Effect of Antiscalant Usage and Air Diffuser Perforation Diameter on Filtration Performance of Submerged Flat Sheet MBR for Treatment of High Salinity and Scaling Propensity Wastewater
Abstract
:1. Introduction
2. Materials and Methods
2.1. ITOB Wastewater Treatment Plant Feed Water Characteristics
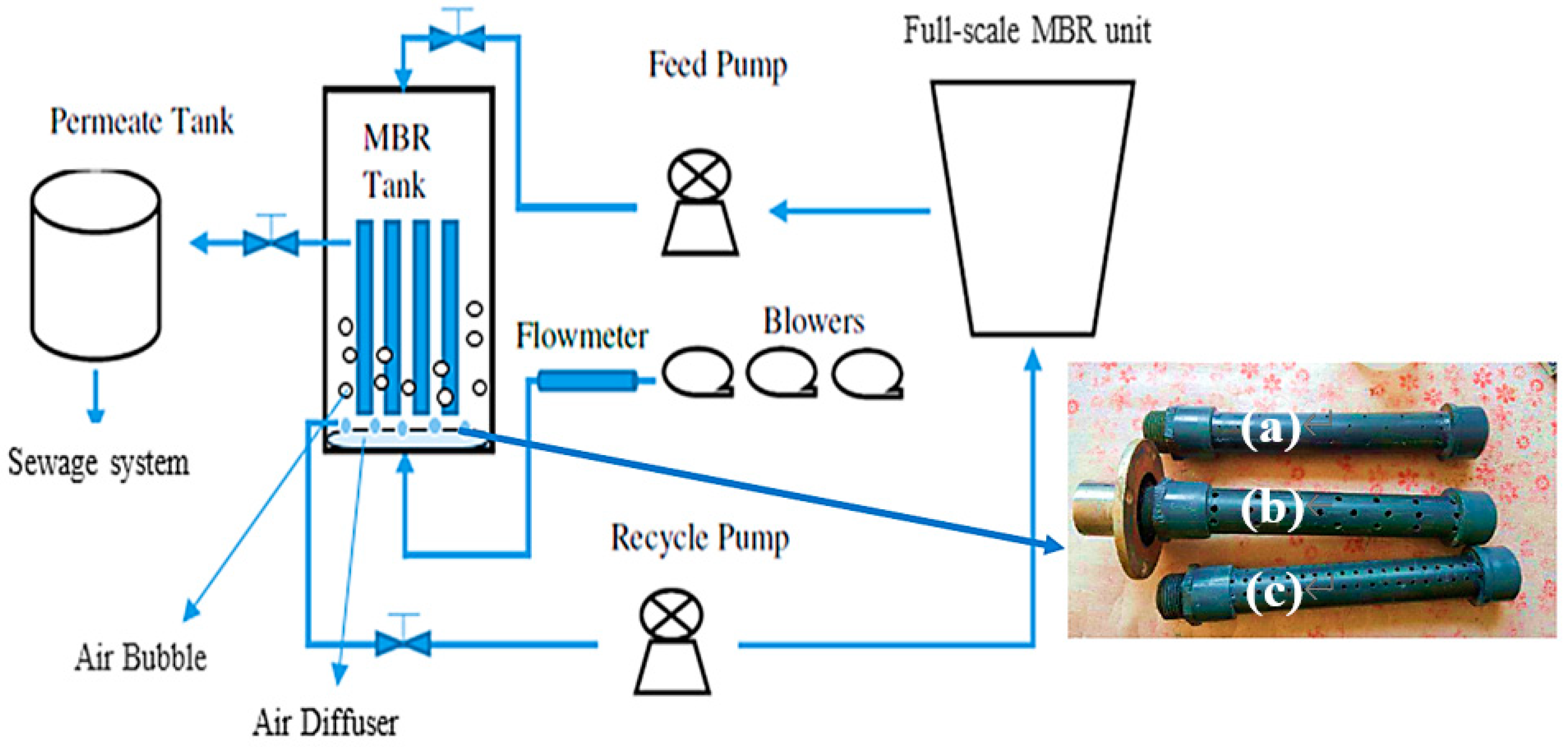
2.2. Pilot MBR System Description
2.3. MBR Tests
- 1.
- MLSS concentration in the bioreactor:
- Low MLSS concentration (10–13 g/L)
- High MLSS concentration (17–21 g/L)
- 2.
- Type and dosage of antiscalant:
- A (40 mg antiscalant/L wastewater)
- B (40 mg antiscalant/L wastewater)
- 3.
- Perforated hole diameter of air diffuser
- 1 mm
- 3 mm
- 5 mm
2.4. MBR Pilot System Performance Evaluation
2.5. Methods
3. Results
3.1. Effect of MLSS Concentration
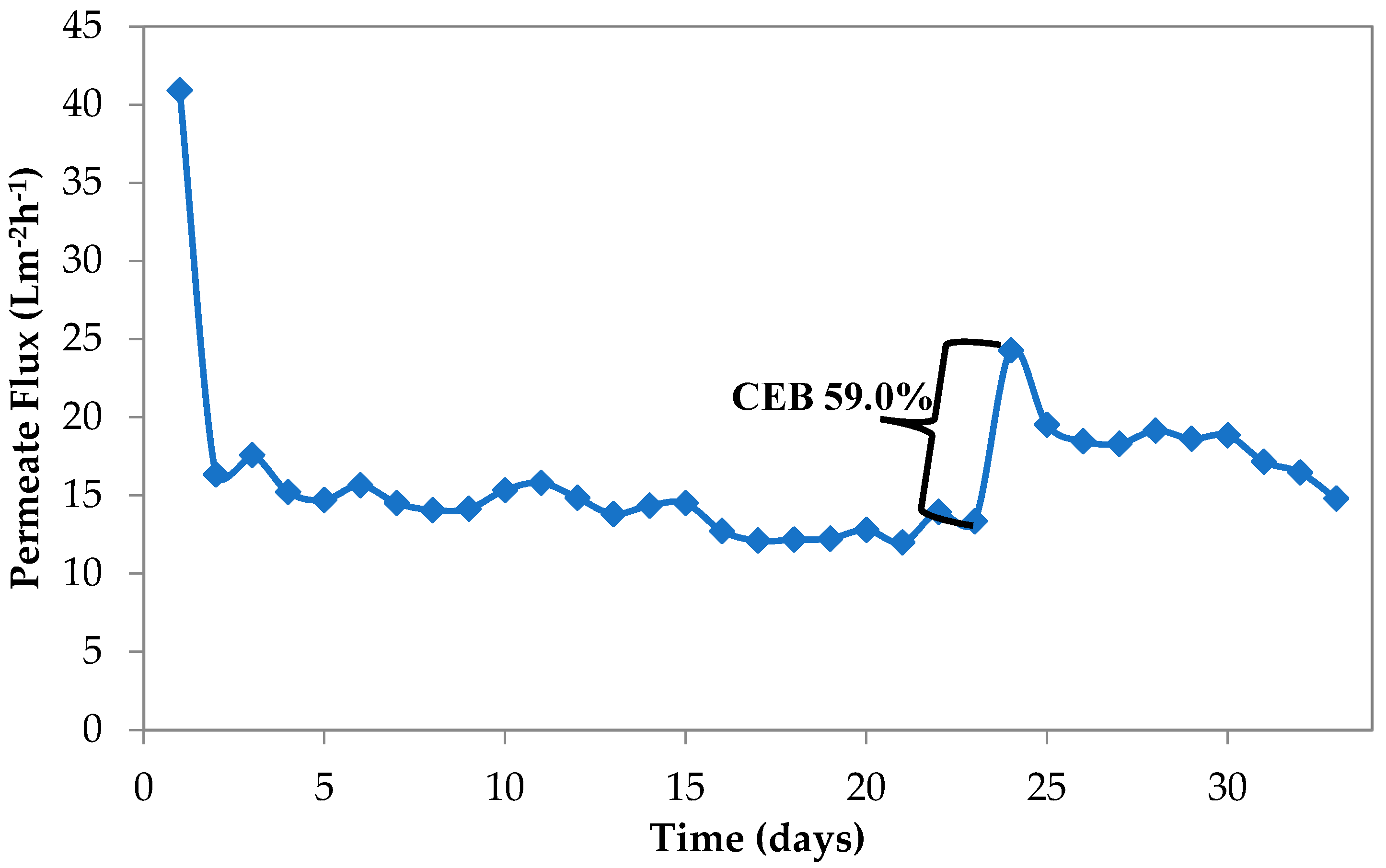
3.1.1. Study 1, Using Low MLSS Levels (10–13 g/L)
3.1.2. Study 2 with High MLSS Concentration (17–21 g/L)
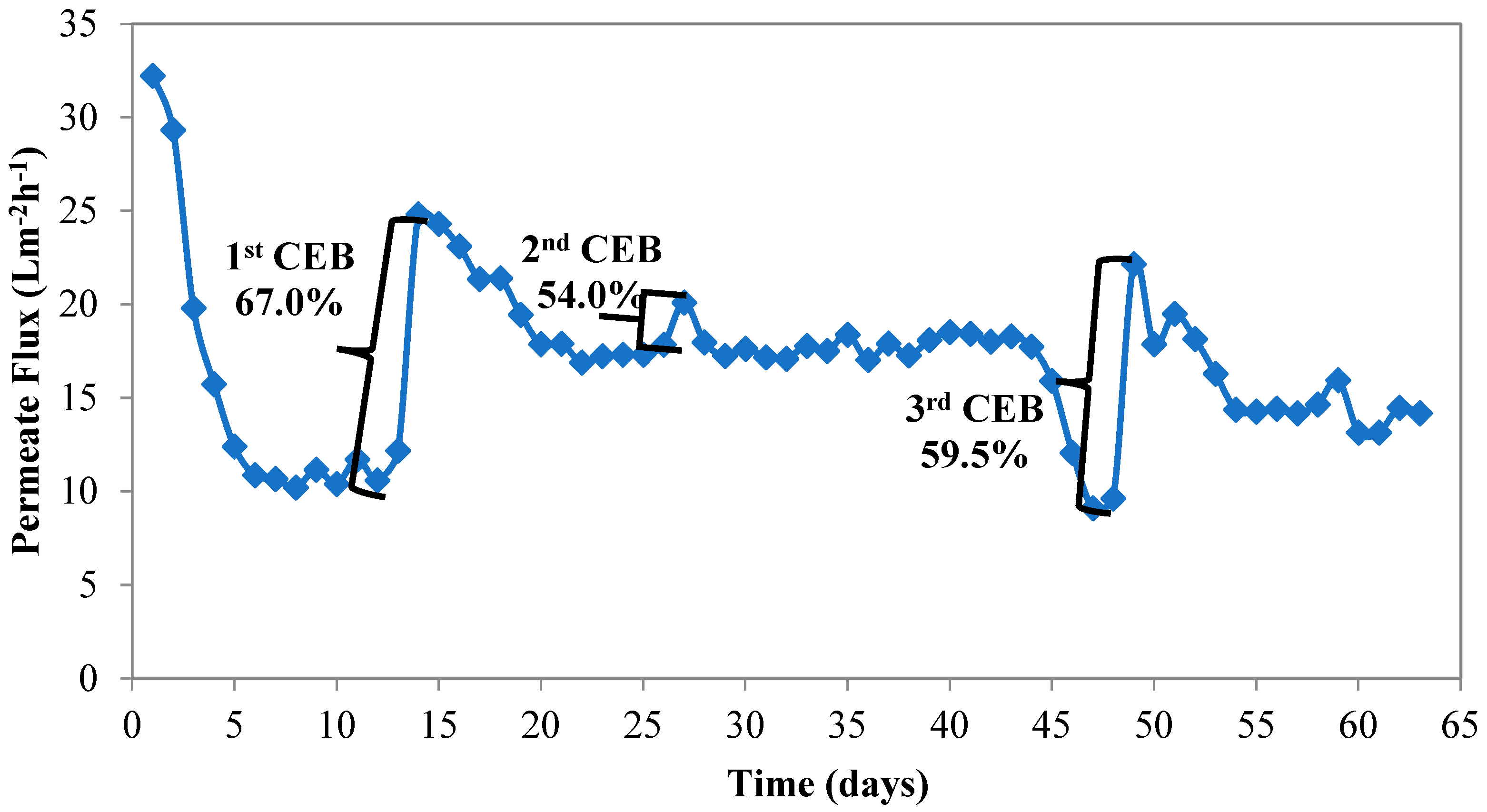
3.2. Type and Dosage of Antiscalant
Study 3-Effect of Antiscalant Usage
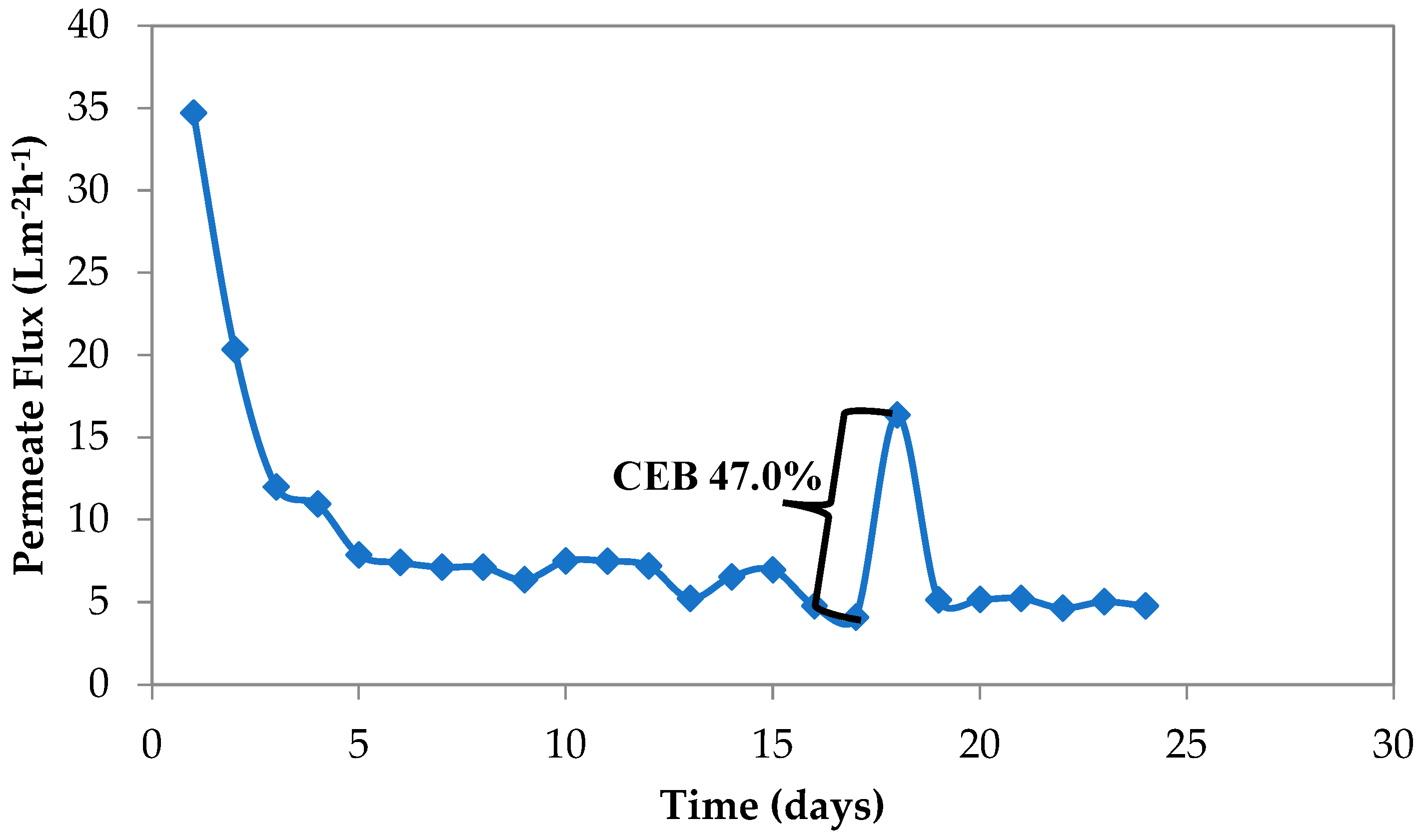
3.3. Effect of Air Diffuser Perforated Hole Diameter
3.3.1. Study 4 Using an Air Diffuser with 5 mm of Perforated Hole Diameter
3.3.2. Effect of an Air Diffuser with 1 mm of Perforated Hole Diameter (Study 5)
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Tang, X.; Fan, C.; Zeng, G.; Zhong, L.; Li, C.; Ren, X.; Song, B.; Liu, X. Phage-host interactions: The neglected part of biological wastewater treatment. Water Res. 2022, 226, 119183. [Google Scholar] [CrossRef]
- Guven, H.; Ersahin, M.E.; Ozgun, H.; Ozturk, I.; Koyuncu, I. Energy and material refineries of future: Wastewater treatment plants. J. Environ. Manag. 2022, 329, 117130. [Google Scholar] [CrossRef]
- Judd, S. The MBR Book: Principles and Applications of Membrane Bioreactors for Water and Wastewater Treatment, 2nd ed.; Elsevier: Amsterdam, The Netherlands, 2011. [Google Scholar]
- Gao, F.; Li, L.; Shi, Y.; Xue, X.; Mao, J.; Xing, L.; Yao, X. Interaction mechanisms of fouling and cleaning protocol for A/O-MBR treatment of coal-to-hydrogen wastewater. Process. Saf. Environ. Prot. 2023, 169, 515–525. [Google Scholar] [CrossRef]
- Wang, C.; Ng, T.C.A.; Ding, M.; Ng, H.Y. Insights on fouling development and characteristics during different fouling stages between a novel vibrating MBR and an air-sparging MBR for domestic wastewater treatment. Water Res. 2022, 212, 118098. [Google Scholar] [CrossRef]
- Emmanuel, E.; Keck, G.; Blanchard, J.M.; Vermande, P.; Perrodin, Y. Toxicological effects of disinfections using sodium hypochlorite on aquatic organisms and its contribution to AOX formation in hospital wastewater. Environ. Int. 2004, 30, 891–900. [Google Scholar] [CrossRef] [PubMed]
- Zheng, X.; Liu, J. Virus rejection with two model human enteric viruses in membrane bioreactor system. Sci. China Ser. B Chem. 2007, 50, 397–404. [Google Scholar] [CrossRef] [Green Version]
- Dogan, E.C.; Yasar, A.; Sen, U.; Aydiner, C. Water recovery from treated urban wastewater by ultrafiltration and reverse osmosis for landscape irrigation. Urban. Water J. 2015, 13, 553–568. [Google Scholar] [CrossRef]
- Fang, Y.; Chen, D.; Zhang, Y.; Field, R.; Wu, J.J.; Wang, B. Wastewater treatment in large-scale novel corrugated-sheet MBR. J. Water Process. Eng. 2022, 50, 103215. [Google Scholar] [CrossRef]
- Wang, C.; Ng, T.C.A.; Ng, H.Y. Comparison between novel vibrating ceramic MBR and conventional air-sparging MBR for domestic wastewater treatment: Performance, fouling control and energy consumption. Water Res. 2021, 203, 117521. [Google Scholar] [CrossRef]
- Kochkodan, V.; Hilal, N. A comprehensive review on surface modified polymer membranes for biofouling mitigation. Desalination 2015, 356, 187–207. [Google Scholar] [CrossRef]
- Malaeb, L.; Le-Clech, P.; Vrouwenvelder, J.S.; Ayoub, G.M.; Saikaly, P.E. Do biological-based strategies hold promise to biofouling control in MBRs? Water Res. 2013, 47, 5447–5463. [Google Scholar] [CrossRef] [PubMed]
- Aslam, M.; Charfi, A.; Lesage, G.; Heran, M.; Kim, J. Membrane bioreactors for wastewater treatment: A review of mechanical cleaning by scouring agents to control membrane fouling. Chem. Eng. J. 2017, 307, 897–913. [Google Scholar] [CrossRef]
- Krzeminski, P.; Leverette, L.; Malamis, S.; Katsou, E. Membrane bioreactors—A review on recent developments in energy reduction, fouling control, novel configurations, LCA and market prospects. J. Membr. Sci. 2017, 527, 207–227. [Google Scholar] [CrossRef] [Green Version]
- Mei, X.; Quek, P.J.; Wang, Z.; Ng, H.Y. Alkali-assisted membrane cleaning for fouling control of anaerobic ceramic membrane bioreactor. Bioresour. Technol. 2017, 240, 25–32. [Google Scholar] [CrossRef]
- Al-Asheh, S.; Bagheri, M.; Aidan, A. Membrane bioreactor for wastewater treatment: A review. Case Stud. Chem. Environ. Eng. 2021, 4, 100109. [Google Scholar] [CrossRef]
- Myat, D.T.; Stewart, M.B.; Mergen, M.; Zhao, O.; Orbell, J.D.; Gray, S. Experimental and computational investigations of the interactions between model organic compounds and subsequent membrane fouling. Water Res. 2014, 48, 108–118. [Google Scholar] [CrossRef] [Green Version]
- Chun, Y.; Jeong, K.; Cho, K.H. Influences of Combined Organic Fouling and Inorganic Scaling on Flux and Fouling Behaviors in Forward Osmosis. Membranes 2020, 10, 115. [Google Scholar] [CrossRef]
- Meng, F.; Chae, S.R.; Drews, A.; Kraume, M.; Shin, H.S.; Yang, F. Recent advances in membrane bioreactors (MBRs): Membrane fouling and membrane material. Water Res. 2009, 43, 1489–1512. [Google Scholar] [CrossRef] [PubMed]
- Decho, A.W. Overview of biopolymer-induced mineralization: What goes on in biofilms? Ecol. Eng. 2010, 36, 137–144. [Google Scholar] [CrossRef]
- Wang, Z.; Ma, J.; Tang, C.Y.; Kimura, K.; Wang, Q.; Han, X. Membrane cleaning in membrane bioreactors: A review. J. Membr. Sci. 2014, 468, 276–307. [Google Scholar] [CrossRef]
- Bohm, L.; Drews, A.; Prieske, H.; Berube, P.R.; Kraume, M. The importance of fluid dynamics for MBR fouling mitigation. Bioresour. Technol. 2012, 122, 50–61. [Google Scholar] [CrossRef]
- Drews, A.; Prieske, H.; Meyer, E.-L.; Senger, G.; Kraume, M. Advantageous and detrimental effects of air sparging in membrane filtration: Bubble movement, exerted shear and particle classification. Desalination 2010, 250, 1083–1086. [Google Scholar] [CrossRef]
- Yang, X.; Wang, R.; Fane, A.G.; Tang, C.Y.; Wenten, I.G. Membrane module design and dynamic shear-induced techniques to enhance liquid separation by hollow fiber modules: A review. Desalination Water Treat. 2013, 51, 3604–3627. [Google Scholar] [CrossRef] [Green Version]
- Wang, Z.; Wu, Z. A Review of Membrane Fouling in MBRs: Characteristics and Role of Sludge Cake Formed on Membrane Surfaces. Sep. Sci. Technol. 2009, 44, 3571–3596. [Google Scholar] [CrossRef]
- Iorhemen, O.T.; Hamza, R.A.; Tay, J.H. Membrane fouling control in membrane bioreactors (MBRs) using granular materials. Bioresour. Technol. 2017, 240, 9–24. [Google Scholar] [CrossRef]
- Aslam, M.; Ahmad, R.; Kim, J. Recent developments in biofouling control in membrane bioreactors for domestic wastewater treatment. Sep. Purif. Technol. 2018, 206, 297–315. [Google Scholar] [CrossRef]
- Chen, W.; Luo, J.; Cao, R.; Li, Y.; Liu, J. Effect of macroporous adsorption resin-membrane bioreactor hybrid system against fouling for municipal wastewater treatment. Bioresour. Technol. 2017, 224, 112–117. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.; Ahn, W.-Y.; Lee, C.-H. Comparison of the filtration characteristics between attached and suspended growth microorganisms in submerged membrane bioreactor. Water Res. 2001, 35, 2435–2445. [Google Scholar] [CrossRef]
- Gkotsis, P.K.; Batsari, E.L.; Peleka, E.N.; Tolkou, A.K.; Zouboulis, A.I. Fouling control in a lab-scale MBR system: Comparison of several commercially applied coagulants. J. Environ. Manag. 2017, 203, 838–846. [Google Scholar] [CrossRef]
- Scholz, W.G.; RougÉ, P.; BÓdalo, A.; Leitz, U. Desalination of Mixed Tannery Effluent with Membrane Bioreactor and Reverse Osmosis Treatment. Environ. Sci. Technol. 2005, 39, 8505–8511. [Google Scholar] [CrossRef]
- Yang, M.; Yu, D.; Liu, M.; Zheng, L.; Zheng, X.; Wei, Y.; Wang, F.; Fan, Y. Optimization of MBR hydrodynamics for cake layer fouling control through CFD simulation and RSM design. Bioresour. Technol. 2017, 227, 102–111. [Google Scholar] [CrossRef] [PubMed]
- Amini, E.; Mehrnia, M.R.; Mousavi, S.M.; Mostoufi, N. Experimental Study and Computational Fluid Dynamics Simulation of a Full-Scale Membrane Bioreactor for Municipal Wastewater Treatment Application. Ind. Eng. Chem. Res. 2013, 52, 9930–9939. [Google Scholar] [CrossRef]
- Du, X.; Liu, X.; Wang, Y.; Radaei, E.; Lian, B.; Leslie, G.; Li, G.; Liang, H. Particle deposition on flat sheet membranes under bubbly and slug flow aeration in coagulation-microfiltration process: Effects of particle characteristic and shear stress. J. Membr. Sci. 2017, 541, 668–676. [Google Scholar] [CrossRef]
- Ma, C.; Liu, Y.; Li, F.; Shen, C.; Huang, M.; Wang, Z.; Cao, C.; Zhou, Q.; Sheng, Y.; Sand, W. CFD simulations of fiber-fiber interaction in a hollow fiber membrane bundle: Fiber distance and position matters. Sep. Purif. Technol. 2019, 209, 707–713. [Google Scholar] [CrossRef]
- Sanguanpak, S.; Chiemchaisri, C.; Chiemchaisri, W.; Yamamoto, K. Influence of operating pH on biodegradation performance and fouling propensity in membrane bioreactors for landfill leachate treatment. Int. Biodeterior. Biodegrad. 2015, 102, 64–72. [Google Scholar] [CrossRef] [Green Version]
- Wu, J.; Zhuang, Y.; Li, H.; Huang, X. pH Adjusting to Reduce Fouling Propensity of Activated Sludge Mixed Liquor in Membrane Bioreactors. Sep. Sci. Technol. 2010, 45, 890–895. [Google Scholar] [CrossRef]
- Zhang, Y.; Zhang, M.; Wang, F.; Hong, H.; Wang, A.; Wang, J.; Weng, X.; Lin, H. Membrane fouling in a submerged membrane bioreactor: Effect of pH and its implications. Bioresour. Technol. 2014, 152, 7–14. [Google Scholar] [CrossRef] [PubMed]
- Rahardianto, A.; McCool, B.C.; Cohen, Y. Reverse Osmosis Desalting of Inland Brackish Water of High Gypsum Scaling Propensity: Kinetics and Mitigation of Membrane Mineral Scaling. Environ. Sci. Technol. 2008, 42, 4292–4297. [Google Scholar] [CrossRef]
- Rahardianto, A.; Shih, W.-Y.; Lee, R.-W.; Cohen, Y. Diagnostic characterization of gypsum scale formation and control in membrane desalination of brackish water. J. Membr. Sci. 2006, 279, 655–668. [Google Scholar] [CrossRef]
- Thompson, J.; Rahardianto, A.; Gu, H.; Uchymiak, M.; Bartman, A.; Hedrick, M.; Lara, D.; Cooper, J.; Faria, J.; Christofides, P.D.; et al. Rapid field assessment of RO desalination of brackish agricultural drainage water. Water Res. 2013, 47, 2649–2660. [Google Scholar] [CrossRef]
- Lyster, E.; Kim, M.-M.; Au, J.; Cohen, Y. A method for evaluating antiscalant retardation of crystal nucleation and growth on RO membranes. J. Membr. Sci. 2010, 364, 122–131. [Google Scholar] [CrossRef]
- Hasson, D.; Drak, A.; Semiat, R. Induction times induced in an RO system by antiscalants delaying CaSO4 precipitation. Desalination 2003, 157, 193–207. [Google Scholar] [CrossRef]
- Ghafour, E.E.A. Enhancing RO system performance utilizing antiscalants. Desalination 2002, 153, 149–153. [Google Scholar] [CrossRef]
- Shih, W.-Y.; Albrecht, K.; Glater, J.; Cohen, Y. A dual-probe approach for evaluation of gypsum crystallization in response to antiscalant treatment. Desalination 2004, 169, 213–221. [Google Scholar] [CrossRef]
- Knoell, T.; Patel, M. The Challenges of Identifying, Evaluating and Implementing Antiscalants in RO Reuse Applications: So Many Choices—So Few Winners. In Proceedings of the AWWA Membrane Technology Conference, Long Beach, CA, USA, 28–31 March 2011. [Google Scholar]
- Jarma, Y.A.; Parlar, I.; Pek, T.Ö.; Kayral, K.; Kabay, N.; Yiğit, N.Ö.; Kitiş, M.; Yüksel, M. Study on Operational Conditions to Minimize Membrane Fouling in Membrane Bioreactor (MBR) System for Wastewater Treatment-Preliminary Pilot Tests. J. Membr. Sci. Res. 2018, 4, 212–217. [Google Scholar] [CrossRef]
- Bunani, S.; Yörükoğlu, E.; Sert, G.; Yüksel, Ü.; Yüksel, M.; Kabay, N. Application of nanofiltration for reuse of municipal wastewater and quality analysis of product water. Desalination 2013, 315, 33–36. [Google Scholar] [CrossRef]
- Sert, G.; Bunani, S.; Kabay, N.; Egemen, Ö.; Arda, M.; Pek, T.Ö.; Yüksel, M. Investigation of mini pilot scale MBR-NF and MBR-RO integrated systems performance—Preliminary field tests. J. Water Process. Eng. 2016, 12, 72–77. [Google Scholar] [CrossRef]
- Gündoğdu, M.; Jarma, Y.A.; Kabay, N.; Pek, T.Ö.; Yüksel, M. Integration of MBR with NF/RO processes for industrial wastewater reclamation and water reuse-effect of membrane type on product water quality. J. Water Process. Eng. 2019, 29, 100574. [Google Scholar] [CrossRef]
- Jarma, Y.A.; Karaoglu, A.; Tekin, O.; Baba, A.; Okten, H.E.; Tomaszewska, B.; Bostanci, K.; Arda, M.; Kabay, N. Assessment of different nanofiltration and reverse osmosis membranes for simultaneous removal of arsenic and boron from spent geothermal water. J. Hazard. Mater. 2021, 405, 124129. [Google Scholar] [CrossRef] [PubMed]
- Dayarathne, H.N.P.; Jeong, S.; Jang, A. Chemical-free scale inhibition method for seawater reverse osmosis membrane process: Air micro-nano bubbles. Desalination 2019, 461, 1–9. [Google Scholar] [CrossRef]
- Zhang, J.; Qin, W.; Ng, D.; Ji, X.; Zhang, X.; Ye, Y.; Xie, Z. Fouling behavior of calcium phosphate in direct contact membrane distillation. Environ. Technol. Innov. 2021, 21, 101203. [Google Scholar] [CrossRef]
- Yigit, N.Ö. Municipal Wastewater Treatment by Membrane Bioreactor (MBR); Suleyman Demirel University: Isparta, Turkey, 2007. [Google Scholar]
- Ansari, A.; Pena-Bahamonde, J.; Fanourakis, S.K.; Hu, Y.; Rodrigues, D.F. Microbially-induced mineral scaling in desalination conditions: Mechanisms and effects of commercial antiscalants. Water Res. 2020, 179, 115863. [Google Scholar] [CrossRef]
- Trukhina, M.; Popov, K.; Oshchepkov, M.; Tkachenko, S. Impact of colloidal iron hydroxide and colloidal silicon dioxide on calcium sulfate crystallization in the presence of antiscalants. Int. J. Corros. Scale Inhib. 2022, 11, 1147–1171. [Google Scholar] [CrossRef]
- Mangal, M.N.; Yangali-Quintanilla, V.A.; Salinas-Rodriguez, S.G.; Dusseldorp, J.; Blankert, B.; Kemperman, A.J.B.; Schippers, J.C.; Kennedy, M.D.; van der Meer, W.G.J. Application of a smart dosing pump algorithm in identifying real-time optimum dose of antiscalant in reverse osmosis systems. J. Membr. Sci. 2022, 658, 120717. [Google Scholar] [CrossRef]
- Yigit, N.O.; Harman, I.; Civelekoglu, G.; Koseoglu, H.; Cicek, N.; Kitis, M. Membrane fouling in a pilot-scale submerged membrane bioreactor operated under various conditions. Desalination 2008, 231, 124–132. [Google Scholar] [CrossRef]
- Skouteris, G.S. Low Energy Membrane Bioreactors for Decentralised Waste Water Treatment; University of Bath: Bath, UK, 2010. [Google Scholar]
- Baek, S.H.; Pagilla, K.R.; Kim, H.-J. Lab-scale study of an anaerobic membrane bioreactor (AnMBR) for dilute municipal wastewater treatment. Biotechnol. Bioprocess. Eng. 2010, 15, 704–708. [Google Scholar] [CrossRef]
- Visvanathan, C.; Aim, R.B.; Parameshwaran, K. Membrane Separation Bioreactors for Wastewater Treatment. Crit. Rev. Environ. Sci. Technol. 2000, 30, 1–48. [Google Scholar] [CrossRef]
- Mutamim, N.S.A.; Noor, Z.Z.; Hassan, M.A.A.; Olsson, G. Application of membrane bioreactor technology in treating high strength industrial wastewater: A performance review. Desalination 2012, 305, 1–11. [Google Scholar] [CrossRef]
- Borchers, A.; Pieler, T. Programming pluripotent precursor cells derived from Xenopus embryos to generate specific tissues and organs. Genes 2010, 1, 413–426. [Google Scholar] [CrossRef] [Green Version]
- Di Trapani, D.; Corsino, S.F.; Torregrossa, M.; Viviani, G. Treatment of high strength industrial wastewater with membrane bioreactors for water reuse: Effect of pre-treatment with aerobic granular sludge on system performance and fouling tendency. J. Water Process. Eng. 2019, 31, 100859. [Google Scholar] [CrossRef]
- Radjenović, J.; Matošić, M.; Mijatović, I.; Petrović, M.; Barceló, D. Membrane Bioreactor (MBR) as an Advanced Wastewater Treatment Technology. In Emerging Contaminants from Industrial and Municipal Waste. The Handbook of Environmental Chemistry, vol 5S/2; Barceló, D., Petrovic, M., Eds.; Springer: Berlin/Heidelberg, Germany, 2008; pp. 37–101. [Google Scholar] [CrossRef]
- Amjad, Z. Scale Inhibition in Desalination Applications: An Overview. In Proceedings of the Nace International Annual Conference and Exposition, Denver, CO, USA, 24 March 1996. [Google Scholar]
- Dudley, L.Y.; Baker, J.S. PermaCare®, The Role of Antiscalants and Cleaning Chemicals to Control Membrane Fouling; Reprint R-79, 77. Available online: https://pdf4pro.com/cdn/the-role-of-antiscalants-and-cleaning-chemicals-to-34a12f.pdf (accessed on 1 January 1999).
- Antiscalant, R. Ropur Antiscalant. 2010. Available online: https://www.lenntech.com/Data-sheets/Ropur-rpi-3000a-e_tdf-L.pdf (accessed on 1 November 2010).
- Macedonio, F.; Katzir, L.; Geisma, N.; Simone, S.; Drioli, E.; Gilron, J. Wind-Aided Intensified eVaporation (WAIV) and Membrane Crystallizer (MCr) integrated brackish water desalination process: Advantages and drawbacks. Desalination 2011, 273, 127–135. [Google Scholar] [CrossRef]
- Popov, K.I.; Kovaleva, N.E.; Rudakova, G.Y.; Kombarova, S.P.; Larchenko, V.E. Recent state-of-the-art of biodegradable scale inhibitors for cooling-water treatment applications (Review). Therm. Eng. 2016, 63, 122–129. [Google Scholar] [CrossRef]
- Fallon, H.P. A Performance-Based Approach to Cooling Water Chemistry Control. PowerPlant Chem. 2004, 4, 6. [Google Scholar]
- Huang, X.; Li, C.; Zuo, K.; Li, Q. Predominant Effect of Material Surface Hydrophobicity on Gypsum Scale Formation. Env. Sci. Technol. 2020, 54, 15395–15404. [Google Scholar] [CrossRef] [PubMed]
- Alshahri, A.H.; Giagnorio, M.; Dehwah, A.H.A.; Obaid, M.; Missimer, T.M.; Leiknes, T.; Ghaffour, N.; Fortunato, L. Advanced coagulation with liquid ferrate as SWRO desalination pretreatment during severe algal bloom. Process performance, environmental impact, and cost analysis. Desalination 2022, 537, 115864. [Google Scholar] [CrossRef]
- AzadiAghdam, M.; Park, M.; Lopez-Prieto, I.J.; Achilli, A.; Snyder, S.A.; Farrell, J. Pretreatment for water reuse using fluidized bed crystallization. J. Water Process. Eng. 2020, 35, 101226. [Google Scholar] [CrossRef]
- Brover, S.; Lester, Y.; Brenner, A.; Sahar-Hadar, E. Optimization of ultrafiltration as pre-treatment for seawater RO desalination. Desalination 2022, 524, 115478. [Google Scholar] [CrossRef]
- Capdevielle, A.; Sýkorová, E.; Béline, F.; Daumer, M.-L. Effects of organic matter on crystallization of struvite in biologically treated swine wastewater. Environ. Technol. 2016, 37, 880–892. [Google Scholar] [CrossRef] [PubMed]
- Molenaar, N.; Venmans, A.A.M. Calcium carbonate cementation of sand: A method for producing artificially cemented samples for geotechnical testing and a comparison with natural cementation processes. Eng. Geol. 1993, 35, 103–122. [Google Scholar] [CrossRef]
- Koutsoukos, P.G.; Kofina, A.N.; Kanellopoulou, D.G. Solubility of salts in water: Key issue for crystal growth and dissolution processes. Pure Appl. Chem. 2007, 79, 825–850. [Google Scholar] [CrossRef]
- Yu, W.; Chen, W.; Yang, H. Evaluation of structural effects on the antiscaling performance of various graft cellulose-based antiscalants in RO membrane scaling control. J. Membr. Sci. 2021, 620, 118893. [Google Scholar] [CrossRef]
- Sofia, A.; Ng, W.J.; Ong, S.L. Engineering design approaches for minimum fouling in submerged MBR. Desalination 2004, 160, 167–174. [Google Scholar] [CrossRef]



| Parameter | Influent |
|---|---|
| EC (mS/cm) | 2.25–3.58 |
| pH | 7.05–7.69 |
| Calcium (mg/L) | 140–160 |
| Salinity (%o) | 1.52–2.05 |
| TSS (mg/L) | 580–1341 |
| NH4-N | 19.7–44.3 |
| PO4-P (mg/L) | 2.1–18.2 |
| TN | 35.8–75.3 |
| NO3-N (mg/L) | 0.5–11.0 |
| NO2-N (mg/L) | 0.112–0.921 |
| COD (mg/L) | 262–1006 |
| Color (Pt-Co) | 1697–2304 |
| Property | Value |
|---|---|
| Reactor capacity (L) | 50 |
| Membrane active area (m2) | 0.11 |
| Nominal pore size of membrane panel (micron) | 0.4 |
| Membrane type | Plate and frame |
| Membrane material | Kubota-PE |
| Diffuser diameter (mm) | 1, 3, 5 |
| MLSS (g/L) | 10–22 |
| HRT (h) | 3–4 |
| SRT (day) | 37–45 |
| Effect of MLSS Concentration | Effect of Type of Antiscalant | Effect of Air Diffuser Hole Diameter | |||
|---|---|---|---|---|---|
| Operational Parameter | Study 1 | Study 2 | Study 3 | Study 4 | Study 5 |
| MLSS (g/L) | 10–13 | 17–21 | 9–13 | 15–18 | 18–22 |
| AS type | AS-1 | AS-1 | AS-2 | AS-1 | AS-1 |
| AS concentration (mg/L) | 40 | ||||
| AS Dosage (mL/min) | 5 | ||||
| Diffuser hole diameter (mm) | 3 | 5 | 1 | ||
| Operation time (month) | 1 | 2 | 1 | 1 | 1 |
| Effect of MLSS Concentration | Effect of Antiscalant Type | Effect of PHD of Air Diffuser | |||
|---|---|---|---|---|---|
| Operation parameters | Study 1 | Study 2 | Study 3 | Study 4 | Study 5 |
| Feed MLSS (g/L) | 17–21 | 10–13 | 9–13 | 15–18 | 18–22 |
| EC (mS/cm) | 3.62–4.59 | 3.65–3.95 | 3.38–3.99 | 3.18–3.94 | 3.92–3.98 |
| Salinity (%o) | 1.91–2.68 | 1.94–2.09 | 1.77–2.10 | 1.98–2.09 | 2.08–2.12 |
| TDS (mg/L) | 1870–2620 | 1890–2050 | 1740–2059 | 1938–2043 | 2034–2074 |
| pH | 7.46–8.29 | 6.73–7.07 | 7.12–7.44 | 7.64–8.02 | 7.93–8.21 |
| DO (mg/L) | 5.59–6.94 | 7.95–8.56 | 6.75–7.41 | 7.45–8.14 | 7.32–7.88 |
| Color (Pt-Co) | 26–53 | 17–44 | 44–92 | 105–122 | 105–123 |
| TSS (mg/L) | 1–8 | 0 | 0 | 5–13 | 7–14 |
| AS type | 1 | 1 | 2 | 1 | 1 |
| PHD (mm) | 3 | 3 | 3 | 5 | 1 |
| Time (month) | 2 | 1 | 1 | 1 | 1 |
| Studies | TSS | Color | COD | NH4-N | PO4-P | NO2-N | TN | |
|---|---|---|---|---|---|---|---|---|
| Effect of MLSS Concentration | Study 1 | 99.7 | 98.9 | 98.4 | 99.9 | 99.2 | 93.9 | 71.5 |
| Study 2 | >99.9 | 99.4 | 98.5 | 99.8 | 99.1 | 86.0 | 59.1 | |
| Effect of AS Type | Study 3 | >99.9 | 98.3 | 97.6 | 99.5 | 99.2 | 88.9 | 47.8 |
| Effect of Diffuser Pore Diameter | Study 4 | 98.47 | 90.25 | 95.34 | 86.90 | 67.01 | 75.03 | 20.36 |
| Study 5 | 99.14 | 84.62 | 96.63 | 99.94 | 72.02 | 89.17 | 71.51 | |
| Literature (Melin et al. [56]) | >99 | NG | 89–98 | 80–90 | 62–97 | NG | 36–80 | |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Parlar, İ.; Jarma, Y.A.; Pek, T.O.; Kabay, N.; Kitis, M.; Yigit, N.O.; Yuksel, M. Effect of Antiscalant Usage and Air Diffuser Perforation Diameter on Filtration Performance of Submerged Flat Sheet MBR for Treatment of High Salinity and Scaling Propensity Wastewater. Water 2023, 15, 1191. https://doi.org/10.3390/w15061191
Parlar İ, Jarma YA, Pek TO, Kabay N, Kitis M, Yigit NO, Yuksel M. Effect of Antiscalant Usage and Air Diffuser Perforation Diameter on Filtration Performance of Submerged Flat Sheet MBR for Treatment of High Salinity and Scaling Propensity Wastewater. Water. 2023; 15(6):1191. https://doi.org/10.3390/w15061191
Chicago/Turabian StyleParlar, İlker, Yakubu A. Jarma, Taylan O. Pek, Nalan Kabay, Mehmet Kitis, Nevzat O. Yigit, and Mithat Yuksel. 2023. "Effect of Antiscalant Usage and Air Diffuser Perforation Diameter on Filtration Performance of Submerged Flat Sheet MBR for Treatment of High Salinity and Scaling Propensity Wastewater" Water 15, no. 6: 1191. https://doi.org/10.3390/w15061191







