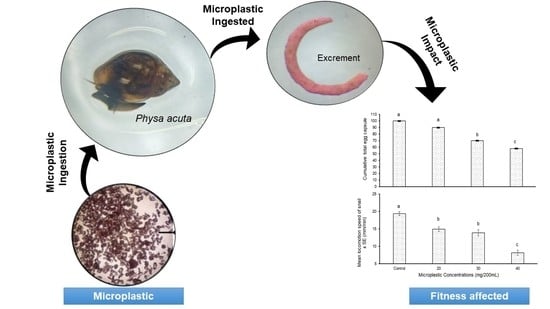
Microplastics Affect Rates of Locomotion and Reproduction via Dietary Uptake in Globally Invasive Snail Physa acuta
Abstract
:1. Introduction
2. Materials and Methods
2.1. Preparation and Characterization of MPs
2.2. Test Organism
2.3. Experimental Setup
2.3.1. Short Duration Experiment
2.3.2. Long Duration Experimental Set up
- (a)
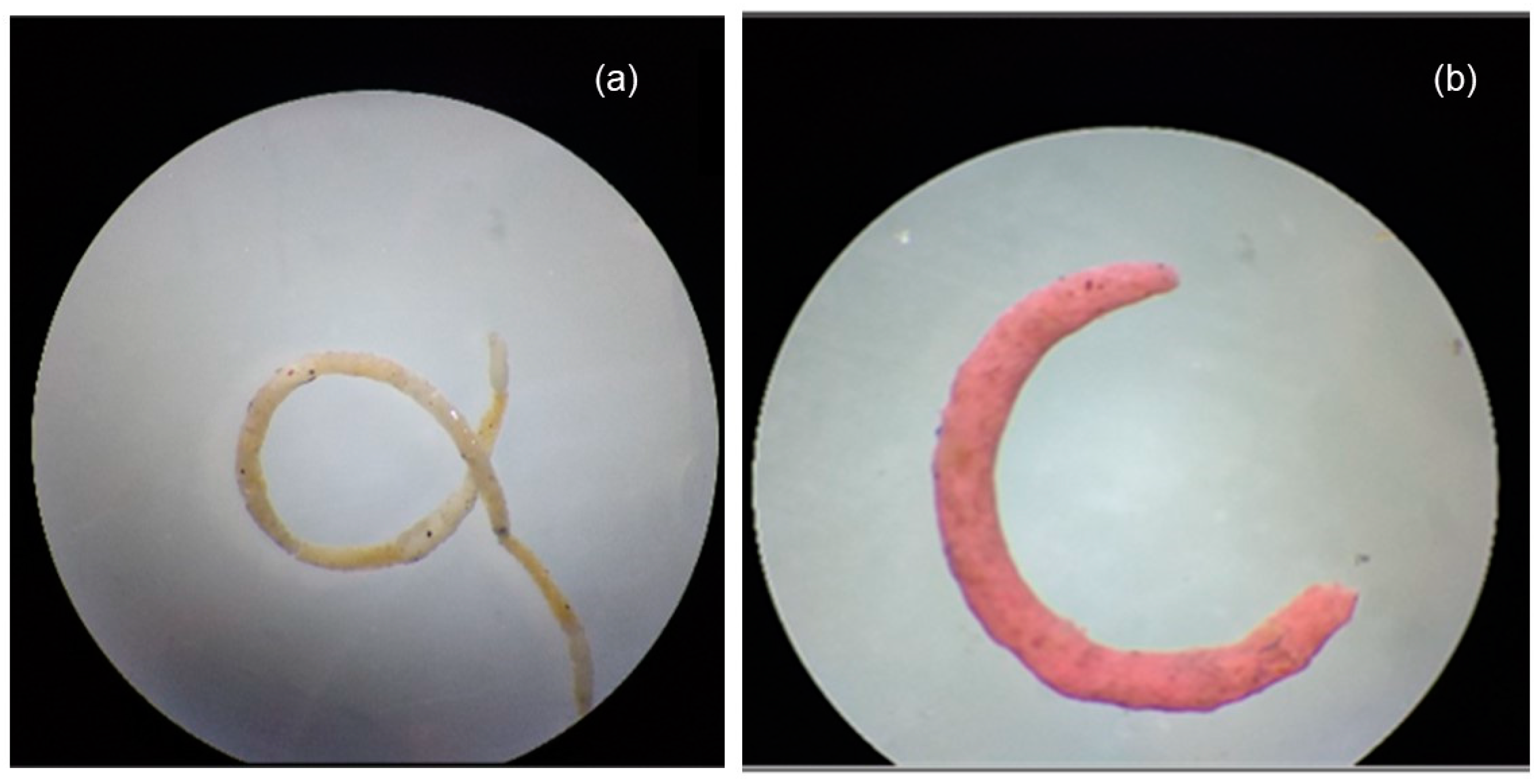
- Egg capsule production
- (b)
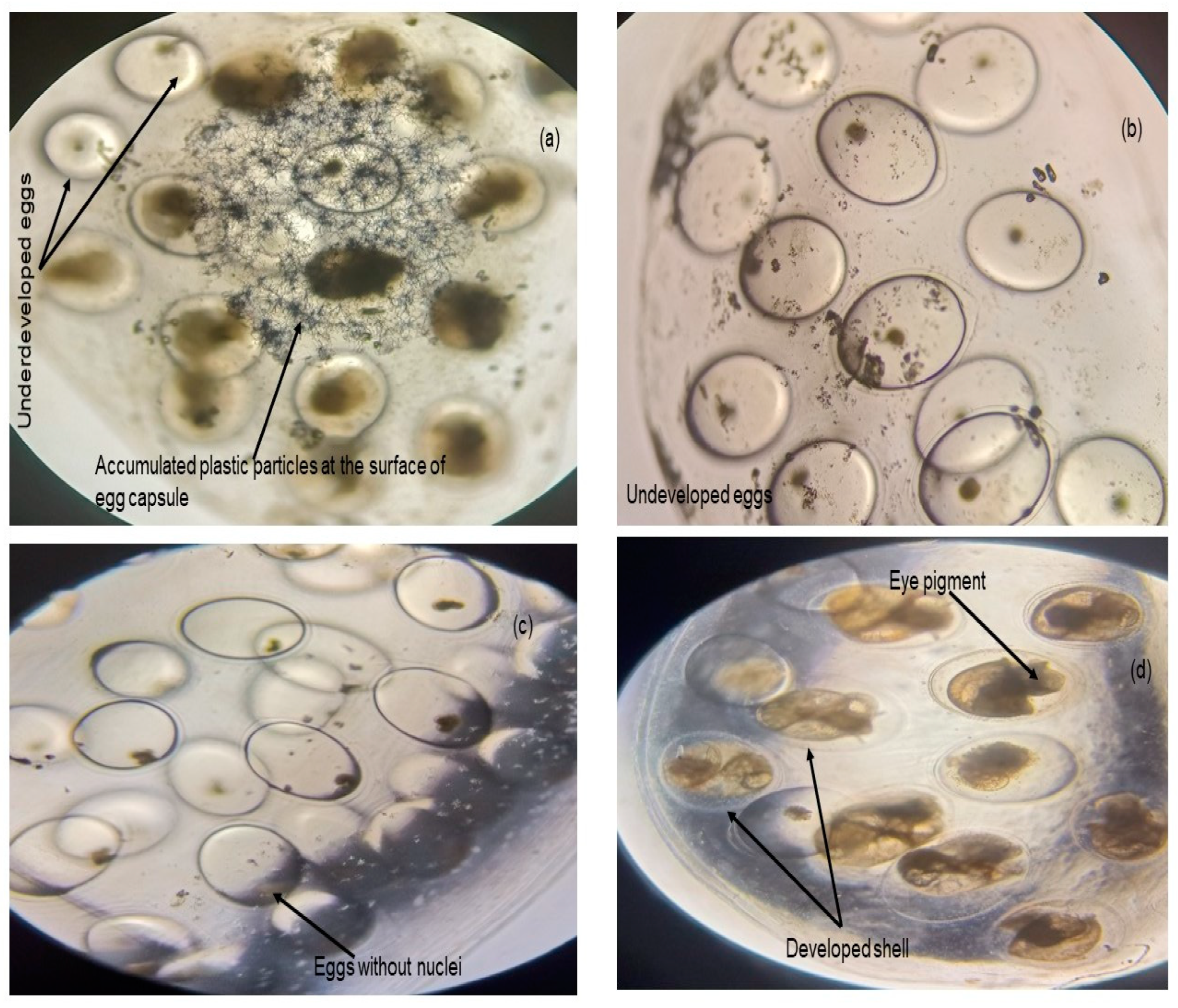
- Egg hatching success
2.4. Statistical Analysis
3. Results
3.1. Short Term Experiment
3.1.1. Experiment I
3.1.2. Experiment II
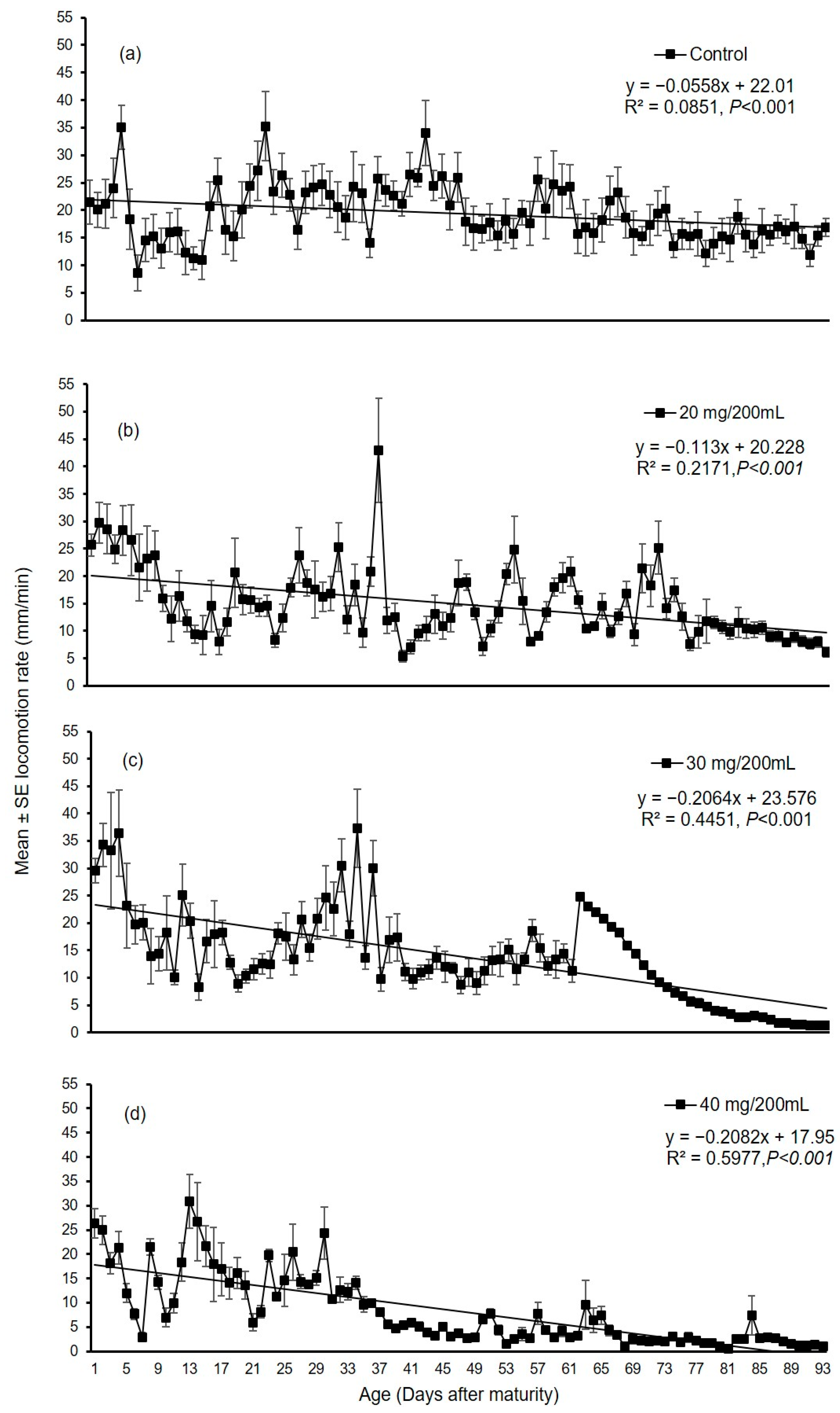
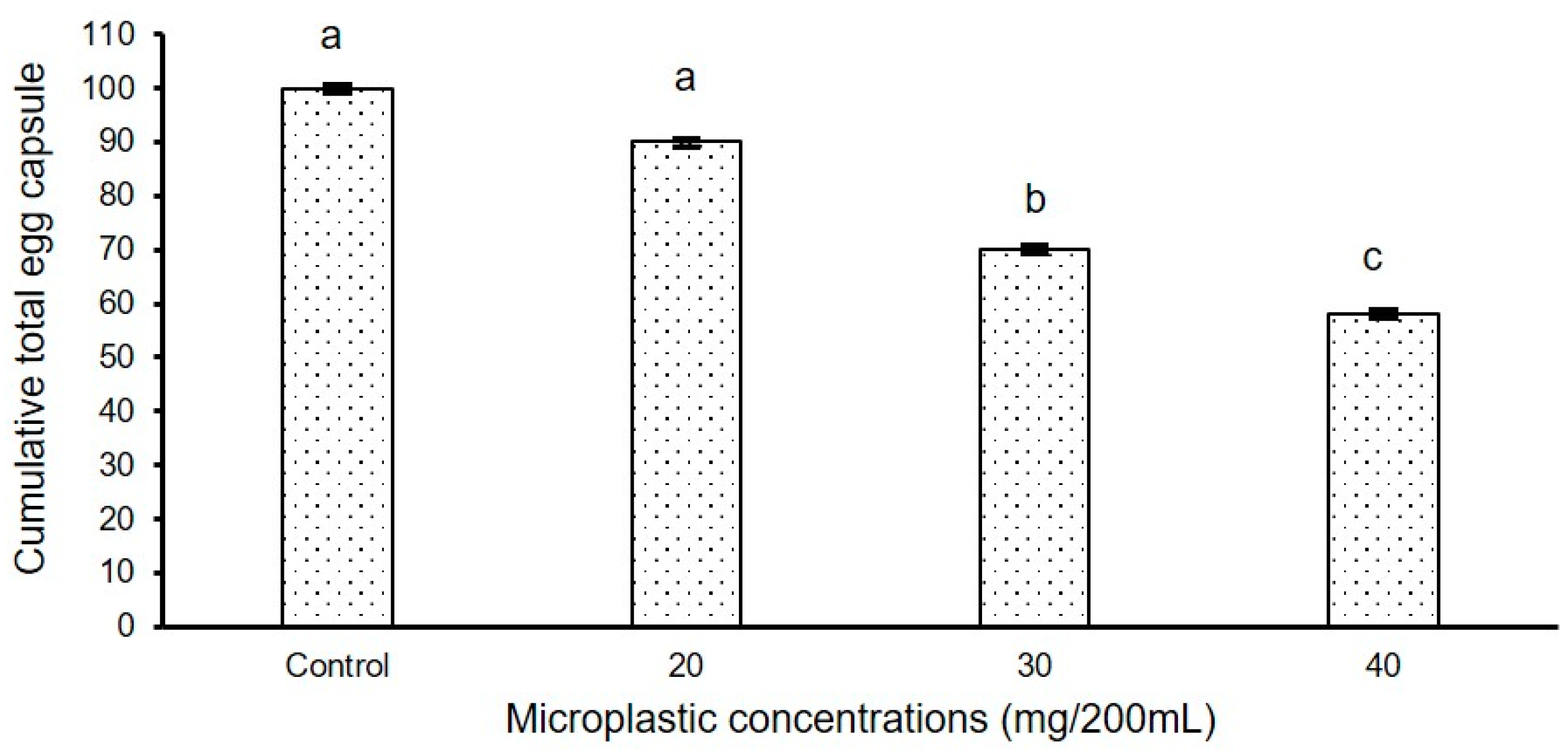
3.2. Long Duration Experiment
3.2.1. Experiment I
3.2.2. Experiment II
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Avio, C.G.; Gorbi, S.; Regoli, F. Plastics and Microplastics in the Oceans: From Emerging Pollutants to Emerged Threat. Mar. Environ. Res. 2017, 128, 2–11. [Google Scholar] [CrossRef]
- Andrady, A.L. Microplastics in the Marine Environment. Mar. Pollut. Bull. 2011, 62, 1596–1605. [Google Scholar] [CrossRef]
- Yuan, W.; Liu, X.; Wang, W.; Di, M.; Wang, J. Microplastic Abundance, Distribution and Composition in Water, Sediments, and Wild Fish from Poyang Lake, China. Ecotoxicol. Environ. Saf. 2019, 170, 180–187. [Google Scholar] [CrossRef]
- Tang, Y.; Liu, Y.; Chen, Y.; Zhang, W.; Zhao, J.; He, S.; Yang, C.; Zhang, T.; Tang, C.; Zhang, C.; et al. A Review: Research Progress on Microplastic Pollutants in Aquatic Environments. Sci. Total Environ. 2021, 766, 142572. [Google Scholar] [CrossRef]
- Cheung, P.K.; Hung, P.L.; Fok, L. River Microplastic Contamination and Dynamics upon a Rainfall Event in Hong Kong, China. Environ. Process. 2019, 6, 253–264. [Google Scholar] [CrossRef]
- Pandey, B.; Pathak, J.; Singh, P.; Kumar, R.; Kumar, A.; Kaushik, S.; Thakur, T.K. Microplastics in the Ecosystem: An Overview on Detection, Removal, Toxicity Assessment, and Control Release. Water 2022, 15, 51. [Google Scholar] [CrossRef]
- Kumar, P.; Inamura, Y.; Bao, P.N.; Abeynayaka, A.; Dasgupta, R.; Abeynayaka, H.D.L. Microplastics in Freshwater Environment in Asia: A Systematic Scientific Review. Water 2022, 14, 1737. [Google Scholar] [CrossRef]
- Kumar, R.; Nesemann, H.; Sharma, G.; Tseng, L.-C.; Prabhakar, A.K.; Roy, S.P. Community Structure of Macrobenthic Invertebrates in the River Ganga in Bihar, India. Aquat. Ecosyst. Health Manag. 2013, 16, 385–394. [Google Scholar] [CrossRef]
- Lusher, A.L.; Welden, N.A.; Sobral, P.; Cole, M. Sampling, Isolating and Identifying Microplastics Ingested by Fish and Invertebrates. In Analysis of Nanoplastics and Microplastics in Food, 1st ed.; Food Analysis and Properties; Nollet, L.M.L., Siddiqi, K.S., Eds.; CRC Press: Boca Raton, FL, USA, 2020; pp. 119–148. ISBN 978-0-429-46959-6. [Google Scholar]
- Nesemann, H.F.; Sharma, G.; Kumar, R.; Sheetal, A.; Roy, S. Do the Rivers of Chota Nagpur Dry Forests (Jharkhand), Damodar and Subarnarekha Differ in Biodiversity of Aquatic Macrozoobenthos, Functional Feeding Groups and Biological Water Quality? Aquat. Ecosyst. Health Manag. 2017, 20, 116–129. [Google Scholar] [CrossRef]
- Schindler, D.E.; Scheuerell, M.D. Habitat Coupling in Lake Ecosystems. Oikos 2002, 98, 177–189. [Google Scholar] [CrossRef] [Green Version]
- Gyllström, M.; Hansson, L.-A. Dormancy in Freshwater Zooplankton: Induction, Termination and the Importance of Benthic-Pelagic Coupling. Aquat. Sci. 2004, 66, 274–295. [Google Scholar] [CrossRef]
- Wagner, M.; Scherer, C.; Alvarez-Muñoz, D.; Brennholt, N.; Bourrain, X.; Buchinger, S.; Fries, E.; Grosbois, C.; Klasmeier, J.; Marti, T.; et al. Microplastics in Freshwater Ecosystems: What We Know and What We Need to Know. Environ. Sci. Eur. 2014, 26, 12. [Google Scholar] [CrossRef] [Green Version]
- Baun, A.; Hartmann, N.B.; Grieger, K.; Kusk, K.O. Ecotoxicity of Engineered Nanoparticles to Aquatic Invertebrates: A Brief Review and Recommendations for Future Toxicity Testing. Ecotoxicology 2008, 17, 387–395. [Google Scholar] [CrossRef]
- Begon, M.; Howarth, R.W.; Townsend, C.R. Essentials of Ecology, 4th ed.; Wiley: Hoboken, NJ, USA, 2014; ISBN 978-0-470-90913-3. [Google Scholar]
- Ibrahim, M.M. Energy Allocation Patterns in Biomphalaria Alexandrina Snails in Response to Cadmium Exposure and Schistosoma Mansoni Infection. Exp. Parasitol. 2006, 112, 31–36. [Google Scholar] [CrossRef]
- Mahmoud, K.M.A.; Abu Taleb, H.M.A. Fresh Water Snails as Bioindicator for Some Heavy Metals in the Aquatic Environment. Afr. J. Ecol. 2013, 51, 193–198. [Google Scholar] [CrossRef]
- Vinarski, M.V. The History of an Invasion: Phases of the Explosive Spread of the Physid Snail Physella Acuta through Europe, Transcaucasia and Central Asia. Biol. Invasions 2017, 19, 1299–1314. [Google Scholar] [CrossRef]
- Sánchez-Argüello, P.; Fernández, C.; Tarazona, J.V. Assessing the Effects of Fluoxetine on Physa acuta (Gastropoda, Pulmonata) and Chironomus riparius (Insecta, Diptera) Using a Two-Species Water–Sediment Test. Sci. Total Environ. 2009, 407, 1937–1946. [Google Scholar] [CrossRef]
- Camargo, J.A.; Alonso, Á. Ecotoxicological Assessment of the Impact of Fluoride (F−) and Turbidity on the Freshwater Snail Physella Acuta in a Polluted River Receiving an Industrial Effluent. Environ. Sci. Pollut. Res. 2017, 24, 15667–15677. [Google Scholar] [CrossRef]
- Spyra, A.; Cieplok, A.; Strzelec, M.; Babczyńska, A. Freshwater Alien Species Physella acuta (Draparnaud, 1805)—A Possible Model for Bioaccumulation of Heavy Metals. Ecotoxicol. Environ. Saf. 2019, 185, 109703. [Google Scholar] [CrossRef]
- Morales, M.; Martínez-Paz, P.; Sánchez-Argüello, P.; Morcillo, G.; Martínez-Guitarte, J.L. Bisphenol A (BPA) Modulates the Expression of Endocrine and Stress Response Genes in the Freshwater Snail Physa acuta. Ecotoxicol. Environ. Saf. 2018, 152, 132–138. [Google Scholar] [CrossRef]
- Cole, M.; Lindeque, P.; Fileman, E.; Halsband, C.; Galloway, T.S. The Impact of Polystyrene Microplastics on Feeding, Function and Fecundity in the Marine Copepod Calanus helgolandicus. Environ. Sci. Technol. 2015, 49, 1130–1137. [Google Scholar] [CrossRef]
- Murphy, F.; Ewins, C.; Carbonnier, F.; Quinn, B. Wastewater Treatment Works (WwTW) as a Source of Microplastics in the Aquatic Environment. Environ. Sci. Technol. 2016, 50, 5800–5808. [Google Scholar] [CrossRef] [Green Version]
- Ziajahromi, S.; Kumar, A.; Neale, P.A.; Leusch, F.D.L. Impact of Microplastic Beads and Fibers on Waterflea (Ceriodaphnia Dubia) Survival, Growth, and Reproduction: Implications of Single and Mixture Exposures. Environ. Sci. Technol. 2017, 51, 13397–13406. [Google Scholar] [CrossRef] [Green Version]
- Bousset, L.; Henry, P.Y.; Sourrouille, P.; Jarne, P. Population Biology of the Invasive Freshwater Snail Physa acuta Approached through Genetic Markers, Ecological Characterization and Demography: Genetics and Demography in a snail. Mol. Ecol. 2004, 13, 2023–2036. [Google Scholar] [CrossRef]
- Kawata, M.; Ishigami, H. The Growth of Juvenile Snails in Water Conditioned by Snails of a Different Species. Oecologia 1992, 91, 245–248. [Google Scholar] [CrossRef]
- Central Pollution Control Board. 2019. Available online: https://cpcb.nic.in/wqstandards/ (accessed on 13 January 2021).
- Case, T.J. An Illustrated Guide to Theoretical Ecology; Oxford University Press: Oxford, NY, USA, 1999; 464p. [Google Scholar]
- Cunningham, E.M.; Cuthbert, R.N.; Coughlan, N.E.; Kregting, L.; Cairnduff, V.; Dick, J.T.A. Microplastics Do Not Affect the Feeding Rates of a Marine Predator. Sci. Total Environ. 2021, 779, 146487. [Google Scholar] [CrossRef]
- Drago, C.; Pawlak, J.; Weithoff, G. Biogenic Aggregation of Small Microplastics Alters Their Ingestion by a Common Freshwater Micro-Invertebrate. Front. Environ. Sci. 2020, 8, 574274. [Google Scholar] [CrossRef]
- Mbedzi, R.; Dalu, T.; Wasserman, R.J.; Murungweni, F.; Cuthbert, R.N. Functional Response Quantifies Microplastic Uptake by a Widespread African Fish Species. Sci. Total Environ. 2020, 700, 134522. [Google Scholar] [CrossRef]
- Santana, M.F.M.; Ascer, L.G.; Custódio, M.R.; Moreira, F.T.; Turra, A. Microplastic Contamination in Natural Mussel Beds from a Brazilian Urbanized Coastal Region: Rapid Evaluation through Bioassessment. Mar. Pollut. Bull. 2016, 106, 183–189. [Google Scholar] [CrossRef]
- Magni, S.; Gagné, F.; André, C.; Della Torre, C.; Auclair, J.; Hanana, H.; Parenti, C.C.; Bonasoro, F.; Binelli, A. Evaluation of Uptake and Chronic Toxicity of Virgin Polystyrene Microbeads in Freshwater Zebra Mussel Dreissena Polymorpha (Mollusca: Bivalvia). Sci. Total Environ. 2018, 631–632, 778–788. [Google Scholar] [CrossRef]
- Sussarellu, R.; Suquet, M.; Thomas, Y.; Lambert, C.; Fabioux, C.; Pernet, M.E.J.; Le Goïc, N.; Quillien, V.; Mingant, C.; Epelboin, Y.; et al. Oyster Reproduction Is Affected by Exposure to Polystyrene Microplastics. Proc. Natl. Acad. Sci. USA 2016, 113, 2430–2435. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pizzurro, F.; Recchi, S.; Nerone, E.; Salini, R.; Barile, N.B. Accumulation Evaluation of Potential Microplastic Particles in Mytilus galloprovincialis from the Goro Sacca (Adriatic Sea, Italy). Microplastics 2022, 1, 303–318. [Google Scholar] [CrossRef]
- Graham, E.R.; Thompson, J.T. Deposit- and Suspension-Feeding Sea Cucumbers (Echinodermata) Ingest Plastic Fragments. J. Exp. Mar. Biol. Ecol. 2009, 368, 22–29. [Google Scholar] [CrossRef]
- Stasolla, G.; Innocenti, G.; Galil, B.S. On the Diet of the Invasive Crab Charybdis Longicollis Leene, 1938 (Brachyura: Portunidae) in the Eastern Mediterranean Sea. Isr. J. Ecol. Evol. 2015, 61, 130–134. [Google Scholar] [CrossRef]
- Castro, G.B.; Bernegossi, A.C.; Felipe, M.C.; Ogura, A.P.; de Lima e Silva, M.R.; Corbi, J.J. Polyethylene Microplastics and Substrate Availability Can Affect Emergence Responses of the Freshwater Insect Chironomus sancticaroli. Ecotoxicology 2022, 31, 679–688. [Google Scholar] [CrossRef] [PubMed]
- Ziajahromi, S.; Kumar, A.; Neale, P.A.; Leusch, F.D.L. Environmentally Relevant Concentrations of Polyethylene Microplastics Negatively Impact the Survival, Growth and Emergence of Sediment-Dwelling Invertebrates. Environ. Pollut. 2018, 236, 425–431. [Google Scholar] [CrossRef]
- Horn, D.A.; Granek, E.F.; Steele, C.L. Effects of Environmentally Relevant Concentrations of Microplastic Fibers on Pacific Mole Crab (Emerita analoga) Mortality and Reproduction. Limnol. Oceanogr. Lett. 2020, 5, 74–83. [Google Scholar] [CrossRef] [Green Version]
- Philips, C.J.; Miller, K.L.; Eberhardt, L. Impact of Microplastic Fiber Pollution on Ramshorn Snail (Panorbella campanulata) Reproduction and Mortality. Symp. Undergrad. Res. Creat. Expr. (SOURCE) 2020, 877. Available online: https://scholar.valpo.edu/cus/877 (accessed on 10 July 2022).
- Scherer, C.; Brennholt, N.; Reifferscheid, G.; Wagner, M. Feeding Type and Development Drive the Ingestion of Microplastics by Freshwater Invertebrates. Sci. Rep. 2017, 7, 17006. [Google Scholar] [CrossRef] [Green Version]
- Redondo-Hasselerharm, P.E.; Falahudin, D.; Peeters, E.T.H.M.; Koelmans, A.A. Microplastic Effect Thresholds for Freshwater Benthic Macroinvertebrates. Environ. Sci. Technol. 2018, 52, 2278–2286. [Google Scholar] [CrossRef] [Green Version]
- Zhao, L.; Qu, M.; Wong, G.; Wang, D. Transgenerational Toxicity of Nanopolystyrene Particles in the Range of Μg L−1 in the Nematode Caenorhabditis elegans. Environ. Sci. Nano 2017, 4, 2356–2366. [Google Scholar] [CrossRef]
- Blarer, P.; Burkhardt-Holm, P. Microplastics Affect Assimilation Efficiency in the Freshwater Amphipod Gammarus Fossarum. Environ. Sci. Pollut. Res. 2016, 23, 23522–23532. [Google Scholar] [CrossRef] [PubMed]
- Straub, S.; Hirsch, P.E.; Burkhardt-Holm, P. Biodegradable and Petroleum-Based Microplastics Do Not Differ in Their Ingestion and Excretion but in Their Biological Effects in a Freshwater Invertebrate Gammarus fossarum. Int. J. Environ. Res. Public Health 2017, 14, 774. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- El-Naggar, H.A.; Khalaf Allah, H.M.M.; Masood, M.F.; Shaban, W.M.; Bashar, M.A.E. Food and Feeding Habits of Some Nile River Fish and Their Relationship to the Availability of Natural Food Resources. Egypt. J. Aquat. Res. 2019, 45, 273–280. [Google Scholar] [CrossRef]
- Ben-Ami, F.; Heller, J. Biological Control of Aquatic Pest Snails by the Black Carp Mylopharyngodon piceus. Biol. Control 2001, 22, 131–138. [Google Scholar] [CrossRef] [Green Version]
- Goeppner, S.R.; Roberts, M.E.; Beaty, L.E.; Luttbeg, B. Freshwater Snail Responses to Fish Predation Integrate Phenotypic Plasticity and Local Adaptation. Aquat. Ecol. 2020, 54, 309–322. [Google Scholar] [CrossRef]
- Turner, A.M.; Montgomery, S.L. Spatial and temporal scales of predator avoidance: Experiments with fish and snails. Ecology 2003, 84, 616–622. [Google Scholar] [CrossRef]
- Lei, L.; Wu, S.; Lu, S.; Liu, M.; Song, Y.; Fu, Z.; Shi, H.; Raley-Susman, K.M.; He, D. Microplastic Particles Cause Intestinal Damage and Other Adverse Effects in Zebrafish Danio rerio and Nematode Caenorhabditis elegans. Sci. Total Environ. 2018, 619–620, 1–8. [Google Scholar] [CrossRef]
- Pous, N.; Hidalgo, M.; Serra, T.; Colomer, J.; Colprim, J.; Salvadó, V. Assessment of Zooplankton-Based Eco-Sustainable Wastewater Treatment at Laboratory Scale. Chemosphere 2020, 238, 124683. [Google Scholar] [CrossRef]
- Bringer, A.; Thomas, H.; Prunier, G.; Dubillot, E.; Bossut, N.; Churlaud, C.; Clérandeau, C.; Le Bihanic, F.; Cachot, J. High Density Polyethylene (HDPE) Microplastics Impair Development and Swimming Activity of Pacific Oyster D-Larvae, Crassostrea Gigas, Depending on Particle Size. Environ. Pollut. 2020, 260, 113978. [Google Scholar] [CrossRef]
- Jemec, A.; Horvat, P.; Kunej, U.; Bele, M.; Kržan, A. Uptake and Effects of Microplastic Textile Fibers on Freshwater Crustacean Daphnia magna. Environ. Pollut. 2016, 219, 201–209. [Google Scholar] [CrossRef]
- Romero-Blanco, A.; Remón-Elola, A.; Alonso, Á. Assessment of the Effects of Environmental Concentrations of Microplastics on the Aquatic Snail Potamopyrgus antipodarum. Water Air Soil Pollut. 2021, 232, 438. [Google Scholar] [CrossRef]
- Imhof, H.K.; Laforsch, C. Hazardous or Not—Are Adult and Juvenile Individuals of Potamopyrgus antipodarum Affected by Non-Buoyant Microplastic Particles? Environ. Pollut. 2016, 218, 383–391. [Google Scholar] [CrossRef] [PubMed]
- Chae, Y.; An, Y.-J. Nanoplastic Ingestion Induces Behavioral Disorders in Terrestrial Snails: Trophic Transfer Effects via Vascular Plants. Environ. Sci. Nano 2020, 7, 975–983. [Google Scholar] [CrossRef]
- Jeyavani, J.; Sibiya, A.; Gopi, N.; Mahboob, S.; Riaz, M.N.; Vaseeharan, B. Dietary Consumption of Polypropylene Microplastics Alter the Biochemical Parameters and Histological Response in Freshwater Benthic Mollusc Pomacea paludosa. Environ. Res. 2022, 212, 113370. [Google Scholar] [CrossRef]
- Au, S.Y.; Bruce, T.F.; Bridges, W.C.; Klaine, S.J. Responses of Hyalella azteca to Acute and Chronic Microplastic Exposures: Effects of Microplastic Exposure on Hyalella azteca. Environ. Toxicol. Chem. 2015, 34, 2564–2572. [Google Scholar] [CrossRef] [PubMed]
- Gallardo, C.S. Developmental pattern and adaptations for reproduction in Nucella crassilabrum and other muricacean gastropods. Biol. Bull. 1979, 157, 453–463. [Google Scholar] [CrossRef] [PubMed]
- De Marco, G.; Conti, G.O.; Giannetto, A.; Cappello, T.; Galati, M.; Iaria, C.; Pulvirenti, E.; Capparucci, F.; Mauceri, A.; Ferrante, M.; et al. Embryotoxicity of Polystyrene Microplastics in Zebrafish Danio rerio. Environ. Res. 2022, 208, 112552. [Google Scholar] [CrossRef]
- Saha, C.; Pramanik, S.; Chakraborty, J.; Parveen, S.; Aditya, G. Life History Features of the Invasive Snail Physa acuta Occurring in Kolkata, India. Natl. Acad. Sci. Lett. 2019, 42, 95–98. [Google Scholar] [CrossRef]
- Bell, G. Measuring the Cost of Reproduction. II. The Correlation Structure of the Life Tables of Five Freshwater Invertebrates. Evolution 1984, 38, 314. [Google Scholar] [CrossRef]
- Plastics Europe Website. 2014. Available online: http://www.plasticseurope.org/Document/plasticsthe-facts-2014.aspx? (accessed on 20 July 2022).
- Plastic Europe Website. 2021. Available online: http://www.plasticseurope.org/Document/plastics-the-Facts2021-web-final.pdf&sa (accessed on 20 July 2022).
- Jambeck, J.R.; Geyer, R.; Wilcox, C.; Siegler, T.R.; Perryman, M.; Andrady, A.; Narayan, R.; Law, K.L. Plastic Waste Inputs from Land into the Ocean. Science 2015, 347, 768–771. [Google Scholar] [CrossRef]
- Martinho, S.D.; Fernandes, V.C.; Figueiredo, S.A.; Delerue-Matos, C. Study of the Potential Accumulation of the Pesticide Alpha-Endosulfan by Microplastics in Water Systems. Polymers 2022, 14, 3645. [Google Scholar] [CrossRef] [PubMed]
- Colomer, J.; Müller, M.F.; Barcelona, A.; Serra, T. Mediated Food and Hydrodynamics on the Ingestion of Microplastics by Daphnia Magna. Environ. Pollut. 2019, 251, 434–441. [Google Scholar] [CrossRef] [PubMed]





Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kumari, N.; Samantaray, B.P.; Patel, A.; Kumar, R. Microplastics Affect Rates of Locomotion and Reproduction via Dietary Uptake in Globally Invasive Snail Physa acuta. Water 2023, 15, 928. https://doi.org/10.3390/w15050928
Kumari N, Samantaray BP, Patel A, Kumar R. Microplastics Affect Rates of Locomotion and Reproduction via Dietary Uptake in Globally Invasive Snail Physa acuta. Water. 2023; 15(5):928. https://doi.org/10.3390/w15050928
Chicago/Turabian StyleKumari, Nisha, Banaja Prakashini Samantaray, Abhishek Patel, and Ram Kumar. 2023. "Microplastics Affect Rates of Locomotion and Reproduction via Dietary Uptake in Globally Invasive Snail Physa acuta" Water 15, no. 5: 928. https://doi.org/10.3390/w15050928