Application of Response Surface Methodology to Optimize Coagulation Treatment Process of Urban Drinking Water Using Polyaluminium Chloride
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
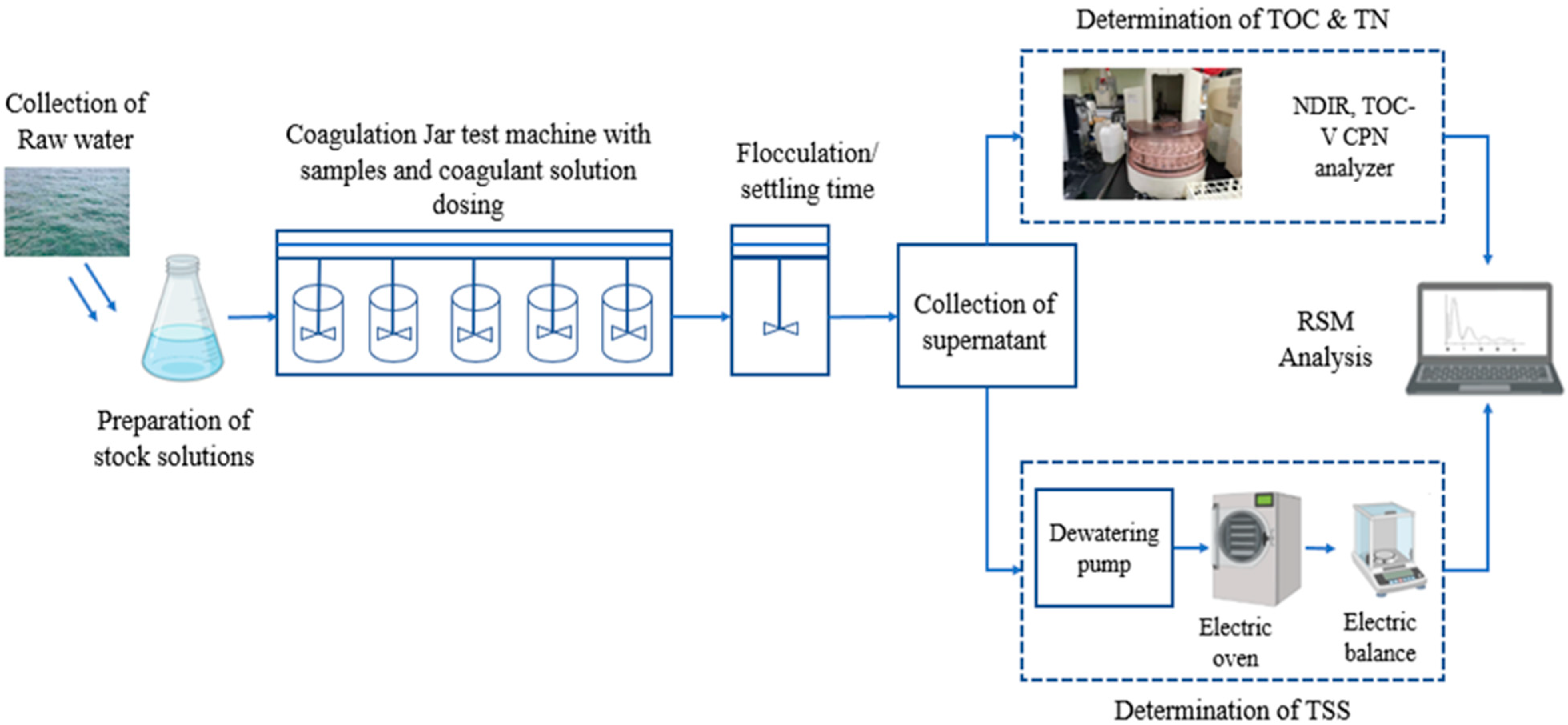
2.2. Jar Test Procedure
2.3. Pre-Experiment
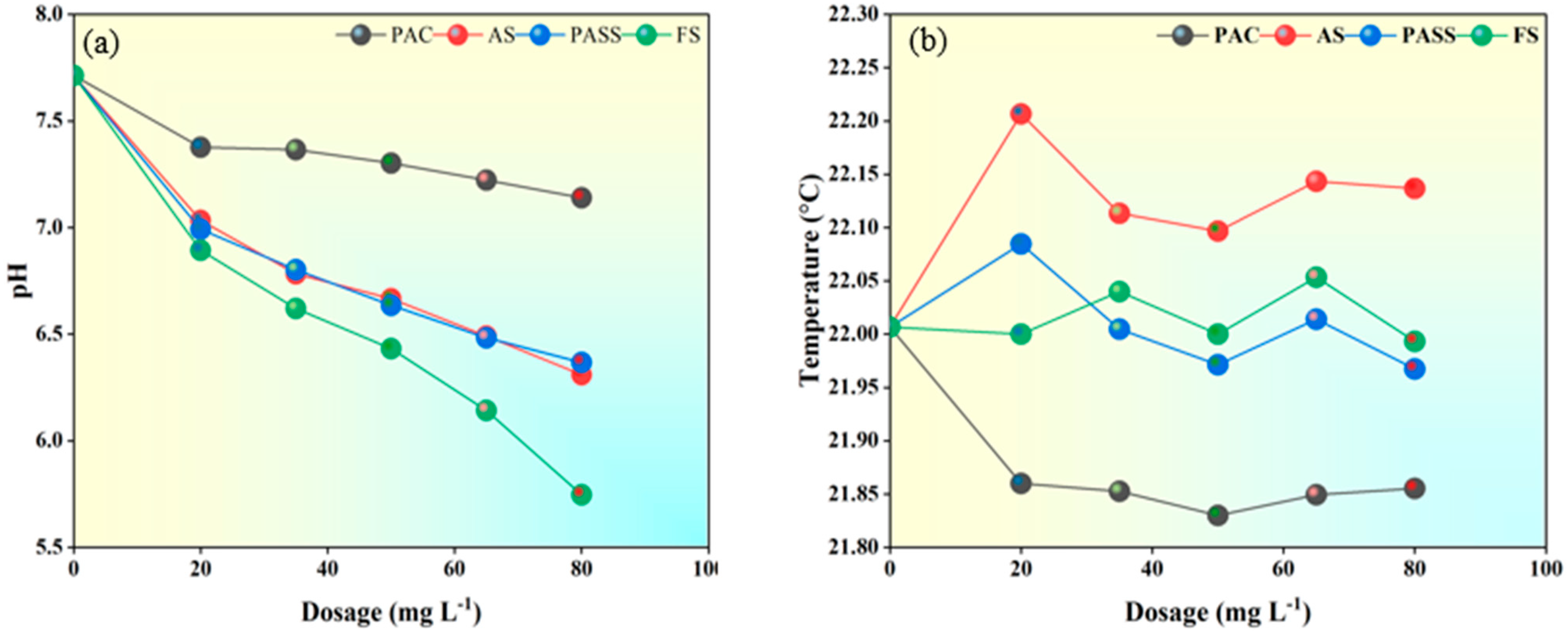
2.3.1. Coagulant Dosage vs. Raw Water pH
2.3.2. Coagulant Dosages vs. Raw Water Temperature
2.4. Data and Statistical Analysis
2.5. Response Surface Methodology and Optimization
3. Results and Discussion
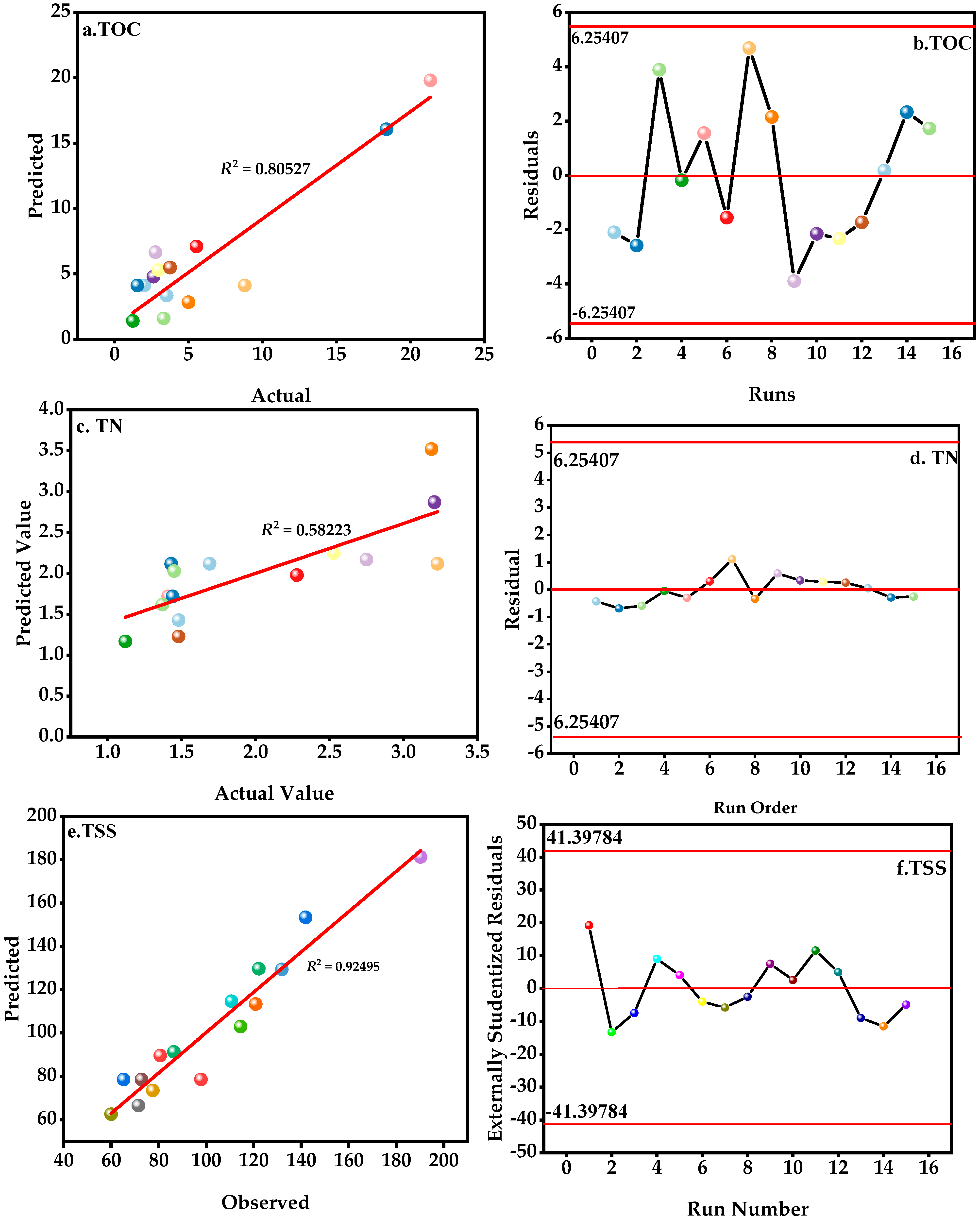
3.1. Fitting Models
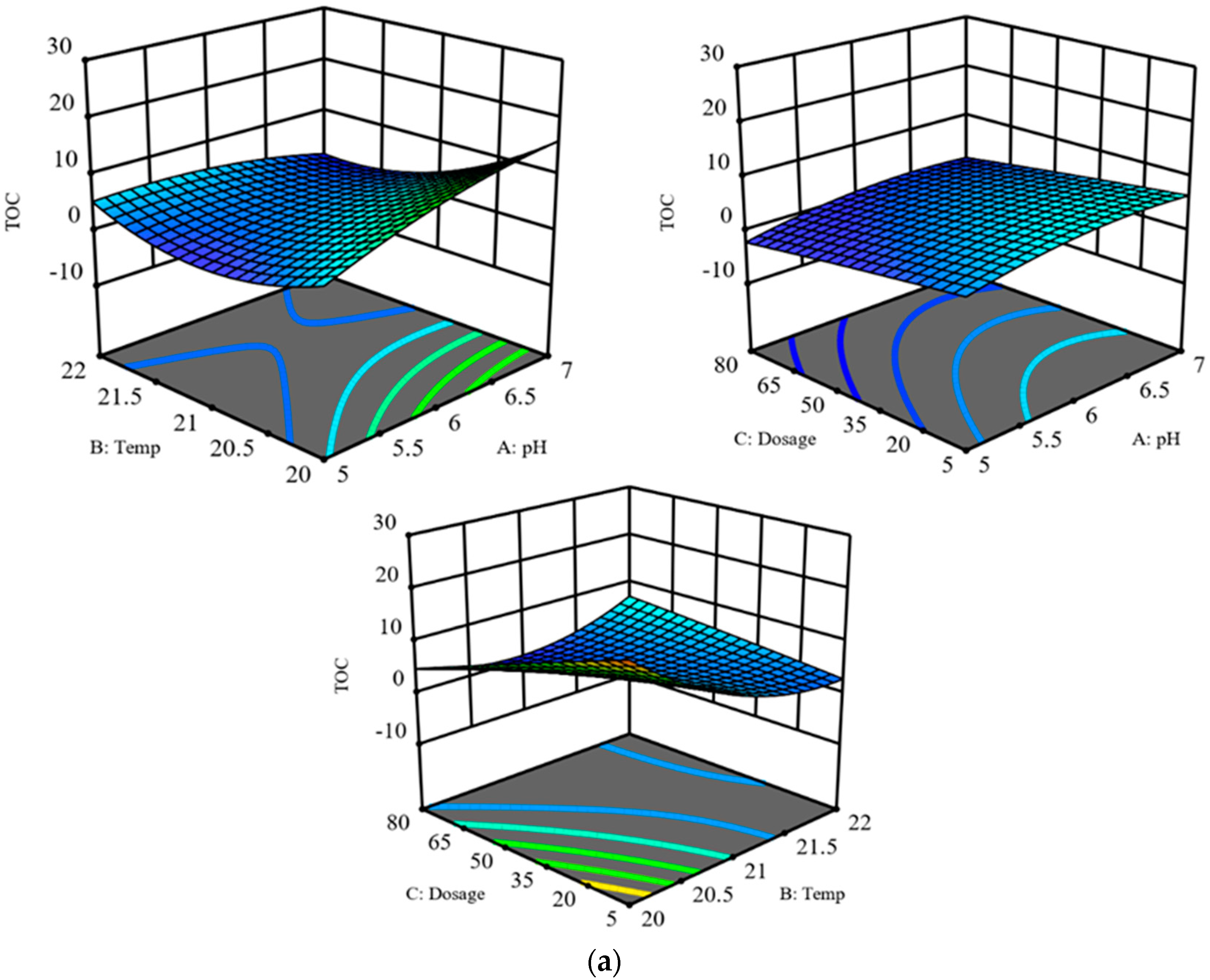
3.2. Process Analysis
3.3. Process Optimization
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Xia, J.; Li, Z.; Zeng, S.; Zou, L.; She, D.; Cheng, D. Perspectives on eco-water security and sustainable development in the Yangtze River Basin. Geosci. Lett. 2021, 8, 18. [Google Scholar] [CrossRef]
- Wang, Y.; Dong, W.; Boelens, L. The Interaction of City and Water in the Yangtze River Delta, a Natural/Artificial Comparison with Euro Delta. Sustainability 2018, 10, 109. [Google Scholar] [CrossRef] [Green Version]
- Wang, Z.; Shao, D.; Westerhoff, P. Wastewater discharge impact on drinking water sources along the Yangtze River (China). Sci. Total Environ. 2017, 599–600, 1399–1407. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Zang, L.; Shen, G.; Liu, M.; Du, W.; Fei, J.; Yang, L.; Chen, L.; Wang, X.; Liu, W.; et al. Resolution of the Ongoing Challenge of Estimating Nonpoint Source Neonicotinoid Pollution in the Yangtze River Basin Using a Modified Mass Balance Approach. Environ. Sci. Technol. 2019, 53, 2539–2548. [Google Scholar] [CrossRef]
- Ding, X.; Liu, L. Long-Term Effects of Anthropogenic Factors on Nonpoint Source Pollution in the Upper Reaches of the Yangtze River. Sustainability 2019, 11, 2246. [Google Scholar] [CrossRef] [Green Version]
- Xu, Z.; Zhang, X.; Xie, J.; Yuan, G.; Tang, X.; Sun, X.; Yu, G. Total Nitrogen Concentrations in Surface Water of Typical Agro- and Forest Ecosystems in China, 2004–2009. PLoS ONE 2014, 9, e92850. [Google Scholar] [CrossRef]
- Villa, A.; Fölster, J.; Kyllmar, K. Determining suspended solids and total phosphorus from turbidity: Comparison of high-frequency sampling with conventional monitoring methods. Environ. Monit. Assess. 2019, 191, 605. [Google Scholar] [CrossRef] [Green Version]
- Ang, W.L.; Mohammad, A.W. State of the art and sustainability of natural coagulants in water and wastewater treatment. J. Clean. Prod. 2020, 262, 121267. [Google Scholar] [CrossRef]
- Corral Bobadilla, M.; Lorza, R.L.; Escribano García, R.; Somovilla Gómez, F.; Vergara González, E.P. Coagulation: Determination of Key Operating Parameters by Multi-Response Surface Methodology Using Desirability Functions. Water 2019, 11, 398. [Google Scholar] [CrossRef] [Green Version]
- Suquet, J.; Godo-Pla, L.; Valentí, M.; Verdaguer, M.; Martin, M.J.; Poch, M.; Monclús, H. Development of an environmental decision support system for enhanced coagulation in drinking water production. Water 2020, 12, 2115. [Google Scholar] [CrossRef]
- Chua, S.-C.; Chong, F.-K.; Malek, M.A.; Ul Mustafa, M.R.; Ismail, N.; Sujarwo, W.; Lim, J.-W.; Ho, Y.-C. Optimized Use of Ferric Chloride and Sesbania Seed Gum (SSG) as Sustainable Coagulant Aid for Turbidity Reduction in Drinking Water Treatment. Sustainability 2020, 12, 2273. [Google Scholar] [CrossRef] [Green Version]
- Guimarães, R.N.; Moreira, V.R.; Amaral, M.C.S. Membrane technology as an emergency response against drinking water shortage in scenarios of dam failure. Chemosphere 2022, 309, 136618. [Google Scholar] [CrossRef] [PubMed]
- Kwiecińska-Mydlaka, A.; Sieradzkib, W.; Jagustyna, B.; Wodzisławskaa, K. The use of membrane filtration for removal of aluminum from drinking water sources. Desalin. Water Treat. 2021, 243, 75–82. [Google Scholar] [CrossRef]
- Zhang, Y.; Peng, S.; Zhao, X.-H. Assessment of the value of NOM treatment processes based on a matter-element model. Water Supply 2016, 17, 870–878. [Google Scholar] [CrossRef]
- Saha, S.; Boro, R.; Das, C. Treatment of tea industry wastewater using coagulation-spinning basket membrane ultrafiltration hybrid system. J. Environ. Manag. 2019, 244, 180–188. [Google Scholar] [CrossRef] [PubMed]
- Dąbrowska, L. Removal of Organic Matter from Surface Water using Coagulants with Various Basicity. J. Ecol. Eng. 2016, 17, 66–72. [Google Scholar] [CrossRef]
- Tian, C.; Zhao, Y.-X. Dosage and pH dependence of coagulation with polytitanium salts for the treatment of Microcystis aeruginosa-laden and Microcystis wesenbergii-laden surface water: The influence of basicity. J. Water Process. Eng. 2021, 39, 101726. [Google Scholar] [CrossRef]
- Naceradska, J.; Pivokonska, L.; Pivokonsky, M. On the importance of pH value in coagulation. J. Water Supply Res. Technol. AQUA 2019, 68, 222–230. [Google Scholar] [CrossRef]
- Arruda, P.M.; Pereira-Filho, E.R.; Libânio, M.; Fagnani, E. Response surface methodology applied to tropical freshwater treatment. Environ. Technol. 2018, 41, 901–911. [Google Scholar] [CrossRef]
- Khairul Zaman, N.; Rohani, R.; Izni Yusoff, I.; Kamsol, M.A.; Basiron, S.A.; Abd Rashid, A.I. Eco-Friendly Coagulant versus Industrially Used Coagulants: Identification of Their Coagulation Performance, Mechanism and Optimization in Water Treatment Process. Int. J. Environ. Res. Public Health 2021, 18, 9164. [Google Scholar] [CrossRef]
- Nti, S.O.; Buamah, R.; Atebiya, J. Polyaluminium chloride dosing effects on coagulation performance: Case study, Barekese, Ghana. Water Pract. Technol. 2021, 16, 1215–1223. [Google Scholar] [CrossRef]
- Zarchi, I.; Friedler, E.; Rebhun, M. Polyaluminium chloride as an alternative to alum for the direct filtration of drinking water. Environ. Technol. 2013, 34, 1199–1209. [Google Scholar] [CrossRef] [PubMed]
- Abdo, S.M.; Mahmoud, R.H.; Youssef, M.; El-Naggar, M.E. Cationic Starch and Polyaluminum Chloride as Coagulants for River Nile Water Treatment. Groundw. Sustain. Dev. 2020, 10, 100331. [Google Scholar] [CrossRef]
- Khuri, A.I.; Cornell, J.A. Response Surfaces-Designs and Analyses: Revised and Expanded; CRC Press: Boca Raton, FL, USA, 2018. [Google Scholar] [CrossRef]
- DB, National Standard of Shanghai City: Living Drinking Water Quality Standard (DB31/T 1091-2018). Available online: https://dbba.sacinfo.org.cn/stdDetail/6930f2db356573fcc8d293c0783ab2e8 (accessed on 31 May 2020).
- GB, Standards for Drinking Water Quality (GB5749-2006): National Standard of the Peoples Republic of China. Available online: https://www.chinesestandard.net/PDF.aspx/GB5749-2006 (accessed on 5 May 2020).
- ASTMD2035, Standard Practice for Coagulation-Flocculation Jar Test of Water. Available online: https://global.ihs.com/doc_detail.cfm?document_name=ASTM%20D2035&item_s_key=00016243 (accessed on 7 June 2021).
- Potter, B.; Wimsatt, J. Method 415.3: Determination of Total Organic Carbon and Specific UV Absorbance at 254 nm in Source Water and Drinking Water; US Environmental Protection Agency: Cincinnati, OH, USA, 2005.
- ASTMD2540, Standard Test Method for Drop-Weight Sensitivity of Liquid Monopropellants. Available online: https://global.ihs.com/doc_detail.cfm?document_name=ASTM%20D2540&item_s_key=00016709 (accessed on 7 June 2021).
- Boughou, N.; Majdy, I.; Cherkaoui, E.; Khamar, M.; Abderrahman, N. Effect of pH and time on the treatment by coagulation from slaughterhouse of the city of Rabat. MATEC Web Conf. 2018, 149, 02091. [Google Scholar] [CrossRef]
- U.S. Environmental Protection Agency. Integrated Science Assessment for Oxides of Nitrogen and Sulfur: Ecological Criteria; EPA/600/R-08/082F; U.S. Environmental Protection Agency: Washington, DC, USA, 2008. Available online: https://www.epa.gov/caddis-vol2/ph (accessed on 17 June 2021).
- Kaji, A.; Taheriyoun, M.; Taebi, A.; Nazari-Sharabian, M. Comparison and optimization of the performance of natural-based non-conventional coagulants in a water treatment plant. J. Water Supply Res. Technol. AQUA 2019, 69, 28–38. [Google Scholar] [CrossRef]
- Lartey-Young, G.; Ma, L. Optimization, equilibrium, adsorption behaviour of Cu/Zn/Fe LDH and LDHBC composites towards atrazine reclamation in an aqueous environment. Chemosphere 2022, 293, 133526. [Google Scholar] [CrossRef]
- Choi, A.E.S.; Roces, S.A.; Dugos, N.P.; Wan, M.W. Ultrasound assisted oxidative desulfurization: A comprehensive optimization analysis using untreated diesel oil. Comput. Chem. Eng. 2022, 166, 107965. [Google Scholar] [CrossRef]
- Barilla, G.R.H.; Chen, C.A.W.; Valencia, M.Z.M.; Dugos, N.P.; Choi, A.E.S. Mixing assisted oxidative desulfurization using a synthesized catalyst of the activated carbon supported phosphotungstic acid: A process optimization study. S. Afr. J. Chem. Eng. 2022, 42, 61–71. [Google Scholar] [CrossRef]
- Dahiru, T. P-value, a true test of statistical significance? A cautionary note. Ann. Ib. Postgrad. Med. 2008, 6, 21–26. [Google Scholar] [CrossRef] [Green Version]
- Ölmez, T. The optimization of Cr(VI) reduction and removal by electrocoagulation using response surface methodology. J. Hazard. Mater. 2009, 162, 1371–1378. [Google Scholar] [CrossRef]
- Ghafari, S.; Aziz, H.A.; Isa, M.H.; Zinatizadeh, A.A. Application of response surface methodology (RSM) to optimize coagulation–flocculation treatment of leachate using poly-aluminum chloride (PAC) and alum. J. Hazard. Mater. 2009, 163, 650–656. [Google Scholar] [CrossRef] [PubMed]

| Samples’ Parameters | Urban Drinking Water Treatment Standards | ||
|---|---|---|---|
| Shanghai Standard (DB31/T1091-2018) [25] | China National Standard (GB5749-2006) [26] | ||
| pH | 7.71 ± 0.20 | ≥6.5 ≤8.5 | 6.5–8.5 |
| Temperature (°C) | 22.01 ± 1.05 | - | - |
| Turbidity (NTU) | 144.89 ± 96.44 | <0.5 | 3 |
| TOC (mgL−1) | 6.54 ± 6.12 | <3 | 5 |
| TN (mgL−1) | 2.52 ± 0.63 | <0.15 | 1 |
| TSS (mgL−1) | 295.14 ± 140.62 | <500 | 1000 |
| Factors | Unit | Code | Levels | |
|---|---|---|---|---|
| Low | High | |||
| pH | - | 5 | 7 | |
| Temperature | °C | 21 | 22 | |
| Dosage | mgL−1 | 5 | 80 | |
| Runs | x1 | x2 | x3 | TOC | TN | TSS | |||
|---|---|---|---|---|---|---|---|---|---|
| O | P | O | P | O | P | ||||
| 1 | 6 | 21 | 42.5 | 2.02 | 4.12 | 1.69 | 2.12 | 97.8 | 78.6 |
| 2 | 6 | 21 | 42.5 | 1.54 | 4.12 | 1.43 | 2.12 | 65.3 | 78.6 |
| 3 | 5 | 21 | 80 | 1.74 | −2.15 | 1.45 | 2.03 | 122.2 | 129.7 |
| 4 | 7 | 21 | 80 | 1.24 | 1.41 | 1.12 | 1.17 | 190.3 | 181.3 |
| 5 | 6 | 20 | 5 | 21.36 | 19.8 | 1.41 | 1.72 | 77.6 | 73.5 |
| 6 | 6 | 22 | 80 | 5.54 | 7.1 | 2.28 | 1.98 | 110.6 | 114.7 |
| 7 | 6 | 21 | 42.5 | 8.81 | 4.12 | 3.23 | 2.12 | 72.8 | 78.6 |
| 8 | 6 | 22 | 5 | 5 | 2.84 | 3.19 | 3.52 | 60.0 | 62.6 |
| 9 | 7 | 21 | 5 | 2.77 | 6.66 | 2.75 | 2.17 | 120.8 | 113.3 |
| 10 | 6 | 20 | 80 | 2.63 | 4.79 | 3.21 | 2.87 | 131.9 | 129.3 |
| 11 | 5 | 22 | 42.5 | 2.97 | 5.3 | 2.53 | 2.25 | 114.5 | 103.0 |
| 12 | 5 | 20 | 42.5 | 3.75 | 5.49 | 1.48 | 1.23 | 71.5 | 66.6 |
| 13 | 5 | 21 | 5 | 3.53 | 3.35 | 1.48 | 1.43 | 80.7 | 89.7 |
| 14 | 7 | 20 | 42.5 | 18.39 | 16.06 | 1.44 | 1.72 | 141.9 | 153.4 |
| 15 | 7 | 22 | 42.5 | 3.33 | 1.6 | 1.37 | 1.62 | 86.5 | 91.4 |
| TOC | TN | TSS | ||||
|---|---|---|---|---|---|---|
| Source | F-Value | p-Value | F-Value | p-Value | F-Value | p-Value |
| A-pH | 1.25 | 0.3143 b | 0.0117 | 0.9181 b | 11.64 | 0.019 a |
| B-Temp | 5.68 | 0.0629 b | 0.6412 | 0.4596 b | 1.35 | 0.2984 b |
| C-Dosage | 3.06 | 0.1406 b | 0.1135 | 0.7499 b | 23.93 | 0.0045 a |
| AB | 2.7 | 0.1615 b | 0.476 | 0.5209 b | 9.95 | 0.0253 a |
| AC | 0.0009 | 0.977 b | 0.9735 | 0.3691 b | 0.8006 | 0.4119 b |
| BC | 4.92 | 0.0774 b | 2.76 | 0.1574 b | 0.0139 | 0.9107 b |
| A2 | 0.5413 | 0.4949 b | 2.15 | 0.2026 b | 12.95 | 0.0156 a |
| B2 | 4.23 | 0.0947 b | 0.2336 | 0.6493 b | 0.2745 | 0.6227 b |
| C2 | 0.0039 | 0.9527 b | 0.2214 | 0.6578 b | 6.47 | 0.0517 b |
| Residual | ||||||
| Lack-of-Fit | 1.24 | 0.4762 | 0.4934 | 0.7226 | 0.7273 | 0.6231 |
| p < 0.05 (significant); p > 0.05 (non-significant); a significant; b non-significant. | ||||||
| Model Parameters | ||||||
| Parameter | R2 | Adjusted R2 | Adequate Precision | PRESS | ||
| TOC | 0.8193 | 0.4941 | 6.1888 | 1055.62 | ||
| TN | 0.6088 | 0.0954 | 3.5497 | 26.68 | ||
| TSS | 0.9303 | 0.8048 | 9.3197 | 11467.34 | ||
| pH | Temperature (°C) | Dosage (mgL−1) | TOC (mgL−1) | TN (mgL−1) | TSS (mgL−1) | Desirability |
|---|---|---|---|---|---|---|
| 6.9 | 20 | 9.7 | 22.174 | 1.753 | 129.358 | 1 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yateh, M.; Lartey-Young, G.; Li, F.; Li, M.; Tang, Y. Application of Response Surface Methodology to Optimize Coagulation Treatment Process of Urban Drinking Water Using Polyaluminium Chloride. Water 2023, 15, 853. https://doi.org/10.3390/w15050853
Yateh M, Lartey-Young G, Li F, Li M, Tang Y. Application of Response Surface Methodology to Optimize Coagulation Treatment Process of Urban Drinking Water Using Polyaluminium Chloride. Water. 2023; 15(5):853. https://doi.org/10.3390/w15050853
Chicago/Turabian StyleYateh, Mohamed, George Lartey-Young, Fengting Li, Mei Li, and Yulin Tang. 2023. "Application of Response Surface Methodology to Optimize Coagulation Treatment Process of Urban Drinking Water Using Polyaluminium Chloride" Water 15, no. 5: 853. https://doi.org/10.3390/w15050853







