Treatment of Fly-Ash-Contaminated Wastewater Loaded with Heavy Metals by Using Fly-Ash-Synthesized Iron Oxide Nanoparticles
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Methods
2.2.1. Synthesis of Iron Oxide Nanoparticles from Fly Ash
2.2.2. Preparation of 20% Fly Ash Aqueous Solution
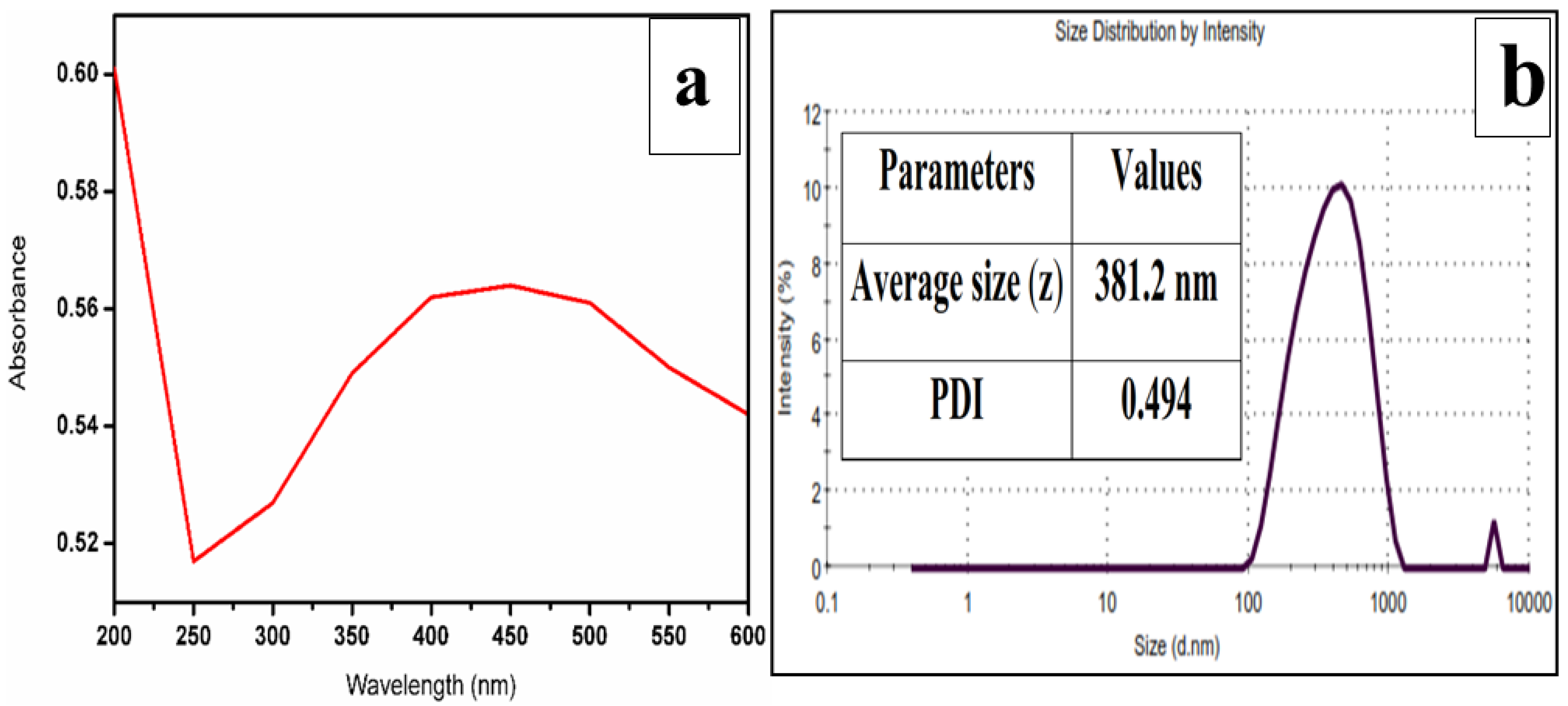
2.2.3. Characterization of Iron Oxide Nanoparticles
2.2.4. Batch Experiment for Remediation of Heavy Metals
- C0 = initial concentration of heavy metal
- Ct = heavy-metal concentration at a particular time
3. Results and Discussion
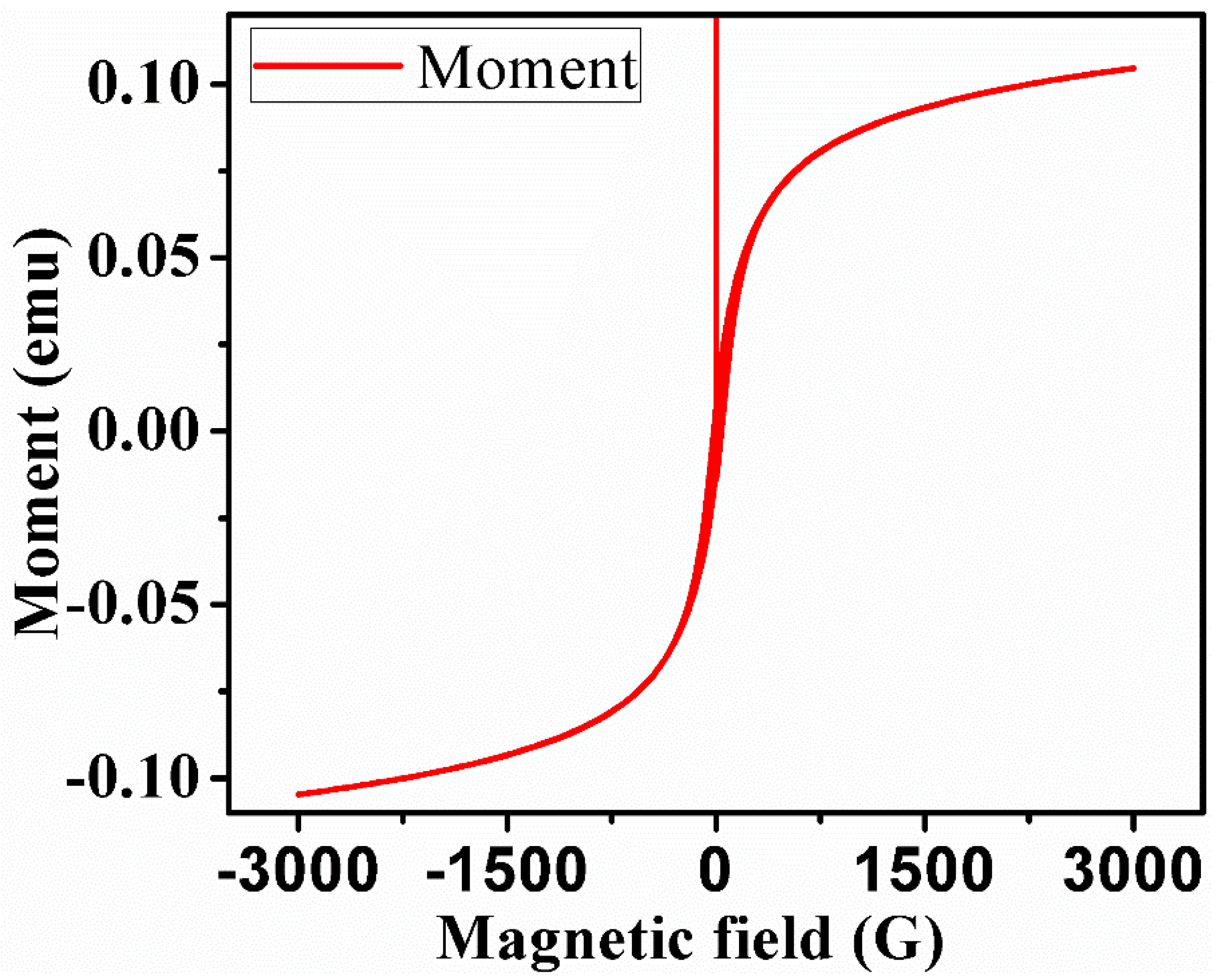
Characterization
4. Effect on Nano-Adsorbent after Remediation of Heavy Metal
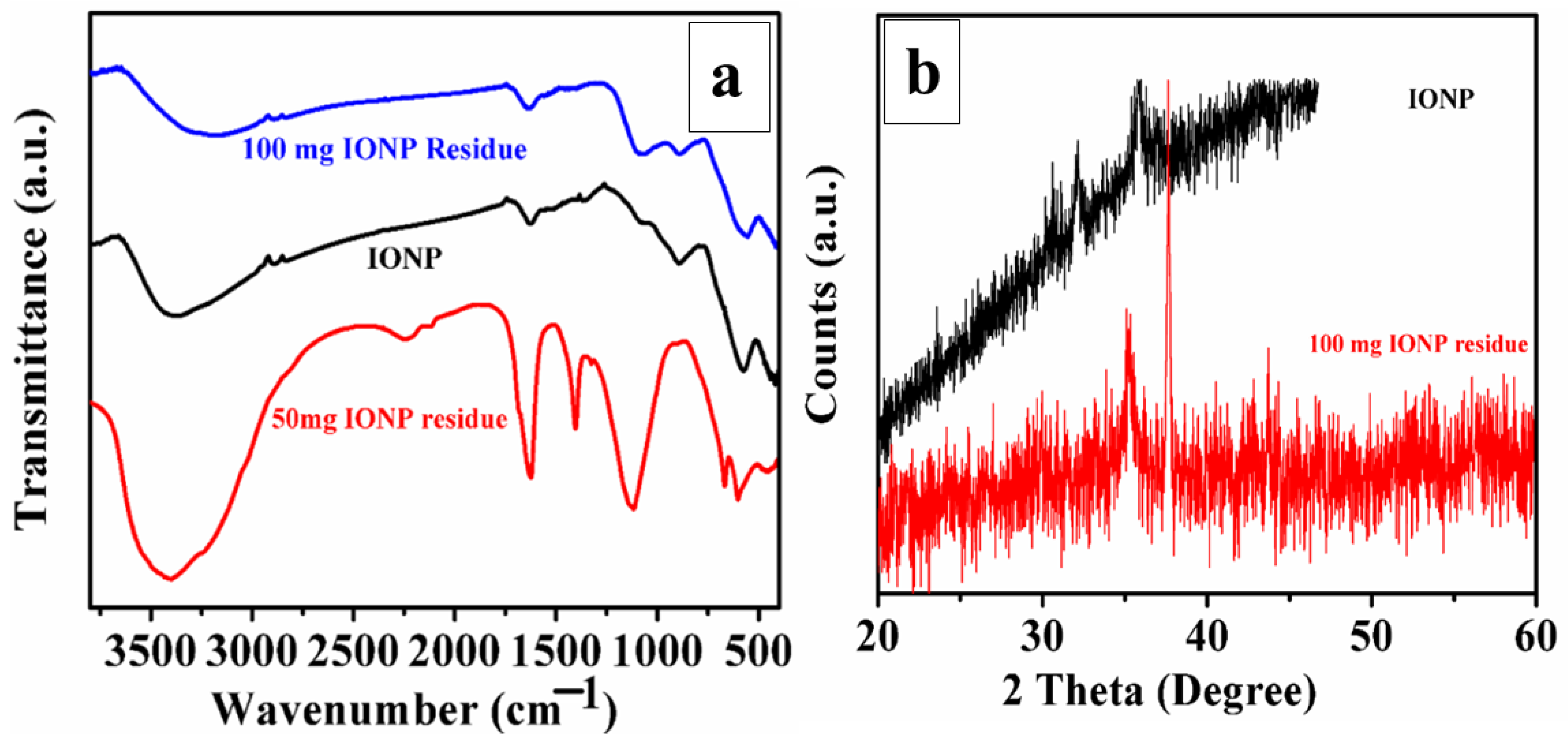
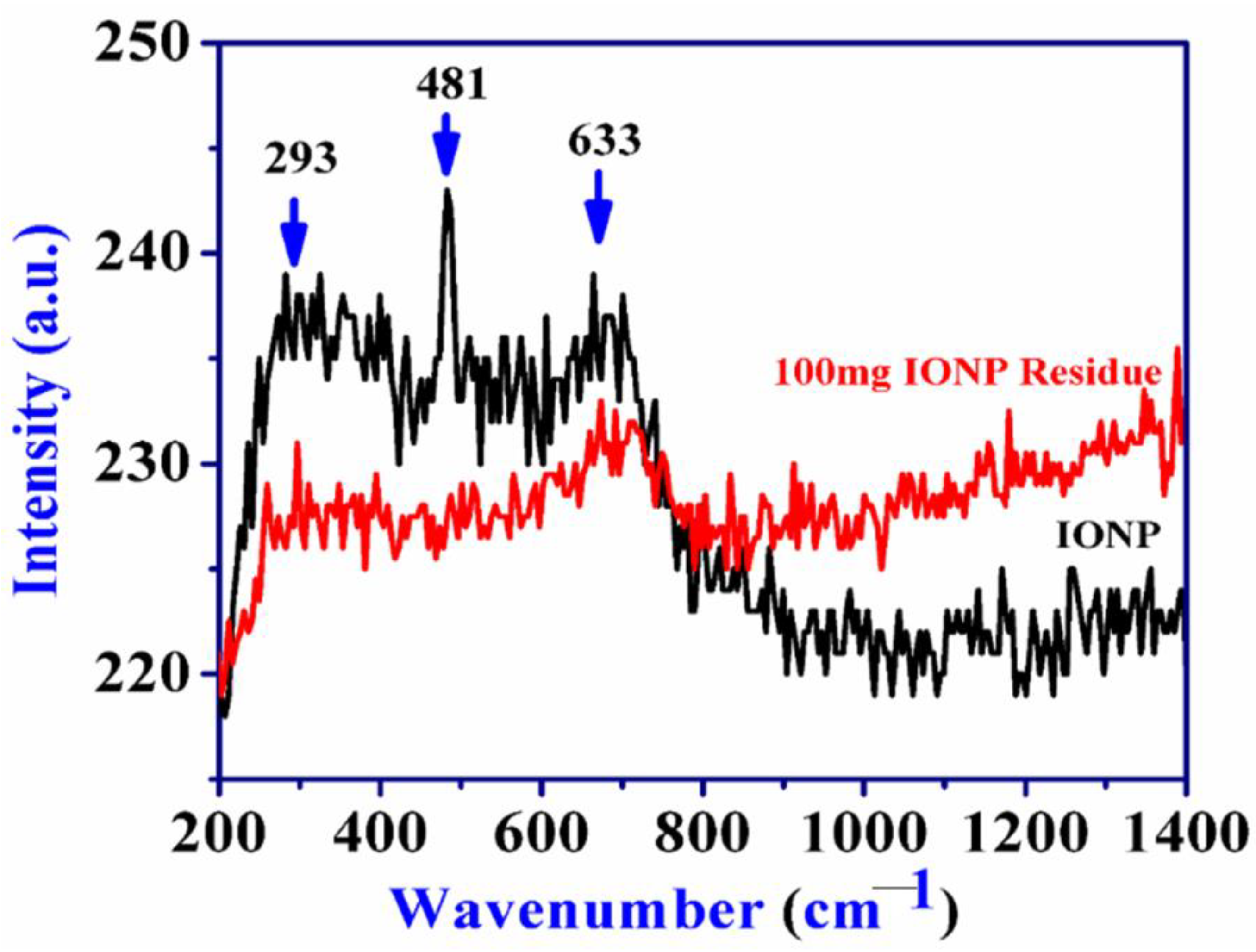
4.1. Mineralogical Changes: XRD, FTIR and Raman
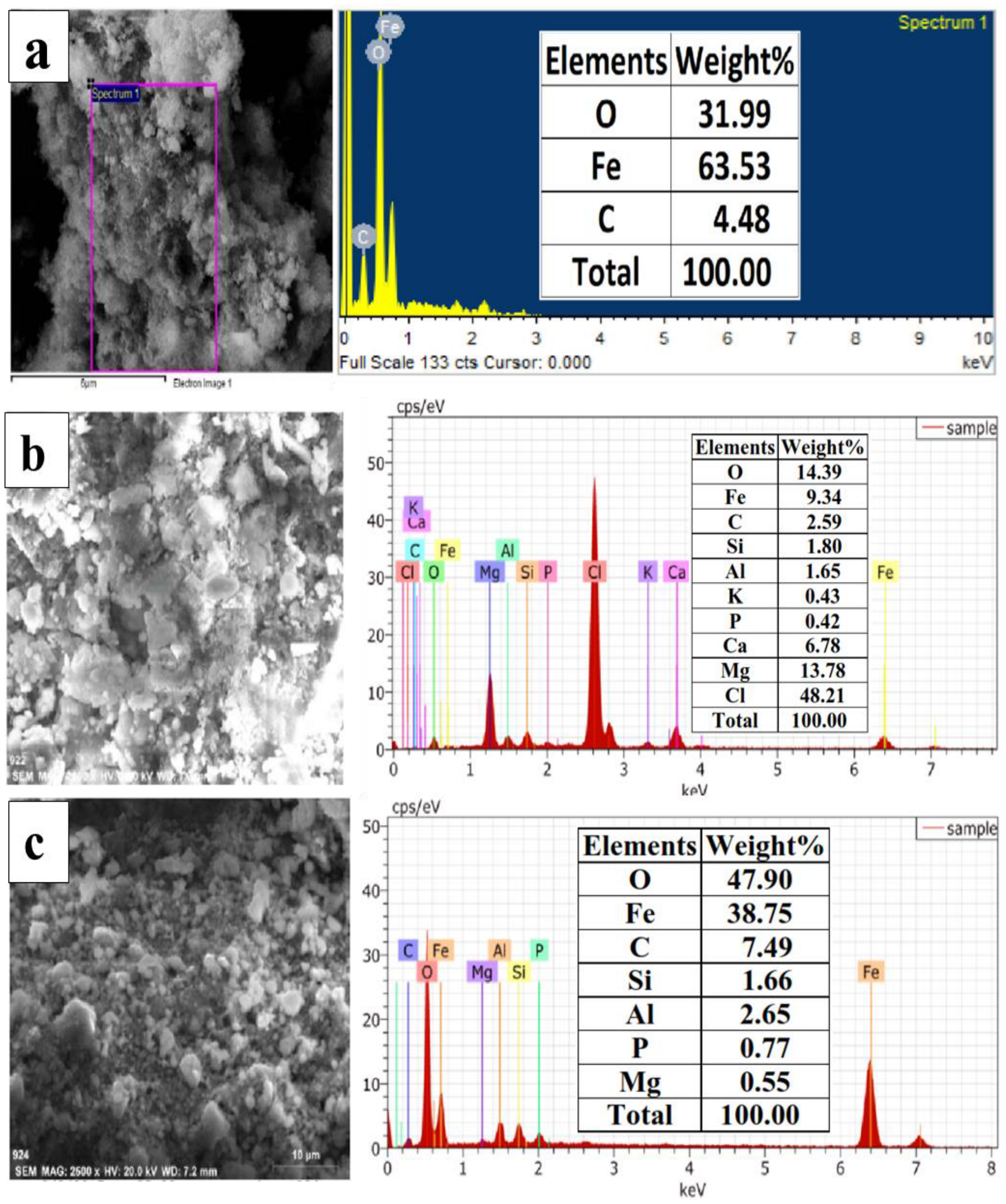
4.2. Morphological and Elemental Changes: FESEM-EDS and ICP-OES
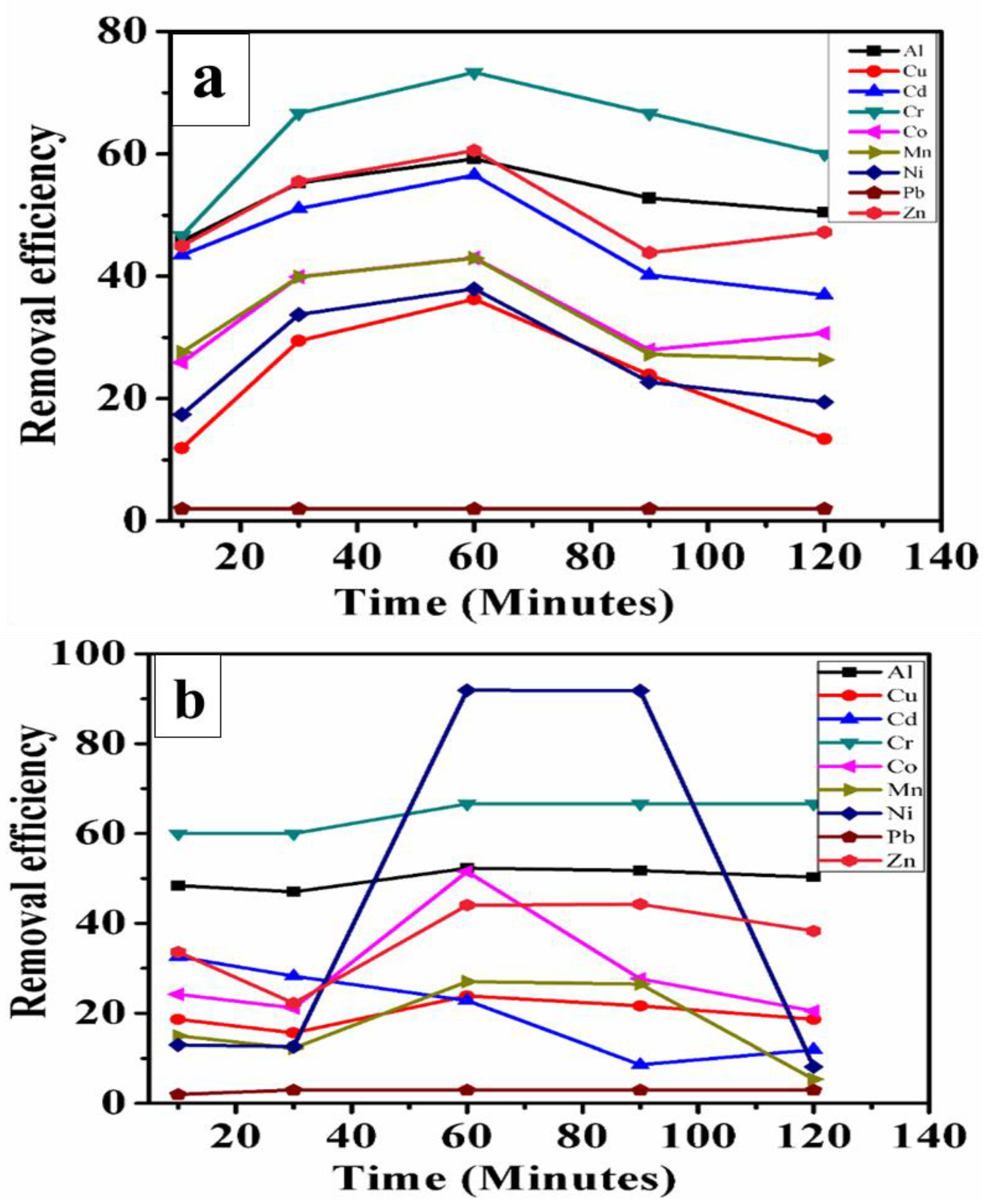
4.3. The Remediation of Heavy Metals by 50 mg IONPs
4.4. The Remediation of Heavy Metals by 100 mg IONPs
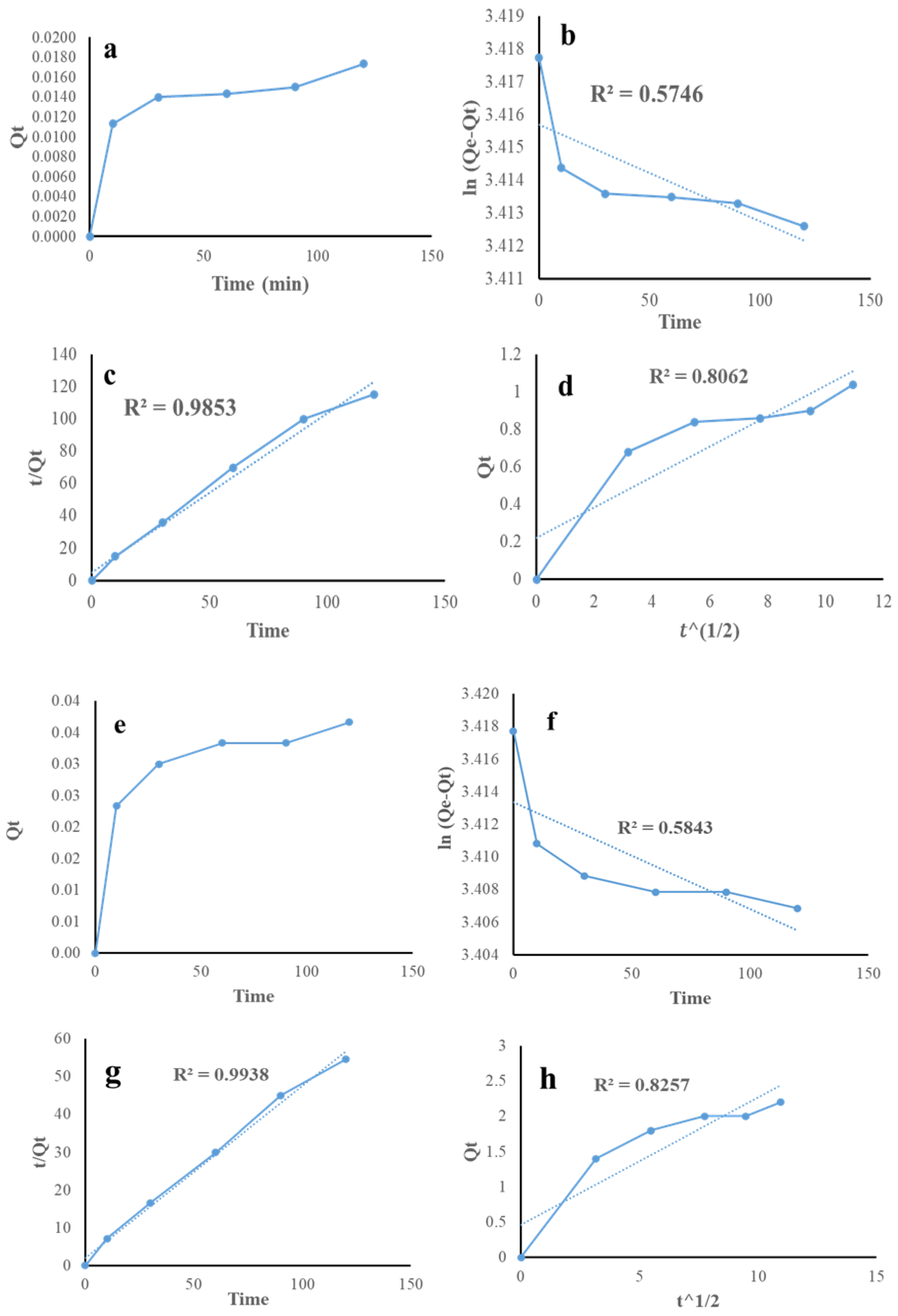
4.5. Kinetics Study of Pb and Cr
4.6. Adsorption Mechanism
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Patel, H.; Yadav, V.K.; Yadav, K.K.; Choudhary, N.; Kalasariya, H.; Alam, M.M.; Gacem, A.; Amanullah, M.; Ibrahium, H.A.; Park, J.-W.; et al. A Recent and Systemic Approach Towards Microbial Biodegradation of Dyes from Textile Industries. Water 2022, 14, 3163. [Google Scholar] [CrossRef]
- Gadore, V. Ahmaruzzaman Fly ash–based nanocomposites: A potential material for effective photocatalytic degradation/elimination of emerging organic pollutants from aqueous stream. Environ. Sci. Pollut. Res. 2021, 28, 46910–46933. [Google Scholar] [CrossRef] [PubMed]
- Ankrah, A.F.; Tokay, B.; Snape, C.E. Heavy Metal Removal from Aqueous Solutions Using Fly-Ash Derived Zeolite NaP1. Int. J. Environ. Res. 2022, 16, 17. [Google Scholar] [CrossRef]
- Yadav, V.K.; Fulekar, M.H. The current scenario of thermal power plants and fly ash: Production and utilization with a focus in India. Int. J. Adv. Eng. Res. Dev. 2018, 5, 768–777. [Google Scholar]
- Park, H.; Wang, L.; Yun, J.-H. Coal beneficiation technology to reduce hazardous heavy metals in fly ash. J. Hazard. Mater. 2021, 416, 125853. [Google Scholar] [CrossRef]
- Yadav, V.K.; Gnanamoorthy, G.; Cabral-Pinto, M.M.S.; Alam, J.; Ahamed, M.; Gupta, N.; Singh, B.; Choudhary, N.; Inwati, G.K.; Yadav, K.K. Variations and similarities in structural, chemical, and elemental properties on the ashes derived from the coal due to their combustion in open and controlled manner. Environ. Sci. Pollut. Res. 2021, 28, 32609–32625. [Google Scholar] [CrossRef]
- Qasem, N.A.A.; Mohammed, R.H.; Lawal, D.U. Removal of heavy metal ions from wastewater: A comprehensive and critical review. Npj Clean Water 2021, 4, 36. [Google Scholar] [CrossRef]
- Puri, N.; Gupta, A.; Mishra, A. Recent advances on nano-adsorbents and nanomembranes for the remediation of water. J. Clean. Prod. 2021, 322, 129051. [Google Scholar] [CrossRef]
- Guerra, F.D.; Attia, M.F.; Whitehead, D.C.; Alexis, F. Nanotechnology for Environmental Remediation: Materials and Applications. Molecules 2018, 23, 1760. [Google Scholar] [CrossRef] [Green Version]
- Nicola, R.; Costişor, O.; Ciopec, M.; Negrea, A.; Lazău, R.; Ianăşi, C.; Picioruş, E.-M.; Len, A.; Almásy, L.; Szerb, E.I.; et al. Silica-Coated Magnetic Nanocomposites for Pb2+ Removal from Aqueous Solution. Appl. Sci. 2020, 10, 2726. [Google Scholar] [CrossRef] [Green Version]
- Wegmann, M.; Scharr, M. Chapter 8—Synthesis of Magnetic Iron Oxide Nanoparticles. In Precision Medicine; Deigner, H.-P., Kohl, M., Eds.; Academic Press: Cambridge, MA, USA, 2018; pp. 145–181. [Google Scholar] [CrossRef]
- Sokol, E.; Kalugin, V.; Nigmatulina, E.; Volkova, N.; Frenkel, A.; Maksimova, N. Ferrospheres from fly ashes of Chelyabinsk coals: Chemical composition, morphology and formation conditions. Fuel 2002, 81, 867–876. [Google Scholar] [CrossRef]
- Yadav, V.K.; Amari, A.; Wanale, S.G.; Osman, H.; Fulekar, M.H. Synthesis of Floral-Shaped Nanosilica from Coal Fly Ash and Its Application for the Remediation of Heavy Metals from Fly Ash Aqueous Solutions. Sustainability 2023, 15, 2612. [Google Scholar] [CrossRef]
- Yadav, V.K.; Yadav, K.K.; Gnanamoorthy, G.; Choudhary, N.; Khan, S.H.; Gupta, N.; Kamyab, H.; Bach, Q.-V. A novel synthesis and characterization of polyhedral shaped amorphous iron oxide nanoparticles from incense sticks ash waste. Environ. Technol. Innov. 2020, 20, 101089. [Google Scholar] [CrossRef]
- Gupta, N.; Yadav, V.K.; Yadav, K.K.; Alwetaishi, M.; Gnanamoorthy, G.; Singh, B.; Jeon, B.-H.; Cabral-Pinto, M.M.; Choudhary, N.; Ali, D.; et al. Recovery of iron nanominerals from sacred incense sticks ash waste collected from temples by wet and dry magnetic separation method. Environ. Technol. Innov. 2021, 25, 102150. [Google Scholar] [CrossRef]
- Yadav, V.K.; Gnanamoorthy, G.; Ali, D.; Bera, S.P.; Roy, A.; Kumar, G.; Choudhary, N.; Kalasariya, H.; Basnet, A. Cytotoxicity, Removal of Congo Red Dye in Aqueous Solution Using Synthesized Amorphous Iron Oxide Nanoparticles from Incense Sticks Ash Waste. J. Nanomater. 2022, 2022, 5949595. [Google Scholar] [CrossRef]
- Wang, C.; Zhou, J.; Wang, Y.; Yang, M.; Li, Y.; Meng, C. Synthesis of zeolite X from low-grade bauxite. J. Chem. Technol. Biotechnol. 2012, 88, 1350–1357. [Google Scholar] [CrossRef]
- Yadav, V.K.; Fulekar, M. Biogenic synthesis of maghemite nanoparticles (γ-Fe2O3) using Tridax leaf extract and its application for removal of fly ash heavy metals (Pb, Cd). Mater. Today: Proc. 2018, 5, 20704–20710. [Google Scholar] [CrossRef]
- Blaha, U.; Sapkota, B.; Appel, E.; Stanjek, H.; Rösler, W. Micro-scale grain-size analysis and magnetic properties of coal-fired power plant fly ash and its relevance for environmental magnetic pollution studies. Atmospheric Environ. 2008, 42, 8359–8370. [Google Scholar] [CrossRef]
- Joos, A.; Rümenapp, C.; Wagner, F.E.; Gleich, B. Characterisation of iron oxide nanoparticles by Mössbauer spectroscopy at ambient temperature. J. Magn. Magn. Mater. 2016, 399, 123–129. [Google Scholar] [CrossRef]
- Adewuyi, Y.G. Recent Advances in Fly-Ash-Based Geopolymers: Potential on the Utilization for Sustainable Environmental Remediation. ACS Omega 2021, 6, 15532–15542. [Google Scholar] [CrossRef]
- Khan, M.A.; Memon, S.A.; Farooq, F.; Javed, M.F.; Aslam, F.; Alyousef, R. Compressive Strength of Fly-Ash-Based Geopolymer Concrete by Gene Expression Programming and Random Forest. Adv. Civ. Eng. 2021, 2021, 6618407. [Google Scholar] [CrossRef]
- Zhao, Y.; Qiu, Z.; Huang, J. Preparation and Analysis of Fe3O4 Magnetic Nanoparticles Used as Targeted-drug Carriers. Chin. J. Chem. Eng. 2008, 16, 451–455. [Google Scholar] [CrossRef]
- Hariharan, S.; Ramar, V.; Joshi, S.P.; Balaya, P. Developing a light weight lithium ion battery—An effective material and electrode design for high performance conversion anodes. RSC Adv. 2013, 3, 6386–6394. [Google Scholar] [CrossRef]
- Ma, J.; Guo, X.; Yan, Y.; Xue, H.; Pang, H. FeOx-Based Materials for Electrochemical Energy Storage. Adv. Sci. 2018, 5, 1700986. [Google Scholar] [CrossRef]
- You, S.; Ho, S.W.; Li, T.; Maneerung, T.; Wang, C.-H. Techno-economic analysis of geopolymer production from the coal fly ash with high iron oxide and calcium oxide contents. J. Hazard. Mater. 2018, 361, 237–244. [Google Scholar] [CrossRef] [Green Version]
- Wang, H.; Wang, M.; Zhang, J.; Wang, N.; Wang, J.; Yang, J. Preparation of fly ash-based cobalt-iron silicate as supercapacitor electrode material. Chem. Eng. J. 2022, 434, 134661. [Google Scholar] [CrossRef]
- Li, X.; Ma, B.; Wang, C.; Chen, Y.; Yang, W.; Zhang, W. A sustainable process to recycle aluminum from coal fly ash for simultaneous removal of iron: Solid waste management and evaluation. Miner. Eng. 2022, 184, 107638. [Google Scholar] [CrossRef]
- Justin, C.; Philip, S.A.; Samrot, A.V. Synthesis and characterization of superparamagnetic iron-oxide nanoparticles (SPIONs) and utilization of SPIONs in X-ray imaging. Appl. Nanosci. 2017, 7, 463–475. [Google Scholar] [CrossRef]
- Kumar, P.; Mathpal, M.C.; Ghosh, S.; Inwati, G.K.; Maze, J.R.; Duvenhage, M.-M. Plasmonic Au nanoparticles embedded in glass: Study of TOF-SIMS, XPS and its enhanced antimicrobial activities. J. Alloys Compd. 2022, 909, 164789. [Google Scholar] [CrossRef]
- Mahmoudi, M.; Sahraian, M.A.; Shokrgozar, M.A.; Laurent, S. Superparamagnetic Iron Oxide Nanoparticles: Promises for Diagnosis and Treatment of Multiple Sclerosis. ACS Chem. Neurosci. 2011, 2, 118–140. [Google Scholar] [CrossRef] [Green Version]
- Modi, S.; Inwati, G.K.; Gacem, A.; Saquib Abullais, S.; Prajapati, R.; Yadav, V.K.; Syed, R.; Alqahtani, M.S.; Yadav, K.K.; Islam, S.; et al. Nanostructured Antibiotics and Their Emerging Medicinal Applications: An Overview of Nanoantibiotics. Antibiotics 2022, 11, 708. [Google Scholar] [CrossRef]
- Yadav, V.; Ali, D.; Khan, S.; Gnanamoorthy, G.; Choudhary, N.; Yadav, K.; Thai, V.; Hussain, S.; Manhrdas, S. Synthesis and Characterization of Amorphous Iron Oxide Nanoparticles by the Sonochemical Method and Their Application for the Remediation of Heavy Metals from Wastewater. Nanomaterials 2020, 10, 1551. [Google Scholar] [CrossRef] [PubMed]
- Rafieizonooz, M.; Khankhaje, E.; Rezania, S. Assessment of environmental and chemical properties of coal ashes including fly ash and bottom ash, and coal ash concrete. J. Build. Eng. 2022, 49, 104040. [Google Scholar] [CrossRef]
- Dash, S.; Chaudhuri, H.; Gupta, R.; Nair, U.G. Adsorption study of modified coal fly ash with sulfonic acid as a potential adsorbent for the removal of toxic reactive dyes from aqueous solution: Kinetics and thermodynamics. J. Environ. Chem. Eng. 2018, 6, 5897–5905. [Google Scholar] [CrossRef]
- Correia, L.M.; Saboya, R.M.A.; Campelo, N.D.S.; Cecilia, J.A.; Rodríguez-Castellón, E.; Cavalcante, C.L.; Vieira, R.S. Characterization of calcium oxide catalysts from natural sources and their application in the transesterification of sunflower oil. Bioresour. Technol. 2014, 151, 207–213. [Google Scholar] [CrossRef] [PubMed]
- Konopacka-Łyskawa, D.; Czaplicka, N.; Kościelska, B.; Łapiński, M.; Gębicki, J. Influence of Selected Saccharides on the Precipitation of Calcium-Vaterite Mixtures by the CO2 Bubbling Method. Crystals 2019, 9, 117. [Google Scholar] [CrossRef] [Green Version]
- Yadav, V.K.; Yadav, K.K.; Gacem, A.; Gnanamoorthy, G.; Ali, I.H.; Khan, S.H.; Jeon, B.-H.; Kamyab, H.; Inwati, G.K.; Choudhary, N.; et al. A novel approach for the synthesis of vaterite and calcite from incense sticks ash waste and their potential for remediation of dyes from aqueous solution. Sustain. Chem. Pharm. 2022, 29, 100756. [Google Scholar] [CrossRef]
- Gnanamoorthy, G.; Yadav, V.K.; Ali, D.; Ramar, K.; Ali, H.; Narayanan, V. New designing (NH4) 2SiP4O13 nanowires and effective photocatalytic degradation of Malachite Green. Chem. Phys. Lett. 2022, 803, 139817. [Google Scholar] [CrossRef]
- Ganesh, A.B.; Shekar, B.C. Synthesis, characterization and plant growth assessment of hybrid calcium oxide nanoparticles. Int. J. pharma Bio Sci. 2017, 8, 193–198. [Google Scholar] [CrossRef]
- Bharathiraja, B.; Sutha, M.; Sowndarya, K.; Chandran, M.; Yuvaraj, D.; Kumar, R.P. Calcium Oxide Nanoparticles as An Effective Filtration Aid for Purification of Vehicle Gas Exhaust. In Advances in Internal Combustion Engine Research. Energy, Environment, and Sustainability; Srivastava, D., Agarwal, A., Datta, A., Maurya, R., Eds.; Springer: Singapore, 2018; pp. 181–192. [Google Scholar] [CrossRef]
- Gnanamoorthy, G.; Yadav, V.K.; Ali, D.; Parthiban, E.; Kumar, G.; Narayanan, V. New orchestrated of X-CuTiAP (en, trien, ETA and DMA) nanospheres with enhanced photocatalytic and antimicrobial activities. J. Ind. Eng. Chem. 2022, 110, 503–519. [Google Scholar] [CrossRef]
- Gnanamoorthy, G.; Ramar, K.; Ali, D.; Yadav, V.K.; Sureshbabu, K.; Narayanan, V. A series of ZnCo2O4/rGO/Pt nanocubes with excellent photocatalytic activity towards visible light. Chem. Phys. Lett. 2020, 759, 137988. [Google Scholar] [CrossRef]
- Yadav, V.K. Nano based approaches techniques and method development for separation of ferrous alumina and silica from waste fly ash. Nanosciences. Ph.D. Thesis, Central University of Gujarat, Gandhinagar, India, 14 October 2019. [Google Scholar]
- Pan, J.; Hassas, B.V.; Rezaee, M.; Zhou, C.; Pisupati, S.V. Recovery of rare earth elements from coal fly ash through sequential chemical roasting, water leaching, and acid leaching processes. J. Clean. Prod. 2020, 284, 124725. [Google Scholar] [CrossRef]
- Rezaei, H.; Shafaei, S.Z.; Abdollahi, H.; Shahidi, A.; Ghassa, S. A sustainable method for germanium, vanadium and lithium extraction from coal fly ash: Sodium salts roasting and organic acids leaching. Fuel 2021, 312, 122844. [Google Scholar] [CrossRef]
- Ma, Y.; Stopic, S.; Xakalashe, B.; Ndlovu, S.; Forsberg, K.; Friedrich, B. A cleaner approach for recovering Al and Ti from coal fly ash via microwave-assisted baking, leaching, and precipitation. Hydrometallurgy 2021, 206, 105754. [Google Scholar] [CrossRef]
- Sahay, D.K.; Bansal, S. Use of Fly Ash—A Resourceful Byproduct in Road Embankment: A Review. In Advances in Construction Materials and Sustainable Environment; Gupta, A.K., Shukla, S.K., Azamathulla, H., Eds.; Springer: Singapore, 2022; pp. 539–550. [Google Scholar]
- Ishwari, B.K.; Kumar, P.S.; Shivaprasad, K.H. Adsorption Studies of Heavy Metals on Coal Fly Ash Samples from Aqueous Solutions. Appl. Ecol. Environ. Sci. 2022, 10, 25–33. [Google Scholar] [CrossRef]
- Chen, Y.; Chen, F.; Zhou, F.; Lu, M.; Hou, H.; Li, J.; Liu, D.; Wang, T. Early solidification/stabilization mechanism of heavy metals (Pb, Cr and Zn) in Shell coal gasification fly ash based geopolymer. Sci. Total. Environ. 2021, 802, 149905. [Google Scholar] [CrossRef]
- Zhang, Z.; Wang, Y.; Zhang, Y.; Shen, B.; Ma, J.; Liu, L. Stabilization of heavy metals in municipal solid waste incineration fly ash via hydrothermal treatment with coal fly ash. Waste Manag. 2022, 144, 285–293. [Google Scholar] [CrossRef]
- Ebrahimi, R.; Hossienzadeh, K.; Maleki, A.; Ghanbari, R.; Rezaee, R.; Safari, M.; Shahmoradi, B.; Daraei, H.; Jafari, A.; Yetilmezsoy, K.; et al. Effects of doping zinc oxide nanoparticles with transition metals (Ag, Cu, Mn) on photocatalytic degradation of Direct Blue 15 dye under UV and visible light irradiation. J. Environ. Heal. Sci. Eng. 2019, 17, 479–492. [Google Scholar] [CrossRef]
- Degefa, A.; Bekele, B.; Jule, L.T.; Fikadu, B.; Ramaswamy, S.; Dwarampudi, L.P.; Nagaprasad, N.; Ramaswamy, K. Green Synthesis, Characterization of Zinc Oxide Nanoparticles, and Examination of Properties for Dye-Sensitive Solar Cells Using Various Vegetable Extracts. J. Nanomater. 2021, 2021, 3941923. [Google Scholar] [CrossRef]



| Elements | Concentration (mg/L) |
|---|---|
| Be | 0.10 |
| Ca | 195.8 |
| Cr | 1.771 |
| Cd | 0.002 |
| Co | 0.305 |
| Cu | 3.602 |
| Fe | 378.0 |
| Li | 0.259 |
| Mg | 323.4 |
| Mn | 24.98 |
| Mo | 0.056 |
| Ni | 1.284 |
| Pb | 0.156 |
| Sb | 0.164 |
| Se | 0.014 |
| Sr | 5.327 |
| Ti | 24.86 |
| V | 0.378 |
| Zn | 2.825 |
| Kinetic | Parameter | Value |
|---|---|---|
| Pseudo-first order | (mg/g) | 30.4382 |
| (min−1) | 2.5 × 10−7 | |
| 0.5746 | ||
| Pseudo-second order | (mg/g) | 1.01 |
| (g/mg. min) | 0.019 × 10−1 | |
| 0.9853 | ||
| Intra-particle diffusion | (mg/g. min3/2) | 0.0813 |
| (mg/g) | 0.2209 | |
| 0.8062 |
| Kinetic | Parameter | Value |
|---|---|---|
| Pseudo-first order | (mg/g) | 20.368 |
| (min−1) | 5.8333 × 10−7 | |
| 0.5843 | ||
| Pseudo-second order | (mg/g) | 2.19 |
| (g/mg. min) | 0.0101 × 10−1 | |
| 0.993 | ||
| Intra-particle diffusion | (mg/g. min3/2) | 0.1805 |
| (mg/g) | 0.4587 | |
| 0.8257 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yadav, V.K.; Amari, A.; Gacem, A.; Elboughdiri, N.; Eltayeb, L.B.; Fulekar, M.H. Treatment of Fly-Ash-Contaminated Wastewater Loaded with Heavy Metals by Using Fly-Ash-Synthesized Iron Oxide Nanoparticles. Water 2023, 15, 908. https://doi.org/10.3390/w15050908
Yadav VK, Amari A, Gacem A, Elboughdiri N, Eltayeb LB, Fulekar MH. Treatment of Fly-Ash-Contaminated Wastewater Loaded with Heavy Metals by Using Fly-Ash-Synthesized Iron Oxide Nanoparticles. Water. 2023; 15(5):908. https://doi.org/10.3390/w15050908
Chicago/Turabian StyleYadav, Virendra Kumar, Abdelfattah Amari, Amel Gacem, Noureddine Elboughdiri, Lienda Bashier Eltayeb, and M. H. Fulekar. 2023. "Treatment of Fly-Ash-Contaminated Wastewater Loaded with Heavy Metals by Using Fly-Ash-Synthesized Iron Oxide Nanoparticles" Water 15, no. 5: 908. https://doi.org/10.3390/w15050908










