Is It Feasible to Preserve a Self-Sustaining Population of Yangtze Finless Porpoise in the Highest Density Section of Yangtze River?
Abstract
:1. Introduction
2. Materials and Methods
3. Results
3.1. Baseline Model
3.2. Sensitivity Analysis
3.3. Scenarios
4. Discussion
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Gao, A.; Zhou, K. Geographical variation of external measurements and three subspecies of Neophocaena phocaenoides in Chinese waters. Acta Theriol. Sin. 1995, 15, 81–92. [Google Scholar]
- Zhou, X.M.; Guang, X.; Sun, D. Population genomics of finless porpoises reveal an incipient cetacean species adapted to freshwater. Nat. Commun. 2018, 9, 1276. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Turvey, S.T.; Pitman, R.; Taylor, B.; Barlow, J.; Akamatsu, T.; Barrett, L.A.; Zhao, X.; Reeves, R.R.; Stewart, B.S.; Wang, K.; et al. First human-caused extinction of a cetacean species? Biol. Lett. 2007, 3, 537–540. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wu, B.; Wang, W.; Wang, H.; Gang, H. A retrospective Analysis on the Population Viability of the Yangtze River Dolphin or Baiji (Lipotes vexillifer). Indian J. Anim. Res. 2020, 56, 775–779. [Google Scholar] [CrossRef]
- Yang, H.L.; Shen, L.; He, Y.F.; Tian, H.W.; Gao, L.; Wu, J.M.; Mei, Z.G.; Wei, N.; Wang, L.; Zhu, T.B.; et al. Status of aquatic organism resources and their environments in Yangtze River system (2017–2021). J. Fish. China 2022, Online. [Google Scholar]
- Liu, S.; Xu, C.X. The Yangtze finless porpoise has been upgraded to first-class state protection. Sci. Technol. Inf. Aquat. Prod. 2021, 48, 117–118. (In Chinese) [Google Scholar]
- Xiao, W.; Zhang, X. Distribution and population size of Yangtze finless porpoise in Poyang Lake and its branches. Acta Theriol. Sin. 2002, 22, 7–14. [Google Scholar]
- Wei, Z.; Wang, D.; Zhang, X.; Kexiong, W.; Xinan, K. Population size, behavior, movement pattern and protection of Yangtze finless porpoise at Balijiang section of the Yangtze River. Resour. Environ. Yangtze Basin. 2002, 11, 427–432. [Google Scholar]
- Dong, S. Studies on the Distribution and Movement of the Yangtze Finless Porpoises (Neophocaena Phocaenoides Asiaeorientalis) in Hukou Area Obtained by Acoustic Method. Master’s Thesis, Institute of Hydrobiology, Chinese Academy of Sciences, Wuhan, China, 2009. [Google Scholar]
- Zhang, X. Population Ecology of Yangtze Finless Porpoise in Dongting Lake and the Adjacent Waters. Ph.D. Dissertation, Institute of Hydrobiology, Chinese Academy of Sciences, Wuhan, China, 2011. [Google Scholar]
- Kimura, S.; Akamatsu, T.; Li, S.; Dong, L.; Wang, K.; Wang, D.; Arai, N. Seasonal changes in the local distribution of Yangtze finless porpoise related to fish presence. Mar. Mammal Sci. 2012, 28, 308–324. [Google Scholar] [CrossRef]
- Wang, Z. Study on the Impacts of Sand Mining Activities in Dongting Lake on Yangtze Finless Porpoise. Master’s Thesis, Institute of Hydrobiology, Chinese Academy of Sciences, Wuhan, China, 2015. [Google Scholar]
- Chen, M.; Fontaine, M.C.; Chehida, Y.; Zheng, J.; Labbé, F.; Mei, Z.; Hao, Y.; Wang, K.; Wu, M.; Zhao, Q.; et al. Genetic footprint of population fragmentation and contemporary collapse in a freshwater cetacean. Sci. Rep. 2017, 7, 14449. [Google Scholar] [CrossRef] [Green Version]
- Huang, J.; Mei, Z.; Chen, M.; Han, Y.; Zhang, X.; Moore, J.E.; Zhao, X.; Hao, Y.; Wang, K.; Wang, D. Population survey showing hope for population recovery of the critically endangered Yangtze finless. Biol. Conserv. 2020, 241, 108315. [Google Scholar] [CrossRef]
- Wang, Z.X.; Chen, M.M.; Wang, K.W.; Lian, Y.; Yu, D. Comparison of Yangtze finless porpoise (Neophocaena asiaeorientalis) clustering in different flow patterns. Acta Theriol. Sin. 2022, 42, 152–158. (In Chinese) [Google Scholar]
- Shaffer, M. Minimum viable populations: Coping with uncertainty. In Viable Populations for Conservation; Soulé, M.E., Ed.; Cambridge University Press: Cambridge, UK, 1987; pp. 69–86. [Google Scholar]
- Brook, B.W.; Burgman, M.; Frankham, R. Differences and congruencies between PVA packages: The importance of sex ratio for predictions of extinction risk. Conserv. Ecol. 2000, 4, 920. [Google Scholar] [CrossRef] [Green Version]
- Brook, B.W.; O’grady, J.; Chapman, A.; Burgman, M.A.; Akcakaya, H.R.; Frankham, R. Predictive accuracy of population viability analysis in conservation biology. Nature 2000, 404, 385–387. [Google Scholar] [CrossRef] [PubMed]
- Lacy, R.C.; Pollak, J.P. VORTEX: A Stochastic Simulation of the Extinction Process; Version 10.2.3; Chicago Zoological Society: Brookfield, IL, USA, 2017. [Google Scholar]
- Harris, R.B.; Metzgar, L.H.; Bevins, C.D. GAPPS: Generalized Animal Population Projection System; Version 3.0; Montana Cooperative Wildlife Research Unit, University of Montana: Missoula, MT, USA, 1986. [Google Scholar]
- Mills, L.S.; Smouse, P.E. Demographic consequences of inbreeding in remnant populations. Am. Nat. 1994, 144, 412–431. [Google Scholar] [CrossRef]
- Akc-akaya, H.R. RAMAS Metapop: Viability Analysis for Stage-Structured Metapopulations; Version 2; Applied Biomathematics: Setauket, NY, USA, 1997. [Google Scholar]
- Ferson, S. RAMAS Stage: Generalized Stage-Based Modelling for Population Dynamics; Applied Biomathematics: Setauket, NY, USA, 1994. [Google Scholar]
- Lacy, R.C. Structure of the VORTEX simulation model for PVA. Ecol. Bull. 2000, 48, 191–203. [Google Scholar]
- Huang, S.L.; Mei, Z.G.; Hao, Y.J.; Zheng, J.; Wang, K.; Wang, D. Saving the Yangtze finless porpoise: Time is rapidly running out. Biol. Conserv. 2017, 210, 40–46. [Google Scholar] [CrossRef]
- Wu, B.; Xu, J.G.; Wang, J.M.; Lie, C.; Weiping, W.; Song, Z. Population Viability Analysis of Yangtze Finless Porpoise in the Yangtze Main Steam Suggesting that a Total Ban on Productive Fishing could be Decisive. Indian J. Anim. Res. 2021, 55, 1515–1520. [Google Scholar]
- Chen, M.M.; Yu, D.; Lian, Y.; Liu, Z. Population abundance and habitat preference of the Yangtze finless porpoise in the highest density section of the Yangtze River. Aquat. Conserv. Mar. Freshw. Ecosyst. 2020, 30, 1088–1097. [Google Scholar] [CrossRef]
- Mei, Z.G.; Zhang, X.; Huang, S.; Zhao, X.; Hao, Y.; Zhang, L.; Qian, Z.; Zheng, J.; Wang, K.; Wang, D.; et al. The Yangtze finless porpoise: On an accelerating path to extinction? Biol. Conserv. 2014, 172, 117–123. [Google Scholar] [CrossRef]
- Zhao, X.; Barlow, J.; Taylor, B.; Pitman, R.L.; Wang, K.; Wei, Z.; Stewart, B.S.; Turvey, S.T.; Akamatsu, S.T.; Reeves, R.R.; et al. Abundance and conservation status of the Yangtze finless porpoise in the Yangtze River. China Biol. Conserv. 2008, 141, 3006–3018. [Google Scholar] [CrossRef] [Green Version]
- Zhao, X.; Wang, D.; Turvey, S.; Taylor, B.; Akamatsu, T. Distribution patterns of Yangtze finless porpoises in the Yangtze River: Implications for reserve management. Anim. Conserv. 2013, 16, 509–518. [Google Scholar] [CrossRef]
- Lacy, R.C. Lessons from 30 years of population viability analysis of wildlife populations. Zoo Biol. 2019, 38, 67–77. [Google Scholar] [CrossRef] [Green Version]
- Zhang, X.F.; Wang, K.X. Viability analysis of finless porpoise populations in the Yangtze River. Chin. Acta Ecol. Sin. 1999, 19, 529–533. [Google Scholar]
- Burgman, M.A.; Ferson, S.; Akçakaya, H. Risk Assessment in Conservation Biology; Chapman and Hall: New York, NY, USA, 1993. [Google Scholar]
- Shaffer, M.L. Minimum population sizes for species conservation. BioScience 1981, 31, 131–134. [Google Scholar] [CrossRef]
- Ginzburg, L.R.; Slobodkin, L.B.; Johnson, K. Quasi-extinction probabilities as a measure of impact on population growth. Risk Anal. 1982, 2, 171–181. [Google Scholar] [CrossRef]
- Ralls, K.; Ballou, J.D.; Templeton, A.R. Estimates of lethal equivalents and the cost of inbreeding in mammals. Conserv. Biol. 1988, 2, 185–189. [Google Scholar] [CrossRef]
- Hao, Y.J.; Wang, D.; Zhang, X. Review on breeding biology of Yangtze finless porpoise (Neophocaena phocaenoides asiaeorientalis). Acta Theriol. Sin. China 2006, 26, 191–200. [Google Scholar]
- Chen, M.M.; Zheng, Y.; Hao, Y.; Mei, Z.; Wang, K.; Zhao, Q.; Zheng, J.; Wang, D. Parentage-Based Group Composition and Dispersal Pattern Studies of the Yangtze Finless Porpoise Population in Poyang Lake. Int. J. Mol. Sci. 2016, 8, 1268. [Google Scholar] [CrossRef] [Green Version]
- Zhang, X.F. Study on age identification, growth and reproduction of finless porpoises. Chin. Acta Hydrobiol. Sin. 1992, 16, 289–297. [Google Scholar]
- Li, Y.T. Study on the Habitat Selection, Environmental Capacity, and Population Viability of the Yangtze Finless Porpoises in Tian-e-zhou Semi-Natural Ex Situ Reserve; Institute of Hydrobiology, the Chinese Academy of Sciences: Wuhan, China, 2017. [Google Scholar]
- Yang, G.; Zhou, K.; Gao, A.; Chang, Q. Study on life table and population dynamics of finless porpoises. Acta Zool. Sin. 1998, 18, 1–7. (In Chinese) [Google Scholar]
- Mei, Z.G.; Huang, S.L.; Hao, Y.J.; Turvey, S.T.; Gong, W.; Wang, D. Accelerating population decline of Yangtze finless porpoise (Neophocaena asiaeorientalis asiaeorientalis). Biol. Conserv. 2012, 153, 192–200. [Google Scholar] [CrossRef]
- Wu, B.; He, G.; Wang, W.P. Preliminary Estimation of Environmental Capacity of Yangtze Finless Porpoise in Poyang Lake through Yangtze River Waterway Based on Ecopath Model. Acta Agric. Jiangxi 2022, 42, 28–35. (In Chinese) [Google Scholar]
- Reed, D.H.; O’Grady, J.J.; Ballou, J.D.; Frankham, R. The frequency and severity of catastrophic die-offs in vertebrates. Anim. Conserv. 2003, 6, 109–114. [Google Scholar] [CrossRef]
- Wang, D.; Hao, Y.; Wang, K.; Zhao, Q.; Chen, D.; Wei, Z.; Zhang, X. Aquatic Resource Conservation. The first Yangtze finless porpoise successfully born in captivity. Environ. Sci. Pollut. Res. 2005, 12, 247–250. [Google Scholar] [CrossRef]
- Wang, R.Y.; Chen, M.M.; Wan, X.L.; Tang, B.; Hao, Y.J.; Mei, Z.G.; Fan, F.; Wang, K.X.; Wang, D.; Zheng, J.S. Microsatellite genetic diversity evaluation and development prediction of the Yangtze finless porpoise Population in the Poyang Lake. Acta Hydrobiol. Sin. 2022, 1–8. Available online: http://ssswxb.ihb.ac.cn/article/app/id/b73e1eaa-3f7a-4489-8578-dc94e8038f32/flg?pageType=en (accessed on 30 November 2022). (In Chinese). [CrossRef]
- Wang, W.P.; Xu, Z.W.; Gong, L.Q.; Zeng, N.J.; Xiong, H.B.; Lin, F.R.; Xu, X.; Huang, J.; Zhang, W.S.; Wu, B. Simultaneous monitoring and population dynamics analysis of Yangtze finless Porpoise in Poyang Lake. J. Hydroecol. 2023. Available online: https://wap.cnki.net/touch/web/Journal/Article/SCAN20230130003.html (accessed on 30 November 2022). (In Chinese).
- Palmer, E.I.; Betty, E.L.; Murphy, S.; Perrott, M.R.; Smith, A.N.; Stockin, K.A. Reproductive biology of female common dolphins (Delphinus delphis) in New Zealand waters. Mar. Biol. 2022, 169, 158. [Google Scholar] [CrossRef]
- He, C.H.; Du, J.; Zhu, D.; Zhang, L. Population viability analysis of small population: A case study for Asian elephant in China. Integr. Zool. 2020, 15, 350–362. [Google Scholar] [CrossRef]
- Rossi, A.; Scordamaglia, E.; Bellingeri, M.; Gnone, G.; Nuti, S.; Salvioli, F.; Manfredi, P.; Santangelo, G. Demography of the bottlenose dolphin Tursiops truncatus (Mammalia: Delphinidae) in the Eastern Ligurian Sea (NW Mediterranean): Quantification of female reproductive parameters. Eur. Zool. J. 2017, 84, 294–302. [Google Scholar] [CrossRef] [Green Version]
- Zhang, J.X.; Chen, M.; Han, Y.; Lei, P.Y.; Wang, K.X.; Mei, Z.G.; Wang, D. River bank habitat assessment of the Yangtze finless porpoise in the main stem of middle-lower Yangtze river. Chin. Acta Hydrobiol. Sin. 2019, 43, 56–61. [Google Scholar]
- He, S.X.; Zhang, D.; Wu, F.X.; Wang, X.Y.; Hu, W.J.; Liang, S.S.; Du, J.G.; Yu, W.W.; Chen, B. Identification of potential ecological corridors for Sousa Chinensisin Xiamen Bay and assessment of anthropogenic disturbance. Acta Ecol. Sin. China 2022, 42, 8555–8567. [Google Scholar]
- Rocha-Loredo, A.G.; Ramírez-Marcial, N.; González-Espinosa, M. Riqueza y diversidad de árboles del bosque tropical caducifolio en la Depresión Central de Chiapas. Bol. Soc. Bot. Mex 2010, 87, 89–103. [Google Scholar]
- Emília, P.M.; Arnaud, L.J.D. Population viability analysis: Using a modeling tool to assess the viability of tapir populations in fragmented landscapes. Integr. Zool. 2012, 7, 356–372. [Google Scholar]
- Cruz-Salazar, S.B.; Ruiz-Montoya, L.; Navarrete-Gutiérrez, D.; Vázquez, L.-B. Influence of the composition and structure of modified landscapes on abundance of two marsupials during the dry season. Therya 2016, 7, 393–406. [Google Scholar] [CrossRef] [Green Version]
- Bárbara, C.S.; Lorena, R.M. Population viability analysis of common marsupials, Didelphis marsupialis and Didelphis virginiana, in a scenario of constant loss of native vegetation. Mammalia 2020, 84, 475–482. [Google Scholar]
- García-Antón, A.; Traba, J. Population viability analysis of the endangered Dupont’s Lark Chersophilus duponti in Spain. Sci. Rep. 2021, 11, 1–15. [Google Scholar] [CrossRef]
- Franklin, I.R. Evolutionary change in small populations. In Conservation Biology: An Evolutionary–Ecological Perspective; Soule, M.E., Wilcox, B.A., Eds.; Sinauer Associates: Sunderland, MA, USA, 1980; pp. 135–150. [Google Scholar]
- Jamieson, I.G.; Allendorf, F.W. How does the 50/500 rule apply to MVPs? Trends Ecol. Evol. 2012, 27, 578–589. [Google Scholar] [CrossRef]
- Li, D. Relationship between Underwater Noise and Population Distribution of Yangtze Finless Porpoise in Zhenjiang Yangtze Dolphin Provincial Nature Reserve; Shanghai Ocean University: Shanghai, China, 2022. [Google Scholar]
- Slooten, E.; Dawson, S.M. Assessing the effectiveness of conservation management decisions: Likely effects of new protection measures for Hector’s dolphin. Aquat. Conserv. Mar. Freshw. Ecosyst. 2010, 20, 334–347. [Google Scholar] [CrossRef]
- Slooten, E.; Davies, N. Hector’s dolphin risk assessments: Old and new analyses show consistent results. J. R. Soc. N. Z. 2011, 42, 49–60. [Google Scholar] [CrossRef] [Green Version]
- Wang, K.X.; Wang, Z.T.; Mei, Z.G.; Zheng, J.S.; Hao, Y.J.; Han, Y.; Duan, P.X.; Chen, Y.W.; Yang, Y.N.; Qiu, J.S.; et al. Ecological assessment indicator of the Yangtze River: Passive acoustic monitor-based population size of the Yangtze finless porpoise. Chin. Acta Hydrobiol. Sin. 2021, 45, 1390–1395. [Google Scholar]
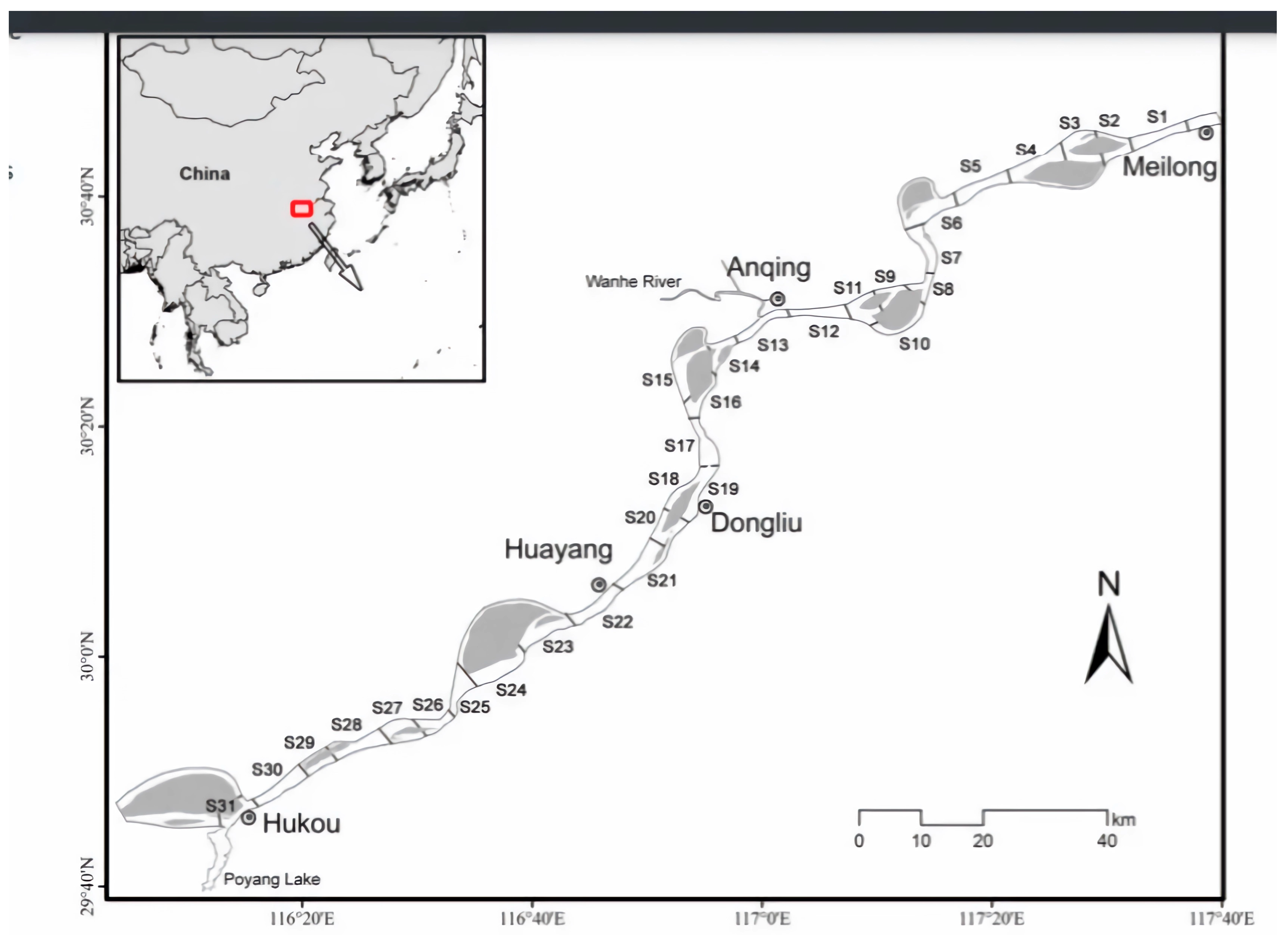
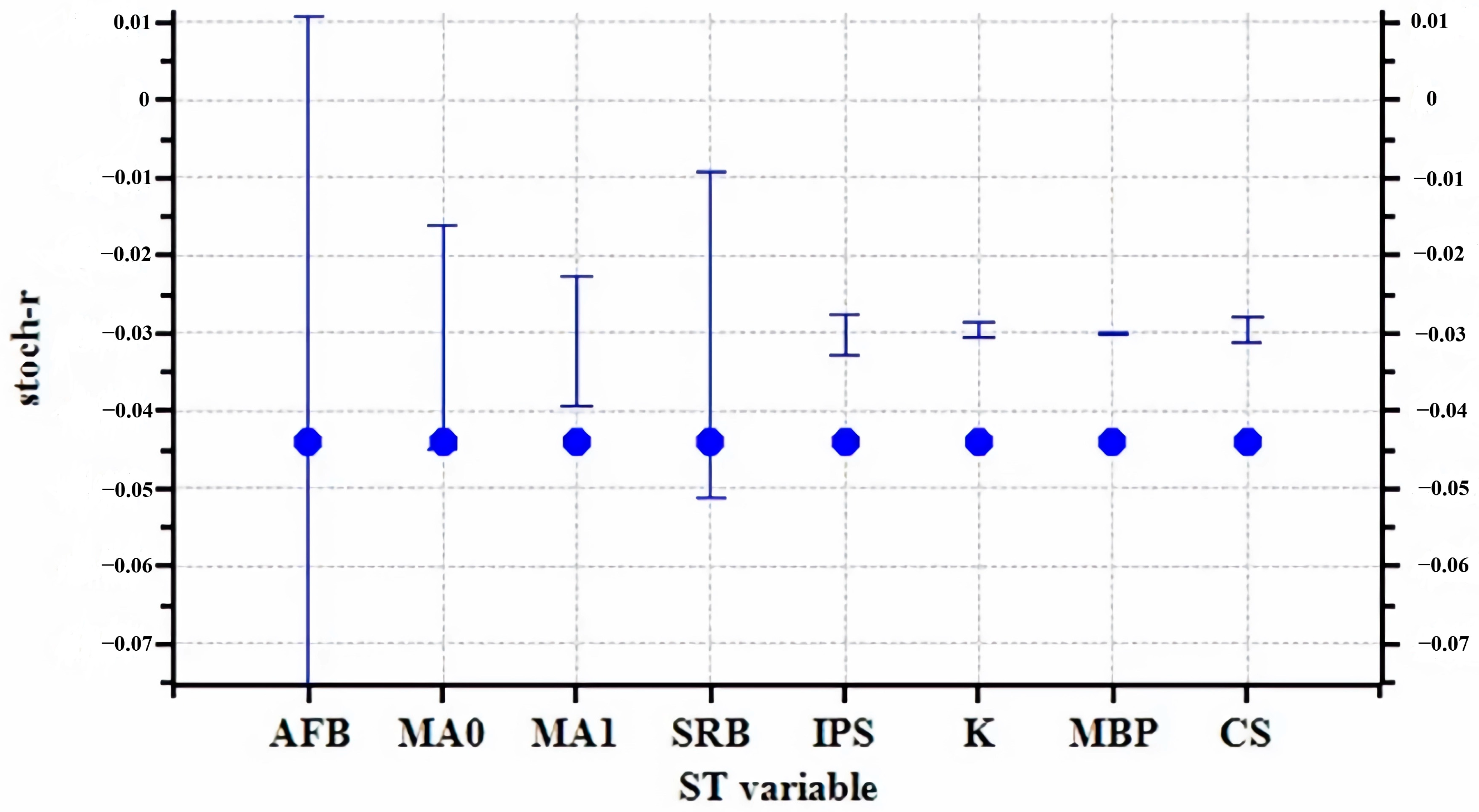
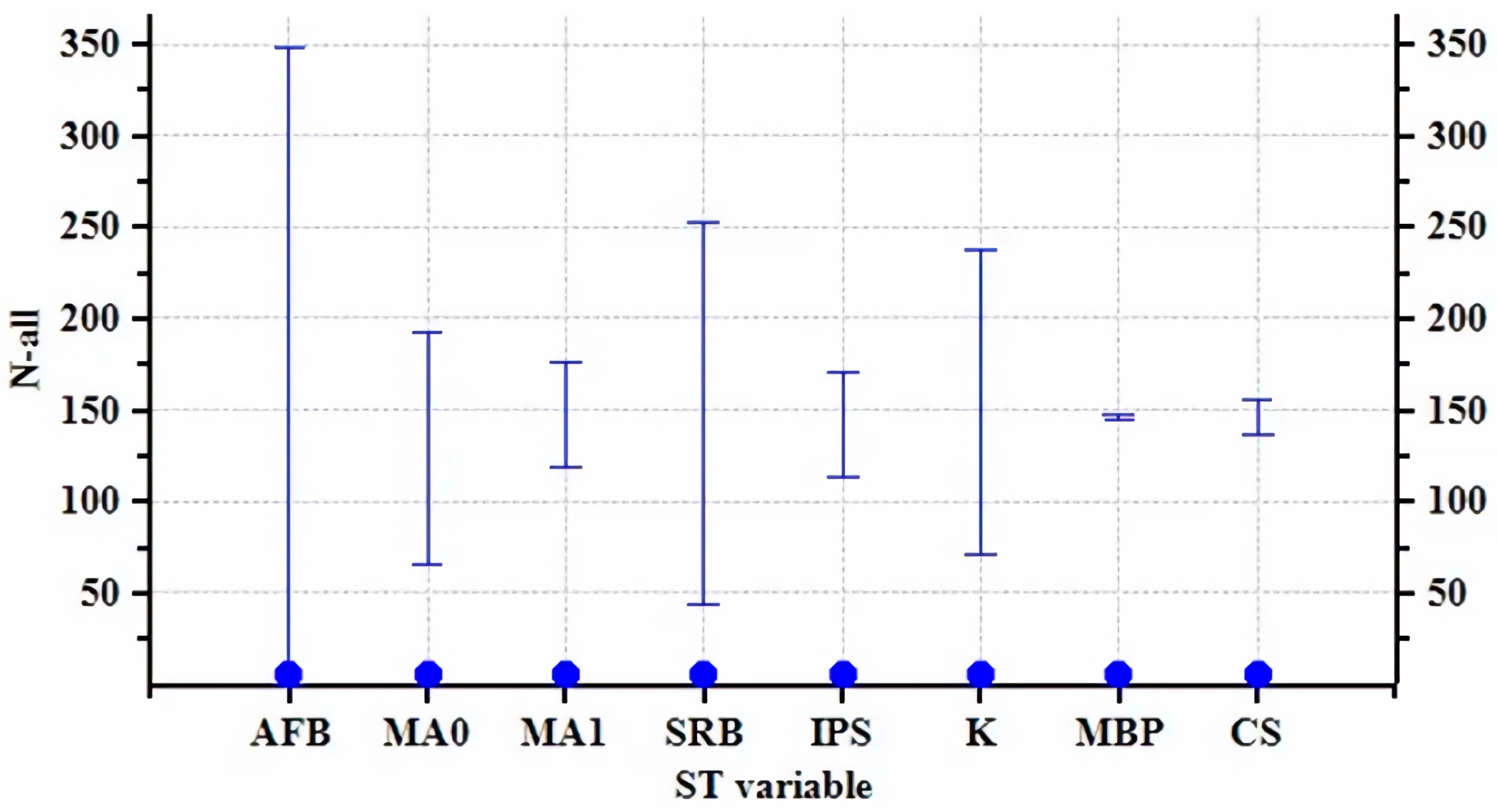
| Parameter | Baseline Value |
|---|---|
| Number of populations Initial population size (N) Carrying capacity (K) Inbreeding depression % of the effect of inbreeding due to recessive lethal alleles Breeding system | 1 181 600 3.14 LE 50 Polygynous |
| Age of first reproduction by males/females | 5/4 years |
| Maximum reproductive age | 18 years |
| Annual % of adult females breeding (SD) Density dependent reproduction? | 50% (10%) No |
| Maximum litter size | 1 |
| Overall offspring sex ratio Adult males in breeding pool (%) Mortality rates: % mortality from age 0–1 (SD) % mortality from age 1–2 (SD) % mortality from other age (SD) | 50:50 70 25 (5) 20 (5) 10 (3) |
| Catastrophe | 1.75%; 50%, 50% 16.7%; 95%, 95% |
| Harvest | None |
| Supplementation | None |
| Projects | Det-r | Stoch-r | PE | GD | N-Extant |
|---|---|---|---|---|---|
| Model 1 (Baseline model) | −0.0230 | −0.0385 | 0.5690 | 0.7698 | 22 |
| Model 2 (Model 1 + immigration 2) | −0.0230 | −0.0273 | 0.3730 | 0.8694 | 68 |
| Model 3 (Model 1 + immigration 4) | −0.0230 | −0.0162 | 0.2440 | 0.9294 | 150 |
| Model 4 (Model 1 + immigration 6) | −0.0230 | −0.0068 | 0.1471 | 0.9603 | 241 |
| Model 5 (Model 1 + immigration 8) | −0.0230 | −0.0004 | 0.0972 | 0.9770 | 322 |
| Model 6 (Model 1 + immigration 10) | −0.0230 | 0.0043 | 0.0685 | 0.9848 | 384 |
| Model 7 (Model 1 + immigration 12) | −0.0230 | 0.0080 | 0.0519 | 0.9894 | 430 |
| Model 8 (Model 1 + emigration 2) | −0.0230 | −0.0438 | 0.6885 | 0.7171 | 15 |
| Model 9 (Model 1 + emigration 4) | −0.0230 | −0.0470 | 0.7555 | 0.6884 | 12 |
| Model 10 (Model 1 + emigration 6) | −0.0230 | −0.0497 | 0.7981 | 0.6766 | 12 |
| Model 11 (Model 1 + emigration 8) | −0.0230 | −0.0508 | 0.8209 | 0.6611 | 12 |
| Model 12 (Model 1 + emigration 10) | −0.0230 | −0.0517 | 0.8234 | 0.6716 | 12 |
| Model 13 (Model 1 + emigration 12) | −0.0230 | −0.0527 | 0.8422 | 0.6580 | 12 |
| Model 14 (Model 1 + individual exchange 8) | −0.0230 | −0.0367 | 0.5320 | 0.8050 | 29 |
| Model 15 (Model 14 + mortality rate of 0–1 age 20%, 1–2 age group 15%) | −0.0092 | −0.0212 | 0.2396 | 0.8838 | 86 |
| Model 16 (Model 15 + breeding rate 55%; general protection model) | 0.0015 | −0.0092 | 0.1058 | 0.9265 | 186 |
| Model 17 (Model 1 + individual exchange 16) | −0.0230 | −0.0244 | 0.3411 | 0.8977 | 88 |
| Model 18 (Model 17 + mortality rate of 0–1 age 15%, 1–2 age group 12.5%) | 0.0009 | −0.0057 | 0.0937 | 0.9438 | 232 |
| Model 19 (Model 18 + breeding rate 60%; medium protection model) | 0.0219 | 0.0144 | 0.0178 | 0.9745 | 421 |
| Model 20 (Model 1 + individual exchange 32 head) | −0.0230 | 0.0006 | 0.0706 | 0.9867 | 300 |
| Model 21 (Model 20 + mortality rate of 0–1 age 12.5%, 1–2 age group 10%) | 0.0074 | 0.0116 | 0.0218 | 0.9874 | 439 |
| Model 22 (Model 21 + breeding rate 65%; optimal protection model) | 0.0383 | 0.0357 | 0.0030 | 0.9926 | 522 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wang, W.; Wang, C.; Yu, J.; Wu, B. Is It Feasible to Preserve a Self-Sustaining Population of Yangtze Finless Porpoise in the Highest Density Section of Yangtze River? Water 2023, 15, 1215. https://doi.org/10.3390/w15061215
Wang W, Wang C, Yu J, Wu B. Is It Feasible to Preserve a Self-Sustaining Population of Yangtze Finless Porpoise in the Highest Density Section of Yangtze River? Water. 2023; 15(6):1215. https://doi.org/10.3390/w15061215
Chicago/Turabian StyleWang, Weiping, Chongrui Wang, Jinxiang Yu, and Bin Wu. 2023. "Is It Feasible to Preserve a Self-Sustaining Population of Yangtze Finless Porpoise in the Highest Density Section of Yangtze River?" Water 15, no. 6: 1215. https://doi.org/10.3390/w15061215
APA StyleWang, W., Wang, C., Yu, J., & Wu, B. (2023). Is It Feasible to Preserve a Self-Sustaining Population of Yangtze Finless Porpoise in the Highest Density Section of Yangtze River? Water, 15(6), 1215. https://doi.org/10.3390/w15061215





