A Comprehensive Approach to the Chemistry, Pollution Impact and Risk Assessment of Drinking Water Sources in a Former Industrialized Area of Romania
Abstract
:1. Introduction
2. Materials and Methods
2.1. Study Area and Sampling
2.2. Sample Preparation and Chemical Analysis
2.3. Statistics and Water Typology
2.4. Pollution Assessment
2.5. Human Health Risk Evaluation
3. Results and Discussion
3.1. Water Chemistry
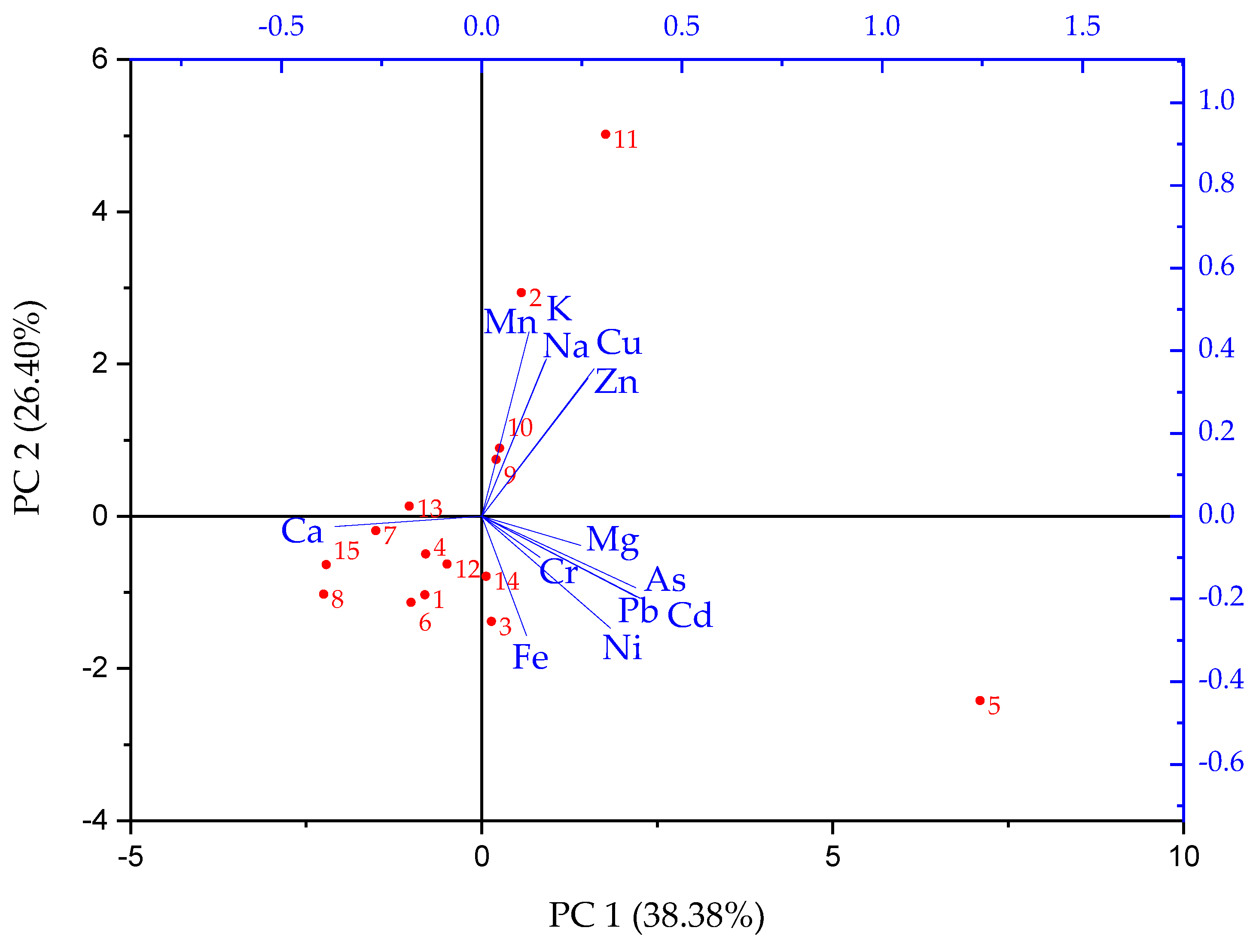
3.1.1. Metal Distribution
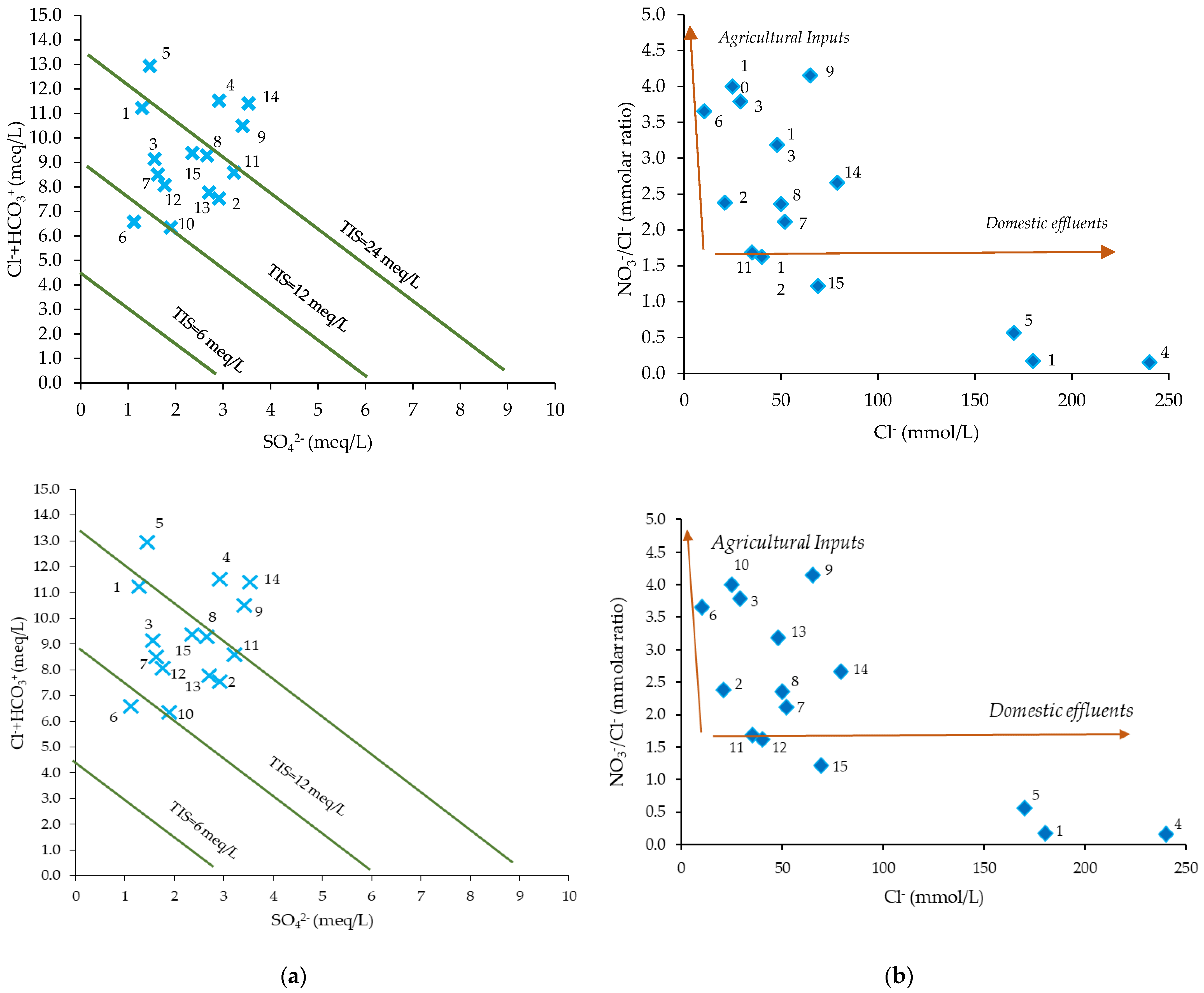
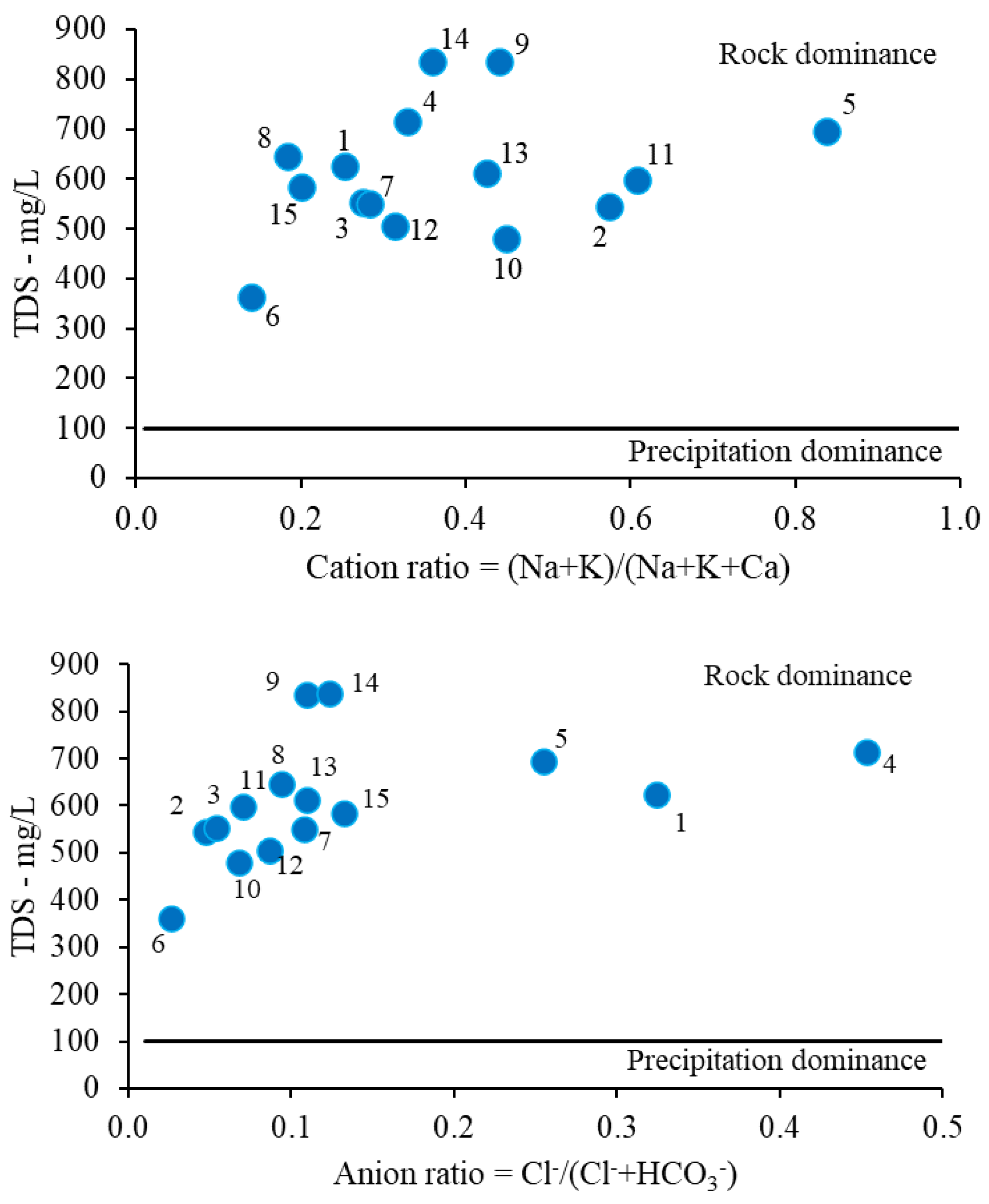
3.1.2. Physico-Chemical Characterization and Water Typology
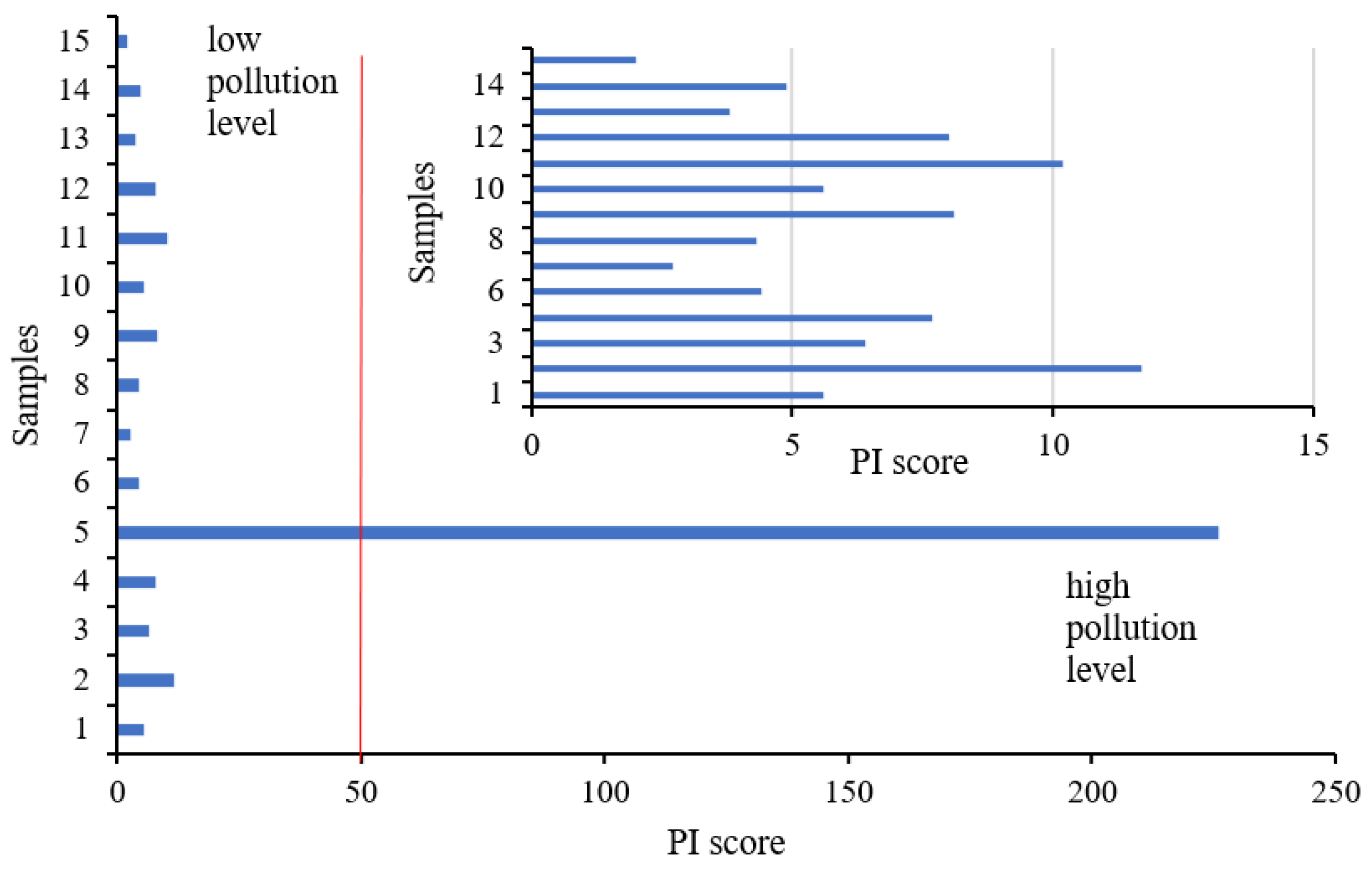
3.2. Contaminants Pollution Assessment
3.3. Exposure and Risk Assessment at Heavy Metals, and Nitrate, Nitrite
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Bouselsal, B.; Saibi, H. Evaluation of groundwater quality and hydrochemical characteristics in the shallow aquifer of El-Oued region (Algerian Sahara). Groundw. Sustain. Dev. 2022, 17, 100747. [Google Scholar] [CrossRef]
- Kom, K.P.; Gurugnanam, B.; Sunitha, V.; Reddy, Y.S.; Kadam, A.K. Hydrogeochemical assessment of groundwater quality for drinking and irrigation purposes in western Coimbatore, South India. Int. J. Energy Water Res. 2022, 6, 475–494. [Google Scholar] [CrossRef]
- Mokarram, M.; Saber, A.; Sheykhi, V. Effects of heavy metal contamination on river water quality due to release of industrial effluents. J. Clean Prod. 2020, 277, 123380. [Google Scholar] [CrossRef]
- Baskaran, P.; Abraham, M. Evaluation of groundwater quality and heavy metal pollution index of the industrial area, Chennai. Phy. Chem. Earth 2022, 128, 103259. [Google Scholar] [CrossRef]
- El-Sorogy, A.S.; Youssef, M.; Al-Hashim, M.H. Water quality assessment and environmental Impact of heavy metals in the red sea coastal seawater of Yanbu, Saudi Arabia. Water 2023, 15, 201. [Google Scholar] [CrossRef]
- Aradpour, S.; Noori, R.; Vesali Naseh, M.R.; Hosseinzadeh, M.; Safavi, S.; Ghahraman-Rozegar, F.; Maghrebi, M. Alarming carcinogenic and non-carcinogenic risk of heavy metals in Sabalan dam reservoir, Northwest of Iran. Environ. Pollut. Bioavail 2021, 33, 278–291. [Google Scholar] [CrossRef]
- Cadar, O.; Miclean, M.; Cadar, S.; Tanaselia, C.; Senila, L.; Senila, M. Assessment of heavy metals in cows milk in Rodnei mountains area, Romania. Environ. Eng. Manag. J. 2015, 14, 2523–2528. [Google Scholar] [CrossRef]
- Vardhan, K.H.; Kumar, P.S.; Panda, R.C. A review on heavy metal pollution, toxicity and remedial measures: Current trends and future perspectives. J. Mol. Liq. 2019, 290, 111197. [Google Scholar] [CrossRef]
- Sankhla, M.S.; Kumari, M.; Nandan, M.; Kumar, R.; Agrawal, P. Heavy metals contamination in water and their hazardous effect on human health-A review. Int. J. Curr. Microbiol. Appl. Sci. 2016, 5, 759–766. [Google Scholar] [CrossRef]
- Jamshaid, M.; Khan, A.A.; Ahmed, K.; Saleem, M. Heavy metal in drinking water its effect on human health and its treatment techniques–a review. Int. J. Biosci. 2018, 12, 223–240. [Google Scholar] [CrossRef]
- Abu-Dief, A.M.; Abdel-Rahman, L.H.; Abd– El Sayed, M.A.; Zikry, M.M. Green synthesis of silver nanoparticles using delonix regia extract, characterization and its application as adsorbent for removal of Cu (II) ions from aqueous solution. Asian J. Appl. Chem. Res. 2021, 9, 1–15. [Google Scholar] [CrossRef]
- Abu-Dief, A.M.; Zikry, M.M. Adsorption of the heavy metal ions onto bio sorbents: A review. Int. J. Nano Chem. 2018, 4, 27–39. [Google Scholar] [CrossRef]
- Abu-Dief, A.M.; Hamdan, S.K. Functionalization of magnetic nano particles: Synthesis, characterization and their application in water purification. Am. J. Nanosci. 2016, 2, 26–40. [Google Scholar] [CrossRef]
- Abdel-Rahman, L.H.; Abu-Dief, A.M.; Abd- El Sayed, M.A.; Zikry, M.M. Nano sized moringa oleifera an effective strategy for Pb(II) ions removal from aqueous Solution. Chem. Mat. Res. 2016, 8, 8–22. [Google Scholar]
- Karaouzas, I.; Kapetanaki, N.; Mentzafou, A.; Kanellopoulos, T.D.; Skoulikidis, N. Heavy metal contamination status in Greek surface waters: A review with application and evaluation of pollution indices. Chemosphere 2021, 263, 128192. [Google Scholar] [CrossRef]
- Bodrud-Doza, M.; Islam, A.R.M.T.; Ahmed, F.; Das, S.; Saha, N.; Rahman, M.S. Characterization of groundwater quality using water evaluation indices, multivariate statistics and geostatistics in central Bangladesh. Water Sci. 2019, 30, 19–40. [Google Scholar] [CrossRef] [Green Version]
- Alexakis, D.E. Meta-evaluation of water quality indices. Application into groundwater resources. Water 2020, 12, 1890. [Google Scholar] [CrossRef]
- Kumar, V.; Parihar, R.D.; Sharma, A.; Bakshi, P.; Singh Sidhu, G.P.; Bali, A.S.; Karaouzas, I.; Bhardwaj, R.; Thukral, A.K.; Gyasi-Agyei, Y.; et al. Global evaluation of heavy metal content in surface water bodies: A meta-analysis using heavy metal pollution indices and multivariate statistical analyses. Chemosphere 2019, 236, 124364. [Google Scholar] [CrossRef] [PubMed]
- Prasad, B.; Sangita, K. Heavy metal pollution index of ground water of an abandoned open cast mine filled with fly ash: A case study. Mine Water Environ. 2008, 27, 265–267. [Google Scholar] [CrossRef]
- Khan, R.; Saxena, A.; Shukla, S.; Sekar, S.; Senapathi, V.; Wu, J. Environmental contamination by heavy metals and associated human health risk assessment: A case study of surface water in Gomti River Basin, India. Environ. Sci. Pollut. Res. Int. 2021, 28, 56105–56116. [Google Scholar] [CrossRef]
- Muhammad, S.; Shah, M.T.; Khan, S. Health risk assessment of heavy metals and their source apportionment in drinking water of Kohistan region, northern Pakistan. Microchem. J. 2011, 98, 334–343. [Google Scholar] [CrossRef]
- Castresana, G.P.; Roldán, E.C.; García Suastegui, W.A.; Morán Perales, J.L.; Cruz Montalvo, A.; Silva, A.H. Evaluation of health risks due to heavy metals in a rural population exposed to atoyac river pollution in Puebla, Mexico. Water 2019, 11, 277. [Google Scholar] [CrossRef] [Green Version]
- Alqarawy, A.; El Osta, M.; Masoud, M.; Elsayed, S.; Gad, M. Use of hyperspectral reflectance and water quality indices to assess groundwater quality for drinking in arid regions, Saudi Arabia. Water 2022, 14, 2311. [Google Scholar] [CrossRef]
- Marculet, V.; Marculet, I. Medias. History, Demography, Personalities. Studies, Communications and Articles; Saint Nicholas Publisher: Braila, Romania, 2011. (In Romanian) [Google Scholar]
- Apollaro, C.; Buccianti, A.; Vespasiano, G.; Vardè, M.; Fuoco, I.; Barca, D.; Bloise, A.; Miriello, D.; Cofone, F.; Servidio, A.; et al. Comparative geochemical study between the tap waters and the bottled mineral waters in Calabria (Southern Italy) by compositional data analysis (CoDA) developments. Appl. Geochem. 2019, 107, 19–33. [Google Scholar] [CrossRef]
- Lyu, M.; Pang, Z.; Yin, L.; Zhang, J.; Huang, T.; Yang, S.; Li, Z.; Wang, X.; Gulbostan, T. The control of groundwater flow systems and geochemical processes on groundwater chemistry: A case study in Wushenzhao basin, NW China. Water 2019, 11, 790. [Google Scholar] [CrossRef] [Green Version]
- Gibbs, R.J. Mechanisms controlling world water chemistry. Science 1970, 170, 1088–1090. [Google Scholar] [CrossRef] [PubMed]
- Piper, A.M. A graphic procedure in the geochemical interpretation of water-analyses. Trans. Am. Geophys. Union 1944, 25, 914–928. [Google Scholar] [CrossRef]
- Law 311 from 6rd June 2004 That Improves and Complements Law 458 from 29 July 2002 Regarding the Quality of Drinking Water. Official Gazette. Part I, no. 582/30.06.2004. Available online: https://www.lege-online.ro/lr-LEGE-311%20-2004-(53106)-(1).html (accessed on 16 January 2023). (In Romanian).
- WHO. Guidelines for Drinking-Water Quality, 4th ed.; Incorporating the 1st Addendum; 2017; Available online: https://www.who.int/publications/i/item/9789241549950 (accessed on 16 January 2023).
- Bhuiyan, M.A.; Islam, M.A.; Dampare, S.B.; Parvez, L.; Suzuki, S. Evaluation of hazardous metal pollution in irrigation and drinking water systems in the vicinity of a coal mine area of northwestern Bangladesh. J. Hazard. Mater. 2010, 179, 1065–1077. [Google Scholar] [CrossRef]
- U.S. Environmental Protection Agency, EPA. Risk Assessment Guidance for Superfund Volume I: Human Health Evaluation Manual (Part E). 2004. Available online: https://www.epa.gov/sites/production/files/2015-09/documents/part_e_final_revision_10-03-07.pdf (accessed on 16 January 2023).
- Alidadi, H.; Tavakoly Sany, S.B.; Zarif Garaati Oftadeh, B.; Mohamad, T.; Shamszade, H.; Fakhari, M. Health risk assessments of arsenic and toxic heavy metal exposure in drinking water in northeast Iran. Environ. Health Prev. Med. 2019, 24, 59. [Google Scholar] [CrossRef] [Green Version]
- U.S. Environmental Protection Agency, EPA. Exposure Assessment Tools by Routes-Ingestion. 1992. Available online: https://www.epa.gov/expobox/exposure-assessment-tools-routes-ingestion (accessed on 16 January 2022).
- Ogarekpe, N.M.; Nnaji, C.C.; Oyebode, O.J.; Ekpenyong, M.G.; Ofem, O.I.; Tenebe, I.T.; Asitok, A.D. Groundwater quality index and potential human health risk assessment of heavy metals in water: A case study of Calabar metropolis, Nigeria. Environ. Nanotechnol. Monit. Manag. 2023, 19, 100780. [Google Scholar] [CrossRef]
- U.S. Environmental Protection Agency, EPA. Integrated Risk Information System, IRIS Assessments. 1987. Available online: https://iris.epa.gov/AtoZ/?list_type=alpha (accessed on 16 January 2023).
- Dippong, T.; Hoaghia, M.-A.; Mihali, C.; Cical, E.; Calugaru, M. Human health risk assessment of some bottled waters from Romania. Environ. Pollut. 2020, 267, 115409. [Google Scholar] [CrossRef] [PubMed]
- Senila, M.; Micu, D.; Hoaghia, A.; Levei, E.; Cadar, O.; Kucsicsa, G.; Balteanu, D. Multivariate statistical analysis of water chemistry in wells from Sub-Carpathians of Curvature, Romania. AES Bioflux 2017, 9, 64–71. [Google Scholar]
- Hoaghia, M.-A.; Cadar, O.; Hognogi, G.-G.; Levei, E.; Moisa, C.; Roman, C. Quality and human health risk assessment of metals and nitrogen compounds in drinking water from an urban area near a former non-ferrous ore smelter. Analyt. Lett. 2019, 52, 1268–1281. [Google Scholar] [CrossRef]
- Mahmoud, N.; Zayed, O.; Petrusevski, B. Groundwater quality of drinking water wells in the West Bank, Palestine. Water 2022, 14, 377. [Google Scholar] [CrossRef]
- Tunc, M.S.; Tepe, O.; Unlu, A.; Dursun, A.Y.; Yaman, M. Assessment of groundwater quality in Uluova, Eazig, Turkey. Fresenius Environ. Bull. 2017, 26, 5092–5102. [Google Scholar]
- Atangana, E.; Oberholster, P.J. Using heavy metal pollution indices to assess water quality of surface and groundwater on catchment levels in South Africa. J. Afr. Earth Sci. 2021, 182, 104254. [Google Scholar] [CrossRef]
- Mitra, S.; Chakraborty, A.J.; Tareq, A.M.; Emran, T.B.; Nainu, F.; Khusro, A.; Idris, A.M.; Khandaker, M.U.; Osman, H.; Alhumaydhi, F.A.; et al. Impact of heavy metals on the environment and human health: Novel therapeutic insights to counter the toxicity. J. King Saud. Univ. Sci. 2022, 34, 101865. [Google Scholar] [CrossRef]
- de Queiroz, T.K.L.; Câmara, V.d.M.; Naka, K.S.; Mendes, L.d.C.d.S.; Chagas, B.R.; de Jesus, I.M.; Meyer, A.; Lima, M.d.O. Human Health Risk Assessment Is Associated with the Consumption of Metal-Contaminated Groundwater around the Marituba Landfill, Amazonia, Brazil. Int. J. Environ. Res. Public Health 2022, 19, 13865. [Google Scholar] [CrossRef]




| pH | EC | TDS | tH | HCO3− | Cl− | SO42− | NH4+ | NO3− | NO2− | |
|---|---|---|---|---|---|---|---|---|---|---|
| Sample | μS/cm | mg/L | German Degree | mg/L | mg/L | mg/L | mg/L | mg/L | mg/L | |
| 1 | 7.44 ± 0.37 | 1250 ± 100 | 625 ± 50.0 | 29 ± 2.32 | 375 ± 30.0 | 180 ± 14.4 | 62 ± 4.96 | 0.64 ± 0.05 | 31 ± 2.48 | 1.48 ± 0.12 |
| 2 | 7.59± 0.38 | 1088 ± 87.0 | 544 ± 43.5 | 14 ± 1.12 | 423 ± 33.8 | 21 ± 1.68 | 140 ± 11.2 | 47 ± 3.76 | 50 ± 4.00 | 2.3 ± 0.18 |
| 3 | 7.33 ± 0.37 | 1105 ± 88.4 | 553 ± 44.2 | 30 ± 2.40 | 508 ± 40.6 | 29 ± 2.32 | 75 ± 6.0 | 0.7 ± 0.06 | 110 ± 8.80 | 0.012 ± 0.001 |
| 4 | 7.25 ± 0.36 | 1430 ± 114 | 715 ± 57.2 | 30 ± 2.40 | 289 ± 23.1 | 240 ± 19.2 | 140 ± 11.2 | 3 ± 0.24 | 37 ± 2.96 | 1.4 ± 0.11 |
| 5 | 7.48 ± 0.37 | 1390 ± 111 | 695 ± 55.6 | 33 ± 2.64 | 497 ± 39.8 | 170 ± 13.6 | 70 ± 5.60 | 0.35 ± 0.03 | 96 ± 7.68 | 0.03 ± 0.002 |
| 6 | 7.46 ± 0.37 | 724 ± 57.9 | 362 ± 29.0 | 21 ± 1.68 | 383 ± 30.6 | 10.4 ± 0.83 | 54 ± 4.32 | 3.4 ± 0.27 | 38 ± 3.04 | 1.38 ± 0.11 |
| 7 | 7.48 ± 0.37 | 1103 ± 88.2 | 552 ± 44.2 | 25 ± 2.00 | 429 ± 34.3 | 52 ± 4.16 | 78 ± 6.24 | 2.45 ± 0.20 | 110 ± 8.80 | 1.35 ± 0.11 |
| 8 | 7.24 ± 0.36 | 1290 ± 103 | 645 ± 51.6 | 33 ± 2.64 | 481 ± 38.5 | 50 ± 4.00 | 128 ± 10.2 | 0.12 ± 0.01 | 118 ± 9.44 | 1.7 ± 0.14 |
| 9 | 7.19 ± 0.36 | 1672 ± 134 | 836 ± 66.9 | 36 ± 2.88 | 528 ± 42.2 | 65 ± 5.20 | 164 ± 13.1 | 38 ± 3.04 | 270 ± 21.60 | 1.81 ± 0.14 |
| 10 | 7.37 ± 0.37 | 959 ± 76.7 | 480 ± 38.4 | 19 ± 1.52 | 344 ± 27.5 | 25 ± 2.00 | 91 ± 7.28 | 2.46 ± 0.20 | 100 ± 8.00 | 0.01 ± 0.001 |
| 11 | 7.64 ± 0.38 | 1195 ± 95.6 | 598 ± 47.8 | 16 ± 1.28 | 464 ± 37.1 | 35 ± 2.80 | 155 ± 12.4 | 48 ± 3.84 | 59 ± 4.72 | 0.09 ± 0.01 |
| 12 | 7.33 ± 0.37 | 1010 ± 80.8 | 505 ± 40.4 | 25 ± 2.00 | 423 ± 33.8 | 40 ± 3.20 | 85 ± 6.80 | 2.8 ± 0.22 | 65 ± 5.20 | 1.53 ± 0.12 |
| 13 | 7.67 ± 0.38 | 1224 ± 97.9 | 612 ± 49.0 | 23 ± 1.84 | 392 ± 31.4 | 48 ± 3.84 | 130 ± 10.4 | 3.79 ± 0.30 | 153 ± 12.24 | 1.02 ± 0.08 |
| 14 | 7.2 ± 0.36 | 1674 ± 134 | 837 ± 67.0 | 38 ± 3.04 | 560 ± 44.8 | 79 ± 6.32 | 170 ± 13.6 | 2.87 ± 0.23 | 210 ± 16.80 | 1.79 ± 0.14 |
| 15 | 7.41 ± 0.37 | 1189 ± 95.1 | 585 ± 46.8 | 19 ± 1.52 | 454 ± 36.3 | 69 ± 5.52 | 113 ± 9.04 | 2.2 ± 0.18 | 84 ± 6.72 | 1.56 ± 0.12 |
| min | 7.19 | 724 | 362 | 14.5 | 289 | 10.4 | 54 | 0.115 | 31 | 0.01 |
| max | 7.67 | 1674 | 837 | 38 | 560 | 240 | 170 | 48 | 270 | 2.3 |
| average | 7.40 | 1220 | 609 | 26 | 437 | 74 | 110 | 10.5 | 102 | 1.16 |
| MAC* | 6.5–9.5 | 2500 | - | <5 | - | 250 | 250 | 0.5 | 50 | 0.5 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Resz, M.-A.; Roman, C.; Senila, M.; Török, A.I.; Kovacs, E. A Comprehensive Approach to the Chemistry, Pollution Impact and Risk Assessment of Drinking Water Sources in a Former Industrialized Area of Romania. Water 2023, 15, 1180. https://doi.org/10.3390/w15061180
Resz M-A, Roman C, Senila M, Török AI, Kovacs E. A Comprehensive Approach to the Chemistry, Pollution Impact and Risk Assessment of Drinking Water Sources in a Former Industrialized Area of Romania. Water. 2023; 15(6):1180. https://doi.org/10.3390/w15061180
Chicago/Turabian StyleResz, Maria-Alexandra, Cecilia Roman, Marin Senila, Anamaria Iulia Török, and Eniko Kovacs. 2023. "A Comprehensive Approach to the Chemistry, Pollution Impact and Risk Assessment of Drinking Water Sources in a Former Industrialized Area of Romania" Water 15, no. 6: 1180. https://doi.org/10.3390/w15061180





