1. Introduction
Synthetic dyes are used in order to color the products of different chemical industries, including textiles, leather, paper, paint, cosmetics and plastics. Each year, more than eight thousand tons of dye is consumed by these industries [
1]. A considerable amount of polluted wastewater is formed, which creates a serious problem of environmental contamination and has an important influence on human health. This has focused the attention of researchers on how to dispose of wastewater polluted with dyes.
Adsorption is reputed to be an efficient, simple, low-cost, versatile and economically sustainable method which gives satisfying results and has high selectivity [
2,
3]. Most investigations in this area have been focused on activated carbon, and have shown promising outcomes. The main disadvantages of this material are its high cost and difficulties in regeneration [
4]. Thus, the potential of other alternative low-cost, efficient and environmentally safe biosorbents is increasingly becoming an important concern. The ability of various solid agricultural wastes to remove dyes from aqueous solutions has been studied, particularly lignocellulosic materials, such as wheat and rice straw, corn stover, rice husk, sugarcane bagasse, fry kernels and walnut shells. It is important that the process of conversion of plant biomass into a high-value biosorbent should have low energy consumption, and minimal cost and waste generation.
Juglans regia L. or walnut is a valuable tree nut in the family
Juglandaceae produced worldwide in the amount of 3.3 million tons per year. In Europe, the largest producers of walnuts are Turkey, Ukraine, Romania and France [
5]. Ukraine is an important exporter of walnuts with an export quantity of about 100 ktonnes/year to the European Union and the Middle East. The majority of Ukrainian walnuts are exported shelled, and a large amount of walnut shells are available in walnut kernel production centers. The shell represents 40% of the weight of the fruit and is an abundant and inexpensive agricultural by-product. Only a small percentage of walnut shells are used as fuel, and the rest is waste. Any possible use of this biomass will have a positive impact from the economic, ecological and social points of view. Therefore, the use of WS as an adsorbent for water purification is a very interesting possibility to valorize this low-cost and easily available product. A recent review by Albatrni et al. [
6] highlights the performance of WS-based adsorbents for the removal of organic compounds and heavy metal ions from wastewater.
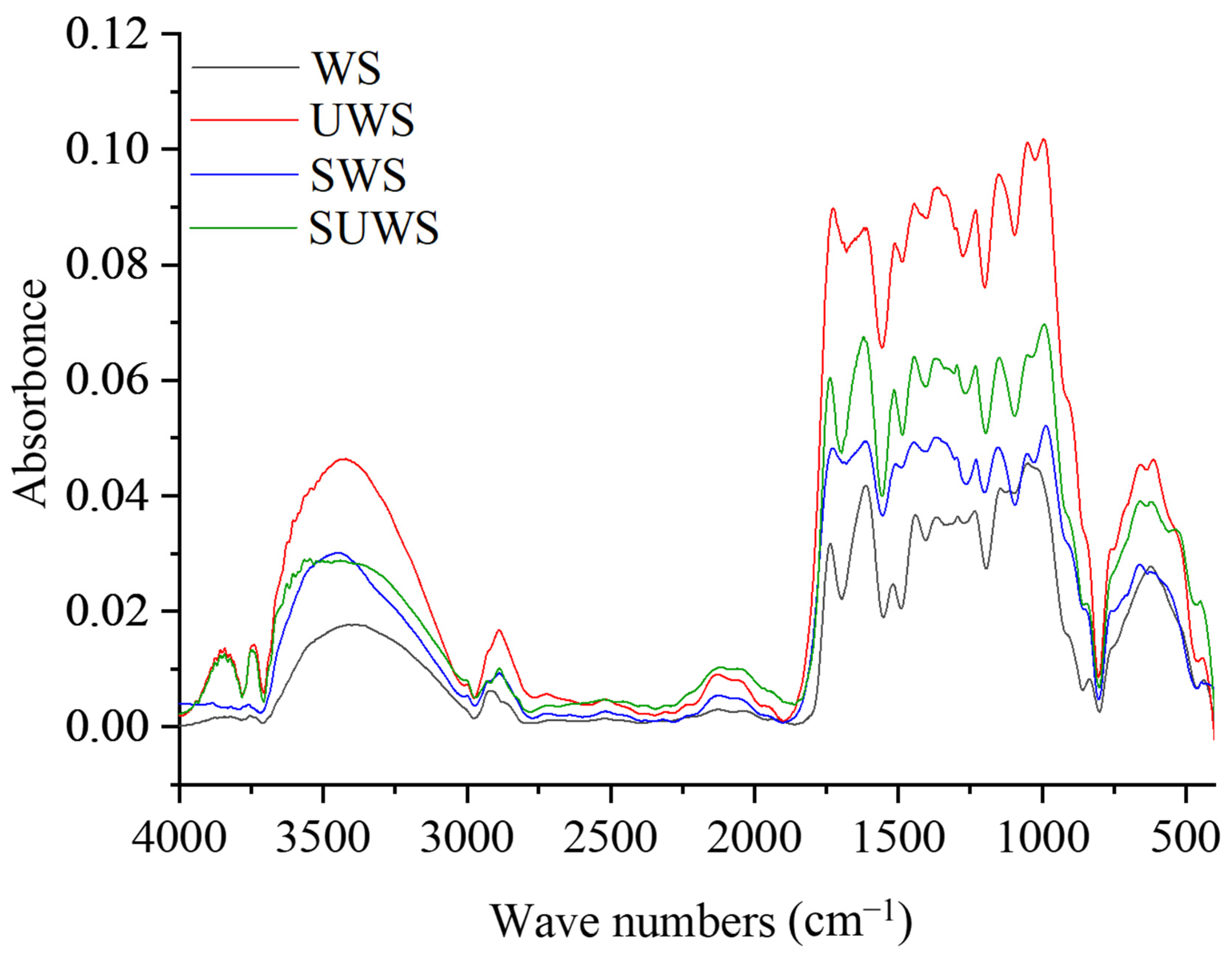
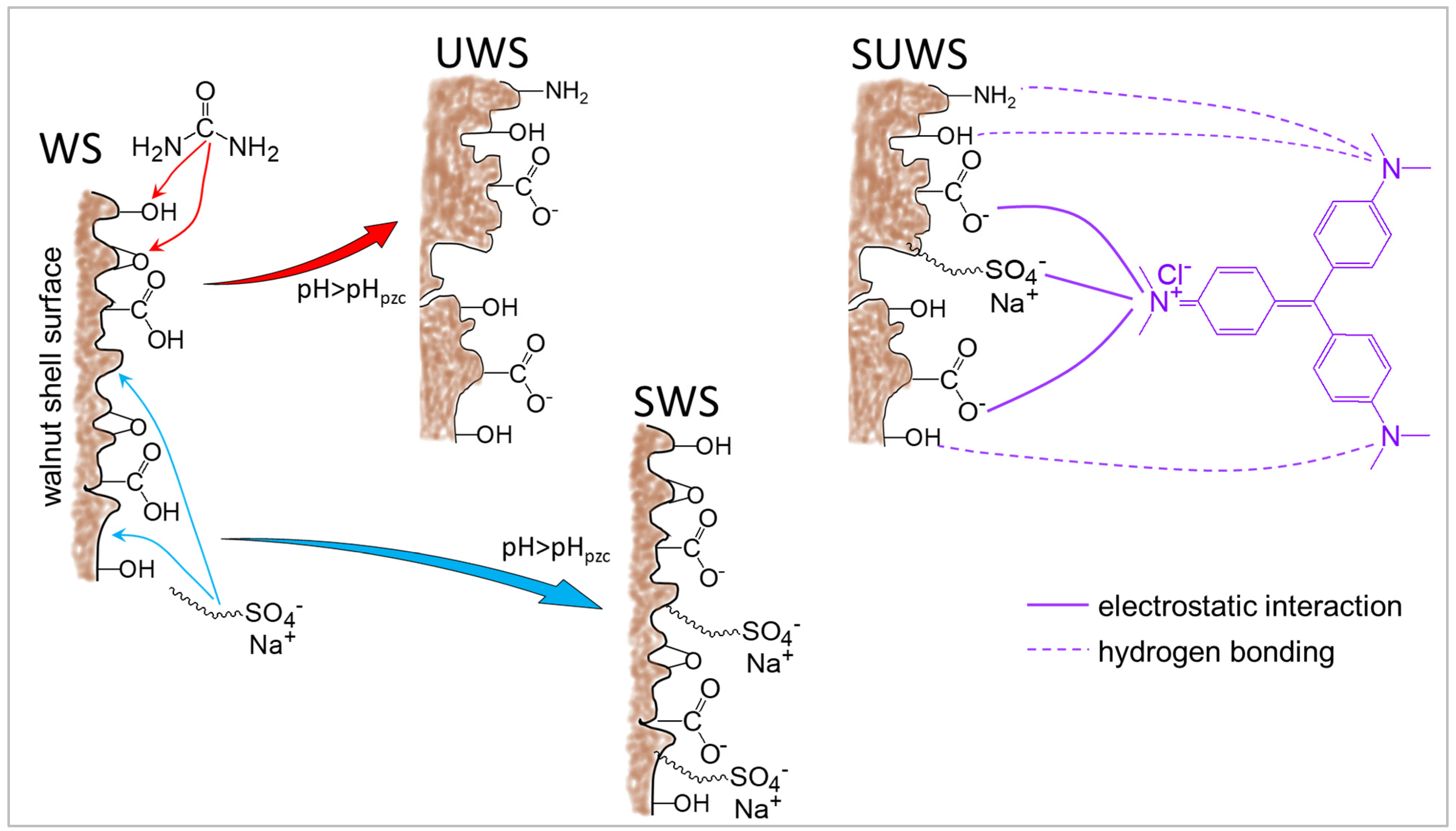
Raw walnut shells mostly consist of cellulose, hemicelluloses and lignin. The functional groups of lignocellulosic biomass that could be potentially involved in the binding of pollutants during the sorption process are hydroxyl (–OH), carbonyl (–C=O) and carboxyl (–COOH) polar groups [
7]. Natural WS adsorbents do not exhibit their full potential, showing slow adsorption kinetics and efficiency. An effective way to use this lignocellulosic adsorbent is to increase its binding potential by using chemical modification with alkali and acid treatment, or by the introduction of polar –NH
2, –COOH and –OH groups via chemical modification [
8]. Alkali and acid methods are characterized by certain disadvantages such as high operational costs and the generation of a huge quantity of liquid wastes which must be regenerated or disposed of [
9]. A proper, eco-friendly and efficient alternative method is required to overcome this waste problem.
In this direction, the use of urea for the pretreatment of lignocellulosic biomass appears to be an interesting method for enhancing adsorption efficiency. Urea is known as a non-toxic low-cost industrial product which is widely used as fertilizer. Modification of biosorbents with urea is an environmentally friendly approach, allowing an increase in nitrogen content in the form of amino (–NH
2) and amide (–CONH
2) groups [
10]. The treatment can be realized under microwave radiation without any use of solvent [
11], and is followed by washing with hot water in order to remove residual urea. A certain amount of urea-rich aqueous solution is thus produced. However, in contrast to alkali and acid wastewater, urea solutions can be efficiently used and offer several environmental benefits, for example as a clean energy source or as an AdBlue additive for vehicles [
12,
13].
Urea treatment of lignocellulosic biomass leads to the destruction of the complex three-dimensional polysaccharide structures, an increase in biosorbent surface area, and improved adsorption properties [
14]. Few research publications exist on the urea treatment of walnut shells. These studies show that combined urea–acid modification of raw walnut shells is efficient for synthetic dye sorption [
15], and carbonized walnut shells N-doped by urea show a very promising results for CO
2 [
16,
17,
18], naphthalene [
19] and perchloroethylene [
20] adsorption.
The amount and charge of functional groups present on the adsorbent surface plays an important role in the surface interaction to enhance the adsorption capacity [
21]. In order to increase the number of active sites on lignocellulosic adsorbents with a well-developed surface structure, the surfactant treatment can be applied. Several studies describe the high efficiency of surfactant-treated lignocellulosic biosorbents, including coconut coir [
22], barley straw [
23], barley husk [
24], fallen leaves of almond [
25], saw-sedge of
Cladium mariscus [
21], sawdust [
26], cork [
27] and peanut shell [
28]. However, to our knowledge, no data on the surfactant modification of walnut shells in their native or urea-pretreated state for the application as biosorbents have been identified in the literature. This research is novel in that it studies the efficiency of a combined urea/surfactant treatment on the adsorption properties of walnut shells.
The objective of the present work was to evaluate the effect of urea and/or anionic surfactant modification on the adsorption capacity of walnut shells originating from Ukraine. A model synthetic dye, crystal violet, was used for the adsorption experiments. The obtained biosorbents were characterized using Fourier transform infrared (FT-IR) analysis, pHpzc measurement and elemental and SEM-EDX analysis. The influence of several operating parameters, such as solution pH, biosorbent dosage and dye concentration, were investigated to determine the optimum conditions for the adsorption process. A kinetic study was also carried out and the experimental data were fitted to existing models in order to better understand the adsorption process.
2. Materials and Methods
2.1. Materials
The walnut shells were collected from a local Ukrainian market as a low-cost lignocellulosic waste. Urea (purity of 99.9%, MW: 60.06 g·mol
−1) and crystal violet (CV, high-purity biological stain, MW: 407.986 g·mol
−1) were purchased from Thermo Fisher Scientific (Villebon-sur-Yvette, France) and used as received. Sodium dodecyl sulfate (SDS, purity of 99%, MW: 288.38 g·mol
−1) was supplied by Sigma-Aldrich (Saint·-Quentin Fallavier, France). The SDS critical micellar concentration (CMC) of 8 mM was previously determined by the Wilhelmy plate method [
29]. All others chemicals were of analytical grade.
2.2. Preparation of Biosorbents
The walnut shells were firstly ground in a high-speed solid crusher (Hukoer, Shenzhen, China) with a rotational speed of 28,000 rpm and motor power of 850 W. The produced powder was sieved with a Retsch AS200 vibratory sieve shaker to obtain particle sizes between 160 and 400 μm. In order to remove the dust particles, walnut shell powder was soaked in deionized water for 24 h and thoroughly washed on a sieve of mesh size 50 μm. Wet walnut shell powder was stored overnight at room temperature and then dried in an oven at 105 °C to constant mass. This adsorbent sample was called WS.
In the first step of adsorbent modification, a simple and environmentally friendly method using urea compound was applied to burst the three-dimensional structure of the cellulose. Urea treatment of WS was realized according to a previously described method [
14]. Dry WS and urea powder were blended in a heat-resistant glass bottle with a weight ratio of 1:2 to ensure homogeneous particle distribution. The mixture was irradiated for 8 min using a microwave laboratory system (230 V–50 Hz, NEOS-GR, Milestone, Italy) operating at a frequency of 2450 MHz and power of 400 W with the temperature controlled. After irradiation, the mixture was boiled in deionized water for 30 min and filtered while hot using a micro-mesh 50 μm sieve. The residues and excess urea were removed by washing in hot water (60 °C). The powder was dried at 80–90 °C in the oven until constant mass. The urea-treated adsorbent was named UWS.
In the second step, an anionic surfactant SDS was used for adsorbent modification in order to increase the number of negatively charged active sites on the surface [
28]. WS and UWS samples were treated with SDS following the procedure described in the literature [
28]. SDS solution in deionized water at a concentration of 8 mM was used for the treatment. This specific concentration corresponding to CMC of SDS was selected in order to maximize the amount of surfactant adsorbed onto the surface of WS and UWS, because of micelle formation above the CMC [
30]. The mixture of adsorbent powder and SDS solution at a ratio 1:50
w/
v was stirred at 240 rpm on the magnetic table for 24 h at room temperature (25 °C). After treatment, the mixture was filtered and washed two times with distilled water to eliminate possible unbound surfactant. Finally, the SDS-modified samples were dried overnight in the oven at 90 ± 5 °C and named SWS and SUWS for walnut shells and urea-treated walnut shells, respectively.
All solid samples were stored in plastic containers at room temperature for future experiments.
2.3. Characterization of Biosorbents
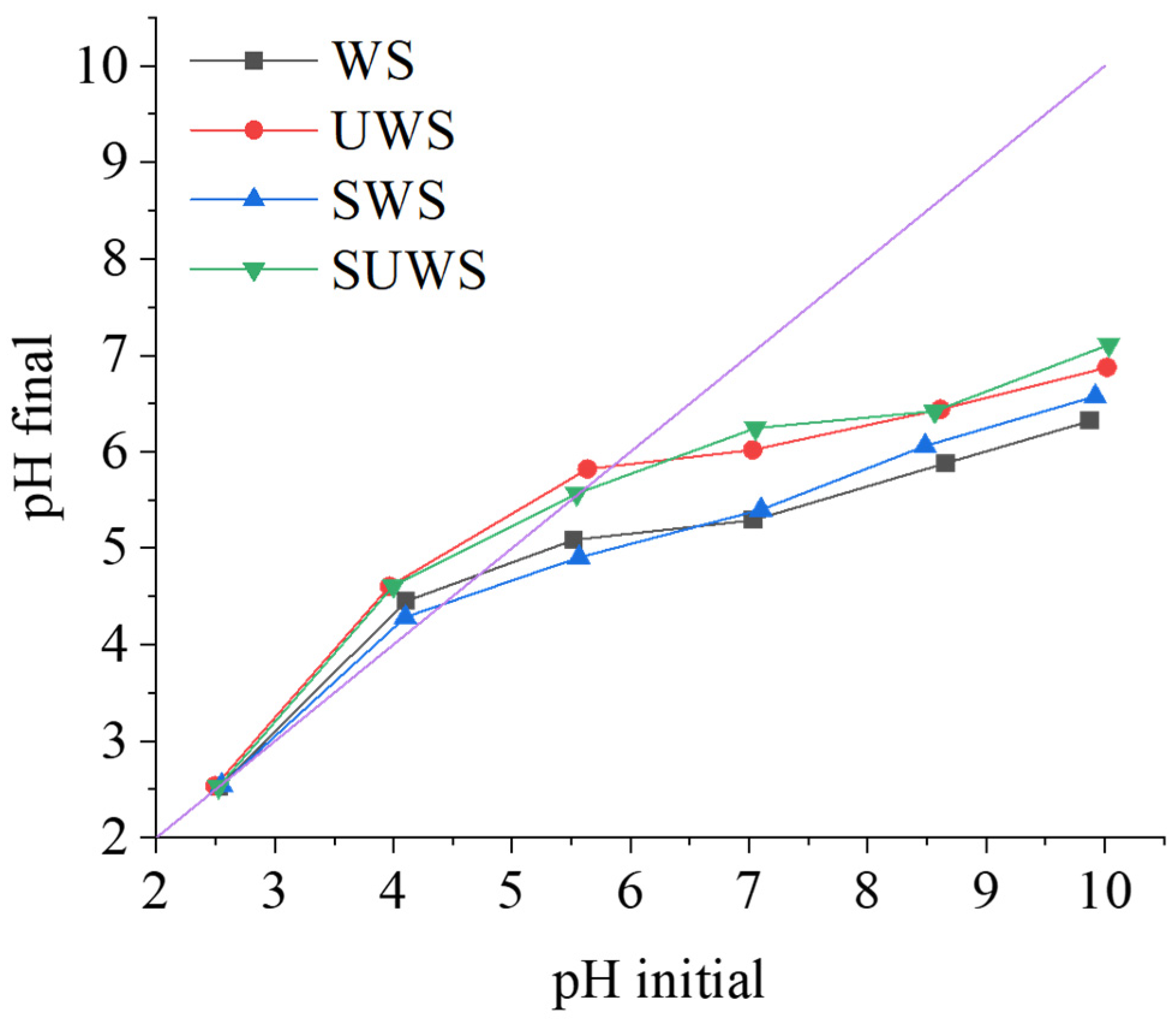
The elemental analysis of raw and modified walnut biosorbents was performed with a FLASH 2000 CHNS/O Analyzer from Thermo Scientific (Waltham, MA, USA). A point of zero charge (pHpzc) was determined using the pH drift method described by Lopez-Ramon et al. [
31] as follows: 50 mL of 0.01 M NaCl solution was placed in a closed glass bottle. The solution pH was adjusted within a range of 2.5–10.0 with 0.1 M NaOH or 0.1 M HCl using a pH probe (SevenCompact S210-Basic, Mettler-Toledo, Shanghai, China). Then, 0.1 g of biosorbent was added to the solution, and the final pH value was measured after 48 h of stirring at room temperature. The average value of three measurements was considered.
Infrared spectra were recorded with a Nicolet iS5 FTIR spectrometer (Thermo Scientific, Waltham, MA, USA) equipped with iD1 Transmission tool. A 2 mg biosorbent sample was mixed with 120 mg of spectroscopic grade KBr then pressed in a standard device to produce pellets with a 13 mm diameter. A background was recorded in ambient conditions with pure KBr pellets. All samples were scanned 16 times in the wavenumber range from 400 to 4000 cm−1 with a resolution of 4 cm−1.
The surface structure of samples was examined using scanning electron microscopy (SEM) using a Quanta FEG 250 (FEI, Hillsboro, OR, USA) equipped with an energy-dispersive spectrometer for X-ray microanalysis (EDX, Bruker Nano GmbH Berlin, Berlin, Germany). SEM micrographs were acquired from the platinum-coated biosorbents (Denton Vacuum Desk V, Moorestown, NJ, USA) in secondary electron mode at low vacuum and 20 kV accelerating voltage. The surface elemental compositions were determined from the EDX analysis.
2.4. Adsorption Experiments
2.4.1. Optimization of Adsorption Conditions
A cationic compound crystal violet (CV) was used as a model dye for adsorption experiments. A stock solution of CV at 100 mg·L−1 was prepared and used to obtain the required concentration range by the dilutions in deionized water. The pH value of the suspensions was adjusted to suitable values for adsorption experiments by using 0.1 M HCl or 0.1 M NaOH solutions.
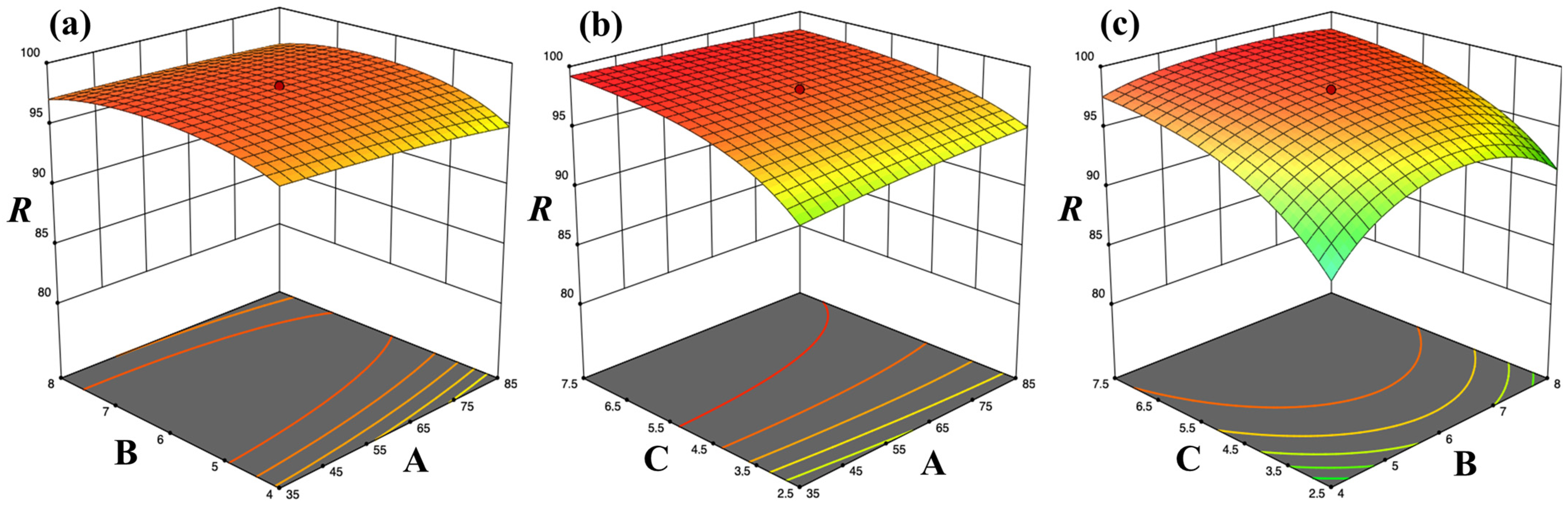
Firstly, the optimization of CV removal was carried out for each of the four adsorbent agents, WS, UWS, SWS and SUWS, by three chosen independent process variables: CV concentration, pH and biosorbent dose. According to the central composite face-centered design (CCFD) [
32,
33,
34], six replicates at the center point and six star point experiments were performed. Each factor was set to five levels: high and low level of axial points, high and low level of factorial points, and the center point, as presented in
Table 1. The zero value of the categorical factor was added, which means that the CCFD was not repeated for every combination of the categorical factor levels. In total, twenty experiments were conducted in duplicate for each biosorbent as summarized in
Table 2.
The adsorption experiments of CV from aqueous solution onto biosorbent samples were performed in 100 mL closed glass bottles with a working volume of 20 mL. A predetermined amount of the biosorbent was added to CV solution of known concentration and pH. The mixture was agitated on a magnetic table at room temperature at a 150 rpm stirring rate for 60 min of contact time. At the end of experiment, the suspension was decanted for 5 min and the residual CV concentration was determined spectrophotometrically at a wavelength of 590 nm using a UV/VIS spectrometer Lambda 12 (PerkinElmer, Waltham, MA, USA).
The response of the design experiments was called removal efficiency (
R) and calculated as the percentage adsorption of CV:
where
is the initial concentration of CV solution (mg·L
−1) and
is the residual concentration of CV solution after adsorption (mg·L
−1).
The optimum values of each experimental variable were obtained using Design-Expert
® version 13 software by solving the regression equations and carrying out graphical analysis of response surface plots, as explained by Garg et al. [
32,
33].
2.4.2. Adsorption Kinetics and Isotherm Models
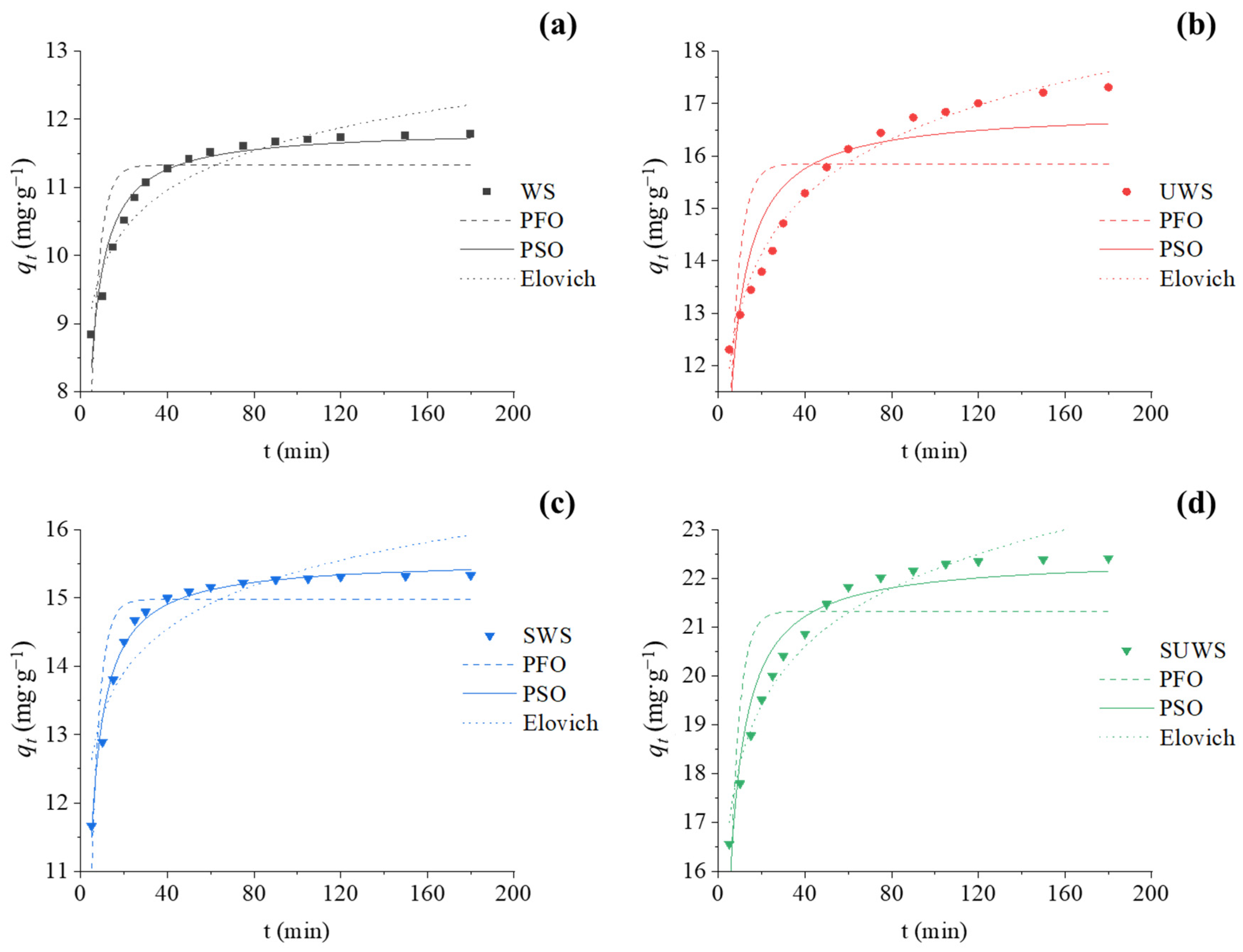
The kinetic studies in batch mode were carried out in a 600 mL glass vessel with a working volume of 300 mL. Different parameters were fixed based on the results of the optimization experiments, namely CV concentration of 60 mg·L−1, biosorbent dose of 5.0 g·L−1, initial pH of solution of 6.5 for UWS and SUWS biosorbents, and 7.5 for WS and SWS biosorbents. The mixture was stirred on a magnetic table at a constant speed of 150 rpm, and the contact times were 5, 10, 15, 20, 25, 30, 40, 50, 60, 75, 90, 105, 120, 150 and 180 min. Aliquots were sampled from the glass at predetermined time intervals in a such way that the material balance of solid phase–liquid was not affected. Using the same experimental conditions of pH, temperature, adsorbent dose and agitation rate, the equilibrium adsorption isotherms were also obtained. Each adsorbent was mixed with 20 mL of CV solution at initial concentrations in the range from 10 to 100 mg·g−1 until equilibrium was achieved. As for optimization experiments, the residual dye concentration was analyzed using UV/VIS spectrophotometry at 590 nm.
The amount of adsorbed dye concentration at any given time,
, was calculated according to mass balance as:
where
is the initial concentration of dye (mg·L
−1),
corresponds to the residual concentration of dye in the aqueous phase at any given time (mg·L
−1),
is the mass of biosorbent (g) and
is the volume of used solution (L). All experiments were performed in triplicate. Experimental data are reported as the average values of at least three measurements.
4. Conclusions
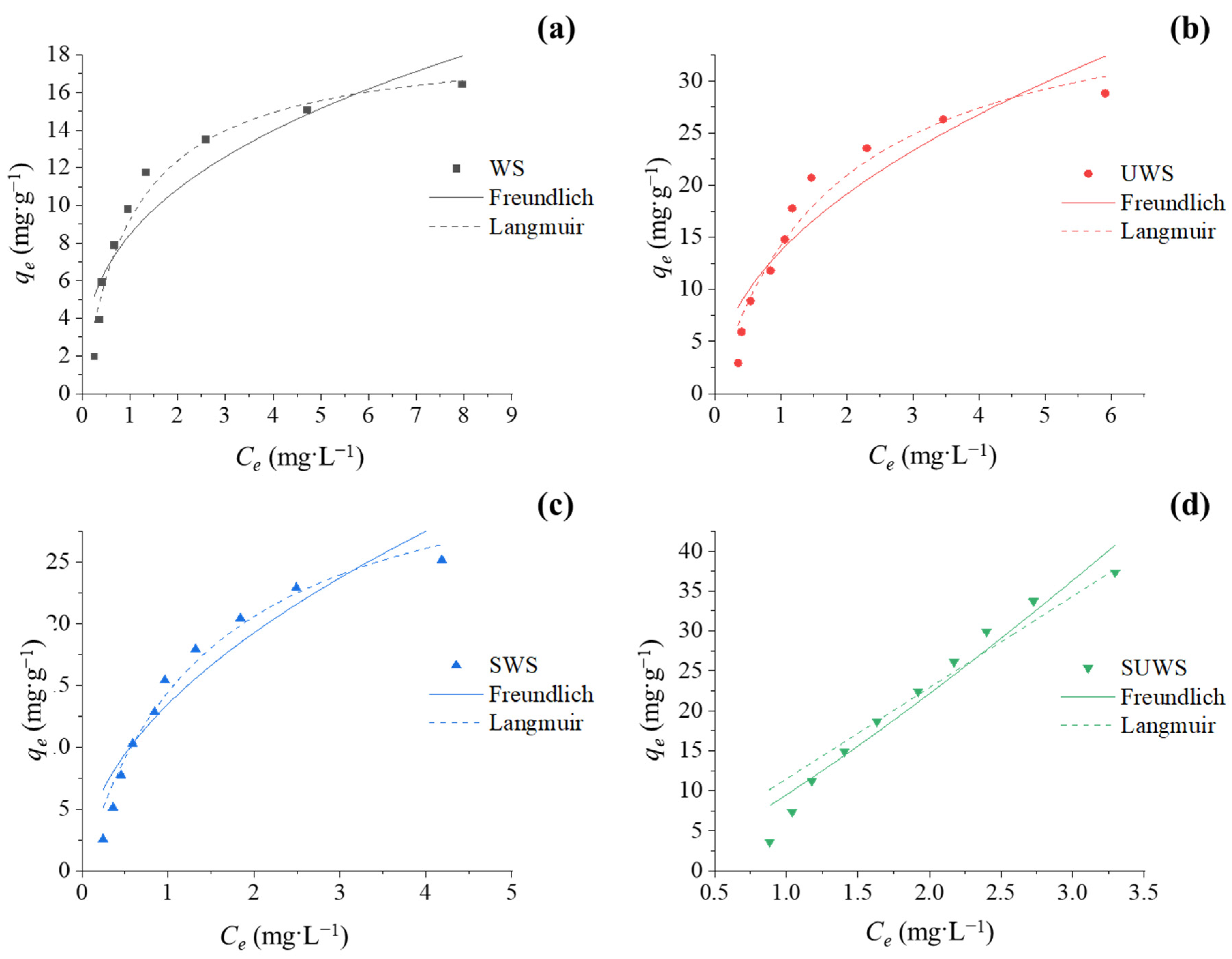
In this study, low cost adsorbents from waste biomass were successfully applied in the treatment of dye effluents. Four kinds of biosorbents were prepared from walnut shells with one- or two-step modification using urea and/or anionic surfactant. The efficiency of native and modified biosorbents for the removal of synthetic cationic CV dye from aqueous solutions was investigated. The optimization experiments showed that pH close to 7 ± 0.5 allowed the highest adsorption efficiency. The biosorbent treated with urea and surfactant demonstrated a high efficiency of CV removal (>95%) and adsorption capacity (37 mg·g−1). The kinetic study of adsorption was observed to follow the pseudo-second-order rate model. Which gave the most accurate correlation between experimental and model data. The adsorption isotherms of walnut biosorbents were better fitted by the Langmuir equation, supporting CV adsorption onto biosorbent in a single layer.
The results confirm that appropriate modification can efficiently increase the performance of adsorbents. The adsorption capacity of walnut shells modified using combined urea–surfactant treatment was twice that of untreated WS. This underlines the efficiency of the proposed eco-friendly modification approach due to the increase in the biosorbent surface area caused by urea treatment and the increase in the number of active negatively charged sites after SDS treatment allowing a larger amount of adsorbed cationic CV.
These investigations contribute to our knowledge of complex adsorption processes using biomass-based adsorbents. In the future, it would be interesting to expand this approach of combined modification to other types of lignocellulosic biomass. From the economical point of view, and for the potential application of industrial effluent treatment, reusability of biosorbent or regeneration process should be explored.