Seasonal and Spatial Variations in Functional Genes and Microbial Community of Feammox and Its Associated Processes in Urban Green Heart Soil
Abstract
:1. Introduction
2. Materials and Methods
2.1. Study Area and Soil Sampling
2.2. Physicochemical Analyses
2.3. DNA Extraction
2.4. Quantitative Polymerase Chain Reaction (qPCR)
2.5. 16S rRNA Gene High-Throughput Sequencing
2.6. Data Analysis
3. Results and Discussion
3.1. Physicochemical Properties
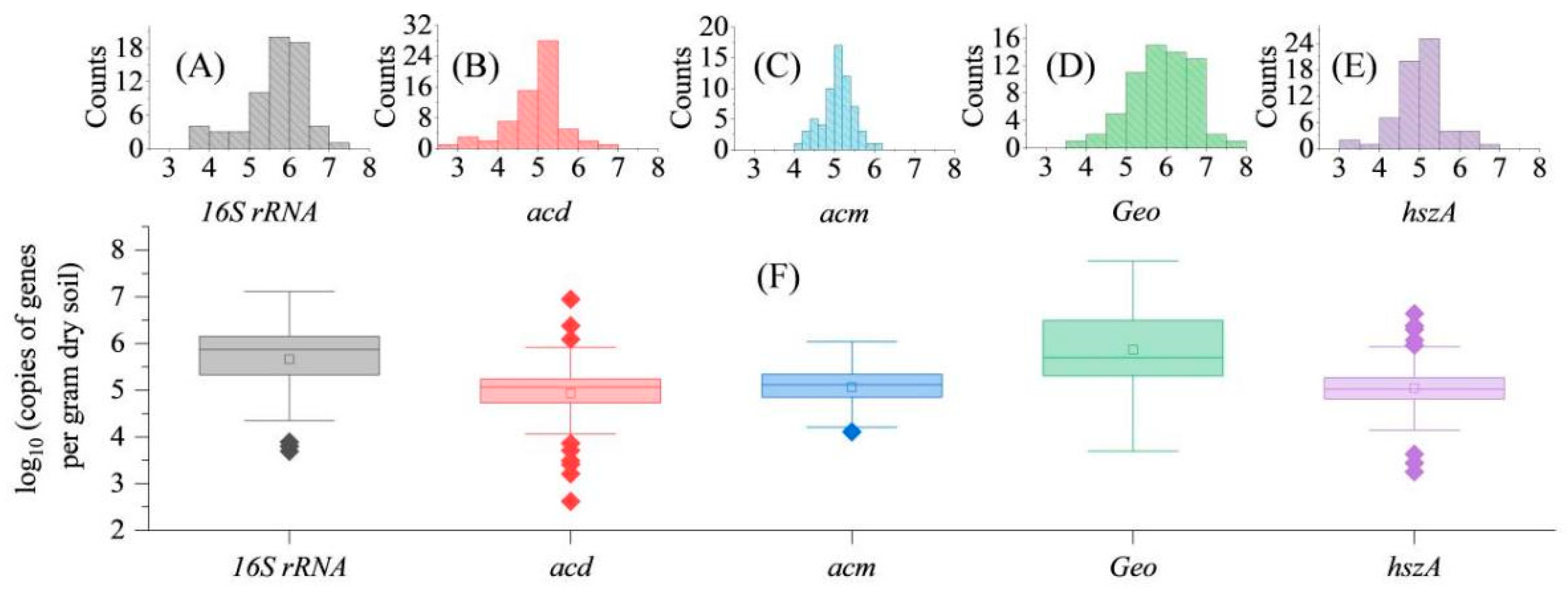
3.2. Absolute and Relative Target Gene Abundance
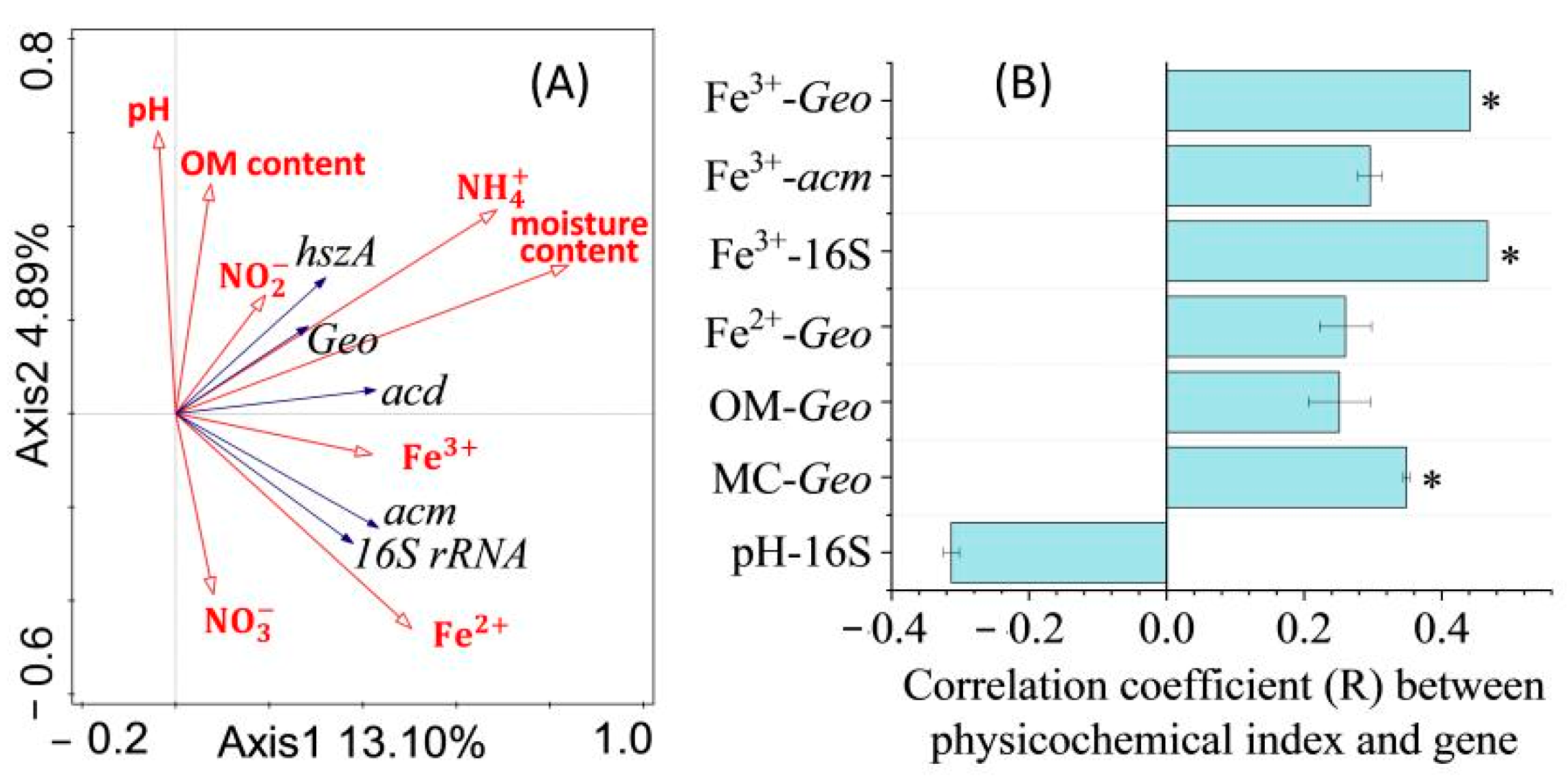
3.3. Effects of Physicochemical Indices on Target Genes
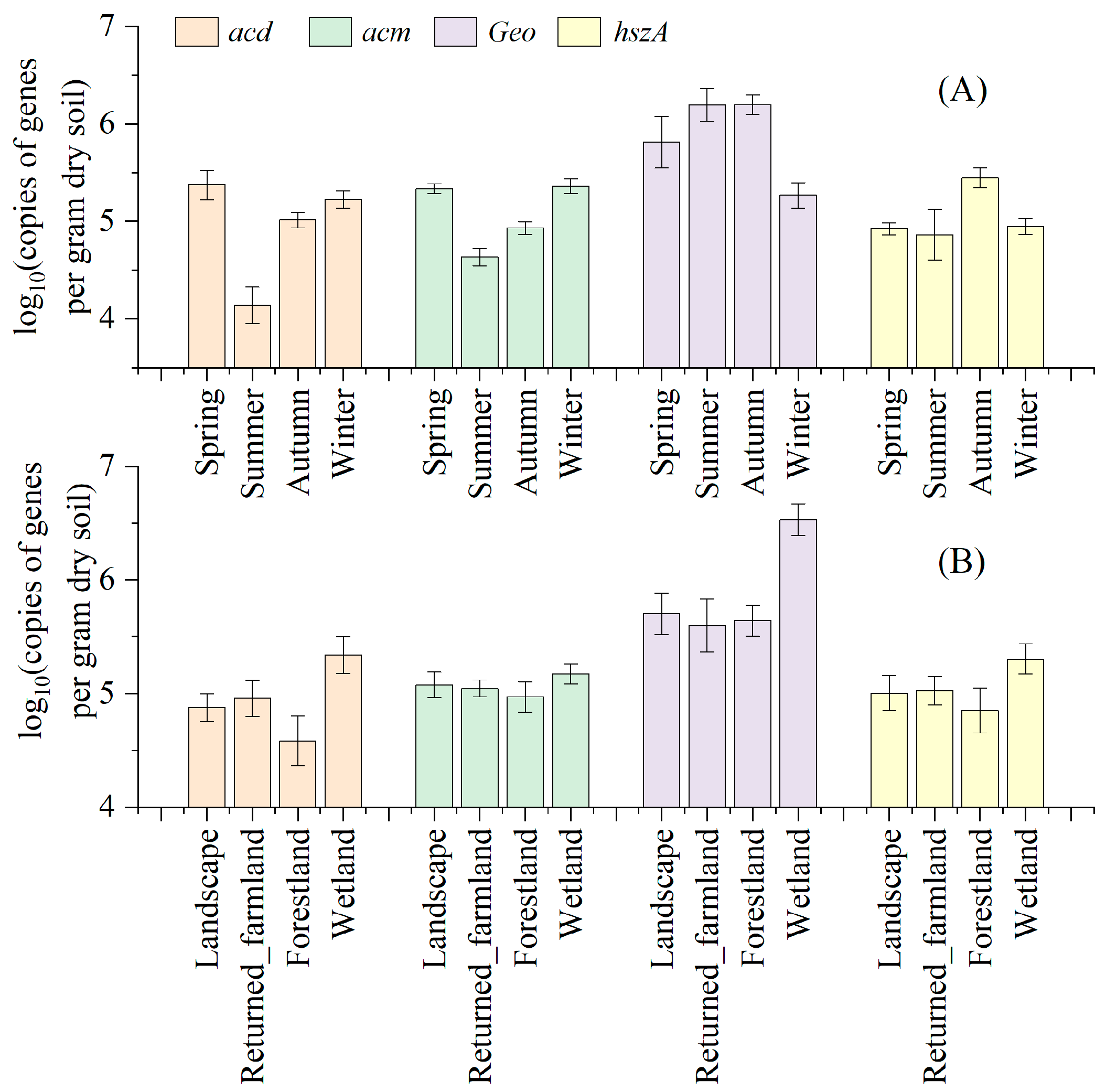
3.4. Seasonal and Spatial Variations in the Target Genes
3.5. Microbial Richness and Diversity
3.6. Microbial Community Structure and Succession
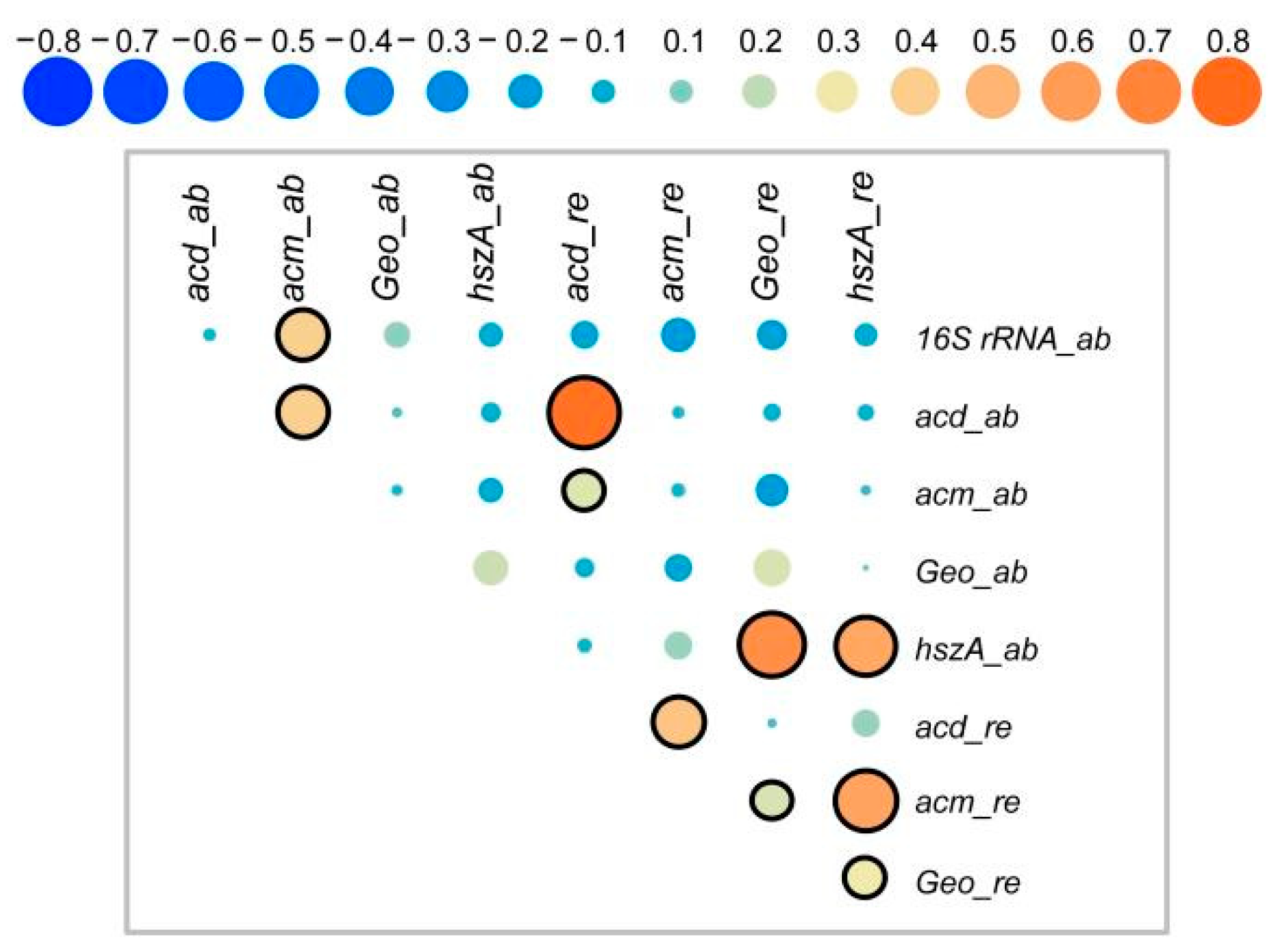
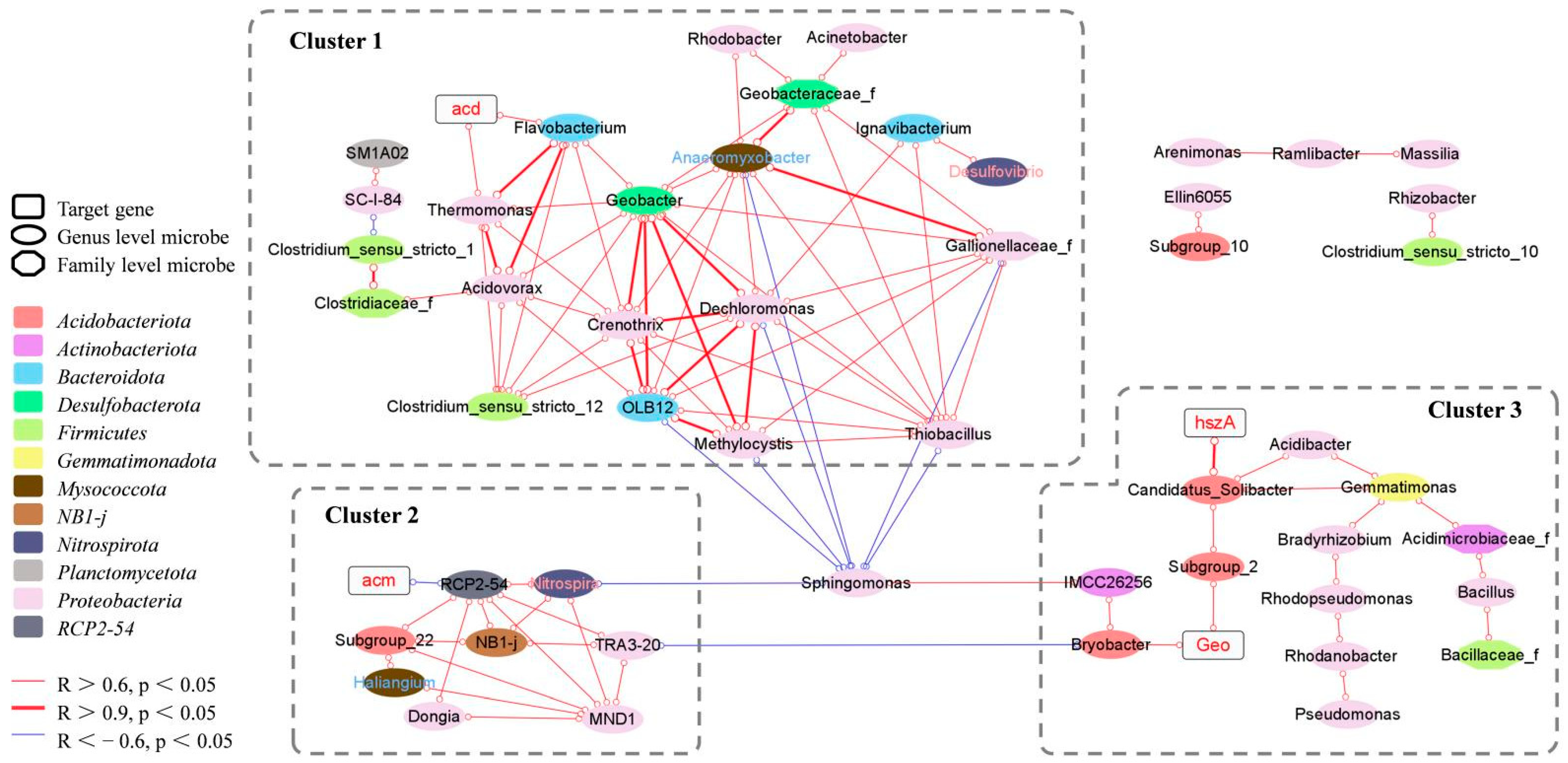
3.7. Correlations between Target Genes and Microbial Community Structure
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Wan, L.; Liu, H.; Wang, X. Anaerobic ammonium oxidation coupled to Fe(III) reduction: Discovery, mechanism and application prospects in wastewater treatment. Sci. Total Environ. 2022, 818, 151687. [Google Scholar] [CrossRef] [PubMed]
- Clement, J.-C.; Shrestha, J.; Ehrenfeld, J.G.; Jaffé, P. Ammonium oxidation coupled to dissimilatory reduction of iron under anaerobic conditions in wetland soils. Soil Biol. Biochem. 2005, 37, 2323–2328. [Google Scholar] [CrossRef]
- Tan, X.; Xie, G.J.; Nie, W.B.; Xing, D.F.; Liu, B.F.; Ding, J.; Ren, N.Q. Fe(III)-mediated anaerobic ammonium oxidation: A novel microbial nitrogen cycle pathway and potential applications. Crit. Rev. Environ. Sci. Tec. 2021, 21, 2112–2120. [Google Scholar] [CrossRef]
- Zhang, L.; Guan, Y.; Jiang, S.C. Investigations of soil autotrophic ammonia oxidizers in farmlands through genetics and big data analysis. Sci. Total Environ. 2021, 777, 146091. [Google Scholar] [CrossRef]
- Gallarotti, N.; Barthel, M.; Verhoeven, E.; Pereira, E.I.P.; Bauters, M.; Baumgartner, S.; Drake, T.W.; Boeckx, P.; Mohn, J.; Longepierre, M.; et al. In-depth analysis of N2O fluxes in tropical forest soils of the Congo Basin combining isotope and functional gene analysis. ISME J. 2021, 15, 3357–3374. [Google Scholar] [CrossRef]
- Toro, E.E.R.-D.; Valenzuela, E.I.; López-Lozano, N.E.; Cortés-Martínez, M.G.; Sánchez-Rodríguez, M.A.; Calvario-Martínez, O.; Sánchez-Carrillo, S.; Cervantes, F.J. Anaerobic ammonium oxidation linked to sulfate and ferric iron reduction fuels nitrogen loss in marine sediments. Biodegradation 2018, 29, 429–442. [Google Scholar] [CrossRef]
- Ding, B.; Li, Z.; Cai, M.; Lu, M.; Liu, W. Feammox is more important than anammox in anaerobic ammonium loss in farmland soils around Lake Taihu, China. Chemosphere 2022, 305, 135412. [Google Scholar] [CrossRef] [PubMed]
- Ding, B.; Zhang, H.; Luo, W.; Sun, S.; Cheng, F.; Li, Z. Nitrogen loss through denitrification, anammox and Feammox in a paddy soil. Sci. Total Environ. 2021, 773, 145601. [Google Scholar] [CrossRef]
- Xiong, Y.; Du, Y.; Deng, Y.; Ma, T.; Wang, Y. Feammox in alluvial-lacustrine aquifer system: Nitrogen/iron isotopic and biogeochemical evidences. Water Res. 2022, 222, 118867. [Google Scholar] [CrossRef]
- Huang, S.; Chen, C.; Peng, X.; Jaffé, P.R. Environmental factors affecting the presence of Acidimicrobiaceae and ammonium removal under iron-reducing conditions in soil environments. Soil Biol. Biochem. 2016, 98, 148–158. [Google Scholar] [CrossRef] [Green Version]
- Yao, Z.; Wang, C.; Song, N.; Wang, C.; Jiang, H. Oxidation of ammonium in aerobic wastewater by anoxic ferric iron-dependent ammonium oxidation (Feammox) in a biofilm reactor. Desalin. Water Treat. 2020, 173, 197–206. [Google Scholar] [CrossRef]
- Yang, Y.; Zhao, Z.; Zhang, Y. Anaerobic Ammonium Removal Pathway Driven by the Fe(II)/Fe(III) Cycle through Intermittent Aeration. Environ. Sci. Technol. 2021, 55, 7615–7623. [Google Scholar] [CrossRef] [PubMed]
- Roussel, F.; Alexandre, F. Landscape ecological enhancement and environmental inequalities in peri-urban areas, using flora as a socio-ecological indicator—The case of the greater Paris area. Landscape Urban Plan 2021, 210, 104062. [Google Scholar] [CrossRef]
- Pouya, S.; Aghlmand, M. Evaluation of urban green space per capita with new remote sensing and geographic information system techniques and the importance of urban green space during the COVID-19 pandemic. Environ. Monit. Assess. 2022, 194, 633. [Google Scholar] [CrossRef] [PubMed]
- Amani-Beni, M.; Zhang, B.; Xie, G.-D.; Xu, J. Impact of urban park’s tree, grass and waterbody on microclimate in hot summer days: A case study of Olympic Park in Beijing, China. Urban For. Urban Green. 2018, 32, 1–6. [Google Scholar] [CrossRef]
- Heo, S.; Bell, M.L. The influence of green space on the short-term effects of particulate matter on hospitalization in the US for 2000–2013. Environ. Res. 2019, 174, 61–68. [Google Scholar] [CrossRef]
- Balwant, P.; Jyothi, V.; Pujari, P.R.; Dhyani, S.; Verma, P.; Padmakar, C.; Quamar, R.; Ramesh, J.; Khare, S.; Mitkari, M. Tree root imaging by electrical resistivity tomography: Geophysical tools to improve understanding of deep root structure and rhizospheric processes. Trop. Eco. 2022, 63, 319–324. [Google Scholar] [CrossRef]
- Howard, E. Garden Cities of Tomorrow; Faber & Faber: London, UK, 1989. [Google Scholar]
- Ma, D.; Wang, J.; Xue, J.; Yue, Z.; Xia, S.; Song, L.; Gao, H. Effects of Soil pH on Gaseous Nitrogen Loss Pathway via Feammox Process. Sustainability 2021, 13, 10393. [Google Scholar] [CrossRef]
- Xiu, W.; Wu, M.; Nixon, S.L.; Lloyd, J.R.; Bassil, N.M.; Gai, R.; Zhang, T.; Su, Z.; Guo, H. Genome-Resolved Metagenomic Analysis of Groundwater: Insights into Arsenic Mobilization in Biogeochemical Interaction Networks. Environ. Sci. Technol. 2022, 56, 14. [Google Scholar] [CrossRef]
- Zhu, J.; Yan, X.; Zhou, L.; Li, N.; Liao, C.; Wang, X. Insight of bacteria and archaea in Feammox community enriched from different soils. Environ. Res. 2022, 203, 111802. [Google Scholar] [CrossRef]
- Yang, X.-R.; Li, H.; Su, J.-Q.; Zhou, G.-W. Anammox bacteria are potentially involved in anaerobic ammonium oxidation coupled to iron(III) reduction in the wastewater treatment system. Front. Microbiol. 2021, 10, 717249. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.; Li, J.; Wang, Y.; Xie, H.; Zhao, W.; Li, J. Feammox: A novel autotrophic nitrogen removal technology. Chem. Ind. Eng. Prog. 2022, 41, 391–399. [Google Scholar] [CrossRef]
- Yang, Y.; Xiao, C.; Lu, J.; Zhang, Y. Fe(III)/Fe(II) forwarding a new anammox-like process to remove high-concentration ammonium using nitrate as terminal electron acceptor. Water Res. 2020, 172, 115528. [Google Scholar] [CrossRef] [PubMed]
- Ding, B.; Chen, Z.; Li, Z.; Qin, Y.; Chen, S. Nitrogen loss through anaerobic ammonium oxidation coupled to Iron reduction from ecosystem habitats in the Taihu estuary region. Sci. Total Environ. 2019, 662, 600–606. [Google Scholar] [CrossRef]
- Ding, B.; Qin, Y.; Luo, W.; Li, Z. Spatial and seasonal distributions of Feammox from ecosystem habitats in the Wanshan region of the Taihu watershed, China. Chemosphere 2020, 239, 124742. [Google Scholar] [CrossRef] [PubMed]
- Ahmed, M.; Anwar, R.; Deng, D.; Garner, E.; Lin, L.S. Functional Interrelationships of Microorganisms in Iron-Based Anaerobic Wastewater Treatment. Microorganisms 2021, 9, 1039. [Google Scholar] [CrossRef]
- Qin, Y.; Ding, B.; Li, Z.; Chen, S. Variation of Feammox following ammonium fertilizer migration in a wheat-rice rotation area, Taihu Lake, China. Environ. Pollut. 2019, 252, 119–127. [Google Scholar] [CrossRef]
- Huang, S.; Jaffé, P.R. Isolation and characterization of an ammonium-oxidizing iron reducer: Acidimicrobiaceae sp. A6. PLoS ONE 2018, 13, e0194007. [Google Scholar] [CrossRef] [Green Version]
- Zhu, J.; Li, T.; Liao, C.; Li, N.; Wang, X. A promising destiny for Feammox: From biogeochemical ammonium oxidation to wastewater treatment. Sci. Total Environ. 2021, 790, 148038. [Google Scholar] [CrossRef]
- Xiong, Y.; Du, Y.; Deng, Y.; Ma, T.; Li, D.; Sun, X.; Liu, G.; Wang, Y. Contrasting sources and fate of nitrogen compounds in different groundwater systems in the Central Yangtze River Basin. Environ. Pollut. 2021, 290, 118119. [Google Scholar] [CrossRef]
- Zhu, T.-T.; Lai, W.-X.; Zhang, Y.-B.; Liu, Y.-W. Feammox process driven anaerobic ammonium removal of wastewater treatment under supplementing Fe(III) compounds. Sci. Total Environ. 2022, 804, 149965. [Google Scholar] [CrossRef] [PubMed]
- Yang, Y.; Peng, H.; Niu, J.; Zhao, Z.; Zhang, Y. Promoting nitrogen removal during Fe(III) reduction coupled to anaerobic ammonium oxidation (Feammox) by adding anthraquinone-2,6-disulfonate (AQDS). Environ. Pollut. 2019, 247, 973–979. [Google Scholar] [CrossRef]
- Li, X.; Hou, L.; Liu, M.; Zheng, Y.; Yin, G.; Lin, X.; Cheng, L.; Li, Y.; Hu, X. Evidence of Nitrogen Loss from Anaerobic Ammonium Oxidation Coupled with Ferric Iron Reduction in an Intertidal Wetland. Envrion. Sci. Technol. 2015, 49, 11560–11568. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Li, J.; Feng, J.; Zhai, S. Effects of Fe(III) on dissimilatory ferric reduction, nitrogen and phosphorus removal in activated sludge process. Environ. Eng. Manag. J. 2013, 12, 1345–1352. [Google Scholar] [CrossRef]
- Wu, J. and Loucks, OL. From balance of nature to hierarchical patch dynamics: A paradigm shift in ecology. Q. Rev. Biol. 1995, 70, 439–466. [Google Scholar] [CrossRef] [Green Version]
- Zhao, S.; Zhang, B.; Sun, X.; Yang, L. Hot spots and hot moments of nitrogen removal from hyporheic and riparian zones: A review. Sci. Total Environ. 2021, 762, 144168. [Google Scholar] [CrossRef]
- Huang, S.; Chen, C.; Jaffé, P. Seasonal distribution of nitrifiers and denitrifiers in urban river sediments affected by agricultural activities. Sci. Total. Environ. 2018, 642, 1282–1291. [Google Scholar] [CrossRef]
- Krichels, A.H.; Sipic, E.; Yang, W.H. Iron Redox Reactions Can Drive Microtopographic Variation in Upland Soil Carbon Dioxide and Nitrous Oxide Emissions. Soil Syst. 2019, 3, 60. [Google Scholar] [CrossRef] [Green Version]
- Yang, W.H.; Weber, K.A.; Silver, W.L. Nitrogen loss from soil through anaerobic ammonium oxidation coupled to iron reduction. Nat. Geosci. 2012, 5, 538–541. [Google Scholar] [CrossRef] [Green Version]
- Guan, Q.S.; Cao, W.Z.; Wang, M.; Wu, G.J.; Wang, F.F.; Jiang, C.; Tao, Y.R.; Gao, Y. Nitrogen loss through anaerobic ammonium oxidation coupled with iron reduction in a mangrove wetland. Eur. J. Siol Sci. 2018, 69, 732–741. [Google Scholar] [CrossRef]
- Ding, B.; Li, Z.; Qin, Y. Nitrogen loss from anaerobic ammonium oxidation coupled to Iron(III) reduction in a riparian zone. Environ. Pollut. 2017, 231, 379–386. [Google Scholar] [CrossRef] [PubMed]
- Ding, L.-J.; An, X.-L.; Li, S.; Zhang, G.-L.; Zhu, Y.-G. Nitrogen Loss through Anaerobic Ammonium Oxidation Coupled to Iron Reduction from Paddy Soils in a Chronosequence. Environ. Sci. Technol. 2014, 48, 10641–10647. [Google Scholar] [CrossRef]
- Huang, J.; Liu, C.; Price, G.; Li, Y.; Wang, Y. Identification of a novel heavy metal resistant Ralstonia strain and its growth response to cadmium exposure. J. Hazard. Mater. 2021, 416, 125942. [Google Scholar] [CrossRef] [PubMed]
- Cho, C.-W.; Zhao, Y.; Choi, J.-W.; Kim, J.-A.; Bediako, J.K.; Lin, S.; Song, M.-H.; Yun, Y.-S. Prediction of organic pollutant removal using Corynebacterium glutamicum fermentation waste. Environ. Res. 2021, 192, 110271. [Google Scholar] [CrossRef] [PubMed]
- Shu, D.; He, Y.; Yue, H.; Yang, S. Effects of Fe(II) on microbial communities, nitrogen transformation pathways and iron cycling in the anammox process: Kinetics, quantitative molecular mechanism and metagenomic analysis. RSC Adv. 2016, 6, 68005–68016. [Google Scholar] [CrossRef]
- Li, W.; Chen, A.; Shang, C.; Zhang, X.; Chai, Y.; Luo, S.; Shao, J.; Peng, L. Remediation of thiamethoxam contaminated wetland soil by Phanerochaete chrysosporium and the response of microorganisms. J. Environ. Chem. Eng. 2022, 10, 108333. [Google Scholar] [CrossRef]
- Wang, C.; Jia, Y.; Wang, Q.; Yan, F.; Wu, M.; Li, X.; Fang, W.; Xu, F.; Liu, H.; Qiu, Z. Responsive change of crop-specific soil bacterial community to cadmium in farmlands surrounding mine area of Southeast China. Environ. Res. 2022, 214 Pt 1, 113748. [Google Scholar] [CrossRef]
- Cao, J.; Li, N.; Jiang, J.; Xu, Y.; Zhang, B.; Luo, X.; Hu, Y. Activated carbon as an insoluble electron shuttle to enhance the anaerobic ammonium oxidation coupled with Fe(III) reduction process. Environ. Res. 2022, 204 Pt A, 111972. [Google Scholar] [CrossRef]
- Fang, Y.; Liu, J.; Yang, J.; Wu, G.; Hua, Z.; Dong, H.; Hedlund, B.P.; Baker, B.J.; Jiang, H. Compositional and Metabolic Responses of Autotrophic Microbial Community to Salinity in Lacustrine Environments. mSystems 2022, 7, e0033522. [Google Scholar] [CrossRef]
- Prakash, A.A.; Rajasekar, A.; Sarankumar, R.K.; AlSalhi, M.S.; Devanesan, S.; Aljaafreh, M.J.; Govarthanan, M.; Sayed, S.R. Metagenomic analysis of microbial community and its role in bioelectrokinetic remediation of tannery contaminated soil. J. Hazard. Mater. 2021, 412, 125133. [Google Scholar] [CrossRef]
- Mohammadipour, Z.; Enayatizamir, N.; Ghezelbash, G.; Abdolamir, M. Bacterial Diversity and Chemical Properties of Wheat Straw-Based Compost Leachate and Screening of Cellulase Producing Bacteria. Waste Biomass Valor. 2021, 12, 1293–1302. [Google Scholar] [CrossRef]
- Li, K.; Wang, Y.; Li, X.; Huang, Y.; Niu, Q.; Meng, Q.; Yang, H.; Li, Q. In-situ generation of H2O2 by zero valent iron to control depolymerization of lignocellulose in composting niche. Chemosphere 2022, 302, 134908. [Google Scholar] [CrossRef]
- Leung, P.M.; Daebeler, A.; Chiri, E.; Iresha, H.; David, L.G.; Ralf, B.S.; Holger, D.; Chris, G. A nitrite-oxidising bacterium constitutively consumes atmospheric hydrogen. ISME J. 2022, 16, 2213–2219. [Google Scholar] [CrossRef]
- Zhang, X.; Wei, D.; Zhang, H.; He, Y.; Zhang, S.; Dai, J.; Wen, X. Comprehensive analysis of the impacts of iron-based nanoparticles and ions on Anammox process. Biochem. Eng. J. 2022, 180, 108371. [Google Scholar] [CrossRef]
- Yang, H.; Deng, L.; Xiao, Y.; Yang, H.; Wang, H.; Zheng, D. Construction of autotrophic nitrogen removal system based on zero-valent iron (ZVI): Performance and mechanism. Water Sci. Technol. 2020, 82, 2990–3002. [Google Scholar] [CrossRef] [PubMed]
- Liu, D.; Zhang, S.; Fei, C.; Ding, X. Impacts of straw returning and N application on NH4+-N loss, microbially reducible Fe(III) and bacterial community composition in saline-alkaline paddy soils. Appl. Soil. Ecol. 2021, 168, 104115. [Google Scholar] [CrossRef]
- Liang, Z.; Shi, J.; Yang, W.; Dai, L.; Dai, X. Coupling anammox and feammox via polymeric ferric sulfate: An efficient and aeration-saving way for nitrogen removal. J. Clean. Prod. 2022, 355, 131788. [Google Scholar] [CrossRef]
- Li, J.; Zeng, W.; Liu, H.; Zhan, M.; Miao, H. Achieving deep autotrophic nitrogen removal in aerated biofilter driven by sponge iron: Performance and mechanism. Environ. Res. 2022, 213, 113653. [Google Scholar] [CrossRef]
- Ding, B.; Luo, W.; Qin, Y.; Li, Z. Effects of the addition of nitrogen and phosphorus on anaerobic ammonium oxidation coupled with iron reduction (Feammox) in the farmland soils. Sci. Total Environ. 2022, 737, 139849. [Google Scholar] [CrossRef]
- Liang, S. Study on Treatment of Nitrogen containing Wastewater by Dissimilar Iron Reduction Coupled with Anaerobic Ammonium Oxidation. Master’s Thesis, Dalian University of Technology, Dalian, China, 2021. [Google Scholar] [CrossRef]
- Wang, W.; Ding, B.; Hu, Y.; Zhang, H.; He, Y.; She, Y.; Li, Z. Evidence for the occurrence of Feammox coupled with nitrate-dependent Fe(II) oxidation in natural enrichment cultures. Chemosphere 2022, 303 Pt 1, 134903. [Google Scholar] [CrossRef]
- Xie, F.; Zhao, B.; Cui, Y.; Ma, X.; Li, D.; Yue, X. Enhancing nitrogen removal performance of anammox process after short-term pH and temperature shocks by coupling with iron-carbon micro-electrolysis. J. Clean. Prod. 2021, 289, 125753. [Google Scholar] [CrossRef]
- Hu, L.; Cheng, X.; Qi, G.; Zheng, M.; Dang, Y.; Li, J.; Xu, K. Achieving ammonium removal through anammox-derived Feammox with low demand of Fe(III). Front. Microbiol. 2022, 27, 918634. [Google Scholar] [CrossRef] [PubMed]
- Hu, Y.; Li, N.; Jiang, J.; Luo, X.; Cao, J. Simultaneous Feammox and anammox process facilitated by activated carbon as an electron shuttle for autotrophic biological nitrogen removal. Front. Environ. Sci. Eng. 2022, 16, 90. [Google Scholar] [CrossRef]
- Li, Y.; Dong, R.; Guo, J.; Wang, L.; Zhao, J. Effects of Mn2+ and humic acid on microbial community structures, functional genes for nitrogen and phosphorus removal, and heavy metal resistance genes in wastewater treatment. J. Environ. Manag. 2022, 313, 115028. [Google Scholar] [CrossRef] [PubMed]
- Thao, T.P.; Hatamoto, M.; Aoki, M.; Watari, T.; Syutsubo, K.; Yamaguchi, T. Effect of inoculum sources on autotrophic nitrogen removal in anaerobic hollow fiber membrane reactors. Environ. Technol. Innov. 2022, 26, 102375. [Google Scholar] [CrossRef]
- Huang, S.; Jaffé, P.R. Characterization of incubation experiments and development of an enrichment culture capable of ammonium oxidation under iron reducing conditions. Biogeosciences 2014, 11, 12295–12321. [Google Scholar] [CrossRef] [Green Version]
- Zhao, Z. Spatiotemporal Distribution of Anaerobic Ammonia Oxidizing Bacteria and Microbial Communities in the Sediments of Xiangxi River, Three Gorges Reservoir Region; China Three Gorges University: Yichang, China, 2020. [Google Scholar] [CrossRef]
- Holmes, D.E.; Finneran, K.T.; O’Neil, R.A.; Lovley, D.R. Enrichment of Members of the Family Geobacteraceae Associated with Stimulation of Dissimilatory Metal Reduction in Uranium-Contaminated Aquifer Sediments. Appl. Environ. Microbiol. 2002, 68, 2300–2306. [Google Scholar] [CrossRef] [Green Version]
- Wang, X.; Wang, W.; Zhang, Y.; Sun, Z.; Zhang, J.; Liang, D.; Hou, L.; Zhang, K.; Li, J. Effect of environmental factors on nitrogen removal and functional genes expression of strain Pseudomonas sp. China Environ. Sci. 2019, 39, 4377–4386. [Google Scholar] [CrossRef]

| Factor | Absolute Abundance | Relative Abundance | |||||||
|---|---|---|---|---|---|---|---|---|---|
| 16S rRNA | acd | acm | Geo | hszA | acd | acm | Geo | hszA | |
| Season | + | + | + | + | + | − | − | + | + |
| Habitat | − | + | − | + | − | − | − | + | − |
| Season–habitat interaction | + | − | − | + | + | − | − | + | + |
| Sample | Chao1 | Shannon | Simpson | Good’s Coverage (%) |
|---|---|---|---|---|
| Spr_Landscape | 1550.71 | 9.64 | 1.00 | 99.90 |
| Spr_Retur_farm | 1388.68 | 9.25 | 1.00 | 99.93 |
| Spr_Forstland | 1317.11 | 9.53 | 1.00 | 99.95 |
| Spr_Wetland | 1537.50 | 9.23 | 0.99 | 99.92 |
| Sum_Landscape | 1599.68 | 9.65 | 1.00 | 99.91 |
| Sum_Retur_farm | 1736.53 | 10.02 | 1.00 | 99.93 |
| Sum_Forstland | 1119.17 | 8.52 | 0.99 | 99.96 |
| Sum_Wetland | 1259.06 | 8.17 | 0.98 | 99.93 |
| Aut_Landscape | 1757.66 | 10.11 | 1.00 | 99.90 |
| Aut_Retur_farm | 1778.11 | 10.04 | 1.00 | 99.89 |
| Aut_Forstland | 1013.08 | 9.01 | 1.00 | 99.98 |
| Aut_Wetland | 1722.62 | 9.64 | 1.00 | 99.91 |
| Win_Landscape | 1583.10 | 9.63 | 1.00 | 99.92 |
| Win_Retur_farm | 904.74 | 6.05 | 0.84 | 99.92 |
| Win_Forstland | 1697.19 | 10.00 | 1.00 | 99.92 |
| Win_Wetland | 1191.20 | 9.02 | 1.00 | 99.96 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chen, M.; Ma, X.; Wei, S.; An, X.; Li, Y.; Liang, L.; Jiang, T. Seasonal and Spatial Variations in Functional Genes and Microbial Community of Feammox and Its Associated Processes in Urban Green Heart Soil. Water 2023, 15, 1024. https://doi.org/10.3390/w15061024
Chen M, Ma X, Wei S, An X, Li Y, Liang L, Jiang T. Seasonal and Spatial Variations in Functional Genes and Microbial Community of Feammox and Its Associated Processes in Urban Green Heart Soil. Water. 2023; 15(6):1024. https://doi.org/10.3390/w15061024
Chicago/Turabian StyleChen, Maoxia, Xuguang Ma, Shiqian Wei, Xin An, Yanjun Li, Liye Liang, and Tao Jiang. 2023. "Seasonal and Spatial Variations in Functional Genes and Microbial Community of Feammox and Its Associated Processes in Urban Green Heart Soil" Water 15, no. 6: 1024. https://doi.org/10.3390/w15061024






