Degradation of Tetracycline Using a Magnetic Gadolinium-Decorated Nanoplatform: A Peroxidase Biomimetic System with Fenton-Like Catalysis
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Synthesis of Nanoparticles
2.3. DTPA-DA Preparation
2.4. Synthesis of PCD
2.5. Coating of Fe3O4
2.6. Loading of Gd3+ Ions to Fe3O4@PCD
2.7. Characterization
2.8. Degradation of TC Using the Magnetite/Gd3+/β-cyclodextrin Nanoplatform
3. Results
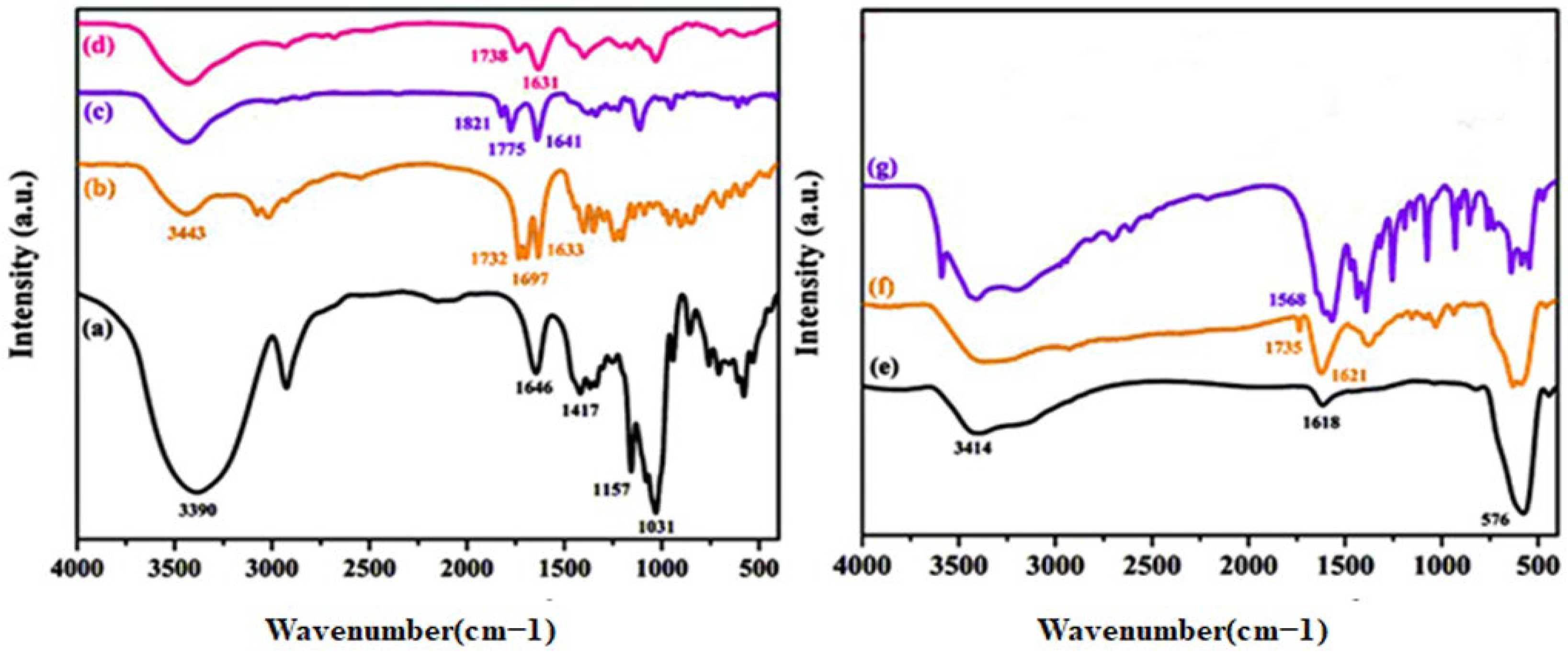
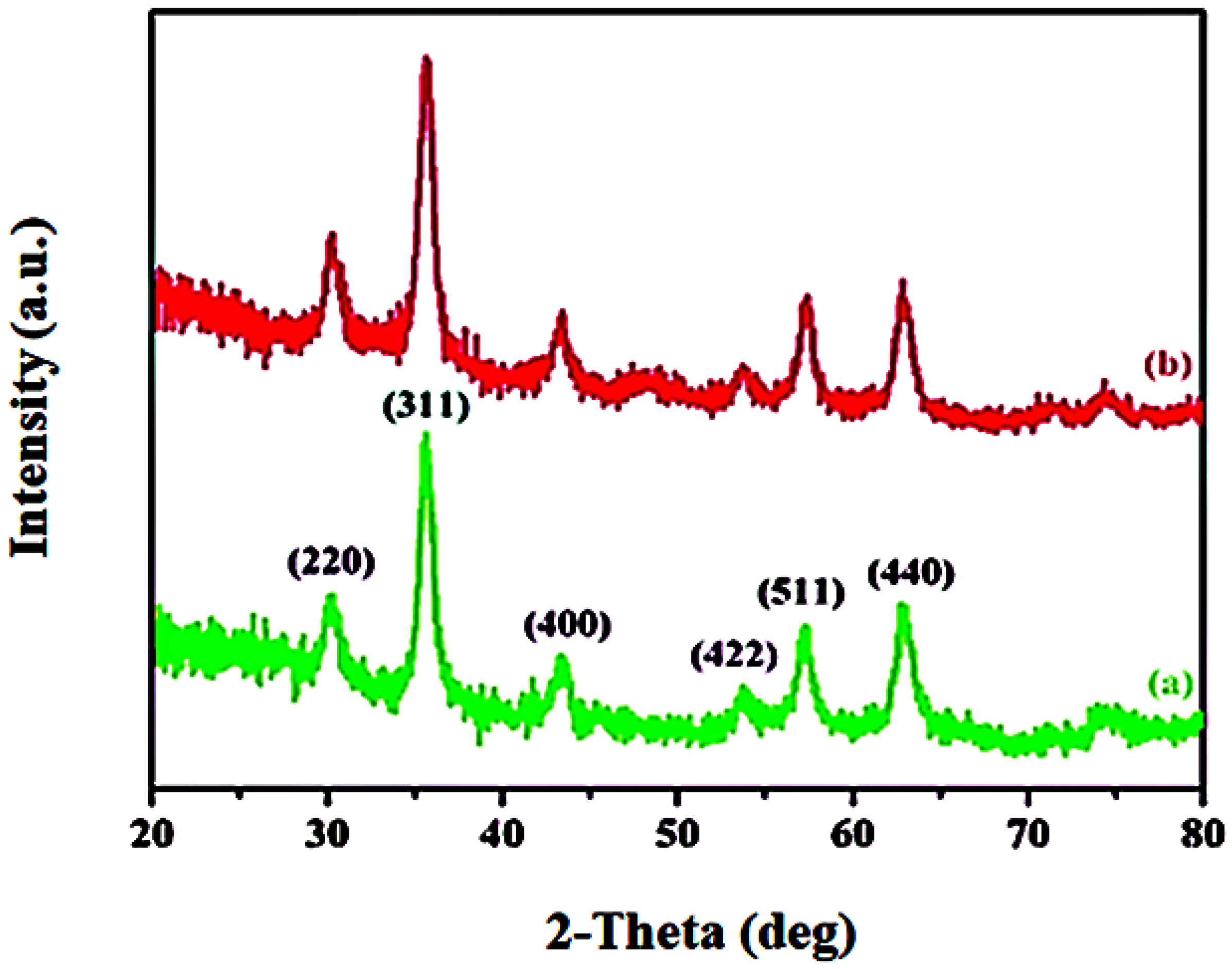
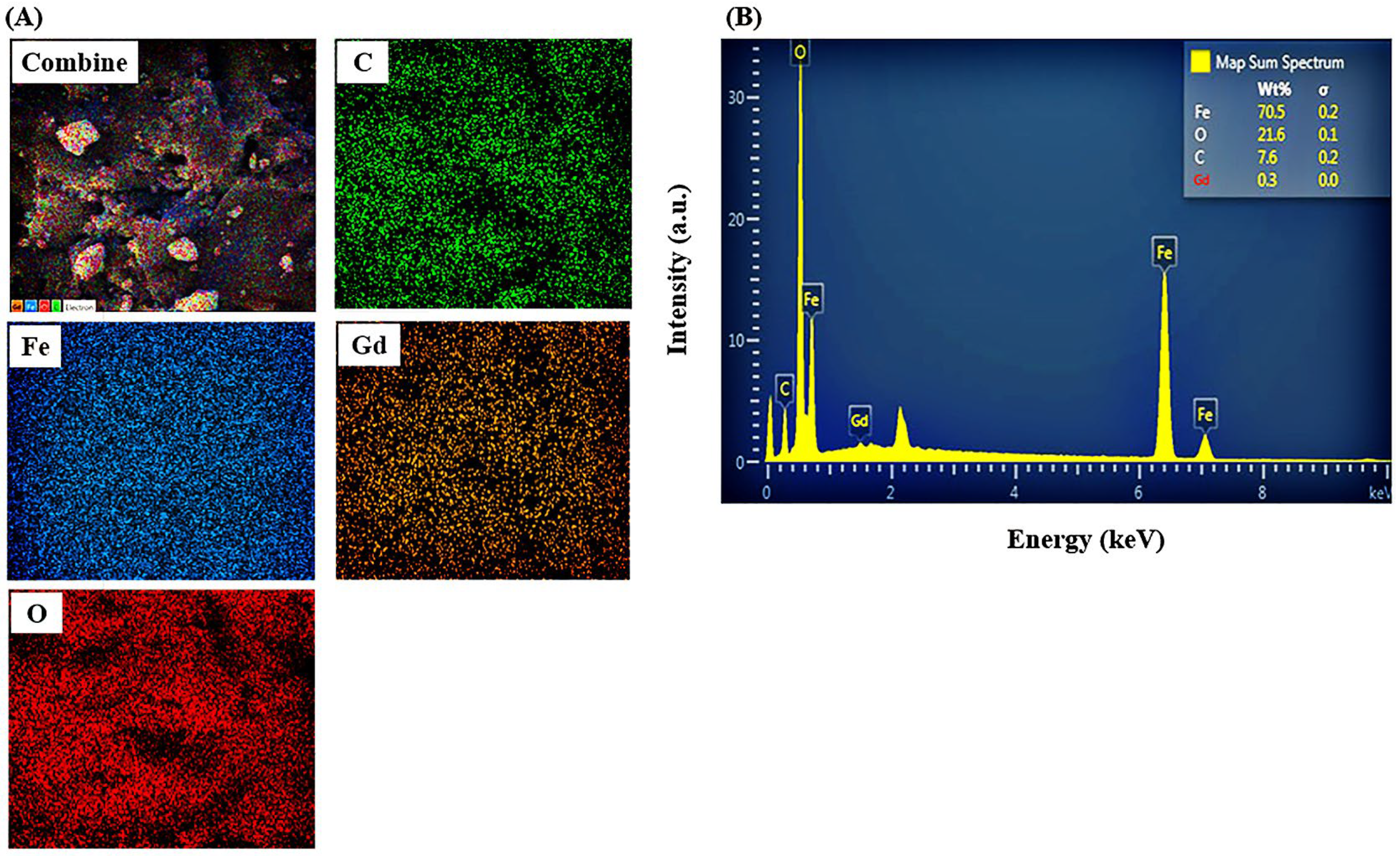
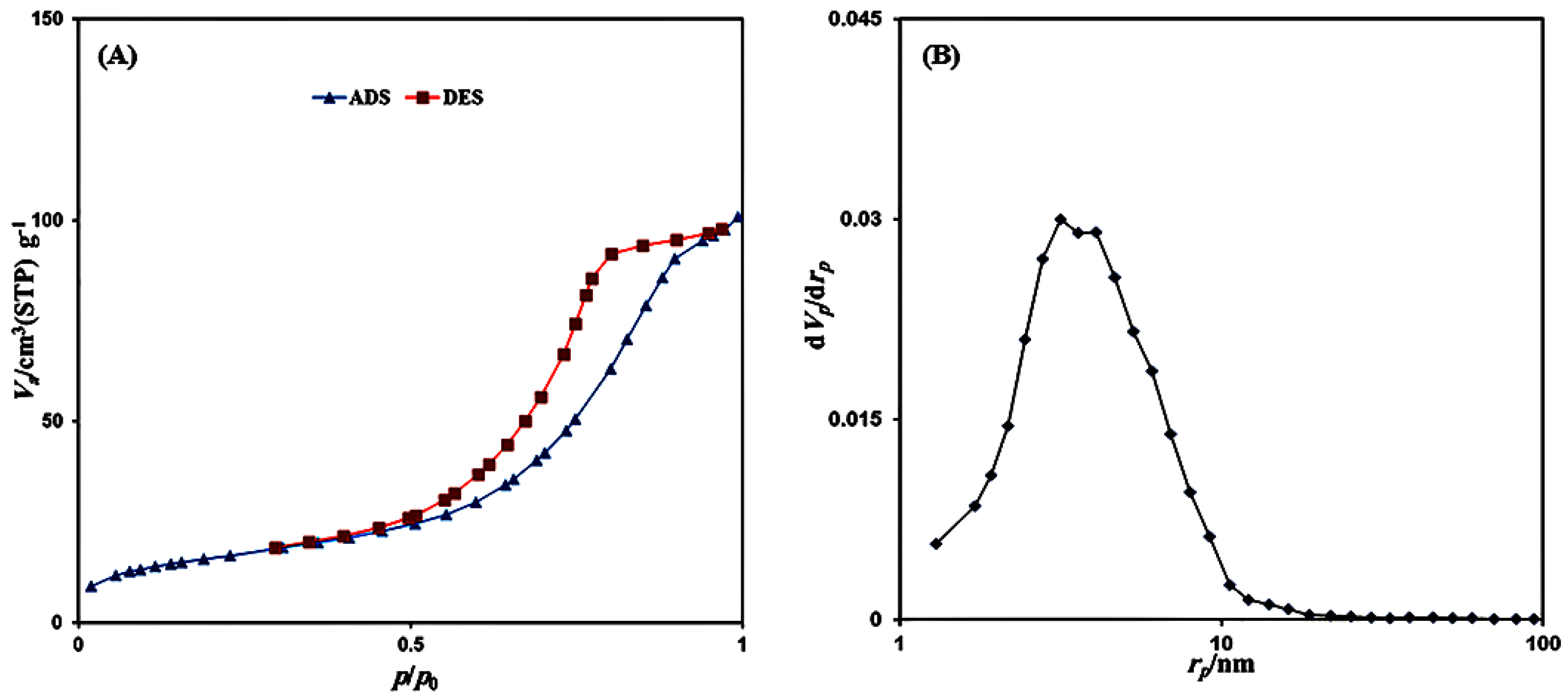
3.1. Characterization Studies
3.2. Enzyme-Like Activity of the Nanoplatform
3.3. Hydroxyl Radical Scavengers
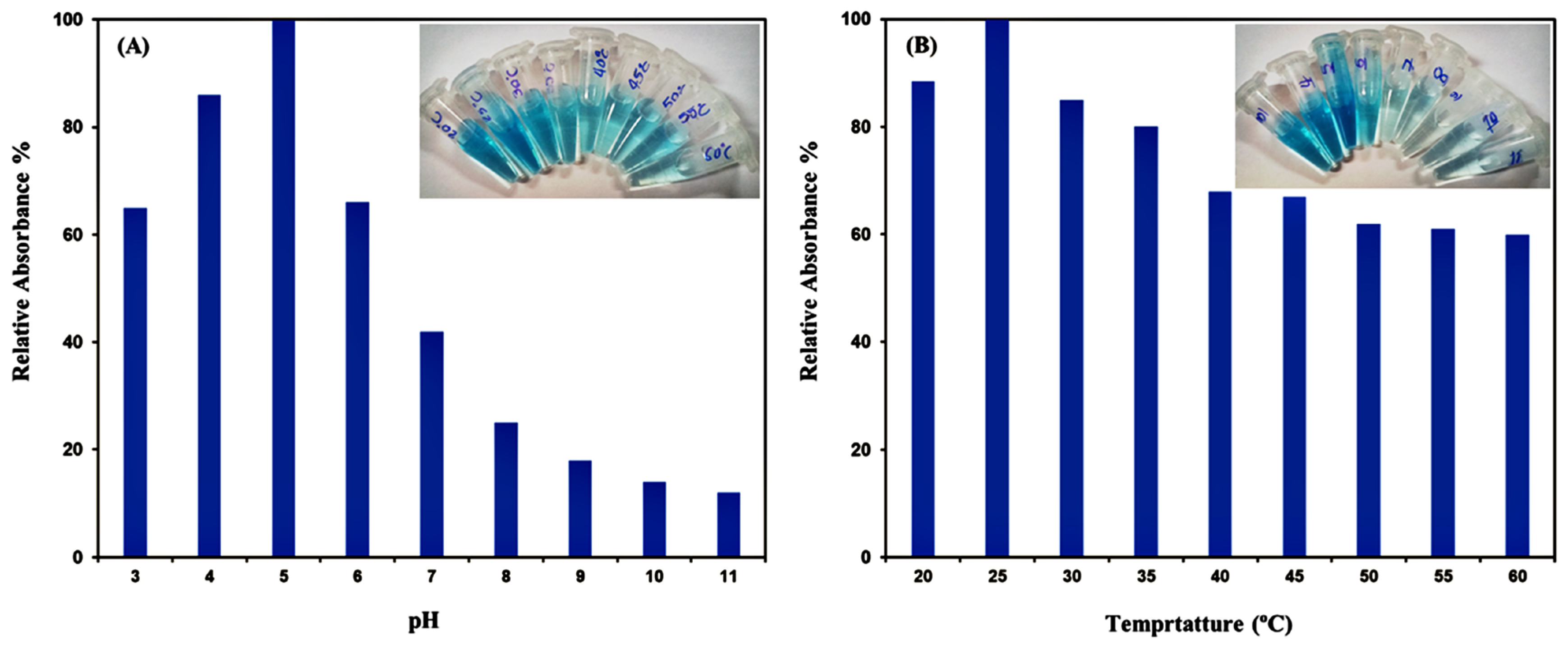
3.4. Optimization
3.5. Kinetic Studies
3.6. Effect of the Nanoplatform Concentration on TC Degradation
3.7. The Effect of H2O2 Concentration on TC Degradation
3.8. Recovery of the Nanoplatform
3.9. Performance of the Catalyst in the Presence of UV and Visible Light
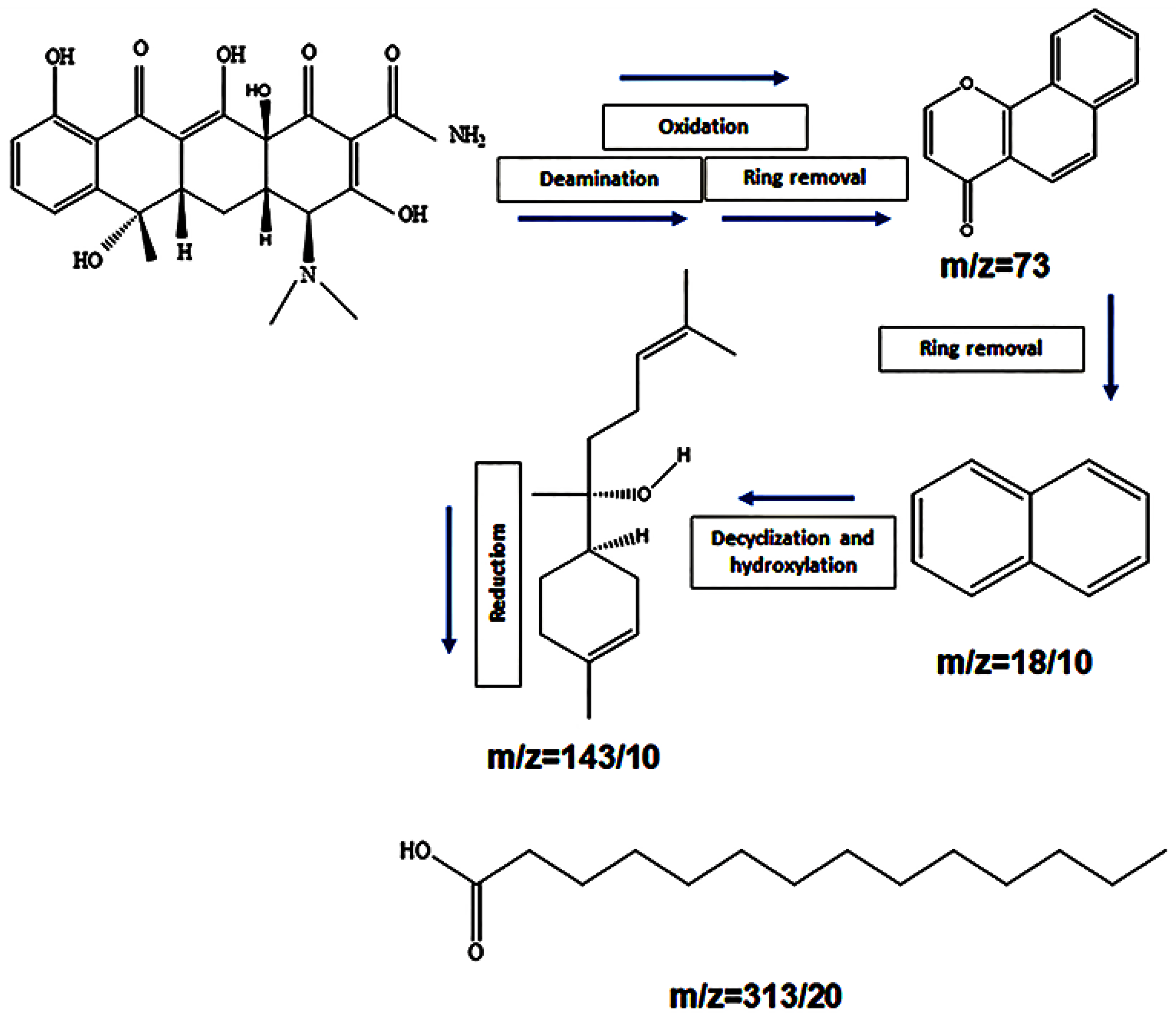
3.10. Proposed Pathway of TC Degradation
3.11. Degradation Kinetic Analysis
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Ahmad, F.; Zhu, D.; Sun, J. Environmental fate of tetracycline antibiotics: Degradation pathway mechanisms, challenges, and perspectives. Environ. Sci. Eur. 2021, 33, 64. [Google Scholar] [CrossRef]
- Eliopoulos, G.M.; Eliopoulos, G.M.; Roberts, M.C. Tetracycline therapy: Update. Clin. Infect. Dis. 2003, 36, 462–467. [Google Scholar]
- Bacanlı, M.; Başaran, N.J.F.; Toxicology, C. Importance of antibiotic residues in animal food. Food Chem. Toxicol. 2019, 125, 462–466. [Google Scholar] [CrossRef]
- Yanamoto, S.; Soutome, S.; Tsuda, S.; Morishita, K.; Hayashida, S.; Harata, S.; Murata, M.; Omori, K.; Rokutanda, S.; Umeda, M. Inhibitory effect of topical antibiotics/antiseptics administration on bacterial growth in the open wound of the jawbone surgery: Randomized controlled, preliminary study. J. Dent. Sci. 2021, 16, 154–159. [Google Scholar] [CrossRef] [PubMed]
- Prescott, J.F.; Baggot, J.D.; Walker, R.D. Antimicrobial Therapy in Veterinary Medicine; Iowa State University Press: Ames, IA, USA, 2000. [Google Scholar]
- Faria, L.V.; Lima, A.P.; Araújo, F.M.; Lisboa, T.P.; Matos, M.A.; Munoz, R.A.; Matos, R.C. High-throughput amperometric determination of tetracycline residues in milk and quality control of pharmaceutical formulations: Flow-injection versus batch-injection analysis. Anal. Methods 2019, 11, 5328–5336. [Google Scholar] [CrossRef]
- Al-Dhabi, N.A.; Esmail, G.A.; Arasu, M.V. Effective degradation of tetracycline by manganese peroxidase producing Bacillus velezensis strain Al-Dhabi 140 from Saudi Arabia using fibrous-bed reactor. Chemosphere 2021, 268, 128726. [Google Scholar] [CrossRef]
- Vagi, M.C.; Petsas, A.S. Recent advances on the removal of priority organochlorine and organophosphorus biorecalcitrant pesticides defined by Directive 2013/39/EU from environmental matrices by using advanced oxidation processes: An overview (2007–2018). J. Environ. Chem. Eng. 2020, 8, 102940. [Google Scholar] [CrossRef]
- Daghrir, R.; Drogui, P. Tetracycline antibiotics in the environment: A review. Environ. Chem. Lett. 2013, 11, 209–227. [Google Scholar] [CrossRef]
- Copeland, R.A. Enzymes: A Practical Introduction to Structure, Mechanism, and Data Analysis; John Wiley & Sons: Hoboken, NJ, USA, 2023. [Google Scholar]
- Wen, X.; Jia, Y.; Li, J. Enzymatic degradation of tetracycline and oxytetracycline by crude manganese peroxidase prepared from Phanerochaete chrysosporium. J. Hazard. Mater. 2010, 177, 924–928. [Google Scholar] [CrossRef]
- Garg, P.; Ghatmale, P.; Tarwadi, K.; Chavan, S. Influence of nanotechnology and the role of nanostructures in biomimetic studies and their potential applications. Biomimetics 2017, 2, 7. [Google Scholar] [CrossRef] [Green Version]
- Dehghan, G.; Rashtbari, S.; Yekta, R.; Sheibani, N. Synergistic inhibition of catalase activity by food colorants sunset yellow and curcumin: An experimental and MLSD simulation approach. Chem. Biol. Interact. 2019, 311, 108746. [Google Scholar]
- Rashtbari, S.; Dehghan, G.; Amini, M. An ultrasensitive label-free colorimetric biosensor for the detection of glucose based on glucose oxidase-like activity of nanolayered manganese-calcium oxide. Anal. Chim. Acta 2020, 1110, 98–108. [Google Scholar] [CrossRef] [PubMed]
- Rashtbari, S.; Dehghan, G.; Khataee, S.; Amini, M.; Khataee, A. Dual enzymes-mimic activity of nanolayered manganese-calcium oxide for fluorometric determination of metformin. Chemosphere 2022, 291, 133063. [Google Scholar] [CrossRef] [PubMed]
- Suliman, M.H.; Yamani, Z.H.; Usman, M. Electrochemical Reduction of CO2 to C1 and C2 Liquid Products on Copper-Decorated Nitrogen-Doped Carbon Nanosheets. Nanomaterials 2023, 13, 47. [Google Scholar] [CrossRef]
- Gkantzou, E.; Chatzikonstantinou, A.V.; Fotiadou, R.; Giannakopoulou, A.; Patila, M.; Stamatis, H. Trends in the development of innovative nanobiocatalysts and their application in biocatalytic transformations. Biotechnol. Adv. 2021, 51, 107738. [Google Scholar] [CrossRef]
- Tang, S.; Zhao, M.; Yuan, D.; Li, X.; Wang, Z.; Zhang, X.; Jiao, T.; Ke, J. Fe3O4 nanoparticles three-dimensional electro-peroxydisulfate for improving tetracycline degradation. Chemosphere 2021, 268, 129315. [Google Scholar] [CrossRef]
- Rashtbari, S.; Dehghan, G.; Khorram, S.; Amini, M.; Khataee, A.; Yoon, Y. Plasma modified Co3O4 nanoparticles for catalytic degradation process through enhanced peroxidase-like activity. J. Ind. Eng. Chem. 2023, 121, 114–123. [Google Scholar] [CrossRef]
- Rashtbari, S.; Dehghan, G. Biodegradation of malachite green by a novel laccase-mimicking multicopper BSA-Cu complex: Performance optimization, intermediates identification and artificial neural network modeling. J. Hazard. Mater. 2021, 406, 124340. [Google Scholar] [CrossRef]
- Jiang, D.; Ni, D.; Rosenkrans, Z.T.; Huang, P.; Yan, X.; Cai, W. Nanozyme: New horizons for responsive biomedical applications. Chem. Soc. Rev. 2019, 48, 3683–3704. [Google Scholar] [CrossRef]
- Thangudu, S.; Su, C.-H. Peroxidase mimetic nanozymes in cancer phototherapy: Progress and perspectives. Biomolecules 2021, 11, 1015. [Google Scholar] [CrossRef]
- Thangudu, S.; Lee, M.T.; Rtimi, S. Tandem synthesis of high yield MoS2 nanosheets and enzyme peroxidase mimicking properties. Catalysts 2020, 10, 1009. [Google Scholar] [CrossRef]
- Thangudu, S.; Chiang, C.-S.; Hwang, K.C. 1550 nm light activatable photothermal therapy on multifunctional CuBi2O4 bimetallic particles for treating drug resistance bacteria-infected skin in the NIR-III biological window. J. Colloid Interface Sci. 2023, 631, 1–16. [Google Scholar] [CrossRef]
- Bao, S.; Wang, Y.; Wei, Z.; Yang, W.; Yu, Y. Highly efficient recovery of heavy rare earth elements by using an amino-functionalized magnetic graphene oxide with acid and base resistance. J. Hazard. Mater. 2022, 424, 127370. [Google Scholar] [CrossRef] [PubMed]
- Vieru, V.; Iwahara, N.; Ungur, L.; Chibotaru, L.F. Giant exchange interaction in mixed lanthanides. Sci. Rep. 2016, 6, 24046. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yu, Y.; Delbianco, M. Synthetic polysaccharides. In Recent Trends in Carbohydrate Chemistry; Elsevier: Amsterdam, The Netherlands, 2020; pp. 333–371. [Google Scholar]
- Petitjean, M.; García-Zubiri, I.X.; Isasi, J.R. History of cyclodextrin-based polymers in food and pharmacy: A review. Environ. Chem. Lett. 2021, 19, 3465–3476. [Google Scholar] [CrossRef]
- Kondiah, P.; Choonara, Y.E.; Hayiyana, Z.; Kondiah, P.J.; Marimuthu, T.; du Toit, L.C.; Kumar, P.; Pillay, V. Multi-Cyclodextrin Supramolecular Encapsulation Entities for Multifaceted Topical Drug Delivery Applications. In Applications of Encapsulation and Controlled Release; CRC Press: Boca Raton, FL, USA, 2019; pp. 1–17. [Google Scholar]
- Khalili, L.; Dehghan, G.; Sheibani, N.; Khataee, A. Smart active-targeting of lipid-polymer hybrid nanoparticles for therapeutic applications: Recent advances and challenges. Int. J. Biol. Macromol. 2022, 213, 166–194. [Google Scholar] [CrossRef]
- Muthukrishnan, L. Disruptive Nanozyme Technology for Futuristic Bio-Medical and Bio-imaging Applications. Curr. Nanosci. 2021, 17, 853–870. [Google Scholar] [CrossRef]
- Dalal, D.S.; Patil, D.R.; Tayade, Y.A. β-Cyclodextrin: A Green and Efficient Supramolecular Catalyst for Organic Transformations. Chem. Rec. 2018, 18, 1560–1582. [Google Scholar] [CrossRef]
- Fiorentin, T.R.; Logan, B.K.; Martin, D.M.; Browne, T.; Rieders, E.F. Assessment of a portable quadrupole-based gas chromatography mass spectrometry for seized drug analysis. Forensic Sci. Int. 2020, 313, 110342. [Google Scholar] [CrossRef]
- Goh, P.S.; Samavati, Z.; Ismail, A.F.; Ng, B.C.; Abdullah, M.S.; Hilal, N. Modification of Liquid Separation Membranes Using Multidimensional Nanomaterials: Revealing the Roles of Dimension Based on Classical Titanium Dioxide. Nanomaterials 2023, 13, 448. [Google Scholar] [CrossRef]
- Norabadi, E.; Jahantiq, A.; Kamani, H. Synthesis of Fe-TiO2@Fe3O4 magnetic nanoparticles as a recyclable sonocatalyst for the degradation of 2, 4-dichlorophenol. Environ. Sci. Pollut. Res. 2022, 30, 31446–31460. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.; Xu, X.; Feng, L.; He, A.; Liu, L.; Li, X.; Khan, S.; Chen, Y. One-step MOF assisted synthesis of SmVO4 nanorods for photocatalytic degradation of tetracycline under visible light. Mater. Lett. 2020, 276, 128213. [Google Scholar] [CrossRef]
- Zhu, G.; Yu, X.; Xie, F.; Feng, W.J.A. Ultraviolet light assisted heterogeneous Fenton degradation of tetracycline based on polyhedral Fe3O4 nanoparticles with exposed high-energy {110} facets. Appl. Surf. Sci. 2019, 485, 496–505. [Google Scholar] [CrossRef]
- Li, X.; Zeng, Z.; Zeng, G.; Wang, D.; Xiao, R.; Wang, Y.; Zhou, C.; Yi, H.; Ye, S.; Yang, Y.; et al. A “bottle-around-ship” like method synthesized yolk-shell Ag3PO4@ MIL-53 (Fe) Z-scheme photocatalysts for enhanced tetracycline removal. J. Colloid Interface Sci. 2020, 561, 501–511. [Google Scholar] [CrossRef]
- Kakavandi, B.; Takdastan, A.; Jaafarzadeh, N.; Azizi, M.; Mirzaei, A.; Azari, A.J.J.; Chemistry, P.A. Application of Fe3O4@ C catalyzing heterogeneous UV-Fenton system for tetracycline removal with a focus on optimization by a response surface method. J. Photochem. Photobiol. A Chem. 2016, 314, 178–188. [Google Scholar] [CrossRef]
- Hassandoost, R.; Kotb, A.; Movafagh, Z.; Esmat, M.; Guegan, R.; Endo, S.; Jevasuwan, W.; Fukata, N.; Sugahara, Y.; Khataee, A. Nanoarchitecturing bimetallic manganese cobaltite spinels for sonocatalytic degradation of oxytetracycline. Chem. Eng. J. 2022, 431, 133851. [Google Scholar] [CrossRef]
- Loeb, S.K.; Alvarez, P.J.; Brame, J.A.; Cates, E.L.; Choi, W.; Crittenden, J.; Dionysiou, D.D.; Li, Q.; Li-Puma, G.; Quan, X. The technology horizon for photocatalytic water treatment: Sunrise or sunset? Environ. Sci. Technol. 2018, 53, 2937–2947. [Google Scholar] [CrossRef] [Green Version]
- Wang, J.; Tang, J. Fe-based Fenton-like catalysts for water treatment: Catalytic mechanisms and applications. J. Mol. Liq. 2021, 332, 115755. [Google Scholar] [CrossRef]
- Cheng, S.; Pan, X.; Zhang, C.; Lin, X.; Zhuang, Q.; Jiao, Y.; Dong, W.; Qi, X. UV-assisted ultrafast construction of robust Fe3O4/polydopamine/Ag Fenton-like catalysts for highly efficient micropollutant decomposition. Sci. Total Environ. 2022, 810, 151182. [Google Scholar] [CrossRef]
- Liu, Y.; Zhao, Y.; Wang, J. Fenton/Fenton-like processes with in-situ production of hydrogen peroxide/hydroxyl radical for degradation of emerging contaminants: Advances and prospects. J. Hazard. Mater. 2021, 404, 124191. [Google Scholar] [CrossRef]
- Zhang, D.; Yin, K.; Tang, Y.; Wei, Y.; Tang, H.; Du, Y.; Liu, H.; Chen, Y.; Liu, C. Hollow sea-urchin-shaped carbon-anchored single-atom iron as dual-functional electro-Fenton catalysts for degrading refractory thiamphenicol with fast reaction kinetics in a wide pH range. Chem. Eng. J. 2022, 427, 130996. [Google Scholar] [CrossRef]
- Sun, Z.; Wang, S.; Chen, W. Metal single-atom catalysts for selective hydrogenation of unsaturated bonds. J. Mater. Chem. A 2021, 9, 5296–5319. [Google Scholar] [CrossRef]
- Du, C.; Zhang, Y.; Zhang, Z.; Zhou, L.; Yu, G.; Wen, X.; Chi, T.; Wang, G.; Su, Y.; Deng, F. Fe-based metal organic frameworks (Fe-MOFs) for organic pollutants removal via photo-Fenton: A review. Chem. Eng. J. 2022, 431, 133932. [Google Scholar] [CrossRef]
- Zhang, B.; Li, X.; Akiyama, K.; Bingham, P.A.; Kubuki, S. Elucidating the Mechanistic Origin of a Spin State-Dependent FeN x–C Catalyst toward Organic Contaminant Oxidation via Peroxymonosulfate Activation. Environ. Sci. Technol. 2021, 56, 1321–1330. [Google Scholar] [CrossRef] [PubMed]
- Yang, T.; Yu, D.; Wang, D.; Yang, T.; Li, Z.; Wu, M.; Petru, M.; Crittenden, J. Accelerating Fe (III)/Fe (II) cycle via Fe (II) substitution for enhancing Fenton-like performance of Fe-MOFs. Appl. Catal. B Environ. 2021, 286, 119859. [Google Scholar] [CrossRef]
- Guo, D.; Liu, Y.; Ji, H.; Wang, C.-C.; Chen, B.; Shen, C.; Li, F.; Wang, Y.; Lu, P.; Liu, W. Silicate-enhanced heterogeneous flow-through electro-Fenton system using iron oxides under nanoconfinement. Environ. Sci. Technol. 2021, 55, 4045–4053. [Google Scholar] [CrossRef]
- Hu, Q.; Zhang, M.; Xu, L.; Wang, S.; Yang, T.; Wu, M.; Lu, W.; Li, Y.; Yu, D. Unraveling timescale-dependent Fe-MOFs crystal evolution for catalytic ozonation reactivity modulation. J. Hazard. Mater. 2022, 431, 128575. [Google Scholar] [CrossRef] [PubMed]
- Mansouri, H.; Gholibegloo, E.; Mortezazadeh, T.; Yazdi, M.H.; Ashouri, F.; Malekzadeh, R.; Najafi, A.; Foroumadi, A.; Khoobi, M. A biocompatible theranostic nanoplatform based on magnetic gadolinium-chelated polycyclodextrin: In vitro and in vivo studies. Carbohydr. Polym. 2021, 254, 117262. [Google Scholar] [CrossRef]
- Gholibegloo, E.; Mortezazadeh, T.; Salehian, F.; Ramazani, A.; Amanlou, M.; Khoobi, M. Improved curcumin loading, release, solubility and toxicity by tuning the molar ratio of cross-linker to β-cyclodextrin. Carbohydr. Polym. 2019, 213, 70–78. [Google Scholar] [CrossRef]
- Gholibegloo, E.; Mortezazadeh, T.; Salehian, F.; Forootanfar, H.; Firoozpour, L.; Foroumadi, A.; Ramazani, A.; Khoobi, M. Folic acid decorated magnetic nanosponge: An efficient nanosystem for targeted curcumin delivery and magnetic resonance imaging. J. Colloid Interface Sci. 2019, 556, 128–139. [Google Scholar] [CrossRef]
- Wu, B.; Yang, T.; Zou, D.; Jin, L.; Liang, X.; Li, T.; Huang, G.; Zhang, J. Nuclear magnetic resonance biosensor based on streptavidin–biotin system and poly-l-lysine macromolecular targeted gadolinium probe for rapid detection of Salmonella in milk. Int. Dairy J. 2020, 102, 104594. [Google Scholar] [CrossRef]
- Jaffray, D.A. World Congress on Medical Physics and Biomedical Engineering, 7–12 June 2015, Toronto, Canada; Springer: Berlin/Heidelberg, Germany, 2015; Volume 51. [Google Scholar]
- De Silva, L.; Fu, J.-Y.; Htar, T.T.; Muniyandy, S.; Kasbollah, A.; Kamal, W.H.B.W.; Chuah, L.-H. Characterization, optimization, and in vitro evaluation of Technetium-99m-labeled niosomes. Int. J. Nanomed. 2019, 14, 1101. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mortezazadeh, T.; Gholibegloo, E.; Alam, N.R.; Dehghani, S.; Haghgoo, S.; Ghanaati, H.; Khoobi, M. Gadolinium (III) oxide nanoparticles coated with folic acid-functionalized poly (β-cyclodextrin-co-pentetic acid) as a biocompatible targeted nano-contrast agent for cancer diagnostic: In vitro and in vivo studies. Magn. Reson. Mater. Phys. Biol. Med. 2019, 32, 487–500. [Google Scholar] [CrossRef] [PubMed]
- Jenjob, R.; Kun, N.; Ghee, J.Y.; Shen, Z.; Wu, X.; Cho, S.K.; Lee, D.H.; Yang, S.-G. Enhanced conjugation stability and blood circulation time of macromolecular gadolinium-DTPA contrast agent. Mater. Sci. Eng. C 2016, 61, 659–664. [Google Scholar] [CrossRef] [PubMed]
- Gholibegloo, E.; Ebrahimpour, A.; Mortezazadeh, T.; Sorouri, F.; Foroumadi, A.; Firoozpour, L.; Ardestani, M.S.; Khoobi, M. pH-Responsive chitosan-modified gadolinium oxide nanoparticles delivering 5-aminolevulinic acid: A dual cellular and metabolic T1-T2* contrast agent for glioblastoma brain tumors detection. J. Mol. Liq. 2022, 368, 120628. [Google Scholar] [CrossRef]
- Thorat, N.D.; Bohara, R.A.; Yadav, H.M.; Tofail, S.A. Multi-modal MR imaging and magnetic hyperthermia study of Gd doped Fe3O4 nanoparticles for integrative cancer therapy. RSC Adv. 2016, 6, 94967–94975. [Google Scholar] [CrossRef]
- Behnajady, M.; Eskandarloo, H.; Modirshahla, N.; Shokri, M. Investigation of the effect of sol–gel synthesis variables on structural and photocatalytic properties of TiO2 nanoparticles. Desalination 2011, 278, 10–17. [Google Scholar] [CrossRef]
- Cao, G.-J.; Jiang, X.; Zhang, H.; Croley, T.R.; Yin, J.-J. Mimicking horseradish peroxidase and oxidase using ruthenium nanomaterials. RSC Adv. 2017, 7, 52210–52217. [Google Scholar] [CrossRef] [Green Version]
- Wang, L.; Liu, Y.; Lu, C.; Yang, Z.; Liu, Y.; Wang, Y.; Rao, H.; Zhang, W.; Wang, X. Ultrasonic synthesis of nano-PrO1. 8 as nanozyme for colorimetric determination of trans-resveratrol. Sci. Rep. 2020, 10, 4432. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hu, J.; Zhao, G.; Long, X.; Wang, Y.; Jiao, F. In situ topotactic fabrication of ZnS nanosheet by using ZnAl-layered double hydroxide template for enhanced tetracycline pollutant degradation activity. Mater. Sci. Semicond. Process. 2021, 134, 106007. [Google Scholar] [CrossRef]
- Chatgilialoglu, C.; Krokidis, M.G.; Masi, A.; Barata-Vallejo, S.; Ferreri, C.; Terzidis, M.A.; Szreder, T.; Bobrowski, K. New insights into the reaction paths of hydroxyl radicals with purine moieties in DNA and double-stranded oligodeoxynucleotides. Molecules 2019, 24, 3860. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Brown, P.; Gill, S.; Allen, S. Metal removal from wastewater using peat. Water Res. 2000, 34, 3907–3916. [Google Scholar] [CrossRef]
- Vakh, C.; Malkova, K.; Syukkalova, E.; Bobrysheva, N.; Voznesenskiy, M.; Bulatov, A.; Osmolovskaya, O. Chemical and computational strategy for design of “switchable” sorbent based on hydroxyapatite nanoparticles for dispersive micro-solid phase extraction of tetracyclines. J. Hazard. Mater. 2021, 419, 126504. [Google Scholar] [CrossRef] [PubMed]
- Ou, J.; Mei, M.; Xu, X. Magnetic adsorbent constructed from the loading of amino functionalized Fe3O4 on coordination complex modified polyoxometalates nanoparticle and its tetracycline adsorption removal property study. J. Solid State Chem. 2016, 238, 182–188. [Google Scholar] [CrossRef]
- Zhu, X.; Qian, F.; Liu, Y.; Matera, D.; Wu, G.; Zhang, S.; Chen, J. Controllable synthesis of magnetic carbon composites with high porosity and strong acid resistance from hydrochar for efficient removal of organic pollutants: An overlooked influence. Carbon 2016, 99, 338–347. [Google Scholar] [CrossRef]


| Sample | Substrate | Km (mM) | Vmax (M s−1) | Ref. |
|---|---|---|---|---|
| HRP | TMB | 0.434 | 2.01 × 10−8 | [66] |
| H2O2 | 3.7 | 3.34 × 10−8 | ||
| Nanoplatform | TMB | 0.5 × 10−4 | 13.6 × 10−7 | This work |
| H2O2 | 2.4 × 10−4 | 17.8 × 10−8 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hamidian, N.; Dehghan, G.; Rashtbari, S.; Khataee, A.; Khoobi, M.; Sheibani, N. Degradation of Tetracycline Using a Magnetic Gadolinium-Decorated Nanoplatform: A Peroxidase Biomimetic System with Fenton-Like Catalysis. Water 2023, 15, 1419. https://doi.org/10.3390/w15071419
Hamidian N, Dehghan G, Rashtbari S, Khataee A, Khoobi M, Sheibani N. Degradation of Tetracycline Using a Magnetic Gadolinium-Decorated Nanoplatform: A Peroxidase Biomimetic System with Fenton-Like Catalysis. Water. 2023; 15(7):1419. https://doi.org/10.3390/w15071419
Chicago/Turabian StyleHamidian, Nesa, Gholamreza Dehghan, Samaneh Rashtbari, Alireza Khataee, Mehdi Khoobi, and Nader Sheibani. 2023. "Degradation of Tetracycline Using a Magnetic Gadolinium-Decorated Nanoplatform: A Peroxidase Biomimetic System with Fenton-Like Catalysis" Water 15, no. 7: 1419. https://doi.org/10.3390/w15071419





