Recent Advances in MXene-Based Nanocomposites for Wastewater Purification and Water Treatment: A Review
Abstract
:1. Introduction
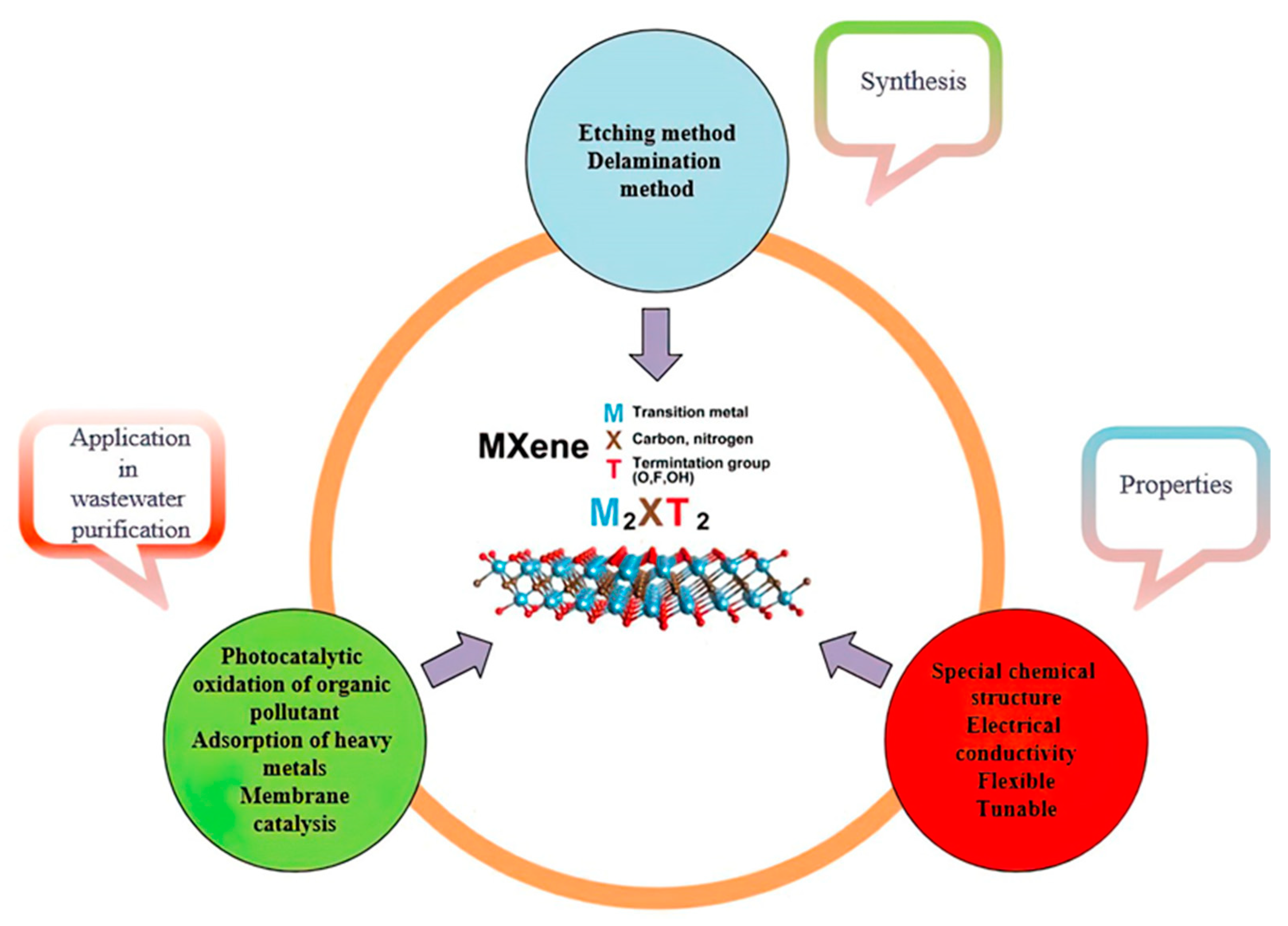
2. MXene and Nanocomposite-Based MXene Synthesis
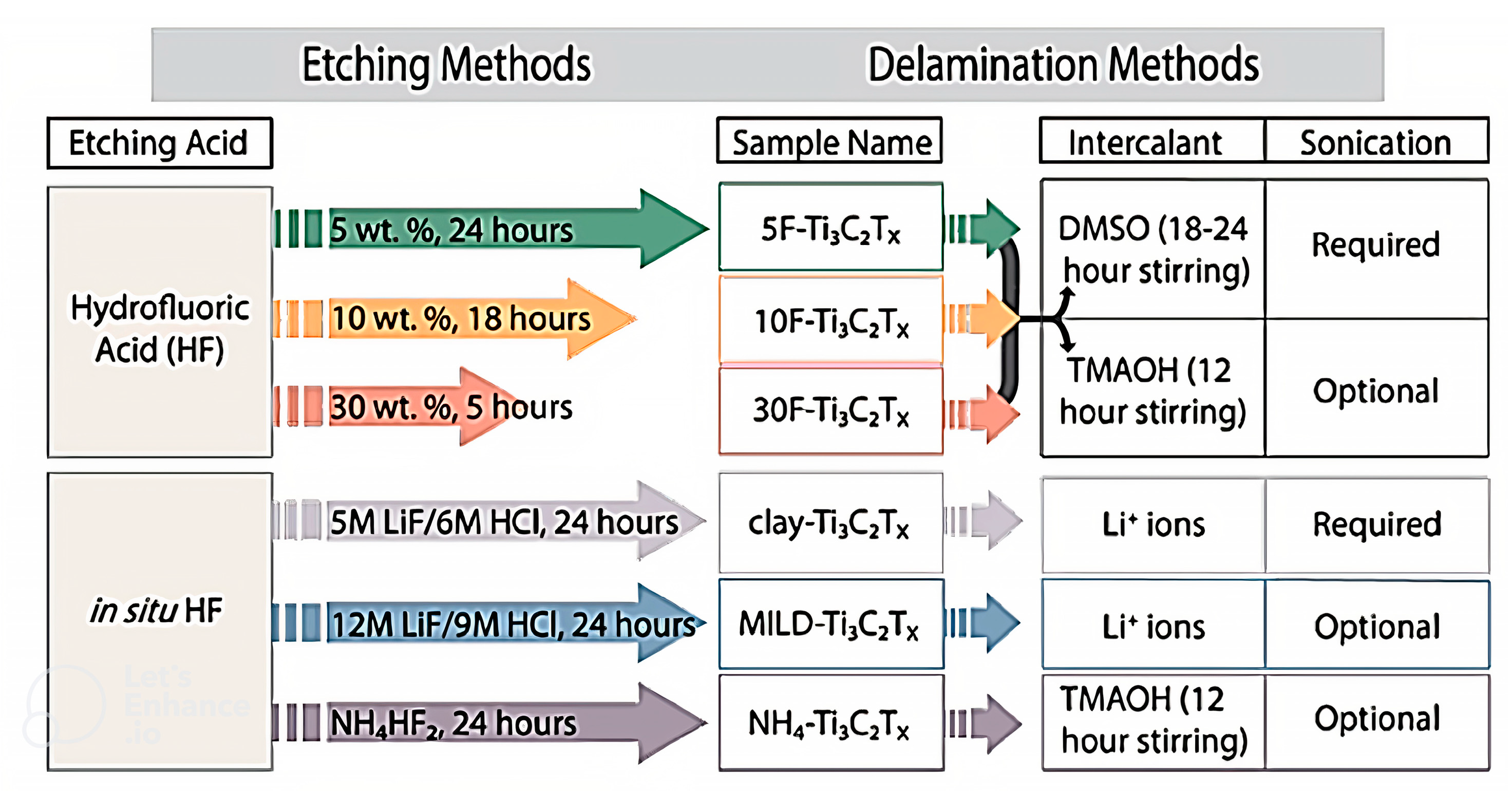
2.1. Etching Approach
2.1.1. HF Approach
2.1.2. HF Solution
2.2. Delamination Method
2.2.1. Intercalation Method
2.2.2. Sonication Method
3. Features of MXenes
4. Removing Pollutants from Wastewater: Challenges and Opportunities
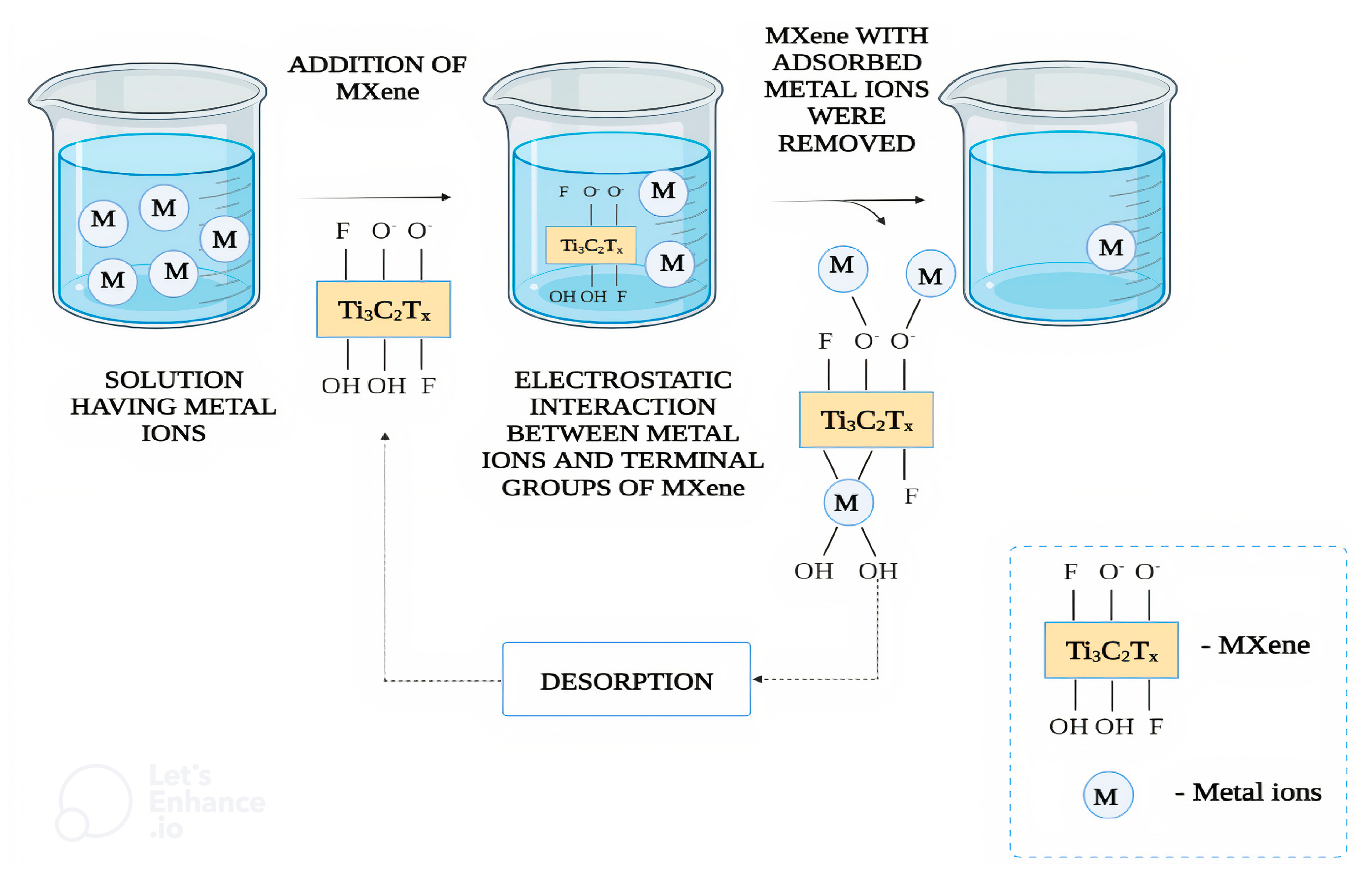
5. Pollutant Removal with MXenes in Wastewater via Adsorption
5.1. Adsorption of Inorganic Contaminant
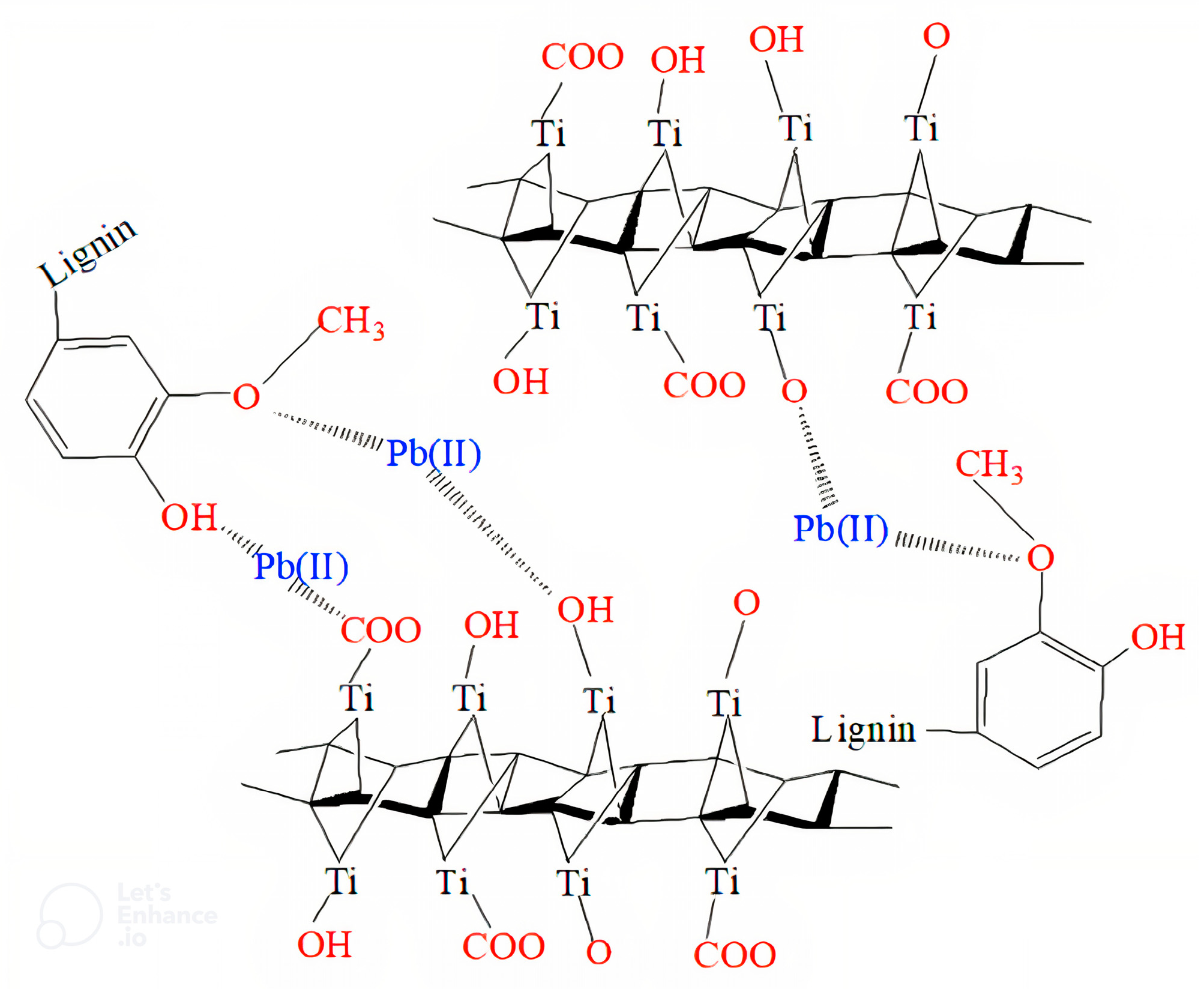
5.1.1. Lead Adsorption
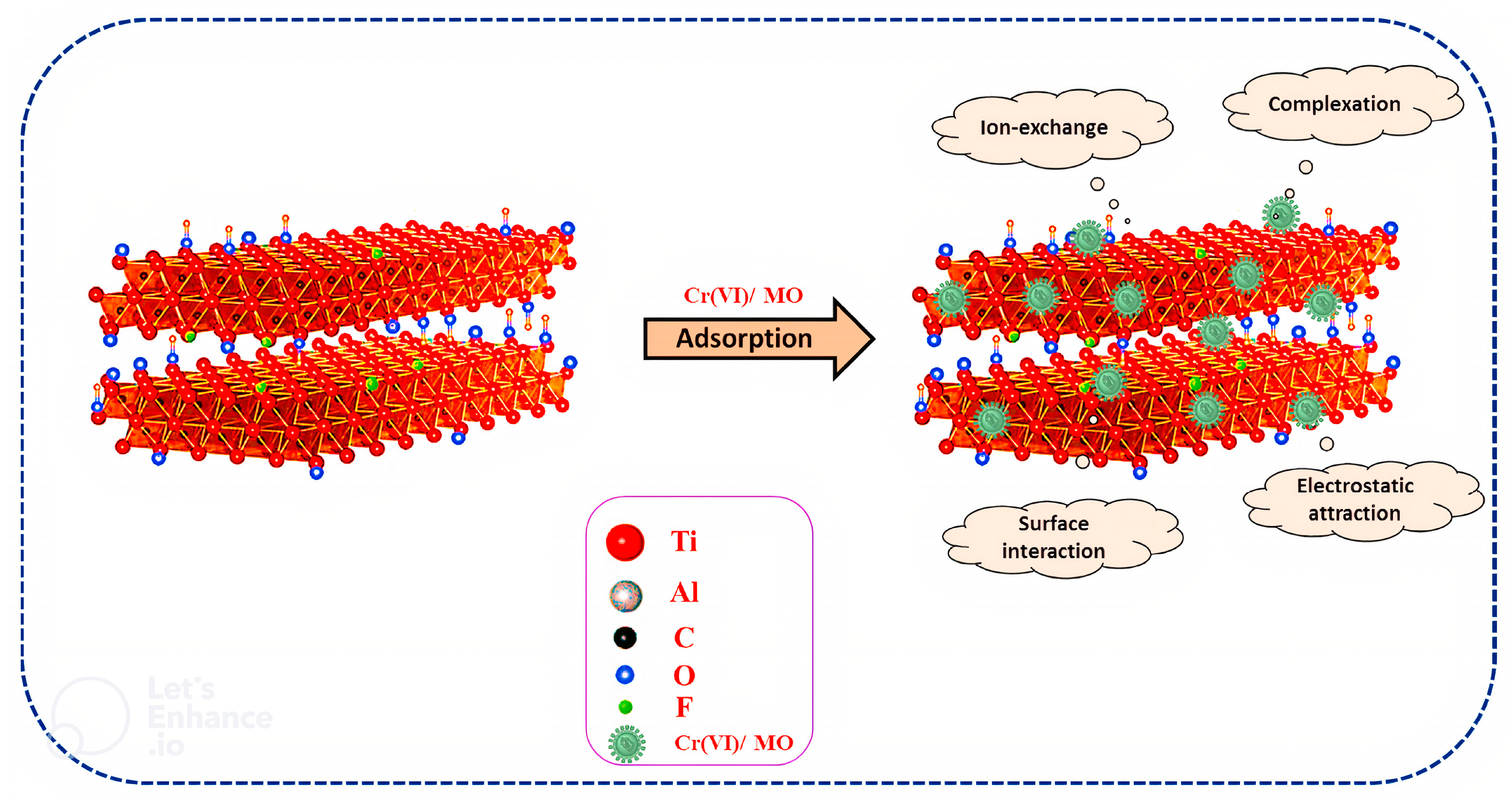
5.1.2. Chromium Adsorption
5.1.3. Copper Adsorption
5.1.4. Mercury Adsorption
5.1.5. Mechanisms of Heavy Metals Degradation
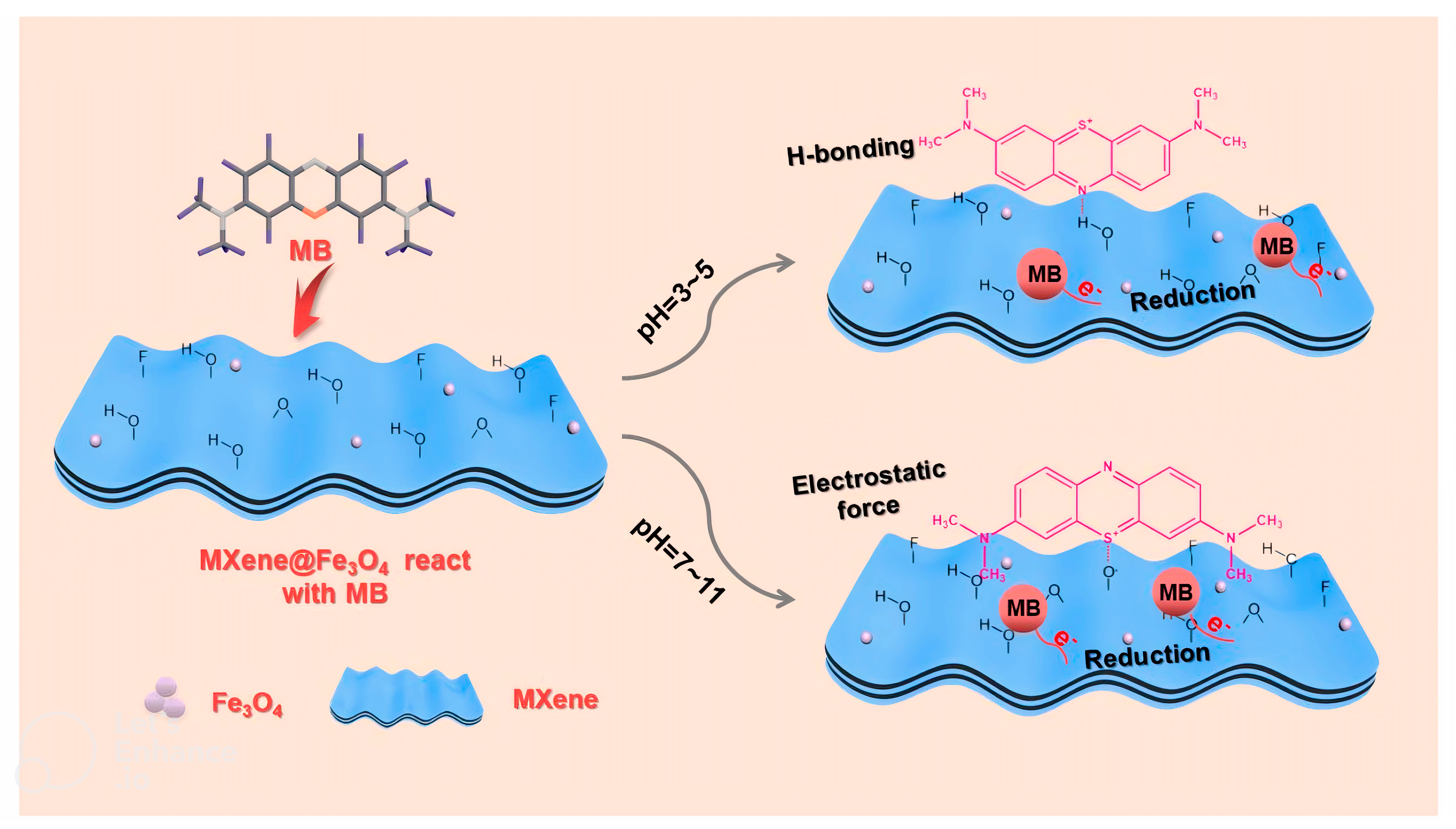
5.2. Adsorption of Organic Contaminant
Mechanisms of Heavy Metals Degradation
6. Combining Fenton Method with MXene-Based Materials for Organic Pollutants Degradation
6.1. Oxidation of H2O2 Activated by MXene
Mechanisms of H2O2 Activation with MXene-Based Materials
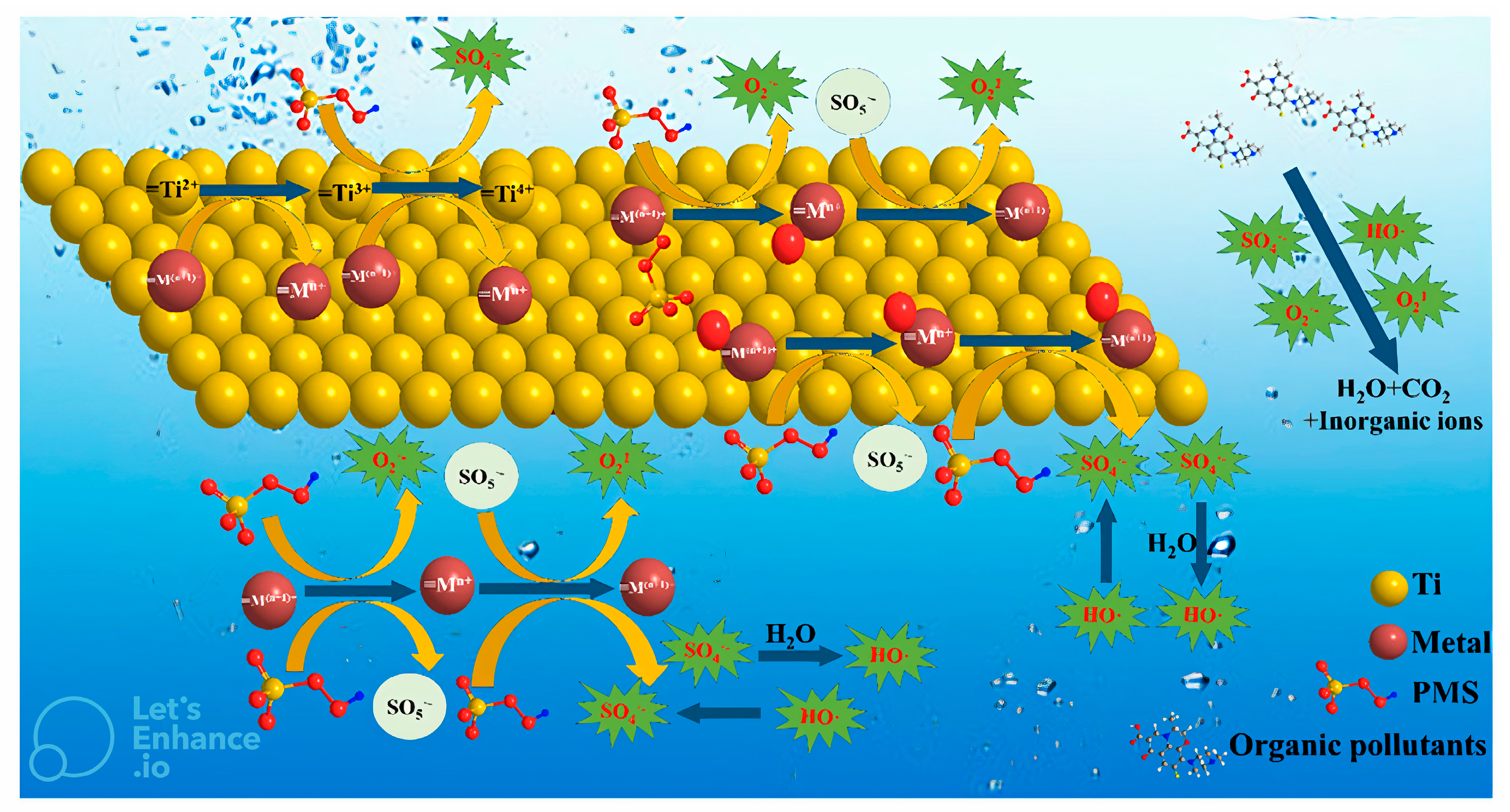
6.2. Utilizing MXene-Based Materials to Activate PS
Mechanisms of Activating PMS
7. An MXene-Based Photocatalysis System for Pollutants Removal in Wastewater
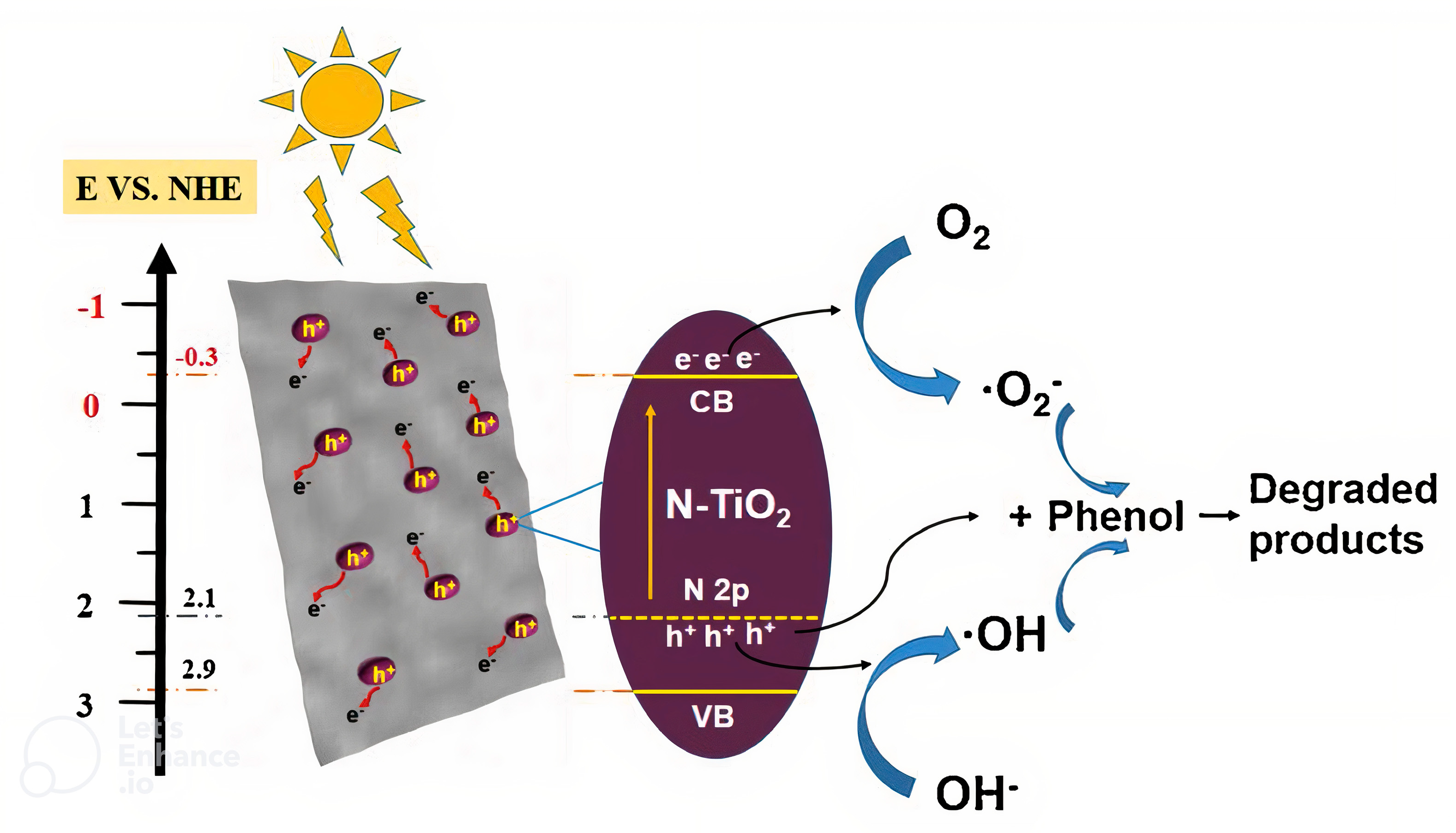
7.1. Composite Based on MXene/TiO2
7.2. MXene and Other Material-Based Composite
8. Treatment of Wastewater with MXene-Based Membranes
8.1. Membrane Made from Pristine MXene
8.2. Hybrid Membranes Based on MXene
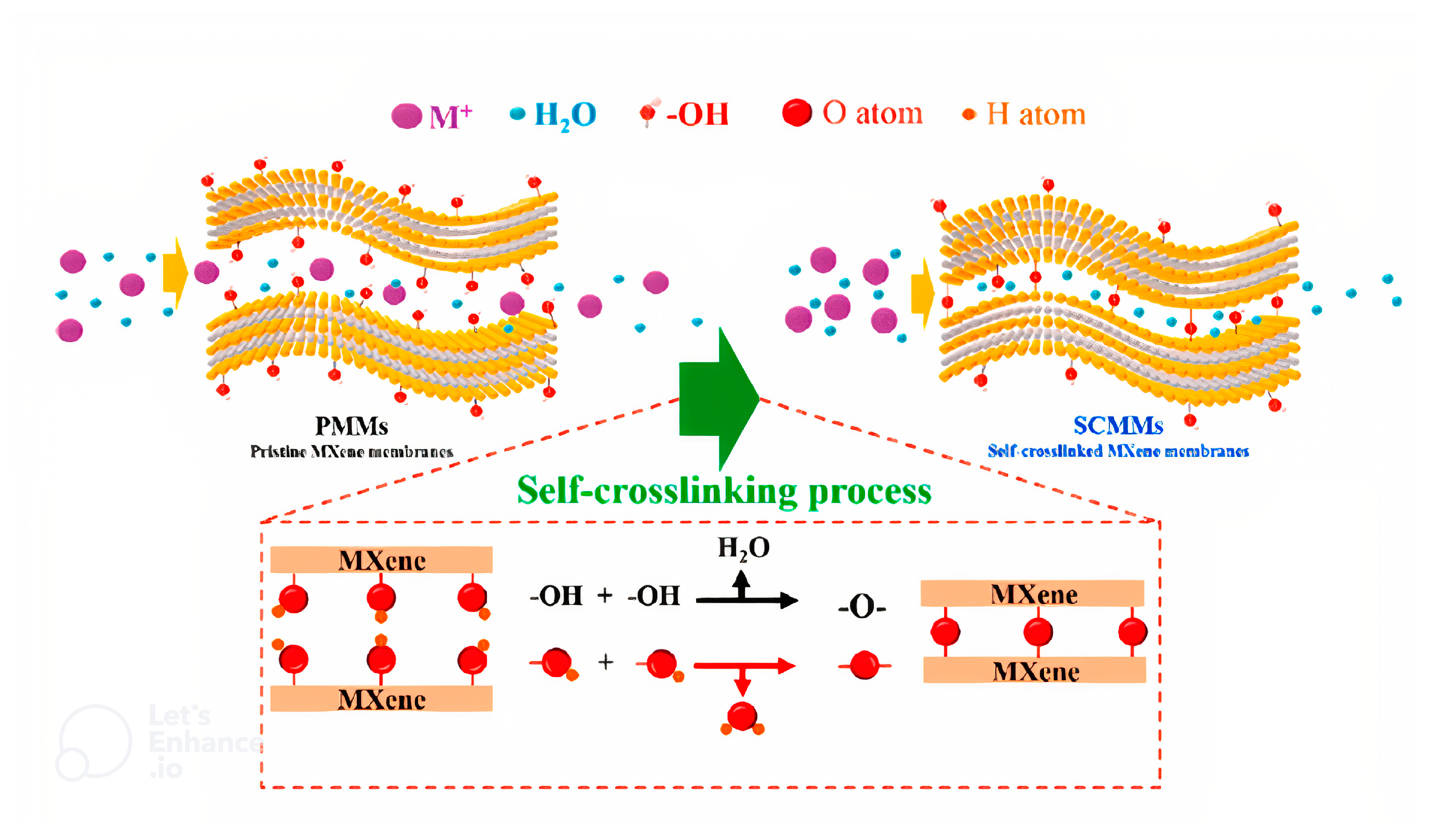
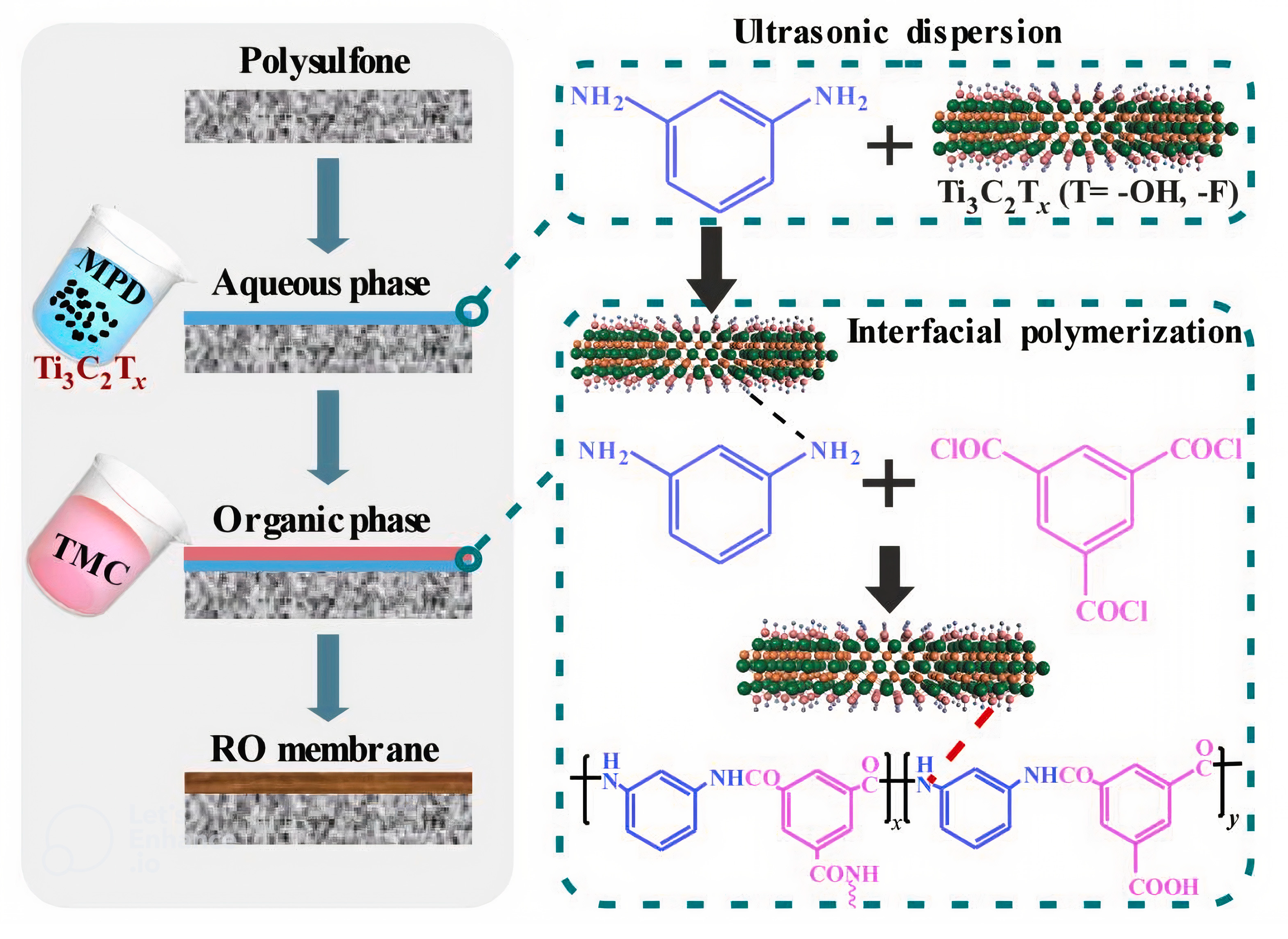
8.3. Composites of MXene and Polymer for High Rejection of Salt
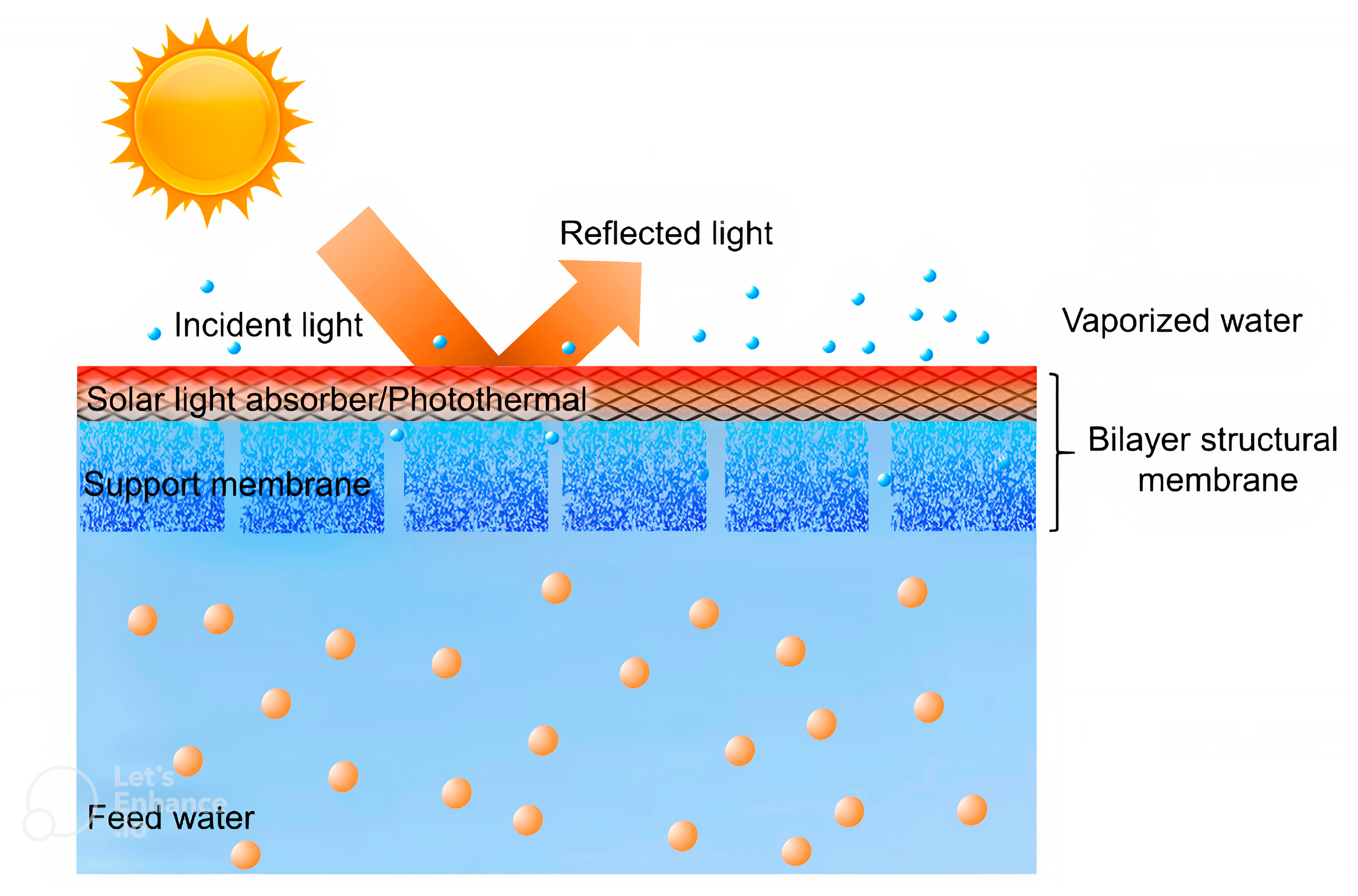
8.4. MXene-Based Photothermal Membranes
9. MXene Nanomaterial Regeneration
10. Issues of Toxicity
11. Challenges and Recommendations
- MXenes should be investigated as an adsorbent in continuous operating systems;
- Hazardous HF is traditionally used to synthesize MXenes, which poses serious health risks. HF can be substituted with greener or less toxic chemicals to ensure an environmentally friendly MXene synthesis;
- It is necessary to study MXene-based nanomaterials’ surface properties to enhance the performance of adsorption mechanisms;
- Designing an environmentally friendly and cost-effective large-scale production system for MXenes leads to opening up new avenues of commercial application for MXenes;
- For further biomedical applications, MXene-based nanomaterials must be tested for their toxicity to both the environment and humans;
- Detaching the photothermal nanoparticles from the MXene-based photothermal membranes surface is another key challenge for practical applications, which reduces the membrane’s photothermal efficiency and causes secondary pollution. In this situation, chemical bonding or grafting could provide a stronger adhesive bond between the photothermal nanoparticles and the membrane surface than physical attachment.
- There are numerous applications of MXene quantum dots (MQDs) in wastewater purification, and they can be viewed as a new generation of smart nanosystems.
12. Future View on MXene
13. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Nguyen, N.T.T.; Nguyen, L.M.; Nguyen, T.T.T.; Liew, R.K.; Nguyen, D.T.C.; Van Tran, T. Recent advances on botanical biosynthesis of nanoparticles for catalytic, water treatment and agricultural applications: A review. Sci. Total Environ. 2022, 827, 154160. [Google Scholar] [CrossRef] [PubMed]
- Amakiri, K.T.; Canon, A.R.; Molinari, M.; Angelis-Dimakis, A. Review of oilfield produced water treatment technologies. Chemosphere 2022, 298, 134064. [Google Scholar] [CrossRef]
- Shi, L.; Pouramini, S. Adaptive façade for building energy efficiency improvement by arithmetical optimization algorithm. Concurr. Comput. Pract. Exp. 2022, 34, e7152. [Google Scholar] [CrossRef]
- Nayebi, B.; Ghalebizade, M.; Niavol, K.P. Removal of Acid Red 131 by Peroxi-Coagulation Using Stainless Steel and Aluminum Electrodes: A Comparative Study. Water Conserv. Sci. Eng. 2021, 6, 201–211. [Google Scholar] [CrossRef]
- Ruiz-Santoyo, V.; Andrade-Espinoza, B.A.; Romero-Toledo, R.; Anaya-Esparza, L.M.; Villagrán, Z.; Guerra-Contreras, A. Use of Nanostructured Photocatalysts for Dye Degradation: A Review. Period. Polytech. Chem. Eng. 2022, 66, 367–393. [Google Scholar] [CrossRef]
- Ramalingam, G.; Nagapandiselvi, P.; Priya, A.; Rajendran, S. A review of graphene-based semiconductors for photocatalytic degradation of pollutants in wastewater. Chemosphere 2022, 300, 134391. [Google Scholar] [CrossRef]
- Pelosato, R.; Bolognino, I.; Fontana, F.; Sora, I.N. Applications of Heterogeneous Photocatalysis to the Degradation of Oxytetracycline in Water: A Review. Molecules 2022, 27, 2743. [Google Scholar] [CrossRef]
- Joseph, C.G.; Taufiq-Yap, Y.H.; Affandi, N.A.; Nga, J.L.H.; Vijayan, V. Photocatalytic treatment of detergent-contaminated wastewater: A short review on current progress. Korean J. Chem. Eng. 2022, 39, 484–498. [Google Scholar] [CrossRef]
- Feng, J.; Luo, X.; Gao, M.; Abbas, A.; Xu, Y.-P.; Pouramini, S. Minimization of energy consumption by building shape optimization using an improved Manta-Ray Foraging Optimization algorithm. Energy Rep. 2021, 7, 1068–1078. [Google Scholar] [CrossRef]
- Abdulhameed, A.S.; Jawad, A.H.; Ridwan, M.; Khadiran, T.; Wilson, L.D.; Yaseen, Z.M. Chitosan/carbon-doped TiO2 composite for adsorption of two anionic dyes in solution and gaseous SO2 capture: Experimental modeling and optimization. J. Polym. Environ. 2022, 30, 4619–4636. [Google Scholar] [CrossRef]
- Jawad, A.H.; Hameed, B.; Abdulhameed, A.S. Synthesis of biohybrid magnetic chitosan-polyvinyl alcohol/MgO nanocomposite blend for remazol brilliant blue R dye adsorption: Solo and collective parametric optimization. Polym. Bull. 2022, 1–21. [Google Scholar] [CrossRef]
- Kuang, P.; Low, J.; Cheng, B.; Yu, J.; Fan, J. MXene-based photocatalysts. J. Mater. Sci. Technol. 2020, 56, 18–44. [Google Scholar] [CrossRef]
- Jang, J.; Shahzad, A.; Woo, S.H.; Lee, D.S. Magnetic Ti3C2Tx (Mxene) for diclofenac degradation via the ultraviolet/chlorine advanced oxidation process. Environ. Res. 2020, 182, 108990. [Google Scholar] [CrossRef] [PubMed]
- Bilal, M.; Mehmood, S.; Rasheed, T.; Iqbal, H.M. Antibiotics traces in the aquatic environment: Persistence and adverse environmental impact. Curr. Opin. Environ. Sci. Health 2020, 13, 68–74. [Google Scholar] [CrossRef]
- Toth, L. Transition Metal Carbides and Nitrides; Elsevier: Amsterdam, The Netherlands, 2014. [Google Scholar]
- Gogotsi, Y.G.; Andrievski, R.A. Materials Science of Carbides, Nitrides and Borides; Springer Science & Business Media: Berlin/Heidelberg, Germany, 2012; Volume 68. [Google Scholar]
- Naguib, M.; Barsoum, M.W.; Gogotsi, Y. Ten years of progress in the synthesis and development of MXenes. Adv. Mater. 2021, 33, 2103393. [Google Scholar] [CrossRef]
- Nair, S.; Gohel, J.V. A Review on Contemporary Hole Transport Materials for Perovskite Solar Cells. In Nanotechnology for Energy and Environmental Engineering; Springer: Cham, Switzerland, 2020; pp. 145–168. [Google Scholar] [CrossRef]
- Jayakumar, A.; Surendranath, A.; Mohanan, P.V. 2D materials for next generation healthcare applications. Int. J. Pharm. 2018, 1–2, 309–321. [Google Scholar] [CrossRef]
- Szuplewska, A.; Kulpińska, D.; Dybko, A.; Chudy, M.; Jastrzębska, A.M.; Olszyna, A.; Brzózka, Z. Future applications of MXenes in biotechnology, nanomedicine, and sensors. Trends Biotechnol. 2020, 38, 264–279. [Google Scholar] [CrossRef]
- Rasool, K.; Pandey, R.P.; Rasheed, P.A.; Buczek, S.; Gogotsi, Y.; Mahmoud, K.A. Water treatment and environmental remediation applications of two-dimensional metal carbides (MXenes). Mater. Today 2019, 30, 80–102. [Google Scholar] [CrossRef]
- Naguib, M.; Kurtoglu, M.; Presser, V.; Lu, J.; Niu, J.; Heon, M.; Hultman, L.; Gogotsi, Y.; Barsoum, M.W. Two-dimensional nanocrystals produced by exfoliation of Ti3AlC2. Adv. Mater. 2011, 23, 4248–4253. [Google Scholar] [CrossRef] [Green Version]
- Al-Hamadani, Y.A.; Jun, B.-M.; Yoon, M.; Taheri-Qazvini, N.; Snyder, S.A.; Jang, M.; Heo, J.; Yoon, Y. Applications of MXene-based membranes in water purification: A review. Chemosphere 2020, 254, 126821. [Google Scholar] [CrossRef]
- Feng, J.; Yang, J.; Shen, Y.; Deng, W.; Chen, W.; Ma, Y.; Chen, Z.; Dong, S. Mesoporous silica nanoparticles prepared via a one-pot method for controlled release of abamectin: Properties and applications. Microporous Mesoporous Mater. 2021, 311, 110688. [Google Scholar] [CrossRef]
- Khandelwal, N.; Darbha, G.K. A decade of exploring MXenes as aquatic cleaners: Covering a broad range of contaminants, current challenges and future trends. Chemosphere 2021, 279, 130587. [Google Scholar] [CrossRef] [PubMed]
- Othman, Z.; Mackey, H.R.; Mahmoud, K.A. A critical overview of MXenes adsorption behavior toward heavy metals. Chemosphere 2022, 295, 133849. [Google Scholar] [CrossRef] [PubMed]
- Zhu, J.; Ha, E.; Zhao, G.; Zhou, Y.; Huang, D.; Yue, G.; Hu, L.; Sun, N.; Wang, Y.; Lee, L.Y.S. Recent advance in MXenes: A promising 2D material for catalysis, sensor and chemical adsorption. Coord. Chem. Rev. 2017, 352, 306–327. [Google Scholar] [CrossRef]
- Zhang, X.; Zhang, Z.; Zhou, Z. MXene-based materials for electrochemical energy storage. J. Energy Chem. 2018, 27, 73–85. [Google Scholar] [CrossRef] [Green Version]
- Nan, J.; Guo, X.; Xiao, J.; Li, X.; Chen, W.; Wu, W.; Liu, H.; Wang, Y.; Wu, M.; Wang, G. Nanoengineering of 2D MXene-based materials for energy storage applications. Small 2021, 17, 1902085. [Google Scholar] [CrossRef]
- Zhang, A.; Liu, R.; Tian, J.; Huang, W.; Liu, J. MXene-based nanocomposites for energy conversion and storage applications. Chem.-A Eur. J. 2020, 26, 6342–6359. [Google Scholar] [CrossRef]
- Huang, H.; Jiang, R.; Feng, Y.; Ouyang, H.; Zhou, N.; Zhang, X.; Wei, Y. Recent development and prospects of surface modification and biomedical applications of MXenes. Nanoscale 2020, 12, 1325–1338. [Google Scholar] [CrossRef]
- Ng, V.M.H.; Huang, H.; Zhou, K.; Lee, P.S.; Que, W.; Xu, Z.J.; Kong, L.B. Correction: Recent progress in layered transition metal carbides and/or nitrides (MXenes) and their composites: Synthesis and applications. J. Mater. Chem. A 2017, 5, 8769. [Google Scholar]
- Deysher, G.; Shuck, C.E.; Hantanasirisakul, K.; Frey, N.C.; Foucher, A.C.; Maleski, K.; Sarycheva, A.; Shenoy, V.B.; Stach, E.A.; Anasori, B. Synthesis of Mo4VAlC4 MAX phase and two-dimensional Mo4VC4 MXene with five atomic layers of transition metals. ACS Nano 2019, 14, 204–217. [Google Scholar] [CrossRef]
- Darvish, M.; Pouramini, M.; Bahador, H. Towards Fine-grained Image Classification with Generative Adversarial Networks and Facial Landmark Detection. In Proceedings of the 2022 International Conference on Machine Vision and Image Processing (MVIP), New York, NY, USA, 25–27 March 2022; pp. 1–6. [Google Scholar]
- Chen, X.; Bei, G. Toughening mechanisms in nanolayered MAX phase ceramics—A review. Materials 2017, 10, 366. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yang, J.; Bao, W.; Jaumaux, P.; Zhang, S.; Wang, C.; Wang, G. MXene-based composites: Synthesis and applications in rechargeable batteries and supercapacitors. Adv. Mater. Interfaces 2019, 6, 1802004. [Google Scholar] [CrossRef]
- Khazaei, M.; Ranjbar, A.; Esfarjani, K.; Bogdanovski, D.; Dronskowski, R.; Yunoki, S. Insights into exfoliation possibility of MAX phases to MXenes. Phys. Chem. Chem. Phys. 2018, 20, 8579–8592. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Alhabeb, M.; Maleski, K.; Anasori, B.; Lelyukh, P.; Clark, L.; Sin, S.; Gogotsi, Y. Guidelines for synthesis and processing of two-dimensional titanium carbide (Ti3C2Tx MXene). Chem. Mater. 2017, 29, 7633–7644. [Google Scholar] [CrossRef]
- Tang, H.; Feng, H.; Wang, H.; Wan, X.; Liang, J.; Chen, Y. Highly conducting MXene–silver nanowire transparent electrodes for flexible organic solar cells. ACS Appl. Mater. Interfaces 2019, 11, 25330–25337. [Google Scholar] [CrossRef] [PubMed]
- Xie, X.; Xue, Y.; Li, L.; Chen, S.; Nie, Y.; Ding, W.; Wei, Z. Surface Al leached Ti3AlC2 as a substitute for carbon for use as a catalyst support in a harsh corrosive electrochemical system. Nanoscale 2014, 6, 11035–11040. [Google Scholar] [CrossRef] [PubMed]
- Lei, J.-C.; Zhang, X.; Zhou, Z. Recent advances in MXene: Preparation, properties, and applications. Front. Phys. 2015, 10, 276–286. [Google Scholar] [CrossRef]
- Lipatov, A.; Alhabeb, M.; Lukatskaya, M.R.; Boson, A.; Gogotsi, Y.; Sinitskii, A. Effect of synthesis on quality, electronic properties and environmental stability of individual monolayer Ti3C2 MXene flakes. Adv. Electron. Mater. 2016, 2, 1600255. [Google Scholar] [CrossRef] [Green Version]
- Mashtalir, O.; Naguib, M.; Mochalin, V.N.; Dall’Agnese, Y.; Heon, M.; Barsoum, M.W.; Gogotsi, Y. Intercalation and delamination of layered carbides and carbonitrides. Nat. Commun. 2013, 4, 1716. [Google Scholar] [CrossRef] [Green Version]
- Mashtalir, O.; Lukatskaya, M.R.; Zhao, M.Q.; Barsoum, M.W.; Gogotsi, Y. Amine-assisted delamination of Nb2C MXene for Li-ion energy storage devices. Adv. Mater. 2015, 27, 3501–3506. [Google Scholar] [CrossRef]
- Naguib, M.; Gogotsi, Y. Synthesis of two-dimensional materials by selective extraction. Acc. Chem. Res. 2015, 48, 128–135. [Google Scholar] [CrossRef] [PubMed]
- Halim, J.; Lukatskaya, M.R.; Cook, K.M.; Lu, J.; Smith, C.R.; Näslund, L.-Å.; May, S.J.; Hultman, L.; Gogotsi, Y.; Eklund, P. Transparent conductive two-dimensional titanium carbide epitaxial thin films. Chem. Mater. 2014, 26, 2374–2381. [Google Scholar] [CrossRef]
- Chang, F.; Li, C.; Yang, J.; Tang, H.; Xue, M. Synthesis of a new graphene-like transition metal carbide by de-intercalating Ti3AlC2. Mater. Lett. 2013, 109, 295–298. [Google Scholar] [CrossRef]
- Ying, Y.; Liu, Y.; Wang, X.; Mao, Y.; Cao, W.; Hu, P.; Peng, X. Two-dimensional titanium carbide for efficiently reductive removal of highly toxic chromium (VI) from water. ACS Appl. Mater. Interfaces 2015, 7, 1795–1803. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Liu, D.; Lian, W.; Hu, Q.; Liu, X.; Zhou, A. The preparation of V2CTx by facile hydrothermal-assisted etching processing and its performance in lithium-ion battery. J. Mater. Res. Technol. 2020, 9, 984–993. [Google Scholar] [CrossRef]
- Sun, W.; Shah, S.; Chen, Y.; Tan, Z.; Gao, H.; Habib, T.; Radovic, M.; Green, M. Electrochemical etching of Ti2AlC to Ti2CTx (MXene) in low-concentration hydrochloric acid solution. J. Mater. Chem. A 2017, 5, 21663–21668. [Google Scholar] [CrossRef]
- Naguib, M.; Mochalin, V.; Barsoum, M.W.; Gogotsi, Y.G. MXenes: A New Family of Two-Dimensional Materials—25th Anniversary Article; Wiley: Hoboken, NJ, USA, 2014; Volume 26, pp. 992–1005. [Google Scholar]
- Ghidiu, M.; Lukatskaya, M.R.; Zhao, M.-Q.; Gogotsi, Y.; Barsoum, M.W. Conductive two-dimensional titanium carbide ‘clay’with high volumetric capacitance. Nature 2014, 516, 78–81. [Google Scholar] [CrossRef]
- Shahzad, A.; Nawaz, M.; Moztahida, M.; Jang, J.; Tahir, K.; Kim, J.; Lim, Y.; Vassiliadis, V.S.; Woo, S.H.; Lee, D.S. Ti3C2Tx MXene core-shell spheres for ultrahigh removal of mercuric ions. Chem. Eng. J. 2019, 368, 400–408. [Google Scholar] [CrossRef]
- Zhang, P.; Wang, L.; Yuan, L.-Y.; Lan, J.-H.; Chai, Z.-F.; Shi, W.-Q. Sorption of Eu (III) on MXene-derived titanate structures: The effect of nano-confined space. Chem. Eng. J. 2019, 370, 1200–1209. [Google Scholar] [CrossRef]
- Wang, X.; Garnero, C.; Rochard, G.; Magne, D.; Morisset, S.; Hurand, S.; Chartier, P.; Rousseau, J.; Cabioc’h, T.; Coutanceau, C. A new etching environment (FeF3/HCl) for the synthesis of two-dimensional titanium carbide MXenes: A route towards selective reactivity vs. water. J. Mater. Chem. A 2017, 5, 22012–22023. [Google Scholar] [CrossRef]
- Zhang, X.; Wang, H.; Hu, R.; Huang, C.; Zhong, W.; Pan, L.; Feng, Y.; Qiu, T.; Zhang, C.J.; Yang, J. Novel solvothermal preparation and enhanced microwave absorption properties of Ti3C2Tx MXene modified by in situ coated Fe3O4 nanoparticles. Appl. Surf. Sci. 2019, 484, 383–391. [Google Scholar] [CrossRef]
- Guo, J.; Peng, Q.; Fu, H.; Zou, G.; Zhang, Q. Heavy-metal adsorption behavior of two-dimensional alkalization-intercalated MXene by first-principles calculations. J. Phys. Chem. C 2015, 119, 20923–20930. [Google Scholar] [CrossRef]
- Liu, G.; Shen, J.; Liu, Q.; Liu, G.; Xiong, J.; Yang, J.; Jin, W. Ultrathin two-dimensional MXene membrane for pervaporation desalination. J. Membr. Sci. 2018, 548, 548–558. [Google Scholar] [CrossRef]
- Malaki, M.; Maleki, A.; Varma, R.S. MXenes and ultrasonication. J. Mater. Chem. A 2019, 7, 10843–10857. [Google Scholar] [CrossRef]
- Li, X.; Huang, Z.; Zhi, C. Environmental stability of MXenes as energy storage materials. Front. Mater. 2019, 6, 312. [Google Scholar] [CrossRef] [Green Version]
- Maleski, K.; Mochalin, V.N.; Gogotsi, Y. Dispersions of two-dimensional titanium carbide MXene in organic solvents. Chem. Mater. 2017, 29, 1632–1640. [Google Scholar] [CrossRef]
- Shah, J.B. Synthesis of MXene-Epoxy Nanocomposites; Drexel University: Philadelphia, PA, USA, 2017. [Google Scholar]
- Lukatskaya, M.R.; Mashtalir, O.; Ren, C.E.; Dall’Agnese, Y.; Rozier, P.; Taberna, P.L.; Naguib, M.; Simon, P.; Barsoum, M.W.; Gogotsi, Y. Cation intercalation and high volumetric capacitance of two-dimensional titanium carbide. Science 2013, 341, 1502–1505. [Google Scholar] [CrossRef] [Green Version]
- Ma, R.; Sasaki, T. Two-dimensional oxide and hydroxide nanosheets: Controllable high-quality exfoliation, molecular assembly, and exploration of functionality. Acc. Chem. Res. 2015, 48, 136–143. [Google Scholar] [CrossRef]
- Bajraktarova-Valjakova, E.; Grozdanov, A.; Guguvcevski, L.; Korunoska-Stevkovska, V.; Kapusevska, B.; Gigovski, N.; Mijoska, A.; Bajraktarova-Misevska, C. Acid etching as surface treatment method for luting of glass-ceramic restorations, part 1: Acids, application protocol and etching effectiveness. Open Access Maced. J. Med. Sci. 2018, 6, 568. [Google Scholar] [CrossRef] [Green Version]
- Zhu, X.; Liu, B.; Hou, H.; Huang, Z.; Zeinu, K.M.; Huang, L.; Yuan, X.; Guo, D.; Hu, J.; Yang, J. Alkaline intercalation of Ti3C2 MXene for simultaneous electrochemical detection of Cd (II), Pb (II), Cu (II) and Hg (II). Electrochim. Acta 2017, 248, 46–57. [Google Scholar] [CrossRef]
- VahidMohammadi, A.; Kayali, E.; Orangi, J.; Beidaghi, M. Techniques for MXene delamination into single-layer flakes. In 2D Metal Carbides and Nitrides (MXenes); Springer: Berlin/Heidelberg, Germany, 2019; pp. 177–195. [Google Scholar]
- Zhou, J.; Gao, S.; Guo, Z.; Sun, Z. Ti-enhanced exfoliation of V2AlC into V2C MXene for lithium-ion battery anodes. Ceram. Int. 2017, 43, 11450–11454. [Google Scholar] [CrossRef]
- Xiao, J.; Wen, J.; Zhao, J.; Ma, X.; Gao, H.; Zhang, X. A safe etching route to synthesize highly crystalline Nb2CTx MXene for high performance asymmetric supercapacitor applications. Electrochim. Acta 2020, 337, 135803. [Google Scholar] [CrossRef]
- Ghidiu, M.; Naguib, M.; Shi, C.; Mashtalir, O.; Pan, L.; Zhang, B.; Yang, J.; Gogotsi, Y.; Billinge, S.J.; Barsoum, M.W. Synthesis and characterization of two-dimensional Nb4C3 (MXene). Chem. Commun. 2014, 50, 9517–9520. [Google Scholar] [CrossRef] [PubMed]
- Naguib, M.; Mashtalir, O.; Carle, J.; Presser, V.; Lu, J.; Hultman, L.; Gogotsi, Y.; Barsoum, M.; Carbides, T.-D.T.M. Two-dimensional transition metal carbides. ACS Nano 2012, 6, 1322–1331. [Google Scholar] [CrossRef] [PubMed]
- Urbankowski, P.; Anasori, B.; Makaryan, T.; Er, D.; Kota, S.; Walsh, P.L.; Zhao, M.; Shenoy, V.B.; Barsoum, M.W.; Gogotsi, Y. Synthesis of two-dimensional titanium nitride Ti4N3 (MXene). Nanoscale 2016, 8, 11385–11391. [Google Scholar] [CrossRef]
- Meshkian, R.; Näslund, L.-Å.; Halim, J.; Lu, J.; Barsoum, M.W.; Rosen, J. Synthesis of two-dimensional molybdenum carbide, Mo2C, from the gallium based atomic laminate Mo2Ga2C. Scr. Mater. 2015, 108, 147–150. [Google Scholar] [CrossRef] [Green Version]
- Zou, D.; Lee, Y.M. Design strategy of poly (vinylidene fluoride) membranes for water treatment. Prog. Polym. Sci. 2022, 128, 101535. [Google Scholar] [CrossRef]
- Fernandes, J.; Nemala, S.S.; De Bellis, G.; Capasso, A. Green Solvents for the Liquid Phase Exfoliation Production of Graphene: The Promising Case of Cyrene. Front. Chem. 2022, 10, 878799. [Google Scholar] [CrossRef]
- Luo, X.-J.; Li, L.; Zhang, H.-B.; Zhao, S.; Zhang, Y.; Chen, W.; Yu, Z.-Z. Multifunctional Ti3C2Tx MXene/Low-Density Polyethylene Soft Robots with Programmable Configuration for Amphibious Motions. ACS Appl. Mater. Interfaces 2021, 13, 45833–45842. [Google Scholar] [CrossRef]
- Liu, H.; Hu, Z.; Liu, Q.; Sun, P.; Wang, Y.; Chou, S.; Hu, Z.; Zhang, Z. Single-atom Ru anchored in nitrogen-doped MXene (Ti3C2Tx) as an efficient catalyst for the hydrogen evolution reaction at all pH values. J. Mater. Chem. A 2020, 8, 24710–24717. [Google Scholar] [CrossRef]
- Kumar, J.; Soomro, R.A.; Neiber, R.R.; Ahmed, N.; Medany, S.S.; Albaqami, M.D.; Nafady, A. Ni nanoparticles embedded Ti3C2Tx-MXene nanoarchitectures for electrochemical sensing of methylmalonic acid. Biosensors 2022, 12, 231. [Google Scholar] [CrossRef] [PubMed]
- Zou, X.; Liu, H.; Xu, H.; Wu, X.; Han, X.; Kang, J.; Reddy, K.M. A simple approach to synthesis Cr2CTx MXene for efficient hydrogen evolution reaction. Mater. Today Energy 2021, 20, 100668. [Google Scholar] [CrossRef]
- Vakili, M.; Cagnetta, G.; Huang, J.; Yu, G.; Yuan, J. Synthesis and regeneration of a MXene-based pollutant adsorbent by mechanochemical methods. Molecules 2019, 24, 2478. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Thirumal, V.; Yuvakkumar, R.; Kumar, P.S.; Ravi, G.; Keerthana, S.; Velauthapillai, D. Facile single-step synthesis of MXene@ CNTs hybrid nanocomposite by CVD method to remove hazardous pollutants. Chemosphere 2022, 286, 131733. [Google Scholar] [CrossRef]
- Rasheed, T.; Rasheed, A.; Munir, S.; Ajmal, S.; Shahzad, Z.M.; Alsafari, I.A.; Ragab, S.A.; Agboola, P.O.; Shakir, I. A cost-effective approach to synthesize NiFe2O4/MXene heterostructures for enhanced photodegradation performance and anti-bacterial activity. Adv. Powder Technol. 2021, 32, 2248–2257. [Google Scholar] [CrossRef]
- Anasori, B.; Lukatskaya, M.R.; Gogotsi, Y. 2D metal carbides and nitrides (MXenes) for energy storage. Nat. Rev. Mater. 2017, 2, 16098. [Google Scholar] [CrossRef]
- Zhang, H.; Yu, D.; Wang, W.; Gao, P.; Zhang, L.; Zhong, S.; Liu, B. Construction of a novel BON-Br-AgBr heterojunction photocatalysts as a direct Z-scheme system for efficient visible photocatalytic activity. Appl. Surf. Sci. 2019, 497, 143820. [Google Scholar] [CrossRef]
- Wang, H.; Wu, Y.; Zhang, J.; Li, G.; Huang, H.; Zhang, X.; Jiang, Q. Enhancement of the electrical properties of MXene Ti3C2 nanosheets by post-treatments of alkalization and calcination. Mater. Lett. 2015, 160, 537–540. [Google Scholar] [CrossRef]
- Wang, H.; Wu, Y.; Yuan, X.; Zeng, G.; Zhou, J.; Wang, X.; Chew, J.W. Clay-inspired MXene-based electrochemical devices and photo-electrocatalyst: State-of-the-art progresses and challenges. Adv. Mater. 2018, 30, 1704561. [Google Scholar] [CrossRef]
- Kurtoglu, M.; Naguib, M.; Gogotsi, Y.; Barsoum, M.W. First principles study of two-dimensional early transition metal carbides. MRS Commun. 2012, 2, 133–137. [Google Scholar] [CrossRef]
- Magnuson, M.; Halim, J.; Näslund, L.-Å. Chemical bonding in carbide MXene nanosheets. J. Electron. Spectrosc. Relat. Phenom. 2018, 224, 27–32. [Google Scholar] [CrossRef] [Green Version]
- Jin, W.; Wu, S.; Wang, Z. Structural, electronic and mechanical properties of two-dimensional Janus transition metal carbides and nitrides. Phys. E Low-Dimens. Syst. Nanostruct. 2018, 103, 307–313. [Google Scholar] [CrossRef]
- Morales-García, Á.; Fernández-Fernández, A.; Viñes, F.; Illas, F. CO2 abatement using two-dimensional MXene carbides. J. Mater. Chem. A 2018, 6, 3381–3385. [Google Scholar] [CrossRef]
- Wang, H.; Peng, R.; Hood, Z.D.; Naguib, M.; Adhikari, S.P.; Wu, Z. Titania Composites with 2D Transition Metal Carbides as Photocatalysts for Hydrogen Production under Visible-Light Irradiation. ChemSusChem 2016, 9, 1490–1497. [Google Scholar] [CrossRef]
- Zhuang, Y.; Liu, Y.; Meng, X. Fabrication of TiO2 nanofibers/MXene Ti3C2 nanocomposites for photocatalytic H2 evolution by electrostatic self-assembly. Appl. Surf. Sci. 2019, 496, 143647. [Google Scholar] [CrossRef]
- Ye, M.; Wang, X.; Liu, E.; Ye, J.; Wang, D. Boosting the photocatalytic activity of P25 for carbon dioxide reduction by using a surface-alkalinized titanium carbide MXene as cocatalyst. ChemSusChem 2018, 11, 1606–1611. [Google Scholar] [CrossRef] [PubMed]
- Hantanasirisakul, K.; Zhao, M.Q.; Urbankowski, P.; Halim, J.; Anasori, B.; Kota, S.; Ren, C.E.; Barsoum, M.W.; Gogotsi, Y. Fabrication of Ti3C2Tx MXene transparent thin films with tunable optoelectronic properties. Adv. Electron. Mater. 2016, 2, 1600050. [Google Scholar] [CrossRef]
- Huang, K.; Li, Z.; Lin, J.; Han, G.; Huang, P. Correction: Two-dimensional transition metal carbides and nitrides (MXenes) for biomedical applications. Chem. Soc. Rev. 2018, 47, 6889. [Google Scholar] [CrossRef] [Green Version]
- Dutta, A.; Basu, S.; Mukherjee, S.K.; Hossain, S.T. Wastewater treatment by microbial biofilm: A distinct possibility. In Microbial Ecology of Wastewater Treatment Plants; Elsevier: Amsterdam, The Netherlands, 2021; pp. 435–468. [Google Scholar]
- Singh, J.; Yadav, P.; Pal, A.K.; Mishra, V. Water pollutants: Origin and status. In Sensors in Water Pollutants Monitoring: Role of Material; Springer: Berlin/Heidelberg, Germany, 2020; pp. 5–20. [Google Scholar]
- Hosseinikhah, M.; Mokhtarani, N. Landfill leachate post-treatment by the photoelectro-peroxone process using a baffled reactor. Sep. Purif. Technol. 2023, 306, 122549. [Google Scholar] [CrossRef]
- Chowdhary, P.; Raj, A.; Bharagava, R.N. Environmental pollution and health hazards from distillery wastewater and treatment approaches to combat the environmental threats: A review. Chemosphere 2018, 194, 229–246. [Google Scholar] [CrossRef]
- Pouramini, Z.; Ayati, B.; Babapoor, A. Enhancing PFC ability to dye removal and power generation simultaneously via conductive spheres in the anodic chamber. J. Electroanal. Chem. 2022, 917, 116410. [Google Scholar] [CrossRef]
- Crini, G.; Lichtfouse, E. Advantages and disadvantages of techniques used for wastewater treatment. Environ. Chem. Lett. 2019, 17, 145–155. [Google Scholar] [CrossRef]
- Jawad, A.H.; Saber, S.E.M.; Abdulhameed, A.S.; Reghioua, A.; ALOthman, Z.A.; Wilson, L.D. Mesoporous activated carbon from mangosteen (Garcinia mangostana) peels by H3PO4 assisted microwave: Optimization, characterization, and adsorption mechanism for methylene blue dye removal. Diam. Relat. Mater. 2022, 129, 109389. [Google Scholar] [CrossRef]
- Kumar, P.S.; Joshiba, G.J.; Femina, C.C.; Varshini, P.; Priyadharshini, S.; Karthick, M.; Jothirani, R. A critical review on recent developments in the low-cost adsorption of dyes from wastewater. Desalin. Water Treat. 2019, 172, 395–416. [Google Scholar] [CrossRef]
- Pouramini, Z.; Mousavi, S.M.; Babapoor, A.; Hashemi, S.A.; Lai, C.W.; Mazaheri, Y.; Chiang, W.-H. Effect of Metal Atom in Zeolitic Imidazolate Frameworks (ZIF-8 & 67) for Removal of Dyes and Antibiotics from Wastewater: A Review. Catalysts 2023, 13, 155. [Google Scholar]
- Jawad, A.H.; Rangabhashiyam, S.; Abdulhameed, A.S.; Syed-Hassan, S.S.A.; ALOthman, Z.A.; Wilson, L.D. Process optimization and adsorptive mechanism for reactive blue 19 dye by magnetic crosslinked chitosan/MgO/Fe3O4 biocomposite. J. Polym. Environ. 2022, 30, 2759–2773. [Google Scholar] [CrossRef]
- TaheriAshtiani, N.; Ayati, B. Using chitosan-based heterogeneous catalyst for degradation of Acid Blue 25 in the effective electro-Fenton process with rotating cathodes. J. Electroanal. Chem. 2022, 905, 115983. [Google Scholar] [CrossRef]
- Nayebi, B.; Niavol, K.P.; Nayebi, B.; Kim, S.Y.; Nam, K.T.; Jang, H.W.; Varma, R.S.; Shokouhimehr, M. Prussian blue-based nanostructured materials: Catalytic applications for environmental remediation and energy conversion. Mol. Catal. 2021, 514, 111835. [Google Scholar] [CrossRef]
- Saravanan, A.; Kumar, P.S.; Jeevanantham, S.; Karishma, S.; Tajsabreen, B.; Yaashikaa, P.; Reshma, B. Effective water/wastewater treatment methodologies for toxic pollutants removal: Processes and applications towards sustainable development. Chemosphere 2021, 280, 130595. [Google Scholar] [CrossRef]
- Pouramini, Z. The VOCs catalytic combustion by perovskite catalysts: A mini-review. Adv. Appl. NanoBio-Technol. 2022, 3, 23–30. [Google Scholar]
- Amini, Z.P. Using nanomembrane to heavy metal removal from wastewater: A mini-review. Adv. Appl. NanoBio-Technol. 2022, 3, 7–13. [Google Scholar]
- Wang, D.; Guo, F.; Wu, Y.; Li, Z.; Wu, G. Technical, economic and environmental assessment of coagulation/filtration tertiary treatment processes in full-scale wastewater treatment plants. J. Clean. Prod. 2018, 170, 1185–1194. [Google Scholar] [CrossRef]
- Nidheesh, P.; Scaria, J.; Babu, D.S.; Kumar, M.S. An overview on combined electrocoagulation-degradation processes for the effective treatment of water and wastewater. Chemosphere 2021, 263, 127907. [Google Scholar] [CrossRef] [PubMed]
- Paździor, K.; Bilińska, L.; Ledakowicz, S. A review of the existing and emerging technologies in the combination of AOPs and biological processes in industrial textile wastewater treatment. Chem. Eng. J. 2019, 376, 120597. [Google Scholar] [CrossRef]
- Zhao, C.; Dong, Y.; Feng, Y.; Li, Y.; Dong, Y. Thermal desorption for remediation of contaminated soil: A review. Chemosphere 2019, 221, 841–855. [Google Scholar] [CrossRef]
- Guo, Z.; Sun, Y.; Pan, S.-Y.; Chiang, P.-C. Integration of green energy and advanced energy-efficient technologies for municipal wastewater treatment plants. Int. J. Environ. Res. Public Health 2019, 16, 1282. [Google Scholar] [CrossRef] [Green Version]
- Udaiyappan, A.F.M.; Hasan, H.A.; Takriff, M.S.; Abdullah, S.R.S. A review of the potentials, challenges and current status of microalgae biomass applications in industrial wastewater treatment. J. Water Process Eng. 2017, 20, 8–21. [Google Scholar] [CrossRef]
- Nanda, S.; Berruti, F. Municipal solid waste management and landfilling technologies: A review. Environ. Chem. Lett. 2021, 19, 1433–1456. [Google Scholar] [CrossRef]
- Karimi-Maleh, H.; Kumar, B.G.; Rajendran, S.; Qin, J.; Vadivel, S.; Durgalakshmi, D.; Gracia, F.; Soto-Moscoso, M.; Orooji, Y.; Karimi, F. Tuning of metal oxides photocatalytic performance using Ag nanoparticles integration. J. Mol. Liq. 2020, 314, 113588. [Google Scholar] [CrossRef]
- Jawad, A.H.; Abdulhameed, A.S.; Selvasembian, R.; ALOthman, Z.A.; Wilson, L.D. Magnetic biohybrid chitosan-ethylene glycol diglycidyl ether/magnesium oxide/Fe3O4 nanocomposite for textile dye removal: Box–Behnken design optimization and mechanism study. J. Polym. Res. 2022, 29, 207. [Google Scholar] [CrossRef]
- Baby, R.; Saifullah, B.; Hussein, M.Z. Carbon nanomaterials for the treatment of heavy metal-contaminated water and environmental remediation. Nanoscale Res. Lett. 2019, 14, 341. [Google Scholar] [CrossRef] [Green Version]
- Lu, Y.C.; Mao, J.H.; Zhang, W.; Wang, C.; Cao, M.; Wang, X.D.; Wang, K.Y.; Xiong, X.H. A novel strategy for selective removal and rapid collection of triclosan from aquatic environment using magnetic molecularly imprinted nano− polymers. Chemosphere 2020, 238, 124640. [Google Scholar] [CrossRef]
- Lakouraj, M.M.; Mojerlou, F.; Zare, E.N. Nanogel and superparamagnetic nanocomposite based on sodium alginate for sorption of heavy metal ions. Carbohydr. Polym. 2014, 106, 34–41. [Google Scholar] [CrossRef]
- El-Naggar, M.E.; Radwan, E.K.; El-Wakeel, S.T.; Kafafy, H.; Gad-Allah, T.A.; El-Kalliny, A.S.; Shaheen, T.I. Synthesis, characterization and adsorption properties of microcrystalline cellulose based nanogel for dyes and heavy metals removal. Int. J. Biol. Macromol. 2018, 113, 248–258. [Google Scholar] [CrossRef]
- Zain, Z.M.; Abdulhameed, A.S.; Jawad, A.H.; ALOthman, Z.A.; Yaseen, Z.M. A pH-sensitive surface of chitosan/sepiolite clay/algae biocomposite for the removal of malachite green and remazol brilliant blue R dyes: Optimization and adsorption mechanism study. J. Polym. Environ. 2023, 31, 501–518. [Google Scholar] [CrossRef]
- Abdulhameed, A.S.; Jawad, A.H.; Kashi, E.; Radzun, K.A.; ALOthman, Z.A.; Wilson, L.D. Insight into adsorption mechanism, modeling, and desirability function of crystal violet and methylene blue dyes by microalgae: Box-Behnken design application. Algal Res. 2022, 67, 102864. [Google Scholar] [CrossRef]
- Razali, N.S.; Abdulhameed, A.S.; Jawad, A.H.; ALOthman, Z.A.; Yousef, T.A.; Al-Duaij, O.K.; Alsaiari, N.S. High-surface-area-activated carbon derived from mango peels and seeds wastes via microwave-induced ZnCl2 activation for adsorption of methylene blue dye molecules: Statistical optimization and mechanism. Molecules 2022, 27, 6947. [Google Scholar] [CrossRef] [PubMed]
- Jani, N.A.; Haddad, L.; Abdulhameed, A.S.; Jawad, A.H.; ALOthman, Z.A.; Yaseen, Z.M. Modeling and optimization of the adsorptive removal of crystal violet dye by durian (Durio zibethinus) seeds powder: Insight into kinetic, isotherm, thermodynamic, and adsorption mechanism. Biomass Convers. Biorefin. 2022, 1–14. [Google Scholar] [CrossRef]
- Suhaimi, A.; Abdulhameed, A.S.; Jawad, A.H.; Yousef, T.A.; Al Duaij, O.; ALOthman, Z.A.; Wilson, L.D. Production of large surface area activated carbon from a mixture of carrot juice pulp and pomegranate peel using microwave radiation-assisted ZnCl2 activation: An optimized removal process and tailored adsorption mechanism of crystal violet dye. Diam. Relat. Mater. 2022, 130, 109456. [Google Scholar] [CrossRef]
- Schultz, T.; Frey, N.C.; Hantanasirisakul, K.; Park, S.; May, S.J.; Shenoy, V.B.; Gogotsi, Y.; Koch, N. Surface termination dependent work function and electronic properties of Ti3C2Tx MXene. Chem. Mater. 2019, 31, 6590–6597. [Google Scholar] [CrossRef] [Green Version]
- Tunesi, M.M.; Soomro, R.A.; Han, X.; Zhu, Q.; Wei, Y.; Xu, B. Application of MXenes in environmental remediation technologies. Nano Converg. 2021, 8, 5. [Google Scholar] [CrossRef]
- Ibrahim, Y.; Kassab, A.; Eid, K.M.; Abdullah, A.; Ozoemena, K.I.; Elzatahry, A. Unveiling fabrication and environmental remediation of MXene-based nanoarchitectures in toxic metals removal from wastewater: Strategy and mechanism. Nanomaterials 2020, 10, 885. [Google Scholar] [CrossRef]
- Velusamy, K.; Chellam, P.; Kumar, P.S.; Venkatachalam, J.; Periyasamy, S.; Saravanan, R. Functionalization of MXene-based nanomaterials for the treatment of micropollutants in aquatic system: A review. Environ. Pollut. 2022, 301, 119034. [Google Scholar] [CrossRef]
- Dong, Y.; Sang, D.; He, C.; Sheng, X.; Lei, L. Mxene/alginate composites for lead and copper ion removal from aqueous solutions. RSC Adv. 2019, 9, 29015–29022. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jin, L.; Chai, L.; Yang, W.; Wang, H.; Zhang, L. Two-dimensional titanium carbides (Ti3C2Tx) functionalized by poly (m-phenylenediamine) for efficient adsorption and reduction of hexavalent chromium. Int. J. Environ. Res. Public Health 2020, 17, 167. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zou, G.; Guo, J.; Peng, Q.; Zhou, A.; Zhang, Q.; Liu, B. Synthesis of urchin-like rutile titania carbon nanocomposites by iron-facilitated phase transformation of MXene for environmental remediation. J. Mater. Chem. A 2016, 4, 489–499. [Google Scholar] [CrossRef]
- Karthikeyan, P.; Ramkumar, K.; Pandi, K.; Fayyaz, A.; Meenakshi, S.; Park, C.M. Effective removal of Cr (VI) and methyl orange from the aqueous environment using two-dimensional (2D) Ti3C2Tx MXene nanosheets. Ceram. Int. 2021, 47, 3692–3698. [Google Scholar] [CrossRef]
- Krstić, V.; Urošević, T.; Pešovski, B. A review on adsorbents for treatment of water and wastewaters containing copper ions. Chem. Eng. Sci. 2018, 192, 273–287. [Google Scholar] [CrossRef]
- Shahzad, A.; Rasool, K.; Miran, W.; Nawaz, M.; Jang, J.; Mahmoud, K.A.; Lee, D.S. Mercuric ion capturing by recoverable titanium carbide magnetic nanocomposite. J. Hazard. Mater. 2018, 344, 811–818. [Google Scholar] [CrossRef] [PubMed]
- Shahzad, A.; Rasool, K.; Miran, W.; Nawaz, M.; Jang, J.; Mahmoud, K.A.; Lee, D.S. Two-dimensional Ti3C2Tx MXene nanosheets for efficient copper removal from water. ACS Sustain. Chem. Eng. 2017, 5, 11481–11488. [Google Scholar] [CrossRef]
- Elumalai, S.; Yoshimura, M.; Ogawa, M. Simultaneous delamination and rutile formation on the surface of Ti3C2Tx MXene for copper adsorption. Chem.-Asian J. 2020, 15, 1044–1051. [Google Scholar] [CrossRef] [PubMed]
- Gan, D.; Huang, Q.; Dou, J.; Huang, H.; Chen, J.; Liu, M.; Wen, Y.; Yang, Z.; Zhang, X.; Wei, Y. Bioinspired functionalization of MXenes (Ti3C2Tx) with amino acids for efficient removal of heavy metal ions. Appl. Surf. Sci. 2020, 504, 144603. [Google Scholar] [CrossRef]
- Shahzad, A.; Jang, J.; Lim, S.-R.; Lee, D.S. Unique selectivity and rapid uptake of molybdenum-disulfide-functionalized MXene nanocomposite for mercury adsorption. Environ. Res. 2020, 182, 109005. [Google Scholar] [CrossRef] [PubMed]
- Fu, K.; Liu, X.; Yu, D.; Luo, J.; Wang, Z.; Crittenden, J.C. Highly efficient and selective Hg (II) removal from water using multilayered Ti3C2Ox MXene via adsorption coupled with catalytic reduction mechanism. Environ. Sci. Technol. 2020, 54, 16212–16220. [Google Scholar] [CrossRef] [PubMed]
- Wu, Y.; Chen, H.; Xiao, J.; Liu, D.; Liu, Z.; Qian, Y.; Xi, H. Adsorptive Separation of Methanol–Acetone on Isostructural Series of Metal–Organic Frameworks M-BTC (M = Ti, Fe, Cu, Co, Ru, Mo): A Computational Study of Adsorption Mechanisms and Metal-Substitution Impacts. ACS Appl. Mater. Interfaces 2015, 7, 26930–26940. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.; Liu, Y.; Lü, Q.-F.; Zhuang, H. Facile preparation of biosurfactant-functionalized Ti2CTX MXene nanosheets with an enhanced adsorption performance for Pb (II) ions. J. Mol. Liq. 2020, 297, 111810. [Google Scholar] [CrossRef]
- Luo, S.; Wang, R.; Yin, J.; Jiao, T.; Chen, K.; Zou, G.; Zhang, L.; Zhou, J.; Zhang, L.; Peng, Q. Preparation and dye degradation performances of self-assembled MXene-Co3O4 nanocomposites synthesized via solvothermal approach. ACS Omega 2019, 4, 3946–3953. [Google Scholar] [CrossRef] [Green Version]
- Li, K.; Zou, G.; Jiao, T.; Xing, R.; Zhang, L.; Zhou, J.; Zhang, Q.; Peng, Q. Self-assembled MXene-based nanocomposites via layer-by-layer strategy for elevated adsorption capacities. Colloids Surf. A Physicochem. Eng. Asp. 2018, 553, 105–113. [Google Scholar] [CrossRef]
- Mashtalir, O.; Cook, K.M.; Mochalin, V.N.; Crowe, M.; Barsoum, M.W.; Gogotsi, Y. Dye adsorption and decomposition on two-dimensional titanium carbide in aqueous media. J. Mater. Chem. A 2014, 2, 14334–14338. [Google Scholar] [CrossRef]
- Gao, Y.; Wang, L.; Zhou, A.; Li, Z.; Chen, J.; Bala, H.; Hu, Q.; Cao, X. Hydrothermal synthesis of TiO2/Ti3C2 nanocomposites with enhanced photocatalytic activity. Mater. Lett. 2015, 150, 62–64. [Google Scholar] [CrossRef]
- Peng, C.; Wei, P.; Chen, X.; Zhang, Y.; Zhu, F.; Cao, Y.; Wang, H.; Yu, H.; Peng, F. A hydrothermal etching route to synthesis of 2D MXene (Ti3C2, Nb2C): Enhanced exfoliation and improved adsorption performance. Ceram. Int. 2018, 44, 18886–18893. [Google Scholar] [CrossRef]
- Yao, C.; Zhang, W.; Xu, L.; Cheng, M.; Su, Y.; Xue, J.; Liu, J.; Hou, S. A facile synthesis of porous MXene-based freestanding film and its spectacular electrosorption performance for organic dyes. Sep. Purif. Technol. 2021, 263, 118365. [Google Scholar] [CrossRef]
- Cai, C.; Wang, R.; Liu, S.; Yan, X.; Zhang, L.; Wang, M.; Tong, Q.; Jiao, T. Synthesis of self-assembled phytic acid-MXene nanocomposites via a facile hydrothermal approach with elevated dye adsorption capacities. Colloids Surf. A Physicochem. Eng. Asp. 2020, 589, 124468. [Google Scholar] [CrossRef]
- Kim, S.; Gholamirad, F.; Yu, M.; Park, C.M.; Jang, A.; Jang, M.; Taheri-Qazvini, N.; Yoon, Y. Enhanced adsorption performance for selected pharmaceutical compounds by sonicated Ti3C2TX MXene. Chem. Eng. J. 2021, 406, 126789. [Google Scholar] [CrossRef]
- Zhang, P.; Xiang, M.; Liu, H.; Yang, C.; Deng, S. Novel two-dimensional magnetic titanium carbide for methylene blue removal over a wide pH range: Insight into removal performance and mechanism. ACS Appl. Mater. Interfaces 2019, 11, 24027–24036. [Google Scholar] [CrossRef]
- da Silva, M.P.; de Souza, A.C.A.; de Lima Ferreira, L.E.; Neto, L.M.P.; Nascimento, B.F.; de Araújo, C.M.B.; Fraga, T.J.M.; da Motta Sobrinho, M.A.; Ghislandi, M.G. Photodegradation of Reactive Black 5 and raw textile wastewater by heterogeneous photo-Fenton reaction using amino-Fe3O4-functionalized graphene oxide as nanocatalyst. Environ. Adv. 2021, 4, 100064. [Google Scholar] [CrossRef]
- Li, Q.; Hu, B.; Yang, Q.; Cai, X.; Nie, M.; Jin, Y.; Zhou, L.; Xu, Y.; Pan, Q.; Fang, L. Interaction mechanism between multi-layered MoS2 and H2O2 for self-generation of reactive oxygen species. Environ. Res. 2020, 191, 110227. [Google Scholar] [CrossRef]
- Tan, C.; Cheng, X.; Xu, T.; Chen, K.; Xiang, H.; Su, L. Crystalline boron significantly accelerates Fe (III)/PMS reaction as an electron donor: Iron recycling, reactive species generation, and acute toxicity evaluation. Chem. Eng. J. 2023, 452, 139154. [Google Scholar] [CrossRef]
- Nayebi, B.; Ayati, B. Degradation of emerging amoxicillin compound from water using the electro-Fenton process with an aluminum anode. Water Conserv. Sci. Eng. 2021, 6, 45–54. [Google Scholar] [CrossRef]
- Cui, Y.; Zhang, D.; Shen, K.; Nie, S.; Liu, M.; Huang, H.; Deng, F.; Zhou, N.; Zhang, X.; Wei, Y. Biomimetic anchoring of Fe3O4 onto Ti3C2 MXene for highly efficient removal of organic dyes by Fenton reaction. J. Environ. Chem. Eng. 2020, 8, 104369. [Google Scholar] [CrossRef]
- Wan, Z.; Wang, J. Ce-Fe-reduced graphene oxide nanocomposite as an efficient catalyst for sulfamethazine degradation in aqueous solution. Environ. Sci. Pollut. Res. 2016, 23, 18542–18551. [Google Scholar] [CrossRef] [PubMed]
- Cui, Y.; Liu, M.; Huang, H.; Zhang, D.; Chen, J.; Mao, L.; Zhou, N.; Deng, F.; Zhang, X.; Wei, Y. A novel one-step strategy for preparation of Fe3O4-loaded Ti3C2 MXenes with high efficiency for removal organic dyes. Ceram. Int. 2020, 46, 11593–11601. [Google Scholar] [CrossRef]
- He, L.; Huang, D.; He, Z.; Yang, X.; Yue, G.; Zhu, J.; Astruc, D.; Zhao, P. Nanoscale zero-valent iron intercalated 2D titanium carbides for removal of Cr (VI) in aqueous solution and the mechanistic aspect. J. Hazard. Mater. 2020, 388, 121761. [Google Scholar] [CrossRef] [PubMed]
- Shi, W.; Fu, Y.; Hao, C.; Guo, F.; Tang, Y. Heterogeneous photo-Fenton process over magnetically recoverable MnFe2O4/MXene hierarchical heterostructure for boosted degradation of tetracycline. Mater. Today Commun. 2022, 33, 104449. [Google Scholar] [CrossRef]
- Ma, Y.; Lv, X.; Xiong, D.; Zhao, X.; Zhang, Z. Catalytic degradation of ranitidine using novel magnetic Ti3C2-based MXene nanosheets modified with nanoscale zero-valent iron particles. Appl. Catal. B Environ. 2021, 284, 119720. [Google Scholar] [CrossRef]
- Wang, C.; Ye, J.; Liang, L.; Cui, X.; Kong, L.; Li, N.; Cheng, Z.; Peng, W.; Yan, B.; Chen, G. Application of MXene-based materials in Fenton-like systems for organic wastewater treatment: A review. Sci. Total Environ. 2022, 862, 160539. [Google Scholar] [CrossRef]
- Li, N.; Wang, Y.; Cheng, X.; Dai, H.; Yan, B.; Chen, G.; Wang, S. Influences and mechanisms of phosphate ions onto persulfate activation and organic degradation in water treatment: A review. Water Res. 2022, 222, 118896. [Google Scholar] [CrossRef]
- Devi, P.; Das, U.; Dalai, A.K. In-situ chemical oxidation: Principle and applications of peroxide and persulfate treatments in wastewater systems. Sci. Total Environ. 2016, 571, 643–657. [Google Scholar] [CrossRef] [PubMed]
- Peng, W.; Dong, Y.; Fu, Y.; Wang, L.; Li, Q.; Liu, Y.; Fan, Q.; Wang, Z. Non-radical reactions in persulfate-based homogeneous degradation processes: A review. Chem. Eng. J. 2021, 421, 127818. [Google Scholar] [CrossRef]
- Yan, X.; Yue, D.; Guo, C.; Wang, S.; Qian, X.; Zhao, Y. Effective removal of chlorinated organic pollutants by bimetallic iron-nickel sulfide activation of peroxydisulfate. Chin. Chem. Lett. 2020, 31, 1535–1539. [Google Scholar] [CrossRef]
- Hu, L.; Xiao, R.; Wang, X.; Wang, X.; Wang, C.; Wen, J.; Gu, W.; Zhu, C. MXene-induced electronic optimization of metal-organic framework-derived CoFe LDH nanosheet arrays for efficient oxygen evolution. Appl. Catal. B Environ. 2021, 298, 120599. [Google Scholar] [CrossRef]
- Li, S.; Gu, M.; Huang, J.; Wang, Y.; Yu, G. Oligolayered Co@ MXene with a Co···SO3 cation-π bridge for ultra-rapid catalytic oxidation of a novel “forever chemical” OBS. Appl. Catal. B Environ. 2022, 311, 121364. [Google Scholar] [CrossRef]
- Song, H.; Zu, D.; Li, C.; Zhou, R.; Wang, Y.; Zhang, W.; Pan, S.; Cai, Y.; Li, Z.; Shen, Y. Ultrafast activation of peroxymonosulfate by reduction of trace Fe3+ with Ti3C2 MXene under neutral and alkaline conditions: Reducibility and confinement effect. Chem. Eng. J. 2021, 423, 130012. [Google Scholar] [CrossRef]
- Song, X.; Wang, H.; Jin, S.; Lv, M.; Zhang, Y.; Kong, X.; Xu, H.; Ma, T.; Luo, X.; Tan, H. Oligolayered Ti3C2Tx MXene towards high performance lithium/sodium storage. Nano Res. 2020, 13, 1659–1667. [Google Scholar] [CrossRef]
- Jin, L.; You, S.; Yao, Y.; Chen, H.; Wang, Y.; Liu, Y. An electroactive single-atom copper anchored MXene nanohybrid filter for ultrafast water decontamination. J. Mater. Chem. A 2021, 9, 25964–25973. [Google Scholar] [CrossRef]
- Ma, Y.; Xiong, D.; Lv, X.; Zhao, X.; Meng, C.; Xie, H.; Zhang, Z. Rapid and long-lasting acceleration of zero-valent iron nanoparticles@Ti3C2-based MXene/peroxymonosulfate oxidation with bi-active centers toward ranitidine removal. J. Mater. Chem. A 2021, 9, 19817–19833. [Google Scholar] [CrossRef]
- Cai, T.; Wang, L.; Liu, Y.; Zhang, S.; Dong, W.; Chen, H.; Yi, X.; Yuan, J.; Xia, X.; Liu, C. Ag3PO4/Ti3C2 MXene interface materials as a Schottky catalyst with enhanced photocatalytic activities and anti-photocorrosion performance. Appl. Catal. B Environ. 2018, 239, 545–554. [Google Scholar] [CrossRef]
- Fayyaz, A.; Saravanakumar, K.; Talukdar, K.; Kim, Y.; Yoon, Y.; Park, C.M. Catalytic oxidation of naproxen in cobalt spinel ferrite decorated Ti3C2Tx MXene activated persulfate system: Mechanisms and pathways. Chem. Eng. J. 2021, 407, 127842. [Google Scholar] [CrossRef]
- Ding, M.; Chen, W.; Xu, H.; Lu, C.; Lin, T.; Shen, Z.; Tao, H.; Zhang, K. Synergistic Features of Superoxide Molecule Anchoring and Charge Transfer on Two-Dimensional Ti3C2Tx MXene for Efficient Peroxymonosulfate Activation. ACS Appl. Mater. Interfaces 2020, 12, 9209–9218. [Google Scholar] [CrossRef]
- Yang, P.; Li, S.; Xiaofu, L.; Xiaojing, A.; Liu, D.; Huang, W. Singlet oxygen-dominated activation of peroxymonosulfate by CuO/MXene nanocomposites for efficient decontamination of carbamazepine under high salinity conditions: Performance and singlet oxygen evolution mechanism. Sep. Purif. Technol. 2022, 285, 120288. [Google Scholar] [CrossRef]
- Song, H.; Pan, S.; Wang, Y.; Cai, Y.; Zhang, W.; Shen, Y.; Li, C. MXene-mediated electron transfer in Cu (II)/PMS process: From Cu (III) to Cu (I). Sep. Purif. Technol. 2022, 12, 121428. [Google Scholar] [CrossRef]
- Zeng, H.; Deng, L.; Yang, L.; Wu, H.; Zhang, H.; Zhou, C.; Liu, B.; Shi, Z. Novel Prussian blue analogues@ MXene nanocomposite as heterogeneous activator of peroxymonosulfate for the degradation of coumarin: The nonnegligible role of Lewis-acid sites on MXene. Chem. Eng. J. 2021, 416, 128071. [Google Scholar] [CrossRef]
- Zheng, H.; Bao, J.; Huang, Y.; Xiang, L.; Ren, B.; Du, J.; Nadagouda, M.N.; Dionysiou, D.D. Efficient degradation of atrazine with porous sulfurized Fe2O3 as catalyst for peroxymonosulfate activation. Appl. Catal. B Environ. 2019, 259, 118056. [Google Scholar] [CrossRef]
- Chen, G.; Nengzi, L.-C.; Gao, Y.; Zhu, G.; Gou, J.; Cheng, X. Degradation of tartrazine by peroxymonosulfate through magnetic Fe2O3/Mn2O3 composites activation. Chin. Chem. Lett. 2020, 31, 2730–2736. [Google Scholar] [CrossRef]
- Yun, E.-T.; Yoo, H.-Y.; Bae, H.; Kim, H.-I.; Lee, J. Exploring the role of persulfate in the activation process: Radical precursor versus electron acceptor. Environ. Sci. Technol. 2017, 51, 10090–10099. [Google Scholar] [CrossRef]
- Zhan, X.; Si, C.; Zhou, J.; Sun, Z. MXene and MXene-based composites: Synthesis, properties and environment-related applications. Nanoscale Horiz. 2020, 5, 235–258. [Google Scholar] [CrossRef]
- Hojjati-Najafabadi, A.; Mansoorianfar, M.; Liang, T.; Shahin, K.; Wen, Y.; Bahrami, A.; Karaman, C.; Zare, N.; Karimi-Maleh, H.; Vasseghian, Y. Magnetic-MXene-based nanocomposites for water and wastewater treatment: A review. J. Water Process Eng. 2022, 47, 102696. [Google Scholar] [CrossRef]
- Sharma, S.K.; Kumar, A.; Sharma, G.; Vo, D.-V.N.; García-Peñas, A.; Moradi, O.; Sillanpää, M. MXenes based nano-heterojunctions and composites for advanced photocatalytic environmental detoxification and energy conversion: A review. Chemosphere 2021, 291, 132923. [Google Scholar] [CrossRef] [PubMed]
- Javaid, A.; Latif, S.; Imran, M.; Hussain, N.; Bilal, M.; Iqbal, H.M. MXene-based hybrid composites as photocatalyst for the mitigation of pharmaceuticals. Chemosphere 2021, 291, 133062. [Google Scholar] [CrossRef]
- Sherryna, A.; Tahir, M. Role of Ti3C2 MXene as prominent schottky barriers in driving hydrogen production through photoinduced water splitting: A comprehensive review. ACS Appl. Energy Mater. 2021, 4, 11982–12006. [Google Scholar] [CrossRef]
- Sinopoli, A.; Othman, Z.; Rasool, K.; Mahmoud, K.A. Electrocatalytic/photocatalytic properties and aqueous media applications of 2D transition metal carbides (MXenes). Curr. Opin. Solid State Mater. Sci. 2019, 23, 100760. [Google Scholar] [CrossRef]
- Huang, H.; Song, Y.; Li, N.; Chen, D.; Xu, Q.; Li, H.; He, J.; Lu, J. One-step in-situ preparation of N-doped TiO2@ C derived from Ti3C2 MXene for enhanced visible-light driven photodegradation. Appl. Catal. B Environ. 2019, 251, 154–161. [Google Scholar] [CrossRef]
- Wang, Y.; Yu, J.; Xiao, W.; Li, Q. Microwave-assisted hydrothermal synthesis of graphene based Au–TiO2 photocatalysts for efficient visible-light hydrogen production. J. Mater. Chem. A 2014, 2, 3847–3855. [Google Scholar] [CrossRef]
- Zhang, C.; Beidaghi, M.; Naguib, M.; Lukatskaya, M.R.; Zhao, M.-Q.; Dyatkin, B.; Cook, K.M.; Kim, S.J.; Eng, B.; Xiao, X. Synthesis and charge storage properties of hierarchical niobium pentoxide/carbon/niobium carbide (MXene) hybrid materials. Chem. Mater. 2016, 28, 3937–3943. [Google Scholar] [CrossRef]
- Wang, H.; Wu, Y.; Xiao, T.; Yuan, X.; Zeng, G.; Tu, W.; Wu, S.; Lee, H.Y.; Tan, Y.Z.; Chew, J.W. Formation of quasi-core-shell In2S3/anatase TiO2@ metallic Ti3C2Tx hybrids with favorable charge transfer channels for excellent visible-light-photocatalytic performance. Appl. Catal. B Environ. 2018, 233, 213–225. [Google Scholar] [CrossRef]
- Li, J.; Wang, S.; Du, Y.; Liao, W. Enhanced photocatalytic performance of TiO2@ C nanosheets derived from two-dimensional Ti2CTx. Ceram. Int. 2018, 44, 7042–7046. [Google Scholar] [CrossRef]
- Liu, M.; Qiu, X.; Miyauchi, M.; Hashimoto, K. Cu (II) oxide amorphous nanoclusters grafted Ti3+ self-doped TiO2: An efficient visible light photocatalyst. Chem. Mater. 2011, 23, 5282–5286. [Google Scholar] [CrossRef]
- Mao, C.; Zuo, F.; Hou, Y.; Bu, X.; Feng, P. In situ preparation of a Ti3+ self-doped TiO2 film with enhanced activity as photoanode by N2H4 reduction. Angew. Chem. 2014, 126, 10653–10657. [Google Scholar] [CrossRef]
- Zhou, Y.; Liu, Y.; Liu, P.; Zhang, W.; Xing, M.; Zhang, J. A facile approach to further improve the substitution of nitrogen into reduced TiO2−x with an enhanced photocatalytic activity. Appl. Catal. B Environ. 2015, 170, 66–73. [Google Scholar] [CrossRef]
- Song, S.M.; Park, J.K.; Sul, O.J.; Cho, B.J. Determination of work function of graphene under a metal electrode and its role in contact resistance. Nano Lett. 2012, 12, 3887–3892. [Google Scholar] [CrossRef]
- Zhao, L.; Dong, B.; Li, S.; Zhou, L.; Lai, L.; Wang, Z.; Zhao, S.; Han, M.; Gao, K.; Lu, M. Interdiffusion reaction-assisted hybridization of two-dimensional metal–organic frameworks and Ti3C2Tx nanosheets for electrocatalytic oxygen evolution. ACS Nano 2017, 11, 5800–5807. [Google Scholar] [CrossRef] [PubMed]
- Romero, E.; Novoderezhkin, V.I.; van Grondelle, R. Quantum design of photosynthesis for bio-inspired solar-energy conversion. Nature 2017, 543, 355–365. [Google Scholar] [CrossRef] [PubMed]
- Long, M.; Brame, J.; Qin, F.; Bao, J.; Li, Q.; Alvarez, P.J. Phosphate changes effect of humic acids on TiO2 photocatalysis: From inhibition to mitigation of electron–hole recombination. Environ. Sci. Technol. 2017, 51, 514–521. [Google Scholar] [CrossRef]
- Iqbal, M.A.; Tariq, A.; Zaheer, A.; Gul, S.; Ali, S.I.; Iqbal, M.Z.; Akinwande, D.; Rizwan, S. Ti3C2-MXene/bismuth ferrite nanohybrids for efficient degradation of organic dyes and colorless pollutants. ACS Omega 2019, 4, 20530–20539. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fang, Y.; Cao, Y.; Chen, Q. Synthesis of an Ag2WO4/Ti3C2 Schottky composite by electrostatic traction and its photocatalytic activity. Ceram. Int. 2019, 45, 22298–22307. [Google Scholar] [CrossRef]
- Zhang, G.; Wu, S.; Li, Y.; Zhang, Q. Significant improvement in activity, durability, and light-to-fuel efficiency of Ni nanoparticles by La2O3 cluster modification for photothermocatalytic CO2 reduction. Appl. Catal. B Environ. 2020, 264, 118544. [Google Scholar] [CrossRef]
- Ghobadifard, M.; Mohebbi, S. Novel nanomagnetic Ag/β-Ag2WO4/CoFe2O4 as a highly efficient photocatalyst under visible light irradiation. New J. Chem. 2018, 42, 9530–9542. [Google Scholar] [CrossRef]
- Zou, Y.; Shi, J.-W.; Sun, L.; Ma, D.; Mao, S.; Lv, Y.; Cheng, Y. Energy-band-controlled ZnxCd1−xIn2S4 solid solution coupled with g-C3N4 nanosheets as 2D/2D heterostructure toward efficient photocatalytic H2 evolution. Chem. Eng. J. 2019, 378, 122192. [Google Scholar] [CrossRef]
- Yuan, X.; Jiang, L.; Liang, J.; Pan, Y.; Zhang, J.; Wang, H.; Leng, L.; Wu, Z.; Guan, R.; Zeng, G. In-situ synthesis of 3D microsphere-like In2S3/InVO4 heterojunction with efficient photocatalytic activity for tetracycline degradation under visible light irradiation. Chem. Eng. J. 2019, 356, 371–381. [Google Scholar] [CrossRef]
- Mi, Y.; Wen, L.; Wang, Z.; Cao, D.; Fang, Y.; Lei, Y. Building of anti-restack 3D BiOCl hierarchitecture by ultrathin nanosheets towards enhanced photocatalytic activity. Appl. Catal. B Environ. 2015, 176, 331–337. [Google Scholar] [CrossRef]
- Shi, X.; Wang, P.; Lan, L.; Jia, S.; Wei, Z. Construction of BiOBrxI1−x/MXene Ti3C2Tx composite for improved photocatalytic degradability. J. Mater. Sci. Mater. Electron. 2019, 30, 19804–19812. [Google Scholar] [CrossRef]
- Tariq, A.; Ali, S.I.; Akinwande, D.; Rizwan, S. Efficient visible-light photocatalysis of 2D-MXene nanohybrids with Gd3+-and Sn4+-codoped bismuth ferrite. ACS Omega 2018, 3, 13828–13836. [Google Scholar] [CrossRef] [Green Version]
- Ran, J.; Gao, G.; Li, F.-T.; Ma, T.-Y.; Du, A.; Qiao, S.-Z. Ti3C2 MXene co-catalyst on metal sulfide photo-absorbers for enhanced visible-light photocatalytic hydrogen production. Nat. Commun. 2017, 8, 13907. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Persson, I.; Näslund, L.-Å.; Halim, J.; Barsoum, M.W.; Darakchieva, V.; Palisaitis, J.; Rosen, J.; Persson, P.O.Å. On the organization and thermal behavior of functional groups on Ti3C2 MXene surfaces in vacuum. 2D Mater. 2017, 5, 015002. [Google Scholar] [CrossRef]
- Pandey, R.P.; Rasheed, P.A.; Gomez, T.; Azam, R.S.; Mahmoud, K.A. A fouling-resistant mixed-matrix nanofiltration membrane based on covalently cross-linked Ti3C2TX (MXene)/cellulose acetate. J. Membr. Sci. 2020, 607, 118139. [Google Scholar] [CrossRef]
- Alessandro, F.; Macedonio, F.; Drioli, E. New Materials and Phenomena in Membrane Distillation. Chemistry 2023, 5, 65–84. [Google Scholar] [CrossRef]
- Gugliuzza, A.; Macedonio, F.; Politano, A.; Drioli, E. Prospects of 2D materials-based membranes in water desalination. Chem. Eng. Trans 2019, 73, 265–270. [Google Scholar]
- Kang, K.M.; Kim, D.W.; Ren, C.E.; Cho, K.M.; Kim, S.J.; Choi, J.H.; Nam, Y.T.; Gogotsi, Y.; Jung, H.-T. Selective molecular separation on Ti3C2Tx–graphene oxide membranes during pressure-driven Filtration: Comparison with graphene oxide and MXenes. ACS Appl. Mater. Interfaces 2017, 9, 44687–44694. [Google Scholar] [CrossRef]
- Ding, L.; Wei, Y.; Wang, Y.; Chen, H.; Caro, J.; Wang, H. A two-dimensional lamellar membrane: MXene nanosheet stacks. Angew. Chem. Int. Ed. 2017, 56, 1825–1829. [Google Scholar] [CrossRef]
- Pandey, R.P.; Rasool, K.; Madhavan, V.E.; Aïssa, B.; Gogotsi, Y.; Mahmoud, K.A. Ultrahigh-flux and fouling-resistant membranes based on layered silver/MXene (Ti3C2Tx) nanosheets. J. Mater. Chem. A 2018, 6, 3522–3533. [Google Scholar] [CrossRef]
- Ding, L.; Li, L.; Liu, Y.; Wu, Y.; Lu, Z.; Deng, J.; Wei, Y.; Caro, J.; Wang, H. Effective ion sieving with Ti3CsTx MXene membranes for production of drinking water from seawater. Nat. Sustain. 2020, 3, 296–302. [Google Scholar] [CrossRef]
- Lu, Z.; Wei, Y.; Deng, J.; Ding, L.; Li, Z.-K.; Wang, H. Self-crosslinked MXene (Ti3C2Tx) membranes with good antiswelling property for monovalent metal ion exclusion. ACS Nano 2019, 13, 10535–10544. [Google Scholar] [CrossRef]
- Sun, Y.; Li, S.; Zhuang, Y.; Liu, G.; Xing, W.; Jing, W. Adjustable interlayer spacing of ultrathin MXene-derived membranes for ion rejection. J. Membr. Sci. 2019, 591, 117350. [Google Scholar] [CrossRef]
- Wang, J.; Zhang, Z.; Zhu, J.; Tian, M.; Zheng, S.; Wang, F.; Wang, X.; Wang, L. Ion sieving by a two-dimensional Ti3C2Tx alginate lamellar membrane with stable interlayer spacing. Nat. Commun. 2020, 11, 3540. [Google Scholar] [CrossRef]
- Sun, Y.; Xu, D.; Li, S.; Cui, L.; Zhuang, Y.; Xing, W.; Jing, W. Assembly of multidimensional MXene-carbon nanotube ultrathin membranes with an enhanced anti-swelling property for water purification. J. Membr. Sci. 2021, 623, 119075. [Google Scholar] [CrossRef]
- Yaroshchuk, A.; Bruening, M.L.; Bernal, E.E.L. Solution-Diffusion–Electro-Migration model and its uses for analysis of nanofiltration, pressure-retarded osmosis and forward osmosis in multi-ionic solutions. J. Membr. Sci. 2013, 447, 463–476. [Google Scholar] [CrossRef]
- Li, K.; Tao, Y.; Li, Z.; Sha, J.; Chen, Y. Selective ion-permeation through strained and charged graphene membranes. Nanotechnology 2017, 29, 035402. [Google Scholar] [CrossRef]
- Wu, X.; Ding, M.; Xu, H.; Yang, W.; Zhang, K.; Tian, H.; Wang, H.; Xie, Z. Scalable Ti3C2Tx mxene interlayered forward osmosis membranes for enhanced water purification and organic solvent recovery. ACS Nano 2020, 14, 9125–9135. [Google Scholar] [CrossRef]
- Wang, X.; Li, Q.; Zhang, J.; Huang, H.; Wu, S.; Yang, Y. Novel thin-film reverse osmosis membrane with MXene Ti3C2Tx embedded in polyamide to enhance the water flux, anti-fouling and chlorine resistance for water desalination. J. Membr. Sci. 2020, 603, 118036. [Google Scholar] [CrossRef]
- Meng, B.; Liu, G.; Mao, Y.; Liang, F.; Liu, G.; Jin, W. Fabrication of surface-charged MXene membrane and its application for water desalination. J. Membr. Sci. 2021, 623, 119076. [Google Scholar] [CrossRef]
- Fuzil, N.S.; Othman, N.H.; Alias, N.H.; Marpani, F.; Othman, M.H.D.; Ismail, A.F.; Lau, W.J.; Li, K.; Kusworo, T.D.; Ichinose, I. A review on photothermal material and its usage in the development of photothermal membrane for sustainable clean water production. Desalination 2021, 517, 115259. [Google Scholar] [CrossRef]
- Liu, Y.; Zhao, J.; Zhang, S.; Li, D.; Zhang, X.; Zhao, Q.; Xing, B. Advances and challenges of broadband solar absorbers for efficient solar steam generation. Environ. Sci. Nano 2022, 9, 2264–2296. [Google Scholar] [CrossRef]
- Huang, L.; Pei, J.; Jiang, H.; Hu, X. Water desalination under one sun using graphene-based material modified PTFE membrane. Desalination 2018, 442, 1–7. [Google Scholar] [CrossRef]
- Li, Z.; Lei, H.; Kan, A.; Xie, H.; Yu, W. Photothermal applications based on graphene and its derivatives: A state-of-the-art review. Energy 2021, 216, 119262. [Google Scholar] [CrossRef]
- Li, W.; Chen, Y.; Yao, L.; Ren, X.; Li, Y.; Deng, L. Fe3O4/PVDF-HFP photothermal membrane with in-situ heating for sustainable, stable and efficient pilot-scale solar-driven membrane distillation. Desalination 2020, 478, 114288. [Google Scholar] [CrossRef]
- Wu, X.; Jiang, Q.; Ghim, D.; Singamaneni, S.; Jun, Y.-S. Localized heating with a photothermal polydopamine coating facilitates a novel membrane distillation process. J. Mater. Chem. A 2018, 6, 18799–18807. [Google Scholar] [CrossRef]
- Politano, A.; Di Profio, G.; Fontananova, E.; Sanna, V.; Cupolillo, A.; Curcio, E. Overcoming temperature polarization in membrane distillation by thermoplasmonic effects activated by Ag nanofillers in polymeric membranes. Desalination 2019, 451, 192–199. [Google Scholar] [CrossRef]
- Li, H.; He, Y.; Liu, Z.; Jiang, B.; Huang, Y. A flexible thin-film membrane with broadband Ag@ TiO2 nanoparticle for high-efficiency solar evaporation enhancement. Energy 2017, 139, 210–219. [Google Scholar] [CrossRef]
- Xu, D.; Li, Z.; Li, L.; Wang, J. Insights into the photothermal conversion of 2D MXene nanomaterials: Synthesis, mechanism, and applications. Adv. Funct. Mater. 2020, 30, 2000712. [Google Scholar] [CrossRef]
- Dongare, P.D.; Alabastri, A.; Pedersen, S.; Zodrow, K.R.; Hogan, N.J.; Neumann, O.; Wu, J.; Wang, T.; Deshmukh, A.; Elimelech, M. Nanophotonics-enabled solar membrane distillation for off-grid water purification. Proc. Natl. Acad. Sci. USA 2017, 114, 6936–6941. [Google Scholar] [CrossRef] [Green Version]
- Bamasag, A.; Almatrafi, E.; Alqahtani, T.; Phelan, P.; Ullah, M.; Mustakeem, M.; Obaid, M.; Ghaffour, N. Recent advances and future prospects in direct solar desalination systems using membrane distillation technology. J. Clean. Prod. 2022, 385, 135737. [Google Scholar] [CrossRef]
- Cao, S.; Wu, X.; Zhu, Y.; Gupta, P.; Martinez, A.; Zhang, Y.; Ghim, D.; Wang, Y.; Liu, L.; Jun, Y.-S. MXene aerogel for efficient photothermally driven membrane distillation with dual-mode antimicrobial capability. J. Mater. Chem. A 2021, 9, 22585–22596. [Google Scholar] [CrossRef]
- Tan, Y.Z.; Wang, H.; Han, L.; Tanis-Kanbur, M.B.; Pranav, M.V.; Chew, J.W. Photothermal-enhanced and fouling-resistant membrane for solar-assisted membrane distillation. J. Membr. Sci. 2018, 565, 254–265. [Google Scholar] [CrossRef]
- Chen, L.; Shi, X.; Yu, N.; Zhang, X.; Du, X.; Lin, J. Measurement and analysis of thermal conductivity of Ti3C2Tx MXene films. Materials 2018, 11, 1701. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gholivand, H.; Fuladi, S.; Hemmat, Z.; Salehi-Khojin, A.; Khalili-Araghi, F. Effect of surface termination on the lattice thermal conductivity of monolayer Ti3C2Tz MXenes. J. Appl. Phys. 2019, 126, 065101. [Google Scholar] [CrossRef] [Green Version]
- Jiang, X.; Kuklin, A.V.; Baev, A.; Ge, Y.; Ågren, H.; Zhang, H.; Prasad, P.N. Two-dimensional MXenes: From morphological to optical, electric, and magnetic properties and applications. Phys. Rep. 2020, 848, 1–58. [Google Scholar] [CrossRef]
- Khan, R.; Andreescu, S. Mxenes-based bioanalytical sensors: Design, characterization, and applications. Sensors 2020, 20, 5434. [Google Scholar] [CrossRef]
- Dong, L.M.; Ye, C.; Zheng, L.L.; Gao, Z.F.; Xia, F. Two-dimensional metal carbides and nitrides (MXenes): Preparation, property, and applications in cancer therapy. Nanophotonics 2020, 9, 2125–2145. [Google Scholar] [CrossRef] [Green Version]
- Peng, Y.-Y.; Akuzum, B.; Kurra, N.; Zhao, M.-Q.; Alhabeb, M.; Anasori, B.; Kumbur, E.C.; Alshareef, H.N.; Ger, M.-D.; Gogotsi, Y. All-MXene (2D titanium carbide) solid-state microsupercapacitors for on-chip energy storage. Energy Environ. Sci. 2016, 9, 2847–2854. [Google Scholar] [CrossRef] [Green Version]
- Zhang, J.; Seyedin, S.; Gu, Z.; Yang, W.; Wang, X.; Razal, J.M. MXene: A potential candidate for yarn supercapacitors. Nanoscale 2017, 9, 18604–18608. [Google Scholar] [CrossRef]
- Zhang, J.; Wan, L.; Gao, Y.; Fang, X.; Lu, T.; Pan, L.; Xuan, F. Highly stretchable and self-healable MXene/polyvinyl alcohol hydrogel electrode for wearable capacitive electronic skin. Adv. Electron. Mater. 2019, 5, 1900285. [Google Scholar] [CrossRef]
- Liu, R.; Li, W. High-thermal-stability and high-thermal-conductivity Ti3C2Tx MXene/poly (vinyl alcohol)(PVA) composites. ACS Omega 2018, 3, 2609–2617. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kim, H.; Alshareef, H.N. MXetronics: MXene-enabled electronic and photonic devices. ACS Mater. Lett. 2019, 2, 55–70. [Google Scholar] [CrossRef] [Green Version]
- Li, J.; Yan, D.; Hou, S.; Li, Y.; Lu, T.; Yao, Y.; Pan, L. Improved sodium-ion storage performance of Ti3C2Tx MXenes by sulfur doping. J. Mater. Chem. A 2018, 6, 1234–1243. [Google Scholar] [CrossRef]
- Liu, B.; Pouramini, S. Multi-objective optimization for thermal comfort enhancement and greenhouse gas emission reduction in residential buildings applying retrofitting measures by an Enhanced Water Strider Optimization Algorithm: A case study. Energy Rep. 2021, 7, 1915–1929. [Google Scholar] [CrossRef]
- Nasrallah, G.K.; Al-Asmakh, M.; Rasool, K.; Mahmoud, K.A. Ecotoxicological assessment of Ti3C2Tx (MXene) using a zebrafish embryo model. Environ. Sci. Nano 2018, 5, 1002–1011. [Google Scholar] [CrossRef]
- Jastrzębska, A.; Szuplewska, A.; Wojciechowski, T.; Chudy, M.; Ziemkowska, W.; Chlubny, L.; Rozmysłowska, A.; Olszyna, A. In vitro studies on cytotoxicity of delaminated Ti3C2 MXene. J. Hazard. Mater. 2017, 339, 1–8. [Google Scholar] [CrossRef]
- Alhussain, H.; Augustine, R.; Hussein, E.A.; Gupta, I.; Hasan, A.; Al Moustafa, A.-E.; Elzatahry, A. MXene nanosheets may induce toxic effect on the early stage of embryogenesis. J. Biomed. Nanotechnol. 2020, 16, 364–372. [Google Scholar] [CrossRef]




| Kind of Precursors | Kinds of Etchants | Duration | MXene | Ref. |
|---|---|---|---|---|
| Ti3AlC2 | 1.3 g of LiF + 6 M HCl | 24 h | Ti3C2Tx | [78] |
| Cr2AlC | 0.2 g LiF + 2 M solution | 30 min | Cr2CTx | [79] |
| Ti3AlC2 | NH3F | 24 h | Ti2C2Tx | [80] |
| Ti3AlC | 48% HF | 30 h | Ti3C2Tx | [81] |
| Ti3AlC2 | 1g MAX + 10 Ml hf | 24 h | Ti3C2Tx | [82] |
| 2D MXene | Contaminants | Adsorption Duration | Adsorption Capacity | Ref. |
|---|---|---|---|---|
| Ti3C2Tx-KH570 | Pb | 2 h | 147.290 (mg/g) | [111] |
| Ti3C2 | Cr(VI) | 14 h | 80 (mg/g) | [112] |
| Ti3C2Tx MXene Nanosheets | Cu | 1 min | 78.4 (mg/g) | [113] |
| Magnetic Ti3C2TX Nanocomposite | Hg | 24 h | 1128.41 (mg/g) | [114] |
| Ti2C2Tx | Pb | 2 h | 36.6 (mg/g) | [115] |
| 2D MXene | Contaminants | Adsorption Duration | Adsorption Capacity | Ref. |
|---|---|---|---|---|
| KOH-Ti3C2Tx | MB | -h | 77 (mg/g) | [136] |
| Ti3C2Tx | AB80 | 20 h | - | [130] |
| Self-assembled MXene (Ti3C2)-Co3O4 nanocomposite | Rhodamine B | 700 min | 47.68 (mg/g) | [128] |
| Ti2C2Tx | MB | 0–24 h | 140 (mg/g) | [115] |
| Sulfonic groups functionalized MXenes (Ti3C2-SO3H) | MB | 70 min | 111.1 (mg/g) | [137] |
| MXene (Ti3C2(OHxF1−x)2) | ST | 180 min | 2.861 | [129] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pouramini, Z.; Mousavi, S.M.; Babapoor, A.; Hashemi, S.A.; Pynadathu Rumjit, N.; Garg, S.; Ahmed, S.; Chiang, W.-H. Recent Advances in MXene-Based Nanocomposites for Wastewater Purification and Water Treatment: A Review. Water 2023, 15, 1267. https://doi.org/10.3390/w15071267
Pouramini Z, Mousavi SM, Babapoor A, Hashemi SA, Pynadathu Rumjit N, Garg S, Ahmed S, Chiang W-H. Recent Advances in MXene-Based Nanocomposites for Wastewater Purification and Water Treatment: A Review. Water. 2023; 15(7):1267. https://doi.org/10.3390/w15071267
Chicago/Turabian StylePouramini, Zahra, Seyyed Mojtaba Mousavi, Aziz Babapoor, Seyyed Alireza Hashemi, Nelson Pynadathu Rumjit, Shivani Garg, Shakeel Ahmed, and Wei-Hung Chiang. 2023. "Recent Advances in MXene-Based Nanocomposites for Wastewater Purification and Water Treatment: A Review" Water 15, no. 7: 1267. https://doi.org/10.3390/w15071267





