Towards Viable Eco-Friendly Local Treatment of Blackwater in Sparsely Populated Regions
Abstract
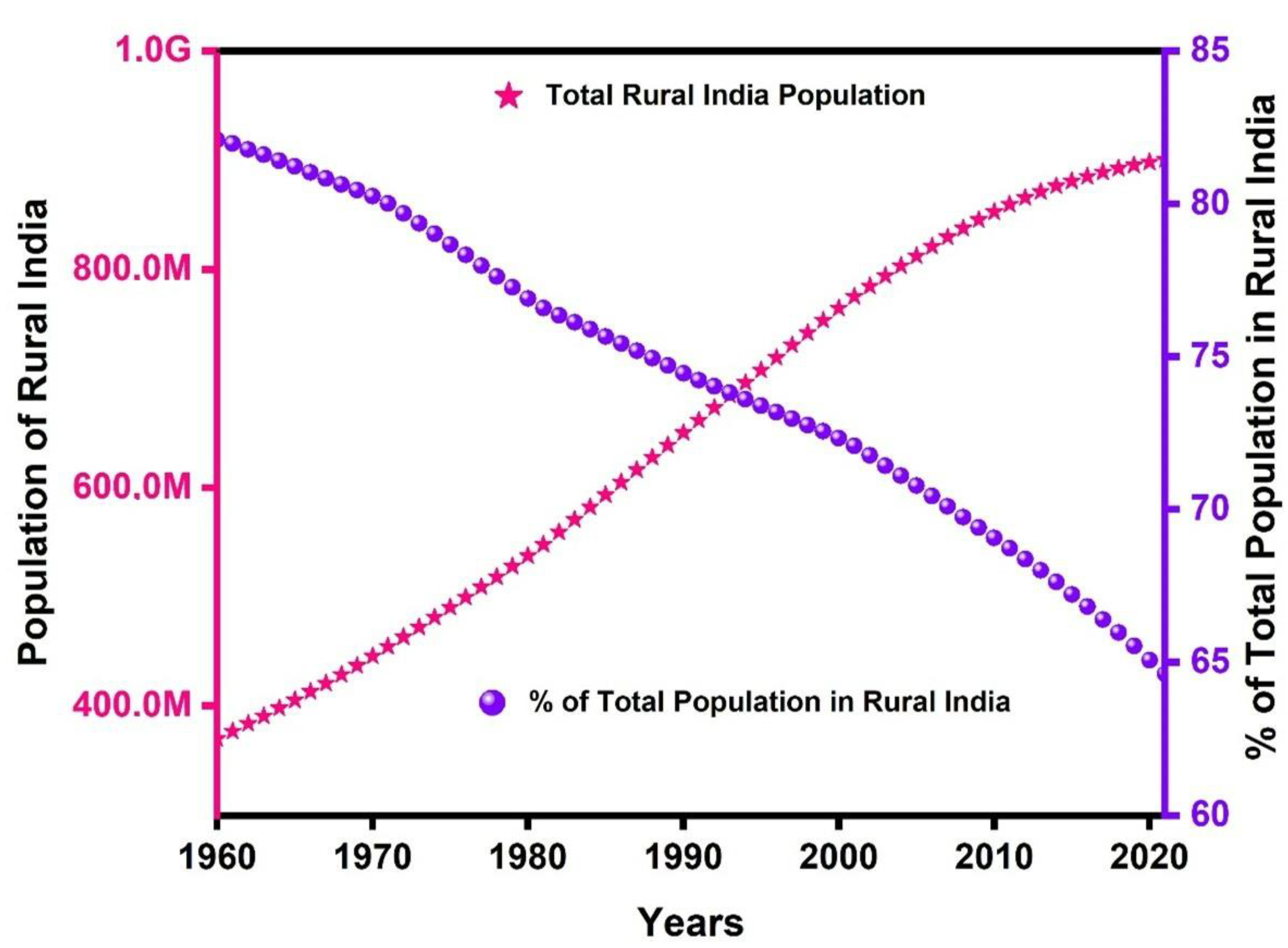
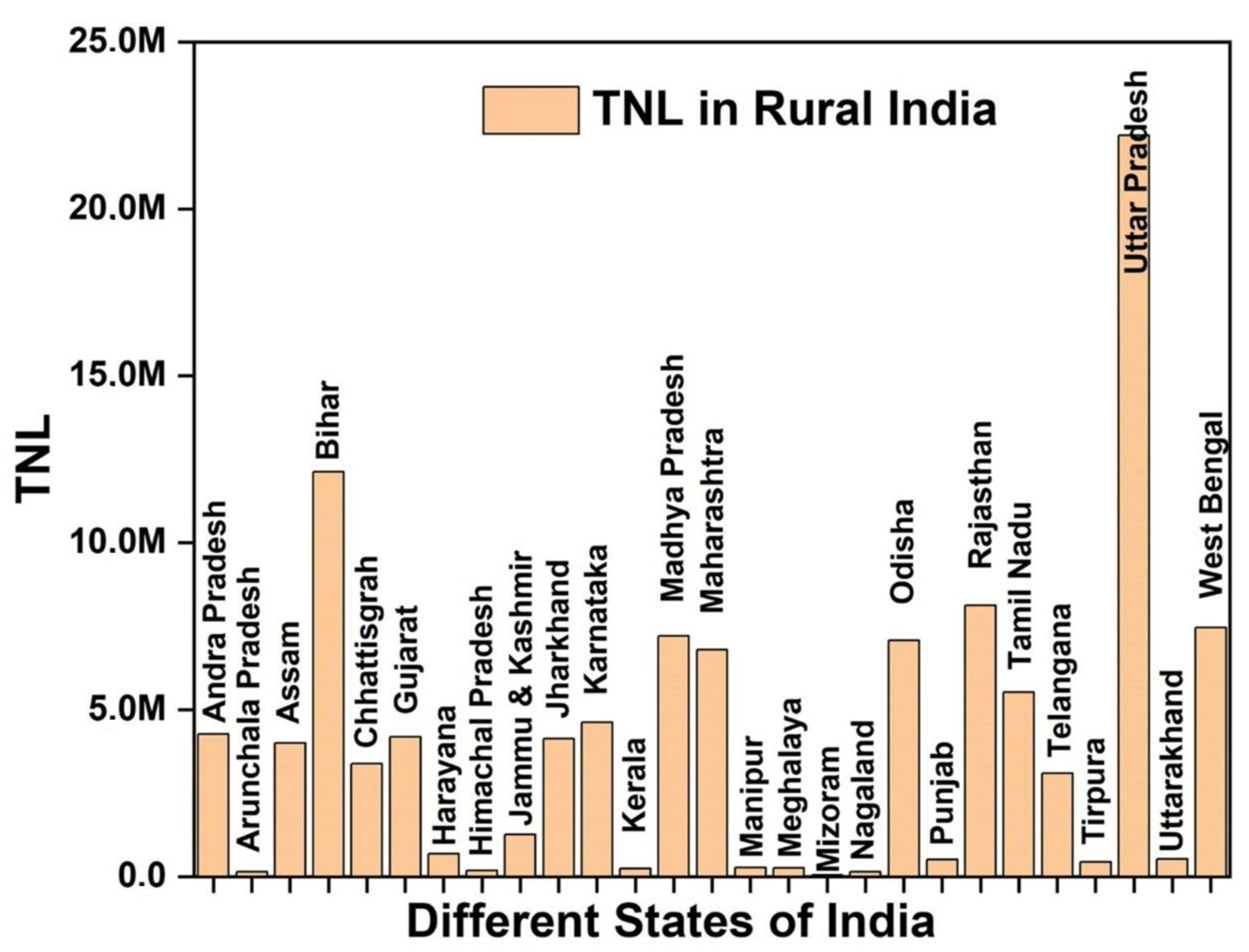
:1. Introduction
2. Goal and Need of On-Site Treatment Processes
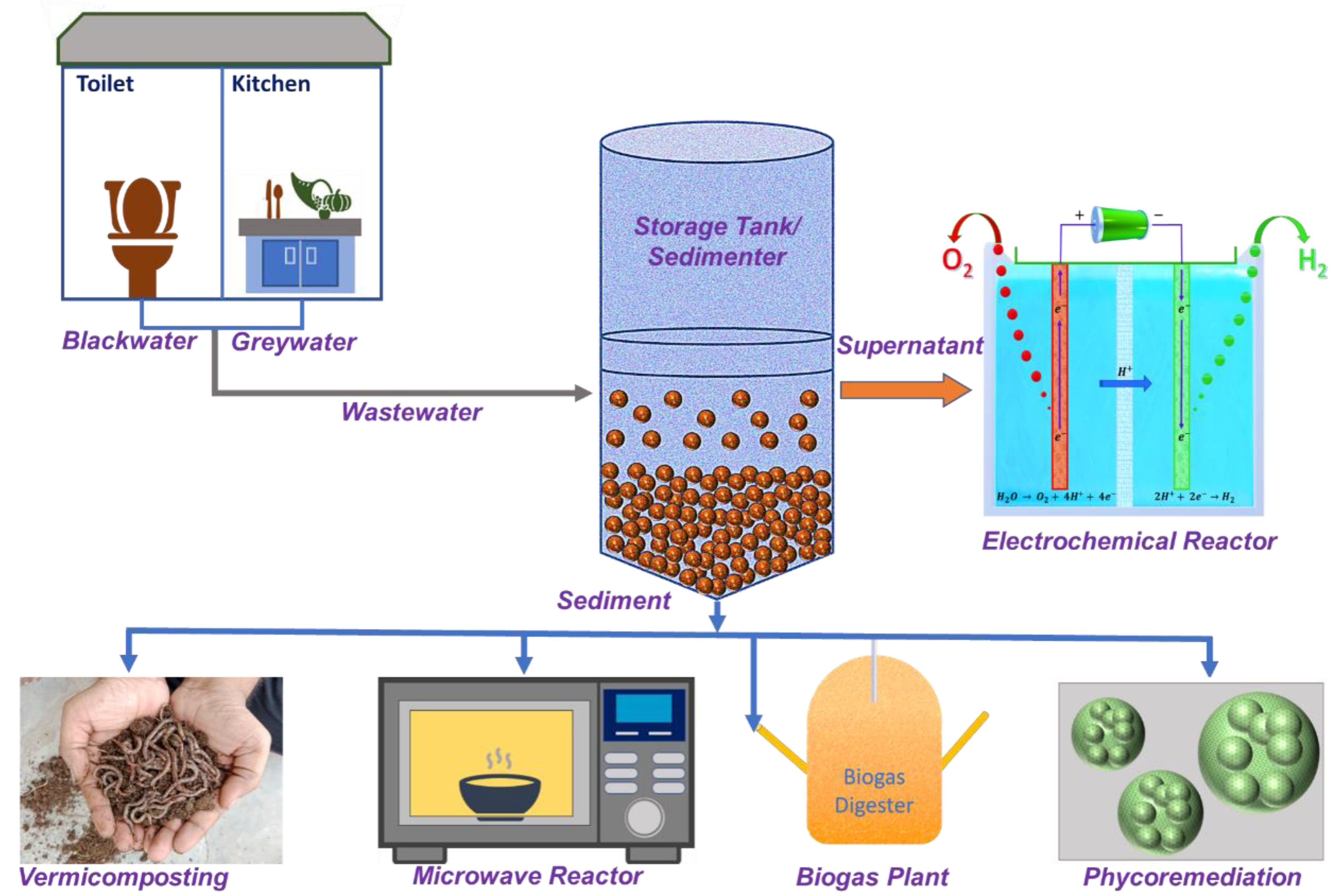
3. Novel Diverse Techniques for the On-Site Treatment of Blackwater (BW)
3.1. Electrochemical Treatment Process
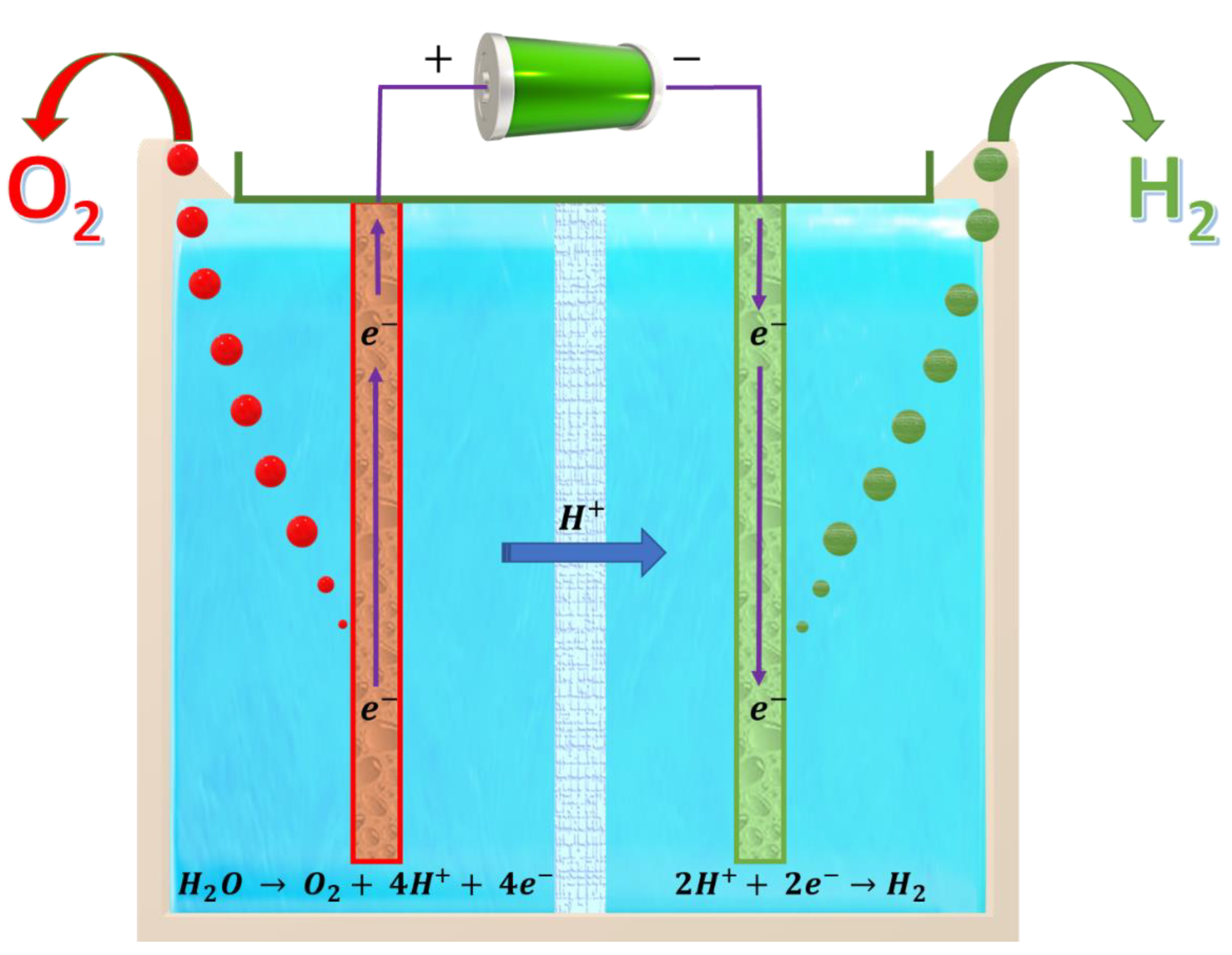
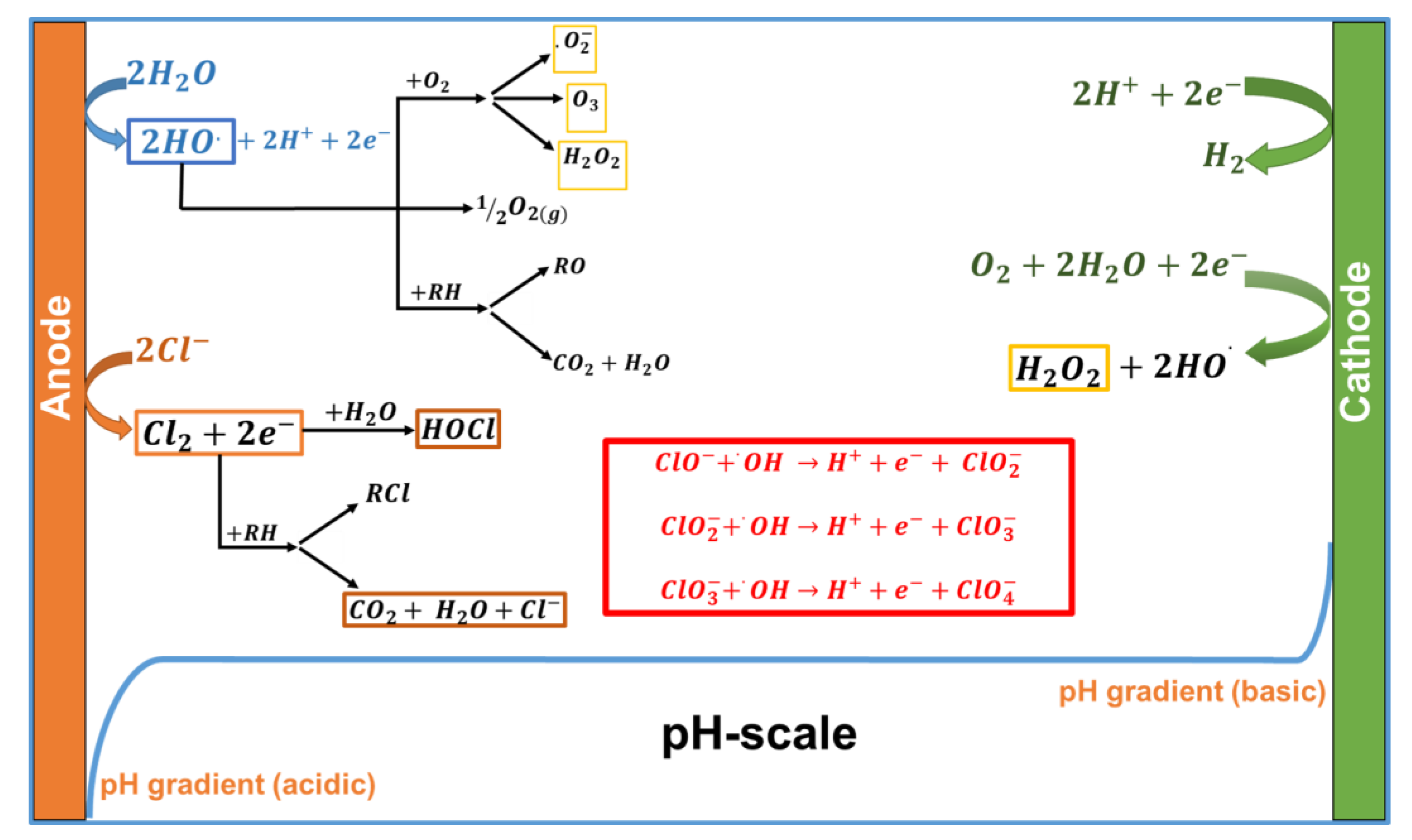
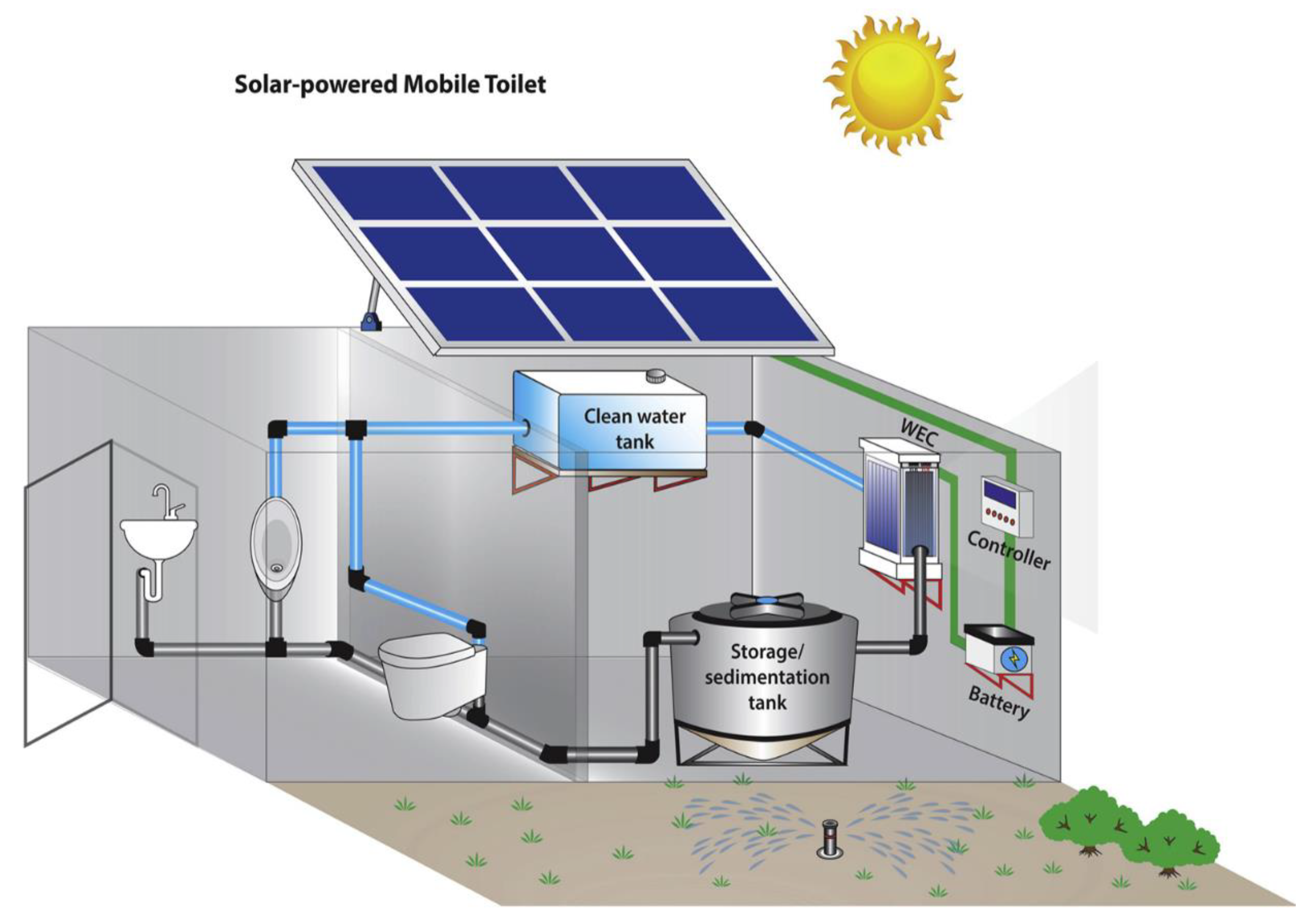

3.2. Vermicomposting

| Parameters | Influent | Effluent | Percentage Reduction (%) |
|---|---|---|---|
| BOD (mg/L) | 260 | 35.5 | 86.3 |
| COD (mg/L) | 345.8 | 102.8 | 70.2 |
| SS (mg/L) | 252.3 | 137 | 45.7 |
| TC (MPN/100 mL) | 7.05 | 3.12 | 55.74 |
| FC (MPN/100 mL) | 5.54 | 2.55 | 53.97 |
| FS (MPN/100 mL) | 6.03 | 2.39 | 60.36 |
3.3. Microwave (MW)-Based Reactor
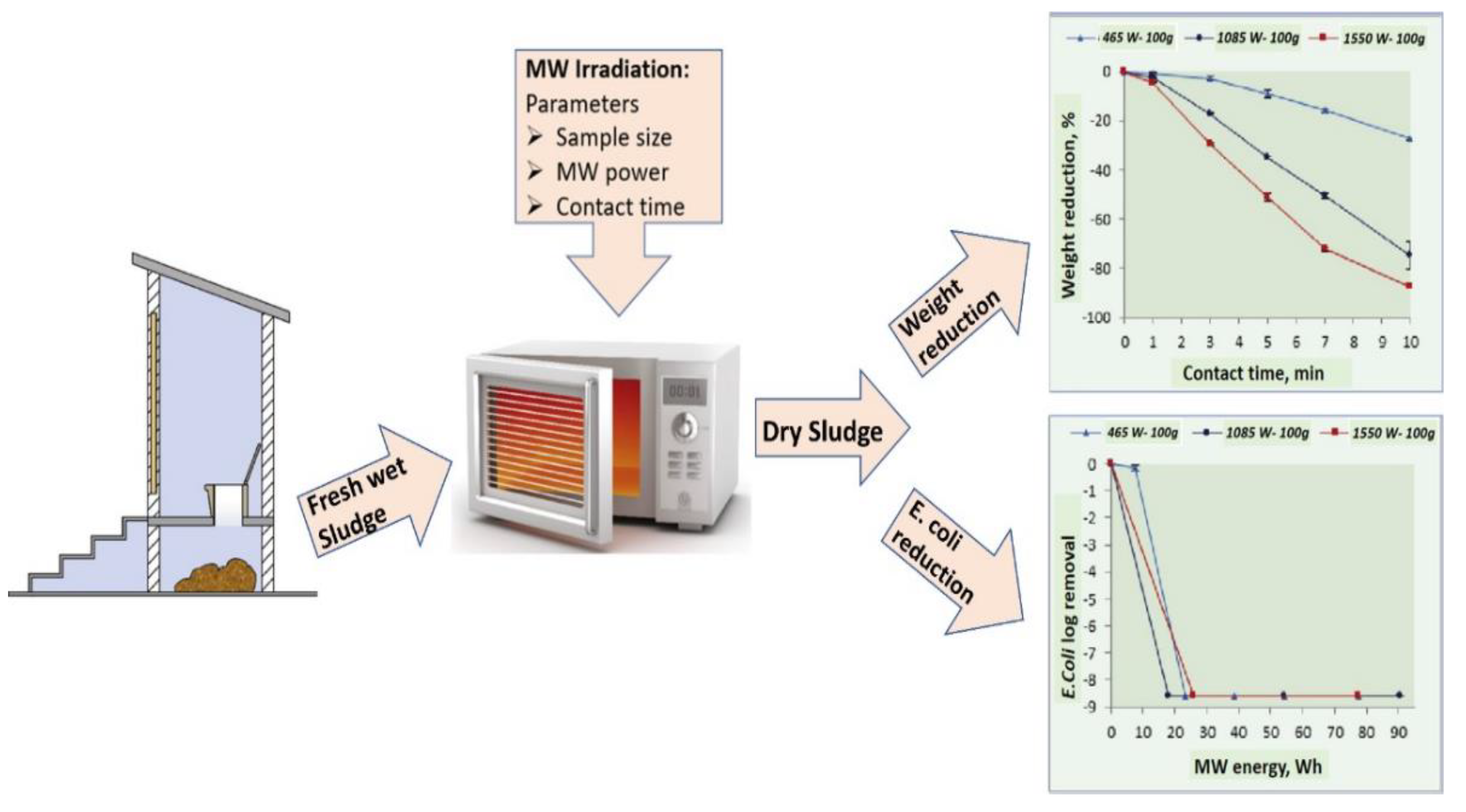

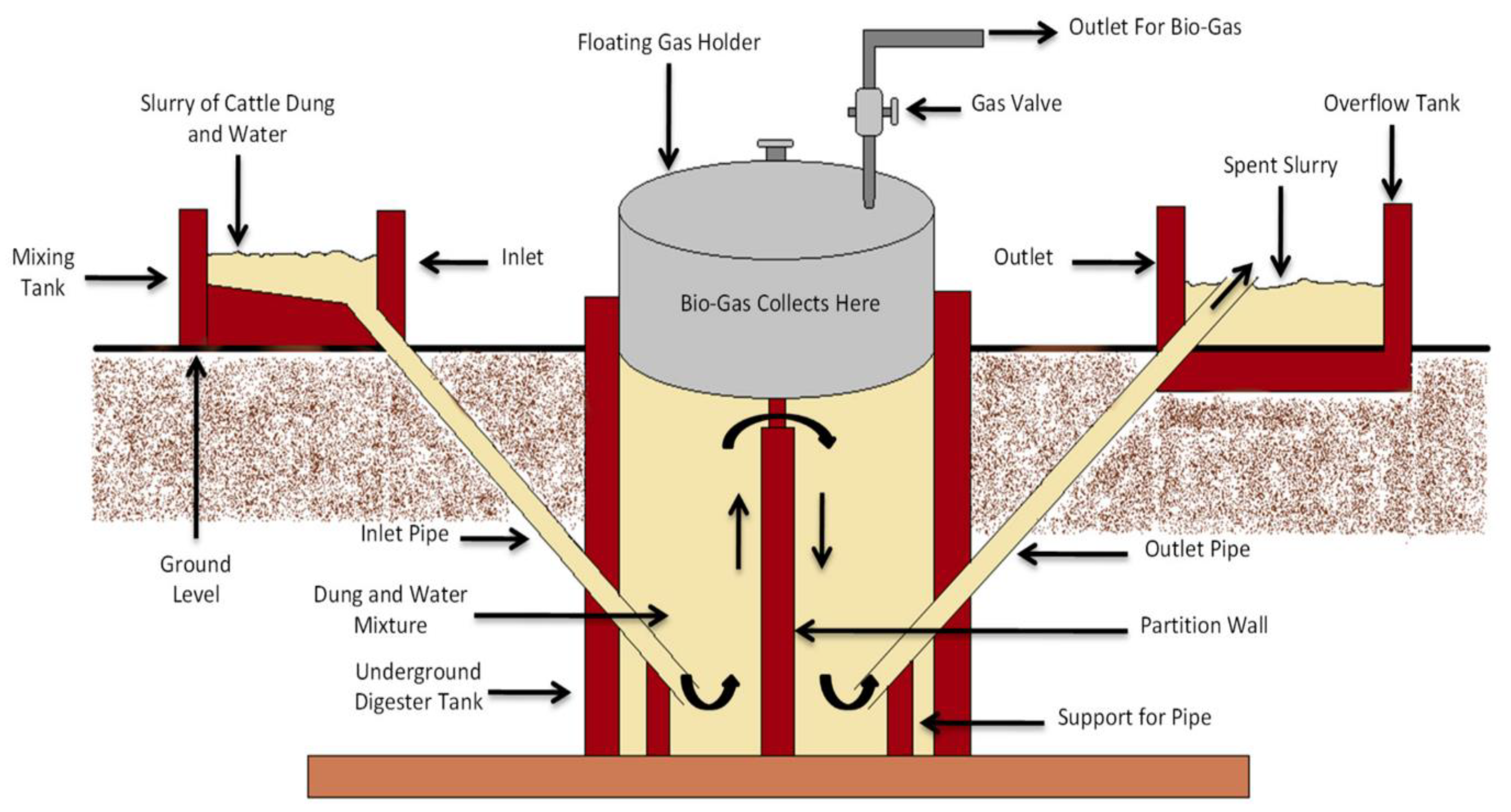
3.4. Biogas-Based Treatment
3.5. Phycoremediation
4. Discussion
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Khanam, K.; Patidar, S.K. Greywater characteristics in developed and developing countries. Mater. Today Proc. 2022, 57, 1494–1499. [Google Scholar] [CrossRef]
- Noori, R.; Farahani, F.; Jun, C.; Aradpour, S.; Bateni, S.M.; Ghazban, F.; Hosseinzadeh, M.; Maghrebi, M.; Vesali Naseh, M.R.; Abolfathi, S. A non-threshold model to estimate carcinogenic risk of nitrate-nitrite in drinking water. J. Clean. Prod. 2022, 363, 132432. [Google Scholar] [CrossRef]
- Dehestaniathar, S.; Nesari, S.; Borzooei, S.; Abolfathi, S. Application of natural biodegradable fiber as biofilm medium and carbon source in DEnitrifying AMmonium OXidation (DEAMOX) process for nitrogen removal from wastewater. J. Taiwan Inst. Chem. Eng. 2021, 119, 108–114. [Google Scholar] [CrossRef]
- Zhou, Y.; Guo, B.; Zhang, L.; Zou, X.; Yang, S.; Zhang, H.; Xia, S.; Liu, Y. Anaerobically digested blackwater treatment by simultaneous denitrification and anammox processes: Feeding loading affects reactor performance and microbial community succession. Chemosphere 2020, 241, 125101. [Google Scholar] [CrossRef]
- Rijsberman, F.R. Water scarcity: Fact or fiction? Agric. Water Manag. 2006, 80, 5–22. [Google Scholar] [CrossRef] [Green Version]
- Shaikh, I.N.; Ahammed, M.M. Quantity and quality characteristics of greywater from an Indian household. Environ. Monit. Assess. 2022, 194, 191. [Google Scholar] [CrossRef]
- Borzooei, S.; Teegavarapu, R.; Abolfathi, S.; Amerlinck, Y.; Nopens, I.; Zanetti, M.C. Impact Evaluation of Wet-Weather Events on Influent Flow and Loadings of a Water Resource Recovery Facility. In New Trends in Urban Drainage Modelling; Springer: Cham, Switzerland, 2019; pp. 706–711. Available online: https://link.springer.com/chapter/10.1007/978-3-319-99867-1_122 (accessed on 25 January 2023).
- Borzooei, S.; Teegavarapu, R.; Abolfathi, S.; Amerlinck, Y.; Nopens, I.; Zanetti, M.C. Data Mining Application in Assessment of Weather-Based Influent Scenarios for a WWTP: Getting the Most Out of Plant Historical Data. Water Air Soil Pollut. 2018, 230, 5. [Google Scholar] [CrossRef] [Green Version]
- Goodarzi, D.; Mohammadian, A.; Pearson, J.; Abolfathi, S. Numerical modelling of hydraulic efficiency and pollution transport in waste stabilization ponds. Ecol. Eng. 2022, 182, 106702. [Google Scholar] [CrossRef]
- Goodarzi, D.; Abolfathi, S.; Borzooei, S. Modelling solute transport in water disinfection systems: Effects of temperature gradient on the hydraulic and disinfection efficiency of serpentine chlorine contact tanks. J. Water Process Eng. 2020, 37, 101411. [Google Scholar] [CrossRef]
- Ahmadi, M.; Ghanbari, F. Optimizing COD removal from greywater by photoelectro-persulfate process using Box-Behnken design: Assessment of effluent quality and electrical energy consumption. Environ. Sci. Pollut. Res. 2016, 23, 19350–19361. [Google Scholar] [CrossRef]
- Quality of Waste Water. Available online: https://www.government.nl/topics/water-management/water-quality/quality-of-waste-water (accessed on 10 November 2022).
- Rural Population. Available online: https://data.worldbank.org/indicator/SP.RUR.TOTL?end=2021&start=1960&type=shaded&view=chart&year=2021 (accessed on 7 October 2022).
- Lal, S.R.S.; Herbert, G.M.J.; Arjunan, P.; Suryan, A. Advancements in renewable energy transition in India: A review. Energy Sources Part A Recovery Util. Environ. Eff. 2022, 1–31. [Google Scholar] [CrossRef]
- Percentage of Total Population in Rural India. Available online: https://www.macrotrends.net/countries/IND/india/rural-population#:~:text=India%20rural%20population%20for%202021,a%200.39%25%20increase%20from%202017 (accessed on 7 October 2022).
- India, G.o. Swachh Bharat Mission. Available online: https://swachhbharatmission.gov.in/sbmcms/index.htm (accessed on 22 January 2023).
- Toilets Built under Swachh Bharat Mission. Available online: https://pib.gov.in/PressReleaseIframePage.aspx?PRID=1797158 (accessed on 10 November 2022).
- Sekher, T.V. Rural Demography of India. In International Handbook of Rural Demography; Kulcsár, L.J., Curtis, K.J., Eds.; Springer Netherlands: Dordrecht, The Netherlands, 2012; pp. 169–189. [Google Scholar]
- Balk, D.; Montgomery, M.R.; Engin, H.; Lin, N.; Major, E.; Jones, B. Urbanization in India: Population and Urban Classification Grids for 2011. Data 2019, 4, 35. [Google Scholar] [CrossRef] [Green Version]
- Census Town. Available online: https://lms.indianeconomy.net/glossary/census-town/ (accessed on 24 December 2022).
- Borzooei, S.; Amerlinck, Y.; Panepinto, D.; Abolfathi, S.; Nopens, I.; Scibilia, G.; Meucci, L.; Zanetti, M.C. Energy optimization of a wastewater treatment plant based on energy audit data: Small investment with high return. Environ. Sci. Pollut. Res. 2020, 27, 17972–17985. [Google Scholar] [CrossRef] [PubMed]
- Borzooei, S.; Miranda, G.H.B.; Abolfathi, S.; Scibilia, G.; Meucci, L.; Zanetti, M.C. Application of unsupervised learning and process simulation for energy optimization of a WWTP under various weather conditions. Water Sci. Technol. 2020, 81, 1541–1551. [Google Scholar] [CrossRef]
- Schmalz, V.; Dittmar, T.; Haaken, D.; Worch, E. Electrochemical disinfection of biologically treated wastewater from small treatment systems by using boron-doped diamond (BDD) electrodes—Contribution for direct reuse of domestic wastewater. Water Res. 2009, 43, 5260–5266. [Google Scholar] [CrossRef]
- Anusuyadevi, P.R.; Svagan, A.J. Role of cellular solids in heterogeneous photocatalytic applications. In Nanostructured Photocatalysts; Elsevier: Stockholm, Sweden, 2021; pp. 305–330. [Google Scholar]
- Subramaniyam, R.; Manickaraj, S.; Anusuyadevi, P.R. Bioinspired Metal Nanoparticles for Microbicidal Activity. Bioinspired Nanomater. Synth. Emerg. Appl. 2021, 111, 36–62. [Google Scholar]
- Anusuyadevi, P.R. Synthesis of Novel Nanophotocatalyst in Micro/Millifludic Supercritical Reactor; Université de Bordeaux: Bordeaux, France, 2018. [Google Scholar]
- Anusuyadevi, P.R.; Riazanova, A.V.; Hedenqvist, M.S.; Svagan, A.J. Floating Photocatalysts for Effluent Refinement Based on Stable Pickering Cellulose Foams and Graphitic Carbon Nitride (g-C3N4). ACS Omega 2020, 5, 22411–22419. [Google Scholar] [CrossRef]
- Xu, C.; Ravi Anusuyadevi, P.; Aymonier, C.; Luque, R.; Marre, S. Nanostructured materials for photocatalysis. Chem. Soc. Rev. 2019, 48, 3868–3902. [Google Scholar] [CrossRef]
- Cui, Y.; Sheng, X.; Anusuyadevi, P.R.; Lawoko, M.; Svagan, A.J. Self-assembled carbon spheres prepared from abundant lignin and urea for photocatalytic and self-propelling applications. Carbon Trends 2021, 3, 100040. [Google Scholar] [CrossRef]
- Thostenson, J.O.; Mourouvin, R.; Hawkins, B.T.; Ngaboyamahina, E.; Sellgren, K.L.; Parker, C.B.; Deshusses, M.A.; Stoner, B.R.; Glass, J.T. Improved blackwater disinfection using potentiodynamic methods with oxidized boron-doped diamond electrodes. Water Res. 2018, 140, 191–199. [Google Scholar] [CrossRef]
- Karak, T.; Bhattacharyya, P. Human urine as a source of alternative natural fertilizer in agriculture: A flight of fancy or an achievable reality. Resour. Conserv. Recycl. 2011, 55, 400–408. [Google Scholar] [CrossRef]
- Cho, K.; Qu, Y.; Kwon, D.; Zhang, H.; Cid, C.A.; Aryanfar, A.; Hoffmann, M.R. Effects of Anodic Potential and Chloride Ion on Overall Reactivity in Electrochemical Reactors Designed for Solar-Powered Wastewater Treatment. Environ. Sci. Technol. 2014, 48, 2377–2384. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhao, K.; Quan, X.; Chen, S.; Yu, H.; Zhang, Y.; Zhao, H. Enhanced electro-Fenton performance by fluorine-doped porous carbon for removal of organic pollutants in wastewater. Chem. Eng. J. 2018, 354, 606–615. [Google Scholar] [CrossRef]
- Barroso-Martínez, J.S.; Romo, A.I.B.; Pudar, S.; Putnam, S.T.; Bustos, E.; Rodríguez-López, J. Real-Time Detection of Hydroxyl Radical Generated at Operating Electrodes via Redox-Active Adduct Formation Using Scanning Electrochemical Microscopy. J. Am. Chem. Soc. 2022, 144, 18896–18907. [Google Scholar] [CrossRef]
- Huang, X.; Qu, Y.; Cid, C.A.; Finke, C.; Hoffmann, M.R.; Lim, K.; Jiang, S.C. Electrochemical disinfection of toilet wastewater using wastewater electrolysis cell. Water Res. 2016, 92, 164–172. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kim, J.; Choi, W.J.K.; Choi, J.; Hoffmann, M.R.; Park, H. Electrolysis of urea and urine for solar hydrogen. Catal. Today 2013, 199, 2–7. [Google Scholar] [CrossRef]
- Bejan, D.; Guinea, E.; Bunce, N.J. On the nature of the hydroxyl radicals produced at boron-doped diamond and Ebonex® anodes. Electrochim. Acta 2012, 69, 275–281. [Google Scholar] [CrossRef]
- Panizza, M.; Cerisola, G. Direct And Mediated Anodic Oxidation of Organic Pollutants. Chem. Rev. 2009, 109, 6541–6569. [Google Scholar] [CrossRef]
- Brown, G.M.; Gu, B. The Chemistry of Perchlorate in the Environment. In Perchlorate: Environmental Occurrence, Interactions and Treatment; Gu, B., Coates, J.D., Eds.; Springer US: Boston, MA, USA, 2006; pp. 17–47. [Google Scholar]
- Chen, L.; Campo, P.; Kupferle, M.J. Identification of chlorinated oligomers formed during anodic oxidation of phenol in the presence of chloride. J. Hazard. Mater. 2015, 283, 574–581. [Google Scholar] [CrossRef]
- Cho, K.; Hoffmann, M.R. Molecular hydrogen production from wastewater electrolysis cell with multi-junction BiOx/TiO2 anode and stainless steel cathode: Current and energy efficiency. Appl. Catal. B Environ. 2017, 202, 671–682. [Google Scholar] [CrossRef] [Green Version]
- Lee, J.; Jun, M.-J.; Lee, M.-H.; Lee, M.-H.; Eom, S.-W.; Zoh, K.-D. Production of various disinfection byproducts in indoor swimming pool waters treated with different disinfection methods. Int. J. Hyg. Environ. Health 2010, 213, 465–474. [Google Scholar] [CrossRef] [PubMed]
- Bougeard, C.M.M.; Goslan, E.H.; Jefferson, B.; Parsons, S.A. Comparison of the disinfection by-product formation potential of treated waters exposed to chlorine and monochloramine. Water Res. 2010, 44, 729–740. [Google Scholar] [CrossRef]
- Talekar, G.V.; Sharma, P.; Yadav, A.; Clauwaert, P.; Rabaey, K.; Mutnuri, S. Sanitation of blackwater via sequential wetland and electrochemical treatment. NPJ Clean Water 2018, 1, 14. [Google Scholar] [CrossRef]
- Lei, Y.; Remmers, J.C.; Saakes, M.; van der Weijden, R.D.; Buisman, C.J.N. Influence of Cell Configuration and Long-Term Operation on Electrochemical Phosphorus Recovery from Domestic Wastewater. ACS Sustain. Chem. Eng. 2019, 7, 7362–7368. [Google Scholar] [CrossRef] [PubMed]
- Talekar, G.V.; Mutnuri, S. Electrochemical removal and recovery of ammonia and phosphates from blackwater and wetland passed blackwater. Sustain. Energy Technol. Assess. 2021, 47, 101374. [Google Scholar] [CrossRef]
- Sharma, K.; Garg, V.K. Chapter 10—Vermicomposting of Waste: A Zero-Waste Approach for Waste Management. In Sustainable Resource Recovery and Zero Waste Approaches; Taherzadeh, M.J., Bolton, K., Wong, J., Pandey, A., Eds.; Elsevier: Amsterdam, The Netherlands, 2019; pp. 133–164. [Google Scholar]
- Dey, M.D.; Das, S.; Kumar, R.; Doley, R.; Bhattacharya, S.S.; Mukhopadhyay, R. Vermiremoval of methylene blue using Eisenia fetida: A potential strategy for bioremediation of synthetic dye-containing effluents. Ecol. Eng. 2017, 106, 200–208. [Google Scholar] [CrossRef]
- Pereira, M.D.G.; Cardoso de Souza Neta, L.; Fontes, M.P.F.; Souza, A.N.; Carvalho Matos, T.; de Lima Sachdev, R.; dos Santos, A.V.; Oliveira da Guarda Souza, M.; de Andrade, M.V.A.S.; Marinho Maciel Paulo, G.; et al. An Overview of the Environmental Applicability of Vermicompost: From Wastewater Treatment to the Development of Sensitive Analytical Methods. Sci. World J. 2014, 2014, 917348. [Google Scholar] [CrossRef]
- Samal, K.; Moulick, S.; Mohapatra, B.G.; Samanta, S.; Sasidharan, S.; Prakash, B.; Sarangi, S. Design of faecal sludge treatment plant (FSTP) and availability of its treatment technologies. Energy Nexus 2022, 7, 100091. [Google Scholar] [CrossRef]
- Special Focus by the Department of Agricultural Research and Education (DARE) on Campaign 2.0 on Swachhta and Reducing Pendency in Government. Available online: https://pib.gov.in/PressReleaseIframePage.aspx?PRID=1871506 (accessed on 12 December 2022).
- Bajsa, O.; Nair, J.; Mathew, K.; Ho, G.E. Vermiculture as a tool for domestic wastewater management. Water Sci. Technol. 2004, 48, 125–132. [Google Scholar] [CrossRef]
- Arora, S.; Rajpal, A.; Bhargava, R.; Kumar, T. An Eco-friendly and Innovative Technology for Wastewater Treatment. Int. J. Environ. Sci. Dev. Monit. 2013, 4, 35–37. [Google Scholar]
- Arora, S.; Saraswat, S. Vermifiltration as a natural, sustainable and green technology for environmental remediation: A new paradigm for wastewater treatment process. Curr. Res. Green Sustain. Chem. 2021, 4, 100061. [Google Scholar] [CrossRef]
- Vermicomposting Toilets. Available online: http://www.vermicompostingtoilets.net/design-construction/ (accessed on 25 December 2022).
- Sudipti, A.; Rajpal, A.; Bhargava, R.; Kumar, T. Vermifiltration: A low-cost and sustainable alternative for wastewater treatment. Res. Gate 2013. Available online: https://www.researchgate.net/publication/257958116_Vermifiltration_A_low-cost_and_sustainable_alternative_for_wastewater_treatment (accessed on 25 January 2023).
- Tyagi, V.K.; Lo, S.-L. Microwave irradiation: A sustainable way for sludge treatment and resource recovery. Renew. Sustain. Energy Rev. 2013, 18, 288–305. [Google Scholar] [CrossRef]
- Mawioo, P.M.; Rweyemamu, A.; Garcia, H.A.; Hooijmans, C.M.; Brdjanovic, D. Evaluation of a microwave based reactor for the treatment of blackwater sludge. Sci. Total Environ. 2016, 548, 72–81. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Elagroudy, S.; El-Gohary, F.A. Microwave Pretreatment of Mixed Sludge for Anaerobic Digestion Enhancement. Int. J. Therm. Environ. Eng. 2013, 5, 105–111. [Google Scholar]
- Mudhoo, A.; Sharma, S.K. Microwave Irradiation Technology In Waste Sludge And Wastewater Treatment Research. Crit. Rev. Environ. Sci. Technol. 2011, 41, 999–1066. [Google Scholar] [CrossRef]
- Vialkova, E.; Obukhova, M.; Belova, L. Microwave Irradiation in Technologies of Wastewater and Wastewater Sludge Treatment: A Review. Water 2021, 13, 1784. [Google Scholar] [CrossRef]
- Martin, D.I.; Margaritescu, I.; Cirstea, E.; Togoe, I.; Ighigeanu, D.; Nemtanu, M.R.; Oproiu, C.; Iacob, N. Application of accelerated electron beam and microwave irradiation to biological waste treatment. Vacuum 2005, 77, 501–506. [Google Scholar] [CrossRef]
- Remya, N.; Lin, J.-G. Current status of microwave application in wastewater treatment—A review. Chem. Eng. J. 2011, 166, 797–813. [Google Scholar] [CrossRef]
- Mawioo, P.M.; Garcia, H.A.; Hooijmans, C.M.; Velkushanova, K.; Simonič, M.; Mijatović, I.; Brdjanovic, D. A pilot-scale microwave technology for sludge sanitization and drying. Sci Total Environ. 2017, 601, 1437–1448. [Google Scholar] [CrossRef]
- Lamble, K.J.; Hill, S.J. Microwave digestion procedures for environmental matrices. Critical Review. Analyst 1998, 123, 103R–133R. [Google Scholar] [CrossRef]
- Uddin, M.N.; Siddiki, S.Y.A.; Mofijur, M.; Djavanroodi, F.; Hazrat, M.A.; Show, P.L.; Ahmed, S.F.; Chu, Y.-M. Prospects of Bioenergy Production From Organic Waste Using Anaerobic Digestion Technology: A Mini Review. Front. Energy Res. 2021, 9, 627093. [Google Scholar] [CrossRef]
- Agrahari, R.P.; Tiwari, G. The Production of Biogas Using Kitchen Waste. Int. J. Energy Sci. 2013, 3, 408. [Google Scholar] [CrossRef]
- Rose, C.; Parker, A.; Jefferson, B.; Cartmell, E. The Characterization of Feces and Urine: A Review of the Literature to Inform Advanced Treatment Technology. Crit. Rev. Env. Sci. Technol. 2015, 45, 1827–1879. [Google Scholar] [CrossRef] [PubMed]
- Andriani, D.; Wresta, A.; Saepudin, A.; Prawara, B. A Review of Recycling of Human Excreta to Energy through Biogas Generation: Indonesia Case. Energy Procedia 2015, 68, 219–225. [Google Scholar] [CrossRef] [Green Version]
- Vögeli, Y. Anaerobic Digestion of Biowaste in Developing Countries: Practical Information and Case Studies; Eawag-Sandec: Dübendorf, Switzerland, 2014. [Google Scholar]
- Pandey, P.; Pandey, A.; Yan, L.; Wang, D.; Pandey, V.; Meikap, B.C.; Huo, J.; Zhang, R.; Pandey, P.K. Dairy Waste and Potential of Small-Scale Biogas Digester for Rural Energy in India. Appl. Sci. 2021, 11, 10671. [Google Scholar] [CrossRef]
- Rao, P.; Kumar, R.; Raghavan, B.; Subramanian, V.; Sivasubramanian, V. Application of phycoremediation technology in the treatment of wastewater from a leather-processing chemical manufacturing facility. Water SA 2011, 37, 64099. [Google Scholar] [CrossRef] [Green Version]
- Koul, B.; Sharma, K.; Shah, M.P. Phycoremediation: A sustainable alternative in wastewater treatment (WWT) regime. Environ. Technol. Innov. 2022, 25, 102040. [Google Scholar] [CrossRef]
- Olguin, E.J. Phycoremediation: Key issues for cost-effective nutrient removal processes. Biotechnol. Adv. 2003, 22, 81–91. [Google Scholar] [CrossRef]
- Shackira, A.M.; Jazeel, K.; Puthur, J.T. Chapter 13—Phycoremediation and phytoremediation: Promising tools of green remediation. In Sustainable Environmental Clean-Up; Kumar Mishra, V., Kumar, A., Eds.; Elsevier: Amsterdam, The Netherlands, 2021; pp. 273–293. [Google Scholar]
- Rath, B. Microalgal bioremediation: Current practices and perspectives. J. Biochem. Technol. 2012, 3, 299–304. [Google Scholar]
- Pavasant, P.; Apiratikul, R.; Sungkhum, V.; Suthiparinyanont, P.; Wattanachira, S.; Marhaba, T.F. Biosorption of Cu2+, Cd2+, Pb2+, and Zn2+ using dried marine green macroalga Caulerpa lentillifera. Bioresour. Technol. 2006, 97, 2321–2329. [Google Scholar] [CrossRef]
- Ahmad, A.; Bhat, A.H.; Buang, A. Biosorption of transition metals by freely suspended and Ca-alginate immobilised with Chlorella vulgaris: Kinetic and equilibrium modeling. J. Clean. Prod. 2018, 171, 1361–1375. [Google Scholar] [CrossRef]
- Guleri, S.; Singh, K.; Kaushik, R.; Dhankar, R.; Tiwari, A. Phycoremediation: A Novel and Synergistic Approach in Wastewater Remediation. J. Microbiol. Biotechnol. Food Sci. 2020, 10, 98–106. [Google Scholar] [CrossRef]
- Moges, M.E.; Heistad, A.; Heidorn, T. Nutrient Recovery from Anaerobically Treated Blackwater and Improving Its Effluent Quality through Microalgae Biomass Production. Water 2020, 12, 592. [Google Scholar] [CrossRef] [Green Version]
- Segovia Bifarini, M.A.; Žitnik, M.; Griessler Bulc, T.; Krivograd Klemenčič, A. Treatment and Re-Use of Raw Blackwater by Chlorella vulgaris-Based System. Water 2020, 12, 2660. [Google Scholar] [CrossRef]
- Škufca, D.; Kovačič, A.; Prosenc, F.; Griessler Bulc, T.; Heath, D.; Heath, E. Phycoremediation of municipal wastewater: Removal of nutrients and contaminants of emerging concern. Sci. Total Environ. 2021, 782, 146949. [Google Scholar] [CrossRef]
- Swachh Bharat Abhiyan (Clean India Mission). Available online: https://sdgs.un.org/partnerships/swachh-bharat-abhiyan-clean-india-mission (accessed on 28 December 2022).
- Swach Bharat Abhiyan. Available online: https://palwal.gov.in/swach-bharat-abhiyan/#:~:text=To%20eliminate%20or%20reduce%20open,mechanism%20of%20monitoring%20latrine%20use (accessed on 28 December 2022).
- Parmar, S. Swachch Bharat Abhiyan- Clean INDIA Initiative: Objectives, Targets & Progress Report. Available online: https://www.theindianwire.com/politics/swachch-bharat-abhiyan-207010/ (accessed on 28 December 2022).
- Indian Government. Municipal Solid Waste Management Manual; Indian Government: New Delhi, India, 2016; Volume 2.
- Pirsaheb, M.; Khosravi, T.; Sharafi, K. Domestic scale vermicomposting for solid waste management. Int. J. Recycl. Org. Waste Agric. 2013, 2, 4. [Google Scholar] [CrossRef] [Green Version]
- Babaei, A.; Shayegan, J. Effects of temperature and mixing modes on the performance of municipal solid waste anaerobic slurry digester. J. Env. Health Sci. Eng. 2019, 17, 1077–1084. [Google Scholar] [CrossRef]


| Source | Waste Amount/Day/kg | Water (%) | Dry Matter (%) | Biogas m3/kg Dry Waste |
|---|---|---|---|---|
| Cow | 20–30 | 80 | 20 | 0.023–0.040 |
| Rooster/Hen | 0.15–0.20 | 72 | 28 | 0.065–0.116 |
| Human | 0.10–0.40 | 77 | 23 | 0.02–0.028 |
| Treatment Techniques | Advantages | Disadvantages |
|---|---|---|
| Electrochemical (EC) |
|
|
| Vermicomposting |
|
|
| Microwave (MW) |
|
|
| Biogas |
|
|
| Phycoremediation |
|
|
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Anusuyadevi, P.R.; Kumar, D.J.P.; Jyothi, A.D.H.V.O.; Patwardhan, N.S.; V., J.; Mol, A. Towards Viable Eco-Friendly Local Treatment of Blackwater in Sparsely Populated Regions. Water 2023, 15, 542. https://doi.org/10.3390/w15030542
Anusuyadevi PR, Kumar DJP, Jyothi ADHVO, Patwardhan NS, V. J, Mol A. Towards Viable Eco-Friendly Local Treatment of Blackwater in Sparsely Populated Regions. Water. 2023; 15(3):542. https://doi.org/10.3390/w15030542
Chicago/Turabian StyleAnusuyadevi, Prasaanth Ravi, Darsi Jaya Prasanna Kumar, A. D. H. V. Omkaara Jyothi, Neha S. Patwardhan, Janani V., and Arjan Mol. 2023. "Towards Viable Eco-Friendly Local Treatment of Blackwater in Sparsely Populated Regions" Water 15, no. 3: 542. https://doi.org/10.3390/w15030542





