anabaenopsis cf. elenkinii; A. cf. elenkinii and Sphaerospermopsis cf. aphanizonemoides accompanied by M. aeruginosa; M. aeruginosa, S. torques-reginae, D. cf. bituri, Cuspidotrix issatschenkoi, S. cf. aphanizonemoides, and R. mediterranea; M. aeruginosa and P. agardhii | 8.5 × 105 colonies/filaments/mL, 670 µg Chl-a/L | l.s. | Extremely low water levels: the effect of water depth, mainly on the acquisition of light and nitrogen. Extreme drought and flood events related to the El Niño Southern Oscillation, which is linked to discharge anomalies in the Paraná River | O’Farrell et al. [77], O’Farrell et al. [78]. |
| Eutrophic/hypertrophic Lake Sakadaš Kopački Rit Danube Floodplain (Croatia) 2003–2008 | Blooms beginning from the development of Nostocales (dominants Dolichospermum spp. and Aphanizomenon flos-aquae) and lasting one month. High variability in patterns of blooming taxa was the main feature of blooms that occurred in summer–autumn periods. C. raciborskii dominanted or co-dominanted in summer. P. agardhii, Pseudanabaena limnetica, L. redekei, Planktolyngbya limnetica was dominant in winter, summer, and autumn. Pseudanabaena limnetica co-occurred with P. agardhii. L. redekei, P. agardhii, and Ps. limnetica were co-dominants or dominants in winter bloom. Only occasionally Merismopedia punctata. | The highest cyanobacterial biomass 61.8 mg/L in the seasons without spring flooding (2003) in comparison with biomass of 10.8 mg/L after the brief spring flood (2007). The bloom absent under conditions of prolonged spring floods (till June 2004 and 2008). The highest biomass equal to 246.6 mg/L | l.s. | High-nutrient concentrations and water column stability, the turbid state during the long-term dry conditions without flooding. The flooding dynamic and intensity as the controlling factor for cyanobacterial blooms in the dynamic river-floodplain system. | Mihaljević, M., and Stević, F. [63]. Stević, F., et al. [54]. |
| Eutrophic/hypertrophic Lakes Dongting and Poyang connected with the river and Lakes Chao and Tai (Taihu) isolated from the river, Yangtze River floodplain (China) 2000–2010 | n.d. * Microcystis spp. | 16.8 µg of Chl-a/L in Poyang Lake 10.0 µg of Chl-a/L in Donting Lake 108 µg of Chl-a/L in Tai Lake 98 µg of Chl-a/L in Poyang Lake | * Dongting Lake: intracellular MC-RR and -LR concentrations were the highest in East Donting Lake * MC-producing Microcystis strains isolated from Lakes Chao and Tai * Lake Tai: First complex study: cyanotoxins predominated by MCs—the highest concentrations were found in November (mean 2.21 µg/L), whereas the lowest amounts were found in February (mean 0.049 µg/L). Cylindrospermopsins concentrations were the highest in May (max. concentration 0.623 µg/L). Saxitoxins only occurred in May (mean concentration 0.019 µg/L) and November (mean 0.199 µg/L). The amount of extracellular T&O was the highest in August—β-cyclocitral predominated (mean 0.241 µg/L) followed by 2-methylisoborneol (mean 0.147 µg/L). | Concentrations of Chl-a considerably higher when relative lake level fluctuations (RLLF) < 100, precipitation < 2.6 mm, wind speed > 2.6 m/s, air temperature > 17.8 °C; Chl-a concentration in river-isolated lakes was more significantly affected by water level amplitude, precipitation, wind speed, and air temperature than in the river-connected lakes | Wu et al. [6]. Huang, J., et al. [60]. Min, et al. [91]. Krüger, et al. [92]. Li, et al. [93] |
| Find floodplain eutrophic lakes including Lake Waaienstein and Lake Ewijk, lower Rhine, (The Netherlands) 1990–1991 | A. flos-aquae predomination in August (summer) only in Lake Waainestein and Lake Ewijk | 110 µg Chl-a/L in Lake Ewijk 55 µg Chl-a/L in Lake Waainetein | l.s. | More frequent events of flood in Lakes Waainestein and Ewijk than in three other studied lakes; floods in winter with river water rich in nutrients | Van den Brink et al. [79]. |
| Eutrophic Iyieke and Ehoma Lakes Cross River floodplain (Nigeria) March 2005–August 2006 | A higher abundance of cyanobacteria during the wet season. Oscillatoria limnetica predominant, O. splendida, Phormidium subcylindrica, Anabaena cylindrica, Aph. flos-aquae, Microcystis flos-aquae, and M. aeruginosa as accompanying taxa | 1.7 × 105 cells/L | l.s. | Nutrient enrichment, water velocity, width, and depth positively influenced cyanobacteria abundance | Okechukwu and Ugwumba Alex O [14]. |
| Smal Winnica 1, Winnica 1a and Martówka Lakes, Larger Lake Port Drzewny, Vistula River (Poland) July and August 2009—before the flood and in July and August 2010-after the flood | Port Drzewny Lake (connected to the river)—annual summer blooms of Aph. flos-aquae, M. wesenbergii and P. agardhii | The highest abundance (17.9 × 106 ind./L) and biomass (4.7 mg/L)in Lake Port Drzewny before the flood | l.s. | Massive flood favored cyanobacterial development in three macrophyte-dominated lakes isolated from the river, but caused a decrease in the cyanobacteria abundance and biomass in lakes that were connected to the river | Dembowska [80]. |
| Eutrophic Lake Poyang Yangtze River floodplain (China) May 2012 to November 2013 | Cyanobacterial dominance in periods of high water levels (from July to August); Microcystis spp. predominated Accompanying taxa: Anabaena spp. Phormidium spp. Planktothrix sp. | 45–93% of the total phytoplankton biomass, the highest peak in August 2013 (93%) | * from January to December 2012: MC-RR was the dominant variant, followed by two isoforms: MC-LR, MC-YR detected in low concentrations. The total concentration of MC (intracellular and extracellular MCs) ranged from 0.0013 to 9.92 µg/L, with an average of 0.47 ng/L. Seasonal variations of MC concentrations. MCs at low concentrations from January to April, then, from June to August, MC concentration rapidly increased and rapidly decreased thereafter. | Large WLFs; high water level observed from April-May to July-August | Liu, X. et al. [58]. Zhang, et al. [94]. |
| Amazonian floodplain eutrophic lakes, Curuai floodplain (Brazil) 2013–2014 | The highest biovolume of cyanobacteria during HW (high water), FL (flushing), and LW (low water) periods. The dominants during HW were Phormidium spp. The dominants during the FL were Dolichospermum spp. and Gleiterinema splendidum. During LW, Oscillatoria spp. and Phormidium spp. predominated. The proportion of cyanobacteria increased along the hydrological cycle from RS to LW | Ca. 98% of the total phytoplankton biomass | l.s. | Nutrient increment in rising periods favored phytoplankton development and increased biomass, which became predominated by cyanobacteria during the low-water period. | Kraus, et al. [81]. |
4. Role of Hydrology in the Occurrence of Cyanobacterial Blooms in the Floodplain Lakes
5. Cyanotoxins in Floodplain Lakes
6. Role of Hydrology in Cyanotoxin Production in the Floodplain Lakes
7. Water Management Practices in Floodplain Lakes
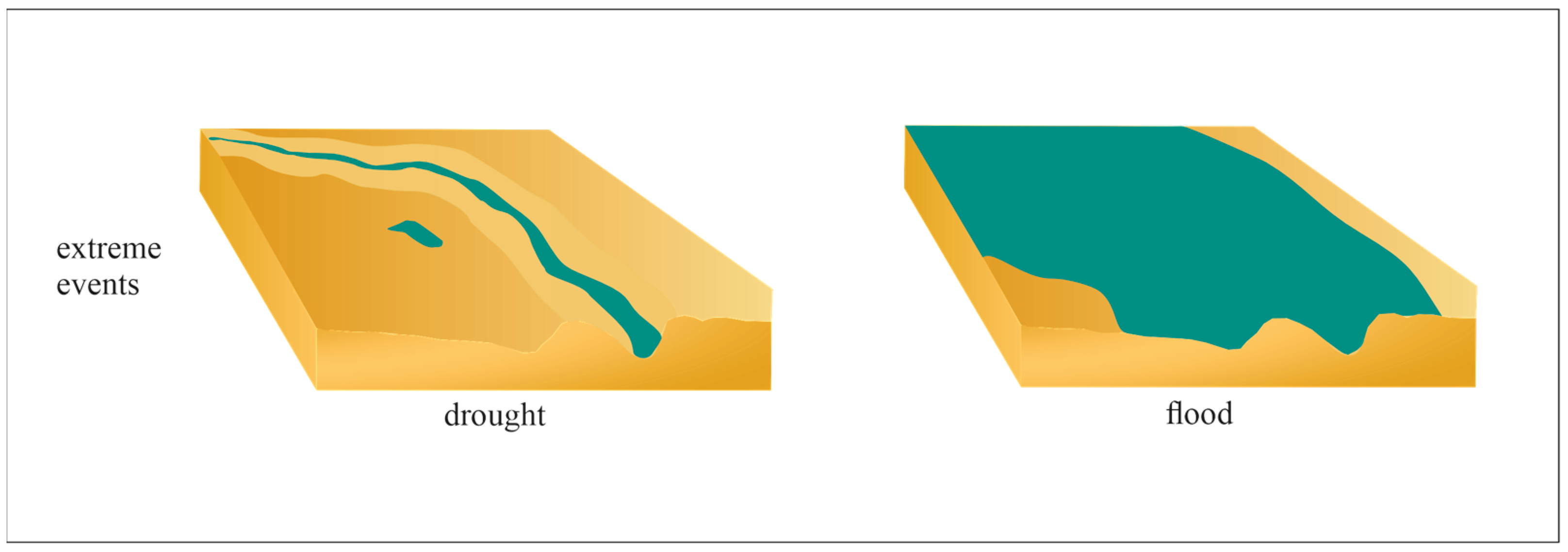
8. Future Changes of Floodplain Lakes Due to Climate Changes
9. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Thomas, R.F.; Kingsford, R.T.; Lu, Y.; Cox, S.J.; Sims, N.C.; Hunter, S.J. Mapping inundation in the heterogeneous floodplain wetlands of the Macquarie Marshes, using Landsat Thematic Mapper. J. Hydrol. 2015, 524, 194–213. [Google Scholar] [CrossRef]
- Ogbeide, O.; Uhunamure, G.; Uwagboe, L.; Osakpamwan, T.; Glory, M.; Chukwuka, A. Comparative gill and liver pathology of tilapia zilli, Clarias gariepinus and Neochanna diversus in owan river (Nigeria): Relative ecological risks of species in a pesticide-impacted river. Chemosphere 2019, 234, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Pettit, N.E.; Naiman, R.J.; Warfe, D.M.; Jardine, T.D.; Douglas, M.M.; Bunn, S.E.; Davies, P.M. Productivity and connectivity in tropical riverscapes of northern Australia: Ecological insights for management. Ecosystems 2017, 20, 492–514. [Google Scholar] [CrossRef]
- Crook, D.A.; Lowe, W.H.; Allendorf, F.W.; Erős, T.; Finn, D.S.; Gillanders, B.M.; Hadwen, W.L.; Harrod, C.; Hermoso, V.; Jennings, S.; et al. Human Effects on Ecological Connectivity in Aquatic Ecosystems: Integrating Scientific Approaches to Support Management and Mitigation. Sci. Total Environ. 2015, 534, 52–64. [Google Scholar] [CrossRef] [PubMed]
- Melack, J.M.; Forsberg, B.R. Biogeochemistry of Amazon Floodplain Lakes and Associated Wetlands: Biogeochemistry of the Amazon Basin; Oxford University Press: Oxford, UK, 2001; pp. 235–274. [Google Scholar]
- Khaki, M.; Awange, J. Improved remotely sensed satellite products for studying Lake Victoria’s water storage changes. Sci. Total Environ. 2018, 652, 915–926. [Google Scholar] [CrossRef]
- Gmitrowicz-Iwan, J.; Ligęza, S.; Pranagal, J.; Smal, H.; Olenderek, H. Floodplain Reservoirs in the Face of Climate Change—Sink or Source of Nutrients? Water 2020, 12, 3423. [Google Scholar] [CrossRef]
- Junk, W.J.; Bayley, P.B.; Sparks, R.E. The floodpulse concept in river-floodplain systems. Can. Fish. Aquat. Sci. Spec. Publ. 1989, 106, 110–127. [Google Scholar]
- Fantin-Cruz, I.; Loverde-Oliveira, S.; Girard, P. Morphometric characterization and its limnological implications in Northern Pantanal lakes. Acta Sci. Biol. Sci. 2008, 30, 133–140. [Google Scholar]
- Neiff, J.J. Ideas para la interpretacion ecológica del Parana. Interciencia 1990, 15, 424–441. [Google Scholar]
- Thomaz, S.M.; Pagioro, T.A.; Bini, L.M.; Roberto, M.C.; Rocha, R.R.A. The Upper Parana River and Itsfloodplain, Physicalaspects, Ecology and Conservation; Thomaz, S.M., Agostinho, A.A., Hahn, N.S., Eds.; Backhuys Publishers: Leiden, The Netherlands, 2004. [Google Scholar]
- Yao, J.; Zhang, Q.; Ye, X.; Zhang, D.; Bai, P. Quantifying the impact of bathymetric changes on the hydrological regimes in a large floodplain lake: Poyang Lake. J. Hydrol. 2018, 561, 711–723. [Google Scholar] [CrossRef]
- Reisenbüchler, M.; Bui, M.D.; Skublics, D.; Rutschmann, P. An integrated approach for investigating the correlation between floods and river morphology: A case study of the Saalach River, Germany. Sci. Total Environ. 2019, 647, 814–826. [Google Scholar] [CrossRef] [PubMed]
- Okechukwu, I.; Ugwumba, O.; Alex, O. Cyanobacteria abundance and its relationship to water quality in the Mid-Cross River floodplain, Nigeria. Rev. Biol. Trop. 2009, 57, 33–43. [Google Scholar] [CrossRef] [PubMed]
- Câmara Dos Reis, M.; Lacativa Bagatini, I.; de Oliveira Vidal, L.; Bonnet, M.P.; da Motta Marques, D.; Sarmento, H. Spatial heterogeneity and hydrological fluctuations drive bacterioplankton community composition in an Amazon floodplain system. PLoS ONE 2019, 14, e0220695. [Google Scholar] [CrossRef]
- Wolf, S.; Esser, V.; Schüttrumpfl, H.; Lehmkuhl, F. Infuence of 200 years of water resource management on a typical central European river. Does industrialization straighten a river? Environ. Sci. Eur. 2021, 33, 15. [Google Scholar] [CrossRef]
- Souza-Filho, E.E.; Zanetti, K.; Peccinini, J.P.; França, V. A hidroquímica do rio Paraná após a barragem de Porto Primavera. Bol. Geogr. 2010, 28, 101–112. [Google Scholar] [CrossRef]
- Hamilton, S.K.; Lewis, W.M. Basin morphology in relation to chemical and ecological characteristics of lakes on the Orinoco River floodplain, Venezuela. Arch. Hydrobiol. 1990, 119, 393–425. [Google Scholar] [CrossRef]
- García de Emiliani, M.O. Effects of water level fluctuations on phytoplankton in a river-floodplain lake system (Paraná River, Argentina). Hydrobiologia 1997, 357, 1–15. [Google Scholar] [CrossRef]
- Dawidek, J.; Ferencz, B. Hydrological processes in the riverine systems, the origin and classifications of floodplain lakes. Ecológia 2012, 31, 331–340. [Google Scholar] [CrossRef]
- Lesack, L.F.W.; Melack, J.M. Flooding hydrology and mixture dynamics of lake water derived from multiple sources in an Amazon floodplain lake. Water Resour. Res. 1995, 31, 329–345. [Google Scholar] [CrossRef]
- Neiff, J.J. Diversity in some tropical wetland systems of South América. In Biodiversity in Wetlands: Assessment, Function and Conservation; Gopal, B., Junk, W.J., Davis, J.A., Eds.; Backhuys Publishers: Leiden, The Netherlands, 2001; pp. 157–186. [Google Scholar]
- Scott, D.T.; Keim, R.F.; Edwards, B.L.; Jones, C.N.; Kroes, D.E. Floodplain Biogeochemical Processing of Floodwaters in the Atchafalaya River Basin during the Mississippi River Flood of 2011. J. Geophys. Res. Biogeosci. 2014, 119, 537–546. [Google Scholar] [CrossRef]
- Jones, C.N.; Scott, D.T.; Guth, C.; Hester, E.T.; Hession, W.C. Seasonal Variation in Floodplain Biogeochemical Processing in a Restored Headwater Stream. Environ. Sci. Technol. 2015, 49, 13190–13198. [Google Scholar] [CrossRef] [PubMed]
- Reid, M.A.; Reid, M.C.; Thoms, M.C. Ecological significance of hydrological connectivity for wetland plant communities on a dryland floodplain river, Macintyre river, Australia. Aquat. Sci. 2016, 78, 139–158. [Google Scholar] [CrossRef]
- Zhang, Y.; Huang, C.; Zhang, W. The concept, approach, and future research of hydrological connectivity and its assessment at multiscales. Environ. Sci. Pollut. Res. 2021, 28, 52724–52743. [Google Scholar] [CrossRef]
- Saha, S.; Chukwuka, A.V.; Mukherjee, D.; Saha, N.C.; Adeogun, A.O. Hydrological connectivity, surface water quality and distribution of fish species within sub-locations of an urban oxbow lake, East India. Watershed Ecol. Environ. 2022, 4, 44–58. [Google Scholar] [CrossRef]
- Napiórkowski, P.; Bąkowska, M.; Mrozińska, N.; Szymańska, M.; Kolarova, N.; Obolewski, K. The Effect of Hydrological Connectivity on the Zooplankton Structure in Floodplain Lakes of a Regulated Large River (the Lower Vistula, Poland). Water 2019, 11, 1924. [Google Scholar] [CrossRef]
- Jacobson, R.B.; Bouska, K.L.; Bulliner, E.A.; Lindner, G.A.; Paukert, C.P. Geomorphic controls on floodplain connectivity, ecosystem services, and sensitivity to climate change: An example from the lower Missouri River. Water Resour. Res. 2022, 58, e2021WR031204. [Google Scholar] [CrossRef]
- López-Vicente, M.; Ben-Salem, N. Computing structural and functional flow and sediment connectivity with a new aggregated index: A case study in a large Mediterranean catchment. Sci. Total Environ. 2019, 651 Pt 1, 179–191. [Google Scholar] [CrossRef]
- De Melo, M.L.; Bertilsson, S.; Henrique, J.; Amaral, F.; Barbosa, P.M.; Forsberg, B.R.; Sarmentoet, H. Flood pulse regulation of bacterioplankton community composition in an Amazonian floodplain lake. Freshw. Biol. 2019, 64, 108–120. [Google Scholar] [CrossRef]
- Huisman, J.; Codd, G.A.; Paerl, H.W.; Ibelings, B.W.; Verspagen, J.M.; Visser, P.M. Cyanobacterial blooms. Nat. Rev. Microbiol. 2018, 16, 471–483. [Google Scholar] [CrossRef]
- Wang, K.; Razzano, M.; Mou, X. Cyanobacterial blooms alter the relative importance of neutral and selective processes in assembling freshwater bacterioplankton community. Sci. Total Environ. 2020, 706, 135724. [Google Scholar] [CrossRef]
- Whitehead, P.G.; Wilby, R.L.; Battarbee, R.W.; Kernan, M.; Wade, A.J. A review of the potential impacts of climate change on surface water quality. Hydrol. Sci. J. 2009, 54, 101–123. [Google Scholar] [CrossRef]
- Gallardo, B.; Cabezas, A.; Gonzalez, E.; Comínet, F.A. Effectiveness of a Newly Created Oxbow Laketo Mitigate Habitat Loss and Increase Biodiversityin a Regulated Floodplain. Restor. Ecol. 2012, 20, 387–394. [Google Scholar] [CrossRef]
- Gallardo, B.; Dolédec, S.; Paillex, A.; Arscott, D.B.; Sheldon, F.; Zilli, F. Response of benthic macroinvertebrates to gradients in hydrological connectivity: A comparison of temperate, subtropical, Mediterranean and semiarid river floodplains. Freshw. Biol. 2014, 59, 630–648. [Google Scholar] [CrossRef]
- Affonso, A.G.; Queiroz, H.L.; Novo, E.M.L.M. Limnological characterization of floodplain lakes in Mamirauá Sustainable Development Reserve, Central Amazon (Amazonas State, Brazil). Acta Limnol. Bras. 2011, 23, 95–108. [Google Scholar] [CrossRef]
- Larned, S.T.; Datry, T.; Arscott, D.B.; Tockner, K. Emerging concepts in temporary river ecology. Freshw. Biol. 2010, 55, 717–738. [Google Scholar] [CrossRef]
- Freeman, M.C.; Pringle, C.M.; Jackson, C.R. Hydrologic connectivity and the contribution of stream headwaters to ecological integrity at regional scales. J. Am. Water Resour. Assoc. 2007, 43, 5–14. [Google Scholar] [CrossRef]
- Knutti, J.; Braunisch, V.; Pellet, J.; Arlettaz, R. Improving longitudinal habitat connectivity in major river restoration projects through farmland re-allocation. J. Nat. Conserv. 2021, 64, 126062. [Google Scholar] [CrossRef]
- Paira, A.R.; Drago, E.C. Origin, Evolution, and Types of Floodplain Water Bodies. In The Middle Paraná River; Iriondo, M.H., Paggi, J.C., Parma, M.J., Eds.; Springer: Berlin/Heidelberg, Germany, 2007. [Google Scholar] [CrossRef]
- Karpack, M.N.; Morrison, R.R.; McManamay, R.A. Quantitative assessment of floodplain functionality using an index of integrity. Ecol. Indic. 2020, 111, 106051. [Google Scholar] [CrossRef]
- Obolewski, K.; Glińska-Lewczuk, K.; Ożgo, M.; Astel, A. Connectivity restoration of floodplain lakes: An assessment based on macroinvertebrate communities. Hydrobiologia 2016, 774, 23–37. [Google Scholar] [CrossRef]
- Thomaz, S.M.; Bini, L.M.; Bozelli, R.L. Floods increase similarity among aquatic habitats in river-floodplain systems. Hydrobiologia 2007, 579, 1–13. [Google Scholar] [CrossRef]
- MacKinnon, B.D.; Sagin, J.; Baulch, H.M.; Lindenschmidt, K.E.; Jardine, T.D. Influence of hydrological connectivity on winter limnology in floodplain of the Saskatchewan River Delta, Saskatchewan. Can. J. Fish. Aquat. Sci. 2016, 73, 140–152. [Google Scholar] [CrossRef]
- Wiklund, J.A.; Hall, R.I.; Wolfe, B.B.; Edwards, T.W.D.; Farwell, A.J.; Dixon, D.G. Has Alberta oil sands development increased far-field delivery of airborne contaminants to the Peace-Athabasca Delta? Sci. Total Environ. 2012, 433, 379–382. [Google Scholar] [CrossRef] [PubMed]
- Seidel, M.; Voigt, M.; Langheinrich, U.; Hoge-Becker, A.; Gersberg, R.M.; Arévalo, J.R.; Lüderitz, V. Reconnection of oxbow lakes as an effective measure of river restoration. CLEAN Soil Air Water 2017, 45, 1600211. [Google Scholar] [CrossRef]
- Paillex, A.; Castella, E.; zu Ergmassen, P.S.E.; Aldridge, D.C. Testing Predictions of Changes in Alien and Native Macroinvertebrate Communities and Their Interaction After the Restoration of a Large River Floodplain (French Rhône). Freshw. Biol. 2015, 60, 1162–1175. [Google Scholar] [CrossRef]
- Wang, Y.; Fan, Z.; Wang, W.; Zhou, Z.; Ye, X. Effects of Flood on Phytoplankton Diversity and Community Structure in Floodplain Lakes Connected to the Yangtze River. Diversity 2022, 14, 581. [Google Scholar] [CrossRef]
- Fonseca, I.A.; Rodrigues, L. Periphytic cyanobacteria in different environments from the upper Paraná river floodplain, Brazil. Acta Limnol. Bras. 2007, 19, 53–65. [Google Scholar]
- Amaral, J.H.F.; Borges, A.V.; Melack, J.M.; Sarmento, H.; Barbosa, P.M.; Kasper, D.; Forsberg, B.R. Influence of plankton metabolism and mixing depth on CO2 dynamics in an Amazon floodplain lake. Sci. Total Environ. 2018, 630, 1381–1393. [Google Scholar] [CrossRef]
- Sokal, M.A.; Hall, R.I.; Wolfe, B.B. The role of flooding on inter-annual and seasonal variability of lake water chemistry, phytoplankton diatom communities and macrophyte biomass in the Slave River Delta (Northwest Territories, Canada). Ecohydrology 2010, 3, 41–54. [Google Scholar] [CrossRef]
- Amezcua, F.; Rajnohova, J.; Flores-de-Santiago, F. The Effect of Hydrological Connectivity on Fish Assemblages in a Floodplain System From the South-East Gulf of California, Mexico. Front. Mar. Sci. 2019, 6, 240. [Google Scholar] [CrossRef]
- Stević, F.; Mihaljević, M.; Špoljarić, D. Changes of phytoplankton functional groups in a floodplain lake associated with hydrological perturbations. Hydrobiologia 2013, 709, 143–158. [Google Scholar] [CrossRef]
- Tockner, K.; Pusch, M.; Borchardt, D.; Lorang, M.S. Multiple stressors in coupled river–floodplain ecosystems. Freshw. Biol. 2010, 55, 135–151. [Google Scholar] [CrossRef]
- Tockner, K.; Malard, F.; Ward, J.V. An extension of the flood pulse concept. Hydrol. Process. 2000, 14, 2861–2883. [Google Scholar] [CrossRef]
- Scheffer, M.; Carpenter, S.R. Catastrophic regime shifts in ecosystems: Linking theory to observation. Trends Ecol. Evol. 2003, 18, 648–656. [Google Scholar] [CrossRef]
- Liu, X.; Qian, K.; Chen, Y. Effects of water level fluctuations on phytoplankton in a Changjiang River floodplain lake (Poyang Lake): Implications for dam operations. J. Great Lakes Res. 2015, 41, 770–779. [Google Scholar] [CrossRef]
- Wu, S.; Wang, S.; Yang, H.; Xie, P.; Ni, L.; Xu, J. Field studies on the environmental factors in controlling microcystin production in the subtropical shallow lakes of the Yangtze River. Bull. Environ. Contam. Toxicol. 2008, 80, 329–334. [Google Scholar] [CrossRef]
- Huang, J.; Xu, Q.; Wang, X.; Ji, H.; Quigley, E.J.; Sharbatmaleki, M.; Li, C. Effects of hydrological and climatic variables on cyanobacterial blooms in four large shallow lakes fed by the Yangtze River. Environ. Sci. Ecotechnol. 2021, 5, 100069. [Google Scholar] [CrossRef] [PubMed]
- Pawlik-Skowronska, B.; Toporowska, M. How to mitigate cyanobacterial blooms and cyanotoxin production in eutrophic water reservoirs? Hydrobiologia 2016, 778, 45–59. [Google Scholar] [CrossRef]
- Coops, H.; Beklioglu, M.; Crisman, T.L. The role of water-level fluctuations in shallow lake ecosystems–workshop conclusions. Hydrobiologia 2003, 506, 23–27. [Google Scholar] [CrossRef]
- Mihaljević, M.; Stević, F. Cyanobacterial blooms in a temperate river-floodplain ecosystem: The importance of hydrological extremes. Aquat. Ecol. 2011, 45, 335–349. [Google Scholar] [CrossRef]
- Oosterberg, W.; Staras, M.; Bogdan, L.; Buijse, A.D.; Constantinescu, A.; Coops, H.; Hanganu, J.; Ibelings, B.W.; Menting, G.A.M.; Nãvodaru, I.; et al. Ecological Gradients in the Danube Delta Lakes: Present State and Man-Induced Changes: RIZA, Danube Delta National Institute Romania and Danube Delta Biosphere Reserve Authority Romania, The Netherlands; RIZA Rapport no. 2000.015; RIZA: Lelystad, The Netherlands, 2000. [Google Scholar]
- Schagerl, M.; Drozdowski, I.; Angeler, D.G.; Hein, T.; Preiner, S. Water age—A major factor controlling phytoplankton community structure in a reconnected dynamic floodplain (Danube, Regelsbrunn, Austria). J. Limnol. 2009, 68, 274–287. [Google Scholar] [CrossRef]
- Reynolds, C.S.; Petersen, A.C. The distribution of planktonic cyanobacteria in Irish lakes in relation to their trophic states. Hydrobiologia 2000, 424, 91–99. [Google Scholar] [CrossRef]
- Mihaljević, M.; Špoljarić, D.; Stević, F.; Cvijanović, V.; Kutuzović, B.H. The influence of extreme floods from the River Danube in 2006 on phytoplankton communities in a floodplain lake: Shift to a clear state. Limnologica 2010, 40, 260–268. [Google Scholar] [CrossRef]
- Whitton, B.A.; Potts, M. (Eds.) The Ecology of Cyanobacteria: Their Diversity in Time and Space; Springer Science & Business Media: Berlin/Heidelberg, Germany, 2007. [Google Scholar]
- Paerl, H.W.; Huisman, J. Blooms like it hot. Science 2008, 320, 57–58. [Google Scholar] [CrossRef] [PubMed]
- Mantzouki, E.; Lürling, M.; Fastner, J.; de Senerpont Domis, L.; Wilk-Woźniak, E.; Koreiviene, J.; Seelen, L.; Teurlincx, S.; Verstijnen, Y.; Krztoń, W.; et al. Temperature effects explain continental scale distributon of cyanobacterial toxins. Toxins 2018, 10, 156. [Google Scholar] [CrossRef] [PubMed]
- Toporowska, M.; Pawlik-Skowrońska, B.; Kalinowska, R. Mass development of diazotrophic cyanobacteria (Nostocales) and production of neurotoxic anatoxin-a in a Planktothrix (Oscillatoriales) dominated temperate lake. Water Air Soil Pollut. 2016, 227, 321. [Google Scholar] [CrossRef] [PubMed]
- Nebaeus, M. Algal water-blooms under ice-cover. Int. Ver. Theor. Angew. Limnol. 1984, 22, 719–724. [Google Scholar] [CrossRef]
- Chorus, I.; Welker, M. Toxic Cyanobacteria in Water: A Guide to Their Public Health Consequences, Monitoring and Management; Taylor & Francis: Abingdon-on-Thames, UK, 2021; 858p. [Google Scholar]
- Codd, G.; Bell, S.; Kaya, K.; Ward, C.; Beattie, K.; Metcalf, J. Cyanobacterial toxins, exposure routes and human health. Eur. J. Phycol. 1999, 34, 405–415. [Google Scholar] [CrossRef]
- Sivonen, K.; Börner, T. Bioactive Compounds Produced by Cyanobacteria. In The Cyanobacteria; Caister Academic Press: Poole, UK, 2008; pp. 159–197. [Google Scholar]
- Toporowska, M.; Mazur-Marzec, H.; Pawlik-Skowrońska, B. The effects of cyanobacterial bloom extracts on the biomass, Chl-a, MC and other oligopeptides contents in a natural Planktothrix agardhii population. Int. J. Environ. Res. Public Health 2020, 17, 2881. [Google Scholar] [CrossRef]
- O’Farrell, I.; Izaguirre, I.; Chaparro, G.; Unrein, F.; Sinistro, R.; Pizarro, H.; Tell, G. Water level as the main driver of the alternation between a free-floating plant and a phytoplankton dominated state: A long-term study in a floodplain lake. Aquat. Sci. 2011, 73, 275–287. [Google Scholar] [CrossRef]
- O’Farrell, I.; Vinocur, A.; de Tezanos Pinto, P. Long-term study of bloom-forming cyanobacteria in a highly fluctuating vegetated floodplain lake: A morpho-functional approach. Hydrobiologia 2015, 752, 91–102. [Google Scholar] [CrossRef]
- Van den Brink, F.W.B.; De Leeuw, J.P.H.M.; Van der Velde, G.; Verheggen, G.M. Impact of hydrology on the chemistry and phytoplankton development in floodplain lakes along the Lower Rhine and Meuse. Biogeochemistry 1993, 19, 103–128. [Google Scholar] [CrossRef]
- Dembowska, E.A. The impact of an extreme flood in spring/summer 2010 on phytoplankton communities in oxbow lakes of the lower Vistula River, central Poland. Ann. Limnol. Int. J. Limnol. 2017, 53, 19–26. [Google Scholar] [CrossRef]
- Kraus, C.N.; Bonnet, M.P.; de Souza Nogueira, I.; Morais Pereira Souza Lobo, M.T.; da Motta Marques, D.; Garnier, J.; Cardoso Galli Vieira, L. Unraveling flooding dynamics and nutrients’ controls upon phytoplankton functional dynamics in Amazonian floodplain lakes. Water 2019, 11, 154. [Google Scholar] [CrossRef]
- Dokulil, M.T.; Teubner, K. Cyanobacterial dominance in lakes. Hydrobiologia 2000, 438, 1–12. [Google Scholar] [CrossRef]
- Reynolds, C.S.; Huszar, V.; Kruk, C.; Naselli-Flores, L.; Melo, S. Towards a functional classification of the freshwater phytoplankton. J. Plankton Res. 2002, 24, 417–428. [Google Scholar] [CrossRef]
- Wang, K.; Mou, X.; Cao, H.; Struewing, I.; Allen, J.; Lu, J. Co-occurring microorganisms regulate the succession of cyanobacterial harmful algal blooms. Environ. Pollut. 2021, 288, 117682. [Google Scholar] [CrossRef]
- Isvánovics, V.; Shafik, H.M.; Présing, M.; Juhos, S. Growth and phosphate uptake kinetics of the cyanobacterium, Cylindrospermopsis raciborskii (Cyanophyceae) in throughflow cultures. Freshw. Biol. 2000, 43, 257–275. [Google Scholar] [CrossRef]
- Scheffer, M.; Rinaldi, S.; Gragnani, A.; Mur, L.R.; van Nes, E.H. On the dominance of filamentous cyanobacteria in shallow, turbid lakes. Ecology 1997, 78, 272–282. [Google Scholar] [CrossRef]
- Romo, S.; Soria, J.; Fernández, F.; Ouahid, J.; Barón-Solá, A. Water residence time and the dynamics of toxic cyanobacteria. Freshw. Biol. 2013, 58, 513–522. [Google Scholar] [CrossRef]
- Briand, J.F.; Robillot, C.; Quiblier-Llobéras, C.; Humbert, J.F.; Coute’, A.; Bernard, C. Environmental context of Cylindrospermopsis raciborskii (Cyanobacteria) blooms in a shallow pond in France. Water Res. 2002, 36, 3183–3192. [Google Scholar] [CrossRef]
- Figueredo, C.C.; Giani, A.; Bird, D.F. Does allelopathy contribute to Cylindrospermopsis raciborskii blooms occurrence and geographic expansion? J. Phycol. 2007, 43, 256–265. [Google Scholar] [CrossRef]
- Padisák, J. Cylindrospermopsis raciborskii (Wołoszynska) Seenayya et Subba Raju, an expanding, highly adaptative cyanobacterium: Worldwide distribution and review of its ecology. Arch. Hydrobiol. Suppl. 1997, 107, 563–593. [Google Scholar]
- Min, X.; Xu, X.D.; Hong, G.; Kong, R.Q. High variability of the gvpA-gvpC region in Microcystis. Prog. Nat. Sci. 2007, 17, 1290–1295. [Google Scholar]
- Krüger, T.; Wiegand, C.; Kun, L.; Luckas, B.; Pflugmacher, S. More and more toxins around–analysis of cyanobacterial strains isolated from Lake Chao (Anhui Province, China). Toxicon 2010, 56, 1520–1524. [Google Scholar] [CrossRef]
- Li, H.; Gu, X.; Chen, H.; Mao, Z.; Shen, R.; Zeng, Q.; Ge, Y. Co-occurrence of multiple cyanotoxins and taste-and-odor compounds in the large eutrophic Lake Taihu, China: Dynamics, driving factors, and challenges for risk assessment. Environ. Pollut. 2022, 294, 118594. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Liu, J.; Zhang, D.; Luo, L.; Liao, Q.; Yuan, L.; Wu, N. Seasonal and spatial variations of microcystins in Poyang Lake, the largest freshwater lake in China. Environ. Sci. Pollut. Res. 2018, 25, 6300–6307. [Google Scholar] [CrossRef]
- Stoyneva, M.P. Steady-state phytoplankton assemblages in shallow Bulgarian wetlands. Hydrobiologia 2003, 502, 169–176. [Google Scholar] [CrossRef]
- Ortega-Mayagoitia, E.; Rojo, C.; Rodrigo, M.A. Controlling factors of phytoplankton taxonomic structure in wetlands: An experimental approach. Hydrobiologia 2003, 502, 177–186. [Google Scholar] [CrossRef]
- Rengefors, K.; Gustafsson, S.; Ståhl-Delbanco, A. Factors regulating the recruitment of cyanobacterial and eukaryotic phytoplankton from littoral and profundal sediments. Aquat. Microb. Ecol. 2004, 36, 213–226. [Google Scholar] [CrossRef]
- Isvánovics, V.; Pettersson, K.; Rodrgio, M.A.; Pierson, D.; Padisák, J.; Colom, E. Gloeotrichia echinulata, a colonial cyanobacterium with a unique phosphorus uptake and life strategy. J. Plankton. Res. 1993, 15, 531–552. [Google Scholar] [CrossRef]
- Nixdorf, B.; Mischke, U.; Rücker, J. Phytoplankton assemblages and steady state in deep and shallow eutrophic lakes—An approach to differentiate the habitat properties of Oscillatoriales. Hydrobiologia 2003, 502, 111–121. [Google Scholar] [CrossRef]
- Pawlik-Skowrońska, B.; Toporowska, M. Blooms of toxin-producing Cyanobacteria—A real threat in small dam reservoirs at the beginning of their operation. Oceanol. Hydrobiol. Stud. 2011, 40, 30–37. [Google Scholar] [CrossRef]
- Grabowska, M. Cyanoprocaryota blooms in the polyhumic Siemianówka dam Reservoir in 1992–2003. Oceanol. Hydrobiol. Stud. 2005, 24, 73–85. [Google Scholar]
- Jöhnk, K.; Huisman, J.; Sharples, J.; Sommeijer, B.; Visser, P.M.; Strooms, J.M. Summer heatwaves promote blooms of harmful cyanobacteria. Glob. Chang. Biol. 2008, 14, 495–512. [Google Scholar] [CrossRef]
- Mankiewicz-Boczek, J.; Urbaniak, M.; Romanowska-Duda, Z.; Izydorczyk, K. Toxic cyanobacteria strains in lowland dam reservoir (Sulejów Res. central Poland): Amplification of MCY genes for detection and identification. Pol. J. Ecol. 2006, 54, 171–180. [Google Scholar]
- Toporowska, M.; Ferencz, B.; Dawidek, J. Impact of lake-catchment processes on phytoplankton community structure in temperate shallow lakes. Ecohydrology 2018, 11, e2017. [Google Scholar] [CrossRef]
- Sivonen, K.; Jones, G. Cyanobacterial toxins. In Toxic Cyanobacteria in Water: A Guide to Their Public Health Consequences, Monitoring and Management; Chorus, I., Bartram, J., Eds.; E and FN Spon: London, UK, 1999; pp. 41–111. [Google Scholar]
- Chen, L.; Giesy, J.P.; Adamovsky, O.; Svirčev, Z.; Meriluoto, J.; Codd, G.A.; Xie, P. Challenges of using blooms of Microcystis spp. in animal feeds: A comprehensive review of nutritional, toxicological and microbial health evaluation. Stoten 2021, 764, 142319. [Google Scholar] [CrossRef]
- Holland, A.; Kinnear, S. Interpreting the possible ecological role (s) of cyanotoxins: Compounds for competitive advantage and/or physiological aide? Mar. Drugs 2013, 11, 2239–2258. [Google Scholar] [CrossRef]
- Welker, M.; Döhren, H. Cyanobacterial peptides—nature’s own combinatorial biosynthesis. FEMS Microbiol. Rev. 2006, 30, 530–563. [Google Scholar] [CrossRef]
- Hu, C.; Rzymski, P. Programmed cell death-like and accompanying release of microcystin in freshwater bloom-forming cyanobacterium Microcystis: From identification to ecological relevance. Toxins 2019, 11, 706. [Google Scholar] [CrossRef]
- Affonso, A.G.; Barbosa, C.; Novo, E.M.L.M. Water quality changes in floodplain lakes due to the Amazon River flood pulse: Lago Grande de Curuaí (Pará). Braz. J. Biol. 2011, 71, 601–610. [Google Scholar] [CrossRef] [PubMed]
- Pawlik-Skowrońska, B.; Toporowska, M.; Mazur-Marzec, H. Toxic oligopeptides in the cyanobacterium Planktothrix agardhii dominated blooms and their effects on duckweed (Lemnaceae) development. KMAE 2018, 419, 41. [Google Scholar] [CrossRef]
- Gilroy, D.J.; Kauffman, K.W.; Hall, R.A.; Huang, X.; Chu, F.S. Assessing potential health risks from microcystin toxins in blue-green algae dietary supplements. Environ. Health Perspect. 2000, 108, 435–439. [Google Scholar] [CrossRef] [PubMed]
- Martins, J.C.; Vasconcelos, V.M. Microcystin dynamics in aquatic organisms. J. Toxicol. Environ. Health Part B 2009, 12, 65–82. [Google Scholar] [CrossRef] [PubMed]
- Foss, A.J.; Aubel, M.T.; Gallagher, B.; Mettee, N.; Miller, A.; Fogelson, S.B. Diagnosing microcystin intoxication of canines: Clinicopathological indications, pathological characteristics, and analytical detection in postmortem and antemortem samples. Toxins 2019, 11, 456. [Google Scholar] [CrossRef] [PubMed]
- Rzymski, P.; Poniedziałek, B. Dermatotoxins synthesized by blue-green algae (Cyanobacteria). Adv. Dermatol. Allergol. 2012, 29, 47–50. [Google Scholar]
- Kaloudis, T.; Triantis, T.M.; Hiskia, A. Taste and odour compounds produced by cyanobacteria. In Handbook of Cyanobacterial Monitoring and Cyanotoxin Analysis; Meriluoto, J., Spoof, L., Codd, G.A., Eds.; Wiley: Hoboken, NJ, USA, 2016; pp. 196–201. [Google Scholar]
- Miller, M.A.; Kudela, R.M.; Mekebri, A.; Crane, D.; Oates, S.C.; Tinker, M.T.; Jessup, D.A. Evidence for a novel marine harmful algal bloom: Cyanotoxin (microcystin) transfer from land to sea otters. PLoS ONE 2010, 5, 12576. [Google Scholar] [CrossRef] [PubMed]
- Schemel, L.E.; Sommer, T.R.; Müller-Solger, A.B.; Harrell, W.C. Hydrologic variability, water chemistry, and phytoplankton biomass in a large floodplain of the Sacramento River, CA, U.S.A. Hydrobiologia 2004, 513, 129–139. [Google Scholar] [CrossRef]
- Dawidek, J.; Ferencz, B. Water Balance of selected floodplain lake basins in the Middle Bug River valley. Hydrol. Earth Syst. Sci. 2014, 18, 1457–1465. [Google Scholar] [CrossRef]
- Dessie, M.; Verhoest, N.E.C.; Pauwels, V.R.N.; Adgo, E.; Deckers, J.; Poesen, J.; Nyssen, J. Water balance of a lake with floodplain buffering: Lake Tana, Blue Nile Basin, Ethiopia. J. Hydrol. 2015, 522, 174–186. [Google Scholar] [CrossRef]
- Brierley, G.J.; Fryirs, K.A.; Jain, V. Landscape connectivity: The geographic basis of geomorphic applications. Area 2006, 38, 165–174. [Google Scholar] [CrossRef]
- Schober, B.; Hauer, C.; Habersack, H. Floodplain losses and increasing flood risk in the context of recent historic land use changes and settlement developments: Austrian case studies. J. Flood Risk Manag. 2020, 13, e12610. [Google Scholar] [CrossRef]
- Shen, M.H.; Liu, X.Q. Assessing the effects of lateral hydrological connectivity alteration on freshwater ecosystems: A meta-analysis. Ecol. Indicat. 2021, 125, 107572. [Google Scholar] [CrossRef]
- Liu, X.Q.; Wang, H.Z. Effect of loss of lateral connectivity of fish functional diversity. Conserv. Biol. J. Soc. Conserv. Biol. 2018, 32, 1336–1345. [Google Scholar] [CrossRef]
- Serra-Llobet, A.; Jähnig, S.C.; Geist, J.; Kondolf, G.M.; Damm, C.; Scholz, M.; Lund, J.; Opperman, J.J.; Yarnell, S.M.; Pawley, A.; et al. Restoring Rivers and Floodplains for Habitat and Flood Risk Reduction: Experiences in Multi-Benefit Floodplain Management from California and Germany. Front. Environ. Sci. 2022, 9, 778568. [Google Scholar] [CrossRef]
- Lemke, M.J.; Hagy, H.M.; Dungey, K.; Casper, A.F.; Lemke, A.M.; VanMiddlesworth, T.D.; Angela Kent, A. Echoes of a flood pulse: Short-term effects of record flooding of the Illinois River on floodplain lakes under ecological restoration. Hydrobiologia 2017, 804, 151–175. [Google Scholar] [CrossRef]
- Kiedrzyńska, E.; Kiedrzyński, M.; Zalewski, M. Sustainable floodplain management for flood prevention and water quality improvement. Nat. Hazards 2015, 76, 955–977. [Google Scholar] [CrossRef]
- Thoms, M.C. Floodplain-river ecosystems: Lateral connections and the implications of human interference. Geomorphology 2003, 56, 335–349. [Google Scholar] [CrossRef]
- Zhang, Z.; Lu, W.X.; Zhao, Y.; Song, W.B. Development tendency analysis and evaluation of the water ecological carrying capacity in the Siping area of Jilin Province in China based on system dynamics and analytic hierarchy process. Ecol. Model. 2014, 275, 9–21. [Google Scholar] [CrossRef]
- Zalewski, M. Ecohydrology for implementation of the EU water framework directive. Proc. Inst. Civ. Eng. Water Manag. 2011, 164, 375–385. [Google Scholar] [CrossRef]
- Opperman, J.J.; Luster, R.; McKenney, B.A.; Roberts, M.; Meadows, A.W. Ecologically functional floodplains: Connectivity, flow regime, and scale. J. Am. Water Resour. Assoc. 2010, 46, 211–226. [Google Scholar] [CrossRef]
- Schilling, K.E.; Kult, K.; Wilke, K.; Streeter, M.; Vogelgesang, J. Nitrate reduction in a reconstructed floodplain oxbow fed by tile drainage. Ecol. Eng. 2017, 102, 98–107. [Google Scholar] [CrossRef]
- Boucek, R.E.; Rehage, J.S. Effects of an Episodic Drought on a Floodplain Subsidy Consumed by Mangrove River Fishes. Trans. Am. Fish. Soc. 2015, 83, 113–116. [Google Scholar]
- Bayley, P.B. Understanding large river-floodplain ecosystems. BioScience 1995, 45, 153–158. [Google Scholar] [CrossRef]
- Mallakpour, I.; Villarini, G. The changing nature of flooding across the central United States. Nat. Clim. Chang. 2015, 5, 250–254. [Google Scholar] [CrossRef]
- Klingelhöfer, D.; Müller, R.; Braun, M.; Brüggmann, D.; Groneberg, D.A. Climate change: Does international research fulfil global demands and necessities? Environ. Sci. Eur. 2020, 32, 137. [Google Scholar] [CrossRef]
- Tabari, H. Climate change impact on flood and extreme precipitation increases with water availability. Sci. Rep. 2020, 10, 13768. [Google Scholar] [CrossRef]
- Kundzewicz, Z.W.; Kanae, S.; Seneviratne, S.I.; Handmer, J.; Nicholls, N.; Peduzzi, P.; Mechler, R.; Bouwer, L.M.; Arnell, N.; Mach, K.; et al. Flood risk and climate change: Global and regional perspectives. Hydrol. Sci. J. 2014, 59, 1–28. [Google Scholar] [CrossRef]
- Mediero, L.; Santillán, D.; Garrote, L.; Granados, A. Detection and attribution of trends in magnitude, frequency and timing of floods in Spain. J. Hydrol. 2014, 517, 1072–1088. [Google Scholar] [CrossRef]
- Ward, R.D.; Friess, D.A.; Day, R.H.; MacKenzie, R.A. Impacts of climate change on mangrove ecosystems: A region by region overview. Ecosyst. Health Sustain. 2016, 2, 11879021. [Google Scholar] [CrossRef]
- Blöschl, G.; Hall, J.; Viglione, A.; Perdigão, R.A.P.; Parajka, J.; Merz, B.; Lun, D.; Arheimer, B.; Aronica, G.T.; Bilibashi, A.; et al. Changing climate both increases and decreases. Eur. River Floods Nat. 2019, 573, 108–111. [Google Scholar] [CrossRef]
- Salimi, S.; Berggren, M.; Scholzet, M. Response of the peatland carbon dioxide sink function to future climate change scenarios and water level management. Glob. Chang. Biol. 2021, 27, 5154–5168. [Google Scholar] [CrossRef] [PubMed]
- Budiyono, Y.; Aerts, J.C.J.H.; Tollenaar, D.; Ward, P.J. River flood risk in Jakarta under scenarios of future change. Nat. Hazard. Earth Sys. 2016, 16, 757–774. [Google Scholar] [CrossRef]
- Hamman, J.J.; Hamlet, A.F.; Lee, S.Y.; Fuller, R.; Grossman, E.E. Combined effects of projected sea level rise, storm surge, and peak river flows on water levels in the Skagit Floodplain. Northwest Sci. 2016, 90, 57–78. [Google Scholar] [CrossRef]
- Berghuijs, W.R.; Aalbers, E.E.; Larsen, J.R.; Trancoso, R.; Woods, R.A. Recent changes in extreme floods across multiple continents. Environ. Res. Lett. 2017, 12, 114035. [Google Scholar] [CrossRef]
- Wiel, K.; Wanders, N.; Selten, F.M.; Bierkens, M.F.P. Added value of large ensemble simulations for assessing extreme river discharge in a 2 °C warmer world. Geophys. Res. Lett. 2019, 46, 2093–2102. [Google Scholar] [CrossRef]
- Wang, A.; Melton, A.E.; Soltis, D.E.; Soltis, P.S. Potential distributional shifts in North America of allelopathic invasive plant species under climate change models. Plant Divers. 2022, 44, 11–19. [Google Scholar] [CrossRef]
- Amaral, J.H.F.; Melack, J.M.; Barbosa, P.M.; MacIntyre, S.; Kasper, D.; Cortés, A.; Silva, T.S.F.; de Sousa, R.N.; Forsberg, B.R. Carbon dioxide fluxes to the atmosphere from waters within flooded forests in the Amazon basin. J. Geophys. Res. Biogeosci. 2020, 125, e2019JG005293. [Google Scholar] [CrossRef]
- Barbosa, P.M.; Melack, J.M.; Amaral, J.H.F.; MacIntyre, S.; Kasper, D.; Cortes, A.C.; Forsberg, B.R. Dissolved CH4 concentrations and fluxes to the atmosphere from a tropical floodplain lake. Biogeochemistry 2020, 148, 129–151. [Google Scholar] [CrossRef]
- Zhang, W.; Zheng, X.; Gu, P.; Wang, N.; Zheng, Z. Distribution and risk assessment of phthalates in water and sediment of the Pearl River Delta. Environ. Sci. Pollut. Res. 2020, 27, 12550–12565. [Google Scholar] [CrossRef]
- Koç, G.; Petrow, T.; Thieken, A.H. Analysis of the Most Severe Flood Events in Turkey (1960–2014): Which Triggering Mechanisms and Aggravating Pathways Can be Identified? Water 2020, 12, 1562. [Google Scholar] [CrossRef]
- Li, Q.; Xiao, J.; Ou, T.; Han, M.S.; Wang, J.F.; Chen, J.G.; Li, Y.L.; Salmaso, N. Impact of water level fluctuations on the development of phytoplankton in a large subtropical reservoir: Implications for the management of cyanobacteria. Environ. Sci. Pollut. Res. 2017, 25, 1306–1318. [Google Scholar] [CrossRef] [PubMed]
- Jeppesen, E.; Meerhoff, M.; Davidson, T.A.; Trolle, D.; Søndergaard, M.; Lauridsen, T.L.; Beklioǧlu, M.; Brucet, S.; Volta, P.; González-Bergonzoni, I.; et al. Climate Change Impacts on Lakes: An Integrated Ecological Perspective Based on a Multi-Faceted Approach, with Special Focus on Shallow Lakes. J. Limnol. 2014, 73, 88–111. [Google Scholar] [CrossRef]
- Giblin, S.M.; Gerrish, G.A. Environmental factors controlling phytoplankton dynamics in a large floodplain river with emphasis on cyanobacteria. River Res. Appl. 2020, 36, 1137–1150. [Google Scholar] [CrossRef]
- Gopfen, M. Relation Significance between Hydrological Residence Time and Phytoplankton Dynamics in Lake Kinneret (Israel) January. Open J. Ecol. 2019, 9, 179–492. [Google Scholar] [CrossRef]
- Sorribas, M.V.; Paiva, R.C.D.; Melack, J.; Jones, C.; Carvalho, L.; Bravo, J.M.; Costa, M.H. Projections of climate change effects on discharge and inundation in the Amazon River basin. Clim. Chang. 2016, 136, 555–570. [Google Scholar] [CrossRef]
- Mantzouki, E.; Visser, P.M.; Bormans, M.; Ibelings, B.W. Understanding the key ecological traits of cyanobacteria as a basis for their management and control in changing lakes. Aquat. Ecol. 2016, 50, 333–350. [Google Scholar] [CrossRef]
- Hodgkins, G.A.; Whitfield, P.H.; Burn, D.H.; Hannaford, J.; Renard, B.; Stahl, K.; Fleig, A.K.; Madsen, H.; Mediero, L.; Korhonen, J.; et al. Climate-driven variability in the occurrence of major floods across North America and Europe. J. Hydrol. 2017, 552, 704–717. [Google Scholar] [CrossRef]
- Roderick, T.P.; Wasko, C.; Sharma, A. Atmospheric moisture measurements explain increases in tropical rainfall extremes. Geophys. Res. Lett. 2019, 46, 1375–1382. [Google Scholar] [CrossRef]
- Carpenter, S.R.; Booth, E.G.; Kucharik, C.J.; Lathrop, R.C. Extreme daily loads: Role in annual phosphorus input to a north temperate lake. Aquat. Sci. 2015, 77, 71–79. [Google Scholar] [CrossRef]
- Muhetaer, G.; Asaeda, T.; Jayasanka, S.M.D.H.; Baniya, M.B.; Abeynayaka, H.D.L.; Rashid, M.H.; Yan, H. Effects of Light Intensity and Exposure Period on the Growth and Stress Responses of Two Cyanobacteria Species: Pseudanabaena galeata and Microcystis aeruginosa. Water 2020, 12, 407. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ferencz, B.; Toporowska, M.; Dawidek, J. Role of Hydrology in Cyanobacterial Blooms in the Floodplain Lakes. Water 2023, 15, 1547. https://doi.org/10.3390/w15081547
Ferencz B, Toporowska M, Dawidek J. Role of Hydrology in Cyanobacterial Blooms in the Floodplain Lakes. Water. 2023; 15(8):1547. https://doi.org/10.3390/w15081547
Chicago/Turabian StyleFerencz, Beata, Magdalena Toporowska, and Jarosław Dawidek. 2023. "Role of Hydrology in Cyanobacterial Blooms in the Floodplain Lakes" Water 15, no. 8: 1547. https://doi.org/10.3390/w15081547
APA StyleFerencz, B., Toporowska, M., & Dawidek, J. (2023). Role of Hydrology in Cyanobacterial Blooms in the Floodplain Lakes. Water, 15(8), 1547. https://doi.org/10.3390/w15081547








