The Effects of Channelization with Low In-Stream Barriers on Macroinvertebrate Communities of Mountain Rivers
Abstract
:1. Introduction
2. Materials and Methods
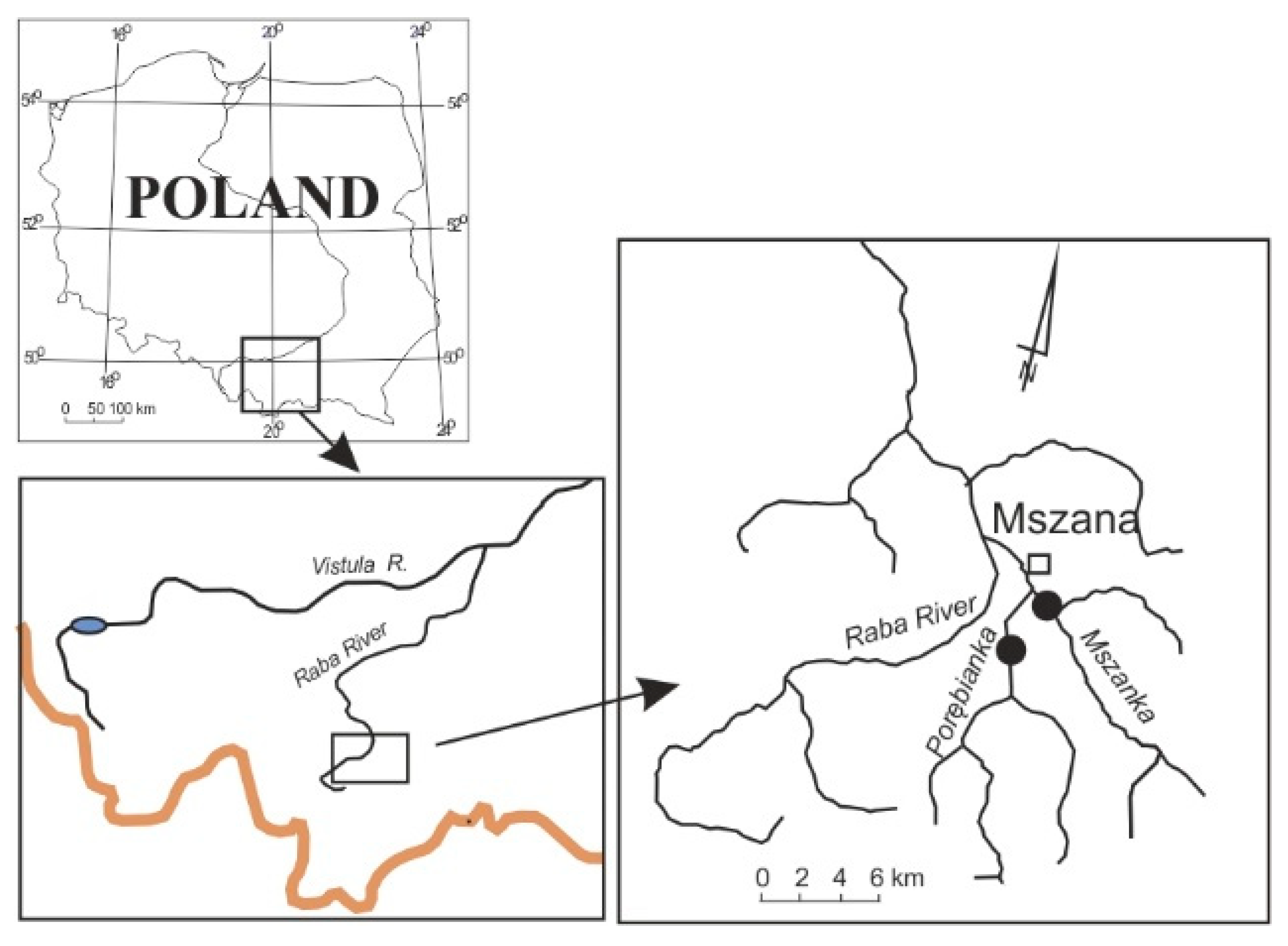
2.1. Study Area
2.2. Methods
2.3. Statistics
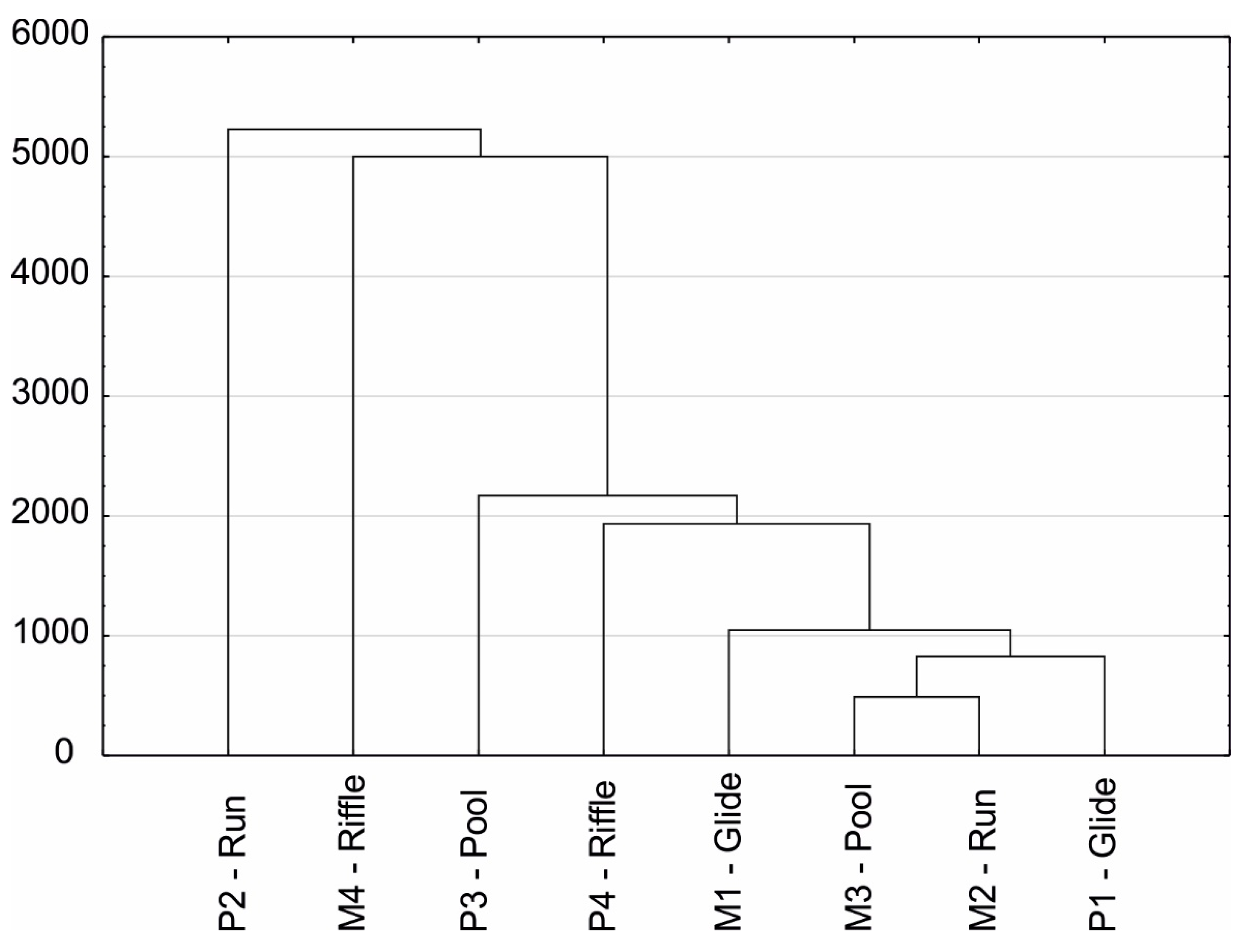
3. Results
3.1. Environmental Parameters
3.2. Benthic Fauna
4. Discussion
4.1. Environmental Parameters
4.2. Benthic Macroinvertebrates
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Brookes, A. Channelized Rivers: Perspectives for Environmental Management; Wiley: Chichester, UK, 1988; p. 326. [Google Scholar]
- Korpak, J.; Lenar-Matyas, A.; Radecki-Pawlik, A.; Plesiński, K. Erosion irregularities from series of grade control structures: The Mszanka River, Western Carpathians. Sci. Total Environ. 2021, 799, 149469. [Google Scholar] [CrossRef] [PubMed]
- Casserly, C.M.; Turner, J.N.; O’Sullivan, J.J.; Bruen, M.; Bullock, C.; Atkinson, S.; Kelly-Quinn, M. Effect of low-head dams on reach-scale suspended sediment dynamics in coarse-bedded streams. J. Environ. Manag. 2021, 277, 111452. [Google Scholar] [CrossRef]
- Conesa-García, C.C.; Lenzi, M.A. (Eds.) Check Dams, Morphological Adjustments and Erosion Control in Torrential Streams; Nova Science Publishers. Inc.: New York, NY, USA, 2013. [Google Scholar]
- Korpak, J. The influence of river training on mountain channel changes (Polish Carpathian Mountains). Geomorphology 2007, 92, 166–181. [Google Scholar] [CrossRef]
- Rohasliney, H.; Jackson, D.C. Lignite and stream channelization influences on aquatic macroinvertebrate assemblages along the Natchez Trave Parkway, Mississippi, USA. Hydrobiologia 2008, 598, 149–162. [Google Scholar] [CrossRef]
- Tenchini, R.; Cazzola, M.; Erba, S.; Ballestrini, R.; Belgiore, C.; Pagnotta, R. Detecting the impact of bank and channel modification on invertebrate communities in Mediterranean temporary streams (Sardinia, SW Italy). Sci. Total Environ. 2016, 565, 1138–1150. [Google Scholar]
- Kownacki, A.; Szarek-Gwiazda, E.; Ligaszewski, M.; Urban, J. Communities of freshwater macroinvertebrate and fish in mountain streams and rivers of the Upper Dunajec Catchment (Western Carpathians) including long-term human impact. In Polish River Basins and Lakes: Biological Status and Water Management p. II.; Handbook of Environmental Chemistry Series; Korzeniewska, E., Hamisz, M., Eds.; Springer Nature Switzerland: Cham, Switzerland, 2020; Volume 87, pp. 269–294. [Google Scholar]
- Mykra, H.; Heino, J. Decreased habitat specialization in macroinvertebrate assemblages in anthropogenically disturbed streams. Ecol. Complex. 2017, 31, 181–188. [Google Scholar] [CrossRef]
- Buffagni, A.; Armanini, D.G.; Stefania, E.R.B.A. Does the lentic-lotic character of rivers affect invertebrate metrics used in the assessment of ecological quality? J. Limnol. 2009, 68, 92. [Google Scholar] [CrossRef]
- Jansen, W.; Böhmer, J.; Kappus, B.; Beiter, T.; Breitinger, B.; Hock, C. Benthic invertebrate and fish communities as indicators of morphological integrity in the Enz River (south-west Germany). Hydrobiologia 2000, 422–423, 331–342. [Google Scholar] [CrossRef]
- Kennedy, T.L.; Turner, T.F. River channelization reduces nutrient flow and macroinvertebrate diversity at the aquatic terrestrial transition zone. Ecosphere 2011, 2, 35. [Google Scholar] [CrossRef]
- Horsak, M.; Bojkova, J.; Zahradkova, S.; Omesova, M.; Helesic, J. Impact of reservoirs and channelization on lowland river macroinvertebrates: A case study from Central Europe. Limnologica 2009, 39, 140–151. [Google Scholar] [CrossRef] [Green Version]
- Palmer, M.A.; Menninger, H.L.; Bernhardt, E. River restoration, habitat heterogeneity and biodiversity: A failure of theory or practice? Frehw. Biol. 2010, 55, 205–222. [Google Scholar] [CrossRef]
- Leps, M.; Sundermann, A.; Tonkin, J.D.; Lorenz, A.W.; Haase, P. Time is no healer: Increasing restoration age does not lead to improved benthic invertebrate communities in restored river reaches. Sci. Total Environ. 2016, 557–558, 722–732. [Google Scholar] [CrossRef] [PubMed]
- Verdonschot, R.C.M.; Kail, J.; McKie, B.G.; Verdonschot, P.F.M. The role of benthic microhabitats in determining the effects of hydromorfological river restoration on macroinvertebrates. Hydrobiologia 2016, 769, 55–66. [Google Scholar] [CrossRef] [Green Version]
- Leps, M.; Tonkin, J.D.; Dahm, V.; Haase, P.; Sundermann, A. Disentangling environmental drivers of benthic invertebrate assemblages: The role of spatial scale and riverscape heterogeneity in a multiple stressor environment. Sci. Total Environ. 2015, 536, 546–556. [Google Scholar] [CrossRef] [PubMed]
- Poulos, H.M.; Miller, K.E.; Heinemann, R.; Kraczkowski, M.L.; Whelchel, A.W.; Chernoff, B. Dam removal effects on benthic macroinvertebrate dynamics: A New England stream case study (Connecticut, USA). Sustainability 2019, 11, 2875. [Google Scholar] [CrossRef] [Green Version]
- Cook, D.R.; Sullivan, S.M.P. Associations between riffle development and aquatic biota following lowhead dam removal. Environ. Monit. Assess. 2018, 190, 339. [Google Scholar] [CrossRef] [Green Version]
- Mbaka, J.G.; Mwaniki, M.W. A global review of the downstream effects of small impoundments on stream habitat conditions and macroinvertebrates. Environ. Rev. 2015, 23, 257–262. [Google Scholar] [CrossRef]
- Parkyn, M.S.; Smith, B.J. Dispersal constraintsfor stream invertebrates: Setting realistic timescales for biodiversity restoration. Environ. Manag. 2011, 48, 602–614. [Google Scholar] [CrossRef]
- Goeller, B.; Wolter, C. Performance of bottom ramps to mitigate gravel habitat bottlenecks in a channelized lowland river. Restor. Ecol. 2015, 23, 595–606. [Google Scholar] [CrossRef]
- Pagliara, S.; Radecki-Pawlik, A.; Palermo, M.; Plesiński, K. Block ramps in curved rivers: Morphology analysis and prototype data supported design criteria for mild bed slopes. River Res. Appl. 2017, 33, 427–437. [Google Scholar] [CrossRef]
- Korpak, J. Geomorphologic effects of river engineering structures in Carpathian fluvial systems. Landf. Anal. 2010, 14, 34–44. [Google Scholar]
- Plesiński, K.; Radecki-Pawlik, A.; Kuboń, P.; Tatara, T.; Pachla, F.; Jurowska, N. Bed load transport and alternation of a gravel-bed river morphology within a vicinity of block ramp: Classical and numerical approach. Sustainability 2022, 14, 4665. [Google Scholar] [CrossRef]
- Kalogianni, E.; Vourka, A.; Karaouzas, I.; Vardakas, L.; Laschou, S.; Skoulikidis, N.T. Combined effects of water stress and pollution on macroinvertebrate and fish assemblages in a Mediterranean intermittent river. Sci. Total Environ. 2017, 603, 639–650. [Google Scholar] [CrossRef] [PubMed]
- Duerdoth, C.P.; Arnold, A.; Murphy, J.F.; Naden, P.S.; Scarlett, P.; Collins, A.L.; Sear, D.A.; Jones, J.I. Assessment of a rapid method for quantitative reach-scale estimates of deposited fine sediment in rivers. Geomorphology 2015, 230, 37–50. [Google Scholar] [CrossRef]
- Kownacki, A. Stream ecosystems in mountain grassland (West Carpathians). 12. General conclusion. Acta Hydrobiol. 1982, 24, 413–422. [Google Scholar]
- Szarek-Gwiazda, E.; Mazurkiewicz-Boroń, G.; Gwiazda, R.; Urban, J. Chemical variability of water and sediment over time and along a mountain river subjected to natural and human impact. Knowl. Manag. Aquat. Ecosyst. 2018, 419, 5. [Google Scholar] [CrossRef] [Green Version]
- Szarek-Gwiazda, E.; Gwiazda, R. Impact of flow and damming on water quality of the mountain Raba River (southern Poland)—Long-term studies. Arch. Environ. Prot. 2022, 48, 31–40. [Google Scholar]
- Regulation of the Minister of Maritime Economy and Inland Navigation of 11 October 2019 on the Classification of Ecological Status, Ecological Potential and Chemical Status as Well as the Method of Classifying the Status of Surface Water Bodies as Well as Environmental Quality Standards for Priority Substances. Dz. U. z dnia 7 November 2019 r., poz. 2149, Warszawa. Available online: https://isap.sejm.gov.pl/isap.nsf/DocDetails.xsp?id=WDU20190002148 (accessed on 20 January 2023). (In Polish)
- Zuber, A.; Chowaniec, J.; Borowiec, M. On the origin of chloride waters in the Polish Flysch Carpathians. Bull. State Geol. Surv. Pol. 2010, 441, 201–2008. [Google Scholar]
- Wang, J.; Ding, C.; Heino, J.; Jiang, X.; Tao, J.; Ding, L.; Su, W.; Huang, M.; He, D. What explains the variation in dam impacts on riverine macroinvertebrates? A global quantitative synthesis. Environ. Res. Lett. 2020, 15, 124028. [Google Scholar]
- Wood, P.J.; Armitage, P.D. Biological effects of fine sediment in the lotic environment. Environ. Manag. 1997, 21, 203–217. [Google Scholar] [CrossRef]
- Graf, W.; Leitner, P.; Hanetseder, I.; Ittner, L.D.; Dossi, F.; Hauer, C. Ecological degradation of meandering river by local channelization effects: A case study in an Austrian lowland river. Hydrobiologia 2016, 772, 145–160. [Google Scholar] [CrossRef]
- Poff, N.L.; Zimmerman, J.K.H. Ecological responses to altered flow regimes: A literature review to inform the science and management of environmental flows. Freshw. Biol. 2010, 55, 194–205. [Google Scholar] [CrossRef]
- Plesinski, K.; Bylak, A.; Radecki-Pawlik, A.; Mikolajczyk, T.; Kukula, K. Possibilities of fish passage trough the block ramp: Model-based estimation of permeability. Sci. Total Environ. 2018, 631–632, 1201–1211. [Google Scholar] [CrossRef] [PubMed]
- Lorenz, A.W. Continuous riverine biodiversity changes in a 10-years-post-restoration-study-impacts and pitfalls. River Res. Appl. 2021, 37, 270–282. [Google Scholar] [CrossRef]
- Kurek, S.; Pawlik-Dobrowolski, J. Stream ecosystems in mountain grassland (West Carpathians). 2. Hydrological characteristics. Acta Hydrobiol. 1982, 24, 375–390. [Google Scholar]
- Bombówna, M. Stream ecosystems in mountain grassland (West Carpathians). 3. Chemical composition of water. Acta Hydrobiol. 1982, 24, 321–335. [Google Scholar]
- Dumnicka, E. Habitat preferences of invertebrates (especially Oligochaeta) in a stream. Acta Hydrobiol. 1994, 30, 91–101. [Google Scholar]
- Syrovátka, V.; Schenková, J.; Brabec, K. The distribution of chironomid larvae and oligochaetes within a stony-bottomed river stretch: The role of substrate and hydraulic characteristics. Fundam. Appl. Limnol. 2009, 174, 43. [Google Scholar] [CrossRef]
- Longing, S.D.; Voshell, J.R.; Dolloff, C.A.; Roghair, C.N. Relationships of sedimentation and benthic macroinvertebrate assemblages in headwater streams using systematic longitudinal sampling at the reach scale. Environ. Monit. Assess. 2010, 161, 517–530. [Google Scholar] [CrossRef]

| Habitat Area in the Rivers [%] | Macroinvertebrate Density [ind./m2] | |||
|---|---|---|---|---|
| Habitat Types | Porębianka | Mszanka | Porębianka | Mszanka |
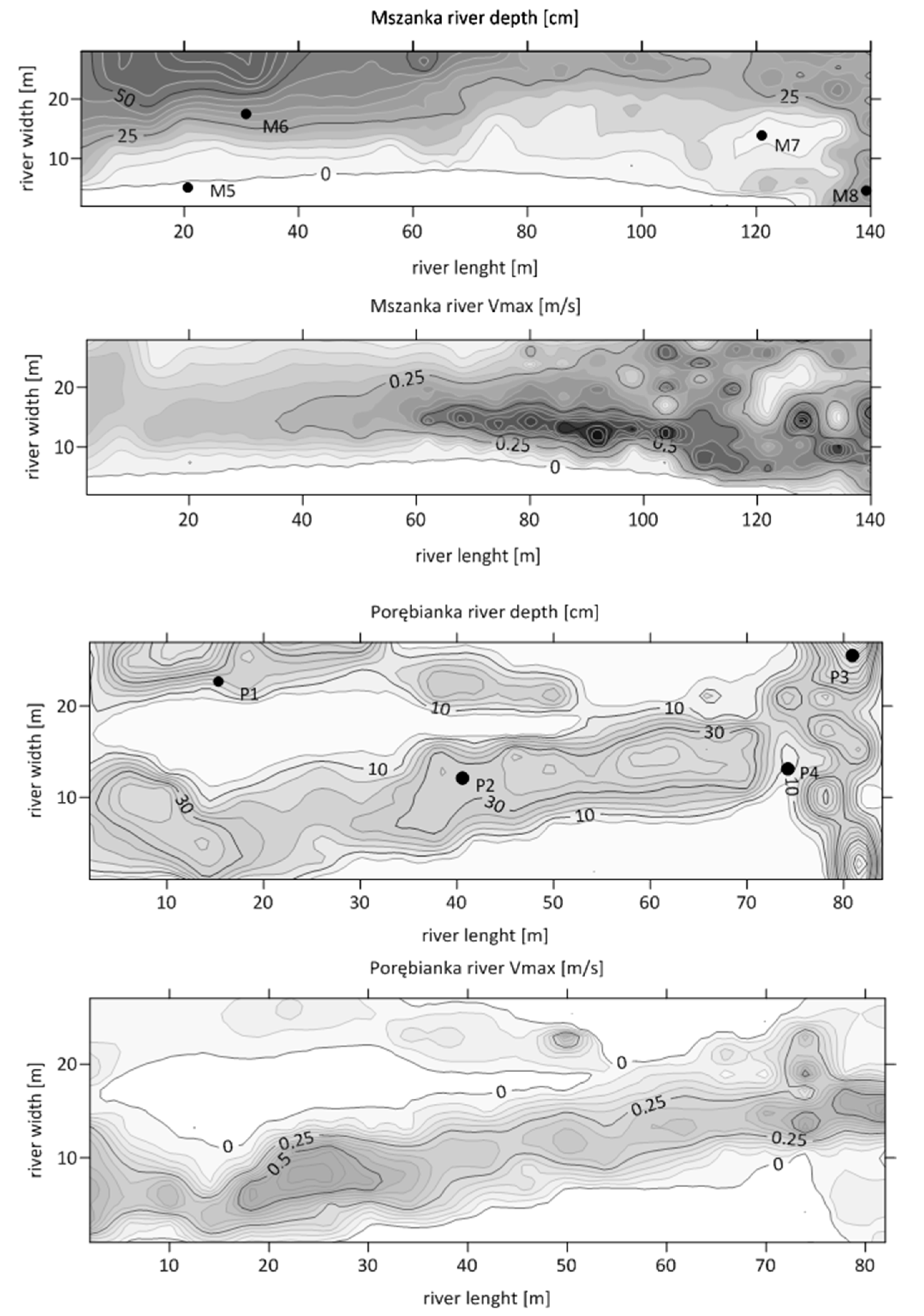
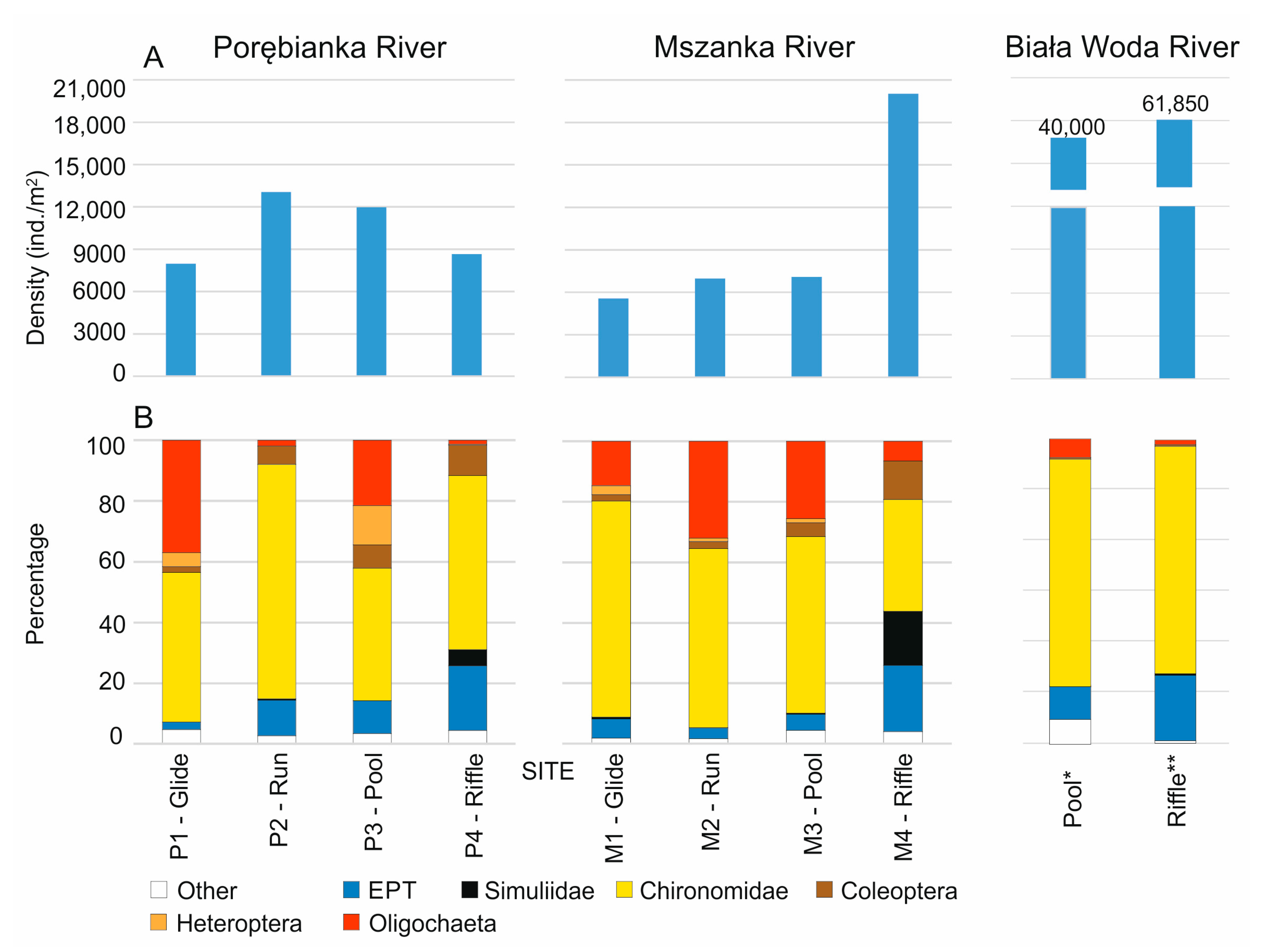
| 1. Glides (relatively shallow areas with slow current < 0.15 m/s) | 53.7 | 26.3 | 7972 | 5571 |
| 2. Runs (areas of relatively moderate depth and current 0.15–0.6 m/s) | 43.3 | 66.3 | 13,058 | 6981 |
| 3. Pools (relatively deep areas, >0.6 m with slow current < 0.15 m/s) | 1.7 | 2.7 | 11,967 | 7088 |
| 4. Riffles (relatively shallow areas with rapid current > 0.6 m/s) | 1.4 | 4.7 | 8662 | 20,029 |
| Median density in the studied stream section | 10,314 | 7034 | ||
| Parameter | Unit | Porębianka | Mszanka |
|---|---|---|---|
| Dissolved oxygen | mg/L | 11.3 ± 1.1 | 11.3 ± 1.1 |
| Oxygen saturation | % | 105.6 ± 10.0 | 108.2 ± 15.2 |
| Conductivity | µS/cm | 281.8 ± 65.5 | 323.0 ± 66.2 |
| pH | 8.0 ± 0.3 | 8.0 ± 0.2 | |
| Cl− | mg/L | 6.5 ± 3.4 | 11.9 ± 2.4 |
| SO42− | mg/L | 12.6 ± 2.2 | 14.8 ± 2.0 |
| HCO3− | mg/L | 118.5 ± 39.2 | 141.0 ± 30.1 |
| Na+ | mg/L | 4.9 ± 2.8 | 8.7 ± 1.4 |
| K+ | mg/L | 1.8 ± 2.0 | 3.0 ± 0.6 |
| Ca2+ | mg/L | 41.2 ± 9.4 | 45.1 ± 10.1 |
| Mg2+ | mg/L | 7.3 ± 1.9 | 8.5 ± 1.9 |
| NO3− | mg/L | 3.5 ± 1.7 | 2.9 ± 0.9 |
| NH4+ | mg/L | 0.028 ± 0.013 | 0.015 ± 0.018 |
| PO43− | mg/L | 0.008 ± 0.011 | 0.007 ± 0.010 |
| Parameter | N | T | Z | p |
|---|---|---|---|---|
| Conductivity | 5 | 0.00 | 2.02 | 0.043 |
| Cl− | 5 | 0.00 | 2.02 | 0.043 |
| SO42− | 5 | 0.00 | 2.02 | 0.043 |
| HCO3− | 5 | 0.00 | 2.02 | 0.043 |
| Na+ | 5 | 0.00 | 2.02 | 0.043 |
| K+ | 5 | 0.00 | 2.02 | 0.043 |
| Ca2+ | 5 | 0.00 | 2.02 | 0.043 |
| Mg2+ | 5 | 0.00 | 2.02 | 0.043 |
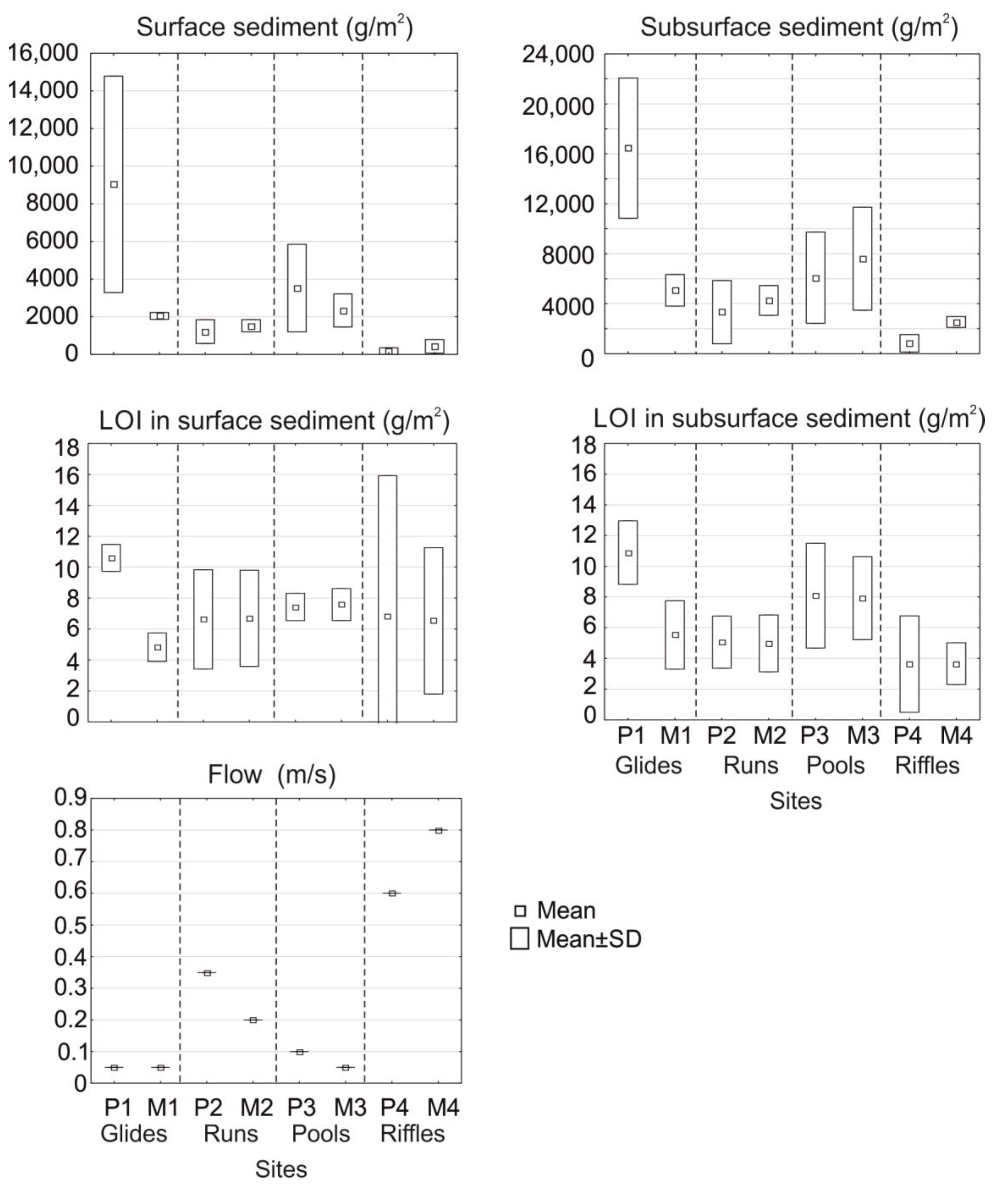
| Parameter/Fauna Group | Flow | Dissolved Oxygen | Surface Sediment | Subsurface Sediment | LOI Surface * | LOI SubSed ** |
|---|---|---|---|---|---|---|
| Surface sediment | −0.88 | - | 0.91 | 0.66 | ||
| Subsurface sediment | −0.81 | 0.91 | - | 0.54 | ||
| LOI Surface * | - | 0.68 | ||||
| LOI SubSed ** | −0.61 | 0.66 | 0.54 | 0.68 | - | |
| Oligochaeta | −0.42 | 0.41 | ||||
| Chironomidae | −0.47 | |||||
| Simullidae | 0.49 | −0.47 | −0.46 | −0.47 | −0.56 | |
| Ephemeroptera | 0.56 | −0.56 | −0.42 | −0.43 | ||
| Plecoptera | 0.46 | −0.47 | ||||
| Trichoptera | 0.57 | −0.48 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Szarek-Gwiazda, E.; Ciszewski, D.; Kownacki, A. The Effects of Channelization with Low In-Stream Barriers on Macroinvertebrate Communities of Mountain Rivers. Water 2023, 15, 1059. https://doi.org/10.3390/w15061059
Szarek-Gwiazda E, Ciszewski D, Kownacki A. The Effects of Channelization with Low In-Stream Barriers on Macroinvertebrate Communities of Mountain Rivers. Water. 2023; 15(6):1059. https://doi.org/10.3390/w15061059
Chicago/Turabian StyleSzarek-Gwiazda, Ewa, Dariusz Ciszewski, and Andrzej Kownacki. 2023. "The Effects of Channelization with Low In-Stream Barriers on Macroinvertebrate Communities of Mountain Rivers" Water 15, no. 6: 1059. https://doi.org/10.3390/w15061059





