Phytoremediation Prospects for Restoration of Contamination in the Natural Ecosystems
Abstract
1. Introduction
2. Phytoremediation
3. Mechanism of the Phytoremediation Technique
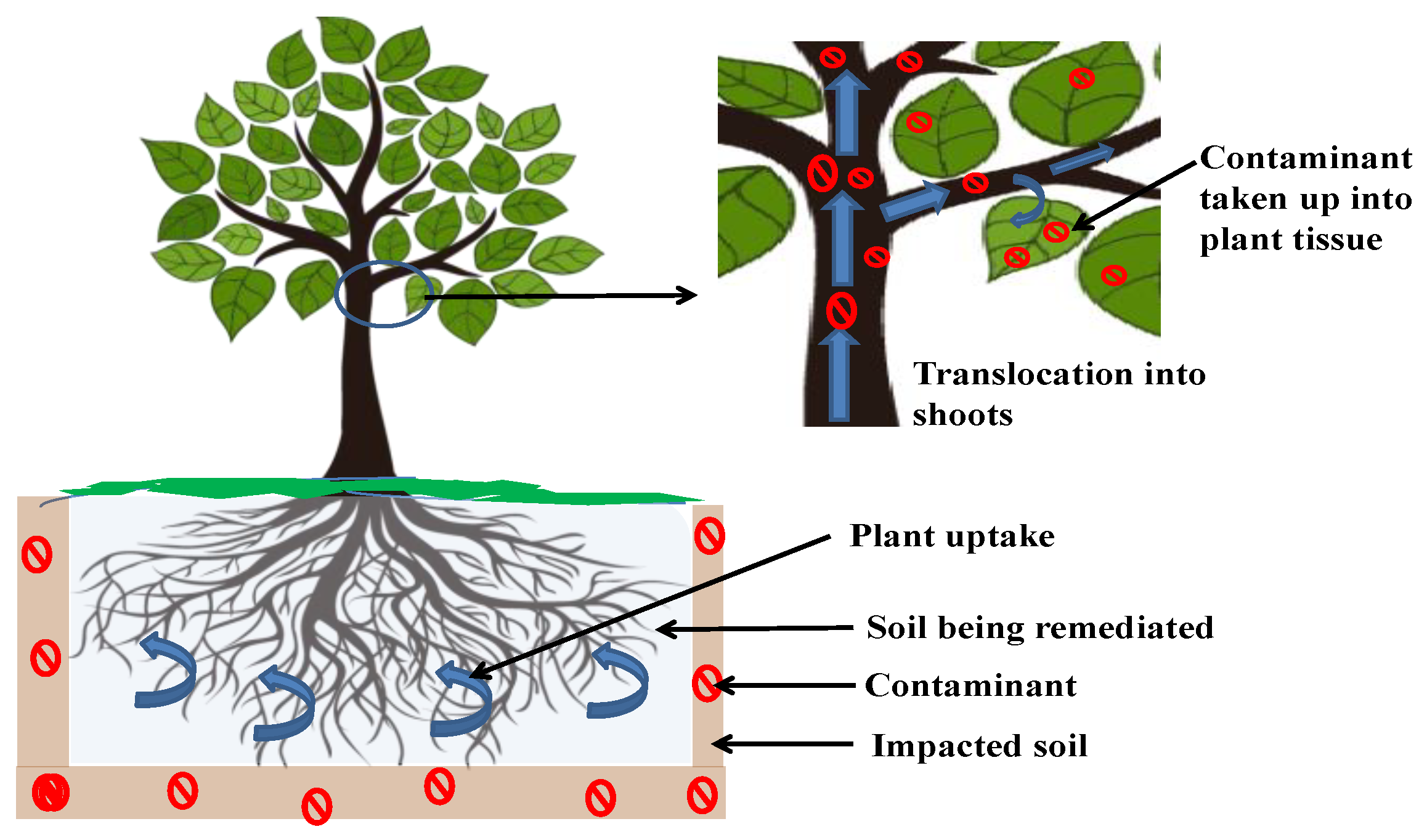
3.1. Phytoextraction
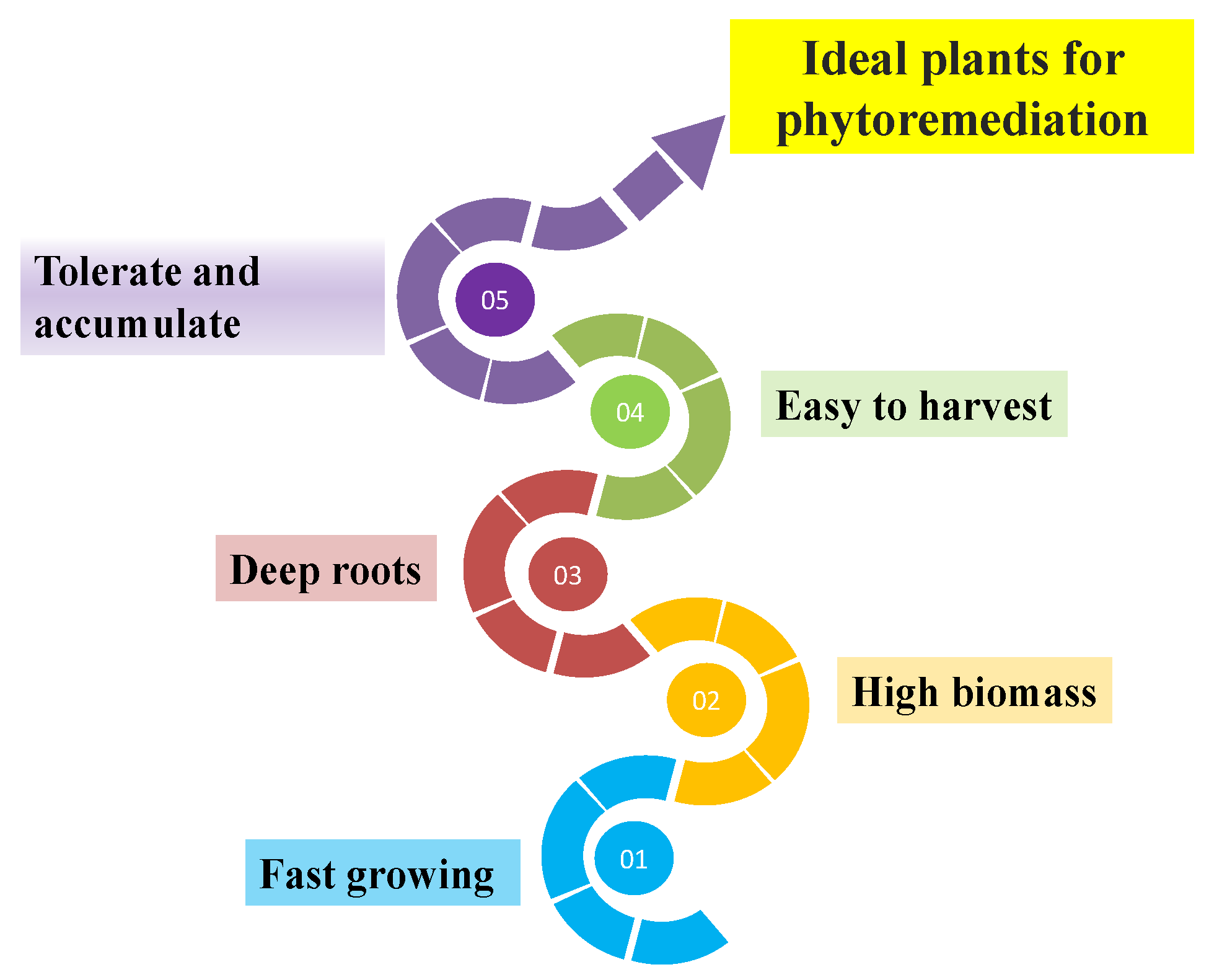
3.1.1. Plant Species
3.1.2. The Properties of the Medium
3.1.3. Root Zone
3.1.4. Environmental Conditions
3.1.5. Chemical Properties of Contaminants
3.1.6. Bioavailability of Metals
3.1.7. Chelating Agent Addition
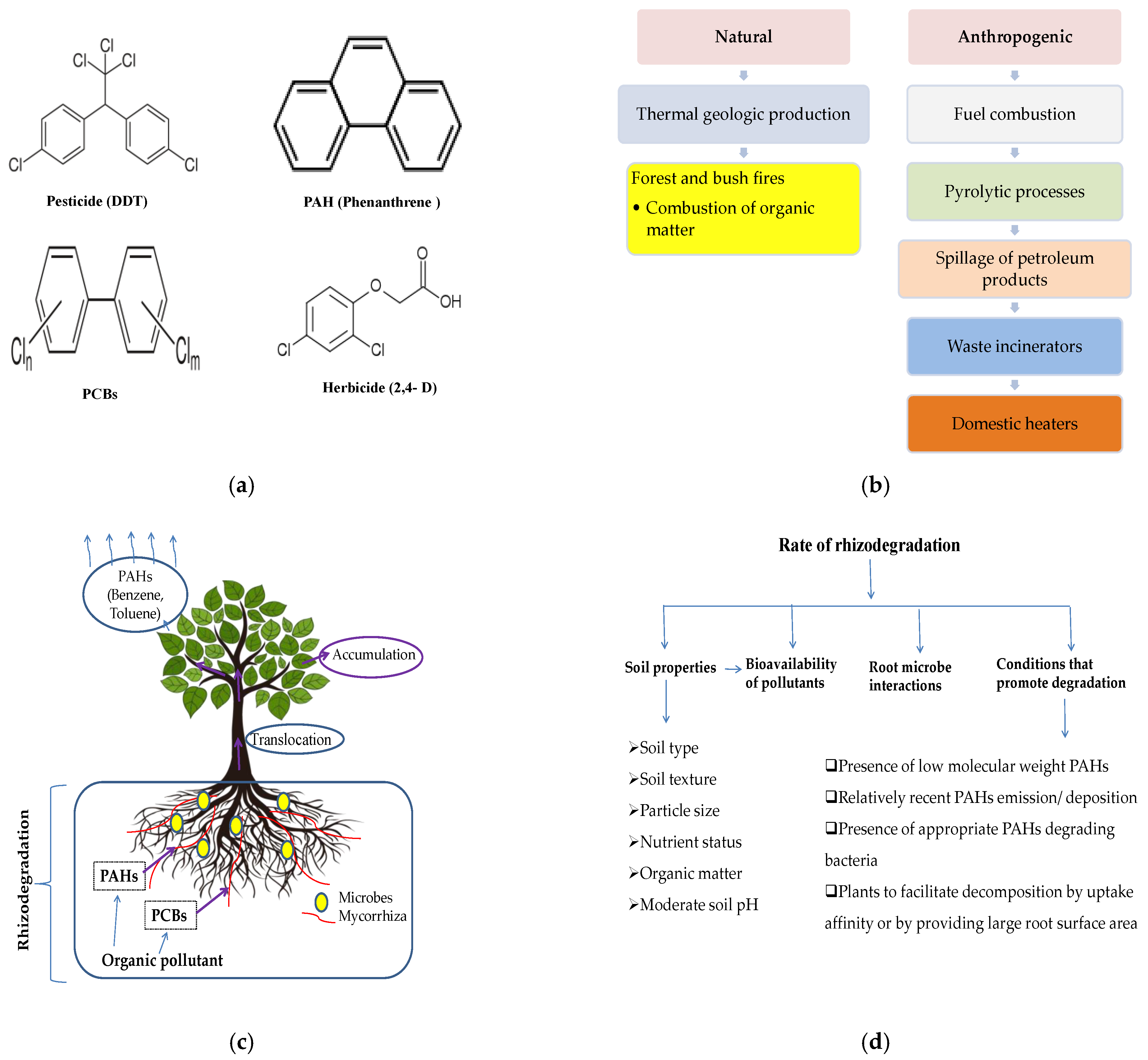
3.2. Rhizodegradation
3.3. Phytostabilization
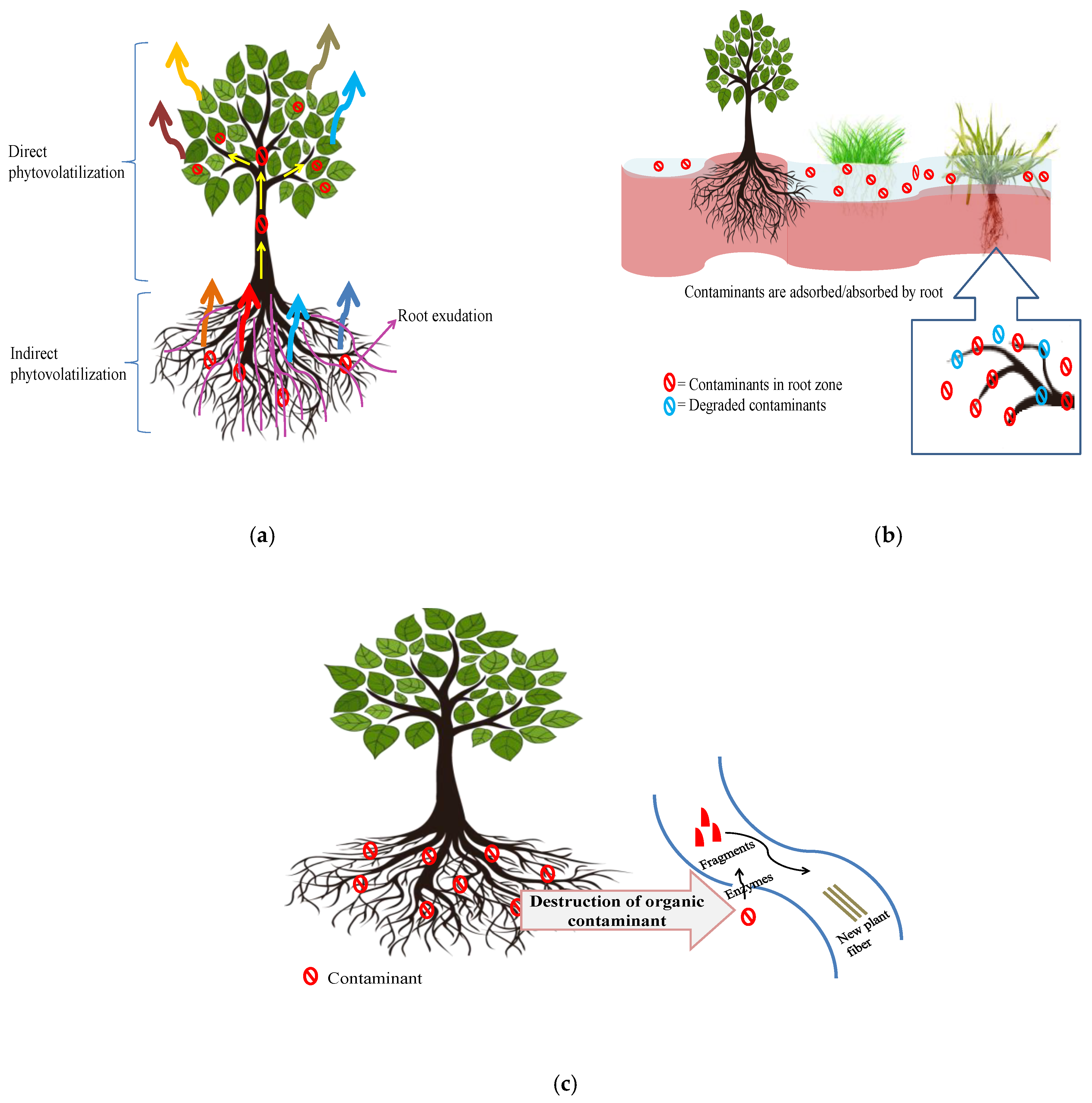
3.4. Phytovolatilization
3.5. Rhizofiltration
3.6. Phytodegradation
| Plant Species | Contaminant | References |
|---|---|---|
| Phytoevaporation | ||
| Arundo donax | AS | [96] |
| Astragalus racemosus | Se | [43] |
| Brassica napus | Se | [97] |
| Medicago sativa | Chlorinated solvents | [98] |
| Nicotiana tabacum | Hg | [99] |
| Salix spp. | Trichloroethylene (TCE), tetrachloroethylene (PCE) | [27] |
| Populus spp. | Trichloroethylene (TCE), tetrachloroethylene (PCE) | [27] |
| Taxodium distichum | Trichloroethylene (TCE) | [27] |
| Rhizofiltration | ||
| Arundo donax L. | Synthetic dye | [10] |
| Echinodorus cordifolius | Cd | [100] |
| Eichhornia crassipes (Mart.) Solms | Ni | [10] |
| Heliconia psittacorum | Zn | [100] |
| Iris pseuda-corus | Cr and Zn | [43] |
| Lepironia articulate | Pb | [43] |
| Pteris vittata | As | [43] |
| Raphanus sativus | U | [101] |
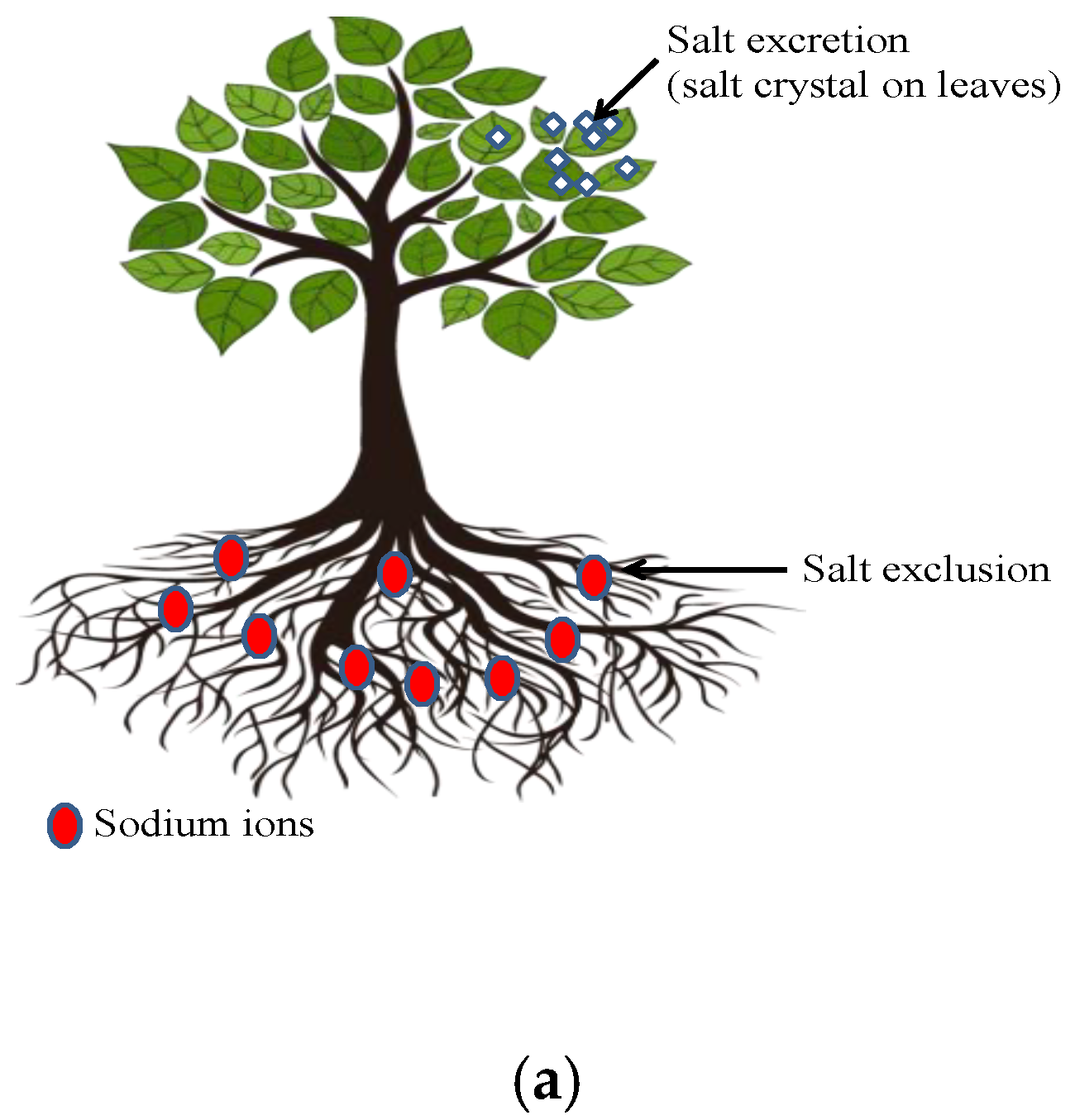
3.7. Phytodesalination
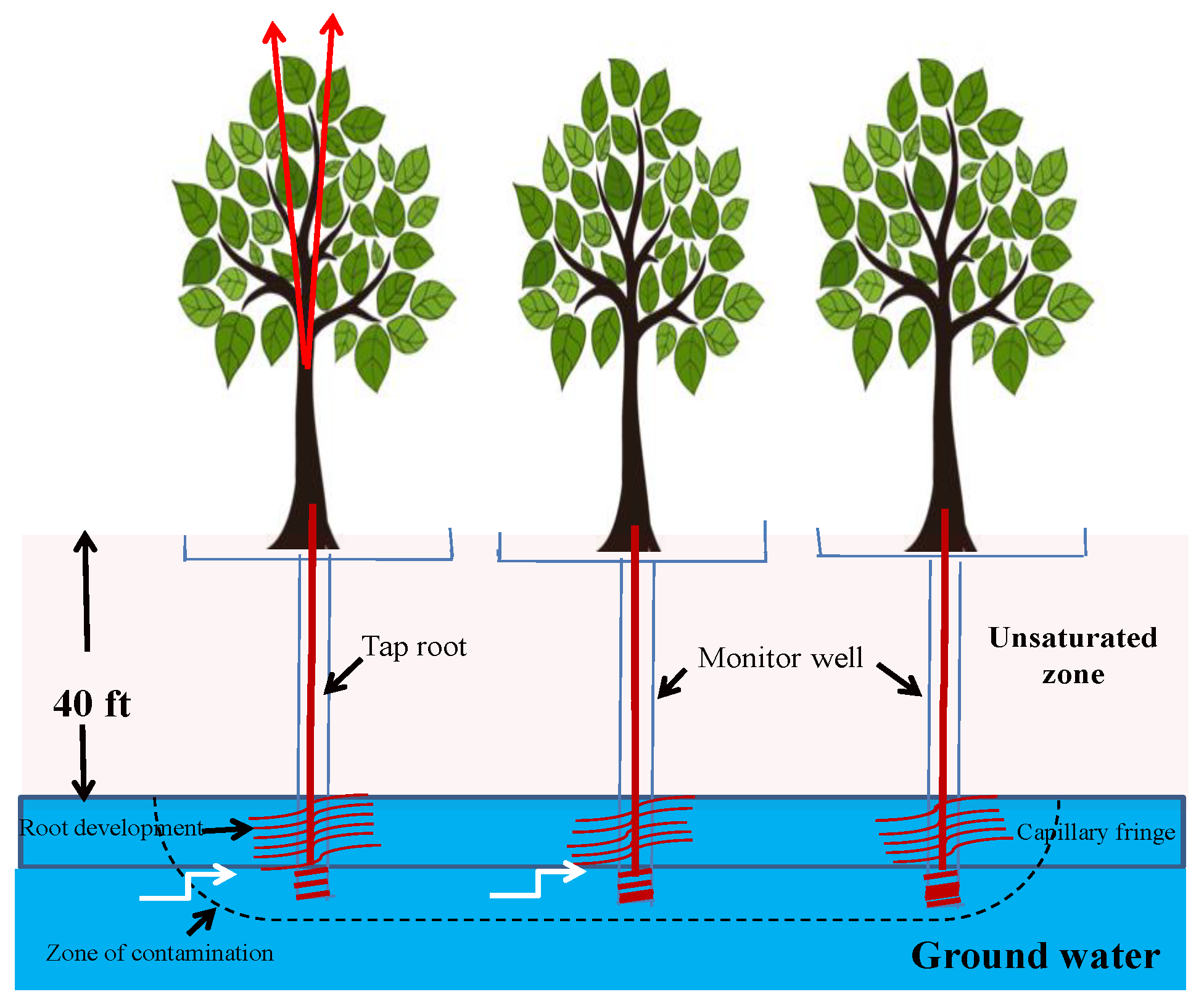
3.8. Phytohydraulics
4. Methods Used for Evaluation of Phytoremediation Potential
4.1. Biological Concentration Factor (BCF)
4.2. Bioaccumulation Coefficient (BAC)
4.3. Translocation Factor (TF)
5. Applications of Phytoremediation Technology
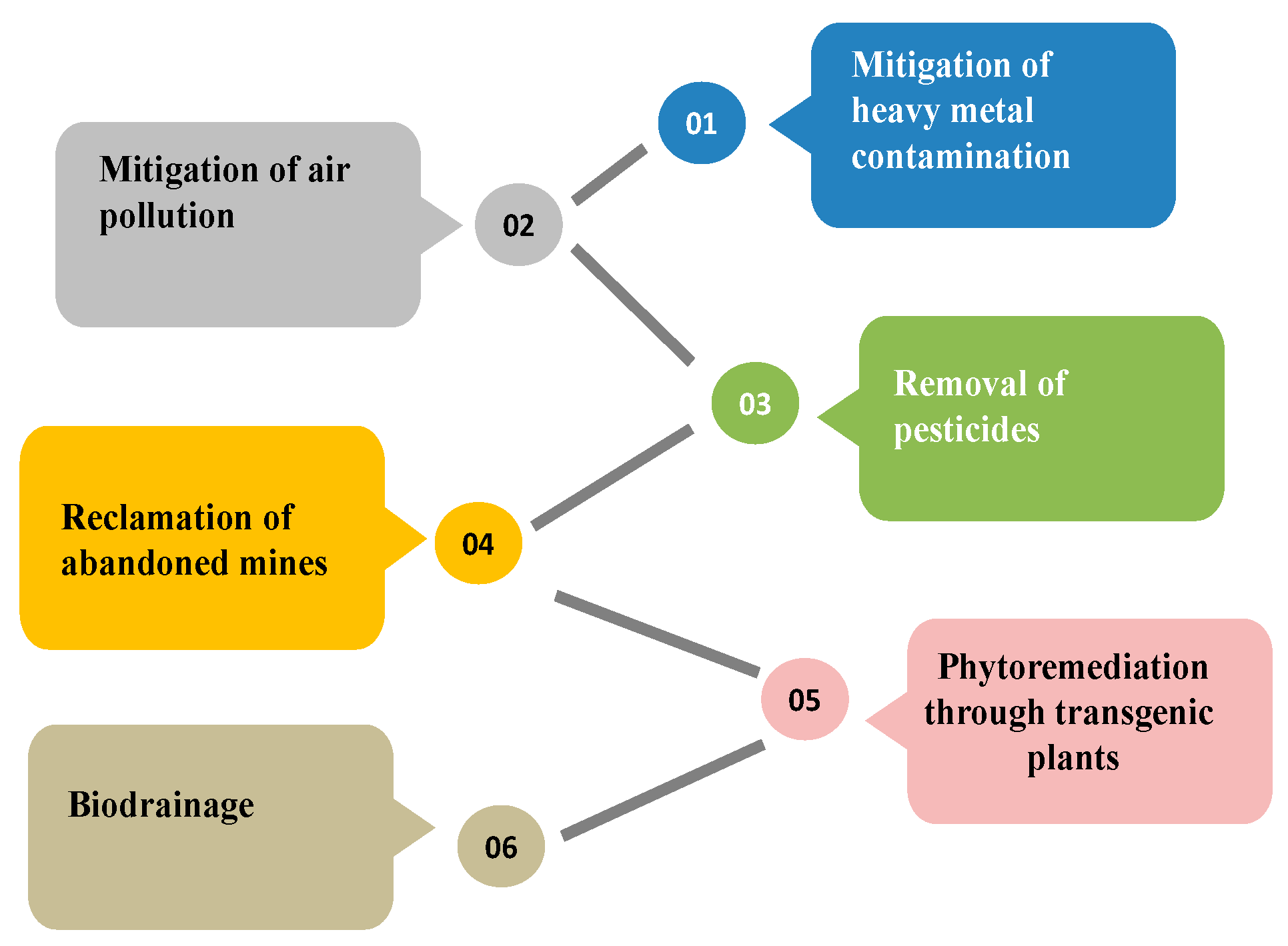
5.1. Mitigation of Heavy Metal Contamination
5.2. Bioremediation of Air Pollutants
5.3. Removal of Pesticides
5.4. Reclamation of Abandoned Mine Sites
5.5. Biodrainage
6. Advantages and Limitations of Phytoremediation
7. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Antoniadis, V.; Shaheen, S.M.; Levizou, E.; Shahid, M.; Niazi, N.K.; Vithanage, M.; Ok, Y.S.; Bolan, N.; Rinklebe, J. A critical prospective analysis of the potential toxicity of trace element regulation limits in soils worldwide: Are they protective concerning health risk assessment?—A review. Environ. Int. 2019, 127, 819–847. [Google Scholar] [CrossRef]
- Mehmood, M.A.; Ganie, S.A.; Bhat, R.A.; Rashid, A.; Rehman, S.; Dar, G.H.; Gulzar, A. Assessment of Some Trace Elements and Heavy Metals in river Jhelum of Kashmir Valley. Int. J. Theor. Appl. Sci. 2018, 10, 27–31. [Google Scholar]
- Rafique, N.; Tariq, S.R. Distribution and source apportionment studies of heavy metals in soil of cotton/wheat fields. Environ. Monit. Assess. 2016, 188, 309. [Google Scholar] [CrossRef]
- Iqbal, M.; Iqbal, N.; Bhatti, A.I.; Ahmad, N.; Zahid, M. Response surface methodology application in optimization of cadmium adsorption by shoe waste: A good option of waste mitigation by waste. Ecol. Eng. 2016, 88, 265–275. [Google Scholar] [CrossRef]
- Farahat, E.; Linderholm, H.W. The effect of long-term wastewater irrigation on accumulation and transfer of heavy metals in Cupressus sempervirens leaves and adjacent soils. Sci. Total Environ. 2015, 15, 1–7. [Google Scholar] [CrossRef]
- Shukla, K.P.; Sharma, S.; Kumar, N.; Singh, V.; Bisht, S.; Kumar, V. Rhizoremediation: A Promising Rhizosphere Technology. In Applied Bioremediation—Active and Passive Approaches; Patil, Y.B., Rao, P., Eds.; IntechOpen Limited: London, UK, 2013. [Google Scholar] [CrossRef]
- Mandal, A.; Purakayastha, T.J.; Ramana, S.; Neenu, S.; Bhaduri, D.; Chakraborty, K.; Manna, M.C.; Rao, A.S. Status on Phytoremediation of Heavy Metals in India—A Review. Int. J. Bio-Resour. Stress Manag. 2014, 5, 553–560. [Google Scholar] [CrossRef]
- Chen, B.; Stein, A.F.; Castell, N.; Gonzalez-Castanedo, Y.; Sanchez de la Campa, A.M.; de la Rosa, J.D. Modeling and evaluation of urban pollution events of atmospheric heavy metals from a large Cu-smelter. Sci. Total Environ. 2016, 1, 17–25. [Google Scholar] [CrossRef] [PubMed]
- Muradoglu, F.; Gundogdu, M.; Ercisli, S.; Encu, T.; Balta, F.; Jaafar, H.Z.E.; Zia-Ul-Haq, M. Cadmium toxicity affects chlorophyll a and b content, antioxidant enzyme activities and mineral nutrient accumulation in strawberry. Biol. Res. 2015, 48, 11. [Google Scholar] [CrossRef]
- Kristanti, R.A.; Ngu, W.J.; Yuniarto, A.; Hadibarata, T. Rhizofiltration for Removal of Inorganic and Organic Pollutants in Groundwater: A Review. Biointerface Res. Appl. Chem. 2021, 11, 12326–12347. [Google Scholar] [CrossRef]
- Fu, F.; Wang, Q. Removal of heavy metal ions from wastewaters: A review. J. Environ. Manag. 2011, 92, 407–418. [Google Scholar] [CrossRef]
- Ahmaruzzaman, M. Industrial wastes as low-cost potential adsorbents for the treatment of wastewater laden with heavy metals. Adv. Colloid Interface Sci. 2011, 10, 36–59. [Google Scholar] [CrossRef]
- Yaqoob, A.; Nasim, F.H.; Sumreen, A.; Munawar, N.; Zia, M.A.; Choudhary, M.S.; Ashraf, M. Current scenario of phytoremediation: Progresses and limitations. Int. J. Biosci. 2019, 14, 191–206. [Google Scholar] [CrossRef]
- Vamerali, T.; Bandiera, M.; Mosca, G. Field crops for phytoremediation of metal-contaminated land. A review. Environ. Chem. Lett. 2010, 8, 1–17. [Google Scholar] [CrossRef]
- Robinson, B.H.; Lombi, E.; Zhao, F.J.; Mcgrath, S.P. Uptake and distribution of nickel and other metals in the hyperaccumulator Berkheya Coddii. New Phytol. 2003, 158, 279–285. [Google Scholar] [CrossRef]
- Borisev, M.; Pajevic, S.; Nikolic, N.; Pilipovic, A.; Krstic, B.; Orlovic, S. Phytoextraction of Cd, Ni, and Pb Using Four Willow Clones (Salix spp.). Pol. J. Environ. Stud. 2009, 18, 553–561. [Google Scholar]
- Tripathi, V.; Edrisi, S.A.; Abhilash, P.C. Towards the coupling of phytoremediation with bioenergy production. Renew. Sustain. Energy Rev. 2016, 57, 1386–1389. [Google Scholar] [CrossRef]
- Bokhari, S.H.; Ahmad, I.; Mahmood-Ul-Hassan, M.; Mohammad, A. Phytoremediation potential of Lemna minor L. for heavy metals. Int. J. Phytoremediat. 2016, 18, 25–32. [Google Scholar] [CrossRef]
- Wani, K.A.; Sofi, Z.M.; Malik, J.A.; Wani, J.A. Phytoremediation of Heavy Metals Using Salix (Willows). In Bioremediation and Biotechnology; Bhat, R., Hakeem, K., Dervash, M., Eds.; Springer: Cham, Switzerland, 2020; p. 2. [Google Scholar] [CrossRef]
- Farraji, H.; Zaman, N.Q.; Tajuddin, R.; Faraji, H. Advantages and disadvantages of phytoremediation: A concise review. Int. J. Environ. Sci. Technol. (IJEST) 2016, 2, 69–75. [Google Scholar]
- Pilon-Smits, E. Phytoremediation. Annu. Reviewof. Plant Biol. 2005, 56, 15–39. [Google Scholar] [CrossRef]
- Cunningham, S.D.; Anderson, T.A.; Schwab, A.P.; Hsu, F.C. Phytoremediation of Soils Contaminated with Organic Pollutants. In Advances in Agronomy; Academic Press Inc.: Cambridge, MA, USA, 1996; Volume 56, pp. 55–114. [Google Scholar] [CrossRef]
- Ghosh, M.; Singh, S.P. A Review on Phytoremediation of Heavy Metals and Utilization of Its Byproducts. Appl. Ecol. Environ. Res. 2005, 3, 1–18. [Google Scholar] [CrossRef]
- Hutchinson, S.L.; Banks, M.K.; Schwab, A.P. Phytoremediation of aged petroleum sludge: Effect of inorganic fertilizer. J. Environ. Qual. 2001, 30, 395–403. [Google Scholar] [CrossRef]
- Joner, E.; Leyval, C. Phytoremediation of organic pollutants using mycorrhizal plants: A new aspect of rhizosphere interactions. Agronomie 2003, 23, 495–502. [Google Scholar] [CrossRef]
- Awa, S.H.; Hadibarata, T. Removal of Heavy Metals in Contaminated Soil by Phytoremediation Mechanism: A review. Water Air Soil Pollut. 2020, 231, 47. [Google Scholar] [CrossRef]
- Limmer, M.; Burken, J. Phytovolatilization of Organic Contaminants. Environ. Sci. Technol. 2016, 50, 6632–6643. [Google Scholar] [CrossRef] [PubMed]
- Bhat, S.A.; Bashir, O.; Haq, S.A.U.; Amin, T.; Rafiq, A.; Ali, M.; Americo-Pinheiro, J.H.P.; Sher, F. Phytoremediation of heavy metals in soil and water: An eco-friendly, sustainable and multidisciplinary approach. Chemosphere 2022, 303, 100774. [Google Scholar] [CrossRef]
- Kumar, A.; Kumar, A.; Mms, C.-P.; Chaturvedi, A.K.; Shabnam, A.A.; Subrahmanyam, G.; Mondal, R.; Gupta, D.K.; Malyan, S.K.; Kumar, S.S.; et al. Lead Toxicity: Health Hazards, Influence on Food Chain, and Sustainable Remediation Approaches. Int. J. Environ. Res. Public Health 2020, 17, 2179. [Google Scholar] [CrossRef] [PubMed]
- Tangahu, B.V.; Abdullah, S.R.S.; Basri, H.; Idris, M.; Anuar, N.; Mukhlisin, M. A Review on Heavy Metals (As, Pb, and Hg) Uptake by Plants through Phytoremediation. Int. J. Chem. Eng. 2011, 2011, 939161. [Google Scholar] [CrossRef]
- Babu, S.M.O.F.; Hossain, M.B.; Rahman, M.S.; Rahman, M.; Ahmed, A.S.S.; Hasan, M.M.; Rakib, A.; Emran, T.B.; Xiao, J.; Simal-Gandara, J. Phytoremediation of Toxic Metals: A Sustainable Green Solution for Clean Environment. Appl. Sci. 2021, 11, 10348. [Google Scholar] [CrossRef]
- Yan, A.; Wang, Y.; Tan, S.N.; Mohd –Yusof, M.L.; Ghosh, S.; Chen, Z. Phytoremediation: A Promising Approach for Revegetation of Heavy Metal-Polluted Land. Front. Plant Sci. 2020, 11, 359. [Google Scholar] [CrossRef]
- Finnegan, P.M.; Chen, W. Arsenic toxicity: The effects on plant metabolism. Front. Physiol. 2012, 6, 182. [Google Scholar] [CrossRef]
- Mandal, P. An insight of environmental contamination of arsenic on animal health. Emerg. Contam. 2017, 3, 17–22. [Google Scholar] [CrossRef]
- Han, J.; Park, H.; Kim, J.; Jeong, D.; Kang, J. Toxic effects of arsenic on growth, hematological parameters, and plasma components of starry flounder, Platichthys stellatus, at two water temperature conditions. Fish. Aquat. Sci. 2019, 22, 3. [Google Scholar] [CrossRef]
- Kumar, A.; Subrahmanyam, G.; Mondal, R.; Cabral-Pinto, M.; Shabnam, A.A.; Jigyasu, D.K.; Malyan, S.K.; Fagodiya, R.K.; Khan, S.A.; Yu, Z.-G. Bio-remediation approaches for alleviation of cadmium contamination in natural resources. Chemosphere 2021, 268, 128855. [Google Scholar] [CrossRef] [PubMed]
- Environmental Protection Agency (EPA). Aquatic Life Ambient Water Quality Criteria Update for Cadmium—2016; U.S. Environmental Protection Agency Office of Water Office of Science and Technology: Washington, DC, USA, 2016. Available online: http://www.epa.gov/wqc/aquatic-life-criteria-cadmium (accessed on 11 December 2022).
- Genchi, G.; Sinicropi, M.S.; Lauria, G.; Carocci, A.; Catalano, A. The Effects of Cadmium Toxicity. Int. J. Environ. Res. Public Health 2020, 26, 3782. [Google Scholar] [CrossRef] [PubMed]
- Haider, F.U.; Liqun, C.; Coulter, J.A.; Cheema, S.A.; Wu, J.; Zhang, R.; Wenjun, M.; Farooq, M. Cadmium toxicity in plants: Impacts and remediation strategies. Ecotoxicol. Environ. Saf. 2011, 211, 111887. [Google Scholar] [CrossRef]
- Outridge, P.M.; Scheuhammer, A.M. Bioaccumulation and toxicology of chromium: Implications for wildlife. Rev. Environ. Contam. Toxicol. 1993, 130, 31–77. [Google Scholar] [CrossRef]
- Aslam, S.; Yousafzai, A.M. Chromium toxicity in fish: A review article. J. Entomol. Zool. Stud. 2017, 5, 1483–1488. [Google Scholar]
- Kapoor, R.T.; Bani- Mfarrej, M.F.; Alam, P.; Rinklebe, J.; Ahmad, P. Accumulation of chromium in plants and its repercussion in animals and humans. Environ. Pollut. 2022, 15, 119044. [Google Scholar] [CrossRef] [PubMed]
- Wani, Z.A.; Ahmad, Z.; Asgher, M.; Bhat, J.A.; Sharma, M.; Kumar, A.; Sharma, V.; Kumar, A.; Pant, S.; Lukatkin, A.S.; et al. Phytoremediation of Potentially Toxic Elements: Role, Status and Concerns. Plants 2023, 12, 429. [Google Scholar] [CrossRef]
- Wang, Z.; He, N.; Wang, Y.; Zhang, J. Effects of Copper on Organisms: A Review. Adv. Mater. Res. 2013, 726–731, 340–343. [Google Scholar] [CrossRef]
- Environmental Protection Agency (EPA). Aquatic Life Ambient Freshwater Quality Criteria—Copper 2007 Revision; U.S. Environmental Protection Agency Office of Water Office of Science and Technology: Washington, DC, USA, 2007.
- Blakley, B.R. Lead Poisoning in Animals. In Toxicology—MSD Veterinary Manual; Saskatoon, SK, Canada, 2022; pp. 1–5. Available online: https://www.msdvetmanual.com/toxicology/lead-poisoning/lead-poisoning-in-animals (accessed on 27 January 2023).
- Milatovic, D.; Gupta, R.C. Manganese. In Veterinary Toxicology, 3rd ed.; Gupta, R.C., Ed.; Academic Press: Cambridge, MA, USA, 2018; pp. 445–454. [Google Scholar] [CrossRef]
- Rodrigues, G.Z.P.; de Souza, M.S.; Gehlen, G. Impacts Caused by Manganese in the Aquatic Environments of Brazil. In Pollution of Water Bodies in Latin America; Gomez-Olivan, L., Ed.; Springer: Cham, Switzerland, 2019. [Google Scholar] [CrossRef]
- Zheng, N.; Wang, S.; Dong, W.; Hua, X.; Li, Y.; Song, X.; Chu, Q.; Hou, S.; Li, Y. The Toxicological Effects of Mercury Exposure in Marine Fish. Bull Environ. Contam. Toxicol. 2019, 102, 714–720. [Google Scholar] [CrossRef]
- Gworek, B.; Dmuchowski, W.; Baczewska-Dąbrowska, A.H. Mercury in the terrestrial environment: A review. Environ. Sci. Eur. 2020, 32, 1–19. [Google Scholar] [CrossRef]
- Bittner, F. Molybdenum metabolism in plants and crosstalk to iron. Front. Plant Sci. 2014, 5, 28. [Google Scholar] [CrossRef]
- Novotny, J.A.; Peterson, C.A. Molybdenum. Adv. Nutr. 2018, 9, 272–273. [Google Scholar] [CrossRef] [PubMed]
- Al-Attar, A.M. The Influences of Nickel Exposure on Selected Physiological Parameters and Gill Structure in the Teleost Fish, Oreochromis niloticus. J. Biol. Sci. 2007, 7, 77–85. Available online: https://scialert.net/abstract/?doi=jbs.2007.77.85 (accessed on 12 November 2022). [CrossRef]
- Kumar, A.; Jigyasu, D.K.; Subrahmanyam, G.; Mondal, R.; Shabnam, A.A.; Cabral-Pinto, M.; Malyan, S.K.; Chaturvedi, A.K.; Gupta, D.K.; Fagodiya, R.K.; et al. Nickel in terrestrial biota: Comprehensive review on contamination, toxicity, tolerance and its remediation approaches. Chemosphere 2021, 275, 129996. [Google Scholar] [CrossRef] [PubMed]
- Hassan, M.U.; Chattha, M.U.; Khan, I.; Chattha, M.B.; Aamer, M.; Nawaz, M.; Ali, A.; Khan, M.A.U.; Khan, T.A. Nickel toxicity in plants: Reasons, toxic effects, tolerance mechanisms, and remediation possibilities—A review. Environ. Sci. Pollut. Res. Int. 2019, 26, 12673–12688. [Google Scholar] [CrossRef] [PubMed]
- Plum, L.M.; Rink, L.; Haase, H. The essential toxin: Impact of zinc on human health. Int. J. Environ. Res. Public Health 2010, 7, 1342–1365. [Google Scholar] [CrossRef]
- Kaur, H.; Garg, N. Zinc toxicity in plants: A review. Planta 2021, 253, 129. [Google Scholar] [CrossRef]
- Mehes-Smith, M.; Nkongolo, K.; Cholew, E. Coping Mechanisms of Plants to Metal Contaminated Soil. In Environmental Change and Sustainability; Silvern, S., Young, S., Eds.; Intech Open: London, UK, 2013. [Google Scholar] [CrossRef]
- Baker, A.J.M.; Walker, P.L. Ecophysiology of metal uptake by tolerant plants: Heavy metal tolerance in plants. In Evolutionary Aspects; Shaw, A.J., Ed.; CRC Press: Boca Raton, FL, USA, 1990. [Google Scholar]
- Mganga, N.; Manoko, M.L.K.; Rulangaranga, Z.K. Classification of Plants According to Their Heavy Metal Content around North Mara Gold Mine, Tanzania: Implication for Phytoremediation. Tanzan. J. Sci. 2011, 37, 109–119. [Google Scholar]
- Cunningham, S.D.; Ow, D.W. Promises and Prospects of Phytoremediation. Plant Physiol. 1996, 110, 715–719. [Google Scholar] [CrossRef] [PubMed]
- Rascio, N.; Navari-Izzo, F. Heavy Metal Hyperaccumulating Plants: How and Why Do They Do It? And What Makes Them So Interesting? Plant Sci. 2011, 180, 169–181. [Google Scholar] [CrossRef]
- Bian, X.; Cui, J.; Tang, B.; Yang, L. Chelant-Induced Phytoextraction of Heavy Metals from Contaminated Soils: A Review. Pol. J. Environ. Stud. 2018, 27, 2417–2424. [Google Scholar] [CrossRef]
- Bolan, N.S.; Park, J.H.; Robinson, B.; Naidu, R.; Huh, K.Y. Phytostabilization: A Green Approach to Contaminant Containment. In Advances in Agronomy; Sparks, D.L., Ed.; Academic Press: Cambridge, MA, USA, 2011; Volume 112, pp. 145–204. [Google Scholar] [CrossRef]
- Singh, S.; Karwadiya, J.; Srivastava, S.; Patra, P.K.; Venugopalan, V.P. Potential of indigenous plant species for phytoremediation of arsenic contaminated water and soil. Ecol. Eng. 2022, 175, 106476. [Google Scholar] [CrossRef]
- Patel, S.J.; Bhattacharya, P.; Banu, S.; Bai, L.; Namratha. Phytoremediation of Copper and Lead by Using Sunflower, Indian Mustard and Water Hyacinth Plants. Int. J. Sci. Res. (IJSR) 2015, 4, 113–115. [Google Scholar]
- Bhattacharya, E.; Biswas, S.M. First Report of the Hyperaccumulating Potential of Cadmium and Lead by Cleome rutidosperma DC. With a Brief Insight into the Chemical Vocabulary of its Roots. Front. Environ. Sci. 2022, 10, 87. [Google Scholar] [CrossRef]
- Reddy, L.C.S.; Reddy, K.V.R.; Humane, S.K.; Damodaram, B. Accumulation of Chromium in Certain plant Species Growing on Mine Dump from Byrapur, Karnataka, India. Res. J. Chem. Sci. 2012, 2, 17–20. [Google Scholar]
- Kumari, P.; Kumar, P.; Kumar, T. An overview of phytomining: A metal extraction process from plant species. J. Emerg. Technol. Innov. Res. (JETIR) 2019, 6, 1367–1376. [Google Scholar]
- Capuana, M. A review of the performance of woody and herbaceous ornamental plants for phytoremediation in urban areas. Iforest—Biogeosci. For. 2020, 13, 139–151. [Google Scholar] [CrossRef]
- Malik, J.A.; Wani, A.A.; Wani, K.A.; Bhat, M.A. Role of White Willow (Salix alba L.) for Cleaning Up the Toxic Metal Pollution. In Bioremediation and Biotechnology; Hakeem, K., Bhat, R., Qadri, H., Eds.; Springer: Cham, Switzerland, 2020. [Google Scholar] [CrossRef]
- Kumar, A.; Mishra, S.; Pandey, R.; Yu, Z.G.; Kumar, M.; Khoo, K.S.; Thakur, T.K.; Show, P.L. Microplastics in terrestrial ecosystems: Un-ignorable impacts on soil characterises, nutrient storage and its cycling. TrAC Trends Anal. Chem. 2023, 158, 116869. [Google Scholar] [CrossRef]
- Lee, S.; Ka, J.O.; Gyu, S.H. Growth promotion of Xanthium italicum by application of rhizobacterial isolates of Bacillus aryabhattai in microcosm soil. J. Microbiol. 2012, 50, 45–49. [Google Scholar] [CrossRef] [PubMed]
- Pimviriyakul, P.; Wongnate, T.; Tinikul, R.; Chaiyen, P. Microbial degradation of halogenated aromatics: Molecular mechanisms and enzymatic reactions. Microb. Biotechnol. 2020, 13, 67–86. [Google Scholar] [CrossRef] [PubMed]
- Sood, S.; Sharma, A.; Sharma, N.; Kanwar, S.S. Carboxylesterases: Sources, Characterization and Broader Applications. Insights Enzyme Res. 2016, 1, 1–11. [Google Scholar] [CrossRef]
- Raunio, H.; Kuusisto, M.; Juvonen, R.O.; Pentikäinen, O.T. Modeling of interactions between xenobiotics and cytochrome P450 (CYP) enzymes. Front. Pharmacol. 2015, 6, 123. [Google Scholar] [CrossRef] [PubMed]
- Lee, M.D.; Odom, J.M.; Buchanan, R.J. New perspectives on microbial dehalogenation of chlorinated solvents: Insights from the field. Annu. Rev. Microbiol. 1998, 52, 423–452. [Google Scholar] [CrossRef]
- Akram, M.S.; Rashid, N.; Basheer, S. Physiological and molecular basis of plants tolerance to linear halogenated hydrocarbons. In Handbook of Bioremediation; Hasanuzzaman, M., Prasad, M.N.V., Eds.; Academic Press: Cambridge, MA, USA, 2021; pp. 591–602. [Google Scholar] [CrossRef]
- Karich, A.; Ullrich, R.; Scheibner, K.; Hofrichter, M. Fungal Unspecific Peroxygenases Oxidize the Majority of Organic EPA Priority Pollutants. Front. Microbiol. 2017, 8, 1463. [Google Scholar] [CrossRef]
- Christian, V.; Shrivastava, R.; Shukla, D.; Modi, H.A.; Vyas, B.R.M. Degradation of xenobiotic compounds by lignin-degrading white-rot fungi: Enzymology and mechanisms involved. Indian J. Exp. Biol. 2005, 43, 301–312. [Google Scholar]
- Balcazar-Lopez, E.; Mendez-Lorenzo, L.H.; Batista-Garcia, R.A.; Esquivel-Naranjo, U.; Ayala, M.; Kumar, V.V.; Savary, O.; Cabana, H.; Herrera-Estrella, A.; Folch-Mallol, J.L. Xenobiotic Compounds Degradation by Heterologous Expression of a Trametes sanguineus Laccase in Trichoderma atroviride. PLoS ONE 2016, 5, e147997. [Google Scholar] [CrossRef]
- Schaffner, A.; Messner, B.; Langebartels, C.; Sandermann, H. Genes and Enzymes for In-Planta Phytoremediation of Air, Water and Soil. Acta Biotechnol. 2002, 22, 141–152. [Google Scholar] [CrossRef]
- Ogunyemi, A.K.; Buraimoh, O.M.; Ogunyemi, B.C.; Samuel, T.A.; Ilori, M.O.; Amund, O.O. Nitrilase gene detection and nitrile metabolism in two bacterial strains associated with waste streams in Lagos, Nigeria. Bull Natl. Res. Cent. 2022, 46, 151. [Google Scholar] [CrossRef]
- Kitts, C.L.; Green, C.E.; Otley, R.A.; Alvarez, M.A.; Unkefer, P.A. Type I nitroreductases in soil enterobacteria reduce TNT (2,4,6-trinitrotoluene) and RDX (hexahydro-1,3,5-trinitro-1,3,5-triazine). Can. J. Microbiol. 2000, 46, 278–282. [Google Scholar] [CrossRef] [PubMed]
- Chen, Q.; Wang, C.H.; Deng, S.K.; Wu, Y.D.; Li, Y.; Yao, L.; Jiang, J.D.; Yan, X.; He, J.; Li, S.P. Novel three-component Rieske non-heme iron oxygenase system catalyzing the N-dealkylation of chloroacetanilide herbicides in sphingomonads DC-6 and DC-2. Appl. Environ. Microbiol. 2014, 80, 5078–5085. [Google Scholar] [CrossRef] [PubMed]
- Karagianni, E.P.; Kontomina, E.; Davis, B.; Kotseli, B.; Tsirka, T.; Garefalaki, V.; Sim, E.; Glenn, A.E.; Boukouvala, S. Homologues of xenobiotic metabolizing N-acetyltransferases in plant-associated fungi: Novel functions for an old enzyme family. Sci. Rep. 2015, 5, 12900. [Google Scholar] [CrossRef]
- Kolhe, P.M.; Ingle, S.T.; Wagh, N.D. Degradation of Phenol Containing Wastewater by Advance Catalysis System—A Review. Annu. Res. Rev. Biol. 2015, 8, 1–15. [Google Scholar] [CrossRef]
- Ambreen, S.; Yasmin, A.; Aziz, S. Isolation and characterization of organophosphorus phosphatases from Bacillus thuringiensis MB497 capable of degrading Chlorpyrifos, Triazophos and Dimethoate. Heliyon 2020, 6, e04221. [Google Scholar] [CrossRef]
- Peco, J.D.; Higueras, P.; Campos, J.A.; Esbri, J.M.; Moreno, M.M.; Battaglia-Brunet, F.; Sandalio, L.M. Abandoned Mine Lands Reclamation by Plant Remediation Technologies. Sustainability 2021, 13, 6555. [Google Scholar] [CrossRef]
- Lorestani, B.; Yousefi, N.; Cheraghi, M.; Farmany, A. Phytoextraction and phytostabilization potential of plants grown in the vicinity of heavy metal-contaminated soils: A case study at an industrial town site. Environ. Monit. Assess. 2013, 185, 10217–10223. [Google Scholar] [CrossRef]
- Wu, B.; Peng, H.; Sheng, M.; Luo, H.; Wang, X.; Zhang, R.; Xu, F.; Xu, H. Evaluation of phytoremediation potential of native dominant plants and spatial distribution of heavy metals in abandoned mining area in Southwest China. Ecotoxicol. Environ. Saf. 2021, 220, 112368. [Google Scholar] [CrossRef] [PubMed]
- Zeng, P.; Guo, Z.; Cao, X.; Xiao, X.; Liu, Y.; Shi, L. Phytostabilization potential of ornamental plants grown in soil contaminated with cadmium. Int. J. Phytoremediat. 2018, 20, 311–320. [Google Scholar] [CrossRef]
- Siyar, R.; Doulati-Ardejani, F.; Norouzi, P.; Maghsoudy, S.; Yavarzadeh, M.; Taherdangkoo, R.; Butscher, C. Phytoremediation Potential of Native Hyperaccumulator Plants Growing on Heavy Metal-Contaminated Soil of Khatunabad Copper Smelter and Refinery, Iran. Water 2022, 14, 3597. [Google Scholar] [CrossRef]
- Greipsson, S. Phytoremediation. Nat. Educ. Knowl. 2011, 3, 7. [Google Scholar]
- Newman, L.A.; Reynolds, C.M. Phytodegradation of organic compounds. Curr. Opin. Biotechnol. 2004, 15, 225–230. [Google Scholar] [CrossRef] [PubMed]
- Guarino, F.; Miranda, A.; Castiglione, S.; Cicatelli, A. Arsenic phytovolatilization and epigenetic modifications in Arundo donax L. assisted by a PGPR consortium. Chemosphere 2020, 251, 126310. [Google Scholar] [CrossRef] [PubMed]
- Nedjimi, B. Phytoremediation: A sustainable environmental technology for heavy metals decontamination. SN Appl. Sci. 2021, 3, 286. [Google Scholar] [CrossRef]
- Narayanan, M.; Davis, L.C.; Erickson, L.E. Fate of volatile chlorinated organic compounds in a laboratory chamber with alfalfa plants. Environ. Sci. Technol. 1995, 29, 2437–2444. [Google Scholar] [CrossRef]
- Ashraf, M.; Ozturk, M.; Ahmad, M.S.A. (Eds.) Toxins and their phytoremediation. In Plant Adaptation and Phytoremediation; Springer: Berlin/Heidelberg, Germany, 2010; pp. 1–32. [Google Scholar]
- Woraharn, S.; Meeinkuirt, W.; Phusantisampan, T.; Avakul, P. Potential of ornamental monocot plants for rhizofiltration of cadmium and zinc in hydroponic systems. Environ. Sci. Pollut. Res. 2021, 28, 35157–35170. [Google Scholar] [CrossRef]
- Han, Y.; Lee, J.; Kim, C.; Park, J.; Lee, M.; Yang, M. Uranium Rhizofiltration by Lactuca sativa, Brassica campestris L., Raphanus sativus L., Oenanthe javanica under Different Hydroponic Conditions. Minerals 2021, 11, 41. [Google Scholar] [CrossRef]
- Kafle, A.; Timilsina, A.; Gautam, A.; Adhikari, K.; Bhattarai, A.; Aryal, N. Phytoremediation: Mechanisms, plant selection and enhancement by natural and synthetic agents. Environ. Adv. 2022, 8, 100203. [Google Scholar] [CrossRef]
- Jha, P.; Jobby, R.; Desai, N.S. Remediation of textile azo dye acid red 114 by hairy roots of Ipomoea carnea Jacq. and assessment of degraded dye toxicity with human keratinocyte cell line. J. Hazard. Mater. 2016, 311, 158–167. [Google Scholar] [CrossRef]
- Doty, S.L.; Shang, T.Q.; Wilson, A.M.; Moore, A.L.; Newman, L.A.; Strand, S.E.; Gordon, M.P. Metabolism of the soil and groundwater contaminants, ethylene dibromide and trichloroethylene, by the tropical leguminous tree, Leuceana leucocephala. Water Res. 2003, 37, 441–449. [Google Scholar] [CrossRef]
- Saddhe, A.A.; Manuka, R.; Nikalje, G.C.; Penna, S. Halophytes as a Potential Resource for Phytodesalination. In Handbook of Halophytes; Grigore, M.N., Ed.; Springer: Cham, Switzerland, 2020. [Google Scholar] [CrossRef]
- Rozema, E.R.; Gordon, R.J.; Zheng, Y. Plant species for the removal of Na+ and Cl from greenhouse nutrient solution. Hort. Sci. 2014, 49, 1071–1075. [Google Scholar] [CrossRef]
- Sarath, N.G.; Sruthi, P.; Shackira, A.M.; Puthur, J.T. Halophytes as effective tool for phytodesalination and land reclamation. In Frontiers in Plant-Soil Interaction; Academic Press: Cambridge, MA, USA, 2021; pp. 459–494. [Google Scholar] [CrossRef]
- Lastiri-Hernandez, M.A.; Alvarez-Bernal, D.; Bermudez-Torres, K.; Cardenas, G.C.; Ceja-Torres, L.F. Phytodesalination of a moderately saline soil combined with two inorganic amendments. Bragantia 2019, 78, 579–586. [Google Scholar] [CrossRef]
- Zorrig, W.; Rabhi, M.; Ferchichi, S.; Smaouti, A.; Abdelly, C. Phytodesalination: A Solution for Salt-affected Soils in Arid and Semi-arid Regions. J. Arid. Land Stud. 2012, 22, 299–302. [Google Scholar]
- Gatliff, E.G. Vegetative Remediation Process Offers Advantages over Traditional Pump and-Treat Technologies. Remediation 1994, 4, 343–352. [Google Scholar] [CrossRef]
- Ahmadpour, P.; Ahmadpour, F.; Mahmud, T.M.M.; Abdu, A.; Soleimani, M.; Tayefeh, F.H. Phytoremediation of heavy metals: A green technology. Afr. J. Biotechnol. 2012, 11, 14036–14043. [Google Scholar]
- Hasnaoui, S.E.; Fahr, M.; Keller, C.; Levard, C.; Angeletti, B.; Chaurand, P.; Triqui, Z.E.A.; Guedira, A.; Rhazi, L.; Colin, F.; et al. Screening of Native Plants Growing on a Pb/Zn Mining Area in Eastern Morocco: Perspectives for Phytoremediation. Plants 2020, 9, 1458. [Google Scholar] [CrossRef] [PubMed]
- Singh, S.N.; Tripathi, R.D. Environmental Bioremediation Technologies; Springer Science & Business Media: Berlin/Heidelberg, Germany, 2007; pp. 1–47. [Google Scholar]
- Henderson, K.L.; Belden, J.B.; Zhao, S.; Coats, J.R. Phytoremediation of pesticide wastes in soil. Z. Für Nat. 2006, 61, 213–221. [Google Scholar] [CrossRef]
- Zhao, S.; Arthur, E.L.; Coats, J.R. Influence of microbial inoculation (Pseudomonas sp. strain ADP), the enzyme atrazine chlorohydrolase, and vegetation on the degradation of atrazine and metolachlor in soil. J. Agric. Food Chem. 2003, 51, 3043–3048. [Google Scholar] [CrossRef]
- Prasad, M.N.V. Phytoremediation in India. In Phytoremediation: Methods and Review; Neil Willey Center for Research in Plant Science, University of the West of England: Bristol, UK, 2007; pp. 435–454. [Google Scholar]
- Tripathi, S.; Singh, V.K.; Srivastava, P.; Singh, R.; Devi, R.S.; Kumar, A.; Bhadouria, R. Phytoremediation of organic pollutants: Current status and future directions. In Abatement of Environmental Pollutants; Singh, P., Kumar, A., Borthakur, A., Eds.; Elsevier: Amsterdam, The Netherlands, 2020; pp. 81–105. [Google Scholar] [CrossRef]
- Raj, D.; Kumar, A.; Maiti, S.K. Mercury remediation potential of Brassica juncea (L.) Czern. for clean-up of flyash contaminated sites. Chemosphere 2020, 248, 125857. [Google Scholar] [CrossRef]
- Raklami, A.; Meddich, A.; Oufdou, K.; Baslam, M. Plants-Microorganisms-Based Bioremediation for Heavy Metal Cleanup: Recent Developments, Phytoremediation Techniques, Regulation Mechanisms, and Molecular Responses. Int. J. Mol. Sci. 2022, 23, 5031. [Google Scholar] [CrossRef]
- Song, H.W.; Wang, C.C.; Kumar, A.; Ding, Y.; Li, S.; Bai, X.; Liu, T.; Wang, J.L.; Zhang, Y.L. Removal of Pb2+ and Cd2+ from contaminated water using novel microbial material (Scoria@UF1). J. Environ. Chem. Eng. 2021, 9, 106495. [Google Scholar] [CrossRef]
- Kumar, A.; Song, H.W.; Mishra, S.; Zhang, W.; Zhang, Y.L.; Zhang, Q.R.; Yu, Z.G. Application of microbial-induced carbonate precipitation (MICP) techniques to remove heavy metal in the natural environment: A critical review. Chemosphere 2023, 318, 137894. [Google Scholar] [CrossRef] [PubMed]
- Song, H.; Kumar, A.; Zhang, Y. Microbial-induced carbonate precipitation prevents Cd2+ migration through the soil profile. Sci. Total Environ. 2022, 844, 157167. [Google Scholar] [CrossRef] [PubMed]


| Phytoremediation Type | Contaminant Nature | Medium | Mechanism | Scope of Application |
|---|---|---|---|---|
| Phytoextraction/phytoaccumulation | Inorganics | Soil, water | Hyperaccumulation | Moderately polluted sites |
| Rhizodegradation/phytostimulation | Organics | Soil | Breakdown inside the rhizosphere through microbial activity | Polycyclic aromatic hydrocarbon (PAH) contaminants |
| Phytostabilization | Inorganics | Soil | Immobilization | Mining contamination |
| Phytovolatilization/phytoevaporation | Organics/inorganics | Soil, water | Volatilization | Volatile contaminants |
| Rhizofiltration | Organics/inorganics | Water | Rhizosphere accumulation | Wastewater |
| Phytodegradation/phytotransformation | Complex organics | Water, soil | Breakdown inside the plant through metabolic processes | Soil and wastewater contamination |
| Phytodesalination | Organics/inorganics | Soil, water | Na hyperaccumulation | Sodic soil and water |
| Phytohydraulics | Organics/inorganics | Ground water | Uptake, sequestering and degradation of groundwater contaminants | Shallow contaminated sites |
| Name of the Metal | Source | Impacts | References | |||
|---|---|---|---|---|---|---|
| Terrestrial | Aquatic | |||||
| Humans | Plants | Animals | ||||
| Arsenic (As) | Fuel burning, pesticides, painting, wood treatment, geothermal processes, natural forces, thermal power plants, smelting | Cancer, skin lesions and increased deaths | Inhibits root extension and proliferation | Abdominal pain, vomiting and diarrhea; subsequently, rapid circulatory collapse | Bioaccumulation, physiological and biochemical disorders | [33,34,35,36] |
| Cadmium (Cd) | Fertilizers, fuel combustion, electroplating, smelting operations, batteries, dead batteries, paint sludge | Lung, prostate, nasopharynx, pancreas, and kidney cancers, as well as itai-itai | Reduces uptake and translocation of nutrients and water and disrupts metabolism | Kidney, lung, bone, liver, blood and nervous system are affected | Increase in mortality rates, deleterious effects on growth and reproduction systems | [37,38,39] |
| Chromium (Cr) | Mining operations, pesticide application, industrial coolant liquids, dyes, timber treatment, leather tanning, chromium salt manufacturing | Cancer, asthma, nose sores, skin infections, kidney and liver problems | Influences crop growth rate, productivity and quality of grains | Detrimental effects on wild birds and mammals | Cytotoxicity and detrimental impact | [40,41,42,43] |
| Copper (Cu) | Fertilizers, pigments, fungicides, mining, painting, electrical sources, lumber treatment, electroplating, smelting practices | Inflammation, cancer and anemia | Growth and development are blocked | Disheveled feathers, gizzard erosion, intestinal inflammation, hematochezia and damaged kidneys | Adverse effects on survival, growth and reproduction | [44,45] |
| Lead (Pb) | Metal products, paints, e-waste products, batteries, petrol additives, preservatives, ceramics, thermal power plants, bangle industry | Weakness, hypertension, brain and kidney damage, impotence and miscarriage | Poor germination, inhibits root growth and biomass production | Salivation, lack of vision, spastic twitching of eyelids, muscle tremors, jaw champing and convulsions | Oxidative stress, neurotoxin, bioaccumulation | [29,43,46] |
| Manganese (Mn) | Application of fertilizer | Deficits and neurodegenerative diseases, including a disorder called manganism | Triggers oxidative stress and disrupt photosynthesis | Reduced feed intake and growth rate and lethargy | Intestinal inflammatory damages, genotoxicity and oxidative stress | [47,48] |
| Mercury (Hg) | Fumigants, geothermal processes, fluorescent lights, chlor-alkali plants, hospital waste (broken thermometers, sphygmomanometers, barometers), thermal power plants | Loss of memory, kidney and nervous system problems and weakened hearing and vision ability | Growth retardation | Anorexia, stomatitis, vomiting, diarrhea, shock, pain and dehydration | Teratogenic, reproductive and neuro-toxicity | [43,46,49,50] |
| Molybdenum (Mo) | Fertilizer, spent catalysts | Pain in joints, gout-like signs and high blood levels of uric acid | Reduces seedling growth, yellowish leaves | Induces secondary copper deficiency in animals | Transformations in the forms of aquatic biota systems and instability in fundamental activities | [51,52] |
| Nickel (Ni) | Alloys, mine tailings, battery manufacturers, smelting processes, thermal power systems | Allergy, kidney disorders, cardiovascular fibrosis, lung and nasal cancer | Lower seed germination, growth, biomass and final yield | Lung disorders in rodents and affects liver, kidney, blood and reproduction processes in rats and mice | Inhibition of respiration, ionoregulatory destruction and enhanced oxidative stress | [53,54,55] |
| Zinc (Zn) | Dyes, paints, fertilizers, galvanization processes, lumber treatment, mining, electroplating, smelting practices | Nausea, back pain, vomiting, anemia and lethargy | Slows down photosynthetic and respiratory rates and leads to unbalanced mineral nutrition | Vomiting, diarrhea, depression, damage to red blood cells and lack of appetite | Kills fish by destroying gill tissues | [56,57] |
| Plant Species | Heavy Metal | Accumulation(A)/ Translocation (T) | Literature Cited |
|---|---|---|---|
| Althernanthera ficoides | As | A, T | [65] |
| Brassica juncea | Pb | A | [66] |
| Cleome rutidosperma DC | Cd and Pb | A | [67] |
| Helianthus annus | Cu | A | [66] |
| Hyptis suaveolens | Cr | A | [68] |
| Berkheya coddii | Ni | A | [69] |
| Phragmites australis | Ni, Mo, Se and Cu | A/T | [43] |
| Populus species | Cd | A | [70] |
| Ricinus communis | Ni | A | [70] |
| Salix species | Cd and Zn | A, T | [71] |
| Senna siamea | Pb | A | [70] |
| Enzyme | Target Pollutant | Biodegradation Pathway | References |
|---|---|---|---|
| Aromatic dehalogenase | Chlorinated aromatics (DDT, PCBs, etc.) | Hydrolytic dehalogenation | [74] |
| Carboxylesterases | Xenobiotics | Hydrolysis | [75] |
| Cytochrome P450 | Xenobiotics (PCBs) | Oxidation, reduction, hydrolysis and conjugation | [76] |
| Dehalogenase | Chlorinated solvents and ethylene | Dehalogenation | [77] |
| Glutathione s-transferase | Xenobiotics | Dehalogenation | [78] |
| Peroxygenases | Xenobiotics | Oxygenations and oxidations | [79] |
| Peroxidases | Xenobiotics | Oxidation and reduction | [80] |
| Laccase | Xenobiotics, degradation of explosives | Oxidation | [81] |
| N-glucosyl transferases | Various xenobiotics | Conjugation | [82] |
| Nitrilase | Herbicides | Degradation of nitrile | [83] |
| Nitroreductase | Explosives (RDX and TNT) | Reduction | [84] |
| N-malonyl transferases | Xenobiotics | Conjugation | [82] |
| O-demethylase | Alachlor, metalachor | N-dealkylation | [85] |
| O-glucosyl transferases | Xenobiotics | Conjugation | [82] |
| O-malonyl transferases | Xenobiotics | Hydrolysis | [86] |
| Peroxdase | Phenols | Elimination or reduction | [87] |
| Phosphatase | Organophosphates | Hydrolase | [88] |
| Plants | Pollutant | Mechanism | References |
|---|---|---|---|
| Arundo donax | Ni, Pb, Hg | Deposition | [89] |
| Atriplex portulacoides | Zn | Adsorption | [43] |
| Cirsium arvense | Pb, Mn, Zn | Absorption/adsorption | [90] |
| Conyza Canadensis | Cr, Ni, Cu, Pb, Cd | Accumulation | [91] |
| Euonymus japonicus | Cd | Deposition | [92] |
| Launaea acanthodes | Ni, Mo | Accumulation | [93] |
| Populus deltoids | As | Deposition | [89] |
| Ricinus communis | Cd, Cu, Mn, Zn | Absorption/adsorption | [43] |
| Salix purpurea | As | Deposition | [89] |
| Species | Pollutant | Biodegradation Pathway | References |
|---|---|---|---|
| Phytodegradation | |||
| Elodea Canadensis | DDT | Catalytic degradation | [102] |
| Ipomoea carnea | Textile azo dyes | Redox reaction | [103] |
| Populus spp. | Trichloroethylene (TCE) | Catalytic degradation | [94] |
| Leucaena leucocephala | Ethylene dibromide | Reduction | [104] |
| Pueraria thunbergiana | DDT | Dehalogenation | [102] |
| Phytodesalination | |||
| Andropogon gerardii | Na+ | EC reduction and salt accumulation | [105] |
| Atriplex prostrate | Na+, Cl− | Accumulate Na+ and Cl− | [106] |
| Phragmites australis | Na+ | Extraction | [105] |
| Typha latifolia | Na+, Cl− | Accumulate Na+ and Cl− | [106] |
| Name of the Plant | Pesticide |
|---|---|
| Populus spp. | Atrazine |
| Corbicula fluminea | Carbaryl, diazinon, carbofuran, glycophosphate, coumphos, parathion |
| Oryza sativa | Carbaryl, parathion, atrazine, carbofuran, diazinon, coumphos, glycophosphate |
| Bassia scoparia | Atrazine |
| Salix spp. | 2,4,5-T, 2,4-D, aldrin |
| Myriophyllum aquaticum | Organo-phosphate pesticides, halogenated pesticides |
| Elodea Canadensis | Organo-phosphate pesticides, halogenated pesticides |
| Spirodela oligorrhiza | Organo-phosphate pesticides |
| Category of Mine Spoils | Plants |
|---|---|
| Coal mine spoils | Eucalyptus hybrid, Pongamia pinnata, Acacia nilotica |
| Limestone mine spoils | Salix tetrasperma, Acacia catechu, Leucaena leucocephala |
| Copper, tungiston, mica, limestone and marble mine spoils | Prosopis juliflora, Acacia Senegal, Cynodon dactylon |
| Iron ore waste | Leucaena leucocephala |
| Manganese, haematite and magnetite spoil | Albizia lebeck |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Khan, S.; Masoodi, T.H.; Pala, N.A.; Murtaza, S.; Mugloo, J.A.; Sofi, P.A.; Zaman, M.U.; Kumar, R.; Kumar, A. Phytoremediation Prospects for Restoration of Contamination in the Natural Ecosystems. Water 2023, 15, 1498. https://doi.org/10.3390/w15081498
Khan S, Masoodi TH, Pala NA, Murtaza S, Mugloo JA, Sofi PA, Zaman MU, Kumar R, Kumar A. Phytoremediation Prospects for Restoration of Contamination in the Natural Ecosystems. Water. 2023; 15(8):1498. https://doi.org/10.3390/w15081498
Chicago/Turabian StyleKhan, Shaista, Tariq H. Masoodi, Nazir A. Pala, Shah Murtaza, Javeed A. Mugloo, Parvez A. Sofi, Musaib U. Zaman, Rupesh Kumar, and Amit Kumar. 2023. "Phytoremediation Prospects for Restoration of Contamination in the Natural Ecosystems" Water 15, no. 8: 1498. https://doi.org/10.3390/w15081498
APA StyleKhan, S., Masoodi, T. H., Pala, N. A., Murtaza, S., Mugloo, J. A., Sofi, P. A., Zaman, M. U., Kumar, R., & Kumar, A. (2023). Phytoremediation Prospects for Restoration of Contamination in the Natural Ecosystems. Water, 15(8), 1498. https://doi.org/10.3390/w15081498







