A Review of Bubble Aeration in Biofilter to Reduce Total Ammonia Nitrogen of Recirculating Aquaculture System
Abstract
1. Introduction
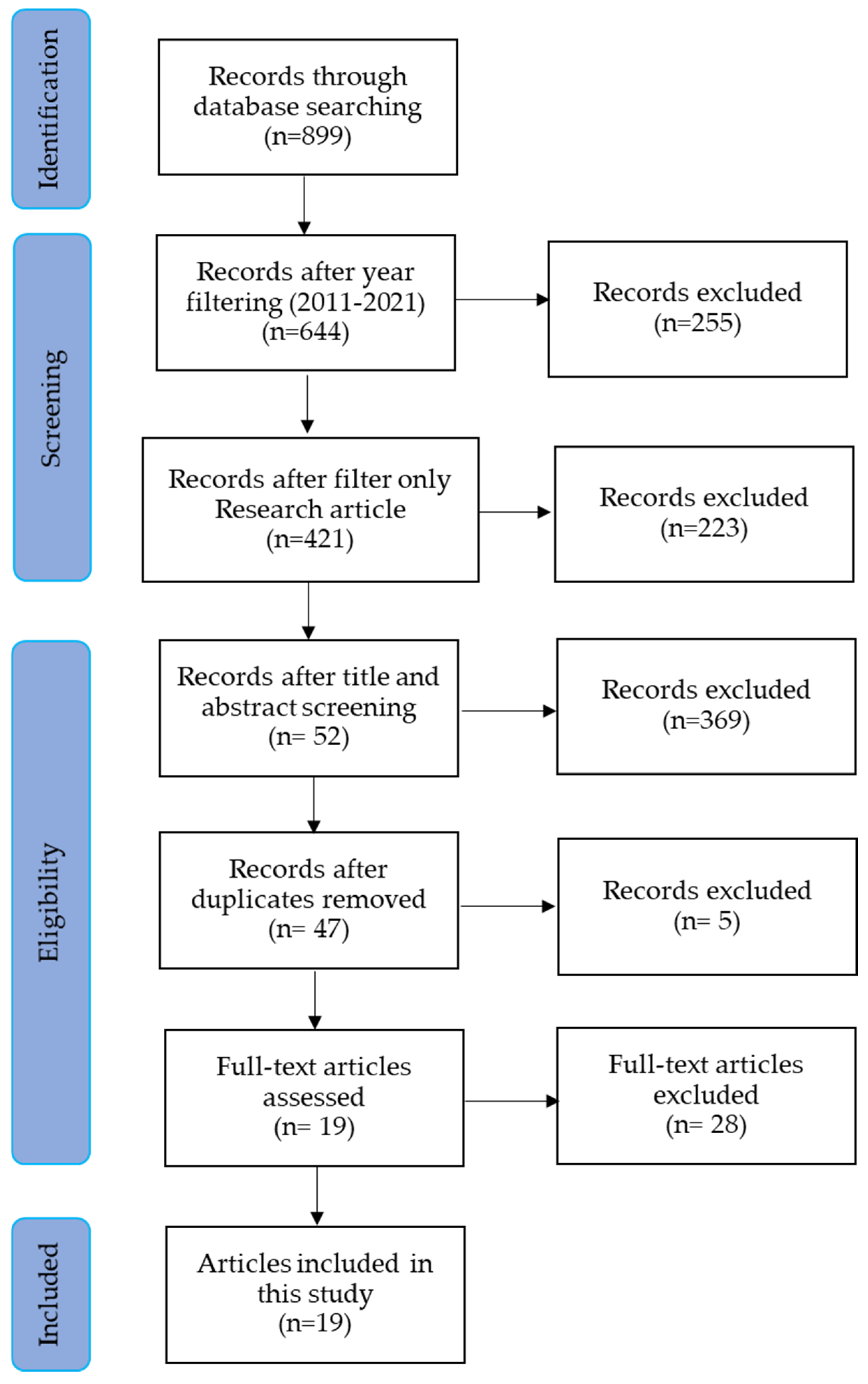
2. Materials and Methods
2.1. Objective
2.2. Screening
2.3. Eligibility
2.4. Inclusion
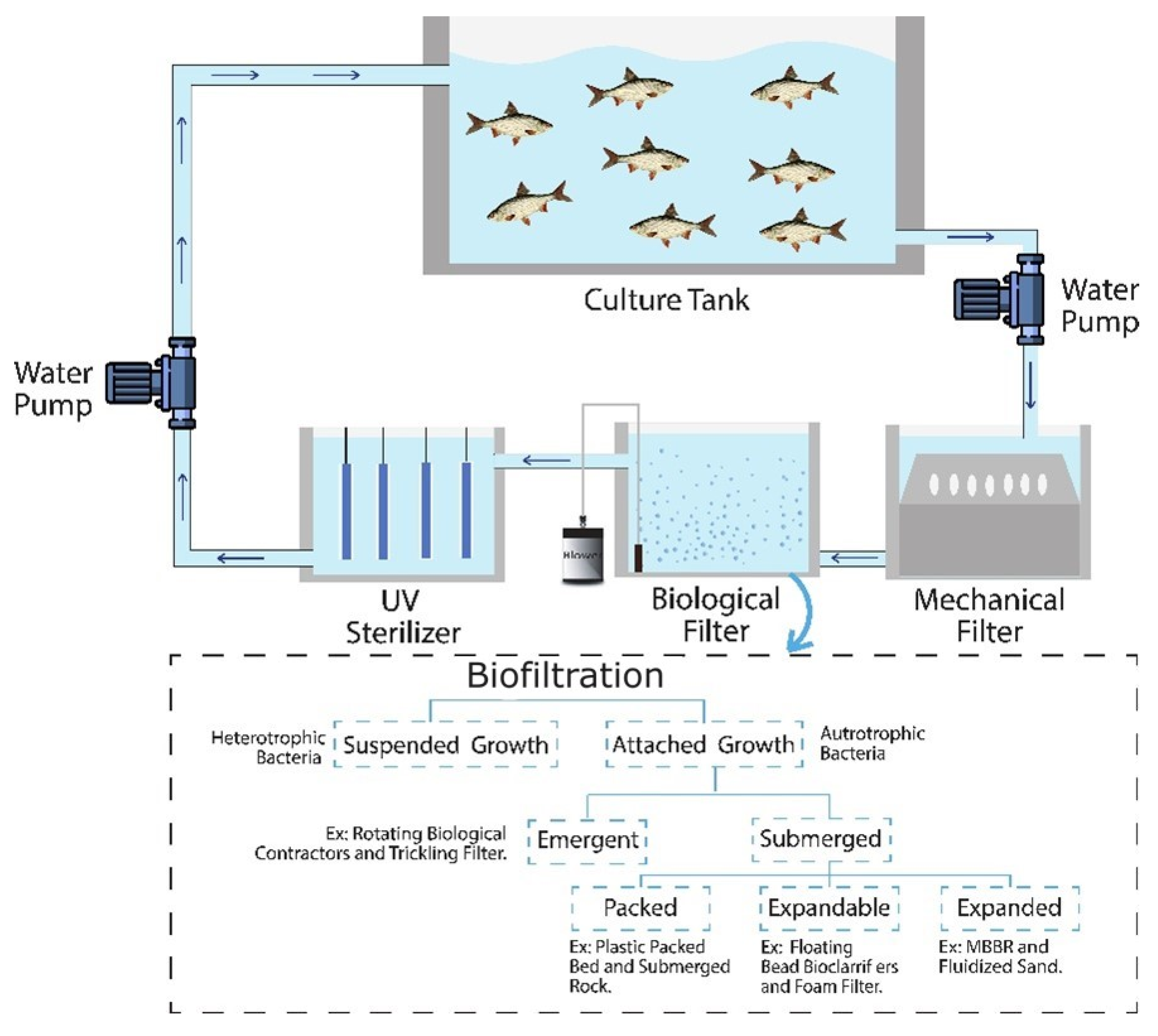
3. Types of Biofilter in RASs and Role of Bubble Aeration in Biofilter
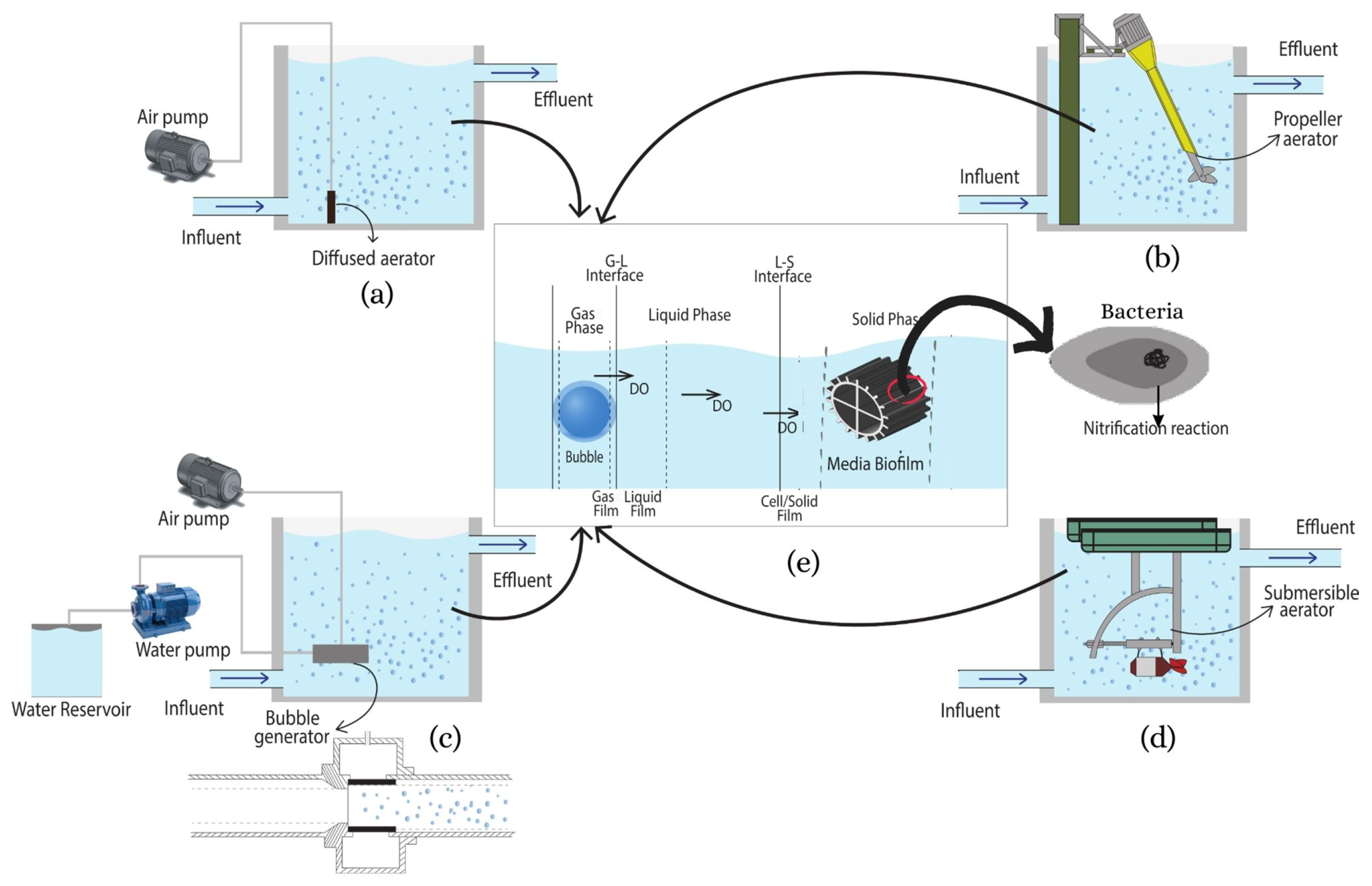
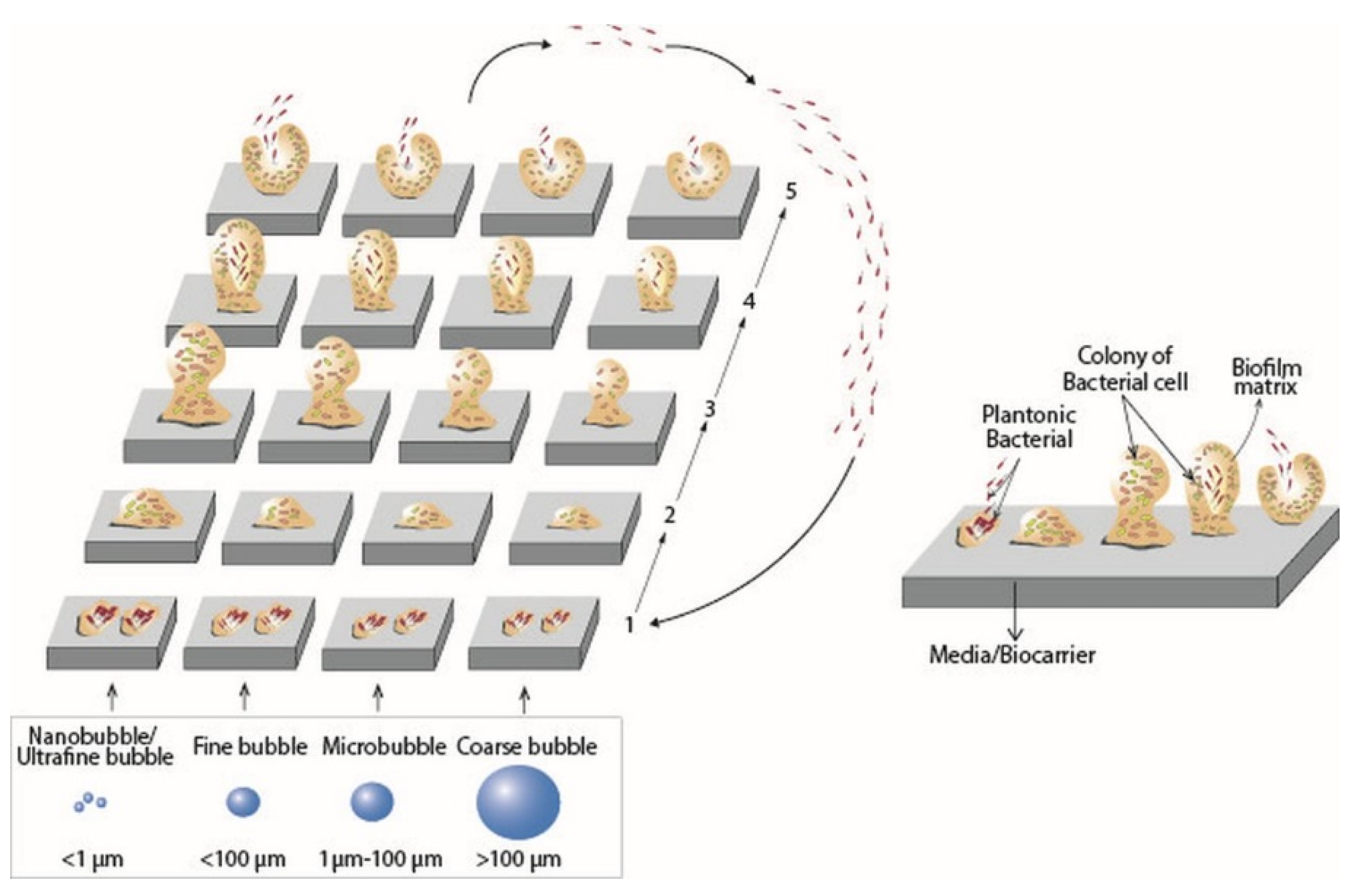
3.1. Bubble Size
3.2. Biofilm Growth
3.3. Nitrification in Biofilm
3.4. Oxygen Transfer
4. Results and Discussion
4.1. Biofilm Performance with Bubble Aeration
4.1.1. Effect of Medium and Coarse Bubble Aeration on the Biofilter Performance
| Type of Aerator | Biofilter Type (Volume in L) | RAS Culture Species | Media Type (Surface Area in m2/m3) | DO (mg/L) | Performance | Reference | ||
|---|---|---|---|---|---|---|---|---|
| Ammonia Removal (%) | Ammonia Concentration (mg/L) | |||||||
| Medium bubble diffuser | MBBR (2000) | N/A | Bioball (220) | 6.1 ± 0.9 | N/A | 64 ± 13 | N/A | [17] |
| Coarse Diffuser | MBBR (6360) | Red drum fingerlings (Sciaenops ocellatus) | MB3 (604) | 9.9 ± 1.9 | N/A | 15.6 ± 0.4 | 1.055 | [50] |
| Coarse bubble diffuser | MBBR (5.5) | Not specified | Novel sponge biocarrier (N/A) | 5.5–7.5 ± 0.5 | N/A | 91.65 ± 2.4 | 0.83 ± 0.31 | [51] |
| Coarse bubble diffuser | MBBR (7000) | Atlantic salmon parr (Salmo salar) | N/A | 7.3 | 1.89 | 50 ± 3 | 0.22 ± 0.03 | [52] |
| Coarse Bubble diffuser | MBBR (410) | Florida pompano (Trachinotus carolinus) | K1 Kaldness (N/A) | 8.2 ± 1.0 | 1.39 | 62.4 | 0.46 ± 0.27 | [53] |
| Coarse bubble diffuser | MBBR (N/A) | European sea bass (Dicentrarchus labrax) | N/A | 10 ± 0.9 | N/A | N/A | 0.11 ± 0.08 | [54] |
| Coarse bubble diffuser | Membrane Filter (N/A) | N/A | Commercial mixed-cellulose (MC) membranes (N/A) | 6.75 | 1.54 | 38 | 7.81 | [12] |
| Microbubble diffuser | airlift fluidized bed reactor (115) | Nile Tilapia (Oreochromis niloticus) | Granular activated carbon (N/A) | 4.16 | N/A | 27 | 0.104 | [2] |
| Fine bubble diffuser | BAF (1.6) | N/A | Puzzolane material (1740) | >2.1 | 0.31 | 50-80 | N/A | [57] |
| Fine bubble diffuser | Fixed bed reactor (3.5) | N/A | Polycaprolactone 114 (PCL) (N/A) | 5.63 ± 0.57 | 1.73 | 57.14 | 1.93 ± 0.37 | [58] |
| Nanobubble generator and combination with coarse bubble diffuser | BAF (4.75) | N/A | Volcanic rock filter (N/A) | 6.52 | 2.06 | 98.02 | N/A | [59] |
| Nanobubbles generator | Membrane Filter (N/A) | N/A | Commercial mixed-cellulose (MC) membranes (N/A) | 8.55 | 1.92 | 75 | 3.15 | [12] |
4.1.2. Effect of Micro, Fine, and Nanobubble Aeration on the Biofilter Performance
4.2. Biofilm Growth
4.2.1. Microbial Community
4.2.2. Biofilm Thickness
5. Author Perspective
6. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Deviller, G.; Aliaume, C.; Nava, M.A.F. High-rate algal pond treatment for water reuse in an integrated marine fish recirculating system: Effect on water quality and sea bass growth. Aquaculture 2004, 235, 331–344. [Google Scholar] [CrossRef]
- Sánchez, O.I.A.; Matsumoto, T. Hydrodynamic characterization and performance evaluation of an aerobic three phase airlift fluidized bed reactor in a recirculation aquaculture system for Nile Tilapia production. Aquac. Eng. 2012, 47, 16–26. [Google Scholar] [CrossRef]
- Cabasso, I. Membrane. In Encyclopedia of Polymer Science and Engineering; Wiley: New York, NY, USA, 1987; Volume 9, pp. 509–579. [Google Scholar]
- Samer, M. Biological and Chemical Wastewater Treatment. In Wastewater Treatment Engineering; Intech: London, UK, 2015. [Google Scholar]
- Holan, A.B.; Wold, P.-A.; Øie, G.; Leiknes, T.O. Integrated Membrane Bioreactor for Water Quality Control in Marine Recirculating Aquaculture Systems. Sep. Sci. Technol. 2013, 48, 1758–1767. [Google Scholar] [CrossRef]
- Tasong, A.C.; Abao, R.P. Design and Development of an IoT Application with Visual Analytics for Water Consumption Monitoring. Procedia Comput. Sci. 2019, 157, 205–213. [Google Scholar] [CrossRef]
- Malone, R.F.; Pfeiffer, T.J. Rating fixed film nitrifying biofilters used in recirculating aquaculture systems. Aquac. Eng. 2006, 34, 389–402. [Google Scholar] [CrossRef]
- Ghangrekar, M.; Behera, M. Suspended Growth Treatment Processes. In Comprehensive Water Quality and Purification; Elsevier Inc.: Amsterdam, The Netherlands, 2014; pp. 75–89. [Google Scholar]
- Gavrilescu, M.; Macoveanu, M. Attached-growth process engineering in wastewater treatment. Bioprocess Biosyst. Eng. 2000, 23, 95–106. [Google Scholar] [CrossRef]
- Wang, X.; Xia, S.; Chen, L.; Zhao, J.; Renault, N.; Chovelon, J. Nutrients removal from municipal wastewater by chemical precipitation in a moving bed biofilm reactor. Process. Biochem. 2006, 41, 824–828. [Google Scholar] [CrossRef]
- Roveto, P.M.; Schuler, A.J. Performance and diversity responses of nitrifying biofilms developed on varied materials and topographies to stepwise increases of aeration. Bioresour. Technol. 2019, 281, 429–439. [Google Scholar] [CrossRef]
- Xiao, W.; Xu, G. Mass transfer of nanobubble aeration and its effect on biofilm growth: Microbial activity and structural properties. Sci. Total Environ. 2019, 703, 134976. [Google Scholar] [CrossRef]
- Havlíček, K.; Nechanická, M.; Lederer, T.; Sirková, B.K. Analysis of nitrifying bacteria growth on two new types of biomass carrier using respirometry and molecular genetic methods. Ecotoxicol. Environ. Saf. 2021, 225, 112795. [Google Scholar] [CrossRef]
- Tang, B.; Song, H.; Bin, L.; Huang, S.; Zhang, W.; Fu, F.; Zhao, Y.; Chen, Q. Determination of the profile of DO and its mass transferring coefficient in a biofilm reactor packed with semi-suspended bio-carriers. Bioresour. Technol. 2017, 241, 54–62. [Google Scholar] [CrossRef] [PubMed]
- Liu, C.; Tang, Y. Application research of micro and nano bubbles in water pollution control. E3S Web Conf. 2019, 136, 06028. [Google Scholar] [CrossRef]
- Ruen-ngam, D.; Wongsuchoto, P.; Limpanuphap, A.; Charinpanitku, T.; Pavasant, P. Influence of salinity on bubble size distribution and gas–liquidmass transfer in airlift contactors. Chem. Eng. J. 2008, 141, 222–232. [Google Scholar] [CrossRef]
- Dias, J.; Bellingham, M.; Hassan, J.; Barrett, M.; Stephenson, T.; Soares, A. Influence of carrier media physical properties on start-up of moving attached growth systems. Bioresour. Technol. 2018, 266, 463–471. [Google Scholar] [CrossRef]
- Kawan, J.A.; Suja, F.; Pramanik, S.K.; Yusof, A.; Rahman, R.A.; Hasan, H.A. Effect of Hydraulic Retention Time on the Performance of a Compact Moving Bed Biofilm Reactor for Effluent Polishing of Treated Sewage. Water 2022, 14, 81. [Google Scholar] [CrossRef]
- Khuntia, S.; Majumder, S.K.; Ghosh, P. Microbubble-aided water and wastewater purification: A review. Rev. Chem. Eng. 2012, 28, 191–221. [Google Scholar] [CrossRef]
- Sakr, M.; Mohamed, M.M.; Maraqa, M.A.; Hamouda, M.A.; Hassan, A.A.; Ali, J.; Jung, J. A critical review of the recent developments in micro–nano bubbles applications for domestic and industrial wastewater treatment. Alex. Eng. J. 2021, 61, 6591–6612. [Google Scholar] [CrossRef]
- Shitu, A.; Liu, G.; Muhammad, A.I.; Zhang, Y.; Tadda, M.A.; Qi, W.; Liu, D.; Ye, Z.; Songming, Z. Recent advances in application of moving bed bioreactors for wastewater treatment from recirculating aquaculture systems: A review. Aquac. Fish. 2022, 7, 244–258. [Google Scholar] [CrossRef]
- Ferreira, C.; Gonçalves, G. A Systematic Review on Life Extension Strategies in Industry. In Proceedings of the IEEE International Conference on Emerging Technologies and Factory Automation, ETFA, Vasteras, Sweden, 7–10 September 2021; Volume 10, pp. 1–17. [Google Scholar]
- Hamza, R.A.; Iorhemen, O.T.; Tay, J.H. Advances in biological systems for the treatment of high-strength wastewater. J. Water Process. Eng. 2016, 10, 128–142. [Google Scholar] [CrossRef]
- Reyes, A.A.D.; Lawson, T.B. Combination of a bead filter and rotating biological contactor in a recirculating fish culture system. Aquac. Eng. 1996, 15, 27–39. [Google Scholar] [CrossRef]
- Kordkandi, S.A.; Khoshfetrat, A.B.; Faramarzi, A. Performance modelling of a partially-aerated submerged fixed-film bioreactor: Mechanistic analysis versus semi data-driven method. J. Ind. Eng. Chem. 2018, 61, 398–406. [Google Scholar] [CrossRef]
- Jakobsen, H.A. Packed Bed Reactors. In Chemical Reactor Modeling: Multiphase Reactive Flows; Springer: Berlin/Heidelberg, Germany, 2008; pp. 953–984. [Google Scholar]
- Liu, B.Y.M.; Pfeffer, J.T. Modeling for Anaerobic Fixed-Bed Biofilm Reactors; University of Illinois Urbana-Champaign: Champaign, IL, USA, 1989. [Google Scholar]
- Malone, R.F. Floating Media Hourglass Biofilter. U.S. Patent 5,232,586, 3 August 1993. [Google Scholar]
- Malone, R.F.; E Beecher, L. Use of floating bead filters to recondition recirculating waters in warmwater aquaculture production systems. Aquac. Eng. 2000, 22, 57–73. [Google Scholar] [CrossRef]
- Wimberly, D.M. Development and Evaluation of a Low-Density Media Biofiltration Unit for Use in Recirculating Fish Culture Systems. Master’s Thesis, Louisiana State University, Baton Rouge, LA, USA, 1990. [Google Scholar]
- Ochoa, F.G.; Gomez, E. Bioreactor scale-up and oxygen transfer rate in microbial processes: An overview. Biotechnol. Adv. 2009, 27, 153–176. [Google Scholar] [CrossRef] [PubMed]
- Xu, X.; Wei, W.; Liu, F.; Wei, W.; Liu, Z. Experimental study on aeration efficiency in a pilot-scale decelerated oxidation ditch equipped with fine bubble diffusers and impellers. Can. J. Chem. Eng. 2020, 991, 1410–1420. [Google Scholar] [CrossRef]
- Kumar, A.; Moulick, S.; Mal, B. Design characteristics of pooled circular stepped cascade aeration system. Aquac. Eng. 2010, 56, 51–58. [Google Scholar] [CrossRef]
- Pambudiarto, B.A.; Mindaryani, A.; Deendarlianto, D.; Budhijanto, W. Evaluation of the Effect of Operating Parameters on the Performance of Orifice/Porous Pipe Type Micro-bubble Generator. J. Eng. Technol. Sci. 2020, 52, 196–207. [Google Scholar] [CrossRef]
- Takahashi, M.; Chiba, K.; Li, P. Free-Radical Generation from Collapsing Microbubbles in the Absence of a Dynamic Stimulus. J. Phys. Chem. B 2007, 111, 1343–1347. [Google Scholar] [CrossRef]
- Ebina, K.; Shi, K.; Hirao, M. Oxygen and Air Nanobubble Water Solution Promote the Growth of Plants, Fishes, and Micer. PLoS ONE 2013, 8, e65339. [Google Scholar] [CrossRef] [PubMed]
- ISO. Fine Bubble Technology—General Principles for Usage and Measurement of Fine Bubbles—Part 1: Terminology; ISO: Geneva, Switzerland, 2017. [Google Scholar]
- Zăbavă, B.Ș.; Ungureanu, N.; Voicu, G.; Dincă, M. Types of Aerators Used in Wastewater Treatment Plants. In Proceedings of the 5th International Conference of Thermal Equipment, Renewable Energy and Rural Development, Golden Sands, Bulgaria, 2–4 June 2016. [Google Scholar]
- Farmer, R.; Arndt, R. Development of an Efficient Aeration System for Aquaculture; University of Minnesota: Minneapolis, MN, USA, 1995. [Google Scholar]
- Flemming, H.C.; Neu, T.R. The EPS matrix: The “house of biofilm cells”. J. Bacteriol. 2007, 189, 7945–7947. [Google Scholar] [CrossRef]
- Karaguler, T.; Kahraman, H.; Tuter, M. Analyzing effects of ELF electromagnetic fields on removing bacterial biofilm. Biocybern. Biomed. Eng. 2017, 37, 336–340. [Google Scholar] [CrossRef]
- Stoodley, P.; Sauer, K.; Davies, D.G.; Costerton, J.W. Biofilms as Complex Differentiated Communities. Annu. Rev. Microbiol. 2002, 56, 187–209. [Google Scholar] [CrossRef] [PubMed]
- Goswami, S.; Sarkar, S.; Mazumder, D. A new approach for development of kinetics of wastewater treatment in aerobic biofilm reactor. Appl. Water Sci. 2016, 7, 2187–2193. [Google Scholar] [CrossRef]
- Bouaifi, M.; Hebrard, G.; Bastoul, D.; Roustan, M. A comparative study of gas hold-up, bubble size, interfacial area and mass transfer coefficients in stirred gas-liquid reactors and bubble columns. Chem. Eng. Process. Process Intensif. 2001, 40, 97–111. [Google Scholar] [CrossRef]
- Larsson, V. Energy Savings with a New Aeration and Control System in a Mid-Size Swedish Wastewater Treatment Plant; Uppsala University: Uppsala, Sweden, 2011. [Google Scholar]
- Akita, K.; Yoshida, F. Bubble Size, Interfacial Area, and Liquid-Phase Mass Transfer Coefficient in Bubble Columns. Ind. Eng. Chem. Process. Des. Dev. 1974, 13, 84–91. [Google Scholar] [CrossRef]
- Yao, G.-J.; Ren, J.-Q.; Zhou, F.; Liu, Y.-D.; Li, W. Micro-nano aeration is a promising alternative for achieving high-rate partial nitrification. Sci. Total Environ. 2021, 795, 148899. [Google Scholar] [CrossRef] [PubMed]
- Woodard & Curran, Inc. Chapter 7–Methods for Treating Wastewaters from Industry. In Industrial Waste Treatment Handbook, 2nd ed.; Butterworth-Heinemann: Oxford, UK, 2006; pp. 149–334. [Google Scholar]
- Liu, K.; Phillips, J.R.; Sun, X.; Mohammad, S.; Huhnke, R.L.; Atiyeh, H.K. Investigation and Modeling of Gas-Liquid Mass Transfer in a Sparged and Non-Sparged Continuous Stirred Tank Reactor with Potential Application in Syngas Fermentation. Fermentation 2019, 5, 75. [Google Scholar] [CrossRef]
- Pfeiffer, T.J.; Wills, P.S. Evaluation of three types of structured floating plastic media in moving bed biofilters for total ammonia nitrogen removal in a low salinity hatchery recirculating aquaculture system. Aquac. Eng. 2011, 45, 51–59. [Google Scholar] [CrossRef]
- Shitu, A.; Zhu, S.; Qi, W.; Tadda, M.A.; Liu, D.; Ye, Z. Performance of novel sponge biocarrier in MBBR treating recirculating aquaculture systems wastewater: Microbial community and kinetic study. J. Environ. Manag. 2020, 275, 111264. [Google Scholar] [CrossRef] [PubMed]
- Summerfelt, S.T.; Zühlke, A.; Kolarevic, J.; Reiten, B.K.M.; Selset, R.; Gutierrez, X.; Terjesen, B.F. Effects of alkalinity on ammonia removal, carbon dioxide stripping, and system pH in semi-commercial scale water recirculating aquaculture systems operated with moving bed bioreactors. Aquac. Eng. 2015, 65, 46–54. [Google Scholar] [CrossRef]
- Pfeiffer, T.J.; Riche, M.A. Evaluation of a Low-head Recirculating Aquaculture System Used for Rearing Florida Pompano to Market Size. J. World Aquac. Soc. 2011, 42, 198–208. [Google Scholar] [CrossRef]
- Torno, J.; Einwächter, V.; Schroeder, J.P.; Schulz, C. Nitrate has a low impact on performance parameters and health status of ongrowing. Aquaculture 2018, 489, 21–27. [Google Scholar] [CrossRef]
- Blancheton, J.P. Developments in recirculation systems for Mediterranean fish species. Aquac. Eng. 2000, 22, 17–31. [Google Scholar] [CrossRef]
- Dosdat, A.; Ruyet, J.P.-L.; Covès, D.; Dutto, G.; Gasset, E.; Le Roux, A.; Lemarié, G. Effect of chronic exposure to ammonia on growth, food utilisation and metabolism of the European sea bass (Dicentrarchus labrax). Aquat. Living Resour. 2003, 16, 509–520. [Google Scholar] [CrossRef]
- Albuquerque, A.; Makinia, J.; Pagilla, K. Impact of aeration conditions on the removal of low concentrations of nitrogen in a tertiary partially aerated biological filter. Ecol. Eng. 2012, 44, 44–52. [Google Scholar] [CrossRef]
- Luo, G.; Xu, G.; Gao, J.; Tan, H. Effect of dissolved oxygen on nitrate removal using polycaprolactone as an organic carbon source and biofilm carrier in fixed-film denitrifying reactors. J. Environ. Sci. 2016, 43, 147–152. [Google Scholar] [CrossRef] [PubMed]
- Xiao, W.; Xu, G.; Li, G. Role of shear stress in biological aerated filter with nanobubble aeration: Performance, biofilm structure and microbial community. Bioresour. Technol. 2021, 325. [Google Scholar] [CrossRef] [PubMed]
- Shannon, C. A mathematical theory of communication. Bell Syst. Tech. J. 1948, 27, 379–423. [Google Scholar] [CrossRef]
- Konopiński, M.K. Shannon diversity index: A call to replace the original Shannon’s formula with unbiased estimator in the population genetics studies. PeerJ 2000, 8, e9391. [Google Scholar] [CrossRef]
- Spohn, S.N.; Young, V.B. Gastrointestinal Microbial Ecology with Perspectives on Health and Disease. In Physiology of the Gastrointestinal Tract; Academic Press: Irvine, CA, USA, 2018; pp. 727–753. [Google Scholar]
- Gonzalez-Silva, B.M.; Jonassen, K.R.; Bakke, I.; Østgaard, K.; Vadstein, O. Nitrification at different salinities: Biofilm community composition and physiological plasticity. Water Res. 2016, 95, 48–58. [Google Scholar] [CrossRef]
- Bakke, I.; Åm, A.L.; Kolarevic, J.; Ytrestøyl, T.; Vadstein, O.; Attramadal, K.J.K.; Terjesen, B.F. Microbial community dynamics in semi-commercial RAS for production of Atlantic salmon post-smolts at different salinities. Aquac. Eng. 2016, 78, 42–49. [Google Scholar] [CrossRef]
- Lee, J.; Kim, I.-S.; Emmanuel, A.; Koh, S.-C. Microbial valorization of solid wastes from a recirculating aquaculture system and the relevant microbial functions. Aquac. Eng. 2019, 87, 102016. [Google Scholar] [CrossRef]
- Lee, D.-E.; Lee, J.; Kim, Y.-M.; Myeong, J.-I.; Kim, K.-H. Uncultured bacterial diversity in a seawater recirculating aquaculture system revealed by 16S rRNA gene amplicon sequencing. J. Microbiol. 2016, 54, 296–304. [Google Scholar] [CrossRef]
- Ni, B.-J.; Yuan, Z. A model-based assessment of nitric oxide and nitrous oxide production in membrane-aerated autotrophic nitrogen removal biofilm systems. J. Membr. Sci. 2013, 428, 163–171. [Google Scholar] [CrossRef]
- Michelin, M.; Mota, A.M.O.; Polizeli, M.L.T.M.; Silva, D.P.; Vicente, A.A.; Teixeira, J.A. Influence of volumetric oxygen transfer coefficient (kLa) on xylanases batch production by Aspergillus niger van Tieghem in stirred tank and internal-loop airlift bioreactors. Biochem. Eng. J. 2013, 80, 19–26. [Google Scholar] [CrossRef]
- Fang, H.; Chen, Y.; Huang, L.; He, G. Analysis of biofilm bacterial communities under different shear stresses using size-fractionated sediment. Sci. Rep. 2017, 7, 1299. [Google Scholar] [CrossRef] [PubMed]
- Agarwal, R.; Singh, S.; Bhilegaonkar, K.; Singh, V. Optimization of microtiter plate assay for the testing of biofilm formation ability in different Salmonella serotypes. Int. Food Res. J. 2011, 18, 1493–1498. [Google Scholar]
- Gomes, I.B.; Meireles, A.; Gonçalves, A.L.; Goeres, D.M.; Sjollema, J.; Simões, L.C.; Simões, M. Standardized reactors for the study of medical biofilms: A review of the principles and latest modifications. Crit. Rev. Biotechnol. 2017, 38, 657–670. [Google Scholar] [CrossRef]
- Gomes, L.C.; Moreira, J.M.; Teodósio, J.S.; Araújo, J.D.P.; Miranda, J.M. 96-well microtiter plates for biofouling simulation in biomedical settings. Biofouling 2014, 30, 535–546. [Google Scholar] [CrossRef]
- Gapes, D.; Keller, J. Impact of oxygen mass transfer on nitrification reactions in suspended carrier reactor biofilms. Process. Biochem. 2009, 44, 43–53. [Google Scholar] [CrossRef]
- Piculell, M.; Welander, P.; Jönsson, K.; Welander, T. Evaluating the effect of biofilm thickness on nitrification in moving bed biofilm reactors. Environ. Technol. 2015, 37, 732–743. [Google Scholar] [CrossRef]
- Torresi, E.; Fowler, S.; Polesel, F.; Bester, K.; Andersen, H.; Smets, B.; Plósz, B.; Christensson, M. Biofilm thickness influences biodiversity in nitrifying MBBRs 1—Implications on micropollutant removal. Environ. Sci. Technol. 2016, 50, 9279–9288. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Capdeville, B. Dynamics of nitrifying biofilm growth in biological nitrogen removal al process. Water Sci. Technol. 1994, 29, 377–380. [Google Scholar] [CrossRef]
- Kumar, C.; Anand, S. Significance of microbial biofilms in food industry: A review. Int. J. Food Microbiol. 1998, 42, 9–27. [Google Scholar] [CrossRef] [PubMed]
| Search Local | Search Expression | Search Result | Types of Documents |
|---|---|---|---|
| ScienceDirect | (bubble! OR nanobubble!) AND (aeration) AND (ammonia) AND (biofilter! OR MBBR!) AND (biofilm!) | 445 |
|
| Google Scholar | (bubble! OR nanobubble!) AND (aeration) AND (ammonia) AND (biofilter! OR MBBR!) AND (biofilm!) AND (RAS!) | 454 |
|
| Aeration Type | Biofilter Type (Volume in L) | Biofilter Volume (L) | Culture Species | Shanon | Performance | Reference | |
|---|---|---|---|---|---|---|---|
| Ammonia Removal (%) | TN Removal (%) | ||||||
| Coarse bubble diffuser | MBBR | 0.7 | N/A | 1.30 | 97.1 | N/A | [63] |
| Coarse bubble diffuser | MBBR | N/A | Atlantic salmon post-smolts | 2.87 | 50 | N/A | [64] |
| Coarse bubble diffuser | BAF | 10,850 | Seawater RAS | 5.04 | 41.7 | 41.3 | [65] |
| Coarse bubble diffuser | BAF | 10,850 | Seawater RAS | 8.36 | 85 | N/A | [66] |
| Coarse bubble diffuser | MBBR | 5 | N/A | 2.30 | 96 | N/A | [13] |
| Coarse bubble diffuser | MBBR | 0.4 | N/A | 2.85 | 36.04 | N/A | [11] |
| Fine bubble diffuser | MBBR | 0.4 | N/A | 2.88 | 51 | N/A | [11] |
| Aeration Type | Biofilter Type (Volume in L) | Biofilter Volume (L) | Culture Species | Biofilm Thickness (µm) | Performance | Reference | |
|---|---|---|---|---|---|---|---|
| Ammonia Removal (%) | TN Removal (%) | ||||||
| Coarse bubble diffuser | MBBR | 27.7 | N/A | 1000 | N/A | ±14.3 | [14] |
| Coarse bubble diffuser | MABR | 1050 | N/A | 1000 | N/A | >70 | [67] |
| Coarse bubble diffuser | Membrane Filter | N/A | N/A | 1016 ± 236 | 38 | N/A | [12] |
| Medium bubble diffuser | MBBR | 2000 | Litopenaeus vannamei | 71 ± 22 | 64 ± 13 | N/A | [17] |
| Fine bubble diffuser | BAF | 1.6 | N/A | 470 ± 50 | 50–80 | N/A | [57] |
| Nanobuble generator | Membrane Filter | N/A | N/A | 1188 ± 322 | 75 | N/A | [12] |
| Nanobubble generator and combination with coarse bubble diffuser | BAF | 19 | N/A | 136.1 | 89.22 | 34.51 | [59] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Suriasni, P.A.; Faizal, F.; Panatarani, C.; Hermawan, W.; Joni, I.M. A Review of Bubble Aeration in Biofilter to Reduce Total Ammonia Nitrogen of Recirculating Aquaculture System. Water 2023, 15, 808. https://doi.org/10.3390/w15040808
Suriasni PA, Faizal F, Panatarani C, Hermawan W, Joni IM. A Review of Bubble Aeration in Biofilter to Reduce Total Ammonia Nitrogen of Recirculating Aquaculture System. Water. 2023; 15(4):808. https://doi.org/10.3390/w15040808
Chicago/Turabian StyleSuriasni, Putu Ayustin, Ferry Faizal, Camellia Panatarani, Wawan Hermawan, and I Made Joni. 2023. "A Review of Bubble Aeration in Biofilter to Reduce Total Ammonia Nitrogen of Recirculating Aquaculture System" Water 15, no. 4: 808. https://doi.org/10.3390/w15040808
APA StyleSuriasni, P. A., Faizal, F., Panatarani, C., Hermawan, W., & Joni, I. M. (2023). A Review of Bubble Aeration in Biofilter to Reduce Total Ammonia Nitrogen of Recirculating Aquaculture System. Water, 15(4), 808. https://doi.org/10.3390/w15040808









