Occurrence Characterization and Contamination Risk Evaluation of Microplastics in Hefei’s Urban Wastewater Treatment Plant
Abstract
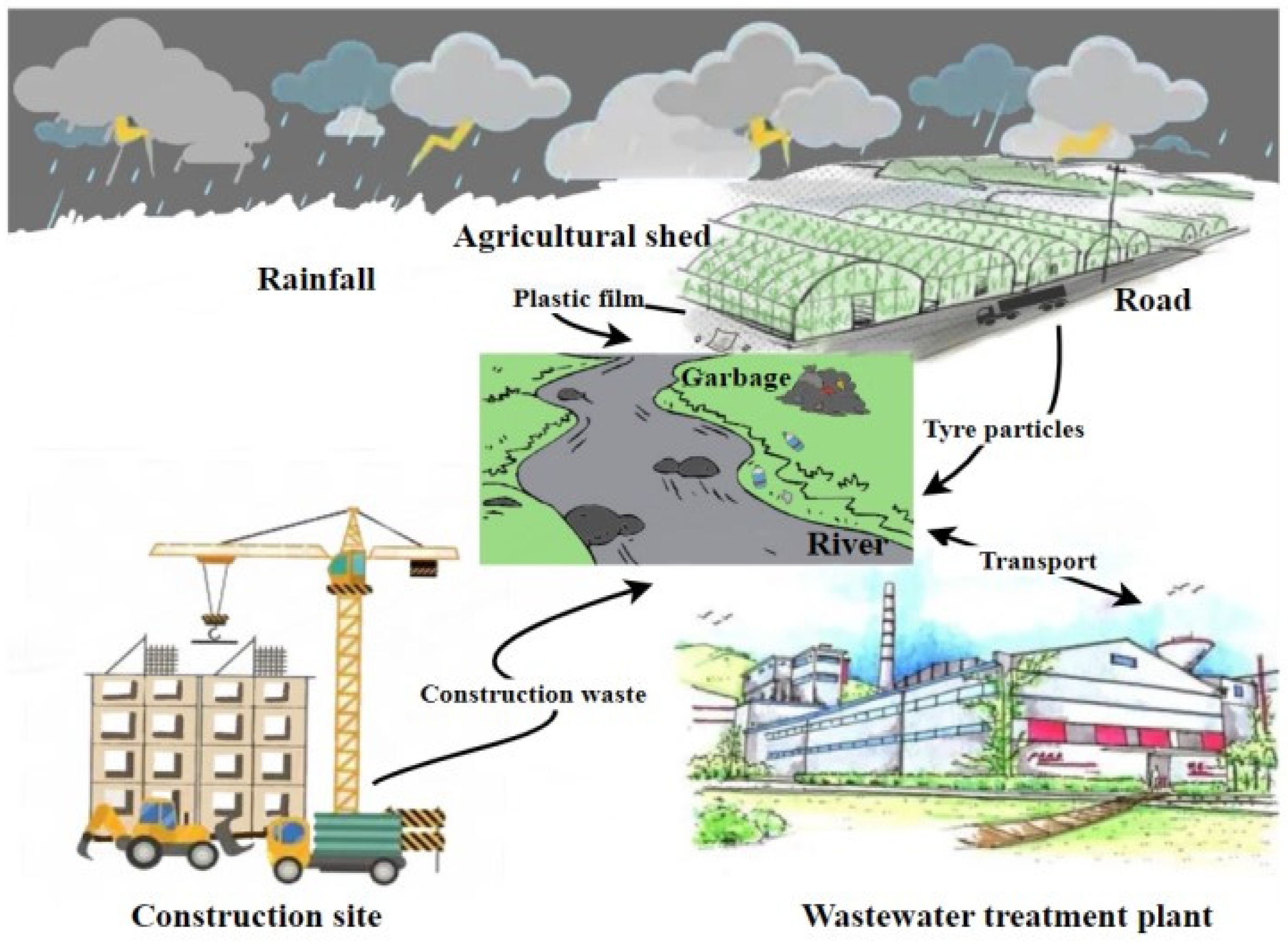
:1. Introduction
2. Materials and Experimental Methods
2.1. Sample Sites
2.2. Sample Methods
2.3. Experimental Scheme
2.3.1. MPs Separation and Extraction
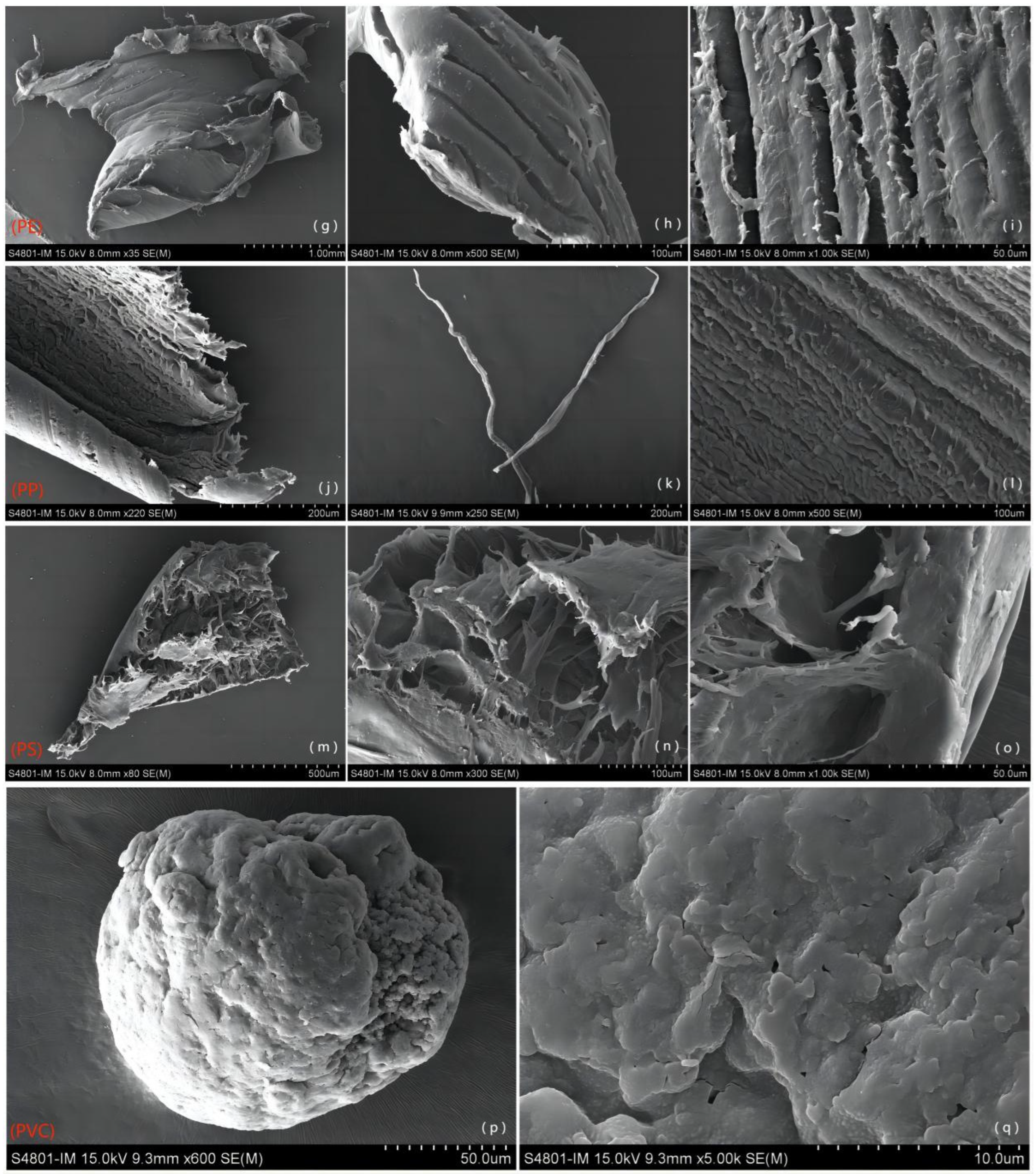
2.3.2. Observation and Identification of Microplastics
2.3.3. Quantitative Methods for Microplastics
2.3.4. Experimental Quality Control
2.3.5. Data Analysis
2.4. Potential Ecological Risk Assessment of MPs
3. Results and Discussion
3.1. Distribution and Reduction of MPS in WWTP
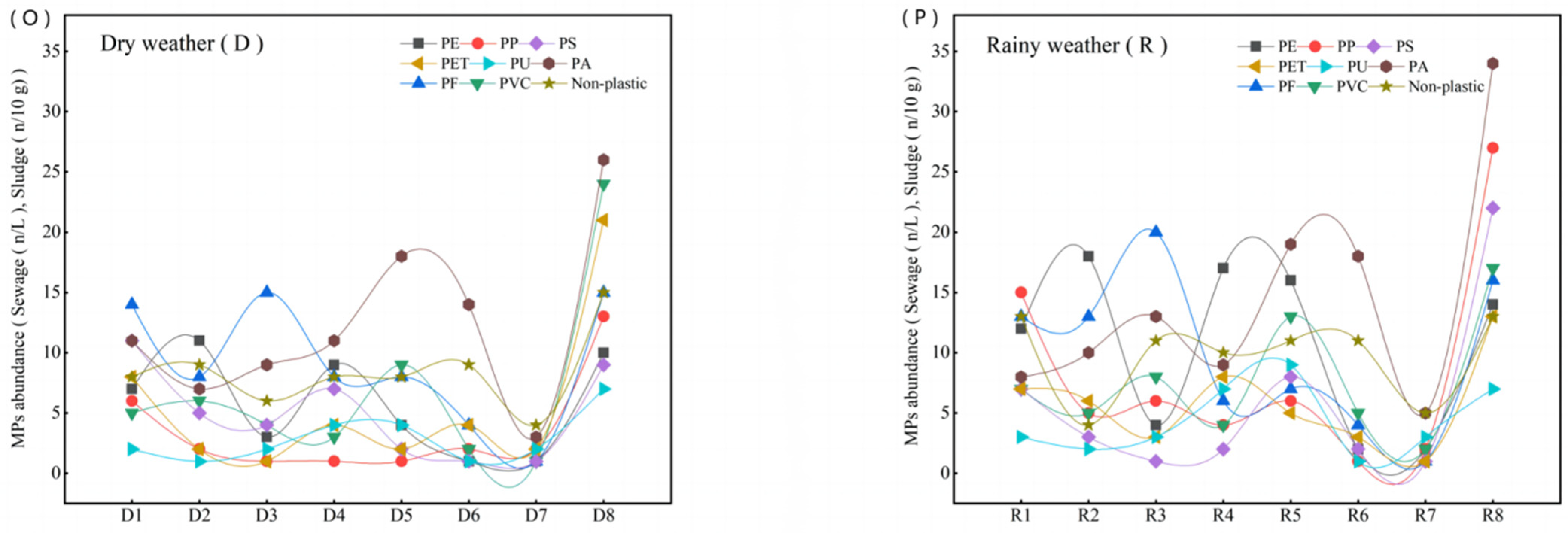
3.2. Source and Variation of MPs in Different Polymer Types in WWTP
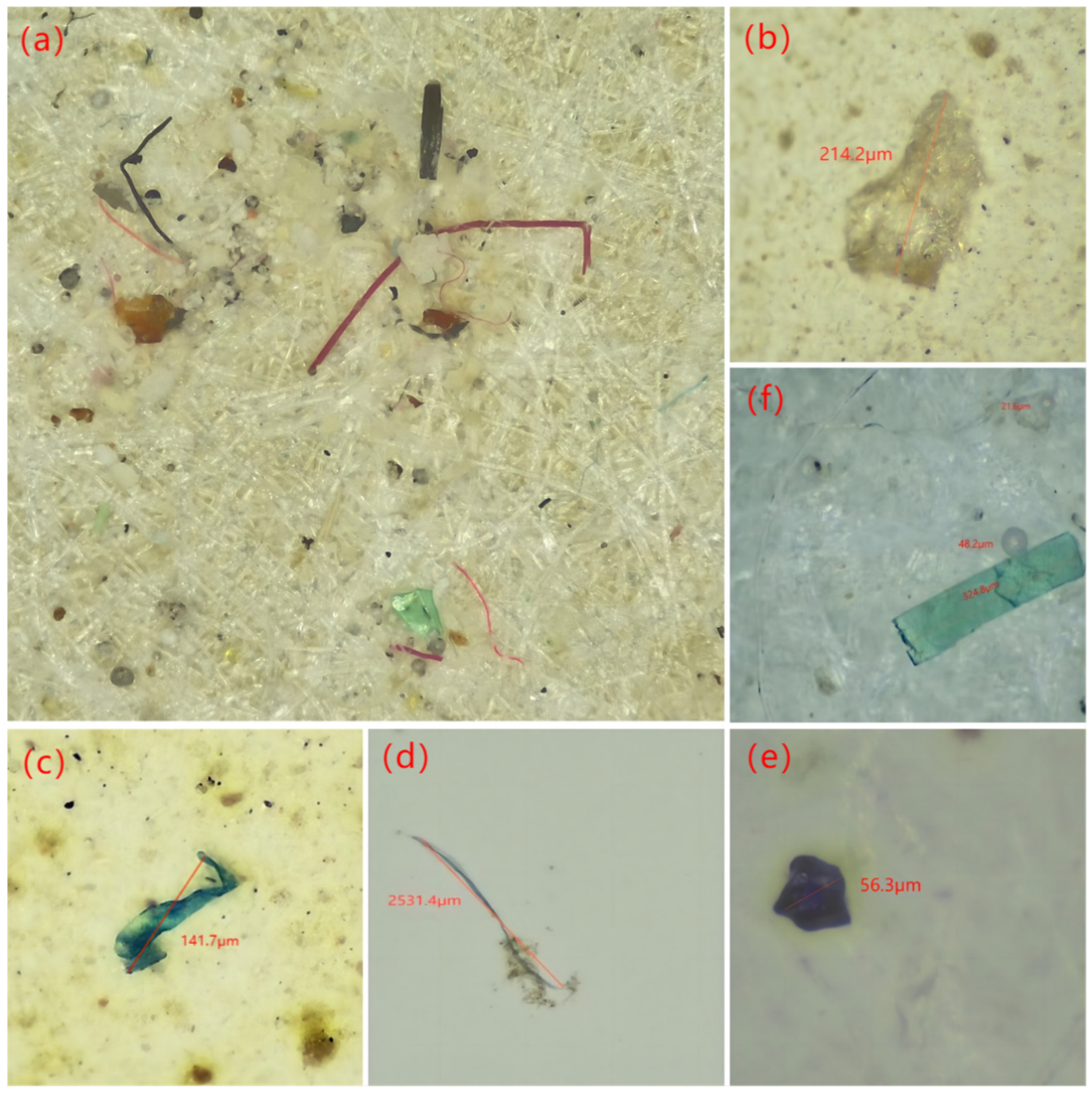
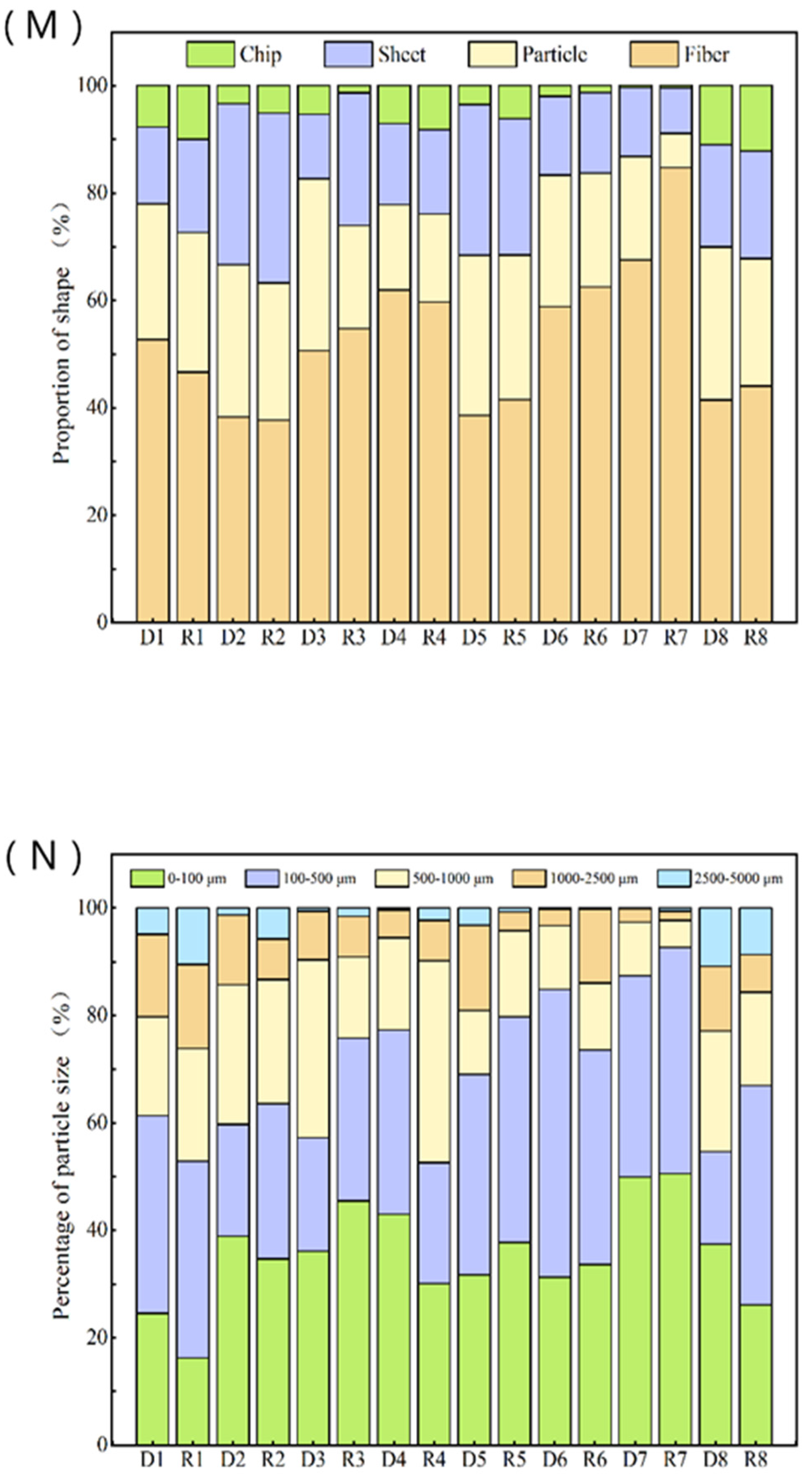
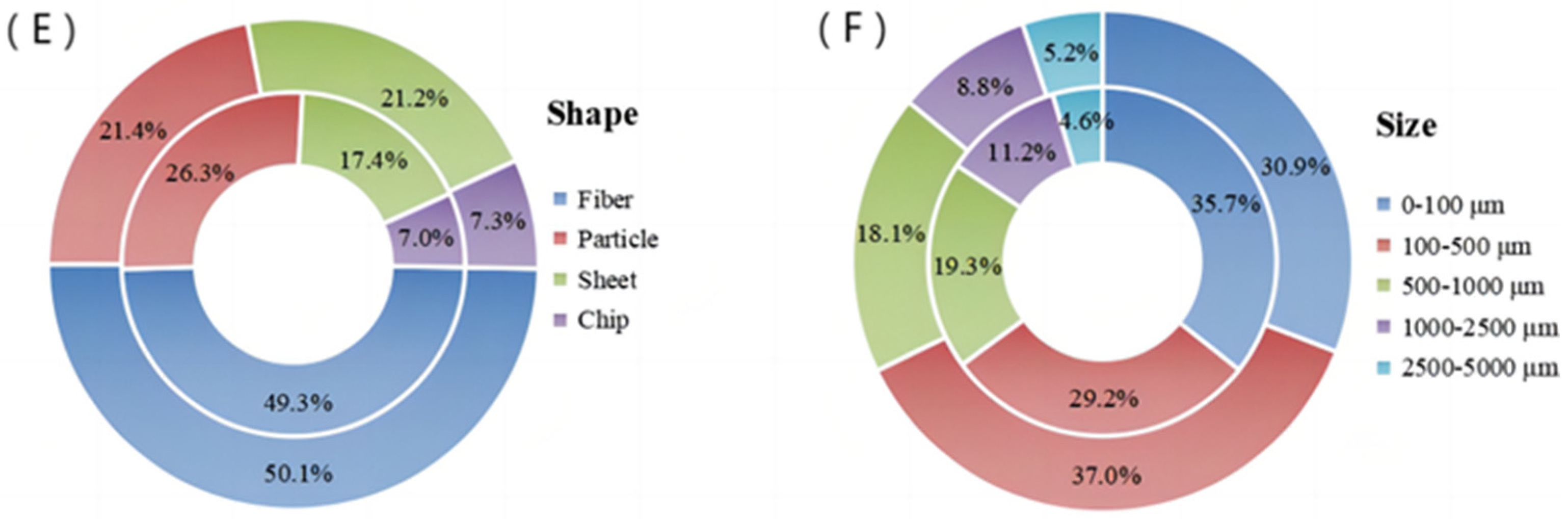
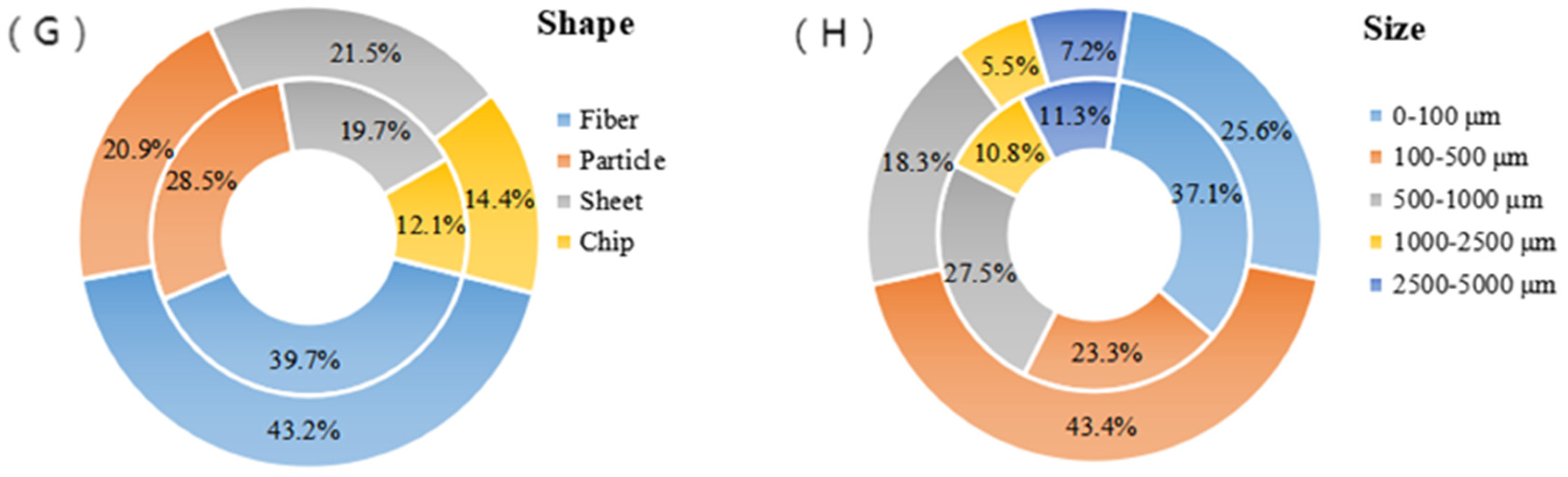
3.3. Shape, Distribution and Size of MPs in Sewage Plants
3.4. Contamination Risk Evaluation of MPs in WWTP
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Wright, S.L.; Kelly, F.J. Plastic and human health: A micro issue? Environ. Sci. Technol. 2017, 51, 6634–6647. [Google Scholar] [PubMed]
- Geyer, R.; Jambeck, J.R.; Law, K.L. Production, Use, and Fate of All Plastics Ever Made. Sci. Adv. 2017, 3, 1700782. [Google Scholar] [CrossRef] [PubMed]
- Chakraborty, P.; Chandra, S.; Dimmen, M.V.; Hurley, R.; Mohanty, S.; Bharat, G.K.; Steindal, E.H.; Olsen, M.; Nizzetto, L. Interlinkage Between Persistent Organic Pollutants and Plastic in the Waste Management System of India: An Overview. Bull. Environ. Contam. Toxicol. 2022, 109, 927–936. [Google Scholar]
- Lee, J.; Hong, S.; Song, Y.K.; Sang, H.H.; Yong, C.J.; Mi, J.; Heo, N.W.; Han, G.M.; Mi, J.L.; Kang, D. Relationships among the abundances of plastic debris in different size classes on beaches in South Korea. Mar. Pollut. Bull. 2013, 77, 349–354. [Google Scholar]
- Liu, X.; Yuan, W.; Di, M.; Li, Z.; Wang, J. Transfer and fate of microplastics during the conventional activated sludge process in one wastewater treatment plant of China. Chem. Eng. J. 2019, 362, 176–182. [Google Scholar] [CrossRef]
- Wagner, M.; Scherer, C.; Alvarez-Muoz, D.; Brennholt, N.; Bourrain, X.; Buchinger, S.; Fries, E.; Grosbois, C.; Klasmeier, J.; Marti, T. Mi-croplastics in freshwater ecosystems: What we know and we need to know. Environ. Sci. Eur. 2014, 26, 12. [Google Scholar] [CrossRef] [PubMed]
- Silva, A.B.; Bastos, A.S.; Justino, C.I.L.; da Costa, J.P.; Duarte, A.C.; Rocha-Santos, T.A.P. Microplastics in the environment: Challenges in analytical chemistry—A review. Anal. Chim. Acta 2018, 1017, 1–19. [Google Scholar]
- Lee, J.; Enfrin, M.; Dum, L.F.; Dumée, L.F.; Lee, J. Nano/microplastics in water and wastewater treatment processes—Origin, impact and potential solutions. Water Res. 2019, 161, 621–638. [Google Scholar]
- Brennecke, D.; Duarte, B.; Paiva, F.; Caçador, I.; Canning-Clode, J. Microplastics as vector for heavy metal contamination from the marine environment. Estuar. Coast. Shelf Sci. 2016, 178, 189–195. [Google Scholar] [CrossRef]
- Kida, M.; Koszelnik, P. Investigation of the Presence and Possible Migration from Microplastics of Phthalic Acid Esters and Polycyclic Aromatic Hydrocarbons. J. Polym. Environ. 2020, 29, 599–611. [Google Scholar]
- Abbasi, S.; Keshavarzi, B.; Moore, F.; Shojaei, N.; Sorooshian, A.; Soltani, N.; Delshab, H. Geochemistry and environmental effects of potentially toxic elements, polycyclic aromatic hydrocarbons and microplastics in coastal sediments of the persian gulf. Environ. Geol. 2019, 492, 2–15. [Google Scholar]
- Mclellan, S.L.; Huse, S.M.; Mueller-Spitz, S.R.; Andreishcheva, E.N.; Sogin, M.L. Diversity and population structure of sewage-derived microorganisms in wastewater treatment plant influent. Environ. Microbiol. 2010, 12, 378–392. [Google Scholar] [PubMed]
- Li, J.; Shen, T.; Wang, H.; Li, S.; Wang, J.; Williams, R.G.; Zhao, Y.; Kong, X.; Zheng, L.; Song, Y. Insights into the Superstable Mineralization of Chromium(III) from Wastewater by CuO. ACS Appl. Mater. Interfaces 2022, 14, 37823–37832. [Google Scholar] [CrossRef] [PubMed]
- He, S.; Jia, M.; Xiang, Y.; Song, B.; Xiong, W.; Cao, J.; Peng, H.; Yang, Y.; Wang, W.; Yang, Z.; et al. Biofilm on microplastics in aqueous environment: Physicochemical properties and environmental implications. J. Hazard. Mater. 2022, 424, 127286. [Google Scholar] [CrossRef] [PubMed]
- Vcs, A.; Pgb, C.; Em, A.; Km, B. First study of its kind on the microplastic contamination ofsoft drinks, cold tea and energy drinks—Future research and envi-ronmental considerations. Sci. Total Environ. 2020, 726, 138580. [Google Scholar]
- Luo, H.; Liu, C.; He, D.; Sun, J.; Li, J.; Pan, X. Effects of aging on environmental behavior of plastic additives: Migration, leaching, and ecotoxicity. Sci. Total Environ. 2022, 849, 157951. [Google Scholar] [CrossRef]
- Botterell, Z.L.R.; Beaumont, N.; Dorrington, T.; Steinke, M.; Thompson, R.C.; Lindeque, P.K. Bioavailability and efects of microplastics on marine zooplankton: A review. Environ. Pollut. 2019, 245, 98–110. [Google Scholar] [CrossRef]
- Wang, L.; Liu, Y.; Kaur, M.; Yao, Z.; Chen, T.; Xu, M. Phytotoxic Effects of Polyethylene Microplastics on the Growth of Food Crops Soybean (Glycine max) and Mung Bean (Vigna radiata). Int. J. Environ. Res. Pub. Health 2021, 18, 10629. [Google Scholar]
- Toussaint, B.; Raffael, B.; Angers-Loustau, A.; Gilliland, D.; Kestens, V.; Petrillo, M.; Rio-Echevarria, I.M.; Van den Eede, G. Review of Micro- and Nanoplastic Contamination in the Food Chain. Food Addit. Contam. Part A 2019, 36, 639–673. [Google Scholar]
- Schwabl, P.; Köppel, S.; Königshofer, P.; Bucsics, T.; Trauner, M.; Reiberger, T.; Liebmann, B. Detection of various microplastics in human stool: A prospective case series. Ann. Intern. Med. 2019, 171, 453–457. [Google Scholar]
- Zhang, J.; Wang, L.; Trasande, L.; Kannan, K. Occurrence of Polyethylene Terephthalate and Polycarbonate Microplastics in Infant and Adult Feces. Environ. Sci. Technol. Lett. 2021, 8, 989–994. [Google Scholar]
- Hal, A.; Mjmvv, A.; Shb, A.; Adva, B.; Gv, C.; Mhl, A. Discovery and quantification of plastic particle pollution in human blood. Environ. Int. 2022, 163, 107199. [Google Scholar]
- Ragusa, A.; Svelato, A.; Santacroce, C.; Catalano, P.; Giorgini, E. Plasticenta: First evidence of microplastics in human placenta. Environ. Int. 2021, 146, 106274. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.K.; Song, N.S. Microplastics in edible Salt: A literature review focusing on Uncertainty related with Measured Minimum Cutoff Sizes. Curr. Opin. Food Sci. 2021, 41, 16–25. [Google Scholar] [CrossRef]
- Jin, M.; Wang, X.; Ren, T.; Wang, J.; Shan, J. Microplastics contamination in food and beverages: Direct exposure to humans. J. Food Sci. 2021, 86, 2816–2837. [Google Scholar] [CrossRef]
- Zhang, Q.; Xu, E.G.; Li, J.; Chen, Q.; Ma, L.; Zeng, E.Y.; Shi, H. A review of microplastics in table salt, drinking water, and air: Direct human exposure. Environ. Sci. Technol. 2020, 54, 3740–3751. [Google Scholar] [CrossRef]
- Jenner, L.C.; Rotchell, J.M.; Bennett, R.T.; Cowen, M.; Tentzeris, V.; Sadofsky, L.R. Detection of microplastics in human lung tissue using μFTIR spectroscopy. Sci. Total Environ. 2022, 831, 154907. [Google Scholar]
- Fournier, S.B.; D’Errico, J.N.; Adler, D.S.; Kollontzi, S.; Stapleton, P.A. Nanopolystyrene translocation and fetal deposition after acute lung exposure during late-stage pregnancy. Part. Fibre Toxicol. 2020, 17, 55. [Google Scholar]
- Murphy, F.; Ewins, C.; Carbonnier, F.; Quinn, B. Wastewatertreatment works (WwTW) as a source of microplastics in theaquatic environment. Environ. Sci. Technol. 2016, 50, 5800–5808. [Google Scholar] [CrossRef]
- Talvitie, J.; Mikola, A.; Koistinen, A.; Setälä, O. Solutions to microplastic pollution–Removal of microplastics from wastewater efuent with advanced wastewater treatment technologies. Water Res. 2017, 123, 401–407. [Google Scholar] [CrossRef]
- Mason, S.A.; Garneau, D.; Sutton, R.; Chu, Y.; Ehmann, K.; Barnes, J.; Fink, P.; Papazissimos, D.; Rogers, D.L. Microplastic pollution is widely detected in US municipal wastewater treatment plant effluent. Environ. Pollut. 2016, 218, 1045–1054. [Google Scholar] [CrossRef] [PubMed]
- Xu, X.; Jian, Y.; Xue, Y.; Hou, Q.; Wang, L.P. Microplastics in the wastewater treatment plants (WWTPs): Occurrence and removal. Chemosphere 2019, 235, 1089–1096. [Google Scholar] [PubMed]
- Zhang, K.; Shi, H.; Peng, J.; Wang, Y.; Xiong, X.; Wu, C.; Lam, P.K.S. Microplastic pollution in China’s inland water systems: A review of findings, methods, characteristics, effects, and management. Sci. Total Environ. 2018, 630, 1641–1653. [Google Scholar] [CrossRef]
- Gies, E.A.; LeNoble, J.L.; Noël, M.; Etemadifar, A.; Bishay, F.; Hall, E.R.; Ross, P.S. Retention of microplastics in a major secondary wastewater treatment plant in Vancouver, Canada Mar. Pollut. Bull. 2018, 133, 553–561. [Google Scholar]
- Paul, K.; Robert, H.; Isobel, M.; Luke, B.; Niamh, M. Wastewater treatment plants as a source of microplastics in river catchments. Environ. Sci. Pollut. Res. 2018, 25, 20265–20266. [Google Scholar]
- Thompson, R.C.; Moore, C.J.; Saal, F.V.; Swan, S.H. Plastics, the environment and human health: Current consensus andfuture trends. Philos. Trans. R. Soc. Lond. B Biol. Sci. 2009, 364, 2153–2166. [Google Scholar] [PubMed]
- Michelbach, S.; Wöhrle, C. Settleable Solids in a Combined Sewer System, Settling Characteristics, Heavy Metals, Efficiency of Storm Water Tanks. Water Sci. Technol. 1993, 27, 153–164. [Google Scholar] [CrossRef]
- Gasperi, J.; Gromaire, M.C.; Kafi, M.; Moilleron, R.; Chebbo, G. Contributions of wastewater, runoff and sewer depositerosion to wet weather pollutant loads in combined sewersystems. Water Res. 2010, 44, 5875–5886. [Google Scholar] [CrossRef]
- Hollender, J.; Zimmermann, S.G.; Koepke, S.; Keaoss, M.; Mcardell, C.S.; Ort, C.; Singer, H.; Gunten, U.V.; Siegrist, H. Elimination of organic micropollutants in a municipal wastewater treatment plant upgraded with a full-scale post-ozonation followed by sand filtration. Environ. Sci. Technol. 2009, 43, 7862–7869. [Google Scholar]
- Noriatsu, O.; Takahiro, Y.; Tomonori, K.; Akiyoshi, O. Stormwater inflow loading of polycyclic aromatic hydrocarbons into urban domestic wastewater treatment plant for separate sewer system. Water Sci. Technol. 2019, 79, 1426–1436. [Google Scholar]
- Hurley, R.; Lusher, A.; Olsen, M.; Nizzetto, L. Validation of a method for extracting microplastics from complex, organic-rich, environmental matrices. Environ. Sci. Technol. 2018, 52, 7409–7417. [Google Scholar] [PubMed]
- Nuelle, M.T.; Dekiff, J.H.; Remy, D.; Fries, E. A new analytical approach for monitoring microplastics in marine sediments. Environ. Pollut. 2014, 184, 161–169. [Google Scholar] [PubMed]
- Carr, S.A.; Liu, J.; Tesoro, A.G. Transport and fate of micro-plastic particles in wastewater treatment plants. Water Res. 2016, 91, 174–182. [Google Scholar] [PubMed]
- Käppler, A.; Fischer, D.; Oberbeckmann, S.; Schernewski, G.; Labrenz, M.; Eichhorn, K.J.; Voit, B. Analysis of environmental microplastics by vibrational microspectroscopy: FTIR, Raman or both? Anal. Bioanal. Chem. 2016, 408, 8377–8391. [Google Scholar]
- Browne, M.A.; Galloway, T.S.; Thompson, R.C. Spatial patterns of plastic debris along estuarine shorelines. Environ. Sci. Technol. 2010, 44, 3404–3409. [Google Scholar]
- Pivokonsky, M.; Cermakova, L.; Novotna, K.; Peer, P.; Cajthaml, T.; Janda, V. Occurrence of microplastics in raw and treated drinking water. Sci. Total Environ. 2018, 643, 1644–1651. [Google Scholar]
- Liu, D.T.; Song, Y.; Li, F.F.; Chen, L.J. A detection method of small-sized microplastics based on micro-raman mapping. CHN Environ. Sci. 2020, 40, 4429–4438. [Google Scholar]
- Tomlinson, D.L.; Wilson, J.G.; Harris, C.R.; Jeffrey, D.W. Problems in the assessment of heavy-metal levels in estuaries and the formation of a pollution index. Helgolnder Meeresuntersuchungen 1980, 33, 566–575. [Google Scholar]
- Lithner, D.; Larsson, A.; Dave, G. Environmental and health hazard ranking and assessment of plastic polymers based on chemical composition. Sci. Total Environ. 2011, 409, 3309–3324. [Google Scholar]
- Wei, W.; Huang, Q.S.; Sun, J.; Wang, J.Y.; Wu, S.L.; Ni, B.J. Polyvinyl Chloride Microplastics Affect Methane Production from the Anaerobic Digestion of Waste Activated Sludge through Leaching Toxic Bisphenol-A. Environ. Sci. Technol. 2019, 53, 2509–2517. [Google Scholar]
- Ghimici, L.; Nichifor, M. Flocculation by cationic amphiphilic polyelectrolyte: Relating efficiency with the association of polyelectrolyte in the initial solution Colloids Surf. A Physicochem. Eng. Asp. 2012, 415, 142–147. [Google Scholar]
- Girish, C.R.; Singh, S.K.; Shenoy, S. Removal of suspended solids from dairy wastewater using flocculation. Int. J. Mech. Eng. Technol. 2017, 8, 99–105. [Google Scholar]
- Zhu, S.; Khan, M.A.; Kameda, T.; Xu, H.; Wang, F.; Xia, M.; Yoshioka, T. New insights into the capture performance and mechanism of hazardous metals cr3+ and cd2+ onto an effective layered double hydroxide based material. J. Hazard. Mater. 2022, 426, 128062. [Google Scholar] [CrossRef] [PubMed]
- He, Z.W.; Yang, W.J.; Ren, Y.X.; Jin, H.Y.; Wang, A.J. Occurrence, effect, and fate of residual microplastics in anaerobic digestion of waste activated sludge: A state-of-the-art review. Bioresour. Technol. 2021, 331, 125035. [Google Scholar] [CrossRef] [PubMed]
- Conley, K.; Clum, A.; Deepe, J.; Lane, H.; Beckingham, B. Wastewater treatment plants as a source of microplastics to an urban estuary: Removal efficiencies and loading per capita over one year. Water Res. X 2019, 3, 100030. [Google Scholar] [PubMed]
- Shima, Z.; Peta, A.N.; Llew, R.; Frederic, L. Wastewater treatment plants as a pathway for microplastics: Development of a new approach to sample wastewater-based microplastics. Water Res. 2017, 112, 93–99. [Google Scholar]
- Koyuncuolu, P.; Gülbin Erden. Sampling, pre-treatment, and identifcation methods of microplastics in sewage sludge and their efects in agricultural soils: A review. Environ. Monit. Assess. 2021, 193, 175–177. [Google Scholar] [CrossRef]
- Li, X.; Chen, L.; Mei, Q.; Dong, B.; Dai, X.; Ding, G.; Zeng, E.Y. Microplastics in sewage sludge from the wastewater treatment plants in China. Water Res. 2018, 142, 75–85. [Google Scholar] [CrossRef]
- Ramasamy, R.; Aragaw, T.A.; Balasaraswathi Subramanian, R. Wastewater treatment plant effluent and microfiber pollution: Focus on industry-specific wastewater. Environ. Sci. Pollut. Res. 2022, 29, 51211–51233. [Google Scholar] [CrossRef]
- Carney Almroth, B.M.; Åström, L.; Roslund, S.; Petersson, H.; Johansson, M.; Persson, N.K. Persson, Nils-Krister. Quantifying shedding of synthetic fibers from textiles, a source of microplastics released into the environment. Environ. Sci. Pollut. Res. 2017, 25, 1191–1199. [Google Scholar] [CrossRef]
- Zhu, J.; Zhang, X.; Liao, K.; Wu, p.; Jin, H. Microplastics in dust from different indoor environments. Sci. Total Environ. 2022, 833, 155256. [Google Scholar] [CrossRef] [PubMed]
- Galgani, F.; Hanke, G.; Werner, S.; De Vrees, L. Marine litter within the european marine strategy framework directive. ICES J Mar. Sci. 2013, 70, 1055–1064. [Google Scholar] [CrossRef]
- Napper, I.E.; Bakir, A.; Rowland, S.J.; Thompson, R.C. Characterisation, quantity and sorptive properties of microplastics extracted from cosmetics. Mar. Pollut. Bull. 2015, 99, 178–185. [Google Scholar] [CrossRef] [PubMed]
- Ng, E.-L.; Lwanga, E.H.; Eldridge, S.M.; Johnston, P.; Hu, H.-W.; Geissen, V.; Chen, D. An overview of microplastic and nanoplastic pollution in agroecosystems. Sci. Total Environ. 2018, 627, 1377–1388. [Google Scholar] [CrossRef] [PubMed]
- Hatinoğlu, M.D.; Sanin, F.D. Sewage sludge as a source of microplastics in the environment: A review of occurrence and fate during sludge treatment. J. Environ. Manag. 2021, 295, 113028. [Google Scholar] [CrossRef]
- Harley-Nyang, D.; Memon, F.A.; Jones, N.; Galloway, T. Investigation and analysis of microplastics in sewage sludge and biosolids: A case study from one wastewater treatment works in the UK. Sci. Total Environ. 2022, 823, 153735. [Google Scholar] [CrossRef]
- Hidayaturrahman, H.; Lee, T.G. A study on characteristics of microplastic in wastewater of South Korea: Identification, quantification, and fate of microplastics during treatment process. Mar. Pollut. Bull. 2019, 146, 696–702. [Google Scholar]
- Senathirajah, K.; Attwood, S.; Bhagwat, G.; Carbery, M.; Wilson, S.; Palanisami, T. Estimation of the mass of microplastics ingested—A pivotal first step towards human health risk assessment. J. Hazard. Mater. 2021, 404, 124004.1–124004.15. [Google Scholar]
- Xu, P.; Peng, G.; Lei, S.; Gao, L.; Li, D. Microplastic risk assessment in surface waters: A case study in the Changjiang Estuary, China. Mar. Pollut. Bull. 2018, 133, 647–654. [Google Scholar]
- Kyriakopoulos, G.L.; Zamparas, M.; Kapsalis, V.C. Eutrophication control: The shif to invasive methods managing the internal nutrient loads. a bibliometric analysis. Desalin. Water Treat. 2022, 267, 177–185. [Google Scholar] [CrossRef]
- Ranjani, M.; Veerasingam, S.; Venkatachalapathy, R.; Mugilarasan, M.; Bagaev, A.; Mukhanov, V.; Vethamony, P. Assessment of potential ecological risk of microplastics in the coastal sedime. Mar. Pollut. Bull. 2021, 163, 111969. [Google Scholar] [PubMed]
- Kyriakopoulos, L.G.; Zamparas, G.M.; Kapsalis, C.V. Investigating the Human Impacts and the Environmental Consequences of Microplastics Disposal into Water Resources. Sustainability 2022, 14, 828. [Google Scholar]
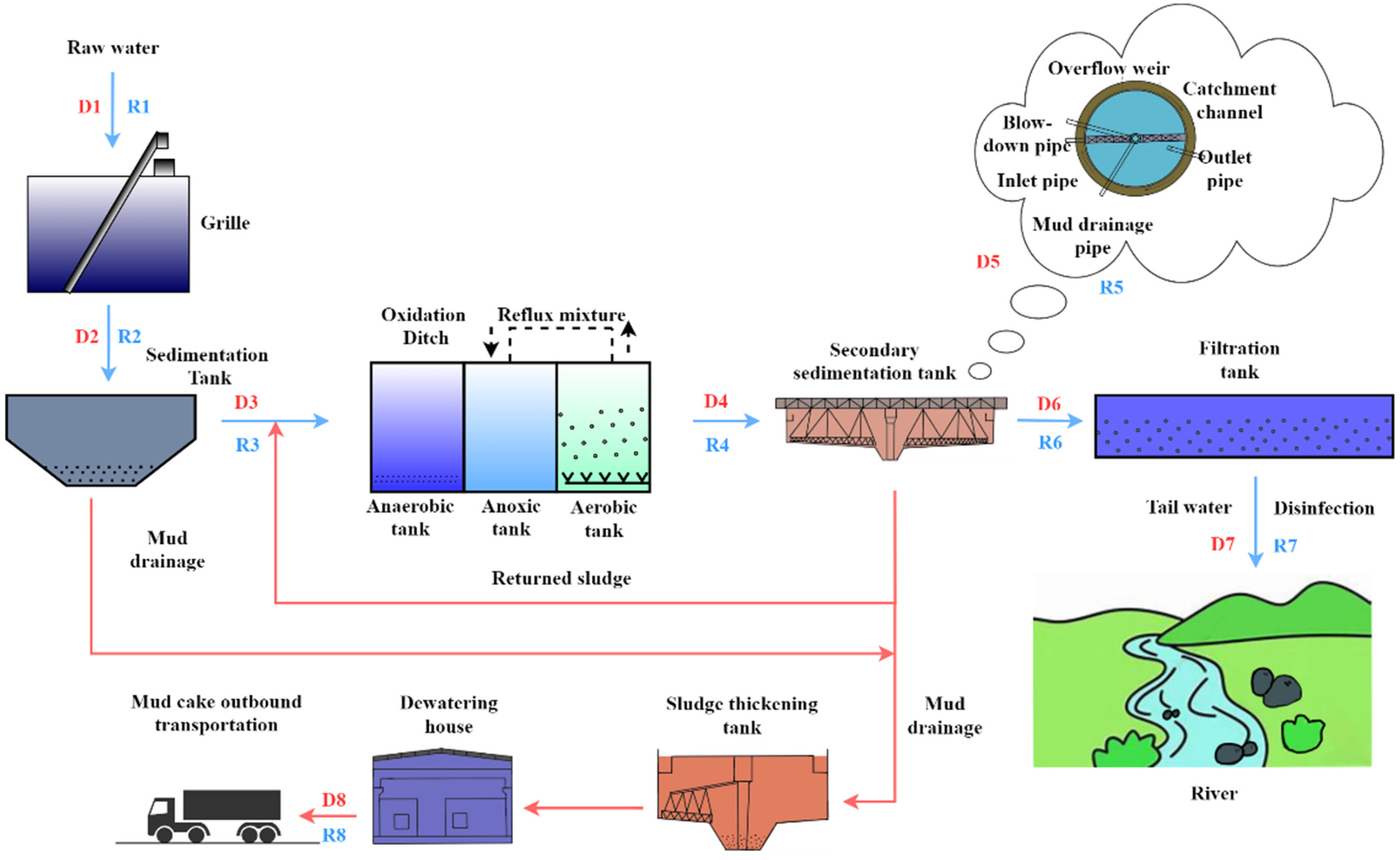

| Sample Points D (Dry Weather), R (Rainy Weather) | Layout Design Description |
|---|---|
| Raw water or Grille Front (D1, R1) | Initial wastewater from WWTP. Sewage samples are collected at the grate. |
| After grille (D2, R2) | The first physical interception processing. Sewage samples are collected behind the grate. |
| After sedimentation Tank (D3, R3) | Gravity deposition of high-density contaminated impurities separates low density suspended matter to the next process. The sewage samples were collected after sand-settling. |
| After oxidation Ditch (D4, R4) | (Anaerobic-Anoxic-Aerobic) to maintain the flow of mixed sewage and activated sludge, initial removal of suspended substances. Sewage samples are collected behind the oxidation ditch. |
| Inside the secondary sedimentation tank (D5, R5) | The flow velocity and amount of water affect the cross-section of rainwater, and the suspended matter rises. Sewage samples are collected in the secondary sedimentation tank. |
| After secondary settling tank (D6, R6) | The mud and water are separated and the suspended impurities form flocculates and sink together. Sewage samples are collected after the secondary sedimentation tank is selected. |
| Tail water or After filtration tank (D7, R7) | WWTP outflow tail water. Sewage samples are collected after the denitrification filter. |
| Dehydrated sludge (D8, R8) | After being dehydrated by an enrichment centrifuge. Sludge samples are collected on the conveyor belt. |
| Treatment Phase | Arrange | MPs Abundance (n/L) | Removal Efficiency (%) | Total Removal Rate of MPs (%) |
|---|---|---|---|---|
| Primary processing | D1 to D3, R1 to R3 | 101.9 ± 17.6 to 51.0 ± 7.3, 108.7 ± 20.1 to 81.2 ± 10.8 | 62.9%, 70.4% | 87.7% (D), 83.5% (R) |
| Secondary treatment | D4 to D6, R4 to R6 | 71.9 ± 15.3 to 44.2 ± 5.5, 87.4 ± 21.3 to 53.6 ± 7.4 | 55.6%, 57.5% | |
| Tertiary or Advanced treatment | D6 to D7, R6 to R7 | 44.2 ± 5.5 to 18.2 ± 3.6, 53.6 ± 7.4 to 26.3 ± 5.1 | 44.9%, 34.6% |
| Process Segment | D1 | D2 | D3 | D4 | D5 | D6 | D7 | D8 |
|---|---|---|---|---|---|---|---|---|
| Scanning points (U1, U2, C3, D4, D5) | 125 | 93 | 62 | 137 | 104 | 78 | 34 | 260 |
| Shape (Formula (1)) | ||||||||
| Fiber | 5.9 | 1.7 | 1.9 | 4.3 | 1.7 | 2.0 | 0.9 | 5.6 |
| Chip | 2.8 | 1.3 | 1.2 | 1.1 | 1.3 | 0.9 | 0.3 | 3.9 |
| Sheet | 1.6 | 1.4 | 0.5 | 1.0 | 0.9 | 0.5 | 0.1 | 2.5 |
| Particle | 0.9 | 0.1 | 0.2 | 0.5 | 0.2 | 0 | 0 | 1.5 |
| Size (Formula (1)) | ||||||||
| 0–100 μm | 2.8 | 1.8 | 1.4 | 3.0 | 1.4 | 1.1 | 0.7 | 4.9 |
| 100–500 μm | 4.1 | 1.0 | 0.8 | 2.4 | 1.6 | 1.8 | 0.5 | 2.3 |
| 500–1000 μm | 2.0 | 1.2 | 1.3 | 1.1 | 0.5 | 0.4 | 0.1 | 3.0 |
| 1000–2500 μm | 1.7 | 0.6 | 0.3 | 0.4 | 0.7 | 0.1 | 0 | 1.7 |
| 2500–5000 μm | 0.6 | 0 | 0 | 0.1 | 0.1 | 0 | 0 | 1.5 |
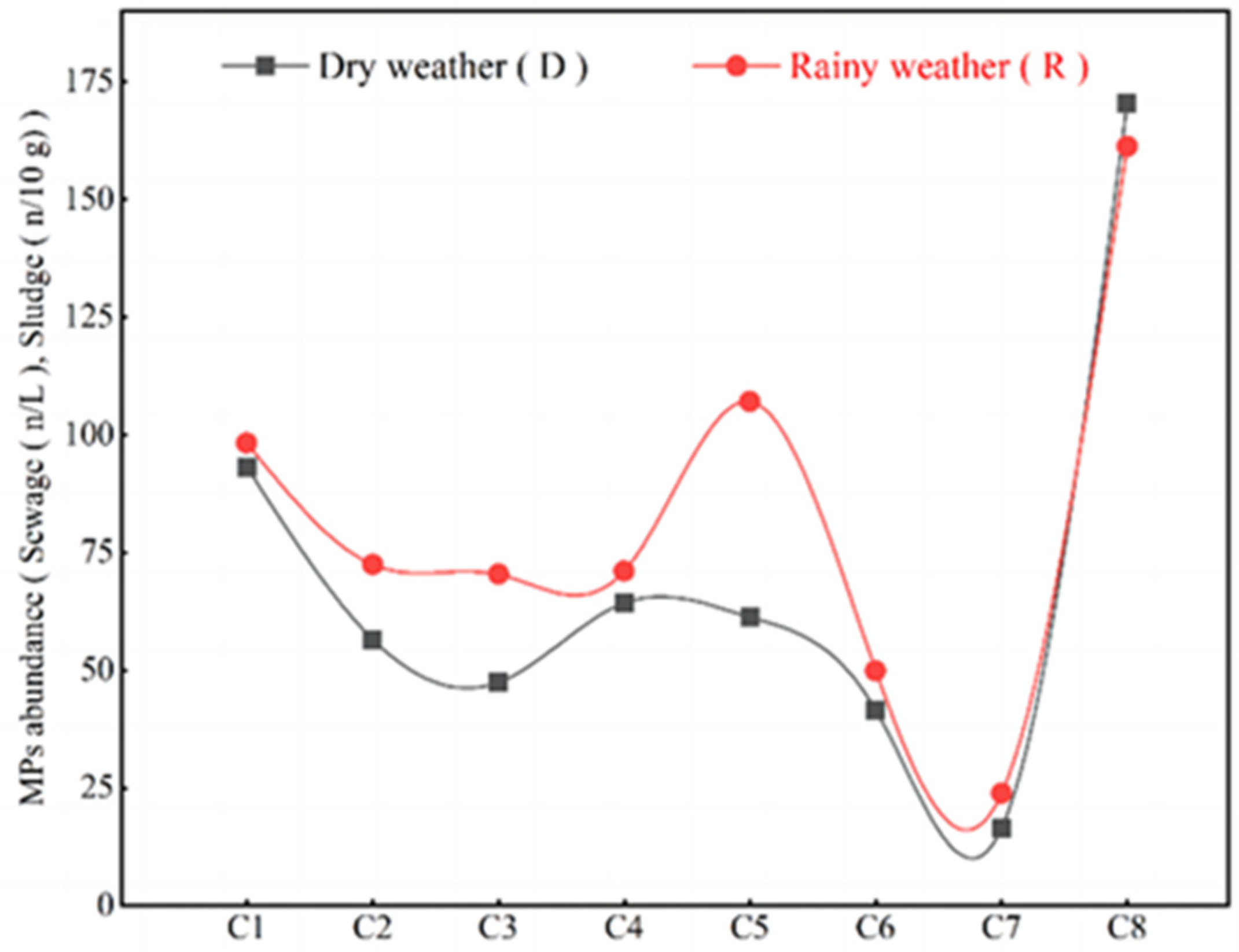
| Actual MPs abundance, Formula (1) | 101.9 ± 17.6 | 61.1 ± 9.3 | 51.0 ± 7.3 | 71.9 ± 15.3 | 68.1 ± 13.6 | 44.2 ± 5.5 | 18.2 ± 3.6 | 184.8 ± 28.6 |
| Process Segment | R1 | R2 | R3 | R4 | R5 | R6 | R7 | R8 |
|---|---|---|---|---|---|---|---|---|
| Scanning points, (U1, U2, C3, D4, D5) | 85 | 93 | 82 | 113 | 131 | 57 | 55 | 277 |
| Shape, (Formula (1)) | ||||||||
| Fiber | 5.0 | 2.3 | 1.9 | 2.3 | 3.8 | 2.8 | 1.7 | 5.5 |
| Chip | 2.8 | 1.6 | 1.2 | 0.1 | 1.0 | 0.9 | 0.1 | 3.0 |
| Sheet | 1.8 | 2.0 | 0.5 | 1.0 | 1.0 | 0.7 | 0.2 | 2.5 |
| Particle | 1.1 | 0.3 | 0.2 | 0 | 0.5 | 0 | 0 | 1.5 |
| Size, (Formula (1)) | ||||||||
| 0–100 μm | 1.7 | 2.1 | 1.9 | 1.9 | 3.3 | 1.5 | 1.0 | 3.3 |
| 100–500 μm | 4.0 | 1.8 | 1.3 | 1.4 | 3.7 | 1.8 | 0.8 | 5.2 |
| 500–1000 μm | 1.3 | 1.4 | 0.6 | 2.4 | 1.4 | 0.5 | 0.1 | 2.1 |
| 1000–2500 μm | 1.7 | 0.5 | 0.3 | 0.5 | 0.3 | 0.6 | 0 | 0.9 |
| 2500–5000 μm | 1.1 | 0.4 | 0 | 0.1 | 0.1 | 0 | 0 | 1.1 |
| Actual MPs abundance, Formula (1) | 108.7 ± 20.1 | 77.9 ± 11 | 81.2 ± 10.8 | 87.4 ± 21.3 | 117.3 ± 22.4 | 53.6 ± 7.4 | 26.3 ± 5.1 | 178.4 ± 34.3 |
| Type of Polymer | PE | PP | PS | PET | ||||
|---|---|---|---|---|---|---|---|---|
| Hazard score (Highest level); Sn | 11 | 1 | 4 | 30 | ||||
| Process Segment | D | R | D | R | D | R | D | R |
| Pn (%) | 11.30 | 15.89 | 6.88 | 12.33 | 9.83 | 8.60 | 10.81 | 8.60 |
| H, (Formula (5)) | 1.24 | 1.75 | 0.07 | 0.12 | 0.39 | 0.34 | 3.24 | 2.58 |
| Potential ecplogical risk level of MPs | I | I | I | I | I | I | I | I |
| PLIi (Formula (2) and (3)) | 2.40 | 2.62 | 1.88 | 2.30 | 1.93 | 2.72 | 1.63 | 1.95 |
| Type of Polymer | PU | PA | PF | PVC | ||||
|---|---|---|---|---|---|---|---|---|
| Hazard score (Highest level); Sn | 871 | 50 | 1450 | 30 | ||||
| Process Segment | D | R | D | R | D | R | D | R |
| Pn (%) | 5.65 | 6.54 | 24.32 | 21.68 | 17.94 | 14.95 | 13.27 | 11.40 |
| H, (Formula (5)) | 49.22 | 56.98 | 352.70 | 314.39 | 1324.40 | 1104.15 | 663.52 | 570.20 |
| Potential ecplogical risk level of MPs | II | II | III | III | III | III | III | III |
| PLIi (Formula (2) and (3)) | 1.00 | 1.51 | 1.04 | 1.00 | 1.73 | 2.39 | 1.97 | 2.42 |
| PLIzone (Formula (4)) | PLIzone (Dry weather) value is 1.63 (moderately pollution), PLIzone (Rainy weather) value is 2.03 (highly pollution) | |||||||
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Meng, X.; Bao, T.; Hong, L.; Wu, K. Occurrence Characterization and Contamination Risk Evaluation of Microplastics in Hefei’s Urban Wastewater Treatment Plant. Water 2023, 15, 686. https://doi.org/10.3390/w15040686
Meng X, Bao T, Hong L, Wu K. Occurrence Characterization and Contamination Risk Evaluation of Microplastics in Hefei’s Urban Wastewater Treatment Plant. Water. 2023; 15(4):686. https://doi.org/10.3390/w15040686
Chicago/Turabian StyleMeng, Xiangwu, Teng Bao, Lei Hong, and Ke Wu. 2023. "Occurrence Characterization and Contamination Risk Evaluation of Microplastics in Hefei’s Urban Wastewater Treatment Plant" Water 15, no. 4: 686. https://doi.org/10.3390/w15040686
APA StyleMeng, X., Bao, T., Hong, L., & Wu, K. (2023). Occurrence Characterization and Contamination Risk Evaluation of Microplastics in Hefei’s Urban Wastewater Treatment Plant. Water, 15(4), 686. https://doi.org/10.3390/w15040686










