Comprehensive Comparison of Various Microplastic Sampling Methods in Sea Water: Implications for Data Compilation
Abstract
:1. Introduction
2. Materials and Methods
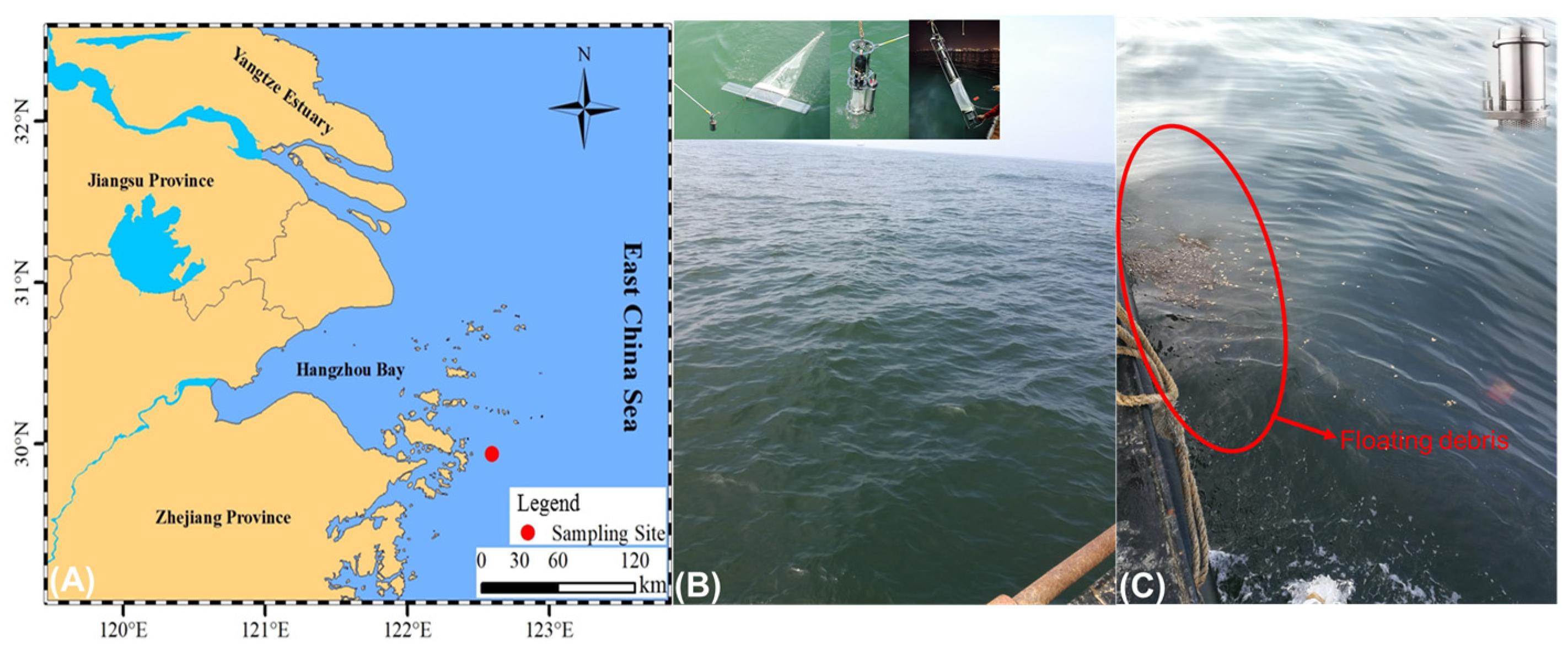
2.1. Study Area and Sampling Methods
2.1.1. Manta Net Sampling
2.1.2. Plankton Pumps
2.1.3. Submersible Pumps
2.2. Laboratory Analysis
2.3. Sample Identification
2.4. Contamination Prevention
2.5. Statistical Analysis
3. Results and Discussions
3.1. MP Abundance
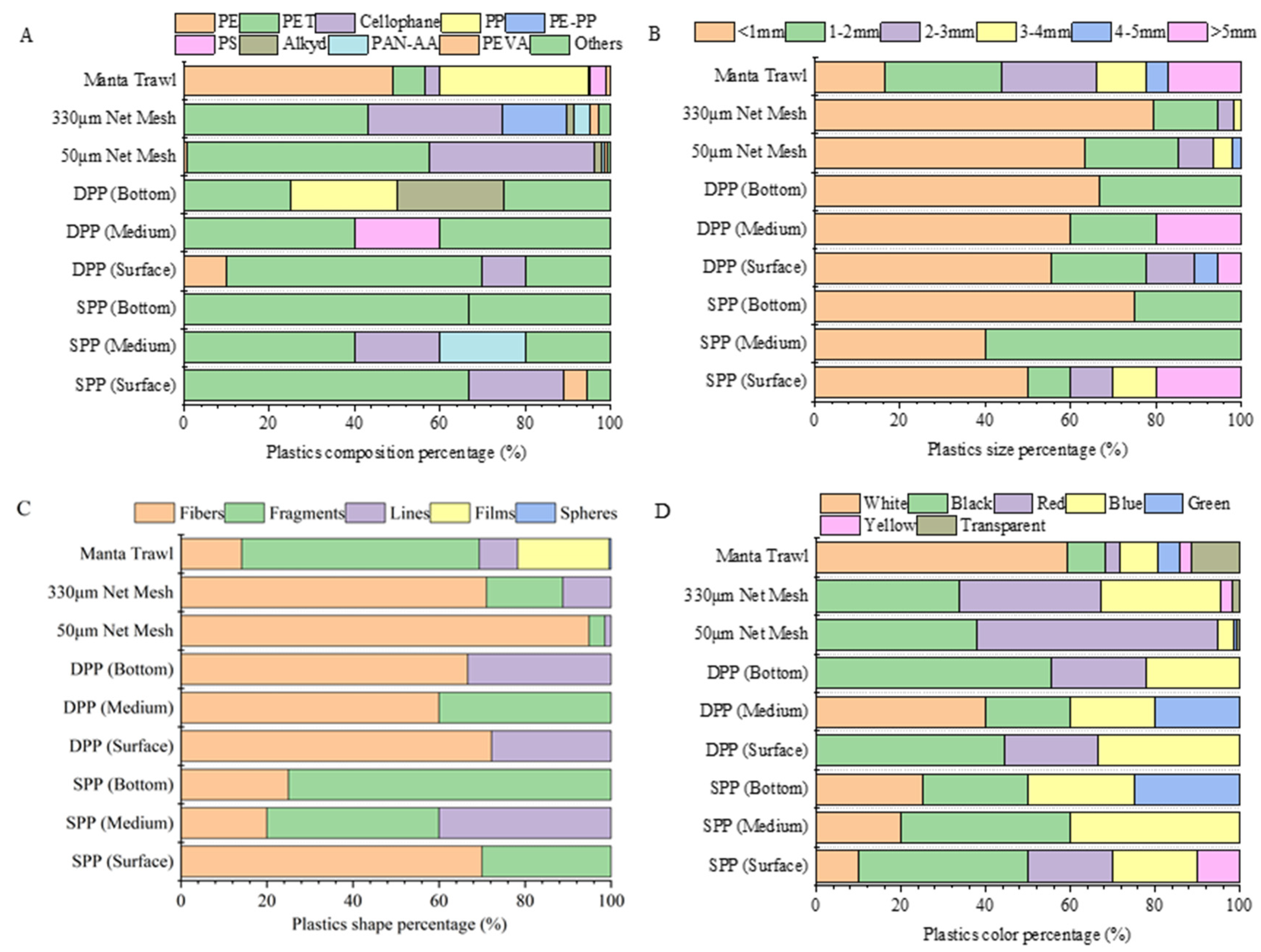
3.2. Characteristics of MP
3.3. MPs Data Comparison between the Manta Trawl and Plankton Pump
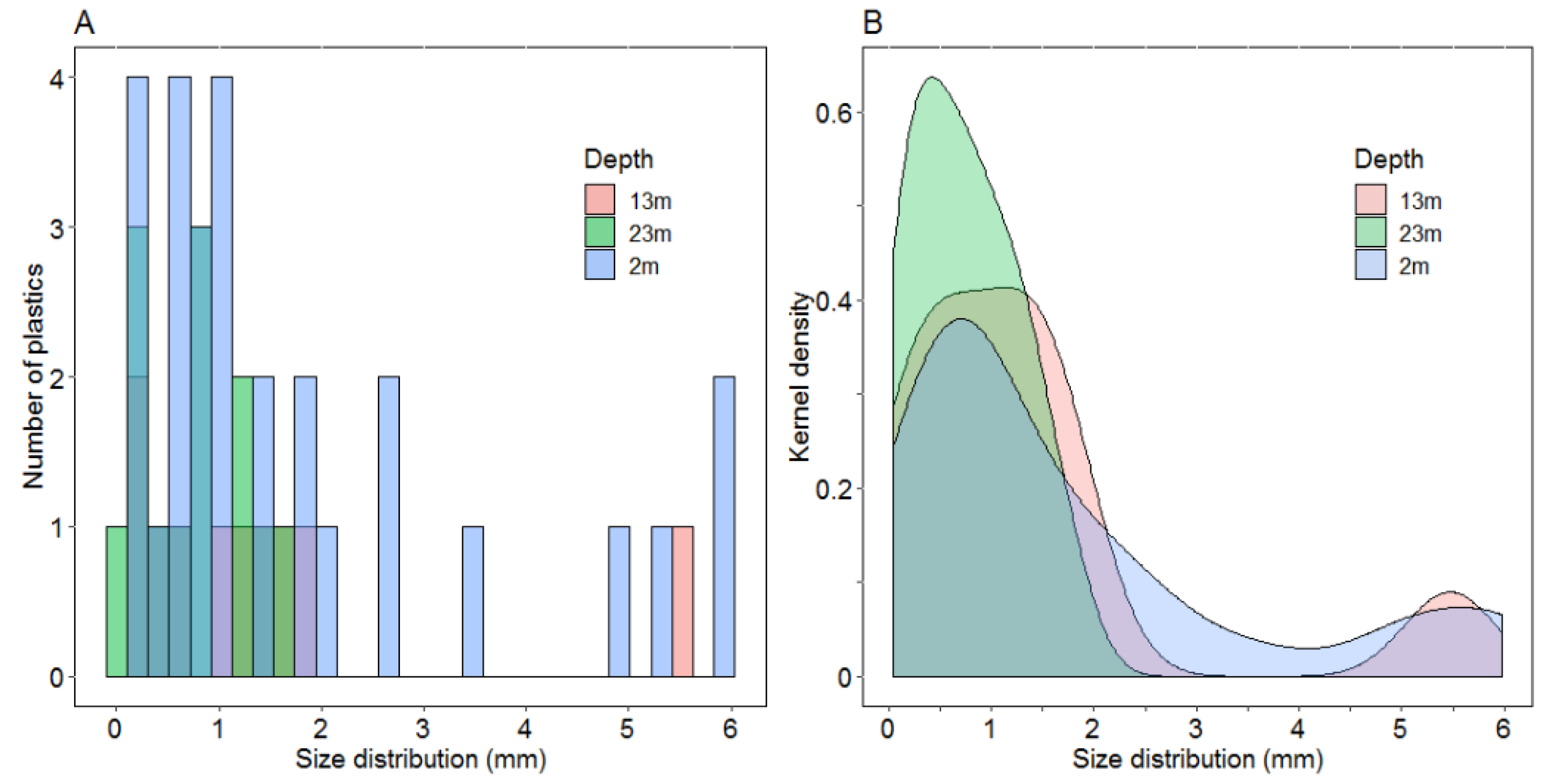
3.4. MPs Distribution in the Water Column
3.5. Impacts of Mesh Size and Floating Debris Hotspot on Microplastics Collection
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- PlasticsEurope. Plastics-the-Facts-2021. 2021. Available online: https://plasticseurope.org/knowledge-hub/plastics-the-facts-2021/ (accessed on 5 May 2022).
- Zhu, L.; Zhao, S.; Bittar, T.B.; Stubbins, A.; Li, D. Photochemical dissolution of buoyant microplastics to dissolved organic carbon: Rates and microbial impacts. J. Hazard. Mater. 2020, 383, 121065. [Google Scholar] [CrossRef] [PubMed]
- Law, K.L. Plastics in the Marine Environment. Annu. Rev. Mar. Sci. 2017, 9, 205–229. [Google Scholar] [CrossRef] [Green Version]
- GESAMP. Guidelines for the Monitoring and Assessment of Plastic Litter in the Ocean; Kershaw, P.J., Turra, A., Galgani, F., Eds.; GESAMP Joint Group of Experts on the Scientific Aspects of Marine Environmental Protection: London, UK, 2019. [Google Scholar]
- Bai, M.; Lin, Y.; Hurley, R.R.; Zhu, L.; Li, D. Controlling Factors of Microplastic Riverine Flux and Implications for Reliable Monitoring Strategy. Environ. Sci. Technol. 2022, 56, 48–61. [Google Scholar] [CrossRef] [PubMed]
- Moore, C.J.; Moore, S.L.; Leecaster, M.K.; Weisberg, S.B. A comparison of plastic and plankton in the north Pacific central gyre. Mar. Pollut. Bull. 2001, 42, 1297–1300. [Google Scholar] [CrossRef]
- Isobe, A.; Uchida, K.; Tokai, T.; Iwasaki, S. East Asian seas: A hot spot of pelagic microplastics. Mar. Pollut. Bull. 2015, 101, 618–623. [Google Scholar] [CrossRef]
- Day, R.H.; Shaw, D.G. Patterns in the abundance of pelagic plastic and tar in the north pacific ocean, 1976–1985. Mar. Pollut. Bull. 1987, 18, 311–316. [Google Scholar] [CrossRef]
- Anastasopoulou, A.; Mytilineou, C.; Smith, C.J.; Papadopoulou, K.N. Plastic debris ingested by deep-water fish of the Ionian Sea (Eastern Mediterranean). Deep. Sea Res. Part I Oceanogr. Res. Pap. 2013, 74, 11–13. [Google Scholar] [CrossRef]
- Christopher, K.P.; Eva, R.L.; Claudia, H.S.A.; Teresa, A.; Melanie, B.; Miquel, C.; Joan, B.C.; Jaime, D.; Gerard, D.; François, G.; et al. Marine litter distribution and density in European seas, from the shelves to deep basins. PLoS ONE 2014, 9, e95839. [Google Scholar]
- Xu, P.; Peng, G.; Su, L.; Gao, Y.; Gao, L.; Li, D. Microplastic risk assessment in surface waters: A case study in the Changjiang Estuary, China. Mar. Pollut. Bull. 2018, 133, 647–654. [Google Scholar] [CrossRef]
- Zhao, S.; Wang, T.; Zhu, L.; Xu, P.; Wang, X.; Gao, L.; Li, D. Analysis of suspended microplastics in the Changjiang Estuary: Implications for riverine plastic load to the ocean. Water Res. 2019, 161, 560–569. [Google Scholar] [CrossRef]
- Li, D.; Liu, K.; Li, C.; Peng, G.; Andrady, A.L.; Wu, T.; Zhang, Z.; Wang, X.; Song, Z.; Zong, C.; et al. Profiling the Vertical Transport of Microplastics in the West Pacific Ocean and the East Indian Ocean with a Novel in Situ Filtration Technique. Environ. Sci. Technol. 2020, 54, 12979–12988. [Google Scholar] [CrossRef]
- Liu, K.; Zhang, F.; Song, Z.; Zong, C.; Wei, N.; Li, D. A novel method enabling the accurate quantification of microplastics in the water column of deep ocean. Mar. Pollut. Bull. 2019, 146, 462–465. [Google Scholar] [CrossRef]
- Wang, T.; Zhao, S.; Zhu, L.; McWilliams, J.C.; Galgani, L.; Amin, R.M.; Nakajima, R.; Jiang, W.; Chen, M. Accumulation, transformation and transport of microplastics in estuarine fronts. Nat. Rev. Earth Environ. 2022, 3, 795–805. [Google Scholar] [CrossRef]
- Lebreton, L.C.M.; Van, D.Z.J.; Damsteeg, J.; Slat, B.; Andrady, A.; Reisser, J. River plastic emissions to the world′s oceans. Nat. Commun. 2017, 8, 15611. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Li, C.; Wang, X.; Liu, K.; Zhu, L.; Wei, N.; Zong, C.; Li, D. Pelagic microplastics in surface water of the Eastern Indian Ocean during monsoon transition period: Abundance, distribution, and characteristics. Sci. Total Environ. 2021, 755, 142629. [Google Scholar] [CrossRef] [PubMed]
- Rech, S.; Macaya, C.V.; Pantoja, J.F.; Rivadeneira, M.M.; Jofre, M.D.; Thiel, M. Rivers as a source of marine litter—A study from the SE Pacific. Mar. Pollut. Bull. 2014, 82, 66–75. [Google Scholar] [CrossRef] [PubMed]
- Gasperi, J.; Dris, R.; Bonin, T.; Rocher, V.; Tassin, B. Assessment of floating plastic debris in surface water along the Seine River. Environ. Pollut. 2014, 195, 163–166. [Google Scholar] [CrossRef] [Green Version]
- Zhao, S.; Zhu, L.; Li, D. Microplastic in three urban estuaries, China. Environ. Pollut. 2015, 206, 597–604. [Google Scholar] [CrossRef]
- Liu, K.; Wu, T.; Wang, X.; Song, Z.; Zong, C.; Wei, N.; Li, D. Consistent Transport of Terrestrial Microplastics to the Ocean through Atmosphere. Environ. Sci. Technol. 2019, 53, 10612–10619. [Google Scholar] [CrossRef]
- Bai, M.; Zhu, L.; An, L.; Peng, G.; Li, D. Estimation and prediction of plastic waste annual input into the sea from China. Acta Oceanol. Sin. 2018, 37, 26–39. [Google Scholar] [CrossRef]
- Zhang, W.; Zhang, S.; Wang, J.; Wang, Y.; Mu, J.; Wang, P.; Lin, X.; Ma, D. Microplastic pollution in the surface waters of the Bohai Sea, China. Environ. Pollut. 2017, 231, 541–548. [Google Scholar] [CrossRef]
- Dris, R.; Imhof, H.; Sanchez, W.; Gasperi, J.; Galgani, F.O.; Tassin, B.; Laforsch, C. Beyond the ocean: Contamination of freshwater ecosystems with (micro-)plastic particles. Environ. Chem. 2015, 12, 539. [Google Scholar] [CrossRef]
- Dris, R.; Gasperi, J.; Saad, M.; Mirande, C.; Tassin, B. Synthetic fibers in atmospheric fallout: A source of microplastics in the environment? Mar. Pollut. Bull. 2016, 104, 290–293. [Google Scholar] [CrossRef]
- Thompson, R.C.; Olsen, Y.; Mitchell, R.P.; Davis, A.; Rowland, S.J.; John, A.W.; McGonigle, D.; Russell, A.E. Lost at sea: Where is all the plastic? Science 2004, 304, 838. [Google Scholar] [CrossRef] [PubMed]
- Fendall, L.S.; Sewell, M.A. Contributing to marine pollution by washing your face: Microplastics in facial cleansers. Mar. Pollut. Bull. 2009, 58, 1225–1228. [Google Scholar] [CrossRef]
- Pan, Z.; Guo, H.; Chen, H.; Wang, S.; Sun, X.; Zou, Q.; Zhang, Y.; Lin, H.; Cai, S.; Huang, J. Microplastics in the Northwestern Pacific: Abundance, distribution, and characteristics. Sci. Total Environ. 2019, 650, 1913–1922. [Google Scholar] [CrossRef]
- Isobe, A.; Kubo, K.; Tamura, Y.; Kako, S.I.; Nakashima, E.; Fujii, N. Selective transport of microplastics and mesoplastics by drifting in coastal waters. Mar. Pollut. Bull. 2014, 89, 324–330. [Google Scholar] [CrossRef] [PubMed]
- Lusher, A.L.; Hernandezmilian, G.; O’Brien, J.; Berrow, S.; O′Connor, I.; Officer, R. Microplastic and macroplastic ingestion by a deep diving, oceanic cetacean: The True’s beaked whale Mesoplodon mirus. Environ. Pollut. 2015, 199, 185. [Google Scholar] [CrossRef]
- Norén, F. Small Plastic Particles in Coastal Swedish Waters; KIMO Report; KIMO: Esbjerg, Denmark, 2007; pp. 1–11. [Google Scholar]
- Dris, R.; Gasperi, J.; Rocher, V.; Saad, M.; Renault, N.; Tassin, B. Microplastic contamination in an urban area: A case study in Greater Paris. Environ. Chem. 2015, 12, 592. [Google Scholar] [CrossRef]
- Kukulka, T.; Proskurowski, G.; Morét, F.S.; Meyer, D.W.; Law, K.L. The effect of wind mixing on the vertical distribution of buoyant plastic debris, Geophys. Res. Lett. 2012, 39, 7. [Google Scholar] [CrossRef] [Green Version]
- Reisser, J.; Slat, B.; Noble, K.; du Plessis, K.; Epp, M.; Proietti, M.; de Sonneville, J.; Becker, T.; Pattiaratchi, C. The vertical distribution of buoyant plastics at sea: An observational study in the North Atlantic Gyre. Biogeosciences 2015, 12, 1249–1256. [Google Scholar] [CrossRef] [Green Version]



| Sampling Method | Depth (m) | Mesh Size (µm) | Filtered Volume (m3) | Abundance (n/m3) | Fiber (%) |
|---|---|---|---|---|---|
| Manta trawl | 0.16 | 330 | 91.85 | 2.31 | 14.2 |
| SPP (Surface) | 1 | 150 | 5 | 2.00 | 70.0 |
| SPP (Medium) | 13 | 150 | 5 | 1.00 | 20.0 |
| SPP (Bottom) | 23 | 150 | 5 | 0.80 | 25.0 |
| DPP (Surface) | 1 | 150 | 3 | 6.00 | 72.2 |
| DPP (Medium) | 13 | 150 | 3 | 1.67 | 66.7 |
| DPP (Bottom) | 23 | 150 | 3 | 3.00 | 94.9 |
| Submersible Pump | 1 | 330 | 0.1 | 357 ± 119 a | 71.0 |
| Submersible Pump | 1 | 50 | 0.1 | 553 ± 19 a | 94.9 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Shi, H.; Wang, X.; Zhu, L.; Li, D. Comprehensive Comparison of Various Microplastic Sampling Methods in Sea Water: Implications for Data Compilation. Water 2023, 15, 1035. https://doi.org/10.3390/w15061035
Shi H, Wang X, Zhu L, Li D. Comprehensive Comparison of Various Microplastic Sampling Methods in Sea Water: Implications for Data Compilation. Water. 2023; 15(6):1035. https://doi.org/10.3390/w15061035
Chicago/Turabian StyleShi, Haochen, Xiaohui Wang, Lixin Zhu, and Daoji Li. 2023. "Comprehensive Comparison of Various Microplastic Sampling Methods in Sea Water: Implications for Data Compilation" Water 15, no. 6: 1035. https://doi.org/10.3390/w15061035
APA StyleShi, H., Wang, X., Zhu, L., & Li, D. (2023). Comprehensive Comparison of Various Microplastic Sampling Methods in Sea Water: Implications for Data Compilation. Water, 15(6), 1035. https://doi.org/10.3390/w15061035






