Odorous Substances in Urban Drainage Pipelines and the Removal Technology: A Review
Abstract
1. Introduction
2. Main Odorous Substances in Drainage Pipelines
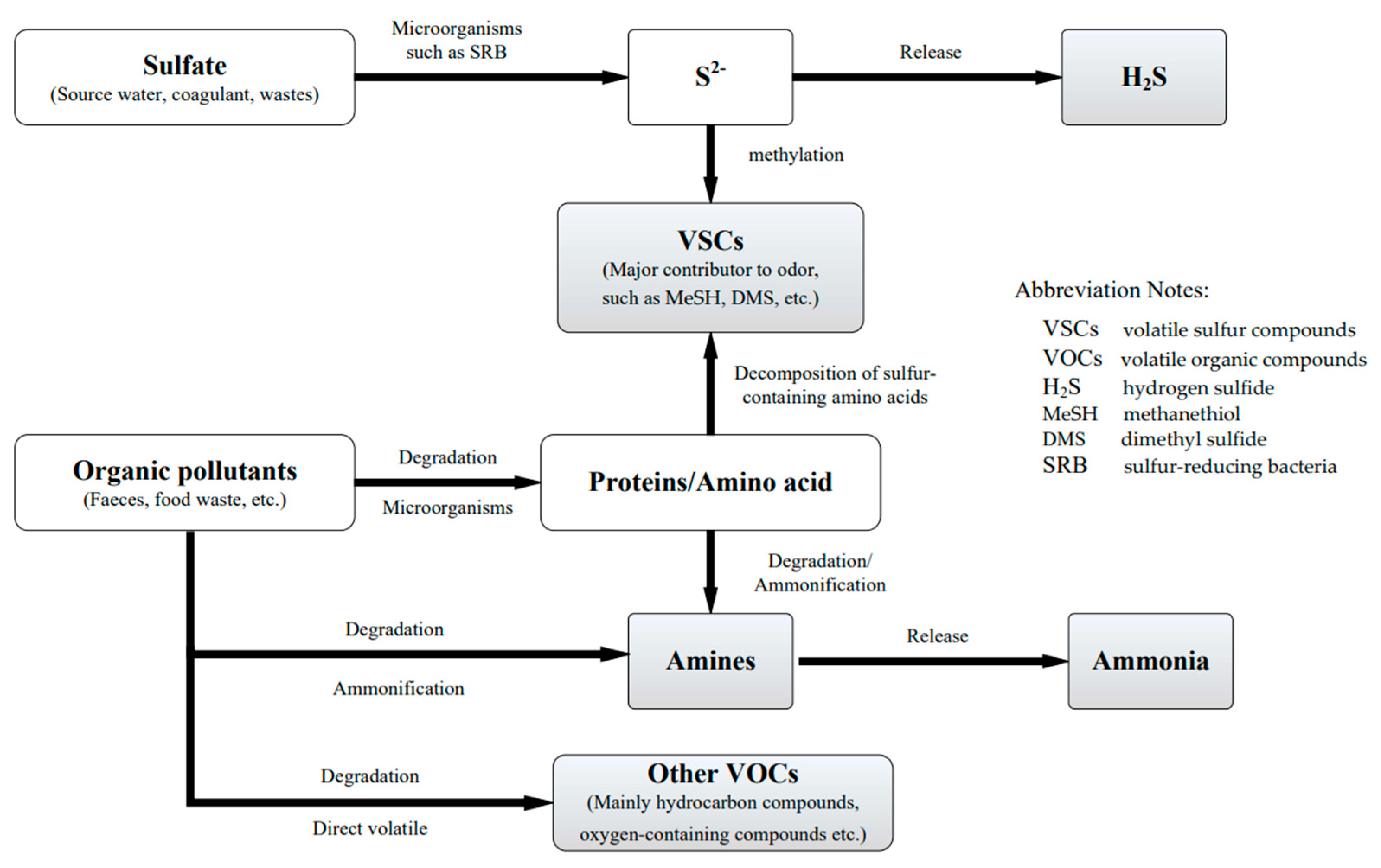
3. Sources of Odorous Substances in Drainage Pipelines
4. In Situ Deodorization Strategies of Urban Drainage Pipelines
4.1. Pipeline Condition Optimization Techniques
4.2. Odor Source Control Techniques
4.3. Chemical Control Techniques
4.3.1. Aeration Oxidation Method
4.3.2. Strong Oxidant Dosing Method
4.3.3. Iron Salt Precipitation Method
4.3.4. Biofilm Activity Inhibition Method
| Experiment Scale | Dosing Strategy | Nitrate-N Dosage Amount (mg·L−1) | Initial S2− Concentration (mg·L−1) | S2− Concentration after Dosing (mg·L−1) | Removal Rate (%) | Reference |
|---|---|---|---|---|---|---|
| Lab-scale | Intermittent | 5 | 12.2 | 8.5 | 30.2 | [98] |
| Lab-scale | Intermittent | 25 | 25 | 10 | 60 | [100] |
| Lab-scale | Intermittent | 35 | 15.5 | 0.8 | 94.8 | [98] |
| Lab-scale | Intermittent | 40 | 8 | 0 | 100 | [99] |
| Lab-scale | Persistent | 15 | 2.5 ± 1.2 | 0.2 ± 0.2 | 92 | [97] |
| Lab-scale | Persistent | 30 | 10 | 2 | 80 | [101] |
| Lab-scale | Persistent | 30 | 17.7 ± 0.8 | 0 | 100 | [94] |
| Field-scale (2.4 km) | Persistent | 10 | 4.2 | 0.2 | 95.2 | [102] |
| Field-scale (5.0 km) | Persistent | 40 | 10–20 | 2–3 | 83.3 | [103] |
| Field-scale (61 km) | Persistent | 5 | 1 | 0 | 100 | [104] |
4.4. Biological Control Techniques
4.4.1. Biological Oxidation Techniques
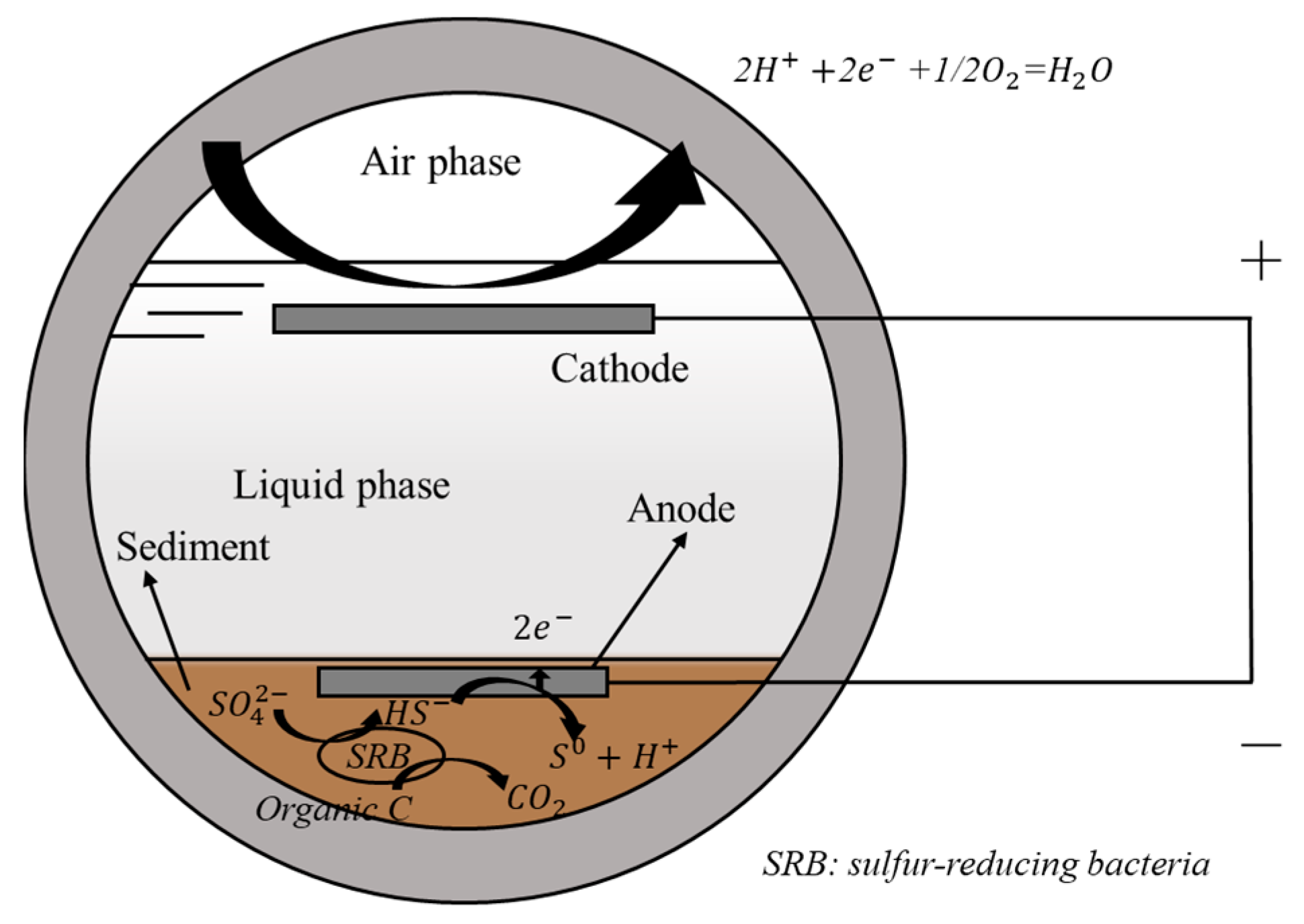
4.4.2. Bioelectrochemical Systems
5. Conclusions and Prospects
- A comprehensive mechanism for odor generation and the distribution pattern of odor along the drainage pipelines needs further research. This future research can form the theoretical basis for a real-time monitoring model, which can aid in the development of a more rational in situ odor control scheme. This includes the selection of deodorization methods, the determination of dosing points, and amounts, among others.
- Greater attention should be given to the overall effectiveness of in situ deodorization technology in removing odors. The research on odor control technology in drainage pipelines is mainly limited to the control and removal of H2S or other certain odorous substances, as well as their precursor substances. There are still research gaps regarding the effects of achieving overall odor control in drainage pipelines. An odor evaluation instrument, similar to an electronic nose, may be introduced to assess overall odor removal efficiency.
- The actual effectiveness of emerging in situ deodorization technologies needs further study through more field-scale experiments. Some of the field-scale experiments conducted on in situ deodorization technologies have yielded different results when compared with corresponding laboratory experiments. Worse still, most studies on in situ deodorization have remained at the laboratory-scale stage and have not been put into actual pipelines to prove their feasibility.
- To prevent sewer biofilm from developing resistance and reducing the effectiveness of deodorants added later, the application of deodorants should be carefully considered after monitoring their intermittent dosing effects over a long period of time. This will also help reduce the burden on WWTPs.
- The use of in situ deodorization techniques should strike a balance between improving control and duration while keeping costs low. Although some deodorization techniques, particularly chemical control methods, can be expensive, they do provide satisfactory results. One potential solution to this problem is to extract necessary chemical and biological materials from waste or use waste directly, such as iron-bearing sludge, for odor control.
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Pan, G.; Wang, B.; Guo, S.; Zhang, W.; Edwini-Bonsu, S. Statistical Analysis of Sewer Odour Based on 10-Year Complaint Data. Water Sci. Technol. 2020, 81, 1221–1230. [Google Scholar] [CrossRef]
- Vollertsen, J.; Revilla, N.; Hvitved-Jacobsen, T.; Nielsen, A.H. Modeling Sulfides, PH and Hydrogen Sulfide Gas in the Sewers of San Francisco. Water Environ. Res. 2015, 87, 1980–1989. [Google Scholar] [CrossRef]
- Kim, Y.-D.; Kwon, W.-T. Development of Complex Module Device for Odor Reduction in Sewage. J. Wellbeing Manag. Appl. Psychol. 2021, 4, 1–9. [Google Scholar] [CrossRef]
- Nielsen, P.H.; Raunkjær, K.; Hvitved-Jacobsen, T. Sulfide Production and Wastewater Quality in Pressure Mains. Water Sci. Technol. 1998, 37, 97–104. [Google Scholar] [CrossRef]
- Giri, B.; Mudliar, S.; Deshmukh, S.; Banerjee, S.; Pandey, R. Treatment of Waste Gas Containing Low Concentration of Dimethyl Sulphide (DMS) in a Bench-Scale Biofilter. Bioresour. Technol. 2010, 101, 2185–2190. [Google Scholar] [CrossRef] [PubMed]
- Fatima, T.; Muntean, A. Sulfate Attack in Sewer Pipes: Derivation of a Concrete Corrosion Model via Two-Scale Convergence. Nonlinear Anal. Real World Appl. 2014, 15, 326–344. [Google Scholar] [CrossRef]
- Li, W.; Zheng, T.; Ma, Y.; Liu, J. Current Status and Future Prospects of Sewer Biofilms: Their Structure, Influencing Factors, and Substance Transformations. Sci. Total Environ. 2019, 695, 133815. [Google Scholar] [CrossRef]
- Hvitved-Jacobsen, T.; Vollertsen, J.; Matos, J.S. The Sewer as a Bioreactor–a Dry Weather Approach. Water Sci. Technol. 2002, 45, 11–24. [Google Scholar] [CrossRef]
- Rauch, W.; Kleidorfer, M. Replace Contamination, Not the Pipes. Science 2014, 345, 734–735. [Google Scholar] [CrossRef]
- Vincke, E. Biogenic Sulfuric Acid Corrosion of Concrete: Microbial Interaction, Simulation and Prevention. Ph.D. Thesis, Ghent University, Ghent, Belgium, 2002. [Google Scholar]
- Anwar, A.; Liu, X.; Zhang, L. Biogenic Corrosion of Cementitious Composite in Wastewater Sewerage System–A Review. Process Saf. Environ. Prot. 2022, 165, 545–585. [Google Scholar] [CrossRef]
- Zhang, L.; De Schryver, P.; De Gusseme, B.; De Muynck, W.; Boon, N.; Verstraete, W. Chemical and Biological Technologies for Hydrogen Sulfide Emission Control in Sewer Systems: A Review. Water Res. 2008, 42, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Talaiekhozani, A.; Bagheri, M.; Goli, A.; Talaei Khoozani, M.R. An Overview of Principles of Odor Production, Emission, and Control Methods in Wastewater Collection and Treatment Systems. J. Environ. Manag. 2016, 170, 186–206. [Google Scholar] [CrossRef] [PubMed]
- Shammay, A.; Sivret, E.C.; Le-Minh, N.; Lebrero Fernandez, R.; Evanson, I.; Stuetz, R.M. Review of Odour Abatement in Sewer Networks. J. Environ. Chem. Eng. 2016, 4, 3866–3881. [Google Scholar] [CrossRef]
- Shammay, A.; Evanson, I.E.J.; Stuetz, R.M. Selection Framework for the Treatment of Sewer Network Emissions. J. Environ. Manag. 2019, 249, 109305. [Google Scholar] [CrossRef] [PubMed]
- Sivret, E.C.; Wang, B.; Parcsi, G.; Stuetz, R.M. Prioritisation of Odorants Emitted from Sewers Using Odour Activity Values. Water Res. 2016, 88, 308–321. [Google Scholar] [CrossRef]
- Yang, Q.; Li, Y.; Cui, B.; Yang, Z.; Liu, Z.; Peng, Y. Research Advances of Odor Released from Municipal Wastewater Treatment Process. Acta Sci. Circumstantiae 2019, 39, 2079–2087. [Google Scholar] [CrossRef]
- Jiang, G.; Melder, D.; Keller, J.; Yuan, Z. Odor Emissions from Domestic Wastewater: A Review. Crit. Rev. Environ. Sci. Technol. 2017, 47, 1581–1611. [Google Scholar] [CrossRef]
- Ryltseva, Y.; Orlov, V. Measures to Prevent Sewerage Odor Emissions into the Atmosphere. IOP Conf. Ser. Mater. Sci. Eng. 2020, 869, 042002. [Google Scholar] [CrossRef]
- Pandey, S.K.; Kim, K.-H.; Kwon, E.E.; Kim, Y.-H. Hazardous and Odorous Pollutants Released from Sewer Manholes and Stormwater Catch Basins in Urban Areas. Environ. Res. 2016, 146, 235–244. [Google Scholar] [CrossRef]
- Wang, B.; Sivret, E.; Parcsi, G.; Le, N.; Kenny, S.; Bustamante, H.; Stuetz, R. Reduced Sulfur Compounds in the Atmosphere of Sewer Networks in Australia: Geographic (and Seasonal) Variations. Water Sci. Technol. 2014, 69, 1167–1173. [Google Scholar] [CrossRef]
- Choi, I.; Lee, H.; Shin, J.; Kim, H. Evaluation of the Effectiveness of Five Odor Reducing Agents for Sewer System Odors Using an On-Line Total Reduced Sulfur Analyzer. Sensors 2012, 12, 16892–16906. [Google Scholar] [CrossRef] [PubMed]
- An, S.-S.; Jang, J.-W.; Cho, M.-C.; Kim, M.-J.; Kim, H.-G.; Park, S.-I.; Bae, S.-J.; Seo, G. The Characteristics of Odor Substances in Sewer Pipes in Urban Areas. J. Odor Indoor Environ. Vol. 2020, 19, 157–165. [Google Scholar] [CrossRef]
- Smit, L.A.M.; Spaan, S.; Heederik, D. Endotoxin Exposure and Symptoms in Wastewater Treatment Workers. Am. J. Ind. Med. 2005, 48, 30–39. [Google Scholar] [CrossRef] [PubMed]
- Pitiriciu, M.; Tansel, B. Volatile Organic Contaminants (VOCs) Emitted from Sewer Networks during Wastewater Collection and Transport. J. Environ. Manag. 2021, 285, 112136. [Google Scholar] [CrossRef]
- Decottignies, V.; Huyard, A.; Kelly, R.; Barillon, B. Development of a Diagnostic Tool: The Wastewater Collection Network Odour Wheel. Water Sci. Technol. 2013, 68, 839–847. [Google Scholar] [CrossRef] [PubMed]
- Pandey, S.K.; Kim, K.-H.; Tang, K.-T. A Review of Sensor-Based Methods for Monitoring Hydrogen Sulfide. TrAC Trends Anal. Chem. 2012, 32, 87–99. [Google Scholar] [CrossRef]
- Higgins, M.J.; Yarosz, D.P.; Chen, Y.-C.; Murthy, S.N.; Mass, N.; Cooney, J. Mechanisms of Volatile Sulfur Compound and Odor Production in Digested Biosolids. Proc. Water Environ. Fed. 2003, 2003, 993–1006. [Google Scholar] [CrossRef]
- Dobrynkin, N.; Batygina, M.; Noskov, A. Water Depollution and the Odor Control by Wet Air Catalytic Oxidation of Ammonia, Sulfides and Mercaptans of Industrial Wastewaters. Chem. Eng. Trans. 2010, 23, 339–344. [Google Scholar] [CrossRef]
- Pikaar, I.; Sharma, K.R.; Hu, S.; Gernjak, W.; Keller, J.; Yuan, Z. Reducing Sewer Corrosion through Integrated Urban Water Management. Science 2014, 345, 812–814. [Google Scholar] [CrossRef]
- Fittschen, I.; Hahn, H.H. Characterization of the Municipal Wastewaterpart Human Urine and a Preliminary Comparison with Liquid Cattle Excretion. Water Sci. Technol. 1998, 38, 9–16. [Google Scholar] [CrossRef]
- Liu, C.; Yang, Y.; Zhou, J.; Chen, Y.; Zhou, J.; Wang, Y.; Fu, D. Migration and Transformation of Nitrogen in Sediment–Water System within Storm Sewers. J. Environ. Manag. 2021, 287, 112355. [Google Scholar] [CrossRef] [PubMed]
- Cao, J.; Sun, Q.; Zhao, D.; Xu, M.; Shen, Q.; Wang, D.; Wang, Y.; Ding, S. A Critical Review of the Appearance of Black-Odorous Waterbodies in China and Treatment Methods. J. Hazard. Mater. 2020, 385, 121511. [Google Scholar] [CrossRef] [PubMed]
- Ren, B.; Zhao, Y.; Lyczko, N.; Nzihou, A. Current Status and Outlook of Odor Removal Technologies in Wastewater Treatment Plant. Waste Biomass Valorization 2019, 10, 1443–1458. [Google Scholar] [CrossRef]
- Zuo, Z.; Zheng, M.; Chang, J.; Ren, D.; Huang, X.; Yuan, Z.; Liu, Y. Free Nitrous Acid-Based Suppression of Sulfide Production in Sewer Sediments: In-Situ Effect Mechanism. Sci. Total Environ. 2020, 715, 136871. [Google Scholar] [CrossRef] [PubMed]
- Pisano, W.C.; O’Riordan, O.C.; Ayotte, F.J.; Barsanti, J.R.; Carr, D.L. Automated Sewer and Drainage Flushing Systems in Cambridge, Massachusetts. J. Hydraul. Eng. 2003, 129, 260–266. [Google Scholar] [CrossRef]
- Fytianos, G.; Tziolas, E.; Papastergiadis, E.; Samaras, P. Least Cost Analysis for Biocorrosion Mitigation Strategies in Concrete Sewers. Sustainability 2020, 12, 4578. [Google Scholar] [CrossRef]
- Ren, D.; Zuo, Z.; Xing, Y.; Ji, P.; Yu, T.; Zhu, D.; Liu, Y.; Huang, X. Simultaneous Control of Sulfide and Methane in Sewers Achieved by a Physical Approach Targeting Dominant Active Zone in Sediments. Water Res. 2022, 211, 118010. [Google Scholar] [CrossRef]
- Lu, J.; Zhou, Y.; Ding, Y.; Zheng, C.; Liao, B.; Song, G.; Chen, J. Release of Harmful Gases in Sewage Collection and Transportation Pipeline System and Its Countermeasures. Chin. J. Environ. Eng. 2019, 13, 757–764. [Google Scholar] [CrossRef]
- Orlov, V.; Zotkin, S. Formation of Harmful Volatile Substances in Sewage Systems and Their Neutralization. E3S Web Conf. 2021, 263, 04003. [Google Scholar] [CrossRef]
- Cyna, B.; Chagneau, G.; Bablon, G.; Tanghe, N. Two Years of Nanofiltration at the Méry-Sur-Oise Plant, France. Desalination 2002, 147, 69–75. [Google Scholar] [CrossRef]
- Liu, Z.; Zhou, L.; Liu, F.; Gao, M.; Wang, J.; Zhang, A.; Liu, Y. Impact of Al-Based Coagulants on the Formation of Aerobic Granules: Comparison between Poly Aluminum Chloride (PAC) and Aluminum Sulfate (AS). Sci. Total Environ. 2019, 685, 74–84. [Google Scholar] [CrossRef] [PubMed]
- Zan, F.; Tang, W.; Jiang, F.; Chen, G. Diversion of Food Waste into the Sulfate-Laden Sewer: Interaction and Electron Flow of Sulfidogenesis and Methanogenesis. Water Res. 2021, 202, 117437. [Google Scholar] [CrossRef] [PubMed]
- Neczaj, E.; Grosser, A. Circular Economy in Wastewater Treatment Plant–Challenges and Barriers. Proceedings 2018, 2, 614. [Google Scholar] [CrossRef]
- Jiang, F.; Chen, Y.; Mackey, H.R.; Chen, G.H.; van Loosdrecht, M.C.M. Urine Nitrification and Sewer Discharge to Realize In-Sewer Denitrification to Simplify Sewage Treatment in Hong Kong. Water Sci. Technol. 2011, 64, 618–626. [Google Scholar] [CrossRef]
- Christiaens, M.E.; De Vrieze, J.; Clinckemaillie, L.; Ganigué, R.; Rabaey, K. Anaerobic Ureolysis of Source-Separated Urine for NH3 Recovery Enables Direct Removal of Divalent Ions at the Toilet. Water Res. 2019, 148, 97–105. [Google Scholar] [CrossRef]
- Chen, C.; Zhang, R.-C.; Xu, X.-J.; Fang, N.; Wang, A.-J.; Ren, N.-Q.; Lee, D.-J. Enhanced Performance of Denitrifying Sulfide Removal Process at High Carbon to Nitrogen Ratios under Micro-Aerobic Condition. Bioresour. Technol. 2017, 232, 417–422. [Google Scholar] [CrossRef]
- Gutierrez, O.; Mohanakrishnan, J.; Sharma, K.R.; Meyer, R.L.; Keller, J.; Yuan, Z. Evaluation of Oxygen Injection as a Means of Controlling Sulfide Production in a Sewer System. Water Res. 2008, 42, 4549–4561. [Google Scholar] [CrossRef]
- Park, K.; Lee, H.; Phelan, S.; Liyanaarachchi, S.; Marleni, N.; Navaratna, D.; Jegatheesan, V.; Shu, L. Mitigation Strategies of Hydrogen Sulphide Emission in Sewer Networks—A Review. Int. Biodeterior. Biodegrad. 2014, 95, 251–261. [Google Scholar] [CrossRef]
- Orlov, V.; Zotkin, S. Developing Physical and Mathematical Models of the Water-Air Mode of Gravity Drainage Network. IOP Conf. Ser. Mater. Sci. Eng. 2021, 1015, 012071. [Google Scholar] [CrossRef]
- Zhang, M.; Qiu, L.; Liu, G. Basic Characteristics and Application of Micro-Nano Bubbles in Water Treatment. IOP Conf. Ser. Earth Environ. Sci. 2020, 510, 042050. [Google Scholar] [CrossRef]
- Xiao, W.; Xu, G. Mass Transfer of Nanobubble Aeration and Its Effect on Biofilm Growth: Microbial Activity and Structural Properties. Sci. Total Environ. 2020, 703, 134976. [Google Scholar] [CrossRef] [PubMed]
- Wei, J.; Jiang, Y.; Li, J.; Kang, H.; Liu, S. Characteristics of Micro-Nano Bubble and Its Application in Environmental Treatment. Environ. Eng. 2018, 36, 75–79. [Google Scholar] [CrossRef]
- Chen, B.; Zhou, S.; Zhang, N.; Liang, H.; Sun, L.; Zhao, X.; Guo, J.; Lu, H. Micro and Nano Bubbles Promoted Biofilm Formation with Strengthen of COD and TN Removal Synchronously in a Blackened and Odorous Water. Sci. Total Environ. 2022, 837, 155578. [Google Scholar] [CrossRef] [PubMed]
- Wu, Y.; Lin, H.; Yin, W.; Shao, S.; Lv, S.; Hu, Y. Water Quality and Microbial Community Changes in an Urban River after Micro-Nano Bubble Technology in Situ Treatment. Water 2019, 11, 66. [Google Scholar] [CrossRef]
- Wang, H.; Zhang, L. Research on the Nitrogen Removal Efficiency and Mechanism of Deep Subsurface Wastewater Infiltration Systems by Fine Bubble Aeration. Ecol. Eng. 2017, 107, 33–40. [Google Scholar] [CrossRef]
- Zhang, Z.; Chang, N.; Wang, S.; Lu, J.; Li, K.; Zheng, C. Enhancing Sulfide Mitigation via the Sustainable Supply of Oxygen from Air-Nanobubbles in Gravity Sewers. Sci. Total Environ. 2022, 808, 152203. [Google Scholar] [CrossRef]
- Guo, K.; Wu, Z.; Chen, C.; Fang, J. UV/Chlorine Process: An Efficient Advanced Oxidation Process with Multiple Radicals and Functions in Water Treatment. Acc. Chem. Res. 2022, 55, 286–297. [Google Scholar] [CrossRef]
- Ksibi, M. Chemical Oxidation with Hydrogen Peroxide for Domestic Wastewater Treatment. Chem. Eng. J. 2006, 119, 161–165. [Google Scholar] [CrossRef]
- Loeb, B.L.; Thompson, C.M.; Drago, J.; Takahara, H.; Baig, S. Worldwide Ozone Capacity for Treatment of Drinking Water and Wastewater: A Review. Ozone Sci. Eng. 2012, 34, 64–77. [Google Scholar] [CrossRef]
- Li, J.; Sharma, K.; Liu, Y.; Jiang, G.; Yuan, Z. Real-Time Prediction of Rain-Impacted Sewage Flow for on-Line Control of Chemical Dosing in Sewers. Water Res. 2019, 149, 311–321. [Google Scholar] [CrossRef]
- Li, J.; Li, W.; Chang, X.; Sharma, K.; Yuan, Z. Real-Time Predictive Control for Chemical Distribution in Sewer Networks Using Improved Elephant Herding Optimization. IEEE Trans. Ind. Inform. 2022, 18, 571–581. [Google Scholar] [CrossRef]
- Zhang, J.; Chen, Y.; Liao, Y.; Wang, Q.; Yu, J. Studies on the Degradation of Trace Phenol and Indole Odorants by Chlorine and Permanganate in Drinking Water Treatment. Chemosphere 2022, 286, 131551. [Google Scholar] [CrossRef] [PubMed]
- Wu, C.-Y.; Chou, M.-S.; Lin, J.-H. Oxidative Scrubbing of DMS-Containing Waste Gases by Hypochlorite Solution. J. Taiwan Inst. Chem. Eng. 2014, 45, 596–602. [Google Scholar] [CrossRef]
- Butt, M.T.; Imtiaz, N.; Abbas, N.; Khan, R.A. Sulphide Removal from Sewage Wastewater by Oxidation Technique. Pak. J. Sci. Ind. Res. Ser. Phys. Sci. 2017, 60, 42–49. [Google Scholar] [CrossRef]
- El Brahmi, A.; Abderafi, S. Hydrogen Sulfide Removal from Wastewater Using Hydrogen Peroxide In-Situ Treatment: Case Study of Moroccan Urban Sewers. Mater. Today Proc. 2021, 45, 7424–7427. [Google Scholar] [CrossRef]
- El Brahmi, A.; Abderafi, S. Performance of Hydrogen Peroxide 35% Treatment for Sulfide Mitigation in Sanitary Sewers: Sewage Characterization and Response Surface Methodology. Int. J. Environ. Sci. Technol. 2022, 20, 2127–2140. [Google Scholar] [CrossRef]
- Rostami, A.; Akradi, J. A Highly Efficient, Green, Rapid, and Chemoselective Oxidation of Sulfides Using Hydrogen Peroxide and Boric Acid as the Catalyst under Solvent-Free Conditions. Tetrahedron Lett. 2010, 51, 3501–3503. [Google Scholar] [CrossRef]
- Feng, D.; Shou, J.; Guo, S.; Ya, M.; Li, J.; Dong, H.; Li, Y. Co-Catalysis of Trace Dissolved Fe(Iii) with Biochar in Hydrogen Peroxide Activation for Enhanced Oxidation of Pollutants. RSC Adv. 2022, 12, 17237–17248. [Google Scholar] [CrossRef]
- Hu, W.; Yang, L.; Shao, P.; Shi, H.; Chang, Z.; Fang, D.; Wei, Y.; Feng, Y.; Huang, Y.; Yu, K.; et al. Proton Self-Enhanced Hydroxyl-Enriched Cerium Oxide for Effective Arsenic Extraction from Strongly Acidic Wastewater. Environ. Sci. Technol. 2022, 56, 10412–10422. [Google Scholar] [CrossRef]
- Yang, L.; Feng, Y.; Wang, C.; Fang, D.; Yi, G.; Gao, Z.; Shao, P.; Liu, C.; Luo, X.; Luo, S. Closed-Loop Regeneration of Battery-Grade FePO4 from Lithium Extraction Slag of Spent Li-Ion Batteries via Phosphoric Acid Mixture Selective Leaching. Chem. Eng. J. 2022, 431, 133232. [Google Scholar] [CrossRef]
- Jiang, J.-Q.; Lloyd, B. Progress in the Development and Use of Ferrate(VI) Salt as an Oxidant and Coagulant for Water and Wastewater Treatment. Water Res. 2002, 36, 1397–1408. [Google Scholar] [CrossRef] [PubMed]
- Lee, Y.; Yoon, J.; Von Gunten, U. Kinetics of the Oxidation of Phenols and Phenolic Endocrine Disruptors during Water Treatment with Ferrate (Fe (VI)). Environ. Sci. Technol. 2005, 39, 8978–8984. [Google Scholar] [CrossRef] [PubMed]
- Sharma, V.K.; Luther, G.W.; Millero, F.J. Mechanisms of Oxidation of Organosulfur Compounds by Ferrate(VI). Chemosphere 2011, 82, 1083–1089. [Google Scholar] [CrossRef] [PubMed]
- Sharma, V.K. Oxidation of Inorganic Contaminants by Ferrates (VI, V, and IV)–Kinetics and Mechanisms: A Review. J. Environ. Manag. 2011, 92, 1051–1073. [Google Scholar] [CrossRef]
- Al-Abduly, A.; Sharma, V.K. Oxidation of Benzothiophene, Dibenzothiophene, and Methyl-Dibenzothiophene by Ferrate (VI). J. Hazard. Mater. 2014, 279, 296–301. [Google Scholar] [CrossRef]
- Chen, J.; Qi, Y.; Pan, X.; Wu, N.; Zuo, J.; Li, C.; Qu, R.; Wang, Z.; Chen, Z. Mechanistic Insights into the Reactivity of Ferrate(VI) with Phenolic Compounds and the Formation of Coupling Products. Water Res. 2019, 158, 338–349. [Google Scholar] [CrossRef]
- Chen, J.; Xu, X.; Zeng, X.; Feng, M.; Qu, R.; Wang, Z.; Nesnas, N.; Sharma, V.K. Ferrate (VI) Oxidation of Polychlorinated Diphenyl Sulfides: Kinetics, Degradation, and Oxidized Products. Water Res. 2018, 143, 1–9. [Google Scholar] [CrossRef]
- Alibabaei, F.; Saebnoori, E.; Fulazzaky, M.A.; Talaeikhozani, A.; Roohi, P.; Moghadas, F.; Abdullah, N.H.; Alian, T. An Evaluation of the Efficiency of Odorant Removal by Sodium Ferrate(VI) Oxidation. Measurement 2021, 179, 109488. [Google Scholar] [CrossRef]
- Rebosura Jr, M.; Salehin, S.; Pikaar, I.; Sun, X.; Keller, J.; Sharma, K.; Yuan, Z. A Comprehensive Laboratory Assessment of the Effects of Sewer-Dosed Iron Salts on Wastewater Treatment Processes. Water Res. 2018, 146, 109–117. [Google Scholar] [CrossRef]
- Zhang, L.; Keller, J.; Yuan, Z. Inhibition of Sulfate-Reducing and Methanogenic Activities of Anaerobic Sewer Biofilms by Ferric Iron Dosing. Water Res. 2009, 43, 4123–4132. [Google Scholar] [CrossRef]
- Yang, Y.; Guo, J.; Hu, Z. Impact of Nano Zero Valent Iron (NZVI) on Methanogenic Activity and Population Dynamics in Anaerobic Digestion. Water Res. 2013, 47, 6790–6800. [Google Scholar] [CrossRef] [PubMed]
- Sun, J.; Wei, L.; Yin, R.; Jiang, F.; Shang, C. Microbial Iron Reduction Enhances In-Situ Control of Biogenic Hydrogen Sulfide by FeOOH Granules in Sediments of Polluted Urban Waters. Water Res. 2020, 171, 115453. [Google Scholar] [CrossRef] [PubMed]
- Yang, Z.; Zhu, D.Z.; Yu, T.; Shypanski, A.; Zhang, G.; Zhou, Y. Effect of Ferric Iron and Nitrate on Hydrogen Sulfide Control in Lab-Scale Reactors. Environ. Sci. Water Res. Technol. 2021, 7, 1806–1818. [Google Scholar] [CrossRef]
- Cao, J.; Zhang, L.; Hong, J.; Sun, J.; Jiang, F. Different Ferric Dosing Strategies Could Result in Different Control Mechanisms of Sulfide and Methane Production in Sediments of Gravity Sewers. Water Res. 2019, 164, 114914. [Google Scholar] [CrossRef]
- Zhang, L.; Keller, J.; Yuan, Z. Ferrous Salt Demand for Sulfide Control in Rising Main Sewers: Tests on a Laboratory-Scale Sewer System. J. Environ. Eng. 2010, 136, 1180–1187. [Google Scholar] [CrossRef]
- Gu, T.; Tan, P.; Zhou, Y.; Zhang, Y.; Zhu, D.; Zhang, T. Characteristics and Mechanism of Dimethyl Trisulfide Formation during Sulfide Control in Sewer by Adding Various Oxidants. Sci. Total Environ. 2019, 673, 719–725. [Google Scholar] [CrossRef] [PubMed]
- Kulandaivelu, J.; Gao, J.; Song, Y.; Shrestha, S.; Li, X.; Li, J.; Doederer, K.; Keller, J.; Yuan, Z.; Mueller, J.F.; et al. Removal of Pharmaceuticals and Illicit Drugs from Wastewater Due to Ferric Dosing in Sewers. Environ. Sci. Technol. 2019, 53, 6245–6254. [Google Scholar] [CrossRef]
- Rathnayake, D.; Bal Krishna, K.C.; Kastl, G.; Sathasivan, A. The Role of PH on Sewer Corrosion Processes and Control Methods: A Review. Sci. Total Environ. 2021, 782, 146616. [Google Scholar] [CrossRef]
- Sun, J.; Pikaar, I.; Sharma, K.R.; Keller, J.; Yuan, Z. Feasibility of Sulfide Control in Sewers by Reuse of Iron Rich Drinking Water Treatment Sludge. Water Res. 2015, 71, 150–159. [Google Scholar] [CrossRef]
- Cipriani, M.; Schiavi, R.; Santos, F. Odour Control: A Successful Experience in Sorocaba City—Brazil. Water Pract. Technol. 2018, 13, 125–133. [Google Scholar] [CrossRef]
- Li, C.-Y.; Zhang, D.; Li, X.-X.; Mbadinga, S.M.; Yang, S.-Z.; Liu, J.-F.; Gu, J.-D.; Mu, B.-Z. The Biofilm Property and Its Correlationship with High-Molecular-Weight Polyacrylamide Degradation in a Water Injection Pipeline of Daqing Oilfield. J. Hazard. Mater. 2016, 304, 388–399. [Google Scholar] [CrossRef] [PubMed]
- Okabe, S.; Ito, T.; Satoh, H.; Watanabe, Y. Effect of Nitrite and Nitrate on Biogenic Sulfide Production in Sewer Biofilms Determined by the Use of Microelectrodes. Water Sci. Technol. 2003, 47, 281–288. [Google Scholar] [CrossRef] [PubMed]
- Jiang, G.; Sharma, K.R.; Yuan, Z. Effects of Nitrate Dosing on Methanogenic Activity in a Sulfide-Producing Sewer Biofilm Reactor. Water Res. 2013, 47, 1783–1792. [Google Scholar] [CrossRef] [PubMed]
- Auguet, O.; Pijuan, M.; Guasch-Balcells, H.; Borrego, C.M.; Gutierrez, O. Implications of Downstream Nitrate Dosage in Anaerobic Sewers to Control Sulfide and Methane Emissions. Water Res. 2015, 68, 522–532. [Google Scholar] [CrossRef] [PubMed]
- Zhang, G.; Gu, T.; Zhou, Y.; Zhu, D.Z.; Zhang, Y. Effects of Intermittent Chemical Dosing on Volatile Sulfur Compounds in Sewer Headspace. Environ. Eng. Res. 2021, 27, 210091. [Google Scholar] [CrossRef]
- Liu, Y.; Sharma, K.R.; Ni, B.-J.; Fan, L.; Murthy, S.; Tyson, G.Q.; Yuan, Z. Effects of Nitrate Dosing on Sulfidogenic and Methanogenic Activities in Sewer Sediment. Water Res. 2015, 74, 155–165. [Google Scholar] [CrossRef]
- Liang, Z.; Wu, D.; Li, G.; Sun, J.; Jiang, F.; Li, Y. Experimental and Modeling Investigations on the Unexpected Hydrogen Sulfide Rebound in a Sewer Receiving Nitrate Addition: Mechanism and Solution. J. Environ. Sci. 2023, 125, 630–640. [Google Scholar] [CrossRef]
- Liang, S.; Zhang, L.; Jiang, F. Indirect Sulfur Reduction via Polysulfide Contributes to Serious Odor Problem in a Sewer Receiving Nitrate Dosage. Water Res. 2016, 100, 421–428. [Google Scholar] [CrossRef]
- Liu, Y.; Wu, C.; Zhou, X.; Zhu, D.Z.; Shi, H. Sulfide Elimination by Intermittent Nitrate Dosing in Sewer Sediments. J. Environ. Sci. 2015, 27, 259–265. [Google Scholar] [CrossRef]
- Jiang, G.; Sharma, K.R.; Guisasola, A.; Keller, J.; Yuan, Z. Sulfur Transformation in Rising Main Sewers Receiving Nitrate Dosage. Water Res. 2009, 43, 4430–4440. [Google Scholar] [CrossRef]
- Bentzen, G.; Smit, A.; Bennett, D.; Webster, N.; Reinholt, F.; Sletholt, E.; Hobsont, J. Controlled Dosing of Nitrate for Prevention of H2S in a Sewer Network and the Effects on the Subsequent Treatment Processes. Water Sci. Technol. 1995, 31, 293–302. [Google Scholar] [CrossRef]
- Saracevic, E.; Bertrán de Lis, F.; Matsché, N. Odour and Corrosion Problems in Pressure Sewers. Water Pract. Technol. 2007, 2, wpt2007028. [Google Scholar] [CrossRef]
- Rodríguez-Gómez, L.E.; Delgado, S.; Álvarez, M.; Elmaleh, S. Inhibition of Sulfide Generation in a Reclaimed Wastewater Pipe by Nitrate Dosage and Denitrification Kinetics. Water Environ. Res. 2005, 77, 193–198. [Google Scholar] [CrossRef]
- Apgar, P.E.D.; Witherspoon, J. Minimization of Odors and Corrosion in Collection Systems; IWA Publishing: London, UK, 2008; ISBN 978-1-78040-372-4. [Google Scholar]
- Gutierrez, O.; Park, D.; Sharma, K.R.; Yuan, Z. Effects of Long-Term PH Elevation on the Sulfate-Reducing and Methanogenic Activities of Anaerobic Sewer Biofilms. Water Res. 2009, 43, 2549–2557. [Google Scholar] [CrossRef] [PubMed]
- Jiang, G.; Keating, A.; Corrie, S.; O’halloran, K.; Nguyen, L.; Yuan, Z. Dosing Free Nitrous Acid for Sulfide Control in Sewers: Results of Field Trials in Australia. Water Res. 2013, 47, 4331–4339. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Zhou, X.; Shi, H. Sulfur Cycle by In Situ Analysis in the Sediment Biofilm of a Sewer System. J. Environ. Eng. 2016, 142, C4015011. [Google Scholar] [CrossRef]
- Zhang, G.; Yang, Z.; Zhou, Y.; Zhu, D.Z.; Zhang, Y.; Yu, T.; Shypanski, A. Combination of Nitrate and Sodium Nitroprusside Dosing for Sulfide Control with Low Carbon Source Loss in Sewer Biofilm Reactors. J. Hazard. Mater. 2022, 424, 127527. [Google Scholar] [CrossRef]
- Yan, X.; Sun, J.; Kenjiahan, A.; Dai, X.; Ni, B.-J.; Yuan, Z. Rapid and Strong Biocidal Effect of Ferrate on Sulfidogenic and Methanogenic Sewer Biofilms. Water Res. 2020, 169, 115208. [Google Scholar] [CrossRef]
- Watsuntorn, W.; Ruangchainikom, C.; Rene, E.R.; Lens, P.N.; Chulalaksananukul, W. Comparison of Sulphide and Nitrate Removal from Synthetic Wastewater by Pure and Mixed Cultures of Nitrate-Reducing, Sulphide-Oxidizing Bacteria. Bioresour. Technol. 2019, 272, 40–47. [Google Scholar] [CrossRef]
- Kodama, Y.; Watanabe, K. Isolation and Characterization of a Sulfur-Oxidizing Chemolithotroph Growing on Crude Oil under Anaerobic Conditions. Appl. Environ. Microbiol. 2003, 69, 107–112. [Google Scholar] [CrossRef]
- Nielsen, A.H.; Vollertsen, J.; Hvitved-Jacobsen, T. Kinetics and Stoichiometry of Aerobic Sulfide Oxidation in Wastewater from Sewers—Effects of PH and Temperature. Water Environ. Res. 2006, 78, 275–283. [Google Scholar] [CrossRef] [PubMed]
- Nielsen, A.H.; Vollertsen, J. Model Parameters for Aerobic Biological Sulfide Oxidation in Sewer Wastewater. Water 2021, 13, 981. [Google Scholar] [CrossRef]
- Rudelle, E.A.; Vollertsen, J.; Hvitved-Jacobsen, T.; Nielsen, A.H. Kinetics of Aerobic Oxidation of Volatile Sulfur Compounds in Wastewater and Biofilm from Sewers. Water Sci. Technol. 2013, 68, 2330–2336. [Google Scholar] [CrossRef] [PubMed]
- Sudarjanto, G.; Gutierrez, O.; Ren, G.; Yuan, Z. Laboratory Assessment of Bioproducts for Sulphide and Methane Control in Sewer Systems. Sci. Total Environ. 2013, 443, 429–437. [Google Scholar] [CrossRef]
- Remote Monitoring and Real-Time Abatement of Odor Emitted from Sewer Using Odor Sensors, Wireless Communication Technique and Microbial Deodorant. Glob. NEST J. 2018, 20, 646–653. [CrossRef]
- Pikaar, I.; Likosova, E.M.; Freguia, S.; Keller, J.; Rabaey, K.; Yuan, Z. Electrochemical Abatement of Hydrogen Sulfide from Waste Streams. Crit. Rev. Environ. Sci. Technol. 2015, 45, 1555–1578. [Google Scholar] [CrossRef]
- Pang, Y.; Gu, T.; Zhang, G.; Yu, Z.; Zhou, Y.; Zhu, D.Z.; Zhang, Y.; Zhang, T. Experimental Study on Volatile Sulfur Compound Inhibition Using a Single-Chamber Membrane-Free Microbial Electrolysis Cell. Environ. Sci. Pollut. Res. 2020, 27, 30571–30582. [Google Scholar] [CrossRef]
- Wang, K.; Sheng, Y.; Cao, H.; Yan, K.; Zhang, Y. A Novel Microbial Electrolysis Cell (MEC) Reactor for Biological Sulfate-Rich Wastewater Treatment Using Intermittent Supply of Electric Field. Biochem. Eng. J. 2017, 125, 10–17. [Google Scholar] [CrossRef]
- Lovley, D.R. Microbial Fuel Cells: Novel Microbial Physiologies and Engineering Approaches. Curr. Opin. Biotechnol. 2006, 17, 327–332. [Google Scholar] [CrossRef]
- Rabaey, K.; Van de Sompel, K.; Maignien, L.; Boon, N.; Aelterman, P.; Clauwaert, P.; De Schamphelaire, L.; Pham, H.T.; Vermeulen, J.; Verhaege, M.; et al. Microbial Fuel Cells for Sulfide Removal. Environ. Sci. Technol. 2006, 40, 5218–5224. [Google Scholar] [CrossRef]
- Hong, S.W.; Chang, I.S.; Choi, Y.S.; Chung, T.H. Experimental Evaluation of Influential Factors for Electricity Harvesting from Sediment Using Microbial Fuel Cell. Bioresour. Technol. 2009, 100, 3029–3035. [Google Scholar] [CrossRef]
- Eaktasang, N.; Min, H.-S.; Kang, C.; Kim, H.S. Control of Malodorous Hydrogen Sulfide Compounds Using Microbial Fuel Cell. Bioprocess Biosyst. Eng. 2013, 36, 1417–1425. [Google Scholar] [CrossRef] [PubMed]
- Valladares Linares, R.; Domínguez-Maldonado, J.; Rodríguez-Leal, E.; Patrón, G.; Castillo-Hernández, A.; Miranda, A.; Diaz Romero, D.; Moreno-Cervera, R.; Camara-chale, G.; Borroto, C.G.; et al. Scale up of Microbial Fuel Cell Stack System for Residential Wastewater Treatment in Continuous Mode Operation. Water 2019, 11, 217. [Google Scholar] [CrossRef]
- Cai, J.; Zheng, P.; Qaisar, M.; Sun, P. Effect of Electrode Types on Simultaneous Anaerobic Sulfide and Nitrate Removal in Microbial Fuel Cell. Sep. Purif. Technol. 2014, 134, 20–25. [Google Scholar] [CrossRef]
- Zhang, Z.; Zhang, K.; Ouyang, H.; Li, M.K.K.; Luo, Z.; Li, Y.; Chen, C.; Yang, X.; Shao, Z.; Yan, D.Y.S. Simultaneous PAHs Degradation, Odour Mitigation and Energy Harvesting by Sediment Microbial Fuel Cell Coupled with Nitrate-Induced Biostimulation. J. Environ. Manag. 2021, 284, 112045. [Google Scholar] [CrossRef] [PubMed]
- Gao, Y.; Sun, D.; Wang, H.; Lu, L.; Ma, H.; Wang, L.; Ren, Z.J.; Liang, P.; Zhang, X.; Chen, X.; et al. Urine-Powered Synergy of Nutrient Recovery and Urine Purification in a Microbial Electrochemical System. Environ. Sci. Water Res. Technol. 2018, 4, 1427–1438. [Google Scholar] [CrossRef]
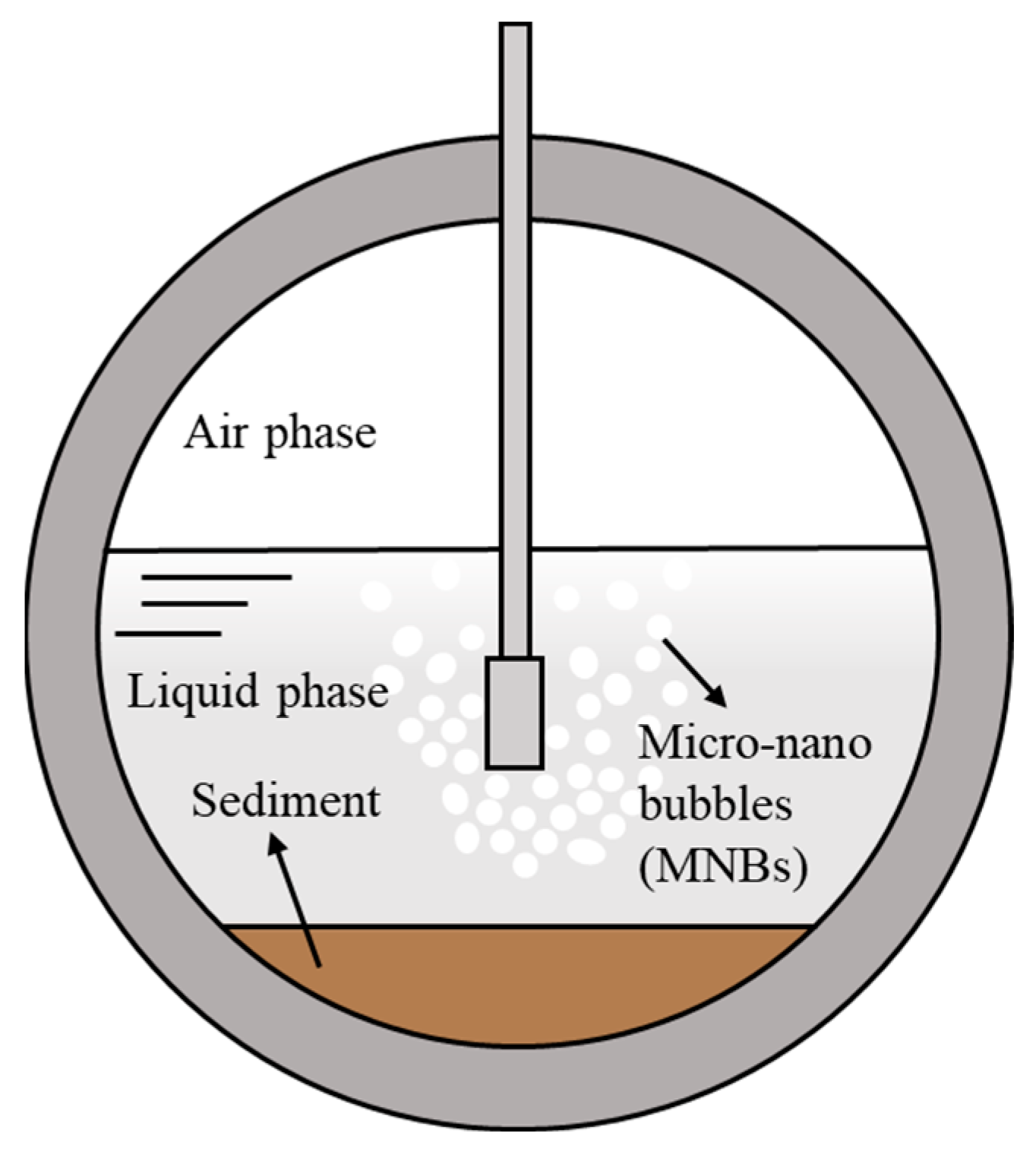

| Category | Representative Substances |
|---|---|
| sulfur-containing compounds | Hydrogen sulfide, mercaptans, thioethers, thiophenes |
| nitrogen-containing compounds | Ammonia, amines, amides, indoles |
| hydrocarbon compounds | Alkanes, alkenes, alkynes, aromatic hydrocarbons |
| oxygen-containing compounds | Alcohols, aldehydes, ketones, phenols, organic acids |
| Category | Compound | Chemical Formula | Characteristics | Health Risks |
|---|---|---|---|---|
| VSCs | Hydrogen sulfide | H2S | Flammable colorless acidic gas, with rotten egg smell at low concentration, sulfur smell at very low concentration | Causes dizziness, weakness, nausea, vomiting, difficulty in breathing, diarrhea, and abdominal pain. High concentrations of inhalation can lead to coma and even death. |
| Methanethiol | CH3SH | Colorless gas, with the odor of rotten cabbage | Causes headaches, nausea, and various degrees of anesthesia. High concentrations of inhalation can cause respiratory paralysis and death. | |
| Ethyl mercaptan | CH3CH2SH | Strong irritating garlic odor, very low OTV, prone to an explosion at high temperatures or in contact with open flames | Causes nausea, dizziness, vomiting, etc., at low concentrations. High concentrations of inhalation can lead to loss of smell, respiratory paralysis, and even death. | |
| Dimethyl sulfide | (CH3)2S | Rotten cabbage odor, easily volatile, low OTV | Very damaging to the central nervous and circulatory systems. | |
| Dimethyl disulfide | C2H6S2 | Colorless or slightly yellowish liquid with a foul odor | Causes headache, nausea, vomiting, irritation of the respiratory tract, eyes, skin, and damage to nerves; large amounts of inhalation can be fatal. | |
| Carbon disulfide | CS2 | Colorless or slightly yellow transparent liquid, easily volatile, with an irritating odor | Causes damage to the human nervous system, cardiovascular system, reproductive system, etc. Short-term exposure to large amounts can lead to acute poisoning. | |
| Nitrogen-containing compounds | Ammonia | NH3 | Colorless, with a strong irritating odor, easily liquefied into colorless liquid | Burns the skin, eyes, and mucous membranes of respiratory organs. If inhaled too much, it can cause lung swelling and even death |
| Methylamine | CH3NH2 | Colorless gas, flammable and explosive, with a strong irritating fishy smell | Causes eye redness and swelling, conjunctival congestion, blurred vision; irritation, edema, and burns in the mucous membranes of the upper respiratory tract, such as the mouth, nose and throat. | |
| Trimethylamine | (CH3)3N | Colorless gas, with a pungent fish smell or cat urine smell | Strong irritant to eyes, nose, throat and respiratory tract. | |
| Indole | C8H7N | White crystals at room temperature, with a strong fecal odor when at high concentrations | Harmful when contact with skin or swallow. Easily irritates eyes. | |
| Skatole | C9H9N | White or slightly brownish crystals with fecal odor; sensitive to light | Causes pulmonary edema, causing nausea, vomiting, dizziness, etc. | |
| Other VOCs | Chloroform | CHCl3 | Colorless, sweet smell, very volatile, but not easily soluble in water | The International Agency for Research on Cancer (IARC) is classified as class 2B, possibly carcinogenic. |
| 1,4-Dichlorobenzene | C6H4Cl2 | Has a strong odor; used in the manufacture of disinfectants, pesticides and deodorants; | Irritates the eyes and respiratory tract, inhibits the nerve center, and is a carcinogen. | |
| Ethylbenzene | C6H5C2H5 | colorless, highly flammable, gasoline-like odor Used in the production of styrene and some products such as pesticides, paints, and inks; usually added to gasoline as an anti-knock agent | Class 2B carcinogen, can cause respiratory and digestive system diseases. | |
| Dichloromethane | CH2Cl2 | Slightly sweet, usually used as solvent in the food industry and manufacturing, paint stripper and degreaser | IARC Category 2A, possibly carcinogenic, can damage the central nervous and respiratory systems. | |
| Tetrachloroethylene | Cl2C=CCl2 | Colorless liquid, volatile, very stable; used for dry cleaning of fabrics, used as paint stripper and degreaser of metal parts in the automobile industry | IARC Category 2A, possibly carcinogenic. Inhalation can cause dizziness, headache, drowsiness, confusion and nausea. | |
| Toluene | C7H8 | Colorless, and smells like paint thinner; used as a solvent and industrial raw material | Causes damage to the skin, eyes, nerves and upper respiratory tract. | |
| Dimethylbenzene | (CH3)2C6H4 | Colorless, highly flammable, sweet-smelling liquid; used as solvent and cleaning agent in printing, rubber, leather and other industries | Irritation to the eyes and upper respiratory tract, anesthesia to the central nervous system at high concentrations, and long-term inhalation may cause cancer. |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jin, S.; Zhang, K.; Cen, C.; Shuai, Y.; Hu, T.; Mao, R. Odorous Substances in Urban Drainage Pipelines and the Removal Technology: A Review. Water 2023, 15, 1157. https://doi.org/10.3390/w15061157
Jin S, Zhang K, Cen C, Shuai Y, Hu T, Mao R. Odorous Substances in Urban Drainage Pipelines and the Removal Technology: A Review. Water. 2023; 15(6):1157. https://doi.org/10.3390/w15061157
Chicago/Turabian StyleJin, Sha, Kejia Zhang, Cheng Cen, Youwen Shuai, Tingting Hu, and Ruyin Mao. 2023. "Odorous Substances in Urban Drainage Pipelines and the Removal Technology: A Review" Water 15, no. 6: 1157. https://doi.org/10.3390/w15061157
APA StyleJin, S., Zhang, K., Cen, C., Shuai, Y., Hu, T., & Mao, R. (2023). Odorous Substances in Urban Drainage Pipelines and the Removal Technology: A Review. Water, 15(6), 1157. https://doi.org/10.3390/w15061157






