Toward a Better Understanding of Phosphorus Nonpoint Source Pollution from Soil to Water and the Application of Amendment Materials: Research Trends
Abstract
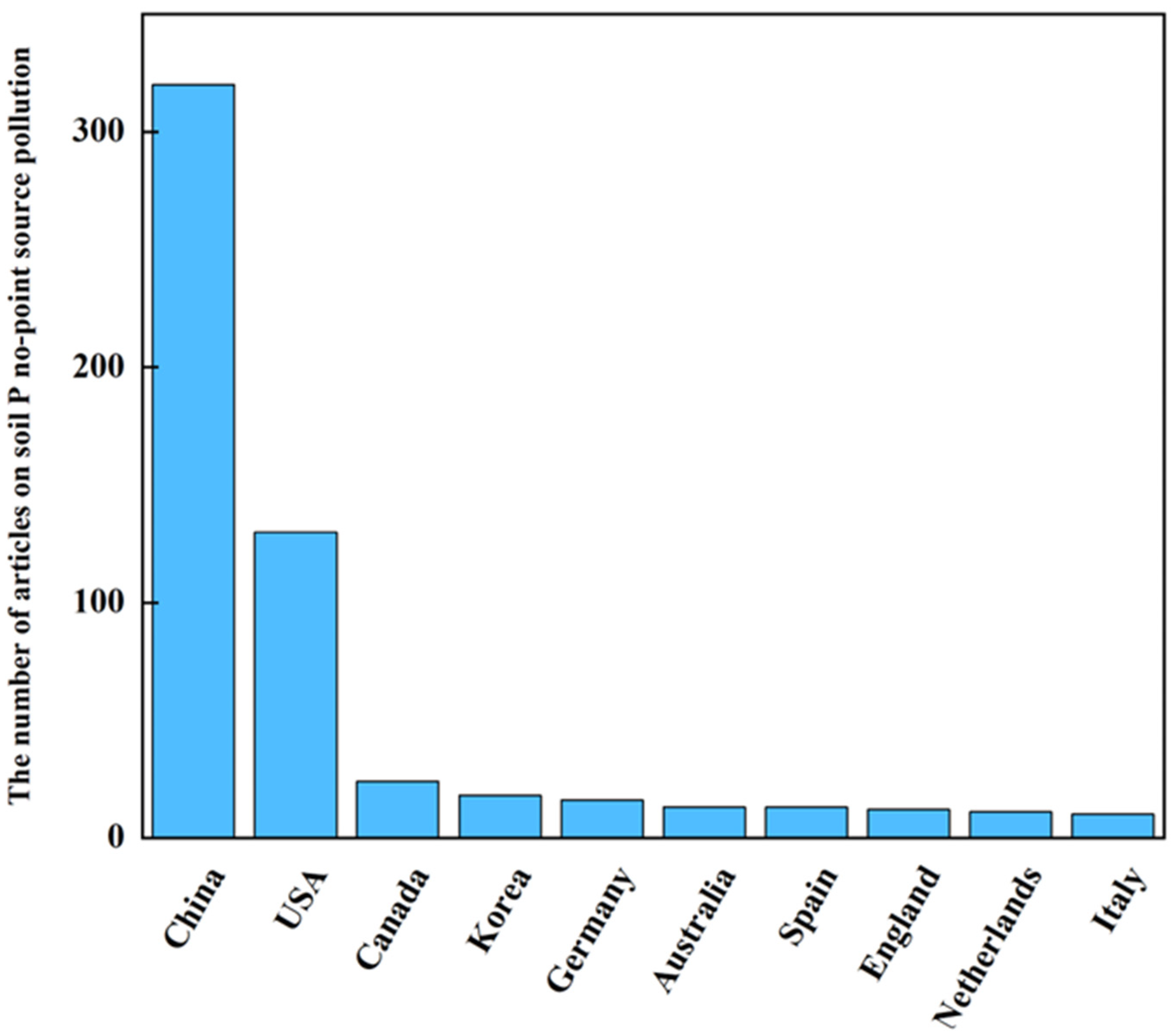
:1. Introduction
2. Phosphorus Nonpoint Source Pollution Management Strategies
2.1. Phosphorus Forms and Accumulation in Soil
2.2. An Important Source of Phosphorus to Water: Soil Phosphorus Loss
2.3. The Management Strategies for Soil Phosphorus Loss to Water Body
3. Application of Amendment Materials for Phosphorus Nonpoint Source Pollution from Soil to Water
3.1. Inorganic Amendment Materials
3.1.1. Calcium and Magnesium Inorganic Amendment Materials
3.1.2. Iron and Aluminum Inorganic Amendment Materials
3.1.3. Clay Mineral Inorganic Amendment Materials
3.1.4. Waste Inorganic Amendment Materials
3.2. Organic Amendment Materials
| Organic Amendment Materials | Applied Soil Type | P Release Reduction | References |
|---|---|---|---|
| polyacrylamide | the total P concentration of the leachate was decreased by 32.4% | [51] | |
| anionic polyacrylamide | tea soil | total P reduced by 54% | [75] |
| maize stover biochar | corn-growing soil | inorganic P reduced by 3.3–59% | [77] |
| polyacrylamide modified biochar | paddy soil | total P reduced by 41.1% | [78] |
| Sugarcane-Derived Biochar | calcareous soil | / | [79] |
| wheat straw biochar | paddy-wheat rotation soil | the P utilization rate is increased by 38–230% | [80] |
| Rice-residue waste biochar | paddy soil | / | [81] |
| reed-biochar | paddy soil | total P reduced by 5.3–13.3% | [82] |
| maple and hickory sawmill waste biochar | forest soil | increases the absorption of a small amount of soluble P | [83] |
3.3. Composite Amendment Materials
4. Soil Retention Phosphorus Reloss to Water Environmental Risk
4.1. Influence of Soil pH
4.2. Influence of Soil Matter
4.3. Influence of Water Condition
5. Conclusions
6. Limitations of Existing Studies
6.1. Watershed Phosphorus Pollution Management
6.2. Management of Phosphorus Pollution in Lakes
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Conflicts of Interest
References
- Springmann, M.; Clark, M.; Croz, D.M.D.; Wiebe, K.; Bodirsky, B.L.; Lassaletta, L.; de Vries, W.; Vermeulen, S.J.; Herrero, M.; Carlson, K.M.; et al. Options for keeping the food system within environmental limits. Nature 2018, 562, 519–525. [Google Scholar] [CrossRef] [PubMed]
- Li, H.H.; Zhang, T.; Shaheen, S.M.; Abdelrahman, H.; Ali, E.F.; Bolan, N.S.; Li, G.X.; Rinklebe, J. Microbial inoculants and struvite improved organic matter humification and stabilized phosphorus during swine manure composting: Multivariate and mul-tiscale investigations. Bioresour. Technol. 2022, 351, 126976. [Google Scholar] [CrossRef]
- Liu, G.F.; Dai, Z.M.; Liu, X.M.; Dahlgren, R.A.; Xu, J.M. Modification of agricultural wastes to improve sorption capacities for pol-lutant removal from water—A review. Carbon Res. 2022, 1, 24. [Google Scholar] [CrossRef]
- Wu, F.C.; Li, F.B.; Zhao, X.L.; Bolan, N.S.; Fu, P.Q.; Lam, S.S.; Masek, O.; Ong, H.C.; Pan, B.; Qiu, X.Q.; et al. Meet the challenges in the “Carbon Age”. Carbon Res. 2022, 1, 1. [Google Scholar] [CrossRef]
- Deng, Y.X.; Zhang, T.; Clark, J.; Aminabhavi, T.; Kruse, A.; Tsang DC, W.; Sharma, B.K.; Zhang, F.S.; Ren, H.Q. Mechanisms and modelling of phosphorus solid–liquid transformation during the hydrothermal processing of swine manure. Green Chem. 2020, 22, 5628–5638. [Google Scholar] [CrossRef]
- Xie, S.Y.; Zhang, T.; Mishra, A.; Tiwari, A.; Bolan, N.S. Assessment of catalytic thermal hydrolysis of swine manure slurry as liquid fertilizer: Insights into nutrients and metals. Front. Environ. Sci. 2022, 10, 1005290. [Google Scholar] [CrossRef]
- Yu, J.X.; Xie, S.Y.; Zhang, T. Influences of hydrothermal carbonization on phosphorus availability of swine manure-derived hydrochar: Insights into reaction time and temperature. Mater. Sci. Energy Technol. 2022, 5, 416–423. [Google Scholar] [CrossRef]
- Ge, X.F.; Zhang, T. Changes in inorganic and organic matters in processed water from hydrothermal-treated biogas slurry. Mater. Sci. Energy Technol. 2023, 6, 145–157. [Google Scholar] [CrossRef]
- Guan, Q.; Zeng, G.; Song, J.; Liu, C.; Wang, Z.; Wu, S. Ultrasonic power combined with seed materials for recovery of phosphorus from swine wastewater via struvite crystallization process. J. Environ. Manag. 2021, 293, 112961. [Google Scholar] [CrossRef]
- Godfray, H.C.J.; Beddington, J.R.; Crute, I.R.; Haddad, L.; Lawrence, D.; Muir, J.F.; Pretty, J.; Robinson, S.; Thomas, S.M.; Toulmin, C. Food security: The challenge of feeding 9 billion people. Science 2010, 327, 812–818. [Google Scholar] [CrossRef] [Green Version]
- Vermeulen, S.J.; Campbell, B.M.; Ingram JS, I. Climate change and food systems. Annu. Rev. Environ. Resour. 2012, 37, 195–222. [Google Scholar] [CrossRef] [Green Version]
- Chen, X.P.; Cui, Z.L.; Fan, M.S. Producing more grain with lower environmental costs. Prod. More Grain Low. Environ. Costs Nat. 2014, 514, 486–489. [Google Scholar] [CrossRef]
- Chen, M.P.; Graedel, T.E. A half-century of global phosphorus flows, stocks, production, consumption, recycling, and environmental impacts. Glob. Environ. Change 2016, 36, 139–152. [Google Scholar] [CrossRef]
- He, X.Y.; Wang, Y.K.; Zhang, Y.Y.; Wang, C.S.; Yu, J.X.; Ohtake, H.; Zhang, T. The potential for livestock manure valorization and phos-phorus recovery by hydrothermal technology—A critical review. Mater. Sci. Energy Technol. 2023, 6, 94–104. [Google Scholar]
- Cordell, D.; White, S. Life’s bottleneck: Sustaining the world’s phosphorus for a food secure future. Annu. Rev. Environ. Resour. 2014, 39, 161–188. [Google Scholar] [CrossRef]
- Wang, Q.M.; Zhang, T.; He, X.Y.; Jiang, R.F. Assessment of phosphorus recovery from swine wastewater in Beijing, China. Sustainability 2017, 9, 1845. [Google Scholar] [CrossRef] [Green Version]
- Zou, L.; Liu, Y.; Wang, Y.; Hu, X. Assessment and analysis of agricultural non-point source pollution loads in China: 1978–2017. J. Environ. Manag. 2020, 263, 110400. [Google Scholar] [CrossRef]
- Hu, X.; Zhou, Y.; Zhou, L.; Zhang, Y.; Wu, L.; Xu, H.; Zhu, G.; Jang, K.; Spencer, R.; Jeppesen, E.; et al. Urban and agricultural land use regulates the molecular composition and bio-lability of fluvial dissolved organic matter in human-impacted southeastern China. Carbon Res. 2022, 1, 19. [Google Scholar] [CrossRef]
- Pradhan, S.N.; Ghosh, A.K.; Seema Ram, S.; Pal, Y.; Pradhan, C. Changes in degree of phosphorus saturation and risk of P loss upon twelve years of manuring and reduced tillage. Geoderma 2021, 404, 115277. [Google Scholar] [CrossRef]
- Li, H.; Huang, G.; Meng, Q.; Ma, L.; Yuan, L.; Wang, F.; Zhang, W.; Cui, Z.; Shen, J.; Chen, X.; et al. Integrated soil and plant phosphorus management for crop and environment in China. A review. Plant Soil 2011, 349, 157–167. [Google Scholar] [CrossRef]
- Sun, H.; Liang, W. Research on phosphorus pollution in China’s water bodies and its monitoring and management technology. J. Zhejiang Norm. Univ. Nat. Sci. Ed. 2007, 30, 201–205. [Google Scholar]
- Ma, E.P.; Cai, J.M.; Lin, J.; Liao, L.W.; Guo, H.; Han, Y. Pattern characteristics of nitrogen and phosphorus emissions from agricultural sources and water environment impacts in China in the last 30 years. J. Nat. Resour. 2021, 36, 752–770. [Google Scholar]
- Tim, U.S.; Jolly, R. Evaluating agricultural nonpoint-source pollution using integrated geographic information systems and hydro-logic/water quality model. J. Environ. Qual. 1994, 23, 25–35. [Google Scholar] [CrossRef]
- Sabo, R.D.; Clark, C.M.; Gibbs, D.A.; Metson, G.S.; Todd, M.J.; LeDuc, S.D.; Greiner, D.; Fry, M.M.; Polinsky, R.; Yang, Q.; et al. Phosphorus inventory for the conterminous United States (2002–2012). J. Geophys. Res. Biogeosci. 2021, 126, e2020JG005684. [Google Scholar] [CrossRef]
- Kronvang, B.; Bruhn, A.J. Choice of sampling strategy and estimation method for calculating nitrogen and phosphorus transport in small lowland streams. Hydrol. Process. 1996, 10, 1483–1501. [Google Scholar] [CrossRef]
- McDowell, R.W. The effectiveness of coal fly-ash to decrease phosphorus loss from grassland soils. Soil Res. 2005, 43, 853–860. [Google Scholar] [CrossRef]
- Eduah, J.O.; Nartey, E.K.; Abekoe, M.K.; Breuning-Madsen, H.; Andersen, M.N. Phosphorus retention and availability in three contrasting soils amended with rice husk and corn cob biochar at varying pyrolysis temperatures. Geoderma 2019, 341, 10–17. [Google Scholar] [CrossRef]
- Jiang, X.; Yan, B.; Chen, J.; Li, W.; Guan, Y. Transport and retention of phosphorus in soil with addition of Mg-Al layered double hydroxides: Effects of material dosage, flow velocity and pH. Chem. Eng. J. 2019, 378, 122154. [Google Scholar] [CrossRef]
- Liang, Z.; Peng, X.; Luan, Z.; Li, W.; Zhao, Y. Reduction of phosphorus release from high phosphorus soil by red mud. Environ. Earth Sci. 2012, 65, 581–588. [Google Scholar] [CrossRef] [Green Version]
- Fan, B.; Fenton, O.; Daly, K.; Ding, J.; Chen, S.; Chen, Q. Alum split applications strengthened phosphorus fixation and phosphate sorption in high legacy phosphorus calcareous soil. J. Environ. Sci. 2021, 101, 87–97. [Google Scholar] [CrossRef]
- Yan, C.; Zhan, H.; Yan, S.; Dong, S.; Ma, C.; Song, Q.; Gong, Z.; Barbie, M. Effects of straw retention and phosphorous fertilizer application on available phosphorus content in the soil solution during rice growth. Paddy Water Environ. 2016, 14, 61–69. [Google Scholar] [CrossRef] [Green Version]
- Liu, L.; Zheng, X.; Wei, X.; Kai, Z.; Xu, Y. Excessive application of chemical fertilizer and organophosphorus pesticides induced total phosphorus loss from planting causing surface water eutrophication. Sci. Rep. 2021, 11, 23015. [Google Scholar] [CrossRef] [PubMed]
- Mardamootoo, T.; Du Preez, C.; Barnard, J.H. Agricultural phosphorus management for environmental protection: A review. J. Geosci. Environ. Prot. 2021, 9, 48–81. [Google Scholar] [CrossRef]
- McDowell, R.W.; Sharpley, A.N. Approximating phosphorus release from soils to surface runoff and subsurface drainage. J. Environ. Qual. 2001, 30, 508–520. [Google Scholar] [CrossRef] [Green Version]
- Hesketh, N.; Brookes, P.C. Development of an indicator for risk of phosphorus leaching. J. Environ. Qual. 2000, 29, 105–110. [Google Scholar] [CrossRef]
- McDowell, R.W.; Condron, L.M. Estimating phosphorus loss from New Zealand grassland soils. N. Z. J. Agric. Res. 2004, 47, 137–145. [Google Scholar] [CrossRef]
- Khan, A.; Lu, G.; Ayaz, M.; Zhang, H.; Wang, R.; Lv, F.; Yang, X.; Sun, B.; Zhang, S. Phosphorus efficiency, soil phosphorus dynamics and critical phosphorus level under long-term fertilization for single and double cropping systems. Agric. Ecosyst. Environ. 2018, 256, 1–11. [Google Scholar] [CrossRef]
- Weihrauch, C.; Weber, C.J. Comparative risk assessment of phosphorus loss from “deep phosphorus stocks” in floodplain subsoils to surface waters. Sci. Total Environ. 2021, 796, 149037. [Google Scholar] [CrossRef]
- Casson, J.P.; Bennett, D.R.; Nolan, S.C.; Olson, B.M.; Ontkean, G.R. Degree of Phosphorus saturation thresholds in manure-amended soils of alberta. J. Environ. Qual. 2006, 35, 2212–2221. [Google Scholar] [CrossRef]
- Zak, D.; Gelbrecht, J.; Steinberg, C. Phosphorus retention at the redox interface of peatlands adjacent to surface waters in northeast Germany. Biogeochemistry 2004, 70, 357–368. [Google Scholar] [CrossRef]
- McDowell, R.; Dodd, R.; Pletnyakov, P.; Noble, A. The ability to reduce soil legacy phosphorus at a country scale. Front. Environ. Sci. 2020, 8, 6. [Google Scholar] [CrossRef] [Green Version]
- Khattab, R.M.; Badr, H.A.; Abo-Almaged, H.H.; Sadek HE, H. Recycling of alum sludge for alpha Al2O3 production using different chemical treatments. Desalination Water Treat. 2018, 113, 148–159. [Google Scholar] [CrossRef]
- Ujaczki, E.; Zimmermann, Y.S.; Gasser, C.A.; Molnar, M.; Feigl, V.; Lenz, M. Red mud as secondary source for critical raw materials—Extraction study. J. Chem. Technol. Biotechnol. 2017, 92, 2835–2844. [Google Scholar] [CrossRef]
- Ma, P.C.; Li, X.; Wen, Z.Y.; Meng, F.H.; Li, Z. Research progress on activation and mechanism of fly ash. Inorg. Chem. Ind. 2021, 53, 28–35. [Google Scholar]
- McDowell, R.; Sharpley, A. Availability of residual phosphorus in high phosphorus soils. Commun. Soil Sci. Plant Anal. 2002, 33, 1235–1246. [Google Scholar] [CrossRef]
- Eslamian, F.; Qi, Z.; Tate, M.J.; Zhang, T.; Prasher, S.O. Phosphorus loss mitigation in leachate and surface runoff from clay loam soil using four lime-based materials. Water Air Soil Pollut. 2018, 229, 91–97. [Google Scholar] [CrossRef]
- Torbert, H.; Watts, D. Impact of flue gas desulfurization gypsum application on water quality in a coastal plain soil. J. Environ. Qual. 2014, 43, 273–280. [Google Scholar] [CrossRef]
- Fan, B.; Ding, J.; Fenton, O.; Daly, K.; Chen, Q. Understanding phosphate sorption characteristics of mineral amendments in relation to stabilising high legacy P calcareous soil. Environ. Pollut. 2020, 261, 114175. [Google Scholar] [CrossRef]
- Miyittah, M.K.; Stanley, C.D.; Mackowiak, C.; Rhue, D.R.; Rechcigl, J.E. Developing a remediation strategy for phosphorus immobilization: Effect of co-blending, Al-residual and Ca-Mg amendments in a manure-impacted spodosol. Soil Sediment Contam. 2011, 20, 337–352. [Google Scholar] [CrossRef]
- Fan, B.; Ding, J.; Fenton, O.; Daly, K.; Chen, S.; Zhang, S.; Chen, Q. Investigation of differential levels of phosphorus fixation in dolomite and calcium carbonate amended red soil. J. Sci. Food Agric. 2021, 102, 740–749. [Google Scholar] [CrossRef]
- Liu, W.; Ji, H.; Kerr, P.; Wu, Y.; Fang, Y. The application of soil amendments benefits to the reduction of phosphorus depletion and the growth of cabbage and corn. Environ. Sci. Pollut. Res. 2015, 22, 16772–16780. [Google Scholar] [CrossRef]
- Brennan, R.B.; Murnane, J.G.; Sharpley, A.N.; Herron, S.; Brye, K.R.; Simmons, T. Soil phosphorus dynamics following land application of unsaturated and partially saturated red mud and water treatment residuals. J. Environ. Manag. 2019, 248, 109296. [Google Scholar] [CrossRef] [PubMed]
- Lin, J.; Kim, M.; Li, D.; Kim, H.; Huang, C. The removal of phosphate by thermally treated red mud from water: The effect of surface chemistry on phosphate immobilization. Chemosphere 2020, 247, 125867. [Google Scholar] [CrossRef] [PubMed]
- Fan, B.; Wang, J.; Fenton, O.; Daly, K.; Ezzati, G.; Chen, Q. Strategic differences in phosphorus stabilization by alum and dolomite amendments in calcareous and red soils. Environ. Sci. Pollut. Res. 2019, 26, 4842–4854. [Google Scholar] [CrossRef] [PubMed]
- Uusi-Kämppä, J.; Turtola, E.; Närvänen, A.; Jauhiainen, L.; Uusitalo, R. Phosphorus mitigation during springtime runoff by amendments applied to grassed soil. J. Environ. Qual. 2012, 41, 420–426. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Callery, O.; Brennan, R.; Healy, M. Use of amendments in a peat soil to reduce phosphorus losses from forestry operations. Ecol. Eng. 2015, 85, 193–200. [Google Scholar] [CrossRef] [Green Version]
- He, X.Y.; Zhang, T.; Niu, Y.Q.; Xue, Q.; Ali, E.F.; Shaheen, S.M.; Tsang DC, W.; Rinklebe, J. Impact of catalytic hydrothermal treatment and Ca/Al-modified hydrochar on lability, sorption, and speciation of phosphorus in swine manure: Microscopic and spectroscopic investigations. Environ. Pollut. 2022, 299, 118877. [Google Scholar] [CrossRef]
- Bacelo, H.; Pintor, A.; Santos, S.; Boaventura, R.; Botelho, C. Performance and prospects of different adsorbents for phosphorus uptake and recovery from water. Chem. Eng. J. 2020, 381, 122566. [Google Scholar] [CrossRef]
- Lim, S.; Lee, D.; Kwak, J.; Park, H.; Kim, H.; Choi, W. Fly ash and zeolite amendments increase soil nutrient retention but decrease paddy rice growth in a low fertility soil. J. Soils Sediments 2016, 16, 756–766. [Google Scholar] [CrossRef]
- Li, X.; Xie, Q.; Chen, S.; Xing, M.; Guan, T.; Wu, D. Inactivation of phosphorus in the sediment of the Lake Taihu by lanthanum modified zeolite using laboratory studies. Environ. Pollut. 2019, 247, 9–17. [Google Scholar] [CrossRef]
- Wu, H.J.; Wang, S.; Gao, L.M.; Zhang, L.; Yuan, Z.W.; Fan, T.Y.; Wei, K.P.; Huang, L. Nutrient-derived environmental impacts in Chinese agriculture during 1978–2015. J. Environ. Manag. 2018, 217, 762–774. [Google Scholar] [CrossRef] [PubMed]
- Jalali, M.; Antoniadis, V. Impact of sewage sludge, nanoparticles, and clay minerals addition on cucumber growth, phosphorus uptake, soil phosphorus status, and potential risk of phosphorus loss. Environ. Technol. Innov. 2021, 23, 101702. [Google Scholar] [CrossRef]
- Moharami, S.; Jalali, M. Effect of time on the sorption and distribution of phosphorus in treated soil with minerals and nanoparticles. Environ. Earth Sci. 2015, 73, 8599–8608. [Google Scholar] [CrossRef]
- Irshad, M.; Hafeez, F.; Naseem, M.; Rizwan, M.; Al-Wabel, M. Effect of coal and wood ash on phosphorus immobilization in different textured soils. Arab. J. Geosci. 2018, 11, 536. [Google Scholar] [CrossRef]
- Faridullah; Malik, N.; Fareed, I.; Irshad, M. Reducing the leachability of nitrate, phosphorus and heavy metals from soil using waste material. Braz. J. Chem. Eng. 2017, 34, 715–726. [Google Scholar] [CrossRef]
- Udeigwe, T.; Wang, J.; Zhang, H. Effectiveness of bauxite residues in immobilizing contaminants in manure-amended soils. Soil Sci. 2009, 174, 676–686. [Google Scholar] [CrossRef]
- Zhang, L.; Chen, J.; Chu, G. Legacy phosphorus in calcareous soil under 33 years of P fertilizer application: Implications for efficient P management in agriculture. Soil Use Manag. 2022, 38, 1380–1393. [Google Scholar] [CrossRef]
- Fang, C.; Zhang, T.; Li, P.; Jiang, R.F.; Wu, S.B.; Nie, H.Y.; Wang, Y.C. Phosphorus recovery from biogas fermentation liquid by Ca-Mg loaded biochar. J. Environ. Sci. 2015, 29, 106–114. [Google Scholar] [CrossRef]
- Zhang, T.; Xu, H.Y.; Li, H.H.; He, X.Y.; Shi, Y.J.; Kruse, A. Microwave digestion-assisted HFO/biochar adsorption to recover phosphorus from swine manure. Sci. Total Environ. 2018, 621, 1512–1526. [Google Scholar] [CrossRef]
- Shaheen, S.M.; Natasha Mosa, A.; El-Naggar, A.; Hossain Md, F.; Abdelrahman, H.; Niazi, N.K.; Shahid, M.; Zhang, T.; Tsang, Y.F.; Trakal, L.; et al. Manganese oxide-modified biochar: Production, characterization and applications for the removal of pollutants from aqueous environments—A review. Bioresour. Technol. 2022, 346, 126581. [Google Scholar] [CrossRef]
- Chen, H.; Gao, Y.; Li, J.; Fang, Z.; Bolan, N.; Bhatnagar, A.; Gao, B.; Hou, D.; Wang, S.; Song, H.; et al. Engineered biochar for environmental decontamination in aquatic and soil systems: A review. Carbon Res. 2022, 1, 4. [Google Scholar] [CrossRef]
- Matin, N.H.; Jalali, M.; Antoniadis, V.; Shaheen, S.M.; Wang, J.; Zhang, T.; Wang, H.; Rinklebe, J. Almond and walnut shell-derived biochars affect sorption-desorption, fractionation, and release of phosphorus in two different soils. Chemosphere 2020, 241, 124888. [Google Scholar] [CrossRef]
- Peng, Y.; Sun, Y.; Fan, B.; Zhang, S.; Tsang DC, W. Fe/Al (hydr)oxides engineered biochar for reducing phosphorus leaching from a fertile calcareous soil. J. Clean. Prod. 2021, 279, 123877. [Google Scholar] [CrossRef]
- Xu, M.; Wu, J.; Yang, G.; Zhang, X.; Peng, H.; Yu, X.; Xiao, Y.; Qi, H. Biochar addition to soil highly increases P retention and decreases the risk of phosphate contamination of waters. Environ. Chem. Lett. 2019, 17, 533–541. [Google Scholar] [CrossRef]
- Niyungeko, C.; Liang, X.; Shan, S.; Zhang, J.; Tiimub, B.; SeyedHamid, H.; Khan, S.; Li, F.; Tian, G. Synergistic effects of anionic polyacrylamide and gypsum to control phosphorus losses from biogas slurry applied soils. Chemosphere 2019, 234, 953–961. [Google Scholar] [CrossRef] [PubMed]
- Liang, X.Q.; Liu, Z.W.; Liu, J.; Chen, L.L.; Tian, G.M. Soil colloidal P release potentials under various polyacrylamide addition levels. Land Degrad. Dev. 2017, 28, 2245–2254. [Google Scholar] [CrossRef]
- Cao, D.; Chen, W.; Yang, P.; Lan, Y.; Sun, D. Spatio-temporal variabilities of soil phosphorus pool and phosphorus uptake with maize stover biochar amendment for 5 years of maize. Environ. Sci. Pollut. Res. 2020, 27, 36350–36361. [Google Scholar] [CrossRef]
- Li, F.; Jin, Y.; He, S.; Jin, J.; Wang, Z.; Khan, S.; Tian, G.; Liang, X. Use of polyacrylamide modified biochar coupled with organic and chemical fertilizers for reducing phosphorus loss under different cropping systems. Agric. Ecosyst. Environ. 2021, 310, 107306. [Google Scholar] [CrossRef]
- Motaghian, H.; Hosseinpur, A.; Safian, M. The effects of sugarcane-derived biochar on phosphorus release characteristics in a calcareous soil. J. Soil Sci. Plant Nutr. 2020, 20, 66–74. [Google Scholar] [CrossRef]
- Zhang, Q.; Song, Y.; Wu, Z.; Yan, X.; Gunina, A.; Kuzyakov, Y.; Xiong, Z. Effects of six-year biochar amendment on soil aggregation, crop growth, and nitrogen and phosphorus use efficiencies in a rice-wheat rotation. J. Clean. Prod. 2020, 242, 118435. [Google Scholar] [CrossRef]
- Mukherjee, S.; Mavi, M.; Singh, J. Differential response of biochar derived from rice-residue waste on phosphorus availability in soils with dissimilar pH. Int. J. Environ. Sci. Technol. 2020, 17, 3065–3074. [Google Scholar] [CrossRef]
- Wang, Y.; Zhang, Y.; Zhao, H.; Hu, W.; Zhang, H.; Zhou, X.; Luo, G. The effectiveness of reed-biochar in mitigating phosphorus losses and enhancing microbially-driven phosphorus dynamics in paddy soil. J. Environ. Manag. 2022, 314, 115087. [Google Scholar] [CrossRef] [PubMed]
- Vanek, S.; Lehmann, J. Phosphorus availability to beans via interactions between mycorrhizas and biochar. Plant Soil 2015, 395, 105–123. [Google Scholar] [CrossRef]
- Fang, C.; Zhang, T.; Li, P.; Jiang, R.F.; Wang, Y.C. Application of magnesium modified corn biochar for phosphorus removal and recovery from Swine wastewater. Int. J. Environ. Res. Public Health 2014, 11, 9217–9237. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- He, X.M.; Zhang, T.; Ren, H.Q.; Li, G.X.; Ding, L.L.; Pawlowski, L. Phosphorus recovery from biogas slurry by ultrasound/H2O2 digestion coupled with HFO/biochar adsorption process. Waste Manag. 2017, 60, 219–229. [Google Scholar] [CrossRef] [PubMed]
- Deng, Y.X.; Zhang, T.; Sharma, B.K.; Nie, H.Y. Optimization and mechanism studies on cell disruption and phosphorus recovery from microalgae with magnesium modified hydrochar in assisted hydrothermal system. Sci. Total Environ. 2019, 646, 1140–1154. [Google Scholar] [CrossRef]
- Chen, Q.; Qin, J.; Cheng, Z.; Huang, L.; Sun, P.; Shen, G. Synthesis of a stable magnesium-impregnated biochar and its reduction of phosphorus leaching from soil. Chemosphere 2018, 199, 402–408. [Google Scholar] [CrossRef]
- Zhao, D.; Luo, Y.; Feng, Y.; He, Q.; Zhang, L.; Zhang, K.; Wang, F. Enhanced adsorption of phosphorus in soil by lanthanum-modified biochar: Improving phosphorus retention and storage capacity. Environ. Sci. Pollut. Res. 2021, 28, 68982–68995. [Google Scholar] [CrossRef]
- Feng, Y.; Lu, H.; Liu, Y.; Xue, L.; Dionysiou, D.; Yang, L.; Xing, B. Nano-cerium oxide functionalized biochar for phosphate retention: Preparation, optimization and rice paddy application. Chemosphere 2017, 185, 816–825. [Google Scholar] [CrossRef]
- Peng, L.; Xue, X.; Tang, Q.; Zhu, Y.; Xiao, L.; Yang, Y.; Lin, Q. Phosphorus retention and loss in three types of soils with implications for geographical pattern of eutrophication in china. Water Environ. J. 2020, 34, 9–18. [Google Scholar] [CrossRef]
- Mng’Ong’o, M.; Munishi, L.K.; Blake, W.; Ndakidemi, P.A.; Comber, S.; Hutchinson, T.H. Characterization of soil phosphate status, sorption and saturation in paddy wetlands in usangu basin-tanzania. Chemosphere 2021, 278, 130466. [Google Scholar] [CrossRef] [PubMed]
- Sun, Q.; Hu, Y.; Chen, X.; Wei, X.; Shen, J.; Ge, T.; Su, Y. Flooding and straw returning regulates the partitioning of soil phosphorus fractions and phoD-harboring bacterial community in paddy soils. Appl. Microbiol. Biotechnol. 2021, 105, 9343–9357. [Google Scholar] [CrossRef] [PubMed]
- Yang, W.; Xia, Y.; Jiang, X.; Yan, X. The influence factors and loss of total phosphorus runoff loss are estimated. J. Agric. Environ. Sci. 2015, 4, 319–325. [Google Scholar]
- Xu, X.; Mao, X.; Van Zwieten, L.; Niazi, N.K.; Wang, H.L. Wetting-drying cycles during a rice-wheat crop rotation rapidly (im)mobilize recalcitrant soil phosphorus. J. Soils Sediments 2020, 20, 3921–3930. [Google Scholar] [CrossRef]
- Zhang, S.; Yang, X.; Hsu, L.; Liu, Y.; Wang, S.; White, J.R.; Shaheen, S.M.; Chen, Q.; Rinklebe, J. Soil acidification enhances the mobilization of phosphorus under anoxic conditions in an agricultural soil: Investigating the potential for loss of phosphorus to water and the associated environmental risk. Sci. Total Environ. 2021, 793, 148531. [Google Scholar] [CrossRef]
- Shaheen, S.M.; Wang, J.; Baumann, K.; Wang, S.L.; Rinklebe, J. Redox-induced mobilization of phosphorus in groundwater affected arable soil profiles. Chemosphere 2021, 275, 129928. [Google Scholar] [CrossRef]
- Zhu, S.J.; Gui, S.B.; Xu, W.; Xiang, P.; Meng, W.; Zhu, J.; Li, K.K. Experimental study on the tolerance of four species of water-holding plants to high concentrations of nitrogen and phosphorus pollution. China Rural Water Hydropower 2022, 2, 34–38. [Google Scholar]
- Wu, H.; Chen, D.J. Progress in the study of legacy effects of non-point source phosphorus pollution in watersheds. J. Agric. Environ. Sci. 2022, 41, 2352–2364. [Google Scholar]
- Cui, J.; Du, Y.; Ding, C.C.; Li, J.F.; Gao, F.S.; Chang, Y.J.; Zhang, J.B.; Liu, X.J.; Yao, D.R. Fugitive patterns of phosphorus in Chinese lake waters and progress of pollution control measures. J. Ecol. Environ. 2022, 31, 621–633. [Google Scholar]

| Ca/Mg Inorganic Materials | Soil Type | pH | Addition Amount (w/w) | Retention Situation | References |
|---|---|---|---|---|---|
| calcium carbonate | red soil | 5.42 | 2.0% | the soil Olsen-P contents increased by 33.9% | [48] |
| dolomite | red soil | 5.42 | 2.0% | the soil Olsen-P contents increased by 66.3% | [48] |
| dolomite | Calcareous soil | 7.56 | 2.0% | the soil CaCl2-P content was reduced by 57.8% | [48] |
| dolomite | Calcareous soil | 7.6 | 5.0% | soil available P decreased by 3.10% | [49] |
| dolomite | Calcareous soil | 7.9 | 1.25% | the total dissolved P of leachate decreased by 68.4% | [50] |
| magnesia | Sandy | 7.1 | 2.0% | soluble P was reduced by 78.6% | [51] |
| Fe/Al Inorganic Materials | Soil Type | pH | Addition Amount (w/w) | Retention Situation | References |
|---|---|---|---|---|---|
| alum | Calcareous soil | 7.56 | 2.0% | the soil CaCl2-P content was reduced by 77.0% | [48] |
| alum | red soil | 6.04 | 2.0% | the soil CaCl2-P content was reduced by 93.8% | [48] |
| alum | 7.6 | 5.0% | the soil CaCl2-P content was reduced by 91.9% | [49] | |
| aluminum sulfate | red soil | 7.25 | 0.2% | the total dissolved P of leachate decreased by 80.6% | [55] |
| ferrous sulfate | red soil | 7.25 | 0.2% | the total dissolved P of leachate decreased by 80.6% | [55] |
| Al-WTR | peat soil | 3.8 | 10% | P adsorption maxima was increased by 11% | [56] |
| Clay Mineral Inorganic Materials | Soil Type | pH | Addition Amount (w/w) | Retention Situation | References |
|---|---|---|---|---|---|
| Mg-Al LDHs | 7.08 | 2% | the P effluent mass balance decreased 82.7% | [28] | |
| natural zeolite | inceptisol | 6.4 | 5% | P was removed from the solution by 3.6% | [59] |
| lanthanum modified zeolite | aquatic soil | 7.9 | the soluble reactive P in decreased by 86.9% | [60] | |
| CFL-Z | aquatic soil | the P content in overlying water was reduced by 97.3% | [61] | ||
| montmorillonite | sandy clay loam | 7.8 | 1% | the calcium chloride-extractable P content was reduced by 62.8% | [62] |
| zeolite | sandy clay loam | 7.8 | 3% | the water-extractable P content was reduced by 9.9% | [62] |
| vermiculite | sandy clay loam | 7.8 | 3% | the Olsen-extractable P content was reduced by 79.8% | [62] |
| bentonite | Sandy loam soil | 8.28 | 10% | P maximum sorption capacity increased by 42.7% | [63] |
| kaolinite | Sandy loam soil | 8.28 | 10% | P maximum sorption capacity increased by 77.5% | [63] |
| zeolite | Sandy loam soil | 8.28 | 5% | P maximum sorption capacity increased by 70.0% | [63] |
| Waste Inorganic Materials | Soil Type | pH | Addition Amount (w/w) | Retention Situation | References |
|---|---|---|---|---|---|
| fly ash | inceptisol | 6.4 | 5% | P was removed from the solution by 97.0% | [59] |
| coal ash | loamy sand | 7.6 | 10% | water-soluble P was reduced by 22.3% | [64] |
| wood ash | sandy loam | 7.4 | 10% | water-soluble P was reduced by 16.5% | [64] |
| wood ash | silt loam | 7.6 | P concentration was reduced by 55.6% | [65] | |
| sawdust | sandy soil | 7.8 | P concentration was reduced by 58.1% | [65] | |
| bauxite residues | 4% | the water-extractable P was reduced 95% | [66] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ge, X.; Chen, X.; Liu, M.; Wang, C.; Zhang, Y.; Wang, Y.; Tran, H.-T.; Joseph, S.; Zhang, T. Toward a Better Understanding of Phosphorus Nonpoint Source Pollution from Soil to Water and the Application of Amendment Materials: Research Trends. Water 2023, 15, 1531. https://doi.org/10.3390/w15081531
Ge X, Chen X, Liu M, Wang C, Zhang Y, Wang Y, Tran H-T, Joseph S, Zhang T. Toward a Better Understanding of Phosphorus Nonpoint Source Pollution from Soil to Water and the Application of Amendment Materials: Research Trends. Water. 2023; 15(8):1531. https://doi.org/10.3390/w15081531
Chicago/Turabian StyleGe, Xiaofei, Xingyu Chen, Mingxin Liu, Chensi Wang, Yingyu Zhang, Yukai Wang, Huu-Tuan Tran, Stephen Joseph, and Tao Zhang. 2023. "Toward a Better Understanding of Phosphorus Nonpoint Source Pollution from Soil to Water and the Application of Amendment Materials: Research Trends" Water 15, no. 8: 1531. https://doi.org/10.3390/w15081531





