Simulation and Key Physical Drivers of Primary Productivity in a Temperate Lake during the Ice-Covered Period: Based on the VGPM Model
Abstract
:1. Introduction
2. Materials and Methods
2.1. Study Site
2.2. Field Methods
2.3. Euphotic Depths
2.4. Primary Productivity
2.5. Statistical Analysis
3. Results
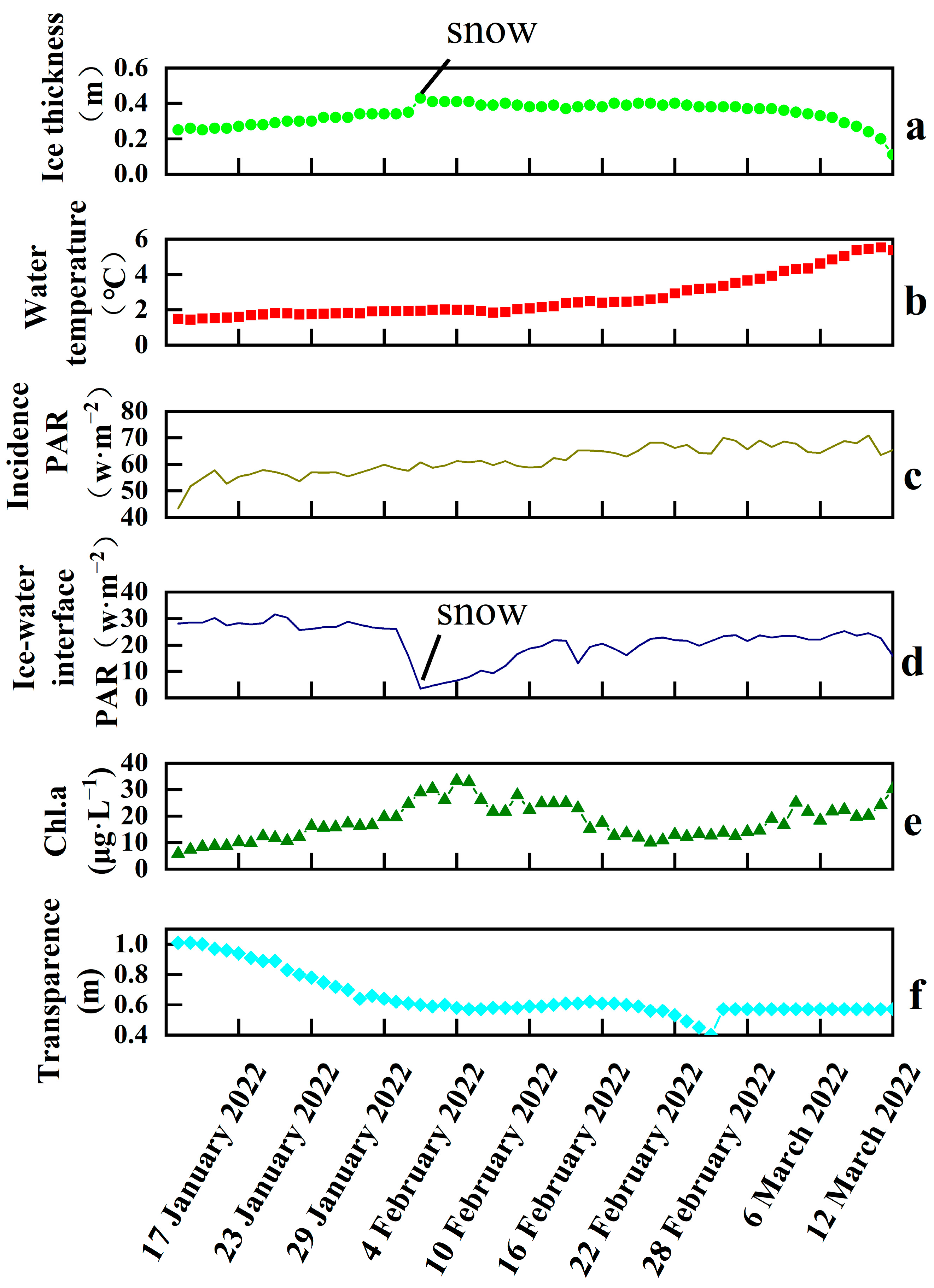
3.1. Ice Thickness, Water Temperature, PAR, Chl.a, and Transparency
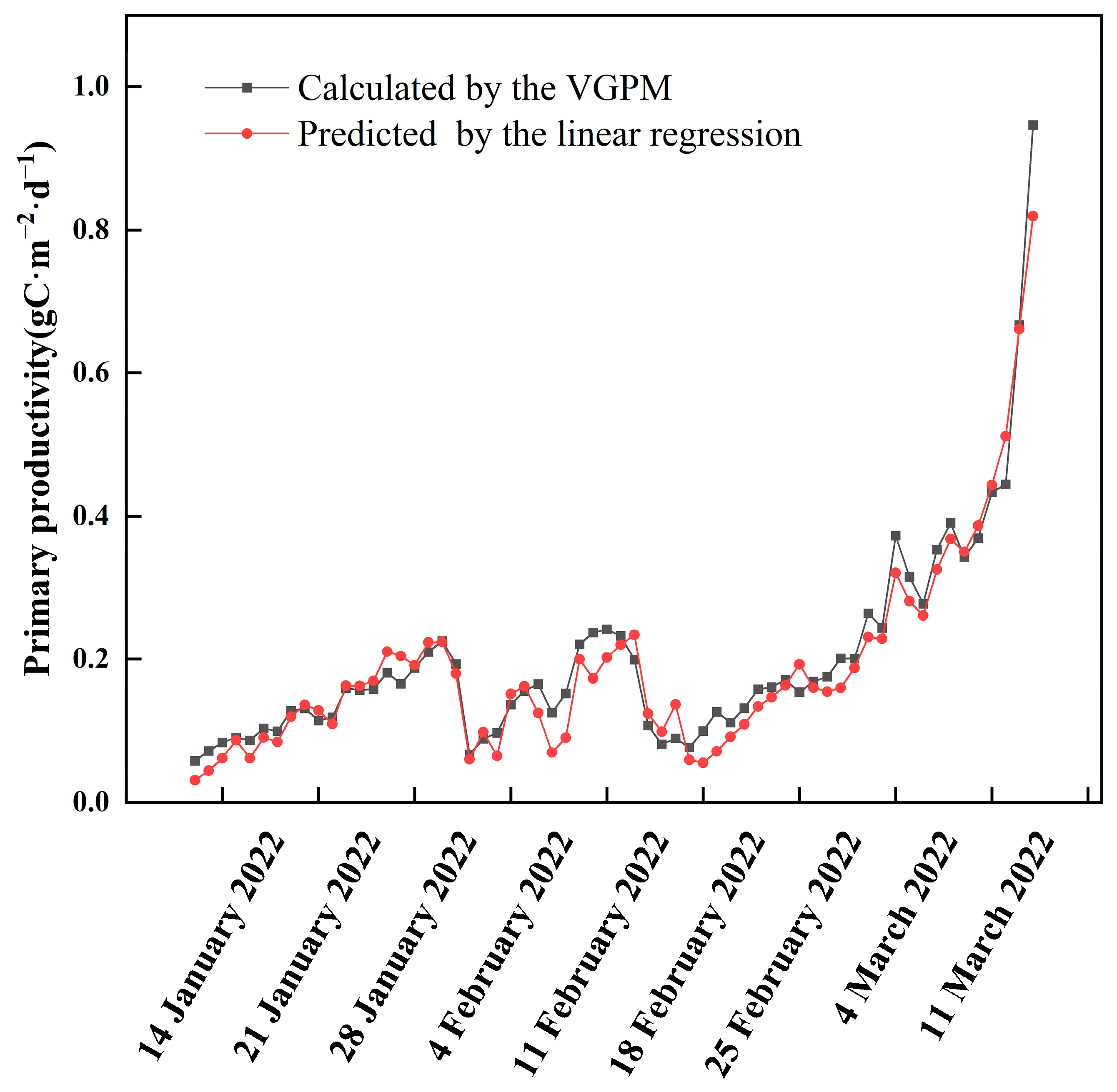
3.2. Primary Productivity
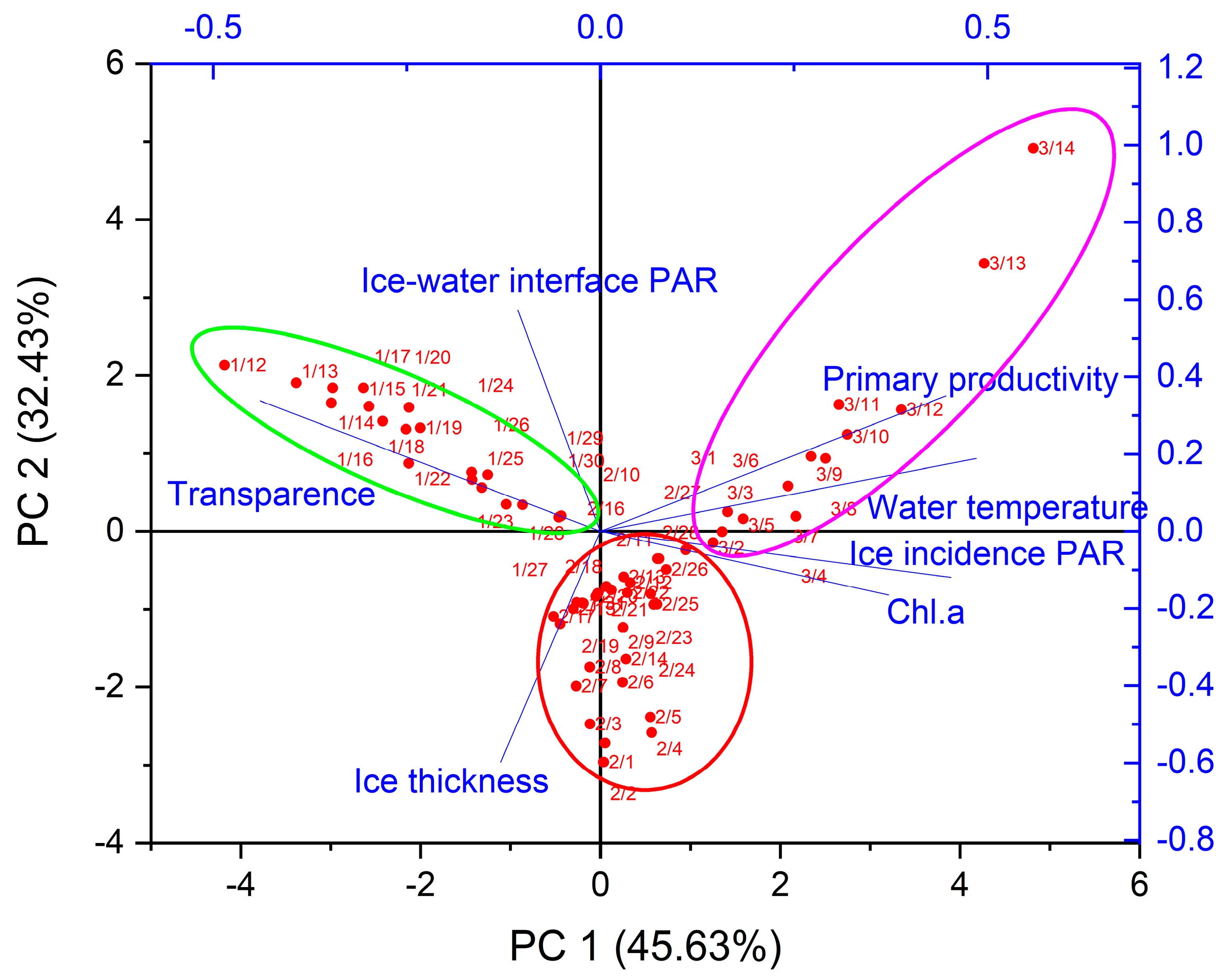
3.3. Key Physical Factors
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Meis, S.; Thackeray, S.J.; Jones, I.D. Effects of recent climate change on phytoplankton phenology in a temperate lake. Freshw. Biol. 2009, 54, 1888–1898. [Google Scholar] [CrossRef]
- Ladwig, R.; Appling, A.P.; Delany, A.; Dugan, H.A.; Gao, Q.T.; Lottig, N.; Stachelek, J.; Hanson, P.C. Long-term change in metabolism phenology in north temperate lakes. Limnol. Oceanogr. 2022, 67, 1502–1521. [Google Scholar] [CrossRef]
- Woolway, R.I.; Kraemer, B.M.; Lenters, J.D.; Merchant, C.J.; O’Reilly, C.M.; Sharma, S. Global Lake responses to climate change. Nat. Rev. Earth Environ. 2020, 1, 388–403. [Google Scholar] [CrossRef]
- Markensten, H.; Moore, K.; Persson, I. Simulated Lake phytoplankton composition shifts toward cyanobacteria dominance in a future warmer climate. Ecol. Appl. 2010, 20, 752–767. [Google Scholar] [CrossRef]
- Kohlb Kohlbach, D.; Graeve, M.; Lange, B.A.; David, C.; Schaafsma, F.L.; van Franeker, J.A.; Vortkamp, M.; Brandt, A.; Flores, H. Dependency of Antarctic zooplankton species on ice algae-produced carbon suggests a sea ice-driven pelagic ecosystem during winter. Glob. Chang. Biol. 2018, 24, 4667–4681. [Google Scholar] [CrossRef] [Green Version]
- Hampton, S.E.; Galloway, A.W.; Powers, S.M. Ecology under lake ice. Ecol. Lett. 2017, 20, 98–111. [Google Scholar] [CrossRef]
- Bramburger, A.J.; Ozersky, T.; Silsbe, G.M.; Crawford, C.J.; Olmanson, L.G.; Shchapov, K. The not-so-dead of winter: Underwater light climate and primary productivity under snow and ice cover in inland lakes. Inland Waters 2022. [Google Scholar] [CrossRef]
- Song, S.; Li, C.Y.; Shi, X.H.; Zhao, S.N.; Tian, W.D.; Li, Z.J.; Bai, Y.L.; Cao, X.W.; Wang, Q.K.; Huotari, J.; et al. Under-ice metabolism in a shallow lake in a cold and arid climate. Freshw. Biol. 2019, 64, 1710–1720. [Google Scholar] [CrossRef]
- Moss, B. Allied attack: Climate change and eutrophication. Inland Waters 2011, 1, 101–105. [Google Scholar] [CrossRef] [Green Version]
- Yang, F.; Cen, R.; Feng, W.Y.; Zhu, Q.H.; Lepparanta, M.; Yang, Y.; Wang, X.H.; Liao, H.Q. Dynamic simulation of nutrient distribution in lakes during ice cover growth and ablation. Chemosphere 2021, 281, 130781. [Google Scholar] [CrossRef]
- Yang, F.; Li, C.; Shi, X.; Zhao, S.; Hao, Y. Impact of seasonal ice structure characteristics on ice cover impurity distributions in Lake Ulansuhai. J. Lake Sci. 2016, 28, 455–462. [Google Scholar]
- Ye, H.B.; Chen, C.Q.; Sun, Z.H.; Tang, S.L.; Song, X.Y.; Yang, C.Y.; Tian, L.Q.; Liu, F.F. Estimation of the Primary Productivity in Pearl River Estuary Using MODIS Data. Estuar. Coasts 2015, 38, 506–518. [Google Scholar] [CrossRef]
- Zainuddin, M.; Kiyofuji, H.; Saitoh, K.; Saitoh, S.I. Using multi-sensor satellite remote sensing and catch data to detect ocean hot spots for albacore (Thunnus alalunga) in the northwestern North Pacific. Deep-Sea Res. Pt. II 2006, 53, 419–431. [Google Scholar] [CrossRef]
- Lomas, M.W.; Moran, S.B.; Casey, J.R.; Bell, D.W.; Tiahlo, M.; Whitefield, J.; Kelly, R.P.; Mathis, J.T.; Cokelet, E.D. Spatial and seasonal variability of primary production on the Eastern Bering Sea shelf. Deep-Sea Res. Pt. II 2012, 65–70, 126–140. [Google Scholar] [CrossRef]
- Huang, L.; Zhou, Y.; Zhou, Q.; Wang, W.; Dong, Y.; Li, K.; Chang, J. Temporal-spatial variation in phytoplankton primary production and its influencing factors in Lake Chenghai on the Yunnan Plateau, China. J. Lake Sci. 2019, 31, 1424–1436. [Google Scholar]
- Yu, H.F.; Shi, X.H.; Zhao, S.N.; Sun, B.; Liu, Y.; Arvola, L.; Li, G.H.; Wang, Y.J.; Pan, X.R.; Wu, R.; et al. Primary productivity of phytoplankton and its influencing factors in cold and arid regions: A case study of Wuliangsuhai Lake, China. Ecol. Indic. 2022, 144, 109545. [Google Scholar] [CrossRef]
- Kirillin, G.; Lepparanta, M.; Terzhevik, A.; Granin, N.; Bernhardt, J.; Engelhardt, C.; Efremova, T.; Golosov, S.; Palshin, N.; Sherstyankin, P.; et al. Physics of seasonally ice-covered lakes: A review. Aquat. Sci. 2012, 74, 659–682. [Google Scholar] [CrossRef]
- Hintz, N.H.; Schulze, B.; Wacker, A.; Striebel, M. Ecological impacts of photosynthetic light harvesting in changing aquatic environments: A systematic literature map. Ecol. Evol. 2022, 12, e8753. [Google Scholar] [CrossRef]
- Xie, F.; Lu, P.; Li, Z.J.; Wang, Q.K.; Zhang, H.; Zhang, Y.W. A floating remote observation system (FROS) for full seasonal lake ice evolution studies. Cold Reg. Sci. Technol. 2022, 199, 103557. [Google Scholar] [CrossRef]
- Holmes, R.W. Secchi disk in turbid coastal waters. Limnol. Oceanogr. 1970, 15, 688–694. [Google Scholar] [CrossRef]
- Ma, J.H. Inversion of Kd (PAR) and Euphotic Zone Depth of Typical Water Bodys in Northeast China with Remote Imagery; Northeast Institute of Geography and Agroecology: Changchun, China; Chinese Academy of Sciences: Beijing, China, 2016. [Google Scholar]
- Behrenfeld, M.J.; Falkowski, P.G. Photosynthetic rates derived from satellite-based chlorophyll concentration. Limnol. Oceanogr. 1997, 42, 1–20. [Google Scholar] [CrossRef]
- Wang, H.; Wang, H.; Li, Y.; Ma, S.; Yu, Q. The control of lake eutrophication: Focusing on phosphorus abatement, or reducing both phosphorus and nitrogen? Acta Hydrobiol. Sin. 2020, 44, 938–960. [Google Scholar]
- Alexander, T.J.; Vonlanthen, P.; Seehausen, O. Does eutrophication-driven evolution change aquatic ecosystems? Philos. Trans. R. Soc. B 2017, 372, 20160041. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Li, H.; Shen, H.; Li, S.; Liang, Y.; Lu, C.; Zhang, L. Effects of eutrophication on the benthic-pelagic coupling food web in Baiyangdian Lake. Acta Ecol. Sin. 2018, 38, 2017–2030. [Google Scholar]
- Saygi-Basbug, Y.; Demirkalp, F.Y. Primary production in shallow eutrophic Yenicaga Lake (Bolu, Turkey). Fresenius Environ. Bull. 2004, 13, 98–104. [Google Scholar]
- Wei, J.; Ji, X.N.; Hu, W. Characteristics of Phytoplankton Production in Wet and Dry Seasons in Hyper-Eutrophic Lake Taihu, China. Sustainability 2022, 14, 11216. [Google Scholar] [CrossRef]
- Stenuite, S.; Pirlot, S.; Hardy, M.A.; Sarmento, H.; Tarbe, A.L.; Leporcq, B.; Descy, J.P. Phytoplankton production and growth rate in Lake Tanganyika: Evidence of a decline in primary productivity in recent decades. Freshw. Biol. 2007, 52, 2226–2239. [Google Scholar] [CrossRef]
- Kennedy, J.T.; Whalen, S.C. Seasonality, and controls of phytoplankton productivity in the middle Cape Fear River, USA. Hydrobiologia 2008, 598, 203–217. [Google Scholar] [CrossRef]
- Hampton, S.E.; Sharma, S.; Brousil, M.R.; Filazzola, A. Winter and summer storms modify chlorophyll relationships with nutrients in seasonally ice-covered lakes. Ecosphere 2022, 13, e4272. [Google Scholar] [CrossRef]
- Xie, C.; Zhou, L.; Ding, S.F.; Liu, R.W.; Zheng, S.J. Experimental and numerical investigation on self-propulsion performance of polar merchant ship in brash ice channel. Ocean Eng. 2023, 269, 113424. [Google Scholar] [CrossRef]
- Sun, Q.Y.; Zhang, M.; Zhou, L.; Garme, K.; Burman, M. A machine learning-based method for prediction of ship performance in ice: Part I. ice resistance. Mar. Struct. 2022, 83, 103181. [Google Scholar] [CrossRef]
- Rautio, M.; Dufresne, F.; Laurion, I.; Bonilla, S.; Vincent, W.F.; Christoffersen, K.S. Shallow freshwater ecosystems of the circumpolar Arctic. Ecoscience 2011, 18, 204–222. [Google Scholar] [CrossRef]
- Ozkundakci, D.; Gsell, A.S.; Hintze, T.; Tauscher, H.; Adrian, R. Winter severity determines functional trait composition of phytoplankton in seasonally ice-covered lakes. Glob. Chang. Biol. 2016, 22, 284–298. [Google Scholar] [CrossRef] [PubMed]
- Katz, S.L.; Izmest’eva, L.R.; Hampton, S.E.; Ozersky, T.; Shchapov, K.; Moore, M.V.; Shimaraeva, S.V.; Silow, E.A. The “Melosira years” of Lake Baikal: Winter environmental conditions at ice onset predict under-ice algal blooms in spring. Limnol. Oceanog. 2015, 60, 1950–1964. [Google Scholar] [CrossRef]
- Shatwell, T.; Thiery, W.; Kirillin, G. Future projections of temperature and mixing regime of European temperate lakes. Hydrol. Earth Syst. Sci. 2019, 23, 1533–1551. [Google Scholar] [CrossRef] [Green Version]
- Obryk, M.K.; Doran, P.T.; Priscu, J.C. The permanent ice cover of Lake Bonney, Antarctica: The influence of thickness and sediment distribution on photosynthetically available radiation and chlorophyll-a distribution in the underlying water column. J. Geophys. Res. Biogeosci. 2014, 119, 1879–1891. [Google Scholar] [CrossRef]

| Lakes and Rivers | Latitudes | Time | Primary Productivity mg C·m−2·d−1 | References |
|---|---|---|---|---|
| Yenicaga Lake | 40°47′ N | December | 319 | [26] |
| Pearl River Estuary | 21°48′–22°27′ | Winter | 224.5 | [12] |
| Taihu | 30°55′40″–31°32′58″ N | Annual | 207.67–2237.71 | [27] |
| Tanganyika | 3°20′–8°48′ S | Wet and Dry Seasons | 110–1410 | [28] |
| Cape Fear River | -- | Annual | 18–2580 | [29] |
| Wuliangsuhai | 40°36′–41°03′ N | Winter | 86.34–96.34 | [16] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhang, J.; Xie, F.; Song, H.; Meng, J.; Zhang, Y. Simulation and Key Physical Drivers of Primary Productivity in a Temperate Lake during the Ice-Covered Period: Based on the VGPM Model. Water 2023, 15, 918. https://doi.org/10.3390/w15050918
Zhang J, Xie F, Song H, Meng J, Zhang Y. Simulation and Key Physical Drivers of Primary Productivity in a Temperate Lake during the Ice-Covered Period: Based on the VGPM Model. Water. 2023; 15(5):918. https://doi.org/10.3390/w15050918
Chicago/Turabian StyleZhang, Jie, Fei Xie, Haoming Song, Jingya Meng, and Yiwen Zhang. 2023. "Simulation and Key Physical Drivers of Primary Productivity in a Temperate Lake during the Ice-Covered Period: Based on the VGPM Model" Water 15, no. 5: 918. https://doi.org/10.3390/w15050918






