Ultraviolet Radiation-Assisted Preparation of a Novel Biomass Fiber to Remove Cadmium from Wastewater
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Synthesis of LF@AA
2.3. Characterization Analysis
2.4. Factors Affecting Adsorption Properties
2.5. Adsorption: Kinetic Study
2.6. Adsorption: Equilibrium Study
2.7. Regeneration
3. Results and Discussion
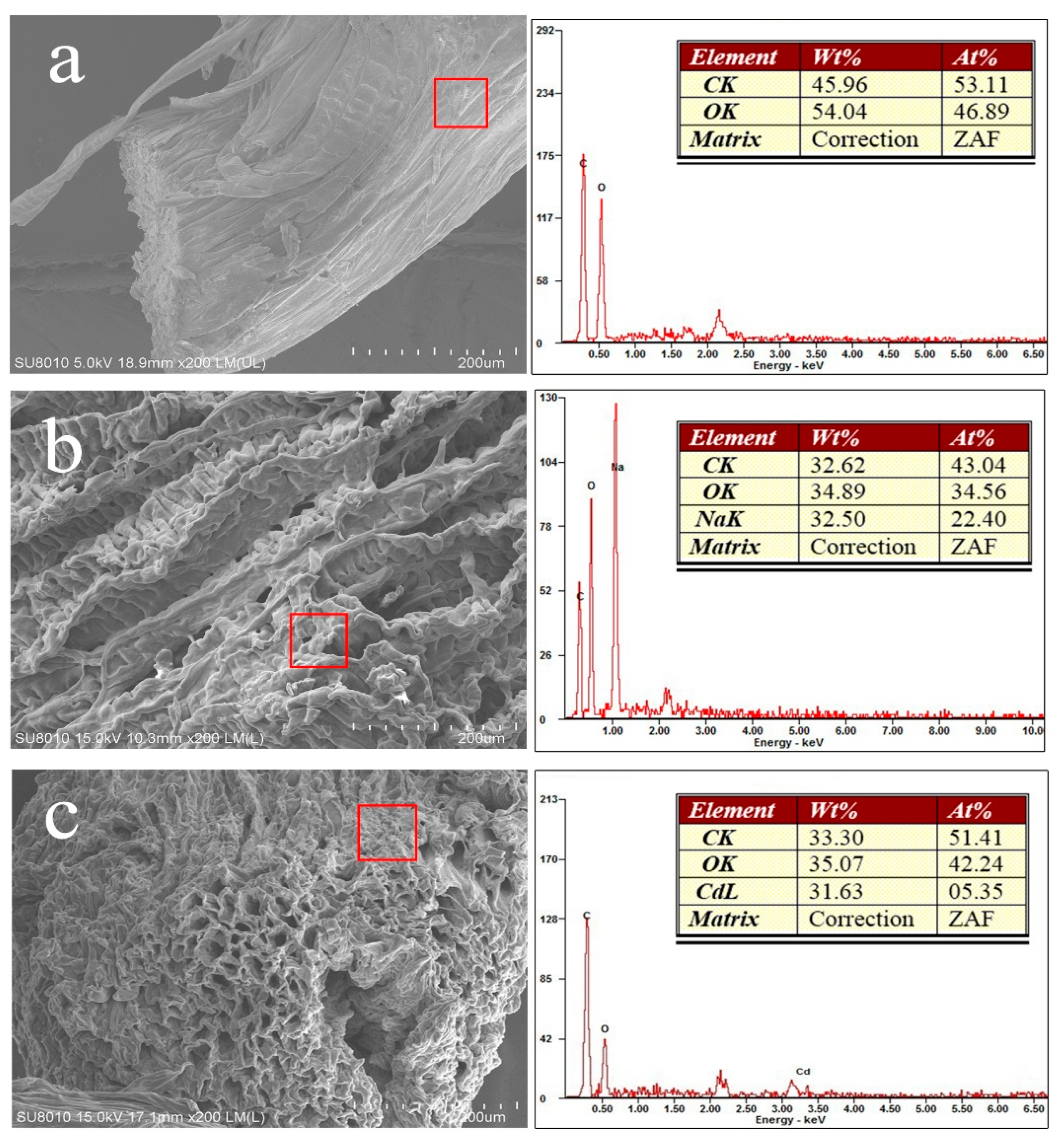
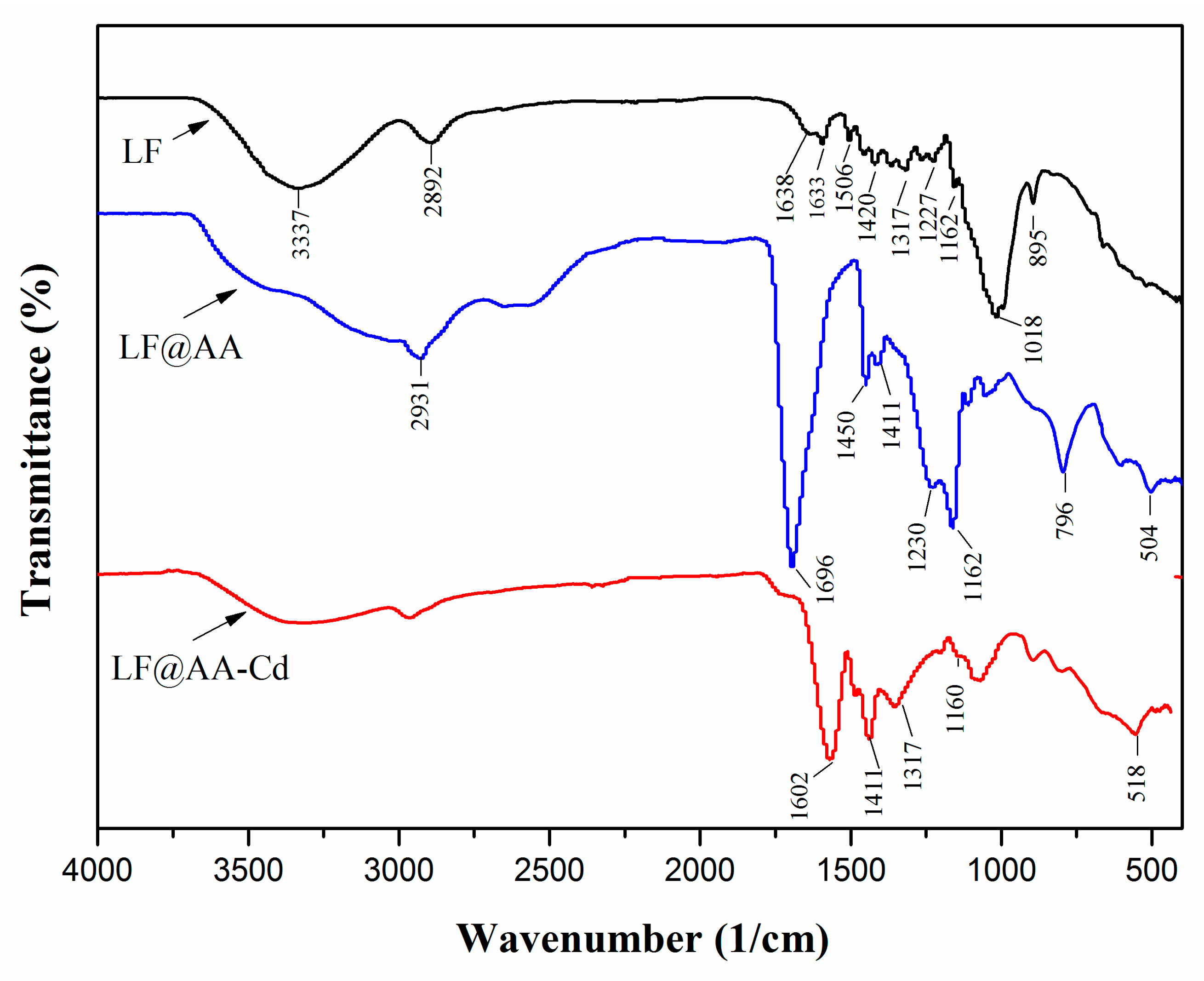
3.1. Adsorbent Characterization
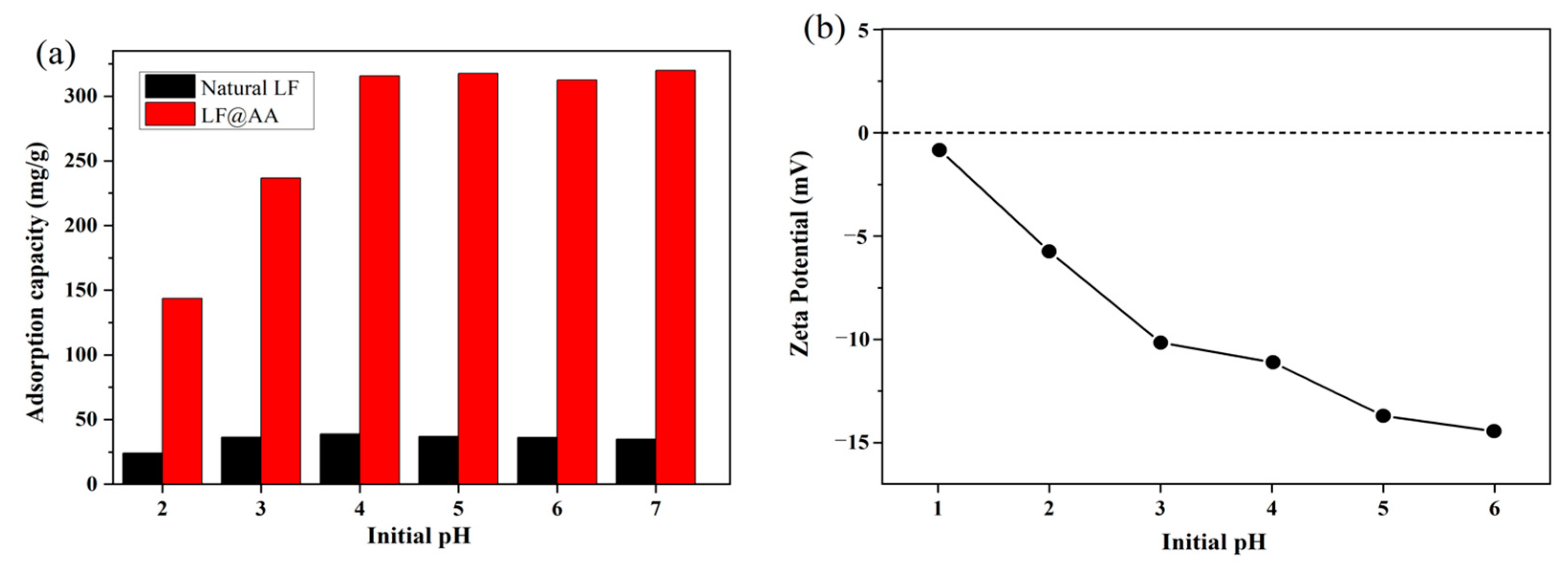
3.2. Effect of pH
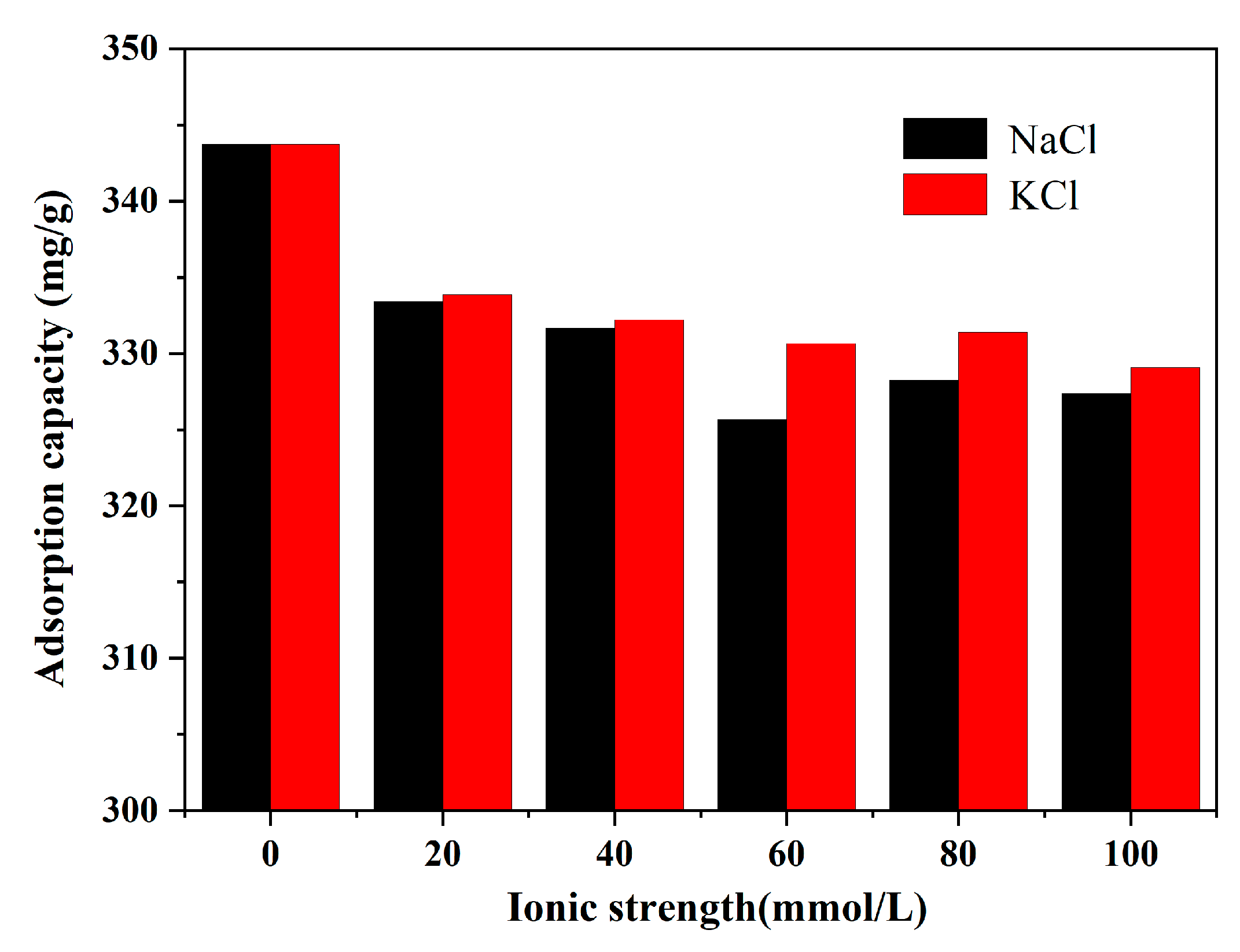
3.3. Effect of Ionic Strength
3.4. Kinetic Study
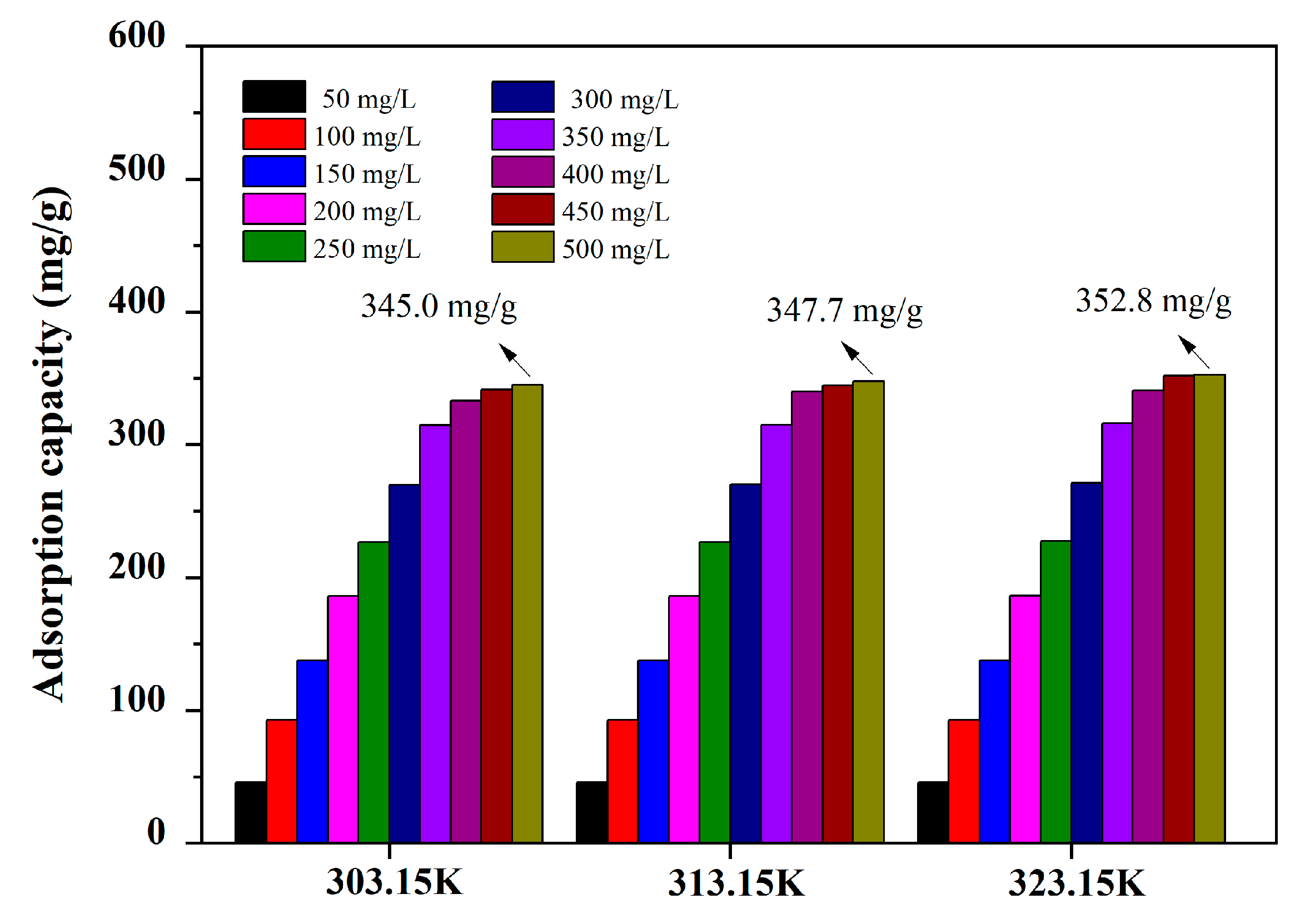
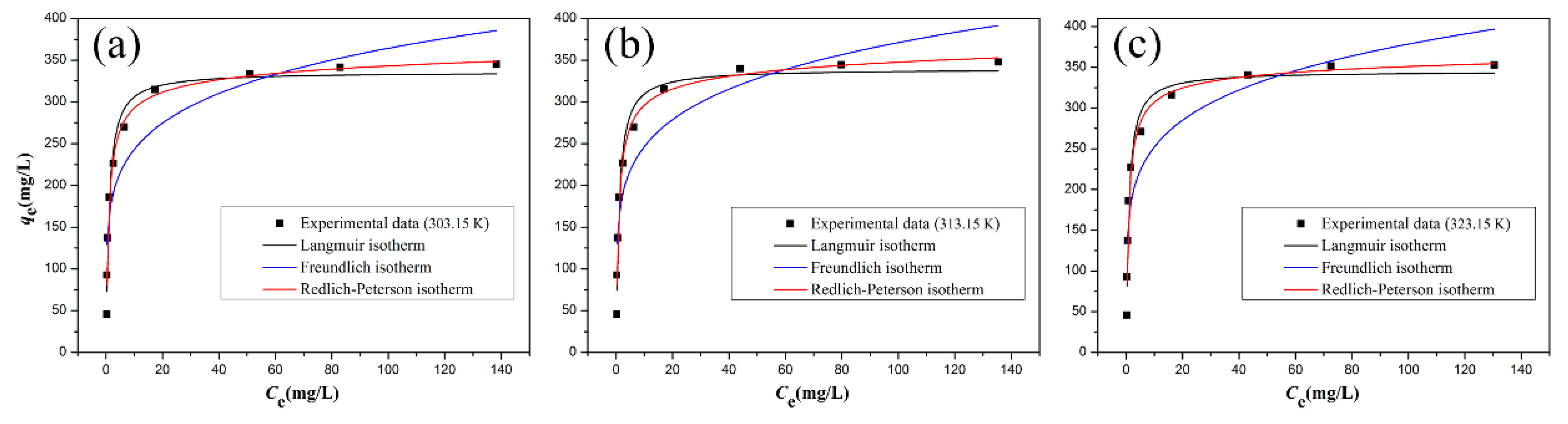
3.5. Adsorption Isotherms
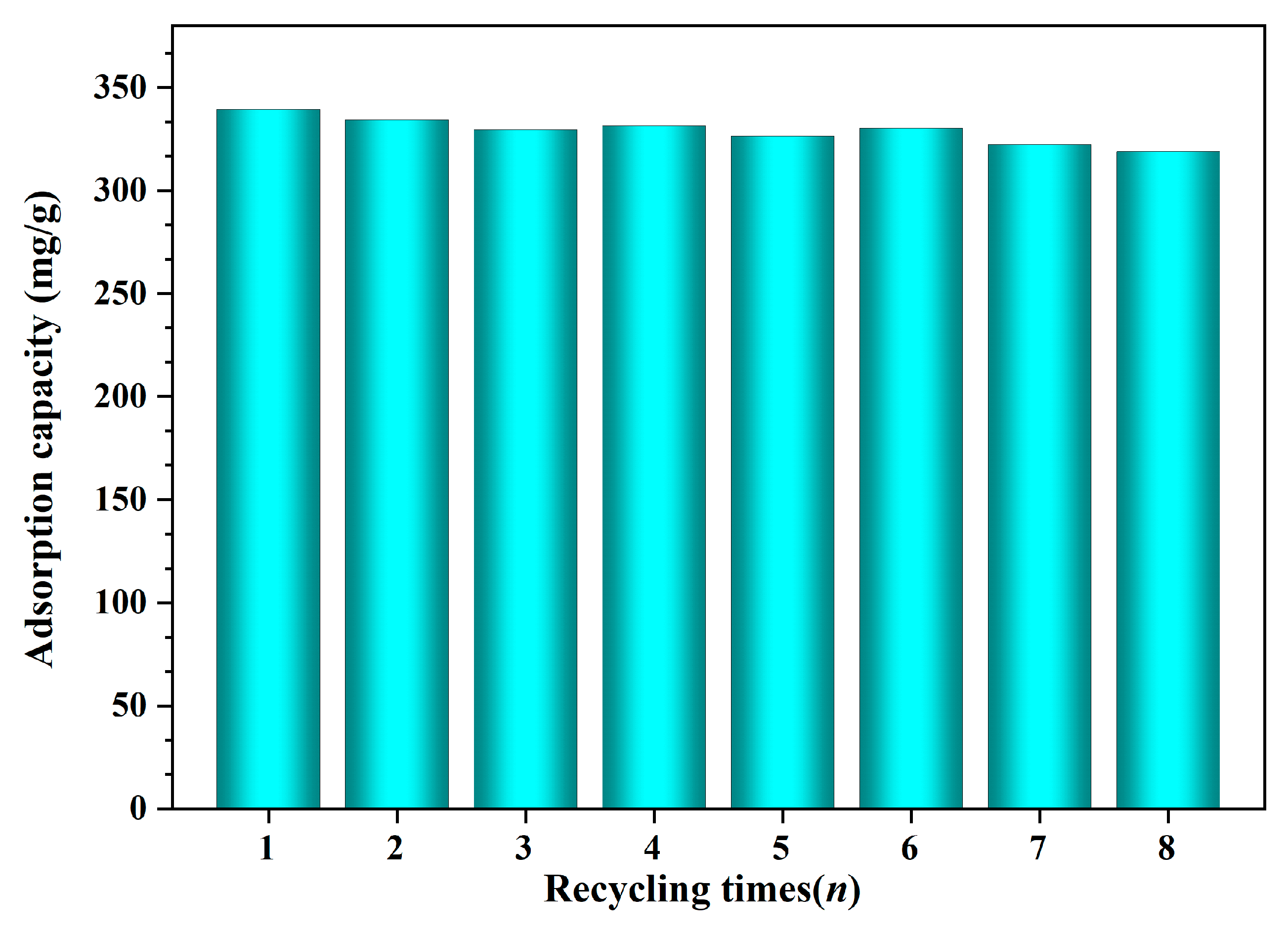
3.6. Regeneration
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Åkesson, A.; Barregard, L.; Bergdahl, I.A.; Nordberg, G.F.; Nordberg, M.; Skerfving, S. Non-Renal effects and the risk assessment of environmental cadmium exposure. Environ. Health Perspect. 2014, 122, 431–438. [Google Scholar] [CrossRef] [Green Version]
- Marques, P.; Rosa, M.F.; Pinheiro, H.M. pH effects on the removal of CU2+, Cd2+ and Pb2+ from aqueous solution by waste brewery biomass. Bioprocess Eng. 2000, 23, 135–141. [Google Scholar] [CrossRef]
- EL-Hendawy, A.A. The role of surface chemistry and solution pH on the removal of Pb2+ and Cd2+ ions via effective adsorbents from low-cost biomass. J. Hazard. Mater. 2009, 167, 260–267. [Google Scholar] [CrossRef]
- Yang, G.; Wang, Z.; Xian, Q.; Shen, F.; Sun, C.; Zhang, Y.; Wu, J. Effects of pyrolysis temperature on the physicochemical properties of biochar derived from vermicompost and its potential use as an environmental amendment. Rsc Adv. 2015, 5, 40117–40125. [Google Scholar] [CrossRef]
- Wan Ngah, W.S.; Hanafiah, M.A.K.M. Removal of heavy metal ions from wastewater by chemically modified plant wastes as adsorbents: A review. Bioresour. Technol. 2008, 99, 3935–3948. [Google Scholar] [CrossRef]
- Feng, N.; Guo, X.; Liang, S.; Zhu, Y.; Liu, J. Biosorption of heavy metals from aqueous solutions by chemically modified orange peel. J. Hazard. Mater. 2011, 185, 49–54. [Google Scholar] [CrossRef]
- Shakya, A.; Agarwal, T. Removal of Cr(VI) from water using pineapple peel derived biochars: Adsorption potential and re-usability assessment. J. Mol. Liq. 2019, 293, 111497. [Google Scholar] [CrossRef]
- Lemraski, E.G.; Sharafinia, S. Kinetics, equilibrium and thermodynamics studies of Pb2+ adsorption onto new activated carbon prepared from Persian mesquite grain. J. Mol. Liq. 2016, 219, 482–492. [Google Scholar] [CrossRef]
- Mayakaduwa, S.S.; Kumarathilaka, P.; Herath, I.; Ahmad, M.; Al-Wabel, M.; Ok, Y.S.; Usman, A.; Abduljabbar, A.; Vithanage, M. Equilibrium and kinetic mechanisms of woody biochar on aqueous glyphosate removal. Chemosphere 2016, 144, 2516–2521. [Google Scholar] [CrossRef]
- Xu, X.; Gao, B.; Huang, X.; Ling, J.; Song, W.; Yue, Q. Physicochemical characteristics of epichlorohydrin, pyridine and trimethylamine functionalized cotton stalk and its adsorption/desorption properties for perchlorate. J. Colloid Interface Sci. 2015, 440, 219–228. [Google Scholar] [CrossRef]
- Hokkanen, S.; Bhatnagar, A.; Sillanpaa, M. A review on modification methods to cellulose-based adsorbents to improve adsorption capacity. Water Res. 2016, 91, 156–173. [Google Scholar] [CrossRef]
- Zhou, Q.; Yang, H.; Yan, C.; Luo, W.; Li, X.; Zhao, J. Synthesis of carboxylic acid functionalized diatomite with a micro-villous surface via UV-induced graft polymerization and its adsorption properties for Lanthanum(III) ions. Colloids Surf. A 2016, 501, 9–16. [Google Scholar] [CrossRef]
- Li, X.J.; Yan, C.J.; Luo, W.J.; Gao, Q.; Zhou, Q.; Liu, C.; Zhou, S. Exceptional cerium(III) adsorption performance of poly(acrylic acid) brushes-decorated attapulgite with abundant and highly accessible binding sites. Chem. Eng. J. 2015, 284, 333–342. [Google Scholar] [CrossRef]
- Zhou, Q.; Yan, C.; Luo, W. Preparation of a novel carboxylate-rich wheat straw through surface graft modification for efficient separation of Ce(III) from wastewater. Mater. Des. 2016, 97, 195–203. [Google Scholar] [CrossRef]
- Liu, C.; Yan, C.; Luo, W.; Li, X.; Ge, W.; Zhou, S. Simple preparation and enhanced adsorption properties of loofah fiber adsorbent by ultraviolet radiation graft. Mater. Lett. 2015, 157, 303–306. [Google Scholar] [CrossRef]
- Chen, Q.; Shi, Q.; Gorb, S.N.; Li, Z. A multiscale study on the structural and mechanical properties of the luffa sponge from Luffa cylindrica plant. J. Biomech. 2014, 47, 1332–1339. [Google Scholar] [CrossRef]
- Demir, H.; Atikler, U.; Balköse, D.; Tıhmınlıoğlu, F. The effect of fiber surface treatments on the tensile and water sorption properties of polypropylene–luffa fiber composites. Compos. Part A Appl. Sci. Manuf. 2006, 37, 447–456. [Google Scholar] [CrossRef] [Green Version]
- Thakur, V.K.; Thakur, M.K. Processing and characterization of natural cellulose fibers/thermoset polymer composites. Carbohydr. Polym. 2014, 109, 102–117. [Google Scholar] [CrossRef]
- Alhijazi, M.; Safaei, B.; Zeeshan, Q.; Asmael, M.; Qin, Z. Recent Developments in Luffa Natural Fiber Composites: Review. Sustainability 2020, 12, 7683. [Google Scholar] [CrossRef]
- Khadir, A.; Motamedi, M.; Pakzad, E.; Sillanpää, M.; Mahajan, S. The prospective utilization of Luffa fibres as a lignocellulosic bio-material for environmental remediation of aqueous media: A review. J. Environ. Chem. Eng. 2020, 9, 104691. [Google Scholar] [CrossRef]
- Shahidi, A.; Jalilnejad, N.; Jalilnejad, E. A study on adsorption of cadmium(II) ions from aqueous solution using Luffa cylindrica. Desalination Water Treat. 2015, 53, 3570–3579. [Google Scholar] [CrossRef]
- Ahmad, R.; Haseeb, S. Kinetic, isotherm and thermodynamic studies for the removal of Pb2+ ion by a novel adsorbent Luffa acutangula (LAPR). Desalination Water Treat. 2015, 57, 17826–17835. [Google Scholar] [CrossRef]
- Zhang, J.; Yang, J.; Tian, Q.; Liang, X.; Zhu, Y.; Sand, W.; Li, F.; Ma, C.; Liu, Y.; Yang, B. Durability and performance of loofah sponge as carrier for wastewater treatment with high ammonium. Water Environ. Res. 2019, 91, 581–587. [Google Scholar] [CrossRef]
- Tang, X.; Zhang, Q.; Liu, Z.; Pan, K.; Dong, Y.; Li, Y. Removal of Cu(II) by loofah fibers as a natural and low-cost adsorbent from aqueous solutions. J. Mol. Liq. 2014, 191, 73–78. [Google Scholar] [CrossRef]
- Liu, C.; Yan, C.; Zhou, S.; Ge, W. Fabrication of sponge biomass adsorbent through UV-induced surface-initiated polymerization for the adsorption of Ce(III) from wastewater. Water Sci. Technol. 2017, 75, 2755–2764. [Google Scholar] [CrossRef]
- Ahmadi, M.; Vahabzadeh, F.; Bonakdarpour, B.; Mehranian, M.; Mofarrah, E. Phenolic removal in olive oil mill wastewater using loofah-immobilized Phanerochaete chrysosporium. World J. Microbiol. Biotechnol. 2006, 22, 119–127. [Google Scholar] [CrossRef]
- Sriharsha, D.V.; Lokesh; Kumar, R.; Savitha, J. Immobilized fungi on Luffa cylindrica: An effective biosorbent for the removal of lead. J. Taiwan Inst. Chem. Eng. 2017, 80, 589–595. [Google Scholar]
- Oun, A.A.; Kamal, K.H.; Farroh, K.; Ali, E.F.; Hassan, M.A. Development of fast and high-efficiency sponge-gourd fibers (Luffa cylindrica)/hydroxyapatite composites for removal of lead and methylene blue. Arabian J. Chem. 2021, 14, 103281. [Google Scholar] [CrossRef]
- Li, S.; Liu, F.; Su, Y.; Shao, N.; Zhang, Z. Luffa sponge-derived hierarchical meso/macroporous boron nitride fibers as superior sorbents for heavy metal sequestration. J. Hazard. Mater. 2019, 378, 120669. [Google Scholar] [CrossRef]
- Khadir, A.; Negarestani, M.; Mollahosseini, A. Sequestration of a non-steroidal anti-inflammatory drug from aquatic media by lignocellulosic material (Luffa cylindrica) reinforced with polypyrrole: Study of parameters, kinetics, and equilibrium. J. Environ. Chem. Eng. 2020, 8, 103734. [Google Scholar] [CrossRef]
- Shih, Y.J.; Dong, C.D.; Huang, Y.H.; Huang, C.P. Electro-sorption of ammonium ion onto nickel foam supported highly microporous activated carbon prepared from agricultural residues (dried Luffa cylindrica). Sci. Total Environ. 2019, 673, 296–305. [Google Scholar] [CrossRef]
- Xiao, F.; Cheng, J.; Cao, W.; Yang, C.; Chen, J.; Luo, Z. Removal of heavy metals from aqueous solution using chitosan-combined magnetic biochars. J. Colloid Interface Sci. 2019, 540, 579–584. [Google Scholar] [CrossRef]
- Azizian, S. Kinetic models of sorption: A theoretical analysis. J. Colloid Interface Sci. 2004, 276, 47–52. [Google Scholar] [CrossRef]
- Ghaedi, M.; Hassanzadeh, A.; Kokhdan, S.N. Multiwalled carbon nanotubes as adsorbents for the kinetic and equilibrium study of the removal of alizarin red s and morin. J. Chem. Eng. Data 2011, 56, 2511–2520. [Google Scholar] [CrossRef]
- Ho, Y.S.; Mckay, G. Sorption of dye from aqueous solution by peat. Chem. Eng. J. 1998, 70, 115–124. [Google Scholar] [CrossRef]
- Tan, I.A.; Hameed, B.H.; Ahmad, A.L. Equilibrium and Kinetic Studies on Basic Dye Adsorption by Oil Palm Fibre Activated Carbon. Chem. Eng. J. 2007, 127, 111–119. [Google Scholar] [CrossRef]
- Redlich, O.; Peterson, D.L. A useful adsorption isotherm. J. Phys. Chem. 2007, 63, 1024. [Google Scholar] [CrossRef]
- Karlsson, J.O.; Henriksson, Å.; Michálek, J.; Gatenholm, P. Control of cellulose-supported hydrogel microstructures by three-dimensional graft polymerization of glycol methacrylates. Polymer 2000, 41, 1551–1559. [Google Scholar] [CrossRef]
- Gupta, V.K.; Agarwal, S.; Singh, P.; Pathania, D. Acrylic acid grafted cellulosic Luffa cylindrical fiber for the removal of dye and metal ions. Carbohydr. Polym. 2013, 98, 1214–1221. [Google Scholar] [CrossRef]
- Tarley, C.R.T.; Arruda, M.A.Z. Biosorption of heavy metals using rice milling by-products. Characterisation and application for removal of metals from aqueous effluents. Chemosphere 2004, 54, 987–995. [Google Scholar] [CrossRef]
- Wang, X.; Zheng, Y.; Wang, A. Fast removal of copper ions from aqueous solution by chitosan- g -poly(acrylic acid)/attapulgite composites. J. Hazard. Mater. 2009, 168, 970–977. [Google Scholar] [CrossRef]
- Zhang, W.M.; Jiang, Y.Q.; Cao, X.Y.; Chen, M.; Ge, D.L.; Sun, Z.X. Synthesis of pore-variable mesoporous CdS and evaluation of its photocatalytic activity in degrading methylene blue. Mater. Res. Bull. 2013, 48, 4379–4384. [Google Scholar] [CrossRef]
- Zhao, Y.; Liu, H.; Wang, F.; Liu, J.; Chul Park, K.; Endo, M. A simple route to synthesize carbon-nanotube/cadmium-sulfide hybrid heterostructures and their optical properties. J. Solid State Chem. 2009, 182, 875–880. [Google Scholar] [CrossRef]
- Badruddoza, A.Z.M.; Tay, A.S.H.; Tan, P.Y.; Hidajat, K.; Uddin, M.S. Carboxymethyl-β-cyclodextrin conjugated magnetic nanoparticles as nano-adsorbents for removal of copper ions: Synthesis and adsorption studies. J. Hazard. Mater. 2011, 185, 1177–1186. [Google Scholar] [CrossRef]
- Lu, D.; Cao, Q.; Li, X.; Cao, X.; Luo, F.; Shao, W. Kinetics and equilibrium of Cu(II) adsorption onto chemically modified orange peel cellulose biosorbents. Hydrometallurgy 2009, 95, 145–152. [Google Scholar] [CrossRef]
- Wang, S.; Zhai, Y.Y.; Gao, Q.; Luo, W.J.; Xia, H.; Zhou, C.G. Highly efficient removal of acid red 18 from aqueous solution by magnetically retrievable chitosan/carbon nanotube: Batch study, isotherms, kinetics, and thermodynamics. J. Chem. Eng. Data 2014, 59, 39–51. [Google Scholar] [CrossRef]
- Gulipalli, C.S.; Prasad, B.; Wasewar, K.L. Batch study, equilibirum and kinetics of adsorption of selenium using rice husk ash (RHA). J. Eng. Sci. Technol. 2011, 6, 590–609. [Google Scholar]
- Luo, S.; Li, X.; Chen, L.; Chen, J.; Wan, Y.; Liu, C. Layer-by-layer strategy for adsorption capacity fattening of endophytic bacterial biomass for highly effective removal of heavy metals. Chem. Eng. J. 2014, 239, 312–321. [Google Scholar] [CrossRef]

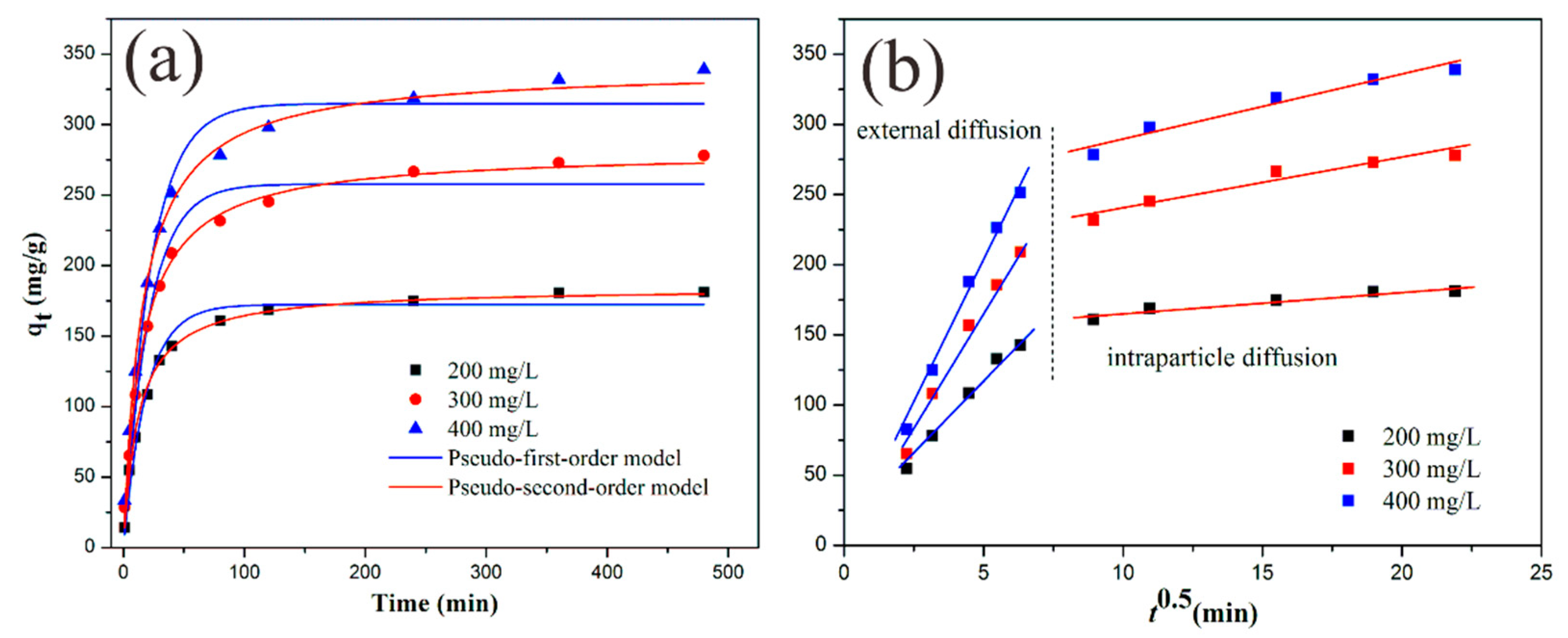
| C0. (mg·L−1) | qe,exp (mg·g−1) | Pseudo-First-Order Model | Pseudo-Second-Order Model | ||||||
|---|---|---|---|---|---|---|---|---|---|
| k1 (min−1) | qe,cal,1 (mg·g−1) | R2 | SSE (%) | k2 (g·mg−1·min−1) | qe,cal,2 (mg·g−1) | R2 | SSE (%) | ||
| 200 | 181.2 | 0.05318 | 172.4 | 0.9794 | 8.8 | 0.000447 | 184.4 | 0.9974 | 3.2 |
| 300 | 279.5 | 0.04791 | 257.9 | 0.9733 | 21.6 | 0.000234 | 281.3 | 0.9964 | 1.8 |
| 400 | 337.0 | 0.04459 | 314.9 | 0.9632 | 22.1 | 0.000188 | 340.1 | 0.9862 | 3.1 |
| C0 (mg·L−1) | First Stage (External Diffusion) | Second Stage (Intraparticle Diffusion) | ||
|---|---|---|---|---|
| ki,1(mg·g−1·min−0.5) | R2 | ki,2 (mg·g−1·min−0.5) | R2 | |
| 200 | 24.23 | 0.9833 | 150.12 | 0.9051 |
| 300 | 34.87 | 0.9862 | 205.12 | 0.9188 |
| 400 | 42.15 | 0.9854 | 244.12 | 0.9469 |
| T (K) | Langmuir Isotherm Constants | Freundlich Isotherm Constants | Redlich−Peterson Isotherm Constants | |||||||
|---|---|---|---|---|---|---|---|---|---|---|
| qm (mg·g−1) | KL (L·mg−1) | R2 | KF (mg1−1/n·L1/n·g−1) | 1/n | R2 | KRP(m·mg−1) | αRP (Lβ·mg-β) | β | R2 | |
| 25 °C | 335.9 | 1.0201 | 0.9706 | 162.28 | 0.1756 | 0.8339 | 421.57 | 1.4757 | 0.9584 | 0.9768 |
| 35 °C | 339.6 | 1.0278 | 0.9699 | 163.80 | 0.1776 | 0.8309 | 427.22 | 1.4781 | 0.9581 | 0.9760 |
| 45 °C | 345.2 | 1.1386 | 0.9723 | 168.22 | 0.1761 | 0.8231 | 447.53 | 1.4540 | 0.9698 | 0.9736 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Xue, H.; Xu, M.; Yu, Y.; Wan, J.; Liu, C.; Meng, F. Ultraviolet Radiation-Assisted Preparation of a Novel Biomass Fiber to Remove Cadmium from Wastewater. Water 2023, 15, 811. https://doi.org/10.3390/w15040811
Xue H, Xu M, Yu Y, Wan J, Liu C, Meng F. Ultraviolet Radiation-Assisted Preparation of a Novel Biomass Fiber to Remove Cadmium from Wastewater. Water. 2023; 15(4):811. https://doi.org/10.3390/w15040811
Chicago/Turabian StyleXue, Hao, Min Xu, Yueyang Yu, Jiancheng Wan, Chen Liu, and Fansheng Meng. 2023. "Ultraviolet Radiation-Assisted Preparation of a Novel Biomass Fiber to Remove Cadmium from Wastewater" Water 15, no. 4: 811. https://doi.org/10.3390/w15040811





